Advancing Research on Chronic Conditions in Women (2024)
Chapter: 6 Chronic Conditions That Predominantly Impact or Affect Women Differently
6
Chronic Conditions That Predominantly Impact or Affect Women Differently
This chapter describes the research progress on and advances in understanding chronic conditions present in both women and men that predominantly impact or affect women differently. As mentioned in Chapter 1, the committee selected certain chronic conditions to highlight to provide an overview of the evidence base and research gaps around them. This chapter covers 16 chronic conditions involving mental health, human immunodeficiency virus/acquired immunodeficiency syndrome (HIV/AIDS), cardiometabolic disorders, pain disorder, autoimmune disorders, neurocognitive disorders, and musculoskeletal conditions. Chapter 4 provided data on the impact of select chronic conditions. This chapter highlights the specific role of biological factors, with specific emphasis on estrogens, sex chromosome effects, and influences of the maternal in utero environment, in the development of the disease or condition; emerging differences in how these disorders and conditions present themselves; diagnosis, treatment, and management in women; disparities by racial and ethnic groups and sexual orientation in terms of effects on susceptibility and outcomes; and research gaps.
DEPRESSION
Depression is more common in women, affecting them across the life course and causing significant morbidity and mortality (Bromet et al., 2011; Kessler et al., 1993). This report primarily discusses major depressive disorder (MDD), with some mention of postpartum depression (PPD) and hormonal-associated depression, which includes premenstrual dysphoric
disorder (PMDD)/premenstrual syndrome (PMS) and perimenopausal depression. Differences in the prevalence and effects of depression are likely related to biological, environmental, and societal factors (Yuan et al., 2023). It is also associated with developing other chronic conditions in women, including Alzheimer’s disease (AD); adverse cardiovascular events, including myocardial infarction, stroke, and death; and post-stroke depression, including greater severity, poorer neurological outcomes, increased risk of stroke recurrence, and mortality (O’Neal, 2023; Sibolt et al., 2013; Volz et al., 2021; Wang et al., 2017; Wu et al., 2019). The co-occurrence of three major chronic diseases—MDD, CVD, and AD—has a significant effect on women (Goldstein et al., 2021).
Biological Factors
Research has yet to fully elucidate the biological mechanisms underlying the susceptibility of women to depression. Several lines of evidence indicate roles for the gamma-aminobutyric acid (GABA), serotonin, and dopamine neurotransmitter systems and the stress-responsive hypothalamus-pituitary-adrenal axis in depression. Studies also show that female gonadal hormones, such as estrogens, and sex chromosome effects modulate the function of these neural systems, which may partly explain why women are more likely to experience depression.
Sex Chromosome Effects
In contrast to the plethora of evidence supporting a role for gonadal hormones in modulating mechanisms that lead to depression, limited evidence shows that sex chromosomal genes play a role in women. One study demonstrated that genetic polymorphisms1 on the X chromosome influence the expression levels of three proteins expressed in GABAergic neurons—somatostatin, a hormone that affects neurotransmission, and two enzymes involved in the synthesis of the neurotransmitter glutamate (Seney et al., 2013a). This study also found that women with depression have significantly lower levels of somatostatin in the brain compared to men with depression. With studies showing less GABA-mediated inhibition of neural activity in depression (Luscher et al., 2011; Sanacora et al., 1999; Sequeira et al., 2009), sex chromosome genes can influence GABAergic neural activity by regulating protein expression in GABAergic neurons. Experiments using the Four Core Genotypes (FCG) mouse model, which decouples sex chromosome and gonadal hormone effects (see Chapter 2),
___________________
1Polymorphisms are two or more variations of the same gene or protein that occur among different individuals or populations.
also support this premise, demonstrating that sex chromosome effects regulate expression of genes related to GABA and the serotonergic and dopaminergic neurotransmitter systems that also modulate depression (Seney et al., 2013b).
The Effects of Estrogens
The menstrual cycle plays an etiological role in depression (Albert, 2015). Fluctuating sex hormone levels are linked to changes in brain morphology, function, and neurochemistry (Kundakovic and Rocks, 2022), with dynamic alterations in the epigenome triggering changes in gene expression that mediate these effects (Kundakovic and Rocks, 2022). Research in mice suggests that hormones and genes interact to change the brain structure in a way that results in an increased risk of depression in women (Kundakovic and Rocks, 2022). Evidence also links depression to a loss of synchronization between estrogens and cortisol phases of the reproductive cycle (Butler, 2018). Some studies demonstrate a genetic association between different estrogen receptor (ER) polymorphisms and depression (Li et al., 2022a).
Animal studies show that the ovarian hormone 17β-estradiol regulates neural activity in key subcortical regions in the stress response circuitry linked to MDD (Jacobs et al., 2015). Furthermore, inflammation has been linked to MDD (Haapakoski et al., 2015), and estrogen deficiency is linked to increases in several inflammatory markers (Klein and Flanagan, 2016). These increases may serve as a viable biomarker for identifying the risk of MDD in women. Perimenopausal depression includes complex relationships between hormonal fluctuations and neurotransmitter activity, brain-derived neurotrophic factors, chronic inflammation, and oxidative stress that warrant further study (Liang et al., 2024).
Other Biological Factors and Mechanisms
Cortisol is an anti-inflammatory hormone whose dysregulation can lead to inflammation, which has been implicated in depression and depressive disorders associated with PMS and PMDD (Bannister, 2019). It is released during stress and modulates activity in distinct brain regions, including the amygdala, prefrontal cortex, and hippocampus. Genetic evidence demonstrates sex differences in cortisol receptor expression and suggests an association between MDD in women and a specific polymorphism in a cortisol receptor (Teo et al., 2023).
Another possible mechanism may involve intestinal dysfunction, such as in inflammatory bowel disease. Experiments have demonstrated that chronic stress in female mice can disrupt the intestinal barrier, which can trigger the release of inflammatory molecules that affect neural circuits in the brain
involved in depression (Doney et al., 2024). A recent scoping review noted sex-specific differences in the composition of the gut microbiome and associations with depression severity in patients (Niemela et al., 2024).
The serotonergic system, the primary target of many antidepressant medications, shows differential expression in women and men of neurotransmitter receptors and some transcription factors. This finding may be related to evidence suggesting that selective serotonin reuptake inhibitors (SSRIs) are more effective in treating depression in women (Seney et al., 2022).
A recent meta-analysis confirmed that PPD involves several genes and is at least partially heritable. This analysis also identified a plausible biological pathway involving GABAergic neurons (Guintivano et al., 2023).
The Maternal in Utero Environment
The maternal in utero environment is an important determinant for susceptibility to depression, especially in female offspring. Clinical studies have linked maternal anxiety (Van den Bergh et al., 2008), higher levels of inflammatory markers (Lipner et al., 2024), and infection during pregnancy (Pallier et al., 2022) to depressive symptoms in adolescent female offspring (Lipner et al., 2024; Van den Bergh et al., 2008). Preclinical rodent models have also demonstrated that exposure to maternal stress-induced inflammation led to an enhanced sensitivity to stresses affecting the hypothalamus-pituitary-adrenal axis in female offspring (García-Cáceres et al., 2010).
Low birthweight and preterm birth are also related to the maternal in utero environment and considered to reflect fetal well-being during pregnancy (Kim et al., 2015). Research examining these features and their relationship to depression later in life have yielded mixed results. One study found no association, but another study comparing outcomes by sex found that low-birthweight female babies had a greater risk of depression than male babies by the age of 21 years (Alati et al., 2007). A recent analysis of data from the Women’s Health Initiative (WHI) supports the idea that low birthweight or preterm birth is associated with depression in women later in life (Rahalkar et al., 2023).
Specific Risk Factors in Women
Reproductive Milestones
Reproductive milestones serve as windows of vulnerability for depression. Clinical and population-based studies have shown that increased prevalence of depression correlates with hormonal changes in women, particularly during puberty, prior to menstruation, after pregnancy,
at perimenopause, and in menopause, suggesting that changes in hormonal levels may explain the differences in increased prevalence of depression in women (Figure 6-1) (Albert, 2015; Kundakovic and Rocks, 2022). Although girls entering puberty do not necessarily have lower levels of estrogen, fluctuations in those levels may play a role in developing depression at this age (Kundakovic and Rocks, 2022). PMDD is associated with cyclical mood, behavioral, and physical indications that correspond to menstruation, specifically the luteal phase in women. PMDD more commonly affects reproductive aged women but can begin to affect women in teen years (Mishra et al., 2024b). Studies suggest that in PMDD, hormonal patterns are normal but that women may have heightened sensitivity to cyclical variations in estrogen and progesterone levels (Hantsoo and Epperson, 2015; Mishra et al., 2024b).
Recently, a population-based cohort study analyzing data from 264,557 women suggested that the use of hormonal contraceptives, particularly in the first 2 years, is associated with a higher risk of depression, and that use during adolescence may elevate depression risk at older ages (Johansson et al., 2023). This is in contrast with a more recent study
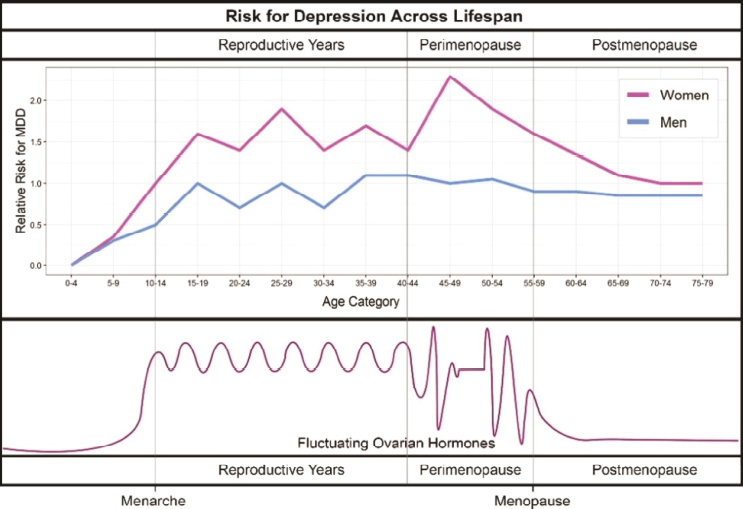
SOURCE: Kundakovic and Rocks, 2022.
in a smaller cohort of 6239 women, which indicated that current hormonal contraceptive use was associated with lower odds of depression (Gawronska et al., 2024). Another study indicated that prior depression associated with hormonal contraceptive use may be linked to a higher likelihood of developing PPD (Larsen et al., 2023). Such findings suggest that individual differences in hormonal sensitivity may influence susceptibility to depression across the life course in some women (Larsen et al., 2023).
The incidence of depression during the menopausal transition increases, even among women who have a history of depression (Maki et al., 2019). Longitudinal studies, such as the Study of Women’s Health Across the Nation (SWAN), Penn Ovarian Aging Study, and Seattle Midlife Women’s Study, have shown that perimenopause has an increased likelihood of depressive symptoms and disorders, with likelihood increasing in the late stage of perimenopause as levels of estrogen continue to drop (Maki et al., 2019). Studies have shown that removing the ovaries increases the likelihood of depression (Bräuner et al., 2022; Hassan et al., 2024; Hickey et al., 2021).
Vulnerabilities
Researchers have used the affective, biological, cognitive model of depression, based on a vulnerability-stress model from childhood to adolescence, to examine gender differences in the developing depression (Hyde and Mezulis, 2020). Evidence shows that affective factors, such as temperament differences between boys and girls, may explain gender differences in depression incidence. However, this correlation is based mainly on cross-sectional rather than longitudinal studies (Hyde and Mezulis, 2020). Cognitive vulnerabilities for depression specific in women include negative cognitive style and objectified body consciousness, an internalization of cultural measures about ideal body and appearance. This model also explains how trauma, sexual harassment and abuse, and victimization, which are more prevalent in women, affect the development of mental disorders, including MDD (Hyde and Mezulis, 2020; Klein and Martin, 2021; Krahé and Berger, 2017).
Gender Roles
The association between gender inequalities and depression has been well studied. Negative early-life experiences resulting from gender inequalities have been associated with the increased vulnerability of women to depression (Remes et al., 2021). Studies have shown that women are more likely to experience complex and varied adverse childhood experiences
(ACEs)2 (Finkelhor, 1987; Silverman et al., 1996; Weiss et al., 1999). These exposures are associated with an increased risk of depression (Bochicchio et al., 2024; Weiss et al., 1999), with inflammation implicated as a possible underlying biological mechanism (Iob et al., 2020).
Certain events in the reproductive life cycle, including menarche, pregnancy, the postpartum period, and menopause, come with new responsibilities, role transitions, body image considerations, societal pressures, and hormonal changes and are also associated with higher rates of depression (Albert, 2015; Kundakovic and Rocks, 2022). Developmental changes in a woman’s life-span, starting from childhood, can lead to shifts in gender roles, societal pressures, vulnerability, discrimination, and variable social support, all of which can influence risk of depression in women (Sachs-Ericsson and Ciarlo, 2000; Vafaei et al., 2016). Special considerations that increase risk include ideas around sexuality, conception, and pregnancy, including carrying out a pregnancy, pregnancy loss, and infertility (Salari et al., 2024).
Changes in body appearance and function starting during adolescence can be associated with societal pressures and experiences of sexual and physical vulnerability. Life events related to reproduction in women, such as miscarriage, are associated with significant trauma and grief (Quenby et al., 2021). Evidence suggests that infertility is a susceptibility factor for depression (Salari et al., 2024), although the association is likely complex (Bagade et al., 2023). For example, female hormonal fluctuations are associated with sleep disorders, which are associated with infertility, which is associated with a higher rate of mood disorders (Baker and Lee, 2022).
Reproductive function itself is not a solely biological prospect; feelings and expectations surround it, and in societies in which motherhood is perceived as an essential component of a woman’s identity, women who do not achieve motherhood may experience significant stress (Bagade et al., 2023). Efforts to conceive are unsuccessful for one in four couples worldwide, and infertility is associated with emotional, social, relational, and financial burdens (Warne et al., 2023). One study demonstrated an association between assisted reproductive technology and current and past mental disorder diagnoses, with onset during assisted reproductive technology in up to one-third of patients undergoing it (Warne et al., 2023).
In addition to hormonal fluctuations, women may experience pressures and expectations about body appearance, which can be conflicting and
___________________
2ACEs are defined as “emotional, sexual, or physical abuse, emotional or physical neglect, and five types of household dysfunction including household substance abuse, a violent home environment, family mental disorders, parental incarceration, and parental separation or divorce” and contribute to the development of risk factors related to morbidity and mortality during adulthood (Christensen et al., 2021; Felitti et al., 1998; Nelson et al., 2017).
contradictory at baseline; pregnancy is associated with noticeable changes, including increased abdominal girth, overall weight gain, and other appearance changes that may be at odds with societal and internalized notions of beauty (Crossland et al., 2023). During pregnancy, women in general are at increased risk for intimate partner violence (IPV). One meta-analysis demonstrated an approximately 9 percent rate of physical and sexual IPV for pregnant women in North America and a nearly 30 percent rate of psychological IPV (Román-Gálvez et al., 2021).
Condition Presentation and Diagnosis
Physical symptoms associated with depression are more common in women and may include fatigue, appetite, weight disturbances, and musculoskeletal pain (Lara-Cinisomo et al., 2020). A major depressive episode during the postpartum period and whether to delineate this from a separate diagnosis of MDD is a point of debate, and the literature varies based on how the investigators defined the postpartum period, epidemiological aspects, and etiology and treatment aspects (Batt et al., 2020). The Edinburgh Postpartum Depression Scale is the most used screening tool for PPD (Sit and Wisner, 2009). Ideally, these tools should incorporate symptoms unique to postpartum experiences that are not captured in MDD. Evidence is mixed that depression with an onset of 8 weeks postpartum might be different than MDD (Batt et al., 2020).
Although sometimes used interchangeably, perinatal depression is different from PPD. Perinatal depression refers to major or minor depression that starts during pregnancy or up to 12 months postpartum (Batt et al., 2020). The U.S. Preventive Services Task Force (USPSTF) recommends screening and identifying women at increased risk of perinatal depression and referring them to interventions involving counseling (Curry et al., 2019).
As with other psychiatric conditions, research has failed to develop consistent and reliable biomarkers for depression. Certain neurotransmitters and inflammatory markers show promise based on physiological findings in persons with depression, but little is known about how sex affects potential biomarkers (Carvalho Silva et al., 2023; Strawbridge et al., 2017).
Treatment and Management
Early and effective treatment of MDD is crucial to achieve positive outcomes, especially in women, but MDD treatments are underutilized (Goodwin et al., 2022). Studies have shown differential responses to certain types of antidepressants and note that many patients do not respond adequately to traditional antidepressants (Dwyer et al., 2020). Several factors, including body composition and hormonal influences, attenuate the
differential response and pharmacokinetics in women. Studies have shown that women respond better to SSRIs, and men to tricyclic antidepressants (Sramek et al., 2016). However, that increased response to SSRIs appears to be age dependent and is not observed in menopause (LeGates et al., 2019). Other studies have shown more rigorous therapeutic benefits of certain medications after hormone therapy (Parry, 2010). Combination therapy has also demonstrated increased benefit in women over men (Parry, 2010). SSRIs in addition to estrogens are usually more beneficial in improving mood than SSRIs or estrogen treatment alone for major depression, whereas the selective norepinephrine and serotonin reuptake inhibitors do not require estrogens to exert their antidepressant effects in menopausal depression (Parry, 2010).
In 2019, Food and Drug Administration (FDA) approved brexanolone, a naturally occurring molecule that modulates GABAergic activity and is associated with significant improvement in depressive symptoms in adult women with PPD (Epperson et al., 2023). It is administered by intravenous infusion in a health care facility over a 60-hour period. In 2023, FDA approved zuranolone as the first oral drug to treat PPD (FDA, 2023).
Other research findings support the benefits of lifestyle-based approaches and alternative therapies in preventing and managing depressive symptoms. Meta-analyses of randomized controlled trials (RCTs) support how exercise improves depressive symptoms and quality of life (Blumenthal and Rozanski, 2023). Numerous studies also indicate that high-quality sleep (Cui et al., 2024), diets low in proinflammatory foods (Choi et al., 2024; Khosravi et al., 2020; Oddy et al., 2018; Tolkien et al., 2019), and the microbiome composition (Burokas et al., 2017; Radjabzadeh et al., 2022) may prevent or reduce risk and symptoms of depression. Interventions such as strong social support, cognitive behavioral therapy, acupuncture, and mindfulness-based therapies have been shown to improve symptoms (Cui et al., 2024; Li et al., 2022b; Witkiewitz and Bowen, 2010; Yang et al., 2023).
Disparities
Racial and Ethnic Disparities
Many studies have shown that hormonal, biological, and psychosocial factors related to depression vulnerability are exacerbated in adolescent girls identifying as lesbian, gay, bisexual, transgender, queer, intersex, asexual (LGBTQIA+) and in certain racial and ethnic groups (Patil et al., 2018). The interplay of gender and racial or ethnic socialization contributes to overall identity formation, which in turn informs coping strategies that are also related to the brain structure and function underlying or protecting against depression (Patil et al., 2018).
Although studies have reported that Black and African American and Hispanic/Latina women report lower levels of depression, symptom presentation differs. A recent narrative synthesis explained that Hispanic/Latina women reported more physical symptoms than White women (Phimphasone-Brady et al., 2023). Furthermore, a systematic review showed that African American women were more likely to express physical symptoms than White women (Lara-Cinisomo et al., 2020). Researchers have not examined symptom presentation in PMS or PMDD in girls and women by race and ethnicity (Phimphasone-Brady et al., 2023).
Lack of culturally competent care for depression also contributes to mental health disparities. For example, women identifying as Black, African American, American Indian, Alaska Native, and Hispanic/Latina placed a strong emphasis on having a health care provider of similar racial and cultural background compared to men of the same racial and ethnic backgrounds (Eken et al., 2021). Several studies support that racial and ethnic concordance between the health care provider and patient may lead to better communication, perceptions of care, and health outcomes, although there are issues of heterogeneity and a lack of quality in the extant evidence (Moore et al., 2023; Shen et al., 2018). Women from these racial and ethnic groups may also experience greater stigma in health care settings, which may be linked to lower access of mental health care and services (Misra et al., 2021). More research specifically focused on women is needed in this area.
Research Gaps
Despite immense strides in acknowledging sex differences in depression and gaining insight as to how sex gonadal hormones, particularly estrogens, heighten the risk in women, significant knowledge gaps remain regarding the mechanisms that underlie sex differences. As most experimental animals do not have a menstrual cycle, alternative models are needed to better understand the molecular basis of the intrinsic, hormonally driven vulnerability to depression (Kundakovic and Rocks, 2022). More research is also needed to study sex hormone and gene interactions (Kundakovic and Rocks, 2022) and understand how sex chromosome effects play a part in the pathophysiology of depression in women. Findings from such research can be leveraged to develop diagnostics and treatments that are most efficacious in women.
As with other chronic conditions, inflammation appears to play a mediating role between depression inducing factors and MDD. More research is needed to understand inflammatory triggers for depression in women (e.g., trauma, ACEs, stress) to guide the development of future therapies.
In addition, more studies on the bidirectional influences of depression and other chronic conditions (e.g. AD and CVD), would yield insight into how negative affectivity and rumination could lead to physiological alterations that increase the risk of developing other chronic conditions. The understanding of how depression, including PMDD and PPD, manifests throughout reproductive milestones and across the life course, such as during the menopausal transition, need to be further explored.
A better understanding of how environmental influences or extrinsic factors influence depression in women is needed. Research on external factors affecting mood and depressive symptoms have multiplied in recent decades, and it is important to understand the factors underlying why some individuals are more susceptible. Further research on biopsychosocial resilience factors, such as nutrition/nutrients, microbiome, lifestyle (e.g., exercise), and family/community/medical caregiving support, and how these counter risk factors for depression in women, is important.
More research is needed to develop appropriate screening tools (e.g., questionnaires and biomarkers) that discern the diagnostic features of perinatal depression, PPD, and MDD to better identify women at risk during the reproductive stages and initiate timely interventions. Adapting routine screening tools for use in racially and ethnically diverse women remains paramount, as research has demonstrated racial differences in depression expression and presentation (Salihu et al., 2022). Similarly, research needs to incorporate racial, ethnic, and cultural considerations into clinical trials to treat depression in women at all stages of the life cycle, since racial and sex differences exist in screening and treatment practices (Hyeouk et al., 2015). Network-based approaches, involving collaborations across multiple institutions, may help facilitate identifying more effective treatment strategies for women (Flynn et al., 2018).
SUBSTANCE USE DISORDER
Women with substance use disorder (SUD) face complex, multifactorial challenges across the life course. Although more men than women meet diagnostic criteria for SUDs (Fonseca et al., 2021; Koob and Moal, 2006)—a difference that has been shrinking for the last few decades (Fonseca et al., 2021; Keyes et al., 2008)—women progress from initial use to dependence at a faster rate and may exhibit more severe symptoms (Brady and Randall, 1999; Kosten et al., 1993).
The early stages of the United States opioid epidemic had a significant increase in nonmedical use of prescription opioids (Hirschtritt et al., 2018; Mazure and Fiellin, 2018). This trend was primarily driven by the treatment of chronic pain, which was more prevalent in women than men
(Racine et al., 2012; Templeton, 2020). Women were found to develop opioid use disorder (OUD) more rapidly than men (Hernandez-Avila et al., 2004; Mazure and Fiellin, 2018), often because clinicians were more likely to prescribe them to women (Mazure and Fiellin, 2018). Research indicates that it typically takes only 5 days to become physically dependent on opioids (Shah et al., 2017).
Biological Factors
Progress in understanding mechanisms linked to addiction-like behaviors have relied on rodents as experimental models. In general, female rodents, compared to male rodents, will self-administer substances of abuse and alcohol more rapidly, are more motivated to take cocaine (Cummings et al., 2011; Quigley et al., 2021a; Roberts et al., 1989), and show a higher drive to self-administer morphine and heroin (Cicero et al., 2003; Lynch and Carroll, 1999; Roth et al., 2002). They also consume more alcohol, relative to body weight, and engage in higher levels of cue-mediated alcohol-seeking behaviors (Cofresí et al., 2019; Crabbe et al., 2009). After periods of extended access to substances of abuse, female rats tend to exhibit greater symptoms of withdrawal, apart from alcohol and possibly opioids. In an animal model of relapse after forced abstinence, female rats compared to male rats exhibited greater cocaine-induced relapse (Lynch and Carroll, 2000), and greater drug- and cue-induced relapse (Cox et al., 2013).
Sex Chromosome Effects
Research in animal models has demonstrated the influence of sex chromosomes, independent of gonadal sex hormones, on addictive behavior. For example, preclinical studies using the FCG mouse model (see Chapter 2) show that after exposure to alcohol, XY mice developed a pattern of alcohol habit formation, and XX mice exhibited reward-seeking addictive behaviors regardless of gonadal phenotype (Barker et al., 2010). Additional research using this model also distinguished between sex chromosome and sex hormone effects on other aspects of alcohol addictive behaviors, including intake, preference, tendency for relapse, and habit formation. The results showed that the XX chromosome complement was associated with higher consumption initially and at relapse (Sneddon et al., 2022).
Studies analyzing cocaine addiction in the FCG animal model found both independent and interactive effects of sex chromosomes and sex gonadal hormones (Martini et al., 2020). Several addiction-associated genes are on the X chromosome—for example, genes encoding monoamine oxidase, an enzyme that regulates various neurotransmitters and implicated
in addiction mechanisms, and receptors for the neurotransmitters, GABA and glutamate (Krueger et al., 2023)—and leaky gene expression from the inactivated X chromosome in females could potentially explain sex differences in addictive behaviors (Krueger et al., 2023).
The Effects of Estrogens
Research has shown that estradiol, the most potent estrogen produced by the ovaries, modulates activity in the brain’s reward system in females and influences the acquisition, cravings, self-administration, and intake of substances. Female rats show enhanced motivation for cocaine at elevated estradiol levels, suggesting that estradiol facilitates their motivation for drug taking (Becker and Hu, 2008; Hu et al., 2003; Nicolas et al., 2019; Roberts et al., 1989). In gonad-intact female rodents, motivation is greatest during periods of the estrous cycle when estradiol is elevated (Becker and Hu, 2008; Becker and Koob, 2016; Roberts et al., 1989). Females in estrus exhibit greater drug-primed relapse for self-administering cocaine compared with those not in estrus and males, suggesting that estradiol enhances drug cravings that may contribute to the persistence of cocaine-seeking behaviors long into abstinence (Kippin et al., 2005). This notion is supported by studies showing that without estradiol replacement, adult females whose ovaries were removed have lower motivation for cocaine than those with estradiol (Becker and Hu, 2008; Hu et al., 2003; Nicolas et al., 2019). Sex differences in cocaine self-administration are evident, however, even in animals without gonadal hormones, with females without ovaries exhibiting greater intake than males (Hu et al., 2003). Exogenous estradiol is sufficient to enhance cocaine acquisition in females without ovaries (Hu and Becker, 2008; Hu et al., 2003; Lynch et al., 2001) but does not facilitate or enhance it in males (Jackson et al., 2005). Finally, sex differences in cocaine self-administration develop in animals that are not exposed to testosterone during the perinatal period and are exposed to ovarian hormones during puberty (Perry et al., 2013), indicating organizational influences of gonadal hormones on the brain that affect sex differences in this behavior.
Administered estradiol also facilitates the opioid acquisition in female rats without ovaries (Cicero et al., 2003; Lynch and Carroll, 1999) and enhances behavioral responses to and reinforcing effects of amphetamine (Robinson and Becker, 1986). During nicotine withdrawal, female rats with intact ovaries exhibit greater anxiety-like behaviors and stress responses compared with rats without ovaries (Torres and O’Dell, 2016). Intake of ethanol is higher during the stage of the estrous cycle in intact females when they are no longer receptive to the male (Forger and Morin, 1982; Roberts et al., 1998; Witt, 2007).
Other Biological Factors and Mechanisms
The transition from casual use of substances to developing a SUD in either sex is mediated by the brain’s reward system, with neurons responsive to the neurotransmitter dopamine activated in response to stimuli necessary for health and reproductive success, such as food consumption, sexual behavior and social interactions (Everitt et al., 1999; Robinson et al., 2015; Schultz, 1986; Wise and Rompre, 1989). Substances act on the reward pathway to induce dopamine neurotransmission either directly or indirectly, and repeated drug use causes physiological changes in the brain that include enhanced dopamine release (Engel and Jerlhag, 2014; Koob and Le Moal, 2008; Robinson and Becker, 1986). Repeated exposure to the acute rewarding effects of drugs can lead to escalated intake and compulsive drug-taking behaviors (Ahmed and Koob, 1998; Chartoff et al., 2009; Wise and Koob, 2014). Sex-related differences occur in neural responses to external stimuli and internal endocrine and physiological signals. Consequently, even when behaviors are the same in both sexes, sex related differences may be present in the neural circuitry activated and the brain’s response to a given situation (Farrell et al., 2013; Orsini et al., 2022; Quigley et al., 2021b).
The Maternal in Utero Environment
Exposure to addictive substances in utero predisposes offspring to developing SUD later in life. Research using animal models has demonstrated that maternal exposure to addictive substances, such as opioids or a high-sugar diet, increases the vulnerability of offspring to drug-seeking behaviors in adulthood (Abu and Roy, 2021; Gawliński et al., 2020). Human studies have shown that prenatal substance exposure plays a critical role in developing addictive behaviors as offspring get older. Alcohol exposure during pregnancy predicts drug dependence behaviors in adolescent and adult offspring after adjusting for certain confounding factors, including family history, prenatal smoking, current parental drinking habits, the environment, and other sociodemographic factors (Baer et al., 1998, 2003; Dodge et al., 2023). Similarly, prenatal cocaine exposure is associated with developing SUD in adolescent and adult offspring.
Although these studies either found no sex differences (Min et al., 2014) or involved small sample sizes that precluded sex-based analysis (Min et al., 2023), more recent studies have provided evidence of sex bias. In one study, prenatal substance use exposure was directly related to substance use progression in girls but not boys during adolescence (Marceau et al., 2021). In addition, findings from the Helsinki Birth Cohort Study have shown that certain measures of prenatal growth—considered a surrogate marker for exposures in utero—was associated with hospitalization for SUD in female adult offspring, suggesting that the fetal period may
be a critical developmental period during which women could develop sensitivities for SUD (Lahti et al., 2014). Overall, several lines of evidence support the maternal in utero environment as a major determinant in SUD susceptibility. However, as developing an addiction involves multiple factors, further research is needed to understand the sex-related vulnerability of women to SUD.
Specific Risk Factors in Women
Higher alcohol intake is associated with higher premenstrual or menstrual distress and negative affective states in women. This supports the hypothesis that women with PMS have higher levels of alcohol use or abuse (Evans and Levin, 2011; Kiesner, 2012; Mello et al., 1990; Svikis et al., 2006). Furthermore, women who have a history of, or are currently using cocaine, opioids, or alcohol have evidence of menstrual cycle disruptions (Emanuele et al., 2002; Mello et al., 1990, 1997; Schmittner et al., 2005). When the menstrual cycle is disrupted, it is more difficult to discern how, or if, ovarian hormones are influencing drug taking or motivation for it. In women aged 18–35, estradiol enhances the positive subjective effects of stimulant drugs, such as amphetamines, similar to findings from studies using animal models (Justice and de Wit, 1999).
Women often report using drugs initially to cope with negative affective states, such as depression (Kuntsche and Müller, 2011; Müller and Kuntsche, 2011), whereas men usually cite peer influence or sensation-seeking (Kuntsche and Müller, 2011). Other sex-related risk factors for substance use and SUD include IPV, which is more likely to affect women, and other social stressors such as death of child or loved one and divorce (NIDA, 2020). Mental health conditions are also more common among women who use substances of abuse. In particular, posttraumatic stress disorder has a strong relationship with developing a SUD, especially among young girls (NIDA, 2020).
Condition Presentation and Diagnosis
Diagnostic criteria for SUD are the same for men and women, despite gender differences in how SUDs manifest. Some research has demonstrated that women entering SUD treatment programs present with a more severe clinical profile and an array of related social, medical, psychological, and multiple mental disorders (Mazure and Fiellin, 2018). Research has shown that depression, which is more prevalent in women, is associated with drug cravings and relapse (Curran et al., 2007; Zilberman et al., 2007), making it important to account for depressive symptoms when women present with concern about an SUD. Furthermore, although women may have used
substances for a shorter duration and in smaller amounts than men, the acceleration of SUD severity and deterioration in functionality occurs more rapidly (Fonseca et al., 2021; Mazure and Fiellin, 2018).
Treatment and Management
Despite women accounting for one-third of substance users, only one-fifth of the individuals receiving SUD treatment are women (UNODC, 2015). Women face greater barriers than men in accessing SUD treatment and harm reduction programs, such as syringe service programs, opioid overdose services, and drug testing. Based on National Survey of Drug Use and Health (NSDUH) data, less than 10 percent of women of reproductive age in need of SUD treatment were receiving it (Martin et al., 2020). Figure 6-2 shows that from 2007 to 2014, treatment receipt for SUD remained relatively stable, whereas it increased for depression. Systemic, structural, social, and cultural barriers that limit access to SUD treatment include stigma, punitive attitudes, discrimination, rigid admission requirements, and inflexible scheduling (see Chapter 7).
Disparities
The literature on differences in prevalence and mortality from overdose by race, ethnicity, and sexual identity is sparse (Schuler et al., 2020). A few studies have reviewed factors that may be associated with SUD for different
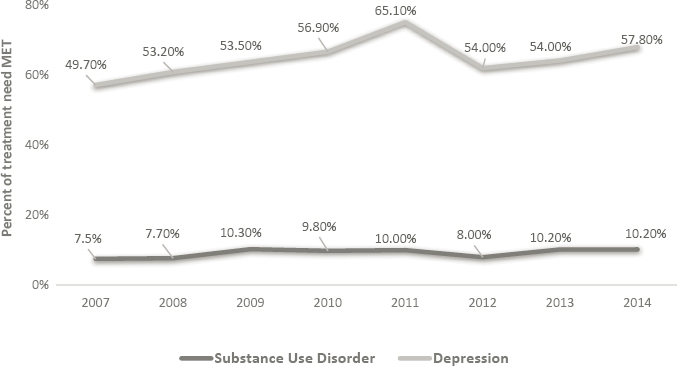
NOTE: SUD = substance use disorder.
SOURCE: Martin, 2020.
racial and ethnic groups. For example, studies have found that Latinas mainly from urban and metropolitan communities and born in the U.S. had greater odds of SUD, including OUD (Castañeda et al., 2019; Takada et al., 2024). A 2019 study based on data from the NSDUH found that Native Hawaiian and Pacific Islander women had a higher prevalence of SUD, whereas Asian American women had higher rates of stimulant use compared to other racial and ethnic groups (Wang et al., 2023a). When comparing the relative risk of heavy episodic drinking and marijuana use, lesbian and bisexual women of color had an increased risk of use (Schuler et al., 2020).
Few studies have considered the role of both gender and race and ethnicity on treatment use (Pinedo et al., 2020). Studies have shown that Black and Hispanic women are less likely to receive treatment for SUD (Martin et al., 2020) and OUD (Iobst et al., 2024; Scheidell et al., 2024). Black, Latina, and other women of color from low-income communities wait longer before entering treatment and have a shorter length of stay in methadone treatment programs compared to men (Marsh et al., 2021).
Research Gaps
Differences in the biology of substance use are evident, influenced by factors such as ovarian hormones, drug type, dose, and exposure history (McHugh et al., 2018). It is also critical to understand how sex chromosome effects differentially influence addiction patterns in male and female individuals, which likely starts early in development via organizational effects on brain anatomy and function. Given the complexity of sex differences across bodily systems, multiple levels of analysis are needed. (McHugh et al., 2018).
Women develop drug dependence faster than men, but most research on risk factors and rates of progression from first use to SUD in women have been retrospective and not yielded a good understanding of the risk and protective factors in women and men. The Adolescent Brain Cognitive Development study, funded by the National Institute on Drug Abuse, will provide prospective data and analysis, and may begin to fill several of these research gaps (NIDA, 2023).
Despite no differences in how SUDs are diagnosed in men versus women, differences in gender and sex appear related to when they present for diagnosis (Mazure and Fiellin, 2018). Women tend to present symptoms of chronic pain and increased pain sensitivity and are thus more likely prescribed painkillers by physicians to alleviate symptoms (Mazure and Fiellin, 2018).
Diagnostic criteria recognize that SUD may manifest differently in women and men, but research is needed to determine if different diagnostic criteria are needed. Research assessing early interventions in women who
experience negative affective states, such as depression, or are exposed to adversities, such as ACEs, that may make them prone to drug use needs to be a priority. Considering the complexity of SUDs and the opioid epidemic, enhancing surveillance systems to encompass the intersectionality of factors such as gender, race, ethnicity, and geographic location will enhance screening and treatment approaches for diverse groups of women (Barbosa-Leiker et al., 2021).
HIV/AIDS
Chapter 3 noted that the rates of new HIV diagnoses and persons living with diagnosed HIV infection are much lower in women than men (CDC, 2023b). Although women represent a small proportion of new cases in the U.S. population living with HIV/AIDS, they have important and distinct characteristics of infection. To reduce the burden of HIV infection in both men and women, the White House Office of National AIDS Policy developed the National HIV/AIDS Strategy and implementation plan for Ending the HIV Epidemic in the United States (CDC, 2023a; HIV.gov, 2023). The stated goal is to reduce the number of new HIV infections and includes recommendations across four pillars of action: test everyone for HIV or DIAGNOSE infection, TREAT those who test positive with antiretrovirals and ensure viral suppression, PREVENT those who test negative from becoming infected by providing pre-exposure prophylaxis (PrEP) medication and syringe services for those who inject drugs, and RESPOND by predicting new outbreaks (CDC, 2023a).
Biological Factors
Sex Chromosome Effects
Several important male versus female sex chromosome differences can influence various aspects of HIV infection. Differences in the sex complement can lead to variations in gene expression patterns, including those related to immune response and susceptibility to infections such as HIV (Moran et al., 2022; Scully, 2018). Furthermore, specific X chromosome genes encode proteins that play a crucial role in the innate immune response, including toll-like receptor-7 (TLR-7) and TLR-8, which are involved in detecting viral infections (Moran et al., 2022). As noted in Chapter 2, females undergo X chromosome inactivation to balance gene expression between the sexes. If the second X chromosome escapes inactivation, that leads to differences in gene dosage, which could contribute to variations in HIV susceptibility, immune response, and disease progression (Moran et al., 2022; Scully, 2018).
Epigenetic mechanisms, such as DNA methylation and histone modifications, can regulate gene expression without altering the underlying DNA sequence. Sex-specific epigenetic patterns may influence immune responses to HIV and contribute to differences in disease outcomes (Chlamydas et al., 2022). Understanding these sex chromosome differences and their interactions with hormonal and epigenetic factors is crucial for elucidating sex-specific disparities in HIV infection and developing tailored prevention and treatment strategies for both sexes.
The Effects of Estrogens
Female sex hormones have profound effects on multiple systems throughout the body and over the life-span. Female sex hormone fluctuations, such as during the menstrual cycle, in pregnancy, and from the use of contraceptives, influence the risk for HIV infection directly (Szotek et al., 2013) or indirectly through modulatory effects on the immune response (Swaims-Kohlmeier et al., 2021). During the menstrual cycle, there are changes in the vaginal tissue physiology and microbiome that can increase susceptibility to infection (Boily-Larouche et al., 2019). High levels of estrogens during the menstrual cycle may be protective and are associated with lower HIV acquisition (Asin et al., 2008; Moran et al., 2022; Wira et al., 2015). Progestin-only contraceptives, however, have been associated with an increased risk of acquiring HIV, although the precise mechanism is unknown (Moran et al., 2022; Ralph et al., 2015; Rodriguez-Garcia et al., 2021).
Pregnancy induces notable alterations in immune responsiveness (Akoto et al., 2021). The proinflammatory immune states observed during the first and third trimesters contrast with the anti-inflammatory state in the second trimester, and these changes could influence susceptibility to HIV infection (Rodriguez-Garcia et al., 2021). In addition, the molecular signals involved in mounting an immune response and the antimicrobial environment of the reproductive tract change significantly during pregnancy, which can also influence susceptibility to HIV infection. The cervical plug formed during pregnancy contains immune factors that exhibit potential anti-HIV activity (Mhlekude et al., 2021). Despite these insights, research has yet to definitively discern whether pregnancy heightens or decreases the risk of HIV infection and indicates an important research gap (Rodriguez-Garcia et al., 2021).
Sex hormone levels fall during menopause, affecting many immune biomarkers in the female reproductive tract. Research using models of cervical tissue has provided evidence of immune activation and increased HIV replication in menopausal tissue, indicating that menopausal women may have specific biological risks for acquiring HIV (Rodriguez-Garcia et al., 2021).
Specific Risk Factors in Women
Sexual behaviors and drug use are major risk factors associated with HIV infection among women. Eighty-four percent of new diagnoses among women are attributed to heterosexual contact and 16 percent to intravenous drug use (CDC, 2021b). A recent study of HIV risk behaviors in women found that 7 percent of cisgender women with HIV had had condomless sex in the past 12 months (CDC, 2022), which increases the risk of not only HIV but also other sexually transmitted infections, such as chlamydia, gonorrhea, and syphilis (NASEM, 2021). It is now well understood that sexually transmitted infections can increase susceptibility to HIV infection and render persons with HIV more infectious (NASEM, 2021).
Intravenous and other drug use has been increasing in the United States, leading to new HIV infections among persons who use drugs (Springer et al., 2020a, 2020b); sharing and reusing needles, syringes, or other injection equipment can increase the risk of getting or transmitting HIV. One study reported that women who share needles had a significantly higher likelihood of contracting HIV compared to men, with the authors attributing that to multiple and overlapping sexual and drug use partners (Riehman et al., 2004; Valente et al., 2001). Substance use can also affect judgment and lead to sexual behaviors that increase the risk of HIV, such as unprotected sex or sex with multiple partners (Riehman et al., 2004; Valente et al., 2001).
Condition Presentation and Diagnosis
In the early stage of infection, women have higher CD4+ T cell counts and lower HIV plasma RNA loads, but both men and women progress to AIDS at similar rates. Women, however, have been seen to progress to AIDS at higher rates compared to men of equivalent HIV RNA load. This has implications for screening, testing, and treatment (Farzadegan et al., 1998; Moran et al., 2022; Ziegler and Altfeld, 2016).
Early in the AIDS epidemic, the Centers for Disease Control and Prevention (CDC) set the case definition of AIDS based primarily on conditions seen in gay men and without fully capturing the experience of women (McGovern, 1994; OTA, 1992). Gynecological conditions, such as cervical dysplasia, pelvic inflammatory disease, and recurrent yeast infections, more common in HIV-infected women were not included. After significant protests by women and others, CDC changed its definition of AIDS; candidiasis and invasive cervical cancer are now among the listed conditions affecting women (CDC, 2021a).
Treatment and Management
PrEP and Treatment
PrEP is the standard approach to HIV prevention for people who are at risk. The regimen consists of three FDA-approved medications that prevent infection by almost 99 percent: two daily oral formulations and one injectable medication administered every 8 weeks. USPSTF released new guidance in August 2023 (Liu et al., 2023; USPSTF et al., 2023) with an “A” recommendation, indicating high certainty of substantial benefit from these medications to prevent HIV. Although the national goal is to increase the estimated people with PrEP indications actually being prescribed PrEP to at least 50 percent by 2025 and remain at 50 percent by 2030, only 10 percent of women who could benefit from PrEP were prescribed it in 2019, indicating major barriers to access for women (CDC, 2021d).
Another goal is to provide life-saving combination antiretroviral therapy3 (ART) to all persons within 7 days of a positive HIV test (Gandhi et al., 2023) and to achieve 100 percent viral suppression. Staying in HIV care is important to achieving and maintaining viral suppression and is improving health and morbidity for the individual and reducing transmission to uninfected individuals (Eisinger et al., 2019). In 2020, 23 percent of cisgender women with HIV reported missing at least one medical appointment in the past 12 months (CDC, 2023c), and approximately 63 percent of them with HIV reported taking all of their doses of ART over the last 30 days (CDC, 2023c). In 2019, only 64 percent of women with diagnosed HIV in 44 states and the District of Columbia were virally suppressed (CDC, 2021d).
Having access to needed ancillary services could reduce barriers to achieving and maintaining viral suppression; the top three services cisgender women with HIV reported needing but not receiving in the past 12 months in 2020 were dental care, the Supplemental Nutrition Assistance Program and Special Supplemental Nutrition Program for Women, Infants, and Children, and shelter and housing services (CDC, 2023c). Six percent of cisgender women with HIV reported homelessness in the past 12 months, and 21 percent reported symptoms of depression or anxiety (CDC, 2023c), both of which research has shown to affect the ability to access health care, HIV care, and ART and maintain viral suppression.
___________________
3ART is a regimen for treating HIV-positive individuals that reduces the levels of HIV in the body. PrEP is meant to prevent infection and taken before a person thinks they might be exposed.
With appropriate ART, women with HIV can live longer. In fact, life expectancy for people with HIV is approaching that of the general population. Some individuals living with HIV experience multiple comorbidities such as cardiovascular disease (CVD), diabetes, renal disease, neurocognitive disease, and cancer. In one study, women with HIV had significantly more ageing-related comorbidities than men with HIV. Women had higher prevalence of diabetes, bone disease, and lung disease than men (Collins et al., 2023). Multiple chronic conditions pose challenges for women, health care providers, and health care systems involved in managing them. These challenges are discussed further in Chapter 8.
Disparities
Disparities in HIV prevention and care for women persist, particularly among sexually, racially, and ethnically minoritized populations. Although these populations are well studied and progress has been made along the HIV care continuum, further work is needed to understand their needs for prevention and treatment, as the majority of research has focused on mother-to-child transmission (Nwangwu-Ike et al., 2023).
Racial and ethnic differences in HIV incidence in women are well known and captured by national surveillance data (see Chapter 4) (CDC, 2023b). Given that the prevalence and incidence is greatest among Black women, followed by multiracial, and Hispanic/Latina women, this disparity clearly needs to be addressed. One study following a prospective cohort of women from the HIV Outpatient Study (Buchacz et al., 2020) found that the odds of using and adhering to ART was lower among Black women compared to Hispanic/Latina and White women (Geter et al., 2019). Furthermore, the rate of achieving viral load suppression was lower among Black and Hispanic/Latina women (see Figure 6-3).
Qualitative studies have described barriers to care encountered by Black and Hispanic/Latina women living with HIV. Among Hispanic/Latina women, a systematic review cited emergent barriers to seeking HIV services that included lack of social support, insurance coverage, out-of-pocket fees, mental health outcomes, HIV-related stigma, and unique barriers such as language; fear of legal consequences, including deportation; and limited access to documentation (Geter Fugerson et al., 2019). Key features of quality of care for Black and Hispanic/Latina women included care that is integrated and coordinated with other support services, compassionate and nonjudgmental care, and shared decision making (Rice et al., 2020). Thus, HIV health care services require tailoring for these populations.
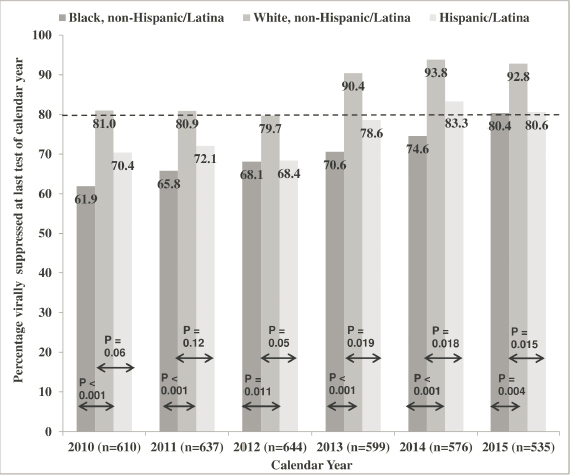
SOURCE: Geter et al., 2018.
Research Gaps
Significant research gaps exist regarding the safety and efficacy in women of the medications used to prevent and treat HIV, in part because women have been and continue to be underrepresented in research studies of PrEP and ART. A 2016 systematic literature review reported that women represent a median of 19.2, 38.1, and 11.1 percent of participants in ART and PrEP, prophylactic vaccine, and HIV cure strategies studies, respectively (Curno et al., 2016). One reason for this low participation is that most PrEP studies focus on enrolling solely men because they represent 80 percent of people living with HIV (CDC, 2021c). Other reasons include greater stigma affecting women with HIV compared to men (Karim et al., 2022) and the lack of women-specific recruitment strategies to educate and enroll women.
Without adequate representation of women in clinical trials of PrEP and ART, significant gaps remain in understanding gender differences in the virological, immunological, and clinical presentation of HIV. Specifically, studies that consider women-specific biological factors and their potential influence on PrEP are needed. Such studies would seek to better understand the role of sex hormones, genital inflammation, genital microbiome, sexually transmitted infection, and sex differences in immune responses
and mechanisms and how they may influence the safety and effectiveness of PrEP and ART (Karim et al., 2022). Clinical studies are also needed to better understand sex-related issues and PrEP, including for transgender women, and women who use drugs during the reproductive period along with hormonal contraception, during pregnancy and breastfeeding, and during menopause (Karim et al., 2022).
Research on improving HIV prevention and treatment for women is lacking, including research focusing on patient preferences regarding oral versus injectable ART or PrEP. Such studies would inform clinicians’ understanding of patient preferences about the settings in which to start ART and how to link and differentiate care by gender, comorbidity, and setting. Randomized prospective trials and studies powered to detect sex differences in access to treatment and prevention are also lacking.
Trials on HIV prevention and treatment that have an insufficient number of women do not have the power to differentiate medication effects by sex, which leave gaps in clinician knowledge around safety and efficacy (Gandhi et al., 2023). Without rigorous published studies that prospectively evaluate the effect of sex differences on preventing and treating HIV, recommendations specific to the needs of women will remain limited.
MIGRAINES/HEADACHES
Migraine disproportionately affects women in the years when they are most productive and active. Studies have shown that it is a complex, multifactorial disease with genetic factors increasing susceptibility and non-genetic factors regulating clinical presentation (Ferrari et al., 2015). The higher prevalence of migraine in women involves many factors, discussed next.
Biological Factors
During a migraine attack, multiple parts of the central and peripheral nervous systems are activated simultaneously (Charles, 2018). Migraine has several key mechanistic features. One mechanism involves central nervous system (CNS) processing of sensory input from the trigeminocervical complex (Bartsch and Goadsby, 2003) that activates the brain’s pain-processing center and the release of neuropeptides (Charles, 2018; Goadsby and Holland, 2019). The developmental differences in male and female pain-sensing systems and the effect of circulating hormones are the main physiological factors driving the higher prevalence of migraine among women (Pavlovic et al., 2017).
Cortical spreading depression—a slowly propagating wave of altered brain activity that involves changes in neuronal, glial, and vascular function (Charles and Baca, 2013)—is another important mechanism. Animal studies have shown a lower threshold to initiate cortical spreading depression in
female compared to male mice, regardless of the phase of estrous cycle (Brennan et al., 2007). Functional magnetic resonance imaging (MRI) studies have found structural changes in the specific brain regions involved in pain processing in women and men with migraine compared to healthy women and men (Maleki et al., 2012). Another study found reduced functional connectivity in sensorimotor networks and one of these same brain regions in women during migraine attack (Araújo et al., 2023).
Genetic factors may contribute to the threshold for triggering migraine in women compared to men. Population-based studies identified 38 genomic loci (Gormley et al., 2016) and genome-wide association studies (GWAS) identified 13 genes (Ferrari et al., 2015) as disease-associated gene variants. However, researchers have not found associations of these genetic variants with sex differences that explain the high prevalence of migraine in women, suggesting that other factors, perhaps hormonal differences, play significant roles in the clinical presentation.
Sex Chromosome Effects
Limited results address sex chromosome effects on the pathobiology of migraine. However, genetic studies have revealed several migraine susceptibility loci on the X chromosome, indicating possible roles of its genes. One study identified such a locus on Xq12, which includes the HEPH gene that encodes an enzyme involved in iron homeostasis, and others have implicated Xp22, Xq27, and Xp28 (Maher et al., 2012a; Maher et al., 2012b; Wieser et al., 2010). A case-control study has shown a link with the X chromosome gene SYN1, which encodes synapsin, a protein involved in neurotransmitter release at neuronal synapses (Quintas et al., 2020).
The Effects of Estrogens
Research has shown that female hormonal changes during puberty, pregnancy, and perimenopause can trigger migraine episodes, suggesting that ovarian hormones, notably estrogens, play an important role (Pavlovic et al., 2017). The “estrogen withdrawal effect” is the most commonly accepted theory for menstrual migraine, and women with histories of migraine exhibit faster declines in estradiol levels during the late luteal phase of the menstrual cycle (Pavlovic et al., 2017). Reductions in circulating estrogens before and during menstruation are associated with migraine episodes, suggesting that estrogens somehow hinder the abnormal neural activity associated with migraine (MacGregor and Hackshaw, 2004; Somerville, 1972).
Estrogens exert multiple effects on neural pathways involved in migraine, including the trigeminovascular pathway and central pain
pathways that traverse the central and peripheral nervous systems. These pathways contain neuronal populations that express ERs (Pavlovic et al., 2017). Animal studies have also identified ERs in trigeminal ganglion neurons (Bereiter et al., 2005; Puri et al., 2006). Estrogens have been reported to affect the excitability of second-order trigeminal sensory neurons involved in craniofacial pain syndromes, which was proposed as a mechanism shared in migraine (Cairns, 2007). In addition, estrogens, as well as progesterone, regulate serotonergic and GABAergic neurotransmitter systems, which are also involved in migraine pathophysiology (Vetvik and MacGregor, 2021).
Research has provided more detailed mechanistic insight into how estrogens are involved in migraine. It is thought to regulate migraine episodes by increasing expression of the oxytocin receptor and levels of the analgesic hormone oxytocin (Amico et al., 1981; Miller et al., 1989; Murata et al., 2014), which has also been implicated in preventing migraine attacks (Krause et al., 2021; Phillips et al., 2006; Tzabazis et al., 2017). Patterns of fluctuating plasma levels of oxytocin and estrogens are similar during the menstrual cycle (Engel et al., 2019), suggesting that reductions in oxytocin may also trigger migraine (Krause et al., 2021). In addition, evidence shows that estrogens modulate levels of calcitonin gene-related peptide (CGRP) (Cetinkaya et al., 2020; Labastida-Ramírez et al., 2019), a known contributor to migraine through its effects on vasodilation and neurogenic inflammation (Karsan and Goadsby, 2015). Research has shown that CGRP release is lower when levels of estrogen are high (Pavlovic et al., 2017), suggesting that estrogen withdrawals during certain phases of the menstrual cycle may increase CGRP and the likelihood of triggering an attack. Estrogens also regulate other analgesic neuropeptides and hormones, such as vasopressin (Lagunas et al., 2019), prolactin (Avona et al., 2021; Franchimont et al., 1976), and orexin (Porkka-Heiskanen et al., 2004), indicating that estrogens may mediate its effects on migraine through multiple mechanisms.
Specific Risk Factors in Women
Reproductive Milestones
The prevalence of migraine changes throughout a woman’s life (Pavlović, 2021). Migraine first appears during puberty, occurs around the time of menstruation, fluctuates through pregnancy, worsens during the perimenopausal stage, and usually improves after menopause (Burch, 2020; Pavlović, 2021). The high prevalence of migraine and severe disability rate that accompanies it during the productive ages can reduce productivity at work and impair family and social functioning (Buse et al., 2019).
Migraine and Oral Contraceptive Use
The risk of stroke is twofold higher in women experiencing migraine with aura as compared to women without migraine and sixfold higher in women using estrogen-containing contraceptives (Lee et al., 2023a). CDC’s U.S. Medical Eligibility Criteria of Contraceptive Use states that estrogen-containing contraceptives have an unacceptable health risk for women who experience migraine with aura, but the benefits for women who have migraine without aura outweigh the risks (CDC, 2016). At one time, research tied estrogen-containing contraceptives to a high risk of stroke (Calhoun, 2017), but today’s contraceptives contain much lower levels of estrogens, which appears to eliminate that excess risk. They can help control menstrual-related migraine and painful menstruation by stabilizing levels of estrogen (Nappi et al., 2022).
Gender Roles and Mental Health Factors
The 2010 ACEs study reported a higher prevalence of frequent headaches in individuals with ACEs (Anda et al., 2010). In the American Migraine Prevalence and Prevention Study, a history of emotional abuse was more common in women who experience migraine even after adjusting for depression and other sociodemographic factors (Tietjen et al., 2015), and data from the 2012 Canadian Community Health Survey-Mental Health showed that migraine was more prevalent in both women and men who experienced violence or sexual or physical abuse as a child (Brennenstuhl and Fuller-Thomson, 2015). Several studies have found that migraine is more prevalent in women who have experienced IPV compared to those who have not (Coker et al., 2000; Cripe et al., 2011; Gelaye et al., 2016; Vives-Cases et al., 2011), although more research is needed.
Despite some studies on the effects of IPV on a woman’s physical and mental health, not enough studies explore its relation to migraine. The known effects of maltreatment on the hypothalamic-pituitary-adrenal axis and stress-mediating homeostatic systems is the presumed mechanism linking childhood maltreatment and migraine (Tietjen et al., 2016).
Condition Presentation and Diagnosis
The International Classification of Headache Disorder third edition (ICHD-3) is the latest system for classifying headache disorders (IHS, 2018; Levin, 2022). The diagnostic criteria in ICHD-3 has been internationally accepted and used in headache-related studies. Migraine is a primary headache disorder with two major types, with and without aura. Migraine without aura for adults is defined as a minimum of 4 hours of headache
with specific symptoms, including nausea and an abnormal sensitivity to light and sound. Migraine with aura is identified with transient neurological symptoms accompanied by or preceding the headache.
Between 18 and 60 percent of women who experience migraine report that they occur most often during menstruation (Vetvik and MacGregor, 2021). Menstrual migraine is included in the appendix of ICHD-3, but the diagnostic criteria still require validation (Cupini et al., 2021). Based on those criteria, menstrual migraine occurs in two forms: pure and menstrual-related (Ceriani and Silberstein, 2023). Women with pure menstrual migraine report having an attack between 2 days before and 3 days after the start of menstruation and none at other times. Women who experience menstrual related migraine report attacks related to their menstruation and also at other times. Menstrual migraine lasts longer, causes more disability, and does not respond as well to acute treatment (Granella et al., 2004; Pinkerman and Holroyd, 2010).
Treatment and Management
“Throughout my life, I have been on and off treatments. Some have been helpful. Some have not. There have been significant side effects with a lot of the treatments and I faced a lot of challenges during graduate school, my working life, particularly during my pregnancies and while breastfeeding my children to find therapies that were safe and hopefully allow[ed] me to function . . . a lot of women delay pregnancy, go without treatments during certain times of life and that can negatively affect our lives and the lives of those around them. I have missed out on moments or times with family and friends.”
—Presenter at Committee Open Session
Acute Migraine Treatment
Migraine causes more than 1.2 million visits to U.S. emergency departments annually and is the third leading cause of emergency department visits in reproductive-aged women (Burch et al., 2018; Minen et al., 2018). Several families of drug—triptans, such as sumatriptan (Imitrex®), and ditans such as Lasmiditan (Reyvow®) are FDA approved as acute treatments (Yang et al., 2021). In March 2023, FDA approved zavegepant (Zavzpret®), a nasal spray that blocks CGRP activity and can bring relief within 30 minutes, to treat acute migraine with or without aura (Dhillon, 2023).
Guidelines based on high-quality, evidence-based approaches are not available for emergency room acute treatment (Robblee and Grimsrud, 2020). In addition, lack of referral or follow-up plan with a neurologist or headache specialist causes high readmission rates to emergency departments (Giamberardino et al., 2020), with one study finding that more than
a quarter of initial visitors for migraine return within 6 months (Minen et al., 2018). More studies are needed on acute care in emergency rooms to develop nationally accepted treatment protocols.
Management has changed with the approval of CGRP-blocking drugs as preventive agents. These new agents include rimegepant (Nurtec®), ubrogepant (Ubrelvy®) and atogepant (Qulipta®). In addition, monoclonal antibody therapies that block CGRP have recently been developed and hold much promise for preventing migraine: three FDA-approved, self-injectable drugs—galcanezumab (Emgality®), erenumab (Aimovig®) and fremanezumab (Ajovy®)—and andeptinezumab-jjmr (Vyepti®), which is an intravenous infusion every 3 months (Pope, 2023).
Migraine Diagnosis and Treatment During Pregnancy
Even though acute headache during pregnancy should always raise a concern about secondary headache disorders, migraine is the most common cause of these headaches (Burch, 2019). Although migraine is primarily a headache disorder, it increases the risk of preeclampsia, cerebral blot clots, and pregnancy-associated stroke compared to women without a history of migraine (Greige et al., 2023).
Up to 10 percent of pregnant women can start to experience migraine without aura, while the onset of migraine with aura has been reported up to 14 percent (Negro et al., 2017), and approximately 60–80 percent report that their migraine symptoms improve during pregnancy (Allais et al., 2019). Based on the data from American Registry for Migraine Research, 20 percent of women who experience migraine avoid pregnancy for fear of their migraines worsening, of having a difficult pregnancy with migraine disability, and the potential adverse effect of their migraine treatment on the fetus (Ishii et al., 2020).
The recommended first-line treatment for migraine during pregnancy is nonpharmacological therapies, including lifestyle changes, cognitive behavioral therapy, and biofeedback (Verhaak et al., 2023). However, accessing these therapies is not easy for pregnant women. Therefore, pharmacological therapies should be considered for those who either do not have access to nonpharmacologic treatments or did not benefit from them. One study supports that triptans are safe to take during pregnancy; however, a control group was not included for comparison (Ephross and Sinclair, 2014).
Based on the 2022 American College of Obstetricians and Gynecologists updated guideline, little high-quality data exist on treating migraine during pregnancy, leaving treatment decisions up to the patient and clinician (ACOG, 2022). Lack of data leads clinicians to hesitate when starting treatment for chronic migraine in pregnant patients. On a recent survey with women’s health care providers in Connecticut, 60 percent reported
they did not feel comfortable starting new preventive treatments during pregnancy, and 40 percent reported referring such patients to headache specialists or neurologists rather than starting treatment (Verhaak et al., 2023).
Disparities
Disparities among racial and ethnic groups exist regarding the severity of headaches. African American individuals reported more frequent and intense headaches and were more likely to discontinue treatment for migraine in specialty clinic settings than White individuals (Kiarashi et al., 2021). American Indian and Alaska Native individuals had the highest prevalence of migraine among all U.S. racial and ethnic groups, with the lowest rate among Asian Americans (Burch et al., 2018). A 2021 literature review found that only 46 percent of Black individuals received care for their migraines compared to 72 percent of White individuals and that only 14 percent of Black patients received a prescription for migraine medication compared to 37 percent of White patients (Kiarashi et al., 2021). The same survey found that Hispanic and Latina/Latino individuals were 50 percent less likely to receive a migraine diagnosis and were less likely to receive a prescription for migraine prevention medication than White individuals.
Transgender and gender-diverse individuals are affected significantly by migraine. Some evidence has suggested that transgender women who have received gender-affirming hormone therapy may have a higher risk of migraine resulting from the effect of estrogens on the nervous system/brain (Ahmad and Rosendale, 2022). A study based on National Health Interview Survey (NHIS) data from 2013 to 2018 found that bisexual women had 33 percent greater odds of headache/migraine than did lesbian women. The same study found the risk of migraine was 25 percent higher in sexual minority women compared to heterosexual women (Heslin, 2020).
Research Gaps
Research has long established that more women than men experience migraine, but the pathophysiological mechanisms and genetic influences underlying sex differences in the presentation and frequency of occurrence throughout the life-span are not well understood. This is in part the result of most preclinical research using animal models that only focus on males (Eisenstein, 2020). One of the very common and debilitating forms for women, menstrual migraine, has only been included in the appendix of ICHD-3 and is still waiting to be validated as a diagnosis. The lack of diagnostic certainty leads to exclusion from treatment trials; there are no specific treatment options available.
Based on the data, 60–80 percent of women with migraine report improvement during pregnancy, but no literature addresses why the 20–40 percent do not. Evidence on the treatment options regarding benefits or risks during pregnancy, breastfeeding, or postpartum is insufficient. Specific data are also lacking on dosing or the effect of comorbidities for medications during pregnancy. New studies are also needed to clarify the specific formulations of combined hormonal contraceptives and the risk of stroke associated with them in different subtypes of migraine. Overall, more data are needed to develop treatment guidelines for pregnancy.
Despite evidence supporting an association between migraine and trauma (e.g., ACEs, interpersonal violence), more research is needed to explore how social factors influence susceptibility to migraine. No data exist on the effect of infertility treatment or gender-affirming hormone therapy on migraine and risk of stroke in patients with history of migraine. The effect of migraine on sexual and gender minority groups is challenging to study, and studies have conflated sex and gender when collecting this information. Nationally accepted treatment approaches in emergency room for acute migraine care are needed, as are strategies to reduce the need for emergency care.
CARDIOMETABOLIC CONDITIONS
“There has to be more attention paid to awareness [of CVD symptoms in women] . . . in the medical community and hospital emergency rooms . . . women talk so often of just being dismissed or diagnosed with GERD [gastroesophageal reflux disease] . . . and not taken seriously that they may be having a heart attack.”
—Presenter at Committee Open Session
Cardiometabolic conditions cause significant morbidity and mortality in women. These conditions often occur together, and causes are multifactorial, in which their development can be explained by various factors. This section focuses on two chronic cardiometabolic conditions with high morbidity and mortality in women—CVD and stroke—and highlight sex and gender differences in known risk factors for both. Other cardiometabolic conditions, including obesity, metabolic syndrome, and Type 2 diabetes, are also discussed as risk factors.
Biological Factors
A discussion of cardiometabolic conditions encompasses multiple major clinical heart and circulatory conditions—including stroke, brain health, complications of pregnancy, kidney disease, congenital heart disease,
rhythm disorders, sudden cardiac arrest, subclinical atherosclerosis, coronary heart disease, cardiomyopathy, heart failure, valvular disease, venous thromboembolism, and peripheral artery disease—the associated risk factors, including diabetes and obesity; and outcomes, including quality of care, procedures, and economic costs. Although many cardiometabolic conditions have overlapping biological mechanisms, some mechanisms are specific to a pathology. To summarize potential key factors that regulate the etiology of cardiometabolic conditions in women, this section highlights findings for the consensus on the more general role of key biological factors that affect women.
Sex Chromosome Effects
The contribution of the X chromosome to developing cardiometabolic conditions is not fully understood, largely because the majority of GWAS studies do not include sex chromosomes (Regitz-Zagrosek and Kararigas, 2017). However, studies in the FCG model have identified specific differences related to metabolism, with the XX, compared to the XY chromosome complement promoting fat accumulation related to increased food intake during the inactive phase of the diurnal cycle and metabolic inflexibility to switch between carbohydrate and fat fuels for energy metabolism (Chen et al., 2012, 2015; Link et al., 2020).
The XX chromosome complement in mice is also associated with elevated cholesterol levels and atherosclerosis (AlSiraj et al., 2019; Link et al., 2015), suggesting that the vascular protective effects for females comes from gonadal hormones rather than the X chromosome. This is consistent with studies showing that in female mice that have no estrogen effects after surgical removal of the ovaries, the X chromosome promoted cardiac injury from elevated expression of genes that promote inflammation, cell death, lipid oxidation, and generation of oxygen-derived free radicals (Brooks and Renaudineau, 2015; Li et al., 2014; Stamova et al., 2012). Together, these findings may provide an additional mechanism that explains how women are protected by estrogens early in life, but become susceptible to CVD, myocardial infarction, and stroke after menopause, resulting from the detrimental effects of the genes encoded on the X chromosome.
Identifying XX chromosome dosage as a risk factor for increased body fat and possibly atherosclerosis and CVD resulted in discovering candidate genes that may underlie this effect (Wiese et al., 2023). Prominent among these are genes that escape X chromosome inactivation and are thus expressed at higher levels in tissues of XX compared to XY individuals (Tukiainen et al., 2017). Two X chromosome genes, KDM5C and KDM6A, encoding histone demethylase enzymes that regulate chromatin accessibility and gene expression have been implicated in transcription of
genes influencing fat development and accumulation (Link et al., 2020) and sex-specific differences related to myocardial infarction remodeling (Li et al., 2014). Nevertheless, further research is needed to clarify how other X chromosome genes increase susceptibilities to cardiometabolic conditions in women.
The Effects of Estrogens
Estrogens play an important role in mediating vascular protective effects in women by promoting normal metabolic function, lowering systemic inflammation, and maintaining the integrity and function of the vascular endothelium—the inner lining of blood vessels—that helps prevent cardiometabolic conditions (Fried et al., 2015; Karastergiou et al., 2012; Mauvais-Jarvis, 2015; Palmer and Clegg, 2015). The role of estrogens start during early development, when they influence gene expression in the brain with lasting effects on behavior and metabolism, including the control of energy metabolism by the hypothalamus (Gegenhuber et al., 2022; Mauvais-Jarvis, 2014). Estrogens have also been shown in human and animal studies to influence appetite, with 17β-estradiol (E2) serving as the primary effector of estrogen signaling. Ovariectomized rats experiencing a sudden decline in estrogens had an increase in their food intake, while the administration of exogenous E2 normalized their feeding, presumably through its effects on appetite control centers in the hypothalamus (Rivera and Stincic, 2018). Estrogen’s anorexigenic effects have been observed in studies conducted in women, with research showing that food intake fluctuates across the menstrual cycle, decreasing most in the peri-ovulatory reproductive phase that coincides with rising estrogen levels (Buffenstein et al., 1995; Rogan and Black, 2023; Vigil et al., 2022). Further evidence is needed to support estrogen’s appetite suppressive effects and how this relates to changes in food intake behaviors during the menstrual cycle and after menopause.
In addition, estrogens act on adipose tissue to influence fat storage. Premenopausal women generally store a greater proportion of fat in subcutaneous depots, facilitating long-term energy storage and resistance to depletion during pregnancy and lactation. However, during perimenopause and after menopause, the decline in estrogens enable fat stores to switch from subcutaneous to visceral and abdominal locations, promoting abdominal obesity, which leads to higher risks of cholesterol and lipid imbalances, insulin resistance, Type 2 diabetes, and hypertension, all of which are associated with CVD (Lizcano and Guzmán, 2014). Estrogens are also a determinant of glucose homeostasis. Healthy premenopausal women demonstrate greater glucose uptake by skeletal muscle (Nuutila et al., 1995) and are more resistant than men to lipid-induced insulin resistance (Frias et al., 2001). However, insulin resistance increases with reduced or abnormally
high levels of estrogens, indicating an optimal range of levels of estrogens required for maintaining proper insulin sensitivity (Perseghin et al., 2001).
Estrogens also have protective effects on the vascular endothelium by preserving vascular tone and promoting anti-inflammatory endothelial responses. In ischemia-reperfusion experiments that model myocardial and cerebral infarction (Chen and Vunjak-Novakovic, 2018), estrogens have been linked to increases in markers indicative of endothelial viability (Chang et al., 2019; Simoncini et al., 2000; Veenema et al., 2019; Yang et al., 2000). Estrogens also activate signaling pathways that block programmed cell death and hypoxia (Jukes and Osawa, 1997; Lazaro et al., 2023; Wang et al., 2006, 2009). Although the majority of studies highlight the benefits of estrogens, others have noted that abnormal concentrations are detrimental (Smith et al., 2000; Zhan et al., 2008). For example, administering estrogens during the first week after a myocardial infarction in female rats without ovaries increases infarct size (Smith et al., 2000). This is line with studies that have shown physiological levels of estrogens improved cardiac physiology, reduced fibrosis, and increased capillary density, albeit with no significant effect on mortality (Zhan et al., 2008). Collectively, these findings are consistent with the notion that an optimal range of estrogens are needed for endothelial homeostasis and protection against injury.
Other Biological Factors and Mechanisms
Inflammation is also linked to CVD. Estrogens and ERs have cardioprotective effects via regulating multiple inflammatory pathways (Horng et al., 2017; Rosenzweig et al., 2021). In a mouse model of ischemic heart failure, levels of one specific ER decreased significantly during acute myocardial infarction but increased during heart failure (Rosenzweig et al., 2021). Estrogen signaling also has age-dependent anti-inflammatory effects. Women with an average age of 73 who developed heart failure after myocardial infarction had an increase in plasma proteins associated with macrophage-mediated production of reactive oxygen species compared to men, indicating that the protection from macrophage-mediated inflammation was lost after menopause (Deleon-Pennell et al., 2018). In a mouse model of myocardial infarction, male and female mice were found to produce specialized anti-inflammatory mediators, although female mice generated higher levels to coordinate infarct healing after myocardial infarction (Pullen et al., 2020).
Aside from estrogens, other ovarian hormones play important roles in regulating cardiometabolic and vascular function. Low levels of anti-Müllerian hormone, produced by ovarian follicles, and relaxin, secreted by the corpus luteum in the ovaries, have been implicated in maintaining cardiovascular health and are correlated with atherosclerotic plaque
accumulation (Lambrinoudaki et al., 2020; Verdiesen et al., 2022) and inflammation (Aragón-Herrera et al., 2022; Brecht et al., 2011). In addition, higher levels of the gonadotropin, follicular stimulating hormone, that increases during menopause, has been linked to increases in visceral adiposity (Liu et al., 2017), which contributes to elevated risks of CVD and stroke.
Experimental stroke models have been crucial in identifying therapeutic targets to improve drug design. Because of the complex nature of the disease, which involves the circulatory system, immune system, and endocrine secretions, multiple possible cellular and molecular targets exist. Basic science studies have shown that cell death pathways are effective targets (Liu et al., 2009), as are composite structures, such as the blood brain barrier (D’Souza et al., 2021; Okada et al., 2020), and cellular targets such as astrocytes (Sampath et al., 2023b). Stroke patients have severe gastrointestinal consequences in the acute phase, and the gut–brain axis has emerged as a new tractable target for stroke (Benakis et al., 2016; El-Hakim et al., 2021; Mani et al., 2023).
The Maternal in Utero Environment
Early-life programming (ELP) includes the idea of the maternal nutritional environment influencing the long-term health trajectory of individuals (Pannia et al., 2016). Exposure to hormonal, metabolic, nutritional, and other maternal factors during sensitive developmental stages affects the structure and function of cells and organs and influence the “programming” of health outcomes with lasting physiological and behavioral effects (Archives of Disease in Childhood, 1994; Barker, 2004; Eriksson, 2016; Warner and Ozanne, 2010). Animal models of ELP show that a maternal high-fat diet led to obesity in offspring later in life (Férézou-Viala et al., 2007). Clinical studies have also implicated elevated maternal fat stores to higher risks of metabolic conditions in offspring later in life. For example, infants born to mothers who are obese exhibit higher risks of obesity, CVD, and allergic conditions (Fleming et al., 2018; Van De Maele et al., 2018). In addition, infants born to women diagnosed with gestational diabetes have a greater risk of Type 2 diabetes, a well-known risk factor for coronary heart disease (Eriksson, 2016). Many experimental models of developmental programming report a sex difference in blood pressure, with males exhibiting a significant increase in young adulthood (Alexander, 2003; Bourque et al., 2013; Ortiz et al., 2001). Birthweight is a risk factor for hypertension and CVD, but its clinical significance is not yet considered when managing cardiovascular health. Overall, early-life exposure factors during critical developmental windows may affect lifetime risks for chronic conditions (Chan et al., 2020; Jašarević and Bale, 2019; Morgan et al., 2019; Morrison et al., 2020).
Specific Risk Factors in Women
Reproductive Milestones
Women’s reproductive milestones have been substantially studied in clinical, longitudinal, and population-based epidemiological studies that span menarche, the reproductive window, pregnancy, postpartum, and menopause with respect to risk for cardiometabolic conditions (O’Kelly et al., 2022). The key roles sex hormones play in CVD development and progression in women have been uniformly understood among clinicians and well studied in the research literature. These factors have been included in epidemiological studies, such as the Nurses’ Health Study, Coronary Artery Risk Development in Young Adults, and SWAN. Key reproductive milestones are described next.
Age at Menarche
Age of menarche has been associated with metabolic syndrome and CVD (Charalampopoulos et al., 2014; Lee et al., 2019). An analysis of 12 cohort studies found that every 1 year increase in age of menarche reduces all-cause mortality by 3 percent, including mortality from CVD, ischemic heart disease, and stroke (Chen et al., 2019). A U-shaped association exists between age of menarche and stroke, with increased risk at early and late menarche (Mishra et al., 2020). The mechanisms for why early menarche may lead to increased risk of cardiometabolic conditions is unclear, and older studies generally attributed it to lifestyle or genetic factors in young girls, such as increased body mass index (BMI), blood pressure, and cholesterol and decreased physical activity. However, age at menarche should be considered as a potential screening tool for risk of cardiometabolic conditions and adverse events associated with CVD and stroke (Lee et al., 2019).
Reproductive Window and Life-Span
During the reproductive window, fluctuations in the menstrual cycle may affect chronic conditions that lead to cardiometabolic conditions. Having a shorter reproductive life-span (the number of years between menarche and menopause), has been associated with an increased risk of CVD in midlife and specifically for women who experienced early age of menarche (Mishra et al., 2020). Menstrual cycle irregularity across the reproductive life-span is known to be associated with CVD risk (O’Kelly et al., 2022). The number of children birthed has also been linked to CVD and stroke risk (D’Errico et al., 2024e; Oliver-Williams et al., 2019; Vladutiu et al., 2017).
Polycystic ovarian syndrome (PCOS) is a gynecological condition affecting women of reproductive age that has a strong association between sex steroids and insulin resistance; at least half of women with PCOS are insulin resistant despite not being obese or overweight (Dunaif et al., 1989) and at a greater risk of hypertension, abnormal lipid levels, and metabolic syndrome (Wild et al., 2010). Despite a greater prevalence of CVD risk factors in PCOS, research has not identified a higher cardiovascular event risk or mortality rate because of study size limitations.
Pregnancy and complications
Adverse pregnancy outcomes, including hypertensive disorders of pregnancy, gestational diabetes, preterm delivery, and intrauterine growth restriction, and absence of breastfeeding are all associated with increased CVD and stroke risk (Miller et al., 2022; O’Kelly et al., 2022; Staff et al., 2024). Women who develop gestational diabetes are reported to have an increased risk of developing Type 2 diabetes later in life. Gestational diabetes is also a major CVD risk factor and is associated with other CVD risk factors, including hypertension and unfavorable changes in cholesterol and triglyceride levels (Rivero et al., 2008). Studies are needed to determine whether early interventions and CVD risk assessment during the reproductive period may reduce CVD risk at midlife.
Hypertension during pregnancy, which can be preeclampsia, eclampsia, chronic hypertension, or gestational hypertension, is associated with maternal stroke (Braunthal and Brateanu, 2019) and stroke later in life (Theilen et al., 2016). Moreover, women who develop hypertension during pregnancy have a higher mortality risk (Theilen et al., 2016), and an approximately fivefold higher odds for future stroke relative to women without this condition (Crovetto et al., 2013; Leffert et al., 2015). Disorders of pregnancy, including preeclampsia, are also associated with increased odds of stroke through midlife (Miller et al., 2022, 2023). In the Population Assessment of Tobacco and Health study, adverse pregnancy outcomes were an independent predictor of later stroke (Manrique-Acevedo et al., 2020; Miller et al., 2022; Miller et al., 2023).
Among women who have given birth, both ever having breastfed and longer duration of breastfeeding is associated with lower risk of stroke, with some evidence that these associations vary by both age and race and ethnicity (Jacobson et al., 2018; Richardson et al., 2022).
Menopausal transition
Research has shown that changes in cardiometabolic health that lead to metabolic disorders accelerate during the menopause transition and have a
significant effect on CVD risk that are independent of chronological aging (El Khoudary et al., 2020). Menopause triggers a dramatic shift in the risk profile for stroke in women, with the decline of estrogens and other female sex steroids thought to be neuroprotective regarding ischemic stroke.
Midlife and menopausal transition are times when women accumulate comorbidities, and cardiometabolic health often worsens, marked by unfavorable changes in lipid profiles, abdominal fat stores, higher risk of diabetes and metabolic syndrome, and endovascular dysfunction (Leeners et al., 2017; Ley et al., 2017; Moreau and Hildreth, 2014; Subramanya et al., 2018; Sun et al., 2019; Thurston et al., 2018). Although research suggests that these issues result from the physiologic changes occurring during the menopausal transition, rather than chronological aging, more data is needed on reproductive aging and how this affects risk of stroke and CVD.
Early menopause is common in women, and those who have experienced early, natural menopause have higher risks of developing Type 2 diabetes (Brand et al., 2013) and CVD mortality, heart failure, and all-cause mortality (Ley et al., 2017). Surgically induced menopause also increases the risk of Type 2 diabetes according to several cohort studies (Honigberg et al., 2019; Rocca et al., 2017). Removal of both ovaries is a common cause of premature or early menopause, and cohort studies have identified an increased risk of CVD outcomes in women who had both ovaries removed before age 45 compared to women who did not (Honigberg et al., 2019; Parker et al., 2009; Rocca et al., 2017). Another cohort study showed steeper increases in weight and BMI in women who had both ovaries removed before age 46 and did not receive estrogen therapy (Gibson et al., 2013; McCarthy et al., 2013). Both premature and early menopause are associated with increased risk of stroke, though this is primarily associated with ischemic stroke and varies between natural and surgical menopause (Lisabeth et al., 2009a; Welten et al., 2021). As surgical removal of the ovaries and fallopian tubes is increasingly being used to reduce risks of genetic forms of breast and ovarian cancers (Daly et al., 2021), appropriate timing for this procedure in order to maximize benefits while minimizing risks needs further investigation (Bertozzi et al., 2023). Clinical guidance for early menopause is lacking and needed for this population (Mishra et al., 2024a).
Clinical Guidelines for CVD and Stroke in Women
Basic science and epidemiological studies have identified risk factors for CVD, stroke, and cardiometabolic conditions, such as obesity, metabolic syndrome, and Type 2 diabetes. Researchers have incorporated these risk factors, along with lifestyle risk factors for CVD and stroke that have distinct gender differences, into the Predicting Risk of CVD Events (PREVENT) calculator (Larkin, 2024).
Clinical guidelines and recommendations for preventing CVD in women have existed since 1999 and emphasize that risk factor management is different for women and men (Mosca et al., 1999). There has been increased recognition that women should have unique recommendations given the evident sex and gender differences in preventive factors that are often overlooked (Mosca et al., 2011). The American Heart Association (AHA) updated its evidence-based guidelines, shifting from pure clinical research to focusing comprehensively on primary prevention of CVD and stroke in women as observed in clinical practice (Mosca et al., 2011). Emerging nontraditional risk factors for women in the 2011 AHA guidelines included preterm delivery, hypertensive pregnancy disorders, gestational diabetes, breast cancer treatments, autoimmune conditions, and depression (Garcia et al., 2016). The updated guidelines have included the “essential eight” risk factors, adding sleep to the list, but they do not contain guidelines specifically for women (Lloyd-Jones et al., 2022).
Clinical guidelines to prevent stroke in women were established in 2014. They highlight female-specific risk factors, such as pregnancy, preeclampsia, and migraines, and factors with a more pronounced effect in women, such as migraine with aura, atrial fibrillation, diabetes, hypertension. The guidelines also address factors with similar prevalence in men and women, such as physical activity, smoking, metabolic syndrome, and diet (Bushnell et al., 2014), for which the differential effects on male versus female risk are unknown. The 2021 guidelines for the prevention of stroke from the AHA and the American Stroke Association outline the continued importance of managing vascular risk factors, including diabetes, smoking, hypertension, and atrial fibrillation, and adopting healthy lifestyle behaviors known to lower risk of stroke, such as a low-salt/Mediterranean diet, exercise, avoidance of prolonged sedentary behavior, and adherence to medication (Kleindorfer et al., 2021). New to these guidelines is the recommendation for patients to receive a “diagnostic workup” after stroke, to identify the etiological cause, and appropriate post-stroke treatment to lower the risk of subsequent stroke (Kleindorfer et al., 2021). Sex and gender considerations were also highlighted; alcohol intake of greater than one drink per day in women and two drinks per day in men should be lowered or altogether prohibited to reduce stroke risk; women with history of gestational diabetes should have their blood glucose levels monitored to lower risk for ischemic stroke; and women with migraine with aura who use estrogen-containing oral contraceptives, particularly smokers, should consider avoiding them and if they have had a previous stroke. Special approaches that address the underlying determinants of inequities in stroke risk, particularly in women, people of color, immigrants, rural populations, older adults, and sexual minority groups, was also emphasized to reduce risk (Kleindorfer et al., 2021).
Other Risk Factors
CVD and stroke risk factors that are disproportionately more severe in women include hypertension, atrial fibrillation, and diabetes (Rexrode et al., 2022). Women have a higher risk of stroke at a lower threshold for systolic blood pressure than men (Howard et al., 2019; Ji et al., 2021; Peters et al., 2020). Atrial fibrillation is the most common type of arrhythmia, and women with it are more likely to have more severe symptoms (Ball et al., 2013), with a greater associated risk of stroke and death (Bushnell et al., 2014; Emdin et al., 2016). Diabetes is a major risk factor for stroke and may be more significant in women (Peters et al., 2014). Women aged 55–64 and who have Type 2 diabetes are significantly more likely to have a stroke than are men (1.5–6-fold vs. 1.5–2-fold) (Almdal et al., 2004). Research is needed to understand the biological mechanisms of why diabetes presents a higher risk of stroke in women and whether this is linked to menopause, as mentioned (Madsen et al., 2021; Peters et al., 2020).
Condition Presentation and Diagnosis
CVD
Emerging evidence from the last 2 decades supports distinct differences in the clinical presentation of CVD in women (Vogel et al., 2021). Compared to men, who typically exhibit features of obstructive coronary artery disease,4 women are more likely to experience atypical symptoms, such as epigastric symptoms, shoulder, jaw and/or neck pain, and palpitations (Lichtman et al., 2018). The cardioprotective effects of estrogens most likely explain the lower prevalence of obstructive coronary artery disease in premenopausal women (Mehta et al., 2016). In women with it, atherosclerotic lesions may be smaller as a result of positive remodeling of coronary arteries, which obscures atherosclerosis (Bairey Merz et al., 2006; Pacheco Claudio et al., 2018). In the Women’s Ischemia Syndrome Evaluation (WISE) study, 79 percent of women with nonobstructive coronary artery disease showed signs of coronary atherosclerosis with positive remodeling (Khuddus et al., 2010). Distinct pathophysiological mechanisms govern atherosclerosis in men and women, with women more often developing features of plaque erosion and men more commonly having disruptions of atherosclerotic plaque (Pacheco et al., 2022b; Pacheco Claudio et al., 2018; Pasupathy et al., 2015).
Although similar proportions of women and men aged 18–55 present with angina, or chest pain, upon diagnosis of acute myocardial infarction
___________________
4Obstructive coronary artery disease is defined as a greater than 50 percent blockage of the coronary arteries.
(Lichtman et al., 2018), women may present additional symptoms and early signs that appear to be distinct. These include angina with absence of coronary artery obstruction; discomfort or pain in the jaw, neck, arms, shoulder or back; weakness; fatigue; difficulty breathing; nausea; indigestion; anxiety; arm weakness or heaviness; cold sweats; and sleep disturbances (Brush et al., 2020; Khan et al., 2017; Pacheco et al., 2022b). Although these differences in clinical presentation have been reported in women for many decades, clinicians still misdiagnose CVD or dismiss symptoms in women (Gendered Innovations, 2023), and only in recent years has recognition increased of cardiovascular syndromes that preferentially affect women (Tamis-Holland et al., 2019). The following section provides an overview of recent findings related to three cardiac conditions primarily observed in women—ischemia with no obstructive coronary artery disease (INOCA), myocardial infarction with no obstructive coronary artery disease (MINOCA), and spontaneous coronary artery dissection (SCAD)—and stroke.
INOCA
Nearly half of all women experiencing angina and undergoing invasive coronary angiography with symptoms and/or signs of ischemia have INOCA (Ford and Berry, 2019), with around two-thirds of angiograms from women with suspected ischemia lacking signs of obstructive coronary artery disease (Merz et al., 2017). INOCA is more prevalent in women, particularly during midlife (Merz et al., 2017). Estimates of INOCA derived from clinical studies vary depending on the study population, inclusion criteria, and diagnostic method. For example, 62 percent versus 34.4 percent of women enrolled in the WISE study and the International Study of Comparative Health Effectiveness with Medical and Invasive Approaches trial, respectively, had no obstructive coronary artery disease (Reynolds et al., 2020; Shaw et al., 2006).
INOCA is not a benign condition, as it is associated with an increase in major adverse cardiovascular outcomes and a significant contributor to CVD mortality in women (Pacheco Claudio et al., 2018). The WISE study identified an elevated risk in all-cause mortality in women with INOCA compared to age-matched control subjects (Kenkre et al., 2017). Considering the morbidity and mortality risks, it is important for both patients and clinicians to be aware of the clinical significance of unconventional CVD symptoms in women.
Angina is a common symptom of INOCA that may be missed by anatomical coronary artery tests alone (Ford and Berry, 2019). Typically, invasive coronary angiography is the standard tool for identifying obstructive coronary artery disease, and its absence in patients presenting with angina may indicate INOCA (Ford and Berry, 2019). It is unclear whether INOCA is a precursor to angiographic stenosis or unrelated (Barsky et al., 2020).
Approximately half of women with INOCA also demonstrate coronary microvascular dysfunction, an abnormal constriction of the heart’s small blood vessels, detected by invasive coronary reactivity testing (Pacheco Claudio et al., 2018). To diagnose this, clinicians typically employ invasive intracoronary measurements or noninvasive positron emission tomography (PET) to assess abnormal coronary flow reserve or coronary blood flow (AlBadri et al., 2019; Merz et al., 2017). Most women with INOCA exhibit diffuse nonobstructive coronary atherosclerosis, likely caused by inflammatory processes (Merz et al., 2017).
The etiological mechanisms of INOCA are numerous and include vascular endothelial dysfunction, inflammation, smooth muscle cell dysfunction, remodeling of small blood vessels, and activation of the sympathetic nervous system. Other symptoms indicating decreased oxygen supply have been observed with INOCA, such as aortic stenosis, severe hypertension, and anemia (Pacheco Claudio et al., 2018). Physicians have used beta-blockers, calcium channel blockers, nitrates, and ranolazine to alleviate INOCA symptoms (Kunadian et al., 2020).
Despite a high prevalence of INOCA in women, there is a lack of clarity, standardization, and uniformity in diagnosis and management. Consequently, women are often discharged with the inaccurate diagnosis of non-cardiac chest pain and repeatedly hospitalized (Ford and Berry, 2019; Pacheco et al., 2022a). No standard definition or a set of unifying consensus guidelines exist regarding diagnostic evaluation and treatment strategies. In a published survey, most cardiologists believed that additional clinical guidance is needed to accurately diagnose and treat patients with INOCA, with over half of them wanting U.S.-specific guidelines or expert consensus for diagnosis and management (Pacheco et al., 2022a). As a result of the knowledge gaps, approximately one in three cardiologists in early to mid-career report being neutral or lacking confidence in their ability to manage patients with INOCA (Luu et al., 2022).
MINOCA
MINOCA is defined as a myocardial infarction attributed to atherosclerotic or non-atherosclerotic causes, with presentation of ischemic symptoms and lesions that occupy less than 50 percent of the epicardial coronary arterial space (Herling de Oliveira et al., 2022; Tamis-Holland et al., 2019). The mortality is approximately 4.7 percent per year (Pacheco Claudio et al., 2018; Pasupathy et al., 2015). MINOCA accounts for between 6–15 percent of all heart attacks (Reynolds et al., 2021), and occurs in 10.5 percent of women compared to 3.4 percent in men (Smilowitz et al., 2017). MINOCA also affects patients at younger ages and who identify as African American or Black (Pacheco Claudio et al., 2018; Pasupathy et al., 2015). A study
using coronary optical coherence tomography and cardiac MRI has shown that MINOCA was the underlying cause in 56 percent of women diagnosed with a heart attack. Within this group, ischemia was more than three times more common than non-ischemia (Reynolds et al., 2021). As several mechanisms may lead to MINOCA, including plaque disruption, coronary vasospasm, coronary microvascular dysfunction, SCAD, clotting disorders, blood clots, cardiomyopathies and myocarditis (Hayes et al., 2018; Reynolds et al., 2021; Tamis-Holland et al., 2019), it serves as a working diagnosis with many possible causes that requires further evaluation of symptoms to administer appropriate therapies (Pasupathy et al., 2015).
SCAD
SCAD is another underdiagnosed and poorly recognized acute coronary syndrome that primarily affects women (Méndez-Eirín et al., 2021). SCAD-triggered myocardial infarction is spontaneous and nonatherosclerotic and is caused by lack of myocardial blood flow resulting from partial or complete occlusion of the coronary artery following a vessel-wall tear or hematoma (Tweet et al., 2018). It occurs frequently in midlife women, with the average age ranging from 45 to 53 years old (Hayes et al., 2018). SCAD shares symptoms with other acute coronary syndrome conditions, and risks include myocardial ischemia, arrhythmias, and sudden cardiac death (Saw et al., 2014a). It accounts for approximately 35 percent of acute myocardial infarctions in women under 50 and is the main cause in women under 40 (Tweet et al., 2018). SCAD is also the most common cause of myocardial infarction associated with pregnancy (Tweet et al., 2017), which is more serious than SCAD in non-pregnant women and can lead to impaired heart function (Tweet et al., 2017).
Diagnosing SCAD continues to be challenging because of limited research on its causes and natural history. No biomarker reliably differentiates SCAD from atherosclerosis (Adlam et al., 2021). Noninvasive imaging, such as computed tomographic coronary angiography, is not recommended routinely because of its limitation in assessing regions of the coronary arteries that SCAD frequently affects, so clinicians use invasive coronary angiography (Adlam et al., 2021). In some confusing circumstances, non-SCAD presentations mimic the angiographic features usually associated with SCAD, which may lead to an inaccurate diagnosis (Adlam et al., 2021).
Stroke
Sex differences in symptom presentation of acute stroke or transient ischemic attack (also known as “minor stroke”) are not well understood
and may underlie issues of misdiagnosis or missed diagnosis in women (Ali et al., 2022). Often, clinical symptoms of stroke presented by women may be mistaken for other conditions, like a migraine attack (Ali et al., 2022). Two recent meta-analyses indicate that, compared to men, women more frequently present with non-localized symptoms, including changes in consciousness or mental status, headache including migraine, and generalized weakness (Ali et al., 2022; Shajahan et al., 2023). However, these two studies differed in their conclusions related to sex differences in focal symptoms. Based on an analysis of 60 studies, the first review indicated that dizziness and speech slurring (with or without nausea/vomiting) occurred more commonly in women, while muscle weakness and visual aberrations were more often experienced by men (Ali et al., 2022). Another review based on 21 studies showed that men more commonly presented with speech slurring and had higher risk of dizziness (Shajahan et al., 2023). These discrepancies and the general lack of high-quality studies warrant further research to distinguish differences in stroke presentation between men and women (Ali et al., 2022), which is a crucial factor in the early recognition and management (Shajahan et al., 2023).
Women are also more likely to arrive at the hospital later than men, which likely excludes more women from clot-busting treatment with tissue plasminogen activator (tPA) (Madsen et al., 2016), and less likely to be conscious upon arrival, which suggests they are experiencing larger strokes and a longer duration of symptoms (Glader et al., 2003). Moreover, the greater prevalence of poorly understood symptoms in women may lead to diagnostic delays and missed treatment opportunities (Lisabeth et al., 2009b).
Treatment and Management
INOCA AND MINOCA
Studies have shown that specialized women’s heart centers improve outcomes in women with INOCA and MINOCA. A prospective study showed that evaluating women at these centers led to more specific diagnoses of INOCA and MINOCA that resulted in therapeutic changes that improved control of CVD risk factors, including blood pressure, total cholesterol, and low-density lipoprotein (LDL) cholesterol; decreased chest pain; and elevated the quality of life after 1 year (Parvand et al., 2022). Specialized women’s heart centers are an important resource for patients and clinicians, providing diagnostic tools, risk stratification, and management relevant to women’s heart health, as well as promoting awareness, education, and multidisciplinary collaboration (Parvand et al., 2022). They may serve as a long-term, sustainable model that can address disparities in cardiovascular risk factors and CVD in women (Pacheco et al., 2022a).
SCAD
The decision to proceed with percutaneous coronary intervention or coronary artery bypass surgery differs widely among clinicians and depends on the acute presentation (Saw et al., 2014a). A conservative approach is typically recommended given no ongoing ischemia, recurrent chest pains, hemodynamic instability, or left main arterial dissection (Saw et al., 2014a). However, this is not based on high-quality randomized data, and the acute and long-term outcomes of a conservative or revascularization approach remain unclear (Saw et al., 2014a).
Pharmacological Treatment for CVD, Stroke, and Risk Factors
Access to treatment and prescribing practices for cardiometabolic conditions differ by gender. Anti-coagulation medication, such as warfarin, is critical for preventing strokes, but clinicians are less likely to prescribe it for women despite showing equal effectiveness in women and rates of similar risk of intracranial hemorrhage between men and women (Rexrode et al., 2022). New oral anticoagulants (e.g., dabigatran) appear to be more efficacious than warfarin, but women are more likely to fill prescriptions for lower doses compared to men (Tsadok et al., 2015). Women are less likely to be prescribed beta-blockers or receive an implantable cardioverter-defibrillator (Butzner et al., 2022). In addition, research shows that women are less consistently prescribed statins, angiotensin-converting enzyme inhibitors, and beta-blockers for managing Type 2 diabetes and the concomitant risk of CVD (Vimalananda et al., 2013; Zhao et al., 2020). Many women with history of Type 2 diabetes and moderate to high CVD risk meet the criteria to initiate statin therapy according to AHA/American College of Cardiology guidelines (Stone et al., 2014). However, clinicians may address CVD risk factors less aggressively in women than men with Type 2 diabetes (Regensteiner et al., 2015). A retrospective cohort study of diabetic women veterans showed that they had lower rates of achieving a target low density lipo-protein (LDL) level than men (Vimalananda et al., 2013). This may be because of no prospective studies in women regarding primary CVD management with statins and no recommendation based on expert opinion for this as a primary prevention approach.
One FDA-approved drug for stroke exists, the clot-busting agent tissue plasminogen activator (tPA). The standard treatment regimen for acute ischemic stroke is to administer tPA within 4.5 hours from symptom onset, which consequently restricts the number of patients eligible to receive treatment (de Los Ríos la Rosa et al., 2012; Schwamm et al., 2013). However, recent studies have shown that the use of imaging tools, such as computed tomographic perfusion or MRI, may give better indications of stroke lesion
progression and could thus be used to identify patients who may benefit from tPA treatment beyond the 4.5-hour window (Broocks et al., 2023; Ma et al., 2019). Further clinical trials are needed to strengthen the evidence base that imaging modalities can help determine if tPA treatment can be safely and effectively given to stroke patients even after 4.5 hours from symptom onset (Ma et al., 2019).
Generally, women are less likely to receive tPA at primary and non-primary stroke centers regardless of stroke presentation and after adjusting for age, socioeconomic class, location, and hospital (Boehme et al., 2017; Kent et al., 2005; Reeves et al., 2009a, 2009b; Reid et al., 2008; Strong et al., 2020). A recent meta-analysis of 24 studies showed that women had 13 percent lower odds of being treated with tPA compared to men; however, disparities in tPA treatment were not consistently observed across different regions (Rexrode et al., 2022). Data from the Get with The Guidelines Stroke registry5, an online, interactive assessment and reporting system, showed that women who arrive at the hospital within 2 hours of symptom onset of acute ischemic stroke were less likely to receive tPA than men (55.8 vs 59.4 percent) (Reeves et al., 2009b). A later retrospective analysis of Get with The Guidelines Stroke data captured between 2003 and 2011 showed that approximately 25 percent of patients presenting with acute ischemic stroke symptoms within 2 hours of onset failed to receive tPA treatment. Based on their findings, researchers suggested that women, older adults, and racial minorities are undertreated despite being eligible for tPA treatment (Messé et al., 2016).
Experimental stroke models have also successfully modeled long-term neuropsychiatric conditions commonly seen in survivors. Poststroke depression is more common in women (Lyu et al., 2022; Poynter et al., 2009), and conventional antidepressants are not effective (Frank et al., 2022; Sun et al., 2017). More than half of stroke patients experience some level of cognitive impairment by 6 months afterward, and ischemic stroke is a leading cause of vascular dementia (Mellon et al., 2015; Vijayan and Reddy, 2016) and AD (Elman-Shina and Efrati, 2022; Filippenkov et al., 2023). Poststroke cognitive impairment is more likely to affect women than men (Pendlebury and Rothwell, 2009), and women are more likely to require care in an assisted living facility because of deteriorating health conditions that include cognitive impairment, frailty, pain, and depression (Yu et al., 2020). Vascular dementia is also worse in women compared to men (Gannon et al., 2022). Experimental stroke models have replicated post-stroke cognitive
___________________
5Additional information available at https://www.heart.org/en/professional/quality-improvement/get-with-the-guidelines/get-with-the-guidelines-stroke/get-with-the-guidelines-stroke-registry-tool
impairment in males and females (Mani et al., 2023; Sampath et al., 2023a; Ward et al., 2018) and aging females (Panta et al., 2020).
Disparities
Research on racial and ethnic disparities regarding factors associated with cardiometabolic conditions is increasing. For example, investigators have examined whether lifestyle modifications and interventions can decrease the risk of morbidity in minoritized populations. In addition to accounting for BMI and central fat stores, researchers have studied how psychosocial and lifestyle factors affect disparities in the incidence of conditions such as diabetes, CVD, and stroke.
Data from the NHIS show that Black women experience higher rates of all the major risk factors associated with cardiometabolic conditions, including diabetes, hypertension, and obesity (Chinn et al., 2021). Compared with White women, Black women had lower incomes and lower rates of education, two social factors that significantly affect stroke risk and attenuated their higher risk (Jiménez et al., 2019). In the Reasons for Geographic and Racial Differences in Stroke study, achieving a higher ideal cardiovascular health score that considers lifestyle behaviors and modifications decreased the risk of diabetes among adults with normal glucose levels, with a stronger association among Black than White adults (Joseph et al., 2019). In a prospective cohort study, the cumulative incidence was greater for Black than for White men and women (Carson et al., 2021). After adjusting for age and prediabetes, this same study found that diabetes affected Black women more than White women (Carson et al., 2021); individual risk factors, such as BMI and waist circumference attenuated the racial disparity (Carson et al., 2021).
As noted, effective treatment of stroke is time sensitive. Studies have identified inequities in administering clot-busting treatments, indicating that Black individuals have less access to key treatments in the acute time window (Otite et al., 2021). Furthermore, data indicate that Black women are disproportionally affected by treatment inequities (Boehme et al., 2014).
CVD significantly affects Hispanic/Latina women. The Hispanic Community Health Study/Study of Latinos found that Hispanic/Latina women face higher than average rates of obesity, elevated cholesterol levels, hypertension, and disorders during pregnancy, such as preeclampsia and gestational diabetes (Gomez et al., 2022; Rodriguez et al., 2021). In addition, these rates differed by background, with higher rates among Cuban, Dominican, and Puerto Rican compared to Mexican American women. Hispanic/Latina women also experience higher rates of heart failure than non-Hispanic White women (Tsao et al., 2023). They were less likely to be aware of having these conditions and to have them under control,
which may result from cultural, socioeconomic, geographic, and linguistic barriers that affect access to care (Gomez et al., 2022).
Studies of risk factors for CVD among Hispanic/Latina women found that workplace exposure to environmental toxins, such as solvent, pesticide, and metal, was associated with greater levels of CVD (Bulka et al., 2019; Gomez et al., 2022). Research has also shown that immigration-related factors, such as level of acculturation, are associated with access to care and engaging in health-promoting behaviors, with Hispanic/Latina women engaging in lower levels of physical activity (Divney et al., 2019).
Despite established clinical guidelines for treating CVD, these have not been validated in Hispanic/Latina women. The AHA/American College of Cardiology Pooled Cohort Equations risk calculator has only been validated in non-Hispanic White and non-Hispanic African American men. These tools may lead to inaccurate and unreliable risk calculations in Hispanic populations (Gomez et al., 2022). Interventions for preventing CVD and addressing associated risk factors that target both youth and adult Hispanic/Latina women are needed, given that obesity rates are high among youth and adults (L’Engle et al., 2023).
Based on limited studies, American Indian and Alaska Native men exhibit higher morbidity and mortality from chronic conditions such as CVD and obesity, and more risk factors, such as smoking, whereas American Indian and Alaska Native women reported higher rates of diabetes (Breathett et al., 2020). To describe the various factors that affect the development of chronic conditions in American Indian and Native Alaska populations, researchers have used the Framework of Historical Oppression, Resilience, and Transcendence (Burnette and Figley, 2016; Burnette et al., 2019; McKinley et al., 2019). One study examining sex differences in various behavioral, physical, and psychological outcomes associated with CVD focused on how factors associated with oppression and trauma affect CVD risk for American Indian and Alaska Native women (Burnette et al., 2020), but more research is needed to understand gender differences in risk factors.
Data disaggregation has remained an issue in identifying cardiometabolic disease disparities among groups of Asian American and Pacific Islander women in risk factors for CVD and stroke (Đoàn et al., 2021). Asian American women have reported lower prevalence of diabetes and being overweight and higher rates of hypertension than men (Đoàn et al., 2021). Among groups of Asian American women, Asian Indian women reported the highest prevalence of being overweight or obese, and Filipino women had the highest prevalence of hypertension (Đoàn et al., 2021). One study found that Asian Indian, Chinese, Filipino, and Korean women had lower levels of high-density lipoprotein and higher triglyceride levels compared to non-Hispanic White individuals (Kwan et al., 2023).
Native Hawaiian and Pacific Islander women report greater prevalence of obesity and hypertension than Native Hawaiian and Pacific Islander men (Đoàn et al., 2021). Studies have found that mortality from CVD and ischemic stroke are higher in Asian American women and men compared to non-Hispanic White individuals (Đoàn et al., 2021). Data from the National Center for Health Statistics have shown that the standardized mortality rate for ischemic heart disease was higher in Asian Indian women, whereas the highest cerebrovascular disease was highest among Asian Vietnamese women compared to other Asian groups and non-Hispanic White groups of men and women (Shah et al., 2022).
In Asian Americans, the level of acculturation is a key factor researchers have studied, but few studies have disaggregated the data by sex. In addition, findings have been mixed on whether living in the U.S. for longer periods was associated with cardiometabolic conditions (Min et al., 2022). Cohort studies in the Asian American community that examined lifestyle factors include Mediators of Atherosclerosis in South Asians Living in America (MASALA) and Multi-Ethnic Study of Atherosclerosis, which only included Chinese Americans (Kanaya et al., 2014; Rodriguez et al., 2020). A study using data from MASALA and focusing predominantly on immigrant women found that self-reported menopausal status was associated with BMI and subcutaneous fat area and increased odds of hypertension (Vijay et al., 2022).
Research Gaps
The prevalence of obesity and metabolic syndrome is increasing in women, with distinct metabolic features compared to similar conditions in men. Understanding the molecular basis for the female-biased propensity to store fat is critical for both prevention and effective use of treatments to combat obesity. Preclinical models will play an instrumental role in achieving this goal. New insights will come from single-cell and spatial transcriptomic approaches to characterize sex differences in adipose tissue structure and function. Recent data on the role of certain X escape genes highlight the important role of epigenetic regulation as a determinant of sex differences and warrant further investigation.
Efforts to determine how gonadal hormones influence fat stores will benefit from studying gonadal hormone ablation in preclinical models throughout the lifespan, particularly in mice at ages similar to the age of menopause in women. Human studies of new diabetes and obesity drugs, such as Ozempic and Wegovy, and sodium glucose cotransporter 2 inhibitors (e.g., Jardiance) should analyze the effects on weight loss and long-term outcomes after stratification by sex/gender, which researchers often fail to do.
Tools to enable early and accurate diagnosis of CVD in women are needed. Myocardial infarction, stroke, and heart failure affect women,
particularly women of color, at a younger age, causing death and disability earlier in the lifetime, and it is important to diagnose CVD in the populations experiencing disparities in diagnosis (Ivey et al., 2022). Knowledge is lacking that would enable accurate diagnosis of the pathophysiological mechanisms of heart disease in women. Improving risk stratification and treatment for women will require differentiating their presentations of coronary artery disease (Pacheco et al., 2022b).
INOCA also lacks standardized diagnostic criteria, with no consensus on how to diagnose INOCA and MINOCA. Establishing diagnostic codes for both would improve treatment management, enable research, and inform policies affecting medical practice (Pacheco et al., 2022a). More research is needed to clarify the mechanisms leading to INOCA and MINOCA in women (Vogel et al., 2021), and more specialized women’s heart centers should be established, as it has been shown that they improve outcomes for both conditions (Parvand et al., 2022).
A knowledge gap continues to exist for clinicians managing SCAD, with a lack of consensus guidelines on evaluation and treatment. In addition, outcomes from conservative therapy and the extent of patient revascularization have been understudied (Saw et al., 2014b).
Disaggregated data are lacking regarding Hispanic, Latina/Latino, and Asian American, Native Hawaiian, and Pacific Islander people, which can mask differences in certain groups that may be more affected by CVD. Furthermore, national data on these populations underreport CVD prevalence and mortality and related deaths as a result of small sample sizes and issues with racial misclassification (Mehta et al., 2023). Conditions such as atrial fibrillation and ventricular arrhythmia are understudied in Hispanic women and Latinas, and more studies are needed to understand these conditions in women and validate diagnostic criteria in these populations.
MULTIPLE SCLEROSIS
“. . . I realize, of course, when I get up, the legs aren’t working quite the way they did . . . Sometimes I use my walker to get to the bathroom or my cane to get around, but then after that reality check, I still try to figure out every single day what it is that I’m able to do and then to try to live my best life around that.”
—Presenter at Committee Open Session
MS is an autoimmune disease6 that causes brain and spinal cord demyelination, axonal damage, and neuronal loss (Gold and Voskuhl, 2016).
___________________
6Systemic lupus erythematosus (SLE) is not discussed in this chapter. The report, Enhancing NIH Research on Autoimmune Disease provides information on other autoimmune diseases including those that predominately impact or affect women differently and have multi-systemic involvement (NASEM, 2022).
Depending on the brain regions affected, diverse clinical symptoms occur, including motor and sensory deficits, urinary incontinence, and cognitive impairment (Voskuhl, 2020). MS is approximately three times more common in women than men, but disease progression appears to be faster in men (Dunn et al., 2015; Voskuhl, 2020). Women with MS experience 70 percent lower rates of relapse of symptoms during later stages of pregnancy, suggesting that pregnancy hormones may alleviate the disease (Gold and Voskuhl, 2016). Both the lower incidence in men and the alleviation during pregnancy represent larger protective effects for MS than are achieved by any conventional therapy. Thus, the clinical observations provide strong rationale for investigating how sex-biasing factors increase or decrease disease incidence and progression and suggest that the effects of sex-related variables are complex, providing relative protection to one sex or the other by acting on different disease mechanisms or phases.
The primary view of MS is that myelin-activated T helper cells expand in the periphery, enter the brain, and elicit the secretion of proinflammatory cytokines. In turn, other immune system cells, such as macrophages, enter the CNS and contribute to demyelination and neuronal injury (Dunn et al., 2015; Petermann and Korn, 2011; Prat and Martin, 2002). In the brain, response to an autoimmune attack involves microglia, oligodendrocytes, astrocytes, and neurons. Evidence suggests that several sex-biasing factors may influence MS via actions on diverse cell types, each of which can protect from or exacerbate disease, affecting incidence and progression independently (Voskuhl, 2020).
The dominant animal model of MS is the experimental autoimmune encephalomyelitis (EAE) model in mice (Dedoni et al., 2023; Voskuhl, 2020). A mouse is injected with a mixture of myelin protein antigens, along with an adjuvant and pertussis toxin, which together activate immune cells and break down the blood–brain barrier. The autoimmune attack on the brain and spinal cord has many features of the MS attack in humans. In specific strains of mice, females show greater susceptibility, and thus can be studied to isolate sex-specific factors that influence EAE outcomes and determine their cellular and molecular mechanisms of action.
Biological Factors
Sex chromosome effects
To investigate a direct role for sex chromosomes on MS, EAE was studied using the FCG model, allowing the comparison of mice with different sex chromosomes (XX vs. XY) but the same type of gonadal hormones (Smith-Bouvier et al., 2008). The mice had their gonads removed to eliminate the possibility of differences in levels of gonadal hormones when EAE was induced. XX mice showed worse clinical disease and neuropathology,
independent of their gonadal sex at birth. XY mice showed increased secretion of Th2 cytokines associated with protection. This sex chromosome effect may be caused in part by a specific X-linked gene, Kdm6a, that escapes X inactivation and is therefore expressed higher in many cell types, including T cells (Itoh et al., 2019). Conditional knock out of Kdm6a in CD4+ T lymphocytes reduces clinical disease and neuropathology in the EAE model. Another X gene escaping X inactivation, Kdm5c, a H3K4me3 histone demethylase, is also implicated in EAE (Doss et al., 2021). EAE can be induced by transferring T helper 17 (Th17) lymphocytes to recipient mice, after the Th17 cells were stimulated by myelin antigens. The resulting EAE is worse if the transferred Th17 cells are XY than if they are XX. Increasing expression of Kdm5c in XY Th17 cells reduces their pathogenicity, but inhibiting Kdm5c in XX Th17 cells increases EAE. Because the two studies implicating opposing actions of the two genes (Doss et al., 2021; Itoh et al., 2019) were performed in different strains and EAE model systems, it is not clear if both occur at the same time, they differentially regulate promoter or suppressor regions, or one relates to worse incidence in females and the other to worse disease progression in males; further work is needed.
The Y chromosome contributes to sex chromosome effects in EAE (Case and Teuscher, 2015; Case et al., 2013). EAE was induced in multiple strains of male mice that differed in the strain background of their Y chromosome but were otherwise genetically identical in their autosomes and X chromosome. Specific types of Y chromosomes had varying effects on severity of EAE. Further work is needed to discover the Y genetic elements that account for these differences and determine if the Y gene effects synergize with or counteract effects of X chromosome genes impacting EAE (Arnold, 2017).
The effect of sex chromosome complement on EAE differs depending on whether the cells affected are in the immune system or brain. Bone marrow chimeras were used to create mice with XX or XY cells in the immune system, in mice composed of XX or XY cells in the brain and rest of the body (Du et al., 2014). When the sex chromosomes in the brain were XY, the clinical response to EAE was worse, independent of the type of sex chromosomes in the immune system. Thus, the direction of the sex chromosome effect is opposite in the brain (XY worse than XX) than in the immune system (XX worse than XY). These results may help explain why progression of EAE is worse in men than women, even when incidence of MS is worse in women than men.
In studies of female bias in autoimmune diseases, increasing attention is given to XIST, the X chromosome gene that is expressed as a noncoding RNA and catalyzes the inactivation of one X chromosome in each somatic XX cell. This gene regulates the expression of a majority of X genes, many of
which regulate immune function. This gene inactivation by XIST is atypical and aberrant in some types of immune system cells, suggesting that greater incidence of autoimmune diseases in females is caused in part by reactivation and aberrant expression of some normally inactivated X genes in XX cells (Forsyth et al., 2024). Another hypothesis is that the XIST RNA forms ribonucleoprotein complexes with its interacting partners in XX cells, which act as an antigen that is attacked by the immune system in autoimmune diseases. Again, the link to this gene may help explain the female predominance of autoimmune diseases. This idea has received recent support from experiments in mice in which this gene was artificially expressed in XY male mice, increasing the amount of disease in a model of SLE (Dou et al., 2024). Further research is needed to understand the role of XIST.
The Effects of Estrogens
The three types of gonadal hormones that might influence MS are estrogens, progestins and androgens. Treatment of either males or females with the androgens testosterone or dihydrotestosterone reduces disease symptoms (Palaszynski et al., 2004). Thus, androgens may be protective, an idea supported by limited analysis of the immune- and neuroprotective effects of androgens in men in a small clinical trial (Voskuhl et al., 2020). Further work is needed to understand how androgen therapy might alleviate MS symptoms in men, including studies of the affected downstream molecular pathways that may be targeted in the clinic in men and women.
The effects of ovarian hormones are generally protective and thus unlikely to explain the greater incidence of MS in women but may explain the alleviation of MS in late pregnancy. In the third trimester, estrogens and progesterone are elevated (Voskuhl, 2020). Women report worse symptoms when levels of estrogen are low before the menstrual period and after menopause. However, removal of the ovaries has an inconsistent effect on the incidence of MS. In EAE mice, treating females with estrogens reduces clinical symptoms, but progesterone has little effect (Voskuhl and Palaszynski, 2001). Selective knockout of ER alpha (α) or beta (β) in mice indicate that ERα expression in astrocytes but not neurons is required for the neuroprotective and anti-inflammatory effects of estrogens, but that estrogens’ effects do not require ERβ expression in astrocytes or neurons (Spence et al., 2013). However, ERβ ligands can offer protection in EAE mice via actions on other cellular targets (Wisdom et al., 2013), which requires ER expression in oligodendrocytes (Kim et al., 2018). Two Phase 2 clinical trials support a protective effect of estrogens in women with MS. In one, oral contraceptives containing estradiol, taken with interferon beta, improved cognition (De Giglio et al., 2017). The other found that an estrogen plus glatiramer acetate (Copaxone®) improved cognitive
processing speed, spared atrophy in cortical regions involved with problem solving and attention, and decreased serum neurofilament light chain levels (MacKenzie-Graham et al., 2018; Voskuhl et al., 2016, 2020, 2022).
Other Biological Factors and Mechanisms
GWAS have provided additional evidence for the role of the immune system in the pathogenesis of MS. A recent study identified more than 200 genetic variants associated with susceptibility to MS, with a large majority related to the immune response (Patsopoulos et al., 2019). In a systematic review of genome-wide transcriptome studies of MS, differential gene expression was analyzed in males and females using Gene Expression Omnibus and ArrayExpress databases that included patient data. Analysis of gene expression in the brain indicated sex-specific differences in immune patterns: females displayed differences in proinflammatory and innate immune responses related to myeloid lineage and males had changes in adaptive responses associated with the lymphocyte lineage. Other notable results were cellular metabolic alterations in females and differences in the stress response to metal ions in males (Català-Senent et al., 2023).
Specific Risk Factors in Women
Sex differences in the prevalence and severity of MS are influenced by the interplay of environmental factors with inherent biological factors. Environmental risk factors implicated in MS are Epstein-Barr viral infection, cigarette smoke, lack of sun exposure, low vitamin D levels, and obesity (Angeloni et al., 2021; Hedström et al., 2021b). A longitudinal analysis in a cohort of military service members indicated that Epstein-Barr viral infection (but not other viral infections) increased the risk of MS by 32-fold (Bjornevik et al., 2022). Environmental exposures to Epstein-Barr virus, obesity, and smoking may interact with a human leukocyte antigen risk allele, further elevating MS risk (Hedström et al., 2021a, 2021b; Olsson et al., 2017). Certain environmental risk factors that mainly affect women, including occupation, smoking, obesity, birth control, and childbirth, may influence disease onset and progression (Filippi et al., 2018).
Condition Presentation and Diagnosis
Most patients present with relapse-remitting MS (RRMS), which is based on at least two occurrences of CNS dysfunction with partial or complete resolution of symptoms. This diagnosis can also include MRI assessment of demyelinating CNS lesions. Patients with RRMS may eventually develop secondary progressive MS (SPMS), a condition marked by permanent neurological impairments with worsening disability.
A small proportion of MS patients will already present with progressive disease, known as primary progressive MS (PPMS), characterized by progressive CNS dysfunction for a minimum of 1 year and demyelinating lesions or oligoclonal bands in cerebrospinal fluid (Filippi et al., 2018). Women are disproportionately affected by RRMS, develop MS earlier, have more relapses, and exhibit an inflammatory pattern when assessed by MRI; men display rapid progression, worse disease outcomes, and less inflammatory MRI patterns (Angeloni et al., 2021).
Treatment and Management
Despite the development and approval of disease-modifying treatments for patients with RRMS, treatment options are still limited for those with PSMS and SPMS. Furthermore, disease-modifying therapies are associated with severe adverse effects, creating an unmet need for developing safe and effective therapies for the different categories of MS (Filippi et al., 2018). Recently, chimeric antigen receptor (CAR) T cell therapy entered U.S. clinical trials for MS. It has been previously shown to effectively target cancer cells in patients with blood cancers; CAR T cells engineered to target B cells that react to myelin will be evaluated as a potential therapeutic for MS (Mullard, 2024).
Disparities
It is unclear whether disparities rooted in racial or ethnic differences affect the risk of MS in women. In a nested, case-control study of an ethnically diverse, population-based cohort from the United Kingdom, female predominance was observed with MS regardless of ethnicity; furthermore, modifiable risk factors associated with MS (e.g., smoking, obesity, infectious mononucleosis, low vitamin D, and head injury) had similar effects across all ethnicities (Jacobs et al., 2024). In contrast, a multicenter study from Italy indicated that individuals with MS from non-White ethnicities experienced a more rapid clinical decline compared to their White counterparts. As data was not disaggregated by sex, it is unclear if these findings also apply to comparisons of White with non-White women (Bianchi et al., 2024). Disparities in care have been observed in a study that showed that women and individuals with lower educational levels were less likely to be treated with disease-modifying therapies (Leavitt et al., 2024).
Research Gaps
Studies of the last 20 years provide strong evidence implicating both hormonal and sex chromosomal mechanisms in sex differences in MS. These studies suggest multiple gene pathways that might be targeted clinically.
Multiple immune system and CNS cell types function differently depending on their hormonal environment and whether they are XX or XY. Little is known about how the hormonal and sex chromosome mechanisms intersect to affect MS or EAE. The investigation of sex chromosome effects in diverse tissues is still in its infancy, and much work is needed to identify X and Y genetic elements that influence disease.
The interactions between genetic predispositions to MS and environmental, or extrinsic, factors determine the likelihood of developing this disease. However, more research is needed to understand how other factors, particularly Epstein-Barr viral infection, obesity, and lack of sun exposure, prime the immune and nervous systems for an autoimmune attack of the myelin sheath, leading to MS. Furthermore, better therapies specifically catering to the unique symptomatology and progression in men and women need to be developed that target sex-specific disease pathways and address the sex- and gender-related environmental risk factors.
CHRONIC PAIN
Like many chronic conditions, chronic pain disproportionately affects women. Given that it can be general or localized, studies have reported a higher prevalence of chronic pain in women, particularly in terms of musculoskeletal pain, such as back pain, body pain, fibromyalgia, and osteoarthritis; abdominal pain; headaches; and pelvic pain.
Biological Factors
The instigation, propagation, and perception of pain involve several key physiological components: a stimulus that triggers the activation of pain-sensing or -modulating receptors on neurons, which then relay pain signaling through neuronal pathways or networks. The pain signals terminate at brain regions that process and perceive pain stimuli, resulting in either evading or tolerating pain. These components of pain processing appear to be regulated differently in males and females. For example, evidence exists that differences in brain structure between males and females commence early in embryonic development, before gonadal differentiation, indicating the important influence of sex chromosome effects (Mallard et al., 2021; Pallier et al., 2022); however, it is unclear if these brain regional differences include pain-processing centers. Depending on the sex, some brain regions may contain different populations of neurons or differentially expressed receptors involved in pain. Less apparent is if alternate pain-processing pathways are recruited according to sex—although past research has indicated sex differences in ascending pain neural transmission pathways and descending pain modulatory neural pathways (Mogil, 2012). As for pain triggers,
perhaps the best studied in the field of sex differences are the molecules released during inflammation by immune cells, a response influenced by gonadal hormones (Rosen et al., 2017).
Sex Chromosome Effects
Experiments using the FCG mouse model have demonstrated sex chromosome effects, independent from gonadal hormone effects, on pain and analgesic pathways. In one study, mice with two copies of the X chromosome7 exhibited higher sensitivity to two types of acute pain-inducing stimuli, regardless of gonadal type, compared to mice with only one X chromosome.8 As these mice had their gonads surgically removed, these findings suggested that the sex chromosomes themselves, rather than gonadal hormones, had a larger role in determining the pain threshold (Gioiosa et al., 2008). In a related study, XX mice showed less activation of pain-suppressing pathways mediated by the kappa opioid receptor irrespective of gonadal hormones, indicating the influence of sex chromosomes on analgesic mechanisms that dull the perception of pain (Taylor et al., 2022). Discovering the precise mechanisms underlying these effects, starting with the specific identification of X chromosome genes that differentially influence pain processing, is ongoing.
Research has uncovered the differential expression of 40 X chromosome genes following a traumatic event (e.g., car collision) in women who later developed chronic musculoskeletal pain and/or posttraumatic stress versus women who recovered from the accident (Yu et al., 2019). Many genes upregulated in the women experiencing longer recovery times with chronic pain were linked to immune and neuronal function (Yu et al., 2019). The investigators also observed increased expression of other X-linked genes that tend to escape X inactivation (Yu et al., 2019). Collectively, these findings suggest the possible involvement of certain X chromosome genes in triggering and perpetuating mechanisms of pain in women.
The Effects of Estrogens
Ample evidence exists that supports the role of gonadal hormones as critical mediators of the sex differences observed for chronic pain. Although the male hormone testosterone is generally associated with pain suppression (Lenert et al., 2021; Lesnak et al., 2020), estrogens have dual, opposing effects, promoting both analgesia and an increased sensitivity to pain, depending on the experimental context (Hernandez-Leon et al., 2018;
___________________
7XX which developed ovaries or XX+SRY which developed testes.
8XY which developed testes or XY-SRY which developed ovaries.
Linher-Melville et al., 2020). Research has linked low levels of estrogens to a greater inflammatory response that aggravates pain signaling (Lenert et al., 2021). However, high levels of estrogen also correlated with enhanced pain sensitivity (Hellman et al., 2021; Lenert et al., 2021), indicating the complexity of estrogen-mediated regulation of pain mechanisms. Although it is difficult to reconcile these seemingly contradictory results, numerous studies have provided evidence that hormonal fluctuations lead to increased pain, but stable hormone levels—after menopause, for example—suppress pain (Lenert et al., 2021).
Numerous studies have shown that gonadal hormones exert sex-specific effects on neural pain pathways and the immune response, which is an important component that triggers and modulates the sensation of pain (Lenert et al., 2021). Estrogens modulate pain transmission in females by binding to estrogen receptors that are widely expressed in the pain processing areas of the peripheral and central nervous systems (Maurer et al., 2016). Research has also shown that estrogens enhance anti-inflammatory responses (Athnaiel et al., 2023). In contrast, testosterone suppresses proinflammatory signaling molecules (Lenert et al., 2021) and decreases signaling mediated by the pain-causing chemical capsaicin, thereby supporting its analgesic effect in males (Athnaiel et al., 2023). Testosterone has similar analgesic effects in women, as evidenced by studies showing that higher levels of testosterone were associated with less neck and shoulder pain (Kaergaard et al., 2000), and that it eased pain associated with vulvodynia upon topical application along with estrogens (Burrows and Goldstein, 2013) (see Chapter 5 on female-specific and gynecologic conditions), ameliorated pain responses after formalin exposure in female rats (Aloisi et al., 2004), and reduced hypersensitivity to muscle pain in female mice by acting on the serotonin transporter (Lesnak et al., 2020).
Sex Differences in Pain Sensitivity
Research has identified sex differences in pain sensitivity, response to analgesics, and propensity to develop chronic pain after injury or disease. A literature search regarding 77 painful disorders revealed female prevalence for 38 of them and male prevalence in 15 (Berkley, 1997). Meta-analyses of epidemiological surveys (Fillingim et al., 2009), analyses of electronic medical record data (Ruau et al., 2012), and meta-analysis of clinical trial data (Li et al., 2023) all confirm that women report more frequent and greater levels of pain than men. In addition, evidence from international collaborations assessing sex and gender inequalities related to chronic pain indicates that a greater proportion of women are affected than men (Bimpong et al., 2022; Tsang et al., 2008). The Global Burden of Diseases, Injuries, and Risk Factors Study 2016 reported that musculoskeletal disorders, which included
lower back and neck pain, were the largest contributors to higher rates of years lived with disability (YLD) in women globally (GBD 2016 Disease and Injury Incidence and Prevalence Collaborators, 2017). In another study, pooled results from 19 European countries showed more women than men reporting chronic pain, with the greatest inequalities in back and neck pain; however, when data was disaggregated by country, there were variations in the extent of sex differences (Bimpong et al., 2022). While the influence of the female sex on chronic pain was consistently observed, the differences in pain inequalities among the studied nations suggest that gender, shaped by social and cultural influences unique to each country, may also play an important role in the expression of pain, and consequently the development of chronic pain in women (Ayres et al., 2023; Bimpong et al., 2022). Because systematic investigation into gender differences in pain is in its infancy, it remains unclear whether sex differences in pain perception result from biological factors or from gender differences in the reporting of chronic pain arising from sociocultural factors (Boerner et al., 2018). Studies on sex differences conducted over the last 10–20 years aimed to determine the mechanisms that link chronic pain and developing other pain-related conditions (chronic overlapping pain conditions), identify the factors that lead to chronic pain, and discern how they differ by sex (Hoffmann et al., 2022).
To clarify sex differences in pain sensitivity from sex and gender differences in disease susceptibility and pain reporting, researchers have conducted hundreds of controlled laboratory studies comparing pain thresholds, tolerances, and ratings between men and women. The variability in the resulting literature has encouraged some to conclude that sex differences in pain are inconsistent and possibly illusory (Racine et al., 2012) or at least more complex than a simple matter of differential “sensitivity” (Hashmi and Davis, 2014). However, when ignoring statistical significance of a study’s results and simply tallying the number of studies in which women were more sensitive to pain than men and vice versa, dramatic overall evidence appears that women are more sensitive to pain, irrespective of measure or modality (Mogil, 2012).
Many potential explanations exist for this sex difference. Theories supported by evidence include those at the level of genetics, neurochemistry, cell- and systems-level processes, organizational and activational effects of gonadal hormones, experiential factors, and gender-related sociocultural factors (Mogil, 2012).
Pain Processing in Female Versus Male Individuals
Regardless of the true explanation(s) of quantitative sex differences in pain (i.e., sex differences in the amount of pain or analgesia), increasing evidence (such as the null mutant phenotypes) suggests robust qualitative
sex differences in the operation of the biological mechanisms underlying pain processing and modulation (Mogil, 2020).
The now much-more-common use of both sexes in preclinical research has led to new revelations regarding sex-specific biological processing of pain. Perhaps the most celebrated and replicated of these findings concerns the role of spinal cord microglia9 in developing chronic pain (Ghazisaeidi et al., 2023; Gregus et al., 2021; Halievski et al., 2020; Midavaine et al., 2021; Rosen et al., 2017; Sorge et al., 2015). One of the main advances in preclinical pain research over the past 20 years has been gradually realizing the critical role of the innate immune system, especially spinal cord microglia, in pain processing (Beggs et al., 2012; De Leo et al., 2006; Ji et al., 2019; Marchand et al., 2005; Scholz and Woolf, 2007; Watkins and Maier, 2003). Investigators conducted virtually all research in this subfield on male mice and rats, as was typical at the time, but in a seminal sex difference finding, one team demonstrated that the microglial dependence of chronic pain was entirely a male-specific phenomenon (Sorge et al., 2015). This same group demonstrated that in female mice and male mice with lowered testosterone levels, T cells of the adaptive immune system were mediating pain signal processing, although the details of female-specific T cell signaling have yet to be fully elucidated. There have been other demonstrations of female-specific pain circuitry, including work demonstrating the role of prolactin—a protein that enables mammals to produce milk—and its receptor in pain sensitization and pain signaling (Chen et al., 2020).
Qualitative sex differences relevant to pain are not only seen in animal studies. A growing proportion of mechanistic pain research is being performed by RNA sequencing of human tissues. The sets of genes such studies have identified tend to depend significantly on the sex of the tissue donor (Ray et al., 2019, 2023; Tavares-Ferreira et al., 2022).
Specific Risk Factors in Women
Researchers have typically viewed chronic pain from a biopsychosocial perspective to identify the factors leading to it and discern the interaction among these factors (Hoffmann et al., 2022). General risk factors include demographics; lifestyle factors, such as physical activity, occupation, and nutrition; clinical factors, and history of injury, abuse, or interpersonal violence (Mills et al., 2019). Lower pain self-efficacy10 and a higher tendency
___________________
9Microglia are cells in the nervous system that share many if not all the properties of immune system macrophages found in other tissues.
10Pain self-efficacy is described as an individual’s belief in their ability to effectively control their pain (Hoffmann et al., 2022).
for pain catastrophizing11 are gender related behaviors associated with chronic pain in women (Hoffmann et al., 2022), although these factors are generally not well established.
ACEs
Research has linked ACEs to an increased risk for several adverse health conditions, including chronic pain (Anda et al., 2010; Felitti et al., 1998; Kerker et al., 2015; Nelson et al., 2017, 2018). Emerging research suggests that exposure to certain ACEs may be more predictive of poor health outcomes, including chronic pain (Atzl et al., 2019; Beal et al., 2020). Evidence suggests that rates of certain ACEs, particularly maltreatment, are higher among parents of youth experiencing chronic pain, suggesting they may confer risk for child pain (Beveridge et al., 2020). Studies have shown that exposure to maltreatment was the driving factor in the relationship between parental ACEs and a gene encoding a specific dopamine receptor in young women. Decreased expression of this gene in young women of parents who were maltreated may result in reduced dopamine signaling, which might explain the increased susceptibility of female chronic pain patients to mental disorders, such as anxiety and depression that are known to have compounding effects on chronic pain (Altemus, 2006; Breslau, 2009; Leadbeater et al., 1999; Noble, 2005; Noel et al., 2016; Piccinelli and Wilkinson, 2000).
Reproductive Milestones
Limited epidemiological data support the idea that chronic pain is likely to develop at certain stages of life in women, such as adolescence and old age; it is thought to be influenced by certain sex-linked factors, such as hormones and reproductive factors (Casale et al., 2021; Osborne and Davis, 2022). How the menstrual cycle influences the perception of pain is not well understood (Piroli et al., 2019), nor is the etiology of underlying chronic pain risk among women in midlife. Research has shown that estrogens and other reproductive hormones have complex interactions with pain modulation and pain sensitivity, with hormonal fluctuations—particularly the decline in estrogens in perimenopause and into post-menopause—potentially influencing vulnerability to developing and exacerbating pain conditions during midlife (Pavlovic and Derby, 2022).
Evidence that the stressors experienced by midlife women affect symptom sensitivity and pain tolerance (Lachman et al., 2015). They face the
___________________
11Pain catastrophizing refers to the continuous, exaggerated, and negative thoughts and feelings occurring during painful experiences (Leung, 2012).
physical effects of aging linked to musculoskeletal pain. A systematic review and meta-analysis of studies on musculoskeletal pain during midlife demonstrated that women in perimenopause were particularly vulnerable to developing it compared to premenopausal women and that postmenopausal women were vulnerable to develop moderate to severe musculoskeletal pain compared to premenopausal and perimenopausal women (Lu et al., 2020).
Weight gain and decreased physical activity at midlife may further contribute to chronic pain morbidity (Gibson et al., 2019). An analysis of data on middle-aged adults from the biannual Health and Retirement Study found that being overweight or obese accounted for 32.1 percent of the increases in severe or limiting pain among women, compared to 19.0 percent in men and as much as 20 percent of the increase in mild or moderate pain in women compared to 10 percent in men (Stokes et al., 2020). Obesity and chronic knee pain are often related to the onset of osteoarthritis in women, with greater risks of developing osteoarthritis as a function of weight gain in women compared to men (Batushansky et al., 2022). Aside from the biomechanical stress elicited by excess physical weight, obesity also alters metabolism and promotes inflammation, which increases the risk of osteoarthritis and chronic pain (Ait Eldjoudi et al., 2022; Batushansky et al., 2022).
In a national sample of midlife women U.S. veterans, those with menopausal symptoms had nearly twofold higher odds of diagnosed chronic pain and chronic pain multimorbidity, accounting for age and other known risk factors (Gibson et al., 2019). These findings may suggest that midlife is a vulnerable and critical period for women to experience chronic pain.
Gender-related factors that influence chronic pain in women
Factors associated with gender (see Chapter 2) also influence pain experiences and related behaviors in women and men (Keogh, 2022). Certain gender-related constructs, such as traditional masculinity, which is often associated with stoicism and independence, and femininity, typically characterized by sensitivity, sociability, compassion and expressiveness, can shape the perceptions and experiences of pain (Keogh, 2022). Greater tolerance to pain in experimental settings was observed in men identifying with traditional male roles compared with women who identified with traditional female roles (Hoffmann et al., 2022; Pool et al., 2007). Associations between high masculine/low feminine traits and pain tolerance have been noted in several studies, regardless of biological sex (Alabas et al., 2012; Applegate et al., 2005; Keogh, 2022). In addition, compared to men, women report their pain more often, demonstrate a greater likelihood for seeking help and for health care utilization (Keogh, 2021, 2022; Mills et al., 2019), and tend to exhibit pain-related catastrophizing and
lower self-efficacy, both of which can aggravate pain (Keogh, 2022). Other evidence has shown that women and men use different coping strategies to manage chronic pain, with women relying more on social support, positive self-affirmation, and self-care, while men resort to avoidance, distraction, and/or problem-solving techniques (Hoffmann et al., 2022; Keogh, 2022). Although gender-based behaviors in response to pain likely originate in early childhood through one’s social environment, pain-related behaviors are also informed by existing socially accepted stereotypes (Keogh, 2021). A better understanding of the impact of gender constructs on the perception and experience of pain is needed to formulate effective treatment strategies for both women and men.
Gender factors influencing chronic pain in women are also shaped by culture (Ayres et al., 2023). For example, a comparison of men and women in the U.S. versus Romania showed that despite receiving the same types and dosages of pain medications, women reported higher pain scores than men in the U.S., but not in Romania (Ayres et al., 2023). These differences in the behavior of reporting pain—reflecting related gender aspects such as coping mechanisms and adherence to social norms—may depend on the cultural context (Ayres et al., 2023). Overall, evidence to date supports a female bias in developing chronic pain, but variability in sex differences across countries are likely influenced by social and cultural differences (Ayres et al., 2023).
Condition Presentation and Diagnosis
Diagnosing pain in women is challenging given its complexity, and studies have investigated differences in symptoms associated with chronic pain. One important point relevant to sex differences in humans is that women are more likely than men to be diagnosed with a musculoskeletal condition leading to pain and have comorbidities or chronic overlapping pain conditions that may affect the risk of chronic pain, but it is not known if these comorbidities contribute to or are a result of heightened sensitivity to noxious stimuli (Templeton, 2020).
Much is known about sex differences in behavioral responses to noxious stimuli that shape pain perception. However, little is known about how to extrapolate the developments evident in epidemiological and preclinical data to clinical settings, resulting in the slow progress of diagnosing and identifying chronic pain in women. For example, data from a meta-analysis of studies that investigated high- and low-intensity pain response among healthy controls and those with depression showed that overall pain tolerance was similar across depressed and control groups, but low-intensity stimulation had small but significantly higher average sensory and pain thresholds in depressed participants (Thompson et al., 2016).
This example led to another development in chronic pain research, “phenotyping,” which recognizes that even people with the same condition have tremendous variability in signs, symptoms, and features (Hoffmann et al., 2022), reflecting the influence of biopsychosocial factors, such as sex, gender, and age (Ocay et al., 2022). Thus, placing an emphasis on clustering individuals with chronic pain in research studies is warranted so that they can identify those whose pain may be driven by different underlying mechanisms (Hoffmann et al., 2022). Fibromyalgia is another example; it presents with variable symptoms, including mood disturbances, fatigue, pain, painful bladder, gastrointestinal issues, sleep issues, and restless legs (Hoffmann et al., 2022). This variability has led researchers to identify subcategories, including pain, physical involvement, psychological function, and social support, that could lead to a more comprehensive assessment of an individual’s symptoms (Yim et al., 2017). These results suggest that phenotyping may be a strategy to better understand how to manage chronic pain in women.
Research has begun to focus on chronic overlapping pain conditions, where one develops with others (Hoffmann et al., 2022), such as chronic lower back pain, chronic migraine and headache, fibromyalgia, irritable bowel syndrome, painful bladder syndrome, endometriosis, chronic tension-type headache, temporomandibular disorders, vulvodynia, and myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), all of which are more common in women (Hoffmann et al., 2022).
Women face a unique challenge in the health care system when it comes to diagnoses of multiple overlapping pain conditions such as fibromyalgia and migraines, both of which are more common in women and perceived to be poorly understood by medical providers (Driscoll et al., 2018). Given the complexity in diagnoses and management of common pain disorders among women, in addition to the lack of multidisciplinary treatment teams and the stigma and bias that serve as complicating factors in pain management, women are left feeling dismissed, disrespected, and frustrated by the health system when it comes to seeking treatment for pain.
A study identified a bias toward underestimation of pain in patients from the perspective of caregivers’ and clinicians’ pain stereotypes. The study demonstrated that caregivers and clinicians estimated women to be in less pain than men at the same level of pain expressiveness and self-report, specifically underestimating women’s pain relative to actual self-reported pain while overestimating men’s (Zhang et al., 2021). They also indicated that women would benefit more than men from psychotherapy versus pharmacotherapy when choosing between the two (Zhang et al., 2021). These findings support the reality that clinicians underestimate women’s pain and see it as less intense than men’s, which leads to ineffective or suboptimal pain treatment recommendations.
Treatment and Management
Treating and managing chronic pain includes pharmacological interventions, such as opioid analgesics and nonopioid analgesics, and nonpharmacological interventions, such as physical therapy, acupuncture, and meditation (Qaseem et al., 2017). Few pharmacological options have been developed specifically for women, and treatment is largely with drugs that were developed for other diseases and have limited effectiveness and problematic tolerability (Casale et al., 2021). Although studies have shown that women are less likely to receive an opioid analgesic, meta-analyses and clinical experimental studies suggest that opioids may provide greater relief from pain for women than for men, although that difference depends on factors such as age and comorbid mental disorders (Pisanu et al., 2019).
In addition, the lack of knowledge regarding how to properly treat pain has led to a knowledge gap when it comes to treating pain in women. Women have reported that they were less informed about the availability of certain treatment modalities, including complementary and integrative health, which is an approach to health care that a majority of non-complementary and integrative health care providers are not familiar with, which means they lack the knowledge needed to provide appropriate counseling (Washington et al., 2011). There are advantages to creating an integrated health care system that prioritizes lifestyle, exercises, self-care, complementary and integrative health, and interdisciplinary modalities as effective and critical treatment options to handle the unmet need in addressing, evaluating, and managing chronic pain disorders among women.
Midlife women experience increasing pain symptoms related to generalized body aches, headaches, and genitourinary pain as reported by a multiethnic cohort of midlife women that identified clear patterns of symptom clustering (Harlow et al., 2017). However, the scarcity of studies regarding pain symptoms have contributed to underdiagnosis of specific pain disorders in the setting of common symptoms endured by midlife women. Thus, poor management of their chronic pain remains a growing national problem, with a critical need to fill this research gap and address pain symptoms; for example, the highest increase in opioid prescribing and opioid-related overdose mortality has been among women in their 40s to 60s (Gibson et al., 2019).
Disparities
Studies suggest that the prevalence of chronic pain disproportionally affects certain racial and ethnic groups, resulting in health disparities (Anderson et al., 2009; Janevic et al., 2017). Some studies have sought to understand how differential exposures to psychosocial and environmental
factors influence these differences (Aroke et al., 2019; Overstreet et al., 2023). Early exposure to psychological and environmental risk factors is associated with chronic pain and contributes to disparities in chronic pain prevalence among people of color (Overstreet et al., 2023). Few studies have focused on identifying the psychological factors that lead to higher levels of pain sensitivity among people of color. For example, although the Hispanic and Latino/Latina population had the most population growth in the U.S. between 2010 and 2020 (Census Bureau, 2021), limited research has focused on their experiences with pain (Zajacova et al., 2022). Population-based studies indicate higher prevalence and greater pain sensitivity among Black, Asian, and Hispanic participants compared to White participants (Zajacova et al., 2022). A higher prevalence of pain was also observed in American Indians and Alaska Natives; however, unlike the other ethnic groups, greater pain sensitivity compared to White individuals was not apparent (Zajacova et al., 2022). These findings suggest that current experimental pain research may not reflect the extent of pain disparities in the general population (Zajacova et al., 2022).
Lesbian, Gay, Bisexual, Transgender, Queer, Intersex, Asexual, and Other (LGBTQIA+) Disparities
Compared to the general population, LGBTQIA+ individuals experience more chronic physical and mental health problems than heterosexual individuals (Moagi et al., 2021; Pinnamaneni et al., 2022). Although poor physical and mental health are known correlates, (Edwards et al., 2016), PRIDE12 is the first cohort study that assessed chronic pain among U.S. sexual and gender minority adults (Chadwick et al., 2024). Compared to cisgender men, transgender women had 1.46 greater odds of widespread chronic pain (Chadwick et al., 2024). Research has also shown that the LGBTQIA+ population is at greater risk for chronic pain than their cisgender, straight counterparts. One study, for example, found that pain prevalence among sexual minority adults was 4.5–11.4 percent higher than among straight adults (Zajacova et al., 2023).
Research has shown that many disparities in chronic conditions, disability, and mental distress observed in younger lesbian, gay, and bisexual adults persist into adulthood compared to other populations (Fredriksen-Goldsen et al., 2017). Studies have also found that LGBTQIA+ adults are more likely to experience more frequent functional limitations from pain and pain from multiple sites compared to heterosexual adults (Case et al., 2004; Chadwick et al., 2024; Fredriksen-Goldsen et al., 2017). Additional studies examining the association between sexual orientation
___________________
12PRIDE is a community-engaged research study conducted in the LGBTQIA+ community.
and pain among a range of age groups and residencies found that sexually minoritized adults have comparable to higher prevalence of pain or painful conditions (Cochran and Mays, 2007; Fredriksen-Goldsen et al., 2017; Katz-Wise et al., 2015; Nagata et al., 2020; Roberts et al., 2013).
Research Gaps
The National Institutes of Health’s Sex as a Biological Variable policy in 2016 forced investigators to account for sex as a biological variable in research study designs and should address the problem that many of the biological mechanisms underlying pain are relevant only to the male subjects that had dominated pain research. It seems likely that sex differences in pain mechanisms (Nemmani et al., 2004; Sorge et al., 2015) may have been responsible for the failure of certain clinical trials of analgesics (Galer et al., 2005). The plethora of sex-dependent pain mechanisms suggests that some therapies will be effective in only one sex. That day may have already arrived. An analysis of publicly available clinical reviews of the New Drug Applications for ubrogepant (Ubrelvy®) and rimegapant (Nurtec ODT®) revealed that they were only effective as migraine treatments in women (Porreca and Dodick, 2023).
Although human studies now typically enroll both men and women, disaggregation of data by sex continues to be a problem, with less than 50 percent of the papers in Pain 2015–2019 featuring any mention of sex differences in the results or discussion sections (Mogil, 2020).
The influence of hormonal factors on chronic pain is nuanced, and more studies are needed. Although studies have investigated sex differences in chronic pain, the primary biological and hormonal mechanisms that explain these differences in prevalence and expression continue to be understudied (Pavlovic and Derby, 2022). In addition, the female-specific risk factors are not well established. The literature suggests that midlife is a critical period for women during which the frequency and intensity of pain symptoms increase, but only a few studies have addressed the epidemiology and mechanisms of pain in midlife women.
Chronic pain diagnosis is a significant research gap, partly due to its similarity with other overlapping pain conditions, with a pressing need for more research to unravel the connections between chronic pain and overlapping pain conditions and understand gender biases in diagnosing pain in women. Studies for establishing consistent definitions of chronic pain are needed; the absence of a clear diagnostic framework for women complicates the ability to synthesize findings across various studies, hindering progress in understanding and treatment.
Moreover, the understanding of effective treatment and management strategies for chronic pain in women is insufficient, posing severe challenges
for women navigating the health care system, with a notable gap in research on how chronic pain medications specifically affect women and a need for more studies on self-care and nonpharmacological interventions. Additionally, the disparities in chronic pain experiences among women of diverse racial and ethnic backgrounds and within the LGBTQIA+ community remain poorly understood. This includes a lack of clarity on the reasons behind the mismatch between measures of pain sensitivity and reported prevalence rates, and a scarcity of research investigating the factors contributing to chronic pain in these groups.
FIBROMYALGIA
Fibromyalgia is a syndrome characterized by chronic musculoskeletal pain and symptoms such as fatigue, sleep disturbances, and mood disorders. It is believed to be caused by CNS dysfunction (Siracusa et al., 2021). Disagreement and ambiguity regarding diagnostic criteria and presence of bias were themes highlighting the historical context of fibromyalgia (Arout et al., 2018; Galvez-Sánchez and Reyes Del Paso, 2020).
A review of the diagnostic criteria for fibromyalgia highlighted the evolution from a singular to a multi-symptom diagnosis (Galvez-Sánchez and Reyes Del Paso, 2020). First described in 1642 as “muscular rheumatism” and later in 1904 as “fibrositis,” it was labeled “fibromyalgia” in 1976. The concept was centered around complaints of pain in at least three areas, no other disorders that may contribute to the symptoms, and at least five consistent tender points (Yunus et al., 1981). It was not until 1990 that the American College of Rheumatology (ACR) established official diagnostic criteria (Wolfe et al., 1990). However, defining and diagnosing fibromyalgia has remained a challenge, and research aimed at understanding how to prevent, diagnose, and treat it has lagged, as has research on disparities among women.
Biological Factors
The etiology of fibromyalgia is largely unknown and may be similar to mechanisms described in chronic pain for central sensitization13 and endogenous pain inhibitory mechanisms (Galvez-Sánchez and Reyes Del Paso, 2020). Greater perception of pain has been a common thread in the diagnosis, perhaps from central sensitization that may result from persistent painful stimuli associated with injured tissue that triggers persistent
___________________
13Central sensitization occurs when the central nervous system becomes hypersensitive to pain-inducing stimuli, including injury, pressure, temperature, and/or movement (Hoffmann et al., 2022).
peripheral sensitization. Some studies have shown peripheral nerve changes in fibromyalgia, including a reduced number of epidermal nerve fibers in skin biopsies and altered heat, cold, and pain thresholds. Researchers have postulated that blocking peripheral sensitization may stop the chronic pain. Other principal processes underlying fibromyalgia include dysfunction of neural circuits and inflammatory, immune, endocrine, genetic, and psychosocial factors (Siracusa et al., 2021). Although the etiology is not well understood, numerous studies have demonstrated that more women have fibromyalgia than men, implicating biological sex as a driving factor (Martínez-Lavín, 2021).
Sex Chromosome Effects
No direct evidence exists for sex chromosome effects on fibromyalgia. Some researchers have noted its co-occurrence with several autoimmune conditions, including autoimmune thyroiditis and Sjogren’s syndrome (Applbaum and Lichtbroun, 2019; Martínez-Lavín, 2021; Tagoe et al., 2019), which hints at an autoimmune-based etiology (Meester et al., 2020). Considering that many autoimmune conditions have recently been attributed to incomplete X chromosome inactivation, leading to higher expression of immune-system-related genes (Pallier et al., 2022), an influence mediated by the X chromosome may exist in comorbidities that include fibromyalgia.
The Effects of Estrogens
Estrogens may exert analgesic effects that depend on a woman’s hormonal milieu. For example, menopausal women with erratic hormonal fluctuations before the final period may be more likely to experience fibromyalgia, and estrogen therapy has been shown to reduce pain (Athnaiel et al., 2023). Estrogen’s analgesic effects were also seen in an experimental animal model of fibromyalgia in which surgically removing the ovaries aggravated pain symptoms in female rats but administering estrogen reduced them (Hernandez-Leon et al., 2018). Together, these preclinical and clinical results suggest that steady levels of estrogens may reduce pain perception, but fluctuations or a sudden drop in levels of estrogen exacerbate pain in women with fibromyalgia (Athnaiel et al., 2023).
In contrast to these findings, other studies suggest that estrogens increase pain symptoms in fibromyalgia. For example, animal model studies demonstrated that estradiol upregulates the expression of an ion channel protein, Acid-Sensing Ion Channel-3 in dorsal root ganglia—a neural population implicated in conducting fibromyalgia pain (Martinez-Lavin and Solano, 2009; Ren et al., 2018) Estradiol also increases the expression
of the sodium channel Nav1.7, in trigeminal ganglion neurons, aggravating the pain response in animal models (Bi et al., 2017). It is interesting to note that a polymorphism in the gene encoding Nav1.7, SCN9A, has been associated with a severe form of fibromyalgia in a small cohort of Mexican women (Vargas-Alarcon et al., 2012), suggesting that estrogen-dependent regulation of ion channels may influence symptoms of fibromyalgia, and other chronic pain disorders predominantly affecting women.
Other studies suggest that variations in ERs may mediate the effects of estrogens on the preponderance of fibromyalgia among women. A study of Turkish women found that variants of estrogen receptor-alpha (ER-α) were more prevalent in fibromyalgia patients compared to controls. The investigators noted that specific genotypes were associated with certain symptoms, such as headache and menstrual cramps (Arslan et al., 2021). Although estrogens appears to modulate the fibromyalgia pain mechanisms, whether it promotes or suppresses pain perception and the mechanisms underlying these dual effects remain unresolved.
Other Biological Factors and Mechanisms
Despite gaps in the understanding of the etiology of fibromyalgia, a growing consensus supports a genetic contribution to it (Ablin and Buskila, 2015). Familial inheritance patterns have been observed, with the odds ratio of fibromyalgia higher in relatives of individuals diagnosed with fibromyalgia versus rheumatoid arthritis (Arnold et al., 2004). Research has shown that genetic polymorphisms in serotonergic, dopaminergic, and catecholaminergic neural transmitter systems involved in pain processing are present in fibromyalgia. One specific genotype in a serotonin transporter gene was associated with fibromyalgia and symptoms of depression and anxiety (Cohen et al., 2002; Offenbaecher et al., 1999). In contrast, the frequency of a polymorphism in a dopamine receptor gene was negatively correlated with fibromyalgia (Buskila et al., 2004). Genome-wide analyses have identified additional genes involved in neurotransmission (Arnold et al., 2013; Docampo et al., 2014). These studies suggest that the genetic vulnerability may account for as much as half of the risk of fibromyalgia (Siracusa et al., 2021).
Studies on the role of several proteins in fibromyalgia pain, including Substance P, nerve growth factor, brain-derived neurotrophic factor, and pentraxin-3 have been inconclusive, with only some studies showing an association (Favretti et al., 2023).
Research is exploring the role of vitamin D and fibromyalgia. Despite some studies suggesting a potential relationship between vitamin D deficiency and fibromyalgia, other studies suggest that these findings may be related to limited exercise and sun exposure, and the effectiveness of
vitamin D supplementation in addressing the pain is unclear. Research is needed on the relationship between changes in the gut microbiome and fibromyalgia severity (Favretti et al., 2023).
Specific Risk Factors in Women
Factors associated with fibromyalgia in women are largely understudied and lack evidence based on studies in U.S. populations. Potential factors are younger age and mental disorders such as depression and anxiety, but these are mainly based on cross-sectional studies (Arout et al., 2018). A history of ACEs, IPV, and physical abuse have been identified as potential factors affecting women, but these findings are based on limited cross-sectional studies (Bacon and White, 2023; Chalder et al., 2021; Chandan et al., 2021). Women veterans are identified as a population at risk of fibromyalgia (D’Aoust et al., 2017).
The role of female gonadal hormones in fibromyalgia is evident from studies conducted at different stages of the life-span. For example, higher incidence of fibromyalgia was observed in girls compared to boys at the onset of puberty (Kashikar-Zuck and Ting, 2014; McLeod, 2014). Another study correlated hormone levels during the menstrual cycle with changes in pain symptoms in women with fibromyalgia and found that high levels of progesterone and testosterone were associated with lower pain severity (Schertzinger et al., 2018). In contrast, estrogen and cortisol levels did not influence pain severity, though a post-hoc analysis showed that the association of high progesterone with low pain severity was significant only when cortisol levels were high (Schertzinger et al., 2018). This study along with separate findings showing how treatment with testosterone gel improved pain symptoms in a small cohort of fibromyalgia patients (White et al., 2015) is in line with testosterone’s purported analgesic effects, previously described in the Chronic Pain section of this report.
Condition Presentation and Diagnosis
Gender differences and potential gender biases in diagnosis are a topic of debate, and study results depend on the use of criteria that are appropriate in research settings versus clinically based fibromyalgia measures (Wolfe et al., 1990). The latter are subject to confirmation and selection bias and present major limitations in validity and reliability in a clinical context. Research studies show less unbiased estimates in research contexts, especially among women, and it has been stated that this condition predominantly affects middle-aged women (Häuser and Fitzcharles, 2018; Wolfe et al., 2018). This situation can lead to challenges of misdiagnosis and overdiagnosis and overtreatment (Galvez-Sánchez and Reyes Del Paso, 2020).
Given these biases and that the diagnosis is primarily clinical, identifying biomarkers would be helpful for both diagnosis and monitoring the benefit of treatment. Despite 25 years of research, no specific biomarker has been identified (Favretti et al., 2023).
The typical age of onset is 30–35 (Siracusa et al., 2021). ACR criteria have provided essential guidance for clinical assessment for decades and were recently updated. The key diagnostic criteria include complaints of pain above and below the waist and generalized pain for at least 3 months (Siracusa et al., 2021). To facilitate diagnosis in the clinical setting, researchers developed survey instruments, such as the Polysymptomatic Distress Scale, which incorporates the Widespread Pain Index. These instruments also provided a means to assess the severity of the disease in any given patient.
Fibromyalgia has other associated characteristics and symptoms. Depression is the most common chronic condition associated with it, with a weighted prevalence of up to 63 percent of patients. Other comorbidities include migraine/headache, irritable bowel syndrome, myofascial pain syndrome, and temporomandibular disorders (Kleykamp et al., 2021). Overactive bladder syndrome is also common in women with fibromyalgia, with as many as 40 percent experiencing it (Salaffi et al., 2022). Sexual dysfunction affects as many as 86.9 percent of women with fibromyalgia (Salaffi et al., 2022). Despite the significant prevalence of these two conditions, they are understudied, as instruments such as the Fibromyalgia Impact Questionnaire used to estimate fibromyalgia in epidemiologic studies do not include items for them (Salaffi et al., 2022).
Treatment and Management
Many studies exist on therapeutic interventions for fibromyalgia. For example, researchers have used animal models of non-inflammatory pain to investigate the efficacy of specific medications in reducing fibromyalgia; antidepressants, opioids, glutamate receptor antagonists were effective. However, nonsteroidal anti-inflammatory agents were not effective (Siracusa et al., 2021).
A systematic review of clinical trials assessed the efficacy of two antidepressants commonly prescribed for treatment. Amitriptyline had a greater effect on quality of life but little evidence that it had a beneficial effect on remedying pain (de Farias et al., 2020).
The literature highlights exercise, massage therapy, and diet as potentially beneficial. Investigation continues into the best medical interventions of associated conditions, such as depression. Given the effects of fibromyalgia over the life course, it is important to appropriately adjust treatment based on not only severity but also the stage of life.
The effect of aquatic training and detraining on the physiology and the quality of life of women with fibromyalgia was assessed in a randomized trial. Aquatic training was associated with improvements in oxygen intake, an increase in pain threshold, and improved quality of life. However, these benefits declined when activity was reduced, suggesting that persistent activity is needed to maintain the benefits of exercise (Andrade et al., 2019).
One study assessed the effects of a tryptophan and magnesium-enriched Mediterranean diet in a randomized trial of 22 women with fibromyalgia over 16 weeks. The participants experienced improvements in sleep quality and psychological metrics compared to controls (Martínez-Rodríguez et al., 2020).
Considering that musculoskeletal pain is a common presenting symptom of fibromyalgia, investigators conducted an RCT to determine if massage therapy is effective in addressing pain and the psychological effects of fibromyalgia. Improvements were noted in the intervention group versus the control group across key variables: fatigue, pain, sleep, stress/anxiety, depression, anger, vigor, and fatigue. This study suggests benefits from massage therapy for fibromyalgia patients (Nadal-Nicolás et al., 2020).
A meta-analysis of RCTs evaluated the effectiveness of virtual-reality therapy in mitigating the impact of fibromyalgia on quality of life. The key findings were that exercise therapy showed significant improvements in functional movement, pain, dynamic balance, aerobic capacity, fatigue, quality of life, anxiety, and depression in women aged 38–55 with a BMI of 25–28. However, few studies and participants, lack of blinding, and potential publication bias limit the generalization and quality of the evidence (Cortés-Pérez et al., 2021).
Disparities
Limited research is available on racial and ethnic disparities in women with fibromyalgia, including how symptoms may vary, pain severity, and differential response to treatment. Findings from a clinical study found that Black women reported higher pain severity and pain interferences with daily functioning compared to White women (Jacobs et al., 2023). The authors mention that factors that may contribute to these disparities observed may be linked to structural and social determinants of health (see Chapter 7). A scoping review to describe the literature on fibromyalgia and other neurological and somatic conditions suggests that the prevalence of fibromyalgia is greater in sexual and gender minority individuals, but this was not broken down by group and was based on limited studies outside of the U.S. (Lerario et al., 2023).
Research Gaps
Research gaps can be identified in every category of the translational spectrum, ranging from the basic sciences to population health. In the basic sciences, developing an animal model that captures the key clinical findings will be important for identifying biomarkers that are predictive of the disease process. Furthermore, the close link between fibromyalgia and co-occurrence of autoimmune diseases suggests the role of incomplete X chromosome inactivation and should be explored further. Greater diversity is needed in publicly available databases used to assess the genetic predisposition to fibromyalgia.
Research to improve the diagnostic characterization of fibromyalgia, that is objective and validated across populations, is needed along with studies of diagnostic bias and strategies to mitigate bias. Moreover, as shown by the omission of overactive bladder in surveys used to diagnose and assess fibromyalgia severity, it is important to include all conditions associated with fibromyalgia in such surveys. Further examination of the role of lifestyle and environmental factors that shape the severity of the disease over the life-span is necessary.
Research is needed on an array of medical therapeutic, complementary and alternative therapies, and lifestyle options. Given that fibromyalgia often emerges in the mid-30s and can persist into postmenopause, a comprehensive understanding of treatment and support through these life stages is essential. Additionally, considering the impact on mobility, research should focus on identifying the environmental and social supports that could enhance quality of life.
MYALGIC ENCEPHALOMYELITIS/CHRONIC FATIGUE SYNDROME
As with fibromyalgia, diagnosing myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) can be challenging. The definition has evolved over several decades and most commonly uses ME/CFS, a descriptive label that captures the most specific symptoms. Patients experience significant adverse effect on their quality of life, reporting high levels of unemployment, permanent inability to work, and significant dependence upon others to carry out activities of daily living (Bested and Marshall, 2015).
Biological Factors
The etiology of ME/CFS remains unknown, despite observations that it may be initiated by an infection (Lacerda et al., 2019), stressful experiences, such as ACEs (Heim et al., 2006), or other events, including immunization,
anesthesia, and exposure to environmental pollutants, chemicals, and heavy metals. Physical trauma, such as a motor vehicle accident, fall, or surgery, may also trigger it (Carruthers et al., 2003). Researchers have estimated that a flu-like illness is the precursor in 50–80 percent of cases (Bested and Marshall, 2015). Some studies have implicated viral illness with the idea that it may be the result of a chronic Epstein-Barr viral infection or infection by other viruses, such as human herpes-viruses 6 and 7, cytomegalovirus, and enterovirus, but the evidence is inconclusive (Grach et al., 2023; Yancey and Thomas, 2012).
Nonetheless, since the December 2019 global emergence of coronavirus disease 2019 (COVID-19), interest has been renewed in the potential link with viruses. In a recent meta-analysis of publications that assessed the prevalence of chronic fatigue following COVID-19 infection, chronic fatigue syndrome (CFS) developed 4 weeks after the initial infection in 45.2 percent of 127,711 patients (Salari et al., 2022). Some studies have shown increased prevalence of chronic fatigue or other long COVID symptoms for women (Huang et al., 2021), including a study that has been retracted because of inconsistencies in data reporting, but others have found no gender associations (Carfì et al., 2020; Chopra et al., 2021).
Despite ongoing research addressing the influence of biological sex on ME/CFS, efforts are hindered by the lack of consistency related to definitions and diagnostic criteria that make it difficult to conduct clinical studies and estimate prevalence in both women and men (Lim et al., 2020). In general, being female appears to be a predictive risk factor (Lacerda et al., 2019; Lim et al., 2020; Reyes et al., 2003; Thomas et al., 2022), and results support a prominent role for sex gonadal hormones underlying this sex bias.
Sex Chromosome Effects
The contribution of sex chromosomal effects to ME/CFS is unknown. Similar to fibromyalgia, indications suggest that an autoimmune component may contribute to the development and progression of ME/CFS (Sotzny et al., 2018), but research is required to concretely establish immune mechanisms and whether the X chromosome is involved.
The few published studies attempting to identify genes associated with ME/CFS have focused mainly on autosomes, not sex chromosomes (Das et al., 2022; Gamer et al., 2023). One exception was a GWAS that identified 35 single nucleotide polymorphisms (SNPs) located on the X chromosome, along with 407 autosomal SNPs, as potential gene candidates for association with ME/CFS (Schlauch et al., 2016).
The Effects of Estrogens
Lines of evidence supporting a role for female gonadal hormones in the pathophysiology of ME/CFS include (1) lower mRNA expression of ER-β, but not ER-α, in ME/CFS patients (Gräns et al., 2007); (2) certain SNPs in CYP2D6, a gene involved in androgen and estrogen biosynthesis and metabolism, at a higher frequency in ME/CFS patients (Perez et al., 2019); and (3) metabolomics analysis using mass spectrometry showing reductions in androgens and progestins in ME/CFS patients compared to controls (Germain et al., 2020).
Specific Risk Factors in Women
Risk factors for ME/CFS in women are not well understood, and very few studies have examined those unique to women. The Nurses Health cohort identified specific risk factors for ME/CFS versus severe fatigue and found that a history of mononucleosis was not associated of ME/CFS. Various factors, such as smoking, caffeine and alcohol intake, were not associated with ME/CFS (Palacios et al., 2023). BMI was marginally associated with ME/CFS, and physical activity, hormone therapy, and menopause status had no association (Palacios et al., 2023). A literature review on how ME/CFS and other related conditions, such as long COVID, develop noted that reproductive milestones, such as pregnancy, and endocrine events, such as early age of menopause, were associated with ME/CFS (Pollack et al., 2023).
The effects of female gonadal hormones likely underlie the preponderance of ME/CFS in women. Hormonal changes during the menstrual cycle and menopause have been reported to worsen symptoms, and similar detrimental effects were observed when women were administered hormone-based birth control and hormone therapy (Allen, 2008; Thomas et al., 2022). Together, these clinical studies suggest that gonadal hormones affect how women experience symptoms. However, it is unclear if pregnancy positively or negatively affects symptoms, or if ME/CFS adversely impacts pregnancy and/or the postpartum period, as findings are limited and have produced contradictory results (Allen, 2008).
No consensus exists yet on whether hormone levels during the menstrual cycle differ between women with ME/CFS and controls. Smaller studies of women with ME/CFS reported higher odds of gynecological conditions, including endometriosis, menstrual abnormalities, pelvic pain unrelated to menstruation, early hysterectomy, and gynecological surgeries, suggesting a relationship with sex hormones, but further research is needed (Boneva et al., 2011, 2015; Pollack et al., 2023). Two small, independent studies have demonstrated differences in estradiol levels (Boneva et al., 2015; Studd and Panay, 1996) and levels of progesterone and progesterone
metabolites (Murphy et al., 2004), but a third study found no notable differences in these hormones, nor in follicle stimulating hormone and luteinizing hormone (Cevik et al., 2004). To resolve this question, researchers should conduct follow-up studies in larger cohorts.
Condition Presentation and Diagnosis
The literature has 25 clinical definitions for this complex chronic condition, indicating a lack of consensus across disciplines (Lim and Son, 2020). These definitions are impacted by etiology, pathophysiology, and exclusion of other conditions (Lim and Son, 2020). Some investigators have favored the Canadian Consensus Criteria, which have been validated using online tools to assess symptoms and function (Bested and Marshall, 2015) and demonstrated to more effectively distinguish ME/CFS from other causes of fatigue, such as depression. Following the exclusion of other diagnoses associated with fatigue and muscle tenderness, patients are assessed based on the following criteria: fatigue considered pathological for a period of 3–6 months, malaise, sleep disorder, pain, and cognitive impairment. Moreover, two symptoms must fall into at least two of the following domains: immune, neuroendocrine, or autonomic (Bested and Marshall, 2015). Given the reliance on clinical symptoms without specific biomarkers, it is not surprising that patients are often not diagnosed and regularly face disbelief that their symptoms are valid.
It has been estimated that as many as 80 percent of patients who are symptomatic have struggled to be diagnosed, largely because providers have not been appropriately educated. ME/CFS has not been as well represented in the medical curriculum compared to other conditions, and targeted continuing education options may close this gap and improve earlier diagnosis (Bested and Marshall, 2015; Brimmer et al., 2009). A number of other conditions may also be reported, such as fibromyalgia, multiple chemical sensitivity, orthostatic intolerance, irritable bowel syndrome, irritable bladder syndrome, interstitial cystitis, sicca syndrome, temporomandibular joint syndrome, migraine headache, allergies, thyroiditis, Raynaud’s phenomenon, and prolapsed mitral valve (Bested and Marshall, 2015).
Treatment and Management
Given the complexity of the symptoms of ME/CFS, treatment requires a customized, integrated approach. Attention must first be given to any comorbid conditions, such as depression, sleep dysfunction, and pain. Research has demonstrated that cognitive behavior therapy and exercise are effective (Turner-Stokes and Wade, 2020). However, according to guidelines published by the National Institute for Health and Care Excellence
in the United Kingdom in 2020, these therapies may not be helpful despite published evidence to the contrary (Turner-Stokes and Wade, 2020). These guidelines were based on quality criteria that assessed the evidence for these therapies, which may not fit well when assessing complex conditions such as ME/CFS. Long-term observational trials would be better suited to assess the efficacy of interventions for ME/CFS (Turner-Stokes and Wade, 2020).
The effect on the lives of patients with ME/CFS is significant. They often feel stigmatized when presenting with an array of symptoms and not believed when sharing their symptoms with providers. Somatization, the conflation of physical and psychological explanation for symptoms, continues to require greater recognition in their care (Wessely, 1997). Providers are not well educated regarding diagnostic criteria, which are often overwhelmed by other comorbidities (Bateman et al., 2021; Bested and Marshall, 2015). Treatable active diseases that contribute to symptoms such as fatigue, pain, sleep dysfunction, and cognitive dysfunction must be ruled out before making a diagnosis of ME/CFS (Bested and Marshall, 2015). Moreover, treatment is embedded in controversy, although those individuals who are guided through a well-structured rehabilitation program may improve (Chambers, 2006; Wearden et al., 2010). The prognosis is variable and dependent upon the severity of the condition at onset and the stage of diagnosis (Bested and Marshall, 2015).
Earlier diagnosis and appropriate management should lead to a better outcome. On average, 5 percent of patients recover, 40 percent improve, 8–30 percent return to work, and 5–20 percent worsen (Bested and Marshall, 2015). Ongoing research exploring mechanisms such as redox imbalance (Paul et al., 2021), genetics, and immune system dysfunction may offer diagnostic and therapeutic paths (Bested and Marshall, 2015).
Disparities
The research on racial and ethnic disparities in ME/CFS is scant. An older meta-analysis of CFS reported on racial and ethnic differences in prevalence, symptom severity, and coping and illness behaviors. Three studies suggested that prevalence was greater in Black and African American and Alaska Native groups. The findings were mixed; greater symptom severity was reported in Latinas compared to Black and African American and White women, but another study indicated that Black and African American individuals reported more severe fatigue than White individuals. However, these findings are based on limited studies available (Dinos et al., 2009).
Research Gaps
Studies are needed to determine the underlying etiological mechanisms of ME/CFS in women and whether sex chromosome and hormonal effects
play a role. The multiple consensus criteria make it difficult to define this condition. A report from the Institute of Medicine14 recommended a new label, but this recommendation does not appear to have had much traction based on a review of the literature. One recommendation was to review the diagnostic criteria 5 years after publication, and the committee suggested that funding may be provided by “nonconflicted sources such as the Agency for Healthcare Research and Quality through its Evidenced-based Practice Centers process and foundations” (IOM, 2015). It is unclear if such a review process has been undertaken. The lack of established diagnostic tools and biomarkers that can rule out other similar chronic conditions present major challenges in properly diagnosing individuals who are experiencing clinical symptoms associated with ME/CFS. Other research needs include addressing the lack of relevant education in physicians and other providers, developing a better understanding of the basic mechanisms specific to it, and conducting long-term observational studies to better understand which patient-centered interventions are the most useful.
ALZHEIMER’S DISEASE
Alzheimer’s disease (AD) causes neurocognitive deficits. Despite advancements in the field characterizing genetic associations, delineating the disease process, identifying diagnostic and prognostic biomarkers, and assessing possible treatments, it still has no cure (Ferretti et al., 2018). Research into disease mechanisms has helped identify pathological biomarkers to gauge disease risk and monitor progression. Although AD is largely a heterogeneous disease, with high variability in timing, type of symptom manifestation, and progression stemming from multiple etiological sources, women have notable differences in risk factors, symptoms, and progression versus men.
Biological Factors
The three main underlying factors for increased AD risk are age, variation in the apolipoprotein E (ApoE) gene, and sex, with age being the most significant (Valencia-Olvera et al., 2023). The contribution of the aging process to AD is beyond the scope of this report. Even though the age-specific incidence rates are similar in women and men, the mechanisms underlying the incidence often vary by sex. Both the frequency and the strength of
___________________
14As of March 2016, the Health and Medicine Division of the National Academies of Sciences, Engineering, and Medicine (the National Academies) continues the consensus studies and convening activities previously carried out by the Institute of Medicine (IOM). The IOM name is used to refer to reports issued prior to July 2015.
the effect of a given risk or protective factor may be different in men and women (Rocca et al., 2014).
Sex Chromosome Effects
Limited data exist to support a direct association of sex chromosome effects with AD, but a few studies indicate an influence of X chromosome number and X chromosomal genes. For example, X chromosome loss may be an underlying factor. One research group demonstrated that the likelihood of X chromosome loss in somatic cells increases 100-fold in women over age 65 compared to girls under age 16 (Russell et al., 2007), which may influence neurodegeneration and aging in the brain (Guo et al., 2022). Specific to AD, the rate of X chromosome loss increased by twofold in the hippocampus and cerebral brain regions of patients (Yurov et al., 2014). Conversely, an extra X chromosome may be neuroprotective, as illustrated by a preclinical study showing that a second X chromosome in a transgenic mouse model for AD reduced mortality and improved cognitive deficits (Davis et al., 2020).
Research has linked several genes on the X chromosome to AD. For example, KDM6A, which encodes a histone demethylase, is associated with cognitive function, as loss-of-function mutations in humans are linked to intellectual disability (Miyake et al., 2013), and a knockout of the same gene in mouse models leads to memory impairments (Tang et al., 2017). Studies have tied a SNP in the PCDH11X gene, known to play a role in CNS function, to higher risks of AD in women (Carrasquillo et al., 2009). GWAS did not replicate this finding (Beecham et al., 2010; Lescai et al., 2010; Miar et al., 2011; Wu et al., 2010), but more recent GWAS confirmed the association (Jiao et al., 2015; Zeng et al., 2021). These inconsistencies may result from differences in cohort size, ethnicity of the study population, environmental factors, and computational method (Bajic et al., 2019). Analysis of data from the Alzheimer’s Disease Neuroimaging Initiative study identified 15 additional SNPs, located within or near genes on the X chromosome—some of which other studies have implicated in neuropsychiatric disorders, brain development, or intellectual disability—as significantly associated with quantitative biomarkers of AD (Wang et al., 2023b). Collectively, the highlighted studies support contributions of sex chromosome effects on the susceptibility of women to AD, although research is needed to discern the individual contributions of sex chromosomes and sex hormones.
The Effects of Estrogens
Estrogens exert neuroprotective effects on the brain, and many studies have shown that reductions in estrogens are associated with a higher
risk of AD in women (Pike, 2017). For example, surgically induced menopause and the resulting abrupt drop in estrogens significantly increase the risk of Alzheimer dementia (Bove et al., 2014; Phung et al., 2010; Rocca et al., 2007). A diagnosis of AD in older women has been correlated with low levels of estrogens in the brain (Rosario et al., 2011). The association between AD and estrogens is also evident in mouse models. Removing the ovaries caused significant increases in beta-amyloid deposits (Jayaraman et al., 2012; Petanceska et al., 2000), and impaired behavioral performance (Carroll et al., 2007) in female animals.
Evidence also suggests that estrogens interact with ApoE, influencing dementia risk in women, especially those carrying the AD-predisposing ApoE4 gene variant that some studies have estimated occurs in approximately 55 percent of the U.S. population (Valencia-Olvera et al., 2023; Ward et al., 2012). Taken together, these findings point to a role of estrogens as a mediator of AD in women.
Studies have shown that ApoE variants appear to modify the role of estrogens in women’s risk of developing AD. ApoE2, the least prevalent variant, is associated with reduced risk (Liu et al., 2013; Serrano-Pozo et al., 2021), but ApoE4 increases the risk by 15-fold in individuals with two copies (Husain et al., 2021; Troutwine et al., 2022; Valencia-Olvera et al., 2023). Several studies have shown that the negative consequences of ApoE4 on brain structure and function before developing dementia are more severe in women compared to men (Altmann et al., 2014; Holland et al., 2013; Liu et al., 2010).
Some evidence suggests that the effect of sex hormones on AD risk may begin in utero. Some studies suggest that women who had an increased exposure to male sex hormones in utero may develop lower risks for dementia later in life (Jiang et al., 2020; Luo et al., 2020). In mice, feminization of males in utero leads to an increase in beta-amyloid, and masculinization of females in utero leads to a reduction in beta-amyloid accumulation (Carroll et al., 2010).
Other Biological Factors and Mechanisms
In the 1990s, research identified mutations in three genes involved in beta-amyloid formation that are causative factors for familial, autosomal dominant, early-onset forms of AD: amyloid precursor protein (APP), presenilin-1, and presenilin-2 (Goate et al., 1991; Lambert et al., 2023; Levy-Lahad et al., 1995; Migliore and Coppedè, 2022; Sherrington et al., 1995). Familial AD affects approximately 1 percent of individuals with the disease, with the remaining 99 percent of cases tied to susceptibility genes and environmental factors (Migliore and Coppedè, 2022; Reitz et al., 2020). Although several transgenic mouse models for familial AD have
demonstrated that females have greater deficits than males (Dubal et al., 2012; Ferretti et al., 2018), it is unknown whether similar differences are present in human familial AD studies.
ApoE4, one of the first characterized genetic susceptibility factors, occurred at a three- to fourfold higher frequency in patients with AD (Strittmatter et al., 1993) that is sex dependent (Neu et al., 2017). Clinical studies have demonstrated that among ApoE4 carriers, women are more likely than men to develop AD (Valencia-Olvera et al., 2023). Specifically, female ApoE4 carriers have increased risks for mild cognitive impairment at ages 55–70 and Alzheimer dementia aged 65–75 (Ferretti et al., 2018; Neu et al., 2017). Clear sex differences were also observed in an ApoE4 transgenic mouse model, with females exhibiting more severe cognitive impairments and faster disease progression than males (Bour et al., 2008; Ferretti et al., 2018; Reverte et al., 2012).
The strong sex dependence associated with ApoE4 suggests that its physiological effect is estrogen dependent and only emerges during menopause, when levels of estrogen fall. The cellular and physiological changes that ApoE4 modulates accumulate over many years before cognitive and behavioral alterations become detectable. ApoE4 is also linked to other conditions and diseases, including frailty, higher levels of LDL, and CVD, with specific risks depending on sex (Bennet et al., 2007; Deelen et al., 2019; Valencia-Olvera et al., 2023); the ApoE2 variant is linked to greater longevity in both sexes (Gerdes et al., 2000; Schächter et al., 1994) and lower CVD risk in women compared to men (Lahoz et al., 2001). As ApoE is the major lipoprotein component expressed in the CNS (Holtzman et al., 2012; Liu et al., 2013; Valencia-Olvera et al., 2023) and involved in cholesterol uptake and sex hormone production, it is not surprising that dysregulation of this protein will have downstream consequences on both cardiovascular and neuronal health.
GWAS have identified over 90 gene locations as significantly associated with AD risk (Lambert et al., 2023). Some of these genetic variants are highly expressed in microglia (Guerreiro et al., 2012; Jonsson et al., 2013; Lambert et al., 2023; Sims et al., 2017), supporting the idea that microglial immune activation contributes to the disease process. GWAS has revealed other genetic variants restricted to certain ancestries. For example, one variant is associated with higher AD in people with African ancestry (Kunkle et al., 2021; Lambert et al., 2023; Logue et al., 2011; Reitz et al., 2013). Sex stratification of GWAS data has identified novel sex-specific associations with specific chromosomal locations. For example, a region on chromosome seven is associated with preserved hippocampal volume, better cognitive function, and later age of onset of AD in men, although research has not identified the genes and mechanisms related to neuroprotection (Dumitrescu et al., 2019). As AD is complex and multifactorial,
genetic variants associated with risk identified from GWAS could be used to generate polygenic risk scores that are sex and ethnicity specific and may serve as a diagnostic predictor and prognostic indicator. This idea is illustrated in a study showing that a higher polygenic risk score based on 85 SNPs is linked to faster rates of tau accumulation—another hallmark of AD—in non-Hispanic White individuals (Rubinski et al., 2023).
Biomarkers for AD
Sex can affect measuring or interpreting of potential AD biomarkers via several mechanisms. For some markers, a measure such as the concentration of a protein in a bio-fluid or volume of a specific brain region, may vary by sex. For others, it is the interpretation that may vary by sex, depending on whether the biomarker is used for diagnosis or prognosis. When interpreting sex differences in biofluid biomarkers, it is important to consider sex differences in the permeability of the blood–brain barrier and the binding of biomarkers to blood proteins (Mielke, 2020). Unfortunately, many studies have considered sex a confounding variable and adjusted for it in their analyses rather than studying it as a major effect modifier (Mielke et al., 2022a).
The cerebrospinal fluid concentration of neurofilament light chain is higher in males than in female individuals across the entire life. It is a marker of large-caliber, subcortical axonal degeneration that is not specific to AD. Therefore, it has been suggested that the reference intervals should be sex specific. In contrast, female individuals have higher concentrations of total tau in the cerebrospinal fluid. Both biomarkers indicate greater neurodegeneration, but the explanation for these sex differences remains unknown (Bridel et al., 2019; Mielke et al., 2022b).
Another example is the higher concentration of blood phosphorylated tau in men, although excluding individuals with chronic kidney disease from the analysis attenuated this difference. Chronic kidney disease was more common in men in that study and may have increased the plasma levels of phosphorylated tau (Mielke et al., 2022b). This example emphasizes the importance of considering comorbidities when interpreting blood biomarkers.
As an example of sex differences in imaging biomarkers, a 2023 study in cognitively unimpaired individuals suggested that women may have higher tau measured by PET in seven brain regions in the temporal, parietal, and occipital lobes compared to age-matched men (Coughlan et al., 2023). However, the findings have not been replicated.
As AD occurs more frequently in women, several large research initiatives have explored whether sex differences related to biomarkers exist (Buckley et al., 2019; Cavedo et al., 2018; Deming et al., 2018). Such information could be useful in determining differential susceptibility of women
to AD, and sex-specific prognostic features and therapeutic responses (Toro et al., 2019).
Two common, highly validated biomarkers indicative of the pathophysiological process of AD are beta-amyloid and tau proteins. In cognitively unimpaired individuals, beta-amyloid formation precedes and modulates the phosphorylation of tau protein (Jack et al., 2013; Milà-Alomà et al., 2022), which is linked to the initial stages of cognitive decline (Hanseeuw et al., 2019; Ossenkoppele et al., 2021, 2022). However, certain individuals with high levels of beta-amyloid do not develop markers for phosphorylated tau and experience cognitive decline (Josephs et al., 2022; Nordberg, 2015; Roberts et al., 2018). A study revealed a possible mechanistic explanation, implicating astrocyte activation as a modulator of beta-amyloid-mediated phosphorylation of tau. Higher plasma levels of a surrogate marker for astrocyte activation correlated with beta-amyloid-dependent tau phosphorylation in cognitively unimpaired individuals, with a more robust association in men than women (Bellaver et al., 2023). Clinical studies have also shown that, compared to men, women exhibit higher tau-related brain pathology (Buckley et al., 2019; Palta et al., 2021) and plasma levels of phosphorylated tau that correlated with higher rates of converting to dementia in beta-amyloid-positive individuals (Tsiknia et al., 2022). Collectively, these findings support that in the early stages of the disease process, nuanced, sex-specific differences are related to mechanisms upstream of beta-amyloid’s effects on tau phosphorylation.
In the advanced stages of AD, postmortem studies have not found sex differences in beta-amyloid in affected brain regions. Measuring it by PET brain imaging and biochemical protein assays of cerebrospinal fluid samples in living patients yielded the same results (Ferretti et al., 2018). Similarly, studies using the same types of samples and methods found no consistent differences by sex in levels of neurofilament tangles—another physiologic hallmark of AD—and tau phosphorylation. However, some studies have shown that women have more neurofilament tangles in certain brain regions compared to men (Barnes et al., 2005; Salehi et al., 1998). It remains unclear why these brain regions are specifically affected in women.
Studies have also observed sex differences in atrophy rates of different brain regions at all AD stages, with faster atrophy rates of 1–1.5 percent per year in women during the early stages of AD and when full-blown AD dementia develops (Ardekani et al., 2016; Ferretti et al., 2018; Hua et al., 2010; Lin et al., 2015; Skup et al., 2011). In addition, a more complex and widespread pathological profile, measured as diffuse plaques and neurofilament tangles across four brain regions, was associated with greater than 20-fold increase in the odds of developing dementia in women compared to a threefold increase in men (Barnes et al., 2005). Other studies have shown that both beta-amyloid and total tau in cerebrospinal fluid are associated
with a greater extent of hippocampal degeneration and rapid cognitive decline in women (Ferretti et al., 2018).
Overall, multiple pathological markers of AD may correlate with disease risk and parallel the accelerated cognitive decline in women. With no way to reverse AD, a possible management approach would be to implement early detection by monitoring blood and cerebrospinal fluid biomarkers and imaging the brain for gross pathological changes. This could change the disease course by creating a larger window of opportunity for therapeutic and lifestyle interventions that could be initiated before irreversible brain pathological changes occur (van der Flier et al., 2023).
Specific Risk Factors in Women
Reproductive Milestones
Cognitive decline in women seems to occur after menopause, with marked dysfunction most evident after premature or early menopause. Studies have shown that the drastic change in cyclical levels of estrogen resulting from the surgical removal of the ovaries and the resulting early menopause makes women more vulnerable to cognitive impairments. In addition, reductions in progesterone and its metabolite, allopregnanolone, during menopause, have been linked to neuroinflammation and risk of AD (Yilmaz et al., 2019), while replacement with exogenous allopregnanolone improves pathological features (Irwin and Brinton, 2014). It is thus important to understand the neuroprotective effects of cyclical estrogens and other female sex hormones; if they are helpful in preserving cognitive function, hormone therapy should be used in women who experience premature or early menopause (Kaunitz et al., 2021).
The rate of decline is also faster in women compared to men at older ages. Several factors may account for this, with one being the propensity for older women to be socially isolated. Several groups have proposed that social and mental activities stimulate neural activity and increase synaptic connections, which help preserve cognitive function as women age.
Women are more susceptible to various AD risk factors compared to men, with differences in both the frequency and the strength of the effect of a given risk or protective factor (Rocca et al., 2014). Certain conditions occurring specifically in women, such as gestational diabetes and preeclampsia during pregnancy (Buhimschi et al., 2014; Carey et al., 2024; Fields et al., 2017; Zhang et al., 2024) and premature or early menopause (Bove et al., 2014; Karamitrou et al., 2023; Rocca et al., 2007), are also associated with higher risks of developing AD and other dementias. One study found that women with hypertension in midlife had an increased risk of dementia compared to men, despite higher prevalence of hypertension
in men, indicating that vascular and metabolic pathways contribute to this disease (Gilsanz et al., 2017; Lorius et al., 2015).
Research has established that sex hormones play a neuroprotective effect in the brain throughout a woman’s lifespan. Analysis of several studies in a recent scoping review suggest that hormonal contraceptive use at younger ages was linked to a lower risk of developing AD later in life (Gregory et al., 2023); however, more data from young women using hormonal contraceptives and longitudinal follow-up are needed. Conversely, hormonal fluctuations and the decline in estrogens in women during the menopause transition may increase the risk of brain changes associated with AD (Rahman et al., 2019). Brain imaging findings indicate a progressively increased risk in women undergoing the menopause transition and suggest that the effect of estrogens outweigh the effects of chronological aging in the female brain by several years, if not decades, before possible clinical symptoms emerge (Scheyer et al., 2018). One imaging study found that menopausal transition has significant effects on brain structure—particularly in regions involved in higher-order cognitive processes—connectivity, and energy metabolism that were not observed in age-matched men and may increase AD risk (Mosconi et al., 2021). Compared to spontaneous menopause, early and surgical menopause are associated with more severe cognitive impairment and higher risk of dementia (Jett et al., 2022). Furthermore, fluctuations in estrogen during perimenopause have been proposed to trigger detrimental changes in metabolic and inflammatory pathways that contribute to AD (Lopez-Lee et al., 2024). Additional studies are needed to understand how fluctuations in other hormones during this reproductive stage affect AD risk.
Data from the Mayo Clinic Study of Aging show that sex-specific comorbidities can predict conversion from normal cognition to mild cognitive impairment. Susceptibility factors included midlife elevated circulating lipid levels, diabetes, and hypertension in women and obesity in men (Pankratz et al., 2015). Gender also plays important roles in AD etiology. For example, lower levels of income and education were more common in women and may increase the risk of AD (Ferretti et al., 2018; Petersen et al., 2010; Rocca et al., 2014; Sharp and Gatz, 2011; Vemuri et al., 2016). Similarly, the social isolation faced by women as they age (Stahl et al., 2017) may contribute to AD, suggesting that mental and social activities that trigger new or maintain established synaptic connections are important for maintaining healthy brain function (Daly, 2023; Sommerlad et al., 2023).
Symptom Presentation and Diagnosis
Depending on the stage of the disease, women and men exhibit sex-specific tendencies related to cognitive performance and behavior.
For example, in preclinical, asymptomatic AD, women scored higher on verbal tasks, whereas men scored better in visuospatial and motor coordination tasks (Ferretti et al., 2018). Among AD patients, women scored lower than men in verbal memory, fluency, and naming tasks, even after controlling for confounding factors such as education, age, and depression (Ferretti et al., 2018). Generally, before menopause, women appear to be protected from AD compared to men, but they display higher rates of cognitive decline and brain atrophy after menopause (Ferretti et al., 2018).
Disease progression also appears to be faster in women. In the longitudinal Alzheimer’s Disease Neuroimaging Initiative study, women with prodromal AD exhibited faster cognitive decline (Petersen et al., 2010). Limited studies with small sample sizes have noted sex-specific behavioral symptoms, with women exhibiting depression, isolation, mood swings, delusions, and affective and manic symptoms, whereas men tended to display apathy, agitation, and abusive and socially inappropriate behavior (Ferretti et al., 2018; Hollingworth et al., 2006; Ott et al., 1996).
Treatment and Management
Despite better representation of both sexes in clinical trials testing drug therapies for AD, published reports typically do not include sex-based data stratification and correlations with outcomes (Ferretti et al., 2018). Applying sex-based analyses to clinical trial data could identify sex differences in drug safety and efficacy and lead to better interventions tailored to men and women (Legato et al., 2016). Currently, there is no therapy that can reverse or cure AD, although three main drug classes are commonly administered to treat AD symptoms, with several studies indicating variable responses between men and women.
Cholinesterase Inhibitors
Cholinesterase inhibitors prevent the breakdown of the neurotransmitter acetylcholine at neuronal synapses, thereby prolonging neural activity. Most clinical trials assessing adverse effects for the cholinesterase inhibitors, donepezil, rivastigmine, and galantamine, did not analyze data disaggregated by sex (Mehta et al., 2017). The two studies found no clear differences in safety and efficacy (Canevelli et al., 2017; Haywood and Mukaetova-Ladinska, 2006). In contrast, studies with another cholinesterase inhibitor, rivastigmine, did find sex differences in clinical outcomes—it delayed progression from the initial appearance of symptoms to later stages of AD in women (Ferris et al., 2009). However, its benefits were less evident in women with advanced dementia (Haywood and Mukaetova-Ladinska, 2006; MacGowan et al., 1998).
Other studies have suggested that in women, the ApoE genotype modulates the response to certain cholinesterase inhibitors, such as tacrine (Farlow et al., 1998). These differences may arise from physiological differences in the cholinergic system in men and women (Counts et al., 2011; Ferretti et al., 2018; Rhodes and Rubin, 1999) or drug interactions with sex hormones, ER 1, and other sex steroid regulatory factors (Ferretti et al., 2018). Cholinesterase inhibitors are the first-line treatment for cognitive deficits, but as efficacy wanes with the progressive neuronal loss characteristic of AD, therapies preserving neuronal function need to be developed.
Intranasal Insulin
Insulin has emerged as a promising treatment because of its pleiotropic effects on multiple organ systems involved in AD pathogenesis, including the vasculature via modulation of glucose and lipid metabolism and inflammation and the nervous system via promotion of neural connections (Kellar and Craft, 2020). Its mechanism of action in AD may also involve modulating beta-amyloid regulation and tau phosphorylation (Kellar and Craft, 2020). Some clinical trials have noted sex differences in intranasal insulin administration. In a secondary analysis of a Phase II clinical trial in patients with mild cognitive impairment, women performed better on activities of daily living, whereas men showed improvements in the delayed story recall task (Claxton et al., 2013). However, the AD-predisposing ApoE4 variant eliminated the benefits of intranasal insulin in women carriers, who exhibited worse outcomes, whereas men carriers showed improvements (Claxton et al., 2013).
Anti-Amyloid Antibody Therapy
In July 2023, FDA approved the first drug to slow AD progression. Lecanemab (Leqembi®) is an antiamyloid monoclonal antibody that reduces amyloid plaques in brain imaging measurements (van Dyck et al., 2022). However, it remains unclear what its effect will be in the general population. It has several disadvantages, including some important contraindications, side effects, high cost, and the requirement of supervised administration and monitoring of side effects via brain imaging (Pittock et al., 2023). In addition, concern arose that the therapeutic effect may be weaker in women than men (Kurkinen, 2023; Valenzuela and Pascual-Leone, 2023).
Hormone Therapy
Hormone therapy for women, involving estrogen alone after hysterectomy or estrogen combined with a progestogen otherwise, is currently
not recommended to treat AD or mild cognitive impairment or prevent dementia in women who have experienced natural menopause (North American Menopause Society, 2022). However, it is currently recommended for women who experienced premature or early menopause. These recommendations are based on observational studies of the risk of AD or dementia in women who underwent premenopausal removal of their ovaries in the premature or early age range (Kaunitz et al., 2021).
It is important to note that different types of estrogens, as well as different routes of administration, may result in variable outcomes related to cognitive function. For example, estradiol and estrone differentially alter cognitive function in post-menopausal women (Hogervorst et al., 2000). Other studies indicate that oral estradiol is converted to estrone, while transdermal estradiol is not (Lobo and Cassidenti, 1992; Tebbens et al., 2022). Consideration of how estrogens are given as treatments has important ramifications and requires further study, especially since compared to oral estrogen formulations, transdermal estradiol has been shown to improve cognition and increase brain white matter in postmenopausal women (Kantarci et al., 2016; Wharton et al., 2011).
Disparities
Comprehensive data are lacking on the impact of AD that consider both sex and their ancestry and ethnicity. However, an analysis of administrative Medicare claims data from a large population-based cohort, which deliberately included a significant number of women and racial and ethnic groups that had been less studied, revealed a 17 percent higher risk of late-onset AD in women than men, after adjusting for age. Incidence rates for both AD and related dementias was provided for women by race and ethnicity. The age-standardized incidence rate for AD per 1,000 person-years was highest among African American (9.54), followed by Latina (7.76), Native Hawaiian (7.65), White (6.85), and Japanese American women (6.60) and lowest among Filipino women (4.58). With respect to AD and related dementias, the age-standardized diagnostic incidence rate per 1,000 person-years was highest for African American (22.92), followed by Native Hawaiian (19.33), Latina (16.79), White (16.38), Japanese American (14.76), and lowest among Filipino women (12.53) (Lim et al., 2022).
The 2020 U.S. Census indicates that the population is more racially and ethnically diverse than a decade ago. Disparities in dementia have been observed in African American, American Indian and Alaska Native, and Latino/Latina individuals. In addition, disparities affecting caregivers and families of patients with dementia are evident (Aranda et al., 2021). Caregivers of those groups have found differences in overall well-being, and studies suggest that they also had poorer health (Aranda et al., 2021).
The racial and ethnic group differences in the incidence and prevalence of AD and related dementias among women is an important research gap that needs to be addressed.
Research Gaps
Much has been learned about AD, but a number of gaps remain. Although studies have pointed to the role of sex chromosome effects on the susceptibility of women to AD, additional research is needed to discern the individual contributions of sex chromosomes and sex hormones. Identifying sex differences in a range of AD biomarkers and their interpretation is another research gap that deserves attention. As AD is no longer viewed as a disease solely associated with aging, it is important to identify and develop preventive measures to address changes in brain biochemistry throughout the life course. By the time AD symptoms are evident, it may be too late to reverse the damage that has occurred in the brain. The only possibility to help patients is to manage the disease. Therefore, it is important to catch the early manifestations by assessing social determinants of health and lifestyle factors and using biomarkers and imaging. Research on how AD differentially affects the lives of racially and ethnically minoritized women is also needed.
OSTEOPOROSIS
Osteoporosis is a chronic condition that affects women more than men and is associated with bone fragility and repeated fracture risk (Weaver et al., 2017). Hip fractures account for a large proportion of the morbidity and mortality associated with osteoporotic fractures. Biological factors, prevention, diagnosis, treatment, and disparities associated with osteoporosis in women are described next.
Biological Factors
Genetic factors substantially influence an individual’s bone strength, microarchitectural properties, and risk of osteoporosis and fractures (Mäkitie et al., 2019). Bone mineral density (BMD) is highly heritable, as demonstrated by twin studies showing that one’s genetic makeup determines up to 80 percent of its variance (Mäkitie et al., 2019). Studies have identified specific genes associated with osteoporosis risk, such as those involved in bone metabolism and regulation of calcium absorption. A family history of osteoporosis or fractures is known to increase fracture risk, indicating a strong genetic component. The most common genetic cause is osteogenesis imperfecta, an inherited disorder also known as “brittle bone disease”
(Mäkitie et al., 2019). A meta-analysis has identified several important genes involved in osteoporosis (Hasan et al., 2021) and several genes and pathways that may be possible to target to develop new treatments for osteoporosis.
Sex Chromosome Effects
Recent findings have identified a gene on the X chromosome that is associated with osteoporosis, plastin 3 (PLS3). This gene codes for a protein involved in the formation of actin bundles and also in the maintenance of bone health. This is supported by human genetic studies demonstrating that variants in this gene caused osteoporosis and osteoporotic fractures in members of 10 different families (van Dijk et al., 2013). In vivo studies using PLS3 knockout mice also led to phenotypic features of osteoporosis, and in vitro cellular experiments indicated the involvement of this gene in osteoclast formation (Neugebauer et al., 2018). More recently, research has linked PLS3 polymorphisms with early-onset osteoporosis in young individuals (Costantini et al., 2020), and mutations in this gene correlate with osteoporosis in postmenopausal women (Wolff et al., 2021).
The Effects of Estrogens
Estrogens maintain bone homeostasis by regulating the metabolic activity of the two main cell types found in bone—osteoblasts, which release bone growth factors, and osteoclasts, which promote bone resorption (Biason-Lauber and Lang-Muritano, 2022). Estrogens preserve bone mass by interfering with the action of inflammatory cytokines that promote osteoclast development (D’Amelio et al., 2008; Jilka et al., 1992; Pietschmann et al., 2016; Weitzmann et al., 2002) and inducing apoptosis, or programmed cell death, of osteoclasts (Kameda et al., 1997). The gradual decline of estrogens during menopause may therefore increase osteoclast activity, leading to higher bone turnover and breakdown and potentially osteoporosis.
As explained in Chapter 2, estrogens act on three different receptors (ER-α, ER-β, and GPR30) to affect numerous cell and tissue types. Given the importance in mediating the effects of estrogens, researchers proposed that ERs could determine osteoporosis risk (Gennari, 2005). Studies have found that genetic variants in two of these receptors occur in women from different racial and ethnic backgrounds, with genetic variants in ERα associated with lower BMD and increased risk of osteoporosis (Gennari, 2005). Although the mechanism by which this occurs is unknown, one group proposed that these variants alter estrogen sensitivity (Khosla et al., 2004). Other studies support the idea that ERα is linked to osteoporosis,
including one in which a man with an ERα mutation had features of osteoporosis (Smith et al., 1994) and in an ERα knockout mouse model that exhibited impaired bone growth (Lindberg et al., 2001; Vidal et al., 2000).
Other Biological Factors and Mechanisms
Osteoclasts and osteoblasts are involved in bone remodeling; they resorb old bone and then form new bone through a well-coordinated mechanism regulated by different factors, such as systemic hormones, local growth factors, cytokines, chemokines, adhesion molecules, and extracellular matrix proteins (Kenkre and Bassett, 2018). These factors are secreted by bone surface cells and produced by osteocytes in the bone matrix, which are the cell type that, together with bone lining cells, is involved in bone remodeling (Robling and Bonewald, 2020).
In addition to hormonal changes, other factors involved in osteoporosis include aging, oxidative stress, and inflammation. Evidence is increasing that oxidative stress is elevated with age, and that the decline in estrogens during menopause is often accompanied by increases in reactive oxygen species and proinflammatory cytokines. The production of reactive oxygen species occurs in parallel with inflammation and is thought to mediate the inflammatory process. These factors influence the functional activity of bone cells and consequently, bone homeostasis. Both oxidative stress and inflammation contribute to the development of osteoporosis by preventing the differentiation of bone-forming osteoblasts, inducing the differentiation and activity of bone-degrading osteoclasts, and enhancing apoptosis of osteocytes. In addition, there are bidirectional influences between certain microRNAs(miRNA) with oxidative stress and inflammation. Reactive oxygen species can alter miRNA expression, and conversely, miRNAs can regulate reactive oxygen species production and inflammatory processes (Iantomasi et al., 2023).
Researchers have used various -omics technologies, including genomics, transcriptomics, epigenomics, proteomics, and metabolomics, to identify the biomolecules involved in the pathogenesis of osteoporosis. The use of multiple -omics approaches can yield a better and more integrated understanding of the complex pathogenesis that could potentially lead to advancements in personalized disease risk prediction, intervention, and treatment, as well as drug development or repurposing (Yang et al., 2020b).
Specific Risk Factors in Women
Reproductive Milestones
Bone mass accumulates progressively from birth, through childhood, and into adolescence, typically reaching 90 percent of peak by late adolescence (Loud and Gordon, 2006; Pitts, 2022). The greatest increase, around
26 percent, occurs during the 4-year pubertal period coinciding with peak growth rate in height (Loud and Gordon, 2006). Genetic factors are crucial, accounting for about 60–80 percent of achieving peak bone mass (Loud and Gordon, 2006; Pitts, 2022). Other factors include hormonal status, nutritional status, physical activity level, overall health, medication use, and illicit substance use (Loud and Gordon, 2006; Pitts, 2022).
Estrogens play a significant role during the pubertal growth spurt by rapidly increasing bone mass and during adolescence to attain maximal peak bone mass (Pitts, 2022). Estrogens, acting via ERs on bone, promotes osteoblast survival while exerting a pro-apoptotic effect on osteoclasts. This results in a positive net balance favoring bone growth. Other hormones influential in this process include insulin-like growth factor, growth hormone, and sex hormones. Adolescent girls with untreated hormonal deficiencies are at risk of low bone density (Pitts, 2022).
Adequate calcium intake is critical for optimal mineralization of bone and cartilage matrix. During the pubertal growth spurt, intestinal calcium absorption increases (NIH, 2024). Vitamin D is also essential, stimulating intestinal calcium absorption and regulating calcium homeostasis (Fleet, 2022; Loud and Gordon, 2006). Adolescents lacking adequate dietary vitamin D should take a supplement (Society for Adolescent Health and Medicine, 2013). Prolonged deficiency of calcium and vitamin D can lead to rickets, particularly concerning in cases of malabsorption where supplementation is advised (NIH, 2024).
Pregnancy constitutes a natural physiological process linked with bone loss (Scioscia and Zanchetta, 2023). Although the precise pathophysiology remains unclear, researchers have hypothesized that molecular factors released during lactation, excessive calcium loss in breast milk, diminished postpartum levels of estrogen, and genetic factors may contribute. Additional risk factors include a family history of the condition, reduced physical activity, smoking, poor nutritional status with low BMI, inadequate intake of calcium and vitamin D, pre-existing factors predisposing to low bone density, and medications known to induce bone loss. Pregnancy- and lactation-associated osteoporosis is rare, but it can lead to multiple vertebral compression fractures. Clinicians should be suspicious with women experiencing mid- to low-back pain during the third trimester or early postpartum phase. Evaluation typically involves thoracic and lumbar spine X-rays or MRI, followed by dual-energy X-ray absorptiometry (DXA). Management strategies include discontinuing breastfeeding, optimizing weight-bearing activities, ensuring adequate calcium and vitamin D intake, managing pain, and considering antiosteoporosis medications (Scioscia and Zanchetta, 2023).
Menopause plays a role in the loss of bone density. Data from SWAN showed that the rates and cumulative amounts of bone loss were greatest during perimenopause, from 1 year before through 2 years after the final menstrual period (Greendale et al., 2019). Bone loss 2–5 years after the
final menstrual period was lower than that observed during the transition. Diminishing levels of estrogens during perimenopause lead to accelerated bone resorption relative to bone formation, predisposing to bone loss and elevating the risk of osteoporosis and fractures in later years. An elevation in levels of the gonadotropin, follicular stimulating hormone, which occurs in response to the decline in estrogen levels during menopause, appears to contribute to bone loss (Liu et al., 2017). Other hormones secreted by the ovaries—inhibin, activin, and relaxin—may also play functional roles in bone formation and resorption (Mills et al., 2021).
BMD starts to decline swiftly approximately 1 year before the final menstrual period and continues for about two years, followed by a slight reduction in bone loss thereafter (Karlamangla et al., 2018). For 1 year before through 2 years after the final menstrual period, women lost 7 and 6 percent of their lumbar spine and femoral neck BMD, respectively (Greendale et al., 2012). Slower BMD loss was observed in African American women, whereas faster BMD loss was observed in Japanese and Chinese women (Greendale et al., 2012).
Lifestyle Behavioral Factors
Physical activity and dietary patterns are two well-recognized lifestyle behaviors that are associated with osteoporosis prevention (Mortensen et al., 2021; Pinheiro et al., 2020). With respect to diet, nutrients such as vitamin D, phosphorus, riboflavin, calcium, protein and fiber intake, and folate from fruits and vegetables are known to contribute to the prevention of osteoporosis and reduction in fractures in postmenopausal women (Ilesanmi-Oyelere and Kruger, 2020). A systematic review found that engaging in physical activity, specifically in higher doses and of multiple types, including resistance training, were beneficial for the prevention of osteoporosis in postmenopausal women (Pinheiro et al., 2020). Larger effects were seen for improvements in lumbar spine and hip BMD (Pinheiro et al., 2020). Sleep fragmentation has an adverse impact of bone health (Swanson, 2021). An increasing body of research indicates that abnormal sleep duration and disturbances in circadian rhythms may impact bone health, including the susceptibility to osteoporosis and fractures. The effects of disrupted sleep may be particularly pertinent for individuals with certain medical conditions predisposing them to both sleep disturbances and skeletal fragility, such as diabetes (Swanson, 2021).
Initial Fracture and Risk of Subsequent Fractures
Among postmenopausal women aged 50–75 at baseline in the WHI Study, every type of initial fracture, even non-hip and non-vertebral fracture, was associated with increased risk of every other type of subsequent fracture.
For example, initial knee fracture was associated with increased risk of subsequent hip fracture, and these associations are evident even in younger postmenopausal women in their 50s (Crandall et al., 2021). Postmenopausal women with incident wrist fracture had elevated risk of experiencing an additional clinical fracture within 10 years after wrist fracture (Crandall et al., 2015).
Symptom Presentation and Diagnosis
The operational definition of osteoporosis is based on BMD assessment. The most commonly used screening test used is lumbar spine and hip DXA, which major drug therapy studies have also used to determine enrollment eligibility (USPSTF et al., 2018). In clinical practice, osteoporosis is diagnosed in individuals with BMD T-score15 of less than or equal to −2.5 at either the lumbar spine or hip (USPSTF et al., 2018). For each 1–standard deviation decrease in BMD measurement, there is an estimated 1.6- to 2.6-fold higher fracture risk (Marshall et al., 1996; Silva et al., 2022). The exact risk depends on various factors including the anatomical location of fracture, prior occurrence of fracture, and number of osteoporotic sites (Fink et al., 2008).
Osteoporosis is significantly underdiagnosed in older U.S. women. Approximately 26 percent of women ages 65–79 years do not receive a DXA test (Gillespie and Morin, 2017). No general agreement exists on the appropriate time to begin DXA for early menopausal women (Jiang et al., 2016).
Currently, a diagnosis of osteoporosis is based on the occurrence of fragility fractures or low BMD scores as assessed by DXA (Lenchik et al., 2018). Numerous professional organizations, including the Office of the Surgeon General, the U.S. Preventive Services Task Force, ACR, American College of Radiology, International Society for Clinical Densitometry, and Bone Health and Osteoporosis Foundations recommend the use of DXA to diagnose osteoporosis (Lenchik et al., 2018; Warriner et al., 2014). However, in the U.S., only approximately one-third of women aged 65 years and older undergo DXA testing (Warriner et al., 2014).
Imaging Bone Density and Quality
Computed Tomography (CT) Scan–BMD Correlation
Novel approaches for identifying patients at risk for osteoporotic fracture who are candidates for pharmacologic therapy are needed.
___________________
15A T-score is the difference between an individual’s BMD and 0, the BMD of a healthy young adult. The lower the T-score, the higher the risk of bone fracture. Health bone produces a T-score of −1 or higher.
“Opportunistic CT” is one such approach that utilizes results from routine CT scans to screen for additional body composition information, such as the attenuation of bone. Regardless of the primary indication, CT scans of the chest, abdomen, pelvis, and spine may thus yield useful information regarding a patient’s risk for osteoporosis, minimizing the burdens related to additional medical diagnoses and interventions, such as examination time and costs (Boutin and Lenchik, 2020; Lenchik et al., 2018).
Biomechanical computed tomography (BCT) is another method used to diagnose osteoporosis and assess fracture risk and measures both bone strength and BMD at the hip or spine. BCT may be helpful for patients without a recent DXA who are undergoing or have previously undergone CT scanning (Keaveny et al., 2020). Advancements in machine learning will provide future opportunities to make it easier to automate measurements and mine data from CT images and could potentially lead to substantial cost savings and improved care for patients at risk for osteoporosis (Lenchik et al., 2018).
Trabecular Bone Score (TBS)
Although DXA provides information about the bone density, TBS provides an estimate of the bone quality. TBS is a software program that integrates information from DXA images of lumbar spine and used as a tool to indirectly assess the microarchitecture and mechanical properties of bone (Goel et al., 2024; Shevroja et al., 2023). TBS measurements have been integrated into the FRAX® tool16 to enhance prediction of fracture independent of BMD (Goel et al., 2024; Shevroja et al., 2023). This method may be useful in determining when to initiate pharmacologic treatments in patients as it can estimate 10-year fracture probability (Goel et al., 2024).
CT Scan–TBS Correlation
The World Health Organization recommends DXA as the gold standard technique for diagnosing osteoporosis. DXA is the most widely used modality for measuring BMD, which is a quantitative measurement of bone used to predict bone strength and fracture risk (Shevroja et al., 2021). However, DXA cannot differentiate trabecular bone from cortical BMD (Lee et al., 2023b).17
Given the limitations of DXA, researchers have investigated tools to assess bone architecture as well as bone density. One approach is to use DXA images of the lumbar spine to evaluate pixel gray-level texture to
___________________
16“The FRAX® tool has been developed to evaluate fracture risk of patients. It is based on individual patient models that integrate the risks associated with clinical risk factors as well as BMD (BMD) at the femoral neck” (https://www.fraxplus.org/).
17Cortical bone is a dense, low-porosity, and less metabolically active tissue, and trabecular bone is a honeycomb-like network with a larger remodeling area and higher turnover rate (Clarke, 2008).
define a TBS, which is an indirect index and qualitative parameter of trabecular microarchitecture.
Quantitative computed tomography (QCT) has some diagnostic advantages over DXA. QCT allows true volumetric measurements of the lumbar spine independent of body size and identifies trabecular and cortical compartments (Link and Kazakia, 2020). Furthermore, QCT is known to be more sensitive than DXA for osteoporosis and has advantages over DXA for rheumatological disorders, such as ankylosing spondylitis (Link and Kazakia, 2020). Nevertheless, QCT has some limitations as a primary diagnostic tool for osteoporosis, given its relatively high cost and larger radiation exposure (Link and Kazakia, 2020). To overcome these limitations, investigators have attempted opportunistic screening for osteoporosis using the Hounsfield unit, a relative quantitative measurement of radio density used by radiologists to interpret CT images (DenOtter and Schubert, 2024). Its value is positively correlated with material density and compressive strength (Schreiber et al., 2011). Opportunistic screening has the advantages of not requiring additional imaging, radiation exposure, or appointments (Boutin and Lenchik, 2020; Lenchik et al., 2018; Schreiber et al., 2011).
MRI–TBS Correlation
Recent attempts have been made to diagnose osteoporosis using MRI-based measurements of the fat fraction of bone (Chang et al., 2020). In the bone marrow, osteoblasts and adipocytes—fat-storing cells—arise from common progenitor cells. Osteoporosis has predominance of differentiation toward adipocytes in progenitor cells, and differentiation toward osteoblasts decreases, leading to reduced bone formation. As a result, the increase in bone marrow fat becomes an indirect indicator of osteoporosis and a target for diagnosis. MRI-based measures of the fat fraction of bone are a reliable tool for evaluating vertebral bone marrow fat quantification without radiation exposure.
Despite attempts to measure BMD with various tools, none have been made to identify a relationship with TBS. Only one study in an Asian population has been published (Lee et al., 2023b), highlighting the need for evaluation in other racial and ethnic populations.
Vertebral Fracture Assessment
Vertebral fractures are commonly observed in postmenopausal women with osteoporosis (Yang et al., 2020a). Vertebral fractures cause substantial morbidity, with detrimental changes that include height loss, progressive and exaggerated forward rounding of the upper back, impaired movement and balance, and chronic pain. It is also associated with higher mortality, with estimates that one-third of patients will die within 5 years following hospitalization. Evidence has shown that prior experience of vertebral
fractures is a predictor of subsequent fractures. Most vertebral fractures are asymptomatic and only approximately one-third of vertebral fractures are identified on radiographs (Yang et al., 2020a).
Spine X-rays are primarily used to identify vertebral fractures, but lateral DXA of the entire spine for vertebral fracture assessment (VFA) is a convenient, low-radiation, and low-cost option to detect vertebral fractures (Lems et al., 2021). Studies have shown that VFA identifies vertebral fractures in one-third of asymptomatic women, although women with prevalent vertebral fractures often do not have osteoporosis (Yang et al., 2020a). VFA can also identify women at high risk of fracture, potentially leading to treatment.
Lateral spine imaging with standard radiography or densitometric VFA is indicated when a T-score is less than −1.0 and of one or more of the following is present (ISCD, 2019): women ages 70 and older and men ages 80 years and older; historical height loss greater than 4 centimeters or 1.5 inches, and self-reported but undocumented prior vertebral fracture.
Microindentation
Impact microindentation is a technique to assess bone material properties of cortical bone at the tibia in a transcutaneous, microinvasive manner. It is used increasingly in human studies evaluating the contribution of tissue material properties to bone fragility and approved for use in the clinic in the U.S. and Europe (Schoeb et al., 2023). Efforts to make this procedure less invasive may result in increased use in clinical settings.
Treatment and Management
Ensuring adequate calcium and vitamin D intake is essential for people with osteoporosis. Clinical trials of drug therapy demonstrated its efficacy in conjunction with calcium and vitamin D supplementation. Drug therapy decreases the risk of fracture and is effective for preventing both the initial fracture in persons with osteoporosis (as diagnosed using BMD criteria), subsequent fractures, and secondary fractures (among persons who already experienced a fracture).
FDA-approved prescription drug therapies include antiresorptive drugs, drugs that promote bone formation, and drugs that do both (Ensrud and Crandall, 2024). Antiresorptive drugs include bisphosphonates, such as alendronate, risedronate, ibandronate, and zoledronic acid; denosumab, an inhibitor of osteoclast development; and raloxifene, a selective ER modulator. Estrogens are a known antiresorptive agent approved to treat bone loss and prevent progression to osteoporosis (Eastell et al., 2019). Research is needed to better understand which patients could safely benefit from temporary cessation of bisphosphonate medication followed by reinitiation, the appropriate duration and monitoring of a bisphosphonate drug
holiday, and reasons for the higher risk of bisphosphonate-associated but rare serious harms in Asian versus White postmenopausal women.
Treatments that promote bone formation include recombinant parathyroid hormone and abaloparatide, a parathyroid-hormone-related peptide analog. Statins, approved for treating high cholesterol levels, may prove to be effective for increasing BMD and treating osteoporosis, with a dose-dependent therapeutic effect (Lin et al., 2018; Wang et al., 2016). However, some studies suggest that high-dose statin therapy can increase the risk of osteoporosis (Leutner et al., 2023).
People with osteoporosis are substantially undertreated (Gillespie and Morin, 2017; Jha et al., 2015; Kim et al., 2016; Singer et al., 2023). Bisphosphonate use within 6 months after hip fracture hospitalization has decreased from 15 percent in 2004 to 3 percent in 2013 (Kim et al., 2016). Among women enrolled in private commercial or Medicare Advantage plans who experience a new hip fracture, those aged 80 years and older are one-third less likely to use recommended services within 6 months compared with those ages 50–79. The most recent examination of commercial and Medicare medical claims data shows that even in 2023, 89 percent remain untreated for at least 1 year after fracture (Singer et al., 2023).
Treatment After Denosumab
Denosumab is a monoclonal antibody therapy approved in 2010 for patients with osteoporosis. It is a potent antiresorptive medication supported by 10 years of clinical trial data that demonstrate its safety and efficacy in reducing fracture risk (Bone et al., 2017; Kendler et al., 2022). Another type of monoclonal antibody therapy is romosozumab, which has also been used to treat postmenopausal women at high risk of fracture (Krupa et al., 2024). Compared to bisphosphonates, another drug class used in the treatment of osteoporosis, denosumab leads to continued gains in bone density (Kendler et al., 2022). Despite aging of the study population, nonvertebral fracture rates were lower after 4–10 years of treatment with denosumab compared to the initial 3 years of therapy (Kendler et al., 2022).
Treatment with denosumab for up to 10 years was associated with a favorable benefit risk profile when comparing fractures prevented per skeletal adverse event, such as osteonecrosis, a condition in which cells in the jawbone die, and atypical hip fracture. In addition, the frequency of adverse events, including infection and malignancy, remained low over time in the aging study population (Bone et al., 2017; Kendler et al., 2022).
If denosumab is discontinued, the gains in BMD may be lost and thus administration of a different class of anti-osteoporosis medication, such as bisphosphonate, can help prevent such bone loss, minimizing future risk of fracture (Kendler et al., 2022). Timing that switch is challenging,
and limited evidence is available. While transition to oral alendronate has been successful (Kendler et al., 2020), most postmenopausal women have contraindications to oral bisphosphonates and would benefit from intravenous infusions of bisphosphonates, such as zoledronic acid (Horikawa et al., 2015; Kendler et al., 2020). The timing of switching to other therapies is crucial for maintaining BMD gains from denosumab (Kendler et al., 2020).
Research is needed to understand how to treat osteoporosis, including the optimal duration of drug therapy, long-term risks and benefits of drug therapy, optimal frequency of BMD monitoring during drug therapy, anti-fracture benefits and harms of drug therapies in women aged 50–64, who are underrepresented in clinical trials compared to women 65 and older, and best means of selecting candidates for initial therapy with bone formation promoting agents instead of antiresorptive drug therapy.
Bone Turnover Markers
Biomolecules released during the bone remodeling cycle are used as bone turnover markers, which are typically detected in blood or urine samples. They are usually secreted by osteoblasts during bone formation or osteoclasts during bone resorption (Vasikaran et al., 2023) and are useful for monitoring the effects of oral therapy in patients with osteoporosis. Further work is needed in exploring their use to determine optimal duration of treatment with antiresorptive therapies, such as bisphosphonates and denosumab and that will yield the most benefit with minimal adverse effects. Large-scale fracture risk prediction studies assessing how bone turnover markers can contribute to risk prediction and consideration of its possible inclusion as a factor in risk calculators, such as FRAX, may help better predict risk. Improving commercial assays that detect bone turnover markers will enable the collation and comparison of data from different studies. Efforts to consolidate methods of detecting bone turnover biomarkers may lead to the uniform application of decision limits and treatment targets in clinical guidelines (Vasikaran et al., 2023).
Physician/Patient Education Tools
Educating patients about the importance of treating osteoporosis is essential for initiating treatment and improving compliance, leading to better outcomes in reducing fractures. However, given the constraints in medical practice, providing detailed education for a single condition is challenging. With the rapid advancement of digital technology, learning has shifted from verbal discussions and paper handouts to digital sources, offering accessibility anytime, anywhere, but the Internet may not always provide reliable information in a patient-friendly format, and social media
discussions often contain conflicting information based on personal experiences. It is also essential to provide culturally competent materials and information that are offered to patients in several languages (Lewiecki and Erb, 2022).
Disparities
Studies examining racial and ethnic disparities have focused on outcomes such as increased mortality (Wright et al., 2020). One recent study, for example, found that Black women experienced a greater 1-year mortality from osteoporosis compared to White women (Wright et al., 2020). After adjusting for age in Medicare beneficiaries, Black women had significantly higher risk of mortality following 1 year of encountering fractures of the femur, hip, humerus, or radius/ulna; debility based on new placement in long-term nursing 1 year following hip fracture; and becoming newly eligible for Medicaid because of limited finances for all fracture types compared to White women (Wright et al., 2020).
Equitable access to bone scans is a major determinant of outcomes associated with osteoporosis and has gender and racial bias (Courtney et al., 2023; Ruiz-Esteves et al., 2022). Black and African American individuals, for example, were more likely to be undiagnosed compared to White individuals based on data from the University of Michigan Health and Retirement Study (Courtney et al., 2023). Although Asian women have similar or lower BMD than White women, they are overrepresented in treatment for osteoporosis and bisphosphonate therapy, conferring a greater risk of atypical femur fractures (Lo et al., 2023). Furthermore, rural communities face inequitable access to DXA due to the reduction in insurance coverage for DXA and decreased availability of screening (Khosla et al., 2023).
BMD is an important risk factor for osteoporosis, and studies have found differences in Black, Asian, and Hispanic populations that vary based on anatomical site (Wu and Dai, 2023). SWAN measured bone density and found that Black and White women had similar rates of bone loss at the tibia, while Chinese and Japanese women had lower BMD values than White women (Cauley et al., 2021; Johannesdottir et al., 2022). There is variation among racial and ethnic groups based on NHANES data for women; non-Hispanic other (Asian) women had lower femoral neck BMD, followed by non-Hispanic White, Hispanic, and non-Hispanic Black women (Wu and Dai, 2023). Similarly, claims data from Kaiser Permanente found that femoral neck BMD was lower among women with Chinese, Filipino, or Japanese ethnicity compared to White women, particularly among older women (Lo et al., 2020); data for Hispanic women and non-Hispanic Black women were not included (Lo et al., 2020). Suggested mechanisms for lower BMD in Asian women have to do with genetic variants of vitamin D receptors
(Wu and Dai, 2023). While more recent NHANES and Kaiser Permanente datasets did not capture risk among American Indian and Alaska Native individuals, an earlier study based on NHANES data found that BMD among Navajo women was low, and they had a higher prevalence of osteoporosis (Miller et al., 2016). Lifestyle factors, including dietary patterns and physical activity, are important targeted areas for intervention to improve BMD in racial and ethnic groups.
Research Gaps
To address and understand disparities in osteoporosis and fracture outcomes, comprehensive research is needed. Disparities exist in postfracture outcomes influenced by social risk factors and fracture rates varying by self-reported race and ethnicity. This underscores the importance of multiethnic longitudinal studies to examine changes in BMD and identify risk factors particularly affecting racially and ethnically minoritized populations.
One critical area of research is determining the most effective risk assessment and screening strategies for young postmenopausal women aged 50–64. They may not have osteoporosis but are at an elevated risk of fractures. Questions about the frequency of osteoporosis screening and optimal intervals for BMD testing in postmenopausal women remain unanswered due to the absence of a standardized approach for assessing BMD risk in women (Gourlay et al., 2015). Moreover, strategies for treating BMD levels that are below normal but not yet in the osteoporotic range need to be developed. This includes determining the right settings and timing for initiating drug therapy in individuals with T-scores between −1 and −2.5, who do not meet the criteria for osteoporosis.
Additionally, the effectiveness of physical activity as a treatment requires further investigation to ascertain any ideal type, intensity, and frequency of exercise that can reduce fracture risk. The research must also explore the systemic barriers to accessing treatment post-fracture. These barriers may include systemic issues within health care, discrepancies among providers regarding who is responsible for initiating and monitoring drug therapy, and other factors at the health system or individual levels, including patient or caregiver characteristics. Addressing these gaps is essential for advancing care and outcomes for those at risk of or living with osteoporosis.
SARCOPENIA
As mentioned in Chapter 4, sarcopenia is primarily defined by a decline in muscle strength (Ooi and Welch, 2024). It is associated with detrimental health consequences: frailty, physical disability, risk of falls, loss of autonomy, poor quality of life, morbidity, and mortality (Santilli et al., 2014). Knowledge about it has evolved, and major insights include that it can
begin early in life, not just with aging; is a muscle disease; and is associated with low muscle quantity and quality (Cruz-Jentoft et al., 2019). In women, it contributes significantly to frailty.
Biological Factors
Sex Chromosome Effects
No evidence exists of sex chromosomal effects on the pathophysiology of sarcopenia. Although GWAS identified potential genes that are associated with this condition, these genetic polymorphisms are all located on autosomal chromosomes, not sex chromosomes (Jin et al., 2022; Jones et al., 2021).
The Effects of Estrogens
The most convincing evidence to support a role for estrogens in the muscular degenerative process of sarcopenia is found in studies of menopausal women. In perimenopause, the gradual decline in estrogens is thought to precipitate changes that lead to a decrease in the quality, mass, and function of muscle. These changes are attributed to several events that are normally regulated by estrogen prior to menopause, including reductions in muscle satellite cells (Collins et al., 2019) and a switch to a proinflammatory immune profile (Cioffi et al., 2002; Geraci et al., 2021).
Specific Risk Factors in Women
Reproductive Milestones
An acceleration of loss of muscle mass is observed with the menopausal transition and menopause (Kim and Kim, 2020). Studies have reported that lean body mass decreases by 0.2 percent a year and fat mass increases by 1.7 percent per year during the menopausal transition (Greendale et al., 2019). Hormonal changes during menopause represent a major risk factor for sarcopenia. For example, decline in estrogens in postmenopausal women is associated with a decline in muscle mass, impaired muscle function, and the development of sarcopenia (Buckinx and Aubertin-Leheudre, 2022). Women who experience premature ovarian insufficiency (<40 years) or early menopause (<45 years) demonstrate lower muscle mass and impaired muscle function compared to women experiencing menopause at normal age (>45 years), suggesting higher risks for sarcopenia in these groups during the post-menopausal years (Divaris et al., 2023).
Population-based studies support the connection between declines in estrogens during menopause and muscle degeneration. One large population-based
cohort study of older English adults recruited from the English Longitudinal Study of Ageing showed that women had a 20 percent higher risk of sarcopenia than men (Yang et al., 2019). Another research group using data from the Estrogenic Regulation of Muscle Apoptosis study also demonstrated that menopausal status was a significant predictor of decreases in lean and muscle mass in women ages 47–55 years old (Juppi et al., 2020). As estrogens have been shown to promote satellite cell growth in muscle tissues by binding to its receptor (Collins et al., 2019) and curb muscle damage triggered by inflammatory signals (Tiidus et al., 2013), the decline in estrogens during menopause would thus contribute to sarcopenia. It is important to prevent and manage sarcopenia in postmenopausal women through nutrition, protein intake, vitamin D intake, and physical activity (Buckinx and Aubertin-Leheudre, 2022).
Nutrition and Physical Activity
The complex interconnection of age-related changes in physical activity and nutritional status between men and women contribute to gender differences in muscle quality and quantity, affecting the risk of sarcopenia (Ning et al., 2021). The complexity of these associations among physical activity, weight, and muscle quality/quantity was demonstrated in the Louisiana Osteoporosis Study (Du et al., 2022), which showed that in both older women and men, only those exercising 3 or more times a week were less likely to be obese and develop sarcopenic obesity (the combination of sarcopenia and obesity) compared to those who were not (Du et al., 2022). Exercising 1–2 times per week in middle-aged women and male adults and any regular exercise in older women and male adults were not related to less sarcopenia (Du et al., 2022). These findings illustrate the importance of nutrition and dietary interventions in sarcopenia prevention and management in middle-aged and older women. A systematic review reported the beneficial effects of protein, vitamin D, vitamin D and magnesium, and fish oil on muscle protein synthesis, muscle strength, and muscle function in addition to exercise (Thornton et al., 2023). Peri- and postmenopausal women can prevent and even combat the negative health outcomes of sarcopenia through healthy lifestyle and dietary habits, regular physical activity, and hormone therapy (Buckinx and Aubertin-Leheudre, 2022). Another study showed that food insecurity was associated with greater odds of sarcopenia based on NHANES data (Lynch et al., 2022).
Obesity
Obesity is a risk factor for sarcopenia. The relationship between insulin resistance and age-related muscle loss is an important factor in sarcopenic obesity (defined as low skeletal muscle mass with low muscle function)
SOURCE: Cruz-Jentoft et al., 2019. Sarcopenia: Revised European Consensus on Definition and Diagnosis. Age and Aging 48(1):16–31, by permission of Oxford University Press.
(Donini et al., 2022). Sarcopenia and CVD also have a bidirectional relationship. Cardiac patients, for example, are in a state of chronic inflammation, malnutrition, and physical inactivity and at the highest risk of accelerated skeletal muscle loss and thus, sarcopenia (Damluji et al., 2023). This supports the understanding that multiple factors are involved in the development of sarcopenia (see Figure 6-4).
Sarcopenic obesity is of significant concern in the United States. In one study of community-dwelling older obese adults, 18 percent had sarcopenic obesity (Dondero et al., 2023). It is linked to geriatric vulnerabilities such as frailty—the same study found that individuals with sarcopenic obesity had a 6.4 times higher odds of frailty compared to those who were nonsarcopenic obese (Dondero et al., 2023).
Condition Presentation and Diagnosis
In 2019, the European Working Group on Sarcopenia in Older People (EWGSOP) developed a revised definition that focuses on muscle strength as the primary diagnostic hallmark for sarcopenia, and that assessments of grip strength or a chair stand test are best ways to assess muscle strength and physical performance and severity associated with sarcopenia (Cruz-Jentoft et al., 2019; Ooi and Welch, 2024). DXA is the most common approach for measuring muscle quantity because of its wide availability, but MRI and CT are considered the gold standards for noninvasive measurement of muscle quantity (Cruz-Jentoft et al., 2019). Gait speed is a quick, safe, and highly reliable test that can predict adverse outcomes related to sarcopenia, such as disability, cognitive impairment, institutionalization, falls, and mortality (Abellan van Kan et al., 2009; Guralnik et al., 2000).
According to EWGSOP, low muscle strength suggests that an individual probably has sarcopenia. Diagnosis is confirmed with documentation of low muscle quantity or quality; if those two criteria and low physical performance are apparent, sarcopenia is considered severe. The revised EWGSOP criteria recognizes that “techniques for evaluating muscle quantity are available in many but not all clinical settings. As instruments and methods to evaluate muscle quality are developed and refined in the future, this parameter is expected to grow in importance as a defining feature of sarcopenia” (Cruz-Jentoft et al., 2019). EWGSOP recommends the five-question SARC-F questionnaire (Malmstrom et al., 2016) to generate self-reports of signs that characterize sarcopenia. Sarcopenia is considered “primary,” or age related, when no other cause is evident but aging and “secondary” given one or more other causes (Cruz-Jentoft et al., 2019).
Treatment and Management
Especially in postmenopausal women, management of sarcopenia includes maintaining proper nutrition, including optimal dietary intake of protein and vitamin D, engaging in physical activity; and using hormone therapy dependent on the individual dose and level of physical activity (Buckinx and Aubertin-Leheudre, 2022). Whole body vibration has been shown to improve features of sarcopenia—specifically, impaired muscle strength and function in postmenopausal women, and has been proposed as an alternative to standard exercises in those with severely impaired functionality (Guedes De Aguiar et al., 2023; Rogan et al., 2015); however, further studies are needed for recommendation as an alternative treatment. Research efforts identifying new drug therapeutics for sarcopenia are ongoing and potential candidates include inhibitors of the myostatin signaling pathway, factors such as irisin released by muscle during exercise (Falsetti et al., 2024), and natural compounds enriched in certain fruits and vegetables, such as ursolic acid and tomatidine (Kwak and Kwon, 2019). No drugs are approved to treat or manage sarcopenia, and although some drugs have improved muscle mass or strength, they did not result in clinically relevant improvements on physical performance (Rolland et al., 2023).
Sarcopenia and Frailty
Frailty, like sarcopenia, increases with age. The two conditions overlap and diagnosing them involves the same physical measures, such as gait speed, grip strength, and muscle mass. Overlap exists between the classic frailty definition and the EWGSOP clinical diagnostic criteria. The Frailty Index reveals how frailty can be quantified based on five criteria: unintentional weight loss of 10 pounds or more in the past year, self-reported exhaustion,
weakness measured by grip strength, slow walking speed, and low physical activity (Fried et al., 2001; Rockwood and Mitnitski, 2007).
A major factor contributing to frailty in women is the deficiency in estrogens at menopause that leads to reductions in bone mass (Khadilkar, 2019). Aging-related hormonal changes also play a role. For example, the amplitude of normal pulsed release of pituitary growth hormone declines with aging (Bartke and Darcy, 2017), leading to decreased liver production of insulin growth factor-1 (Elmlinger et al., 2004; Juul et al., 1994), the hormone that stimulates bone and muscle production. Another phenomenon in older adults that contributes to frailty is aging-related appetite loss that results in reduced intake of protein, calcium, vitamin D, and other nutrients essential for bone and muscle formation and in weight maintenance (Landi et al., 2016; Molfino et al., 2021; Picca et al., 2022). Furthermore, an age-related, generalized decline in physical activity contributes to losses of bone and muscle. According to the 2020 NHIS, the adherence rate to exercise recommendations, was 22 percent in men and 17.2 percent in women aged 65 and older (CDC, 2020; HHS, 2018).
Sex differences in frailty prevalence or risk have been well studied, with robust evidence that supports that women are frailer than men at all ages, and this is evident in a range of populations with the Frailty Index (Kane and Howlett, 2021). A meta-analysis consistently showed higher scores in women compared to men at any age (Gordon et al., 2017). However, the underlying mechanisms of the higher prevalence in women despite being at lower risk for frailty mortality are poorly understood. Studies have suggested that sociobehavioral factors may play a role, as women are more likely than men to report health concerns and seek medical help (Kane and Howlett, 2021). Moreover, data from preclinical models indicate that, at least in some studies, female mice and dogs do have higher levels of frailty than males, suggesting a biological contribution to sex differences in prevalence (Bisset and Howlett, 2019).
Inflammation and immune cell changes, hormone dysregulation, cell senescence, mitochondrial dysfunction, genetic instability, and epigenetic changes have all been proposed as mechanisms of frailty (Bisset and Howlett, 2019). Evidence is still lacking on frailty in ethnic minority women, but it has been reported that this is likely influenced and exacerbated by biopsychosocial factors (Reid et al., 2021). Figure 6-5 describes the various factors that lead to frailty and the resulting outcomes.
Several clinical studies have indicated that the risk and prevalence and incidence of frailty are higher in women than men (Kane and Howlett, 2021). Major national databases that have looked at frailty and its association with many other chronic conditions include NHANES. In a recent study using NHANES from 1999–2018, the prevalence of frailty in women was 35.4 percent (Guo et al., 2024). Sex differences in frailty
SOURCE: Morley, J. E. 2016. Frailty and sarcopenia in elderly. Wiener Klinische Wochenschrift 128(S7):439–445, Springer Nature.
may be attributed to the role of chronic inflammation, a key factor in the pathophysiology of frailty in women, and the role of estrogens (Gordon and Hubbard, 2019).
In general, a universal definition for frailty is lacking (Morley, 2011). The frailty phenotype developed using data from the Cardiovascular Health Study has been used to measure frailty (Fried et al., 2001; Morley, 2011). This assessment is characterized as having three out of five components: weight loss, exhaustion, weakness, slowness, and low physical activity (Fried et al., 2001).
Frailty is known to be at the intersection of various chronic conditions, including CVD, hypertension, osteoarthritis, obesity, and chronic pain (Chen et al., 2019a; Crow et al., 2019; Davis et al., 2021; Huang et al., 2023; Li et al., 2023b). Gender differences in frailty have also indicated that women with heart failure are at a greater risk (Davis et al., 2021). Sarcopenia is a major contributor to frailty.
Disparities
Data from the Louisiana Osteoporosis Study show that the prevalence of sarcopenia and sarcopenic obesity is significantly associated with social
determinants of health, such as income and education (Jeng et al., 2018). For women, age had the largest association with sarcopenic obesity, with the odds of developing sarcopenic obesity increasing from 2.3-fold for women in their 50s to fivefold for women older than 70, in comparison to women in their 40s (Jeng et al., 2018). Significant associations were also evident between race and ethnicity and sarcopenic obesity for Black compared to White adults. Furthermore, Asian adults had the highest rates of both sarcopenia and sarcopenic obesity compared to White, Black, and Hispanic adults (Jeng et al., 2018). In another U.S. study, Hispanic and Asian individuals had more severe sarcopenia as measured by grip strength and gait speed, but this study did not report gender differences (Bigman and As, 2021).
The literature shows that individuals with sarcopenia or who have evidence of a decline in muscle mass but do not yet meet the criteria for sarcopenia were more likely to have lower BMD (presarcopenia). These associations vary by race, ethnicity, and gender (Ning et al., 2021). In a study of non-Hispanic White, non-Hispanic Black, and non-Hispanic Asian men and women, older individuals with presarcopenia and sarcopenia were more likely to have lower BMD compared with those without presarcopenia and sarcopenia (Ning et al., 2021). The strongest association between sarcopenia and BMD was found in non-Hispanic Black followed by non-Hispanic White and non-Hispanic Asian individuals (Ning et al., 2021). In addition, significant differences of BMD across sarcopenia stages were found in more subgroups in women than in men after adjusting for age and BMI, with a negative linear relationship between sarcopenia and regional or whole-body BMD in non-Hispanic White, non-Hispanic Black, and non-Hispanic Asian men but not in non-Hispanic Asian women (Ning et al., 2021). The authors concluded that for outcomes that involve BMD, sarcopenia had a larger effect in women than in men.
Research Gaps
Many gaps remain in understanding sarcopenia, especially its initiation and progression, diagnostic tools and cutoff points, and outcomes and their applicability in different racial and ethnic groups and by gender (Cruz-Jentoft et al., 2019). An area lacking in research is the exploration of sex and gender differences in sarcopenia. Studies are also needed examining how structural and social determinants of health, which intersect with gender, are associated with sarcopenia. This includes investigating risk factors such as obesity, physical activity, and diet and nutrition. The link between sarcopenia and frailty needs to be explored further. In relation to frailty, areas of research opportunity include investigating the influence of inflammation, hormone dysregulation, cell senescence, and biopsychosocial factors.
SUMMARY
Biological and Mechanistic Factors
Further attention is required to understand how sex influences cellular and molecular processes in addition to physiology and pathophysiology to improve strategies in disease prevention, detection and screening, diagnosis, treatment, monitoring, and women’s specific response to drug therapies. Biological mechanisms are not well understood for certain conditions, and sex chromosome and gonadal hormone effects are not well understood for many conditions.
Understanding Risk Factors
Female-specific biological factors need to be included in both clinical and basic studies on topics such as the oral contraceptives, hormonal fluctuations, number of pregnancies, menstruation, age of menopause transition, and stage of menopause, as this will allow research to better reflect on human diseases that affect women. Understanding the influence of female-specific biological factors will facilitate developing criteria and diagnostic testing, risk assessment tools, and effective treatment strategies and options relevant to women. Sex- and gender-specific differences in pharmacokinetics play an important role in determining the safety of treatment options for women and their adverse drug reactions. According to FDA, women experience more adverse events than men, and those events are more serious in women; the reported reasons include that women are more frequently overdosed; are more sensitive; and take a greater number of medications. The strength of animal and cellular models is the ability to dissect the influence of potential biological factors by focusing on one potential sex-associated pathological driver to dissect disease mechanisms. When addressing the health conditions that plague women, researchers need to not only ensure data availability but also start evaluating the role of sex hormones and chromosomes.
Improving Diagnosis, Treatment, and Management
In terms of diagnostic tools, diagnoses are lacking for certain chronic conditions, such as fibromyalgia, ME/CFS, and chronic pain. Diagnosing sarcopenia is largely understudied compared to osteoporosis. Diagnosing mental disorders is a challenge among racial and ethnic groups.
Preclinical research for some diseases or conditions has inadequate biomarkers for diagnosis. For some biomarkers, the reference ranges have not been studied separately for men and women. Future studies should consider
sex as a major effect modifier of biomarkers rather than a confounder to be removed by adjustment. Preliminary findings on biomarkers obtained from convenience samples should be validated in population-based samples.
Comparisons of blood biomarkers in men and women should consider possible mechanisms for differences, such as sex differences in the binding of biomarkers to blood proteins, permeability of the blood–brain barrier, and effect of comorbidities. For some of the existing biomarkers, a major obstacle to clinical translation is the high cost and the invasiveness of the measure. Although research has advanced for CVD in women, diagnosis and treatment of INOCA, MINOCA, and SCAD are major gaps in research.
Gaps in research exist on how to improve treatment and management of several chronic conditions. New treatments, such as Ozempic for weight loss and Zurzuvae for PPD, have reached the market, but research on access to these medications is needed. Access to treatment is lacking for many conditions, such as HIV and SUD, and disparities are evident among different groups of women by race, ethnicity, sexual orientation, and incarceration.
Disparities
Despite advances in the last 2 decades to promote better representation of male and female sexes in basic, preclinical, and clinical research related to female-predominant conditions, disparities are still evident, and women continue to be underrepresented. Furthermore, glaring disparities related to access to care, prescribing practices, and social determinants of health are governed by a woman’s race, ethnicity, and gender. Future studies need to consider these differences to be able to identify the most efficacious ways to prevent, treat, and manage chronic conditions in women.
REFERENCES
Abellan van Kan, G., Y. Rolland, S. Andrieu, J. Bauer, O. Beauchet, M. Bonnefoy, M. Cesari, L. M. Donini, S. Gillette Guyonnet, M. Inzitari, F. Nourhashemi, G. Onder, P. Ritz, A. Salva, M. Visser, and B. Vellas. 2009. Gait speed at usual pace as a predictor of adverse outcomes in community-dwelling older people an International Academy on Nutrition and Aging (IANA) task force. The Journal of Nutrition, Health & Aging 13(10):881–889.
Abu, Y., and S. Roy. 2021. Prenatal opioid exposure and vulnerability to future substance use disorders in offspring. Experimental Neurology 339:113621.
ACOG (American College of Obstetricians and Gynecologists). 2022. Headaches in pregnancy and postpartum: ACOG clinical practice guideline no. 3. Obstetrics and Gynecology 139(5):944–972.
Adlam, D., M. S. Tweet, R. Gulati, D. Kotecha, P. Rao, A. J. Moss, and S. N. Hayes. 2021. Spontaneous coronary artery dissection: Pitfalls of angiographic diagnosis and an approach to ambiguous cases. JACC Cardiovascular Interventions 14(16):1743–1756.
Ahmad, S. R., and N. Rosendale. 2022. Sex and gender considerations in episodic migraine. Current Pain and Headache Reports 26(7):505–516.
Ahmed, S. H., and G. F. Koob. 1998. Transition from moderate to excessive drug intake: Change in hedonic set point. Science 282(5387):298–300.
Ait Eldjoudi, D., A. Cordero Barreal, M. Gonzalez-Rodríguez, C. Ruiz-Fernández, Y. Farrag, M. Farrag, F. Lago, M. Capuozzo, M. A. Gonzalez-Gay, A. Mera Varela, J. Pino, and O. Gualillo. 2022. Leptin in osteoarthritis and rheumatoid arthritis: Player or bystander? International Journal of Molecular Sciences 23(5).
Akoto, C., S. A. Norris, and J. Hemelaar. 2021. Maternal HIV infection is associated with distinct systemic cytokine profiles throughout pregnancy in South African women. Scientific Reports 11(1):10079.
Alabas, O. A., O. A. Tashani, G. Tabasam, and M. I. Johnson. 2012. Gender role affects experimental pain responses: A systematic review with meta-analysis. European Journal of Pain (London, England) 16(9):1211–1223.
Alati, R., D. A. Lawlor, A. A. Mamun, G. M. Williams, J. M. Najman, M. O’Callaghan, and W. Bor. 2007. Is there a fetal origin of depression? Evidence from the Mater University Study of pregnancy and its outcomes. American Journal of Epidemiology 165(5):575–582.
AlBadri, A., C. N. Bairey Merz, B. D. Johnson, J. Wei, P. K. Mehta, G. Cook-Wiens, S. E. Reis, S. F. Kelsey, V. Bittner, G. Sopko, L. J. Shaw, C. J. Pepine, and B. Ahmed. 2019. Impact of abnormal coronary reactivity on long-term clinical outcomes in women. Journal of the American College of Cardiology 73(6):684–693.
Albert, P. R. 2015. Why is depression more prevalent in women? Journal of Psychiatry and Neuroscience 40(4):219–221.
Alexander, B. T. 2003. Placental insufficiency leads to development of hypertension in growth-restricted offspring. Hypertension 41(3):457–462.
Ali, M., H. J. A. van Os, N. van der Weerd, J. W. Schoones, M. W. Heymans, N. D. Kruyt, M. C. Visser, and M. J. H. Wermer. 2022. Sex differences in presentation of stroke: A systematic review and meta-analysis. Stroke 53(2):345–354.
Allais, G., G. Chiarle, S. Sinigaglia, O. Mana, and C. Benedetto. 2019. Migraine during pregnancy and in the puerperium. Neurological Sciences 40(Suppl 1):81–91.
Allen, P. R. 2008. Chronic fatigue syndrome: Implications for women and their health care providers during the childbearing years. Journal of Midwifery & Women’s Health 53(4):289–301.
Almdal, T., H. Scharling, J. S. Jensen, and H. Vestergaard. 2004. The independent effect of Type 2 diabetes mellitus on ischemic heart disease, stroke, and death: A population-based study of 13,000 men and women with 20 years of follow-up. Archives Internal Medicine 164(13):1422–1426.
Aloisi, A. M., I. Ceccarelli, P. Fiorenzani, A. M. De Padova, and C. Massafra. 2004. Testosterone affects formalin-induced responses differently in male and female rats. Neuroscience Letters 361(1–3):262–264.
AlSiraj, Y., X. Chen, S. E. Thatcher, R. E. Temel, L. Cai, E. Blalock, W. Katz, H. M. Ali, M. Petriello, P. Deng, A. J. Morris, X. Wang, A. J. Lusis, A. P. Arnold, K. Reue, K. Thompson, P. Tso, and L. A. Cassis. 2019. XX sex chromosome complement promotes atherosclerosis in mice. Nature Communications 10(1):2631. https://doi.org/10.1038/s41467–019–10462–z
Altemus, M. 2006. Sex differences in depression and anxiety disorders: Potential biological determinants. Hormones and Behavior 50(4):534–538.
Altmann, A., L. Tian, V. W. Henderson, M. D. Greicius, and Alzheimer’s Disease Neuroimaging Initiative Investigators. 2014. Sex modifies the ApoE-related risk of developing Alzheimer disease. Annals of Neurology 75(4):563–573.
Amico, J. A., S. M. Seif, and A. G. Robinson. 1981. Oxytocin in human plasma: Correlation with neurophysin and stimulation with estrogen. Journal of Clinical Endocrinology and Metabolism 52(5):988–993.
Anda, R., G. Tietjen, E. Schulman, V. Felitti, and J. Croft. 2010. Adverse childhood experiences and frequent headaches in adults. Headache 50(9):1473–1481.
Anderson, K. O., C. R. Green, and R. Payne. 2009. Racial and ethnic disparities in pain: Causes and consequences of unequal care. Journal of Pain 10(12):1187–1204.
Andrade, C. P., A. R. Zamunér, M. Forti, N. Y. Tamburús, and E. Silva. 2019. Effects of aquatic training and detraining on women with fibromyalgia: Controlled randomized clinical trial. European Journal of Physical and Rehabilitation Medicine 55(1): 79–88.
Angeloni, B., R. Bigi, G. Bellucci, R. Mechelli, C. Ballerini, C. Romano, E. Morena, G. Pellicciari, R. Reniè, V. Rinaldi, M. C. Buscarinu, S. Romano, G. Ristori, and M. Salvetti. 2021. A case of double standard: Sex differences in multiple sclerosis risk factors. International Journal of Molecular Sciences 22(7):3696.
Applbaum, E., and A. Lichtbroun. 2019. Novel Sjögren’s autoantibodies found in fibromyalgia patients with SICCA and/or xerostomia. Autoimmune Reviews 18(2):199–202.
Applegate, K. L., F. J. Keefe, I. C. Siegler, L. A. Bradley, D. C. McKee, K. S. Cooper, and P. Riordan. 2005. Does personality at college entry predict number of reported pain conditions at mid-life? A longitudinal study. Journal of Pain 6(2):92–97.
Aragón-Herrera, A., S. Feijóo-Bandín, L. Anido-Varela, S. Moraña-Fernández, E. Roselló-Lletí, M. Portolés, E. Tarazón, O. Gualillo, J. R. González-Juanatey, and F. Lago. 2022. Relaxin-2 as a potential biomarker in cardiovascular diseases. Journal of Personalized Medicine 12(7).
Aranda, M. P., I. N. Kremer, L. Hinton, J. Zissimopoulos, R. A. Whitmer, C. H. Hummel, L. Trejo, and C. Fabius. 2021. Impact of dementia: Health disparities, population trends, care interventions, and economic costs. Journal of the American Geriatrics Society 69(7):1774–1783.
Araújo, R. P., P. Figueiredo, J. Pinto, P. Vilela, I. P. Martins, and R. Gil-Gouveia. 2023. Altered functional connectivity in a sensorimotor-insular network during spontaneous migraine attacks: A resting-state FMRI study. Brain Research 1818:148513.
Archives of Disease in Childhood. 1994. Role of nutritional programming in determining adult morbidity. Archives of Disease in Childhood 71:288–290.
Ardekani, B. A., A. Convit, and A. H. Bachman. 2016. Analysis of the Miriad data shows sex differences in hippocampal atrophy progression. Journal of Alzheimer’s Disease 50(3):847–857.
Arnold, A. P. 2017. Y chromosome’s roles in sex differences in disease. Proceedings of the National Academy of Sciences of the United States of America 114(15):3787–3789.
Arnold, L. M., J. I. Hudson, E. V. Hess, A. E. Ware, D. A. Fritz, M. B. Auchenbach, L. O. Starck, and P. E. Keck, Jr. 2004. Family study of fibromyalgia. Arthritis and Rheumatism 50(3):944–952.
Arnold, L. M., J. Fan, I. J. Russell, M. B. Yunus, M. A. Khan, I. Kushner, J. M. Olson, and S. K. Iyengar. 2013. The fibromyalgia family study: A genome-wide linkage scan study. Arthritis and Rheumatism 65(4):1122–1128.
Aroke, E. N., P. V. Joseph, A. Roy, D. S. Overstreet, T. O. Tollefsbol, D. E. Vance, and B. R. Goodin. 2019. Could epigenetics help explain racial disparities in chronic pain? Journal of Pain Research 12:701–710.
Arout, C. A., M. Sofuoglu, L. A. Bastian, and R. A. Rosenheck. 2018. Gender differences in the prevalence of fibromyalgia and in concomitant medical and psychiatric disorders: A National Veterans Health Administration study. Journal of Women’s Health 27(8):1035–1044.
Arslan, H., A. Nursal, A. Inanir, and S. Yigit. 2021. Influence of ESR1 variants on clinical characteristics and fibromyalgia syndrome in Turkish women. Endocrine Metabolic & Immune Disorders—Drug Targets 21:1326–1332.
Asin, S. N., A. M. Heimberg, S. K. Eszterhas, C. Rollenhagen, and A. L. Howell. 2008. Estradiol and progesterone regulate HIV Type 1 replication in peripheral blood cells. AIDS Research and Human Retroviruses 24(5):701–716.
Athnaiel, O., S. Cantillo, S. Paredes, and N. N. Knezevic. 2023. The role of sex hormones in pain-related conditions. International Journal of Molecular Sciences 24(3):1866.
Atzl, V. M., A. J. Narayan, L. M. Rivera, and A. F. Lieberman. 2019. Adverse Childhood Experiences and Prenatal Mental Health: Type of ACEs and Age of Maltreatment Onset. Arlington, VA: American Psychological Association.
Avona, A., B. N. Mason, C. Burgos-Vega, A. H. Hovhannisyan, S. N. Belugin, J. Mecklenburg, V. Goffin, N. Wajahat, T. J. Price, A. N. Akopian, and G. Dussor. 2021. Meningeal CGRP-prolactin interaction evokes female-specific migraine behavior. Annals of Neurology 89(6):1129–1144.
Ayres, J. M., J. Dallman, J. A. Nolte, N. Higginbotham, J. Baker, G. Horton, J. Salava, J. Sojka, K. J. Templeton, R. I. Malancea, and A. Heddings. 2023. Managing post-operative pain in orthopedic patients: An international comparison. Kansas Journal of Medicine 16:56–60.
Bacon, A. M., and L. White. 2023. The association between adverse childhood experiences, self-silencing behaviours and symptoms in women with fibromyalgia. Psychology, Health & Medicine 28(8):2073–2083.
Baer, J. S., H. M. Barr, F. L. Bookstein, P. D. Sampson, and A. P. Streissguth. 1998. Prenatal alcohol exposure and family history of alcoholism in the etiology of adolescent alcohol problems. Journal of Studies on Alcohol 59(5):533–543.
Bagade, T., A. G. Mersha, and T. Majeed. 2023. The social determinants of mental health disorders among women with infertility: A systematic review. BMC Women’s Health 23(1):668.
Bairey Merz, C. N., L. J. Shaw, S. E. Reis, V. Bittner, S. F. Kelsey, M. Olson, B. D. Johnson, C. J. Pepine, S. Mankad, B. L. Sharaf, W. J. Rogers, G. M. Pohost, A. Lerman, A. A. Quyyumi, G. Sopko, and WISE Investigators. 2006. Insights from the NHLBI-sponsored Women’s Ischemia Syndrome Evaluation (WISE) study: Part II: Gender differences in presentation, diagnosis, and outcome with regard to gender-based pathophysiology of atherosclerosis and macrovascular and microvascular coronary disease. Journal of the American College of Cardiology 47(3 Suppl):S21–29.
Bajic, V. P., M. Essack, L. Zivkovic, A. Stewart, S. Zafirovic, V. B. Bajic, T. Gojobori, E. Isenovic, and B. Spremo-Potparevic. 2019. The X files: “The mystery of X chromosome instability in Alzheimer’s disease.” Frontiers Genetics 10:1368.
Baker, F. C., and K. A. Lee. 2022. Menstrual cycle effects on sleep. Sleep Medicine Clinics 17(2):283–294.
Ball, J., M. J. Carrington, K. A. Wood, and S. Stewart. 2013. Women versus men with chronic atrial fibrillation: Insights from the Standard Versus Atrial Fibrillation Specific Management Study (safety). PloS One 8(5):e65795.
Bannister, E. 2019. There is increasing evidence to suggest that brain inflammation could play a key role in the aetiology of psychiatric illness. Could inflammation be a cause of the premenstrual syndromes PMS and PMDD? Post Reproductive Health 25(3): 157–161.
Barbosa-Leiker, C., A. N. C. Campbell, R. K. McHugh, C. Guille, and S. F. Greenfield. 2021. Opioid use disorder in women and the implications for treatment. Psychiatric Research and Clinical Practice 3(1):3–11.
Barker, D. J. P. 2004. Developmental origins of adult health and disease. Journal of Epidemiology and Community Health 58(2):114–115.
Barker, J. M., M. M. Torregrossa, A. P. Arnold, and J. R. Taylor. 2010. Dissociation of genetic and hormonal influences on sex differences in alcoholism-related behaviors. Journal of Neuroscience 30(27):9140–9144.
Barnes, L. L., R. S. Wilson, J. L. Bienias, J. A. Schneider, D. A. Evans, and D. A. Bennett. 2005. Sex differences in the clinical manifestations of Alzheimer disease pathology. Archives of General Psychiatry 62(6):685–691.
Barsky, L., C. N. B. Merz, J. Wei, C. Shufelt, E. Handberg, C. Pepine, T. Rutledge, S. Reis, M. Doyle, W. Rogers, L. Shaw, and G. Sopko. 2020. Even “WISE-r?”—an update on the NHLBI-sponsored Women’s Ischemia Syndrome Evaluation. Current Atherosclerosis Reports 22(8):35.
Bartke, A., and J. Darcy. 2017. Gh and ageing: Pitfalls and new insights. Best Practice & Research: Clinical Endocrinology & Metabolism 31(1):113–125.
Bartsch, T., and P. J. Goadsby. 2003. The trigeminocervical complex and migraine: Current concepts and synthesis. Current Pain and Headache Reports 7(5):371–376.
Bateman, L., A. C. Bested, H. F. Bonilla, B. V. Chheda, L. Chu, J. M. Curtin, T. T. Dempsey, M. E. Dimmock, T. G. Dowell, D. Felsenstein, D. L. Kaufman, N. G. Klimas, A. L. Komaroff, C. W. Lapp, S. M. Levine, J. G. Montoya, B. H. Natelson, D. L. Peterson, R. N. Podell, I. R. Rey, I. S. Ruhoy, M. A. Vera-Nunez, and B. P. Yellman. 2021. Myalgic encephalomyelitis/chronic fatigue syndrome: Essentials of diagnosis and management. Mayo Clinic Proceedings 96(11):2861–2878.
Batt, Melissa M., Korrina A. Duffy, Andrew M. Novick, Christina A. Metcalf, and C. Neill Epperson. 2020. Is postpartum depression different from depression occurring outside of the perinatal period? A review of the evidence. Focus 18(2):106–119.
Batushansky, A., S. Zhu, R. K. Komaravolu, S. South, P. Mehta-D’souza, and T. M. Griffin. 2022. Fundamentals of oa. An initiative of osteoarthritis and cartilage. Obesity and metabolic factors in oa. Osteoarthritis and Cartilage 30(4):501–515.
Beal, S. J., S. Kashikar-Zuck, C. King, W. Black, J. Barnes, and J. G. Noll. 2020. Heightened risk of pain in young adult women with a history of childhood maltreatment: A prospective longitudinal study. Pain 161(1):156–165.
Becker, J. B., and M. Hu. 2008. Sex differences in drug abuse. Frontiers in Neuroendocrinology 29(1):36–47.
Becker, J. B., and G. F. Koob. 2016. Sex differences in animal models: Focus on addiction. Pharmacological Reviews 68(2):242–263.
Beecham, G. W., A. C. Naj, J. R. Gilbert, J. L. Haines, J. D. Buxbaum, and M. A. Pericak-Vance. 2010. PCDH11X variation is not associated with late-onset Alzheimer disease susceptibility. Psychiatric Genetics 20(6):321–324.
Beggs, S., T. Trang, and M. W. Salter. 2012. P2X4r+ microglia drive neuropathic pain. Nature: Neuroscience 15(8):1068–1073.
Bellaver, B., G. Povala, P. C. L. Ferreira, J. P. Ferrari-Souza, D. T. Leffa, F. Z. Lussier, A. L. Benedet, N. J. Ashton, G. Triana-Baltzer, H. C. Kolb, C. Tissot, J. Therriault, S. Servaes, J. Stevenson, N. Rahmouni, O. L. Lopez, D. L. Tudorascu, V. L. Villemagne, M. D. Ikonomovic, S. Gauthier, E. R. Zimmer, H. Zetterberg, K. Blennow, H. J. Aizenstein, W. E. Klunk, B. E. Snitz, P. Maki, R. C. Thurston, A. D. Cohen, M. Ganguli, T. K. Karikari, P. Rosa-Neto, and T. A. Pascoal. 2023. Astrocyte reactivity influences amyloid-β effects on tau pathology in preclinical Alzheimer’s disease. Nature Medicine 29(7):1775–1781.
Benakis, C., D. Brea, S. Caballero, G. Faraco, J. Moore, M. Murphy, G. Sita, G. Racchumi, L. Ling, E. G. Pamer, C. Iadecola, and J. Anrather. 2016. Commensal microbiota affects ischemic stroke outcome by regulating intestinal γδ t cells. Nature Medicine 22(5):516–523.
Bennet, A. M., E. Di Angelantonio, Z. Ye, F. Wensley, A. Dahlin, A. Ahlbom, B. Keavney, R. Collins, B. Wiman, U. de Faire, and J. Danesh. 2007. Association of apolipoprotein E genotypes with lipid levels and coronary risk. JAMA 298(11):1300–1311.
Bereiter, D. A., J. L. Cioffi, and D. F. Bereiter. 2005. Oestrogen receptor-immunoreactive neurons in the trigeminal sensory system of male and cycling female rats. ArcHIVes of Oral Biology 50(11):971–979.
Berkley, K. J. 1997. Sex differences in pain. Behavioral and Brain Sciences 20(3):371–380; discussion 435–513.
Bertozzi, S., A. P. Londero, A. Xholli, G. Azioni, R. Di Vora, M. Paudice, I. Bucimazza, C. Cedolini, and A. Cagnacci. 2023. Risk-reducing breast and gynecological surgery for BRCA mutation carriers: A narrative review. Journal of Clinical Medicine 12(4):1422.
Bested, A. C., and L. M. Marshall. 2015. Review of myalgic encephalomyelitis/chronic fatigue syndrome: An evidence-based approach to diagnosis and management by clinicians. Reviews on Environmental Health 30(4):223–249.
Beveridge, J. K., K. S. Dobson, S. Madigan, K. O. Yeates, A. L. Stone, A. C. Wilson, S. Salberg, R. Mychasiuk, and M. Noel. 2020. Adverse childhood experiences in parents of youth with chronic pain: Prevalence and comparison with a community-based sample. Pain Reports 5(6):e866.
Bi, R.-Y., Z. Meng, P. Zhang, X.-D. Wang, Y. Ding, and Y.-H. Gan. 2017. Estradiol upregulates voltage-gated sodium channel 1.7 in trigeminal ganglion contributing to hyperalgesia of inflamed tmj. PloS One 12(6):e0178589.
Bianchi, A., D. Matranga, F. Patti, L. Maniscalco, S. Pilotto, M. Di Filippo, M. Zaffaroni, P. Annovazzi, A. Bertolotto, C. Gasperini, E. Quartuccio, D. Centonze, R. Fantozzi, A. Gajofatto, F. Gobbin, D. Landi, F. Granella, M. Buccafusca, G. A. Marfia, C. Chisari, P. Naldi, R. Bergamaschi, G. Greco, I. R. Zarbo, V. Rizzo, M. Ulivelli, D. Bezzini, L. Florio, M. Turazzini, M. Di Gregorio, M. Pugliatti, G. Salemi, and P. Ragonese. 2024. The role of ethnicity and native-country income in multiple sclerosis: The Italian Multicentre Study (MS-MIGIT). Journal of Neurology 271(5):2182–2194.
Biason-Lauber, A., and M. Lang-Muritano. 2022. Estrogens: Two nuclear receptors, multiple possibilities. Molecular and Cellular Endocrinology 554:111710.
Bigman, G., and R. As. 2021. Implications of race and ethnicity in sarcopenia U.S. national prevalence of sarcopenia by muscle mass, strength, and function indices. Gerontology and Geriatric Research 4(1).
Bimpong, K., K. Thomson, C. L. McNamara, M. Balaj, N. Akhter, C. Bambra, and A. Todd. 2022. The gender pain gap: Gender inequalities in pain across 19 european countries. Scandinavian Journal of Public Health 50(2):287–294.
Bisset, E. S., and S. E. Howlett. 2019. The biology of frailty in humans and animals: Understanding frailty and promoting translation. Aging Medicine 2(1):27–34.
Bjornevik, K., M. Cortese, B. C. Healy, J. Kuhle, M. J. Mina, Y. Leng, S. J. Elledge, D. W. Niebuhr, A. I. Scher, K. L. Munger, and A. Ascherio. 2022. Longitudinal analysis reveals high prevalence of Epstein-Barr virus associated with multiple sclerosis. Science 375(6578):296–301.
Blumenthal, J. A., and A. Rozanski. 2023. Exercise as a therapeutic modality for the prevention and treatment of depression. Progress in Cardiovascular Diseases 77:50–58.
Bochicchio, L., L. Porsch, S. Zollweg, A. K. Matthews, and T. L. Hughes. 2024. Health outcomes of sexual minority women who have experienced adverse childhood experiences: A scoping review. Trauma, Violence & Abuse 25(1):764–794.
Boehme, A. K., J. E. Siegler, M. T. Mullen, K. C. Albright, M. J. Lyerly, D. J. Monlezun, E. M. Jones, R. Tanner, N. R. Gonzales, T. M. Beasley, J. C. Grotta, S. I. Savitz, and S. Martin-Schild. 2014. Racial and gender differences in stroke severity, outcomes, and treatment in patients with acute ischemic stroke. Journal of Stroke and Cerebrovascular Diseases 23(4):e255–261.
Boehme, A., B. Carr, S. Kasner, K. Albright, M. Kallan, M. Elkind, C. Branas, and M. Mullen. 2017. Sex differences in RT-PA utilization at hospitals treating stroke: The national inpatient sample. Frontiers in Neurology 8:500.
Boerner, K. E., C. T. Chambers, J. Gahagan, E. Keogh, R. B. Fillingim, and J. S. Mogil. 2018. Conceptual complexity of gender and its relevance to pain. Pain 159(11):2137–2141.
Boily-Larouche, G., J. Lajoie, B. Dufault, K. Omollo, J. Cheruiyot, J. Njoki, M. Kowatsch, M. Kimani, J. Kimani, J. Oyugi, and K. R. Fowke. 2019. Characterization of the genital mucosa immune profile to distinguish phases of the menstrual cycle: Implications for HIV susceptibility. Journal of Infectious Diseases 219(6):856–866.
Bone, H. G., R. B. Wagman, M. L. Brandi, J. P. Brown, R. Chapurlat, S. R. Cummings, E. Czerwiński, A. Fahrleitner-Pammer, D. L. Kendler, K. Lippuner, J. Y. Reginster, C. Roux, J. Malouf, M. N. Bradley, N. S. Daizadeh, A. Wang, P. Dakin, N. Pannacciulli, D. W. Dempster, and S. Papapoulos. 2017. 10 years of denosumab treatment in postmenopausal women with osteoporosis: Results from the Phase 3 randomised freedom trial and open-label extension. Lancet Diabetes Endocrinology 5(7):513–523.
Boneva, R. S., E. M. Maloney, J.-M. Lin, J. F. Jones, F. Wieser, U. M. Nater, C. M. Heim, and W. C. Reeves. 2011. Gynecological history in chronic fatigue syndrome: A population-based case-control study. Journal of Women’s Health 20(1):21–28.
Boneva, R. S., J.-M. S. Lin, and E. R. Unger. 2015. Early menopause and other gynecologic risk indicators for chronic fatigue syndrome in women. Menopause 22(8):826–834.
Bour, A., J. Grootendorst, E. Vogel, C. Kelche, J.-C. Dodart, K. Bales, P.-H. Moreau, P. M. Sullivan, and C. Mathis. 2008. Middle-aged human APOE4 targeted-replacement mice show retention deficits on a wide range of spatial memory tasks. Behavioural Brain Research 193(2):174–182.
Bourque, S. L., F. S. Gragasin, A. L. Quon, Y. Mansour, J. S. Morton, and S. T. Davidge. 2013. Prenatal hypoxia causes long-term alterations in vascular endothelin-1 function in aged male, but not female, offspring. Hypertension 62(4):753–758.
Boutin, R. D., and L. Lenchik. 2020. Value-added opportunistic CT: Insights into osteoporosis and sarcopenia. American Journal of Roentgenology 215(3):582–594.
Bove, R., E. Secor, L. B. Chibnik, L. L. Barnes, J. A. Schneider, D. A. Bennett, and P. L. De Jager. 2014. Age at surgical menopause influences cognitive decline and Alzheimer pathology in older women. Neurology 82(3):222–229.
Brady, K. T., and C. L. Randall. 1999. Gender differences in substance use disorders. Psychiatric Clinics of North America 22(2):241–252.
Brand, J. S., Y. T. Van Der Schouw, N. C. Onland-Moret, S. J. Sharp, K. K. Ong, K.-T. Khaw, E. Ardanaz, P. Amiano, H. Boeing, M.-D. Chirlaque, F. Clavel-Chapelon, F. L. Crowe, B. De Lauzon-Guillain, E. J. Duell, G. Fagherazzi, P. W. Franks, S. Grioni, L. C. Groop, R. Kaaks, T. J. Key, P. M. Nilsson, K. Overvad, D. Palli, S. Panico, J. R. Quirós, O. Rolandsson, C. Sacerdote, M.-J. Sánchez, N. Slimani, B. Teucher, A. Tjonneland, R. Tumino, D. L. Van Der A, E. J. M. Feskens, C. Langenberg, N. G. Forouhi, E. Riboli, and N. J. Wareham. 2013. Age at menopause, reproductive life span, and Type 2 diabetes risk. Diabetes Care 36(4):1012–1019.
Bräuner, E. V., L. F. Wilson, T. Koch, J. Christensen, C. Dehlendorff, A. K. Duun-Henriksen, L. Priskorn, J. Abildgaard, M. K. Simonsen, J. T. Jørgensen, Y. H. Lim, Z. J. Andersen, A. Juul, and M. Hickey. 2022. The long-term association between bilateral oophorectomy and depression: A prospective cohort study. Menopause 29(3):276–283.
Braunthal, S., and A. Brateanu. 2019. Hypertension in pregnancy: Pathophysiology and treatment. SAGE Open Medicine 7:2050312119843700.
Breathett, K., M. Sims, M. Gross, E. A. Jackson, E. J. Jones, A. Navas-Acien, H. Taylor, K. L. Thomas, and B. V. Howard. 2020. Cardiovascular health in American Indians and Alaska Natives: A scientific statement from the American Heart Association. Circulation 141(25):e948–e959.
Brecht, A., C. Bartsch, G. Baumann, K. Stangl, and T. Dschietzig. 2011. Relaxin inhibits early steps in vascular inflammation. Regulatory Peptides 166(1–3):76–82.
Brennan, K. C., M. Romero Reyes, H. E. López Valdés, A. P. Arnold, and A. C. Charles. 2007. Reduced threshold for cortical spreading depression in female mice. Annals of Neurology 61(6):603–606.
Brennenstuhl, S., and E. Fuller-Thomson. 2015. The painful legacy of childhood violence: Migraine headaches among adult survivors of adverse childhood experiences. Headache 55(7):973–983.
Breslau, N. 2009. The epidemiology of trauma, PTSD, and other posttrauma disorders. Trauma, Violence & Abuse 10(3):198–210.
Bridel, C., W. N. van Wieringen, H. Zetterberg, B. M. Tijms, C. E. Teunissen, J. C. Alvarez-Cermeño, U. Andreasson, M. Axelsson, D. C. Bäckström, A. Bartos, M. Bjerke, K. Blennow, A. Boxer, L. Brundin, J. Burman, T. Christensen, L. Fialová, L. Forsgren, J. L. Frederiksen, M. Gisslén, E. Gray, M. Gunnarsson, S. Hall, O. Hansson, M. K. Herbert, J. Jakobsson, J. Jessen-Krut, S. Janelidze, G. Johannsson, M. Jonsson, L. Kappos, M. Khademi, M. Khalil, J. Kuhle, M. Landén, V. Leinonen, G. Logroscino, C. H. Lu, J. Lycke, N. K. Magdalinou, A. Malaspina, N. Mattsson, L. H. Meeter, S. R. Mehta, S. Modvig, T. Olsson, R. W. Paterson, J. Pérez-Santiago, F. Piehl, Y. A. L. Pijnenburg, O. T. Pyykkö, O. Ragnarsson, J. C. Rojas, J. Romme Christensen, L. Sandberg, C. S. Scherling, J. M. Schott, F. T. Sellebjerg, I. L. Simone, T. Skillbäck, M. Stilund, P. Sundström, A. Svenningsson, R. Tortelli, C. Tortorella, A. Trentini, M. Troiano, M. R. Turner, J. C. van Swieten, M. Vågberg, M. M. Verbeek, L. M. Villar, P. J. Visser, A. Wallin, A. Weiss, C. Wikkelsø, and E. J. Wild. 2019. Diagnostic value of cerebrospinal fluid neurofilament light protein in neurology: A systematic review and meta-analysis. JAMA Neurology 76(9):1035–1048.
Brimmer, D. J., K. K. McCleary, T. A. Lupton, K. M. Faryna, and W. C. Reeves. 2009. Continuing medical education challenges in chronic fatigue syndrome. BMC Medical Education 9(1):70.
Bromet, E., L. H. Andrade, I. Hwang, N. A. Sampson, J. Alonso, G. de Girolamo, R. de Graaf, K. Demyttenaere, C. Hu, N. Iwata, A. N. Karam, J. Kaur, S. Kostyuchenko, J. P. Lépine, D. Levinson, H. Matschinger, M. E. Mora, M. O. Browne, J. Posada-Villa, M. C. Viana, D. R. Williams, and R. C. Kessler. 2011. Cross-national epidemiology of DSM-IV major depressive episode. BMC Medicine 9:90.
Broocks, G., L. Meyer, M. Bechstein, U. Hanning, H. C. Kniep, E. Schlemm, A. A. Kyselyova, L. Winkelmeier, G. Schön, J. Fiehler, And A. Kemmling. 2023. Investigating Neurologic Improvement After IV Thrombolysis: The Effect Of Time From Stroke Onset vs Imaging-Based Tissue Clock. Neurology 101(17):E1678–E1686.
Brooks, W. H., and Y. Renaudineau. 2015. Epigenetics and autoimmune diseases: The X chromosome-nucleolus nexus. Frontiers Genetics 6:22.
Brush, J. E., H. M. Krumholz, E. J. Greene, and R. P. Dreyer. 2020. Sex differences in symptom phenotypes among patients with acute myocardial infarction. Circulation: Cardiovascular Quality and Outcomes 13(2).
Buchacz, K., C. Armon, F. J. Palella, Jr., R. M. Novak, J. Fuhrer, E. Tedaldi, D. Ward, C. Mayer, L. Battalora, K. Carlson, S. Purinton, M. Durham, J. Li, and HIV Outpatient Study (HOS) Investigators. 2020. The HIV Outpatient Study—25 years of HIV patient care and epidemiologic research. Open Forum Infectious Diseases 7(5).
Buckinx, F., and M. Aubertin-Leheudre. 2022. Sarcopenia in menopausal women: Current perspectives. International Journal of Women’s Health 14:805–819.
Buckley, R. F., E. C. Mormino, J. S. Rabin, T. J. Hohman, S. Landau, B. J. Hanseeuw, H. I. L. Jacobs, K. V. Papp, R. E. Amariglio, M. J. Properzi, A. P. Schultz, D. Kirn, M. R. Scott, T. Hedden, M. Farrell, J. Price, J. Chhatwal, D. M. Rentz, V. L. Villemagne, K. A. Johnson, and R. A. Sperling. 2019. Sex differences in the association of global amyloid and regional tau deposition measured by positron emission tomography in clinically normal older adults. JAMA Neurology 76(5):542–551.
Buhimschi, I. A., U. A. Nayeri, G. Zhao, L. L. Shook, A. Pensalfini, E. F. Funai, I. M. Bernstein, C. G. Glabe, and C. S. Buhimschi. 2014. Protein misfolding, congophilia, oligomerization, and defective amyloid processing in preeclampsia. Science Translational Medicine 6(245):245ra292–245ra292.
Buffenstein, R., S. D. Poppitt, R. M. McDevitt, and A. M. Prentice. 1995. Food intake and the menstrual cycle: A retrospective analysis, with implications for appetite research. Physiology and Behavior 58(6):1067–1077.
Bulka, C. M., M. L. Daviglus, V. W. Persky, R. A. Durazo-Arvizu, J. P. Lash, T. Elfassy, D. J. Lee, A. R. Ramos, W. Tarraf, and M. Argos. 2019. Association of occupational exposures with cardiovascular disease among U.S. Hispanics/Latinos. Heart 105(6):439–448.
Burch, R. 2019. Headache in pregnancy and the puerperium. Neurologic Clinics 37(1):31–51.
Burch, R. 2020. Epidemiology and treatment of menstrual migraine and migraine during pregnancy and lactation: A narrative review. Headache 60(1):200–216.
Burch, R., P. Rizzoli, and E. Loder. 2018. The prevalence and impact of migraine and severe headache in the United States: Figures and trends from government health studies. Headache 58(4):496–505.
Burnette, C. E., and C. R. Figley. 2016. Historical oppression, resilience, and transcendence: Can a holistic framework help explain violence experienced by indigenous people? Social Work 62(1):37–44.
Burnette, C. E., L. M. Renner, and C. R. Figley. 2019. The framework of historical oppression, resilience and transcendence to understand disparities in depression amongst indigenous peoples. British Journal of Social Work 49(4):943–962.
Burnette, C. E., K. Ka’apu, J. M. Scarnato, and J. Liddell. 2020. Cardiovascular health among U.S. Indigenous peoples: A holistic and sex-specific systematic review. Journal of Evidence Based Social Work (2019) 17(1):24–48
Burokas, A., S. Arboleya, R. D. Moloney, V. L. Peterson, K. Murphy, G. Clarke, C. Stanton, T. G. Dinan, and J. F. Cryan. 2017. Targeting the microbiota-gut-brain axis: Prebiotics have anxiolytic and antidepressant-like effects and reverse the impact of chronic stress in mice. Biological Psychiatry 82(7):472–487.
Burrows, L. J., and A. T. Goldstein. 2013. The treatment of vestibulodynia with topical estradiol and testosterone. Sexual Medicine 1(1):30–33.
Buse, D. C., K. M. Fanning, M. L. Reed, S. Murray, P. K. Dumas, A. M. Adams, and R. B. Lipton. 2019. Life with migraine: Effects on relationships, career, and finances from the Chronic Migraine Epidemiology and Outcomes (CAMEO) study. Headache 59(8):1286–1299.
Bushnell, C., L. D. McCullough, I. A. Awad, M. V. Chireau, W. N. Fedder, K. L. Furie, V. J. Howard, J. H. Lichtman, L. D. Lisabeth, and I. L. Piña. 2014. Guidelines for the prevention of stroke in women: A statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 45(5):1545–1588.
Buskila, D., H. Cohen, L. Neumann, and R. P. Ebstein. 2004. An association between fibromyalgia and the dopamine D4 receptor exon III repeat polymorphism and relationship to novelty seeking personality traits. Molecular Psychiatry 9(8):730–731.
Butler, K. G. 2018. Relationship between the cortisol-estradiol phase difference and affect in women. Journal of Circadian Rhythms 16(1).
Butzner, M., D. Leslie, Y. Cuffee, C. S. Hollenbeak, C. Sciamanna, and T. P. Abraham. 2022. Sex differences in clinical outcomes for obstructive hypertrophic cardiomyopathy in the USA: A retrospective observational study of administrative claims data. BMJ Open 12(3):e058151.
Cairns, B. E. 2007. The influence of gender and sex steroids on craniofacial nociception. Headache 47(2):319–324.
Calhoun, A. H. 2017. Hormonal contraceptives and migraine with aura—is there still a risk? Headache 57(2):184–193.
Canevelli, M., F. Quarata, F. Remiddi, F. Lucchini, E. Lacorte, N. Vanacore, G. Bruno, and M. Cesari. 2017. Sex and gender differences in the treatment of Alzheimer’s disease: A systematic review of randomized controlled trials. Pharmacological Research 115:218–223.
Carey, C., E. Mulcahy, F. P. McCarthy, E. Jennings, K. Kublickiene, A. Khashan, and P. Barrett. 2024. Hypertensive disorders of pregnancy and the risk of maternal dementia: A systematic review and meta-analysis. American Journal of Obstetrics and Gynecology.
Carfì, A., R. Bernabei, and F. Landi. 2020. Persistent symptoms in patients after acute COVID-19. JAMA 324(6):603–605.
Carrasquillo, M. M., F. Zou, V. S. Pankratz, S. L. Wilcox, L. Ma, L. P. Walker, S. G. Younkin, C. S. Younkin, L. H. Younkin, G. D. Bisceglio, N. Ertekin-Taner, J. E. Crook, D. W. Dickson, R. C. Petersen, N. R. Graff-Radford, and S. G. Younkin. 2009. Genetic variation in PCDH11X is associated with susceptibility to late-onset Alzheimer’s disease. Nature Genetics 41(2):192–198.
Carroll, J. C., E. R. Rosario, L. Chang, F. Z. Stanczyk, S. Oddo, F. M. LaFerla, and C. J. Pike. 2007. Progesterone and estrogen regulate Alzheimer-like neuropathology in female 3XTG-AD mice. Journal of Neuroscience 27(48):13357–13365.
Carroll, J. C., E. R. Rosario, S. Kreimer, A. Villamagna, E. Gentzschein, F. Z. Stanczyk, and C. J. Pike. 2010. Sex differences in β-amyloid accumulation in 3XTG-AD mice: Role of neonatal sex steroid hormone exposure. Brain Research 1366:233–245.
Carruthers, B. M., A. K. Jain, K. L. De Meirleir, D. L. Peterson, N. G. Klimas, A. M. Lerner, A. C. Bested, P. Flor-Henry, P. Joshi, A. C. P. Powles, J. A. Sherkey, and M. I. van de Sande. 2003. Myalgic encephalomyelitis/chronic fatigue syndrome. Journal of Chronic Fatigue Syndrome 11(1):7–115.
Carson, A. P., D. L. Long, A. L. Cherrington, G. R. Dutton, V. J. Howard, T. M. Brown, G. Howard, M. M. Safford, and M. Cushman. 2021. Sex differences in factors contributing to the racial disparity in diabetes risk. American Journal of Preventive Medicine 60(4):e169–e177.
Carvalho Silva, R., C. Pisanu, E. Maffioletti, V. Menesello, M. Bortolomasi, M. Gennarelli, B. T. Baune, A. Squassina, and A. Minelli. 2023. Biological markers of sex-based differences in major depressive disorder and in antidepressant response. European Neuropsychopharmacology 76:89–107.
Casale, R., F. Atzeni, L. Bazzichi, G. Beretta, E. Costantini, P. Sacerdote, and C. Tassorelli. 2021. Pain in women: A perspective review on a relevant clinical issue that deserves prioritization. Pain and Therapy 10(1):287–314.
Case, L. K., and C. Teuscher. 2015. Y genetic variation and phenotypic diversity in health and disease. Biology of Sex Differences 6:6.
Case, P., S. B. Austin, D. J. Hunter, J. E. Manson, S. Malspeis, W. C. Willett, and D. Spiegelman. 2004. Sexual orientation, health risk factors, and physical functioning in the Nurses’ Health Study II. Journal of Women’s Health 13(9):1033–1047.
Case, L. K., E. H. Wall, J. A. Dragon, N. Saligrama, D. N. Krementsov, M. Moussawi, J. F. Zachary, S. A. Huber, E. P. Blankenhorn, and C. Teuscher. 2013. The Y chromosome as a regulatory element shaping immune cell transcriptomes and susceptibility to autoimmune disease. Genome Research 23(9):1474–1485.
Castañeda, S. F., M. L. Garcia, M. Lopez-Gurrola, M. Stoutenberg, K. Emory, M. L. Daviglus, R. Kaplan, A. L. Giachello, K. M. Molina, K. M. Perreira, M. E. Youngblood, D. C. Vidot, and G. A. Talavera. 2019. Alcohol use, acculturation and socioeconomic status among Hispanic/Latino men and women: The Hispanic Community Health Study/Study of Latinos. PloS One 14(4):e0214906.
Català-Senent, J. F., Z. Andreu, M. R. Hidalgo, I. Soler-Sáez, F. J. Roig, N. Yanguas-Casás, A. Neva-Alejo, A. López-Cerdán, M. de la Iglesia-Vayá, B. E. Stranger, and F. García-García. 2023. A deep transcriptome meta-analysis reveals sex differences in multiple sclerosis. Neurobiology of Disease 181:106113.
Cauley, J. A., A. S. Karlamangla, K. Ruppert, Y. Lian, M. Huang, S. Harlow, J. S. Finkelstein, and G. A. Greendale. 2021. Race/ethnic difference in trabecular bone score in midlife women: The Study of Women’s Health Across the Nation (SWAN). Archives of Osteoporosis 16(1):91.
Cavedo, E., P. A. Chiesa, M. Houot, M. T. Ferretti, M. J. Grothe, S. J. Teipel, S. Lista, M. O. Habert, M. C. Potier, B. Dubois, and H. Hampel. 2018. Sex differences in functional and molecular neuroimaging biomarkers of Alzheimer’s disease in cognitively normal older adults with subjective memory complaints. Alzheimer’s Dementia 14(9):1204–1215.
CDC (Centers for Disease Control and Prevention). 2016. U.S. medical eligibility criteria for contraceptive use, 2016 (U.S. MEC).
CDC. 2020. Quickstats: Percentage of adults aged ≥18 years who met the federal guidelines for muscle-strengthening physical activity, by age group and sex—National Health Interview Survey United States, 2020. MMWR 71(642).
CDC. 2021a. AIDS and Opportunistic Infections. https://www.cdc.gov/hiv/basics/livingwithhiv/opportunisticinfections.html (accessed February 20, 2024).
CDC. 2021b. Diagnoses of HIV Infection in the United States and Dependent Areas 2019. https://www.cdc.gov/hiv/library/reports/hiv-surveillance/vol-32/index.html (accessed December 7, 2023).
CDC. 2021c. Diagnoses of HIV Infection in the United States and Dependent Areas 2020. https://www.cdc.gov/hiv/library/reports/hiv-surveillance/vol-33/index.html (accessed 04/04, 2024).
CDC. 2021d. Monitoring selected national HIV prevention and care objectives by using HIV surveillance data—United States and 6 dependent areas, 2019. Atlanta, Georgia: Centers for Disease Control and Prevention.
CDC. 2022. Behavioral and clinical characteristics of persons with diagnosed HIV infection—medical monitoring project, United States 2020 cycle (June 2020–May 2021). Atlanta, Georgia: Centers for Disease Control and Prevention.
CDC. 2023a. About the Ending the HIV Epidemic in the U.S. Initiative. https://www.cdc.gov/endhiv/about-ehe/index.html (accessed February 20, 2024).
CDC. 2023b. Diagnoses of HIV Infection in the United States and Dependent Areas, 2021: Tables. https://www.cdc.gov/hiv/library/reports/hiv-surveillance/vol-34/content/tables.html (accessed February 20, 2024).
CDC. 2023c. HIV and Women: Viral Suppression and Barriers to Care. https://www.cdc.gov/hiv/group/gender/women/viral-suppression.html (accessed April 2, 2024).
Census Bureau. 2020. Annual estimates of the resident population by sex, age, race alone of in combination, and Hispanic origin for the United States: April 1, 2010 to July 1, 2019 (nc-est2019–asr5h). Washington, DC: U.S. Census Bureau, Population Division.
Ceriani, C. E. J., and S. D. Silberstein. 2023. Current and emerging pharmacotherapy for menstrual migraine: A narrative review. Expert Opinion on Pharmacotherapy 24(5):617–627.
Cetinkaya, A., E. Kilinc, C. Camsari, and M. N. Ogun. 2020. Effects of estrogen and progesterone on the neurogenic inflammatory neuropeptides: Implications for gender differences in migraine. Experimental Brain Research 238(11):2625–2639.
Cevik, R., A. Gur, S. Acar, K. Nas, and A. J. Sarac. 2004. Hypothalamic-pituitary-gonadal axis hormones and cortisol in both menstrual phases of women with chronic fatigue syndrome and effect of depressive mood on these hormones. BMC Musculoskeletal Disorders 5:47.
Chadwick, A. L., N. E. Lisha, M. E. Lubensky, Z. Dastur, M. R. Lunn, J. Obedin-Maliver, and A. Flentje. 2024. Localized and widespread chronic pain in sexual and gender minority people-an analysis of the pride study. Pain Medicine.
Chalder, T., A. E. Cullen, R. Evans, T. Harris, N. Kaleycheva, and T. Nicholson. 2021. The role of lifetime stressors in adult fibromyalgia: Systematic review and meta-analysis of case-control studies. Psychological Medicine 51(2):177–193.
Chambers, D., A. M. Bagnall, S. Hempel, and C. Forbes. 2006. Interventions for the treatment, management and rehabilitation of patients with chronic fatigue syndrome/myalgic encephalomyelitis: An updated systematic review. J R Soc Med 99(10):506–520.
Chan, J. C., C. P. Morgan, N. Adrian Leu, A. Shetty, Y. M. Cisse, B. M. Nugent, K. E. Morrison, E. Jašarević, W. Huang, N. Kanyuch, A. B. Rodgers, N. V. Bhanu, D. S. Berger, B. A. Garcia, S. Ament, M. Kane, C. Neill Epperson, and T. L. Bale. 2020. Reproductive tract extracellular vesicles are sufficient to transmit intergenerational stress and program neurodevelopment. Nature Communications 11(1):1499.
Chandan, J. S., T. Thomas, K. Raza, C. Bradbury-Jones, J. Taylor, S. Bandyopadhyay, and K. Nirantharakumar. 2021. Intimate partner violence and the risk of developing fibromyalgia and chronic fatigue syndrome. Journal of Interpersonal Violence 36(21–22): NP12279–NP12298.
Chang, R., X. Ma, Y. Jiang, D. Huang, X. Chen, M. Zhang, and D. Hao. 2020. Percentage fat fraction in magnetic resonance imaging: Upgrading the osteoporosis-detecting parameter. BMC Medical Imaging 20(1).
Chang, Y., Z. Han, Y. Zhang, Y. Zhou, Z. Feng, L. Chen, X. Li, L. Li, and J. Q. Si. 2019. G protein-coupled estrogen receptor activation improves contractile and diastolic functions in rat renal interlobular artery to protect against renal ischemia reperfusion injury. Biomedicine and Pharmacotherapy 112:108666.
Charalampopoulos, D., A. McLoughlin, C. E. Elks, and K. K. Ong. 2014. Age at menarche and risks of all-cause and cardiovascular death: A systematic review and meta-analysis. American Journal of Epidemiology 180(1):29–40.
Charles, A. C., and S. M. Baca. 2013. Cortical spreading depression and migraine. Nature Reviews: Neurology 9(11):637–644.
Charles, A. 2018. The pathophysiology of migraine: Implications for clinical management. Lancet Neurology 17(2):174–182.
Chartoff, E. H., M. Papadopoulou, M. L. MacDonald, A. Parsegian, D. Potter, C. Konradi, and W. A. Carlezon, Jr. 2009. Desipramine reduces stress-activated dynorphin expression and CREB phosphorylation in NAC tissue. Molecular Pharmacology 75(3):704–712.
Chen, T., and G. Vunjak-Novakovic. 2018. In vitro models of ischemia-reperfusion injury. Regenerative Engineering and Translational Medicine 4(3):142–153.
Chen, C., A. G. Winterstein, R. B. Fillingim, and Y.-J. Wei. 2019. Body weight, frailty, and chronic pain in older adults: A cross-sectional study. BMC Geriatrics 19(1).
Chen, X., R. McClusky, J. Chen, S. W. Beaven, P. Tontonoz, A. P. Arnold, and K. Reue. 2012. The number of X chromosomes causes sex differences in adiposity in mice. PLOS Genetics 8(5):e1002709.
Chen, X., L. Wang, D. H. Loh, C. S. Colwell, Y. Taché, K. Reue, and A. P. Arnold. 2015. Sex differences in diurnal rhythms of food intake in mice caused by gonadal hormones and complement of sex chromosomes. Hormones and Behavior 75:55–63.
Chen, X., Y. Liu, X. Sun, Z. Yin, H. Li, X. Liu, D. Zhang, C. Cheng, L. Liu, F. Liu, Q. Zhou, C. Wang, L. Li, B. Wang, Y. Zhao, D. Liu, M. Zhang, and D. Hu. 2019. Age at menarche and risk of all-cause and cardiovascular mortality: A systematic review and dose–response meta-analysis. Menopause 26(6).
Chen, Y., E. Navratilova, D. W. Dodick, and F. Porreca. 2020. An emerging role for prolactin in female-selective pain. Trends in Neuroscience 43(8):635–648.
Chinn, J. J., I. K. Martin, and N. Redmond. 2021. Health equity among Black women in the United States. Journal of Women’s Health 30(2):212–219.
Chlamydas, S., M. Markouli, D. Strepkos, and C. Piperi. 2022. Epigenetic mechanisms regulate sex-specific bias in disease manifestations. Journal of Molecular Medicine 100(8): 1111–1123.
Choi, J. Y., S. J. Park, and H. J. Lee. 2024. Healthy and unhealthy dietary patterns of depressive symptoms in middle-aged women. Nutrients 16(6).
Chopra, V., S. A. Flanders, M. O’Malley, A. N. Malani, and H. C. Prescott. 2021. Sixty-day outcomes among patients hospitalized with COVID-19. Annals of Internal Medicine 174(4):576–578.
Christensen, J., J. K. Beveridge, M. Wang, S. L. Orr, M. Noel, and R. Mychasiuk. 2021. A pilot study investigating the role of gender in the intergenerational relationships between gene expression, chronic pain, and adverse childhood experiences in a clinical sample of youth with chronic pain. Epigenomes 5(2):9.
Cicero, T. J., S. C. Aylward, and E. R. Meyer. 2003. Gender differences in the intravenous self-administration of mu opiate agonists. Pharmacology Biochemistry and Behavior 74(3):541–549.
Cioffi, M., K. Esposito, M. T. Vietri, P. Gazzerro, A. D’Auria, I. Ardovino, G. A. Puca, and A. M. Molinari. 2002. Cytokine pattern in postmenopause. Maturitas 41(3):187–192.
Clarke, B. 2008. Normal bone anatomy and physiology. Clinical Journal of the American Society of Nephrology 3(Supplement_3):S131–S139.
Claxton, A., L. D. Baker, C. W. Wilkinson, E. H. Trittschuh, D. Chapman, G. S. Watson, B. Cholerton, S. R. Plymate, M. Arbuckle, and S. Craft. 2013. Sex and ApoE genotype differences in treatment response to two doses of intranasal insulin in adults with mild cognitive impairment or Alzheimer’s disease. Journal of Alzheimer’s Disease 35(4):789–797.
Cochran, S. D., and V. M. Mays. 2007. Physical health complaints among lesbians, gay men, and bisexual and homosexually experienced heterosexual individuals: Results from the California Quality of Life survey. American Journal of Public Health 97(11):2048–2055.
Cofresí, R. U., M.-H. Monfils, N. Chaudhri, R. A. Gonzales, and H. J. Lee. 2019. Cue-alcohol associative learning in female rats. Alcohol 81:1–9.
Cohen, H., D. Buskila, L. Neumann, and R. P. Ebstein. 2002. Confirmation of an association between fibromyalgia and serotonin transporter promoter region (5-HTTLPR) polymorphism, and relationship to anxiety-related personality traits. Arthritis and Rheumatism 46(3):845–847.
Coker, A. L., P. H. Smith, L. Bethea, M. R. King, and R. E. McKeown. 2000. Physical health consequences of physical and psychological intimate partner violence. Archives of Family Medicine 9(5):451–457.
Collins, B. C., R. W. Arpke, A. A. Larson, C. W. Baumann, N. Xie, C. A. Cabelka, N. L. Nash, H. K. Juppi, E. K. Laakkonen, S. Sipilä, V. Kovanen, E. E. Spangenburg, M. Kyba, and D. A. Lowe. 2019. Estrogen regulates the satellite cell compartment in females. Cell Reports 28(2):368–381.e366.
Collins, L. F., F. J. Palella, Jr., C. C. Mehta, J. Holloway, V. Stosor, J. E. Lake, T. T. Brown, E. F. Topper, S. Naggie, K. Anastos, T. N. Taylor, S. Kassaye, A. L. French, A. A. Adimora, M. A. Fischl, M. C. Kempf, S. L. Koletar, P. C. Tien, I. Ofotokun, and A. N. Sheth. 2023. Aging-related comorbidity burden among women and men with or at-risk for HIV in the us, 2008–2019. JAMA Network Open 6(8):e2327584.
Cortés-Pérez, I., N. Zagalaz-Anula, M. D. R. Ibancos-Losada, F. A. Nieto-Escámez, E. Obrero-Gaitán, and M. C. Osuna-Pérez. 2021. Virtual reality-based therapy reduces the disabling impact of fibromyalgia syndrome in women: Systematic review with meta-analysis of randomized controlled trials. Journal of Personalized Medicine 11(11):1167.
Costantini, A., R. E. Mäkitie, M. A. Hartmann, N. Fratzl-Zelman, M. C. Zillikens, U. Kornak, K. Søe, and O. Mäkitie. 2020. Early-onset osteoporosis: Rare monogenic forms elucidate the complexity of disease pathogenesis beyond type I collagen. Journal of Bone and Mineral Research 37(9):1623–1641.
Coughlan, G. T., T. J. Betthauser, R. Boyle, R. L. Koscik, H. M. Klinger, L. B. Chibnik, E. M. Jonaitis, W. W. Yau, A. Wenzel, B. T. Christian, C. E. Gleason, U. G. Saelzler, M. J. Properzi, A. P. Schultz, B. J. Hanseeuw, J. E. Manson, D. M. Rentz, K. A. Johnson, R. Sperling, S. C. Johnson, and R. F. Buckley. 2023. Association of age at menopause and hormone therapy use with tau and β-amyloid positron emission tomography. JAMA Neurology 80(5):462–473.
Counts, S. E., S. Che, S. D. Ginsberg, and E. J. Mufson. 2011. Gender differences in neurotrophin and glutamate receptor expression in cholinergic nucleus basalis neurons during the progression of Alzheimer’s disease. Journal of Chemical Neuroanatomy 42(2):111–117.
Courtney, M. G., J. Roberts, Y. Quintero, and K. Godde. 2023. Childhood family environment and osteoporosis in a population-based cohort study of middle- to older-age Americans. JBMR Plus 7(5):e10735.
Cox, B. M., A. B. Young, R. E. See, and C. M. Reichel. 2013. Sex differences in methamphetamine seeking in rats: Impact of oxytocin. Psychoneuroendocrinology 38(10):2343–2353.
Crabbe, J. C., P. Metten, J. S. Rhodes, C.-H. Yu, L. L. Brown, T. J. Phillips, and D. A. Finn. 2009. A line of mice selected for high blood ethanol concentrations shows drinking in the dark to intoxication. Biological Psychiatry 65(8):662–670.
Crandall, C. J., K. M. Hovey, J. A. Cauley, C. A. Andrews, J. R. Curtis, J. Wactawski-Wende, N. C. Wright, W. Li, and M. S. Leboff. 2015. Wrist fracture and risk of subsequent fracture: Findings from the Women’s Health Initiative study. Journal of Bone and Mineral Research 30(11):2086–2095.
Crandall, C. J., R. P. Hunt, A. Z. LaCroix, J. A. Robbins, J. Wactawski-Wende, K. C. Johnson, M. Sattari, K. L. Stone, J. C. Weitlauf, T. R. Gure, and J. A. Cauley. 2021. After the initial fracture in postmenopausal women, where do subsequent fractures occur? eClinicalMedicine 35:100826.
Cripe, S. M., S. E. Sanchez, B. Gelaye, E. Sanchez, and M. A. Williams. 2011. Association between intimate partner violence, migraine and probable migraine. Headache 51(2):208–219.
Crossland, A. E., L. Munns, E. Kirk, and C. E. J. Preston. 2023. Comparing body image dissatisfaction between pregnant women and non-pregnant women: A systematic review and meta-analysis. BMC Pregnancy and Childbirth 23(1):709.
Crovetto, F., E. Somigliana, A. Peguero, and F. Figueras. 2013. Stroke during pregnancy and pre-eclampsia. Current Opinion in Obstetrics and Gynecology 25(6):425–432.
Crow, R. S., M. C. Lohman, A. J. Titus, S. B. Cook, M. L. Bruce, T. A. Mackenzie, S. J. Bartels, and J. A. Batsis. 2019. Association of obesity and frailty in older adults: NHANES 1999–2004. The Journal of Nutrition, Health & Aging 23(2):138–144.
Cruz-Jentoft, A. J., G. Bahat, J. Bauer, Y. Boirie, O. Bruyère, T. Cederholm, C. Cooper, F. Landi, Y. Rolland, A. A. Sayer, S. M. Schneider, C. C. Sieber, E. Topinkova, M. Vandewoude, M. Visser, M. Zamboni, I. Bautmans, J.-P. Baeyens, M. Cesari, A. Cherubini, J. Kanis, M. Maggio, F. Martin, J.-P. Michel, K. Pitkala, J.-Y. Reginster, R. Rizzoli, D. Sánchez-Rodríguez, and J. Schols. 2019. Sarcopenia: Revised European consensus on definition and diagnosis. Age and Ageing 48(1):16–31.
Cui, L., S. Li, S. Wang, X. Wu, Y. Liu, W. Yu, Y. Wang, Y. Tang, M. Xia, and B. Li. 2024. Major depressive disorder: Hypothesis, mechanism, prevention and treatment. Signal Transduction and Targeted Therapy 9(1):30.
Cummings, J. A., B. A. Gowl, C. Westenbroek, S. M. Clinton, H. Akil, and J. B. Becker. 2011. Effects of a selectively bred novelty-seeking phenotype on the motivation to take cocaine in male and female rats. Biology of Sex Differences 2:3–3.
Cupini, L. M., I. Corbelli, and P. Sarchelli. 2021. Menstrual migraine: What it is and does it matter? Journal of Neurology 268(7):2355–2363.
Curno, M. J., S. Rossi, I. Hodges-Mameletzis, R. Johnston, M. A. Price, and S. Heidari. 2016. A systematic review of the inclusion (or exclusion) of women in HIV research: From clinical studies of antiretrovirals and vaccines to cure strategies. Journal of Acquired Immune Deficiency Syndromes 71(2):181–188.
Curran, G. M., B. M. Booth, J. E. Kirchner, and D. E. Deneke. 2007. Recognition and management of depression in a substance use disorder treatment population. American Journal of Drug and Alcohol Abuse 33(4):563–569.
Curry, S. J., A. H. Krist, D. K. Owens, M. J. Barry, A. B. Caughey, K. W. Davidson, C. A. Doubeni, J. W. Epling, Jr., D. C. Grossman, A. R. Kemper, M. Kubik, C. S. Landefeld, C. M. Mangione, M. Silverstein, M. A. Simon, C. W. Tseng, and J. B. Wong. 2019. Interventions to prevent perinatal depression: U.S. Preventive Services Task Force recommendation statement. JAMA 321(6):580–587.
Daly, M. B., T. Pal, M. P. Berry, S. S. Buys, P. Dickson, S. M. Domchek, A. Elkhanany, S. Friedman, M. Goggins, M. L. Hutton, B. Y. Karlan, S. Khan, C. Klein, W. Kohlmann, A. W. Kurian, C. Laronga, J. K. Litton, J. S. Mak, C. S. Menendez, S. D. Merajver, B. S. Norquist, K. Offit, H. J. Pederson, G. Reiser, L. Senter-Jamieson, K. M. Shannon, R. Shatsky, K. Visvanathan, J. N. Weitzel, M. J. Wick, K. B. Wisinski, M. B. Yurgelun, S. D. Darlow, and M. A. Dwyer. 2021. Genetic/familial high-risk assessment: Breast, ovarian, and pancreatic, version 2.2021, nccn clinical practice guidelines in oncology. Journal of the National Comprehensive Cancer Network 19(1):77–102.
Daly, T. 2023. If deprivation worsens dementia outcomes, stimulation should improve them. Current Medical Research and Opinion 39(10):1391–1394.
D’Amelio, P., A. Grimaldi, S. Di Bella, S. Z. M. Brianza, M. A. Cristofaro, C. Tamone, G. Giribaldi, D. Ulliers, G. P. Pescarmona, and G. Isaia. 2008. Estrogen deficiency increases osteoclastogenesis up-regulating T cells activity: A key mechanism in osteoporosis. Bone 43(1):92–100.
Damluji, A. A., M. Alfaraidhy, N. AlHajri, N. N. Rohant, M. Kumar, C. Al Malouf, S. Bahrainy, M. Ji Kwak, W. B. Batchelor, D. E. Forman, M. W. Rich, J. Kirkpatrick, A. Krishnaswami, K. P. Alexander, G. Gerstenblith, P. Cawthon, C. R. deFilippi, and P. Goyal. 2023. Sarcopenia and cardiovascular diseases. Circulation 147(20):1534–1553.
D’Aoust, R. F., A. G. Rossiter, A. Elliott, M. Ji, C. Lengacher, and M. Groer. 2017. Women veterans, a population at risk for fibromyalgia: The associations between fibromyalgia, symptoms, and quality of life. Military Medicine 182(7):e1828–e1835.
Das, S., K. Taylor, J. Kozubek, J. Sardell, and S. Gardner. 2022. Genetic risk factors for ME/CFS identified using combinatorial analysis. Journal of Translational Medicine 20(1):598.
Davis, E. J., L. Broestl, S. Abdulai-Saiku, K. Worden, L. W. Bonham, E. Miñones-Moyano, A. J. Moreno, D. Wang, K. Chang, G. Williams, B. I. Garay, I. Lobach, N. Devidze, D. Kim, C. Anderson-Bergman, G.-Q. Yu, C. C. White, J. A. Harris, B. L. Miller, D. A. Bennett, A. P. Arnold, P. L. De Jager, J. J. Palop, B. Panning, J. S. Yokoyama, L. Mucke, and D. B. Dubal. 2020. A second X chromosome contributes to resilience in a mouse model of Alzheimer’s disease. Science Translational Medicine 12(558):eaaz5677.
Davis, M. R., C. S. Lee, A. Corcoran, N. Gupta, I. Uchmanowicz, and Q. E. Denfeld. 2021. Gender differences in the prevalence of frailty in heart failure: A systematic review and meta-analysis. International Journal of Cardiology 333:133–140.
Dawson, A., and E. Dennison. 2016. Measuring the musculoskeletal aging phenotype. Maturitas 93:13–17.
Dedoni, S., M. Scherma, C. Camoglio, C. Siddi, L. Dazzi, R. Puliga, J. Frau, E. Cocco, and P. Fadda. 2023. An overall view of the most common experimental models for multiple sclerosis. Neurobiology of Disease 184:106230.
Deelen, J., D. S. Evans, D. E. Arking, N. Tesi, M. Nygaard, X. Liu, M. K. Wojczynski, M. L. Biggs, A. van der Spek, G. Atzmon, E. B. Ware, C. Sarnowski, A. V. Smith, I. Seppälä, H. J. Cordell, J. Dose, N. Amin, A. M. Arnold, K. L. Ayers, N. Barzilai, E. J. Becker, M. Beekman, H. Blanché, K. Christensen, L. Christiansen, J. C. Collerton, S. Cubaynes, S. R. Cummings, K. Davies, B. Debrabant, J. F. Deleuze, R. Duncan, J. D. Faul, C. Franceschi, P. Galan, V. Gudnason, T. B. Harris, M. Huisman, M. A. Hurme, C. Jagger, I. Jansen, M. Jylhä, M. Kähönen, D. Karasik, S. L. R. Kardia, A. Kingston, T. B. L. Kirkwood, L. J. Launer, T. Lehtimäki, W. Lieb, L. P. Lyytikäinen, C. Martin-Ruiz, J. Min, A. Nebel, A. B. Newman, C. Nie, E. A. Nohr, E. S. Orwoll, T. T. Perls, M. A. Province, B. M. Psaty, O. T. Raitakari, M. J. T. Reinders, J. M. Robine, J. I. Rotter, P. Sebastiani, J. Smith, T. I. A. Sørensen, K. D. Taylor, A. G. Uitterlinden, W. van der Flier, S. J. van der Lee, C. M. van Duijn, D. van Heemst, J. W. Vaupel, D. Weir, K. Ye, Y. Zeng, W. Zheng, H. Holstege, D. P. Kiel, K. L. Lunetta, P. E. Slagboom, and J. M. Murabito. 2019. A meta-analysis of genome-wide association studies identifies multiple longevity genes. Nature Communications 10(1):3669.
de Farias Á, D., L. Eberle, T. A. Amador, and T. da Silva Dal Pizzol. 2020. Comparing the efficacy and safety of duloxetine and amitriptyline in the treatment of fibromyalgia: Overview of systematic reviews. Advances in Rheumatology 60(1):35.
De Giglio, L., F. Marinelli, V. T. Barletta, V. A. Pagano, F. De Angelis, F. Fanelli, N. Petsas, P. Pantano, V. Tomassini, and C. Pozzilli. 2017. Effect on cognition of estroprogestins combined with interferon beta in multiple sclerosis: Analysis of secondary outcomes from a randomised controlled trial. CNS Drugs 31(2):161–168.
De Leo, J. A., V. L. Tawfik, and M. L. LaCroix-Fralish. 2006. The tetrapartite synapse: Path to CNS sensitization and chronic pain. Pain 122(1–2):17–21.
de Los Ríos la Rosa, F., J. Khoury, B. M. Kissela, M. L. Flaherty, K. Alwell, C. J. Moomaw, P. Khatri, O. Adeoye, D. Woo, S. Ferioli, and D. O. Kleindorfer. 2012. Eligibility for intravenous recombinant tissue-type plasminogen activator within a population: The effect of the European Cooperative Acute Stroke Study (ECASS) III trial. Stroke 43(6):1591–1595.
Deleon-Pennell, K. Y., A. J. Mouton, O. K. Ero, Y. Ma, R. Padmanabhan Iyer, E. R. Flynn, I. Espinoza, S. K. Musani, R. S. Vasan, M. E. Hall, E. R. Fox, and M. L. Lindsey. 2018. LXR/RXR signaling and neutrophil phenotype following myocardial infarction classify sex differences in remodeling. Basic Research in Cardiology 113(5).
Deming, Y., L. Dumitrescu, L. L. Barnes, M. Thambisetty, B. Kunkle, K. A. Gifford, W. S. Bush, L. B. Chibnik, S. Mukherjee, P. L. De Jager, W. Kukull, M. Huentelman, P. K. Crane, S. M. Resnick, C. D. Keene, T. J. Montine, G. D. Schellenberg, J. L. Haines, H. Zetterberg, K. Blennow, E. B. Larson, S. C. Johnson, M. Albert, A. Moghekar, J. L. del Aguila, M. V. Fernandez, J. Budde, J. Hassenstab, A. M. Fagan, M. Riemenschneider, R. C. Petersen, L. Minthon, M. J. Chao, V. M. Van Deerlin, V. M. Y. Lee, L. M. Shaw, J. Q. Trojanowski, E. R. Peskind, G. Li, L. K. Davis, J. M. Sealock, N. J. Cox, Alzheimer’s Disease Neuroimaging Initiative, The Alzheimer Disease Genetics Consortium, A. M. Goate, D. A. Bennett, J. A. Schneider, A. L. Jefferson, C. Cruchaga, T. J. Hohman. 2018. Sex-specific genetic predictors of Alzheimer’s disease biomarkers. Acta Neuropathologica 136(6):857–872.
DenOtter, T. D., and J. Schubert. 2024. Hounsfield unit. In Statpearls. Treasure Island, FL: StatPearls Publishing LLC.
Dhillon, S. 2023. Zavegepant: First approval. Drugs 83(9):825–831.
Dinos, S., B. Khoshaba, D. Ashby, P. D. White, J. Nazroo, S. Wessely, and K. S. Bhui. 2009. A systematic review of chronic fatigue, its syndromes and ethnicity: Prevalence, severity, co-morbidity and coping. International Journal of Epidemiology 38(6):1554–1570.
Divaris, E., P. Anagnostis, N. K. Gkekas, E. Kouidi, and D. G. Goulis. 2023. Early menopause and premature ovarian insufficiency may increase the risk of sarcopenia: A systematic review and meta-analysis. Maturitas 175:107782.
Divney, A. A., R. Murillo, F. Rodriguez, C. A. Mirzayi, E. K. Tsui, and S. E. Echeverria. 2019. Diabetes prevalence by leisure-, transportation-, and occupation-based physical activity among racially/ethnically diverse U.S. Adults. Diabetes Care 42(7):1241–1247.
Đoàn, L. N., Y. Takata, K. Hooker, C. Mendez-Luck, and V. L. Irvin. 2021. Trends in cardiovascular disease by Asian American, Native Hawaiian, and Pacific Islander ethnicity, Medicare Health Outcomes Survey 2011–2015. The Journals of Gerontology: Series A 77(2):299–309.
Docampo, E., G. Escaramís, M. Gratacòs, S. Villatoro, A. Puig, M. Kogevinas, A. Collado, J. Carbonell, J. Rivera, J. Vidal, J. Alegre, X. Estivill, and R. Rabionet. 2014. Genome-wide analysis of single nucleotide polymorphisms and copy number variants in fibromyalgia suggest a role for the central nervous system. Pain 155(6):1102–1109.
Dodge, N. C., J. L. Jacobson, L. H. Lundahl, and S. W. Jacobson. 2023. Prenatal alcohol exposure and attention-deficit/hyperactivity disorder independently predict greater substance use in young adulthood. Alcohol Clinical and Experimental Research 47(6):1143–1155.
Dondero, K. R., J. R. Falvey, B. A. Beamer, and O. Addison. 2023. Geriatric vulnerabilities among obese older adults with and without sarcopenia: Findings from a nationally representative cohort study. Journal of Geriatric Physical Therapy 46(3).
Doney, E., L. Dion-Albert, F. Coulombe-Rozon, N. Osborne, R. Bernatchez, S. E. J. Paton, F. N. Kaufmann, R. O. Agomma, J. L. Solano, R. Gaumond, K. A. Dudek, J. K. Szyszkowicz, M. Lebel, A. Doyen, A. Durand, F. Lavoie-Cardinal, M. C. Audet, and C. Menard. 2024. Chronic stress exposure alters the gut barrier: Sex-specific effects on microbiota and jejunum tight junctions. Biological Psychiatry: Global Open Science 4(1):213–228.
Donini, L. M., L. Busetto, S. C. Bischoff, T. Cederholm, M. D. Ballesteros-Pomar, J. A. Batsis, J. M. Bauer, Y. Boirie, A. J. Cruz-Jentoft, D. Dicker, S. Frara, G. Frühbeck, L. Genton, Y. Gepner, A. Giustina, M. C. Gonzalez, H. S. Han, S. B. Heymsfield, T. Higashiguchi, A. Laviano, A. Lenzi, I. Nyulasi, E. Parrinello, E. Poggiogalle, C. M. Prado, J. Salvador, Y. Rolland, F. Santini, M. J. Serlie, H. Shi, C. C. Sieber, M. Siervo, R. Vettor, D. T. Villareal, D. Volkert, J. Yu, M. Zamboni, and R. Barazzoni. 2022. Definition and diagnostic criteria for sarcopenic obesity: ESPEN and EASO consensus statement. Obesity Facts 15(3):321–335.
Doss, P., M. Umair, J. Baillargeon, R. Fazazi, N. Fudge, I. Akbar, A. P. Yeola, J. B. Williams, M. Leclercq, C. Joly-Beauparlant, P. Beauchemin, G. F. Ruda, M. Alpaugh, A. C. Anderson, P. E. Brennan, A. Droit, H. Lassmann, C. S. Moore, and M. Rangachari. 2021. Male sex chromosomal complement exacerbates the pathogenicity of TH17 cells in a chronic model of central nervous system autoimmunity. Cell Reports 34(10):108833.
Dou, D. R., Y. Zhao, J. A. Belk, Y. Zhao, K. M. Casey, D. C. Chen, R. Li, B. Yu, S. Srinivasan, B. T. Abe, K. Kraft, C. Hellström, R. Sjöberg, S. Chang, A. Feng, D. W. Goldman, A. A. Shah, M. Petri, L. S. Chung, D. F. Fiorentino, E. K. Lundberg, A. Wutz, P. J. Utz, and H. Y. Chang. 2024. Xist ribonucleoproteins promote female sex-biased autoimmunity. Cell 187(3):733–749.e716.
Driscoll, M. A., M. T. Knobf, D. M. Higgins, A. Heapy, A. Lee, and S. Haskell. 2018. Patient experiences navigating chronic pain management in an integrated health care system: A qualitative investigation of women and men. Pain Medicine 19(suppl_1):S19–S29.
D’Souza, A., K. M. Dave, R. A. Stetler, and S. M. D. 2021. Targeting the blood–brain barrier for the delivery of stroke therapies. Advanced Drug Delivery Review 171:332–351.
Du, S., N. Itoh, S. Askarinam, H. Hill, A. P. Arnold, and R. R. Voskuhl. 2014. XY sex chromosome complement, compared with XX, in the CNS confers greater neurodegeneration during experimental autoimmune encephalomyelitis. Proceedings of the National Academy of Sciences of the United States of America 111(7):2806–2811.
Du, Y., T. Xu, Z. Yin, S. Espinoza, Y. Xie, C. Gentry, Q. Tian, L.-J. Zhao, H. Shen, Z. Luo, and H.-W. Deng. 2022. Associations of physical activity with sarcopenia and sarcopenic obesity in middle-aged and older adults: The Louisiana Osteoporosis Study. BMC Public Health 22(1).
Dubal, D. B., L. Broestl, and K. Worden. 2012. Sex and gonadal hormones in mouse models of Alzheimer’s disease: What is relevant to the human condition? Biology of Sex Differences 3(1):24.
Dumitrescu, L., L. L. Barnes, M. Thambisetty, G. Beecham, B. Kunkle, W. S. Bush, K. A. Gifford, L. B. Chibnik, S. Mukherjee, P. L. De Jager, W. Kukull, P. K. Crane, S. M. Resnick, C. D. Keene, T. J. Montine, G. D. Schellenberg, Y. Deming, M. J. Chao, M. Huentelman, E. R. Martin, K. Hamilton-Nelson, L. M. Shaw, J. Q. Trojanowski, E. R. Peskind, C. Cruchaga, M. A. Pericak-Vance, A. M. Goate, N. J. Cox, J. L. Haines, H. Zetterberg, K. Blennow, E. B. Larson, S. C. Johnson, M. Albert, Alzheimer’s Disease Genetics Consortium and the Alzheimer’s Disease Neuroimaging Initiative, D. A. Bennett, J. A. Schneider, A. L. Jefferson, and T. J. Hohman. 2019. Sex differences in the genetic predictors of Alzheimer’s pathology. Brain 142(9):2581–2589.
Dunaif, A., K. R. Segal, W. Futterweit, and A. Dobrjansky. 1989. Profound peripheral insulin resistance, independent of obesity, in polycystic ovary syndrome. Diabetes 38(9):1165–1174.
Dunn, S. E., E. Gunde, and H. Lee. 2015. Sex-based differences in multiple sclerosis (MS): Part II: Rising incidence of multiple sclerosis in women and the vulnerability of men to progression of this disease. Current Topics in Behavioral Neurosciences 26:57–86.
Dwyer, J. B., A. Aftab, R. Radhakrishnan, A. Widge, C. I. Rodriguez, L. L. Carpenter, C. B. Nemeroff, W. M. McDonald, and N. H. Kalin. 2020. Hormonal treatments for major depressive disorder: State of the art. American Journal of Psychiatry 177(8):686–705.
Eastell, R., C. J. Rosen, D. M. Black, A. M. Cheung, M. H. Murad, and D. Shoback. 2019. Pharmacological management of osteoporosis in postmenopausal women: An endocrine society clinical practice guideline. Journal of Clinical Endocrinology and Metabolism 104(5):1595–1622.
Edwards, R. R., R. H. Dworkin, M. D. Sullivan, D. C. Turk, and A. D. Wasan. 2016. The role of psychosocial processes in the development and maintenance of chronic pain. The Journal of Pain 17(9):T70–T92.
Eisenstein, M. 2020. Closing the gender gap in migraine research. Nature 586(7829):S16–S17.
Eisinger, R. W., C. W. Dieffenbach, and A. S. Fauci. 2019. HIV viral load and transmissibility of HIV infection: Undetectable equals untransmittable. JAMA 321(5):451–452.
Eken, H. N., E. C. Dee, A. R. Powers, and A. Jordan. 2021. Racial and ethnic differences in perception of provider cultural competence among patients with depression and anxiety symptoms: A retrospective, population-based, cross-sectional analysis. The Lancet Psychiatry 8(11):957–968.
El-Hakim, Y., K. K. Mani, A. Eldouh, S. Pandey, M. T. Grimaldo, A. Dabney, R. Pilla, and F. Sohrabji. 2021. Sex differences in stroke outcome correspond to rapid and severe changes in gut permeability in adult sprague-dawley rats. Biology of Sex Differences 12(1):14.
El Khoudary, S. R., B. Aggarwal, T. M. Beckie, H. N. Hodis, A. E. Johnson, R. D. Langer, M. C. Limacher, J. E. Manson, M. L. Stefanick, and M. A. Allison. 2020. Menopause transition and cardiovascular disease risk: Implications for timing of early prevention: A scientific statement from the American Heart Association. Circulation 142(25).
Elman-Shina, K., and S. Efrati. 2022. Ischemia as a common trigger for Alzheimer’s disease. Frontiers in Aging Neuroscience 14:1012779.
Elmlinger, M. W., W. Kühnel, M. M. Weber, and M. B. Ranke. 2004. Reference ranges for two automated chemiluminescent assays for serum insulin-like growth factor I (IGF-I) and IGF-binding protein 3 (IGFBP-3). Clinical Chemistry and Laboratory Medicine 42(6):654–664.
Emanuele, M. A., F. Wezeman, and N. V. Emanuele. 2002. Alcohol’s effects on female reproductive function. Alcohol Research & Health 26(4):274–281.
Emdin, C. A., C. X. Wong, A. J. Hsiao, D. G. Altman, S. A. Peters, M. Woodward, and A. A. Odutayo. 2016. Atrial fibrillation as risk factor for cardiovascular disease and death in women compared with men: Systematic review and meta-analysis of cohort studies. BMJ 532:h7013.
Engel, J. A., and E. Jerlhag. 2014. Chapter 9, Alcohol: Mechanisms along the mesolimbic dopamine systeme. In Progress in Brain Research, Dopamine. 1st ed., Vol 211, edited by M. Diana, G. Di Chara, and P. Spanco. Amsterdam, The Netherlands: Elsevier. Pp. 201–234.
Engel, S., H. Klusmann, B. Ditzen, C. Knaevelsrud, and S. Schumacher. 2019. Menstrual cycle-related fluctuations in oxytocin concentrations: A systematic review and meta-analysis. Frontiers in Neuroendocrinology 52:144–155.
Ensrud, K. E., and C. J. Crandall. 2024. Osteoporosis. Annals of Internal Medicine 177(1): Itc1–itc16.
Ephross, S. A., and S. M. Sinclair. 2014. Final results from the 16-year sumatriptan, saratriptan, and treximet pregnancy registry. Headache 54(7):1158–1172.
Epperson, C. N., D. R. Rubinow, S. Meltzer-Brody, K. M. Deligiannidis, R. Riesenberg, A. D. Krystal, K. Bankole, M. Y. Huang, H. Li, C. Brown, S. J. Kanes, and R. Lasser. 2023. Effect of brexanolone on depressive symptoms, anxiety, and insomnia in women with postpartum depression: Pooled analyses from 3 double-blind, randomized, placebo-controlled clinical trials in the Hummingbird clinical program. Journal of Affective Disorders 320:353–359.
Eriksson, J. G. 2016. Developmental origins of health and disease—from a small body size at birth to epigenetics. Annals of Medicine 48(6):456–467.
Evans, S. M., and F. R. Levin. 2011. Response to alcohol in women: Role of the menstrual cycle and a family history of alcoholism. Drug and Alcohol Dependence 114(1): 18–30.
Everitt, B. J., J. A. Parkinson, M. C. Olmstead, M. Arroyo, P. Robledo, and T. W. Robbins. 1999. Associative processes in addiction and reward the role of amygdala-ventral striatal subsystems. Annals of the New York Academy of Sciences 877(1):412–438.
Falsetti, I., G. Palmini, S. Donati, C. Aurilia, T. Iantomasi, and M. L. Brandi. 2024. Irisin and its role in postmenopausal osteoporosis and sarcopenia. Biomedicines 12(4).
Farlow, M. R., D. K. Lahiri, J. Poirier, J. Davignon, L. Schneider, and S. L. Hui. 1998. Treatment outcome of tacrine therapy depends on apolipoprotein genotype and gender of the subjects with Alzheimer’s disease. Neurology 50(3):669–677.
Farrell, M. R., D. R. Sengelaub, and C. L. Wellman. 2013. Sex differences and chronic stress effects on the neural circuitry underlying fear conditioning and extinction. Physiology and Behavior 122:208–215.
Farzadegan, H., D. R. Hoover, J. Astemborski, C. M. Lyles, J. B. Margolick, R. B. Markham, T. C. Quinn, and D. Vlahov. 1998. Sex differences in HIV-1 viral load and progression to AIDS. Lancet 352(9139):1510–1514.
Favretti, M., C. Iannuccelli, and M. Di Franco. 2023. Pain biomarkers in fibromyalgia syndrome: Current understanding and future directions. International Journal of Molecular Sciences 24(13):10443.
FDA (U.S. Food and Drug Administration). 2023. FDA News Release: FDA Approves First Oral Treatment for Postpartum Depression. https://www.fda.gov/news-events/press-announcements/fda-approves-first-oral-treatment-postpartum-depression/ (accessed June 18, 2024).
Felitti, V. J., R. F. Anda, D. Nordenberg, D. F. Williamson, A. M. Spitz, V. Edwards, M. P. Koss, and J. S. Marks. 1998. Relationship of childhood abuse and household dysfunction to many of the leading causes of death in adults. The Adverse Childhood Experiences (ACE) study. American Journal of Preventive Medicine 14(4):245–258.
Férézou-Viala, J., A.-F. Roy, C. Sérougne, D. Gripois, M. Parquet, V. Bailleux, A. Gertler, B. Delplanque, J. Djiane, M. Riottot, and M. Taouis. 2007. Long-term consequences of maternal high-fat feeding on hypothalamic leptin sensitivity and diet-induced obesity in the offspring. American Journal of Physiology—Regulatory, Integrative and Comparative Physiology 293(3):R1056–R1062.
Ferrari, M. D., R. R. Klever, G. M. Terwindt, C. Ayata, and A. M. van den Maagdenberg. 2015. Migraine pathophysiology: Lessons from mouse models and human genetics. Lancet Neurology 14(1):65–80.
Ferretti, M. T., M. F. Iulita, E. Cavedo, P. A. Chiesa, A. Schumacher Dimech, A. Santuccione Chadha, F. Baracchi, H. Girouard, S. Misoch, E. Giacobini, H. Depypere, H. Hampel, for the Women’s Brain, and the Alzheimer Precision Medicine Initiative. 2018. Sex differences in Alzheimer disease—the gateway to precision medicine. Nature Reviews Neurology 14(8):457–469.
Ferris, S., R. Lane, N. Sfikas, B. Winblad, M. Farlow, and H. H. Feldman. 2009. Effects of gender on response to treatment with rivastigmine in mild cognitive impairment: A post hoc statistical modeling approach. Gender Medicine 6(2):345–355.
Fields, J. A., V. D. Garovic, M. M. Mielke, K. Kantarci, M. Jayachandran, W. M. White, A. M. Butts, J. Graff-Radford, B. D. Lahr, K. R. Bailey, and V. M. Miller. 2017. Preeclampsia and cognitive impairment later in life. American Journal of Obstetrics and Gynecology 217(1):74.e71–74.e11.
Filippenkov, I. B., A. V. Khrunin, I. V. Mozgovoy, L. V. Dergunova, and S. A. Limborska. 2023. Are ischemic stroke and alzheimer’s disease genetically consecutive pathologies? Biomedicines 11(10).
Filippi, M., A. Bar-Or, F. Piehl, P. Preziosa, A. Solari, S. Vukusic, and M. A. Rocca. 2018. Multiple sclerosis. Nature Reviews Disease Primers 4(1):43.
Fillingim, R. B., C. D. King, M. C. Ribeiro-Dasilva, B. Rahim-Williams, and J. L. Riley, 3rd. 2009. Sex, gender, and pain: A review of recent clinical and experimental findings. Pain 10(5):447–485.
Fink, H. A., S. L. Harrison, B. C. Taylor, S. R. Cummings, J. T. Schousboe, M. A. Kuskowski, K. L. Stone, and K. E. Ensrud. 2008. Differences in site-specific fracture risk among older women with discordant results for osteoporosis at hip and spine: Study of osteoporotic fractures. Journal of Clinical Densitometry 11(2):250–259.
Finkelhor, D. P. 1987. The sexual abuse of children: Current research reviewed. Psychiatric Annals 17(4):233–237,241.
Fleet, J. C. 2022. Vitamin D-mediated regulation of intestinal calcium absorption. Nutrients 14(16).
Fleming, T. P., A. J. Watkins, M. A. Velazquez, J. C. Mathers, A. M. Prentice, J. Stephenson, M. Barker, R. Saffery, C. S. Yajnik, J. J. Eckert, M. A. Hanson, T. Forrester, P. D. Gluckman, and K. M. Godfrey. 2018. Origins of lifetime health around the time of conception: Causes and consequences. The Lancet 391(10132):1842–1852.
Flynn, H. A., C. Spino, C. Guille, K. M. Deligiannidis, P. Maki, J. Jahnke, K. L. Rosenblum, C. N. Epperson, and S. J. Weiss. 2018. A collaborative, network-based approach to advance women’s depression research in the United States: Preliminary findings. Journal of Women’s Health 27(1):51–57.
Fonseca, F., M. Robles-Martínez, J. Tirado-Muñoz, M. Alías-Ferri, J.-I. Mestre-Pintó, A. M. Coratu, and M. Torrens. 2021. A gender perspective of addictive disorders. Current Addiction Reports 8(1):89–99.
Ford, T. J., and C. Berry. 2019. How to diagnose and manage angina without obstructive coronary artery disease: Lessons from the British Heart Foundation Cormica trial. Interventional Cardiology 14(2):76–82.
Forger, N. G., and L. P. Morin. 1982. Reproductive state modulates ethanol intake in rats: Effects of ovariectomy, ethanol concentration, estrous cycle and pregnancy. Pharmacology Biochemistry and Behavior 17(2):323–331.
Forsyth, K. S., N. Jiwrajka, C. D. Lovell, N. E. Toothacre, and M. C. Anguera. 2024. The conneXion between sex and immune responses. Nature Reviews: Immunology. https://doi.org/10.1038/s41577-024-00996-9/ (accessed June 18, 2024).
Franchimont, P., C. Dourcy, J. J. Legros, A. Reuter, Y. Vrindts-Gevaert, J. R. Van Cauwenberge, and U. Gaspard. 1976. Prolactin levels during the menstrual cycle. Clinical Endocrinology 5(6):643–650.
Frank, D., B. F. Gruenbaum, A. Zlotnik, M. Semyonov, A. Frenkel, and M. Boyko. 2022. Pathophysiology and current drug treatments for post-stroke depression: A review. International Journal of Molecular Sciences 23(23).
Fredriksen-Goldsen, K. I., H. J. Kim, C. Shui, and A. E. B. Bryan. 2017. Chronic health conditions and key health indicators among lesbian, gay, and bisexual older U.S. adults, 2013–2014. American Journal of Public Health 107(8):1332–1338.
Frias, J. P., G. B. Macaraeg, J. Ofrecio, J. G. Yu, J. M. Olefsky, and Y. T. Kruszynska. 2001. Decreased susceptibility to fatty acid-induced peripheral tissue insulin resistance in women. Diabetes 50(6):1344–1350.
Fried, L. P., C. M. Tangen, J. Walston, A. B. Newman, C. Hirsch, J. Gottdiener, T. Seeman, R. Tracy, W. J. Kop, G. Burke, and M. A. McBurnie. 2001. Frailty in older adults: Evidence for a phenotype. The Journals of Gerontology: Series A 56(3):M146–M157.
Fried, S. K., M.-J. Lee, and K. Karastergiou. 2015. Shaping fat distribution: New insights into the molecular determinants of depot- and sex-dependent adipose biology. Obesity 23(7):1345–1352.
Galer, B. S., D. Lee, T. Ma, B. Nagle, and T. G. Schlagheck. 2005. Morphidex (morphine sulfate/dextromethorphan hydrobromide combination) in the treatment of chronic pain: Three multicenter, randomized, double-blind, controlled clinical trials fail to demonstrate enhanced opioid analgesia or reduction in tolerance. Pain 115(3):284–295.
Galvez-Sánchez, C. M., and G. A. Reyes Del Paso. 2020. Diagnostic criteria for fibromyalgia: Critical review and future perspectives. Journal of Clinical Medicine 9(4).
Gamer, J., D. J. Van Booven, O. Zarnowski, S. Arango, M. Elias, A. Kurian, A. Joseph, M. Perez, F. Collado, N. Klimas, E. Oltra, and L. Nathanson. 2023. Sex-dependent transcriptional changes in response to stress in patients with myalgic encephalomyelitis/chronic fatigue syndrome: A pilot project. International Journal of Molecular Sciences 24(12):10255.
Gandhi, R. T., R. Bedimo, J. F. Hoy, R. J. Landovitz, D. M. Smith, E. F. Eaton, C. Lehmann, S. A. Springer, P. E. Sax, M. A. Thompson, C. A. Benson, S. P. Buchbinder, C. Del Rio, J. J. Eron, Jr., H. F. Günthard, J. M. Molina, D. M. Jacobsen, and M. S. Saag. 2023. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2022 recommendations of the International Antiviral Society—USA Panel. JAMA 329(1):63–84.
Gannon, O. J., L. S. Robison, A. E. Salinero, C. Abi-Ghanem, F. M. Mansour, R. D. Kelly, A. Tyagi, R. R. Brawley, J. D. Ogg, and K. L. Zuloaga. 2022. High-fat diet exacerbates cognitive decline in mouse models of Alzheimer’s disease and mixed dementia in a sex-dependent manner. Journal of Neuroinflammation 19(1):110.
García-Cáceres, C., N. Lagunas, I. Calmarza-Font, I. Azcoitia, Y. Diz-Chaves, L. M. García-Segura, E. Baquedano, L. M. Frago, J. Argente, and J. A. Chowen. 2010. Gender differences in the long-term effects of chronic prenatal stress on the HPA axis and hypothalamic structure in rats. Psychoneuroendocrinology 35(10):1525–1535.
Garcia, M., S. L. Mulvagh, C. N. Merz, J. E. Buring, and J. E. Manson. 2016. Cardiovascular disease in women: Clinical perspectives. Circulation Research 118(8):1273–1293.
Gawliński, D., K. Gawlińska, M. Frankowska, and M. Filip. 2020. Maternal diet influences the reinstatement of cocaine-seeking behavior and the expression of melanocortin-4 receptors in female offspring of rats. Nutrients 12(5):1462.
Gawronska, J., C. Meads, L. Smith, C. Cao, N. Wang, and S. Walker. 2024. Association of oral contraceptive pill use and depression among US women. Journal of Affective Disorders 344:132–140.
GBD 2016 Disease and Injury Incidence and Prevalence Collaborators. 2017. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: A systematic analysis for the global burden of disease study 2016. Lancet 390(10100):1211–1259.
Gegenhuber, B., M. V. Wu, R. Bronstein, and J. Tollkuhn. 2022. Gene regulation by gonadal hormone receptors underlies brain sex differences. Nature 606(7912):153–159.
Gelaye, B., N. Do, S. Avila, J. Carlos Velez, Q. Y. Zhong, S. E. Sanchez, B. Lee Peterlin, and M. A. Williams. 2016. Childhood abuse, intimate partner violence and risk of migraine among pregnant women: An epidemiologic study. Headache 56(6):976–986.
Gendered Innovations. 2023. Heart Disease in Diverse Populations: Analyzing Sex and Gender. Stanford University. https://genderedinnovations.stanford.edu/case-studies/heart.html#tabs-2 (accessed June 18, 2024.
Gennari, L. 2005. Estrogen receptor gene polymorphisms and the genetics of osteoporosis: A huge review. American Journal of Epidemiology 161(4):307–320.
Geraci, A., R. Calvani, E. Ferri, E. Marzetti, B. Arosio, and M. Cesari. 2021. Sarcopenia and menopause: The role of estradiol. Frontiers in Endocrinology 12:682012.
Gerdes, L. U., B. Jeune, K. A. Ranberg, H. Nybo, and J. W. Vaupel. 2000. Estimation of apolipoprotein E genotype-specific relative mortality risks from the distribution of genotypes in centenarians and middle-aged men: Apolipoprotein E gene is a “frailty gene,” not a “longevity gene.” Genetic Epidemiology 19(3):202–210.
Germain, A., D. K. Barupal, S. M. Levine, and M. R. Hanson. 2020. Comprehensive circulatory metabolomics in ME/CFS reveals disrupted metabolism of acyl lipids and steroids. Metabolites 10(1):34.
Geter, A., M. Y. Sutton, C. Armon, and K. Buchacz. 2019. Disparities in viral suppression and medication adherence among women in the USA, 2011–2016. AIDS and Behavior 23(11):3015–3023.
Geter Fugerson, A., M. Y. Sutton, and D. Hubbard McCree. 2019. Social and structural determinants of HIV treatment and care among Hispanic women and Latinas living with HIV infection in the United States: A qualitative review: 2008–2018. Health Equity 3(1):581–587.
Ghazisaeidi, S., M. M. Muley, and M. W. Salter. 2023. Neuropathic pain: Mechanisms, sex differences, and potential therapies for a global problem. Annual Reviews of Pharmacology and Toxicology 63:565–583.
Giamberardino, M. A., G. Affaitati, R. Costantini, M. Guglielmetti, and P. Martelletti. 2020. Acute headache management in emergency department. A narrative review. Internal and Emergency Medicine 15(1):109–117.
Gibson, C. J., Y. Li, D. Bertenthal, A. J. Huang, and K. H. Seal. 2019. Menopause symptoms and chronic pain in a national sample of midlife women veterans. Menopause 26(7):708–713.
Gibson, C. J., R. C. Thurston, S. R. El Khoudary, K. Sutton-Tyrrell, and K. A. Matthews. 2013. Body mass index following natural menopause and hysterectomy with and without bilateral oophorectomy. International Journal of Obesity (London) 37(6):809–813.
Gillespie, C. W., and P. E. Morin. 2017. Trends and disparities in osteoporosis screening among women in the United States, 2008–2014. The American Journal of Medicine 130(3): 306–316.
Gilsanz, P., E. R. Mayeda, M. M. Glymour, C. P. Quesenberry, D. M. Mungas, C. DeCarli, A. Dean, and R. A. Whitmer. 2017. Female sex, early-onset hypertension, and risk of dementia. Neurology 89(18):1886–1893.
Gioiosa, L., X. Chen, R. Watkins, N. Klanfer, C. D. Bryant, C. J. Evans, and A. P. Arnold. 2008. Sex chromosome complement affects nociception in tests of acute and chronic exposure to morphine in mice. Hormones and Behavior 53(1):124–130.
Glader, E. L., B. Stegmayr, B. Norrving, A. Terént, K. Hulter-Asberg, P. O. Wester, and K. Asplund. 2003. Sex differences in management and outcome after stroke: A Swedish national perspective. Stroke 34(8):1970–1975.
Goadsby, P. J., and P. R. Holland. 2019. An update: Pathophysiology of migraine. Neurologic Clinics 37(4):651–671.
Goate, A., M.-C. Chartier-Harlin, M. Mullan, J. Brown, F. Crawford, L. Fidani, L. Giuffra, A. Haynes, N. Irving, L. James, R. Mant, P. Newton, K. Rooke, P. Roques, C. Talbot, M. Pericak-Vance, A. Roses, R. Williamson, M. Rossor, M. Owen, and J. Hardy. 1991. Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer’s disease. Nature 349(6311):704–706.
Goel, H., N. Binkley, M. Boggild, W. P. Chan, W. D. Leslie, E. McCloskey, S. L. Morgan, B. C. Silva, and A. M. Cheung. 2024. Clinical use of trabecular bone score: The 2023 ICSD official positions. Journal of Clinical Densitometry 27(1):101452.
Gold, S. M., and R. R. Voskuhl. 2016. Pregnancy and multiple sclerosis: From molecular mechanisms to clinical application. Seminars in Immunopathology 38(6):709–718.
Goldstein, J. M., A. Langer, and J. A. Lesser. 2021. Sex differences in disorders of the brain and heart—a global crisis of multimorbidity and novel opportunity. JAMA Psychiatry 78(1):7–8.
Gomez, S., V. Blumer, and F. Rodriguez. 2022. Unique cardiovascular disease risk factors in Hispanic individuals. Current Cardiovascular Risk Reports 16(7):53–61.
Goodwin, R. D., L. C. Dierker, M. Wu, S. Galea, C. W. Hoven, and A. H. Weinberger. 2022. Trends in U.S. depression prevalence from 2015 to 2020: The widening treatment gap. American Journal of Preventive Medicine 63(5):726–733.
Gordon, E. H., N. M. Peel, M. Samanta, O. Theou, S. E. Howlett, and R. E. Hubbard. 2017. Sex differences in frailty: A systematic review and meta-analysis. Experimental Gerontology 89:30–40.
Gordon, E. H., and R. E. Hubbard. 2019. Do sex differences in chronic disease underpin the sex-frailty paradox? Mechanisms of Ageing and Development 179:44–50.
Gormley, P., V. Anttila, B. S. Winsvold, P. Palta, T. Esko, T. H. Pers, K.-H. Farh, E. Cuenca-Leon, M. Muona, N. A. Furlotte, T. Kurth, A. Ingason, G. McMahon, L. Ligthart, G. M. Terwindt, M. Kallela, T. M. Freilinger, C. Ran, S. G. Gordon, A. H. Stam, S. Steinberg, G. Borck, M. Koiranen, L. Quaye, H. H. H. Adams, T. Lehtimäki, A.-P. Sarin, J. Wedenoja, D. A. Hinds, J. E. Buring, M. Schürks, P. M. Ridker, M. G. Hrafnsdottir, H. Stefansson, S. M. Ring, J.-J. Hottenga, B. W. J. H. Penninx, M. Färkkilä, V. Artto, M. Kaunisto, S. Vepsäläinen, R. Malik, A. C. Heath, P. A. F. Madden, N. G. Martin, G. W. Montgomery, M. I. Kurki, M. Kals, R. Mägi, K. Pärn, E. Hämäläinen, H. Huang, A. E. Byrnes, L. Franke, J. Huang, E. Stergiakouli, P. H. Lee, C. Sandor, C. Webber, Z. Cader, B. Muller-Myhsok, S. Schreiber, T. Meitinger, J. G. Eriksson, V. Salomaa, K. Heikkilä, E. Loehrer, A. G. Uitterlinden, A. Hofman, C. M. Van Duijn, L. Cherkas, L. M. Pedersen, A. Stubhaug, C. S. Nielsen, M. Männikkö, E. Mihailov, L. Milani, H. Göbel, A.-L. Esserlind, A. F. Christensen, T. F. Hansen, T. Werge, J. Kaprio, A. J. Aromaa, O. Raitakari, M. A. Ikram, T. Spector, M.-R. Järvelin, A. Metspalu, C. Kubisch, D. P. Strachan, M. D. Ferrari, A. C. Belin, M. Dichgans, M. Wessman, A. M. J. M. Van Den Maagdenberg, J.-A. Zwart, D. I. Boomsma, G. D. Smith, K. Stefansson, N. Eriksson, M. J. Daly, B. M. Neale, J. Olesen, D. I. Chasman, D. R. Nyholt, and A. Palotie. 2016. Meta-analysis of 375,000 individuals identifies 38 susceptibility loci for migraine. Nature Genetics 48(8):856–866.
Gourlay, M. L., R. A. Overman, and K. E. Ensrud. 2015. Bone density screening and re-screening in postmenopausal women and older men. Current Osteoporosis Reports 13(6): 390–398.
Grach, S. L., J. Seltzer, T. Y. Chon, and R. Ganesh. 2023. Diagnosis and management of myalgic encephalomyelitis/chronic fatigue syndrome. Mayo Clinic Proceedings 98(10):1544–1551.
Granella, F., G. Sances, G. Allais, R. E. Nappi, A. Tirelli, C. Benedetto, B. Brundu, F. Facchinetti, and G. Nappi. 2004. Characteristics of menstrual and nonmenstrual attacks in women with menstrually related migraine referred to headache centres. Cephalalgia 24(9):707–716.
Gräns, H., M. Nilsson, K. Dahlman-Wright, and B. Evengård. 2007. Reduced levels of oestrogen receptor beta mRNA in Swedish patients with chronic fatigue syndrome. Journal of Clinical Pathology 60(2):195–198.
Greendale, G. A., M. Sowers, W. Han, M. H. Huang, J. S. Finkelstein, C. J. Crandall, J. S. Lee, and A. S. Karlamangla. 2012. Bone mineral density loss in relation to the final menstrual period in a multiethnic cohort: Results from the study of women’s health across the nation (swan). Journal of Bone and Mineral Research 27(1):111–118.
Greendale, G. A., B. Sternfeld, M. Huang, W. Han, C. Karvonen-Gutierrez, K. Ruppert, J. A. Cauley, J. S. Finkelstein, S. F. Jiang, and A. S. Karlamangla. 2019. Changes in body composition and weight during the menopause transition. JCI Insight 4(5).
Gregory, S., L. Booi, N. Jenkins, K. Bridgeman, G. Muniz-Terrera, and F. R. Farina. 2023. Hormonal contraception and risk for cognitive impairment or Alzheimer’s disease and related dementias in young women: A scoping review of the evidence. Frontiers in Global Women’s Health 4:1289096.
Gregus, A. M., I. S. Levine, K. A. Eddinger, T. L. Yaksh, and M. W. Buczynski. 2021. Sex differences in neuroimmune and glial mechanisms of pain. Pain 162(8):2186–2200.
Greige, T., L. A. Bilello, J. M. Singleton, and J. A. Edlow. 2023. Acute headache in pregnant and post-partum patients: A clinical review. American Journal of Emergency Medicine 72:16–19.
Guedes De Aguiar, E. O., E. Moreira Marconi, B. B. Monteiro-Oliveira, A. C. Gomes-Santos, A. C. Coelho Oliveira, L. L. Paineiras-Domingos, D. C. Sá-Caputo, and M. Bernardo Filho. 2023.
Guerreiro, R., A. Wojtas, J. Bras, M. Carrasquillo, E. Rogaeva, E. Majounie, C. Cruchaga, C. Sassi, J. S. K. Kauwe, S. Younkin, L. Hazrati, J. Collinge, J. Pocock, T. Lashley, J. Williams, J.-C. Lambert, P. Amouyel, A. Goate, R. Rademakers, K. Morgan, J. Powell, P. St. George-Hyslop, A. Singleton, and J. Hardy. 2012. TREM2 variants in Alzheimer’s disease. New England Journal of Medicine 368(2):117–127.
Guintivano, J., E. M. Byrne, J. Kiewa, S. Yao, A. E. Bauer, K. A. Aberg, M. J. Adams, A. Campbell, M. L. Campbell, K. W. Choi, E. C. Corfield, A. Havdahl, D. Hucks, N. Koen, Y. Lu, M. L. Mægbæk, J. Mullaert, R. E. Peterson, L. M. Raffield, H. M. Sallis, J. M. Sealock, A. Walker, H. J. Watson, Y. Xiong, J. M. K. Yang, R. J. L. Anney, K. Gordon-Smith, L. Hubbard, L. A. Jones, R. Mihaescu, M. Nyegaard, A. F. Pardiñas, A. Perry, N. Saquib, A. H. Shadyab, A. Viktorin, O. A. Andreassen, T. B. Bigdeli, L. K. Davis, C.-L. Dennis, A. Di Florio, C. Dubertret, Y.-C. A. Feng, B. N. Frey, S. Grigoriadis, E. Gloaguen, I. Jones, J. L. Kennedy, H. Krohn, T. Kunovac Kallak, Y. Li, N. G. Martin, A. M. McIntosh, J. Milgrom, T. Munk-Olsen, T. Oberlander, C. M. Olsen, N. Ramoz, T. Reichborn-Kjennerud, E. Robertson Blackmore, D. Rubinow, A. Skalkidou, J. W. Smoller, D. J. Stein, Z. N. Stowe, V. Taylor, S. Tebeka, M. Tesli, R. J. Van Lieshout, E. J. C. G. van den Oord, S. N. Vigod, T. Werge, L. T. Westlye, D. C. Whiteman, H. J. Zar, N. Wray, S. Meltzer-Brody, and P. Sullivan. 2023. Meta-analyses of genome-wide association studies for postpartum depression. American Journal of Psychiatry 180(12):884–895.
Guo, H.-J., Y.-L. Ye, Y.-F. Gao, and Z.-H. Liu. 2024. Age at first birth is associated with the likelihood of frailty in middle-aged and older women: A population-based analysis from NHANES 1999–2018. Maturitas 181:107904.
Guo, L., M. B. Zhong, L. Zhang, B. Zhang, and D. Cai. 2022. Sex differences in Alzheimer’s disease: Insights from the multiomics landscape. Biological Psychiatry 91(1):61–71.
Guralnik, J. M., L. Ferrucci, C. F. Pieper, S. G. Leveille, K. S. Markides, G. V. Ostir, S. Studenski, L. F. Berkman, and R. B. Wallace. 2000. Lower extremity function and subsequent disability: Consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. Journals of Gerontology: Series A 55(4):M221–231.
Haapakoski, R., J. Mathieu, K. P. Ebmeier, H. Alenius, and M. Kivimäki. 2015. Cumulative meta-analysis of interleukins 6 and 1β, tumour necrosis factor α and C-reactive protein in patients with major depressive disorder. Brain, Behavior, and Immunity 49:206–215.
Halievski, K., S. Ghazisaeidi, and M. W. Salter. 2020. Sex-dependent mechanisms of chronic pain: A focus on microglia and P2X4r. Journal of Pharmocology and Experimential Therapeutics 375(1):202–209.
Hanseeuw, B. J., R. A. Betensky, H. I. L. Jacobs, A. P. Schultz, J. Sepulcre, J. A. Becker, D. M. O. Cosio, M. Farrell, Y. T. Quiroz, E. C. Mormino, R. F. Buckley, K. V. Papp, R. A. Amariglio, I. Dewachter, A. Ivanoiu, W. Huijbers, T. Hedden, G. A. Marshall, J. P. Chhatwal, D. M. Rentz, R. A. Sperling, and K. Johnson. 2019. Association of amyloid and tau with cognition in preclinical Alzheimer disease. JAMA Neurology 76(8):915.
Hantsoo, L., and C. N. Epperson. 2015. Premenstrual dysphoric disorder: Epidemiology and treatment. Current Psychiatry Reports 17(11):87.
Harlow, S. D., C. Karvonen-Gutierrez, M. R. Elliott, I. Bondarenko, N. E. Avis, J. T. Bromberger, M. M. Brooks, J. M. Miller, and B. D. Reed. 2017. It is not just menopause: Symptom clustering in the Study of Women’s Health Across the Nation. Women’s Midlife Health 3(1).
Hasan, L. K., J. Aljabban, M. Rohr, M. Mukhtar, N. Adapa, R. Salim, N. Aljabban, S. Syed, S. Syed, M. Panahiazar, D. Hadley, and W. Jarjour. 2021. Metaanalysis reveals genetic correlates of osteoporosis pathogenesis. The Journal of Rheumatology 48(6): 940–945.
Hashmi, J. A., and K. D. Davis. 2014. Deconstructing sex differences in pain sensitivity. Pain 155(1):10–13.
Hassan, H., I. Allen, E. Sofianopoulou, Y. Walburga, C. Turnbull, D. M. Eccles, M. Tischkowitz, P. Pharoah, and A. C. Antoniou. 2024. Long-term outcomes of hysterectomy with bilateral salpingo-oophorectomy: A systematic review and meta-analysis. American Journal of Obstetrics and Gynecology 230(1):44–57.
Häuser, W., and M. A. Fitzcharles. 2018. Facts and myths pertaining to fibromyalgia. Dialogues in Clinical Neuroscience 20(1):53–62.
Hayes, S. N., E. S. H. Kim, J. Saw, D. Adlam, C. Arslanian-Engoren, K. E. Economy, S. K. Ganesh, R. Gulati, M. E. Lindsay, J. H. Mieres, S. Naderi, S. Shah, D. E. Thaler, M. S. Tweet, and M. J. Wood. 2018. Spontaneous coronary artery dissection: Current state of the science: A scientific statement from the American Heart Association. Circulation 137(19):e523–e557.
Haywood, W. M., and E. B. Mukaetova-Ladinska. 2006. Sex influences on cholinesterase inhibitor treatment in elderly individuals with Alzheimer’s disease. The American Journal of Geriatric Pharmacotherapy 4(3):273–286.
Hedström, A. K., N. Brenner, J. Butt, J. Hillert, T. Waterboer, T. Olsson, and L. Alfredsson. 2021a. Overweight/obesity in young adulthood interacts with aspects of EBV infection in MS etiology. Neurology Neuroimmunology Neuroinflammation 8(1).
Hedström, A. K., J. Huang, N. Brenner, J. Butt, I. Kockum, T. Waterboer, T. Olsson, and L. Alfredsson. 2021b. Low sun exposure acts synergistically with high Epstein-Barr nuclear antigen 1 (EBNA-1) antibody levels in multiple sclerosis etiology. European Journal of Neurology 28(12):4146–4152.
Heim, C., D. Wagner, E. Maloney, D. A. Papanicolaou, L. Solomon, J. F. Jones, E. R. Unger, and W. C. Reeves. 2006. Early adverse experience and risk for chronic fatigue syndrome. Archives of General Psychiatry 63(11):1258.
Hellman, K. M., F. A. Oladosu, E. F. Garrison, G. E. Roth, K. E. Dillane, and F. F. Tu. 2021. Circulating sex steroids and bladder pain sensitivity in dysmenorrhea. Molecular Pain 17:174480692110352.
Herling de Oliveira, L. L., V. M. Correia, P. F. G. Nicz, P. R. Soares, and T. L. Scudeler. 2022. Minoca: One size fits all? Probably not-a review of etiology, investigation, and treatment. Journal of Clinical Medicine 11(19).
Hernandez-Leon, A., Y. E. De la Luz-Cuellar, V. Granados-Soto, M. E. González-Trujano, and A. Fernández-Guasti. 2018. Sex differences and estradiol involvement in hyperalgesia and allodynia in an experimental model of fibromyalgia. Hormones and Behavior 97:39–46.
HHS (Department of Health and Human Services). 2018. Physical Activity Guidelines for Americans. Washington, DC: Department of Health and Human Services.
Hickey, M., K. M. Moss, A. Brand, C. D. Wrede, S. M. Domchek, B. Meiser, G. D. Mishra, and H. Joffe. 2021. What happens after menopause? (WHAM): A prospective controlled study of depression and anxiety up to 12 months after premenopausal risk-reducing bilateral salpingo-oophorectomy. Gynecologic Oncology 161(2):527–534.
Hirschtritt, M. E., K. L. Delucchi, and M. Olfson. 2018. Outpatient, combined use of opioid and benzodiazepine medications in the United States, 1993–2014. Preventive Medicine Reports 9:49–54.
HIV.gov. 2023. National HIV/AIDS Strategy (2022–2025). https://www.hiv.gov/federal-response/national-hiv-aids-strategy/national-hiv-aids-strategy-2022-2025 (accessed February 29, 2024).
Hoffmann, D. E., R. B. Fillingim, and C. Veasley. 2022. The woman who cried pain: Do sex-based disparities still exist in the experience and treatment of pain? Journal of Law, Medicine, and Ethics 50(3):519–541.
Hogervorst, E., J. Williams, M. Budge, W. Riedel, and J. Jolles. 2000. The nature of the effect of female gonadal hormone replacement therapy on cognitive function in post-menopausal women: A meta-analysis. Neuroscience 101(3):485–512.
Holland, D., R. S. Desikan, A. M. Dale, and L. K. McEvoy. 2013. Higher rates of decline for women and apolipoprotein Eε4 carriers. American Journal of Neuroradiology 34(12):2287–2293.
Hollingworth, P., M. L. Hamshere, V. Moskvina, K. Dowzell, P. J. Moore, C. Foy, N. Archer, A. Lynch, S. Lovestone, C. Brayne, D. C. Rubinsztein, B. Lawlor, M. Gill, M. J. Owen, and J. Williams. 2006. Four components describe behavioral symptoms in 1,120 individuals with late-onset Alzheimer’s disease. Journal of the American Geriatrics Society 54(9):1348–1354.
Holtzman, D. M., J. Herz, and G. Bu. 2012. Apolipoprotein E and apolipoprotein E receptors: Normal biology and roles in Alzheimer disease. Cold Spring Harbor Perspectives in Medicine 2(3).
Honigberg, M. C., S. M. Zekavat, K. Aragam, P. Finneran, D. Klarin, D. L. Bhatt, J. L. Januzzi, Jr., N. S. Scott, and P. Natarajan. 2019. Association of premature natural and surgical menopause with incident cardiovascular disease. JAMA 322(24):2411–2421.
Horikawa, A., N. Miyakoshi, Y. Shimada, Y. Sugimura, and H. Kodama. 2015. A comparative study between intravenous and oral alendronate administration for the treatment of osteoporosis. Springerplus 4:675.
Horng, H. C., W. H. Chang, C. C. Yeh, B. S. Huang, C. P. Chang, Y. J. Chen, K. H. Tsui, and P. H. Wang. 2017. Estrogen effects on wound healing. International Journal of Molecular Sciences 18(11).
Howard, V. J., T. E. Madsen, D. O. Kleindorfer, S. E. Judd, J. D. Rhodes, E. Z. Soliman, B. M. Kissela, M. M. Safford, C. S. Moy, L. A. McClure, G. Howard, and M. Cushman. 2019. Sex and race differences in the association of incident ischemic stroke with risk factors. JAMA Neurology 76(2):179–186.
Hu, M., and J. B. Becker. 2008. Acquisition of cocaine self-administration in ovariectomized female rats: Effect of estradiol dose or chronic estradiol administration. Drug and Alcohol Dependence 94(1–3):56–62.
Hu, M., H. S. Crombag, T. E. Robinson, and J. B. Becker. 2003. Biological basis of sex differences in the propensity to self-administer cocaine. Neuropsychopharmacology 29(1):81–85.
Hua, X., D. P. Hibar, S. Lee, A. W. Toga, C. R. Jack, Jr., M. W. Weiner, and P. M. Thompson. 2010. Sex and age differences in atrophic rates: An ADNI study with n=1368 MRI scans. Neurobiology of Aging 31(8):1463–1480.
Huang, C., L. Huang, Y. Wang, X. Li, L. Ren, X. Gu, L. Kang, L. Guo, M. Liu, X. Zhou, J. Luo, Z. Huang, S. Tu, Y. Zhao, L. Chen, D. Xu, Y. Li, C. Li, L. Peng, Y. Li, W. Xie, D. Cui, L. Shang, G. Fan, J. Xu, G. Wang, Y. Wang, J. Zhong, C. Wang, J. Wang, D. Zhang, and B. Cao. 2021. 6–month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 397(10270):220–232.
Huang, G., D. Qian, Y. Liu, G. Qu, Y. Qian, and B. Pei. 2023. The association between frailty and osteoarthritis based on the NHANES and Mendelian Randomization Study. Archives of Medical Science 19(5):1545–1550.
Husain, M. A., B. Laurent, and M. Plourde. 2021. APOE and Alzheimer’s disease: From lipid transport to physiopathology and therapeutics. Frontiers in Neuroscience 15:630502.
Hyde, J. S., and A. H. Mezulis. 2020. Gender differences in depression: Biological, affective, cognitive, and sociocultural factors. Harvard Review of Psychiatry 28(1).
Hyeouk, Chris Hahm, Benjamin Lê Cook, Andrea Ault-Brutus, and Margarita Alegría. 2015. Intersection of race-ethnicity and gender in depression care: Screening, access, and minimally adequate treatment. Psychiatric Services 66(3):258–264.
Iantomasi, T., C. Romagnoli, G. Palmini, S. Donati, I. Falsetti, F. Miglietta, C. Aurilia, F. Marini, F. Giusti, and M. L. Brandi. 2023. Oxidative stress and inflammation in osteoporosis: Molecular mechanisms involved and the relationship with microRNAs. International Journal of Molecular Sciences 24(4):3772.
Ilesanmi-Oyelere, B. L., and M. C. Kruger. 2020. Nutrient and dietary patterns in relation to the pathogenesis of postmenopausal osteoporosis-a literature review. Life (Basel) 10(10).
IHS (International Headache Society). 2018. Headache Classification Committee of the International Headache Society (IHS) the International Classification of Headache Disorders, 3rd edition. Cephalalgia 38(1):1–211.
Iob, E., R. Lacey, and A. Steptoe. 2020. Adverse childhood experiences and depressive symptoms in later life: Longitudinal mediation effects of inflammation. Brain, Behavior, and Immunity 90:97–107.
IOM (Institute of Medicine). 2015. Beyond Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Redefining an Illness. Washington, DC: The National Academies Press.
ISCD (International Society for Clinical Densitometry). 2019. Indications for Bone Mineral Density (BMD) Testing. https://iscd.org/learn/official-positions/adult-positions/ (accessed March 14, 2024).
Ishii, R., T. J. Schwedt, S. K. Kim, G. Dumkrieger, C. D. Chong, and D. W. Dodick. 2020. Effect of migraine on pregnancy planning: Insights from the American Registry for Migraine Research. Mayo Clinic Proceedings 95(10):2079–2089.
Itoh, Y., L. C. Golden, N. Itoh, M. A. Matsukawa, E. Ren, V. Tse, A. P. Arnold, and R. R. Voskuhl. 2019. The X-linked histone demethylase KDM6A in CD4+ T lymphocytes modulates autoimmunity. Journal of Clinical Investigation 129(9):3852–3863.
Ivey, S. L., H. R. Hanley, C. Taylor, E. Stock, N. Vora, J. Woo, S. Johnson, C. N. Bairey Merz, and Right Care Women’s Cardiovascular Writing Group. 2022. Early identification and treatment of women’s cardiovascular risk factors prevents cardiovascular disease, saves lives, and protects future generations: Policy recommendations and take action plan utilizing policy levers. Clinical Cardiology 45(11):1100–1106.
Jack, C. R., D. S. Knopman, W. J. Jagust, R. C. Petersen, M. W. Weiner, P. S. Aisen, L. M. Shaw, P. Vemuri, H. J. Wiste, S. D. Weigand, T. G. Lesnick, V. S. Pankratz, M. C. Donohue, and J. Q. Trojanowski. 2013. Tracking pathophysiological processes in Alzheimer’s disease: An updated hypothetical model of dynamic biomarkers. The Lancet Neurology 12(2):207–216.
Jackson, L. R., T. E. Robinson, and J. B. Becker. 2005. Sex differences and hormonal influences on acquisition of cocaine self-administration in rats. Neuropsychopharmacology 31(1):129–138.
Jacobs, B. M., P. Tank, J. P. Bestwick, A. J. Noyce, C. R. Marshall, R. Mathur, G. Giovannoni, and R. Dobson. 2024. Modifiable risk factors for multiple sclerosis have consistent directions of effect across diverse ethnic backgrounds: A nested case-control study in an English population-based cohort. Journal of Neurology 271(1):241–253.
Jacobs, E. G., L. M. Holsen, K. Lancaster, N. Makris, S. Whitfield-Gabrieli, A. Remington, B. Weiss, S. Buka, A. Klibanski, and J. M. Goldstein. 2015. 17β-estradiol differentially regulates stress circuitry activity in healthy and depressed women. Neuropsychopharmacology 40(3):566–576.
Jacobs, M. M., E. Crall, and V. Menzies. 2023. Racial disparities in pain among women with fibromyalgia: Secondary data analysis of severity, interference with function, and response to guided imagery. Journal of Integrative and Complementary Medicine 29(11): 757–766.
Jacobson, L. T., E. M. Hade, T. C. Collins, K. L. Margolis, M. E. Waring, L. V. Van Horn, B. Silver, M. Sattari, C. E. Bird, K. Kimminau, K. Wambach, and M. L. Stefanick. 2018. Breastfeeding history and risk of stroke among parous postmenopausal women in the Women’s Health Initiative. Journal of the American Heart Association 7(17):e008739.
Janevic, M. R., S. J. McLaughlin, A. A. Heapy, C. Thacker, and J. D. Piette. 2017. Racial and socioeconomic disparities in disabling chronic pain: Findings from the health and retirement study. Journal of Pain 18(12):1459–1467.
Jašarević, E., and T. L. Bale. 2019. Prenatal and postnatal contributions of the maternal microbiome on offspring programming. Frontiers in Neuroendocrinology 55:100797.
Jayaraman, A., J. C. Carroll, T. E. Morgan, S. Lin, L. Zhao, J. M. Arimoto, M. P. Murphy, T. L. Beckett, C. E. Finch, R. D. Brinton, and C. J. Pike. 2012. 17β-estradiol and progesterone regulate expression of β-amyloid clearance factors in primary neuron cultures and female rat brain. Endocrinology 153(11):5467–5479.
Jeng, C., L.-J. Zhao, K. Wu, Y. Zhou, T. Chen, and H.-W. Deng. 2018. Race and socioeconomic effect on sarcopenia and sarcopenic obesity in the Louisiana Osteoporosis Study (LOS). JCSM Clinical Reports 3(2):1–8.
Jett, S., E. Schelbaum, G. Jang, C. Boneu Yepez, J. P. Dyke, S. Pahlajani, R. Diaz Brinton, and L. Mosconi. 2022. Ovarian steroid hormones: A long overlooked but critical contributor to brain aging and Alzheimer’s disease. Frontiers in Aging Neuroscience 14:948219.
Jha, S., Z. Wang, N. Laucis, and T. Bhattacharyya. 2015. Trends in media reports, oral bisphosphonate prescriptions, and hip fractures 1996–2012: An ecological analysis. Journal of Bone and Mineral Research 30(12):2179–2187.
Ji, H., T. J. Niiranen, F. Rader, M. Henglin, A. Kim, J. E. Ebinger, B. Claggett, C. N. B. Merz, and S. Cheng. 2021. Sex differences in blood pressure associations with cardiovascular outcomes. Circulation 143(7):761–763.
Ji, Y., A. Rizk, P. Voulalas, H. Aljohani, S. Akerman, G. Dussor, A. Keller, and R. Masri. 2019. Sex differences in the expression of calcitonin gene-related peptide receptor components in the spinal trigeminal nucleus. Neurobiology of Pain 6:100031.
Jiang, J., K. Young, and C. J. Pike. 2020. Second to fourth digit ratio (2d:4d) is associated with dementia in women. Early Human Development 149:105152.
Jiang, X., L. E. Good, R. Spinka, and P. F. Schnatz. 2016. Osteoporosis screening in postmenopausal women aged 50–64 years: BMI alone compared with current screening tools. Maturitas 83:59–64.
Jiao, B., X. Liu, L. Zhou, M. H. Wang, Y. Zhou, T. Xiao, W. Zhang, R. Sun, M. M. Waye, B. Tang, and L. Shen. 2015. Polygenic analysis of late-onset Alzheimer’s disease from mainland China. PloS One 10(12):e0144898.
Jilka, R. L., G. Hangoc, G. Girasole, G. Passeri, D. C. Williams, J. S. Abrams, B. Boyce, H. Broxmeyer, and S. C. Manolagas. 1992. Increased osteoclast development after estrogen loss: Mediation by interleukin-6. Science 257(5066):88–91.
Jiménez, M. C., J. E. Manson, N. R. Cook, I. Kawachi, S. Wassertheil-Smoller, B. Haring, R. Nassir, J. J. Rhee, S. Sealy-Jefferson, and K. M. Rexrode. 2019. Racial variation in stroke risk among women by stroke risk factors. Stroke 50(4):797–804.
Jin, H., H. J. Yoo, Y. A. Kim, J. H. Lee, Y. Lee, S. H. Kwon, Y. J. Seo, S. H. Lee, J. M. Koh, Y. Ji, A. R. Do, S. Won, and J. H. Seo. 2022. Unveiling genetic variants for age-related sarcopenia by conducting a genome-wide association study on Korean cohorts. Scientific Reports 12(1):3501.
Johannesdottir, F., M. S. Putman, S. M. Burnett-Bowie, J. S. Finkelstein, E. W. Yu, and M. L. Bouxsein. 2022. Age-related changes in bone density, microarchitecture, and strength in postmenopausal Black and White women: The SWAN longitudinal HR-PQCT study. Journal of Bone and Mineral Research 37(1):41–51.
Johansson, T., S. Vinther Larsen, M. Bui, W. E. Ek, T. Karlsson, and Å. Johansson. 2023. Population-based cohort study of oral contraceptive use and risk of depression. Epidemiology and Psychiatric Sciences 32:e39.
Jones, G., K. Trajanoska, A. J. Santanasto, N. Stringa, C. L. Kuo, J. L. Atkins, J. R. Lewis, T. Duong, S. Hong, M. L. Biggs, J. Luan, C. Sarnowski, K. L. Lunetta, T. Tanaka, M. K. Wojczynski, R. Cvejkus, M. Nethander, S. Ghasemi, J. Yang, M. C. Zillikens, S. Walter, K. Sicinski, E. Kague, C. L. Ackert-Bicknell, D. E. Arking, B. G. Windham, E. Boerwinkle, M. L. Grove, M. Graff, D. Spira, I. Demuth, N. van der Velde, L. de Groot, B. M. Psaty, M. C. Odden, A. E. Fohner, C. Langenberg, N. J. Wareham, S. Bandinelli, N. M. van Schoor, M. Huisman, Q. Tan, J. Zmuda, D. Mellström, M. Karlsson, D. A. Bennett, A. S. Buchman, P. L. De Jager, A. G. Uitterlinden, U. Völker, T. Kocher, A. Teumer, L. Rodriguéz-Mañas, F. J. García, J. A. Carnicero, P. Herd, L. Bertram, C. Ohlsson, J. M. Murabito, D. Melzer, G. A. Kuchel, L. Ferrucci, D. Karasik, F. Rivadeneira, D. P. Kiel, and L. C. Pilling. 2021. Genome-wide meta-analysis of muscle weakness identifies 15 susceptibility loci in older men and women. Nature Communications 12(1):654.
Jonsson, T., H. Stefansson, S. Steinberg, I. Jonsdottir, P. V. Jonsson, J. Snaedal, S. Bjornsson, J. Huttenlocher, A. I. Levey, J. J. Lah, D. Rujescu, H. Hampel, I. Giegling, O. A. Andreassen, K. Engedal, I. Ulstein, S. Djurovic, C. Ibrahim-Verbaas, A. Hofman, M. A. Ikram, C. M. van Duijn, U. Thorsteinsdottir, A. Kong, and K. Stefansson. 2013. Variant of TREM2 associated with the risk of Alzheimer’s disease. New England Journal of Medicine 368(2):107–116.
Joseph, J. J., A. Bennett, J. B. Echouffo Tcheugui, V. S. Effoe, J. B. Odei, B. Hidalgo, A. Dulin, M. M. Safford, D. M. Cummings, M. Cushman, and A. P. Carson. 2019. Ideal cardiovascular health, glycaemic status and incident Type 2 diabetes mellitus: The Reasons for Geographic and Racial Differences in Stroke (REGARDS) Study. Diabetologia 62(3):426–437.
Josephs, K. A., S. D. Weigand, and J. L. Whitwell. 2022. Characterizing amyloid-positive individuals with normal tau PET levels after 5 years: An ADNI study. Neurology 98(22):e2282–e2292.
Jukes, T. H., and S. Osawa. 1997. Further comments on codon reassignment. Journal of Molecular Evolution 45(1):1–3.
Juppi, H. K., S. Sipilä, N. J. Cronin, S. Karvinen, J. E. Karppinen, T. H. Tammelin, P. Aukee, V. Kovanen, U. M. Kujala, and E. K. Laakkonen. 2020. Role of menopausal transition and physical activity in loss of lean and muscle mass: A follow-up study in middle-aged Finnish women. Journal of Clinical Medicine 9(5).
Justice, A. J., and H. de Wit. 1999. Acute effects of d-amphetamine during the follicular and luteal phases of the menstrual cycle in women. Psychopharmacology 145(1):67–75.
Juul, A., P. Bang, N. T. Hertel, K. Main, P. Dalgaard, K. Jørgensen, J. Müller, K. Hall, and N. E. Skakkebaek. 1994. Serum insulin-like growth factor-i in 1030 healthy children, adolescents, and adults: Relation to age, sex, stage of puberty, testicular size, and body mass index. The Journal of Clinical Endocrinology & Metabolism 78(3):744–752.
Kaergaard, A., A. M. Hansen, K. Rasmussen, and J. H. Andersen. 2000. Association between plasma testosterone and work-related neck and shoulder disorders among female workers. Scandinavian Journal of Work, Environment and Health 26(4):292–298.
Kameda, T., H. Mano, T. Yuasa, Y. Mori, K. Miyazawa, M. Shiokawa, Y. Nakamaru, E. Hiroi, K. Hiura, A. Kameda, N. N. Yang, Y. Hakeda, and M. Kumegawa. 1997. Estrogen inhibits bone resorption by directly inducing apoptosis of the bone-resorbing osteoclasts. The Journal of Experimental Medicine 186(4):489–495.
Kanaya, A. M., D. Herrington, E. Vittinghoff, S. K. Ewing, K. Liu, M. J. Blaha, S. S. Dave, F. Qureshi, and N. R. Kandula. 2014. Understanding the high prevalence of diabetes in U.S. South Asians compared with four racial/ethnic groups: The MASALA and MESA studies. Diabetes Care 37(6):1621–1628.
Kane, A. E., and S. E. Howlett. 2021. Sex differences in frailty: Comparisons between humans and preclinical models. Mechanisms of Ageing and Development 198:111546.
Kantarci, K., V. J. Lowe, T. G. Lesnick, N. Tosakulwong, K. R. Bailey, J. A. Fields, L. T. Shuster, S. M. Zuk, M. L. Senjem, M. M. Mielke, C. Gleason, C. R. Jack, W. A. Rocca, and V. M. Miller. 2016. Early postmenopausal transdermal 17β-estradiol therapy and amyloid-β deposition. Journal of Alzheimer’s Disease 53(2):547–556.
Karamitrou, E. K., P. Anagnostis, K. Vaitsi, L. Athanasiadis, and D. G. Goulis. 2023. Early menopause and premature ovarian insufficiency are associated with increased risk of dementia: A systematic review and meta-analysis of observational studies. Maturitas 176:107792.
Karastergiou, K., S. R. Smith, A. S. Greenberg, and S. K. Fried. 2012. Sex differences in human adipose tissues—the biology of pear shape. Biology of Sex Differences 3(1):13.
Karim, Q. A., D. Archary, F. Barré-Sinoussi, K. Broliden, C. Cabrera, F. Chiodi, S. J. Fidler, T. N. Gengiah, C. Herrera, A. B. M. Kharsany, L. J. P. Liebenberg, S. Mahomed, E. Menu, C. Moog, G. Scarlatti, N. Seddiki, A. Sivro, and M. Cavarelli. 2022. Women for science and science for women: Gaps, challenges and opportunities towards optimizing preexposure prophylaxis for HIV-1 prevention. Frontiers in Immunology 13:1055042.
Karlamangla, A. S., S. M. Burnett-Bowie, and C. J. Crandall. 2018. Bone health during the menopause transition and beyond. Obstetrics and Gynecology Clinics of North America 45(4):695–708.
Karsan, N., and P. J. Goadsby. 2015. Calcitonin gene-related peptide and migraine. Current Opinion in Neurology 28(3):250–254.
Kashikar-Zuck, S., and T. V. Ting. 2014. Juvenile fibromyalgia: Current status of research and future developments. Nature Reviews Rheumatology 10(2):89–96.
Katz-Wise, S. L., B. Everett, E. A. Scherer, H. Gooding, C. E. Milliren, and S. B. Austin. 2015. Factors associated with sexual orientation and gender disparities in chronic pain among U.S. adolescents and young adults. Preventive Medicine Rep 2:765–772.
Kaunitz, A. M., E. Kapoor, and S. Faubion. 2021. Maintaining cognitive function in surgically menopausal women: The importance of estrogen. Menopause 28(4):349–351.
Keaveny, T. M., B. L. Clarke, F. Cosman, E. S. Orwoll, E. S. Siris, S. Khosla, and M. L. Bouxsein. 2020. Biomechanical computed tomography analysis (BCT) for clinical assessment of osteoporosis. Osteoporosis International 31(6):1025–1048.
Kellar, D., and S. Craft. 2020. Brain insulin resistance in Alzheimer’s disease and related disorders: Mechanisms and therapeutic approaches. Lancet Neurology 19(9):758–766.
Kendler, D., A. Chines, P. Clark, P. R. Ebeling, M. McClung, Y. Rhee, S. Huang, and R. K. Stad. 2020. Bone mineral density after transitioning from denosumab to alendronate. The Journal of Clinical Endocrinology & Metabolism 105(3):e255–e264.
Kendler, D. L., F. Cosman, R. K. Stad, and S. Ferrari. 2022. Denosumab in the treatment of osteoporosis: 10 years later: A narrative review. Advances in Therapy 39(1):58–74.
Kenkre, J. S., and J. Bassett. 2018. The bone remodelling cycle. Annals of Clinical Biochemistry 55(3):308–327.
Kenkre, T. S., P. Malhotra, B. D. Johnson, E. M. Handberg, D. V. Thompson, O. C. Marroquin, W. J. Rogers, C. J. Pepine, C. N. Bairey Merz, and S. F. Kelsey. 2017. Ten-year mortality in the WISE study (Women’s Ischemia Syndrome Evaluation). Circulation: Cardiovascular Quality and Outcomes 10(12).
Kent, D. M., L. L. Price, P. Ringleb, M. D. Hill, and H. P. Selker. 2005. Sex-based differences in response to recombinant tissue plasminogen activator in acute ischemic stroke: A pooled analysis of randomized clinical trials. Stroke 36(1):62–65.
Keogh, E. 2021. The gender context of pain. Health Psychology Review 15(3):454–481.
Keogh, E. 2022. Sex and gender differences in pain: Past, present, and future. Pain 163(S1): S108–S116.
Kerker, B. D., J. Zhang, E. Nadeem, R. E. K. Stein, M. S. Hurlburt, A. Heneghan, J. Landsverk, and S. McCue Horwitz. 2015. Adverse childhood experiences and mental health, chronic medical conditions, and development in young children. Academic Pediatrics 15(5):510–517.
Kessler, R. C., K. A. McGonagle, M. Swartz, D. G. Blazer, and C. B. Nelson. 1993. Sex and depression in the National Comorbidity Survey. I: Lifetime prevalence, chronicity and recurrence. Journal of Affective Disorders 29(2–3):85–96.
Keyes, K. M., B. F. Grant, and D. S. Hasin. 2008. Evidence for a closing gender gap in alcohol use, abuse, and dependence in the United States population. Drug and Alcohol Dependence 93(1–2):21–29.
Khadilkar, S. S. 2019. Musculoskeletal disorders and menopause. The Journal of Obstetrics and Gynecology of India 69(2):99–103.
Khan, N. A., S. S. Daskalopoulou, I. Karp, M. J. Eisenberg, R. Pelletier, M. A. Tsadok, K. Dasgupta, C. M. Norris, and L. Pilote. 2017. Sex differences in prodromal symptoms in acute coronary syndrome in patients aged 55 years or younger. Heart 103(11):863–869.
Khosla, S., B. L. Riggs, E. J. Atkinson, A. L. Oberg, C. Mavilia, F. Del Monte, L. J. Melton, and M. L. Brandi. 2004. Relationship of estrogen receptor genotypes to bone mineral density and to rates of bone loss in men. The Journal of Clinical Endocrinology & Metabolism 89(4):1808–1816.
Khosla, S., N. C. Wright, A. L. Elderkin, and D. P. Kiel. 2023. Osteoporosis in the USA: Prevention and unmet needs. The Lancet Diabetes & Endocrinology 11(1):19–20.
Khosravi, M., G. Sotoudeh, M. Amini, F. Raisi, A. Mansoori, and M. Hosseinzadeh. 2020. The relationship between dietary patterns and depression mediated by serum levels of folate and vitamin b12. BMC Psychiatry 20(1):63.
Khuddus, M. A., C. J. Pepine, E. M. Handberg, C. N. Bairey Merz, G. Sopko, A. A. Bavry, S. J. Denardo, S. P. McGorray, K. M. Smith, B. L. Sharaf, S. J. Nicholls, S. E. Nissen, and R. D. Anderson. 2010. An intravascular ultrasound analysis in women experiencing chest pain in the absence of obstructive coronary artery disease: A substudy from the National Heart, Lung and Blood Institute–sponsored Women’s Ischemia Syndrome Evaluation (WISE). Journal of Interventional Cardiology 23(6):511–519.
Kiarashi, J., J. Vanderpluym, C. L. Szperka, S. Turner, M. T. Minen, S. Broner, A. C. Ross, A. E. Wagstaff, M. Anto, M. Marzouk, T. S. Monteith, N. Rosen, S. L. Manrriquez, E. Seng, A. Finkel, and L. Charleston. 2021. Factors associated with, and mitigation strategies for, health care disparities faced by patients with headache disorders. Neurology 97(6):280–289.
Kiesner, J. 2012. Affective response to the menstrual cycle as a predictor of self-reported affective response to alcohol and alcohol use. Archives of Women’s Mental Health 15(6): 423–432.
Kim, D. R., T. L. Bale, and C. N. Epperson. 2015. Prenatal programming of mental illness: Current understanding of relationship and mechanisms. Current Psychiatry Reports 17(2).
Kim, H., M. Kim, S.-K. Im, and S. Fang. 2018. Mouse CRE-LOXP system: General principles to determine tissue-specific roles of target genes. Laboratory Animal Research 34(4):147.
Kim, S. C., D. H. Kim, H. Mogun, W. Eddings, J. M. Polinski, J. M. Franklin, and D. H. Solomon. 2016. Impact of the U.S. Food and Drug Administration’s safety-related announcements on the use of bisphosphonates after hip fracture. Journal of Bone and Mineral Research 31(8):1536–1540.
Kim, S. W., and R. Kim. 2020. The association between hormone therapy and sarcopenia in postmenopausal women: The Korea National Health and Nutrition Examination Survey, 2008–2011. Menopause 27(5):506–511.
Kippin, T. E., R. A. Fuchs, R. H. Mehta, J. M. Case, M. P. Parker, H. A. Bimonte-Nelson, and R. E. See. 2005. Potentiation of cocaine-primed reinstatement of drug seeking in female rats during estrus. Psychopharmacology 182(2):245–252.
Klein, L. B., and S. L. Martin. 2021. Sexual harassment of college and university students: A systematic review. Trauma, Violence, & Abuse 22(4):777–792.
Klein, S. L., and K. L. Flanagan. 2016. Sex differences in immune responses. Nature Reviews: Immunology 16(10):626–638.
Kleindorfer, D. O., A. Towfighi, S. Chaturvedi, K. M. Cockroft, J. Gutierrez, D. Lombardi-Hill, H. Kamel, W. N. Kernan, S. J. Kittner, E. C. Leira, O. Lennon, J. F. Meschia, T. N. Nguyen, P. M. Pollak, P. Santangeli, A. Z. Sharrief, S. C. Smith, T. N. Turan, and L. S. Williams. 2021. 2021 guideline for the prevention of stroke in patients with stroke and transient ischemic attack: A guideline from the American Heart Association/American Stroke Association. Stroke 52(7).
Kleykamp, B. A., M. C. Ferguson, E. McNicol, I. Bixho, L. M. Arnold, R. R. Edwards, R. Fillingim, H. Grol-Prokopczyk, D. C. Turk, and R. H. Dworkin. 2021. The prevalence of psychiatric and chronic pain comorbidities in fibromyalgia: An ACTTION systematic review. Seminars in Arthritis and Rheumatism 51(1):166–174.
Koob, G. F., and M. Le Moal. 2006. Chapter 1, What is addiction? In Neurobiology of Addiction: 1–22. Elsevier Inc.
Koob, G. F., and M. Le Moal. 2008. Review. Neurobiological mechanisms for opponent motivational processes in addiction. Philosophical Transactions of the Royal Society of London: Series B 363(1507):3113–3123.
Kosten, T. A., F. H. Gawin, T. R. Kosten, and B. J. Rounsaville. 1993. Gender differences in cocaine use and treatment response. Journal of Substance Abuse Treatment 10(1):63–66.
Krahé, B., and A. Berger. 2017. Longitudinal pathways of sexual victimization, sexual self-esteem, and depression in women and men. Psychological Trauma: Theory, Research, Practice, and Policy 9(2):147–155.
Krause, D. N., K. Warfvinge, K. A. Haanes, and L. Edvinsson. 2021. Hormonal influences in migraine—interactions of oestrogen, oxytocin and CGRP. Nature Reviews: Neurology 17(10):621–633.
Krueger, K., F. Lamenza, H. Gu, H. El-Hodiri, J. Wester, J. Oberdick, A. J. Fischer, and S. Oghumu. 2023. Sex differences in susceptibility to substance use disorder: Role for X chromosome inactivation and escape? Molecular and Cellular Neurosciences 125:103859.
Krupa, K., M. Parmar, and L. F. Delo. 2024. Romosozumab. In Statpearls. Treasure Island, FL: StatPearls Publishing LLC.
Kunadian, V., A. Chieffo, P. G. Camici, C. Berry, J. Escaned, A. H. E. M. Maas, E. Prescott, N. Karam, Y. Appelman, C. Fraccaro, G. Louise Buchanan, S. Manzo-Silberman, R. Al-Lamee, E. Regar, A. Lansky, J. D. Abbott, L. Badimon, D. J. Duncker, R. Mehran, D. Capodanno, and A. Baumbach. 2020. An EAPCI expert consensus document on ischaemia with non-obstructive coronary arteries in collaboration with European Society of Cardiology Working Group on Coronary Pathophysiology & Microcirculation endorsed by Coronary Vasomotor Disorders International Study Group. European Heart Journal 41(37):3504–3520.
Kundakovic, M., and D. Rocks. 2022. Sex hormone fluctuation and increased female risk for depression and anxiety disorders: From clinical evidence to molecular mechanisms. Frontiers in Neuroendocrinology 66:101010.
Kunkle, B. W., M. Schmidt, H.-U. Klein, A. C. Naj, K. L. Hamilton-Nelson, E. B. Larson, D. A. Evans, P. L. De Jager, P. K. Crane, J. D. Buxbaum, N. Ertekin-Taner, L. L. Barnes, M. D. Fallin, J. J. Manly, R. C. P. Go, T. O. Obisesan, M. I. Kamboh, D. A. Bennett, K. S. Hall, A. M. Goate, T. M. Foroud, E. R. Martin, L.-S. Wang, G. S. Byrd, L. A. Farrer, J. L. Haines, G. D. Schellenberg, R. Mayeux, M. A. Pericak-Vance, C. Reitz, Writing Group for the Alzheimer’s Disease Genetics Consortium, N. R. Graff-Radford, I. Martinez, T. Ayodele, M. W. Logue, L. B. Cantwell, M. Jean-Francois, A. B. Kuzma, L. D. Adams, J. M. Vance, M. L. Cuccaro, J. Chung, J. Mez, K. L. Lunetta, G. R. Jun, O. L. Lopez, H. C. Hendrie,
E. M. Reiman, N. W. Kowall, J. B. Leverenz, S. A. Small, A. I. Levey, T. E. Golde, A. J. Saykin, T. D. Starks, M. S. Albert, B. T. Hyman, R. C. Petersen, M. Sano. T. Wisniewski, R. Vassar, J. A. Kaye, V. W. Henderson, C. DeCarli, F. M. LaFerla, J. B. Brewer, B. L. Miller, R. H. Swerdlow, L. J. Van Eldik, H. L. Paulson, J. Q. Trojanowski, S. Asthana, J. C. Morris, S. M. Strittmatter, W. A. Kukull. 2021. Novel Alzheimer disease risk loci and pathways in African American individuals using the African Genome Resources Panel: A meta-analysis. JAMA Neurology 78(1):102–113.
Kuntsche, E., and S. Müller. 2011. Why do young people start drinking? Motives for first-time alcohol consumption and links to risky drinking in early adolescence. European Addiction Research 18(1):34–39.
Kurkinen, M. 2023. Lecanemab (leqembi) is not the right drug for patients with Alzheimer’s disease. Advances in Clinical and Experimental Medicine 32(9):943–947.
Kwak, J. Y., and K. S. Kwon. 2019. Pharmacological interventions for treatment of sarcopenia: Current status of drug development for sarcopenia. Annals of Geriatric Medicine and Research 23(3):98-104.
Kwan, T. W., S. S. Wong, Y. Hong, A. M. Kanaya, S. S. Khan, L. L. Hayman, S. H. Shah, F. K. Welty, P. C. Deedwania, A. Khaliq, and L. P. Palaniappan. 2023. Epidemiology of diabetes and atherosclerotic cardiovascular disease among Asian American adults: Implications, management, and future directions: A scientific statement from the American Heart Association. Circulation 148(1):74–94.
L’Engle, K., E. Trejo, A. Landeros, E. Zúñiga Sandoval, J. Jauregui, and S. Yang. 2023. Brief peer coaching complements daily digital messages for chronic disease prevention among young adult Latinas. Translational Behavioral Medicine 14(2):80–88.
Labastida-Ramírez, A., E. Rubio-Beltrán, C. M. Villalón, and A. MaassenVanDenBrink. 2019. Gender aspects of CGRP in migraine. Cephalalgia 39(3):435–444.
Lacerda, E. M., K. Geraghty, C. C. Kingdon, L. Palla, and L. Nacul. 2019. A logistic regression analysis of risk factors in ME/CFS pathogenesis. BMC Neurology 19(1):275.
Lachman, M. E., S. Teshale, and S. Agrigoroaei. 2015. Midlife as a pivotal period in the life course. International Journal of Behavioral Development 39(1):20–31.
Lagunas, N., M. Marraudino, M. de Amorim, H. Pinos, P. Collado, G. Panzica, L. M. Garcia-Segura, and D. Grassi. 2019. Estrogen receptor beta and G protein–coupled estrogen receptor 1 are involved in the acute estrogenic regulation of arginine–vasopressin immunoreactive levels in the supraoptic and paraventricular hypothalamic nuclei of female rats. Brain Research 1712:93–100.
Lahoz, C., E. J. Schaefer, L. A. Cupples, P. W. F. Wilson, D. Levy, D. Osgood, S. Parpos, J. Pedro-Botet, J. A. Daly, and J. M. Ordovas. 2001. Apolipoprotein E genotype and cardiovascular disease in the Framingham Heart Study. Atherosclerosis 154(3):529–537.
Lahti, J., M. Lahti, A.-K. Pesonen, K. Heinonen, E. Kajantie, T. Forsén, K. Wahlbeck, C. Osmond, D. J. P. Barker, J. G. Eriksson, and K. Räikkönen. 2014. Prenatal and childhood growth, and hospitalization for alcohol use disorders in adulthood: The Helsinki Birth Cohort Study. PloS One 9(1):e87404.
Lambert, J. C., A. Ramirez, B. Grenier-Boley, and C. Bellenguez. 2023. Step by step: Towards a better understanding of the genetic architecture of Alzheimer’s disease. Molecular Psychiatry 28(7):2716–2727.
Lambrinoudaki, I., S. Stergiotis, P. Chatzivasileiou, A. Augoulea, P. Anagnostis, E. Armeni, D. Rizos, G. Kaparos, A. Alexandrou, G. Georgiopoulos, C. Kontogiannis, and K. Stamatelopoulos. 2020. Anti-Müllerian hormone concentrations are inversely associated with subclinical atherosclerosis in premenopausal women. Angiology 71(6):552–558.
Landi, F., R. Calvani, M. Tosato, A. Martone, E. Ortolani, G. Savera, A. Sisto, and E. Marzetti. 2016. Anorexia of aging: Risk factors, consequences, and potential treatments. Nutrients 8(2):69.
Lara-Cinisomo, S., T. D. Akinbode, and J. Wood. 2020. A systematic review of somatic symptoms in women with depression or depressive symptoms: Do race or ethnicity matter? Journal of Women’s Health 29(10):1273–1282.
Larkin, H. 2024. What to know about prevent, the AHA’s new cardiovascular disease risk calculator. JAMA 331(4):277.
Larsen, S. V., A. P. Mikkelsen, Ø. Lidegaard, and V. G. Frokjaer. 2023. Depression associated with hormonal contraceptive use as a risk indicator for postpartum depression. JAMA Psychiatry 80(7):682–689.
Lazaro, C. M., J. A. Victorio, A. P. Davel, and H. C. F. Oliveira. 2023. CETP expression ameliorates endothelial function in female mice through estrogen receptor-α and endothelial nitric oxide synthase pathway. American Journal of Physiology: Heart and Circulatory Physiology 325(3):H592–h600.
Leadbeater, B. J., G. P. Kuperminc, S. J. Blatt, and C. Hertzog. 1999. A multivariate model of gender differences in adolescents’ internalizing and externalizing problems. Developmental Psychology 35(5):1268–1282.
Leavitt, V. M., J. D. Dworkin, R. Galioto, and A. S. Ratzan. 2024. Disparities in DMT treatment: Demographic and neurocognitive differences between MS patients currently treated versus not treated with disease-modifying therapies. Multiple Sclerosis and Related Disorders 85:105508.
Lee, C. D., R. E. Nappi, and C. Cwiak. 2023a. Oral contraceptives for menstrual migraine with aura. New England Journal of Medicine 389(22):2102–2104.
Lee, H., S. Park, K.-S. Kwack, and J. S. Yun. 2023b. CT and MR for bone mineral density and trabecular bone score assessment in osteoporosis evaluation. Scientific Reports 13(1).
Lee, J. J., G. Cook-Wiens, B. D. Johnson, G. D. Braunstein, S. L. Berga, F. Z. Stanczyk, C. J. Pepine, C. N. B. Merz, and C. L. Shufelt. 2019. Age at menarche and risk of cardiovascular disease outcomes: Findings from the National Heart Lung and Blood Institute–sponsored Women’s Ischemia Syndrome Evaluation. Journal of the American Heart Association 8(12):e012406.
Leeners, B., N. Geary, P. N. Tobler, and L. Asarian. 2017. Ovarian hormones and obesity. Human Reproduction Update 23(3):300–321.
Leffert, L. R., C. R. Clancy, B. T. Bateman, A. S. Bryant, and E. V. Kuklina. 2015. Hypertensive disorders and pregnancy-related stroke: Frequency, trends, risk factors, and outcomes. Obstetrics and Gynecology 125(1):124–131.
LeGates, T. A., M. D. Kvarta, and S. M. Thompson. 2019. Sex differences in antidepressant efficacy. Neuropsychopharmacology 44(1):140–154.
Legato, M. J., P. A. Johnson, and J. E. Manson. 2016. Consideration of sex differences in medicine to improve health care and patient outcomes. JAMA 316(18):1865–1866.
Lems, W. F., J. Paccou, J. Zhang, N. R. Fuggle, M. Chandran, N. C. Harvey, C. Cooper, K. Javaid, S. Ferrari, and K. E. Akesson. 2021. Vertebral fracture: Epidemiology, impact and use of DXA vertebral fracture assessment in fracture liaison services. Osteoporosis International 32(3):399–411.
Lenchik, L., A. A. Weaver, R. J. Ward, J. M. Boone, and R. D. Boutin. 2018. Opportunistic screening for osteoporosis using computed tomography: State of the art and argument for paradigm shift. Current Rheumatology Reports 20(12).
Lenert, M. E., A. Avona, K. M. Garner, L. R. Barron, and M. D. Burton. 2021. Sensory neurons, neuroimmunity, and pain modulation by sex hormones. Endocrinology 162(8).
Lerario, M. P., M. Fusunyan, C. D. Stave, V. Roldán, A. S. Keuroghlian, J. Turban, D. L. Perez, T. Maschi, and N. Rosendale. 2023. Functional neurological disorder and functional somatic syndromes among sexual and gender minority people: A scoping review. Journal of Psychosomatic Research 174:111491.
Lescai, F., C. Pirazzini, G. D’Agostino, A. Santoro, R. Ghidoni, L. Benussi, D. Galimberti, E. Federica, F. Marchegiani, M. Cardelli, F. Olivieri, B. Nacmias, S. Sorbi, S. Bagnoli, F. Tagliavini, D. Albani, F. Martinelli Boneschi, G. Binetti, G. Forloni, P. Quadri, E. Scarpini, and C. Franceschi. 2010. Failure to replicate an association of RS5984894 SNP in the PCDH11X gene in a collection of 1,222 Alzheimer’s disease affected patients. Journal of Alzheimer’s Disease 21(2):385–388.
Lesnak, J. B., S. Inoue, L. Lima, L. Rasmussen, and K. A. Sluka. 2020. Testosterone protects against the development of widespread muscle pain in mice. Pain 161(12):2898–2908.
Leung, L. 2012. Pain catastrophizing: An updated review. Indian Journal of Psychological Medicine 34(3):204–217.
Leutner, M., M. Butylina, C. Matzhold, P. Klimek, C. Cuhaj, L. Bellach, S. Baumgartner-Parzer, B. Reiter, K. Preindl, A. Kautzky, T. Stimpfl, S. Thurner, P. Pietschmann, C. Fürnsinn, and A. Kautzky-Willer. 2023. Simvastatin therapy in higher dosages deteriorates bone quality: Consistent evidence from population-wide patient data and interventional mouse studies. Biomedicine and Pharmacotherapy 158:114089.
Levin, M. 2022. Classification and diagnosis of primary headache disorders. Seminars in Neurology 42(4):406–417.
Levy-Lahad, E., W. Wasco, P. Poorkaj, D. M. Romano, J. Oshima, W. H. Pettingell, C.-e. Yu, P. D. Jondro, S. D. Schmidt, K. Wang, A. C. Crowley, Y.-H. Fu, S. Y. Guenette, D. Galas, E. Nemens, E. M. Wijsman, T. D. Bird, G. D. Schellenberg, and R. E. Tanzi. 1995. Candidate gene for the chromosome 1 familial Alzheimer’s disease locus. Science 269(5226):973–977.
Lewiecki, E. M., and S. F. Erb. 2022. Racial disparities and inequalities in the management of patients with osteoporosis. Orthopaedic Nursing 41(2):125–134.
Ley, S. H., Y. Li, D. K. Tobias, J. E. Manson, B. Rosner, F. B. Hu, and K. M. Rexrode. 2017. Duration of reproductive life span, age at menarche, and age at menopause are associated with risk of cardiovascular disease in women. Journal of American Heart Association 6(11).
Li, C., M. Xie, W. Wang, Y. Liu, D. Liao, J. Yin, and H. Huang. 2022a. Association between polymorphisms in estrogen receptor genes and depression in women: A meta-analysis. Frontiers in Genetics 13.
Li, J., X. Chen, R. McClusky, M. Ruiz-Sundstrom, Y. Itoh, S. Umar, A. P. Arnold, and M. Eghbali. 2014. The number of X chromosomes influences protection from cardiac ischaemia/reperfusion injury in mice: One X is better than two. Cardiovascular Research 102(3):375–384.
Li, J. X. L., X. Wang, A. Henry, C. S. Anderson, N. Hammond, K. Harris, H. Liu, K. Loffler, J. Myburgh, J. Pandian, B. Smyth, B. Venkatesh, C. Carcel, and M. Woodward. 2023. Sex differences in pain expressed by patients across diverse disease states: Individual patient data meta-analysis of 33,957 participants in 10 randomized controlled trials. Pain 164(8):1666–1676.
Li, L., Y. Wang, C. Yang, C. Huang, L. Duan, J. Zhou, Y. Lu, and G. Zhao. 2023. Frailty in hypertensive population and its association with all-cause mortality: Data from the National Health and Nutrition Examination Survey. Frontiers in Cardiovascular Medicine 10:945468.
Li, P., L. Mao, M. Hu, Z. Lu, X. Yuan, Y. Zhang, and Z. Hu. 2022b. Mindfulness on rumination in patients with depressive disorder: A systematic review and meta-analysis of randomized controlled trials. International Journal of Environmental Research and Public Health 19(23):16101.
Liang, G., A. S. F. Kow, R. Yusof, C. L. Tham, Y. C. Ho, and M. T. Lee. 2024. Menopause-associated depression: Impact of oxidative stress and neuroinflammation on the central nervous system—a review. Biomedicines 12(1):184.
Lichtman, J. H., E. C. Leifheit, B. Safdar, H. Bao, H. M. Krumholz, N. P. Lorenze, M. Daneshvar, J. A. Spertus, and G. D’Onofrio. 2018. Sex differences in the presentation and perception of symptoms among young patients with myocardial infarction. Circulation 137(8):781–790.
Lim, E.-J., and C.-G. Son. 2020. Review of case definitions for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Journal of Translational Medicine 18(1):289.
Lim, E.-J., Y.-C. Ahn, E.-S. Jang, S.-W. Lee, S.-H. Lee, and C.-G. Son. 2020. Systematic review and meta-analysis of the prevalence of chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME). Journal of Translational Medicine 18(1):100.
Lim, U., S. Wang, S. Y. Park, D. Bogumil, A. H. Wu, I. Cheng, C. A. Haiman, L. Le Marchand, L. R. Wilkens, L. White, and V. W. Setiawan. 2022. Risk of Alzheimer’s disease and related dementia by sex and race/ethnicity: The multiethnic cohort study. Alzheimer’s Dementia 18(9):1625–1634.
Lin, K. A., K. R. Choudhury, B. G. Rathakrishnan, D. M. Marks, J. R. Petrella, and P. M. Doraiswamy. 2015. Marked gender differences in progression of mild cognitive impairment over 8 years. Alzheimer’s Dementia 1(2):103–110.
Lin, T.-K., P. Chou, C.-H. Lin, Y.-J. Hung, and G.-P. Jong. 2018. Long-term effect of statins on the risk of new-onset osteoporosis: A nationwide population-based cohort study. PloS One 13(5):e0196713.
Lindberg, M., S. Alatalo, J. Halleen, S. Mohan, J. Gustafsson, and C. Ohlsson. 2001. Estrogen receptor specificity in the regulation of the skeleton in female mice. Journal of Endocrinology 171(2):229–236.
Linher-Melville, K., A. Shah, and G. Singh. 2020. Sex differences in neuro(auto)immunity and chronic sciatic nerve pain. Biology of Sex Differences 11(1):62.
Link, J. C., X. Chen, C. Prien, M. S. Borja, B. Hammerson, M. N. Oda, A. P. Arnold, and K. Reue. 2015. Increased high-density lipoprotein cholesterol levels in mice with XX versus XY sex chromosomes. Arteriosclerosis, Thrombosis, and Vascular Biology 35(8):1778–1786.
Link, T. M., and G. Kazakia. 2020. Update on imaging-based measurement of bone mineral density and quality. Current Rheumatology Reports 22(5):13.
Lipner, E., N. Mac Giollabhui, E. C. Breen, B. A. Cohn, N. Y. Krigbaum, P. M. Cirillo, T. M. Olino, L. B. Alloy, and L. M. Ellman. 2024. Sex-specific pathways from prenatal maternal inflammation to adolescent depressive symptoms. JAMA Psychiatry. 81(5): 498–505.
Lisabeth, L. D., A. S. Beiser, D. L. Brown, J. M. Murabito, M. Kelly-Hayes, and P. A. Wolf. 2009a. Age at natural menopause and risk of ischemic stroke: The Framingham Heart Study. Stroke 40(4):1044–1049.
Lisabeth, L. D., D. L. Brown, R. Hughes, J. J. Majersik, and L. B. Morgenstern. 2009b. Acute stroke symptoms: Comparing women and men. Stroke 40(6):2031–2036.
Liu, A. Y., H. M. Scott, and S. P. Buchbinder. 2023. New USPSTF guidelines for HIV preexposure prophylaxis: Will more choices lead to greater impact? JAMA 330(8):699–701.
Liu, C.-C., T. Kanekiyo, H. Xu, and G. Bu. 2013. Apolipoprotein E and Alzheimer disease: Risk, mechanisms and therapy. Nature Reviews Neurology 9(2):106–118.
Liu, P., Y. Ji, T. Yuen, E. Rendina-Ruedy, V. E. DeMambro, S. Dhawan, W. Abu-Amer, S. Izadmehr, B. Zhou, A. C. Shin, R. Latif, P. Thangeswaran, A. Gupta, J. Li, V. Shnayder, S. T. Robinson, Y. E. Yu, X. Zhang, F. Yang, P. Lu, Y. Zhou, L. L. Zhu, D. J. Oberlin, T. F. Davies, M. R. Reagan, A. Brown, T. R. Kumar, S. Epstein, J. Iqbal, N. G. Avadhani, M. I. New, H. Molina, J. B. van Klinken, E. X. Guo, C. Buettner, S. Haider, Z. Bian, L. Sun, C. J. Rosen, and M. Zaidi. 2017. Blocking fsh induces thermogenic adipose tissue and reduces body fat. Nature 546(7656):107–112.
Liu, F., Z. Li, J. Li, C. Siegel, R. Yuan, and L. D. McCullough. 2009. Sex differences in caspase activation after stroke. Stroke 40(5):1842–1848.
Liu, P., Y. Ji, T. Yuen, E. Rendina-Ruedy, V. E. DeMambro, S. Dhawan, W. Abu-Amer, S. Izadmehr, B. Zhou, A. C. Shin, R. Latif, P. Thangeswaran, A. Gupta, J. Li, V. Shnayder, S. T. Robinson, Y. E. Yu, X. Zhang, F. Yang, P. Lu, Y. Zhou, L. L. Zhu, D. J. Oberlin, T. F. Davies, M. R. Reagan, A. Brown, T. R. Kumar, S. Epstein, J. Iqbal, N. G. Avadhani, M. I. New, H. Molina, J. B. van Klinken, E. X. Guo, C. Buettner, S. Haider, Z. Bian, L. Sun, C. J. Rosen, and M. Zaidi. 2017. Blocking fsh induces thermogenic adipose tissue and reduces body fat. Nature 546(7656):107–112.
Liu, Y., T. Paajanen, E. Westman, L.-O. Wahlund, A. Simmons, C. Tunnard, T. Sobow, P. Proitsi, J. Powell, P. Mecocci, M. Tsolaki, B. Vellas, S. Muehlboeck, A. Evans, C. Spenger, S. Lovestone, H. Soininen, and AddNeuroMed Consortium. 2010. Effect of ApoE ε4 allele on cortical thicknesses and volumes: The AddNeuroMed study. Journal of Alzheimer’s Disease 21:947–966.
Lizcano, F., and G. Guzmán. 2014. Estrogen deficiency and the origin of obesity during menopause. Biomedical Research International 2014:757461.
Lloyd-Jones, D. M., N. B. Allen, C. A. M. Anderson, T. Black, L. C. Brewer, R. E. Foraker, M. A. Grandner, H. Lavretsky, A. M. Perak, G. Sharma, and W. Rosamond. 2022. Life’s essential 8: Updating and enhancing the American Heart Association’s construct of cardiovascular health: A presidential advisory from the American Heart Association. Circulation 146(5):e18–e43.
Lo, J. C., M. Chandra, C. Lee, J. A. Darbinian, M. Ramaswamy, and B. Ettinger. 2020. Bone mineral density in older U.S. Filipino, Chinese, Japanese, and White women. Journal of the American Geriatrics Society 68(11):2656–2661.
Lo, J. C., W. Yang, J. J. Park-Sigal, and S. M. Ott. 2023. Osteoporosis and fracture risk among older U.S. Asian adults. Current Osteoporosis Reports 21(5):592–608.
Lobo, R. A., and D. L. Cassidenti. 1992. Pharmacokinetics of oral 17 beta-estradiol. Journal of Reproductive Medicine 37(1):77–84.
Logue, M. W., M. Schu, B. N. Vardarajan, J. Buros, R. C. Green, R. C. P. Go, P. Griffith, T. O. Obisesan, R. Shatz, A. Borenstein, L. A. Cupples, K. L. Lunetta, M. D. Fallin, C. T. Baldwin, L. A. Farrer; Multi-Institutional Research on Alzheimer Genetic Epidemiology Study Group. 2011. A comprehensive genetic association study of Alzheimer disease in African Americans. Archives of Neurology 68(12):1569–1579.
Lopez-Lee, C., E. R. S. Torres, G. Carling, and L. Gan. 2024. Mechanisms of sex differences in alzheimer’s disease. Neuron 112(8):1208–1221.
Lorius, N., J. J. Locascio, D. M. Rentz, K. A. Johnson, R. A. Sperling, A. Viswanathan, and G. A. Marshall. 2015. Vascular disease and risk factors are associated with cognitive decline in the Alzheimer disease spectrum. Alzheimer’s Disease & Associated Disorders 29(1):18–25.
Loud, K. J., and C. M. Gordon. 2006. Adolescent bone health. Archives of Pediatrics and Adolescent Medicine 160(10):1026.
Lu, C.-B., P.-F. Liu, Y.-S. Zhou, F.-C. Meng, T.-Y. Qiao, X.-J. Yang, X.-Y. Li, Q. Xue, H. Xu, Y. Liu, Y. Han, and Y. Zhang. 2020. Musculoskeletal pain during the menopausal transition: A systematic review and meta-analysis. Neural Plasticity 2020:1–10.
Luo, J., C. R. Beam, I. K. Karlsson, C. J. Pike, C. A. Reynolds, and M. Gatz. 2020. Dementia risk in women higher in same-sex than opposite-sex twins. Alzheimer’s & Dementia: Diagnosis, Assessment & Disease Monitoring 12(1):e12049.
Luscher, B., Q. Shen, and N. Sahir. 2011. The GABAergic deficit hypothesis of major depressive disorder. Molecular Psychiatry 16(4):383–406.
Luu, J. M., J. Wei, C. L. Shufelt, A. Asif, B. Tjoe, P. Theriot, and C. N. Bairey Merz. 2022. Clinical practice variations in the management of ischemia with no obstructive coronary artery disease. Journal of the American Heart Association 11(19):e022573.
Lynch, D. H., C. L. Petersen, M. J. Van Dongen, H. B. Spangler, S. A. Berkowitz, and J. A. Batsis. 2022. Association between food insecurity and probable sarcopenia: Data from the 2011–2014 National Health and Nutrition Examination Survey. Clinical Nutrition 41(9):1861–1873.
Lynch, W. J., and M. E. Carroll. 1999. Sex differences in the acquisition of intravenously self-administered cocaine and heroin in rats. Psychopharmacology 144(1):77–82.
Lynch, W. J., and M. E. Carroll. 2000. Reinstatement of cocaine self-administration in rats: Sex differences. Psychopharmacology 148(2):196–200.
Lynch, W. J., M. E. Roth, J. L. Mickelberg, and M. E. Carroll. 2001. Role of estrogen in the acquisition of intravenously self-administered cocaine in female rats. Pharmacology Biochemistry and Behavior 68(4):641–646.
Lyu, Y., W. Li, and T. Tang. 2022. Prevalence trends and influencing factors of post-stroke depression: A study based on the national health and nutrition examination survey. Medical Science Monitor 28:e933367.
Ma, H., B. C. V. Campbell, M. W. Parsons, L. Churilov, C. R. Levi, C. Hsu, T. J. Kleinig, T. Wijeratne, S. Curtze, H. M. Dewey, F. Miteff, C. H. Tsai, J. T. Lee, T. G. Phan, N. Mahant, M. C. Sun, M. Krause, J. Sturm, R. Grimley, C. H. Chen, C. J. Hu, A. A. Wong, D. Field, Y. Sun, P. A. Barber, A. Sabet, J. Jannes, J. S. Jeng, B. Clissold, R. Markus, C. H. Lin, L. M. Lien, C. F. Bladin, S. Christensen, N. Yassi, G. Sharma, A. Bivard, P. M. Desmond, B. Yan, P. J. Mitchell, V. Thijs, L. Carey, A. Meretoja, S. M. Davis, and G. A. Donnan. 2019. Thrombolysis guided by perfusion imaging up to 9 hours after onset of stroke. New England Journal of Medicine 380(19):1795–1803.
MacGowan, S. H., G. K. Wilcock, and M. Scott. 1998. Effect of gender and apolipoprotein E genotype on response to anticholinesterase therapy in Alzheimer’s disease. International Journal of Geriatric Psychiatry 13(9):625–630.
MacGregor, E. A., and A. Hackshaw. 2004. Prevalence of migraine on each day of the natural menstrual cycle. Neurology 63(2):351–353.
MacKenzie-Graham, A., J. Brook, F. Kurth, Y. Itoh, C. Meyer, M. J. Montag, H. J. Wang, R. Elashoff, and R. R. Voskuhl. 2018. Estriol-mediated neuroprotection in multiple sclerosis localized by voxel-based morphometry. Brain and Behavior 8(9):e01086.
Madsen, T. E., H. Sucharew, B. Katz, K. A. Alwell, C. J. Moomaw, B. M. Kissela, M. L. Flaherty, D. Woo, P. Khatri, S. Ferioli, J. Mackey, S. Martini, F. De Los Rios La Rosa, and D. Kleindorfer. 2016. Gender and time to arrival among ischemic stroke patients in the Greater Cincinnati/Northern Kentucky stroke study. Journal of Stroke and Cerebrovascular Disease 25(3):504–510.
Madsen, T. E., D. L. Long, A. P. Carson, G. Howard, D. O. Kleindorfer, K. L. Furie, J. E. Manson, S. Liu, and V. J. Howard. 2021. Sex and race differences in the risk of ischemic stroke associated with fasting blood glucose in regards. Neurology 97(7):e684–e694.
Maher, B. H., M. Kerr, H. C. Cox, J. C. Macmillan, P. J. Brimage, T. Esposito, F. Gianfrancesco, L. M. Haupt, D. R. Nyholt, R. A. Lea, and L. R. Griffiths. 2012a. Confirmation that XQ27 and XQ28 are susceptibility loci for migraine in independent pedigrees and a case-control cohort. Neurogenetics 13(1):97–101.
Maher, B. H., R. A. Lea, M. Benton, H. C. Cox, C. Bellis, M. Carless, T. D. Dyer, J. Curran, J. C. Charlesworth, J. E. Buring, T. Kurth, D. I. Chasman, P. M. Ridker, M. Schürks, J. Blangero, and L. R. Griffiths. 2012b. An X chromosome association scan of the Norfolk Island genetic isolate provides evidence for a novel migraine susceptibility locus at XQ12. PloS One 7(5):e37903.
Maki, P. M., S. G. Kornstein, H. Joffe, J. T. Bromberger, E. W. Freeman, G. Athappilly, W. V. Bobo, L. H. Rubin, H. K. Koleva, L. S. Cohen, and C. N. Soares. 2019. Guidelines for the evaluation and treatment of perimenopausal depression: Summary and recommendations. Journal of Women’s Health 28(2):117–134.
Mäkitie, R. E., A. Costantini, A. Kämpe, J. J. Alm, and O. Mäkitie. 2019. New insights into monogenic causes of osteoporosis. Frontiers in Endocrinology 10:70.
Maleki, N., C. Linnman, J. Brawn, R. Burstein, L. Becerra, and D. Borsook. 2012. Her versus his migraine: Multiple sex differences in brain function and structure. Brain 135(8):2546–2559.
Mallard, T. T., S. Liu, J. Seidlitz, Z. Ma, D. Moraczewski, A. Thomas, and A. Raznahan. 2021. X-chromosome influences on neuroanatomical variation in humans. Nature Neuroscience 24(9):1216–1224.
Malmstrom, T. K., D. K. Miller, E. M. Simonsick, L. Ferrucci, and J. E. Morley. 2016. SARC-F: A symptom score to predict persons with sarcopenia at risk for poor functional outcomes. Journal of Cachexia, Sarcopenia and Muscle 7(1):28–36.
Mani, K. K., Y. El-Hakim, T. E. Branyan, N. Samiya, S. Pandey, M. T. Grimaldo, A. Habbal, A. Wertz, and F. Sohrabji. 2023. Intestinal epithelial stem cell transplants as a novel therapy for cerebrovascular stroke. Brain, Behavior, and Immunity 107:345–360.
Manrique-Acevedo, C., B. Chinnakotla, J. Padilla, L. A. Martinez-Lemus, and D. Gozal. 2020. Obesity and cardiovascular disease in women. International Journal of Obesity 44(6):1210–1226.
Marceau, K., G. Horvath, A. M. Loviska, and V. S. Knopik. 2021. Developmental cascades from polygenic and prenatal substance use to adolescent substance use: Leveraging severity and directionality of externalizing and internalizing problems to understand pubertal and harsh discipline-related risk. Behavior Genetics 51(5):559–579.
Marchand, F., M. Perretti, and S. B. McMahon. 2005. Role of the immune system in chronic pain. Nature Reviews Neuroscience 6(7):521–532.
Marsh, J. C., H. Amaro, Y. Kong, T. Khachikian, and E. Guerrero. 2021. Gender disparities in access and retention in outpatient methadone treatment for opioid use disorder in low-income urban communities. Journal of Substance Abuse Treatment 127:108399.
Marshall, D., O. Johnell, and H. Wedel. 1996. Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. BMJ 312(7041):1254–1259.
Martin, C. E., A. Scialli, and M. Terplan. 2020. Addiction and depression: Unmet treatment needs among reproductive age women. Maternal and Child Health Journal 24(5):660–667.
Martínez-Lavín, M. 2021. Fibromyalgia in women: Somatisation or stress-evoked, sex-dimorphic neuropathic pain? Clinical and Experimental Rheumatology 39(2):422–425.
Martinez-Lavin, M., and C. Solano. 2009. Dorsal root ganglia, sodium channels, and fibromyalgia sympathetic pain. Medical Hypotheses 72(1):64–66.
Martínez-Rodríguez, A., J. Rubio-Arias, D. J. Ramos-Campo, C. Reche-García, B. Leyva-Vela, and Y. Nadal-Nicolás. 2020. Psychological and sleep effects of tryptophan and magnesium-enriched Mediterranean diet in women with fibromyalgia. International Journal of Environmental Research and Public Health 17(7):2227.
Martini, M., J. W. Irvin, C. G. Lee, W. J. Lynch, and E. F. Rissman. 2020. Sex chromosome complement influences vulnerability to cocaine in mice. Hormones and Behavior 125:104821.
Maurer, A. J., A. Lissounov, I. Knezevic, K. D. Candido, and N. N. Knezevic. 2016. Pain and sex hormones: A review of current understanding. Pain Management 6(3):285–296.
Mauvais-Jarvis, F. 2014. Developmental androgenization programs metabolic dysfunction in adult mice: Clinical implications. Adipocyte 3(2):151–154.
Mauvais-Jarvis, F. 2015. Sex differences in metabolic homeostasis, diabetes, and obesity. Biology of Sex Differences 6(1):14.
Mazure, C. M., and D. A. Fiellin. 2018. Women and opioids: Something different is happening here. Lancet 392(10141):9–11.
McCarthy, A. M., A. Menke, and K. Visvanathan. 2013. Association of bilateral oophorectomy and body fatness in a representative sample of us women. Gynecologic Oncology 129(3):559–564.
McGovern, T. M. 1994. S.P. v. Sullivan: The effort to broaden the Social Security Administration’s definition of AIDS. Fordham Urban Law Journal 21(4):1083–1096.
McHugh, R. K., V. R. Votaw, D. E. Sugarman, and S. F. Greenfield. 2018. Sex and gender differences in substance use disorders. Clinical Psychology Review 66:12–23.
McKinley, C. E., C. R. Figley, S. M. Woodward, J. L. Liddell, S. Billiot, N. Comby, and S. Sanders. 2019. Community-engaged and culturally relevant research to develop behavioral health interventions with american indians and alaska natives. American Indian and Alaska Native Mental Health Research 26(3):79–103.
McLeod, J. D. 2014. Juvenile fibromyalgia syndrome and improved recognition by pediatric primary care providers. Journal of Pediatric Health Care 28(2):e9–18.
Society for Adolescent Health and Medicine. 2013. Recommended vitamin D intake and management of low vitamin D status in adolescents: A position statement of the Society for Adolescent Health and Medicine. Journal of Adolescent Health 52(6):801–803.
Meester, I., G. F. Rivera-Silva, and F. González-Salazar. 2020. Immune system sex differences may bridge the gap between sex and gender in fibromyalgia. Frontiers in Neuroscience 13:1414.
Mehta, L. S., T. M. Beckie, H. A. DeVon, C. L. Grines, H. M. Krumholz, M. N. Johnson, K. J. Lindley, V. Vaccarino, T. Y. Wang, K. E. Watson, and N. K. Wenger. 2016. Acute myocardial infarction in women. Circulation 133(9):916–947.
Mehta, L. S., G. P. Velarde, J. Lewey, G. Sharma, R. M. Bond, A. Navas-Acien, A. M. Fretts, G. S. Magwood, E. Yang, R. S. Blumenthal, R. M. Brown, and J. H. Mieres. 2023. Cardiovascular disease risk factors in women: The impact of race and ethnicity: A scientific statement from the American Heart Association. Circulation 147(19):1471–1487.
Mehta, N., C. Rodrigues, M. Lamba, W. Wu, S. E. Bronskill, N. Herrmann, S. S. Gill, A.-W. Chan, R. Mason, S. Day, J. H. Gurwitz, and P. A. Rochon. 2017. Systematic review of sex-specific reporting of data: Cholinesterase inhibitor example. Journal of the American Geriatrics Society 65(10):2213–2219.
Mello, N. K., J. H. Mendelson, and B. W. Lex. 1990. Alcohol use and premenstrual symptoms in social drinkers. Psychopharmacology 101(4):448–455.
Mello, N. K., J. H. Mendelson, M. Kelly, N. Diaz-Migoyo, and J. W. Sholar. 1997. The effects of chronic cocaine self-administration on the menstrual cycle in rhesus monkeys. Journal of Pharmacology and Experimental Therapeutics 281(1):70–83.
Mellon, L., L. Brewer, P. Hall, F. Horgan, D. Williams, and A. Hickey. 2015. Cognitive impairment six months after ischaemic stroke: A profile from the ASPIRE-S study. BMC Neurology 15:31.
Méndez-Eirín, E., Y. Suárez-Ouréns, and J. Rodríguez-Fernández. 2021. Spontaneous coronary artery dissection. Revista Clínica Española 221(5):297–305.
Merz, C. N. B., C. J. Pepine, M. N. Walsh, J. L. Fleg, P. G. Camici, W. M. Chilian, J. A. Clayton, L. S. Cooper, F. Crea, M. D. Carli, P. S. Douglas, Z. S. Galis, P. Gurbel, E. M. Handberg, A. Hasan, J. A. Hill, J. S. Hochman, E. Iturriaga, R. Kirby, G. N. Levine, P. Libby, J. Lima, P. Mehta, P. Desvigne-Nickens, M. Olive, G. D. Pearson, A. A. Quyyumi, H. Reynolds, B. Robinson, G. Sopko, V. Taqueti, J. Wei, and N. Wenger. 2017. Ischemia and no obstructive coronary artery disease (INOCA). Circulation 135(11):1075–1092.
Messé, S. R., P. Khatri, M. J. Reeves, E. E. Smith, J. L. Saver, D. L. Bhatt, M. V. Grau-Sepulveda, M. Cox, E. D. Peterson, G. C. Fonarow, and L. H. Schwamm. 2016. Why are acute ischemic stroke patients not receiving iv tpa? Results from a national registry. Neurology 87(15):1565–1574.
Mhlekude, B., A. Lenman, P. Sidoyi, J. Joseph, J. Kruppa, C. B. Businge, M. L. Mdaka, F. Konietschke, A. Pich, G. Gerold, C. Goffinet, and A. S. Mall. 2021. The barrier functions of crude cervical mucus plugs against HIV-1 infection in the context of cell-free and cell-to-cell transmission. AIDS 35(13):2105–2117.
Miar, A., V. Alvarez, A. I. Corao, B. Alonso, M. Díaz, M. Menéndez, C. Martínez, M. Calatayud, G. Morís, and E. Coto. 2011. Lack of association between protocadherin 11–X/Y (PCDH11X and PCDH11Y) polymorphisms and late onset Alzheimer’s disease. Brain Research 1383:252–256.
Midavaine, É., J. Côté, S. Marchand, and P. Sarret. 2021. Glial and neuroimmune cell choreography in sexually dimorphic pain signaling. Neuroscience and Biobehavior Reviews 125:168–192.
Mielke, M. M. 2020. Consideration of sex differences in the measurement and interpretation of Alzheimer disease-related biofluid-based biomarkers. Journal of Applied Laboratory Medicine 5(1):158–169.
Mielke, M. M., N. T. Aggarwal, C. Vila-Castelar, P. Agarwal, E. M. Arenaza-Urquijo, B. Brett, A. Brugulat-Serrat, L. E. DuBose, W. S. Eikelboom, J. Flatt, N. S. Foldi, S. Franzen, P. Gilsanz, W. Li, A. J. McManus, D. M. van Lent, S. A. Milani, C. E. Shaaban, S. D. Stites, E. Sundermann, V. Suryadevara, J. F. Trani, A. D. Turner, J. M. J. Vonk, Y. T. Quiroz, and G. M. Babulal. 2022a. Consideration of sex and gender in Alzheimer’s disease and related disorders from a global perspective. Alzheimers & Dementia 18(12):2707–2724.
Mielke, M. M., J. L. Dage, R. D. Frank, A. Algeciras-Schimnich, D. S. Knopman, V. J. Lowe, G. Bu, P. Vemuri, J. Graff-Radford, C. R. Jack, Jr., and R. C. Petersen. 2022b. Performance of plasma phosphorylated tau 181 and 217 in the community. Nature Medicine 28(7):1398–1405.
Migliore, L., and F. Coppedè. 2022. Gene–environment interactions in Alzheimer disease: The emerging role of epigenetics. Nature Reviews Neurology 18(11):643–660.
Milà-Alomà, M., N. J. Ashton, M. Shekari, G. Salvadó, P. Ortiz-Romero, L. Montoliu-Gaya, A. L. Benedet, T. K. Karikari, J. Lantero-Rodriguez, E. Vanmechelen, T. A. Day, A. González-Escalante, G. Sánchez-Benavides, C. Minguillon, K. Fauria, J. L. Molinuevo, J. L. Dage, H. Zetterberg, J. D. Gispert, M. Suárez-Calvet, and K. Blennow. 2022. Plasma P-tau231 and P-tau217 as state markers of amyloid-β pathology in preclinical Alzheimer’s disease. Nature Medicine 28(9):1797–1801.
Miller, E. C., N. A. Bello, R. Davis, A. M. Friedman, M. S. V. Elkind, R. Wapner, and S. E. Tom. 2022. Women with adverse pregnancy outcomes have higher odds of midlife stroke: The Population Assessment of Tobacco and Health study. Journal of Women’s Health 31(4):503–512.
Miller, E. C., A. Kauko, S. E. Tom, H. Laivuori, T. Niiranen, and N. A. Bello. 2023. Risk of midlife stroke after adverse pregnancy outcomes: The FinnGen study. Stroke 54(7): 1798–1805.
Miller, F. D., G. Ozimek, R. J. Milner, and F. E. Bloom. 1989. Regulation of neuronal oxytocin mRNA by ovarian steroids in the mature and developing hypothalamus. Proceedings of the National Academy of Sciences of the United States of America 86(7):2468–2472.
Miller, K., T. Frech, T. Greene, K. N. Ma, M. McFadden, L. Tom-Orme, M. L. Slattery, and M. A. Murtaugh. 2016. Bone mineral density in Navajo men and women and comparison to non-Hispanic Whites from NHANES (2005–2008). Journal of Health Care for the Poor and Underserved 27(2):644–662.
Mills, E. G., L. Yang, M. F. Nielsen, M. Kassem, W. S. Dhillo, and A. N. Comninos. 2021. The relationship between bone and reproductive hormones beyond estrogens and androgens. Endocrine Reviews 42(6):691–719.
Mills, S. E. E., K. P. Nicolson, and B. H. Smith. 2019. Chronic pain: A review of its epidemiology and associated factors in population-based studies. British Journal of Anaesthesia 123(2):e273–e283.
Min, L. Y., R. B. Islam, N. Gandrakota, and M. K. Shah. 2022. The social determinants of health associated with cardiometabolic diseases among Asian American subgroups: A systematic review. BMC Health Services Research 22(1):257.
Min, M. O., S. Minnes, A. Lang, P. Weishampel, E. J. Short, S. Yoon, and L. T. Singer. 2014. Externalizing behavior and substance use related problems at 15 years in prenatally cocaine exposed adolescents. Journal of Adolescence 37(3):269–279.
Min, M. O., S. Minnes, S. K. Kim, J. Y. Kim, and L. T. Singer. 2023. Prenatal cocaine exposure and substance use disorder in emerging adulthood at age 21. Drug and Alcohol Dependence 242:109736.
Minen, M. T., A. Boubour, A. Wahnich, C. Grudzen, and B. W. Friedman. 2018. A retrospective nested cohort study of emergency department revisits for migraine in New York City. Headache 58(3):399–406.
Mishra, G. D., M. C. Davies, S. Hillman, H.-F. Chung, S. Roy, K. Maclaran, and M. Hickey. 2024a. Optimising health after early menopause. The Lancet 403(10430):958–968.
Mishra, S., H. Elliott, and R. Marwaha. 2024b. Premenstrual dysphoric disorder. In Statpearls. Treasure Island, FL: StatPearls Publishing LLC.
Mishra, S. R., H. F. Chung, M. Waller, A. J. Dobson, D. C. Greenwood, J. E. Cade, G. G. Giles, F. Bruinsma, M. K. Simonsen, R. Hardy, D. Kuh, E. B. Gold, S. L. Crawford, C. A. Derby, K. A. Matthews, P. Demakakos, J. S. Lee, H. Mizunuma, K. Hayashi, L. L. Sievert, D. E. Brown, S. Sandin, E. Weiderpass, and G. D. Mishra. 2020. Association between reproductive life span and incident nonfatal cardiovascular disease: A pooled analysis of individual patient data from 12 studies. JAMA Cardiology 5(12):1410–1418.
Misra, S., V. Jackson, J. Chong, K. Choe, C. Tay, J. Wong, and L. Yang. 2021. Systematic review of cultural aspects of stigma and mental illness among racial and ethnic minority groups in the United States: Implications for interventions. American Journal of Community Psychology 68.
Miyake, N., S. Mizuno, N. Okamoto, H. Ohashi, M. Shiina, K. Ogata, Y. Tsurusaki, M. Nakashima, H. Saitsu, N. Niikawa, and N. Matsumoto. 2013. KDM6A point mutations cause Kabuki syndrome. Human Mutation 34(1):108–110.
Moagi, M. M., A. E. van Der Wath, P. M. Jiyane, and R. S. Rikhotso. 2021. Mental health challenges of lesbian, gay, bisexual and transgender people: An integrated literature review. Health SA 26:1487.
Mogil, J. S. 2012. Sex differences in pain and pain inhibition: Multiple explanations of a controversial phenomenon. Nature Reviews Neuroscience 13(12):859–866.
Mogil, J. S. 2020. Qualitative sex differences in pain processing: Emerging evidence of a biased literature. Nature Reviews Neuroscience 21(7):353–365.
Molfino, A., G. Imbimbo, and M. Muscaritoli. 2021. Endocrinological and nutritional implications of anorexia of aging. Endocrines 2(4):439–448.
Moore, C., E. Coates, A. Watson, R. de Heer, A. McLeod, and A. Prudhomme. 2023. “It’s important to work with people that look like me”: Black patients’ preferences for patient-provider race concordance. Journal of Racial and Ethnic Health Disparities 10(5):2552–2564.
Moran, J. A., S. R. Turner, and M. D. Marsden. 2022. Contribution of sex differences to HIV immunology, pathogenesis, and cure approaches. Frontiers in Immunology 13.
Moreau, K. L., and K. L. Hildreth. 2014. Vascular aging across the menopause transition in healthy women. Advanced Vascular Medicine 204390.
Morgan, C. P., J. C. Chan, and T. L. Bale. 2019. Driving the next generation: Paternal lifetime experiences transmitted via extracellular vesicles and their small RNA cargo. Biological Psychiatry 85(2):164–171.
Morley, J. E. 2011. Frailty: Diagnosis and management. The Journal of Nutrition, Health & Aging 15(8):667–670.
Morrison, K. E., A. B. Cole, P. J. Kane, V. E. Meadows, S. M. Thompson, and T. L. Bale. 2020. Pubertal adversity alters chromatin dynamics and stress circuitry in the pregnant brain. Neuropsychopharmacology 45(8):1263–1271.
Mortensen, S. J., I. Beeram, J. Florance, K. Momenzadeh, A. Mohamadi, E. K. Rodriguez, A. von Keudell, and A. Nazarian. 2021. Modifiable lifestyle factors associated with fragility hip fracture: A systematic review and meta-analysis. Journal of Bone and Mineral Metabolism 39(5):893-902.
Mosca, L., S. M. Grundy, D. Judelson, K. King, M. Limacher, S. Oparil, R. Pasternak, T. A. Pearson, R. F. Redberg, S. C. Smith, M. Winston, and S. Zinberg. 1999. Guide to preventive cardiology for women. Circulation 99(18):2480–2484.
Mosca, L., E. J. Benjamin, K. Berra, J. L. Bezanson, R. J. Dolor, D. M. Lloyd-Jones, L. K. Newby, I. L. Piña, V. L. Roger, L. J. Shaw, D. Zhao, T. M. Beckie, C. Bushnell, J. D’Armiento, P. M. Kris-Etherton, J. Fang, T. G. Ganiats, A. S. Gomes, C. R. Gracia, C. K. Haan, E. A. Jackson, D. R. Judelson, E. Kelepouris, C. J. Lavie, A. Moore, N. A. Nussmeier, E. Ofili, S. Oparil, P. Ouyang, V. W. Pinn, K. Sherif, S. C. Smith, G. Sopko, N. Chandra-Strobos, E. M. Urbina, V. Vaccarino, and N. K. Wenger. 2011. Effectiveness-based guidelines for the prevention of cardiovascular disease in women—2011 update. Circulation 123(11):1243–1262.
Mosconi, L., V. Berti, J. Dyke, E. Schelbaum, S. Jett, L. Loughlin, G. Jang, A. Rahman, H. Hristov, S. Pahlajani, R. Andrews, D. Matthews, O. Etingin, C. Ganzer, M. De Leon, R. Isaacson, and R. D. Brinton. 2021. Menopause impacts human brain structure, connectivity, energy metabolism, and amyloid-beta deposition. Scientific Reports 11(1).
Mullard, A. 2024. CAR-T therapy for multiple sclerosis enters U.S. trials for first time. Nature News. https://www.nature.com/articles/d41586-024-00470-5 (accessed June 18, 2024).
Müller, S., and E. Kuntsche. 2011. Do the drinking motives of adolescents mediate the link between their parents’ drinking habits and their own alcohol use? Journal of Studies on Alcohol and Drugs 72(3):429–437.
Murata, T., K. Narita, and T. Ichimaru. 2014. Rat uterine oxytocin receptor and estrogen receptor α and β mRNA levels are regulated by estrogen through multiple estrogen receptors. Journal of Reproductive Development 60(1):55–61.
Murphy, B. E., F. V. Abbott, C. M. Allison, C. Watts, and A. M. Ghadirian. 2004. Elevated levels of some neuroactive progesterone metabolites, particularly isopregnanolone, in women with chronic fatigue syndrome. Psychoneuroendocrinology 29(2):245–268.
Nadal-Nicolás, Y., J. Rubio-Arias, M. Martínez-Olcina, C. Reche-García, M. Hernández-García, and A. Martínez-Rodríguez. 2020. Effects of manual therapy on fatigue, pain, and psychological aspects in women with fibromyalgia. International Journal of Environmental Research and Public Health 17(12):4611.
Nagata, J. M., K. T. Ganson, J. Tabler, A. J. Blashill, and S. B. Murray. 2020. Disparities across sexual orientation in migraine among U.S. adults. JAMA Neurology 78(1):117–118.
Nappi, R. E., L. Tiranini, S. Sacco, E. De Matteis, R. De Icco, and C. Tassorelli. 2022. Role of estrogens in menstrual migraine. Cells 11(8):1355.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2021. Sexually Transmitted Infections: Adopting a Sexual Health Paradigm. Edited by S. H. Vermund, A. B. Geller, and J. S. Crowley. Washington, DC: The National Academies Press.
NASEM. 2022. Enhancing NIH Research on Autoimmune Disease. Washington, DC: The National Academies Press.
Negro, A., Z. Delaruelle, T. A. Ivanova, S. Khan, R. Ornello, B. Raffaelli, A. Terrin, U. Reuter, and D. D. Mitsikostas. 2017. Headache and pregnancy: A systematic review. Journal of Headache and Pain 18(1):106.
Nelson, S., L. E. Simons, and D. Logan. 2018. The incidence of adverse childhood experiences (ACEs) and their association with pain-related and psychosocial impairment in youth with chronic pain. Clinical Journal of Pain 34(5):402–408.
Nelson, S. M., N. R. Cunningham, and S. Kashikar-Zuck. 2017. A conceptual framework for understanding the role of adverse childhood experiences in pediatric chronic pain. The Clinical Journal of Pain 33(3):264–270.
Nemmani, K. V. S., J. E. Grisel, J. R. Stowe, R. Smith-Carliss, and J. S. Mogil. 2004. Modulation of morphine analgesia by site-specific n-methyl-d-aspartate receptor antagonists: Dependence on sex, site of antagonism, morphine dose, and time. Pain 109(3):274–283.
Neu, S. C., J. Pa, W. Kukull, D. Beekly, A. Kuzma, P. Gangadharan, L. S. Wang, K. Romero, S. P. Arneric, A. Redolfi, D. Orlandi, G. B. Frisoni, R. Au, S. Devine, S. Auerbach, A. Espinosa, M. Boada, A. Ruiz, S. C. Johnson, R. Koscik, J. J. Wang, W. C. Hsu, Y. L. Chen, and A. W. Toga. 2017. Apolipoprotein E genotype and sex risk factors for Alzheimer disease: A meta-analysis. JAMA Neurology 74(10):1178–1189.
Neugebauer, J., J. Heilig, S. Hosseinibarkooie, B. C. Ross, N. Mendoza-Ferreira, F. Nolte, M. Peters, I. Hölker, K. Hupperich, T. Tschanz, V. Grysko, F. Zaucke, A. Niehoff, and B. Wirth. 2018. Plastin 3 influences bone homeostasis through regulation of osteoclast activity. Human Molecular Genetics 27(24): 4249–4262.
Nicolas, C., T. I. Russell, A. F. Pierce, S. Maldera, A. Holley, Z.-B. You, M. M. McCarthy, Y. Shaham, and S. Ikemoto. 2019. Incubation of cocaine craving after intermittent-access self-administration: Sex differences and estrous cycle. Biological Psychiatry 85(11): 915–924.
NIDA (National Institute on Drug Abuse). 2020. Substance Use in Women Research Report: Summary. https://nida.nih.gov/publications/research-reports/substance-use-in-women/summary (accessed January 3, 2024).
NIDA. 2023. Adolescent Brain Cognitive Development Study (ABCD Study). https://nida.nih.gov/research-topics/adolescent-brain/longitudinal-study-adolescent-brain-cognitive-development-abcd-study (accessed February 23, 2024).
Niemela, L., G. Lamoury, S. Carroll, M. Morgia, A. Yeung, and B. Oh. 2024. Exploring gender differences in the relationship between gut microbiome and depression—A scoping review. Frontiers in Psychiatry 15:1361145.
NIH (National Institutes of Health). Calcium: Fact Sheet for Health Professionals. https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional/ (accessed March 14, 2024).
Ning, H. T., Y. Du, L. J. Zhao, Q. Tian, H. Feng, and H. W. Deng. 2021. Racial and gender differences in the relationship between sarcopenia and bone mineral density among older adults. Osteoporosis International 32(5):841–851.
Noble, R. E. 2005. Depression in women. Metabolism: Clinical and Experimental 54(5 Suppl 1): 49–52.
Noel, M., C. B. Groenewald, S. E. Beals-Erickson, J. T. Gebert, and T. M. Palermo. 2016. Chronic pain in adolescence and internalizing mental health disorders. Pain 157(6):1333–1338.
Nordberg, A. 2015. Towards early diagnosis in Alzheimer disease. Nature Reviews Neurology 11(2):69–70.
North American Menopause Society. 2022. The 2022 hormone therapy position statement of the North American Menopause Society. Menopause 29(7):767–794.
Nuutila, P., M. J. Knuuti, M. Mäki, H. Laine, U. Ruotsalainen, M. Teräs, M. Haaparanta, O. Solin, and H. Yki-Järvinen. 1995. Gender and insulin sensitivity in the heart and in skeletal muscles. Studies using positron emission tomography. Diabetes 44(1):31–36.
Nwangwu-Ike, N., M. Kapaya, K. Oladapo, and D. V. DAngelo. 2023. HIV testing during pregnancy among women with a recent live birth—seven U.S. states, 2016–2019. Women’s Health 19:17455057231199571.
Ocay, D. D., C. L. Larche, N. Betinjane, A. Jolicoeur, M. J. Beaulieu, N. Saran, J. A. Ouellet, P. M. Ingelmo, and C. E. Ferland. 2022. Phenotyping chronic musculoskeletal pain in male and female adolescents: Psychosocial profiles, somatosensory profiles and pain modulatory profiles. Journal of Pain Research 15:591–612.
Oddy, W. H., K. L. Allen, G. S. A. Trapp, G. L. Ambrosini, L. J. Black, R. C. Huang, P. Rzehak, K. C. Runions, F. Pan, L. J. Beilin, and T. A. Mori. 2018. Dietary patterns, body mass index and inflammation: Pathways to depression and mental health problems in adolescents. Brain, Behavior, and Immunity 69:428–439.
Offenbaecher, M., B. Bondy, S. D. Jonge, K. Glatzeder, M. Krüger, P. Schoeps, and M. Ackenheil. 1999. Possible association of fibromyalgia with a polymorphism in the serotonin transporter gene regulatory region. Arthritis and Rheumatism 42(11):2482–2488.
Okada, T., H. Suzuki, Z. D. Travis, and J. H. Zhang. 2020. The stroke-induced blood-brain barrier disruption: Current progress of inspection technique, mechanism, and therapeutic target. Current Neuropharmacology 18(12):1187–1212.
O’Kelly, A. C., E. D. Michos, C. L. Shufelt, J. V. Vermunt, M. B. Minissian, O. Quesada, G. N. Smith, J. W. Rich-Edwards, V. D. Garovic, S. R. El Khoudary, and M. C. Honigberg. 2022. Pregnancy and reproductive risk factors for cardiovascular disease in women. Circulation Research 130(4):652–672.
Oliver-Williams, C., C. J. Vladutiu, L. R. Loehr, W. D. Rosamond, and A. M. Stuebe. 2019. The association between parity and subsequent cardiovascular disease in women: The Atherosclerosis Risk in Communities Study. Journal of Women’s Health 28(5):721–727.
Olsson, T., L. F. Barcellos, and L. Alfredsson. 2017. Interactions between genetic, lifestyle and environmental risk factors for multiple sclerosis. Nature Reviews: Neurology 13(1):25–36.
Ooi, H., and C. Welch. 2024. Obstacles to the early diagnosis and management of sarcopenia: Current perspectives. Clinical Interventions in Aging 19:323–332.
Orsini, C. A., T. E. Brown, T. E. Hodges, Y. Alonso-Caraballo, C. A. Winstanley, and J. B. Becker. 2022. Neural mechanisms mediating sex differences in motivation for reward: Cognitive bias, food, gambling, and drugs of abuse. The Journal of Neuroscience 42(45):8477–8487.
Ortiz, L. A., A. Quan, A. Weinberg, and M. Baum. 2001. Effect of prenatal dexamethasone on rat renal development. Kidney International 59(5):1663–1669.
Osborne, N. R., and K. D. Davis. 2022. Sex and gender differences in pain. In International Review of Neurobiology, Vol. 164, edited by E. Moro, G. Arabia, M. C. Tartaglia, and M. T. Ferretti. Academic Press. Pp. 277–307.
Ossenkoppele, R., R. Smith, N. Mattsson-Carlgren, C. Groot, A. Leuzy, O. Strandberg, S. Palmqvist, T. Olsson, J. Jögi, E. Stormrud, H. Cho, Y. H. Ryu, J. Y. Choi, A. L. Boxer, M. L. Gorno-Tempini, B. L. Miller, D. Soleimani-Meigooni, L. Iaccarino, R. La Joie, S. Baker, E. Borroni, G. Klein, M. J. Pontecorvo, M. D. Devous, Sr, W. J. Jagust, C. H. Lyoo, G. D. Rabinovici, and O. Hansson. 2021. Accuracy of tau positron emission tomography as a prognostic marker in preclinical and prodromal Alzheimer disease: A head-to-head comparison against amyloid positron emission tomography and magnetic resonance imaging. JAMA Neurology 78(8):961–971.
Ossenkoppele, R., A. Pichet Binette, C. Groot, R. Smith, O. Strandberg, S. Palmqvist, E. Stomrud, P. Tideman, T. Ohlsson, J. Jögi, K. Johnson, R. Sperling, V. Dore, C. L. Masters, C. Rowe, D. Visser, B. N. M. van Berckel, W. M. van der Flier, S. Baker, W. J. Jagust, H. J. Wiste, R. C. Petersen, C. R. Jack, and O. Hansson. 2022. Amyloid and tau PET-positive cognitively unimpaired individuals are at high risk for future cognitive decline. Nature Medicine 28(11):2381–2387.
OTA (Office of Technology Assessment). 1992. The CDC’s case definition of AIDS: Implications of Proposed Revisions. Washington, DC.: U.S. Government Publishing Office.
Otite, F. O., V. Saini, N. B. Sur, S. Patel, R. Sharma, E. O. Akano, N. Anikpezie, K. Albright, E. Schmidt, H. Hoffman, G. Gould, P. Khandelwal, J. G. Latorre, A. M. Malik, R. L. Sacco, and S. Chaturvedi. 2021. Ten-year trend in age, sex, and racial disparity in TPA (alteplase) and thrombectomy use following stroke in the United States. Stroke 52(8):2562–2570.
Ott, B. R., C. A. Tate, N. M. Gordon, and W. C. Heindel. 1996. Gender differences in the behavioral manifestations of Alzheimer’s disease. Journal of the American Geriatrics Society 44(5):583–587.
Overstreet, D. S., B. D. Pester, J. M. Wilson, K. M. Flowers, N. K. Kline, and S. M. Meints. 2023. The experience of BIPOC living with chronic pain in the USA: Biopsychosocial factors that underlie racial disparities in pain outcomes, comorbidities, inequities, and barriers to treatment. Current Pain and Headache Reports 27(1):1–10.
Pacheco, C., J. Luu, P. K. Mehta, J. Wei, M. Gulati, and C. N. Bairey Merz. 2022a. INOCA and MINOCA: Are women’s heart centres the answer to understanding and management of these increasing populations of women (and men)? Canadian Journal of Cardiology 38(10):1611–1614.
Pacheco, C., K.-A. Mullen, T. Coutinho, S. Jaffer, M. Parry, H. G. C. Van Spall, M.-A. Clavel, J. D. Edwards, T. Sedlak, C. M. Norris, A. Dhukai, J. Grewal, and S. L. Mulvagh. 2022b. The Canadian Women’s Heart Health Alliance atlas on the epidemiology, diagnosis, and management of cardiovascular disease in women—Chapter 5: Sex- and gender-unique manifestations of cardiovascular disease. CJC Open 4(3):243–262.
Pacheco Claudio, C., O. Quesada, C. J. Pepine, and C. Noel Bairey Merz. 2018. Why names matter for women: MINOCA/INOCA (myocardial infarction/ischemia and no obstructive coronary artery disease). Clinical Cardiology 41(2):185–193.
Palacios, N., S. Molsberry, K. C. Fitzgerald, and A. L. Komaroff. 2023. Different risk factors distinguish myalgic encephalomyelitis/chronic fatigue syndrome from severe fatigue. Scientific Reports 13(1):2469.
Palaszynski, K. M., K. K. Loo, J. F. Ashouri, H. B. Liu, and R. R. Voskuhl. 2004. Androgens are protective in experimental autoimmune encephalomyelitis: Implications for multiple sclerosis. Journal of Neuroimmunology 146(1–2):144–152.
Pallier, P. N., M. Ferrara, F. Romagnolo, M. T. Ferretti, H. Soreq, and A. Cerase. 2022. Chromosomal and environmental contributions to sex differences in the vulnerability to neurological and neuropsychiatric disorders: Implications for therapeutic interventions. Progress in Neurobiology 219:102353.
Palmer, B. F., and D. J. Clegg. 2015. The sexual dimorphism of obesity. Molecular and Cellular Endocrinology 402:113–119.
Palta, P., B. Rippon, M. Tahmi, M. Pardo, A. Johnson, Z. Tomljanovic, H. He, K. K. Laing, Q. R. Razlighi, J. A. Teresi, H. Moreno, A. M. Brickman, W. C. Kreisl, and J. A. Luchsinger. 2021. Sex differences in in vivo tau neuropathology in a multiethnic sample of late middle-aged adults. Neurobiology of Aging 103:109–116.
Pankratz, V. S., R. O. Roberts, M. M. Mielke, D. S. Knopman, C. R. Jack, Jr., Y. E. Geda, W. A. Rocca, and R. C. Petersen. 2015. Predicting the risk of mild cognitive impairment in the Mayo Clinic Study of Aging. Neurology 84(14):1433–1442.
Pannia, E., C. E. Cho, R. Kubant, D. Sánchez-Hernández, P. S. P. Huot, and G. Harvey Anderson. 2016. Role of maternal vitamins in programming health and chronic disease. Nutrition Reviews 74(3):166–180.
Panta, A., K. Montgomery, M. Nicolas, K. K. Mani, D. Sampath, and F. Sohrabji. 2020. MIR363–3p treatment attenuates long-term cognitive deficits precipitated by an ischemic stroke in middle-aged female rats. Frontiers in Aging Neuroscience 12:586362.
Parry, B. L. 2010. Optimal management of perimenopausal depression. International Journal of Women’s Health 2:143–151.
Parvand, M., L. Cai, S. Ghadiri, K. H. Humphries, A. Starovoytov, P. Daniele, N. Prodan-Bhalla, and T. L. Sedlak. 2022. One-year prospective follow-up of women with INOCA and MINOCA at a Canadian Women’s Heart Centre. Canadian Journal of Cardiology 38(10):1600–1610.
Pasupathy, S., T. Air, R. P. Dreyer, R. Tavella, and J. F. Beltrame. 2015. Systematic review of patients presenting with suspected myocardial infarction and nonobstructive coronary arteries. Circulation 131(10):861–870.
Patil, P. A., M. V. Porche, N. A. Shippen, N. T. Dallenbach, and L. R. Fortuna. 2018. Which girls, which boys? The intersectional risk for depression by race and ethnicity, and gender in the U.S. Clinical Psychology Review 66:51–68.
Patsopoulos, N. A., S. E. Baranzini, A. Santaniello, P. Shoostari, C. Cotsapas, G. Wong, A. H. Beecham, T. James, J. Replogle, I. S. Vlachos, C. McCabe, T. H. Pers, A. Brandes, C. White,
B. Keenan, M. Cimpean, P. Winn, I.-P. Panteliadis, A. Robbins, T. F. M. Andlauer, O. Zarzycki, B. Dubois, A. Goris, H. B. Søndergaard, F. Sellebjerg, P. S. Sorensen, H. Ullum, L. W. Thørner, J. Saarela, I. Cournu-Rebeix, V. Damotte, B. Fontaine, L. Guillot-Noel, M. Lathrop, S. Vukusic, A. Berthele, V. Pongratz, D. Buck, C. Gasperi, C. Graetz, V. Grummel, B. Hemmer, M. Hoshi, B. Knier, T. Korn, C. M. Lill, F. Luessi, M. Mühlau, F. Zipp, E. Dardiotis, C. Agliardi, A. Amoroso, N. Barizzone, M. D. Benedetti, L. Bernardinelli, P. Cavalla, F. Clarelli, G. Comi, D. Cusi, F. Esposito, L. Ferrè, D. Galimberti, C. Guaschino, M. A. Leone, V. Martinelli, L. Moiola, M. Salvetti, M. Sorosina, D. Vecchio, A. Zauli, S. Santoro, N. Mancini, M. Zuccalà, J. Mescheriakova, C. Van Duijn, S. D. Bos, E. G. Celius, A. Spurkland, M. Comabella, X. Montalban, L. Alfredsson, I. L. Bomfim, D. Gomez-Cabrero, J. Hillert, M. Jagodic, M. Lindén, F. Piehl, I. Jelčić, R. Martin, M. Sospedra, A. Baker, M. Ban, C. Hawkins, P. Hysi, S. Kalra, F. Karpe, J. Khadake, G. Lachance, P. Molyneux, M. Neville, J. Thorpe, E. Bradshaw, S. J. Caillier, P. Calabresi, B. A. C. Cree, A. Cross, M. Davis, P. W. I. De Bakker, S. Delgado, M. Dembele, K. Edwards, K. Fitzgerald, I. Y. Frohlich, P.-A. Gourraud, J. L. Haines, H. Hakonarson, D. Kimbrough, N. Isobe, I. Konidari, E. Lathi, M. H. Lee, T. Li, D. An, A. Zimmer, L. Madireddy, C. P. Manrique, M. Mitrovic, M. Olah, E. Patrick, M. A. Pericak-Vance, L. Piccio, C. Schaefer, H. Weiner, K. Lage, A. Compston, D. Hafler, H. F. Harbo, S. L. Hauser, G. Stewart, S. D’Alfonso, G. Hadjigeorgiou, B. Taylor, L. F. Barcellos, D. Booth, R. Hintzen, I. Kockum, F. Martinelli-Boneschi, J. L. McCauley, J. R. Oksenberg, A. Oturai, S. Sawcer, A. J. Ivinson, T. Olsson, and P. L. De Jager. 2019. Multiple sclerosis genomic map implicates peripheral immune cells and microglia in susceptibility. Science 365(6460):eaav7188.
Paul, B. D., M. D. Lemle, A. L. Komaroff, and S. H. Snyder. 2021. Redox imbalance links COVID-19 and myalgic encephalomyelitis/chronic fatigue syndrome. Proceedings of the National Academy of Sciences of the United States of America 118(34):e2024358118.
Pavlović, J. M. 2021. Headache in women. Continuum 27(3):686–702.
Pavlovic, J. M., D. Akcali, H. Bolay, C. Bernstein, and N. Maleki. 2017. Sex-related influences in migraine. Journal of Neuroscience Research 95(1–2):587–593.
Pavlović, J. M., and C. A. Derby. 2022. Pain in midlife women: A growing problem in need of further research. Women’s Midlife Health 8(1).
Pendlebury, S. T., and P. M. Rothwell. 2009. Prevalence, incidence, and factors associated with pre-stroke and post-stroke dementia: A systematic review and meta-analysis. Lancet Neurology 8(11):1006–1018.
Perez, M., R. Jaundoo, K. Hilton, A. Del Alamo, K. Gemayel, N. G. Klimas, T. J. A. Craddock, and L. Nathanson. 2019. Genetic predisposition for immune system, hormone, and metabolic dysfunction in myalgic encephalomyelitis/chronic fatigue syndrome: A pilot study. Frontiers in Pediatrics 7:206.
Perry, A. N., C. Westenbroek, and J. B. Becker. 2013. Impact of pubertal and adult estradiol treatments on cocaine self-administration. Hormones and Behavior 64(4):573–578.
Perseghin, G., P. Scifo, E. Pagliato, A. Battezzati, S. Benedini, L. Soldini, G. Testolin, A. Del Maschio, and L. Luzi. 2001. Gender factors affect fatty acids–induced insulin resistance in nonobese humans: Effects of oral steroidal contraception. Journal of Clinical Endocrinology and Metabolism 86(7):3188–3196.
Petanceska, S. S., V. Nagy, D. Frail, and S. Gandy. 2000. Ovariectomy and 17beta-estradiol modulate the levels of Alzheimer’s amyloid beta peptides in brain. Experimental Gerontology 35(9–10):1317–1325.
Petermann, F., and T. Korn. 2011. Cytokines and effector T cell subsets causing autoimmune CNS disease. FEBS Letters 585(23):3747–3757.
Peters, S. A., R. R. Huxley, and M. Woodward. 2014. Diabetes as a risk factor for stroke in women compared with men: A systematic review and meta-analysis of 64 cohorts, including 775,385 individuals and 12,539 strokes. Lancet 383(9933):1973–1980.
Peters, S. A. E., C. Carcel, E. R. C. Millett, and M. Woodward. 2020. Sex differences in the association between major risk factors and the risk of stroke in the U.K. Biobank Cohort Study. Neurology 95(20):e2715–e2726.
Petersen, R. C., P. S. Aisen, L. A. Beckett, M. C. Donohue, A. C. Gamst, D. J. Harvey, C. R. Jack, W. J. Jagust, L. M. Shaw, A. W. Toga, J. Q. Trojanowski, and M. W. Weiner. 2010. Alzheimer’s Disease Neuroimaging Initiative (ADNI). Neurology 74(3):201–209.
Phillips, W. J., O. Ostrovsky, R. L. Galli, and S. Dickey. 2006. Relief of acute migraine headache with intravenous oxytocin: Report of two cases. Journal of Pain & Palliative Care Pharmacotherapy 20(3):25–28.
Phimphasone-Brady, P., C. E. Page, D. A. Ali, H. C. Haller, and K. A. Duffy. 2023. Racial and ethnic disparities in women’s mental health: A narrative synthesis of systematic reviews and meta-analyses of the U.S.-based samples. Fertility and Sterility 119(3):364–374.
Phung, T. K., B. L. Waltoft, T. M. Laursen, A. Settnes, L. V. Kessing, P. B. Mortensen, and G. Waldemar. 2010. Hysterectomy, oophorectomy and risk of dementia: A nationwide historical cohort study. Dementia and Geriatric Cognitive Disorders 30(1):43–50.
Picca, A., R. Calvani, H. J. Coelho-Júnior, F. Landi, and E. Marzetti. 2022. Anorexia of aging: Metabolic changes and biomarker discovery. Clinical Interventions in Aging Volume 17:1761–1767.
Piccinelli, M., and G. Wilkinson. 2000. Gender differences in depression. British Journal of Psychiatry 177(6):486–492.
Pietschmann, P., D. Mechtcheriakova, A. Meshcheryakova, U. Föger-Samwald, and I. Ellinger. 2016. Immunology of osteoporosis: A mini-review. Gerontology 62(2):128–137.
Pike, C. J. 2017. Sex and the development of Alzheimer’s disease. Journal of Neuroscience Research 95(1–2):671–680.
Pinedo, M., S. Zemore, J. Beltrán-Girón, P. Gilbert, and Y. Castro. 2020. Women’s barriers to specialty substance abuse treatment: A qualitative exploration of racial/ethnic differences. Journal of Immigrant and Minority Health 22(4):653–660.
Pinheiro, M. B., J. Oliveira, A. Bauman, N. Fairhall, W. Kwok, and C. Sherrington. 2020. Evidence on physical activity and osteoporosis prevention for people aged 65+ years: A systematic review to inform the who guidelines on physical activity and sedentary behaviour. The International Journal of Behavioral Nutrition and Physical Activity 17(1):150.
Pinkerman, B., and K. Holroyd. 2010. Menstrual and nonmenstrual migraines differ in women with menstrually-related migraine. Cephalalgia 30(10):1187–1194.
Pinnamaneni, M., L. Payne, J. Jackson, C.-I. Cheng, and M. A. Cascio. 2022. Disparities in chronic physical health conditions in sexual and gender minority people using the United States Behavioral Risk Factor Surveillance System. Preventive Medicine Reports 28:101881.
Piroli, A., A. Mattei, G. Carta, A. D’Alfonso, P. Palermo, F. Marinangeli, B. Alfonsi, A. Testa, C. Pasquale, G. Varrassi, and A. Paladini. 2019. Influence of the menstrual cycle phase on pain perception and analgesic requirements in young women undergoing gynecological laparoscopy. Pain Practice 19(2):140–148.
Pisanu, C., F. Franconi, G. L. Gessa, S. Mameli, G. M. Pisanu, I. Campesi, L. Leggio, and R. Agabio. 2019. Sex differences in the response to opioids for pain relief: A systematic review and meta-analysis. Pharmacological Research 148:104447.
Pittock, R. R., J. A. Aakre, A. M. Castillo, V. K. Ramanan, W. K. Kremers, J. Clifford R. Jack, P. Vemuri, V. J. Lowe, D. S. Knopman, R. C. Petersen, J. Graff-Radford, and M. Vassilaki. 2023. Eligibility for anti-amyloid treatment in a population-based study of cognitive aging. Neurology 101(19):e1837–e1849.
Pitts, S. 2022. Hormonal contraception’s effect on adolescent bone health. Contemporary OB/GYN 67(10).
Pollack, B., E. Von Saltza, L. McCorkell, L. Santos, A. Hultman, A. K. Cohen, and L. Soares. 2023. Female reproductive health impacts of long COVID and associated illnesses including ME/CFS, POTS, and connective tissue disorders: A literature review. Frontiers in Rehabilitation Sciences 4:1122673.
Pool, G. J., A. F. Schwegler, B. R. Theodore, and P. N. Fuchs. 2007. Role of gender norms and group identification on hypothetical and experimental pain tolerance. Pain 129(1–2): 122–129.
Pope, C. 2023. CGRP Inhibitors. https://www.drugs.com/drug-class/cgrp-inhibitors.html (accessed March 27, 2024).
Porkka-Heiskanen, T., A. Kalinchuk, L. Alanko, I. Huhtaniemi, and D. Stenberg. 2004. Orexin A and B levels in the hypothalamus of female rats: The effects of the estrous cycle and age. European Journal of Endocrinology of the European Federation of Endocrine Societies 150(5):737–742.
Porreca, F., and D. W. Dodick. 2023. Considering patient sex in prescribing CGRP receptor antagonists for short-term treatment of migraine. JAMA Neurology 80(9):885–886.
Poynter, B., M. Shuman, N. Diaz-Granados, M. Kapral, S. L. Grace, and D. E. Stewart. 2009. Sex differences in the prevalence of post-stroke depression: A systematic review. Psychosomatics 50(6):563–569.
Prat, E., and R. Martin. 2002. The immunopathogenesis of multiple sclerosis. Journal of Rehabilitation Research and Development 39(2):187–199.
Pullen, A. B., V. Kain, C. N. Serhan, and G. V. Halade. 2020. Molecular and cellular differences in cardiac repair of male and female mice. Journal of the American Heart Association 9(8):e015672.
Puri, V., S. Puri, S. R. Svojanovsky, S. Mathur, R. R. Macgregor, R. M. Klein, K. M. Welch, and N. E. Berman. 2006. Effects of oestrogen on trigeminal ganglia in culture: Implications for hormonal effects on migraine. Cephalalgia 26(1):33–42.
Qaseem, A., T. J. Wilt, R. M. McLean, M. A. Forciea, T. D. Denberg, M. J. Barry, C. Boyd, R. D. Chow, N. Fitterman, R. P. Harris, L. L. Humphrey, and S. Vijan. 2017. Noninvasive treatments for acute, subacute, and chronic low back pain: A clinical practice guideline from the American College of Physicians. Annals of Internal Medicine 166(7): 514–530.
Quenby, S., I. D. Gallos, R. K. Dhillon-Smith, M. Podesek, M. D. Stephenson, J. Fisher, J. J. Brosens, J. Brewin, R. Ramhorst, E. S. Lucas, R. C. McCoy, R. Anderson, S. Daher, L. Regan, M. Al-Memar, T. Bourne, D. A. MacIntyre, R. Rai, O. B. Christiansen, M. Sugiura-Ogasawara, J. Odendaal, A. J. Devall, P. R. Bennett, S. Petrou, and A. Coomarasamy. 2021. Miscarriage matters: The epidemiological, physical, psychological, and economic costs of early pregnancy loss. The Lancet 397(10285):1658–1667.
Quigley, J. A., M. K. Logsdon, B. C. Graham, K. G. Beaudoin, and J. B. Becker. 2021a. Activation of G protein–coupled estradiol receptor 1 in the dorsolateral striatum enhances motivation for cocaine and drug-induced reinstatement in female but not male rats. Biology of Sex Differences 12(1):46–46.
Quigley, J. A., M. K. Logsdon, C. A. Turner, I. L. Gonzalez, N. B. Leonardo, and J. B. Becker. 2021b. Sex differences in vulnerability to addiction. Neuropharmacology 187:108491–108491.
Quintas, M., J. L. Neto, J. Sequeiros, A. Sousa, J. Pereira-Monteiro, C. Lemos, and I. Alonso. 2020. Going deep into synaptic vesicle machinery genes and migraine susceptibility—a case-control association study. Headache 60(10):2152–2165.
Racine, M., Y. Tousignant-Laflamme, L. A. Kloda, D. Dion, G. Dupuis, and M. Choinière. 2012. A systematic literature review of 10 years of research on sex/gender and experimental pain perception—part 1: Are there really differences between women and men? Pain 153(3):602–618.
Radjabzadeh, D., J. A. Bosch, A. G. Uitterlinden, A. H. Zwinderman, M. A. Ikram, J. B. J. van Meurs, A. I. Luik, M. Nieuwdorp, A. Lok, C. M. van Duijn, R. Kraaij, and N. Amin. 2022. Gut microbiome-wide association study of depressive symptoms. Nature Communications 13(1):7128.
Rahalkar, N., A. Holman-Vittone, C. Daniele, R. Wacks, A. Gagnon, A. D’Agata, N. Saquib, P. F. Schnatz, M. C. Sullivan, R. Wallace, and C. N. Spracklen. 2023. Preterm birth, birthweight, and subsequent risk for depression. Journal of Developmental Origins of Health and Disease 14(5):623–630.
Rahman, A., H. Jackson, H. Hristov, R. S. Isaacson, N. Saif, T. Shetty, O. Etingin, C. Henchcliffe, R. D. Brinton, and L. Mosconi. 2019. Sex and gender driven modifiers of Alzheimer’s: The role for estrogenic control across age, race, medical, and lifestyle risks. Frontiers in Aging Neuroscience 11.
Ralph, L. J., S. I. McCoy, K. Shiu, and N. S. Padian. 2015. Hormonal contraceptive use and women’s risk of HIV acquisition: A meta-analysis of observational studies. The Lancet Infectious Diseases 15(2):181–189.
Ray, P. R., J. Khan, A. Wangzhou, D. Tavares-Ferreira, A. N. Akopian, G. Dussor, and T. J. Price. 2019. Transcriptome analysis of the human tibial nerve identifies sexually dimorphic expression of genes involved in pain, inflammation, and neuro-immunity. Frontiers Molecular Neuroscience 12:37.
Ray, P. R., S. Shiers, J. P. Caruso, D. Tavares-Ferreira, I. Sankaranarayanan, M. L. Uhelski, Y. Li, R. Y. North, C. Tatsui, G. Dussor, M. D. Burton, P. M. Dougherty, and T. J. Price. 2023. RNA profiling of human dorsal root ganglia reveals sex differences in mechanisms promoting neuropathic pain. Brain 146(2):749–766.
Reeves, M., A. Bhatt, P. Jajou, M. Brown, and L. Lisabeth. 2009a. Sex differences in the use of intravenous RT-PA thrombolysis treatment for acute ischemic stroke: A meta-analysis. Stroke 40(5):1743–1749.
Reeves, M. J., G. C. Fonarow, X. Zhao, E. E. Smith, and L. H. Schwamm. 2009b. Quality of care in women with ischemic stroke in the GWTG program. Stroke 40(4):1127–1133.
Regensteiner, J. G., S. Golden, A. G. Huebschmann, E. Barrett-Connor, A. Y. Chang, D. Chyun, C. S. Fox, C. Kim, N. Mehta, J. F. Reckelhoff, J. E. B. Reusch, K. M. Rexrode, A. E. Sumner, F. K. Welty, N. K. Wenger, and B. Anton. 2015. Sex differences in the cardiovascular consequences of diabetes mellitus. Circulation 132(25):2424–2447.
Regitz-Zagrosek, V., and G. Kararigas. 2017. Mechanistic pathways of sex differences in cardiovascular disease. Physiology Reviews 97(1):1–37.
Reid, J. M., D. Dai, G. J. Gubitz, M. K. Kapral, C. Christian, and S. J. Phillips. 2008. Gender differences in stroke examined in a 10-year cohort of patients admitted to a Canadian teaching hospital. Stroke 39(4):1090–1095.
Reid, N., S. Weerasekera, R. E. Hubbard, and E. H. Gordon. 2021. Frailty in ethnic minority women. Maturitas 152:26–31.
Reitz, C., G. Jun, A. Naj, R. Rajbhandary, B. N. Vardarajan, L.-S. Wang, O. Valladares, C.-F. Lin, E. B. Larson, N. R. Graff-Radford, D. Evans, P. L. De Jager, P. K. Crane, J. D. Buxbaum, J. R. Murrell, T. Raj, N. Ertekin-Taner, M. Logue, C. T. Baldwin, R. C. Green, L. L. Barnes, L. B. Cantwell, M. D. Fallin, R. C. P. Go, P. Griffith, T. O. Obisesan, J. J. Manly, K. L. Lunetta, M. I. Kamboh, O. L. Lopez, D. A. Bennett, H. Hendrie, K. S. Hall, A. M. Goate, G. S. Byrd, W. A. Kukull, T. M. Foroud, J. L. Haines, L. A. Farrer, M. A. Pericak-Vance, G. D. Schellenberg, R. Mayeux, and Alzheimer Disease Genetics Consortium. 2013. Variants in the ATP-binding cassette transporter (ABCA7), apolipoprotein E ε4, and the risk of late-onset Alzheimer disease in African Americans. JAMA 309(14):1483–1492.
Reitz, C., E. Rogaeva, and G. W. Beecham. 2020. Late-onset vs. nonMendelian early-onset Alzheimer disease: A distinction without a difference? Neurology Genetics 6(5):e512.
Remes, O., J. F. Mendes, and P. Templeton. 2021. Biological, psychological, and social determinants of depression: A review of recent literature. Brain Science 11(12).
Ren, P., W. B. Wang, H. H. Pan, C. Y. Qiu, and W. P. Hu. 2018. Up-regulation of ASIC3 expression by β-estradiol. Neuroscience Letters 684:200–204.
Reverte, I., A. B. Klein, C. Ratner, J. L. Domingo, and M. T. Colomina. 2012. Behavioral phenotype and BDNF differences related to ApoE isoforms and sex in young transgenic mice. Experimental Neurology 237(1):116–125.
Rexrode, K. M., T. E. Madsen, A. Y. X. Yu, C. Carcel, J. H. Lichtman, and E. C. Miller. 2022. The impact of sex and gender on stroke. Circulation Research 130(4):512–528.
Reyes, M., R. Nisenbaum, D. C. Hoaglin, E. R. Unger, C. Emmons, B. Randall, J. A. Stewart, S. Abbey, J. F. Jones, N. Gantz, S. Minden, and W. C. Reeves. 2003. Prevalence and incidence of chronic fatigue syndrome in Wichita, Kansas. Archives of Internal Medicine 163(13):1530–1536.
Reynolds, H. R., L. J. Shaw, J. K. Min, J. A. Spertus, B. R. Chaitman, D. S. Berman, M. H. Picard, R. Y. Kwong, C. N. Bairey-Merz, D. D. Cyr, R. D. Lopes, J. L. Lopez-Sendon, C. Held, H. Szwed, R. Senior, G. Gosselin, R. G. Nair, A. Elghamaz, O. Bockeria, J. Chen, A. M. Chernyavskiy, B. Bhargava, J. D. Newman, S. B. Hinic, J. Jaroch, A. Hoye, J. Berger, W. E. Boden, S. M. O’Brien, D. J. Maron, and J. S. Hochman. 2020. Association of sex with severity of coronary artery disease, ischemia, and symptom burden in patients with moderate or severe ischemia: Secondary analysis of the ischemia randomized clinical trial. JAMA Cardiology 5(7):773–786.
Reynolds, H. R., A. Maehara, R. Y. Kwong, T. Sedlak, J. Saw, N. R. Smilowitz, E. Mahmud, J. Wei, K. Marzo, M. Matsumura, A. Seno, A. Hausvater, C. Giesler, N. Jhalani, C. Toma, B. Har, D. Thomas, L. S. Mehta, J. Trost, P. K. Mehta, B. Ahmed, K. R. Bainey, Y. Xia, B. Shah, M. Attubato, S. Bangalore, L. Razzouk, Z. A. Ali, N. B. Merz, K. Park, E. Hada, H. Zhong, and J. S. Hochman. 2021. Coronary optical coherence tomography and cardiac magnetic resonance imaging to determine underlying causes of myocardial infarction with nonobstructive coronary arteries in women. Circulation 143(7): 624–640.
Rhodes, M. E., and R. T. Rubin. 1999. Functional sex differences (“sexual diergism”) of central nervous system cholinergic systems, vasopressin, and hypothalamic-pituitary-adrenal axis activity in mammals: A selective review. Brain Research Reviews 30(2):135–152.
Rice, W. S., F. E. Fletcher, B. Akingbade, M. Kan, S. Whitfield, S. Ross, C. A. Gakumo, I. Ofotokun, D. J. Konkle-Parker, M. H. Cohen, G. M. Wingood, B. W. Pence, A. A. Adimora, T. N. Taylor, T. E. Wilson, S. D. Weiser, M.-C. Kempf, B. Turan, and J. M. Turan. 2020. Quality of care for Black and Latina women living with HIV in the U.S.: A qualitative study. International Journal for Equity in Health 19(1):115.
Richardson, G. M., E. Mitchell, K. Stratton, L. R. Kair, and L. M. Marshall. 2022. History of breastfeeding and stroke prevalence among parous females: Findings from the National Health and Nutrition Examination Survey, 1999–2012. Breastfeeding Medicine 17(9):768–775.
Riehman, K. S., A. H. Kral, R. Anderson, N. Flynn, and R. N. Bluthenthal. 2004. Sexual relationships, secondary syringe exchange, and gender differences in HIV risk among drug injectors. Journal of Urban Health 81(2):249–259.
Rivera, H. M., and T. L. Stincic. 2018. Estradiol and the control of feeding behavior. Steroids 133:44–52.
Rivero, K., V. L. Portal, M. Vieira, and I. Behle. 2008. Prevalence of the impaired glucose metabolism and its association with risk factors for coronary artery disease in women with gestational diabetes. Diabetes Research and Clinical Practice 79(3):433–437.
Robblee, J., and K. W. Grimsrud. 2020. Emergency department and inpatient management of headache in adults. Current Neurology and Neuroscience Reports 20(4):7.
Roberts, A. J., A. D. Smith, F. Weiss, C. Rivier, and G. F. Koob. 1998. Estrous cycle effects on operant responding for ethanol in female rats. Alcoholism: Clinical and Experimental Research 22(7):1564–1569.
Roberts, A. L., M. Rosario, H. L. Corliss, D. Wypij, J. R. Lightdale, and S. B. Austin. 2013. Sexual orientation and functional pain in U.S. young adults: The mediating role of childhood abuse. PloS One 8(1):e54702.
Roberts, D. C. S., S. A. L. Bennett, and G. J. Vickers. 1989. The estrous cycle affects cocaine self-administration on a progressive ratio schedule in rats. Psychopharmacology 98(3): 408–411.
Roberts, R. O., J. A. Aakre, W. K. Kremers, M. Vassilaki, D. S. Knopman, M. M. Mielke, R. Alhurani, Y. E. Geda, M. M. Machulda, P. Coloma, B. Schauble, V. J. Lowe, C. R. Jack, Jr., and R. C. Petersen. 2018. Prevalence and outcomes of amyloid positivity among persons without dementia in a longitudinal, population-based setting. JAMA Neurology 75(8):970–979.
Robinson, M. J. F., A. M. Fischer, A. Ahuja, E. N. Lesser, and H. Maniates. 2015. Roles of “wanting” and “liking” in motivating behavior: Gambling, food, and drug addictions. In Behavioral Neuroscience of Motivation. Cham: Springer International Publishing.
Robinson, T. E., and J. B. Becker. 1986. Enduring changes in brain and behavior produced by chronic amphetamine administration: A review and evaluation of animal models of amphetamine psychosis. Brain Research Reviews 11(2):157–198.
Robling, A. G., and L. F. Bonewald. 2020. The osteocyte: New insights. Annual Review of Physiology 82:485–506.
Rocca, W. A., J. H. Bower, D. M. Maraganore, J. E. Ahlskog, B. R. Grossardt, M. de Andrade, and L. J. M. III. 2007. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology 69(11):1074–1083.
Rocca, W. A., M. M. Mielke, P. Vemuri, and V. M. Miller. 2014. Sex and gender differences in the causes of dementia: A narrative review. Maturitas 79(2):196–201.
Rockwood, K., and A. Mitnitski. 2007. Frailty in relation to the accumulation of deficits. Journals of Gerontology. Series A: Biological Sciences and Medical Sciences 62(7):722–727.
Rodriguez-Garcia, M., K. Connors, and M. Ghosh. 2021. HIV pathogenesis in the human female reproductive tract. Current HIV/AIDS Reports 18(2):139–156.
Rocca, W. A., L. Gazzuola Rocca, C. Y. Smith, B. R. Grossardt, S. S. Faubion, L. T. Shuster, E. A. Stewart, M. M. Mielke, K. Kantarci, and V. M. Miller. 2017. Cohort profile: The Mayo Clinic Cohort Study of Oophorectomy and Aging-2 (MOA-2) in Olmsted County, Minnesota (USA). BMJ Open 7(11):e018861.
Rodriguez, F., U. J. Lee, N. Barone, K. Swett, L. Lopez, S. Cheng, M. L. Daviglus, D. B. Hanna, R. A. Espinoza Giacinto, W. Arguelles, J. Cai, G. A. Talavera, and C. J. Rodriguez. 2021. Risk factor control across the spectrum of cardiovascular risk: Findings from the Hispanic Community Health Study/Study of Latinos (HCHS/SOL). American Journal of Preventive Cardiology 5:100147.
Rogan, M. M., and K. E. Black. 2023. Dietary energy intake across the menstrual cycle: A narrative review. Nutrition Reviews 81(7):869–886.
Rogan, S., E. D. de Bruin, L. Radlinger, C. Joehr, C. Wyss, N. J. Stuck, Y. Bruelhart, R. A. de Bie, and R. Hilfiker. 2015. Effects of whole-body vibration on proxies of muscle strength in old adults: A systematic review and meta-analysis on the role of physical capacity level. European Review of Aging and Physical Activity 12:12.
Rolland, Y., C. Dray, B. Vellas, and P. S. Barreto. 2023. Current and investigational medications for the treatment of sarcopenia. Metabolism: Clinical and Experimental 149:155597.
Román-Gálvez, R. M., S. Martín-Peláez, B. M. Fernández-Félix, J. Zamora, K. S. Khan, and A. Bueno-Cavanillas. 2021. Worldwide prevalence of intimate partner violence in pregnancy. A systematic review and meta-analysis. Frontiers in Public Health 9:738459.
Rosario, E. R., L. Chang, E. H. Head, F. Z. Stanczyk, and C. J. Pike. 2011. Brain levels of sex steroid hormones in men and women during normal aging and in Alzheimer’s disease. Neurobiology of Aging 32(4):604–613.
Rosen, S., B. Ham, and J. S. Mogil. 2017. Sex differences in neuroimmunity and pain. Journal of Neuroscience Research 95(1–2):500–508.
Rosenzweig, R., S. Gupta, V. Kumar, R. J. Gumina, and S. S. Bansal. 2021. Estrogenic bias in T-lymphocyte biology: Implications for cardiovascular disease. Pharmacological Research 170:105606.
Roth, M. E., A. G. Casimir, and M. E. Carroll. 2002. Influence of estrogen in the acquisition of intravenously self-administered heroin in female rats. Pharmacology, Biochemistry and Behavior 72(1–2):313–318.
Ruau, D., L. Y. Liu, J. D. Clark, M. S. Angst, and A. J. Butte. 2012. Sex differences in reported pain across 11,000 patients captured in electronic medical records. Journal of Pain 13(3):228–234.
Rubinski, A., S. Frerich, R. Malik, N. Franzmeier, A. Ramirez, M. Dichgans, M. Ewers, and Alzheimer’s Disease Neuroimaging Initiative. 2023. Polygenic effect on tau pathology progression in Alzheimer’s disease. Annals of Neurology 93(4):819–829.
Ruiz-Esteves, K. N., J. Teysir, D. Schatoff, E. W. Yu, and S. M. Burnett-Bowie. 2022. Disparities in osteoporosis care among postmenopausal women in the United States. Maturitas 156:25–29.
Russell, L. M., P. Strike, C. E. Browne, and P. A. Jacobs. 2007. X chromosome loss and ageing. Cytogenetic and Genome Research 116(3):181–185.
Sachs-Ericsson, N., and J. A. Ciarlo. 2000. Gender, social roles, and mental health: An epidemiological perspective. Sex Roles 43:605–628.
Salaffi, F., M. Di Carlo, S. Farah, V. Giorgi, N. Mosca, and P. Sarzi-Puttini. 2022. Overactive bladder syndrome and sexual dysfunction in women with fibromyalgia and their relationship with disease severity. Clinical and Experimental Rheumatology 40(6):1091–1101.
Salari, N., Y. Khodayari, A. Hosseinian-Far, H. Zarei, S. Rasoulpoor, H. Akbari, and M. Mohammadi. 2022. Global prevalence of chronic fatigue syndrome among long COVID-19 patients: A systematic review and meta-analysis. Biopsychosocial Medicine 16(1):21.
Salari, N., F. Babajani, A. Hosseinian-Far, R. Hasheminezhad, N. Abdoli, P. Haydarisharaf, and M. Mohammadi. 2024. Global prevalence of major depressive disorder, generalized anxiety, stress, and depression among infertile women: A systematic review and meta-analysis. Archives of Gynecology and Obstetrics 309(5):1833–1846.
Salehi, A., V. Gonzalez Martinez, and D. F. Swaab. 1998. A sex difference and no effect of APOE type on the amount of cytoskeletal alterations in the nucleus basalis of Meynert in Alzheimer’s disease. Neurobiology of Aging 19(6):505–510.
Salihu, H. M., D. Dongarwar, E. Oduguwa, J. O. Atkinson, and T. B. Harris. 2022. Racial/ethnic disparity in suicidal ideation, suicide attempt and non-suicidal intentional self-harm among pregnant women in the United States. Journal of Immigrant and Minority Health 24(3):588–596.
Sampath, D., T. E. Branyan, K. G. Markowsky, R. Gunda, N. Samiya, A. Obenaus, and F. Sohrabji. 2023a. Sex differences in cognitive impairment after focal ischemia in middle-aged rats and the effect of IV MIR-20A-3P treatment. Neurobiology of Aging 129:168–177.
Sampath, D., M. E. Zardeneta, and F. Sohrabji. 2023b. Synopsis theme: Targeting astrocytes for poststroke recovery. Stroke 54(12):e503–e504.
Sanacora, G., G. F. Mason, D. L. Rothman, K. L. Behar, F. Hyder, O. A. Petroff, R. M. Berman, D. S. Charney, and J. H. Krystal. 1999. Reduced cortical gamma-aminobutyric acid levels in depressed patients determined by proton magnetic resonance spectroscopy. Archives of General Psychiatry 56(11):1043–1047.
Santilli, V., A. Bernetti, M. Mangone, and M. Paoloni. 2014. Clinical definition of sarcopenia. Clinical Cases in Mineral and Bone Metabolism 11(3):177–180.
Saw, J., E. Aymong, G. B. Mancini, T. Sedlak, A. Starovoytov, and D. Ricci. 2014a. Nonatherosclerotic coronary artery disease in young women. Canadian Journal of Cardiology 30(7):814–819.
Saw, J., E. Aymong, T. Sedlak, C. E. Buller, A. Starovoytov, D. Ricci, S. Robinson, T. Vuurmans, M. Gao, K. Humphries, and G. B. Mancini. 2014b. Spontaneous coronary artery dissection: Association with predisposing arteriopathies and precipitating stressors and cardiovascular outcomes. Circulation Cardiovascular Interventions 7(5):645–655.
Schächter, F., L. Faure-Delanef, F. Guénot, H. Rouger, P. Froguel, L. Lesueur-Ginot, and D. Cohen. 1994. Genetic associations with human longevity at the ApoE and ACE loci. Nature Genetics 6(1):29–32.
Scheidell, J. D., M. Pitre, and B. Andraka-Christou. 2024. Racial and ethnic inequities in substance use treatment among women with opioid use disorder. American Journal of Drug and Alcohol Abuse 50(1):106–116.
Schertzinger, M., K. Wesson-Sides, L. Parkitny, and J. Younger. 2018. Daily fluctuations of progesterone and testosterone are associated with fibromyalgia pain severity. The Journal of Pain 19(4):410–417.
Scheyer, O., A. Rahman, H. Hristov, C. Berkowitz, R. S. Isaacson, R. Diaz Brinton, and L. Mosconi. 2018. Female sex and Alzheimer’s risk: The menopause connection. The Journal of Prevention of Alzheimer’s Disease 5(4):224–230.
Schmittner, J., J. R. Schroeder, D. H. Epstein, and K. L. Preston. 2005. Menstrual cycle length during methadone maintenance. Addiction 100(6):829–836.
Schoeb, M., T. M. Avci, E. M. Winter, and N. M. Appelman-Dijkstra. 2023. Safety outcomes of impact microindentation: A prospective observational study in the Netherlands. JBMR Plus 7(10).
Scholz, J., and C. J. Woolf. 2007. The neuropathic pain triad: Neurons, immune cells and glia. Nature Neuroscience 10(11):1361–1368.
Schreiber, J. J., P. A. Anderson, H. G. Rosas, A. L. Buchholz, and A. G. Au. 2011. Hounsfield units for assessing bone mineral density and strength: A tool for osteoporosis management. Journal of Bone and Joint Surgery (American Volume) 93(11):1057–1063.
Schuler, M. S., D. M. Prince, J. Breslau, and R. L. Collins. 2020. Substance use disparities at the intersection of sexual identity and race/ethnicity: Results from the 2015–2018 National Survey on Drug Use and Health. LGBT Health 7(6):283–291.
Schultz, W. 1986. Responses of midbrain dopamine neurons to behavioral trigger stimuli in the monkey. Journal of Neurophysiology 56(5):1439–1461.
Schwamm, L. H., S. F. Ali, M. J. Reeves, E. E. Smith, J. L. Saver, S. Messe, D. L. Bhatt, M. V. Grau-Sepulveda, E. D. Peterson, and G. C. Fonarow. 2013. Temporal trends in patient characteristics and treatment with intravenous thrombolysis among acute ischemic stroke patients at get with the guidelines-stroke hospitals. Circulation: Cardiovascular Quality and Outcomes 6(5):543–549.
Scioscia, M. F., and M. B. Zanchetta. 2023. Recent insights into pregnancy and lactation-associated osteoporosis (PLO). International Journal of Women’s Health 15:1227–1238.
Scully, E. P. 2018. Sex differences in HIV infection. Current HIV/AIDS Reports 15(2): 136–146.
Seney, M. L., L. C. Chang, H. Oh, X. Wang, G. C. Tseng, D. A. Lewis, and E. Sibille. 2013a. The role of genetic sex in affect regulation and expression of GABA-related genes across species. Frontiers in Psychiatry 4:104.
Seney, M. L., K. I. Ekong, Y. Ding, G. C. Tseng, and E. Sibille. 2013b. Sex chromosome complement regulates expression of mood-related genes. Biology of Sex Differences 4(1):20.
Seney, M. L., J. Glausier, and E. Sibille. 2022. Large-scale transcriptomics studies provide insight into sex differences in depression. Biological Psychiatry 91(1):14–24.
Sequeira, A., F. Mamdani, C. Ernst, M. P. Vawter, W. E. Bunney, V. Lebel, S. Rehal, T. Klempan, A. Gratton, C. Benkelfat, G. A. Rouleau, N. Mechawar, and G. Turecki. 2009. Global brain gene expression analysis links glutamatergic and GABAergic alterations to suicide and major depression. PloS One 4(8):e6585.
Serrano-Pozo, A., S. Das, and B. T. Hyman. 2021. ApoE and Alzheimer’s disease: Advances in genetics, pathophysiology, and therapeutic approaches. Lancet Neurology 20(1):68–80.
Shah, A., C. J. Hayes, and B. C. Martin. 2017. Characteristics of initial prescription episodes and likelihood of long-term opioid use—United States, 2006–2015. Morbidity and Mortality Weekly Report 66(10):265–269.
Shah, N. S., K. Xi, K. I. Kapphahn, M. Srinivasan, T. Au, V. Sathye, V. Vishal, H. Zhang, and L. P. Palaniappan. 2022. Cardiovascular and cerebrovascular disease mortality in Asian American subgroups. Circulation: Cardiovascular Quality and Outcomes 15(5):e008651.
Shajahan, S., L. Sun, K. Harris, X. Wang, E. C. Sandset, A. Y. Yu, M. Woodward, S. A. Peters, and C. Carcel. 2023. Sex differences in the symptom presentation of stroke: A systematic review and meta-analysis. International Journal of Stroke 18(2):144-153.
Sharp, E. S., and M. Gatz. 2011. Relationship between education and dementia: An updated systematic review. Alzheimer Disease and Associated Disorders 25(4):289–304.
Shaw, L. J., C. N. B. Merz, C. J. Pepine, S. E. Reis, V. Bittner, K. E. Kip, S. F. Kelsey, M. Olson, B. D. Johnson, S. Mankad, B. L. Sharaf, W. J. Rogers, G. M. Pohost, and G. Sopko. 2006. The economic burden of angina in women with suspected ischemic heart disease. Circulation 114(9):894–904.
Shen, M. J., E. B. Peterson, R. Costas-Muñiz, M. H. Hernandez, S. T. Jewell, K. Matsoukas, and C. L. Bylund. 2018. The effects of race and racial concordance on patient-physician communication: A systematic review of the literature. Journal of Racial and Ethnic Health Disparities 5(1):117–140.
Sherrington, R., E. I. Rogaev, Y. Liang, E. A. Rogaeva, G. Levesque, M. Ikeda, H. Chi, C. Lin, G. Li, K. Holman, T. Tsuda, L. Mar, J. F. Foncin, A. C. Bruni, M. P. Montesi, S. Sorbi, I. Rainero, L. Pinessi, L. Nee, I. Chumakov, D. Pollen, A. Brookes, P. Sanseau, R. J. Polinsky, W. Wasco, H. A. R. Da Silva, J. L. Haines, M. A. Pericak-Vance, R. E. Tanzi, A. D. Roses, P. E. Fraser, J. M. Rommens, and P. H. St. George-Hyslop. 1995. Cloning of a gene bearing missense mutations in early-onset familial Alzheimer’s disease. Nature 375(6534):754–760.
Shevroja, E., F. P. Cafarelli, G. Guglielmi, and D. Hans. 2021. DXA parameters, trabecular bone score (TBS) and bone mineral density (BMD), in fracture risk prediction in endocrine-mediated secondary osteoporosis. Endocrine 74(1):20–28.
Shevroja, E., J.-Y. Reginster, O. Lamy, N. Al-Daghri, M. Chandran, A.-L. Demoux-Baiada, L. Kohlmeier, M.-P. Lecart, D. Messina, B. M. Camargos, J. Payer, S. Tuzun, N. Veronese, C. Cooper, E. V. McCloskey, and N. C. Harvey. 2023. Update on the clinical use of trabecular bone score (TBS) in the management of osteoporosis: Results of an expert group meeting organized by the European Society for Clinical and Economic aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Disease. Osteoporosis International 34(9):1501–1529.
Sibolt, G., S. Curtze, S. Melkas, T. Pohjasvaara, M. Kaste, P. J. Karhunen, N. K. Oksala, R. Vataja, and T. Erkinjuntti. 2013. Post-stroke depression and depression-executive dysfunction syndrome are associated with recurrence of ischaemic stroke. Cerebrovascular Diseases 36(5–6):336–343.
Silva, B. C., M. Madeira, C. B. d’Alva, S. S. Maeda, N. C. P. de Holanda, M. N. Ohe, V. Szejnfeld, C. A. F. Zerbini, F. J. A. de Paula, and F. Bandeira. 2022. Definition and management of very high fracture risk in women with postmenopausal osteoporosis: A position statement from the brazilian society of endocrinology and metabolism (sbem) and the brazilian association of bone assessment and metabolism (abrasso). Archives of Endocrinolology and Metabolism 66(5):591–603.
Silverman, A. B., H. Z. Reinherz, and R. M. Giaconia. 1996. The long-term sequelae of child and adolescent abuse: A longitudinal community study. Child Abuse and Neglect 20(8):709–723.
Simoncini, T., A. Hafezi-Moghadam, D. P. Brazil, K. Ley, W. W. Chin, and J. K. Liao. 2000. Interaction of oestrogen receptor with the regulatory subunit of phosphatidylinositol-3–oh kinase. Nature 407(6803):538–541.
Sims, R., S. J. van der Lee, A. C. Naj, C. Bellenguez, N. Badarinarayan, J. Jakobsdottir, B. W. Kunkle, A. Boland, R. Raybould, J. C. Bis, E. R. Martin, B. Grenier-Boley, S. Heilmann-Heimbach, V. Chouraki, A. B. Kuzma, K. Sleegers, M. Vronskaya, A. Ruiz, R. R. Graham, R. Olaso, P. Hoffmann, M. L. Grove, B. N. Vardarajan, M. Hiltunen, M. M. Nöthen, C. C. White, K. L. Hamilton-Nelson, J. Epelbaum, W. Maier, S. H. Choi, G. W. Beecham, C. Dulary, S. Herms, A. V. Smith, C. C. Funk, C. Derbois, A. J. Forstner, S. Ahmad, H. Li, D. Bacq, D. Harold, C. L. Satizabal, O. Valladares, A. Squassina, R. Thomas, J. A. Brody, L. Qu, P. Sánchez-Juan, T. Morgan, F. J. Wolters, Y. Zhao, F. S. Garcia, N. Denning, M. Fornage, J. Malamon, M. C. D. Naranjo, E. Majounie, T. H. Mosley, B. Dombroski, D. Wallon, M. K. Lupton, J. Dupuis, P. Whitehead, L. Fratiglioni, C. Medway, X. Jian, S. Mukherjee, L. Keller, K. Brown, H. Lin, L. B. Cantwell, F. Panza, B. McGuinness, S. Moreno-Grau, J. D. Burgess, V. Solfrizzi, P. Proitsi, H. H. Adams, M. Allen, D. Seripa, P. Pastor, L. A. Cupples, N. D. Price, D. Hannequin, A. Frank-García, D. Levy, P. Chakrabarty, P. Caffarra, I. Giegling, A. S. Beiser, V. Giedraitis, H. Hampel, M. E. Garcia, X. Wang, L. Lannfelt, P. Mecocci, G. Eiriksdottir, P. K. Crane, F. Pasquier, V. Boccardi, I. Henández, R. C. Barber, M. Scherer, L. Tarraga, P. M. Adams, M. Leber, Y. Chen, M. S. Albert, S. Riedel-Heller, V. Emilsson, D. Beekly, A. Braae, R. Schmidt, D. Blacker, C. Masullo, H. Schmidt, R. S. Doody, G. Spalletta, W. T. Longstreth, Jr., T. J. Fairchild, P. Bossù, O. L. Lopez, M. P. Frosch, E. Sacchinelli, B. Ghetti, Q. Yang, R. M. Huebinger, F. Jessen, S. Li, M. I. Kamboh, J. Morris, O. Sotolongo-Grau, M. J. Katz, C. Corcoran, M. Dunstan, A. Braddel, C. Thomas, A. Meggy, R. Marshall, A. Gerrish, J. Chapman, M. Aguilar, S. Taylor, M. Hill, M. D. Fairén, A. Hodges, B. Vellas, H. Soininen, I. Kloszewska, M. Daniilidou, J. Uphill, Y. Patel, J. T. Hughes, J. Lord, J. Turton, A. M. Hartmann, R. Cecchetti, C. Fenoglio, M. Serpente, M. Arcaro, C. Caltagirone, M. D. Orfei, A. Ciaramella, S. Pichler, M. Mayhaus, W. Gu, A. Lleó, J. Fortea, R. Blesa, I. S. Barber, K. Brookes, C. Cupidi, R. G. Maletta, D. Carrell, S. Sorbi, S. Moebus, M. Urbano, A. Pilotto, J. Kornhuber, P. Bosco, S. Todd, D. Craig, J. Johnston, M. Gill, B. Lawlor, A. Lynch, N. C. Fox, J. Hardy, R. L. Albin, L. G. Apostolova, S. E. Arnold, S. Asthana, C. S. Atwood, C. T. Baldwin, L. L. Barnes, S. Barral, T. G. Beach, J. T. Becker, E. H. Bigio, T. D. Bird, B. F. Boeve, J. D. Bowen, A. Boxer, J. R. Burke, J. M. Burns, J. D. Buxbaum, N. J. Cairns, C. Cao, C. S. Carlson, C. M. Carlsson, R. M. Carney, M. M. Carrasquillo, S. L. Carroll, C. C. Diaz, H. C. Chui, D. G. Clark, D. H. Cribbs, E. A. Crocco, C. DeCarli, M. Dick, R. Duara, D. A. Evans, K. M. Faber, K. B. Fallon, D. W. Fardo, M. R. Farlow, S. Ferris, T. M. Foroud, D. R. Galasko, M. Gearing, D. H. Geschwind, J. R. Gilbert, N. R. Graff-Radford, R. C. Green, J. H. Growdon, R. L. Hamilton, L. E. Harrell, L. S. Honig, M. J. Huentelman, C. M. Hulette, B. T. Hyman, G. P. Jarvik, E. Abner, L. W. Jin, G. Jun, A. Karydas, J. A. Kaye, R. Kim, N. W. Kowall, J. H. Kramer, F. M. LaFerla, J. J. Lah, J. B. Leverenz, A. I. Levey, G. Li, A. P. Lieberman, K. L. Lunetta, C. G. Lyketsos, D. C. Marson, F. Martiniuk, D. C. Mash, E. Masliah, W. C. McCormick, S. M. McCurry, A. N. McDavid, A. C. McKee, M. Mesulam, B. L. Miller, C. A. Miller, J. W. Miller, J. C. Morris, J. R. Murrell, A. J. Myers, S. O’Bryant, J. M. Olichney, V. S. Pankratz, J. E. Parisi, H. L. Paulson, W. Perry, E. Peskind, A. Pierce, W. W. Poon, H. Potter, J. F. Quinn, A. Raj, M. Raskind, B. Reisberg, C. Reitz, J. M. Ringman, E. D. Roberson, E. Rogaeva, H. J. Rosen, R. N. Rosenberg, M. A. Sager, A. J. Saykin, J. A. Schneider, L. S. Schneider, W. W. Seeley, A. G. Smith, J. A. Sonnen, S. Spina, R. A. Stern, R. H. Swerdlow, R. E. Tanzi, T. A. Thornton-Wells, J. Q. Trojanowski, J. C. Troncoso, V. M. Van Deerlin, L. J. Van
Eldik, H. V. Vinters, J. P. Vonsattel, S. Weintraub, K. A. Welsh-Bohmer, K. C. Wilhelmsen, J. Williamson, T. S. Wingo, R. L. Woltjer, C. B. Wright, C. E. Yu, L. Yu, F. Garzia, F. Golamaully, G. Septier, S. Engelborghs, R. Vandenberghe, P. P. De Deyn, C. M. Fernadez, Y. A. Benito, H. Thonberg, C. Forsell, L. Lilius, A. Kinhult-Stählbom, L. Kilander, R. Brundin, L. Concari, S. Helisalmi, A. M. Koivisto, A. Haapasalo, V. Dermecourt, N. Fievet, O. Hanon, C. Dufouil, A. Brice, K. Ritchie, B. Dubois, J. J. Himali, C. D. Keene, J. Tschanz, A. L. Fitzpatrick, W. A. Kukull, M. Norton, T. Aspelund, E. B. Larson, R. Munger, J. I. Rotter, R. B. Lipton, M. J. Bullido, A. Hofman, T. J. Montine, E. Coto, E. Boerwinkle, R. C. Petersen, V. Alvarez, F. Rivadeneira, E. M. Reiman, M. Gallo, C. J. O’Donnell, J. S. Reisch, A. C. Bruni, D. R. Royall, M. Dichgans, M. Sano, D. Galimberti, P. St. George-Hyslop, E. Scarpini, D. W. Tsuang, M. Mancuso, U. Bonuccelli, A. R. Winslow, A. Daniele, C. K. Wu, O. Peters, B. Nacmias, M. Riemenschneider, R. Heun, C. Brayne, D. C. Rubinsztein, J. Bras, R. Guerreiro, A. Al-Chalabi, C. E. Shaw, J. Collinge, D. Mann, M. Tsolaki, J. Clarimón, R. Sussams, S. Lovestone, M. C. O’Donovan, M. J. Owen, T. W. Behrens, S. Mead, A. M. Goate, A. G. Uitterlinden, C. Holmes, C. Cruchaga, M. Ingelsson, D. A. Bennett, J. Powell, T. E. Golde, C. Graff, P. L. De Jager, K. Morgan, N. Ertekin-Taner, O. Combarros, B. M. Psaty, P. Passmore, S. G. Younkin, C. Berr, V. Gudnason, D. Rujescu, D. W. Dickson, J. F. Dartigues, A. L. DeStefano, S. Ortega-Cubero, H. Hakonarson, D. Campion, M. Boada, J. K. Kauwe, L. A. Farrer, C. Van Broeckhoven, M. A. Ikram, L. Jones, J. L. Haines, C. Tzourio, L. J. Launer, V. Escott-Price, R. Mayeux, J. F. Deleuze, N. Amin, P. A. Holmans, M. A. Pericak-Vance, P. Amouyel, C. M. van Duijn, A. Ramirez, L. S. Wang, J. C. Lambert, S. Seshadri, J. Williams, and G. D. Schellenberg. 2017. Rare coding variants in PLCG2, ABI3, and TREM2 implicate microglial-mediated innate immunity in Alzheimer’s disease. Nature Genetics 49(9):1373–1384.
Singer, A. J., S. A. Williams, L. Pearman, Y. Wang, N. Pyrih, and K. Jeray. 2023. An evaluation of treatment patterns for osteoporosis and outcomes after a fragility fracture in a real-world setting. Journal of Orthopaedic Trauma 37(4):e159–e164.
Siracusa, R., R. D. Paola, S. Cuzzocrea, and D. Impellizzeri. 2021. Fibromyalgia: Pathogenesis, mechanisms, diagnosis and treatment options update. International Journal of Molecular Sciences 22(8).
Sit, D. K., and K. L. Wisner. 2009. Identification of postpartum depression. Clinical Obstetrics and Gynecology 52(3):456–468.
Skup, M., H. Zhu, Y. Wang, K. S. Giovanello, J. A. Lin, D. Shen, F. Shi, W. Gao, W. Lin, Y. Fan, and H. Zhang. 2011. Sex differences in grey matter atrophy patterns among AD and AMCI patients: Results from ADNI. Neuroimage 56(3):890–906.
Smilowitz, N. R., A. M. Mahajan, M. T. Roe, A. S. Hellkamp, K. Chiswell, M. Gulati, and H. R. Reynolds. 2017. Mortality of myocardial infarction by sex, age, and obstructive coronary artery disease status in the action registry—GWTG (acute coronary treatment and intervention outcomes network registry—get with the guidelines). Circulation: Cardiovascular Quality and Outcomes 10(12):e003443.
Smith-Bouvier, D. L., A. A. Divekar, M. Sasidhar, S. Du, S. K. Tiwari-Woodruff, J. K. King, A. P. Arnold, R. R. Singh, and R. R. Voskuhl. 2008. A role for sex chromosome complement in the female bias in autoimmune disease. Journal of Experimental Medicine 205(5):1099–1108.
Smith, E. P., J. Boyd, G. R. Frank, H. Takahashi, R. M. Cohen, B. Specker, T. C. Williams, D. B. Lubahn, and K. S. Korach. 1994. Estrogen resistance caused by a mutation in the estrogen-receptor gene in a man. New England Journal of Medicine 331(16): 1056–1061.
Smith, P. J., O. Ornatsky, D. J. Stewart, P. Picard, F. Dawood, W. H. Wen, P. P. Liu, D. J. Webb, and J. C. Monge. 2000. Effects of estrogen replacement on infarct size, cardiac remodeling, and the endothelin system after myocardial infarction in ovariectomized rats. Circulation 102(24):2983–2989.
Sneddon, E. A., L. N. Rasizer, N. G. Cavalco, A. H. Jaymes, N. J. Ostlie, B. L. Minshall, B. M. Masters, M. R. Hughes, H. Hrncir, A. P. Arnold, and A. K. Radke. 2022. Gonadal hormones and sex chromosome complement differentially contribute to ethanol intake, preference, and relapse-like behaviour in Four Core Genotypes mice. Addiction Biology 27(5):e13222.
Somerville, B. W. 1972. The role of estradiol withdrawal in the etiology of menstrual migraine. Neurology 22(4):355–365.
Sommerlad, A., M. Kivimäki, E. B. Larson, S. Röhr, K. Shirai, A. Singh-Manoux, and G. Livingston. 2023. Social participation and risk of developing dementia. Nature Aging 3(5):532–545.
Sorge, R. E., J. C. Mapplebeck, S. Rosen, S. Beggs, S. Taves, J. K. Alexander, L. J. Martin, J. S. Austin, S. G. Sotocinal, D. Chen, M. Yang, X. Q. Shi, H. Huang, N. J. Pillon, P. J. Bilan, Y. Tu, A. Klip, R. R. Ji, J. Zhang, M. W. Salter, and J. S. Mogil. 2015. Different immune cells mediate mechanical pain hypersensitivity in male and female mice. Nature Neuroscience 18(8):1081–1083.
Sotzny, F., J. Blanco, E. Capelli, J. Castro-Marrero, S. Steiner, M. Murovska, and C. Scheibenbogen. 2018. Myalgic encephalomyelitis/chronic fatigue syndrome—evidence for an autoimmune disease. Autoimmune Reviews 17(6):601–609.
Spence, R. D., A. J. Wisdom, Y. Cao, H. M. Hill, C. R. Mongerson, B. Stapornkul, N. Itoh, M. V. Sofroniew, and R. R. Voskuhl. 2013. Estrogen mediates neuroprotection and anti-inflammatory effects during eae through Erα signaling on astrocytes but not through erβ signaling on astrocytes or neurons. Journal of Neuroscience 33(26): 10924–10933.
Springer, S. A., J. A. Barocas, A. Wurcel, A. Nijhawan, K. Thakarar, R. Lynfield, H. Hurley, J. Snowden, A. Thornton, C. del Rio. 2020a. Federal and state action needed to end the infectious complications of illicit drug use in the United States: IDSA and HIVMA’s advocacy agenda. The Journal of Infectious Diseases 222(Supplement 5):S230–S238.
Springer, S. A., B. E. Biondi, C. Frank, and N. El-Bassel. 2020b. A call to action to combat the opioid epidemic among women. Journal of Addication and Medicine 14(5): 364–366.
Sramek, J. J., M. F. Murphy, and N. R. Cutler. 2016. Sex differences in the psycho-pharmacological treatment of depression. Dialogues in Clinical Neuroscience 18(4): 447–457.
Staff, A. C., M. L. Costa, R. Dechend, D. P. Jacobsen, and M. Sugulle. 2024. Hypertensive disorders of pregnancy and long-term maternal cardiovascular risk: Bridging epidemiological knowledge into personalized postpartum care and follow-up. Pregnancy Hypertension 36:101127.
Stahl, S. T., S. R. Beach, D. Musa, and R. Schulz. 2017. Living alone and depression: The modifying role of the perceived neighborhood environment. Aging & Mental Health 21(10):1065–1071.
Stamova, B., Y. Tian, G. Jickling, C. Bushnell, X. Zhan, D. Liu, B. P. Ander, P. Verro, V. Patel, W. C. Pevec, N. Hedayati, D. L. Dawson, E. C. Jauch, A. Pancioli, J. P. Broderick, and F. R. Sharp. 2012. The X-chromosome has a different pattern of gene expression in women compared with men with ischemic stroke. Stroke 43(2):326–334.
Strittmatter, W. J., A. M. Saunders, D. Schmechel, M. Pericak-Vance, J. Enghild, G. S. Salvesen, and A. D. Roses. 1993. Apolipoprotein E: High-avidity binding to beta-amyloid and increased frequency of type 4 allele in late-onset familial Alzheimer disease. Proceedings of the National Academy of Sciences 90(5):1977-1981.
Stokes, A. C., W. Xie, D. J. Lundberg, K. Hempstead, A. Zajacova, Z. Zimmer, D. A. Glei, E. Meara, and S. H. Preston. 2020. Increases in BMI and chronic pain for U.S. adults in midlife, 1992 to 2016. SSM Population Health 12:100644.
Stone, N. J., J. G. Robinson, A. H. Lichtenstein, C. N. Bairey Merz, C. B. Blum, R. H. Eckel, A. C. Goldberg, D. Gordon, D. Levy, D. M. Lloyd-Jones, P. McBride, J. S. Schwartz, S. T. Shero, S. C. Smith, Jr., K. Watson, P. W. Wilson, K. M. Eddleman, N. M. Jarrett, K. LaBresh, L. Nevo, J. Wnek, J. L. Anderson, J. L. Halperin, N. M. Albert, B. Bozkurt, R. G. Brindis, L. H. Curtis, D. DeMets, J. S. Hochman, R. J. Kovacs, E. M. Ohman, S. J. Pressler, F. W. Sellke, W. K. Shen, S. C. Smith, Jr., and G. F. Tomaselli. 2014. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines. Circulation 129(25 Suppl 2):S1–45.
Strawbridge, R., A. H. Young, and A. J. Cleare. 2017. Biomarkers for depression: Recent insights, current challenges and future prospects. Neuropsychiatric Disease and Treatment 13:1245–1262.
Strong, B., L. D. Lisabeth, and M. Reeves. 2020. Sex differences in IV thrombolysis treatment for acute ischemic stroke: A systematic review and meta-analysis. Neurology 95(1): e11–e22.
Studd, J., and N. Panay. 1996. Chronic fatigue syndrome. Lancet 348(9038):1384.
Subramanya, V., B. Ambale-Venkatesh, Y. Ohyama, D. Zhao, C. C. Nwabuo, W. S. Post, E. Guallar, P. Ouyang, S. J. Shah, M. A. Allison, C. E. Ndumele, D. Vaidya, D. A. Bluemke, J. A. Lima, and E. D. Michos. 2018. Relation of sex hormone levels with prevalent and 10-year change in aortic distensibility assessed by MRI: The Multi-Ethnic Study of Atherosclerosis. American Journal of Hypertension 31(7):774–783.
Sun, Y., Y. Liang, Y. Jiao, J. Lin, H. Qu, J. Xu, and C. Zhao. 2017. Comparative efficacy and acceptability of antidepressant treatment in poststroke depression: A multiple-treatments meta-analysis. BMJ Open 7(8):e016499.
Sun, Y., B. Liu, L. G. Snetselaar, R. B. Wallace, B. J. Caan, T. E. Rohan, M. L. Neuhouser, A. H. Shadyab, R. T. Chlebowski, J. E. Manson, and W. Bao. 2019. Association of normal-weight central obesity with all-cause and cause-specific mortality among postmenopausal women. JAMA Network Open 2(7):e197337.
Svikis, D. S., D. R. Miles, N. A. Haug, B. Perry, R. Hoehn-Saric, and D. McLeod. 2006. Premenstrual symptomatology, alcohol consumption, and family history of alcoholism in women with premenstrual syndrome. Journal of Studies on Alcohol 67(6):833–836.
Swaims-Kohlmeier, A., A. N. Sheth, J. Brody, F. P. Hardnett, S. Sharma, E. W. Bonning, I. Ofotokun, I. Massud, and J. G. García-Lerma. 2021. Proinflammatory oscillations over the menstrual cycle drives bystander CD4 T cell recruitment and SHIV susceptibility from vaginal challenge. EBioMedicine 69:103472.
Swanson, C. M. 2021. Sleep disruptions and bone health: What do we know so far? Current Opinion in Endocrinology, Diabetes, and Obesity 28(4):348–353.
Szotek, E. L., S. D. Narasipura, and L. Al-Harthi. 2013. 17β-estradiol inhibits HIV-1 by inducing a complex formation between β-catenin and estrogen receptor α on the HIV promoter to suppress HIV transcription. Virology 443(2):375–383.
Tagoe, C. E., T. Sheth, E. Golub, and K. Sorensen. 2019. Rheumatic associations of autoimmune thyroid disease: A systematic review. Clinical Rheumatology 38(7):1801–1809.
Takada, S., M. J. Seamans, M. Javanbakht, C. Bone, R. Ijadi-Maghsoodi, S. Shoptaw, and L. Gelberg. 2024. Nativity and the risk of opioid use disorder among Hispanic/Latinx women in primary care in Los Angeles, CA. Journal of Ethnicity in Substance Abuse: 1–15.
Tamis-Holland, J. E., H. Jneid, H. R. Reynolds, S. Agewall, E. S. Brilakis, T. M. Brown, A. Lerman, M. Cushman, D. J. Kumbhani, C. Arslanian-Engoren, A. F. Bolger, and J. F. Beltrame. 2019. Contemporary diagnosis and management of patients with myocardial infarction in the absence of obstructive coronary artery disease: A scientific statement from the American Heart Association. Circulation 139(18):e891–e908.
Tang, G. B., Y. Q. Zeng, P. P. Liu, T. W. Mi, S. F. Zhang, S. K. Dai, Q. Y. Tang, L. Yang, Y. J. Xu, H. L. Yan, H. Z. Du, Z. Q. Teng, F. Q. Zhou, and C. M. Liu. 2017. The histone H3k27 demethylase UTX regulates synaptic plasticity and cognitive behaviors in mice. Frontiers in Molecular Neuroscience 10:267.
Tavares-Ferreira, D., S. Shiers, P. R. Ray, A. Wangzhou, V. Jeevakumar, I. Sankaranarayanan, A. M. Cervantes, J. C. Reese, A. Chamessian, B. A. Copits, P. M. Dougherty, R. W. t. Gereau, M. D. Burton, G. Dussor, and T. J. Price. 2022. Spatial transcriptomics of dorsal root ganglia identifies molecular signatures of human nociceptors. Science Translational Medicine 14(632):eabj8186.
Taylor, A. M. W., C. I. Chadwick, S. Mehrabani, H. Hrncir, A. P. Arnold, and C. J. Evans. 2022. Sex differences in kappa opioid receptor antinociception is influenced by the number of X chromosomes in mouse. Journal of Neuroscience Research 100(1):183–190.
Tebbens, M., A. C. Heijboer, G. T’Sjoen, P. H. Bisschop, and M. den Heijer. 2022. The role of estrone in feminizing hormone treatment. Journal of Clinical Endocrinology and Metabolism 107(2):e458–e466.
Templeton, K. J. 2020. Sex and gender issues in pain management. JBJS 102(Suppl 1).
Teo, C. H., A. C. H. Wong, R. N. Sivakumaran, I. Parhar, and T. Soga. 2023. Gender differences in cortisol and cortisol receptors in depression: A narrative review. International Journal of Molecular Sciences 24(8).
Theilen, L. H., A. Fraser, M. S. Hollingshaus, K. C. Schliep, M. W. Varner, K. R. Smith, and M. S. Esplin. 2016. All-cause and cause-specific mortality after hypertensive disease of pregnancy. Obstetrics and Gynecology 128(2):238–244.
Thomas, N., C. Gurvich, K. Huang, P. R. Gooley, and C. W. Armstrong. 2022. The underlying sex differences in neuroendocrine adaptations relevant to myalgic encephalomyelitis chronic fatigue syndrome. Frontiers in Neuroendocrinology 66:100995.
Thompson, T., C. U. Correll, K. Gallop, D. Vancampfort, and B. Stubbs. 2016. Is pain perception altered in people with depression? A systematic review and meta-analysis of experimental pain research. The Journal of Pain 17(12):1257–1272.
Thornton, M., M. Sim, M. A. Kennedy, K. Blodgett, R. Joseph, and R. Pojednic. 2023. Nutrition interventions on muscle-related components of sarcopenia in females: A systematic review of randomized controlled trials. Calcified Tissue International 114(1):38–52.
Thurston, R. C., C. A. Karvonen-Gutierrez, C. A. Derby, S. R. El Khoudary, H. M. Kravitz, and J. E. Manson. 2018. Menopause versus chronologic aging: Their roles in women’s health. Menopause 25(8):849–854.
Tietjen, G. E., D. C. Buse, K. M. Fanning, D. Serrano, M. L. Reed, and R. B. Lipton. 2015. Recalled maltreatment, migraine, and tension-type headache: Results of the AMPP study. Neurology 84(2):132–140.
Tietjen, G. E., D. C. Buse, and S. A. Collins. 2016. Childhood maltreatment in the migraine patient. Current Treatment Options in Neurology 18(7).
Tiidus, P. M., D. A. Lowe, and M. Brown. 2013. Estrogen replacement and skeletal muscle: Mechanisms and population health. Journal of Applied Physiology (1985) 115(5): 569–578
Tolkien, K., S. Bradburn, and C. Murgatroyd. 2019. An anti-inflammatory diet as a potential intervention for depressive disorders: A systematic review and meta-analysis. Clinical Nutrition 38(5):2045–2052.
Toro, C. A., L. Zhang, J. Cao, and D. Cai. 2019. Sex differences in Alzheimer’s disease: Understanding the molecular impact. Brain Research 1719:194–207.
Torres, O. V., and L. E. O’Dell. 2016. Stress is a principal factor that promotes tobacco use in females. Progress in Neuro-Psychopharmacology and Biological Psychiatry 65:260–268.
Troutwine, B. R., L. Hamid, C. R. Lysaker, T. A. Strope, and H. M. Wilkins. 2022. Apolipoprotein E and Alzheimer’s disease. Acta Pharmaceutica Sinica B 12(2):496–510.
Tsang, A., M. Von Korff, S. Lee, J. Alonso, E. Karam, M. C. Angermeyer, G. L. Borges, E. J. Bromet, K. Demytteneare, G. de Girolamo, R. de Graaf, O. Gureje, J. P. Lepine, J. M. Haro, D. Levinson, M. A. Oakley Browne, J. Posada-Villa, S. Seedat, and M. Watanabe. 2008. Common chronic pain conditions in developed and developing countries: Gender and age differences and comorbidity with depression-anxiety disorders. Journal of Pain 9(10):883–891.
sadok, M. A., C. A. Jackevicius, E. Rahme, K. H. Humphries, and L. Pilote. 2015. Sex differences in dabigatran use, safety, and effectiveness in a population-based cohort of patients with atrial fibrillation. Circulation: Cardiovascular Quality and Outcomes 8(6):593–599.
Tsao, C. W., A. W. Aday, Z. I. Almarzooq, C. A. M. Anderson, P. Arora, C. L. Avery, C. M. Baker-Smith, A. Z. Beaton, A. K. Boehme, A. E. Buxton, Y. Commodore-Mensah, M. S. V. Elkind, K. R. Evenson, C. Eze-Nliam, S. Fugar, G. Generoso, D. G. Heard, S. Hiremath, J. E. Ho, R. Kalani, D. S. Kazi, D. Ko, D. A. Levine, J. Liu, J. Ma, J. W. Magnani, E. D. Michos, M. E. Mussolino, S. D. Navaneethan, N. I. Parikh, R. Poudel, M. Rezk-Hanna, G. A. Roth, N. S. Shah, M. P. St-Onge, E. L. Thacker, S. S. Virani, J. H. Voeks, N. Y. Wang, N. D. Wong, S. S. Wong, K. Yaffe, and S. S. Martin. 2023. Heart disease and stroke statistics—2023 update: A report from the American Heart Association. Circulation 147(8):e93–e621.
Tsiknia, A. A., S. D. Edland, E. E. Sundermann, E. T. Reas, J. B. Brewer, D. Galasko, S. J. Banks, and Alzheimer’s Disease Neuroimaging Initiative. 2022. Sex differences in plasma p-tau181 associations with Alzheimer’s disease biomarkers, cognitive decline, and clinical progression. Molecular Psychiatry 27(10):4314–4322.
Tukiainen, T., A.-C. Villani, A. Yen, M. A. Rivas, J. L. Marshall, R. Satija, M. Aguirre, L. Gauthier, M. Fleharty, A. Kirby, B. B. Cummings, S. E. Castel, K. J. Karczewski, F. Aguet, A. Byrnes, T. Lappalainen, A. Regev, K. G. Ardlie, N. Hacohen, and D. G. Macarthur. 2017. Landscape of X chromosome inactivation across human tissues. Nature 550(7675):244–248.
Turner-Stokes, L., and D. T. Wade. 2020. Updated NICE guidance on chronic fatigue syndrome. BMJ 371:m4774.
Tweet, M. S., S. N. Hayes, E. Codsi, R. Gulati, C. H. Rose, and P. J. M. Best. 2017. Spontaneous coronary artery dissection associated with pregnancy. Journal of the American College of Cardiology 70(4):426–435.
Tweet, M. S., S. N. Kok, and S. N. Hayes. 2018. Spontaneous coronary artery dissection in women: What is known and what is yet to be understood. Clinical Cardiology 41(2):203–210.
Tzabazis, A., S. Kori, J. Mechanic, J. Miller, C. Pascual, N. Manering, D. Carson, M. Klukinov, E. Spierings, D. Jacobs, J. Cuellar, W. H. Frey, II, L. Hanson, M. Angst, and D. C. Yeomans. 2017. Oxytocin and migraine headache. Headache 57 (Suppl 2):64–75.
UNODC (United Nation Office on Drugs and Crime). 2015. World Drug Report 2015. Vienna, Austria: United Nations publication.
USPSTF (U.S. Preventive Services Task Force), M. J. Barry, W. K. Nicholson, M. Silverstein, D. Chelmow, T. R. Coker, E. M. Davis, K. E. Donahue, C. R. Jaén, M. Kubik, L. Li, G. Ogedegbe, G. Rao, J. M. Ruiz, J. J. Stevermer, J. Tsevat, S. M. Underwood, and J. B. Wong. 2023. Preexposure prophylaxis to prevent acquisition of HIV: U.S. Preventive Services Task Force recommendation statement. JAMA 330(8):736–745.
Vafaei, A., T. Ahmed, N. Freire Ado, M. V. Zunzunegui, and R. O. Guerra. 2016. Depression, sex and gender roles in older adult populations: The International Mobility in Aging Study (IMIAS). PloS One 11(1):e0146867.
Valencia-Olvera, A. C., J. Maldonado Weng, A. Christensen, M. J. LaDu, and C. J. Pike. 2023. Role of estrogen in women’s Alzheimer’s disease risk as modified by ApoE. Journal of Neuroendocrinology 35(2):e13209.
Valente, T. W., R. K. Foreman, B. Junge, and D. Vlahov. 2001. Needle-exchange participation, effectiveness, and policy: Syringe relay, gender, and the paradox of public health. Journal of Urban Health 78(2):340–349.
Valenzuela, M. J., and A. Pascual-Leone. 2023. Lecanemab in early Alzheimer’s disease. New England Journal of Medicine 388(17):1630.
Van De Maele, K., R. Devlieger, and I. Gies. 2018. In utero programming and early detection of cardiovascular disease in the offspring of mothers with obesity. Atherosclerosis 275:182–195.
Van den Bergh, B. R., B. Van Calster, T. Smits, S. Van Huffel, and L. Lagae. 2008. Antenatal maternal anxiety is related to HPA-axis dysregulation and self-reported depressive symptoms in adolescence: A prospective study on the fetal origins of depressed mood. Neuropsychopharmacology 33(3):536–545.
van der Flier, W. M., M. E. de Vugt, E. M. A. Smets, M. Blom, and C. E. Teunissen. 2023. Towards a future where Alzheimer’s disease pathology is stopped before the onset of dementia. Nature Aging 3(5):494–505.
van Dijk, F. S., M. C. Zillikens, D. Micha, M. Riessland, C. L. Marcelis, C. E. de Die-Smulders, J. Milbradt, A. A. Franken, A. J. Harsevoort, K. D. Lichtenbelt, H. E. Pruijs, M. E. Rubio-Gozalbo, R. Zwertbroek, Y. Moutaouakil, J. Egthuijsen, M. Hammerschmidt, R. Bijman, C. M. Semeins, A. D. Bakker, V. Everts, J. Klein-Nulend, N. Campos-Obando, A. Hofman, G. J. te Meerman, A. J. Verkerk, A. G. Uitterlinden, A. Maugeri, E. A. Sistermans, Q. Waisfisz, H. Meijers-Heijboer, B. Wirth, M. E. Simon, and G. Pals. 2013. PLS3 mutations in X-linked osteoporosis with fractures. New England Journal of Medicine 369(16):1529–1536.
van Dyck, C. H., C. J. Swanson, P. Aisen, R. J. Bateman, C. Chen, M. Gee, M. Kanekiyo, D. Li, L. Reyderman, S. Cohen, L. Froelich, S. Katayama, M. Sabbagh, B. Vellas, D. Watson, S. Dhadda, M. Irizarry, L. D. Kramer, and T. Iwatsubo. 2022. Lecanemab in early Alzheimer’s disease. New England Journal of Medicine 388(1):9–21.
Vargas-Alarcon, G., E. Alvarez-Leon, J.-M. Fragoso, A. Vargas, A. Martinez, M. Vallejo, and M. Martinez-Lavin. 2012. A SCN9A gene-encoded dorsal root ganglia sodium channel polymorphism associated with severe fibromyalgia. BMC Musculoskeletal Disorders 13(1):23.
Vasikaran, S. D., M. Miura, R. Pikner, H. P. Bhattoa, and E. Cavalier. 2023. Practical considerations for the clinical application of bone turnover markers in osteoporosis. Calcified Tissue International 112(2):148–157.
Veenema, R., K. M. Casin, P. Sinha, R. Kabir, N. Mackowski, N. Taube, D. Bedja, R. Chen, A. Rule, and M. J. Kohr. 2019. Inorganic arsenic exposure induces sex-disparate effects and exacerbates ischemia-reperfusion injury in the female heart. American Journal of Physiology—Heart & Circulatory Physiology 316(5):H1053–h1064.
Verdiesen, R. M. G., N. C. Onland-Moret, C. H. van Gils, and Y. T. van der Schouw. 2022. Circulating anti-Müllerian hormone levels and markers of subclinical cardiovascular disease in middle-aged and older men. Maturitas 163:38–45.
Verhaak, A., S. Bakaysa, A. Johnson, M. Veronesi, A. Williamson, and B. Grosberg. 2023. Migraine treatment in pregnancy: A survey of comfort and treatment practices of women’s healthcare providers. Headache 63(2):211–221.
Vetvik, K. G., and E. A. MacGregor. 2021. Menstrual migraine: A distinct disorder needing greater recognition. Lancet Neurology 20(4):304–315.
Vidal, O., M. K. Lindberg, K. Hollberg, D. J. Baylink, G. Andersson, D. B. Lubahn, S. Mohan, J.-Å. Gustafsson, and C. Ohlsson. 2000. Estrogen receptor specificity in the regulation of skeletal growth and maturation in male mice. Proceedings of the National Academy of Sciences 97(10):5474–5479.
Vigil, P., J. Meléndez, G. Petkovic, and J. P. Del Río. 2022. The importance of estradiol for body weight regulation in women. Frontiers in Endocrinology 13:951186.
Vijay, A., N. R. Kandula, A. M. Kanaya, S. S. Khan, and N. S. Shah. 2022. Relation of menopause with cardiovascular risk factors in South Asian American women (from the MASALA study). The American Journal of Cardiology 171:165–170.
Vijayan, M., and P. H. Reddy. 2016. Stroke, vascular dementia, and Alzheimer’s disease: Molecular links. Journal of Alzheimer’s Disease 54(2):427–443.
Vimalananda, V. G., D. R. Miller, T. P. Hofer, R. G. Holleman, M. L. Klamerus, and E. A. Kerr. 2013. Accounting for clinical action reduces estimates of gender disparities in lipid management for diabetic veterans. Journal of General Internal Medicine 28 (Suppl 2):S529–535.
Vives-Cases, C., M. T. Ruiz-Cantero, V. Escriba-Aguir, and J. J. Miralles. 2011. The effect of intimate partner violence and other forms of violence against women on health. Journal of Public Health 33(1):15–21.
Vladutiu, C. J., M. L. Meyer, A. M. Malek, A. M. Stuebe, A. Mosher, S. Aggarwal, D. Kleindorfer, and V. J. Howard. 2017. Racial differences in the association between parity and incident stroke: Results from the Reasons for Geographic and Racial Differences in Stroke Study. Journal of Stroke and Cerebrovascular Diseases 26(4):749–755.
Vogel, B., M. Acevedo, Y. Appelman, C. N. Bairey Merz, A. Chieffo, G. A. Figtree, M. Guerrero, V. Kunadian, C. S. P. Lam, A. H. E. M. Maas, A. S. Mihailidou, A. Olszanecka, J. E. Poole, C. Saldarriaga, J. Saw, L. Zühlke, and R. Mehran. 2021. The Lancet Women and Cardiovascular Disease Commission: Reducing the global burden by 2030. The Lancet 397(10292):2385–2438.
Voskuhl, R. R. 2020. The effect of sex on multiple sclerosis risk and disease progression. Multiple Sclerosis 26(5):554–560.
Voskuhl, R. R., and K. Palaszynski. 2001. Sex hormones in experimental autoimmune encephalomyelitis: Implications for multiple sclerosis. Neuroscientist 7(3):258–270.
Voskuhl, R. R., H. Wang, T. C. Wu, N. L. Sicotte, K. Nakamura, F. Kurth, N. Itoh, J. Bardens, J. T. Bernard, J. R. Corboy, A. H. Cross, S. Dhib-Jalbut, C. C. Ford, E. M. Frohman, B. Giesser, D. Jacobs, L. H. Kasper, S. Lynch, G. Parry, M. K. Racke, A. T. Reder, J. Rose, D. M. Wingerchuk, A. J. MacKenzie-Graham, D. L. Arnold, C. H. Tseng, and R. Elashoff. 2016. Estriol combined with glatiramer acetate for women with relapsing-remitting multiple sclerosis: A randomised, placebo-controlled, Phase 2 trial. Lancet Neurology 15(1):35–46.
Voskuhl, R. R., K. Patel, F. Paul, S. M. Gold, M. Scheel, J. Kuchling, G. Cooper, S. Asseyer, C. Chien, A. U. Brandt, C. E. Meyer, and A. MacKenzie-Graham. 2020. Sex differences in brain atrophy in multiple sclerosis. Biology of Sex Differences 11(1):49.
Voskuhl, R., J. Kuhle, P. Siddarth, N. Itoh, K. Patel, and A. MacKenzie-Graham. 2022. Decreased neurofilament light chain levels in estriol-treated multiple sclerosis. Annals of Clinical and Translational Neurology 9(8):1316–1320.
Wang, C. L., M. Kanamori, A. Moreland-Capuia, S. F. Greenfield, and D. E. Sugarman. 2023a. Substance use disorders and treatment in Asian American and Pacific Islander women: A scoping review. American Journal on Addictions 32(3):231–243.
Wang, K. W., Y. X. Yuan, B. Zhu, Y. Zhang, Y. F. Wei, F. S. Meng, S. Zhang, J. X. Wang, and J. Y. Zhou. 2023b. X chromosome-wide association study of quantitative biomarkers from the Alzheimer’s Disease Neuroimaging Initiative study. Frontiers in Aging Neuroscience 15:1277731.
Wang, M., B. M. Tsai, K. M. Reiger, J. W. Brown, and D. R. Meldrum. 2006. 17–beta-estradiol decreases P38 MAPK-mediated myocardial inflammation and dysfunction following acute ischemia. Journal of Molecular and Cellular Cardiology 40(2):205–212.
Wang, M., Y. Wang, B. Weil, A. Abarbanell, J. Herrmann, J. Tan, M. Kelly, and D. R. Meldrum. 2009. Estrogen receptor beta mediates increased activation of PI3K/AKT signaling and improved myocardial function in female hearts following acute ischemia. American Journal of Physiology—Regulatory, Integrative, Comparative Physiology 296(4):R972–978.
Wang, Z., Y. Li, F. Zhou, Z. Piao, and J. Hao. 2016. Effects of statins on bone mineral density and fracture risk: A PRISMA-compliant systematic review and meta-analysis. Medicine 95(22):e3042.
Wang, Z., M. Zhu, Z. Su, B. Guan, A. Wang, Y. Wang, N. Zhang, and C. Wang. 2017. Poststroke depression: Different characteristics based on follow-up stage and gender—a cohort perspective study from mainland China. Neurological Research 39(11): 996–1005.
Ward, A., S. Crean, C. J. Mercaldi, J. M. Collins, D. Boyd, M. N. Cook, and H. M. Arrighi. 2012. Prevalence of apolipoprotein E4 genotype and homozygotes (ApoE E4/4) among patients diagnosed with Alzheimer’s disease: A systematic review and meta-analysis. Neuroepidemiology 38(1):1–17.
Ward, R., J. P. Valenzuela, W. Li, G. Dong, S. C. Fagan, and A. Ergul. 2018. Poststroke cognitive impairment and hippocampal neurovascular remodeling: The impact of diabetes and sex. American Journal of Physiology: Heart and Circulatory Physiology 315(5): H1402–h1413.
Warne, E., M. Oxlad, and T. Best. 2023. Consulting patients and providers of assisted reproductive technologies to inform the development of a group psychological intervention for women with infertility. PEC Innovation 3:100206.
Warner, Matthew J., and Susan E. Ozanne. 2010. Mechanisms involved in the developmental programming of adulthood disease. Biochemical Journal 427(3):333–347.
Warriner, A. H., R. C. Outman, A. C. Feldstein, D. W. Roblin, J. J. Allison, J. R. Curtis, D. T. Redden, M. M. Rix, B. E. Robinson, A. G. Rosales, M. M. Safford, and K. G. Saag. 2014. Effect of self-referral on bone mineral density testing and osteoporosis treatment. Medical Care 52(8):743–750.
Washington, D. L., B. Bean-Mayberry, D. Riopelle, and E. M. Yano. 2011. Access to care for women veterans: Delayed healthcare and unmet need. Journal of General Internal Medicine 26(S2):655–661.
Watkins, L. R., and S. F. Maier. 2003. Glia: A novel drug discovery target for clinical pain. Nature Reviews Drug Discoveries 2(12):973–985.
Wearden, A. J., C. Dowrick, C. Chew-Graham, R. P. Bentall, R. K. Morriss, S. Peters, L. Riste, G. Richardson, K. Lovell, and G. Dunn. 2010. Nurse led, home based self help treatment for patients in primary care with chronic fatigue syndrome: Randomised controlled trial. BMJ 340:c1777.
Weaver, J., S. Sajjan, E. M. Lewiecki, S. T. Harris, and P. Marvos. 2017. Prevalence and cost of subsequent fractures among U.S. patients with an incident fracture. Journal of Managed Care & Specialty Pharmacy 23(4):461–471.
Weiss, E. L., J. G. Longhurst, and C. M. Mazure. 1999. Childhood sexual abuse as a risk factor for depression in women: Psychosocial and neurobiological correlates. American Journal of Psychiatry 156(6):816–828.
Weitzmann, M. N., C. Roggia, G. Toraldo, L. Weitzmann, and R. Pacifici. 2002. Increased production of IL-7 uncouples bone formation from bone resorption during estrogen deficiency. Journal of Clinical Investigation 110(11):1643–1650.
Welten, S., N. C. Onland-Moret, J. M. A. Boer, W. M. M. Verschuren, and Y. T. van der Schouw. 2021. Age at menopause and risk of ischemic and hemorrhagic stroke. Stroke 52(8): 2583–2591.
Wessely, S. 1997. Chronic fatigue syndrome: A 20th century illness? Scandinavian Journal of Work, Environment and Health 23(Suppl 3):17–34.
Wharton, W., L. D. Baker, C. E. Gleason, M. Dowling, J. H. Barnet, S. Johnson, C. Carlsson, S. Craft, and S. Asthana. 2011. Short-term hormone therapy with transdermal estradiol improves cognition for postmenopausal women with Alzheimer’s disease: Results of a randomized controlled trial. Journal of Alzheimer’s Disease 26(3):495–505.
White, H. D., L. A. Brown, R. J. Gyurik, P. D. Manganiello, T. D. Robinson, L. S. Hallock, L. D. Lewis, and K. T. Yeo. 2015. Treatment of pain in fibromyalgia patients with testosterone gel: Pharmacokinetics and clinical response. International Immunopharmacology 27(2):249–256.
Wiese, C. B., R. Avetisyan, and K. Reue. 2023. The impact of chromosomal sex on cardiometabolic health and disease. Trends in Endocrinology and Metabolism 34(10): 652–665.
Wieser, T., J. Pascual, A. Oterino, M. Soso, M. Barmada, and K. L. Gardner. 2010. A novel locus for familial migraine on XP22. Headache 50(6):955–962.
Wild, R. A., E. Carmina, E. Diamanti-Kandarakis, A. Dokras, H. F. Escobar-Morreale, W. Futterweit, R. Lobo, R. J. Norman, E. Talbott, and D. A. Dumesic. 2010. Assessment of cardiovascular risk and prevention of cardiovascular disease in women with the polycystic ovary syndrome: A consensus statement by the Androgen Excess and Polycystic Ovary Syndrome (AE-PCOS) society. Journal of Clinical Endocrinology and Metabolism 95(5):2038–2049.
Wira, C. R., M. Rodriguez-Garcia, and M. V. Patel. 2015. The role of sex hormones in immune protection of the female reproductive tract. Nature Reviews: Immunology 15(4):217–230.
Wisdom, A. J., Y. Cao, N. Itoh, R. D. Spence, and R. R. Voskuhl. 2013. Estrogen receptor-β ligand treatment after disease onset is neuroprotective in the multiple sclerosis model. Journal of Neuroscience Research 91(7):901–908.
Wise, R. A., and G. F. Koob. 2014. The development and maintenance of drug addiction. Neuropsychopharmacology 39(2):254–262.
Wise, R. A., and P. P. Rompre. 1989. Brain dopamine and reward. Annual Review of Psychology 40(1):191–225.
Witkiewitz, K., and S. Bowen. 2010. Depression, craving, and substance use following a randomized trial of mindfulness-based relapse prevention. Journal of Consulting and Clinical Psychology 78(3):362–374.
Witt, E. D. 2007. Puberty, hormones, and sex differences in alcohol abuse and dependence. Neurotoxicology and Teratology 29(1):81–95.
Wolfe, F., B. Walitt, S. Perrot, J. J. Rasker, and W. Häuser. 2018. Fibromyalgia diagnosis and biased assessment: Sex, prevalence and bias. PloS One 13(9):e0203755.
Wolfe, F., H. A. Smythe, M. B. Yunus, R. M. Bennett, C. Bombardier, D. L. Goldenberg, P. Tugwell, S. M. Campbell, M. Abeles, P. Clark, et al. 1990. The American College of Rheumatology 1990 criteria for the classification of fibromyalgia. Report of the Multicenter Criteria Committee. Arthritis and Rheumatism 33(2):160–172.
Wolff, L., E. A. Strathmann, I. Müller, D. Mählich, C. Veltman, A. Niehoff, and B. Wirth. 2021. Plastin 3 in health and disease: A matter of balance. Cellular and Molecular Life Sciences 78(13):5275–5301.
Wright, N. C., L. Chen, K. G. Saag, C. J. Brown, J. M. Shikany, and J. R. Curtis. 2020. Racial disparities exist in outcomes after major fragility fractures. Journal of the American Geriatrics Society 68(8):1803–1810.
Wu, Q., and J. Dai. 2023. Racial/ethnic differences in bone mineral density for osteoporosis. Current Osteoporosis Reports 21(6):670–684.
Wu, Q. E., A. M. Zhou, Y. P. Han, Y. M. Liu, Y. Yang, X. M. Wang, and X. Shi. 2019. Poststroke depression and risk of recurrent stroke: A meta-analysis of prospective studies. Medicine 98(42):e17235.
Wu, Z. C., J. T. Yu, N. D. Wang, N. N. Yu, Q. Zhang, W. Chen, W. Zhang, Q. X. Zhu, and L. Tan. 2010. Lack of association between PCDH11X genetic variation and late-onset Alzheimer’s disease in a Han Chinese population. Brain Research 1357:152–156.
Yancey, J. R., and S. M. Thomas. 2012. Chronic fatigue syndrome: Diagnosis and treatment. American Family Physician 86(8):741–746.
Yang, J., Y. Mao, and J. W. Nieves. 2020a. Identification of prevalent vertebral fractures using vertebral fracture assessment (VFA) in asymptomatic postmenopausal women: A systematic review and meta-analysis. Bone 136:115358.
Yang, T. L., H. Shen, A. Liu, S-S. Dang, L. Zhang, F-Y. Deng, Q. Zhao, H-W. Deng. 2020b. A road map for understanding molecular and genetic determinants of osteoporosis. Nature Reviews Endocrinology 16(2):91–103.
Yang, L., L. Smith, and M. Hamer. 2019. Gender-specific risk factors for incident sarcopenia: 8-year follow-up of the English Longitudinal Study of Ageing. Journal of Epidemiology and Community Health 73(1):86–88.
Yang, M., B. Song, Y. Jiang, Y. Lin, and J. Liu. 2023. Mindfulness-based interventions for postpartum depression: A systematic review and meta-analysis. Iranian Journal of Public Health 52(12):2496–2505.
Yang, S., L. Bae, and L. Zhang. 2000. Estrogen increases ENOS and NOX release in human coronary artery endothelium. Journal of Cardiovascular Pharmacology 36(2): 242–247.
Yim, Y. R., K. E. Lee, D. J. Park, S. H. Kim, S. S. Nah, J. H. Lee, S. K. Kim, Y. A. Lee, S. J. Hong, H. S. Kim, H. S. Lee, H. A. Kim, C. I. Joung, S. H. Kim, and S. S. Lee. 2017. Identifying fibromyalgia subgroups using cluster analysis: Relationships with clinical variables. European Journal of Pain 21(2):374–384.
Yu, A. Y. X., L. C. Maclagan, C. Diong, P. C. Austin, M. K. Kapral, R. H. Swartz, and S. E. Bronskill. 2020. Sex differences in care need and survival in patients admitted to nursing home poststroke. Canadian Journal of Neurological Sciences 47(2):153–159.
Yu, S., C. Chen, Y. Pan, M. C. Kurz, E. Datner, P. L. Hendry, M. A. Velilla, C. Lewandowski, C. Pearson, R. Domeier, S. A. McLean, and S. D. Linnstaedt. 2019. Genes known to escape X chromosome inactivation predict co-morbid chronic musculoskeletal pain and posttraumatic stress symptom development in women following trauma exposure. American Journal of Medical Genetics: Part B 180(6):415–427.
Yuan, M., B. Yang, G. Rothschild, J. J. Mann, L. D. Sanford, X. Tang, C. Huang, C. Wang, and W. Zhang. 2023. Epigenetic regulation in major depression and other stress-related disorders: Molecular mechanisms, clinical relevance and therapeutic potential. Signal Transduction and Targeted Therapy 8(1):309.
Yang, C. P., C. S. Liang, C. M. Chang, C. C. Yang, P. H. Shih, Y. C. Yau, K. T. Tang, and S. J. Wang. 2021. Comparison of new pharmacologic agents with triptans for treatment of migraine: A systematic review and meta-analysis. JAMA Network Open 4(10):e2128544.
Yilmaz, C., K. Karali, G. Fodelianaki, A. Gravanis, T. Chavakis, I. Charalampopoulos, and V. I. Alexaki. 2019. Neurosteroids as regulators of neuroinflammation. Frontiers in Neuroendocrinology 55:100788.
Yunus, M., A. T. Masi, J. J. Calabro, K. A. Miller, and S. L. Feigenbaum. 1981. Primary fibromyalgia (fibrositis): Clinical study of 50 patients with matched normal controls. Seminars in Arthritis and Rheumatism 11(1):151–171.
Yurov, Y. B., S. G. Vorsanova, T. Liehr, A. D. Kolotii, and I. Y. Iourov. 2014. X chromosome aneuploidy in the Alzheimer’s disease brain. Molecular Cytogenetics 7(1):20.
Zajacova, A., H. Grol-Prokopczyk, and R. Fillingim. 2022. Beyond Black vs. White: Racial/ethnic disparities in chronic pain including Hispanic, Asian, Native American, and multiracial U.S. adults. Pain 163(9):1688–1699.
Zeng, A., H. Rong, D. Pan, L. Jia, Y. Zhang, F. Zhao, and S. Peng. 2021. Discovery of genetic biomarkers for Alzheimer’s disease using adaptive convolutional neural networks ensemble and genome-wide association studies. Interdisciplinary Sciences 13(4):787–800.
Zhan, E., T. Keimig, J. Xu, E. Peterson, J. Ding, F. Wang, and X. P. Yang. 2008. Dose-dependent cardiac effect of oestrogen replacement in mice post-myocardial infarction. Experimental Physiology 93 (8):982–993.
Zhang, L., E. A. R. Losin, Y. K. Ashar, L. Koban, and T. D. Wager. 2021. Gender biases in estimation of others’ pain. The Journal of Pain 22(9):1048–1059.
Zhao, M., M. Woodward, I. Vaartjes, E. R. C. Millett, K. Klipstein-Grobusch, K. Hyun, C. Carcel, and S. A. E. Peters. 2020. Sex differences in cardiovascular medication prescription in primary care: A systematic review and meta-analysis. Journal of the American Heart Association 9(11):e014742.
Ziegler, S., and M. Altfeld. 2016. Sex differences in HIV-mediated immunopathology. Current Opinion in HIV and AIDS 11(2):209–215.
Zilberman, M. L., H. Tavares, D. C. Hodgins, and N. el-Guebaly. 2007. The impact of gender, depression, and personality on craving. Journal of Addictive Diseases 26(1):79–84.
This page intentionally left blank.

















































































































































































