Review and Evaluation of Alternative Chemical Disposal Technologies (1996)
Chapter: REFERENCES
References
Bowles, A. 1996. Personal communication from Alvin Bowles, Hazardous and Solid Waste Management Administration, Maryland Department of the Environment, to the AltTech Panel. April 8, 1996.
Bradbury, J., K. Branch, J. Heerwagen, and E. Liebow. 1994. Community Viewpoints of the Chemical Stockpile Disposal Program. Washington, D.C.: Battelle Pacific Northwest Laboratories.
Brenner, A., R. Chozick, and R.L. Irvine. 1992. Treatment of a high-strength mixed phenolic waste in an SBR. Water and Environmental Resources. 64: 128-133.
Bretherick, L. 1985. Handbook of Reactive Chemical Hazards. Third Edition. London: Butterworths. Pp. 1,100-1,124.
Brubaker, J., C. Maggio, G. Young, J. Henson, D. Grieder, L.L. Szafraniec, and W.T. Beaudry. 1995. Proceedings, ERDEC [Edgewood Research Development and Engineering Center] Scientific Conference on Chemical Defense Research, November 14, 1995, Aberdeen Proving Grounds, Maryland.
Clinton Herald. 1994. The Clinton Herald. Clinton, Indiana. January 26, 1994.
Douglass, J.J. 1996. Personal communication from J.J. Douglass, Dupont Environmental Treatment, Deepwater, New Jersey, to NRC Panel on Review and Evaluation of Alternative Chemical Technologies. August 1, 1996.
ECO LOGIC. 1996a. System Safety Program and System Hazard Analysis. The ECO LOGIC Gas-Phase Chemical Reduction Process for the Destruction of Bulk HD and VX Chemical Agent and Decontamination of Their Containers and Associated Materiel. March 27, 1996. Prepared for U.S. Army product manager for alternative technologies and approaches, Aberdeen, Maryland. Rockwood, Ontario: ECO LOGIC.
ECO LOGIC. 1996b. Report by ECO LOGIC, Inc., on Public Consultation Activities. April 4, 1996. Submitted to National Research Council, Panel on Review and Evaluation of Alternative Chemical Disposal Technologies. Rockwood, Ontario: ECO LOGIC.
Farmer, J.C., F.T. Wang, R.A. Hawley-Fedder, P.R. Lewis, L.J. Summers, and L. Foiles. 1992. Journal of the Electrochemical Society 139(3): 654-662.
Flamm, K.J., Q. Kwan, and W.B. McNulty. 1987. Chemical Agent and Munition Disposal: Summary of the U. S. Army's Experience. Report SAPEO-CDE-IS-87005. Aberdeen Proving Ground, Maryland: U.S. Army Program Manager for Chemical Demilitarization.
Gibbs, K. 1996. Personal correspondence to the panel from Kathy Gibbs, CSEPP Public Affairs Office. April 3, 1996.
Gill, D.M. 1996. Letter from D.M. Gill, business development manager, Defense Land, AEA Technology, U.S. DOD Chemical Demilitarization Project-Alternative Technology Program. April 4, 1996.
Haley, M.V. 1996. Toxicity Testing by ERDEC [Edgewood Research, Development and Engineering Center]: HD Neutralization and Biodegradation Products. October. (Informal results presented to the AltTech panel, April 1996) Aberdeen Proving Ground, Maryland: Edgewood Arsenal.
Harvey, S.P. 1994. Report to the U.S. Army Program Manager for Chemical Demilitarization, Agent Neutralization. I. Hydrolysis of Sulfur Mustard. March 1994. Aberdeen Proving Ground, Maryland: Edgewood Research, Development and Engineering Center and Geo-Centers, Inc.
Harvey, S.P., T.A. Blades, L.L. Szafraniec, W.T. Beaudry, M.V. Haley, T. Rosso, G.P. Young, J.P. Earley, and R.L. Irvine. 1996. Kinetics and toxicological parameters of HD hydrolysis and biodegradation. Presented at NATO Advanced Research Workshop on Chemical Problems Associated with Old Arsenical and Mustard Munitions, March 17-19, 1996. Lodz, Poland.
Harvey, S.P., ERDEC Research Biologist, 1995. Oral communication to G.W. Parshall. May 5, 1995.
Hosseinzadeh, K. and N. Sachdeva. 1996. Personal communication from Kaveh Hosseinzadeh and Nand Sachdeva to Harold "Butch" Dye, and Alvin Bowles,
all from Hazardous and Solid Waste Management Administration of the Maryland Department of the Environment. March 15, 1996.
Indiana, 1992a. Indiana Code, Section 13-7-8.5-3(b).
Indiana, 1992b. Indiana Code, Section 13-7-8.5-13(b).
Irvine, D.A., J.P. Earley, D.P. Cassidy, and S.P. Harvey. In press. Biodegradation of sulfur mustard hydrolysate in the sequencing batch reactor. To be published in Proceedings of the First International Water Quality Specialized Conference on Sequencing Batch Reactor Technology.
Irvine, R.L., and L.H. Ketchum. 1988. Sequencing batch reactors for biological wastewater treatment. Critical Reviews in Environmental Control. 18: 255-294.
Keane, P. 1996. Verbal communication from Patrick Keane, Illinois CSEPP, during an AltTech Panel meeting, March 13, 1996, Indianapolis, Indiana.
Koch, M., and Z. Wertejuk. 1995. Scientific advances in alternative demilitarization technologies. Presentation at the NATO Advanced Research Workshop, April 25, 1995, Warsaw, Poland.
Kosson, D.S., T.T. Kosson, and H. van der Sloot. 1993. Evaluation of Solidification/Stabilization Treatment Processes for Municipal Waste Combustion Residues. Springfield, Virgina: National Technical Information Service. (NTIS) Report PB93-229 870/AS.
Lehmani, A., P.Turq, and J.P Simonin. 1996. Oxidation kinetics of water and organic compounds by Silver (II) using a potentiometric method. Journal of the Electrochemical Society 143(6): 1860-1866.
Lovrich, J.W. 1996. Personal communication from J.W. Lovrich, coordinator for VX research, Office of the Program Manager for Chemical Demilitarization, to G. W. Parshall , NRC Panel on Review and Evaluation of Alternative Chemical Technologies. July 22, 1996.
Mackay, D., and W.Y. Shui. 1981. Critical review of Henry's Law constants for chemicals of environmental interest. Journal of Physical and Chemical Reference Data 10(4): 1175-1198.
Maryland. 1996. Annotated Code of the Public General Laws of Maryland, Environment, 7-239.3 (1987, as amended 1996).
Maryland Citizens Advisory Commission (CAC). 1994. Comments on the Recommendations for the Disposal of Chemical Agents and Munitions. Report issued February 21, 1994.
Maryland CAC. 1996. Meeting of the Maryland Citizens Advisory Commission with the NRC Panel on Review and Evaluation of Alternative Chemical Disposal Technologies, March 15, 1996, Chestertown, Maryland.
Massalski, T.B. 1986. Binary Alloy Phase Diagrams. Metals Park, Ohio: American Society for Metals.
Mentasti, E., C. Baiocchi, and J.S. Coe. 1984. Mechanical aspects of reactions involving AG (II) as an oxidant. Coordination Chemistry Reviews 54: 131-157.
Metcalf & Eddy, Inc. 1979. Wastewater Engineering: Treatment/Disposal/Reuse. 2nd ed. New York: McGraw-Hill.
Morales, M. 1996. Personal correspondence from Mickey Morales, public affairs specialist, Office of the Program Manager for Chemical Demilitarization, Alternative Technology. May 10, 1996.
M4 Environmental L.P. 1996a. News Release for Immediate Release, March 20, 1996. Oak Ridge, Tennessee.
M4 Environmental L.P. 1996b. Alternative Technologies for Chemical Demilitarization. Response to NRC Questions and Conceptual Design Baseline Updates. Submitted to National Research Council. Panel on Review and Evaluation of Alternative Chemical Disposal Technologies. Oak Ridge, Tennessee: M4 Environmental L.P. April 4, 1996.
M4 Environmental L.P. 1996c. Correspondence to Michael Clarke, study director, from T.J. Abraham, M4 Environmental L.P. May 10, 1966.
M4 Environmental L.P. 1996d. Alternative Technologies for Chemical Demilitarization: National Program Plan. Submitted to U.S. Army Chemical, Biological and Defense Command. Oak Ridge, Tennessee: M4 Environmental L.P. May 31, 1996.
M4 Environmental L.P. 1996e. Chemical Demilitarization Utilizing Catalytic Extraction Processing: Hazard Analysis. Prepared by H&R Technical Associates, Inc., Oak Ridge Tennessee. Submitted to U.S. Army Program Manager for Chemical Demilitarization. Oak Ridge, Tennessee: M4 Environmental L.P. March 1996.
M4 Environmental L.P. 1996f. Public Participation Document. Submitted to National Research Council Panel on Review and Evaluation of Alternative Chemical Disposal Technologies. Oak Ridge, Tennessee: M4 Environmental L.P. April 4, 1996.
Nagel, C.J., C.A. Chanenchuk, E.W. Wong, and R.D. Bach. 1996. Catalytic extraction processing: an elemental recycling technology. Environmental Science and Technology 30(7): 2155-2167.
Novad, J. 1996. Personal communication from J. Novad, coordinator for HD neutralization studies, U.S. Army Alternative Technology Program, Aberdeen
Proving Ground, Maryland, to G. W. Parshall, NRC Panel on Review and Evaluation of Alternative Chemical Technologies. July 23, 1996.
NRC (National Research Council). 1984. Disposal of chemical Munitions and Agents. National Research Council. Committee on Demilitarizing Chemical Munitions and Agents. Washington, D.C.: National Academy Press.
NRC. 1993. Alternative Technologies for the Destruction of Chemical Agents and Munitions. National Research Council. Committee on Alternative Chemical Demilitarization Technologies, Washington, D.C.: National Academy Press.
NRC. 1994a. Evaluation of the Johnston Atoll Chemical Agent Disposal System Operational Verification Testing, Part II. National Research Council. Committee on the Review and Evaluation of the Army Chemical Stockpile Disposal Program. Washington, D.C.: National Academy Press.
NRC. 1994b. Recommendations for the Disposal of Chemical Agents and Munitions. National Research Council. Committee on the Review and Evaluation of the Army Chemical Stockpile Disposal Program. Washington, D.C.: National Academy Press.
NRC. 1995. Evaluation of the Army's Draft Assessment Criteria to Aid in the Selection of Alternative Technologies for Chemical Demilitarization. National Research Council . Committee on Review and Evaluation of the Army Chemical Stockpile Disposal Program. Washington, D.C.: National Academy Press.
NRC. 1996. Review of Systemization of the Tooele Chemical Agent Disposal Facility. National Research Council. Committee on Review and Evaluation of the Army Chemical Stockpile Disposal Program, Washington, D.C.: National Academy Press.
Nunn, J. 1996a. Letter from John E. Nunn, III, cochair of the Maryland CAC, to the AltTech Panel. March 15, 1996.
Nunn, J. 1996b. Letter from John E. Nunn, III, cochair of the Maryland CAC, to the AltTech Panel. April 11, 1996.
O'Brien, G.J. and E.W. Teather. 1995. A dynamic model for predicting effluent concentrations for organic priority pollutants from an industrial wastewater treatment plant. Water Environment Research 67(6): 935-942.
OTA (Office of Technology Assessment). U.S. Congress. 1992. Disposal of Chemical Weapons: An Analysis of Alternatives to Incineration. Washington, D.C.: U.S. Government Printing Office.
Po, H.N., J.H. Swinehart, and T.L. Allen. 1968. Kinetics and mechanism of the oxidation of water by Silver II in concentrted nitric acid solution. Inorganic Chemistry 7: 244-249.
Rao, Y. K. 1985. Stoichiometry and Thermodynamics of Metallurgical Processes. New York: Cambridge University Press.
Ray, A. 1996. Letter from A. Ray to W. McGowan, M4 Environmental L.P. April 1, 1996.
Satterfield, C.N. 1991. Heterogeneous Catalysis in Industrial Practice. 2nd ed. New York: McGraw-Hill.
SBR Technologies. 1996. Laboratory Feasibility Studies: Biodegradation of HD Hydrolysate in Sequencing Batch Reactors. Prepared for Edgewood Research, Development, and Engineering Center, Aberdeen Proving Grounds, Maryland. Contract DAAL03-91-0034. TCN No. 94353. South Bend, Indiana: SBR Technologies, Inc.
Solarchem Environmental Systems. 1996. Report on the Rayox® design test to mineralize organic by-products in HD-caustic hydrolysate. Markham, Ontario. March 13, 1996.
Steele, D.F. 1990. Electrochemical destruction of toxic organic industrial waste platinum. Metal Review 34: 10-14.
U.S. Army. 1988. Chemical Stockpile Disposal Program Final Programmatic Environmental Impact Statement (PEIS). Aberdeen Proving Ground, Maryland: U.S. Army Program Manager for Chemical Demilitarization.
U.S. Army. 1994. U.S. Army's Alternative Demilitarization Technology Report to Congress. 11 April 94. Department of the Army. Aberdeen Proving Ground, Maryland: U.S. Army Program Manager for Chemical Demilitarization.
U.S. Army. 1995a. Assessment Criteria to Aid in Selection of Alternative Technologies for Chemical Demilitarization. 26 April 95. Department of the Army, Alternative Technology Branch. Aberdeen Proving Ground, Maryland: U.S. Army Program Manager for Chemical Demilitarization.
U.S. Army. 1995b. Concept Design Package: Stand-Alone Neutralization. 21 November 1995. Aberdeen Proving Grounds, Maryland: U.S. Army Program Manager for Chemical Demilitarization.
U.S. Army. 1996a. Alternative Technology Program Summary Status Report, Period 1 October 1995 through 31 December 1995. Aberdeen Proving Ground, Maryland: U.S. Army Program Manager for Chemical Demilitarization.
U.S. Army. 1996b. Concept Design Package for HD Neutralization Followed by Biodegradation, 4 April 1996. Product Manager for Alternative Technologies and Approaches. Aberdeen Proving Ground, Maryland: U.S. Army Program Manager for Chemical Demilitarization.
U.S. Army. 1996c. HD and VX Effluent Management Interim Summary Report. 5 April 1996. (Draft version made available to the NRC.) Aberdeen Proving Grounds, Maryland: U.S. Army Program Manager for Chemical Demilitarization.
U.S. Army. 1996d. Memorandum from Robert B. Perry, chief, Risk Management and Quality Assurance Office, Chemical Demilitarization Program, for the chief of staff, ATTN: DACS-SF. March 18. Memorandum from James A. Gibson, senior safety manager, Army Safety Office, Office of the Chief of Staff, to the program manager for chemical demilitarization. March 29, 1996.
U.S. Army. 1996e. Mustard Agent, Neutralization Followed by Biodegradation: Findings Since 4 April 1996. May 31, 1996. Aberdeen Proving Ground, Maryland: U.S. Army Program Manager for Chemical Demilitarization.
U.S. Army. 1996f. Newport Chemical Activity Concept Design Package for VX Neutralization Followed by Off-Site Biodegradation. April 4, 1996. Product Manager for Alternative Technologies and Approaches. Aberdeen Proving Grounds, Maryland: U.S. Army Program Manager for Chemical Demilitarization.
U.S. Army. 1996g. Cumulative Sample Analysis Data Report, HD Ton Container Survey Results. Dated March 14, 1996. Aberdeen Proving Grounds, Maryland: U.S. Army Program Manager for Chemical Demilitarization.
U.S. Army. 1996h. U.S. Chemical Weapons Stockpile Information Declassified, News Release, Office of Assistant Secretary of Defense (Public Affairs). Washington, D.C. January 22, 1996.
U.S. Army. 1996i. Summary Report on Laboratory- and Bench-Scale Biodegradation Testing of Neutralized HD Mustard. Prelease draft dated March 13, 1996. Aberdeen Proving Ground, Maryland: U.S. Army Program Manager for Chemical Demilitarization.
U.S. Army. 1996j. Alternative Technologies Public Information Sessions. [undated white paper summarizing public sessions from January 25-27, 1966. Aberdeen Proving Ground, Maryland: Office of the Product Manager for Alternative Technologies and Approaches.
U.S. Army. 1996k. Nerve Agent VX: Neutralization Followed by Offsite Biodegradation. Concept Design Package Supplement
: Findings since 4 April 1996. May 31, 1996. Product Manager for Alternative Technologies and Approaches. Aberdeen Proving Ground, Maryland: U.S. Army Program Manager for Chemical Demilitarization.Valenti, M. 1996. Ironing out industrial wastes. Mechanical Engineering 118 (March): 106-110.
Yang, Y-C. 1995. Chemical reactions for neutralizing chemical warfare agents. Chemistry and Industry, n.9 (May 1): 334-337.
Yang, Y-C., J.A. Baker, and J.R. Ward. 1992. Decontamination of chemical warfare agents. Chemical Reviews 92: 1729-1743.
Yang, Y-C., L.L. Szafraniec, W.T. Beaudry, D.K. Rohrbaugh, L.R. Procell, and J.B. Samuel. 1995. Proceedings, ERDEC Scientific Conference on Chemical Defense Research, November 14, 1995. Aberdeen Proving Grounds, Maryland.
Zulty, J.J., J.J. DeFrank, and S.P. Harvey. 1994. Abstract 130, Scientific Conference on Chemical and Biological Defense Research, November 16, 1994. Aberdeen Proving Ground, Maryland.
| This page in the original is blank. |
Appendix A
Commerce Business Daily Announcement
August 14, 1995
Alternative Technologies for Chemical Demilitarization
- The U.S. Army, through the Office of the Program Manager for Chemical Demilitarization is responsible for the demilitarization and disposal of chemical agents and munitions. Eight demilitarization facilities are proposed for construction and operation in the continental United States.
- The Army has demonstrated the operational effectiveness of incineration at its Johnston Atoll Chemical Agent Disposal System facility. The first demilitarization facility for the continental United States has been constructed at Tooele Army Depot and is scheduled to be operational in 1995. In the spring of 1981, the Army began testing at the Chemical Agent Munitions Disposal System (CAMDS) at Tooele, Utah. The mission of CAMDS is to test and evaluate equipment and processes proposed for chemical agent munitions demilitarization facilities.
- The National Research Council's (NRC) Committee on Review and Evaluation of the Army Chemical Stockpile Disposal Program (Stockpile Committee) was formed in 1987 at the request of the Undersecretary of the Army to monitor the disposal program and to review and comment on relevant technical issues. The Stockpile Committee is a standing committee which remains in service with rotating membership until the demilitarization program is completed.
- As a consequence of public concern over the use of incineration for chemical warfare agent disposal, the Army commissioned in November 1991, the National Research Council to conduct a study to evaluate alternatives to the reverse assembly (baseline) incineration process for use in destroying the U.S. chemical stockpile. In January 1992, the National Research Council established the Committee on Alternative Chemical Demilitarization Technologies (Alternatives Committee) to develop a comprehensive list of alternative technologies and to review their capabilities and potential as agent and munitions disposal technologies. The Defense Authorization Act for FY93 directed the Army to submit to Congress a report on potential alternative technologies.
- The NRC report on recommendations for the disposal of chemical agents and munitions was published in 1994. The NRC recommended that the Army continue the current baseline incineration program, since, at that time, no other technologies were mature enough to meet the Army's requirements. However, the NRC did recommend that the Army investigate alternative technologies based on chemical neutralization for the bulk-only sites.
- In August 1994, the Army initiated an aggressive RDT&E program to investigate, develop, and support testing of two technologies based on chemical neutralization for the destruction of mustard (agent HD) at Aberdeen Proving Ground, MD, and nerve agent VX at the Newport Chemical Activity at Newport, IN. The two alternative technologies are stand-alone chemical neutralization and neutralization followed by biodegradation. The purpose of the RDT&E program is to determine whether an alternative technology warrants pursuing a pilot-scale facility based on one or both technologies. The decision to proceed to pilot-testing will be made by the Defense Acquisition Board (DAB) in October 1996.
- The NRC also was aware that there would be ongoing development of the various research programs involving
- potential alternatives subsequent to the publication of the NRC report in 1993 on alternative technologies. Thus, the NRC recommended that the Army continue to monitor research developments.
- The Army agrees with this NRC recommendation, and the Army has been exploring developments in technologies with potential application to chemical demilitarization as part of the RDT&E program.
- The Army will be conducting a survey to determine if there are any technologies other than the two already being evaluated by the Army as part of the Alternative Technology Program which are capable, within the Chemical Stockpile Disposal Program (CSDP) schedule of meeting chemical demilitarization requirements for the HD (mustard) and VX (nerve) agents stored at the Aberdeen Proving Ground, MD, and Newport Chemical Activity, IN storage sites, respectively. This announcement requests information from industry on any alternative technology that a firm believes is mature enough to meet the needs of the Army program. The Army will conduct a preliminary 30 day screening to determine whether any of the technologies identified pursuant to this announcement warrant further review by the NRC. The Army will identify up to a maximum of three of the most promising technologies in addition to neutralization and neutralization followed by biodegradation. The evaluators will determine whether the technology meets the following screening criteria.
- Any proposed alternative technology should not resemble incineration (high temperature oxidation) or produce effluents characteristic of incineration;
- The technology must utilize processes and equipment that are developed or capable of being developed in time to meet the requirements of the Chemical Weapons Convention;
- Laboratory-scale testing must have been completed with agent or chemicals with similar properties to agent. Data must be available to provide an initial indication of performance characteristics and destruction efficiency.
- Interested firms are asked to provide information in the form of a conceptual design package within 60 days from the date of this announcement. The purpose of the conceptual design package is to demonstrate the feasibility of using an alternative set of process unit operations to conduct the total activities that are required to complete the program, and to provide a basis for its comparison with the baseline system. At a minimum, it should include the following:
- Process description. The information package should include a description of the total process, detailing how actual experience or test results have been used to project equipment performance, and how the various agent destruction, decontamination, and waste processing steps are conducted. The description should also provide an adequate basis for establishing that the process has a high probability of success, after pilot-testing, to perform the necessary agent destruction and waste disposal functions.
- Process data. Chemical and physical properties of all process materials should be provided to the extent that data is needed to design each unit operation in the overall process.
- Flow sheets, showing all proposed equipment, piping, and general control methods, including:
- Material and energy balances, projections showing all material flow rates, and energy requirements, such as heat generation and removal rates for each step of the process
- Process monitoring and control, showing all proposed process monitoring instrumentation and describing the methods used to control the process
- A description and characterization of all process waste streams.
- A description of storage facilities for all feed materials and all wastes prior to the final disposition.
- A description of facilities for packaging and handling wastes prior to off-site shipping.
- Utility requirements, including process requirements for both fuel and electricity. Also include need for backup requirements to allow for emergency shutdown of the process and related pollution control systems.
- Feed materials requirements, including both quantities and qualities of all chemicals that are required, and the need for any special feed preparation.
- Equipment lists for all major pieces of equipment for the destruction process, secondary treatment systems, and pollution control systems.
- Any data generated from agent or simulant tests or data resulting from destruction of similar chemicals by the proposed processes.
- Equipment designs, including design sketches, sizing calculations and materials of construction for all major pieces of process equipment.
- Plant layout. The design should show the layout and working space for the major pieces of equipment, plot plans for the current storage facilities, and planned means for transport of agent containers from the storage area to the destruction facilities.
- On written request, firms will be provided with information on: the baseline incineration system; the chemical stockpile disposal program schedule; and the current program for developing neutralization and neutralization followed by biodegradation. Firms may write or fax their requests to Dr. Francis W. Holm, Science Applications International Corporation, 9 Aberdeen Shopping Plaza, Aberdeen, MD 21001. Fax: (410) 273-1001.
- The NRC will review those promising alternative technologies, if any, identified by the Army as well as neutralization and neutralization-biodegradation. Concurrently, proponents of technologies identified by the Army will be asked to furnish a notional program plan including: a rough, order of magnitude estimate of the projected cost and schedule and chemical agent destruction test data. Firms must perform testing to obtain actual chemical agent test data at an Army approved surety laboratory at the firm's expense. The test data must be available to the NRC for review by 31 May 1996.
- As a note of caution, those considering participation should understand that chemical agents and munitions are significantly more toxic than many substances normally referred to as "hazardous and toxic material." Therefore, high standards of employee, public, and environmental protection are required.
- This announcement is meant to offer industry the opportunity to make the Army aware of potential alternative technologies which can meet the needs of the chemical demilitarization program. The process outlined herein will not necessarily lead to any request for proposals (RFP) or contract awards. The government does not intend to reimburse firms for the cost of providing data originally submitted pursuant to this request.
- Mr. Eric W. Braerman, Procurement Directorate, CBDCOM, is the point of contact for this announcement, (410) 671-4469.
ROBERT D. ORTON
MAJOR GENERAL, U.S. ARMY PROGRAM MANAGER FOR CHEMICAL DEMILITARIZATION
Appendix B
Input from the Public
Chapter 9 discusses the rationale for public involvement in the panel's deliberations and describes interactions with the communities neighboring the Newport and Aberdeen sites and meetings with regulators. This appendix includes samples of the letters of invitation sent to individuals and organizations in Indiana and Maryland prior to the public forums and summarizes the categories of stakeholders contacted.
The Army Program Office, located in Maryland with established communications links to stakeholders interested in the chemical demilitarization process, assisted with the notification process within Maryland. Because of the independent notification by the Army, the panel sent fewer letters to Maryland stakeholders than to Indiana stakeholders.
|
Indiana |
|
|
Letters to government officials (federal, state, and city) |
85 |
|
Letters to Indiana Citizens Advisory Commission (CAC) members |
2 |
|
Letters to other citizens, media, companies, etc. |
254 |
|
Maryland |
|
|
Letters to government officials (federal, state, and city) |
7 |
|
Letters to Maryland Citizens Advisory Commission (CAC) members |
9 |
|
Letters to other citizens, media, companies, etc. |
95 |
Letter of Invitation to the Citizens of Newport, Indiana
March 4, 1996
The National Research Council (NRC) has been asked by the Army to evaluate alternative technologies (alternatives to the Army's baseline incineration process) for the destruction of bulk chemical warfare agents stored at facilities near Aberdeen, Maryland and Newport, Indiana.
In August through October, 1995, the Army conducted an evaluation of chemical destruction processes that resulted in the selection of three technologies, plus the Army's two neutralization technologies, to be evaluated by the NRC. A Panel on Review and Evaluation of Alternative Chemical Disposal Technologies, called the AltTech Panel, was formed by the NRC. The AltTech panel will provide a report to the Army in August, 1996 that will make recommendations on whether any of the five technologies is suitable for pilot plant demonstration. In the Fall of 1996, the Army will present its recommendations to the Defense Acquisition Board (DAB) on which, if any, of the technologies should move forward to the pilot plant demonstration phase.
On Tuesday, March 12 at 7:00 p.m. representatives of the AltTech Panel will be present at North Vermillion High School, RR 1, Cayuga, Indiana, to solicit the public's views on these technologies. As Chairman of the AltTech Panel, I am writing to inform you of this information-gathering meeting. In the past, public meetings like this have added greatly to the knowledge base of other NRC committees and have ensured views of all interested parties are heard and considered.
At the meeting you will be provided an opportunity to state your views about the five technologies. The AltTech panel has been informed that the Army provided information on the alternatives during its earlier meeting, and that you have also had the opportunity to review vendor-provided information and information placed by the Army in libraries. Therefore, the panel will not spend valuable time describing the technologies again at this meeting so that the time can be applied to the most important objective, hearing your input.
I will begin the public meeting by making a short presentation that describes the NRC panel schedule and data gathering methodology. After my presentation, you may make your statements. To enable as many as possible with an opportunity to speak, you will be asked to limit your remarks to five minutes or less. If you intend to speak, please ensure you have signed in prior to the meeting. You are also encouraged to submit your statements in written form at the meeting, whether you speak or not. If you cannot attend the meeting, and you wish the AltTech panel to consider your views, please provide a written statement to the National Research Council, 2101 Constitution Avenue, N.W., Washington, DC 20418, Attn.: Mr. Michael A. Clarke, HA258, by March 31, 1996
The sole purpose of this meeting is to provide the public an opportunity to state its insights, observations, concerns, and feelings about the various technologies under consideration. You should also know that the panel will not share its assessment of the technologies with you at this meeting. That would be premature and is reserved for the panel's final report in August. Therefore, it is very important that you state only your views when you address the panel. Please do not address questions to vendor or Army personnel present. This meeting is intended to be a dialog between the NRC and the public. Conversations with the Army or vendors present should take place in other locations than the formal meeting.
Your opinions on these important local and national issues are important to us. The panel members and I look forward to hearing from you.
Sincerely,
![]()
Richard S. Magee, Chair
AltTech Panel
Letter of Invitation to the Citizens of Aberdeen, Maryland
March 5, 1996
The National Research Council (NRC) has been asked by the Army to evaluate alternative technologies (alternatives to the Army's baseline incineration process) for the destruction of bulk chemical warfare agents stored at facilities near Aberdeen, Maryland and Newport, Indiana.
In August through October, 1995, the Army conducted an evaluation of chemical destruction processes that resulted in the selection of three technologies, plus the Army's two neutralization technologies, to be evaluated by the NRC. A Panel on Review and Evaluation of Alternative Chemical Disposal Technologies, called the AltTech Panel, was formed by the NRC. The AltTech panel will provide a report to the Army in August, 1996 that will make recommendations on whether any of the five technologies is suitable for pilot plant demonstration. In the Fall of 1996, the Army will present its recommendations to the Defense Acquisition Board (DAB) on which, if any, of the technologies should move forward to the pilot plant demonstration phase.
On Friday, March 15 at 8:00 p.m. representatives of the AltTech Panel will be present at the Kent County Courthouse, County Commissioner's Room, 103 Cross Street, Chestertown, MD and on Saturday, March 16, 1996 at 10:00 a.m. at Edgewood High School, Willoughby Beach Road, Edgewood, Maryland, to solicit the public's views on these technologies. As Chairman of the AltTech Panel, I am writing to inform you of this information-gathering meeting. In the past, public meetings like this have added greatly to the knowledge base of other NRC committees and have ensured views of all interested parties are heard and considered.
At the meeting you will be provided an opportunity to state your views about the five technologies. The AltTech panel has been informed that the Army provided information on the alternatives during its earlier meeting, and that you have also had the opportunity to review vendor-provided information and information placed by the Army in libraries. Therefore, the panel will not spend valuable time describing the technologies again at this meeting so that the time can be applied to the most important objective, hearing your input.
I will begin the public meeting by making a short presentation that describes the NRC panel schedule and data gathering methodology. After my presentation, you may make your statements. To enable as many as possible with an opportunity to speak, you will be asked to limit your remarks to five minutes or less. If you intend to speak, please ensure you have signed in prior to the meeting. You are also encouraged to submit your statements in written form at the meeting, whether you speak or not. If you cannot attend the meeting, and you wish the AltTech panel to consider your views, please provide a written statement to the National Research Council, 2101 Constitution Avenue, N.W., Washington, DC 20418, Attn.: Mr. Michael A. Clarke, HA258, by March 31, 1996
The sole purpose of this meeting is to provide the public an opportunity to state its insights, observations, concerns, and feelings about the various technologies under consideration. You should also know that the panel will not share its assessment of the technologies with you at this meeting. That would be premature and is reserved for the panel's final report in August. Therefore, it is very important that you state only your views when you address the panel. Please do not address questions to vendor or Army personnel present. This meeting is intended to be a dialog between the NRC and the public. Conversations with the Army or vendors present should take place in other locations than the formal meeting.
Your opinions on these important local and national issues are important to us. The panel members and I look forward to hearing from you.
Sincerely,
![]()
Richard S. Magee, Chair
AltTech Panel
Appendix C
Meetings and Site Visits
Panel Meeting: October 11-12, 1995
Washington, D.C.
Participants. Panel chair: Richard S. Magee. Panel members: Joan B. Berkowitz, Gene H. Dyer, Frederick T. Harper, Joseph A. Heintz, David A. Hoecke, David S. Kosson, Walter G. May, Alvin H. Mushkatel, Laurance Oden, George W. Parshall, L. David Pye, William Tumas; BAST liaison: Robert A. Beaudet; NRC staff members: Bruce Braun, Michael Clarke, Jacqueline Johnson, and Deborah Randall. Briefers.
Objectives. Welcome and introduce new members; complete administrative matters; complete composition and balance discussion; discuss and develop prototype criteria checklist; organize panel into subpanel teams; perform historical review for new members; receive status briefings from applicable Army officials on call for alternative technologies; and discuss November meeting requirements.
Panel Meeting: November 20-21, 1995
Washington, D.C.
Participants. Panel members, NRC staff, and briefers
Objectives. Welcome and introduce two new members; complete administrative matters; complete composition and balance discussion; discuss and develop prototype criteria checklist; develop and approve report concept; organize panel into technology assessment teams; receive briefings from applicable Army officials on alternative technology selection process; receive briefings from technology proponent company finalists; and discuss future meetings/vendor visits.
Site Visit: January 8-9, 1996
Fall River, Massachusetts
Participants. Panel members: Gene Dyer, and Laurance Oden. BAST liaison: Robert Beaudet.
Objectives. Receive presentations and information from technology proponent company for catalytic extraction process technology and perform site tour.
Site Visit: January 8-9, 1996,
Ontario, Canada
Participants. Panel members: Walter May, Roger Staehle, and William Tumas
Objectives. Receive presentations and data from technology proponent company for gas-phase reduction technology and perform site tour.
Site Visit: January 14-16, 1996
Aberdeen/Dounreay, Scotland
Participants. Panel members: Roger Staehle, Joan Berkowitz, and Walter May. NRC staff member: Michael Clarke.
Objectives. Receive information from AEA and SubSea on the status of the electrochemical oxidation process.
Site Visit: January 18-19, 1996
Oak Ridge, Tennessee
Participants. Panel members: Gene Dyer and Laurance Oden. NRC staff member: James Zucchetto.
Objectives. Receive presentations and information from technology proponent company for catalytic extraction process technology and perform site tour.
Site Visit: January 18-19, 1996,
Edgewood, Maryland
Participants. Panel members: George Parshall and David Kosson. NRC staff members: Bruce Braun and Donald Siebenaler.
Objectives. Receive presentations and data from Army and Army contractors on neutralization technologies.
Panel Meeting: February 1-2, 1996
Irvine, California
Participants. Panel. NRC staff members: Bruce Braun, Michael Clarke, Deborah Randall, and Shirel Smith.
Objectives. Welcome and introduce three new members; complete administrative matters; complete composition and balance discussion; receive Army briefing on AltTech program status; receive briefings from team leaders on vendor assessment visits; discuss plan for public meetings and meetings with state and federal agencies; and discuss report status and future activities.
Panel Meeting: March 14-15, 1996
Washington, D.C.
Participants. Panel chair: Richard S. Magee. Panel members: Joan B. Berkowitz, Gene H. Dyer, Frederick T. Harper, Joseph A. Heintz, David A. Hoecke, David S. Kosson, Walter G. May, Alvin H. Mushkatel, Laurance Oden, George W. Parshall. BAST liaison: Robert A. Beaudet. NRC staff members: Bruce Braun, Michael Clarke, and Deborah Randall.
Objectives. Complete composition and balance discussion for three new members; receive panel assessment team briefings on chapter draft status and data requirements; discuss comparison criteria for chapter 8; assemble first full message draft of report; discuss results of Newport public and regulator meetings and plan for Aberdeen meetings; discuss AltTech program status; and discuss future report activities and goals for April meeting.
Site Visit: March 16-17, 1996
Oak Ridge, Tennessee
Participants. Panel chair: Richard S. Magee. Panel members: Joseph Heintz, David Hoecke, and Laurance Oden.
Objectives. Receive follow-up information on status of catalytic extraction process technology.
Site Visit: April 2, 1996
Ontario, Canada
Participant. Panel member: Frederick Harper
Objectives. Receive presentations data from technology proponent company for gas-phase reduction technology and perform site tour.
Site Visit: April 8-9, 1996
Edgewood, Maryland
Participants. Panel members: David Kosson and George Parshall
Objectives. Receive follow-up information from Army and Army contractors on the status of neutralization technologies.
Writing Session: April 11-12, 1996
Washington, D.C.
Participants. Panel members: Alvin Mushkatel and Richard Magee. NRC staff member: Michael Clarke.
Objectives. Organize report and draft community and regulator chapter of report.
Panel Meeting: April 18-20, 1996
Washington, D.C.
Participants. Panel chair: Richard S. Magee. Panel members: Joan B. Berkowitz, Gene H. Dyer, Frederick T. Harper, Joseph A. Heintz, David A. Hoecke, David S. Kosson, Walter G. May, Alvin H. Mushkatel, George W. Parshall, L. David Pye, William Tumas. BAST liaison: Robert A. Beaudet. NRC staff members: Bruce Braun, Michael Clarke, and Deborah Randall.
Objectives. Assemble first full message draft of report; develop a strategy for the rapid development of a concurrence draft; discuss AltTech program status, including final data acquisition and surety testing; review preliminary hazard and operability report status; settle panel indemnity issue; and set goals for the May meeting.
Site Visit: April 26, 1996
Oak Ridge, Tennessee
Participants. Panel member: Frederick Harper
Objectives. Tour facilities and orient risk assessment panel member; discuss risk issues with the technology proponent company.
Site Visit: May 5-8, 1996
London, England
Participants. Panel members: Joan Berkowitz and Walter May
Objectives. Evaluate and assess the electrochemical reduction alternative technology as a candidate for pilot-
plant demonstration by the U.S. Department of the Army for destruction of chemical agents; receive presentations from the technology proponent company on that technology and perform site tour.
Panel Meeting: May 15-17, 1996
Washington, D.C.
Participants. Panel chair: Richard S. Magee. Panel members: Joan B. Berkowitz, Gene H. Dyer, Frederick T. Harper, David A. Hoecke, David S. Kosson, Walter G. May, Alvin H. Mushkatel, Laurance Oden, William Tumas. BAST liaison: Robert A. Beaudet. NRC staff members: Bruce Braun, Michael Clarke, and Deborah Randall. Technical writer/consultant: Robert Katt.
Objectives. Assemble and sign off on concurrence draft of report; discuss AltTech program status and methodology for including surety testing data; settle panel indemnity issue; and discuss milestones leading to report review and publication.
Appendix E
Electrochemical Oxidation
Water Balance Issues
When HD is treated in the Silver II process, 75 kg·mols will be decomposed during the course of a campaign as the source of oxygen for agent oxidation. An additional amount of water will be lost by the parasitic reaction in which water is decomposed and 02 gas evolves. Approximately 176 kg·mols of water will be carried from the anode compartment to the cathode compartment by electrical diffusion of hydrated hydrogen ions. The total of these water losses, more than 251 kg·mols, should be compared to the initial water content of the anode compartment of 2.5 m3 or 139 kg·mols. Part of the loss, as yet unquantified, is made up by spontaneous osmotic diffusion from the cathode compartment back to the anode department, induced by the large difference in acid concentration between the two. (The anolyte is maintained at 8 molar in nitric acid, the catholyte at 4 molar.)
When VX is treated, the water losses are about 116 kg·mols from agent oxidation and 307 kg·mols from the transport of hydrated hydrogen ions. Total losses therefore exceed 423 kg·mols during the course of a VX campaign, compared with the initial water content in the anode compartment of 139 kg·mols. As in the HD case, there is an as yet undetermined osmotic flow of water back from the cathode compartment to the anode compartment.
Mass Balance Data for Silver II Process
Tables E-1 and E-2 provide an elemental analysis of the mass balance data provided by the TPC for the Silver II process for treating VX and HD, respectively.
TABLE E-1 Elemental Breakdown of Mass Balances for VX Destruction
|
|
Metric Tons |
|
||||||
|
Input |
Total |
C |
H |
S |
N |
P |
O |
Na |
|
VX |
2 |
0.99 |
0.19 |
0.24 |
0.1 |
0.23 |
0.24 |
|
|
HNO3 |
0.7 |
|
0.01 |
|
0.16 |
|
0.53 |
|
|
H2O |
0.3 |
|
0.03 |
|
|
|
0.27 |
|
|
H2O2 |
1.9 |
|
0.11 |
|
|
|
1.78 |
|
|
H2O |
3.6 |
|
0.4 |
|
|
|
3.2 |
|
|
NaOH |
1.8 |
|
0.05 |
|
|
|
0.72 |
1.04 |
|
H2O |
0.1 |
|
0.01 |
|
|
|
0.09 |
|
|
O2 |
4.9 |
|
|
|
|
|
4.9 |
|
|
N2 |
0.5 |
|
|
|
0.5 |
|
|
|
|
Total In |
15.8 |
1.0 |
0.8 |
0.2 |
0.8 |
0.2 |
11.7 |
1.0 |
|
Output |
Total |
C |
H |
S |
N |
P |
O |
Na |
|
CO2 |
3.8 |
1.04 |
|
|
|
|
2.76 |
|
|
O2 |
0.1 |
|
|
|
|
|
0.1 |
|
|
N2 |
0.5 |
|
|
|
0.5 |
|
|
|
|
NOx |
0.004 |
|
|
|
|
|
|
|
|
HNO3 |
1.12 |
|
0.02 |
|
0.25 |
|
0.85 |
|
|
H2O |
3.9 |
|
0.43 |
|
|
|
3.47 |
|
|
H2O |
3.6 |
|
0.42 |
|
|
|
3.2 |
|
|
NaNO3 |
0.6 |
|
|
|
0.1 |
|
0.34 |
0.16 |
|
Na2SO4 |
1.07 |
|
|
0.24 |
|
|
0.48 |
0.35 |
|
Na3PO4 |
1.23 |
|
|
|
|
0.23 |
0.48 |
0.52 |
|
Total Out |
15.9 |
1.0 |
0.9 |
0.2 |
0.9 |
0.2 |
11.7 |
10 |
TABLE E-2 Elemental Breakdown of Mass Balances for HD Destruction
|
|
Metric Tons |
|
|||||||
|
Inputs |
Total |
C |
H |
S |
Cl |
N |
O |
Na |
|
|
HD (mustard) |
2 |
0.6 |
0.1 |
0.4 |
0.89 |
|
|
|
|
|
HNO3 |
0.4 |
|
0.01 |
|
|
0.09 |
0.3 |
|
|
|
H2O |
0.2 |
|
0.02 |
|
|
|
0.18 |
|
|
|
H2O2 |
1.1 |
|
0.06 |
|
|
|
1.04 |
|
|
|
H2O |
2 |
|
0.22 |
|
|
|
1.78 |
|
|
|
NaOH |
2 |
|
0.05 |
|
|
|
0.8 |
1.15 |
|
|
H2O |
0.1 |
|
0.01 |
|
|
|
0.09 |
|
|
|
O2 |
2.8 |
|
|
|
|
|
2.8 |
|
|
|
N2 |
0.3 |
|
|
|
|
0.3 |
|
|
|
|
Total In |
10.9 |
0.6 |
0.5 |
0.4 |
0.9 |
0.4 |
7.0 |
1.2 |
|
|
Outputs |
Total |
C |
H |
S |
Cl |
N |
O |
Na |
|
|
CO2 |
2.2 |
0.6 |
|
|
|
|
1.6 |
|
|
|
O2 |
0.1 |
|
|
|
|
|
0.1 |
|
|
|
N2 |
0.3 |
|
|
|
|
0.3 |
|
|
|
|
NOx |
0.002 |
|
|
|
|
|
|
|
|
|
HNO3 |
0.63 |
|
0.01 |
|
|
0.14 |
0.48 |
|
|
|
H2O |
2.2 |
|
0.24 |
|
|
|
1.96 |
|
|
|
Na2SO4 |
1.8 |
|
|
0.4 |
|
|
0.81 |
0.58 |
|
|
NaCl |
1.5 |
|
|
|
0.9 |
|
|
0.59 |
|
|
H2O |
2.2 |
|
0.24 |
|
|
|
1.96 |
|
|
|
Total Out |
10.9 |
0.6 |
0.5 |
0.4 |
0.9 |
0.4 |
6.9 |
1.2 |
|
Appendix F
Gas-Phase Reduction
Thermodynamic Calculations
The ECO LOGIC process is described as a gas-phase chemical reduction process in which waste materials react with hydrogen and steam at high temperature. The reaction conditions are very different from the reaction conditions in industrial hydrogenation processes, which are usually carried out at much higher hydrogen pressure and lower temperature than the ECO LOGIC conditions and require a catalyst because of the lower temperature. In this appendix, the AltTech Panel has used thermodynamic data to examine likely chemical reactions and reaction products that will result from processing agent.
Data on free energy of formation were used for these calculations. The data were taken primarily from the JANAF Thermochemical Tables (JANAF, 1985); a few of the data are from Perry's Chemical Engineers Handbook (Perry et al., 1984). Data at 1100 K were used as representative of reactor conditions; data at 298 K were used as representative of quenched reactor products.
Feed material in the main reactor is at a high enough temperature for cracking (breakup of the carbon chain into smaller fragments) to occur rapidly. Molecular fragments can then react with the hydrogen and steam in the reactor environment. The end products indicated by thermodynamic considerations are discussed below for carbon and for each of the heteroatoms.
Carbon
Methane is the only hydrocarbon with significant thermodynamic stability at 1100 K in the presence of hydrogen as illustrated by the following possible product reactions:
|
|
|
Equilibrium Constant |
|
Ethylene |
C2H4 + 2H2 = 2CH4 |
K1100 K = 1417 |
|
Acetylene |
C2H2 + 3H2 = 2CH4 |
K1100 K = 4.2 x 1012 |
|
Benzene |
C6H6 + 9H2 = 6CH4 |
K1123 K = 2 x 1017 |
The panel concludes that at this, reaction temperature and with this hydrogen content in the main reactor, these hydrocarbons would react almost completely to form methane. Methane itself, however, is not expected at high concentration; reaction with hydrogen should result in solid carbon and only low concentration of methane.
C + 2H2 = CH4 K1100 K = 0.0356
With a hydrogen content of 70 percent in the product gas, the equilibrium methane concentration is calculated to be only 1.7 percent. This does not conform to experimental observation, however. The observed methane content, which is reported to be as high as 15 percent, probably represents a nonequilibrium, rate-controlled product. A possible alternative explanation is that carbon formed in high temperature reactions sometimes has a higher free energy than graphite (so-called "Dent" carbon). At 500°C, the free energy of this carbon form may be 15 KJ above the free energy of graphite. This difference would lead to a larger equilibrium constant for the reaction (K1100 K = 0.1859) and a possible equilibrium methane content of 9 percent.
This calculation suggests that high methane content is probably a result of the reaction sequence during the decomposition process. It also suggests that solid carbon should be expected as a product. The TPC assumes that 10 percent of the carbon in the feed will show up as solid elemental carbon in the reactor effluent gas. Precursors to the solid carbon, such as polycyclic aromatics, would then also be expected.
Steam can also react with carbon (and methane), and in fact thermodynamic equilibrium would result in complete conversion.
C + H2O = CO + H2 K1100 K = 11.16
However, reaction rates with industrial carbon at 1100 K are very slow and are inhibited by the presence of hydrogen (Gadsby et al., 1946; May et al., 1958).
Reaction with carbon from a decomposing hydrocarbon could be faster, however, and significant CO would be expected.
Much of the CO produced would react via the water gas shift reaction to form CO2.
CO + H2O = CO2 + H2 K1100 K = 0.988
The rate for this reaction is high enough to approach equilibrium.
Heteroatoms
Chlorine
Chlorine (in mustard) should react almost completely to HCl.
H2 + Cl2 = 2HCl K1100 K = 66000
Sulfur
Sulfur should go primarily to H2S in the reactor.
H2 + S = H2S K1100 K = 51.9
A very small amount of H2S could react with steam.
H2S + H2O = 3H2 + SO2 K1100 K = 7.6 x 10-7
A small amount of sulfur should be expected in the quench of the product stream-probably in the HCl product solution.
Phosphorus
Phosphine and oxides of P(II) and P(IV) do not appear to be very stable relative to elemental phosphorus at either reactor (1100 K) or quench (298 K) conditions; only very small concentrations would therefore be expected. For example:
|
Phosphine: |
P + 1.5H2 = PH3 |
|
|
K1100 K = 5.2 × 10-4 |
|
|
K298 K = 3.8 × 10-6 |
|
P-Oxides: |
P + H2O = PO + H2 |
|
|
K1100 K = 1 × 10-4 |
|
|
K298 K = 1.4 × 10-32 |
|
|
P + 2H2O = PO2 + 2H2 |
|
|
K1100 K = 0.0016 |
|
|
K298 K = 2 × 10-25 |
Higher-valence oxides, P4O6 in particular, are much more stable under both reactor and quench conditions.
4P + 6H2O = P4O6 + 6H2
K1100 K= 2.8 × 1028
K298 K >10100
The trivalent oxide appears to be the most stable oxide under the reducing conditions of the process. It is considerably more stable than the divalent and tetravalent oxides, as well as the pentavalent oxide, at both reactor and quench conditions.
P4O6 + 4H2O = P4O10 + 4H2
K1100 K = 6.7 × 10-22
K298 K = 1.5 × 10-58
Under oxidizing conditions, P4O10 would be the stable species.
P4O6 + 2O2 = P4O10
K298 K >10100
It appears likely that the phosphorus species produced in the reactor will be the oxide of trivalent phosphorus, P4O6.
A number of phosphorus acids might form in solution when the reactor vapor is quenched. The stable one appears to be the orthophosphorous acid (Moeller, 1952).
P4O6 + 6H2O = 4H3PO3
This form is unstable in an oxidizing atmosphere and would presumably convert to the pentavalent orthophosphoric acid, H3PO4. The rate of conversion to the higher oxide is not known.
Nitrogen
Nitrogen would be expected in the form of molecular nitrogen, ammonia, and possibly some N-oxide species. An interesting possibility that will need further examination is the potential to produce hydrogen cyanide (HCN). At reactor conditions, this material would be expected at parts per million concentration, though at virtually zero concentration at room temperature.
1/2H2 + 1/2N2 + C = HCN
K1100 K = 2.2 × 10-5
K298 K = 14 × 10-22
The rates of reaction are unknown. Because nitrogen is associated with carbon (in VX), HCN would probably be formed in the reactor. Whether it will be at its equilibrium level and whether it will persist (at above equilibrium level) during the quench are questions that will need evaluation.
Material Balances
Two sets of material and energy balances were submitted by the TPC, the first on January 30, 1996, the second on April 4, 1996. The panel has examined the balances for HD. The two balances differed in the feed rate of HD: 5.0 liter/min. for the first, 2.736 liter/min. for the second. The numbers that follow are taken from the second balance unless otherwise stated. The feed rate of 2.736 liter/min. corresponds to a destruction rate of 5 metric tons per day (5.5 English tons per day).
The feed to the reactor consists of four streams: feed HD; gas from the steam reformer; waste steam; and waste water. A stream from the SBV (sequencing batch vaporizer) would also go to the reactor when the SBV is operating. Some product gas from the product gas blower might also be recycled directly back to the reactor, bypassing the catalytic reformer.
The largest gas stream is the reformer gas, which constitutes approximately 85 percent of the total gram-mols of feed. This gas is at a high temperature (775°C) and has a large H2 content.
|
Reformer Gas |
g-mols/min. |
vol% |
|
H2 |
755 |
74.0 |
|
CH4 |
15.3 |
1.5 |
|
CO |
35.3 |
3.5 |
|
CO2 |
55.3 |
5.4 |
|
H2O |
159.8 |
15.7 |
Table F-1 shows flow rates and compositions into and out of the reactor. The hydrogen content of the product gas is kept high, above 55 percent (wet basis) in this case. There may also be trace quantities (parts per million) of other materials not shown in the product gas analysis above are possible (SO2, for example).
The TPC assumes that 10 percent of the carbon in the HD feed will be solid carbon in the product. Most of the TPC's experience has been with aromatic feed stocks, such as PCBs (polychlorinated biphenyls), which would presumably yield relatively large carbon residues. The carbon residue from HD (or VX) might be lower than 10 percent.
The methane content of the product gas is well above the thermodynamic equilibrium value. It may simply represent a nonequilibrium product limited by the reaction rate. The methane presumably forms from CH2 radicals (see section on Thermodynamic Calculations).
Most of the gas feed to the reactor is at high temperature; the reformer gas, which is 85 percent of the total, is at 775°C, and the direct recirculation gas is heated to
TABLE F-1 Material Flows to and from GPCR Reactor
|
|
Feeds to Reactor |
|
Products from Reactor |
|
|
Material Species |
g-mols/min. |
vol% |
g-mols/min. |
vol% |
|
H2Oa |
270.2 |
23.4 |
239.0 |
20.9 |
|
H2 |
755 |
65.4 |
654.0 |
57.2 |
|
CH4 |
15.3 |
1.3 |
77.8 |
6.8 |
|
Hydrocarbon |
0.8 |
.07 |
— |
— |
|
HD (mustard) |
21.84 |
1.89 |
— |
— |
|
CO |
35.3 |
3.06 |
43.5 |
3.8 |
|
CO2 |
55.3 |
4.79 |
62.9 |
5.5 |
|
H2S |
— |
— |
21.8 |
1.9 |
|
HCl |
— |
— |
43.6 |
3.8 |
|
Solid Carbon |
— |
— |
8.7 |
— |
|
a The hydrogen and oxygen in feed and product do not balance exactly. |
||||
TABLE F-2 Material Balance for HD in the ECO LOGIC Process
|
Material Ina |
|
Material Out |
|
|
|
Material Species |
g-atoms/min. |
Material Species |
g-atoms/min. |
|
|
Carbon from HD |
87.35 |
Solid carbon |
8.7 |
|
|
|
|
CO2 in HCl solution |
5.5 |
|
|
|
|
CO2 in MEA offgas |
51.2 |
|
|
|
|
CH4/CO/CO2 in gas to burner |
22.0 |
|
|
|
|
Total carbon out |
87.4 |
|
|
Hydrogen |
|
Hydrogen |
|
|
|
from HD |
174.7 |
Gas to Burner |
|
|
|
from H2O |
|
H2 |
235.6 |
|
|
Reactor |
62.4 |
H in CH4 |
53.7 |
|
|
Catalytic Reformer |
-130 |
HCl solution |
43.45 |
|
|
|
|
H2S from MEA |
43.45 |
|
|
Total H2 in |
367.1 |
Total H2 out |
366.2 |
|
|
Sulfur from HD |
21.84 |
H2S in MEA offgas |
21.84 |
|
|
Chlorine in HD |
43.68 |
HCl in quench solution |
43.68 |
|
|
a Based on HD feed rate of 21.84 g-mols/min. |
||||
600°C. The electric heaters in the reactor then supply energy to raise the gas mixture to between 850 and 900°C. The reaction itself is a combination of hydrocracking (to produce methane), which is exothermic, and steam reforming (to produce CO), which is endothermic. Overall the reaction appears to be slightly exothermic (about 1,400 kJ/kg of HD processed, equivalent to less than 10 percent of the heat of combustion of HD).
The product gas from the reactor is quenched with water to produce an HCl solution of moderate concentration together with suspended carbon. The quench will also dissolve some of the H2S (and the possible low concentration [ppm range] of SO2), as well as some CO2. The suspended carbon must be filtered out before disposal of the HCl solution. The TPC has estimated that the quench will remove 43.8 g-mols/min. of HCl and 5.5 g-mols/min. of CO2. (On the assumption that the HCl solution will be fairly concentrated, perhaps 30 percent, the CO2 removal rate appears too high.)
The H2S and most of the CO2 will be recovered in the methanolamine scrubber. The product gas from this scrubber will be:
|
H2S |
21.8 g-mols/min.; 29.9 vol% |
|
CO2 |
51.2 g-mols/min.; 70.1 vol% |
The scrubbed gas will have the following composition (dry basis).
|
|
Composition |
|
|
Gas |
g-mols/min |
vol% |
|
H2 |
654.0 |
83.7 |
|
CH4 |
77.8 |
10.0 |
|
CO |
43.5 |
5.6 |
|
CO2 |
6.2 |
0.8 |
In the material balances submitted by the TPC, no aromatic hydrocarbons are shown for the product gas, and the submitted design makes no provision for hydrocarbon scrubbing. The TPC does recognize that some high-molecular-weight hydrocarbon may be present (a precursor to solid carbon) and that a scrubber for removal may be necessary.
Part of the scrubbed product gas is recycled (mostly via the catalytic reformer); part is burned to supply steam. Overall, the material balance indicates that approximately 17 percent of the scrubbed gas will be burned. The products of HD destruction then show up in the streams shown in Table F-2. The scrubbed product
gas, which consists mainly of hydrogen (83 percent) and methane (10 percent), should burn cleanly, that is, with negligible products of incomplete combustion.
References
Gadsby, J., C.N. Hinshelwood, and K.W. Sykes. 1946. Kinetics of the Reactions of the Steam-Carbon System. Proceedings of the Royal Society, London, Series A. 187: 129-151.
JANAF. 1985. JANAF Thermochemical Tables. 3rd Ed. Journal of Physical and Chemical Reference Data 14, Suppl.1.
May, W.G., R.H. Mueller, and S.B. Sweetser. 1958, Carbon steam reaction kinetics from pilot plant data. Industrial and Engineering Chemistry 50: 1289-1296.
Moeller, T. 1952. Inorganic Chemistry. New York: John Wiley and Sons, Inc.
Perry; R. H., D.W. Green, and J.O. Maloney (eds). 1984. Perry's Chemical Engineers Handbook. 6th Ed. New York: McGraw-Hill.
Appendix G
Mass Balances for HD Neutralization
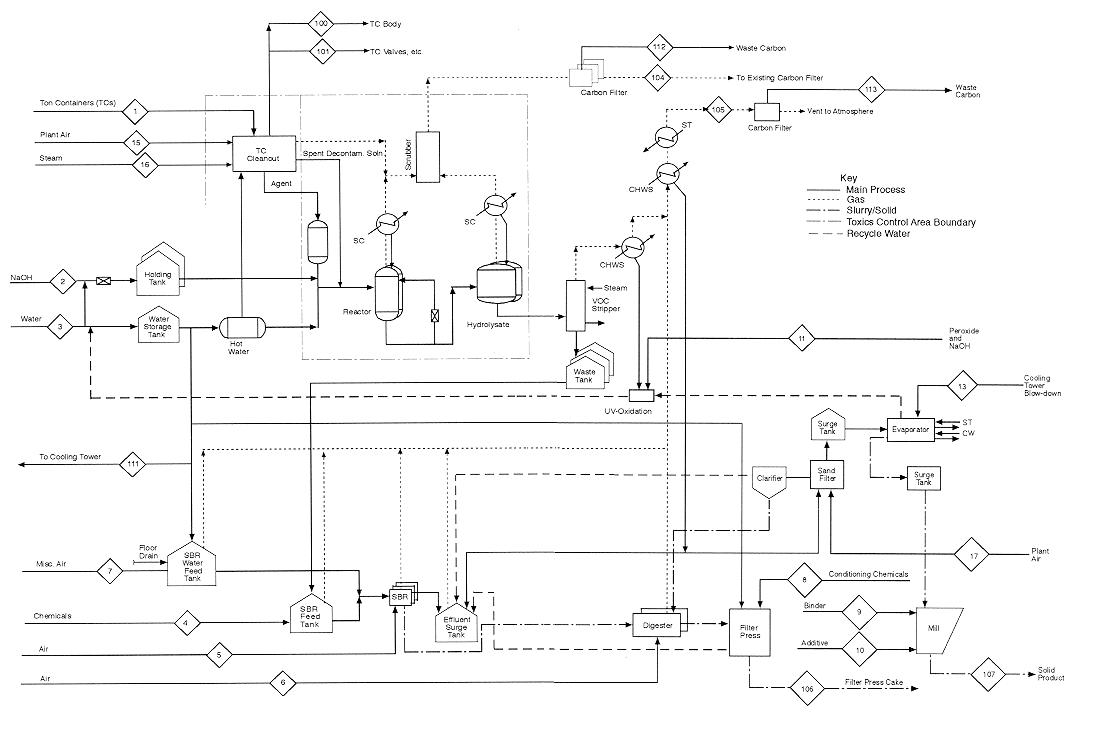
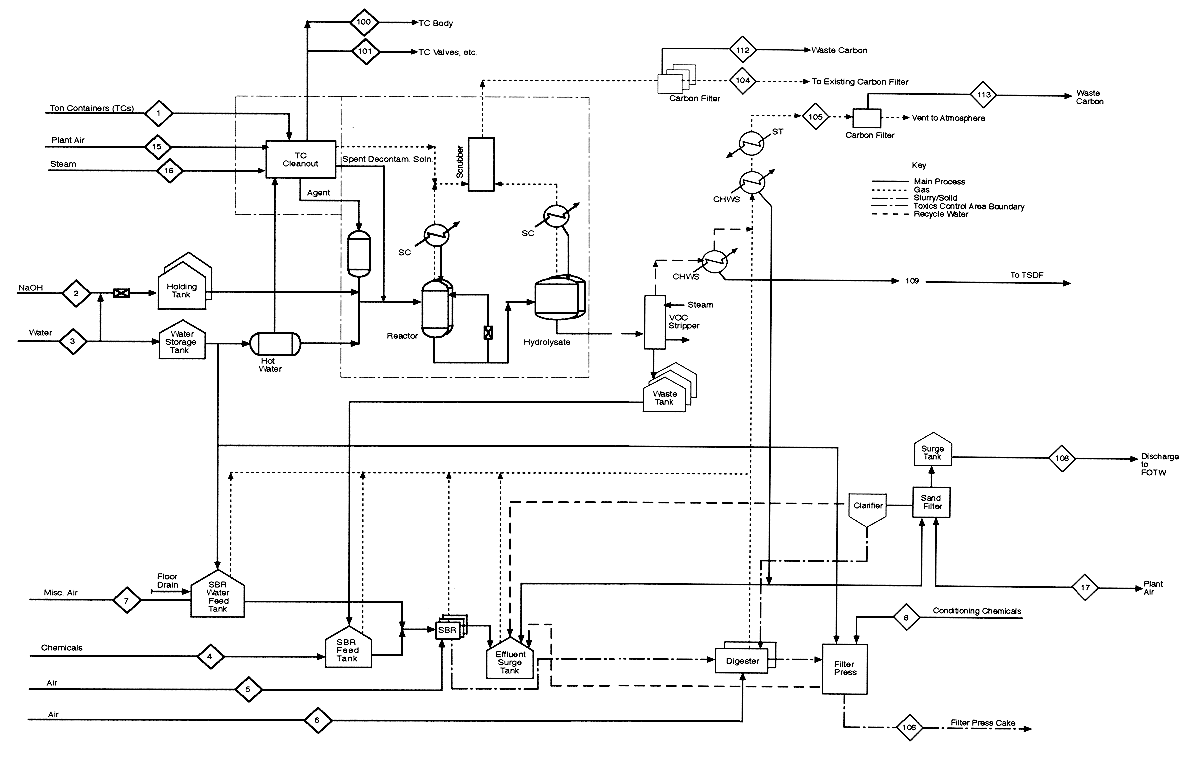
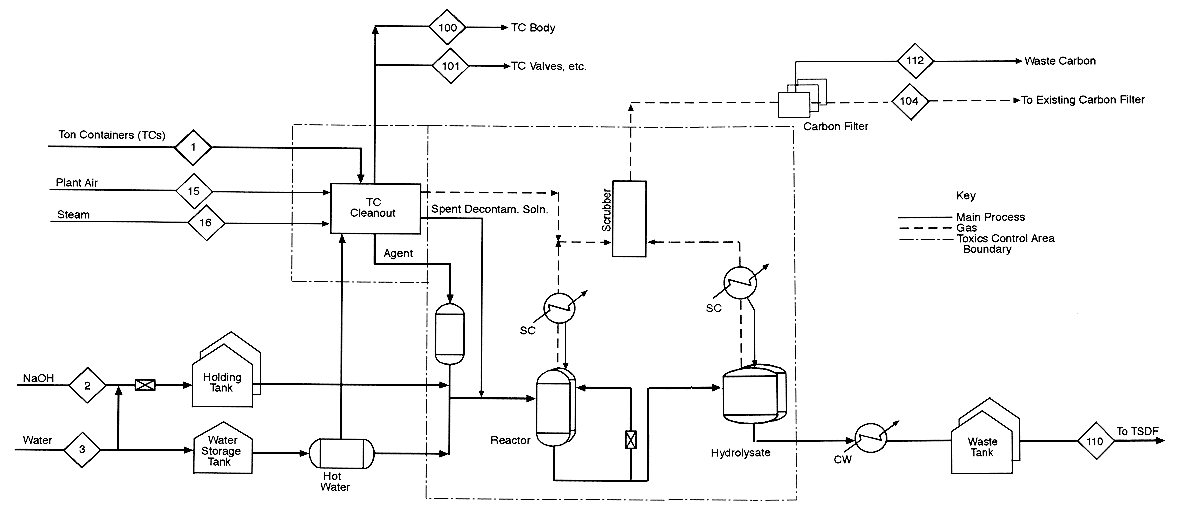
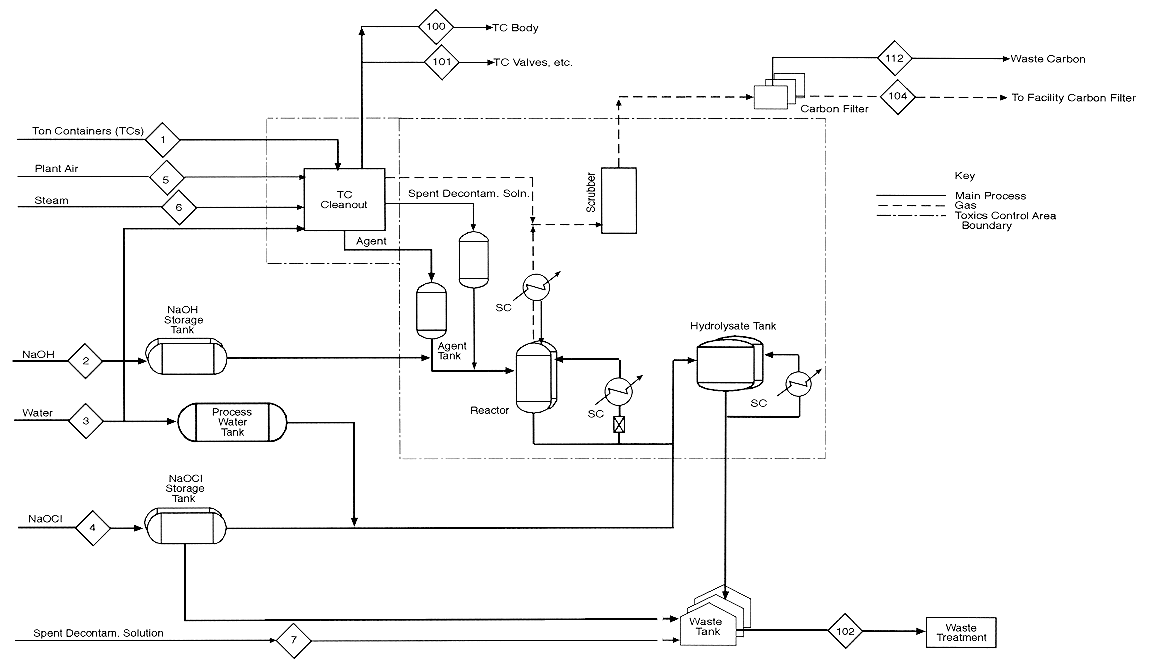
This appendix contains mass balance matrices for the four HD neutralization configurations. For each configuration, there is a matrix for process inputs and one for process outputs. The stream numbers in the column headings are keyed to the numbered input and output streams shown in the process diagrams preceding each set of matrices. Each process diagram consists of two sheets: sheet 1 is the left side of a full diagram, sheet 2 is the right side. Input streams are numbered from 1; output streams are numbered from 100.
The diagrams and the mass balance data are derived either from the April 4, 1996, design package submitted by the Army Alternative Technology Program or from more recent data.
TABLE G-1 Process Inputs for HD Neutralization, Configuration 1
|
|
Neutralization |
Biodegradation |
Solidify & Stabilize |
Photochem. Oxidation |
Ton Container Cleanout |
Water Recycle |
|
|||||||||
|
Stream Number (see figures) |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
11 |
15 |
16 |
13 |
17 |
|
|
Description |
TCs with Agent |
NaOH (aq.) |
Water |
Nutrients and Buffer |
Air |
Air |
Air |
Conditioning Chemicals |
Binder |
Additives |
|
Air |
Steam |
Cooling Tower |
Sand Filter Air |
Total Inputs |
|
Process Conditions |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total flow, kg/1,000 kg |
1,020 |
1,002 |
0 |
1,262 |
62,296 |
6,671 |
2,409 |
62 |
2,736 |
547 |
171 |
1,648 |
227 |
34,166 |
31 |
114,248 |
|
Pressure, psig |
|
15 |
60 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
30 |
80 |
125 |
0 |
25 |
|
|
Temperature, ºF |
70 |
70 |
47 |
70 |
70 |
70 |
70 |
70 |
70 |
70 |
70 |
100 |
351 |
75 |
110 |
|
|
Physical state, solid (S), liquid (L), or gas (G) |
S, L & G |
L |
L |
S & L |
G |
G |
G |
L |
S |
S |
L |
G |
G |
L |
G |
|
|
Feeds and Components |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
HD (C4H8Cl2S), kg/1,000 kg |
904 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
904 |
|
Sodium hydroxide (NaOH), kg/1,000 kg |
|
501 |
|
|
|
|
|
|
|
|
11 |
|
|
|
|
512 |
|
Water (H2O), kg/1,000 kg |
|
501 |
0 |
174 |
921 |
105 |
36 |
40 |
|
|
106 |
|
227 |
34,166 |
|
36,276 |
|
Sulfur-containing impurities, kg/1,000 kg |
82 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
82 |
|
Chlorinated aliphatic hydrocarbons, kg/1,000 kg |
7 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
7 |
|
Process Chemicals, Other |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
NaHCO3, kg/1,000 kg |
|
|
|
1,008 |
|
|
|
|
|
|
|
|
|
|
|
1,008 |
|
KNO3, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
0 |
|
KCl, kg/1,000 kg |
|
|
|
15 |
|
|
|
|
|
|
|
|
|
|
|
15 |
|
Na2SO4, kg/1000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
0 |
|
NH3, kg/1,000 kg |
|
|
|
37 |
|
|
|
|
|
|
|
|
|
|
|
37 |
|
Na3PO4, kg/1000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
0 |
|
Wolin salts, kg/1,000 kg |
|
|
|
8 |
|
|
|
|
|
|
|
|
|
|
|
8 |
|
H3PO4, kg/1,000 kg |
|
|
|
20 |
|
|
|
|
|
|
|
|
|
|
|
20 |
|
H2O2, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
54 |
|
|
|
|
54 |
|
Organics, kg/1000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
0 |
|
|
Neutralization |
|
Biodegradation |
Solidify & Stabilize |
Photochem. Oxidation |
Ton Container Cleanout |
Water Recycle |
|
||||||||
|
Stream Number (see figures) |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
11 |
15 |
16 |
13 |
17 |
|
|
Description |
TCs with Agent |
NaOH (aq.) |
Water |
Nutrients and Buffer |
Air |
Air |
Air |
Conditioning Chemicals |
Binder |
Additives |
|
Air |
Steam |
Cooling Tower |
Sand Filter Air |
Total Inputs |
|
Polymer, kg/1,000 kg |
|
|
|
|
|
|
|
4 |
|
|
|
|
|
|
|
4 |
|
Fe+2, Fe+3, kg/1,000 kg |
10 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10 |
|
Cl-, kg/1,000 kg |
17 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
17 |
|
Activated carbon (estim.), kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
0 |
|
Biosolids, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
0 |
|
Fe(OH)3, kg/1000 kg |
|
|
|
|
|
|
|
18 |
|
|
|
|
|
|
|
18 |
|
Binder compound (TBD), kg/1,000 kg |
|
|
|
|
|
|
|
|
2,736 |
|
|
|
|
|
|
2,736 |
|
Cement additive (TBD), kg/1,000 kg |
|
|
|
|
|
|
|
|
|
547 |
|
|
|
|
|
547 |
|
Gases |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O2, kg/1,000 kg |
|
|
|
|
14,208 |
1,520 |
549 |
|
|
|
|
380 |
|
|
7 |
16,664 |
|
N2, kg/1000 kg |
|
|
|
|
44,239 |
4,733 |
1,711 |
|
|
|
|
1,189 |
|
|
23 |
51,895 |
|
CO2, kg/1,000 kg |
|
|
|
|
2,928 |
313 |
113 |
|
|
|
|
78 |
|
|
2 |
3,434 |
|
TC Shells |
1.23 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1.23 |
|
TC Valves |
2.49 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2.49 |
|
TC Plugs |
7.45 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
7.45 |
|
TC Cuttings (3 lb/TC, estim.), kg/1,000 kg |
1.69 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1.69 |
TABLE G-2 Process Outputs for HD Neutralization, Configuration 1
|
|
Ton Container Clean-Out and Neutralization |
Biodegradation Process |
|
Solidify & Stabilize |
Water Recycle |
|
||||
|
Stream Number (see figures) |
100 |
101 |
104 |
112 |
105 |
106 |
113 |
107 |
111 |
|
|
Description |
Ton Container Bodies |
Valves Plugs, etc. |
Vent Gas |
Activated Carbon |
Vent Gas |
Biomass (from filter) |
Activated Carbon |
Solid Product |
To Cooling Tower |
Total Outputs |
|
Process Conditions |
|
|
|
|
|
|
|
|
|
|
|
Total flow, kg/1,000 kg |
|
|
1,651 |
2 |
71,339 |
9724 |
8,754 |
31,478 |
114,200 |
|
|
Pressure, psig |
|
|
-5 |
0 |
0 |
0 |
0 |
|
5 |
|
|
Temperature, °F |
70 |
70 |
110 |
|
100 |
90 |
|
70 |
125 |
|
|
Physical state, solid (S), liquid (L), or gas (G) |
S |
S |
G |
S |
G |
|
S |
S |
L |
|
|
Feeds and Components |
|
|
|
|
|
|
|
|
|
|
|
HD (C4H8Cl2S), kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
Sodium hydroxide (NaOH), kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
Water (H2O), kg/1,000 kg |
|
|
|
|
811 |
|
|
|
31,478 |
32,289 |
|
Sulfur-containing impurities, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
Chorinated aliphatic hydrocarbons, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
Process Residuals, Other |
|
|
|
|
|
|
|
|
|
|
|
NaHCO3, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
NaCl, kg/1,000 kg |
|
|
|
|
|
7 |
|
740 |
|
747 |
|
KNO3, kg/1,000 kg |
|
|
|
|
|
|
|
2 |
|
2 |
|
KCl, kg/1,000 kg |
|
|
|
|
|
|
|
7 |
|
7 |
|
Na2SO4, kg/1,000 kg |
|
|
|
|
|
7 |
|
792 |
|
799 |
|
NH3, kg/1,000 kg |
|
|
|
|
|
|
|
1 |
|
1 |
|
NaNO3, kg/1,000 kg |
|
|
|
|
|
1 |
|
28 |
|
29 |
|
NaNO2, kg/1,000 kg |
|
|
|
|
|
|
|
8 |
|
8 |
|
Na3PO4, kg/1,000 kg |
|
|
|
|
|
|
|
7 |
|
7 |
|
Wolin salts, kg/1,000 kg |
|
|
|
|
|
|
|
8 |
|
8 |
|
H3PO4, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
|
Ton Container Clean-Out and Neutralization |
Biodegradation Process |
|
Solidify & Stabilize |
Water Recycle |
|
||||
|
Stream Number (see figures) |
100 |
101 |
104 |
112 |
105 |
106 |
113 |
107 |
111 |
|
|
Description |
Ton Container Bodies |
Valves Plugs, etc. |
Vent Gas |
Activated Carbon |
Vent Gas |
Biomass (from filter) |
Activated Carbon |
Solid Product |
To Cooling Tower |
Total Outputs |
|
H2O2, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
Organics, kg/1,000 kg |
|
|
|
|
|
1 |
|
66 |
|
67 |
|
Polymer, kg/1,000 kg |
|
|
|
|
|
4 |
|
|
|
4 |
|
Fe+2, Fe+3, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
Cl-, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
Activated carbon (estim.), kg/1,000 kg |
|
|
|
2 |
|
|
4 |
|
|
6 |
|
Biosolids, kg/1,000 kg |
|
|
|
|
|
163 |
|
|
|
163 |
|
Fe(OH)3, kg/1,000 kg |
|
|
|
|
|
32 |
|
|
|
32 |
|
Binder compound (TBD), kg/1,000 kg |
|
|
|
|
|
|
|
2,736 |
|
2,736 |
|
Cement additive (TBD), kg/1,000 kg |
|
|
|
|
|
|
|
547 |
|
547 |
|
Gases |
|
|
|
|
|
|
|
|
|
|
|
O2, kg/1,000 kg |
|
|
378 |
|
50,720 |
|
|
|
|
51,098 |
|
N2, kg/1,000 kg |
|
|
1,273 |
|
15,246 |
|
|
|
|
16,519 |
|
CO2, kg/1,000 kg |
|
|
|
|
4,563 |
|
|
|
|
4,563 |
|
TC Shells |
1.23 |
|
|
|
|
|
|
|
|
1.23 |
|
TC Valves |
|
2.49 |
|
|
|
|
|
|
|
2.49 |
|
TC Plugs |
|
7.45 |
|
|
|
|
|
|
|
7.45 |
|
TC Cuttings (3 lb/TC, estim.), kg/1,000 kg |
|
1.69 |
|
|
|
|
|
|
|
1.69 |
TABLE G-3 Process Inputs for HD Neutralization, Configuration 2
|
|
Neutralization |
|
Biodegradation |
Photochem. Oxidation |
Ton Container Cleanout |
Sand Filtration |
|
|||||||
|
Stream Number (see figures) |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
11 |
14 |
15 |
16 |
17 |
|
|
Description |
TCs with Agent |
NaOH (aq.) |
Water |
Nutrients and Buffer |
Air |
Air |
Air |
Conditioning Chemicals |
Oxidation Chemicals |
Dilution Water |
Air |
Steam |
Air |
Total Inputs |
|
Process Conditions |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total flow, kg/1,000 kg |
1,020 |
1,002 |
6,727 |
1,262 |
62,296 |
6,671 |
2,409 |
62 |
34 |
78,571 |
1,648 |
227 |
31 |
161,960 |
|
Pressure, psig |
|
15 |
60 |
0 |
0 |
0 |
0 |
0 |
30 |
60 |
80 |
125 |
25 |
|
|
Temperature, ºF |
70 |
70 |
47 |
70 |
70 |
70 |
70 |
70 |
70 |
47 |
100 |
351 |
110 |
|
|
Physical state, solid (S), liquid (L), or gas (G) |
S, L & G |
L |
L |
S & L |
G |
G |
G |
L |
L |
|
G |
G |
G |
|
|
Feeds and Components |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
HD (C4H8Cl2S), kg/1,000 kg |
904 |
|
|
|
|
|
|
|
|
|
|
|
|
904 |
|
Sodium hydroxide (NaOH), kg/1,000 kg |
|
501 |
|
|
|
|
|
|
4 |
|
|
|
|
505 |
|
Water (H2O), kg/1,000 kg |
|
501 |
6,727 |
174 |
921 |
105 |
36 |
40 |
24 |
78,571 |
|
227 |
|
87,326 |
|
Sulfur-containing impurities, kg/1,000 kg |
82 |
|
|
|
|
|
|
|
|
|
|
|
|
82 |
|
Chlorinated aliphatic hydrocarbons, kg/1,000 kg |
7 |
|
|
|
|
|
|
|
|
|
|
|
|
7 |
|
Process Chemicals, Other |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
NaHCO3, kg/1,000 kg |
|
|
|
1,008 |
|
|
|
|
|
|
|
|
|
1,008 |
|
KNO3, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
0 |
|
KCl, kg/1,000 kg |
|
|
|
15 |
|
|
|
|
|
|
|
|
|
15 |
|
Na2SO4, kg/1000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
0 |
|
NH3, kg/1,000 kg |
|
|
|
37 |
|
|
|
|
|
|
|
|
|
37 |
|
Na3PO4, kg/1000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
0 |
|
Wolin salts, kg/1,000 kg |
|
|
|
8 |
|
|
|
|
|
|
|
|
|
8 |
|
H3PO4, kg/1,000 kg |
|
|
|
20 |
|
|
|
|
|
|
|
|
|
20 |
|
H2O2, kg/1,000 kg |
|
|
|
|
|
|
|
|
6 |
|
|
|
|
6 |
|
Organics, kg/1000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
0 |
|
Polymer, kg/1,000 kg |
|
|
|
|
|
|
|
4 |
|
|
|
|
|
4 |
|
|
Neutralization |
|
Biodegradation |
Solidify & Stabilize |
Photochem. Oxidation |
Ton Container Cleanout |
Water Recycle |
|
||||||||
|
Stream Number (see figures) |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
11 |
15 |
16 |
13 |
17 |
|
|
Description |
TCs with Agent |
NaOH (aq.) |
Water |
Nutrients and Buffer |
Air |
Air |
Air |
Conditioning Chemicals |
Binder |
Additives |
|
Air |
Steam |
Cooling Tower |
Sand Filter Air |
Total Inputs |
|
Fe+2, Fe+3, kg/1,000 kg |
10 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10 |
|
Cl-, kg/1,000 kg |
17 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
17 |
|
Activated carbon (estim.), kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
0 |
|
Biosolids, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
0 |
|
Fe(OH)3, kg/1000 kg |
|
|
|
|
|
|
|
18 |
|
|
|
|
|
|
|
18 |
|
Gases |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O2, kg/1,000 kg |
|
|
|
|
14,208 |
1,520 |
549 |
|
|
|
|
380 |
|
|
7 |
16,664 |
|
N2, kg/1000 kg |
|
|
|
|
44,239 |
4,733 |
1,711 |
|
|
|
|
1,189 |
|
|
23 |
51,895 |
|
CO2, kg/1,000 kg |
|
|
|
|
2,928 |
313 |
113 |
|
|
|
|
78 |
|
|
2 |
3,434 |
|
TC Shells |
1.23 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TC Valves |
2.49 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TC Plugs |
7.45 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TC Cuttings (3 lb/TC, estim.), kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TABLE G-4 Process Outputs for HD Neutralization, Configuration 2
|
|
Ton Container Cleanout and Neutralization |
Biodegradation Process |
Sand Filtration |
|
|||||
|
Stream Number (see figures) |
100 |
101 |
104 |
112 |
105 |
106 |
113 |
108 |
|
|
Description |
Ton Container Bodies |
Valves, Plugs, etc. |
Vent Gas |
Activated Carbon |
Vent Gas |
Biomass (from filter) |
Activated Carbon |
Effluent (to FOTW) |
Total Outputs |
|
Process Conditions |
|
|
|
|
|
|
|
|
|
|
Total flow, kg/1,000 kg |
|
|
1,651 |
2 |
71,339 |
972 |
4 |
87,947 |
161,915 |
|
Pressure, psig |
|
|
-5 |
0 |
0 |
0 |
0 |
0 |
|
|
Temperature, °F |
70 |
70 |
110 |
|
100 |
90 |
|
94 |
|
|
Physical state, solid (S), liquid (L), or gas (G) |
S |
S |
G |
S |
G |
|
S |
L |
|
|
Feeds and Components |
|
|
|
|
|
|
|
|
|
|
HD (C4H8Cl2S), kg/l,000 kg |
|
|
|
|
|
|
|
|
0 |
|
Sodium hydroxide (NaOH), kg/1,000 kg |
|
|
|
|
|
|
|
|
0 |
|
Water (H2O), kg/1,000 kg |
|
|
|
|
811 |
757 |
|
86,278 |
87,846 |
|
Sulfur-containing impurities, kg/1,000 kg |
|
|
|
|
|
|
|
|
0 |
|
Chlorinated aliphatic hydrocarbons, kg/1,000 kg |
|
|
|
|
|
|
|
|
0 |
|
Process Residuals, Other |
|
|
|
|
|
|
|
|
|
|
NaHCO3, kg/1,000 kg |
|
|
|
|
|
|
|
|
0 |
|
NaCl, kg/1,000 kg |
|
|
|
|
|
7 |
|
740 |
747 |
|
KNO3, kg/1,000 kg |
|
|
|
|
|
|
|
2 |
2 |
|
KCl, kg/1,000 kg |
|
|
|
|
|
|
|
7 |
7 |
|
Na2SO4, kg/1,000 kg |
|
|
|
|
|
7 |
|
792 |
799 |
|
NH3, kg/1,000 kg |
|
|
|
|
|
|
|
|
0 |
|
NaNO3, kg/1,000 kg |
|
|
|
|
|
1 |
|
28 |
29 |
|
NaNO2, kg/1,000 kg |
|
|
|
|
|
|
|
8 |
8 |
|
Na3PO4, kg/1,000 kg |
|
|
|
|
|
|
|
7 |
7 |
|
Wolin salts, kg/1,000 kg |
|
|
|
|
|
|
|
8 |
8 |
|
H3PO4, kg/1,000 kg |
|
|
|
|
|
|
|
|
0 |
|
H2O2, kg/1,000 kg |
|
|
|
|
|
|
|
|
0 |
|
Organics, kg/1,000 kg |
|
|
|
|
|
1 |
|
76 |
77 |
|
Polymer, kg/1,000 kg |
|
|
|
|
|
4 |
|
|
4 |
|
Fe+2, Fe+3, kg/1,000 kg |
|
|
|
|
|
|
|
|
0 |
|
Cl-, kg/1,000 kg |
|
|
|
|
|
|
|
|
0 |
|
Activated carbon (estim.), kg/1,000 kg |
|
|
|
2 |
|
|
4 |
|
6 |
|
|
Ton Container Cleanout and Neutralization |
Biodegradation Process |
Sand Filtration |
|
|||||
|
Stream Number (see figures) |
100 |
101 |
104 |
112 |
105 |
106 |
113 |
108 |
|
|
Description |
Ton Container Bodies |
Valves, Plugs, etc. |
Vent Gas |
Activated Carbon |
Vent Gas |
Biomass (from filter) |
Activated Carbon |
Effluent (to FOTW) |
Total Outputs |
|
Biosolids, kg/1,000 kg |
|
|
|
|
|
163 |
|
|
163 |
|
Fe(OH)3, kg/1,000 kg |
|
|
|
|
|
32 |
|
|
32 |
|
Binder compound (TBD), kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
Cement additive (TBD), kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
Gases |
|
|
|
|
|
|
|
|
|
|
O2, kg/1,000 kg |
|
|
378 |
|
50,720 |
|
|
|
51,098 |
|
N2, kg/1,000 kg |
|
|
1,273 |
|
15,246 |
|
|
|
16,519 |
|
CO2, kg/1,000 kg |
|
|
|
|
4,563 |
|
|
|
4,563 |
|
TC Shells |
1.23 |
|
|
|
|
|
|
|
1.23 |
|
TC Valves |
|
2.49 |
|
|
|
|
|
|
2.49 |
|
TC Plugs |
|
7.45 |
|
|
|
|
|
|
7.45 |
|
TC Cuttings (3 lb/TC, estim.), kg/1,000 kg |
|
1.69 |
|
|
|
|
|
|
1.69 |
TABLE G-5 Process Inputs for HD Neutralization, Configuration 3
|
|
Neutralization |
Biodegradation |
Ton Container Cleanout |
Sand Filtration |
|
|||||||
|
Stream Number (see figures) |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
15 |
16 |
17 |
|
|
Description |
TCs with Agent |
NaOH (aq.) |
Water |
Nutrients and Buffer |
Air |
Air |
Air |
Conditioning Chemicals |
Air |
Steam |
Air |
Total Inputs |
|
Process Conditions |
|
|
|
|
|
|
|
|
|
|
|
|
|
Total flow, kg/l,000 kg |
1,020 |
1,002 |
6,727 |
1,262 |
62,296 |
6,671 |
2,409 |
62 |
1,648 |
227 |
31 |
161,960 |
|
Pressure, psig |
|
15 |
60 |
0 |
0 |
0 |
0 |
0 |
80 |
125 |
25 |
|
|
Temperature, ºF |
70 |
70 |
47 |
70 |
70 |
70 |
70 |
70 |
100 |
351 |
110 |
|
|
Physical state, solid (S), liquid (L), or gas (G) |
S, L & G |
L |
L |
S & L |
G |
G |
G |
L |
G |
G |
G |
|
|
Feeds and Components |
|
|
|
|
|
|
|
|
|
|
|
|
|
HD (C4H8Cl2S), kg/1,000 kg |
904 |
|
|
|
|
|
|
|
|
|
|
904 |
|
Sodium hydroxide (NaOH), kg/ 1,000 kg |
|
501 |
|
|
|
|
|
|
|
|
|
501 |
|
Water (H2O), kg/1,000 kg |
|
501 |
87,691 |
174 |
921 |
105 |
36 |
40 |
|
227 |
|
87,326 |
|
Sulfur-containing impurities, kg/1,000 kg |
82 |
|
|
|
|
|
|
|
|
|
|
82 |
|
Chlorinated aliphatic hydrocarbons, kg/1,000 kg |
7 |
|
|
|
|
|
|
|
|
|
|
7 |
|
Process Chemicals, Other |
|
|
|
|
|
|
|
|
|
|
|
|
|
NaHCO3, kg/1,000 kg |
|
|
|
1,008 |
|
|
|
|
|
|
|
1,008 |
|
KNO3, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
0 |
|
KCl, kg/1,000 kg |
|
|
|
15 |
|
|
|
|
|
|
|
15 |
|
Na2SO4, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
0 |
|
NH3, kg/1,000 kg |
|
|
|
37 |
|
|
|
|
|
|
|
37 |
|
Na3PO4, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
0 |
|
Wolin salts, kg/1,000 kg |
|
|
|
8 |
|
|
|
|
|
|
|
8 |
|
H3PO4, kg/1,000 kg |
|
|
|
20 |
|
|
|
|
|
|
|
20 |
|
Organics, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
0 |
|
Polymer, kg/1,000 kg |
|
|
|
|
|
|
|
4 |
|
|
|
4 |
|
Fe+2, Fe+3, kg/1,000 kg |
10 |
|
|
|
|
|
|
|
|
|
|
10 |
|
|
Neutralization |
Biodegradation |
Ton Container Cleanout |
Sand Filtration |
|
|||||||
|
Stream Number (see figures) |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
15 |
16 |
17 |
|
|
Description |
TCs with Agent |
NaOH (aq.) |
Water |
Nutrients and Buffers |
Air |
Air |
Air |
Conditioning Chemicals |
Air |
Steam |
Air |
Total Inputs |
|
Cl-, kg/1,000 kg |
17 |
|
|
|
|
|
|
|
|
|
|
17 |
|
Activated carbon (estim.), kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
0 |
|
Biosolids, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
0 |
|
Fe(OH)3, kg/1,000 kg |
|
|
|
|
|
|
|
18 |
|
|
|
18 |
|
Gases |
|
|
|
|
|
|
|
|
|
|
|
|
|
O2, kg/1,000 kg |
|
|
|
|
14,208 |
1,520 |
549 |
|
380 |
|
7 |
16,664 |
|
N2, kg/1,000 kg |
|
|
|
|
44,239 |
4,733 |
1,711 |
|
1,189 |
|
23 |
51,895 |
|
CO2, kg/1,000 kg |
|
|
|
|
2,928 |
313 |
113 |
|
78 |
|
2 |
3,434 |
|
TC Shells |
1.23 |
|
|
|
|
|
|
|
|
|
|
|
|
TC Valves |
2.49 |
|
|
|
|
|
|
|
|
|
|
|
|
TC Plugs |
7.45 |
|
|
|
|
|
|
|
|
|
|
|
|
TC Cuttings (3 lb/TC, estim.), kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
TABLE G-6 Process Outputs for HD Neutralization, Configuration 3
|
|
Ton Container Cleanout and Neutralization |
Biodegradation Process |
|
Sand Filtration |
Stripped VOCs |
|
||||
|
Stream Number (see figures) |
100 |
101 |
104 |
112 |
105 |
106 |
113 |
108 |
109 |
|
|
Description |
Ton container bodies |
Valves, plugs, etc. |
Vent gas |
Activated carbon |
Vent gas |
Biomass (from filter) |
Activated carbon |
Effluent (to FOTW) |
VOCs to TSDF |
Total Outputs |
|
Process Conditions |
|
|
|
|
|
|
|
|
|
|
|
Total flow, kg/1,000 kg |
|
|
1,651 |
2 |
71,339 |
972 |
4 |
87,947 |
2,352 |
164,267 |
|
Pressure, psig |
|
|
-5 |
0 |
0 |
0 |
0 |
0 |
0 |
|
|
Temperature, ºF |
70 |
70 |
110 |
|
100 |
90 |
|
94 |
35 |
|
|
Physical state, solid (S), liquid (L), or gas (G) |
S |
S |
G |
S |
G |
S |
S |
L |
L |
|
|
Feeds and Components |
|
|
|
|
|
|
|
|
|
|
|
HD (C4H8Cl2S), kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
Sodium hydroxide (NaOH), kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
Water (H2O), kg/1,000 kg |
|
|
|
|
811 |
|
|
86,278 |
2,347 |
89,436 |
|
Sulfur-containing impurities, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
Chlorinated aliphatic hydrocarbons, kg/1,000 kg |
|
|
|
|
|
|
|
|
5 |
5 |
|
Process Residuals, Other |
|
|
|
|
|
|
|
|
|
|
|
NaHCO3, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
NaCl, kg/1,000 kg |
|
|
|
|
|
7 |
|
740 |
|
747 |
|
KNO3, kg/1,000 kg |
|
|
|
|
|
|
|
2 |
|
2 |
|
KCl, kg/1,000 kg |
|
|
|
|
|
|
|
7 |
|
7 |
|
Na2SO4, kg/1,000 kg |
|
|
|
|
|
7 |
|
792 |
|
799 |
|
NH3, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
NaNO3, kg/1,000 kg |
|
|
|
|
|
1 |
|
28 |
|
29 |
|
NaNO2, kg/1,000 kg |
|
|
|
|
|
|
|
8 |
|
8 |
|
Na3PO4, kg/1,000 kg |
|
|
|
|
|
|
|
7 |
|
7 |
|
Wolin salts, kg/1,000 kg |
|
|
|
|
|
|
|
8 |
|
8 |
|
H3PO4, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
|
Ton Container Cleanout and Neutralization |
Biodegradation Process |
|
Sand Filtration |
Stripped VOCs |
|
||||
|
Stream Number (see figures) |
100 |
101 |
104 |
112 |
105 |
106 |
113 |
108 |
109 |
|
|
Description |
Ton container bodies |
Valves, plugs, etc. |
Vent gas |
Activated carbon |
Vent gas |
Biomass (from filter) |
Activated carbon |
Effluent (to FOTW) |
VOCS to TSDF |
Total Outputs |
|
H2O2, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
Organics, kg/1,000 kg |
|
|
|
|
|
1 |
|
76 |
|
77 |
|
Polymer, kg/1,000 kg |
|
|
|
|
|
4 |
|
|
|
4 |
|
Fe+2, Fe+3, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
|
|
Cl-, kg/1,000 kg |
|
|
|
|
|
|
|
|
|
0 |
|
Activated carbon (estim.), kg/1,000 kg |
|
|
|
2 |
|
|
4 |
|
|
6 |
|
Biosolids, kg/1,000 kg |
|
|
|
|
|
163 |
|
|
|
163 |
|
Fe(OH)3, kg/1,000 kg |
|
|
|
|
|
32 |
|
|
|
32 |
|
Gases |
|
|
|
|
|
|
|
|
|
|
|
O2, kg/1,000 kg |
|
|
378 |
|
50,720 |
|
|
|
|
51,098 |
|
N2, kg/1,000 kg |
|
|
1,273 |
|
15,246 |
|
|
|
|
16,519 |
|
CO2, kg/1,000 kg |
|
|
|
|
4,563 |
|
|
|
|
4,563 |
|
TC Shells |
1.23 |
|
|
|
|
|
|
|
|
1.23 |
|
TC Valves |
|
2.49 |
|
|
|
|
|
|
|
2.49 |
|
TC Plugs |
|
7.45 |
|
|
|
|
|
|
|
7.45 |
|
TC Cuttings (3 lb/TC, estim.), kg/1,000 kg |
|
1.69 |
|
|
|
|
|
|
|
1.69 |
TABLE G-7 Process Inputs for HD Neutralization, Configuration 4
|
|
Neutralization |
Ton Container Clean-Out |
|
|||
|
Stream Number (see figures) |
1 |
2 |
3 |
15 |
16 |
|
|
Description |
TCs with Agent |
NaOH (aq.) |
Water |
Air |
Steam |
Total Inputs |
|
Process Conditions |
|
|
|
|
|
|
|
Total flow, kg/1,000 kg |
1,020 |
1,002 |
27,304 |
1,648 |
227 |
31,201 |
|
Pressure, psig |
|
15 |
60 |
80 |
125 |
|
|
Temperature, °F |
70 |
70 |
47 |
100 |
351 |
|
|
Physical state, solid (S), liquid (L), or gas (G) |
S, L & G |
L |
L |
G |
G |
|
|
Feeds and Components |
|
|
|
|
|
|
|
HD (C4H8Cl2S), kg/1,000 kg |
904 |
|
|
|
|
904 |
|
Sodium hydroxide (NaOH), kg/1,000 kg |
|
501 |
|
|
|
501 |
|
Water (H2O), kg/1,000 kg |
|
501 |
27,304 |
|
227 |
28,032 |
|
Sulfur-containing impurities, kg/1,000 kg |
82 |
|
|
|
|
82 |
|
Chlorinated aliphatic hydrocarbons, kg/1,000 kg |
7 |
|
|
|
|
7 |
|
Process Chemicals, Other |
|
|
|
|
|
|
|
Fe+2, Fe+3, kg/1,000 kg |
10 |
|
|
|
|
10 |
|
Cl-, kg/1,000 kg |
17 |
|
|
|
|
17 |
|
Activated carbon (estim.), kg/1,000 kg |
|
|
|
|
|
0 |
|
Gases |
|
|
|
|
|
|
|
O2, kg/1,000 kg |
|
|
|
380 |
|
380 |
|
N2, kg/1,000 kg |
|
|
|
1,189 |
|
1,189 |
|
CO2, kg/1,000 kg |
|
|
|
78 |
|
78 |
|
TC Shells |
1.23 |
|
|
|
|
1.23 |
|
TC Valves |
2.49 |
|
|
|
|
2.49 |
|
TC Plugs |
7.45 |
|
|
|
|
7.45 |
|
TC Cuttings (3 lb/TC, estim.), kg/1,000 kg |
|
|
|
|
|
|
TABLE G-8 Process Outputs for HD Neutralization, Configuration 4
|
|
Ton Container Clean-Out and Neutralization |
|
|
|||
|
Stream Number (see figures) |
100 |
101 |
104 |
110 |
112 |
|
|
Total Inputs |
Ton Container Bodies |
Valves, Plugs, etc. |
Vent Gas |
Hydrolysate to TSDF |
Activated Carbon |
Total Outputs |
|
Process Conditions |
|
|
|
|
|
|
|
Total flow, kg/1,000 kg |
|
|
1,651 |
29,468 |
2 |
31,121 |
|
Pressure, psig |
|
|
-5 |
20 |
0 |
|
|
Temperature, °F |
70 |
70 |
110 |
120 |
|
|
|
Physical state, solid (S), liquid (L), or gas (G) |
S |
S |
G |
L |
S |
|
|
Feeds and Components |
|
|
|
|
|
|
|
HD (C4H8Cl2S), kg/1,000 kg |
|
|
|
|
|
0 |
|
Sodium hydroxide (NaOH), kg/1,000 kg |
|
|
|
12 |
|
12 |
|
Water (H2O), kg/1,000 kg |
|
|
|
27,949 |
|
27,949 |
|
Sulfur-containing impurities, kg/1,000 kg |
|
|
|
17 |
|
17 |
|
Chlorinated aliphatic hydrocarbons, kg/1,000 kg |
|
|
|
7 |
|
7 |
|
Process Residuals, Other |
|
|
|
|
|
|
|
Thiodiglycol, kg/1,000 kg |
|
|
|
624 |
|
624 |
|
Other hydrolysis products, kg/1,000 kg |
|
|
|
125 |
|
125 |
|
NaCl, kg/1,000 kg |
|
|
|
715 |
|
715 |
|
Fe+2, Fe+3, kg/1,000 kg |
|
|
|
|
|
0 |
|
Cl-, kg/1,000 kg |
|
|
|
|
|
0 |
|
Activated carbon (estim.), kg/1,000 kg |
|
|
|
|
2 |
2 |
|
Gases |
|
|
|
|
|
|
|
O2, kg/1,000 kg |
|
|
378 |
|
|
378 |
|
N2, kg/1,000 kg |
|
|
1,273 |
|
|
1,273 |
|
CO2, kg/1,000 kg |
|
|
|
|
|
0 |
|
TC Shells |
1.23 |
|
|
|
|
1.23 |
|
TC Valves |
|
2.49 |
|
|
|
2.49 |
|
TC Plugs |
|
7.45 |
|
|
|
7.45 |
|
TC Cuttings (3 lb/TC, estim.), kg/1,000 kg |
|
1.69 |
|
|
|
1.69 |
Appendix H
Mass Balances for VX Neutralization
This appendix contains mass balance matrices for neutralization of VX followed by off-site treatment of oxidized hydrolysate, as described in Chapter 8. There is one matrix for process inputs (Table H-1) and one for process outputs (Table H-2). The stream numbers in the column headings are keyed to the numbered input and output streams shown in the process diagram (Figure H-1). Input streams are numbered from 1; output streams are numbered from 100.
The process diagram and the mass balance data are derived either from the April 4, 1996, design package submitted by the Army Alternative Technology Program or from more recent data.
TABLE H-1 Process Inputs for VX Neutralization
|
|
Neutralization |
Ton Container Cleanout |
|
|||||
|
Stream Number (see figures) |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
|
|
Description |
Ton Containers with Agent |
NaOH (aq.) |
Water |
NaOCl (aq) |
Air |
Steam |
Decontam. Fluid |
Total Inputs |
|
Process Conditions |
|
|
|
|
|
|
|
|
|
Total flow, kg/1,000 kg |
1,000 |
1,028 |
2,660 |
4,958 |
1,728 |
174 |
510 |
12,058 |
|
Pressure, psig |
|
15 |
65 |
15 |
80 |
125 |
15 |
|
|
Temperature, °F |
70 |
70 |
47 |
70 |
70 |
351 |
70 |
|
|
Physical State, solid (S), liquid (L), or gas (G) |
S, L |
L |
L |
L |
G |
G |
L |
|
|
Major Feed Components |
|
|
|
|
|
|
|
|
|
VX, C11H26NO2PS, kg/1,000 kg |
937 |
|
|
|
|
|
|
937 |
|
Water, kg/1,000 kg |
|
617 |
2,660 |
4,214 |
|
174 |
495 |
8,160 |
|
NaOH, kg/1,000 kg |
|
411 |
|
|
|
|
|
411 |
|
NaOCl, kg/1,000 kg |
|
|
|
744 |
|
|
15 |
759 |
|
NaCl, kg/1,000 kg |
|
|
|
|
|
|
|
0 |
|
Air, kg/1,000 kg |
|
|
|
|
1,728 |
|
|
1,728 |
|
Agent Impurities |
|
|
|
|
|
|
|
|
|
Diisopropylamine, kg/1,000 kg |
1 |
|
|
|
|
|
|
1 |
|
Diisopropylcarbodiimide (stabilizer), kg/1,000 kg |
17 |
|
|
|
|
|
|
17 |
|
O-Ethyl methylethylphosphinate, kg/1,000 kg |
2 |
|
|
|
|
|
|
2 |
|
Diethyl methylphosphonate, kg/1,000 kg |
1 |
|
|
|
|
|
|
1 |
|
2-(Diisopropylamino)ethanethiol, kg/1,000 kg |
9 |
|
|
|
|
|
|
9 |
|
O,O-Diethyl methylphosphonothioate, kg/1,000 kg |
2 |
|
|
|
|
|
|
2 |
|
O,S-Diethyl methylphosphonothioate, kg/1,000 kg |
1 |
|
|
|
|
|
|
1 |
|
2-(Diisopropylamino)ethyl ethyl sulfide, kg/1,000 kg |
1 |
|
|
|
|
|
|
1 |
|
Diethyl dimethylpyrophosphonate ("Pyro"), kg/1,000 kg |
10 |
|
|
|
|
|
|
10 |
|
O,O-Diethyl dimethylpyrophosphonothioate, kg/1,000 kg |
2 |
|
|
|
|
|
|
2 |
|
O-(2-Diisopropylaminoethyl) O-ethylmethylphosphonate, kg/1,000 kg |
3 |
|
|
|
|
|
|
3 |
|
1,2-bis(ethyl methylphosphonothiolo)ethane, kg/1,000 kg |
6 |
|
|
|
|
|
|
6 |
|
Unknowns, kg/1,000 kg |
7 |
|
|
|
|
|
|
7 |
|
Ton Containers, no./1000 kg |
1.52 |
|
|
|
|
|
|
1.52 |
|
TC Valves, no./1000 kg |
3.06 |
|
|
|
|
|
|
3.06 |
|
TC Plugs, no./1000 kg |
9.19 |
|
|
|
|
|
|
9.19 |
|
TC Cuttings (3 lb/TC estimated), kg/1,000 kg |
|
|
|
|
|
|
|
0 |
|
Activated Carbon (estimated), kg/1,000 kg |
|
|
|
|
|
|
|
0 |
TABLE H-2 Process Outputs for VX Neutralization
|
|
Ton Container Cleanout and Neutralization |
|
||||
|
Stream Number (see figures) |
100 |
101 |
102 |
103 |
104 |
|
|
Description |
Ton Container Bodies |
Valves, Plugs, etc. |
Hydrolysate to TSDF |
Vent Gas |
Activated Carbon |
Total Outputs |
|
Process Conditions |
|
|
|
|
|
|
|
Total Flow, kg/1,000 kg |
see below |
see below |
10,330 |
1,736 |
2 |
12,068 |
|
Pressure, psig |
|
|
10 |
-1 |
|
|
|
Temperature, °F |
70 |
70 |
70 |
110 |
110 |
|
|
Physical State, solid (S), liquid (L), or gas (G) |
S |
S |
L |
G |
S |
|
|
Major Feed Components |
|
|
|
|
|
|
|
VX, C11H26NO2PS, kg/1,000 kg |
|
|
|
|
|
0 |
|
Water, kg/1,000 kg |
|
|
7,575 |
|
|
7,575 |
|
NaOH, kg/1,000 kg |
|
|
120 |
|
|
120 |
|
NaOCl, kg/1,000 kg |
|
|
|
|
|
0 |
|
NaCl, kg/1,000 kg |
|
|
1,192 |
|
|
1,192 |
|
Air, kg/1,000 kg |
|
|
|
1,736 |
|
1,736 |
|
Agent Impurities |
|
|
|
|
|
|
|
Diisopropylamine, kg/1,000 kg |
|
|
1 |
|
|
1 |
|
O-Ethyl methylethylphosphinate, kg/1,000 kg |
|
|
2 |
|
|
2 |
|
O,O-Diethyl methylphosphonothioate, kg/1,000 kg |
|
|
2 |
|
|
2 |
|
O,S-Diethyl methylphosphonothioate, kg/1,000 kg |
|
|
1 |
|
|
1 |
|
2-(Diisopropylamino)ethyl ethyl sulfide, kg/1,000 kg |
|
|
1 |
|
|
1 |
|
O,O-Diethyl dimethylpyrophosphonothioate, kg/1,000 kg |
|
|
2 |
|
|
2 |
|
1,2-bis(ethyl methylphosphonothiolo)ethane, kg/1,000 kg |
|
|
6 |
|
|
6 |
|
Unknowns, kg/1,000 kg |
|
|
7 |
|
|
7 |
|
Process Residuals, Other |
|
|
|
|
|
|
|
EMPA-Na, kg/1,000 kg |
|
|
463 |
|
|
463 |
|
MPA-2Na, kg/1,000 kg |
|
|
49 |
|
|
49 |
|
EA-2192, Na salt, kg/1,000 kg |
|
|
2 |
|
|
2 |
|
Chloroform, kg/1,000 kg |
|
|
43 |
|
|
43 |
|
Chloroamine, kg/1,000 kg |
|
|
2 |
|
|
2 |
|
Diisopropylamino ethylsulfonic acid, kg/1,000 kg |
|
|
251 |
|
|
251 |
|
Diisopropylamino ethylsulfinic acid, kg/1,000 kg |
|
|
534 |
|
|
534 |
|
Dicyclohexylurea (DCHU) or Diisopropylurea, kg/1,000 kg |
|
|
14 |
|
|
14 |
|
Sodium methylphosphinate, kg/1,000 kg |
|
|
1 |
|
|
1 |
|
EMPSA, Na salt, kg/1,000 kg |
|
|
5 |
|
|
5 |
|
Methyl phosphonothioates, as salts, kg/1,000 kg |
|
|
10 |
|
|
10 |
|
Other methylphosphonates, as salts, kg/1,000 kg |
|
|
2 |
|
|
2 |
|
Disulfide, kg/1,000 kg |
|
|
11 |
|
|
11 |
|
Trisulfide, kg/1,000 kg |
|
|
16 |
|
|
16 |
|
Other sulfides and amines, kg/1,000 kg |
|
|
18 |
|
|
18 |
|
TC, no/1000 kg |
1.52 |
|
|
|
|
1.52 |
|
TC Valves, no./1000 kg |
|
3.06 |
|
|
|
3.06 |
|
TC Plugs, no./1000 kg |
|
9.19 |
|
|
|
9.19 |
|
TC Cuttings (3 lb/TC estimated, kg/1,000 kg |
|
2.08 |
|
|
|
2.08 |
|
Activated Carbon (estimated), kg/1,000 kg |
|
|
|
|
2 |
2.00 |
Appendix I
Biographical Sketches of Panel Members
Richard S. Magee, chair, is a professor in the Department of Mechanical Engineering and the Department of Chemical Engineering, Chemistry, and Environmental Science and is executive director of the Center for Environmental Engineering and Science at New Jersey Institute of Technology (NJIT). He also directs the U.S. Environmental Protection Agency Northeast Hazardous Substance Research Center as well as the Hazardous Substance Management Research Center, which is jointly sponsored by the National Science Foundation and the New Jersey Commission on Science and Technology, both headquartered at NJIT. He is a fellow of the ASME (American Society of Mechanical Engineers) and a diplomate of the American Academy of Environmental Engineers. Dr. Magee's research expertise is in combustion, with a major interest in the incineration of municipal and industrial wastes. He has served as vice chairman of the ASME Research Committee on Industrial and Municipal Wastes and as a member of the United Nations Special Commission (under Security Council Resolution 687) Advisory Panel on the Destruction of Iraq's Chemical Weapons Capabilities. He is presently a member of the North Atlantic Treaty Organization Science Committee Priority Area Panel on Disarmament Technologies. Dr. Magee is also the current chair of the NRC Committee on Review and Evaluation of the Army Chemical Stockpile Disposal Program (Stockpile Committee).
Joan B. Berkowitz graduated from the University of Illinois with a Ph.D. in physical chemistry and from the Sloan School Senior Executive Program at M.I.T. Dr. Berkowitz is currently the managing director of Farkas Berkowitz and Company. She has extensive experience in the area of environmental and hazardous waste management, a knowledge of available technologies for the cleanup of contaminated soils and groundwater, and a background in physical and electrochemistry. She has contributed to several EPA studies, been a consultant on remediation techniques, and assessed various destruction technologies. Dr. Berkowitz has written numerous publications on hazard waste treatment and environmental subjects.
Gene H. Dyer graduated with a bachelor of science degree in chemistry, mathematics, and physics from the University of Nebraska. Over a 12-year period, he worked for General Electric as a process engineer, the U.S. Navy as a research and development project engineer, and the U.S. Atomic Energy Commission as a project engineer. In 1963, he began a more than 20-year career with the Bechtel Corporation, first as a consultant on advanced nuclear power plants and later as a program supervisor for nuclear facilities. From 1969 to 1983, he was manager of the Process and Environmental Department, which provided engineering services related to research and development projects, including technology probes, environmental assessment, air pollution control, water pollution control, process development, nuclear fuel process development, and regional planning. As a senior staff consultant for several years, he was responsible for identifying and evaluating new technologies and managing further development and testing for practical applications. Mr. Dyer is a member of the American Institute of Chemical Engineers and a registered professional engineer. He recently served as a member of the NRC Committee on Alternative Chemical Demilitarization Technologies and is currently a member of the NRC Committee on Review and Evaluation of the Army Chemical Stockpile Disposal Program (Stockpile Committee).
Frederick T. Harper is the manager of the Accident Analysis and Consequence Assessment Department at the Sandia National Laboratory, Albuquerque, New Mexico. His areas of expertise are the probabilistic assessment of accident progression, including the physical response of systems to accident conditions and the transport of toxicological and radiological contaminants; assessment of the release of contaminants; and the structural and thermal response of systems to fire and explosion. Dr. Harper has served on an international
committee in the area of consequence uncertainty and has been a prime developer of computer codes for assessing toxicological consequences and accident progression. Dr. Harper earned a bachelor's degree from Yale University in physics, a master's degree from the University of Virginia in nuclear engineering, and a doctorate, also in nuclear engineering, from the University of New Mexico. He is a member of Tau Beta Pi, the American Physical Society, and the American Nuclear Society.
Joseph A. Heintz recently retired from the Atlantic Richfield Oil Corporation where he was engineering manager for many years. Mr. Heintz attended the University of Illinois and Purdue University where he received degrees in electrical engineering. He is an expert in mechanical design, plant layout, process configuration, and process monitoring. He has supervised the designing of pressure vessels, overseen stress analysis studies, coordinated engineering standards and instrumentation groups responsible for developing process control strategies, prepared detailing piping and instrumentation diagrams, identified control system components, and prepared control system functional specifications. In addition, he has participated in the selection of control system vendors. His is a member of the Instrument Society of America.
David A. Hoecke, president and CEO of Enercon Systems, Inc., is an expert in the fields of waste combustion, pyrolysis, heat transfer, and gas cleaning. He graduated with a B.S.M.E from the Cooper Union in 1960 and rose from project engineer to R&D manager to chief engineer for incineration at Midland-Ross Corporation and later founded his own company. Mr. Hoecke has been responsible for the design and construction of numerous combustion systems, including solid waste incinerators, thermal oxidizers, heat recovery systems, gas-to-air heat exchangers, and high velocity drying ovens. This hands-on experience gives him the expertise needed to participate in the assessment of alternative destruction technologies for chemical agents. He has served as the cochair of the ASME Subcommittee on Vitrification Systems. He also recently served on the ASME Board on Research and Technology Development.
David S. Kosson graduated with a bachelor of science degree in chemical engineering, a master's degree in chemical and biochemical engineering, and a doctorate in chemical and biochemical engineering from Rutgers-The State University of New Jersey. He joined the faculty at Rutgers in 1986 as an associate professor, with tenure in 1990. He became a full professor in 1996. Dr. Kosson teaches graduate and undergraduate chemical engineering courses and conducts research for the Department of Chemical and Biochemical Engineering, where considerable work is under way in developing microbial, chemical, and physical treatment methods for hazardous waste. He is responsible for project planning and coordination, from basic research through full-scale design and implementation. He has published extensively in the fields of chemical engineering, waste management and treatment, and contaminant fate and transport in soils and groundwater. Dr. Kosson is a participant in several Environmental Protection Agency advisory panels involved in waste research and is the director of the Physical Treatment Division of the Hazardous Substances Management Research Center in New Jersey. He is a member of the American Institute of Chemical Engineers. He recently served as a member of the NRC Committee on Alternative Chemical Demilitarization Technologies and is currently a member of the NRC Committee on Review and Evaluation of the Army Chemical Stockpile Disposal Program (Stockpile Committee).
Walter G. May graduated with a bachelor of science degree in chemical engineering and master of science degree in chemistry from the University of Saskatchewan and a doctor of science degree in chemical engineering from the Massachusetts Institute of Technology. He joined the faculty of the University of Saskatchewan as a professor of chemical engineering in 1943. In 1948, he began a distinguished career with Exxon Research and Engineering Company, where he was a senior science advisor from 1976 to 1983. He was professor of chemical engineering at the University of Illinois from 1983 until his retirement in 1991. There he taught courses in process design, thermodynamics, chemical reactor design, separation processes, and industrial chemistry and stoichiometry. Dr. May has published extensively, served on the editorial boards of Chemical Engineering Reviews and Chemical Engineering Progress, and has obtained numerous patents in his field. He is a member of the National Academy of Engineering and a fellow of the American Institute of Chemical Engineers, and he has received special awards from the American Institute of Chemical Engineers and ASME. Dr. May's particular interest is in separations research. He is a registered professional engineer in the state of
Illinois and recently served as a member of the NRC Committee on Alternative Chemical Demilitarization Technologies. He is currently a member of the NRC Committee on Review and Evaluation of the Army Chemical Stockpile Disposal Program (Stockpile Committee).
Alvin H. Mushkatel, professor and director of the Office of Hazards Studies, and Professor of the School of Planning and Landscape Architecture, Arizona State University, is an expert in emergency response and risk perceptions. His research interests include emergency management, natural and technological hazards policy, and environmental policy. He has been a member of the NRC Committee on Earthquake Engineering and the Committee on the Decontamination and Decomissioning of Uranium Enrichment Facilities. His most recent research focuses on conflicts in intergovernmental policy involving high level nuclear waste disposal and the role of citizens in technological policy decision-making. He has published extensively on issues relating to siting controversies. Dr. Mushkatel is currently a member of the NRC Committee on Review and Evaluation of the Army Chemical Stockpile Disposal Program (Stockpile Committee).
Laurance Oden is a retired senior researcher in the Pyrometallurgy Subdivision of the Process Metallurgy Division of the Albany Research Center, U.S. Bureau of Mines, Albany, Oregon. Dr. Oden's expertise is in the fields of high temperature phase equilibria, superconductivity, the corrosion chemistry of metals and nonmetals, the thermochemistry of high temperature reactions, and the processing of metals and slags. He has written or co-written 94 publications and formal presentations and is the holds 15 patents. Dr. Oden received his bachelor's degree in chemistry from Oregon State University and his Ph.D. from Oregon State in mathematics and metallurgy.
George W. Parshall is a member of the National Academy of Sciences and a retired member of the Central Research Department of E.I. du Pont de Nemours & Company where he served for nearly 40 years, including 13 years as director of chemical science. Dr. Parshall is an expert in conducting and supervising chemical research, particularly in the area of catalysis and inorganic chemistry. He is a past member of the NRC Board on Chemical Science and Technology and has played an active role in NRC and National Science Foundation activities. He is currently a member of the NRC Committee on Review and Evaluation of the Army Chemical Stockpile Disposal Program (Stockpile Committee).
L. David Pye is currently dean of the College of Ceramics at Alfred University. Having received his undergraduate degree at Alfred, Dr. Pye started as a research engineer in the Melting and Forming Laboratory of PPG Industries, followed by Army service and a stint at Bausch and Lomb. After completing graduate studies at the University of Rochester and Alfred, he embarked on a long and distinguished career at Alfred University. In the course of his rise from assistant professor to dean, Dr. Pye has published more than 70 technical articles, presented more than 100 lectures and papers, established numerous international symposia, and set up the first Ph.D program in glass science in the United States. Dr. Pye is a fellow of the American Ceramic Society and the American Institute of Chemists and many other professional societies. He received the Dominick Labino Award from the Glass Art Society in 1995 and numerous other awards.
Roger W. Staehle is currently an industrial consultant and adjunct professor of chemical engineering and materials science at the University of Minnesota. He is a member of the National Academy of Engineering and has received the Whitney Award from the National Association of Corrosion Engineers (NACE) for outstanding work in corrosion science. He was a dean of the Institute of Technology and professor of chemical engineering and materials science at the University of Minnesota. Before that, Dr. Staehle was a professor at Ohio State University. Dr. Staehle has organized the two largest centers of corrosion science in the United States, one at Ohio State, called the Fontana Corrosion Center, and the other at the University of Minnesota. He was appointed first chair in corrosion science and technology at Ohio State when he received the International Nickel Chair. He was an editor of Corrosion Journal and Advances in Corrosion Science and Technology, has edited 23 books, and has written 160 papers. He is a fellow of NACE and the American Society for Metals. He has been a reactor engineer with the nuclear submarine program and a consultant on the subject of corrosion and degradation for industries in all major fields in the United States and many foreign countries.
William Tumas is currently the group leader for the Waste Treatment and Minimization Science and
Technology Group at Los Alamos National Laboratory. He is a senior chemist known primarily for his science and engineering research on waste treatment and minimization. His work has included research and development technology, industrial waste applications, and environmental restoration for DOE. At Los Alamos he has studied supercritical fluids, oxidation, and organic transformations. Dr. Tumas has written numerous papers and is a member of several professional organizations.
Appendix J
Questionnaires Sent to Technology Proponent Companies and Environmental Regulators
The AltTech Panel developed a questionnaire to guide panel members as they gathered information during visits and subsequent interactions with the three TPCs (technology proponent companies) and the Army's Alternative Technology Program, which was treated as the proponent for the neutralization technologies. This appendix includes samples of the cover memo sent to the TPCs and the memo sent to the Army, as well as the questionnaire.
|
December 15, 1995 MEMORANDUM TO: Technology Firms FROM: Mike Clarke, AltTech Panel, Study Director As currently planned, representatives of the NRC's AltTech Panel will be visiting each of you during the month of January. These visits will necessarily be brief and to the point, as the assessment team's time is limited. Thank you all for the support you have already provided. The list of questions that follows is provided to each of you to facilitate discussion and to ensure that you have the opportunity to plan for the requisite company representation at the meetings. I make no assertion that the list is all-inclusive, that there are no redundancies, or that some of the information is included in your submissions; only that these represent the body of data sought. The assessment teams are free to range over a wide spectrum of pertinent subjects, but, clearly, if they receive clear and concise answers to this list, they will have achieved most of their data gathering goals. In preparing an agenda for this visit, please allow adequate time for this purpose, even if it is at the expense of other important activities such as tours or company briefings. Thanks in advance for your help. Recognizing that the holidays are rapidly approaching, and I'd like to take this opportunity to wish you "Happy Holidays" and a safe and prosperous New Year, if you choose to answer some or all of these questions in writing either in advance or for delivery at the meetings, that would be very much appreciated. It might help you with your responses and reduce the amount of note taking the assessment teams will have to do. There are, of course, other areas that will be investigated that do not involve the companies, including meetings with the Army, state and federal regulators, and the interested public. This process should be completed by March. Attachment: Questionnaire |
|
December 15, 1995 MEMORANDUM TO: LTC Steve Landry, Chief Applied Technology Branch FROM: Mike Clarke, AltTech Panel, Study Director As currently planned, representatives of the NRC's AltTech Panel will be visiting you during the month of January. For review of the Army's neutralization technologies, this visit is scheduled for 18 and 19 January at Aberdeen. The visit will necessarily be brief and to the point, as the assessment team's time is limited. Thank you all for the support you have already provided. The list of questions that follows is provided to each technology proponent to facilitate discussion and to ensure that you have the opportunity to plan for the requisite representation at the meeting. I make no assertion that the list is all-inclusive, that there are no redundancies, or that some of the information is included in your submissions; only that these represent the body of data sought. The assessment team is free to range over a wide spectrum of pertinent subjects, but, clearly, if it receives clear and concise answers to this list, it will have achieved most of its data gathering goals. In preparing an agenda for this visit, please allow adequate time for this purpose, even if it is at the expense of other important activities such as tours or technology briefings. Thanks in advance for your help. Recognizing that the holidays are rapidly approaching, and I'd like to take this opportunity to wish you "Happy Holidays" and a safe and prosperous New Year, if you choose to answer some or all of these questions in writing either in advance or for delivery at the meetings, that would be very much appreciated. It might help you with your responses and reduce the amount of note taking the assessment team will have to do. Attachment: Questionnaire |
Questionnaire for Technology Assessment
1. Operational Requirements and Considerations
1.1 Feed Streams
- Has waste handling received attention so that one can be confident that there will be no surprises?
- What equipment is necessary for waste feeding and handling? At what scale has it been demonstrated?
- Is any pretreatment required? How are gels, solids, and other inhomogeneities handled and fed?
- How are the ton containers handled and what are the feed requirements to clean them?
1.2 Process Operation
For agent detoxification:
- What is the maximum residual concentration of agent in each process effluent?
- Materials and Energy Balance: What is the quantity (per unit of agent), physical state (gas, liquid, solid, slurry) and chemical composition (major components, unreacted reactants, organic reaction products, inorganic reaction products) for each process effluent? Specify for each agent type to be processed. What are the analytical detection limits for each species in each phase?
- Are any of the process reactions reversible to the extent that agent can be reformed?
- What type of toxicity evaluation, if any, has been carried out on process residuals?
For ton container cleanout:
- What is the proposed method for removal and detoxification of residual agent in bulk containers?
- How is detoxification/cleaning of ton containers ensured to 3X? 5X? What analytical methods will be necessary?
- How will the ton containers be managed (recycled, landfilled, etc.) after cleanout?
- Materials and Energy Balance:
- What is the quantity (per unit of agent), physical state (gas, liquid, solid, slurry) and chemical composition (major components, unreacted reactants, organic reaction products, inorganic reaction products) for each process effluent (e.g. decontamination fluid)? Specify for each agent type to be processed. What are the analytical detection limits for each species in each phase?
1.3 Process Effluent Streams
For bulk agent and ton container cleanout:
- What is total amount of solid, liquid, aqueous, slurry and gaseous waste products produced from treatment?
- What is the proposed management scenario (e.g. aqueous discharge to wastewater treatment facility, solidification/stabilization, landfill, atmospheric emission, recycling) for each process effluent?
- What additional treatment will be required to achieve disposal requirements under the proposed management scenario? What testing has been carried out for these treatment requirements and at what scale and on what wastes?
- What commercial facilities have been identified as potential recipients for each effluent waste stream? What are the permit requirements for the proposed management option?
For nonprocess wastes:
- How will nonprocess wastes (e.g. entry suits, dunnage, facility decontamination fluids) be managed?
- What additional treatment will be required to achieve disposal requirements under the proposed management scenario? What testing has been carried out for these treatment requirements? What commercial facilities have been identified as potential recipients for each nonprocess waste type?
1.4 Process Instrumentation and Controls
- What are the process monitoring requirements, e.g. detection limits for the feed and product streams? For process control? For effluents?
-
- How stringent are the process monitoring and control requirements?
- Does proven monitoring technology exist to meet process control and effluent discharge requirements? What is the operational experience with these monitoring systems?
- If new monitoring technology is required, what is the status of its development?
2. Materials of Construction
2.1 System and Materials
- What is the overall system diagram of piping and components?
- What are the materials of construction of the piping and components? Alloys, specifications.
- Where are the welds and what is the state of their stress relief?
- What kinds of inspections of welds and joints are being made?
2.2 Environmental Chemistry
- What are the nominal chemical environments, temperatures, pressures, residual stresses, and flow rates in each of the pipes and components?
- What are the exterior environments for the piping and components, i.e., the environments on the side opposite the process side? E.g., insulation, relative humidity, atmospheric contamination, and leached chemicals?
- What is the major environment and its nominal composition?
- What impurities are in the environment?
- What kinds of crevices are there in the piping and components in terms of gaskets, tight geometries, thermal sleeves, weld under penetrations, surface deposits, and/or bottom deposits?
- Where are heat transfer surfaces? What are the heat fluxes? Is there any heat flux in crevice geometries such as at tube supports?
- What are the startup and shutdown procedures?
- What are the procedures for deoxygenating or similar steps on startup? What is the temperature change rate on startup?
- What is the design life of the system and materials?
2.3 Qualification of Materials in the Application
-
•
What work has been done to qualify materials of construction for the design life in the way of corrosion and mechanical testing?
- If no laboratory work has been done, what literature references support the application of the materials?
2.4 Failure Definition
- What modes of failure have you considered for the various materials and components in your total system?
- What are the bases for considering the various failure modes?
2.5 Monitoring and Inspection
- What factors are you planning to monitor in the operating system? E.g., chemistry (what species?), temperature, pressure? Is the monitoring continuous or batch?
- How frequently will the system be inspected, and what locations are inspected for what observations?
2.6 Previous Experience
- What similar engineering or field experience is available on this or similar systems? What failures have occurred? What have been the results of inspections?
- What prototype facilities or laboratory systems have been operated using your system? What is their experience, operation time, failures, inspections?
3. Process Stability, Reliability and Robustness
3.1 Stability
- Can deviation from "normal" operation lead to an out-of-control situation where the system will find another operating regime that is quite different from the one desired?
- Are there process mechanisms, e.g. uncontrolled reactions, that could lead to a catastrophic facility failure? What are the safeguards against such events?
-
- How does the system respond/adjust to modest reaction condition changes, e.g. will a temperature rise lead to uncontrolled temperature increases.
- What is the total amount of stored energy in the system at any one time?
3.2 Reliability
- Does the mechanical equipment have a good record of performance?
- Are there backup systems to rescue the operation in case of failure of a component?
- How quickly will the backup system respond?
3.3 Robustness
- Will the process operate satisfactorily over a wide and varying range of operating conditions: temperature, pressure, energy input (mechanical, electrical, thermal) and composition of feed. How does the system respond to upsets in feed, reaction conditions or energy input?
- What control mechanisms are necessary to ensure operation with varying conditions and feeds?
- Will operation be continuous (days?, weeks?, months?) or intermittent? Which is better? Can other modes be employed?
4.1 Operations:
- What are the staffing requirements for normal operation? for normal shutdown/restart? for emergency shutdown/restart?
- What are the training requirements for staff (e.g. Ph.D electrical engineering vs. chemical plant operator vs. municipal sewage treatment operator)?
- What is the operational experience (documented) of the technology? On what kinds of wastes has the operational experience been obtained?
- What operational safeguards are built into the system?
- What control systems are necessary? What control systems have previously been demonstrated/employed? What does the control room look like?
- What experience is available on downtime vs. operational time? on what types of waste streams and at what scale?
4.2 8 hour versus 24 hour operation:
- Can the operation be reasonably run 8 hours a day? continuously for 24 hours a day?
- Does the system work better continuously or in an 8 hour operation shift?
- What are the requirements for shutdown/ready mode?
4.3 Startup/Shutdown:
- What is the procedure and can the system be shutdown and restarted with minimal upsets during normal operations?
- What are the procedures for emergency shutdown?
- What is the procedure for restarting after emergency shutdown?
4.4 Maintenance:
- What routine maintenance is required for normal operation?
- What documented record of performance is available concerning operation and maintenance of equipment? How much downtime is typical for normal operation? What is the operation/maintenance history of the technology?
- Are maintenance manuals and documented procedures available?
- What is the lifetime of equipment and what are the main consumables? What is the documented record of performance of equipment? How is the equipment replaced or maintained?
- What measures are taken to assure worker safety/exposure during routine maintenance?
- What staffing is required for normal maintenance?
5. Utility requirements
- What are the electrical, water and fuel requirements for the process?
6. Scale-Up Requirements
- What is the state of development of the process? How novel is the process?
- What scale of operation has been demonstrated and on what types of waste? To what extent will the plant be modified from the largest operation demonstrated?
- To what extent is the process, or parts of it, demonstrated commercial technology?
- Has the process been demonstrated with agent, i.e. feedstock and range of feedstock anticipated for the plant?
- To what extent have processes that would be used for ton container cleanout been demonstrated and on what types of waste?
- Very high conversions, e.g. 6 nines, will be required. How does the reactor design allow for this, e.g. batch reaction, staged reactors, etc. Will it change with scale-up? How does the system scale with mass, volume? What are the economic scaling factors?
- How much is understood about mass and energy transfer and will there be differences in "mixing" and "heat transfer" between small and large scale equipment (e.g. impellers and vessel size/shape, flow Reynolds number, Froude number (2-phase))?
- Has any catalysis been adequately demonstrated over reasonable time of operation with the range of feeds and possible poisons that will be encountered? How is system regenerated after poisoning?
- How well are the reaction mechanisms and intermediates understood for the destruction process? Frequently a reaction requires an "intermediate" that is built up during the reaction itself; the reaction may exhibit an "induction period" as a consequence. Is the reaction mechanism understood well enough to anticipate this?
- How many unit operations are involved in the entire treatment process, including treatment of secondary wastes?
7. Facility Decommissioning
- How will the disposal facility be decommissioned?
- What wastes (type and quantity) will be generated from facility decommissioning and how will they be managed?
8. Process Safety
8.1 Plant Safety and Health Risks
Risk of catastrophic failure and agent release:
- What are the possible modes of failure in feed systems, equipment, process operations, and monitoring systems that could give rise to a sudden release of agent?
- What influence could external factors have on the possibility of agent release (e.g. earthquake, vibration, ambient temperature, humidity, electrostatic discharge)?
- What measures can be taken to prevent the sudden release of agent and/or processing products? What is the proposal for secondary containment?
- What measures can be taken to mitigate the effects of an agent release on base personnel and the surrounding population if an agent release does occur?
Risk of exposing plant workers to agent:
- What are the possible modes of exposure of workers to agent over the duration of the disposal program?
- What is the expected level and duration of exposure for each of the identified modes?
- What are the known human and health effects of such exposures?
- What can be done to prevent worker exposure?
- Risk of plant worker exposure to other hazardous chemicals:
- What other hazardous chemicals could workers be exposed to?
- What are the associated human health effects at the possible levels and duration of exposure?
- What can be done to prevent worker exposure, and to mitigate the effects of exposure if it does occur?
8.2 Community Safety, Health and Environmental Risks
Risks of agent release and exposure due to normal operations:
- What are the possible sources and duration of agent release during normal operations?
- Following release, what are the pathways of agent migration outside of plant boundaries?
- What are the possible routes, levels and duration of exposure?
- What are the health effects that might result?
- What damages might result to natural resources and man-made structures?
Other risks due to normal operations:
- What other hazardous chemicals could be released during normal operations?
- What are the possible sources and duration of such releases?
- What are the pathways of migration outside of plant boundaries?
- What are the possible routes, levels and duration of exposure?
- What are the health effects that might result?
- What damages might result to natural resources and man-made structures?
Risks due to abnormal events:
- What is the largest possible release of agent?
- What is the largest possible release of other hazardous chemicals?
- How large an area would be affected?
- What are the possible adverse effects on human health and the environment?
- What emergency preparedness and emergency response measures can be taken to mitigate adverse effects?
Accident risk assessment:
- Has an accident risk assessment been done? If so, what were the results?
Health and environmental risk assessment:
- Has a health and environmental risk assessment been done? If so, what were the results?
Liability insurance:
- What type of liability insurance, if any, covers the use of the proposed technology?
- Has a risk assessment been conducted in support of an application for insurance? If so, what were the results?
9. Schedule
- What is the schedule for pilot-scale design and construction?
- What is the schedule for pilot-scale testing and evaluation?
- What is the effect on facility construction of scale-up requirements from pilot plant to commercial operations?
- What is the time required for facility construction?
- What is the time required for facility systemization?
- What is the effect on facility construction imposed by regulatory requirements? Permitting requirements?
- What is the effect of public acceptance on technology implementation?
- What is the expected duration of operations?
- What is the schedule for facility closure and site remediation?
Regulatory Review and Permitting Impacts
The following questions were solicited from the AltTech panel members as those they would most like to discuss in meetings with state environmental officials in Maryland and Indiana. There are redundancies, but to avoid the omission of any subtleties, the questions are included as written. The general intent is to determine the extent to which regulatory and permitting impacts may affect the eventual use of the five alternative technologies under consideration by the Army and being evaluated by the NRC. There is no prioritization to the questions.
- What are the primary restrictions on quantity, composition and toxicity for aqueous waste disposal?
- What are the primary restrictions on quantity, composition and toxicity for solid waste disposal from a chemical agent destruction facility at a land disposal facility? What testing is required to verify attainment of requirements?
- What are the primary restrictions on quantity, composition and toxicity for atmospheric emissions from a chemical agent destruction process (combustion and non-combustion emissions)? What testing is required to verify attainment of requirements?
- What information will the regulatory agencies require to approve the use of these technologies?
- What are the permitting requirements and schedule for treatment technology systems? Have these systems been permitted on other wastes and at what scale?
- How can the AltTech Panel obtain a copy of the state or federal regulations governing the management of hazardous wastes, water effluents, and air emissions?
- Has your state been granted authority to administer: the RCRA program? NPDES permits? Air permits? What role does the EPA play?
- Are mustard and/or VX listed as hazardous under state regulations?
- If not, what would each state need to know to determine whether they are characteristically hazardous under state regulations?
- Would a state RCRA permit be required to treat mustard (Maryland) or VX (Indiana) for each of the following processes:
- neutralization
- high temperature/high pressure hydrogen reduction
- low temperature/ambient pressure electrochemical oxidation
- molten metal bath agent destruction with recoverable by-products
- What specific regulatory subtitles would apply to each of the above treatment processes?
- If any of the treatment processes would have to be permitted under the state equivalent of RCRA Subtitle X, what experience has the state had in Subtitle X permitting?
- What are the steps involved in applying for a RCRA permit? How long does the process typically take from submission of an application to final approval?
- Are there different regulatory requirements for full-scale treatment and for bench or pilot-scale treatment for purposes of R&D? If so, what are the limitations on throughput under an R&D permit?
- What are the steps involved in applying for an air, NPDES or SPDES permit? How long does the process typically take from application submission to final approval?
- What are the steps involved in applying for a construction permit?
- What additional permits or approvals would be required prior to startup of operations?
- M4 Environmental asserts that its technology is a recycling process. Do the states of Maryland and
- Indiana concur with this characterization, or is a RCRA permit required? What about the other technologies?
- What air permits are required for a gas turbine/power generator fired with syngas (from agent)?
- Would it allay concerns if the agents were neutralized before being treated by the alternative technologies?
- Are there reasons to believe that any of these technologies would be prohibited in Maryland or Indiana?
- Would your state permit shipment of hydrolysate produced by agent neutralization to a toxic waste treatment facility?
- How does the Clean Water Act provision restricting disposal of agent-derived waste into navigable waters affect the disposal of agent hydrolysate in your state?
- How will the combustion of offgases from the Eco Logic process be regulated? What restrictions will be placed on the NOx emissions from the AEA process?