Description and Analysis of the VA National Formulary (2000)
Chapter: Appendix C: Additional Cost Information
APPENDIX C
Additional Cost Information
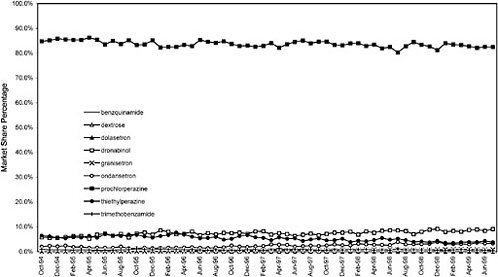
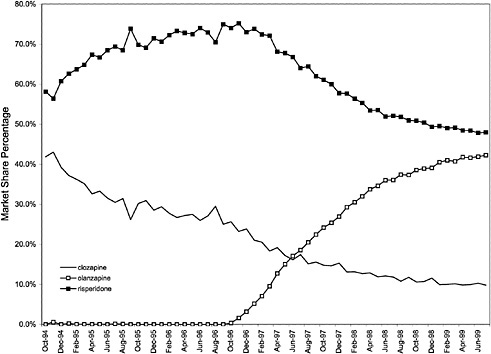
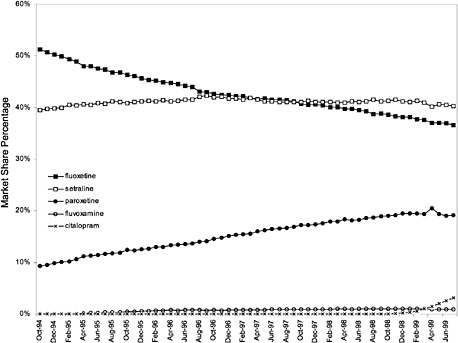
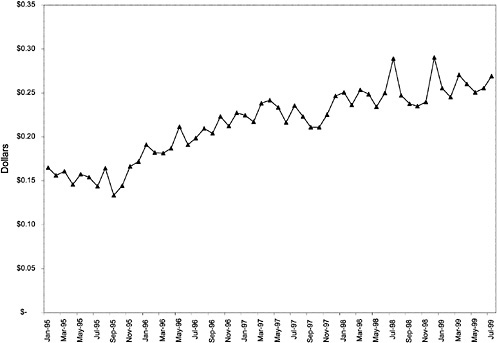
This appendix contains additional graphs that are relevant to the analysis of the potential cost effects associated with the National Formulary (see chapter 3 for discussion).

FIGURE C.4 Inhaled antiasthma agents—open class: Market share of pharmaceuticals within the class. NOTE: Some drug products (triamcinolone, salmeterol, pirbuterol, nedocromil, montelukast, fluticasone, mometasone, levalbuterol, isoetharine, flunisolide, and epinephrine) had negligible market share and were removed for clarity.

FIGURE C.6 Nonsteroidal anti-inflammatory drugs—open class: Market share of pharmaceuticals within the class. NOTE: Some drug products (tolmetin, phenylbutazone, meclofen, ketoprofen, flurbiprofen, fenoprofen, diflunisal, sulindac, and nabumetone) had negligible market share and were removed for clarity.
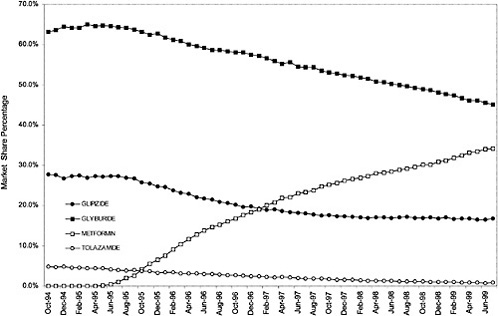
FIGURE C.7 Oral diabetic—open class: Market share of pharmaceuticals within the class. NOTE: Some drug products (dextrose, diazoxide, tolbutamide, acarbose, troglitazone, rosiglitazone, repaglinide, miglitol, chlorpropamide, glucose, glucagon, acetohexamide, and glimepirode) had negligible market share and were removed for clarity.
TABLE C.1 Implementation Date of VHA National Contracts Across the VISNs
|
ACE inhibitors |
|||||||
|
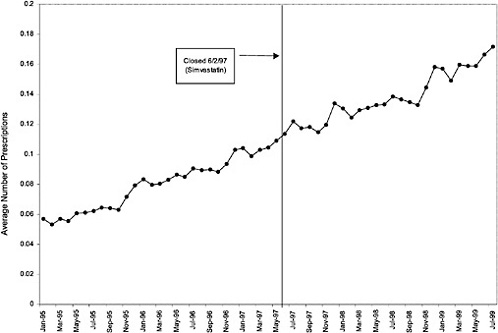
HMG CoA Ris lovastatin/simvastatin |
LHRHs goserelin |
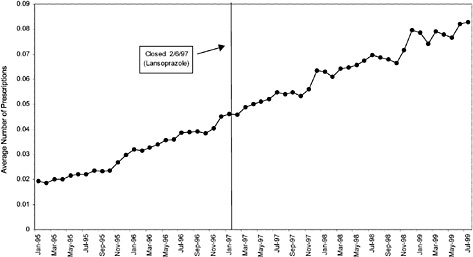
PPIs lansoprazole |
lisinopril |
fosinopril |
H2R blockera famotidine |
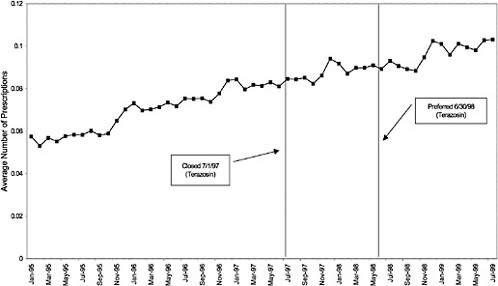
Alpha blockersb terazosin |
|
|
National |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
7/1/1997–6/30/1998 |
|
VISN 1 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN2 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN3 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN4 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN 5 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN 6 |
06/22/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN7 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN 8 |
07/02/1997 |
02/13/1997 |
03/06/1997 |
11/20/1997 |
02/12/1998 |
9/9/1996–9/8/1998 |
08/01/1997 |
|
VISN 9 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN 10 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
08/09/1996 |
07/01/1997 |
|
VISN 11 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN 12 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN 13 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN 14 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN 15 |
10/28/1997 |
06/11/1996 |
06/11/1996 |
10/28/1997 |
06/11/1996 |
01/02/1997 |
07/17/1997 |
|
VISN 16 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN 17 |
06/02/1997 |
01/01/1997 |
01/01/1997 |
10/01/1997 |
12/01/1998 |
08/09/1996 |
07/01/1997 |
|
VISN 18 |
06/02/1997 |
01/13/1997 |
02/06/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
07/01/1997 |
|
VISN 19 |
7/1/1997–9/1/1997 |
3/1/1997–7/1/1997 |
3/1/1997–4/1/1997 |
2/1/1997–12/1/1997 |
2/1/1997–12/1/1997 |
9/1/1996–12/1/1996 |
7/1/1997–10/1/1997 |
|
VISN 20 |
06/01/1997 |
12/01/1996 |
05/01/1997 |
10/20/1997 |
01/12/1998 |
8/9/1996–8/8/1998 |
7/1/1997–6/30/1998 |
|
VISN 21 |
06/02/1997 |
12/20/1996 |
01/24/1997 |
09/19/1997 |
09/19/1997 |
07/26/1996 |
08/15/1998 |
|
VISN 22 |
07/02/1997 |
02/13/1997 |
03/06/1997 |
11/20/1997 |
05/12/1998 |
9/9/1996–9/8/1998 |
08/01/1997 |
|
a H2R blockers were closed (8/9/1996) and reclassified as open (8/8/1998). b Alpha-blockers were closed (7/1/1997) and reclassified as preferred (6/30/1998). |
|||||||
TABLE C.2 Regression Results on Natural Logarithm of Spending per Veteran Outpatient User
|
Variable |
ACEI |
Alpha |
HMG |
PPI |
CCB |
H2R |
|
Closed |
-0.185 (8.60) |
-0.193 (9.10) |
-0.085 (5.92) |
-0.076 (5.45) |
-0.529 (11.26) |
|
|
Subsequent change in formulary status* |
-0.089 (3.93) |
-0.195 (4.91) |
-0.0605 (7.73) |
|||
|
preferred |
0.076 (1.83) |
|||||
|
% Male |
-0.007 (1.99) |
0.013 (2.69) |
0.017 (4.87) |
0.041 (12.03) |
0.010 (2.20) |
0.005 (0.46) |
|
%<45 years |
0.0001 (0.06) |
0.011 (3.33) |
-0.002 (0.85) |
0.003 (1.11) |
0.004 (1.32) |
0.022 (3.13) |
|
% 45–65 |
0.019 (2.96) |
-0.001 (0.14) |
-0.005 (0.79) |
-0.050 (7.99) |
-0.020 (2.38) |
0.011 (0.63) |
|
Time |
-0.019 (15.27) |
-0.046 (24.45) |
0.036 (29.43) |
0.054 (43.48) |
0.010 (6.06) |
0.014 (3.15) |
|
Time2 |
0.0002 (8.55) |
-0.0004 (10.86) |
-0.0002 (10.56) |
-0.003 (21.11) |
-0.0002 (8.57) |
-0.0004 (5.74) |
|
Constant |
1.203 (3.01) |
-1.819 (3.39) |
-0.642 (1.66) |
-1.941 (5.16) |
1.362 (2.71) |
-0.388 (0.35) |
|
VISN fixed-effects included |
||||||
|
R2 |
0.83 |
0.83 |
0.92 |
0.96 |
0.66 |
0.66 |
|
F |
198.43 |
201.2 |
507.5 |
944.3 |
85.86 |
80.12 |
|
NOTE: ACEI = angiotensin converting enzyme inhibitor; alpha = alpha blocer; CCB = calcium channel blocker; HMG = hydroxymethylglutaryl coenzyme A reductase inhibitor; PPI = proton pump inhibitor; VISN = veteran integrated service network; t statistics in parentheses. * For ACEIs, this variable represents the addition of a second closed drug, fosinopril, to the National Formulary. For alpha blockers, this variable represents the change in formulary status from closed to preferred. For H2R blockers, this variable represents the reopening of the class. |
||||||
TABLE C.3 Regression Results on Natural Logarithm of Inpatient Discharge Rates *