Using Systematic Reviews to Support Future Dietary Reference Intakes: A Letter Report (2023)
Chapter: Summary
Summary
The Standing Committee for the Review of the Dietary Reference Intake (DRI) Framework was convened to consider past experiences and learnings from previous DRI reports and other relevant documents related to the DRI process in response to questions about the DRI framework and structuring of new DRI reviews. To answer questions posed by the sponsors (the U.S. Department of Agriculture and Health Canada), the committee provides overarching responses that could apply across future DRI reviews. These responses are submitted as letter reports to the sponsors.
THE COMMITTEE’S CHARGE
In this letter report, the committee was asked by the Federal DRI Joint U.S.-Canadian Working Group to consider the application of systematic reviews (SRs) to support the development of the Dietary Reference Intakes (see Box S-1). The committee was asked to consider the following three overarching questions:
- Are de novo systematic reviews needed in future DRI reviews or can qualified systematic reviews be used?
- If qualified systematic reviews can be used, what are the appropriate inclusion and exclusion criteria?
- Can previously published systematic reviews be updated?
The committee was further asked to develop a decision tree to guide the process and to comment on the need for meta-analysis questions as part of the systematic review process. The standing committee notes that its response to this task was to focus its recommendations on the systematic review questions that occur in preparation for, but prior to, the deliberations of a DRI committee. It was not the standing committee’s task to address issues related to the interpretation or application of reviews that would occur during a DRI study.
In discussion, the sponsors requested that the committee, as part of its task, comment on the series of papers on the Nordic Nutrition Recommendations 2022 (Arnesen et al., 2020a,b; Christensen et al., 2020; Høyer et al., 2021) and a report from the European Food Safety Authority (EFSA), “Scientific Opinion on a Tolerable Upper Intake Level for Dietary Sugars” (NDA, 2022).
USE OF SYSTEMATIC REVIEWS TO SUPPORT DRI REVIEWS
The use of systematic reviews in nutrition research and related applications is advantageous because it enhances the ability to evaluate the rigor and overall strength of a body of evidence. Further, the process enhances transparency by documenting the decision-making process. Inclusion of an analytic framework provides clarity to the research questions and con-
tributes to the supporting logic. A high-quality and transparent systematic review that would meet the needs of a DRI panel should encompass the following key characteristics:
- the research questions and content are relevant to the Populations, Interventions and/or Exposures, Comparators, and Outcomes of interest (PI[E]CO criteria) for the DRI decisions of interest;
- the systematic review was conducted by qualified systematic review methodologists who are both knowledgeable and independent of the user or sponsoring groups;1
- the systematic review has been peer reviewed and is made publicly available;
- the systematic review was not sponsored by parties that have substantial conflicts of interest, including financial conflicts of interest.2
The standing committee notes that non-peer-reviewed reports, theses, dissertations, official documents, informal communication, research-in-progress, or clinical trials produced by government agencies, academic institutions, or business do not meet the standard of evidence for a DRI review and thus would not be included in the process.
Criteria for Systematic Reviews
The standing committee was asked to consider application of systematic reviews to support the conduct of a DRI study. This would include reviews performed de novo as well as existing and updates to existing systematic reviews. The committee used the series of papers on the Nordic Nutrition Recommendations (NNR2022) as a guide to identify the following criteria that it recommends be considered for systematic reviews:
- Systematic reviews should document how they were conducted.
- The PI(E)CO criteria in an existing systematic review need to be closely aligned with the DRI research questions. Specific attention should be given to the methodologies used to assess nutrient or food substance intakes or status.
___________________
1 Qualified methodologists cannot be employees or paid associates of the sponsoring group and such individuals should not participate in the systematic review process.
2 Conflicts of interest and any policies regarding conflicts of interest will be defined by the user or sponsoring group that is commissioning the systematic review.
- The decision about eligibility of studies for inclusion in the systematic review should be made and agreed to by more than one reviewer.
- The publication range of dates used in an existing systematic review must be considered to ensure that the most relevant and up-to-date evidence has been captured.
- Consideration should be given to heterogeneity across studies, including outcomes, interventions, intake response, and methodological heterogeneity.
- The systematic review should include discussion of biases that affect the evidence base, including publication and time-lag bias, and the reporting of individual study results (reporting biases).
- The systematic review includes an assessment of risk of bias using an appropriate tool.
- The systematic review includes a strength of evidence evaluation.
- The scientific quality of the systematic review should be assessed using a recognized standard quality assessment tool such as AMSTAR.3
Nordic Nutrition Recommendation’s Criteria for “Qualified” Systematic Reviews
The standing committee used the NNR2022 requirements as a guide in developing its criteria. However, the committee suggests using the term “eligible” instead of “qualified” to avoid confusion with the use of “qualified health claims” on food labels for which the available evidence does not meet the “significant scientific agreement” standard.
The NNR2022 identified six criteria needed in a “qualified” or “eligible” systematic review that was designed and conducted for purposes other than evaluating causal relationships between nutrients and health outcomes (Arnesen et al, 2020b, p.2). These are:
- “a clearly stated set of objectives and research questions with predefined eligibility criteria for the studies;
- an explicit, reproducible methodology;
- a systematic search that attempts to identify all studies that would meet the eligibility criteria;
- an assessment of the validity of the findings of the included studies through an assessment of risk of bias of the studies;
___________________
3 AMSTAR = Assessment of Multiple Systematic Reviews. This tool would be used at the level of assessment by a DRI committee as part of its overall assessment of the SR quality.
- a systematic presentation and synthesis of the characteristics and findings of the included studies; and
- a grading of the overall evidence.”
Interpreting and applying these criteria to the decision about whether and how to use a systematic review should be made by the DRI review committee.
When is a Meta-Analysis Needed in a Systematic Review?
A meta-analysis is a quantitative synthesis of results from separate studies. It can be used to evaluate heterogeneity and estimate commonality among the dose-response relationships when there are multiple published scientific studies addressing the same question. A meta-analysis that uses the results of a systematic review should only be conducted when adequate high-quality data are available. Systematic reviews with or without a meta-analysis, due to inadequate or poor data, are useful because they provide an unbiased evaluation of the data. The appropriateness of a meta-analysis would be determined on the basis of available high-quality data.
DECISION TREE TO DETERMINE THE NEED FOR A DE NOVO OR UPDATE OF AN EXISTING SYSTEMATIC REVIEW
The NNR2022 served as a resource to determine the need for a de novo or update of an existing systematic review that could support a DRI study. The decision tree recommended by this committee for determining whether a de novo or updated systematic review is needed is shown in Figure S-1. Specific factors such as high-profile topics, or systematic reviews that are inconsistent in their results may need to be considered on a case-by-case base by the user or sponsoring group as they decide whether to update or conduct a de novo systematic review. The standing committee notes that it is possible that a systematic review may describe results that are different from conclusions. Thus, the user or sponsoring group may choose to disregard the interpretive elements of the systematic review and focus on the results.
EUROPEAN FOOD SAFETY AUTHORITY REPORT
The standing committee was also asked to review and comment on the European Food Safety Authority (EFSA) Panel on Nutrition, Novel Foods and Food Allergens (NDA Panel) report, “Scientific Opinion on a Tolerable Upper Intake Level for Dietary Sugars” (NDA, 2022). The com-
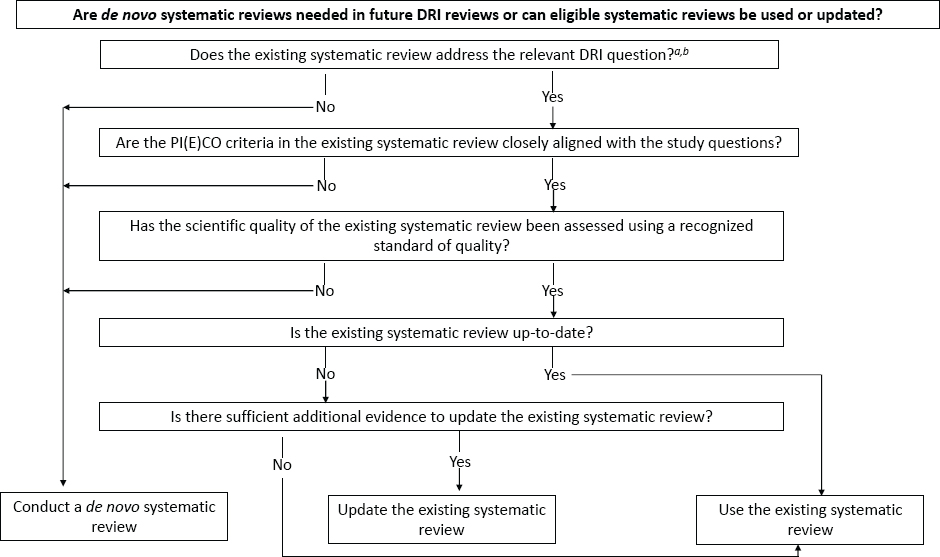
NOTE: PI(E)CO = population, intervention (exposure), comparator, outcome.
a A relevant DRI question would include specific DRI values to be addressed in the review such as setting a Tolerable Upper Intake Level (UL) vs. Chronic Disease Risk Reduction (CDRR) value.
b The process of setting the study questions and development of the PI(E)CO criteria falls to the group that is commissioning the systematic review.
mittee evaluated the applicability of the approach that was employed in the EFSA assessment for possible use in a future DRI review on carbohydrates, but not fiber.
The Acceptable Macronutrient Distribution Range (AMDR) was developed to specifically provide guidance about the relative range of intakes from carbohydrates, proteins, and fats associated with reduced risk of chronic diseases while concurrently ensuring that intakes of micro-nutrients were nutritionally sufficient. A new Chronic Disease Risk Reduction (CDRR) category was created during the DRI update of sodium and potassium to provide an approach for evaluating relationships between those nutrients and chronic disease risk and for future DRI reviews. Based on this previous work, the standing committee concludes that the EFSA report on setting a Tolerable Upper Intake Level (UL) for added sugars is not an appropriate application for the U.S.-Canadian DRI process because it is not based on chronic disease risk.
When evaluating sugar intakes and chronic disease outcomes, the standing committee recommends that future DRI committees use the CDRR model. Additionally, the standing committee recommends that a future DRI committee on macronutrients should reevaluate the necessity of continued use of the AMDR when developing its recommendations.
This page intentionally left blank.







