Wolves, Bears, and Their Prey in Alaska: Biological and Social Challenges in Wildlife Management (1997)
Chapter: 3 ALASKA'S PEOPLE, BIOMES, AND WILDLIFE SPECIES OF CONCERN
3
Alaska's People, Biomes, and Wildlife Species of Concern
INTRODUCTION
To evaluate a science-based program, one must understand the contexts in which the program is carried out, including the ecological, economic, and political contexts. This chapter provides an overview of the physical and biological environments in which wolves, bears, and their prey interact. The two following chapters discuss what is known about predator-prey interactions in general (chapter 4) and what has been learned from different attempts to alter predator-prey interactions through wolf and/or bear reductions (chapter 5). The socioeconomic environment is covered in chapter 6.
An overview of the climate, vegetation, and soils of Alaska and the major biomes into which Alaska has been divided serves 2 purposes. First, by illustrating the great diversity of Alaskan environments, it demonstrates why management programs must be based on area-specific information. Second, it shows how the vastness of Alaska and the limitations of personnel and financial resources available for biological research make it inevitable that management decisions are based on less-complete information than is desirable and than would be possible if resources were less limited or the area smaller.
This survey is followed by a review of the ecology and natural history of wolves, bears, and their primary prey—moose and caribou. The review demonstrates the substantial differences among the species of concern and provides a basis for designing management programs that are tailored to the life-history traits of the managed species and how they respond to changes in their environment.
THE PEOPLE OF ALASKA
The first humans in the Western Hemisphere are believed to have come from Asia across the Beringian land bridge into Alaska 12,000–15,000 years ago. The first to arrive were the Paleoindians, who spread throughout North America and South America and from whom most native American cultures derived, including the Haida and Tlingit Indians of the southeastern coast of Alaska (Greenberg 1987). Later migrations of people are believed responsible for the Athabascan Indian cultures that are present throughout the interior and south-central regions of Alaska and in parts of northwestern Canada. The marine-oriented Eskimos of Arctic, western, and southwestern Alaska (represented today by the Inupiat, Yup'ik, and Koniak cultures) arrived much later, apparently by boat across Bering Strait. The Aleut culture of the Aleutian Islands and adjacent Alaska Peninsula has its closest affinity to early Eskimo cultures.
Today, the human population of Alaska is about 610,000, with the majority concentrated in and around Anchorage, Fairbanks, Juneau, and smaller southcoastal cities of a few thousand each. The Alaskan population is younger than the rest of the United States (median age 30 years versus 33.4 years for the whole United States), and its rate of population increase in recent years is second only to that of Nevada (Alaska Bureau of Vital Statistics 1995). The nonindigenous residents of Alaska (those who are not Alaska Natives of Eskimo, Indian, or Aleut descent as defined by the Alaska Native Claims Settlement Act of 1971) make up about 84% of the Alaskan population, and about 80% of them live in urban communities. The non-Native residents of Alaska are primarily first-or second-generation immigrants from the other states and reflect the racial and ethnic diversity that characterizes the United States. There are some differences in the racial make-up between Alaska and the United States as a whole. Alaska's population is 4.1% black (12.1% for the entire United States), 16% Native American (0.8%), and 3.2% Hispanic of any race (9%), according to the 1990 US census. Alaska Natives currently make up 16.5% of the state's population and most live in rural communities (Wolfe 1996). There are about 225 rural communities of fewer than 500 residents scattered throughout the state but concentrated in southeastern Alaska. The residents of all but a few of those communities are predominantly Alaska Natives.
Human activities have had less effect on the ecosystems of Alaska than elsewhere in the United States. Conversion of land to agricultural use has been minimal, as is the extent of land alteration through mining and petroleum development. The greatest alteration of ecosystems has been through extensive logging of forests in southeastern Alaska. More than 40% of Alaska is managed by federal agencies through the National Park Service, Fish and Wildlife Service, and Bureau of Land Management.
BIOMES: CLIMATE, VEGETATION, SOILS, PERMAFROST
Alaska is one-fifth the size of the lower 48 states and occupies 1,477,270 km2. It extends more than 20° in latitude from Pt. Barrow to Amatiguak Island in the Aleutians; it spans 42° in longitude from Portland Canal in southeastern Alaska to Attu Island in the western Aleutians. The topography, climate, and ecosystems of Alaska are characterized by great diversity (Selkregg 1976; Klein and others 1997). Alaska's coastline extends more than 54,700 km, bordering on the North Pacific, Bering Sea, and Arctic Ocean. The average length of the frost-free period varies from 40 days in the Arctic to more than 200 days in parts of southeastern Alaska. The Alaskan interior, because of its relatively warm, very long summer days, has relatively high plant productivity, except where permafrost is present. Permafrost is ground that remains perennially frozen except for a shallow summer-active layer. Annual precipitation ranges from less than 25 cm in the Arctic to 500 cm in parts of the Alexander Archipelago of southeastern Alaska. The low precipitation of interior Alaska would result in desert conditions at lower latitudes, but most of the winter precipitation remains on the land as snow until spring, and low summer evaporation rates and drainage (impeded by permafrost) retain soil moisture throughout the summer in most areas. Permafrost is present throughout most of the Arctic and northwestern Alaska except beneath lakes, rivers, and adjacent riparian zones. South of the Brooks Range in the interior and in southwestern Alaska, permafrost is discontinuous and confined mostly to lowlands, north-facing slopes, and higher elevations.
The position of Alaska between the cold Arctic Ocean and the relatively warm North Pacific, its extensive coastline and southern islands, and its high mountain ranges and associated ice fields with intervening and extensive lowlands are responsible for the ecological diversity that characterizes the state. Alaska can be divided into 6 major biogeographic regions or biomes (figure 3.1).
Coastal Temperate Rain Forest and Coast Range Mountains
The coastal temperate rain forest grows under the moderating maritime influence of relatively warm ocean currents of the North Pacific and Gulf of Alaska. It is a continuation of the temperate coniferous rain forest that extends from northern California along the northwestern coast to northern Kodiak Island. The distribution of large mammals in this region, because of its many islands and the relatively short time since it was heavily glaciated, is more complex than that in the remainder of Alaska. Sitka black-tailed deer (Odocoileus hemionus sitkensis) are common on the islands and adjacent mainland of southeastern Alaska and have been successfully introduced to the islands of Prince William Sound and to Kodiak Island. Moose (Alces alces) occupy the major river valleys that penetrate the Coast Range Mountains—such as the Stikine, Taku, and Chilkat rivers—but do not normally occur in the coniferous forests of the islands. Mountain goats
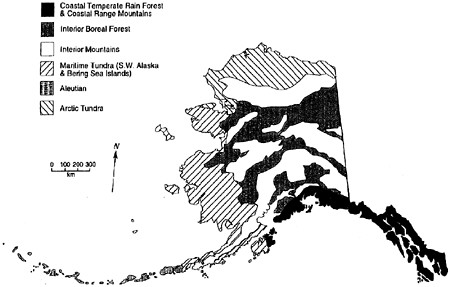
FIGURE 3.1 Major biogeographic regions of Alaska.
(Oreamnos americanus) occur in scattered localities in the mainland mountains and have been introduced to Baranof and Revilla islands. Brown, or grizzly, bears (Ursus arctos) are present on the mainland; Admiralty, Baranof, and Chichagof islands of southeastern Alaska; the islands of Prince William Sound; and Kodiak and Afognak islands. Black bears (Ursus americanus) and wolves (Canis lupus) are present throughout the mainland and on the islands of southeastern Alaska south of Frederick Sound. Densities of bears are high on the islands and mainland, especially in the vicinity of salmon spawning streams. Wolves vary in density in relation to the availability of their primary prey, blacktailed deer. Deer densities fluctuate with the frequency and severity of winters of deep snow. Deer habitat has been greatly modified by forest harvesting.
Interior Boreal Forest
Much of interior Alaska, which is sheltered by high mountains from the moist maritime air to the south and the cold Arctic air to the north, has a continental climate. Winters are cold and long; summers are warm and short. Seasonal changes are rapid. Altitude strongly influences plant growth, the presence and composition of forests, and the extent of permafrost. Fire, caused mostly by lightning, is a natural feature of the ecology of the interior boreal forest. The pattern of vegetation is a complex juxtaposition of plant communities that vary with fire history, soil temperatures, drainage, and exposure. The present boreal forest in interior Alaska is part of the northern boreal forest that extends from the
Atlantic coast of Canada across the northern portion of the continent into Alaska. Mammals characteristic of this major biome are similar throughout this vast area.
The extensive interior boreal forest region is broken by several mountain complexes that support typical mountain species, such as caribou (Rangifer tarandus) and brown bear. The lowland areas of mixed forest, shrub zones, and wetlands support moose and black bears. Moose also venture into the mountains, especially in summer. Wolves are present throughout the region, varying in density with the distribution and availability of prey, primarily moose and caribou. Mammal populations in this largely intact ecosystem undergo natural fluctuations, primarily in relation to variations in winter snow depth, plant succession after wildfire, and variation in rates of predation by wolves and bears.
Interior Mountains (Montane Habitats)
This biogeographic region is a complex of mountain ranges characterized by extreme physiographic variability. Elevation, slope steepness, and exposure vary widely locally and between major mountain masses. The distribution of vegetation, although dominated primarily by alpine forms, reflects the terrain variability, and vegetation at lower elevations includes elements from the boreal forest. Climatic variability is pronounced. Because oceanic air masses lose much of their moisture on the seaward sides of mountain ranges as rain or snow, interior sides are characterized by more arid conditions. The progression of summer plant growth is also highly variable, a condition that favors mammals—such as mountain sheep (Ovis dalli), moose, and caribou—that are able to move extensively over the variable terrain to forage. The depth of winter snows usually limits the availability of winter habitat for mountain sheep, moose, and caribou in these mountain areas. Brown bears and wolves are common, especially in areas of high prey densities.
Maritime Tundra (Southwestern Alaska and Bering Sea Islands)
The maritime tundra that dominates southwestern Alaska and the Bering Sea islands is the product of the cool climate generated by the cold Bering Sea waters. However, there is a gradation from the more-humid and milder conditions prevailing in Bristol Bay and the coastal Alaska peninsula bordering the Aleutian region to the Seward Peninsula, where the adjacent seas are ice-covered for 8 months of the year. Coastal wetlands are extensive throughout the region and dominate the broad expanse of the delta of the Yukon and Kuskokwim rivers (Selkregg 1976). In its climate and vegetation, the region is transitional between the Aleutian and Arctic biogeographic regions.
Moose, caribou, brown bears, and wolves have been absent or rare throughout much of this region in the past. In recent years, caribou re-entered the area from the expanding Mulchatna Herd, a re-established herd in the Kilbuck Mountains,
and segments of the Western Arctic Herd, which winters on the western Seward Peninsula and in Kotzebue and Norton Sound drainages. About 30,000 reindeer (domesticated caribou introduced from Scandinavia) in several herds privately owned by Alaska Natives are grazed on the Seward Peninsula and have come into increasing conflict with wintering caribou from the Western Arctic Herd. Moose have become established in recent decades in riparian areas on the Seward Peninsula. Their populations appear to have peaked, with some possible declines, perhaps because the sparse winter habitats along the major rivers have become heavily browsed. Introduced muskoxen live on Nunivak Island, on Nelson Island on the Yukon Delta, and on the Seward Peninsula. Their expanding populations are hunted under permit systems by both subsistence and sport hunters. Brown bears are common in coastal areas of the Alaska Peninsula and present at lower densities on the Seward Peninsula.
Aleutian Region
The Aleutian Islands and adjacent Alaska Peninsula, an interface between the North Pacific and the Bering Sea, includes the southernmost land area in Alaska. The Aleutians, which extend nearly 1,900 km from the Alaska Peninsula to Attu Island, are renowned for their cool, foggy, and windy weather and temperature consistency. The mean daily temperature of 3.9°C has an annual range of only 9.4°C.
Native large terrestrial mammals are absent from virtually all the Aleutian Islands. In the Aleutian biogeographic region, only the southern portion of the Alaska Peninsula and closely adjacent Unimak Island support caribou (currently at a low density), brown bears, and wolves. The bears are moderately abundant and depend heavily on the productive salmon streams in the area. Wolf numbers are low, presumably because of the low prey density.
Arctic Tundra
The Brooks Range mountains separate the boreal forest and the Arctic tundra biogeographic regions. The Arctic tundra of Alaska, on the higher and drier ground at its southern limit, descends to the broad and wet coastal plain that continues to the Arctic Ocean. Coastal plain tundra is interspersed with thousands of shallow lakes. The Arctic tundra experiences strong northeasterly winds in winter that are generated by the Arctic high-pressure system over the frozen Arctic Ocean. The little snow that falls throughout the long winter is redistributed by winds into drifts wherever there is variation in the terrain.
In spite of climatic extremes of the Arctic tundra—which results in a short plant growth season, low mean annual temperature, and cold soils underlain by permafrost—this region is extremely productive of life, much of which (such as nesting birds and caribou) migrates out of the region during winter. The Arctic
tundra supports Alaska's largest caribou herds, the Western Arctic Herd (about 500,000) and Porcupine Herd (about 170,000), as well as the smaller Central Arctic and Teshekpuk herds, each numbering around 20,000. In the latter half of the 20th century, moose moved into riparian habitats along the Collville, Sagavanirktok, Canning, and other larger rivers of the Arctic. Barren-ground brown bears occur at low densities, being more numerous in the southern foothills and western Arctic where they are important predators on young caribou during calving. Wolf densities are relatively low in the Arctic, largely because of the low density of ungulate prey in winter. Their numbers are highest in the northern foothills and adjacent Brooks Range mountains, where mountain sheep, moose, and some wintering caribou can be present. Muskoxen have been reestablished in the Arctic through introductions in the 1960s and 1970s in the eastern and western areas. They are increasing and dispersing into unoccupied habitats. The total numbers in the Arctic tundra appear to be approaching 1,000.
ECOLOGY OF LARGE MAMMALS IN NORTHERN ECOSYSTEMS
The population dynamics of large mammals in southern Canada and the lower 48 states have been dramatically altered over the past two or three centuries. Not only have most of the natural habitats been converted to agricultural lands, managed forests, or high density human use areas, but dominant species such as wolves, grizzly bears, and millions of migratory bison have largely been eliminated. The population dynamics of large mammals in the pre-Columbian system are unknown. What remains of the system can be managed carefully to provide predictable harvests for hunters and trappers.
In northern Canada and Alaska, however, natural habitats have not been substantially altered. Alaskan ecosystems are still much the same as they were when Europeans first arrived in North America and caribou, moose, wolves, and bears are not threatened with extirpation. The large mammal system remains largely intact and is highly volatile. Most caribou migrate over large, and often unpredictable areas, and their numbers fluctuate enormously. For example, in Alaska, the Mulchatna herd increased from about 30,000 to 110,000 individuals between 1984 and 1993. In Quebec and Labrador, the George River herd increased from about 10,000 in the 1950s to approximately 800,000 in the 1990s. Such enormous shifts in one species influence the dynamics of the entire system, and large changes in moose abundance and distribution have also been evident. Biologists have debated how and when factors such as the very slow growth of terrestrial lichens, the highly variable Arctic weather, and predation at different caribou densities, influence caribou and moose populations. What isn't debated is that the northern systems are much more dynamic than what is left of the southern systems. Consequently, we cannot expect to manage northern systems to provide predictable numbers of ungulates and predictable harvests; Alaska is
not Colorado, and caribou are not elk. Alaskans should not expect as constant a supply of game as people expect in the southern portion of the continent.
Many factors influence the population dynamics of moose, caribou, wolves, and bears. Among the most important are quantity and quality of food, weather, diseases, parasites, predation, intraspecific strife, and human harvest. These factors can act individually, but often they act in concert. For example, animals weakened by poor nutrition are more likely to succumb to disease or predators than well-fed animals are. Together, these factors determine the ecological carrying capacity of the environment—that is, the number of animals that can be supported on a long term basis by the resources of the environment (see box). Another definition of carrying capacity is the number that will persist if disease organisms and predators are present. Because the goal of predator control and management in Alaska is to increase prey numbers, we use the former definition of carrying capacity in the report. Economic carrying capacity, the density of animals that will allow maximum sustained harvest, is always lower than ecological carrying capacity (Caughley 1976 in Krebs 1994). Carrying capacities fluctuate because abiotic and biotic environmental factors change. Assessing environmental carrying capacity is important because success or failure of management programs often depends on the environmental conditions under which they are carried out.
Ecological carrying capacities of Alaskan environments for ungulates are low because arctic, alpine, and subalpine soils are typically poor in nutrients. In combination with short growing seasons, this limits the potential quality and production of forage. Shallow, high-latitude soils underlain by permafrost are fragile and easily damaged and vegetation may recover slowly from overgrazing.
Arctic and sub-Arctic plants grow rapidly during the short growing season, and growth rates are higher during warm, sunny summers than during cloudy, cold summers (Klein 1970). Plant species are adapted to different soils, terrain, exposure, and water availability (Maessen and others 1983) and are distributed in a mosaic throughout Alaska (Maessen and others 1983). This mosaic distribution influences the foraging patterns of large herbivores and omnivorous bears. Large herbivores, in turn, can strongly affect plant succession, species composition, and productivity. High densities of large herbivores, such as moose and caribou, can cause both short-term declines in aboveground plant biomass and long-term declines in the quality of plants (Leader-Williams and others 1981). Lower biomass or poorer-quality plants (for example, plants that have been severely browsed) reduce the carrying capacity of the habitat for ungulates (Klein 1968).
Plants respond to being fed on by moose or caribou in a variety of ways. Light grazing can stimulate growth, and plants that grow beyond the reach of browsers can complete their annual growth more rapidly (Edenius 1993; Edenius and others 1993; Molvar and others 1993; Robbins and others 1987). Browsing and grazing can also stimulate chemical changes that alter plant palatability (Edenius 1993; Bryant and Kuropat 1980). Thus, plants and herbivores are
|
Carrying Capacity Ecological carrying capacity is the number of organisms that the resources of the environment can sustain in a particular region (Pianka 1978, Sharkey 1970). Populations that exceed their carrying capacity must ultimately decline. Populations below their carrying capacity will tend to increase toward it. Carrying capacity itself can vary from year to year because the availability of resources for a population varies from year to year. Because carrying capacities are themselves dynamic, they are difficult to estimate (Dhondt 1988, Pulliam and Haddad 1994). The notion of carrying capacity is crucial in studying predator-prey systems because the effect of predator removal depends largely on how much below its ecological carrying capacity a prey population is. If prey are far below their carrying capacity and if predators are the cause, predator removal can be effective in enhancing the abundance of prey. In contrast, regardless of how many prey are killed by predators, if prey populations are close to their ecological carrying capacity, removal of predators will not produce a marked increase in prey abundance. |
involved in a coevolutionary process of defense against and response to predation (for example, Coley and others 1985; Edenius 1993; Rosenthal and Janzen 1979).
Fire is a natural part of northern ecosystems and has a major impact on plant community organization and structure, and, hence, carrying capacity for ungulates (Viereck 1973; Zackrisson 1977). After a fire, short-lived and fast-growing deciduous shrubs and trees colonize the area, and long-lived and slow-growing species decline (Scotter 1967). Lichens recover from fire much more slowly than shrubs, but, over a long time period, fire rejuvenates lichen communities and helps to create the mosaic of habitats that is characteristic of Arctic and sub-Arctic environments (Miller 1980; Zackrisson 1977). Fires might have increased with settlement and have been blamed for the decline of some caribou herds (Bergerud 1974; Miller 1980). However, after about 200–300 years without a fire, lichens become senescent and grow more slowly (Klein 1982).
Because abundance and distribution of animal populations depends on the availability of suitable habitat, habitat management should be explored as a management tool, especially where studies of habitat and surrogate measures of habitat quality (responses of ungulates such as age at first reproduction, reproductive rates, and growth rates) indicate that habitat quality is depressing the growth of ungulate populations. If the data indicate that a population is limited by habitat (especially food), habitat management programs (such as burning or crushing of vegetation) might be effective. Habitat management is a long-term process and requires long-term commitment. It will not solve short-term ''emergency" situations, but it can improve food availability and it might buffer populations during catastrophic events, such as deep persistent snow. Finally, because habitat management is generally socially acceptable, it offers the potential to decrease or
avoid the use of controversial methods (such as killing predators) to increase ungulate populations.
Wolf Ecology
Distribution and Density
Wolves occur throughout the Northern Hemisphere wherever large ungulates occur, from about 20° N latitude to the polar ice pack. They are present in habitats ranging from deserts to tundra, and although they still occur as far south as Saudi Arabia and India, they are most common in more-northern areas of Alaska, Canada, and Russia. In Alaska, wolves are found over most of their historical range, occupying about 85% of the state's 1.5 x 106 km2 (figure 3.2; Stephenson and others 1995). Wolves are absent from areas that they did not colonize after the last glacial recession, including the Aleutian, Kodiak, Admiralty, Baranof, and Chichagof islands.
Wolf densities vary geographically. In Alaska, wolf densities range from about 2 to 20/1,000 km2; total numbers were estimated at 5,900–7,200 during the winter of 1989–90 (Stephenson and others 1995). In the Arctic, densities of wolf populations are often less than 5/1,000 km2, but maximal midwinter wolf densities in southern populations often exceed 40/1,000 km2 (Fuller 1989a).
On the basis of data from more than 20 intensive studies that measured total average ungulate biomass (often more than 1 ungulate species) and average wolf populations for a period of several years, variations in wolf density in all of North America seem to be strongly correlated with variations in ungulate biomass (Fuller 1989a, 1995; Messier 1985). The relationship between prey abundance and wolf numbers can vary in areas with migratory versus nonmigratory prey or where prey concentrate seasonally. However, all available data suggest that, unless artificially depressed by humans, wolf numbers are typically limited by ungulate numbers and availability. Food availability is the dominant natural factor that limits wolf abundance.
Although the correlation between wolf density and prey abundance is high, the ratio of ungulate biomass to number of wolves is highest for heavily exploited wolf populations (Ballard and others 1987; Peterson and others 1984) or newly protected populations (for example, Fritts and Mech 1981; Wydeven and others 1995) and lowest for unexploited populations (Mech 1986; Oosenbrug and Carbyn 1982) or those where ungulates are heavily harvested (Kolenosky 1972). In Alaska, lightly harvested wolf populations occurred at much higher densities per unit of ungulate availability than heavily harvested populations (Gasaway and others 1992).
Changes in wolf density in response to varying prey density have been documented by long-term studies in northeastern Minnesota (Mech 1977, 1986), Isle Royale (Peterson and Page 1988), and southwestern Quebec (Messier and
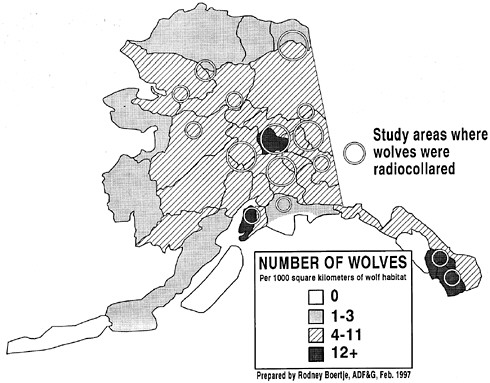
FIGURE 3.2 Alaska wolf population densities as estimated by Alaska Department of Fish and Game. Management biologists with aerial winter surveys and by contacting area trappers and pilots. Research biologists have radio-collared wolves from several packs in 11 Alaska study areas to verify estimates of wolf densities.
Crête 1985). Because the numerical response of an individual wolf population lags behind a change in prey density by up to several years, the ratio of ungulate biomass to wolves often differs among years and areas (Peterson and Page 1983).
Wolf densities can also vary with prey type. Population densities per unit of prey biomass are lower in areas where wolves prey mainly on moose than where they prey mainly on deer. The reason appears to be that moose are, on the average, less vulnerable to wolf predation (harder to catch) than are deer. Vulnerability of individual prey species may depend on which other prey species also are found in the area. For example, caribou are more vulnerable to wolf predation when they co-exist with moose, but moose and sheep are less vulnerable when caribou are present (Bergurud 1974, Dale and others 1995, Seip 1992).
In Alaska, wolf populations are estimated at different levels of precision, depending on management needs. The least-precise estimates are derived from a combination of information resulting from aerial surveys of wolf tracks, incidental observations, reports from the public, and sealing (mandatory registration)
records. Such assessments often are made for a given game-management unit or subunit (for example, Hicks 1994) in the fall (pre-trapping season) and spring (post-trapping season) to make use of harvest data and to identify the annual population low that occurs in the early spring before pups are born. Aerial track surveys are conducted 1-3 days after a fresh snowfall (Stephenson 1975). Flight patterns concentrate on terrain on which tracks are visible and over which wolves are likely to travel. Wolf tracks are followed until wolves are sighted or until wolf numbers can be estimated from tracks. Such surveys provide an estimate of the minimal number of wolves in an area, and their accuracy depends on ease of observation and the intensity of search effort. Observations of wolf packs, by whatever method, that note location, pack size, and color composition, and harvest information obtained from sealing of wolf pelts can augment aerial-survey data and indicate the location of packs that were not detected by aerial surveys. These low-precision estimates are used throughout Alaska (except southeastern Alaska), where populations of neither wolves nor their prey are thought to be changing dramatically and where no management is contemplated in the near future.
The most-precise estimates come from censuses of radio-collared packs (as in Fuller and Snow 1988). During the last 25 years, nearly every well-studied wolf population has been studied by radio-marking one or more wolves in most of or all the packs in a study area of 1,000-5,000 km2. During the winter, all packs are counted by radio-tracking and searching for tracks or wolves in areas where no packs are radio-collared but that are large enough to have a resident pack. Numbers of lone wolves are estimated by using the rate at which local pack members are known to disperse and settle, or by using rates determined for other populations (usually 10–15‰). In Alaska, such intensive censuses have been conducted over a number of years in at least 9 areas where detailed predator-prey interaction studies were being conducted (figure 3.3 ).
Rigorous, statistically designed aerial track-sampling surveys have recently been conducted in several areas of Alaska (Ballard and others 1995; Becker and others in press). The method requires snow in which wolves leave continuous tracks, and a recent snow or windstorm is required to identify fresh tracks. Tracks are followed, and the size of the group making the tracks is identified. Comparisons with telemetry-derived census data have shown the technique to be accurate. It holds promise for making statistically valid estimates of wolf abundance over large areas that cannot be studied with radiotelemetry or do not merit the expense of more-intensive censuses.
Habitat and Diet
Four factors dominate wolf population dynamics: wolf density, ungulate density, human exploitation, and ungulate vulnerability (Fuller 1989a, 1995; Keith 1983). Rabies and other diseases or parasites might infrequently limit local wolf
numbers, but no studies have suggested that they are a common limiting factor. Food habits of wolves have traditionally been assessed by analyzing scats (feces) during snow-free seasons and by finding kills in the winter. Although beavers, hares, and other small mammals can be seasonally important in some areas, they have not been shown to control either the distribution or the abundance of wolves. Vegetation type makes little difference to wolves as long as populations of hoofed prey are available. Prey availability is determined by both abundance and vulnerability. Deep snow or disease can make some prey more vulnerable and thus more "available" (Mech and others 1995). Availability of food ultimately affects nutritional levels and thus wolf reproduction, survival, and behavior.
Wolf populations are also influenced by hunting, trapping, and other control activities, but they seem little affected by snowmobiles, cars, trucks, logging, mining, and other human activities, except for accidental killing by humans or intentional killing where hunting or trapping for wolves or other species occurs.
Because wolves are great dispersers and can move to new areas fairly easily, wolf populations in marginal habitats (that is, where food resources are poor because of competition with humans or where there is high human-caused mortality) can be augmented by individuals from adjacent source or reservoir populations. Thus, the distance between populations plays an important role in wolf population ecology.
In sum, good wolf habitat is where ungulates are abundant and available, and where adverse human effects on wolves are low.
Social Structure and Movements
Packs originate when male and female wolves pair and produce pups. These wolves typically are dispersers from different packs that meet, travel together, find an "unoccupied" territory, and settle down. One of the wolves might have already settled in an area or might be the sole survivor of a pack with which a dispersing wolf of the opposite sex pairs. Packs vary in size from 2 to 25 or more. Average pack sizes, which usually range from 3 to 10, do not differ substantially among wolf populations whose major prey are different; that is, packs feeding mainly on black-tail deer are not usually larger than those feeding on caribou. Pack sizes also are similar at both high and low prey densities (but see Messier 1085).
Reproduction
Female wolves older than 22 months are capable of producing pups every year, but in a given year many potentially reproductive females in a pack do not produce pups. In particular, most wolf packs produce only 1 litter of pups per year (Packard and Mech 1980; Packard and others 1983), although 2 litters per pack have been reported (Ballard and others 1987; Harrington and others 1983;
Mech and Nelson 1989; Murie 1944; Peterson and others 1984; Van Ballenberghe 1983). If there are more than 2 female wolves of reproductive age in a pack, some do not breed. Consequently, populations with larger packs have a lower proportion of breeders (Ballard and others 1987; Peterson and others 1984). Peterson and others (1984) found that on the Kenai Peninsula in Alaska, increased harvest of wolves resulted in smaller packs and territories and establishment of new packs in vacated areas; as a result, breeders made up a higher proportion of the population, and the rate of pup production increased. It is not known why 2 females per pack produce pups in some areas more frequently than others (for example, Denali Park, Alaska). Pack sizes are not characteristically higher in these areas than in other geographically similar areas with similar prey bases.
In the 1960s, litter size was small in an unexploited population in Ontario (average, 4.9; Pimlott and others 1969) but large in exploited populations in Alaska (average, 6.5; Rausch 1969) and northeastern Minnesota (average, 6.4; Stenlund 1955). These reports led Van Ballenberghe and others (1975) and Keith (1983) to suggest that litter size might increase with greater ungulate biomass per wolf. More recent data from Alaska strongly confirm the assertion: litter sizes increased by 50% when available ungulate biomass per wolf increased by a factor of 6 (Boertje and Stephenson 1992).
Measurements of reproductive rates are based on examination of carcasses of adult females and, more rarely, observation of wild wolves (usually radio-collared) at den sites. The proportion of pups in early-winter populations is sometimes assessed by aerial observations (Peterson 1977).
Survival Rates and Causes of Mortality
Wolf survival is usually assessed in 2 ways. Indirectly, differences in consecutive population counts and known deaths due to "harvest" are used to estimate the proportion of wolves that have died. More directly, radio-collared wolves are monitored, and the rates at which they die are extrapolated to obtain an annual estimate of survival.
Survival of wolf pups in summer is difficult to measure, but data strongly suggest that survival is positively correlated with prey availability. Pup survival over the summer was almost double (89% compared with 48%) where per capita ungulate biomass was 4 times greater (Fuller 1989b). In n northeastern Minnesota, pup survival decreased in years when the ungulate food base declined (Mech 1977; Seal and others 1975; Van Ballenberghe and Mech 1975). The percentage of pups in the population or in packs is highest in newly protected populations (Fritts and Mech 1981) and heavily exploited populations (Ballard and others 1987)—situations in which litters are large, pup survival is good, and ungulates are abundant (Harrington and others 1983; Keith 1974, 1983; Pimlott and others 1969).
Wolves die of a variety of natural factors, such as starvation, accidents, disease, and intraspecific strife. In populations where some human-caused deaths occur (and thus compensate for natural mortality), about 8% of individuals greater than 6 months of age can be lost each year (Ballard and others 1987; Fuller 1989a). On Isle Royale, where there are no human-caused deaths, annual-mortality due to starvation and intraspecific strife (both related to relatively low food availability) ranged from 18% to 57% during a 20-year period (Peterson and Page 1988). Rabies, canine distemper, parvovirus, and such parasites as heartworm and sarcoptic mange, might be important causes of death, but documentation is scarce (Brand and others 1995).
Legally regulated killing of wolves can reduce or eliminate a population. Where focused wolf reduction programs have occurred, populations have been reduced by more than 60% in some years. In a few cases, site-specific control programs have eliminated entire packs (Fritts and others 1992). Even in some legally protected populations in Minnesota and Wisconsin, human-caused wolf deaths reached 20%–30% per year (Berg and Kuehn 1982; Fritts and Mech 1981; Fuller 1989a; Wydeven 1993).
Rates of Population Change
Under favorable conditions, wolf populations can increase as rapidly as 50% per year (Hayes 1995). Potential rates of increase are higher after exploitation because increased per capita food availability results in increased pup production and survival (Keith 1983). Immigrations from surrounding areas could push the rate of increase even higher.
Given normal levels of natural mortality, sustainable harvest of wolves is probably less than 30% of the early winter population (Keith 1983). Wolf density on the Kenai Peninsula declined after 2 annual kills of greater than 40% but increased after harvests of less than 35% (Peterson and others 1984). Wolf populations remained stable after harvests of 16–24% of the early-winter population but declined by 20–52% after harvests of 42–61% of the population (Gasaway and others 1983). In the unexploited wolf population on Isle Royale in Michigan, annual mortality of adult-sized wolves was 13–16% when numbers were increasing or stable, 51% during a population crash, and 33–34% when the population later stabilized (Peterson and Page 1988). Wolf populations might be able to withstand harvests as high as 40% before the harvest causes a population decline. Determining the exact magnitude of harvest that will cause a wolf decline is difficult and depends on the methods of analysis used (Ballard and others 1987; Fuller 1989a).
Dispersal is a major means by which wolves expand their geographic range or immigrate to populations that could not otherwise sustain themselves. Radiotelemetry studies show that a wolf usually leaves home by itself, finds a mate or another pack that accepts it, settles down, and either begins its own pack or lives
in the new pack (Fritts and Mech 1981; Rothman and Mech 1979). The proportion of dispersing lone wolves in a population varies seasonally, but a variety of studies have documented or estimated that lone wolves typically make up about 10–15% of a wolf population (Fuller 1989a).
Dispersing wolves typically establish territories or join packs within 50–100 km of the pack in which they were born (Fritts and Mech 1981; Fuller 1989a; Gese and Mech 1991; Wydeven and others 1995). However, dispersing wolves sometimes move longer distances. Fritts (1983) observed a wolf that traveled at least 550 km. Radio-collared wolves have traveled long distances between Wisconsin and Minnesota or Ontario (Mech and others 1994; Wydeven and others 1995), and a number of wolves have dispersed from western Minnesota far out onto the Dakota Plains (Licht and Fritts 1994). In Alaska, it is fairly common for wolves to disperse hundreds of kilometers from their natal range (figure 3.3; Stephenson and others 1995). Thus "connectivity" of populations is relatively high (Fritts and Carbyn 1995).
Consequences of Control on Wolf Populations
When wolves have been intentionally harvested heavily in 1 or more years during a control effort, their numbers have declined precipitously; but when control efforts have ceased, wolf numbers have rebounded to 88–112% of pre-control densities within 3–5 years (table 3.1). In south-central Alaska (GMU 13) wolf numbers increased from minimum post-control density by only 85%, but legal harvest was continued after control ended to keep wolf numbers from reaching pre-control density. Annual rates of increase during such recoveries, and others rates for which no corresponding pre-control densities were calculated, averaged about 26% and were the result of dispersal of young wolves into new territories, rapid pair bonding, territorial establishment and breeding, and shifts of pack ranges. During population recovery, survival rates of wolves are typically very high, dispersal rates are low, reproduction is normal, and packs are likely to split (for example, Hayes 1995). It has been suggested that harvest of wolves can actually result in an increase in the number of packs in an area, but there are few data to support this (but see Peterson and others 1984). Given relatively abundant prey and an annual increase of 26% per year, wolf populations potentially can recover to pre-control numbers in as little as 3 years. When wolves have recolonized areas from which they were extirpated, their populations have also increased rapidly (for example, Fritts and Mech 1981; Wydeven and others 1995).
Although wolf populations can recover numerically from major control efforts within a few years, it has been suggested that managers need to consider the special features of wolf social organization and related behavior (Haber 1996). Some critics of wolf control have argued that human harvest of ungulates is biologically justifiable because ungulates have evolved as prey but that it is not justifiable for humans to kill wolves because wolves are highly evolved social
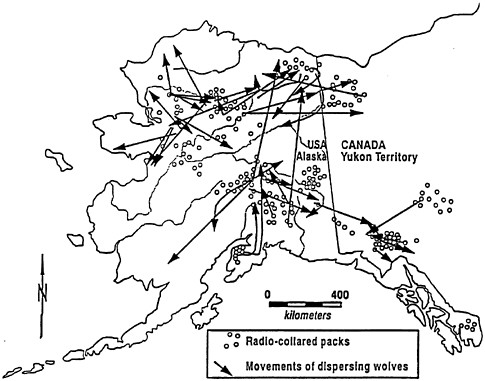
FIGURE 3.3 General location of radio-marked wolf packs. Wolf packs were studied in Alaska and the adjacent Yukon from 1975 to 1991, and known long-range movements of dispersing wolves. Packs were monitored for periods of 2 to 8 years (Stephenson and others 1995).
animals that have no evolutionary history of having been preyed on. In addition, it is suggested that harvesting wolves has lingering effects on the size, number, stability, and persistence of family-group social units (packs); on reproductive, hunting, and territorial behavior; on the role of learning and related traditions in wolf packs; on within-group and between-group patterns of genetic variation; and on overall mortality (Haber 1996).
Human hunting might be "unnatural" but nonetheless similar to other sources of wolf mortality. One way to evaluate the claim that hunting is more damaging to wolves than to ungulates is to compare the rates at which wolves die in hunted and unhunted populations. The demographic data on unharvested wolves presented by Haber (1996:1079-81) indicate that population turnover is high. Given an average pack size of 8, 1 litter per pack per year, a mean litter size of 5 (Boertje and Stephenson 1992), and 38% pups in packs in winter, 42% of pups and 36% of adults must die if the population is to remain stable. Where humans are not present, wolves typically die of starvation and intraspecific strife (wolves killing
TABLE 3.1 Changes in Winter Wolf Density After Various Amounts of Wolf Control
|
|
Precontrol Density |
Minimum Postcontrol Density |
|
|
|
Study Location |
Wolves/1,000 km 2 |
Wolves/1,000 km 2 |
Years Since control |
Percentage of Precontrol Density |
|
Areas where wolf control was carried out with quantitative precontrol data: |
||||
|
South-central Alaska (GMU 13) |
7.1 |
2.6 |
6 |
37% |
|
East-central Alaska (GMU 20E) |
8.1 |
4.1 |
1 |
51% |
|
East-central Yukon |
9.3 |
1.3 |
6 |
14% |
|
Southern Yukon |
12.4 |
3.6 |
3 |
29% |
|
East-central Alaska (GMU 20A) |
14.0 |
4.4 |
4 |
31% |
|
Areas where extensive wolf control was carried out without quantitative precontrol data: |
||||
|
South-central Alaska (GMU 13) |
NA a |
0.5 |
NA |
NA |
|
Western Alberta |
NA |
10.9 |
NA |
NA |
|
Kenai Peninsula, Alaska (GMU 15) |
NA |
0.4 |
NA |
NA |
|
a NA, Not Available. |
||||
wolves), at rates nearly as high as those during most control efforts. This suggests that much mortality resulting from control programs substitutes for mortality that would have occurred for other reasons. Haber (1996) described cases of long-term social stability among wolves in Denali National Park, such as one female who retained her alpha-female status within the pack for 13–14 years. However, based on their analysis based on genetic fingerprinting of wolf packs in and adjacent to Denali, Meier and others (1995) concluded that individual longevity and stability of sizes and composition of packs were less than previously thought, even where human disturbance is low.
Inevitably, the social organization and behavior of a species is influenced by
|
Density after Control Ended |
||||
|
Wolves/1,000 km(2) |
Years Since Control Ended |
Percentage of Precontrol Density |
Annual Rate of Increase, % |
References |
|
4.8 |
ó 8 |
68% |
8 |
Ballard and others 1987; Hicks 1994 |
|
7.4 |
4 |
91% |
16 |
Gasaway and others 1992 |
|
10.4 |
5 |
112% |
51 |
Farnell and others unpublished; Hayes 1995 |
|
10.9 |
3 |
88% |
17 |
Hayes and others 1991 |
|
15.7 |
12 |
112% |
11 |
Boertje and others 1995 |
|
6.6 |
12 |
NA |
24 |
Burkholder 1959; Rausch 1969 |
|
23.8 |
4 |
NA |
22 |
Bjorge and Gunson 1983, 1989 |
|
5.0 |
8 |
NA |
37 |
Peterson and others 1984 |
a natural annual turnover rate of more than one-third. Haber (1996) argues that because wolves are a highly social species, harvesting them ''implies a high potential for eventually reducing these complex societies to much simpler, more primitive forms, particularly when it is so likely to scramble their unusual genetic and cultural information transfer processes." He suggests that such problems will "ultimately translate into a major, long-term declining in numbers." However, no rigorous review of ecological or genetic data has indicated any long-term demographic consequences of human harvest, even when control efforts result in annual mortality higher than the average for unexploited populations (compare Haber 1994; Lehman and others 1992; Mech 1994; Meier and others 1995; Wayne
and others 1995). The reason might be that local control efforts actually mimic epidemics of disease that historically must have eliminated whole subpopulations of wolves. If that is so, it should not be surprising that wolf populations can recover quickly from control efforts with no evident genetic effects.
Different methods of wolf reductions might affect social structure. For example, hunting, snaring, and trapping tend to take subordinates and dispersing individuals, whereas air-assisted wolf reductions can eliminate whole packs (McNay, ADFG, personnel communication). However, there are no published data with which to assess changes in social structure resulting from different harvest methods.
Arguments that harvest of wolves is ethically unacceptable because of the intelligence and social complexity of wolves (Haber 1996) can be neither supported nor rejected on scientific grounds. However, no available data suggest that the killing of wolves by humans has adversely affected the long-term social organization, reproductive rates, or population dynamics of the species.
Summary
The most important factor is the availability of ungulates such as moose, caribou and sheep for food. Alternative prey, such as hares and beaver can be important when ungulate populations are extremely low. The sources of wolf mortality are intraspecific strife, disease, parasites, and legal and illegal harvest by humans. Wolf populations have high reproductive rates and dispersal rates, so populations can recover rapidly from increased mortality or decreased prey availability.
Bear Ecology
Although bears are omnivores, they are members of the mammalian order Carnivora. Black bears are relatively docile and are almost always closely associated with forests, where cubs and even adults can climb trees to escape from danger. They have inhabited almost all forests of North America. Brown, or grizzly, bears are larger and generally more aggressive than black bears, a trait essential for defending their offspring or food in the more-treeless areas they inhabit.
The distribution of both species has decreased dramatically since Europeans began settling North America about 400 years ago. When pioneers settled the West, bears were viewed as a threat to be sought out and destroyed (Brown 1985; Storer and Tevis 1955). Wherever people and their livestock settled, bears were either eliminated or greatly reduced. In the lower 48 states, brown bears are found in only about 2% of their former range (Servheen 1990); and they have been extirpated in large portions of southern Canada (Banci and others 1994). Thus, the conservation of brown bears in regions inhabited by people is a major
management challenge. Black bears remain in many portions of the West, but they were extirpated over much of the eastern United States and parts of Canada. In portions of the eastern states and even areas of the West, maintaining black bear populations is an increasing conservation issue. In more-remote northern and mountainous areas, where bear populations are more secure, the management of garbage and other attractants, human settlement, habitat, acceptable hunting limits, and bear effects on ungulates are issues that face wildlife managers.
Distribution and Density
The 3 most important variables in determining bear distribution and numbers appear to be habitat quality, human density, and human behavior (McLellan in press). Habitat quality is closely related to the abundance, quality, and spatial, and temporal distribution of food. Because of the complexity of bears' diets, quantifying habitat quality on the basis of the availability of food is difficult. Body size has been used as a surrogate measure of habitat quality (Stringham 1980, 1990; Wielgus 1993), but it is also influenced by bear population density, genetic constraints, and seasonal availability of high-protein foods in otherwise-poor habitats.
Human density is relatively easy to measure; human behavior is much more difficult. In such areas as national parks, where human densities can be high but their behavior is closely controlled, people have relatively little effect on the numbers and distribution of bears. At the other extreme, a small number of armed people that practice poor garbage management can have serious effects on local bear populations; bears that are attracted by the garbage will be particularly vulnerable to human-caused mortality. Between those extremes lie a multitude of combinations of human densities and behaviors that influence bear numbers.
The density of brown and black bears varies widely across North America and in Alaska. The highest density of brown bears recorded was 551 bears/1,000 km 2, in Katmai National Park at the base of the Alaska Peninsula, where the abundance of salmon combined with good vegetation make optimal habitat conditions for bears (Miller and others 1997). Because Katmai is a national park, there is no legal hunting, and human visitors are strictly controlled. The other extreme of brown bear density is found along the fringe of brown bear distribution, where people are numerous and their behavior is anything but conducive to maintaining even a few bears. In other areas, low brown bear densities are due more to poor habitat than to the abundance and behavior of people. Such areas include the Tuktoyaktuk Peninsula in the Northwest Territories (Nagy and others 1983a) and the Arctic National Wildlife Refuge in northeastern Alaska, where there are only 4 bears/1,000 km2 (figure 3.4, Miller and others 1997). Brown bear densities in Alaska and elsewhere are greatly influenced by their access to salmon. Coastal populations that feed on salmon have densities of 191–551 bears/1,000 km2 (Miller and others 1997). Interior populations without salmon usually have
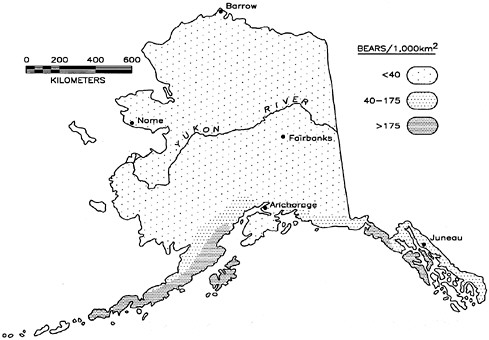
FIGURE 3.4 Distribution and density of Alaska brown bears.
densities of 4–47 bears/1,000 km2 (McLellan 1994, Miller and others 1997). However, southern interior areas with an exceptional diversity of bear foods (McLellan and Hovey 1994) had an estimated density of 80 bears/1,000 km2 (McLellan 1989).
Black bears are often found at much-higher densities than brown bears. On a small coastal, unhunted island of Washington state, the density of black bears (excluding cubs) reached 1,760/1,000 km 2 when the largely clearcut logged habitat was in optimal condition (Garshelis 1994; Lindzey and others 1986). On the mainland, density (excluding cubs) was estimated at 860 bears/1,000 km 2 in the Shenandoah National Park, a park with excellent bear habitat and where bears are protected (Carney 1985). The lowest recorded estimates of black bear densities have been 90/1,000 km 2, in the Susitna drainage of Alaska and in White Rock, Arkansas (Clark 1991; Miller and others 1997). The Susitna drainage is both poor black bear habitat, and has a substantial brown bear population that might restrict the black bears to a finger of streamside spruce forests (Miller and others 1997). The Arkansas study area supports a reintroduced population in which many females are shot by hunters.
Brown, or grizzly, bears are distributed over most of Alaska; exceptions are Kupreanof, Prince of Wales, and associated islands, Aleutian Islands (except Uniniak), Nunivak, and St. Lawrence Island. Except for Admiralty, Baranof,
Chichagof, and associated islands, black bears are found in most forested portions of Alaska (figure 3.4). Because they are found at low densities, move over large areas, sometimes live in densely forested areas, and hibernate in the winter, when tracks would otherwise be left, they are difficult to count. However, the Alaska Department of Fish and Game biologists have developed a successful procedure for estimating black and brown bear density in open or sparsely timbered areas. They establish a census area that is ecologically representative of the area of interest and then estimate density with a capture-mark-resight (CMR) method. Bears that are captured and radio-collared at least a year before the census constitute the marked sample. Several independent visual searches of the entire census area are conducted from a fixed-wing aircraft. On each search, the number of independent bears observed, the number of these that are radio-collared, and the total number of collared animals in the census area are recorded. Density estimates have been calculated by summing results of each search and correcting for the number of days searching (Miller and others 1987; Seber 1982). More recently, Miller and others (1997) have used statistical advances (maximal-likelihood algorithms for combining research results), which adjust estimates to account for emigration and immigration (White 1983). CMR estimates have been obtained for 15 brown bear and 3 black bear populations in 17 areas of Alaska (Miller and others 1987). Because a substantial proportion of the population must be marked in areas with low densities, the census areas are smaller than those for other large mammals on which it is less difficult to observe a significant portion of the population.
Habitat and Diet
Black and brown bears survive during the winter by retiring to dens, reducing their body temperature, and living off accumulated fat. They must ingest sufficient nutrients to meet the demands of growth and reproduction, and they must accumulate enough fat stores in a half-year to provide them with energy for the other half. Thus, during the active part of their year, feeding is their dominant activity.
Although bears have a carnivore's digestive anatomy and thus are relatively poor at digesting plant material, black and brown bears eat both plant and animal matter. The nutritional value of animal matter is higher and more consistent than plant matter, but plants are much more abundant and easier to obtain. When foraging for food, bears make tradeoffs between feeding on readily available but low-quality plant foods and seeking rare but high-quality animal matter. The quality and availability of plant food and the availability of animal matter vary widely among areas and over the seasons, and bear foraging behavior is consequently complex and dynamic.
In spring, the dominant plant foods of bears are new shoots of green vegetation, such as grasses, sedges, horsetails, and broad-leaved plants. These plants
are relatively abundant but contain only about one-fifth the digestible energy per unit of dry weight as ungulates (McLellan and Hovey 1994; Pritchard and Robbins 1990). Spring is also when ungulates are born, and both black and brown bears are relatively efficient at catching newborn ungulates during their first few weeks of life (Ballard and others 1990; Boertie and others 1988; Franzmann and Schwartz 1986; Franzmann and others 1980; Gunther and Renkin 1990). In some areas, adult ungulates are also more easily captured during spring than later in summer or fall (Boertje and others 1988). In portions of coastal British Columbia and Alaska that have abundant spring plant foods but relatively few ungulates, bears expend little effort seeking calves and tend to forage more at estuaries, riparian areas, and avalanche paths (Hamilton 1988; Schoen and Beier 1990). Where both ungulates and spring plant foods are abundant, bears might forage on vegetation and, once satiated, search for ungulate calves and vulnerable adults. When spring plant foods are not readily available, bears expend more effort to find ungulate calves and adults.
As the summer progresses and ungulate calves mature, they become quicker and more difficult for bears to catch (Gunther and Renkin 1990). Concurrently, the quality of plant foods increases greatly in many areas. Over most of western North America, fruits of huckleberries (Vaccinium spp.), soap berries (Shepherdia canadensis), and a great variety of other species become ripe. Fruits, although low in protein, are rich in energy and are highly digestible (McLellan and Hovey 1994; Pritchard and Robbins 1990). In some more southern and eastern areas, seeds from white bark pine (Pinus albicaulis) and oaks (Quercus spp.) are a rich source of energy. During the summer and fall, when bears acquire the bulk of the fat needed for winter dormancy, they feed mostly on salmon in coastal areas (Jonkel and Cowan 1971; Kingsley and others 1988; Reiner 1996). Feeding on ungulates can increase in the fall during the rutting season (Boertje and others 1988; Hamer and Herrero 1991) or when hunters wound their quarry or leave gut piles (McLellan and Hovey 1994).
Social Structure and Movements
Male bears spend most of their life as solitary individuals; females are either alone or with their latest litter of cubs. Exceptions are during mating and at concentrated food sources, such as salmon spawning streams. Individuals have home ranges or areas in which they live year after year. Within their home range, bears gain knowledge of the location and seasonality of resources, such as food. The size of a home range depends largely on the sex and species of the bear and the productivity of the habitat. Brown bears generally have much larger ranges than black bears, and male bears have much larger ranges than females. Subadult male bears ranger over large areas when they disperse and seek permanent home ranges.
Home ranges are typically smaller in high-quality than in poorer-quality habitats. For example, average male and female brown bear home ranges were 100 and 37 km 2, respectively, on the productive coastal Admiralty Island (Schoen and Beier 1990) but 3,757 and 884 km 2 respectively in Yellowstone National Park (Blanchard and Knight 1991). In most cases, the home ranges of individuals of both sexes overlap extensively. In some areas, some female black bears maintain exclusive home ranges or territories (Rogers 1987).
Reproduction
It is well established that small species tend to reproduce much faster than large species (Charnov 1982), but bears have even lower reproductive rates than their body size would predict. Bears have long interbirth intervals and are slow to mature. Female brown bears occasionally produce their first litter when they are 4 years old, but usually they are 5–8 years old. Female black bears can have their first litter when they are 3 years old, but more commonly not until when they are 4–6 years old. In the Susitna drainage of Alaska and the Flathead drainage of British Columbia, where research has been conducted on sympatric black and brown bear populations, the ages at first litter are similar in the two species (Garshelis 1994; Hovey and McLellan unpublished data; McLellan 1994).
An average interbirth interval of 2.6 years was recorded for brown bears on the East Front of Montana, where some extremely productive females had 2-year intervals. However, average intervals of 3–4 years are more common, and average intervals of more than 4 years are recorded in some areas of Alaska, including Kodiak Island and Black Lake on the Alaska Peninsula (Aune and Kasworm 1989; McLellan 1994). Black bear interbirth intervals are about a year shorter than those of brown bears; study averages range from 2 to 3 years.
Average interbirth intervals are probably longer than have been reported. Because of the duration of research projects and the battery life of radio collars, intervals of 2 or 3 years are more commonly recorded than longer intervals (McLellan 1994; Schoen and Beier 1990). That bias is particularly evident in some brown bear studies, in which females tracked for 9 years failed to produce a second litter (Sellers 1994).
Brown bear litter sizes range from an average of 1.7 cubs/litter (Glacier National Park Montana) to 2.5 cubs/litter (Kodiak Island and Black Lake), but litters of 4 are recorded (Martinka 1974; McLellan 1989, 1994; Pearson 1975). Average black bear litter size varied greatly among study areas across North America. In Arkansas, an average of 1.4 was recorded; in Pennsylvania, the average was 3.0 (Alt 1989; Clark 1991). In summary, brown bears usually do not produce a litter until they are 5–7 years old and produce only 0.45–0.85 cub/year, and black bears reach maturity about 1 year earlier and produce 0.55 to more than 1 cub/year.
Survival Rates and Causes of Mortality
The low reproductive rate of bears is balanced by their high annual adult survival rate of more than 92% (Eberhardt and others 1994; Hovey and McLellan 1996; McLellan 1989; Sellers 1994; Sellers and others 1993; Titus and Beier 1994). Survival rates of cubs are much more variable among study areas. Annual survival of brown bear cubs range from just over 35% in Katmai National Park (Sellers and others 1993) to 87% in Montana and southern British Columbia (Aune and others 1986, Hovey and McLellan 1996). Cub survival might vary with population density, but such relationships are unclear. Brown bear densities are high in both Katmai National Park and the Admiralty Islands, but cub survival rates are 35% and 80%, respectively (Schoen and Beier 1990). Reported annual black bear cubs has ranged from as low as 30% in some study areas to 90% in others (Garshelis 1994).
Bears die of a variety of causes. Natural causes include predation by other bears and other carnivores, accidents, and old age. Starvation of adult bears is rare but appears to be relatively common for young bears after years of poor food production (Schwartz and Franzmann 1991). In almost all study areas, people are the primary source of mortality for bears, legally or otherwise.
Dispersal
Dispersal rates and distances of bears are poorly known. In a Minnesota study, all male black bears dispersed 33–324 km from the study area when they were 2–4 years old (Rogers 1987). Similarly, all marked male black bears dispersed from a study area in Idaho (Beecham 1983), and male dispersal was common in Washington (Lindzey and Meslow 1977) and Arizona (LeCount 1982). In Arkansas, male black bears did not disperse from the study area (Smith and Pelton 1990). Female black bears typically remain in the maternal home range (LeCount 1982).
On the Tuktoyaktuk Peninsula, 3 subadult male brown bears were shot 25, 31, and 92 km from their mothers' home ranges (Nagy and others 1983a). In the Alaska Range, 1 subadult male brown bear was shot 32 km and a second shed his collar 37 km from maternal ranges (Reynolds and Boudreau 1990). Four subadult males moved an average of 70 km from their maternal ranges, but 1 was shot only 15 km from his maternal range (Blanshard and Knight 1991). As is apparent with black bears, subadult female brown bears tend to remain in or overlap their mothers' home range (Blanshard and Knight 1991; Nagy and others 1983b), but some females dispersed from a high-density population in the Flathead Valley (McLellan and Hovey unpublished data).
Rates of Population Change
The maximal reported rate of annual increase in a brown bear population is 8.5% in the Flathead drainage of British Columbia, an area with a great diversity of bear foods (Hovey and McLellan 1996; McLellan 1989; McLellan and Hovey 1994); this population is apparently increasing after overharvesting. Annual rates of change of 4.6% and 1.0% have been reported in National Park and the South Fork of the Flathead in Montana, respectively (Eberhardt and others 1994; Servheen and others 1994). Black bears reproduce at higher rates than brown bears and thus have a higher potential rate of increase; however, because of human harvest, this rate is rarely realized.
Given the great variation in brown bear reproductive rates in Alaska, there is potential for great variation in rates of increase or sustainable harvest. Population decreases due to intentional overharvest in areas with road access to hunters have been reported, as has a lack of reproduction in a poorly accessed area (F. Miller 1990; SD Miller 1995; Reynolds 1994).
Consequences of Control on Bear Populations
The results of bear control by lengthening of hunting seasons, translocations, or other methods are difficult to determine because bears are difficult to count. In addition, bear population densities are highly variable. Bears are easily overexploited and, unlike wolves, recover slowly from low densities. Coastal bears that have access to salmon have higher reproductive rates and can sustain higher harvest rates than interior bears, which lack this food source. It is the interior bears, however, that feed on ungulate calves. The low densities and low reproductive rates of these bears make bear control a delicate management decision. It is well established that bear populations can be reduced and even eradicated by overharvest. In addition, reduced reproduction due to social disruption by young immigrant male bears has been suggested in areas where brown bear numbers were reduced (Wielgus 1993; Wielgus and Bunnell 1994). It is clear that if bear control is conducted, it must be in conjunction with detailed monitoring of the bear populations to be controlled, to reduce the risk of local extirpation.
If given the opportunity, most (or all) bears would kill and eat an ungulate calf, but individual bears vary widely in predation success. Boertje and others (1988) found that 4 of the 9 bears they tracked killed 72% of moose calves that were killed by the 9. Ballard and others (1990) found wide variation in individual bear's predation rates. Many black and brown bears killed no moose calves while they were monitored, whereas 1 female brown bear killed 1 moose calf every 2.8 days and a black bear killed 1 moose calf every 5.7 days. Ballard and Miller (1990) noted that 2 identifiable brown bears were known to have killed 6 of the 12 radio-collared moose calves that died in their study. In a foraging study conducted in Yellowstone National Park, French and French (1990) observed
that some brown bears never fed on elk, whereas others fed almost exclusively on elk. Given this variation among bears, the outcome of bear control programs is highly unpredictable. It will depend on which bears are removed and the feeding habits of the removed bears. Not surprisingly, bear control experiments have yielded inconsistent and short-term effects on ungulate calf survival (Ballard and Miller 1990; Crête and Jolicoeur 1987; Miller and Ballard 1992; Schlegel 1983).
Summary
The diet of bears includes neonate and adult ungulates, especially moose and caribou. Salmon are are important when they are available, as well as ground squirrels and other small mammals. Bears also eat grasses, sedges, broad-leaved plants, fruits and seeds. Sources of mortality besides legal and illegal hunting by humans include intraspecific strife among bears, accidents, and rarely predation by wolves and disease. Humans affect the distribution of bears by their hunting pressure and how they manage garbage.
Caribou Ecology
Caribou (Rangifer tarandus) inhabit the high Arctic, tundra, boreal and subboreal forests, and wet interior mountains across the Northern Hemisphere. Caribou are found in most regions of Alaska (figure 3.5, table 3.2). All North American caribou are wild, except in a few areas where semi-domesticated varieties called reindeer have been introduced for herding by native peoples. In Eurasia, there are large herds of reindeer (Andreyev 1977). During the 1980s, most caribou populations in Alaska were increasing (Davis and Valkenburg 1991). In the early 1990s, most of the interior Alaskan herds began declining, while many of the smaller herds and the Western Arctic and Mulchatna herds (two of the largest herds in Alaska) continued to increase (Valkenburg and others 1996, table 3.2).
Social Structure and Movements
Caribou generally exist in separate herds or populations that annually move over extensive areas, sometimes migrating hundreds of kilometers between wintering areas and calving-summering grounds, behavior that is probably an antipredator strategy (Heard and others 1996; Seip and Brown 1996). Female caribou usually show strong fidelity to the specific calving grounds of the herd, returning each year to the same general area to give birth (Skoog 1968). Some herds migrate between summer and winter ranges, and all caribou shift their ranges in response to forage conditions. Short-term variation in habitat use and movements also can be stimulated by harassment by wolves (Scotter 1995).
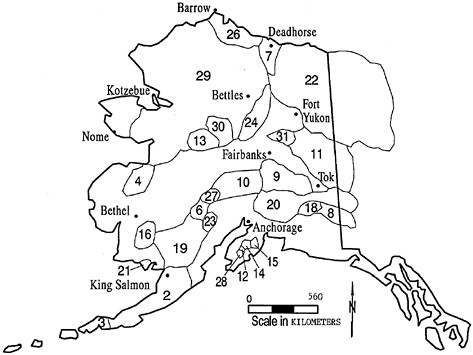
FIGURE 3.5 Approximate ranges of caribou herds in Alaska. SOURCE: Valkenburg and others 1996. Printed with permission from the author.
Caribou of the Western Arctic Caribou Herd in Alaska select calving grounds that have low snow cover and early snow melt (Lent 1980).
Knowing whether caribou regularly switch between herds is important to wildlife managers because if substantial numbers of caribou emigrate from herds that are increasing to areas where herds are small, resources used to increase the size of small herds might be wasted; immigration would ''rescue" the depressed herds. However, although caribou are highly migratory and herds sometimes intermingle, direct evidence of more than the occasional individual leaving one herd to join another is lacking. Exchange of a few individuals between herds is important from a genetic standpoint, but periodic exchanges of large numbers of individuals would be necessary to alter herd population dynamics. If large shifts of animals were regular, they should have been detected from the extensive radio-collaring of caribou in the Nelchina, Delta, Ashinik, and Fortymile herds.
Nevertheless, caribou herds can undergo rapid population fluctuations. For example, the Mulchatna Herd increased from about 30,000 to 110,000 animals between 1984 and 1993, and its range shifted to the west. When the Fortymile herd had 570,000 animals and ranged over an area 5 times as large as it occupies today (Boertje and Gardner 1996; Valkenburg and Davis 1986), it might have engulfed other herds. Maps in Valkenburg and Davis (1986) show that the
TABLE 3.2 Estimated Size and Approximate Density of Alaskan Caribou Herds
|
Herd No. |
Herd Name |
1993 Population Estimate |
Population Trend Since 1989 |
|
|
1 |
Adak |
750 |
up |
|
|
2 |
Alaska Peninsula (North) |
18,000 |
down |
|
|
3 |
Alaska Peninsula (South) |
2,500 |
stable |
|
|
4 |
Andreafsky |
<50 |
unknown |
|
|
5 |
Beaver Mountains |
649 |
unknown |
|
|
6 |
Big River |
750 |
unknown |
|
|
7 |
Central Arctic |
23,444 |
stable |
|
|
8 |
Chisana |
850 |
down |
|
|
9 |
Delta |
3,661 |
down |
|
|
10 |
Denali |
1,890 |
down |
|
|
11 |
Fortymile |
21,884 |
down |
|
|
12 |
Fox River |
75 |
up |
|
|
13 |
Galena Mountain |
275 |
up |
|
|
14 |
Kenai Lowlands |
100 |
stable |
|
|
15 |
Kenai Mountains |
300 |
stable |
|
|
16 |
Kilbuck Mountains |
2,500 |
up |
|
|
17 |
Macomb |
500 |
down |
|
|
18 |
Mentasta |
880 |
down |
|
|
19 |
Mulchatna |
110,000 |
up |
|
|
20 |
Nelchina |
40,361 |
stable |
|
|
21 |
Nushugak Peninsula |
750 |
up |
|
|
22 |
Porcupine |
165,000 |
stable |
|
|
23 |
Rainy Pass |
500–1,000 |
unknown |
|
|
24 |
Ray Mountains |
700 |
up |
|
|
25 |
Sunshine Mountains |
800 |
unknown |
|
|
26 |
Teshepuk |
27,630 |
up |
|
|
27 |
Tonzona |
800 |
down |
|
|
28 |
Killey River |
100 |
up |
|
|
29 |
Western Arctic |
450,000 |
up |
|
|
30 |
Wolf Mountain |
650 |
stable |
|
|
31 |
White Mountains |
1,000 |
up |
|
|
|
TOTAL (approximate) |
880,000 |
|
|
|
SOURCE: Valkenburg and others 1996. |
||||
Fortymile Herd historically occupied much of the current wintering area of the Porcupine Herd in the Ogilvie Mountains. Heard and Calef (1986) suggested, in the absence of reliable census and management data, that the increase in the Kaminuriak Herd on the calving grounds from 13,000 in 1980 to 41,000 in 1982 must have included immigration from other herds. Continued extensive monitoring of radio-collared animals and genetic analyses will assist in determining the
frequency and extent of exchanges of individual animals between herds. Meanwhile, it appears imprudent to rely on immigration as a substantial source of growth of depressed herds during time frames of a few years.
Habitat and Diet
Caribou are primarily grazers, but they occasionally browse. They eat a variety of species—lichens, willows, forbs, sedges, grasses, mosses, blueberries, and mushrooms (Klein 1970; White and Truddell 1980). In summer, caribou forage primarily in areas with high species diversity and in microhabitats that provide high-quality forage (White and Trudell 1980). In the winter, they move to areas dominated by lichens (Boertje 1984; Holleman and Luick 1977; Klein 1982). In southern boreal forests or in subalpine habitats in Canada, arboreal lichens often make up the bulk of the caribou winter diet; in tundra areas and in the northern fringes of the boreal forest, terrestrial lichens dominate (Boertje 1984; Klein 1982). Lichens can be adversely affected by overgrazing and trampling (Pegau 1970, 1975). Stands of lichens have deteriorated from excessive grazing by caribou and trampling by caribou, moose, and domesticated reindeer (Gaare and Skogland 1980; Pegau 1975; White and Trudell 1980).
Caribou have especially high nutritional requirements during the summer because their offspring grow very rapidly (McEwan 1968). Consequently, in summer, caribou are selective feeders, seeking out new leaves of willow and sedges and buds and flowers of the sedge known as cotton grass and other flowering plants of the tundra (Klein 1970). Caribou move to southern exposures at the beginning of the season, where plant growth begins sooner. By late summer, nutritional needs of caribou have shifted from a high protein requirement for milk production and body and antler growth to an emphasis on carbohydrates needed to lay down fat stores for the coming winter.
In winter, caribou dig through snow to obtain food; because they are constantly on the move in very cold weather, they require forage of high energy value (Klein 1992). Lichens are an ideal winter food for caribou because they are high in carbohydrates that provide the energy to meet their metabolic needs in winter. Although lichens are poorly digested and avoided by most other herbivores, caribou are well adapted to digest them and have unique microorganisms to aid the process in their rumens.
Caribou can maintain their body temperatures at ambient temperatures of at least -35°C without increasing their metabolic rates (Hart and others 1961). Moose are less well insulated, but they can maintain their body temperatures without increasing metabolic rates when ambient temperature is as low as -32°C (Renecker and Hudson 1986). At still lower air temperatures, they must expend extra energy to stay warm, and strong winds effectively lower ambient temperatures (Bergerud 1983).
During summer, caribou are attacked by mosquitoes, warble and bot flies,
and biting flies, particularly when weather conditions are favorable for insect flight. Caribou respond to mild harassment by mosquitoes by feeding while facing the wind (White and Milan 1981), but when insect harassment is severe, caribou stop feeding and seek relief by running, standing in water or on snow patches, forming tight herds (with constant jostling), or moving to windy areas (Batzli and others 1981; Helle and others 1992; White and others 1975). Those activities interfere with growth and fattening (Reimers 1980) during summer, when energy requirements are highest (Boertje 1985). Body fat reserves of caribou in the Northwest Territories decline to almost zero in July; when insect numbers decline in August caribou reach their highest yearly levels of body fat (Heard and others 1996).
The effect of snow on wildlife has been studied extensively in Europe and North America (Peek 1974; Pruitt 1981). Snow accumulations vary with climate, storm tracks, moisture content, redistribution by wind, topography, and forest cover. Snow adversely affects moose and caribou by burying plants, by reducing the animals' ability to locate plants by smell (Leader-Williams and others 1981), and by increasing the energy cost of locomotion (Parker and Robbins 1984; Telfer and Kelsall 1984). Deep snow that has a hard-packed surface can increase ungulate vulnerability to wolf predation because wolves, but not moose and caribou, can run over the surface without sinking (Mech and others 1995; Nelson and Mech 1986).
Caribou populations are often regulated by winter food supplies. In addition, reproductive rates are lower and juvenile survival rates are higher when summer food supplies are low. Caribou are smaller where they live at high densities (Skogland 1985).
Summary
In summer caribou eat broad-leaved plants, willows and sedges and their supply and quality may vary in relation to both previous range conditions and the number of shady days in the current growing season. Nutrient quality of some forage plants is improved by more shady days. The winter diet is largely slow-growing lichens in areas that have not been burned for more than 50 years. Caribou are vulnerable to starvation and predation by wolves in severe winters and to predation on neonates and adults on calving grounds. Brown bears and black bears also take neonate caribou, as do eagles. Caribou numbers can also be affected by outbreaks of mosquitoes, flies and disease, and by other human disturbances associated with roads and oil fields.
Moose Ecology
Distribution and Density
Moose occur throughout northern mountains, boreal forest, and riparian areas of the Northern Hemisphere. Moose appear to have been absent or rare in many areas of Alaska as recently as 75 years ago (Huntington and Rearden 1993, Spalding 1990). Today moose are most abundant on the Kenai Peninsula, the lower Matanuska and Susitna drainages, and locally throughout the interior and western Alaska in riparian communities associated with major river systems.
Social Structure and Movements
Most of the year moose are solitary or live in small groups of bulls or of females and offspring (Miquelle and others 1992). The breeding season of moose varies with locality, but generally begins in late September and continues to early November (Van Ballenberghe and Miquelle 1993). During this period, mature bulls devote most of their time to seeking copulation and can lose up to 25% of their body mass. Females continue to feed and gain body condition that will be needed for pregnancy over the lean winter months. During the rut, female moose in open habitats may be aggregated into larger groups of breeding harems by dominant bulls. In winter, moose sometimes assemble into larger herds. Moose are typically not migratory, although they can shift between summer and winter ranges. Some moose that summer on alpine ranges winter at lower elevations.
Habitat and Diet
Moose are most abundant in early successional-stage habitats that develop after fires, floods, logging, and land-clearing. Moose eat primarily leaves and tender stems of woody plants, but during summer they also eat herbaceous plants. In winter, they prefer to browse on species that are relatively high in protein and carbohydrates, such as willow, aspen, and birch; the former are found in thickets along rivers. During late summer, moose accumulate fat deposits for use in winter and, among adult males, for use in the rut. The winter diet of moose is dominated by twigs of woody shrubs grown during the previous summer. The bark and buds of these young woody twigs contain high concentrations of available nutrients.
Summary
Moose depend on access to shrubby plant growth, especially willows, whose availability is best 10–30 years after a fire, but is also affected by browsing history by moose, snowshoe hares and the recent history of snow depth and
condition. Plant nutrient quality is improved if there have been more shady days in the growing season (Molvar and others 1993). During plant succession conifers crowd out deciduous browse. Improvement of resources for moose must be considered in their management.
Long winters and deep crusted snow can increase mortality or weaken moose so that they are more vulnerable to predation. Levels of predation by wolves are affected in turn by human harvest of wolves and other factors that affect wolf populations. If moose have access to wetland habitats, they can more readily escape from predation by wolves. Brown and black bears often prey heavily on moose, especially newly born calves in some areas.
Comparative Ecology of Moose and Caribou
Reproduction, Survival Rates, and Causes of Mortality
Moose and caribou differ strikingly in their basic reproductive biology (table 3.3). In habitats of moderate to high quality, many female moose reach breeding age in their second year and give birth to their first calf at the age of 2 years (Franzmann 1981). In later pregnancies, twinning is common and triplets are occasionally born. Female moose with more than 1 calf, however, are more likely to lose a calf to predators, accidents, or other causes than are females with a single calf.
Caribou, in contrast, always give birth to a single calf, and most females do
TABLE 3.3 Comparative Population Characteristics of Moose and Caribou a
not reach breeding age until their third year, although under favorable nutritional conditions some female caribou bear their first calves at the age of 2 years. In both caribou and moose, the pregnancy rate increases until females reach the age of 4 or 5 years and declines in very old animals (Dauphine 1976; Franzmann 1981). Because of those differences, moose populations can increase and recover from depression faster than caribou if the quality and quantity of forage are adequate.
The ratio of calves to cows is relatively easy to obtain, but identifying the cause of a low ratio is difficult and expensive. Calves that are born dead are more easily discovered than calves taken by predators. Nevertheless, predation rates are known to be high among calves during their first few weeks of life (tables 3.4 and 3.5). Compared to wolves, predation by bears is a significantly greater source of mortality for moose calves and, in some cases, for caribou calves (tables 3.4 and 3.5; Osborne and others 1996).
The high annual survival rates of adult female moose and caribou, which are usually above 85 %, mean that these species can thrive even when calf mortality is substantial, because most cows will live to reproduce another year, but excessive calf mortality can cause a population collapse (Bergerud 1996; Larsen and others 1989).
TABLE 3.4 Cause-Specific Mortality (%) in Radio-Collared Caribou Calves in Areas of Southern Alaska and British Columbia
|
|
|
Predator |
Other Mortality |
|
||||
|
Area |
No. of Calves |
Wolf (%) |
Brown Bear (%) |
Other (%) |
Unknown (%) |
Not Predation (%) |
Unknown Cause (%) |
TOTAL (%) |
|
Denalia |
226 |
14 |
20 |
2 |
9 |
1 |
4 |
50 |
|
Deltab |
93 |
25 |
22 |
15 |
0 |
0 |
4 |
66 |
|
Fortymilec |
109 |
24 |
17 |
12 |
0 |
6 |
6 |
65 |
|
Spatsizid |
34 |
24 |
9 |
0 |
0 |
15 |
9 |
57 |
|
Average |
|
22.0 |
17.0 |
7.3 |
2.3 |
5.5 |
5.8 |
59.5 |
|
a Adams and others (1995): birth to 1 yr of age. b Valkenburg and others (1996): birth to September 30. c Boertje and Gardner (1996): birth to 1 yr of age. d Page (1985): birth to 4 months of age. |
||||||||
TABLE 3.5 Cause-Specific Mortality (%) in Radio-Collared Moose Calves in Alaska and the Yukon
|
|
|
Predator |
Other Mortality |
|
|||||
|
Area |
No. of Calves |
Wolf (%) |
Brown Bear (%) |
Black Bear (%) |
Other (%) |
Unknown (%) |
Not Predation (%) |
Unknown (%) |
TOTAL (%) |
|
Susitna, June–Oct.a |
198 |
3 |
44 |
2 |
1 |
2 |
8 |
3 |
63 |
|
Susitna, Nov.–May |
114 |
0 |
2 |
0 |
0 |
1 |
10 |
0 |
13 |
|
Yukonb |
117 |
18 |
42 |
3 |
0 |
3 |
6 |
9 |
81 |
|
Tananac |
40 |
0 |
10 |
13 |
0 |
0 |
0 |
10 |
33 |
|
GMU20Ed |
33 |
12 |
52 |
0 |
0 |
0 |
12 |
0 |
79 |
|
Kenai, 1947e |
47 |
6 |
6 |
34 |
0 |
2 |
4 |
4 |
69 |
|
Kenai, 1969f |
74 |
1 |
3 |
35 |
0 |
5 |
1 |
50 |
|
|
Averageg |
|
6.7 |
26.2 |
15.0 |
0.2 |
2.0 |
5.8 |
4.5 |
60.3 |
|
a Ballard and others (1991): wolves had been decreased, bears also decreased late in neonatal mortality study. b Larsen and others (1989): annual survival rates were measured during a period when 55% of the wolves were removed annually. 29.9 calves/100 cowsin fall and 19.5 calves/100 cows in spring. c Keech (in preparation): calf survival was measured from birth to 4 mo of age. d Gasaway 1992): wolves had been reduced by 60%; 22 calves/100 cows in winter; adult mortality of 9% to 11 months of age. e Franzmann and others (1980): 1947 burn; survival to about 2-2.5 mo of age. f Franzmann and Schwartz (1986): 1969 burn; survival to 4 mo of age. g Average excludes Susitna, November–May. |
|||||||||
Habitat Quality and Ungulate Population Dynamics
Habitat quality is an important determinant of dynamics of populations of large mammalian herbivores (Caughley 1981; Crête and others 1990; Jeffries and others 1994; Pastor 1988). Knowledge of seasonal and annual variability in forage quality and quantity is important for development of management plans for ungulate populations. In Alaska, habitat carrying capacities have been assessed primarily by such indirect indicators as the length of the lower jaw, marrow fat, and body weight of individual animals, or such demographic data as birth rate, calf survival, or population age structure. Those indirect measures, which assume that the status of the animals reflects the status of their habitats, have proved useful when time, expertise, or funding was not available for direct assessment of the habitats. However, those measures are often slow to respond to habitat changes, can be unduly influenced by short-term weather anomalies, and are influenced by factors other than habitat quality.
A few long-term studies involving experimental manipulations and monitoring of habitat changes caused by these and natural disturbances, such as fire, have been undertaken. The collaborative research on moose-habitat relationships by ADFG and the Fish and Wildlife Service (FWS) in the Kenai National Wildlife Refuge (formerly the Kenai National Moose Range) is an excellent example. ADFG, with management responsibility for wildlife, and FWS, with responsibility for habitat, entered into a cooperative agreement that yielded basic information on responses of moose habitat to fire and other manipulations in the Kenai Peninsula and on the physiological, nutritional, and population consequences of habitat changes, constraints, and stocking levels (Oldemeyer and Regelin 1987; Regelin and others 1987; Schwartz and others 1988). Information gained from this long-term effort has been extremely valuable in the management of moose in Alaska and especially on the Kenai Peninsula.
Intensive habitat-relationship studies like those carried out on the Kenai need not be duplicated in each area where ADFG plans to intensify management activities, but some level of knowledge of the existing habitat is essential if wise decisions are to be made. Desirable information includes description of vegetation, fire history, proportions of habitat in different successional stages after fire, rough biomass estimates of critical forage types (for example, lichens on caribou winter ranges and primary winter browse species in moose habitats), and distribution and depth of snow cover in the winter.
REFERENCES
Adams LG, BW Dale, and LD Mech. 1995. Wolf predation on caribou calves in Denali National Park, Alaska. In LN Carbyn, SH Fritts, and DR Seip, Eds. Ecology and Conservation of wolves in a changing world: proceedings of the second North American symposium on wolves. Canadian Circumpolar Inst. Univ. Alberta, Edomonton, 1995.
Alaska Bureau of Vital Statistics. 1995. Population trends in Alaska.
Alt GL. 1989. Reproductive biology of female black bears and early growth and development of cubs in Northeastern Pennsylvania. Ph.D. thesis, WV Univ, Morgantown. 116 Pp.
Andreyev VN. 1977. Reindeer pastures in the subarctic territories of the USSR. Pp. 275–313 in W Krause, Ed. Handbook of vegetation science, part XIII. Application of vegetation science to grassland husbandry. Dr. W. Junk BV publishers, The Hague, Netherlands.
Aune K and W Kasworm. 1989. Final report. East front grizzly studies. Mont Dep Wildl Parks, Helena. 332 Pp.
Aune M Madel, and C Hunt. 1986. Rocky mountain front grizzly bear monitoring and investigation. Mont Dep Wildl Parks, Helena. 175 Pp.
Ballard WB, JS Whitman, and CL Gardner. 1987. Ecology of an exploited wolf population in southcentral Alaska. Wildlife Monographs 98:1–54.
Ballard WB and SL Miller. 1990. Effects of reducing brown bear density on moose calf survival in south-central Alaska. Alces 26:9–13.
Ballard WB, SD Miller, and JS Whitman. 1990. Brown and black bear predation on moose in southcentral Alaska. Alces 26:1–8.
Ballard WB, JS Whitman, and DJ Reed. 1991. Population dynamics of moose in southcentral Alaska. Wildlife Monographs 114:1–49.
Ballard WB, CL Gardner, and DJ Reed. 1995. Use of line-intercept transects for estimating wolf densities. In LN Carbyn, SH Fritts, and DR Seip, Eds. Ecology and conservation of wolves in a changing world. Canadian Circumpolar Inst., Univ. Alberta, Edmonton.
Banci V, DA Demarchi, and WR Archibald. 1994. Evaluation of the population status of grizzly bears in Canada. Int Conf Bear Res and Management 9:129–142.
Batzli GO, RG White, and FL Bunnell. 1981. Herbivory: a strategy of tundra consumers. International Biological Programme 25:359–375.
Becker E and others. In press. A population estimator based on network sampling of tracks in the snow. J Wildlife Management.
Beecham JJ. 1983. Population characteristics of black bears in west central Idaho. J Wildlife Management 47:405–412.
Berg WE and DW Kuehn. 1982. Ecology of wolves in northcentral Minnesota. In Pacquet, Eds. Wolves: a worldwide perspective of their behavior, ecology and conservation. Noyes Publications, Park Ridge, NJ.
Bergerud AT. 1974. Decline of caribou in North America following settlement. J Wildlife Management 38:757–770.
Bergerud AT. 1980. A review of the population dynamics of caribou and wild reindeer in North America. Proc Int Reindeer/Caribou Symp 2:556–581.
Bergerud AT. 1983. The natural population control of caribou. In FL Bunnell, DS Eastman, and JM Peek, Eds. Symposium on natural regulation of wildlife populations. Proc No. 14. Forest Management and Range Exp Stn, Univ Idaho, Moscow.
Bergerud AT. 1996. Evolving perspectives on caribou population dynamics, have we got it right yet? Rangifer Special Issue 9.
Bjorge RR and JR Gunson. 1983. Wolf predation of cattle in the Simonette River Pastures in northwestern Alberta. In LN Carbyn, Ed. Wolves in Canada and Alaska. Canadian Wildlife Service Report Series 45. 135 Pp.
Bjorge RR and JR Gunson. 1989. Wolf, Canis lupud, population characteristics and prey relationships near Simonette River, Alberta. Canadian Field-Naturalist 103:327–334
Blanchard BM and RR Knight. 1991. Movements of Yellowstone grizzly bear. Biol Cons 58:41–67.
Boertje RD. 1984. Seasonal diets of the Denali caribou herd, Alaska. Arctic 37:161–165. Boertje RD. 1985. Seasonal activity of the Denali Caribou herd, Alaska. Rangifer 5:32–42.
Boertje RD, WC Gasaway, DV Grangaard, and DG Kellyhouse. 1988. Predation on moose and caribou by radio-collared grizzly bears in east central Alaska. Canadian J Zoology 66:2492–2492.
Boertje RD and RO Stephenson. 1992. Effects of ungulate availability on wolf reproductive potential in Alaska. Canadian J Zoology 70:2441–2443.
Boertje RD, DG Kellyhouse, and RD Hayes. 1995. Methods for reducing natural predation on moose: an evaluation. In LN Carbyn, SH Fritts, and DR Seip, Eds. Ecology and conservation of wolves in a changing world. Canadian Circumpolar Inst, Univ. Alberta, Edomonton.
Boertje RD and CL Gardner. 1996. Factors limiting the Fortymile caribou herd. Alaska Dep Fish and Game. Fed Aid in Wildlife Restor. Prog Rep. Proj W-24-4. Study 3.38. Juneau. 79 Pp.
Brand CJ, MJ Pybus, WB Ballard, and RO Peterson. 1995. Infectious and parasitic diseases of the gray wolf and their potential effects on wolf populations in North America. In LN Carbyn, SH Fritts, and DR Seip, Eds. Ecology and conservation of wolves in a changing world. Canadian Circumpolar Inst. Univ Alberta, Edmonton.
Brown DE. 1985. The grizzly in the southwest; documentary of an extinction. Univ of Okla Press, Norman. 274 Pp.
Bryant JP and PJ Kuropat. 1980. Selection of winter forage by subarctic browsing vertebrates: the role of plant chemistry. Annual Review of Ecology and Systematics 11:261–286.
Burkholder BL. 1959. Movements and behavior of a wolf pack in Alaska. J Wildlife Management 23:1–11.
Carney DW. 1985. 1985. Population dynamics and denning ecology of black bears in Shenandoah National Park, Virginia. M.S. thesis, VA Polytech Inst and State Univ, Blacksburg. 84 Pp.
Caughley G. 1981. Overpopulation. In Problems in management of locally abundant wild mammals. R Jewell and S Holt, Eds. Academic Press, NY
Charnov EL. 1982. The theory of sex allocation. Monographs in population biology. Vol. 18. Princeton, NJ, Princeton University Press,
Clark. 1991. Ecology of two black bear (Ursus americanus) populations in the interior highlands of Arkansas. Ph.D. thesis, Univ Ark, Fayetteville. 228 Pp.
Coley PD, JP Bryant, and FS Chapin III. 1985. Resource availability and plant anti-herbivore defense. Science 230:895–899.
Crête M and H Jolicoeur. 1987. Impact of wolf and black bear removal on cow: calf ratio and moose density in southwestern Québec. Alces 23:61–87.
Crête M and others. 1990. Food selection during early location by caribou calving in the tundra in Quebec. Arctic 43:60–65.
Dale BW, LG Adams, and RT Bowyer. 1995. Winter wolf predation in a multiple ungulate prey system, Gates of the Arctic National Park, Alaska. In LN Carbyn, SH Fritts, and DR Seip, Eds. Ecology and conservation of wolves in a changing world. Canadian Circumpolar Inst. Univ Alberta, Edmonton.
Dauphine TC, Jr. 1976. Biology of the Kaminuriak population of barren-ground caribou. Part 4. Growth, reproduction and energy reserves. Canadian Wildl Serv Rep. Ser No. 38. 69 Pp.
Davis JL and P Valkenburg. 1991. A review of caribou population dynamics in Alaska emphasizing limiting factors, theory, and management implications. Pp. 184–209 in Proceedings of the Fourth North American Caribou Workshop. C Butler and SP Mahoney, Eds. Newfoundland and Labrador Wildlife Division, St. John's.
Dhondt AA. 1988. Carrying capacity: a confusing concept. Acta Oecologia 9:337–346.
Eberhardt LL, BM Blanchard, and RR Knight. 1994. Population trend of the Yellowstone grizzly bear as estimated from reproductive and survival rates. Canadian J Zoology. 72:360–363.
Edenius L. 1993. Browsing by moose on scots pine in relation to plant resource availability. Ecology 74:2261–2269.
Edenius L, K Danell, and R Bergstrom. 1993. Impact of herbivory and competition on compensatory growth in woody plants winter browsing by moose on Scots Pine. Oikos 66:286–292
Franzmann AW, CC Schwartz, and RO Peterson. 1980. Moose calf mortality in summer on the Kenai Peninsula. J Wildlife Management 44:764–768.
Franzmann AW. 1981. Alces alces. Mammalian Species 154:1–7.
Franzmann AW and CC Schwartz. 1986. Black bear predation on moose calves in highly productive versus marginal moose habitats on the Kenai Peninsula, Alaska. Alces 22:139–153.
French SP and MG French. 1990. Predatory behavior of grizzly bears feeding on elk calves in Yellowstone National Park, 1986–88. Int Conf Bear Res and Management 8:335–341.
Fritts SH and LD Mech. 1981. Dynamics, movements, and feeding ecology of a newly-protected wolf population in northwestern Minnesota. Wildlife Monographs 80. 79 Pp.
Fritts SH. 1983. Record dispersal by a wolf from Minnesota. J Mammalogy 64:166–167.
Fritts SH, WJ Paul, LD Mech, and DP Scott. 1992. Trends and management of wolf-livestock conflicts in Minnesota. US Department of Interior, US Fish and Wildlife Service Resource Publication 181. 27 Pp.
Fritts SH and LN Carbyn. 1995. Population viability, nature reserves, and the outlook for gray wolf conservation in North America. Restoration Ecology 3:26–38.
Fuller TK and WJ Snow. 1988. Estimating wolf densities from radiotelemetry data. Wildlife Soc Bull 16:367–370.
Fuller TK. 1989a. Population dynamics of wolves in north-central Minnesota. Wildlife Monographs 105:1–41.
Fuller TK. 1989b. Denning behavior of wolves in north-central Minnesota. American Midland Naturalist 121:184–188.
Fuller TK. 1995. Guidelines for gray wolf management in the Northern Great Lakes Region. International Wolf Center Technical Pub 271, Ely, MN. 19 Pp.
Gaare E and T Skogland. 1980. Lichen-reindeer interaction studies in a simple case model. In E Reimers, E Gaare, and S. Skjenneberg, Eds. Proceedings of the Second International Reindeer/Caribou Symposium, Direktoratet for vilt og ferskvannfisk, Trodheim, Norway.
Garshelis DL. 1994. Density dependent population regulation of black bears. In M Taylor, Ed. Density dependent population regulation in black, grizzly and polar bears. Int Conf Bear Res and Management. Monogr Series No. 3. 43 Pp.
Gasaway WC, RO Stephenson, JL Davis, PEK Shepherd, and OE Burris. 1983. Interrelationships of wolves, prey and man in interior Alaska. Wildlife Monographs 84:1–50.
Gasaway WC, RD Boertje, DV Grangaard, DG Kellyhouse, RO Stephenson, and DG Larsen. 1992. The role of predation in limiting moose at low densities in Alaska and Yukon and implications for conservation. Wildlife Monographs 120:1–59.
Gese EM, and LD Mech. 1991. Dispersal of wolves (Canis lupus) in northeastern Minnesota, 1969–1989. Canadian J Zoology 69:2946–2955.
Greenberg JH. 1987. Language in the Americas. Stanford University Press, Stanford, CA.
Gunther KA and RA Renkin. 1990. Grizzly bear predation on elk calves and other fauna of Yellowstone National Park. Int Conf Bear Res and Management 8:329–334.
Haber GC. 1996. Biological impacts on wolves of exploitation and control. P. 36 in First Annual Conference of the Wildlife Society, Albuquerque, NM (abstract).
Haber GC. 1996. Biological, conservation, and ethical implications of exploiting and controlling wolves. Conservation Biology 10:1068–1081.
Hamer D and S Herrero. 1991. Elk, Cervus elaphus, calves as food for grizzly bears, Ursus arctos, in Banff National Park, Alberta, Canada. Canadian Field-Naturalist 105:101–103.
Hamilton AN. 1988. Classification of coastal grizzly bear habitat for forestry interpretations and the role of food in habitat use by coastal grizzly bears. M.Sc. thesis, University of British Columbia.
Harrington FH, LD Mech, and SH Fritts. 1993. Pack size and wolf pup survival: their relationship under varying ecological conditions. Behavioral Ecology and Sociobiology 13:19–26.
Hart JS, O Heroux, WH Cottle, and CA Mills. 1961. The influence of climate on metabolic and thermal responses of infant caribou. Canadian J Zoology 39:845–856.
Hayes RD, A Baer, and DL Larsen. 1991. Population dynamics and prey relationships of an exploited and recovering wolf population in the southern Yukon. Yukon Fish and Wildlife Br Final Rep TR-91-1. Whitehorse. 67 Pp.
Hayes RD. 1995. Numerical and functional response of wolves, and regulation of moose in the Yukon. M.Sc. thesis, Simon Fraser University, Burnaby, British Columbia.
Heard DC and GW Calef. 1986. Population dynamics of the Kaminuriak northwest territories Canada caribou herd 1968–1985. Rangifer. Special Issue 1:159–166.
Heard DC, TM Williams, and DA Melton. 1996. The relationship between food intake and predation risk in migratory caribou and implications to caribou and wolf population dynamics. Rangifer. Special Issue 9:37–44.
Helle T, J Aspi, K Lempa, and E Taskinen. 1992. Strategies to avoid biting flies by reindeer: field experiments with silhouette traps. Annales Zoologici Fennici 29:69–74.
Hicks MV. 1994. Federal aid in wildlife restoration survey-inventory management report. Alaska Department of Fish and Game, Div Wildlife Cons, Juneau, AK.
Holleman DF and JR Luick. 1977. Lichen species preference by reindeer. Canadian J Zoology 55:14–26.
Hovey FW and BN McLellan. 1996. Estimating population growth of grizzly bears from the Flathead River drainage using computer simulations of reproduction and survival rates. Can J Zool 74:1409–1416.
Huntington S and Rearden 1993. Shadows on the Koyukuk. Alaska Northwest Books. Anchorage, AK
Jeffries R and others. 1994. Vertebrate herbivores and northern plant communities: reciprocal influences and responses. Oikos 71:193–206.
Jonkel CJ and IM Cowan. 1971. The black bear in the spruce-fir forest. Wildlife Monographs 27:1–57.
Keith LB. 1974. Some features of population dynamics in mammals. Trans Int Congr Game Bio 11:17–58.
Keith LB. 1983. Population dynamics of wolves. Pp. 66–77 in LN Carbyn, Ed. Wolves in Canada and Alaska, Canadian Wildlife Service Report Series 45. 135 Pp.
Kingsley MCS, JA Nagy, and HV Reynolds. 1988. Growth in length and weight of northern brown bears—Differences between sexes and populations. Canadian J Zoology 66:981–986.
Klein DR. 1968. The introduction, increase and crash of reindeer on St. Matthew Island. J Wildlife Management 32:350–367.
Klein DR. 1970. Interactions of Rangifer-Tarandus reindeer and caribou with its habitat in Alaska. Riistatteteellisia Julkaisuja. 289–293.
Klein DR. 1982. Fire, lichens, and caribou. J Range Management 35:390–395.
Klein DR. 1992. Comparative ecological and behavioral adaptations of Ovibos-Moschatus and Rangifer Tarandus. Rangifer 12:13–25.
Klein DR, DF Murray, RH Armstrong, and BA Anderson. 1997. Alaska. In MJ Mac, PA Opler, and PD Doran, Eds. National status and trends report. US National Biological Service, Washington, DC.
Kolenosky GB. 1972. Wolf predation on wintering deer in east-central Ontario. J Wildlife Management 36:357–369.
Krebs CJ. 1994. Ecology, the experimental analysis of distribution and abundance, 4th ed. Harper Collins College Publishers, NY, NY. 651 Pp.
Larsen DG, DA Gauthier, and RL Markel. 1989. Causes and rate of moose mortality in the southwest Yukon. J Wildlife Management 53:548–557.
Leader-Williams N, TA Scott, and RM Pratt. 1981. Forage selection by introduced reindeer on South Georgia, South Atlantic Ocean and its consequences for the flora. J Applied Ecology 18:83–106.
LeCount AL. 1982. Characteristics of a central Arizona black bear population. J Wildlife Management 46:861–868.
Lehman NE, P Clarkson, LD Mech, TJ Meier, and RK Wayne. 1992. A study of the genetic relationships within and among wolf packs using DNA fingerprinting and mitochondrial DNA. Behavioral Ecology and Sociobiology 30:83–94.
Lent PC. 1980. Synoptic snowmelt patterns in Arctic Alaska in relation to caribou habitat use. Pp. 71–77 in E Reimers, E Gaare, and S. Skjenneberg, Eds. Proceedings of the Second International Reindeer/Caribou Symposium, Direktoratet for vilt og ferskvannfisk, Trondheim, Norway.
Licht DS and SH Fritts. 1994. Gray wolf (Canis lupus) occurrences in the Dakotas. American Midland Naturalist 132:74–81.
Lindzey FG, KR Barber, RD Peters, and EC Meslow. 1986. Responses of a black bear population to a changing environment. Int Conf Bear Res and Management 6:57–63.
Lindzey FG and EC Meslow. 1977. Population characteristics of black bears on an island in Washington. J Wildlife Management 41:408–412.
Maessen O, B Freedman, J Svovoda, and MLN Nam. 1983. Resource allocation in high arctic vascular plants of differing growth form. Canadian J Botany 61:1680–1691.
Martinka CJ. 1974. Population characteristics of grizzly bears in Glacier National Park, Montana. J. Mammal. 55:21–29.
McEwan EH. 1968. Growth and development of the barren-ground caribou. II. Postnatal growth rates. Can J Zool 46:1023–1029.
McLellan B. 1989. Dynamics of a grizzly bear population during a period of industrial resource extraction. III. Natality and rate of increase. Can J Zool 67:1865–1868.
McLellan B. 1994. Density-dependent population regulation of brown bears. Pp. 15–24 In M Taylor, Ed. Density-dependent population regulation on black, brown and polar bears. Int Conf Bears Res and Management. Monogr Series No. 3. 43 Pp.
McLellan BN and FW Hovey. 1994. The diet of grizzly bears in the Flathead river drainage of southeastern British Columbia. Can J Zool 73:704–712.
McLellan BN. In press. Maintaining viability of brown bears along the southern fringe of their distribution. Int Conf Bear Res and Management.
Mech LD. 1977. Wolf pack buffer zones as pre reservoirs. Science 198:320–321.
Mech LD. 1986. Wolf numbers and population trend in the central Superior National Forest, 1967–1985. US Dept Agric for Serv Res Pap No. NC-270. 6 Pp.
Mech LD and ME Nelson. 1989. Polygyny in a wild wolf pack. J Mammalogy 70:675–676.
Mech LD. 1994. Biological impacts on wolves of exploitation and control: a response. In First Annual Conference of the Wildlife Society, Albuquerque, NM (abstract).
Mech LD, SH Fritts, and D Wagner. 1994. Minnesota wolf dispersal to Wisconsin and Michigan. US National Biological Survey, Minneapolis, MN. Typewritten manuscript 6 Pp.
Mech LD, TJ Meier, JW Burch, and LG Adams. 1995. Patterns of prey selection by wolves in Denali National Park, Alaska. In LN Carbyn, SH Fritts, and DR Seip, Eds. Ecology and conservation of wolves in a changing world. Canadian Circumpolar Institute, Occasional Papers 35, Edmonton, Alberta.
Meier T, J Burch, LD Mech, and L Adams. 1995. Pack structure and genetic relatedness among wolf packs in a naturally regulated population. Pp. 293–302 In LN Carbyn, SH Fritts, and DR Seip, Eds. Ecology and conservation of wolves in a changing world. Canadian Circumpolar Institute, Occasional Papers 35, Edmonton, Alberta.
Messier F and M Crête. 1985. Moose-wolf dynamics and the natural regulation of moose populations. Oecologia 65:503–512.
Messier F. 1985. Social organization, spatial distribution, and population density of wolves in relation to moose density. Canadian J Zoology 63:1068–1077.
Miller DR. 1980. Wildfire effects of barren-ground caribou wintering on the taiga of northcentral Canada: a reassessment. Pp. 84–98 in Reimers, E, Gaare E and S. Skjenneberg, Eds. Proceedings of the Second International Reindeer/Caribou Symposium, Direktoratet for vilt og ferskvannfisk, Trondheim, Norway.
Miller F. 1990. Peary caribou status report. Canadian Wildlife Service. West and North Reg Rep. 64 Pp.
Miller SD, EF Becker, and WB Ballard. 1987. Black and brown bear density estimates using modified capture-recapture techniques in Alaska. Int Conf Bear Research and Management 7:23–35.
Miller SD and WB Ballard. 1992. Analysis of an effort to increase moose calf survivorship by increasing hunting of brown bears in southcentral Alaska. Wildlife Society Bulletin 20:445–454.
Miller SD. 1995. Impacts of heavy hunting pressure on the density and demographics of brown bear populations in southcentral Alaska. Alaska Dep Fish and Game. Fed Aid in Wildlife Res Grant W-24-3, Study 4.26. Fairbanks. AK. 28 Pp.
Miller SD, GC White, RA Sellers, HV Reynolds, JW Schoen, K Titus, VG Barnes Jr., RB Smith, RR Nelson, WB Ballard, and CC Schwartz. 1997. Brown and black bear density estimation in Alaska using radio telemetry and replicated mark-resight techniques. Wildlife Monographs 113:1–55.
Miquelle DG, JM Peek, and V Van Ballenberghe. 1992. Sexual segration in Alaskan moose. Wildlife Monographs 122:1–43.
Molvar EM, RT Bowyer, and V Van Ballenberghe. 1993. Moose herbivory, browse quality, and nutrient cycling in an Alaskan treeline community. Oecologia 94:472–479.
Murie AO. 1994. The wolves of Mount McKinley. Fauna of the national parks of the US. Fauna Ser No. 5. US Gov Print Off, Washington, DC. 238 Pp.
Nagy JA, RH Russel, AM Pearson, MC Kingsley, and CB Larsen. 1983a. A study of grizzly bears on the barren-grounds of Tuktoyaktuk Peninsula and Richards Island, Northwest Territories, 1974–1978. Can Wildlife Ser Rep. 136 Pp.
Nagy JA, RH Russel, AM Pearson, MC Kingsley, and BC Goski. 1983b. Ecological studies of the grizzly bear in the Arctic mountains, northern Yukon territory, 1972 to 1975. Can Wildlife Ser Rep. 104 Pp.
Nelson ME and LD Mech. 1986. Relationship between snow depth and gray wolf predation on white-tailed deer. J Wildlife Management 50:90–91.
Oldemeyer JL and WL Regelin. 1987. Forest succession, habitat management, and moose in the Kenai National Wildlife Refuge. Viltrevy (Swedish Wildlife Journal) Supplement 1:163–180.
Osborne TO and others. 1996. Extent, cause, and timing of moose calf mortality in western interior Alaska. Alces 27:24–30.
Oosenbrug S and LN Carbyn. 1982. Winter predation on bison and activity pattern of a pack of wolves in Wood Buffalo National Park. Pp 43–53 in FM Harrington and PC Paquet, Eds. Wolves: a worldwide perspective of their behavior, ecology, and conservation. Noyes Publications, Park Ridge, NJ.
Packard JM and LD Mech. 1980. Population regulation in wolves. Pp. 135–150 in MN Cohens, RS Malpass, and HG Klein, Eds. Biosocial mechanisms of population regulation. New Haven: Yale Univ Press.
Packard JM, LD Mech, and US Seal. 1983. Social influences on reproduction in wolves. Pp. 78–85 in LN Carbyn, Ed. Wolves in Canada and Alaska: their status, biology, and management Canadian Widlife Serv. Rep Ser No. 45.
Page RE. 1985. Early calf mortality in northwestern British Columbia. M.Sc. thesis, Univ of Victoria, Victoria, BC 129 Pp.
Parker KL and CT Robbins. 1984. Thermoregulation in mule deer (Odocoileus hemionus hemionus) and elk (Cervus elaphus nelsoni). Canadian J Zoology 62:1409–1422.
Pastor J. 1988. Moose, microbes, and the boreal forest. Bioscience 38:770–777.
Pearson AM. 1975. The northern interior grizzly bear. Can. Wildlife. Serv. Rep. Ser. No. 34. 86 Pp.
Peek JM. 1974. Initial response of moose to a forest fire in northeastern Minnesota. Am Midland Naturalist 91:435–438.
Pegau RE. 1970. Succession in two exclosures near Unalakleet, Alaska. Canadian Field-Naturalist 84:12–25
Pegau RE. 1975. Analysis of the Nelchina caribou range. Biol Pap Univ Alaska special report 1.
Peterson RO and RE Page. 1983. Wolf-moose fluctuation in Isle Royale National Park, Michigan USA. Annales Zoologici Fennica 74:251–253.
Peterson RO, JD Woolington, and TN Bailey. 1984. Wolves of the Kenai Peninusla. Alaska Wildlife Monographs 88. 52 Pp.
Peterson RO and RE Page. 1988. The rise and fall of Isle Royale wolves, 1975–1986. J. Mammalogy 69:89–99.
Peterson RO. 1977. Wolf ecology and prey relationships on Isle Royale. National Park Service Scientific Monograph 11. 210Pp.
Pianka ER. 1978. Evolutionary Ecology. 2nd Ed. Harper & Row, New York.
Pimlott DH, JA Shannon, and GB Kolenosky. 1969. The ecology of the timber wolf in Algonquin Provincial Park. Ontario Dept Lands and For Res Rep (Wildl) No. 87. 92 Pp.
Pritchard GT and CT Robbins. 1990. Digestive efficiencies of grizzly and black bears. Can J Zool 68:1645–1651.
Pruitt WO. 1981. Application of the varrio snow index to over wintering North American barren-ground caribou (Rangifer tarandus arcticus). Canadian Field-Naturalist 95:363–365.
Pulliam HR and NM Haddad. 1994. Human population growth and the carrying capacity concept. Bull. of the Ecological Society of America 75:141–157.
Rausch RA. 1969. A summary of wolf studies in southcentral Alaska, 1957–1968. Trans North Am Wildlife and Nat Resour Conf 34:117–131.
Regelin WL, CC Schwartz, and AW Franzmann. 1987. Effects of forest succession on the nutritional dynamics of moose forage. Viltrevy (Swedish Wildlife Journal). Supplement. 1:247–264.
Reimers E. 1980. Activity pattern; the major determinant for growth fattening in Rangifer. Pp. 466–474 in E Reimers, E Gaare, and S Skjenneberg, Eds. Proceedings of the Second International Reindeer/Caribou Symposium, Direktoratet for vilt og ferskvannfisk, Trondheim, Norway.
Reiner DC. 1996. Estimating bear habitat quality in southeastern British Columbia by measuring changes in bear body condition. M.S. thesis, Univ Montana, Missoula, MT. 81 Pp.
Renecker LA and RJ Hudson. 1986. Seasonal energy expenditures and thermoregulatory responses of moose (Alces alces). Canadian J Zoology 64:322–327
Reynolds HV and TA Boudreau. 1990. Effects of harvest rates on grizzly bear population dynamics in the northcentral range. Alaska Dep Fish and Game. Div Wildlife Cons. Fed Aid in Wildlife Restoration Research progress report Project W-23-3, study 4.19.
Reynolds HV. 1994. Effects of harvest on grizzly bear population dynamics in the northcentral Alaska Range . Alaska Dep Fish and Game. Fed Aid in Wildlife Res Grant W-24-3, Study 4.26. Fairbanks, AK. 28 Pp.
Robbins CT, S Mole, AE Hagerman, and TA Hanley. 1987. Role of tannins in defending plants against ruminants reduction in dry matter digestion. Ecology 68:1606–1615.
Rogers LL. 1987. Effects of food supply and kinship on social behavior, movements, and population growth of black bears in Northeastern Minnesota. Wildlife Monographs 97:1–72.
Rosenthal GA and DH Janzen, Eds. 1979. Herbivores: their interaction with secondary plant metabolites. Academic Press, New York, NY.
Rothman RJ and LD Mech. 1979. Scent-marking in lone wolves and newly formed pairs. Animal Behavior. 27:750–760.
Schlegel M. 1983. Factors affecting calf survival in the Lochsa elk herd. Job Completion Report, Federal aid project W-1600-R, Study 1, Job 3. Idaho Dept of Fish and Game, Boise, ID.
Schoen JW and LR Beier. 1990. Brown bear habitat preferences and brown bear logging and mining relationships in southeast Alaska. Alaska Department of Fish and Game, Fed Aid in Wildlife Restor Final Report. Project W22:1–6 and W 23:1–3. Juneau, AK 90 Pp.
Schwartz CC, WL Regelin, and AW Franzmann. 1988. Estimates of digestibility of birch, willow and aspen in moose. J Wildlife Management 52:33–37.
Schwartz CC and AW Franzmann. 1991. Interrelationship of black bears to moose and forest succession in the northern coniferous forest. Wildlife Monographs 113:1–58.
Scotter GW. 1967. Effects of fire on barren-ground caribou and their forest habitat in northern Canada. Trans North Amer Wildlife Nat Res Conf 32:246–259.
Scotter GW. 1995. Influence of harassment by wolves, Canis lupus, on barren-ground caribou Rangifer tarandus groenlandicus, movements near the Burnside River, Northwest Territories. Canadian Field-Naturalist 109:452–453.
Seal US, LD Mech, and V VanBallenberghe. 1975. Blood analyses of wolf pups and their ecological and metabolic interpretation. J Mammalogy 56:64–75.
Seber GAF. 1982. The estimation of animal abundance and related parameters, 2nd ed. Griffin, London. 654 Pp.
Seip DR. 1992. Factors limiting woodland caribou populations and their interrelationships with wolves and moose in southeastern British Columbia. Canadian J Zoology 70:1494–1503.
Seip DR and K Brown. 1996. Introduction to the population ecology of North American caribou. Rangifer 9:11–12.
Selkregg LL. 1976. 1974–1976 Alaska Regional Profiles: Vol. I Southcentral Alaska, 225 Pp.; Vol. II Arctic, 218 Pp.; Vol. III Southwest, 313 Pp.; Vol. VI Southeast, 233 Pp.; Vol. V Northwest, 265 Pp.; Vol. VI Yukon, 346 Pp. Arctic Information and Data Center, Anchorage, AK.
Sellers RA. 1994. Dynamics of a hunted brown bear population at Black Lake, Alaska. 1993 Annual Progress Report. Alaska Department of Fish and Game. 61 Pp.
Sellers RA, SD Miller, TS Smith, and R Potts. 1993. Population dynamics and habitat partitioning of a naturally regulated brown bear population on the coast of Katmai National Park: 1993 Annual progress report. Alaska Dept of Fish and Game, Div of Wildlife Conservation, Juneau.
Servheen C 1990. The status and conservation of the bears of the world. Eighth Int Conf Bear Res and Management 9:4–16.
Servheen C, FW Hovey, BN McLellan, R Mace, W Wakkinen, W Kasworm, D Carney, T Manley, K Kendall, and R Wielgus. 1994. Report of the northern ecosystems researches on grizzly bear population trends in the north and south forks of the Flathead, and the Blackfeet Indian Reservation of the NCDE; the Cabinet-Yank ecosystem; and the Selkirk ecosystem; and future data needs to improve trend estimates. US Fish and Wildlife Service. Missoula, MT.
Sharkey MJ. 1970. The carrying capacity of natural and improved land in different climatic zones. Mammalia 34:564–572.
Skogland T. 1985. The effect of density-dependent resource limitations on the demography of wild reindeer. J Animal Ecology 54:359–374.
Skoog R. 1968. Ecology of the caribou (Rangifer tarandus granti) in Alaska. Ph.D. thesis, Univ CA, Berkeley.
Smith TR and MR Pelton. 1990. Home ranges and movements of black bears in a bottomland hardwood forest in Arkansas. Pp. 213–218 in LM Darling and WR Archibald, Eds. Bears: their biology and management. International Association for Bear Research and Management, Coeur d'Alene.
Spalding DJ. 1990. The early history of moose (Alces alces) distribution and relative abundance in British Columbia, Victoria BC. Royal British Columbia Museum.
Stenlund MH. 1955. A field study of the timber wolf, Canis lupus, on the superior National Forest. MN. MN Dept Conservation Tech Bull 4:1–55.
Stephenson RO. 1975. Wolf Report. Alaska Sept of Fish and Game, Juneau.
Stephenson RO, WB Ballard, CA Smith, and K Richardson. 1995. Wolf biology and management in Alaska 1981–92. Pp. 43–54 in LN Carbyn, SH Fritts, and DR Seip, Eds. Ecology and conservation of wolves in a changing world. Canadian Circumpolar Institute, Occasional Papers 35, Edmonton, Alberta.
Storer TI and LP Tevis. 1955. California grizzly. University of California Press, Berkeley.
Stringham SF. 1980. Possible impacts of hunting on the grizzly/brown bear, a threatened species. Int Conf Bear Res and Management 4:337–349.
Stringham SF. 1990. Grizzly bear reproductive rate relative to body size. Int Conf Bear Res and Management 8:433–443.
Telfer ES and JP Kelsall. 1984. Adaptation of some large North American mammals for survival in snow. Ecology 65:1828–1834.
Titus K and LR Beier. 1994. Population and habitat ecology of brown bears on Admiralty and Chichagof Islands, Alaska Dept of Fish and Game, Div of Wildlife Cons, Grant W-24-2 study 4.22, Juneau, AK.
US Department of Agriculture. 1994. Wolf depredation on livestock in Minnesota annual update of statistics. USDA, Animal and Plant Health Inspection Service, Animal Damage Control, Grand Rapids, MN.
Valkenburg P and JL Davis. 1986. Calving distribution of Alaska's Steese-Fortymile herd: a case of infidelity? Rangifer (special issue) 1:315–325.
Valkenburg P, JL Davis, JM Ver Hoef, RD Boertje, ME McNay, RM Egan, DJ Reed, CL Gardner and RW Tobey. 1996. Population decline in the Delta caribou herd with reference to other Alaskan herds. Rangifer (special issue) 9:53–62.
Van Ballenberghe V and LD Mech. 1975. Weights, growth, and survival of timber wolf pups in Minnesota. J Mammalogy 56:44–63.
Van Ballenberghe V, AW Erickson, and D Byman. 1975. Ecology of the timber wolf in northeastern Minnesota. Wildlife Monographs 43.
Van Ballenberghe V and DG Miquelle. 1993. Mating in moose: timing, behavior, and male access patterns. Canadian J. Zoology 71:1687–1690.
Van Ballenberghe V. 1983. Extraterritorial movement and dispersal of wolves in southcentral Alaska . J Mammalogy 64:168–171.
Viereck LA. 1973. Wildfire in the taiga of Alaska. Quat Res (NY) 3:465 –495.
Wayne RK, DA Gilbert, N Lehman, K Hansen, A Eisenhower, D Girman, LD Mech, PJP Gogan, US Seal, and RJ Krumenaker. 1991. Conservation genetics of the endangered Isle Royale gray wolf. Conservation Biology 5:41–51.
Wayne RK, N Lehman, and TK Fuller. 1995. Conservation genetics of the gray wolf. Pp. 399–408 in LN Carbyn, SH Fritts, and DR Seip, Eds. Ecology and conservation of wolves in a changing world. Canadian Circumpolar Institute, Occasional Papers 35, Edmonton, Alberta.
White GC. 1983. Evaluation of radio tagging, marking, and sighting estimators of population size using Monte Carlo simulations. Pp. 91–103 in JD Lebreton and PM North, Eds. Marked individuals in the study of bird populations. Biskhauser Verlog Basel, Switzerland.
White RG, BR Thompson, T Skogland, SJ Person, DE Russel, D Holleman, and JR Luick. 1975. Ecology of caribou at Prudhoe Bay, Alaska. Biol Pap Univ Alaska, Special Rep 2. Pp 151–201.
White RG and J Trudell. 1980. Habitat preference and forage consumption by reindeer (Rangifer tarandus tarandus) and caribou (Rangifer tarandus granti) near Atkasook, Alaska. Arctic Alpine Research 12:511–530.
White RG and FA Milan. 1981. Reindeer herds of the Kuskowin, Alaska. Proc Alaska Sci Conf 32:1–10.
Wielgus RB. 1993. Causes and consequences of sexual habitat segregation in grizzly bears. Ph.D. thesis, University of British Columbia.
Wielgus RB and FL Bunnell. 1994. Sexual segregation and female bear avoidance of males. J Wildlife Management 58:405–413.
Wolfe RJ. 1996. Subsistence food harvests in rural Alaska, and food safety issues. Paper presented to the Institute of Medicine. National Academy of Sciences Committee on Environmental Justice, Spokane, WA, August 13.
Wydeven AP, RN Schultz, and RP Thiel. 1995. Monitoring of a recovering gray population in Wisconsin 1979–1991. Pp. 147–156 in LN Carbyn, SH Fritts, and DR Seip, Eds. Ecology and conservation of wolves in a changing world. Canadian Circumpolar Institute, Edmonton. Alberta.
Wydeven AP. 1993. Wolves in Wisconsin: reconolization under way. International Wolf 3:18–19.
Zackrisson O. 1977. Influence of forest fires on the North Swedish boreal forest. Oikos 29:22–32.