PFAS in Agricultural Systems: Guidance for Conservation Programs at USDA (2026)
Chapter: 2 PFAS in Agricultural Systems
2
PFAS in Agricultural Systems
Among chemicals of concern, per- and polyfluoroalkyl substances (PFAS) have garnered a spotlight due to their persistence, links to adverse health effects, high usage, and the difficulty in identifying adequate replacements in a timeframe that will not affect U.S. economic competitiveness and national security. To provide an initial framework to guide the efforts of the Farm Production and Conservation (FPAC) programs that directly deal with conservation on the land, it is essential to understand the underlying science of how PFAS enter and interact with environmental media. This chapter provides foundational information on the structure, classification, and environmental behavior of PFAS—key elements that influence occurrence, mobility, persistence, and potential impacts of PFAS in agricultural systems. The committee uses agricultural systems as a shorthand to describe all the lands on which relevant FPAC agencies—namely, the Natural Resources Conservation Service (NRCS) and the Farm Service Agency—may work. As described by NRCS at one of the committee’s meetings, the term agricultural system applies to a farm-level system and does not extend beyond the physical boundary of a farm operation.1
Understanding the characteristics and behavior of PFAS is critical for evaluating how PFAS contamination may affect the natural resource base—that is, soil, water, air, and plant and animal systems—and how PFAS may enter and cycle within agricultural systems. This chapter outlines the entry pathways for PFAS into agricultural settings, including point and nonpoint sources, and examines how PFAS move through agricultural landscapes and potentially leave the farm through surface water, groundwater, air, and food and other farm products.
___________________
1 Personal communication, B. Reck, National Environmental Engineer, Natural Resources Conservation Service, U.S. Department of Agriculture. “Presentation to the committee,” April 3, 2025. https://www.nationalacademies.org/projects/DELS-BANR-24-03/event/44748.
PFAS STRUCTURE
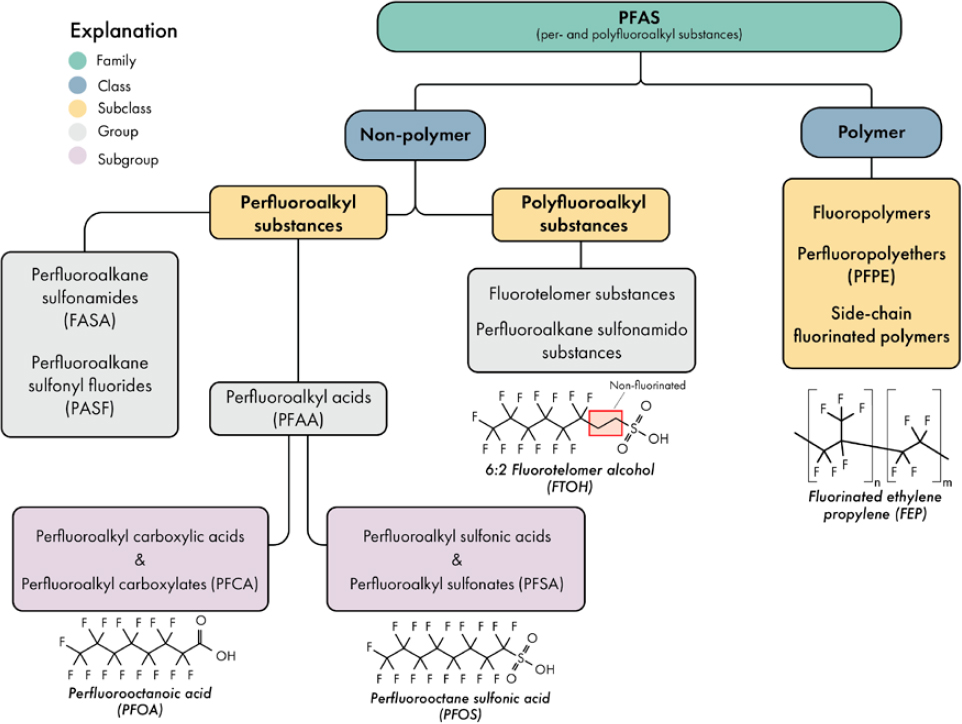
PFAS can be differentiated from other synthetic compounds by the presence of one or more perfluorinated carbons, typically present as or within a branched or linear alkyl chain of varying carbon chain lengths, bound to a polar functional group, such as a carboxylate, sulfonate, alcohol, phosphate, amino, or other group (Figure 2-1). Polyfluorinated substances also have non-fluorinated, hydrocarbon moieties present within the alkyl chain or attached to the polar functional group. PFAS are manufactured primarily through (1) fluorotelomerization (FT), which results in a –CH2CH2– linkage between the perfluoroalkyl chain and the polar functional group, and (2) electrochemical fluorination (ECF), which directly connects the perfluoroalkyl chain and the polar functional group (Buck et al. 2011). In addition, perfluoroalkyl ethers are manufactured through esterification and dehalocarbonylation. These processes, the type of polar functional group, and additional hydrocarbon moieties lead to several unique PFAS classes and varied environmental fate.
There are two broad classes of PFAS, namely polymers and non-polymers (Figure 2-2). Non-polymers are further divided into two subclasses: perfluoroalkyl substances (fully fluorinated) and polyfluoroalkyl substances (not fully fluorinated). Each of these subclasses contain different PFAS groups (Buck et al. 2011). Within perfluoroalkyl substances, the group perfluoroalkyl acids (PFAAs) contains the most well-known and well-studied PFAS—perfluorooctanoic acid (PFOA) and perfluorooctane sulfonic acid (PFOS)—which fall, respectively, into the two subgroups of PFAAs: perfluoroalkyl carboxylic acids (PFCAs) and perfluoroalkyl sulfonic acids (PFSAs) (Box 2-1). There are other PFAA classes, such as phosphonic (PFPAs, -PO3H2), sulfinic (PFSiAs, -SO2H), and phosphinic (PFPiAs, -PO2H) acids. Notably, these acids exist as anions in
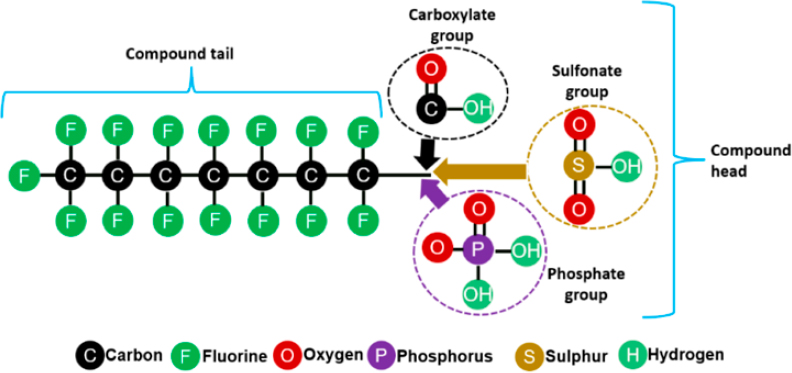
NOTE: PFAS structures shown here are present in the environment as anions due to the low pKa of their acidic functional groups.
SOURCE: © 2023 The Pennsylvania State University. All rights reserved. See https://extension.psu.edu/understanding-pfas-what-they-are-their-impact-and-what-we-can-do.
BOX 2-1
Legacy PFAS: PFOA and PFOS
Of the thousands of PFAS that exist, the two most studied are perfluorooctanoic acid (PFOA) and perfluorooctane sulfonic acid (PFOS). These were widely used for decades in products as processing aids for some nonstick coatings, stain-resistant fabrics, and aqueous film-forming foam (Lindstrom et al. 2011). With mounting evidence of the toxicity, persistence, and bioaccumulation of these long-chain PFAS, the U.S. Environmental Protection Agency (EPA) began working with manufacturers in 2000 to end production of these substances (EPA 2000; Lindstrom et al. 2011). Most U.S. production of PFOA and PFOS was phased out by 2015 (EPA 2024a). Because they are no longer in mass production, these compounds are often referred to as legacy PFAS.
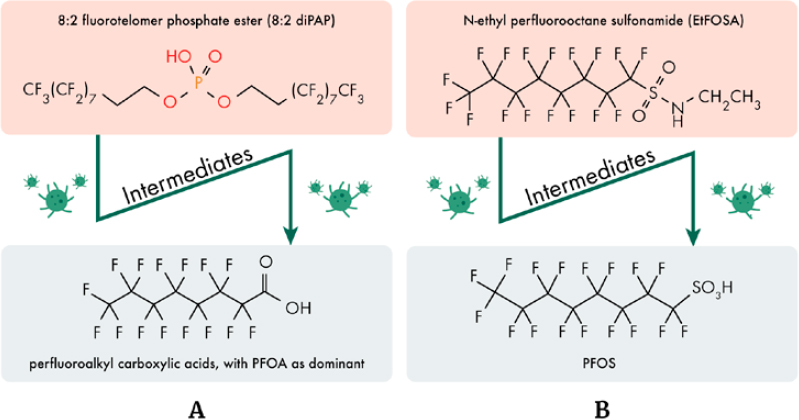
PFOA and PFOS are detected in the blood of nearly all participants in surveys of the U.S. general population, although the amount of PFOS and PFOA in samples has declined between the first survey at the beginning of the 21st century and surveys conducted two decades later. The downward trend might be expected with the phasing out of these substances from consumer products (Botelho et al. 2025). Nevertheless, detectable amounts of these compounds were still found in the blood of most adolescents, even though many in this cohort were born after phase-out efforts began (Botelho et al. 2025). The detection of legacy PFAS in this subpopulation, and its continued detection in the U.S. adult general population, likely results from the compound persistence and because PFOA and PFOS are terminal transformation products of precursor PFAS degradation. Precursor PFAS are polyfluoroalkyl substances that can degrade into terminal PFAS such as PFOA and PFOS. Examples include perfluorooctane sulfonamide and N-ethyl perfluorooctane sulfonamide, which degrade ultimately to PFOS, and fluorotelomer PFAS like 8:2 fluorotelomer alcohol and 8:2 diPAPs, which degrade ultimately to PFOA. In some cases, precursor PFAS were introduced to replace legacy PFAS. Degradation can occur in the environment or through biotransformation in the body (Sunderland et al. 2019; ITRC 2023).
the environment because of their low acid dissociation constants (pKa); for example, PFOS exists and is often referred to in the literature as perfluorooctane sulfonate. Additional perfluoroalkyl substances include perfluoroalkane sulfonamides (FASAs), perfluoroalkyl aldehydes (PFALs), perfluoroalkyl iodides (PFAIs), perfluoroalkane sulfonyl fluorides (PASF), and perfluoroalkyl ethers (PFEAs).
Groups within the subclass of polyfluoroalkyl substances include FT-based PFAS and semi-fluorinated n-alkanes and alkenes, and ECF-based perfluoroalkane sulfonamido derivatives.2 Polyfluoroalkyl substances differ from perfluoroalkyl substances
___________________
2 Greater detail of the PFAS family tree can be found in Appendix C.
because they contain both perfluorinated carbons and hydrocarbon segments. In ECF-derived polyfluoroalkyl substances, a perfluorinated chain is typically bonded to one side of a central functional group (e.g., a sulfonamide) with hydrocarbon moieties on the other. As noted above, FT-based PFAS consist of a single alkyl chain with a hydrocarbon spacer separating the functional group from the perfluorinated carbons. As a result, FT-based PFAS use nomenclature such as n:2 or n:3, indicating the ratio of perfluorinated carbons (n) to hydrocarbon carbons (2 or 3). For example, 6:2 fluorotelomer sulfonic acid (6:2 FTS) contains six perfluorinated carbons and two hydrocarbon carbons in one alkyl chain linked to a sulfonic acid group.
Polymer PFAS include fluoropolymers, polymeric perfluoropolyethers (PFPE), and side-chain fluorinated polymers. Most polymer PFAS are not easily broken down into monomer PFAS that can mobilize or bioaccumulate (Russell et al. 2008; Washington et al. 2009; Russell et al. 2010; Rankin et al. 2014; Washington and Jenkins 2015; Washington et al. 2015). Exceptions exist for certain side-chain fluoropolymers, some of which have relatively short environmental half-lives of less than 1 year (OECD 2022). Beyond these cases, there remains considerable debate regarding the persistence of polymeric PFAS, with reported half-life estimates ranging from several decades to millennia (Russell et al. 2008, 2010; Rankin et al. 2014; Washington and Jenkins 2015; Washington et al. 2015; Dasu and Lee 2016). Therefore, exposure pathways leading to adverse impacts on human and animal health are assumed to be lower compared with non-polymer PFAS. However, environmental data are sparse, and there is a lack of consensus on this topic (Lohmann et al. 2020). Occurrence data on side-chain fluorinated polymer surfactants is limited, but they have been found at up to several hundred parts per billion (ppb; equivalent at µg/kg) in Canadian biosolids (Letcher et al. 2020). In addition, the residual monomer PFAS that may be present in fluorinated polymers can be released during product use (OECD 2022).
Concerns with polymer PFAS are more about occupational exposure to and environmental emissions of non-polymer PFAS during polymer manufacturing processes as well as potential non-polymer PFAS residuals in finished products containing polymer PFAS. For example, atmospheric release of PFOA during application of dispersed fluoropolymer coatings and subsequent deposition has caused soil and groundwater impacts in regions such as eastern New York and western Vermont (Schroeder et al. 2021). Studies have also found that non-polymeric fluorotelomer alcohols (FTOHs) can be released from polymers (Dinglasan-Panlilio and Mabury 2006), which have been shown to contribute substantially to long-range, atmospheric PFAS transport (Wallington et al. 2006). Dispersal of non-polymer PFAS residuals in finished products containing polymer PFAS has been intentionally reduced over the past two decades. Some groups are also concerned about the unknown longevity of polymer PFAS after product disposal (Lohmann et al. 2020; Lohman and Letcher 2023).
Regulatory agencies have added chain length as a delineator of PFAS because the length of the carbon chain is a primary factor that influences the behavior of a PFAS in the environment and its potential to accumulate in living tissue (Figure 2-3). For example, the U.S. Environmental Protection Agency (EPA) characterizes PFCAs (Cn–1F2n–1–COOH) as long chain if the substances have a chain of at least seven perfluorocarbon atoms; PFSAs (CnF2n+1SO3H) are long chain with six or more perfluorocarbon
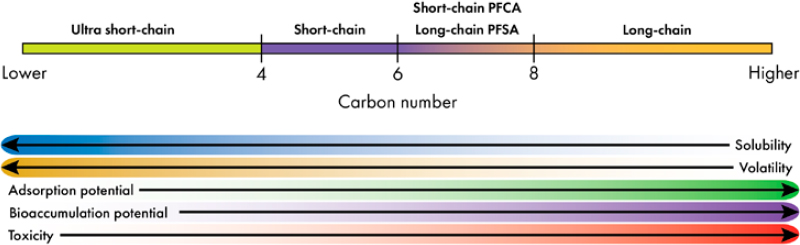
NOTE: The toxicity trends with chain length are not robust due to limited toxicity data for short-chain PFAS and the growing evidence of different toxicity mechanisms between short-chain and long-chain PFAS (Solan et al. 2023; Wang et al. 2024).
SOURCE: McAdoo et al. 2022.
atoms (Pulster et al. 2024). Ultra short-chain PFAS have fewer than four carbons (e.g., trifluoroacetic acid to perfluoropropionic acid). While the approach described here designates short-chain versus long-chain PFAS, there is no universally accepted definition for these two groupings.
OCCURRENCE OF PFAS IN U.S. SOILS
The scope of the PFAS challenge in U.S. agriculture is not well characterized as there are no national systematic surveys of PFAS concentrations in soil, groundwater, or surface water. EPA maintains an integrative map of data available on PFAS manufacturers, release, regional monitoring data, and occurrence in drinking water.3 It contains only 234 soil sample data points—all taken from one location in Minnesota in 2008. Furthermore, as described below, there are datasets in addition to those compiled by EPA that suggest PFAS will be ubiquitous at low levels in many, if not most, soils. The widespread detection of PFAS raises key questions about what soil concentrations should be considered impacted or potentially harmful and what levels should be considered background or unimpacted.
Determining background PFAS concentrations and identifying a range of PFAS concentrations in contaminated ecosystems can help NRCS or a private landowner evaluate whether PFAS present on agricultural lands result from ambient deposition or represent impacts of nearby sources or use of PFAS-impacted materials on site. Studies have assessed PFAS in impacted soils and at sites without a known PFAS source. Rankin et al. (2016) analyzed soil samples collected across the United States (including Hawaii, Alaska, and Puerto Rico), Canada, and Mexico, from locations with no or limited human impact. In North America, total PFCA and PFSA concentrations were found in the 0.15–6.08 µg/kg and 0.04–2.0 µg/kg ranges, respectfully. There were quantifiable concentrations of at least three PFCAs in all locations, and PFOA and PFOS were detected in samples from all 29 locations in the United States. Washington et al. (2019)
___________________
3 See EPA PFAS Analytic Tools, https://awsedap.epa.gov/public/extensions/PFAS_Tools/PFAS_Tools.html.
subsequently implemented a statistical analysis of the Rankin et al. (2016) dataset and found that mean PFCA background concentrations in the northern hemisphere were 0.0114 µg/kg (perfluorodecanoic acid [PFDA]) to 0.0583 µg/kg (perfluorohexanoic acid [PFHxA]). The mean background concentration of PFOS in the northern hemisphere was 0.0555 µg/kg. These data support the conclusion that, even without direct application of PFAS-contaminated inputs, PFAS can accumulate in the environment through long-range air transport (see section “Transport Via Water and Air” below; Rankin et al. 2016).
Background PFAS levels have also been assessed in some states. In Vermont, data were generated from 68 surface soil samples collected from state and municipal parks, forests, greens, and lawns within urban areas (Zhu et al. 2019). Perfluorononanoic acid (PFNA) and PFOS were detected in 100 percent of soil samples at concentrations of 0.051–5.0 µg/kg and 0.106–9.7 µg/kg, respectively. EPA statistical software ProUCL was used to establish background threshold values for 10 PFAS detection frequencies greater than 10 percent. The proposed values were 0.150 µg/kg (perfluorodecanesulfonic acid [PFDS]) to 3.40 µg/kg (PFOS). These values are higher than those estimated by Washington et al. (2019), which may reflect analysis of samples from urban areas rather than those with little to no human impact.
In Massachusetts, samples were collected from 100 sites that were distant from known or potential PFAS sources (e.g., state and municipal parks, woodlands, and other conservation areas; McIntosh et al. 2025). PFOA and PFOS were detected in 100 percent of soil samples at concentrations of 0.072–4.20 µg/kg and 0.190–6.00 µg/kg, respectively. Four other PFAS were detected in at least 80 percent of samples. Background threshold values for 12 PFAS-detection frequencies greater than 10 percent were estimated as the upper 95-percent confidence level on the 95th percentile of the concentration distribution (known as the upper tolerance level). The resulting values were 0.079 µg/kg (PFBS) to 3.25 µg/kg (PFOS). The study authors concluded that, based on the types and relative abundances of PFAS present, these background PFAS likely originated from long-range atmospheric transport, not from regional industrial or other sources. Based on samples collected from Maine and New Hampshire, McIntosh et al. (2025) also proposed background threshold values of 0.080 µg/kg (perfluoropentanoic acid [PFPeA]) to 0.648 µg/kg (PFOS) for Maine and 0.061 µg/kg (hexafluoropropylene oxide dimer acid [HFPO-DA]) to 2.41 µg/kg (PFOS) for New Hampshire.
Brusseau et al. (2020) compiled published literature data from more than 30,000 soil samples and from more than 2,500 sites throughout the world focused on maximum reported total PFAS, PFOA, and PFOS concentrations. The data included background soil concentrations from more than 1,400 sites (including Rankin et al. 2016) and from hundreds of primary source sites (e.g., PFAS manufacturing sites, fire-training areas, and locations where aqueous film-forming foam [AFFF] had been used) and secondary source sites (e.g., sites adjacent to primary sites or sites where PFAS-contaminated materials were utilized) where PFAS were directly or indirectly utilized. Data compiled by Brusseau et al. (2020) included samples from urban areas (e.g., parks) similar to those described in the Vermont study (Zhu et al. 2019) and samples from pristine areas with minimal human impact as described in Rankin et al. (2016). Maximum reported
PFOA and PFOS concentrations from background sites in the compiled datasets were 0.5–33 µg/kg and 3.1–126 µg/kg, respectively. Maximum PFOA or PFOS concentrations from locations categorized as background by Brusseau et al. (2020) tended to be one to three orders of magnitude lower than those found in primary or secondary contaminated sites (Table 2-1). However, the range of reported background concentrations is 1–2 orders of magnitude higher than studies summarized above. This difference likely reflects the inclusion of varied site types and the focus on maximum reported concentrations. Importantly, no statistical analyses of this compiled dataset have been implemented to understand what concentrations are likely to be most representative of background PFAS.
Information on background PFAS in soil and other media can help inform whether a site contains PFAS from ambient PFAS deposition (e.g., precipitation) or PFAS sources (e.g., manufacturing discharge, biosolids) and may help contextualize PFAS detections at agricultural facilities. However, results of studies to date highlight challenges in establishing background PFAS concentrations. There is no consensus regarding sampling location types that are defined as background, and studies summarized above suggest different results will be obtained from relatively pristine locations versus urban sampling locations that are not proximal to known PFAS sources. Additionally, studies have used varied sample preparation approaches and analytical methods, included different PFAS analyte lists, achieved different detection limits, and used different
TABLE 2-1 Maximum Reported PFAS in Soils Worldwide and in the United States
| Background Sites | Primary-Source Contaminated Sites | Secondary-Source Contaminated Sites | |
|---|---|---|---|
| ------------------------------------- µg/kg ------------------------------------- | |||
| Global | |||
| Total PFAS | <0.001–237 | ND | ND |
| Min. PFOA | 0.01 | 2 | 0.8 |
| Max. PFOA | 123.6 | 50,000 | 2,531 |
| Median PFOA | 2.7 | 83 | 38 |
| Min. PFOS | 0.003 | 0.4 | 0.4 |
| Max. PFOS | 162 | 460,000 | 5,500 |
| Median PFOS | 2.7 | 8,722 | 680.5 |
| United States | |||
| Total PFAS | <0.2–135 | ND | ND |
| Min. PFOA | 0.5 | 58 | 23.6 |
| Max. PFOA | 33 | 50,000 | 2,531 |
| Min. PFOS | 3.1 | 9,700 | 483 |
| Max. PFOS | 126 | 373,000 | 1,409 |
NOTE: ND = no data.
DATA SOURCE: Created using data from Tables 1, 2, and 3 in Brusseau et al. 2020.
quality assurance and quality control protocols; these differences challenge comparison of results across studies. As a result, caution should be exercised before using data from prior work to establish concentrations considered impacted versus unimpacted at agricultural facilities.
FATE AND TRANSPORT OF PFAS IN THE ENVIRONMENT
Once in the landscape, the behavior of PFAS in the soil and their transport outcomes depend on prevailing soil and PFAS properties, as well as topography, climate, and land management. These factors affect PFAS sorption to soils and air–water interfaces, leaching, runoff, wet and dry deposition, uptake by plants, and bioaccumulation. These effects on environmental media and plant and animal life influence what NRCS terms the resource base—that is, the condition of soil, water, air, plants, and animals, which NRCS often refers to as SWAPA.
Abiotic and Microbially Mediated PFAS Transformation
Though the carbon–fluorine bond is extremely difficult to break, most polyfluoroalkyl substances are susceptible to biotransformation via oxidation and microbial degradation. Microbial degradation of PFAS is considered a co-metabolic process (Wackett 2025). A few studies have claimed degradation with specific PFAAs as the sole carbon source; however, only partial defluorination was observed and other evidence within the studies was not consistent (e.g., presence of fluoride but no loss of parent compound; Chetverikov and Loginov 2019; Harris et al. 2022; Smorada et al. 2024).
The PFAS that break down into terminal PFAAs are known as precursors. Though intermediate PFAS transformation products may also persist in the environment for some time, precursor degradation ultimately yields PFAAs, including PFOA and PFOS (Box 2-2; Figure 2-4).
For FT-derived precursors, degradation typically ends in the formation of a suite of PFCAs, with the dominant PFCA corresponding to the length of the precursor’s perfluorinated carbon tail (Liu and Mejia Avendaño 2013; Figure 2-4A). For example, degradation of 8:2 fluorotelomer alcohol (8:2 FTOH) primarily yields PFOA as the major end product but also includes other shorter-chain PFCA products. In contrast, ECF-derived precursors ultimately degrade to PFSAs of the same perfluoroalkyl chain length originally present in the precursor (Mejia Avendaño and Liu 2015; Zhang et al. 2017; Figure 2-4B).
The half-lives of precursors and their intermediates in soils vary widely, often spanning several orders of magnitude (Guelfo et al. 2021). Degradation occurs significantly faster under aerobic conditions than in anaerobic settings. While environmental factors such as soil properties, climate, and vegetation do influence microbial degradation rates, the PFAS class itself is often the most important determinant of degradation half-life under a given set of conditions. Generally, FT-derived precursors and some ECF-derived compounds fall on the shorter end of the half-life spectrum because of the relatively easily attacked non-fluorinated linkages, such as the –CH2CH2– group found
BOX 2-2
PFAS and Persistence
For many legacy PFAS, such as PFOA and PFOS, no environmentally relevant degradation pathways are known under ambient conditions. These compounds persist indefinitely once released. Some PFAS, specifically polyfluoroalkyl substances, can undergo a degree of transformation in the environment, but their intermediate and terminal products are themselves PFAS. Stated differently, parent compounds may not be persistent in their original form, but their transformation products remain fluorinated and resistant to mineralization, and they ultimately transform into fully recalcitrant PFAS such as perfluoroalkyl acids (PFAAs). Thus, even when the parent molecule is not inherently a “forever chemical,” transformation processes yield formation of PFAS that are.
Polyfluoroalkyl substances that contain a trifluoromethyl (CF3) group and are capable of environmental transformation can form PFAAs. In some cases, such as molecules with a single CF3 group attached to a partially fluorinated or non-fluorinated structure, transformation may yield ultrashort-chain PFAAs such as trifluoroacetate (TFA). In this sense, PFAS have redefined the understanding of molecular persistence in environmental systems. Even when parent PFAS are not persistent in their initial form, they gain persistence through transformation into fully stable, recalcitrant endpoints.
in many FT-derived PFAS. Among fluorotelomers, for example, fluorotelomer alcohols degrade relatively quickly, with aerobic half-lives ranging from a few hours to less than 1 month. In contrast, fluorotelomer sulfonates degrade more slowly, with aerobic half-lives ranging from several months to a few years. For ECF-derived PFAS, certain intermediates—such as ethylfluorosulfonyloxyacetic acid (EtFOSAA) and methylfluorosulfonyloxyacetic acid (MeFOSAA)—can persist in soils for well over a year under laboratory conditions and for multiple years under field conditions. These persistent intermediates can lead to continued, in situ production of PFOS, contributing to long-term environmental contamination (see Figure 2-2).
The unfavorable thermodynamics of breaking the carbon–fluorine bond also leads to natural abiotic processes being limited for transforming PFAS. Soil minerals can react with natural constituents to form reactive oxygen species, such as hydroxyl radicals, that can then degrade other constituents. Potential abiotic processes include hydrolysis, hydroxylation, decarboxylation, and oxidation–reduction, all of which will be PFAS-dependent and only lead to a partial transformation result similar to biotransformation processes (Fang et al. 2025). These processes under natural soil conditions are slow and considered insignificant for PFAS mineralization. Considerable attention has been given to the role of biological enzymes and the use of added enzymes to promote PFAS degradation (Amin et al. 2025; Harris et al. 2025; Mekureyaw et al. 2025) or covalent binding of PFAS to organic matter, known as humification (Munir et al. 2025).
NOTE: Precursors of PFAAs may go through many intermediate steps, pathways, and rates before ultimately transforming into terminal microbial metabolites.
SOURCE: Original from Linda S. Lee, modified with permission.
Nevertheless, to date, only lab-scale studies have shown partial degradation similar to trends in biotransformation or humification. Applications at the field scale are likely to be made difficult by stability under environmental conditions, enzyme specificity, limited substrate range, and a lack of understanding of the enzymatic pathways.
Sorption
Sorption of PFAS by soils occurs through several mechanisms, including hydrophobic and electrostatic interactions, as well as hydrogen and covalent bonding (Higgins and Luthy 2006; Figure 2-5). Hydrophobic interactions take place between the perfluorocarbon tails of PFAS and soil organic matter (SOM), with increasing ionic strength increasing sorption. Electrostatic interactions, which are affected by pH, occur directly between the polar functional groups of PFAS and the charged components of soil, such as SOM or metal oxides (e.g., iron or aluminum oxides), or indirectly through cation bridging with divalent cations (Mejia-Avendaña et al. 2020; Mei et al. 2021; Kookana et al. 2023). Sorption to metal oxides can also occur via ligand exchange of hydroxyl groups, for example, the carboxylate head of PFCAs (Du et al. 2014). PFAA affinity to soil proteins has been attributed to electrostatic interactions between anionic PFAAs and positively charged amino and amide groups in proteins (F. Li et al. 2019). Hydrogen
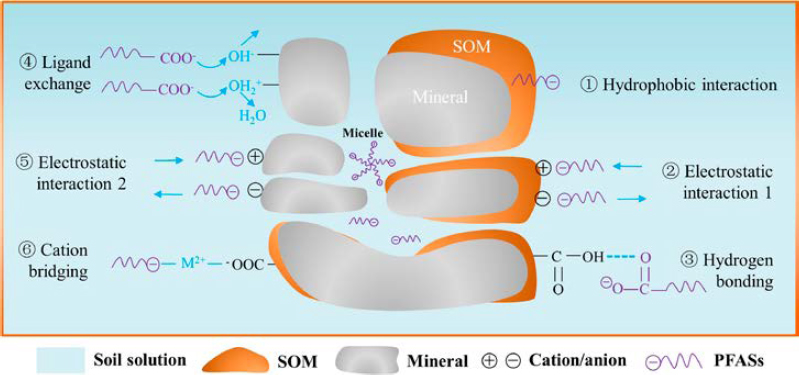
NOTE: SOM = soil organic matter.
SOURCE: Mei et al. 2021. CC BY-ND 4.0.
bonding of PFAAs with soil functional groups and covalent bonding with SOM have also been demonstrated in lab-scale spectroscopic studies (Zhu et al. 2021). In addition, PFAS can self-assemble into micelles, hemi-micelles, and versicular structures (Krafft 2025; Yan et al. 2025), which affects their interaction with soil surfaces and air–water interfaces (AWI), including potential entrapment in soil aggregates.
Perfluorocarbon chain length and the type of polar functional group are the main PFAS structural properties that influence sorption. Most sorption data to date are for PFAAs. Sorption is generally stronger as the chain length increases for PFAS with the same functional group (e.g., PFCAs or PFSAs) because the longer the chain, the more hydrophobic the PFAS (Sharifan et al. 2021). Different functional groups have different sorption behavior; for example, among the PFAAs, PFSAs exhibit stronger sorption than PFCAs of the same chain length. Zwitterionic and cationic PFAS may bind to greater magnitudes than PFAAs because of the dominance of negative charge sites on soils.
The charge state of a PFAS also affects the extent of sorption. Most PFAS, especially PFCAs and PFSAs, have relatively low acid dissociation constants (pKa values below 1.6 and 0.3, respectively) (Burns et al. 2008; Goss 2008; Rayne and Forest 2010; Vierke et al. 2013; Murillo-Gelvez et al. 2023), which means they exist as negatively charged anions under most environmental conditions (i.e., pH 4–9). Exceptions include FASAs, which have a pKa of ~6 and may exist in neutral or anionic form at environmentally relevant pH. In contrast, zwitterionic and cationic PFAS—many of which have been used historically in AFFF—exhibit different sorption behavior because of their positive or mixed charges and may bind more strongly to soil surfaces. Although soil organic carbon has traditionally been viewed as the primary driver of sorption for
hydrophobic chemicals, it alone often does not accurately predict PFAS sorption to many soil types and within soil profiles because of the charge of the polar functional group (Li et al. 2018; Kookana et al. 2023; Evich et al. 2025).
In addition, PFAS with polar functional groups are surfactants and therefore tend to accumulate at interfaces, especially the AWI in the vadose (unsaturated) zone (Brusseau 2018; Sharifan et al. 2021), akin to how soap suds collect at the AWI. The extent of this interfacial sorption (Kaw) increases with PFAS chain length (thus molar volume), as longer-chain PFAS have higher affinity for the AWI (Brusseau 2023; Endo et al. 2023). Decreasing soil particle size and moisture content both lead to higher interfacial surface area and thus more air–water interfacial partitioning of PFAS; interfacial partitioning also increases with increasing ionic strength of the porewater (Lyu and Brusseau 2020). Thus, the magnitude of sorption at the AWI is determined by soil properties, moisture content, and the PFAS AWI sorption coefficient. PFAS concentration can also affect sorption in the vadose zone because of both PFAS self-assembly mechanisms (micelle formation) and competitive sorption (Silva et al. 2021). Fate and transport models with PFAS-specific considerations for sorption to soil and the AWI are increasingly being used to understand and predict PFAS fate and transport (see Box 4-1 in Chapter 4), including in agricultural scenarios (Guo et al. 2022; Silva et al. 2022; Brusseau and Guo 2023; Smith et al. 2024; Liao et al. 2025; Doria-Manzur et al. 2026).
Plant Uptake
Many PFAS can move within plants through both passive and active transport mechanisms, allowing them to translocate from one part of the plant to another. The extent and pathway of PFAS uptake depend on several key factors, including the chemical structure of the PFAS (especially functional group and alkyl chain length), the initial concentration and profile of PFAS in environmental media (such as soil or water), plant type, and the chemical characteristics of the media—notably organic carbon content, surface charge, and pH (Ghisi et al. 2019; Costello and Lee 2024). Measured transfer factors of PFAS from soil to plant are also affected by the presence of precursors that can transform during the growing season. The PFAAs generated through precursor degradation are not captured in the soil concentration but are equally subject to plant uptake. In field studies, Simones et al. (2024) found PFOS precursor presence to be significantly associated with increasing PFOS transfer factors. Likewise, in a forage greenhouse study, Openiyi et al. (2025) attributed high transfer factors to measured precursor transformation. In addition, it is well established that uptake and accumulation of PFAS are distinctly higher in the vegetative parts of plants (leaves) than in storage organs (fruits, grains, tubers) (Stahl et al. 2009; Blaine et al. 2014; Wen et al. 2014; Wang et al. 2020; Lesmeister et al. 2021).
PFAS typically accumulate passively in root tissues, primarily via diffusion from high-concentration zones in the surrounding environment. This diffusion is strongly influenced by PFAS sorption to soil particles, which affects bioavailable concentrations in porewater, and by soil moisture content, which mediates PFAS mobility and access
to the root surface. Once at the root surface, PFAS accumulation is further influenced by the protein and lipid content of root tissues, which may enhance or inhibit binding. Root density and architecture affect the root’s contact area with soil and soil porewater where PFAS reside, as well how much of the soil is explored by the roots, thus affecting the extent of PFAS accumulation. However, molecular size and structure play a critical role in whether a PFAS can penetrate the root barrier. Some larger or more complex PFAS molecules may be restricted from entering root tissues or prevented from trans-locating further into the rest of the plant. PFAS interaction with the roots is defined by the root concentration factor—the ratio of PFAS concentration in the root to that in the surrounding media—and will be PFAS, plant, and media dependent. Additionally, in one greenhouse study, mixtures of PFAS were also noted to affect uptake, with PFOA uptake increasing when present in a mixture of PFAAs (Zhang et al. 2022).
Once PFAS have entered the root, translocation to above-ground plant parts (such as stems, leaves, and reproductive tissues) can occur and is often quantified by a translocation factor—the ratio of PFAS concentration in the above-ground tissues to that found in the root. This process happens primarily for PFAAs and is driven by transpiration-induced flow through the xylem, the vascular system that transports water and nutrients upward from the roots. Shorter-chain PFAS, which are less likely to sorb to soil particles, remain more freely dissolved in porewater and thus are more readily available for uptake and translocation through transpiration (Krippner et al. 2015; Wang et al. 2020; Costello and Lee 2024). Consequently, as chain length increases, both overall plant uptake and movement into above-ground tissues tend to decrease.
Nonetheless, some long-chain PFAS, including PFOS, have been detected in the leaves, stems, and seeds of grasses and other plants. For PFAS to reach reproductive tissues such as seeds (e.g., grains, beans, or corn kernels), they must be transferred from the xylem to the phloem, which transports nutrients throughout the plant. This xylem-to-phloem offloading may occur via nonselective substrate transporters, allowing certain PFAS to move into reproductive tissues (Yao et al. 2020; Gill et al. 2021). For example, Lazo and Lee (2024) observed primarily short-chain perfluorobutanoic acid (PFBA) and perfluoropentanoic acid (PFPeA) and did not observe PFSAs in the bean of soybean plants although PFOS soil concentrations were several hundred ppb (µg/kg). This result indicates that PFCAs, which mimic fatty acids, are likely to be inadvertently transported into the phloem, whereas the PFSAs are not. Trends from greenhouse and field studies to date show that levels of long-chain PFAS are half to three orders of magnitude lower in storage organs (fruits, grains, tubers) than in plant vegetative parts (Stahl et al. 2009; Blaine et al. 2014; Wen et al. 2014; Krippner et al. 2015; Wang et al. 2020; Lesmeister et al. 2021; Lazo and Lee 2024; Ortiz and Mallory 2025).
Transport Via Water and Air
PFAS present in soil can be transported through the environment via water by vertical leaching into deeper soil layers and groundwater and by surface runoff during precipitation and snowmelt events. The extent to which PFAS are transported by these
processes is strongly influenced by their sorption to soil surfaces as well as their accumulation at the AWI within the vadose (unsaturated) zone. The degree of PFAS leaching is determined by a combination of climatic and hydrologic conditions, soil hydraulic and chemical properties, and PFAS sorption, which is discussed above. Environmental factors—such as rainfall intensity and duration and snowmelt, as well as irrigation rates—play a particularly critical role in mobilizing PFAS via leaching and runoff (Borthakur et al. 2021c). These effects can be amplified by the release and downward transport of soil colloids during infiltration events, which may carry sorbed PFAS deeper into the soil profile (Borthakur et al. 2021a). Additionally, natural weathering and seasonal drainage cycles can remobilize PFAS that had previously been bound to soil particles (Borthakur et al. 2021b).
As discussed above, short-chain PFAS sorb less than longer-chain PFAS, leading to their higher propensity to be in soil porewater. Short-chain PFAS are thus more available for plant uptake or leach more readily than long-chain PFAS. These characteristics create an inverse relationship between soil depth and the long-chain PFAS concentration. Specifically, long-chain PFAS tend to accumulate in surface soils, while short-chain PFAS migrate deeper or are more likely to be absorbed by vegetation (Alvarez-Ruiz et al. 2024; Peter et al. 2025). Time since application and the balance of rainfall events leading to leaching versus to runoff control what PFAS may remain in surface soils and thus be subject to runoff. The persistence of long-chain PFAS in surface soils makes them more susceptible to overland transport via runoff, particularly during high-intensity or frequent precipitation events (Peter et al. 2025). However, any PFAS present in surface soils are subject to runoff. Therefore, short-chain PFAS are also frequently found in runoff; they have been observed often in urban water systems (Kali et al. 2025; Saleh et al. 2025; Zhang et al. 2025). Often attached to suspended solids, PFAS can be carried into nearby surface water bodies. PFAS-laden runoff that enters streams or closed or semi-closed aquatic systems, such as ponds and lakes, can increase exposure of animals that use those water bodies as drinking sources (e.g., livestock) or habitat (e.g., fish) (see section “Bioaccumulation” below). In tile-drained agricultural fields (perforated corrugated piping placed approximately 1 meter below the surface), vertical PFAS transport to groundwater is short-circuited and diverted to the agricultural ditch network, which flows into larger flowing surface water bodies (Gottschall et al. 2017; Peter and Lee 2025).
PFAS enter the atmosphere through manufacturing emissions, landfill waste combustion, volatilization, and wind-blown particles from PFAS-contaminated lands and through aerosols produced during the use of AFFFs or spraying PFAS-containing pesticides, as well as from water bodies including lakes, the ocean, and aeration basins (Faust 2023; Kourtchev et al. 2023; Lin et al. 2024; Pandamkulangara Kizhakkethil et al. 2024). Once in the atmosphere, precursor PFAS such as FTOHs can undergo oxidative transformation to the terminal PFAAs (Ellis et al. 2004; Wallington et al. 2006; D’eon et al. 2006; Nielsen et al. 2007; Faust 2023). Airborne PFAS undergo wet/dry deposition through rain events and particle settling. For example, PFAS concentrations up to 200 µg/kg have been reported in agricultural soils near a fluorochemical manufacturing plant
(Liu et al. 2019). Additionally, airborne PFAS (whether sorbed to particles or present as aerosols) can be transported via air currents for several months and well beyond initial emission locations (Kourtchev et al. 2024), thus contributing to background levels of PFAS in terrestrial and aquatic ecosystems.
The myriad of PFAS uses coupled to long-range atmospheric transport has led to an increasing frequency of PFAS detections in rainwater, in some instances above prevailing regulatory limits or guidelines (Shimizu et al. 2021; Cousins et al. 2022; Pfotenhauer et al. 2022). Natural precipitation has been shown to contribute to PFAS accumulation in North American and Asian topsoils (Scott et al. 2006; Chen et al. 2019; Gewurtz et al. 2019). A study by Shimizu et al. (2021) showed that the annual flux contribution of PFAS was over an order of magnitude greater via wet versus dry deposition. Shimizu et al. (2021) suggested that PFAS incorporation into natural precipitation may be localized, yet PFAS have been found at both poles of the globe (Del Vento et al. 2012; Xie et al. 2015).
Bioaccumulation
Because PFAS are hydrophobic, lipophobic, and proteinophilic, as well as environmentally persistent, they tend to accumulate in biota, including human and animal tissues (De Silva et al. 2021; Savoca and Pace 2021). As with their stronger sorption to soils, longer-chain PFAS have a higher bioaccumulation propensity and take longer to be eliminated from the body (body t1/2). PFAS affinity for proteins has made some PFAS highly bioaccumulative, particularly as perfluorocarbon chain length increases. PFAS accumulation has been found in muscle tissue, liver, kidneys, and breast milk of humans and animals globally (Custer et al. 2014; Death et al. 2021; Wood et al. 2021; Coy et al. 2022; Khan et al. 2023; Witt et al. 2024). PFAS have even been found in brains (Khalid et al. 2025), most likely transported through phospholipids. PFAS have been found in the blood samples of nearly all human beings (Jian et al. 2018; Kuo et al. 2023) and are frequently detected in wildlife, domesticated animal products (milk, meat, eggs, fish) (Göckener et al. 2020; Lasters et al. 2022), and a variety of foods. Some PFAS, such as PFOS, tend to bioaccumulate effectively in animal tissue, even when feed or forage contain low PFAS concentrations, resulting in livestock animal products (e.g., milk and meat) with high levels of PFAS (Fitzgerald et al. 2025). Likewise, relatively low PFAS concentrations in water can lead to elevated concentrations in humans and other biota.
PFAS that bioaccumulate in one species can transfer to another through food webs. New Mexico issued a health advisory in January 2025 to hunters and anyone consuming game from Holloman Lake after samples of waterfowl and rodent tissue collected from the habitat were found to contain the highest PFAS levels ever recorded in wild animals (Witt et al. 2024; Daniel B. Stephens & Associates, Inc. 2025; New Mexico Department of Health 2025). Stormwater runoff and treated sewage from nearby Holloman Air Force Base were found to outflow into the lake, potentially contributing to this problem. In a
separate investigation, a freshwater food web in a North Carolina/South Carolina river likely contaminated by municipal and agricultural sources identified high concentrations of several PFAS in biofilm, aquatic insects, and fish, suggesting that insects may bioaccumulate PFAS from feeding on biofilm, and fish may do the same by feeding on insects (Penland et al. 2020). However, bioaccumulation of PFAS varied by type of PFAS, and organisms were likely exposed via water in addition to diet. Within tributaries of the Hudson River in New York, elevated levels of PFAS have been measured in benthic macroinvertebrates, even though concentrations of PFAS in surrounding surface water and sediment were not exceedingly high; these results indicate bioaccumulation in the macroinvertebrates, with implications for the fish that feed on them (Brase et al. 2022). PFAS bioaccumulation has also been reported in feeding experiments involving crickets (McDermett et al. 2022), earthworms (Rich et al. 2015), and toads (East et al. 2025). In studies conducted in Europe, PFAS have been found in honeybees (Müller et al. 2025) and honey (Surma et al. 2016).
Biomagnification
Bioaccumulation of PFAS through food webs often leads to biomagnification. Biomagnification results when ingested compounds accumulate and become more concentrated in the animal’s tissues because elimination rates are slower than accumulation rates (Death et al. 2021; Fremlin et al. 2023; George et al. 2023; Khan et al. 2023; Miranda et al. 2023; Adeogun et al. 2024). Biomagnification is affected by both PFAS properties and ecological factors such as habitat (e.g., temperature) and organism characteristics (e.g., size, age, and sex; Burkhard 2021; Cara et al. 2022; Adeogun et al. 2024). Trophic magnification factors (TMFs) are used to describe quantitatively the increase in concentration with increasingly higher trophic levels (Death et al. 2021; Adeogun et al. 2024; Kelly et al. 2024; Ricolfi et al. 2025). TMFs are higher for longer-chain PFAS and tend to be higher for PFSAs compared to PFCAs with a similar chain length (Adeogun et al. 2024). For PFAS-contaminated areas, similar to the legacy chemicals mercury and polychlorinated biphenyls, fish consumption recommendations include restricting the amount of higher-trophic-level fish consumed per unit time (ECOS 2025). Of particular concern in the livestock industry are dairy and meat products. The high affinity of PFAS for proteins (Martin et al. 2013; Ng and Hungerbühler 2013), which are high in meat and milk, leads to bioaccumulation and subsequent biomagnification. Livestock consume large amounts of plant material (e.g., typically 25 pounds or more per day for cows), which can lead to unacceptable PFAS levels in meat and milk even for low PFAS-containing feed. There are currently no formal maximum contaminant levels for PFAS in milk and meat, but Maine has set action levels when PFOS exceed 210 parts per trillion (ng/kg) in milk (Maine Center for Disease Control and Prevention 2017) and 3.4 ppb (µg/kg) in meat (Maine Center for Disease Control and Prevention 2020), based on potential risk to children. Another concern due to biomagnification is PFAS levels in higher-trophic-level fish in surface water bodies receiving PFAS-contaminated runoff or deposition. For example, Hoskins
et al. (2023) observed PFOS at several hundred ppb (µg/kg) in largemouth bass collected in a pond receiving runoff from nearby farms that had received biosolids over time.
ENTRY AND CYCLING OF PFAS IN AGRICULTURAL SYSTEMS
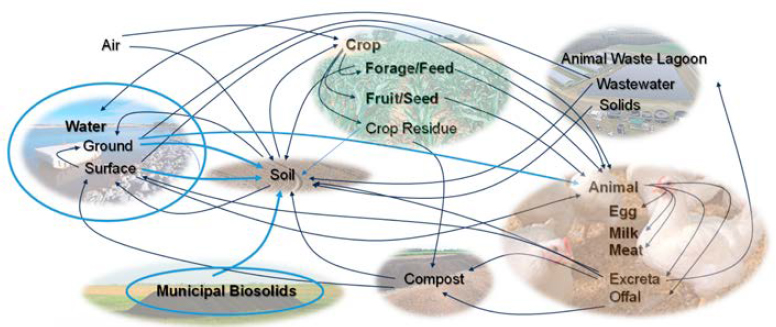
Like all contaminants, PFAS can enter an agricultural system through a point or a nonpoint source. An example of point-source PFAS contamination is the release by military installations of AFFF into groundwater that is eventually used by farmers for livestock watering or crop irrigation.4 Documented cases of such contamination include Highland Dairy near Cannon Air Force Base in New Mexico (Amec Foster Wheeler Programs, Inc. 2018; NASEM 2022; State of New Mexico 2022) and Venetucci Farm near Peterson Air Force Base in Colorado (Aerostar SES LLC 2017; Gaulke 2020). Nonpoint-source contamination can enter via any number of routes (Figure 2-6). PFAS from off-site emissions from any unknown source(s) may be introduced via fate and transport processes, including atmospheric transport (e.g., wind-born particulate deposition, precipitation), impacted groundwater, land-applied wastewater effluent, and impacted surface water. Products applied to agricultural fields may also introduce PFAS impacts. Organic soil amendments (e.g., biosolids, manure, compost; Box 2-3) are increasingly scrutinized as a source of introduction to agricultural operations. PFAS have also been detected in pesticides, but there is debate about whether the container or formulation is the source of the PFAS as well as which PFAS occur and at what levels, even within a single pesticidal formulation (PEER 2020). Additionally, fluorinated pesticides can degrade to ultra short-chain PFAS trifluoroacetate (TFA) (Ellis and Mabury 2000; EFSA 2014). Other potential sources include synthetic fertilizers, commercial livestock feed (Choi et al. 2023), and animal bedding (Fernandes et al. 2019), which can include paper and paper industry residuals that may contain PFAS (Choi et al. 2019). Lastly, PFAS-impacted water sources can affect aquaculture fish, and studies have identified PFAS in commercial fish feed used in freshwater aquaculture (X. Li et al. 2019; Rushing et al. 2023).
Once PFAS are introduced to agricultural facilities, there is the potential for on-farm cycling (Figures 2-6 and 2-7). Animal manure represents one potential source of PFAS cycling at facilities following exposure of livestock to PFAS via contaminated feed or drinking water. A recent study identified PFAS in 17 of 21 manure samples from European dairy cows, pigs, and poultry (Munoz et al. 2022). The management of as much as 1.4 billion tons (wet weight) of animal manure generated in the United States each year is already a significant challenge (Pagliari et al. 2020). High manure volumes, low value, and high disposal costs sometimes lead to over application of manure on site (Pagliari et al. 2020; Lim et al. 2023). If PFAS are present in the manure, PFAS will be distributed in the field and potentially be taken up by plants, leach into groundwater, and leave the field through runoff. This movement into plants and water can then lead
___________________
4 This groundwater is also often a source of drinking water for farmers, other rural residents, and municipalities.
BOX 2-3
Organic Soil Amendments
Organic soil amendments are natural materials added to soil to improve its physical, chemical, and biological properties. They can increase the amount of available organic material in soils; improve the structure, water retention, and nutrient availability of soils; and support beneficial soil microbesa (Siedt et al. 2021). Organic soil amendments are primarily derived from plant, animal, or microbial sources and are used to enhance soil quality, structure, and health (Larney and Angers 2012). Common types include animal manure, biochar, biosolids, compost, and crop residues (Urra et al. 2019; Sakhiya et al. 2020; Malone et al. 2023).
Animal manure is everything that would be considered animal waste, both solid and liquid, in addition to any animal bedding materials (Urra et al. 2019). In U.S. agricultural systems, manure is primarily from pigs, cows, and poultry (Lim et al. 2023). Manure may be applied raw or after undergoing a management process, such as composting or heat treatment (Lim et al. 2023).
Biosolids are nutrient-rich organic materials produced from the treatment of sewage. Solid waste is separated from liquid waste and then stabilized and treated to reduce pathogens and contaminants (Lu et al. 2012; Poornima et al. 2022; Elgarahy et al. 2024). They are often used as soil amendments in agriculture because of their high content of essential plant nutrients and organic matter and their affordability. In the United States, biosolids cannot be applied to land unless heavy metal and pathogen levels are under specified thresholds (Lu et al. 2012). However, there are currently no thresholds for PFAS in biosolids at the federal level.
Biochar, a stable, carbon-rich material made by pyrolyzing organic matter under low-oxygen conditions, helps build long-lasting soil organic carbon. Its high surface area enhances water and nutrient retention, improving soil health and plant nutrient availability (Weber and Quicker 2018; Gao et al. 2019). Made from materials such as wood and crop residues, its properties vary with feedstock and pyrolysis conditions, influencing its effects on soil (Joseph et al. 2021).
Additional examples of organic soil amendments are compost, crop residue, and green manure (Urra et al. 2019). Compost can come from aerobic and anaerobic sources, and it can be made from a variety of organic materials, including biosolids from municipal wastewater (Urra et al. 2019) and manures. Crop residues are the parts of the plant that remain in the fields after harvest (Lal 2005). Green manure refers to plants that are incorporated into soil while plants are green or soon after they mature (Goss et al. 2013).
a See Role of Organic Matter, https://www.nrcs.usda.gov/conservation-basics/natural-resource-concerns/soils/soil-health/role-of-organic-matter.
to further livestock exposure to PFAS via feed and water. Land application of biosolids or compost that is contaminated with PFAS can cause similar movement of PFAS within an agricultural facility, including into on-site water sources that may be used for irrigation or livestock feed and water.
Furthermore, manure is not always economical to distribute as a fertilizer, so the U.S. Department of Agriculture (USDA) recommends management practices such as the separation of liquids and solids, composting manure, and using manure to generate biogas through anaerobic digestion to raise its value (Lim et al. 2023). The latter may be coupled with the use of digestate as fertilizer, separation of digestate fibers for use in products such as bedding, or both. However, little is known about the impact of these strategies on PFAS composition and concentrations. Studies with municipal biosolids have shown that treatments such as digestion and composting can cause transformation of polyfluoroalkyl substances (i.e., precursors) to terminal, PFAA daughter products (Thompson et al. 2023; Alukkal et al. 2024a,b). Temperature, pH, and oxygen content will affect the rates of PFAS degradation, with aerobic digestion promoting faster degradation of precursors to intermediates to terminal PFAAs than anaerobic digestion (Alukkal et al. 2024a,b). For municipal solids treated in storage nitrification–denitrification reactors, most quantifiable PFAS increased in concentration from both breakdown of known precursors and reduced analytical matrix effects (Alukkal et al. 2024b). Therefore, while PFAS-specific data for manure are still limited, on-site applications of liquid or solid manure fractions or compost or use of digestate fiber as bedding may also promote on-site PFAS cycling.
Another example of on-farm PFAS cycling is plant uptake of PFAS from impacted soils followed by re-release during plant senescence. When the plant decomposes, PFAS reenters the soil and can eventually be taken up by subsequent living plants or leached into groundwater. Plants may be consumed by humans or animals, and groundwater may be used as a source of irrigation for crops or forage or for human and livestock consumption.
PFAS can also migrate off farm via water, soil, or farm products. PFAS from agricultural facilities that drain into groundwater, tile drainage, and surface water can migrate off site. Similarly, wildlife may consume PFAS-impacted water or forage from affected agricultural facilities and migrate off site. PFAS-impacted particulates from farm activities can undergo off-site atmospheric transport. Although not a primary focus of this report, it is also important to consider that PFAS-impacted agricultural facilities have implications for U.S. consumers, who may consume produce, meat, eggs, and dairy that have been contaminated with PFAS; a recent study detected PFAS in fresh and canned vegetables and milk purchased from grocery stores (Yang et al. 2023). Lastly, it is possible to transfer PFAS impacts between agricultural facilities via sale of agricultural products. For example, sale of PFAS-impacted feed may transfer impacts to livestock of another facility. Although some of these considerations (e.g., consumer exposure) are beyond the direct purview of USDA, off-farm considerations facilitate a comprehensive understanding of the problems associated with PFAS at U.S. agricultural facilities.
SOURCE: Photo courtesy of U.S. Department of Agriculture–Agricultural Research Service.
SCOPE OF THE CHALLENGE
As discussed above, the scope of the PFAS challenge in U.S. agriculture is not well characterized (see section “Occurrence of PFAS in U.S. Soils”). However, geospatial data on PFAS sources5 and detections compiled by EPA show that the footprint of PFAS on the landscape is large (Figure 2-8). Assuming PFAS contamination on agricultural lands is widespread, the severity of the problem is still difficult to assess. Data from various sources collected by EPA indicate where detection has occurred, but most data points do not convey the amount of one or more PFAS detected or the degree to which the detected PFAS present harm to human, livestock, or environmental health. Maximum contaminant levels in drinking water have been set for some PFAS,6 but no such threshold currently exists on a federal level for soil, groundwater, surface water, livestock, milk, or food in general.
As discussed in Chapter 1, there is no easy way to treat PFAS-impacted media. With no readily available, affordable way to remediate, the question then for NRCS is how can the agency best use its capabilities to address on-farm PFAS contamination and mitigation? The next chapter reviews the relevant programs within NRCS’s remit, the conservation practices it supports, and the intersection of both programs and practices with PFAS. Chapter 4 then explores how decisions could be made, given limited information and, presumably, constrained financial resources.
___________________
5 Sources include drinking water samples, industry and wastewater treatment facility discharge reports, gaseous emissions from large emitting facilities, groundwater and surface water, sediment, and fish tissue analysis, military installations, PFAS spill sites or release events, Superfund sites with PFAS detections, and PFAS manufacturing facilities.
6 In 2024, EPA established maximum contaminant levels (MCLs) for six PFAS in drinking water under the Safe Water Drinking Act (EPA 2024b). In May 2025, EPA announced its intention to rescind the MCLs for all but PFOS and PFOA (EPA 2025).
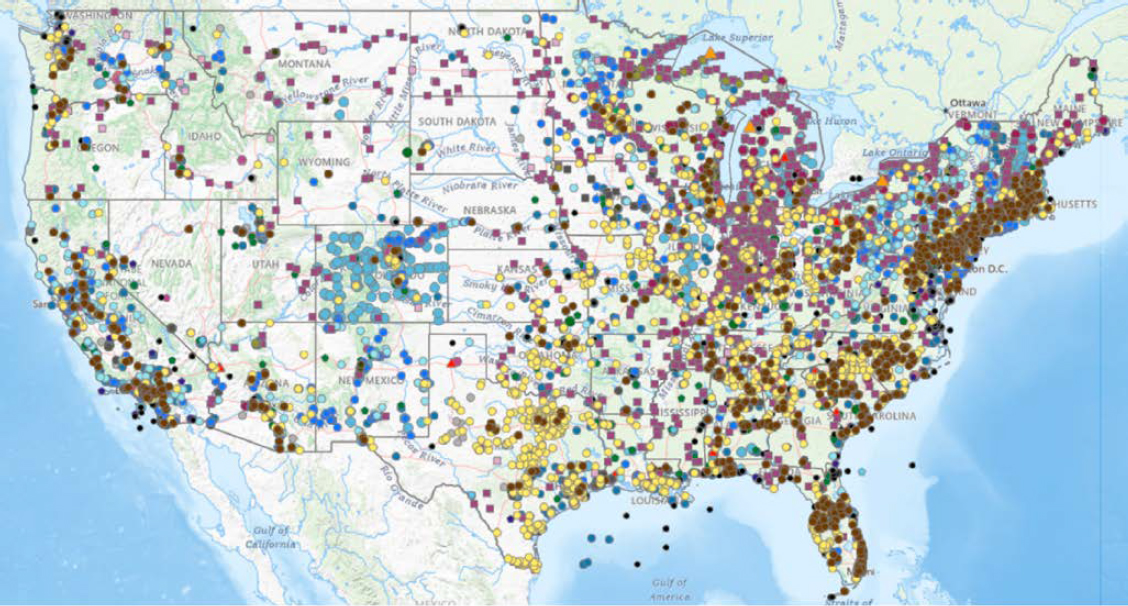
NOTE: Sites with no detection of PFAS are not displayed. MRL = minimum reporting level.
SOURCE: Generated with data from EPA PFAS Analytics Tools, https://echo.epa.gov/trends/pfas-tools (accessed September 25, 2025).

REFERENCES
Adeogun, Aina O., Azubuike V. Chukwuka, Oju R. Ibor, Alexandros G. Asimakopoulos, Junjie Zhang, and Augustine Arukwe. 2024. “Occurrence, Bioaccumulation and Trophic Dynamics of Per- and Polyfluoroalkyl Substances in Two Tropical Freshwater Lakes.” Environmental Pollution 346: 123575. https://doi.org/10.1016/j.envpol.2024.123575.
Aerostar SES LLC. 2017. Final Site Inspection Report of Aqueous Film Forming Foam Areas at Peterson Air Force Base El Paso County, Colorado. Air Force Civil Engineer Center.
Alukkal, Caroline Rose, Linda S. Lee, and Dana J. Gonzalez. 2024a. “Understanding the Impact of Pre-Digestion Thermal Hydrolysis Process on PFAS in Anaerobically Digested Biosolids.” Chemosphere 365: 143406. https://doi.org/10.1016/j.chemosphere.2024.143406.
Alukkal, Caroline Rose, Linda S. Lee, and Kevin Staton. 2024b. “Per- and Polyfluoroalkyl Substances Behavior: Insights from Autothermal Thermophilic Aerobic Digestion - Storage Nitrification-Denitrification Reactors.” Chemosphere 365: 143357. https://doi.org/10.1016/j.chemosphere.2024.143357.
Alvarez-Ruiz, Rodrigo, Linda S. Lee, and YounJeong Choi. 2024. “Fate of Per- and Polyfluoroalkyl Substances at a 40-Year Dedicated Municipal Biosolids Land Disposal Site.” Science of The Total Environment 954: 176540. https://doi.org/10.1016/j.scitotenv.2024.176540.
Amec Foster Wheeler Programs, Inc. 2018. Site Inspection of Aqueous Film Forming Foam (AFFF) Release Areas. Final Site Inspection Report, Cannon Air Force Base. Air Force Civil Engineer Center. Joint Base Antonio–Lackland, Texas.
Amin, Khadije Ahmad, Ashfaq Ahmad, Sumayya Al Ali, Aryam Alkaabi, Ghaliah Alazem, Hilu Amreen, Habiba AlSafar, and Syed Salman Ashraf. 2025. “Enzyme-Mediated Biodegradation of Per- and Polyfluoroalkyl Substances (PFAS): Challenges, Opportunities, and Future Directions.” Chemical Engineering Journal Advances 24: 100910. https://doi.org/10.1016/j.ceja.2025.100910.
Blaine, Andrea C., Courtney D. Rich, Erin M. Sedlacko, Lakhwinder S. Hundal, Kuldip Kumar, Christopher Lau, Marc A. Mills et al. 2014. “Perfluoroalkyl Acid Distribution in Various Plant Compartments of Edible Crops Grown in Biosolids-Amended Soils.” Environmental Science & Technology 48 (14): 7858–7865. https://doi.org/10.1021/es500016s.
Borthakur, Annesh, Brian K. Cranmer, Gregory P. Dooley, Jens Blotevogel, Shaily Mahendra, and Sanjay K. Mohanty. 2021a. “Release of Soil Colloids during Flow Interruption Increases the Pore-Water PFAS Concentration in Saturated Soil.” Environmental Pollution 286: 117297. https://doi.org/10.1016/j.envpol.2021.117297.
Borthakur, Annesh, Patience Olsen, Gregory P. Dooley, Brian K. Cranmer, Unnati Rao, Eric M. V. Hoek, Jens Blotevogel et al. 2021b. “Dry-Wet and Freeze-Thaw Cycles Enhance PFOA Leaching from Subsurface Soils.” Journal of Hazardous Materials Letters 2: 100029. https://doi.org/10.1016/j.hazl.2021.100029.
Borthakur, Annesh, Meng Wang, Meng He, Katia Ascencio, Jens Blotevogel, David T. Adamson, Shaily Mahendra, and Sanjay K. Mohanty. 2021c. “Perfluoroalkyl Acids on Suspended Particles: Significant Transport Pathways in Surface Runoff, Surface Waters, and Subsurface Soils.” Journal of Hazardous Materials 417: 126159. https://doi.org/10.1016/j.jhazmat.2021.126159.
Botelho, Julianne Cook, Kayoko Kato, Lee-Yang Wong, and Antonia M. Calafat. 2025. “Per- and Polyfluoroalkyl Substances (PFAS) Exposure in the U.S. Population: NHANES 1999–March 2020.” Environmental Research 270: 120916. https://doi.org/10.1016/j.envres.2025.120916.
Brase, Richard A., Holly E. Schwab, Lingyun Li, and David C. Spink. 2022. “Elevated Levels of Per- and Polyfluoroalkyl Substances (PFAS) in Freshwater Benthic Macroinvertebrates from the Hudson River Watershed.” Chemosphere 291: 132830. https://doi.org/10.1016/j.chemosphere.2021.132830.
Brusseau, Mark L. 2018. “Assessing the Potential Contributions of Additional Retention Processes to PFAS Retardation in the Subsurface.” Science of The Total Environment 613–614: 176–185. https://doi.org/10.1016/j.scitotenv.2017.09.065.
Brusseau, Mark L. 2023. “QSPR-Based Prediction of Air-Water Interfacial Adsorption Coefficients for Nonionic PFAS with Large Headgroups.” Chemosphere 340: 139960. https://doi.org/10.1016/j.chemosphere.2023.139960.
Brusseau, Mark L., and Bo Guo. 2023. “Revising the EPA Dilution-Attenuation Soil Screening Model for PFAS.” Journal of Hazardous Materials Letters 4: 100077. https://doi.org/10.1016/j.hazl.2023.100077.
Brusseau, Mark L., R. Hunter Anderson, and Bo Guo. 2020. “PFAS Concentrations in Soils: Background Levels Versus Contaminated Sites.” Science of The Total Environment 740: 140017. https://doi.org/10.1016/j.scitotenv.2020.140017.
Buck, Robert C., James Franklin, Urs Berger, Jason M. Conder, Ian T. Cousins, Pim de Voogt, Allan Astrup Jensen et al. 2011. “Perfluoroalkyl and Polyfluoroalkyl Substances in the Environment: Terminology, Classification, and Origins.” Integrated Environmental Assessment and Management 7 (4): 513–541. https://doi.org/10.1002/ieam.258.
Burkhard, Lawrence P. 2021. “Evaluation of Published Bioconcentration Factor (BCF) and Bioaccumulation Factor (BAF) Data for Per- and Polyfluoroalkyl Substances across Aquatic Species.” Environmental Toxicology and Chemistry 40 (6): 1530–1543. https://doi.org/10.1002/etc.5010.
Burns, Darcy C., David A. Ellis, Hongxia Li, Colin J. McMurdo, and Eva Webster. 2008. “Experimental pKa Determination for Perfluorooctanoic Acid (PFOA) and the Potential Impact of pKa Concentration Dependence on Laboratory-Measured Partitioning Phenomena and Environmental Modeling.” Environmental Science & Technology 42 (24): 9283–9288. https://doi.org/10.1021/es802047v.
Cara, Byns, Teunen Lies, Groffen Thimo, Lasters Robin, and Bervoets Lieven. 2022. “Bioaccumulation and Trophic Transfer of Perfluorinated Alkyl Substances (PFAS) in Marine Biota from the Belgian North Sea: Distribution and Human Health Risk Implications.” Environmental Pollution 311: 119907. https://doi.org/10.1016/j.envpol.2022.119907.
Chen, Hao, Lu Zhang, Mengqi Li, Yiming Yao, Zhen Zhao, Gabriel Munoz, and Hongwen Sun. 2019. “Per- and Polyfluoroalkyl Substances (PFASs) in Precipitation from Mainland China: Contributions of Unknown Precursors and Short-Chain (C2–C3) Perfluoroalkyl Carboxylic Acids.” Water Research 153: 169–177. https://doi.org/10.1016/j.watres.2019.01.019.
Chetverikov, S. P., and O. N. Loginov. 2019. “A New Ensifer adhaerens Strain M1 is Capable of Transformation of Perfluorocarboxylic Acids.” Microbiology 88 (1): 115–117. https://doi.org/10.1134/S0026261718060085.
Choi, Youn Jeong, Rooney Kim Lazcano, Peyman Yousefi, Heather Trim, and Linda S. Lee. 2019. “Perfluoroalkyl Acid Characterization in U.S. Municipal Organic Solid Waste Composts.” Environmental Science & Technology Letters 6 (6): 372–377. https://doi.org/10.1021/acs.estlett.9b00280.
Choi, Youn Jeong, Linda S. Lee, Tyler D. Hoskins, Mahsa Modiri Gharehveran, and Maria S. Sepúlveda. 2023. “Occurrence and Implications of Per and Polyfluoroalkyl Substances in Animal Feeds Used in Laboratory Toxicity Testing.” Science of The Total Environment 867: 161583. https://doi.org/10.1016/j.scitotenv.2023.161583.
Costello, M. Christina Schilling, and Linda S. Lee. 2024. “Sources, Fate, and Plant Uptake in Agricultural Systems of Per- and Polyfluoroalkyl Substances.” Current Pollution Reports 10 (4): 799–819. https://doi.org/10.1007/s40726-020-00168-y.
Cousins, Ian T., Jana H. Johansson, Matthew E. Salter, Bo Sha, and Martin Scheringer. 2022. “Outside the Safe Operating Space of a New Planetary Boundary for Per- and Polyfluoroalkyl Substances (PFAS).” Environmental Science & Technology 56 (16): 11172–11179. https://doi.org/10.1021/acs.est.2c02765.
Coy, Carrie O., Alexandra N. Steele, Sara A. Abdulelah, Rachelle M. Belanger, Karen G. Crile, Louise M. Stevenson, and Paul A. Moore. 2022. “Differing Behavioral Changes in Crayfish and Bluegill under Short- and Long-Chain PFAS Exposures: Field Study in Northern Michigan, USA.” Ecotoxicology and Environmental Safety 247: 114212. https://doi.org/10.1016/j.ecoenv.2022.114212.
Custer, Christine M., Thomas W. Custer, Paul M. Dummer, Matthew A. Etterson, Wayne E. Thogmartin, Qian Wu, Kurunthachalam Kannan et al. 2014. “Exposure and Effects of Perfluoroalkyl Substances in Tree Swallows Nesting in Minnesota and Wisconsin, USA.” Archives of Environmental Contamination and Toxicology 66 (1): 120–138. https://doi.org/10.1007/s00244-013-9934-0.
Daniel B. Stephens & Associates, Inc. 2025. Ecological Research on PFAS Contamination of Wildlife at Holloman Lake. New Mexico Environment Department.
Dasu, Kavitha, and Linda S. Lee. 2016. “Aerobic Biodegradation of Toluene-2,4-Di(8:2 Fluorotelomer Urethane) and Hexamethylene-1,6-Di(8:2 Fluorotelomer Urethane) Monomers in Soils.” Chemosphere 144: 2482–2488. https://doi.org/10.1016/j.chemosphere.2015.11.021.
Death, Clare, Cameron Bell, David Champness, Charles Milne, Suzie Reichman, and Tarah Hagen. 2021. “Per- and Polyfluoroalkyl Substances (PFAS) in Livestock and Game Species: A Review.” Science of The Total Environment 774: 144795. https://doi.org/10.1016/j.scitotenv.2020.144795.
D’eon, Jessica C., Michael D. Hurley, Timothy J. Wallington, and Scott A. Mabury. 2006. “Atmospheric Chemistry of N-Methyl Perfluorobutane Sulfonamidoethanol, C4F9SO2N(CH3) CH2CH2OH: Kinetics and Mechanism of Reaction with OH.” Environmental Science & Technology 40 (6): 1862–1868. https://doi.org/10.1021/es0520767.
De Silva, Amila O., James M. Armitage, Thomas A. Bruton, Clifton Dassuncao, Wendy Heiger-Bernays, Xindi C. Hu, Anna Kärrman et al. 2021. “PFAS Exposure Pathways for Humans and Wildlife: A Synthesis of Current Knowledge and Key Gaps in Understanding.” Environmental Toxicology and Chemistry 40 (3): 631–657. https://doi.org/10.1002/etc.4935.
Del Vento, Sabino, Crispin Halsall, Rosalinda Gioia, Kevin Jones, and Jordi Dachs. 2012. “Volatile Per- and Polyfluoroalkyl Compounds in the Remote Atmosphere of the Western Antarctic Peninsula: An Indirect Source of Perfluoroalkyl Acids to Antarctic Waters?” Atmospheric Pollution Research 3 (4): 450–455. https://doi.org/10.5094/APR.2012.051.
Dinglasan-Panlilio, Mary Joyce A., and Scott A. Mabury. 2006. “Significant Residual Fluorinated Alcohols Present in Various Fluorinated Materials.” Environmental Science & Technology 40 (5): 1447–1453. https://doi.org/10.1021/es051619+.
Doria-Manzur, Alonso, Evan P. Gray, Summer S. Streets, and Jennifer L. Guelfo. 2026. “Per- and Polyfluoroalkyl Substances (PFAS) Transport from Biosolids-Amended Soils: An Experimental and Numerical Approach.” Water Research 288: 124674. https://doi.org/10.1016/j.watres.2025.124674.
Du, Ziwen, Shubo Deng, Yue Bei, Qian Huang, Bin Wang, Jun Huang, and Gang Yi. 2014. “Adsorption Behavior and Mechanism of Perfluorinated Compounds on Various Adsorbents—a Review.” Journal of Hazardous Materials 274: 443–454. https://doi.org/10.1016/j.jhazmat.2014.04.038.
East, Andrew G., Mike Simini, Emily E. Stricklin, Guilherme R. Lotufo, Jennifer L. Guelfo, Zhao Yang, Travis Gallo, Michael J. Quinn, and Roman G. Kuperman. 2025. “Dietary Kinetics of a PFAS Mixture in the American Toad (Anaxyrus americanus): Laboratory Insights into Trophic Transfer of PFAS.” Environmental Toxicology and Chemistry 44 (10): 3051–3066. https://doi.org/10.1093/etojnl/vgaf180.
ECOS (Environmental Council of States). 2025. ECOS Compendium of State PFAS Actions. https://www.ecos.org/documents/ecos-compendium-of-state-pfas-actions.
EFSA (European Food Safety Authority). 2014. “Reasoned Opinion on the Setting of MRLs for Saflufenacil in Various Crops, Considering the Risk Related to the Metabolite Trifluoroacetic Acid (TFA).” EFSA Journal 12 (2): 3585. https://doi.org/10.2903/j.efsa.2014.3585.
Elgarahy, Ahmed M., M. G. Eloffy, A. K. Priya, V. Yogeshwaran, Zhen Yang, Khalid Z. Elwakeel, and Eduardo Alberto Lopez-Maldonado. 2024. “Biosolids Management and Utilizations: A Review.” Journal of Cleaner Production 451: 141974. https://doi.org/10.1016/j.jclepro.2024.141974.
Ellis, David A., and Scott A. Mabury. 2000. “The Aqueous Photolysis of TFM and Related Trifluoromethylphenols. An Alternate Source of Trifluoroacetic Acid in the Environment.” Environmental Science & Technology 34 (4): 632–637. https://doi.org/10.1021/es990422c.
Ellis, David A., Jonathan W. Martin, Amila O. De Silva, Scott A. Mabury, Michael D. Hurley, Mads P. Sulbaek Andersen, and Timothy J. Wallington. 2004. “Degradation of Fluorotelomer Alcohols:A Likely Atmospheric Source of Perfluorinated Carboxylic Acids.” Environmental Science & Technology 38 (12): 3316–3321. https://doi.org/10.1021/es049860w.
Endo, Satoshi, Jort Hammer, and Sadao Matsuzawa. 2023. “Experimental Determination of Air/Water Partition Coefficients for 21 Per- and Polyfluoroalkyl Substances Reveals Variable Performance of Property Prediction Models.” Environmental Science & Technology 57 (22): 8406–8413. https://doi.org/10.1021/acs.est.3c02545.
EPA (U.S. Environmental Protection Agency). 2000. “EPA and 3M Announce Phase Out of PFOS.” May 16. https://www.epa.gov/archive/epapages/newsroom_archive/newsreleases/33aa946e6cb11f35852568e1005246b4.html.
EPA. 2024a. “Designation of Perfluorooctanoic Acid (PFOA) and Perfluorooctanesulfonic Acid (PFOS) as CERCLA Hazardous Substances.” Federal Register 89 (90): 39124–39192.
EPA. 2024b. “PFAS National Primary Drinking Water Regulation Rulemaking.” Federal Register 89 (82): 32532–32757.
EPA. 2025. “EPA Announces It Will Keep Maximum Contaminant Levels for PFOA, PFOS.” May 14. https://www.epa.gov/newsreleases/epa-announces-it-will-keep-maximum-contaminant-levels-pfoa-pfos.
Evich, Marina G., James Ferreira, Oluwaseun Adeyemi, Paul A. Schroeder, Jason C. Williams, Brad Acrey, Diana Burdette et al. 2025. “Mineralogical Controls on PFAS and Anthropogenic Anions in Subsurface Soils and Aquifers.” Nature Communications 16 (1): 3118. https://doi.org/10.1038/s41467-025-58040-w.
Fang, Bo, Hao Chen, Maosen Zhao, Biting Qiao, Yue Zhou, Yulong Wang, Yaozhi Zhang et al. 2025. “Biotic and Abiotic Transformations of Aqueous Film-Forming Foam (AFFF)-Derived Emerging Polyfluoroalkyl Substances in Aerobic Soil Slurry.” Water Research 276: 123284. https://doi.org/10.1016/j.watres.2025.123284.
Faust, Jennifer A. 2023. “PFAS on Atmospheric Aerosol Particles: A Review.” Environmental Science: Processes & Impacts 25 (2): 133–150. https://doi.org/10.1039/D2EM00002D.
Fernandes, A. R., I. R. Lake, A. Dowding, M. Rose, N. R. Jones, R. Petch, F. Smith et al. 2019. “The Potential of Recycled Materials Used in Agriculture to Contaminate Food through Uptake by Livestock.” Science of The Total Environment 667: 359–370. https://doi.org/10.1016/j.scitotenv.2019.02.211.
Fitzgerald, C., Polly Shyka, and Ellen Mallory. 2025. Guide to Investigating PFAS Risk on Your Farm. University of Maine Cooperative Extension. https://extension.umaine.edu/agriculture/guide-to-investigating-pfas-risk-on-your-farm/.
Fremlin, Katharine M., John E. Elliott, Robert J. Letcher, Tom Harner, and Frank A. P. C. Gobas. 2023. “Developing Methods for Assessing Trophic Magnification of Perfluoroalkyl Substances within an Urban Terrestrial Avian Food Web.” Environmental Science & Technology 57 (34): 12806–12818. https://doi.org/10.1021/acs.est.3c02361.
Gao, Si, Thomas H. DeLuca, and Cory C. Cleveland. 2019. “Biochar Additions Alter Phosphorus and Nitrogen Availability in Agricultural Ecosystems: A Meta-Analysis.” Science of The Total Environment 654: 463–472. https://doi.org/10.1016/j.scitotenv.2018.11.124.
Gaulke, Eric Patrick. 2020. “Analysis of PFAS over Time in Soil at Venetucci Farm.” Master’s. thesis, University of Colorado, Colorado Springs.
George, Serena E., Tracie R. Baker, and Bridget B. Baker. 2023. “Nonlethal Detection of PFAS Bioaccumulation and Biomagnification within Fishes in an Urban- and Wastewater-Dominant Great Lakes Watershed.” Environmental Pollution 321: 121123. https://doi.org/10.1016/j.envpol.2023.121123.
Gewurtz, Sarah B., Lisa E. Bradley, Sean Backus, Alice Dove, Daryl McGoldrick, Hayley Hung, and Helena Dryfhout-Clark. 2019. “Perfluoroalkyl Acids in Great Lakes Precipitation and Surface Water (2006–2018) Indicate Response to Phase-Outs, Regulatory Action, and Variability in Fate and Transport Processes.” Environmental Science & Technology 53 (15): 8543–8552. https://doi.org/10.1021/acs.est.9b01337.
Ghisi, Rossella, Teofilo Vamerali, and Sergio Manzetti. 2019. “Accumulation of Perfluorinated Alkyl Substances (PFAS) in Agricultural Plants: A Review.” Environmental Research 169: 326–341. https://doi.org/10.1016/j.envres.2018.10.023.
Gill, Rafaqat Ali, Sunny Ahmar, Basharat Ali, Muhammad Hamzah Saleem, Muhammad Umar Khan, Weijun Zhou, and Shengyi Liu. 2021. “The Role of Membrane Transporters in Plant Growth and Development, and Abiotic Stress Tolerance.” International Journal of Molecular Sciences 22 (23): 12792. https://www.mdpi.com/1422-0067/22/23/12792.
Göckener, Bernd, Maria Eichhorn, René Lämmer, Matthias Kotthoff, Janine Kowalczyk, Jorge Numata, Helmut Schafft et al. 2020. “Transfer of Per- and Polyfluoroalkyl Substances (PFAS) from Feed into the Eggs of Laying Hens. Part 1: Analytical Results Including a Modified Total Oxidizable Precursor Assay.” Journal of Agricultural and Food Chemistry 68 (45): 12527–12538. https://doi.org/10.1021/acs.jafc.0c04456.
Goss, Kai-Uwe. 2008. “The pKa Values of PFOA and Other Highly Fluorinated Carboxylic Acids.” Environmental Science & Technology 42 (2): 456–458. https://doi.org/10.1021/es702192c.
Goss, Michael J., Ashraf Tubeileh, and Dave Goorahoo, eds. 2013. “Chapter Five—A Review of the Use of Organic Amendments and the Risk to Human Health.” In Advances in Agronomy, vol. 120, 275–379. Academic Press.
Gottschall, N., E. Topp, M. Edwards, M. Payne, S. Kleywegt, and D. R. Lapen. 2017. “Brominated Flame Retardants and Perfluoroalkyl Acids in Groundwater, Tile Drainage, Soil, and Crop Grain Following a High Application of Municipal Biosolids to a Field.” Science of The Total Environment 574: 1345–1359. https://doi.org/10.1016/j.scitotenv.2016.08.044.
Guelfo, Jennifer L., Stephen Korzeniowski, Marc A. Mills, Janet Anderson, Richard H. Anderson, Jennifer A. Arblaster, Jason M. Conder et al. 2021. “Environmental Sources, Chemistry, Fate, and Transport of Per- and Polyfluoroalkyl Substances: State of the Science, Key Knowledge Gaps, and Recommendations Presented at the August 2019 SETAC Focus Topic Meeting.” Environmental Toxicology and Chemistry 40 (12): 3234–3260. https://doi.org/10.1002/etc.5182.
Guo, Bo, Jicai Zeng, Mark L. Brusseau, and Yonggen Zhang. 2022. “A Screening Model for Quantifying PFAS Leaching in the Vadose Zone and Mass Discharge to Groundwater.” Advances in Water Resources 160: 104102. https://doi.org/10.1016/j.advwatres.2021.104102.
Harris, Benjamin A., Jinpeng Zhou, Bradley O. Clarke, and Ivanhoe K. H. Leung. 2025. “Enzymatic Degradation of PFAS: Current Status and Ongoing Challenges.” ChemSusChem 18 (2): e202401122. https://doi.org/10.1002/cssc.202401122.
Harris, Jackson D., Collin M. Coon, Megan E. Doherty, Eamon A. McHugh, Margaret C. Warner, Conley L. Walters, Olivia M. Orahood et al. 2022. “Engineering and Characterization of Dehalogenase Enzymes from Delftia acidovorans in Bioremediation of Perfluorinated Compounds.” Synthetic and Systems Biotechnology 7 (2): 671–676. https://doi.org/10.1016/j.synbio.2022.02.005.
Higgins, Christopher P., and Richard G. Luthy. 2006. “Sorption of Perfluorinated Surfactants on Sediments.” Environmental Science & Technology 40 (23): 7251–7256. https://doi.org/10.1021/es061000n.
Hoskins, T. D., A. L. Pendleton, Y. J. Choi, J. T. Hoverman, L. S. Lee, and M. S. Sepulveda 2023. “Are Biosolid-Impacted, Stocked Farm Ponds Understudied PFAS Hotspots?,” Paper presented at SETAC North America 44th Annual Meeting, November 12–16, Louisville, KY.
ITRC (Interstate Technology & Regulatory Council). 2023. PFAS Technical and Regulatory Guidance Document and Fact Sheets PFAS-1. Washington, D.C.: Interstate Technology & Regulatory Council, PFAS Team. https://pfas-1.itrcweb.org.
Jian, Jun-Meng, Da Chen, Fu-Juan Han, Ying Guo, Lixi Zeng, Zingwen Lu, and Fei Wang. 2018. “A Short Review on Human Exposure to and Tissue Distribution of Per- and Polyfluoroalkyl Substances (PFASs).” Science of The Total Environment 636: 1058–1069. https://doi.org/10.1016/j.scitotenv.2018.04.380.
Joseph, Stephen, Annette L. Cowie, Lukas Van Zwieten, Nanthi Bolan, Alice Budai, Wolfram Buss, Maria Luz Cayuela et al. 2021. “How Biochar Works, and When It Doesn’t: A Review of Mechanisms Controlling Soil and Plant Responses to Biochar.” GCB Bioenergy 13 (11): 1731–1764. https://doi.org/10.1111/gcbb.12885.
Kali, Suna Ekin, Heléne Österlund, Maria Viklander, and Godecke-Tobias Blecken. 2025. “Stormwater Discharges Affect PFAS Occurrence, Concentrations, and Spatial Distribution in Water and Bottom Sediment of Urban Streams.” Water Research 271: 122973. https://doi.org/10.1016/j.watres.2024.122973.
Kelly, Barry C., Jennifer M. Sun, Mandy R. R. McDougall, Elsie M. Sunderland, and Frank A. P. C. Gobas. 2024. “Development and Evaluation of Aquatic and Terrestrial Food Web Bioaccumulation Models for Per- and Polyfluoroalkyl Substances.” Environmental Science & Technology 58 (40): 17828–17837. https://doi.org/10.1021/acs.est.4c02134.
Khalid, Nejumal Kannankeril, Amira Aker, Stéphane Lair, and Sébastien Sauvé. 2025. “Presence of Per- and Poly-Fluoroalkyl Substances (PFAS) in Brain Samples of Marine Mammals from the St. Lawrence Estuary and Gulf, Canada.” Environmental Science: Advances 10. https://doi.org/10.1039/D5VA00061K.
Khan, Bushra, Robert M. Burgess, and Mark G. Cantwell. 2023. “Occurrence and Bioaccumulation Patterns of Per- and Polyfluoroalkyl Substances (PFAS) in the Marine Environment.” ACS ES&T Water 3 (5): 1243–1259. https://doi.org/10.1021/acsestwater.2c00296.
Kookana, Rai S., Divina A. Navarro, Shervin Kabiri, and Mike J. McLaughlin. 2023. “Key Properties Governing Sorption–Desorption Behaviour of Poly- and Perfluoroalkyl Substances in Saturated and Unsaturated Soils: A Review.” Soil Research 61 (2): 107–125. https://doi.org/10.1071/SR22183.
Kourtchev, Ivan, Bruna G. Sebben, Anna Bogush, Ana Flavia L. Godoi, and Ricardo H. M. Godoi. 2023. “Per- and Polyfluoroalkyl Substances (PFASs) in Urban PM2.5 Samples from Curitiba, Brazil.” Atmospheric Environment 309: 119911. https://doi.org/10.1016/j.atmosenv.2023.119911.
Kourtchev, Ivan, Bruna G. Sebben, Sebastian Brill, G. G. Barbosa Cybelli, Bettina Weber, Rosaria R. Ferreira, Flávio Agusto Farias D’Oliveira et al. 2024. “Occurrence of a ‘Forever Chemical’ in the Atmosphere above Pristine Amazon Forest.” Science of The Total Environment 944: 173918. https://doi.org/10.1016/j.scitotenv.2024.173918.
Krafft, Marie Pierre. 2025. “From Fluorine’s Position in the Periodic Table to PFAS Environmental Issues.” Comptes Rendus Chimie 28: 423–438. https://doi.org/10.5802/crchim.391.
Krippner, Johanna, Sandy Falk, Hubertus Brunn, Sebastian Georgii, Sven Schubert, and Thorsten Stahl. 2015. “Accumulation Potentials of Perfluoroalkyl Carboxylic Acids (PFCAs) and Perfluoroalkyl Sulfonic Acids (PFSAs) in Maize (Zea mays).” Journal of Agricultural and Food Chemistry 63 (14): 3646–3653. https://doi.org/10.1021/acs.jafc.5b00012.
Kuo, Kuan-Yu, Yu Chen, Yi Chuang, Pinpin Lin, and Yi-Jun Lin. 2023. “Worldwide Serum Concentration-Based Probabilistic Mixture Risk Assessment of Perfluoroalkyl Substances among Pregnant Women, Infants, and Children.” Ecotoxicology and Environmental Safety 268: 115712. https://doi.org/10.1016/j.ecoenv.2023.115712.
Lal, R. 2005. “World Crop Residues Production and Implications of Its Use as a Biofuel.” Environment International 31 (4): 575–584. https://doi.org/10.1016/j.envint.2004.09.005.
Larney, Francis J., and Denis A. Angers. 2012. “The Role of Organic Amendments in Soil Reclamation: A Review.” Canadian Journal of Soil Science 92 (1): 19–38. https://doi.org/10.4141/cjss2010-064.
Lasters, Robin, Thimo Groffen, Marcel Eens, Dries Coertjens, Wouter A. Gebbink, Jelle Hofman, and Lieven Bervoets. 2022. “Home-Produced Eggs: An Important Human Exposure Pathway of Perfluoroalkylated Substances (PFAS).” Chemosphere 308: 136283. https://doi.org/10.1016/j.chemosphere.2022.136283.
Lazo, Ariana J., and Linda S. Lee. 2024. “Plant Uptake of PFAS in Soybeans.” Paper presented at the ASA, CSSA, and SSSA International Annual Meeting, November 13, San Antonio, TX.
Lesmeister, Lukas, Frank Thomas Lange, Jörn Breuer, Annegret Biegel-Engler, Evelyn Giese, and Marco Scheurer. 2021. “Extending the Knowledge About PFAS Bioaccumulation Factors for Agricultural Plants – A Review.” Science of The Total Environment 766: 142640. https://doi.org/10.1016/j.scitotenv.2020.142640.
Letcher, Robert J., Shaogang Chu, and Shirley-Anne Smyth. 2020. “Side-Chain Fluorinated Polymer Surfactants in Biosolids from Wastewater Treatment Plants.” Journal of Hazardous Materials 388: 122044. https://doi.org/10.1016/j.jhazmat.2020.122044.
Li, Yasong, Danielle P. Oliver, and Rai S. Kookana. 2018. “A Critical Analysis of Published Data to Discern the Role of Soil and Sediment Properties in Determining Sorption of Per and Polyfluoroalkyl Substances (PFASs).” Science of The Total Environment 628-629: 110–120. https://doi.org/10.1016/j.scitotenv.2018.01.167.
Li, Fei, Xinliang Fang, Zhenming Zhou, Xiaobin Liao, Jing Zou, Baoling Yuan, and Wenjie Sun. 2019. “Adsorption of Perfluorinated Acids onto Soils: Kinetics, Isotherms, and Influences of Soil Properties.” Science of The Total Environment 649: 504–514. https://doi.org/10.1016/j.scitotenv.2018.08.209.
Li, Xiaomin, Shujun Dong, Wei Zhang, Xia Fan, Ruiguo Wang, Peilong Wang, and Xiaoou Su. 2019. “The Occurrence of Perfluoroalkyl Acids in an Important Feed Material (Fishmeal) and Its Potential Risk through the Farm-to-Fork Pathway to Humans.” Journal of Hazardous Materials 367: 559–567. https://doi.org/10.1016/j.jhazmat.2018.12.103.
Liao, Shuchi, Uriel Garza-Rubalcava, Linda M. Abriola, Heather E. Preisendanz, Linda S. Lee, and Kurt D. Pennell. 2025. “Simulating PFAS Transport in Effluent-Irrigated Farmland Using PRZM5, LEACHM, and HYDRUS-1D Models.” Journal of Environmental Quality 54 (1): 54–65. https://doi.org/10.1002/jeq2.20639.
Lim, Teng, Ray Massey, Laura McCann, Timothy Canter, Seabrook Omura, Cammy Willett, Alice Roach, Nigel Key, and Laura Dodson. 2023. Increasing the Value of Animal Manure for Farmers, AP-109. U.S. Department of Agriculture, Economic Research Service. https://www.ers.usda.gov/publications/pub-details?pubid=106088.
Lin, Ashley M., Jake T. Thompson, Jeremy P. Koelmel, Yalan Liu, John A. Bowden, and Timothy G. Townsend. 2024. “Landfill Gas: A Major Pathway for Neutral Per- and Polyfluoroalkyl Substance (PFAS) Release.” Environmental Science & Technology Letters 11 (7): 730–737. https://doi.org/10.1021/acs.estlett.4c00364.
Lindstrom, Andrew B., Mark J. Strynar, and E. Laurence Libelo. 2011. “Polyfluorinated Compounds: Past, Present, and Future.” Environmental Science & Technology 45 (19): 7954–7961. https://doi.org/10.1021/es2011622.
Liu, Jinxia, and Sandra Mejia Avendaño. 2013. “Microbial Degradation of Polyfluoroalkyl Chemicals in the Environment: A Review.” Environment International 61: 98–114. https://doi.org/10.1016/j.envint.2013.08.022.
Liu, Zhaoyang, Yonglong Lu, Xin Song, Kevin Jones, Andrew J. Sweetman, Andrew C. Johnson, Meng Zhang et al. 2019. “Multiple Crop Bioaccumulation and Human Exposure of Perfluoroalkyl Substances around a Mega Fluorochemical Industrial Park, China: Implication for Planting Optimization and Food Safety.” Environment International 127: 671–684. https://doi.org/10.1016/j.envint.2019.04.008.
Lohmann, Rainer, Ian T. Cousins, Jamie C. DeWitt, Juliane Glüge, Gretta Goldenman, Dorte Herzke, Andrew B. Lindstrom et al. 2020. “Are Fluoropolymers Really of Low Concern for Human and Environmental Health and Separate from Other PFAS?” Environmental Science & Technology 54 (20): 12820–12828. https://doi.org/10.1021/acs.est.0c03244.
Lohmann, Rainer, and Robert J. Letcher. 2023. “The Universe of Fluorinated Polymers and Polymeric Substances and Potential Environmental Impacts and Concerns.” Current Opinion in Green and Sustainable Chemistry 41: 100795. https://doi.org/10.1016/j.cogsc.2023.100795.
Lu, Qin, Zhenli L. He, and Peter J. Stoffella. 2012. “Land Application of Biosolids in the USA: A Review.” Applied and Environmental Soil Science 2012 (1): 201462. https://doi.org/10.1155/2012/201462.
Lyu, Ying, and Mark L. Brusseau. 2020. “The Influence of Solution Chemistry on Air-Water Interfacial Adsorption and Transport of PFOA in Unsaturated Porous Media.” Science of The Total Environment 713: 136744. https://doi.org/10.1016/j.scitotenv.2020.136744.
Maine Center for Disease Control and Prevention. 2017. “Memorandum: Action Levels for PFOS in Cow’s Milk.” March 28. https://www.maine.gov/dep/spills/topics/pfas/Derivation-of-Action-Levels-for-PFOS-in-Cows-Milk-03.28.17.pdf.
Maine Center for Disease Control and Prevention. 2020. “Memorandum: Action Levels for PFOS in Beef for Use in Determining Whether Beef at a Farm is Adulterated.” August 4. https://www.maine.gov/dep/spills/topics/pfas/PFOS-Action-Levels-for-Beef-Derivation-Memo-08.04.20.pdf.
Malone, Zachary, Asmeret Asefaw Berhe, and Rebecca Ryals. 2023. “Impacts of Organic Matter Amendments on Urban Soil Carbon and Soil Quality: A Meta-Analysis.” Journal of Cleaner Production 419: 138148. https://doi.org/10.1016/j.jclepro.2023.138148.
Martin, Jonathan W., Scott A. Mabury, Keith R. Solomon, and Derek C. G. Muir. 2013. “Progress toward Understanding the Bioaccumulation of Perfluorinated Alkyl Acids.” Environmental Toxicology and Chemistry 32 (11): 2421–2423. https://doi.org/10.1002/etc.2376.
McAdoo, Mitchell A., Gregory T. Connock, and Terrence Messinger. 2022. Occurrence of Per- and Polyfluoroalkyl Substances and Inorganic Analytes in Groundwater and Surface Water Used as Sources for Public Water Supply in West Virginia. U.S. Geological Survey Scientific Investigations Report 2022–5067. https://doi.org/10.3133/sir20225067.
McDermett, Kaylin, Todd Anderson, W. Andrew Jackson, and Jennifer Guelfo. 2022. “Assessing Potential Perfluoroalkyl Substances Trophic Transfer to Crickets (Acheta domesticus).” Environmental Toxicology and Chemistry 41 (12): 2981–2992. https://doi.org/10.1002/etc.5478.
McIntosh, Lisa, Catharine Rockwell, Samantha Olney, Lisa Campe, R. Duff Collins, J. Daniel Bryant, Victoria Ward, Piper Harring, and James Occhialini. 2025. “Background PFAS Concentrations in Surface Soil of Massachusetts and Northern New England: Regional and Global Source Patterns and Regulatory Relevance.” Remediation Journal 35 (2): e70013. https://doi.org/10.1002/rem.70013.
Mei, Weiping, Hao Sun, Mengke Song, Longfei Jiang, Yongtao Li, Weisheng Lu, Guang-Guo Ying et al. 2021. “Per- and Polyfluoroalkyl Substances (PFASs) in the Soil–Plant System: Sorption, Root Uptake, and Translocation.” Environment International 156: 106642. https://doi.org/10.1016/j.envint.2021.106642.
Mejia Avendaño, Sandra, and Jinxia Liu. 2015. “Production of PFOS from Aerobic Soil Biotransformation of Two Perfluoroalkyl Sulfonamide Derivatives.” Chemosphere 119: 1084–1090. https://doi.org/10.1016/j.chemosphere.2014.09.059.
Mejia-Avendaño, Sandra, Yue Zhi, Bei Yan, and Jinxia Liu. 2020. “Sorption of Polyfluoroalkyl Surfactants on Surface Soils: Effect of Molecular Structures, Soil Properties, and Solution Chemistry.” Environmental Science & Technology 54 (3): 1513–1521. https://doi.org/10.1021/acs.est.9b04989.
Mekureyaw, Mengistu F., Allyson Leigh Junker, Lu Bai, Yan Zhang, Zongsu Wei, and Zheng Guo. 2025. “Laccase Based Per- and Polyfluoroalkyl Substances Degradation: Status and Future Perspectives.” Water Research 271: 122888. https://doi.org/10.1016/j.watres.2024.122888.
Miranda, Daniele A., Alison M. Zachritz, Heather D. Whitehead, Shannon R. Cressman, Graham F. Peaslee, and Gary A. Lamberti. 2023. “Occurrence and Biomagnification of Perfluoroalkyl Substances (PFAS) in Lake Michigan Fishes.” Science of The Total Environment 895: 164903. https://doi.org/10.1016/j.scitotenv.2023.164903.
Müller, Viktoria, Jörg Feldmann, Eileen Prieler, and Robert Brodschneider. 2025. “PFAS in the Buzz: Seasonal Biomonitoring with Honey Bees (Apis mellifera) and Bee-Collected Pollen.” Environmental Pollution 382: 126750. https://doi.org/10.1016/j.envpol.2025.126750.
Munir, Umar, Yifei Wang, and Qingguo Huang. 2025. “Enzyme Catalyzed Oxidative Humification Reactions (ECOHRs): PFAS Remediation and Thatch Management.” Frontiers in Environmental Engineering 4. https://doi.org/10.3389/fenve.2025.1673461.
Munoz, Gabriel, Aurélia Marcelline Michaud, Min Liu, Sung Vo Duy, Denis Montenach, Camille Resseguier, Françoise Watteau et al. 2022. “Target and Nontarget Screening of PFAS in Biosolids, Composts, and Other Organic Waste Products for Land Application in France.” Environmental Science & Technology 56 (10): 6056–6068. https://doi.org/10.1021/acs.est.1c03697.
Murillo-Gelvez, Jimmy, Olga Dmitrenko, Tifany L. Torralba-Sanchez, Paul G. Tratnyek, and Dominic M. Di Toro. 2023. “pKa Prediction of Per- and Polyfluoroalkyl Acids in Water Using in Silico Gas Phase Stretching Vibrational Frequencies and Infrared Intensities.” Physical Chemistry Chemical Physics 25 (36): 24745–24760. https://doi.org/10.1039/D3CP01390A.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2022. Guidance on PFAS Exposure, Testing, and Clinical Follow-Up. The National Academies Press. https://doi.org/10.17226/26156,
New Mexico Department of Health. 2025. “Health Advisory Issued for Holloman Lake.” January 27. https://www.nmhealth.org/news/alert/2025/1/?view=2173.
Ng, Carla A., and Konrad Hungerbühler. 2013. “Bioconcentration of Perfluorinated Alkyl Acids: How Important Is Specific Binding?” Environmental Science & Technology 47 (13): 7214–7223. https://doi.org/10.1021/es400981a.
Nielsen, O. J., M. S. Javadi, M. P. Sulbaek Andersen, M. D. Hurley, T. J. Wallington, and R. Singh. 2007. “Atmospheric Chemistry of CF3CF=CH2: Kinetics and Mechanisms of Gas-Phase Reactions with Cl Atoms, OH Radicals, and O3.” Chemical Physics Letters 439 (1): 18–22. https://doi.org/10.1016/j.cplett.2007.03.053.
OECD (Organisation for Economic Co-operation and Development). 2022. Synthesis Report on Understanding Side-Chain Fluorinated Polymers and Their Life Cycle. https://www.oecd.org/content/dam/oecd/en/publications/reports/2022/11/synthesis-report-on-understanding-side-chain-fluorinated-polymers-and-their-life-cycle_5c473fc6/e13559f7-en.pdf.
Openiyi, Elijah O., Linda S. Lee, Hailey E. Young, Andrew Carpenter, and Romy Carpenter. 2025. “High Carbon Wood Ash Impact on Grass Uptake of Per- and Polyfluoroalkyl Substances from Contaminated Agricultural Soils.” Journal of Agricultural and Food Chemistry 73 (50): 31794–31803. https://doi.org/10.1021/acs.jafc.5c08985.
Ortiz, Sonora, and Ellen B. Mallory. 2025. “Management Strategies to Reduce PFOS Uptake by Forages.” Paper presented at the ASA, CSSA, and SSSA International Annual Meeting, November 10, Salt Lake City, UT.
Pagliari, Paulo, Melissa Wilson, and Zhongqi He. 2020. “Animal Manure Production and Utilization: Impact of Modern Concentrated Animal Feeding Operations.” In Animal Manure, edited by H. M. Waldrip, P. H. Pagliari, and Z. He. https://doi.org/10.2134/asaspecpub67.c1.
Pandamkulangara Kizhakkethil, Jishnu, Zongbo Shi, Anna Bogush, and Ivan Kourtchev. 2024. “Aerosolisation of Per- and Polyfluoroalkyl Substances (PFAS) during Aeration of Contaminated Aqueous Solutions.” Atmospheric Environment 334: 120716. https://doi.org/10.1016/j.atmosenv.2024.120716.
PEER (Public Employees for Environmental Responsibility). 2020. “Summary of Public Employees for Environmental Responsibility’s (PEER’s) PFAS Tests on Anvil 10+10.” https://peer.org/wp-content/uploads/2020/11/Summary-of-PEER-PFAS-Anvil-test-results-fnl-11-25-20-2.pdf.
Penland, Tiffany N., W. Gregory Cope, Thomas J. Kwak, Mark J. Strynar, Casey A. Grieshaber, Ryan J. Heise, and Forrest W. Sessions. 2020. “Trophodynamics of Per- and Polyfluoroalkyl Substances in the Food Web of a Large Atlantic Slope River.” Environmental Science & Technology 54 (11): 6800–6811. https://doi.org/10.1021/acs.est.9b05007.
Peter, Lynda Godwin, and Linda S. Lee. 2025. “Sources and Pathways of PFAS Occurrence in Water Sources: Relative Contribution of Land-Applied Biosolids in an Agricultural Dominated Watershed.” Environmental Science & Technology 59 (2): 1344–1353. https://doi.org/10.1021/acs.est.4c09490.
Peter, Lynda Godwin, Linda S. Lee, Chris Burbage, Kenneth Hoffman, and April Richardson. 2025. “PFAS Retention and Distribution in the Vadose Zone of Three Soil Types Impacted by Biosolids Application.” Journal of Environmental Management 396: 128137. https://doi.org/10.1016/j.jenvman.2025.128137.
Pfotenhauer, David, Emily Sellers, Mark Olson, Katie Praedel, and Martin Shafer. 2022. “PFAS Concentrations and Deposition in Precipitation: An Intensive 5-Month Study at National Atmospheric Deposition Program – National Trends Sites (NADP-NTN) across Wisconsin, USA.” Atmospheric Environment 291: 119368. https://doi.org/10.1016/j.atmosenv.2022.119368.
Poornima, Ramesh, Kathirvel Suganya, and Selvaraj Paul Sebastian. 2022. “Biosolids Towards Back–to–Earth Alternative Concept (BEA) for Environmental Sustainability: A Review.” Environmental Science and Pollution Research 29 (3): 3246–3287. https://doi.org/10.1007/s11356-021-16639-8.
Pulster, Erin L., Sarah R. Bowman, Landon Keele, and Jeffery Steevens. 2024. Guide to Per- and Polyfluoroalkyl Substances (PFAS) Sampling within Natural Resource Damage Assessment and Restoration. U.S. Geological Survey Open-File Report 2024–1001. https://doi.org/10.3133/ofr20241001.
Rankin, Keegan, Holly Lee, Pablo J. Tseng, and Scott A. Mabury. 2014. “Investigating the Biodegradability of a Fluorotelomer-Based Acrylate Polymer in a Soil–Plant Microcosm by Indirect and Direct Analysis.” Environmental Science & Technology 48 (21): 12783–12790. https://doi.org/10.1021/es502986w.
Rankin, Keegan, Scott A. Mabury, Thomas M. Jenkins, and John W. Washington. 2016. “A North American and Global Survey of Perfluoroalkyl Substances in Surface Soils: Distribution Patterns and Mode of Occurrence.” Chemosphere 161: 333–341. https://doi.org/10.1016/j.chemosphere.2016.06.109.
Rayne, Sierra, and Kaya Forest. 2010. “Theoretical Studies on the pKa Values of Perfluoroalkyl Carboxylic Acids.” Journal of Molecular Structure: THEOCHEM 949 (1): 60–69. https://doi.org/10.1016/j.theochem.2010.03.003.
Ricolfi, Lorenzo, Yefeng Yang, Patrice Pottier, Kyle Morrison, Coralie Williams, Pietro Pollo, Daniel Hesselson et al. 2025. “Unravelling the Magnitude and Drivers of PFAS Trophic Magnification: A Meta-Analysis.” Nature Communications 16 (1): 10720. https://doi.org/10.1038/s41467-025-65746-4.
Rich, Courtney D., Andrea C. Blaine, Lakhwinder Hundal, and Christopher P. Higgins. 2015. “Bioaccumulation of Perfluoroalkyl Acids by Earthworms (Eisenia fetida) Exposed to Contaminated Soils.” Environmental Science & Technology 49 (2): 881–888. https://doi.org/10.1021/es504152d.
Rushing, Rosie, Christopher Schmokel, Bryan W. Brooks, and Matt F. Simcik. 2023. “Occurrence of Per- and Polyfluoroalkyl Substance Contamination of Food Sources and Aquaculture Organisms Used in Aquatic Laboratory Experiments.” Environmental Toxicology and Chemistry 42 (7): 1463–1471. https://doi.org/10.1002/etc.5624.
Russell, Mark H., William R. Berti, Bogdan Szostek, and Robert C. Buck. 2008. “Investigation of the Biodegradation Potential of a Fluoroacrylate Polymer Product in Aerobic Soils.” Environmental Science & Technology 42 (3): 800–807. https://doi.org/10.1021/es0710499.
Russell, Mark, William Berti, Bogdan Szostek, Ning Wang, and Robert Buck. 2010. “Evaluation of PFO Formation from the Biodegradation of a Fluorotelomer-Based Urethane Polymer Product in Aerobic Soils.” Polymer Degradation and Stability 95: 79–85. https://doi.org/10.1016/j.polymdegradstab.2009.10.004.
Sakhiya, Anil Kumar, Abhijeet Anand, and Priyanka Kaushal. 2020. “Production, Activation, and Applications of Biochar in Recent Times.” Biochar 2 (3): 253–285. https://doi.org/10.1007/s42773-020-00047-1.
Saleh, Hadeer, Dibyendu Sarkar, Zhiming Zhang, Michel Boufadel, and Rupali Datta. 2025. “Per- and Polyfluoroalkyl Substances (PFAS) in Urban Stormwater Runoff: Insights from a Roadside Rain Garden.” Water 17 (20): 2982. https://www.mdpi.com/2073-4441/17/20/2982.
Savoca, Dario, and Andrea Pace. 2021. “Bioaccumulation, Biodistribution, Toxicology and Biomonitoring of Organofluorine Compounds in Aquatic Organisms.” International Journal of Molecular Sciences 22 (12): 6276. https://www.mdpi.com/1422-0067/22/12/6276.
Schroeder, Tim, David Bond, and Janet Foley. 2021. “PFAS Soil and Groundwater Contamination Via Industrial Airborne Emission and Land Deposition in SW Vermont and Eastern New York State, USA.” Environmental Science: Processes & Impacts 23 (2): 291–301. https://doi.org/10.1039/D0EM00427H.
Scott, Brian F., Christine Spencer, Scott A. Mabury, and Derek C. G. Muir. 2006. “Poly and Perfluorinated Carboxylates in North American Precipitation.” Environmental Science & Technology 40 (23): 7167–7174. https://doi.org/10.1021/es061403n.
Sharifan, Hamidreza, Majid Bagheri, Dan Wang, Joel G. Burken, Christopher P. Higgins, Yanna Liang, Jinxia Liu et al. 2021. “Fate and Transport of Per- and Polyfluoroalkyl Substances (PFASs) in the Vadose Zone.” Science of The Total Environment 771: 145427. https://doi.org/10.1016/j.scitotenv.2021.145427.
Shimizu, Megumi S., Rachael Mott, Ariel Potter, Jiaqi Zhou, Karsten Baumann, Jasen D. Surratt, Barbara Turpin et al. 2021. “Atmospheric Deposition and Annual Flux of Legacy Perfluoroalkyl Substances and Replacement Perfluoroalkyl Ether Carboxylic Acids in Wilmington, NC, USA.” Environmental Science & Technology Letters 8 (5): 366–372. https://doi.org/10.1021/acs.estlett.1c00251.
Siedt, Martin, Andreas Schäffer, Kilian E. C. Smith, Moritz Nabel, Martina Roß-Nickoll, and Joost T. van Dongen. 2021. “Comparing Straw, Compost, and Biochar Regarding Their Suitability as Agricultural Soil Amendments to Affect Soil Structure, Nutrient Leaching, Microbial Communities, and the Fate of Pesticides.” Science of The Total Environment 751: 141607. https://doi.org/10.1016/j.scitotenv.2020.141607.
Silva, Jeff A. K., William A. Martin, and John E. McCray. 2021. “Air-Water Interfacial Adsorption Coefficients for PFAS When Present as a Multi-Component Mixture.” Journal of Contaminant Hydrology 236: 103731. https://doi.org/10.1016/j.jconhyd.2020.103731.
Silva, Jeff A. K., Jennifer L. Guelfo, Jiří Šimůnek, and John E. McCray. 2022. “Simulated Leaching of PFAS from Land-Applied Municipal Biosolids at Agricultural Sites.” Journal of Contaminant Hydrology 251: 104089. https://doi.org/10.1016/j.jconhyd.2022.104089.
Simones, Thomas L., Chris Evans, Caleb P. Goossen, Richard Kersbergen, Ellen B. Mallory, Susan Genualdi, Wendy Young et al. 2024. “Uptake of Per- and Polyfluoroalkyl Substances in Mixed Forages on Biosolid-Amended Farm Fields.” Journal of Agricultural and Food Chemistry 72 (42): 23108–23117. https://doi.org/10.1021/acs.jafc.4c02078.
Smith, Jacob, Mark L. Brusseau, and Bo Guo. 2024. “An Integrated Analytical Modeling Framework for Determining Site-Specific Soil Screening Levels for PFAS.” Water Research 252: 121236. https://doi.org/10.1016/j.watres.2024.121236.
Smorada, Chiara M., Matthew W. Sima, and Peter R. Jaffé. 2024. “Bacterial Degradation of Perfluoroalkyl Acids.” Current Opinion in Biotechnology 88: 103170. https://doi.org/10.1016/j.copbio.2024.103170.
Solan, Megan E., Camryn P. Koperski, Sanjanaa Senthilkumar, and Ramon Lavado. 2023. “Short-Chain Per- and Polyfluoralkyl Substances (PFAS) Effects on Oxidative Stress Biomarkers in Human Liver, Kidney, Muscle, and Microglia Cell Lines.” Environmental Research 223: 115424. https://doi.org/10.1016/j.envres.2023.115424.
Stahl, T., J. Heyn, H. Thiele, J. Hüther, K. Failing, S. Georgii, and H. Brunn. 2009. “Carryover of Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonate (PFOS) from Soil to Plants.” Archives of Environmental Contamination and Toxicology 57 (2): 289–298. https://doi.org/10.1007/s00244-008-9272-9.
State of New Mexico. 2022. “New Mexico Assists Clovis Family Dairy Farm with PFAS Contamination.” Environment Department, May 19. https://www.env.nm.gov/wp-content/uploads/2022/05/2022-05-19-COMMS-New-Mexico-assists-Clovis-family-dairy-farm-with-PFAS-contamination-Final.pdf.
Sunderland, Elsie M., Xindi C. Hu, Clifton Dassuncao, Andrea K. Tokranov, Charlotte C. Wagner, and Joseph G. Allen. 2019. “A Review of the Pathways of Human Exposure to Poly- and Perfluoroalkyl Substances (PFASs) and Present Understanding of Health Effects.” Journal of Exposure Science & Environmental Epidemiology 29 (2): 131–147. https://doi.org/10.1038/s41370-018-0094-1.
Surma, Magdalena, Henryk Zieliński, and Mariusz Piskuła. 2016. “Levels of Contamination by Perfluoroalkyl Substances in Honey from Selected European Countries.” Bulletin of Environmental Contamination and Toxicology 97 (1): 112–118. https://doi.org/10.1007/s00128-016-1840-5.
Thompson, Jake T., Nicole M. Robey, Thabet M. Tolaymat, John A. Bowden, Helena M. Solo-Gabriele, and Timothy G. Townsend. 2023. “Underestimation of Per- and Polyfluoroalkyl Substances in Biosolids: Precursor Transformation during Conventional Treatment.” Environmental Science & Technology 57 (9): 3825–3832. https://doi.org/10.1021/acs.est.2c06189.
Urra, Julen, Itziar Alkorta, and Carlos Garbisu. 2019. “Potential Benefits and Risks for Soil Health Derived from the Use of Organic Amendments in Agriculture.” Agronomy 9 (9): 542. https://www.mdpi.com/2073-4395/9/9/542.
Vierke, Lena, Urs Berger, and Ian T. Cousins. 2013. “Estimation of the Acid Dissociation Constant of Perfluoroalkyl Carboxylic Acids through an Experimental Investigation of Their Water-to-Air Transport.” Environmental Science & Technology 47 (19): 11032–11039. https://doi.org/10.1021/es402691z.
Wackett, Lawrence P. 2025. “PFAS Biodegradation and the Constraints of Thermodynamics.” Microbial Biotechnology 18 (6): e70181. https://doi.org/10.1111/1751-7915.70181.
Wallington, T. J., M. D. Hurley, J. Xia, D. J. Wuebbles, S. Sillman, A. Ito, J. E. Penner et al. 2006. “Formation of C7F15COOH (PFOA) and Other Perfluorocarboxylic Acids during the Atmospheric Oxidation of 8:2 Fluorotelomer Alcohol.” Environmental Science & Technology 40 (3): 924–930. https://doi.org/10.1021/es051858x.
Wang, Lixi, Tong Yang, Xinglu Liu, Jinxia Liu, and Wenxin Liu. 2024. “Critical Evaluation and Meta-Analysis of Ecotoxicological Data on Per- and Polyfluoroalkyl Substances (PFAS) in Freshwater Species.” Environmental Science & Technology 58 (40): 17555–17566. https://doi.org/10.1021/acs.est.4c04818.
Wang, Wenfeng, Geoff Rhodes, Jing Ge, Xiangyang Yu, and Hui Li. 2020. “Uptake and Accumulation of Per- and Polyfluoroalkyl Substances in Plants.” Chemosphere 261: 127584. https://doi.org/10.1016/j.chemosphere.2020.127584.
Washington, John W., and Thomas M. Jenkins. 2015. “Abiotic Hydrolysis of Fluorotelomer-Based Polymers as a Source of Perfluorocarboxylates at the Global Scale.” Environmental Science & Technology 49 (24): 14129–14135. https://doi.org/10.1021/acs.est.5b03686.
Washington, John W., J. Jackson Ellington, Thomas M. Jenkins, John J. Evans, Hoon Yoo, and Sarah C. Hafner. 2009. “Degradability of an Acrylate-Linked, Fluorotelomer Polymer in Soil.” Environmental Science & Technology 43 (17): 6617–6623. https://doi.org/10.1021/es9002668.
Washington, John W., Thomas M. Jenkins, Keegan Rankin, and Jonathan E. Naile. 2015. “Decades-Scale Degradation of Commercial, Side-Chain, Fluorotelomer-Based Polymers in Soils and Water.” Environmental Science & Technology 49 (2): 915–923. https://doi.org/10.1021/es504347u.
Washington, John W., Keegan Rankin, E. Laurence Libelo, David G. Lynch, and Mike Cyterski. “Determining Global Background Soil PFAS Loads and the Fluorotelomer-Based Polymer Degradation Rates That Can Account for These Loads.” Science of The Total Environment 651 (Pt 2): 2444–2449. https://doi.org/10.1016/j.scitotenv.2018.10.071.
Weber, Kathrin, and Peter Quicker. 2018. “Properties of Biochar.” Fuel 217: 240–261. https://doi.org/10.1016/j.fuel.2017.12.054.
Wen, Bei, Longfei Li, Hongna Zhang, Yibing Ma, Xiao-Quan Shan, and Shuzhen Zhang. 2014. “Field Study on the Uptake and Translocation of Perfluoroalkyl Acids (PFAAs) by Wheat (Triticum aestivum L.) Grown in Biosolids-Amended Soils.” Environmental Pollution 184: 547–554. https://doi.org/10.1016/j.envpol.2013.09.040.
Witt, Christopher C., Chauncey R. Gadek, Jean-Luc E. Cartron, Michael J. Andersen, Mariel L. Campbell, Marialejandra Castro-Farias, Ethan F. Gyllenhaal et al. 2024. “Extraordinary Levels of Per- and Polyfluoroalkyl Substances (PFAS) in Vertebrate Animals at a New Mexico Desert Oasis: Multiple Pathways for Wildlife and Human Exposure.” Environmental Research 249: 118229. https://doi.org/10.1016/j.envres.2024.118229.
Wood, Cathryn, George H. Balazs, Marc Rice, Thierry M. Work, T. Todd Jones, Eleanor Sterling, Tammy M. Summers et al. 2021. “Sea Turtles across the North Pacific Are Exposed to Perfluoroalkyl Substances.” Environmental Pollution 279: 116875. https://doi.org/10.1016/j.envpol.2021.116875.
Xie, Zhiyong, Zhen Wang, Wenying Mi, Axel Möller, Hendrik Wolschke, and Ralf Ebinghaus. 2015. “Neutral Poly-/Perfluoroalkyl Substances in Air and Snow from the Arctic.” Scientific Reports 5 (1): 8912. https://doi.org/10.1038/srep08912.
Yan, Bei, Riccardo Alessandri, Siewert J. Marrink, Linda S. Lee, and Jinxia Liu. 2025. “Insight into the Self-Assembly Behaviors of Per- and Polyfluoroalkyl Substances Using a ‘Computational Microscope’.” Environmental Science & Technology Letters 12 (5): 626–631. https://doi.org/10.1021/acs.estlett.4c01081.
Yang, Zhao, Marzieh Shojaei, and Jennifer L. Guelfo. 2023. “Per- and Polyfluoroalkyl Substances (PFAS) in Grocery Store Foods: Method Optimization, Occurrence, and Exposure Assessment.” Environmental Science: Processes & Impacts 25 (12): 2015–2030. https://doi.org/10.1039/D3EM00268C.
Yao, Xuehui, Jing Nie, Ruoxue Bai, and Xiaolei Sui. 2020. “Amino Acid Transporters in Plants: Identification and Function.” Plants 9 (8): 972. https://www.mdpi.com/2223-7747/9/8/972.
Zhang, Kunfeng, Abdul Qadeer, Sheng Chang, Xiang Tu, Hongru Shang, Moonis Ali Khan, Yingying Zhu et al. 2025. “Short-Chain PFASs Dominance and Their Environmental Transport Dynamics in Urban Water Systems: Insights from Multimedia Transport Analysis and Human Exposure Risk.” Environment International 202: 109602. https://doi.org/10.1016/j.envint.2025.109602.
Zhang, Lilan, Linda S. Lee, Junfeng Niu, and Jinxia Liu. 2017. “Kinetic Analysis of Aerobic Biotransformation Pathways of a Perfluorooctane Sulfonate (PFOS) Precursor in Distinctly Different Soils.” Environmental Pollution 229: 159–167. https://doi.org/10.1016/j.envpol.2017.05.074.
Zhang, Weilan, Nina Tran, and Yanna Liang. 2022. “Uptake of Per- and Polyfluoroalkyl Substances (PFAS) by Soybean across Two Generations.” Journal of Hazardous Materials Advances 8: 100170. https://doi.org/10.1016/j.hazadv.2022.100170.
Zhu, Wendy, Harrison Roakes, Stephen G. Zemba, and Appala Raju Badireddy. 2019. PFAS Background in Vermont Shallow Soils. https://anrweb.vt.gov/PubDocs/DEC/PFOA/Soil-Background/PFAS-Background-Vermont-Shallow-Soils-03-24-19.pdf.
Zhu, Xiaojing, Xin Song, and Jan Schwarzbauer. 2021. “First Insights into the Formation and Long-Term Dynamic Behaviors of Nonextractable Perfluorooctanesulfonate and Its Alternative 6:2 Chlorinated Polyfluorinated Ether Sulfonate Residues in a Silty Clay Soil.” Science of The Total Environment 761: 143230. https://doi.org/10.1016/j.scitotenv.2020.143230.