Reference Manual on Scientific Evidence: Fourth Edition (2025)
Chapter: Reference Guide on Exposure Science and Exposure Assessment
Reference Guide on Exposure Science and Exposure Assessment
M. ELIZABETH MARDER AND JOSEPH V. RODRICKS
M. Elizabeth Marder, Ph.D., is Staff Toxicologist, California Department of Public Health (previously Senior Environmental Scientist, California Environmental Protection Agency’s Office of Environmental Health Hazard Assessment), and Assistant Adjunct Professor of Environmental Toxicology, University of California at Davis.
[Note that the views expressed are those of the author and do not represent the California Department of Public Health, the California Environmental Protection Agency, or the State of California.]
Joseph V. Rodricks, Ph.D., is founding principal of ENVIRON (now Ramboll).
[Note that the views expressed are those of the author and do not represent Ramboll.]
CONTENTS
Introduction to Exposure Science
Relevance to Disease Causation and Risk Assessment
Understanding Human Exposure: Key Concepts
Characteristics of the Exposed Individual or Population
Introduction to Exposure Assessment
Exposure Assessment Considerations for Chemical Stressors
Problem Formulation: Why Conduct a Given Exposure Assessment?
Quantification of Exposure to Chemical Stressors
Integrated Exposure Assessment
Exposure Assessment of Biologic and Physical Stressors
How Exposure Science Informs Evaluation of Disease Causation in Individuals
How Exposure Science Informs Evaluation of Risk in Populations
Evaluating the Scientific Quality of an Exposure Assessment
Qualifications of Exposure Scientists or Other Exposure Assessors
Is the Proposed Expert Established Professionally in the Field?
Does the Proposed Expert Have Experience Relevant to the Topic or Method of Interest?
Appendix A: Units of Exposure and Dose
References on Law and Exposure Science
References on Exposure Science
Select Guidelines, Reports, and Other Technical Documents
FIGURES
1. Illustration of source-to-outcome framework for exposure science
2. Examples of exposure sources across a product life cycle
TABLES
Introduction to Exposure Science
Humans and other organisms regularly come into contact with a wide range of agents that directly or indirectly result in some form of adverse effect or harm. Generally referred to as stressors, these agents are typically chemical, physical, or biologic in nature but may also be psychosocial. Exposure science is a distinct discipline that studies individuals and populations and their behaviors related to contact with stressors, the nature and extent of such contact, and the fate of these stressors in the environment and in organisms over space and time. Though the origins of exposure science date to the ancient Greek physician Hippocrates, modern exposure science is rooted in the industrial-hygiene and radiation health-physics practices of the last century, and the importance of this field is increasingly recognized.1
Exposure science has a complementary role with epidemiology2 and toxicology,3 two disciplines devoted to understanding the inherent potential hazard of a given stressor. This is particularly important in the context of risk assessment, which is a formal process by which the nature and probability of adverse health effects in organisms exposed to a given stressor can be estimated. Given that a dose only results from exposure, exposure science is intrinsically linked with one of the fundamental tenets of human health risk assessment, the toxicological concept attributed to Paracelsus that “the dose makes the poison.” Characterization of exposure plays a key role in environmental epidemiology, as such studies are designed to ascertain whether or not there is an association between exposure to a particular stressor of interest and a health effect in humans. More generally, in order to evaluate whether individuals or populations exposed to a stressor are at risk of harm,4 or have actually been harmed, information on toxicity or hazard derived from epidemiological and toxicological studies is needed, as is information
1. In 2012, the National Research Council (NRC) published a seminal report on the field, Exposure Science in the 21st Century: A Vision and a Strategy. This report defines exposure science more formally as “the collection and analysis of quantitative and qualitative information needed to understand the nature of contact between receptors and physical, chemical, or biologic stressors.” Nat’l Rsch. Council, Exposure Science in the 21st Century: A Vision and a Strategy 20 (2012), https://doi.org/10.17226/13507. Following release of this report, the Exposure Science in the 21st Century Federal Working Group (ES21 FWG), consisting of twenty federal organizations, was established to create a common understanding of exposure science, expand partnerships, and drive research across the federal government. In the past decade, federal exposure science programs have rapidly expanded to address increasing demand for exposure data and methods, including programs within the National Exposure Research Laboratory and the National Center for Computational Toxicology of the U.S. Environmental Protection Agency (EPA).
2. See Steve C. Gold et al., Reference Guide on Epidemiology, in this manual.
3. See David L. Eaton et al., Reference Guide on Toxicology, in this manual.
4. See, e.g., Rhodes v. E.I. du Pont de Nemours & Co., 253 F.R.D. 365 (S.D. W. Va. 2008) (suit for medical monitoring costs because exposure to perfluorooctanoic acid (PFOA) in drinking water allegedly caused an increased risk of developing certain diseases in the future); In re Welding
on the exposures incurred by those individuals or populations. This interrelationship is a central theme in a report by the National Academies of Sciences, Engineering, and Medicine (the National Academies) that examines how data being generated today across disciplines can be used in risk assessment applications.5
This reference guide emphasizes assessment of exposure to chemical stressors, which serves as a model for considerations of approaches to characterization (e.g., direct measurement, indirect estimation, exposure reconstruction) and quantification of exposure (see sections titled “Exposure Assessment Considerations for Chemical Stressors” and “Quantification of Exposure to Chemical Stressors”). However, some considerations specific to exposure of certain biological and physical stressors are discussed briefly (see “Exposure Assessment of Biologic and Physical Stressors”).
Key Concepts
Exposure science describes the environment, the behavior of stressors in the environment, the characteristics and activities of individuals and populations, and the processes that lead to human contact with and uptake of these stressors. For exposure to occur, the stressor and an individual (or population) need to come together in both space and time. The time of continuous contact between the stressor and individual (or population) is the exposure period. Exposure can be described in terms of intensity or magnitude (how much), frequency (how often) and duration (how long) of contact at an external boundary (those characterized by external exposure surfaces, such as the surface of the skin). For most stressors, both intensity or magnitude and route of exposure are critical characteristics in determining adverse effects. In addition, factors such as the frequency, duration, and timing of exposure (e.g., life-stage considerations, acute versus chronic exposure) are influential in determining adverse effects. These factors depend on the source of the stressor, its transport and fate, its persistence in the environment, and the activities of individuals that lead to contact with the stressor.
Exposure science also extends beyond the exposure event itself (i.e., the point of contact with a stressor) to study and describe the processes that affect the transport and transformation of a stressor, or agent, from its source to a dose at a target internal organ, tissue, or toxicity pathway associated with a disease process. Exposure scientists use a broad range of data and information (empirically derived and modeled) to describe conditions in the real world that could lead to human health risks, typically by conducting an exposure assessment. An exposure
Fume Prods. Liab. Litig., 245 F.R.D. 279 (N.D. Ohio 2007) (exposure to manganese fumes allegedly increased the risk of later developing brain damage).
5. Nat’l Acads. of Scis., Eng’g & Med., Using 21st Century Science to Improve Risk-Related Evaluations (2017) [hereinafter National Academies 2017 Report], https://doi.org/10.17226/24635.
assessment is a process of estimating or measuring the magnitude, frequency, and duration of exposure to a stressor, and the characteristics of the population exposed. These exposure assessments, which link exposure to health outcomes and help regulators evaluate various options to manage exposures effectively, can be for current, prospective, and retrospective exposures.6 Ideally, an exposure assessment describes the sources, routes, pathways, and uncertainty; describes contact with agents as they occur in the real world at various life stages; and provides data to understand and quantify health outcomes as they occur in various populations.7 A source-to-outcome framework, as illustrated in Figure 1, helps to visualize the information and processes important for exposure science, including the major types of experimental and computational approaches to exposure characterization commonly utilized in exposure assessment. Historically, exposure assessment may have ended with the description of the exposure itself (sometimes framed as the potential dose to the body, e.g., prior to crossing a relevant absorption barrier like the skin), but it is now common for practitioners to quantify stressors present in blood, urine, or various tissues of the body as a result of current or past exposure—referred to as biomarkers of exposure—and even to quantify certain biological responses of concern triggered by exposure, referred to as biomarkers of effect.
Practitioners
It is important to note that not all exposure assessments are sufficiently complex to require an exposure scientist or similar exposure expert. Indeed, some assessments are relatively simple, with epidemiologists or toxicologists likely capable of estimating exposure in relatively straightforward scenarios, such as consumption of a residue in food or ingestion of a medication. In more complex cases, such as when
6. See, e.g., National Academies 2017 Report, supra note 5 (presents recommendations for integrating new scientific approaches into risk-based evaluations; chapter 2 covers advances in exposure science); Nat’l Rsch. Council, Risk Assessment in the Federal Government: Managing the Process (1983), https://doi.org/10.17226/366 (discusses past efforts to develop and use risk assessment guidelines and evaluates various proposals to modify the risk assessment procedures, including exposure assessment, used by regulatory agencies); Nat’l Rsch. Council, supra note 1; U.S. Env’t Prot. Agency, Guidelines for Human Exposure Assessment (2019) [hereinafter EPA Guidelines], https://perma.cc/7XGA-BFKJ (these present the current policies and practices of exposure assessors within EPA from scoping and problem formulation to presentation of results); Int’l Programme on Chem. Safety, World Health Org., WHO Human Health Risk Assessment Toolkit: Chemical Hazards (2d ed. 2021) [hereinafter WHO HHRA Toolkit], https://iris.who.int/handle/10665/350206 (section 3.3.4 covers exposure assessment, including a generic roadmap for exposure assessment in the context of human health risk assessment).
7. See, e.g., Nat’l Rsch. Council, supra note 1; Linda S. Sheldon & Elaine A. Cohen Hubal, Exposure as Part of a Systems Approach for Assessing Risk, 117 Env’t Health Persp. 1181, https://doi.org/10.1289/ehp.080040; EPA Guidelines, supra note 6; WHO HHRA Toolkit supra note 6.
 Source: Adapted from Figure 2-1 (“Conceptual overview of the scope of and common methods for exposure science”), National Academies of Sciences, Engineering, and Medicine. 2017. Using 21st Century Science to Improve Risk-Related Evaluations. Washington, DC: The National Academies Press. https://doi.org/10.17226/24635.
Source: Adapted from Figure 2-1 (“Conceptual overview of the scope of and common methods for exposure science”), National Academies of Sciences, Engineering, and Medicine. 2017. Using 21st Century Science to Improve Risk-Related Evaluations. Washington, DC: The National Academies Press. https://doi.org/10.17226/24635.exposures result from a stressor moving from sources through one or more environmental media, or when historic exposure profiles must be constructed, it is unlikely that toxicologists or epidemiologists will be able to offer appropriate qualifications, because more advanced modeling and integration of various exposure data are needed to characterize exposures. Given the heterogeneity of exposure science, it should not be surprising that practitioners (exposure scientists as well as related experts) come from a wide range of academic backgrounds. These backgrounds may include environmental health sciences (including some specializing in exposure science), industrial hygiene, environmental and analytical chemistry, chemical and environmental engineering, geology and hydrogeology, toxicology (toxicokinetic applications in particular), epidemiology, and even behavioral sciences (pertaining to those aspects of human behavior that affect exposures). As noted in Figure 1, exposure scientists may address issues of exposure and dose from a source to a biologically effective dose, but these experts are not typically qualified to address health effects of such exposure, which is typically the domain of experts in epidemiology, toxicology, and medicine. Further details on the qualifications of experts are offered in the last section of this reference guide (section titled “Qualifications of Exposure Scientists or Other Exposure Assessors” below).
Relevance to Disease Causation and Risk Assessment
In the evaluation of disease causation for an individual, exposure science is applied to characterize the individual’s contact with a stressor, either qualitatively or quantitatively, which is then linked to evidence of disease causation. Exposure assessment is also a major component of risk assessment. In this context, exposure assessments typically must identify and quantify the exposure of populations that are most highly exposed and populations that are most vulnerable, including all relevant exposure pathways (while ideally allowing the pathways to be identified and defined individually), and quantifying inherent uncertainty in the assessment itself.
Exposure Science and the Law
Exposure is the bridge between the presence of a stressor (or agent) in an environment and its ability to cause harm. Therefore, either contact or the plausibility of contact8 with a stressor by the individual or population in question must be established before a harmful level of exposure can be definitively determined to have occurred, which is a central challenge in legal cases. Exposure science evidence is particularly relevant in claims of toxic tort or product liability, in which exposure information is critical to effectively assess the liability of defendants in situations where plaintiffs are suing for damages to their health, or the health of their family members, or a group in a class-action claim. Legal actions to protect consumers are usually brought by agencies and groups, including nongovernmental organizations and affected industries, and less frequently by members of the public. Other areas where exposure science is necessary are in formulating a national environmental- or occupational-health-standard control strategy, in developing or updating state or local regulations to protect human health, and in setting safety standards used to protect our food supply or consumer products from contaminants. Not all legal questions concerning human exposures to potentially harmful substances require expert testimony. For example, when the magnitude of exposure is not relevant or is clearly evident (e.g., because a plaintiff
8. See, e.g., Kitzmiller v. Jefferson, No. 2:05-CV-22, 2006 WL 2473399 (N.D. W. Va. Aug. 25, 2006) (defendants offered expert testimony that plaintiff’s use of liquid cleaning agents containing benzalkonium chloride failed to show that she was exposed to benzalkonium chloride in the air); Hawkins v. Nicholson, No. 02-1578, 2006 WL 954654, at *4 (Vet. App. Mar. 2, 2006) (noting that “a veteran who served on active duty in Vietnam between January 9, 1962, and May 7, 1975, is entitled to a rebuttable presumption of exposure to Agent Orange”); In re Stand’n Seal, 623 F. Supp. 2d 1355 (N.D. Ga. 2009) (consumer use of spray-on product allegedly resulted in inhalation exposure to toxic substances, causing respiratory injuries).
was observed to take the prescribed amount of a prescription medicine or was observed being covered in a powder), expert testimony is not indicated. However, expert testimony may be needed in cases involving complex exposure scenarios or in which the magnitude of exposure itself is not a simple question of fact. Such expert testimony—in which a qualitative or quantitative estimate of the magnitude, duration, and frequency of the exposure—is critical to understanding the potential presence or absence of causality between the agent and the health effect, the latter of which typically is based on toxicological, clinical, or epidemiological evidence.
Understanding Human Exposure: Key Concepts
This section of the reference guide is descriptive rather than quantitative. It covers the various physical processes that lead to human exposures to stressors and introduces the terms that exposure scientists apply to those processes, as well as exposure-related characteristics of exposed individuals and populations. Given that exposure science overlaps with other disciplines, many of which use different terms for the same concepts, it is important to note that the definitions used in this reference guide reflect the field of exposure science.
Exposure vs. Dose
Exposure refers to the contact between an agent and the external boundary (exposure surface) of the body for a specific duration. In toxicology, this metric is sometimes also referred to as administered dose or external dose. In this reference guide the general term dose is used to refer to the amount of a stressor (e.g., a toxicant such as benzene) that, over a specified time period, enters the body after crossing an external exposure surface, such as the lungs. In certain contexts, dose may also be referred to as internal exposure, internal dose, or absorbed dose. However, the concept of dose is generally understood to represent the amount of the stressor that gets past the absorption barrier (e.g., lungs) and into circulation (e.g., into blood), a process referred to as uptake,9 while the amount of the contaminant that can interact with organs and tissues to cause biological
9. The capacity for a substance to be absorbed via uptake processes is a reflection of its bioavailability. Chemical properties, the physical state of the material to which an individual is exposed, the ability of the individual to physiologically absorb the chemical (e.g., nutritional status, gut flora activity), and even the exposure medium can all affect bioavailability.
effects can be referred to as a biologically effective dose or target dose depending on the context. Note that dose profiles are a function of time, and over time are dependent on factors described for exposure and pharmacokinetics/toxicokinetics (e.g., absorption into the body, distribution throughout the body, metabolism within the body, and elimination from the body).
Characteristics of the Exposed Individual or Population
The focus for human exposure science in the context of legal evidence is often the exposed individual, group, or population (sometimes referred to as the receptor or receptor population) and not the sources of the stressor, considering potential contact based on the individual or population’s location and behaviors. In this context, understanding the characteristics of human receptors, their behaviors, and the relationship between these factors and exposure is crucial for what is known as a receptor-based approach. There is inherent variability in exposure that occurs because of factors related to human behavior and characteristics that help determine an individual’s exposure to a stressor, such as location, occupation, activities within a location, socioeconomic status, consumer preferences, dietary habits, and other lifestyle choices. Such exposure factors include rates of ingestion (e.g., foods, water) or inhalation, factors affecting dermal exposure (e.g., skin surface area), activity factors (e.g., time spent in indoor environment, time spent exercising outdoors), or other factors (e.g., body weight, amount and frequency of consumer product use). Where appropriate, exposure assessors also consider characteristics that might increase exposure or predispose an individual, life stage,10 specific group, or population to greater health risk. These include factors associated with susceptibility, those in which there is an increased likelihood to be more affected by exposure to a stressor than the general population because of intrinsic factors, such as biological sex, life stage, and genetic polymorphisms. Other factors considered include those associated with vulnerability, such as economic, demographic, social, cultural, psychological, and physical states of the human receptor that influence patterns of exposure to environmental contaminants or alter the relationship between the exposure to environmental contaminants and the health effect of concern (e.g., built environment, access to health care, racism, discrimination). Behaviors relative to life stage can also be particularly influential determinants for exposure, especially for
10. An exposure assessor often needs to establish a dialogue with toxicologists/health scientists to consider whether a specific “window of susceptibility” during a given life stage is important to a particular risk assessment.
infants and toddlers and for a developing embryo/fetus during pregnancy. Note that in most cases, drivers for human activities are complex and cannot be predicted readily. Instead, human activities are often treated as variables described by population distributions based on available (e.g., observational or modeled) data.
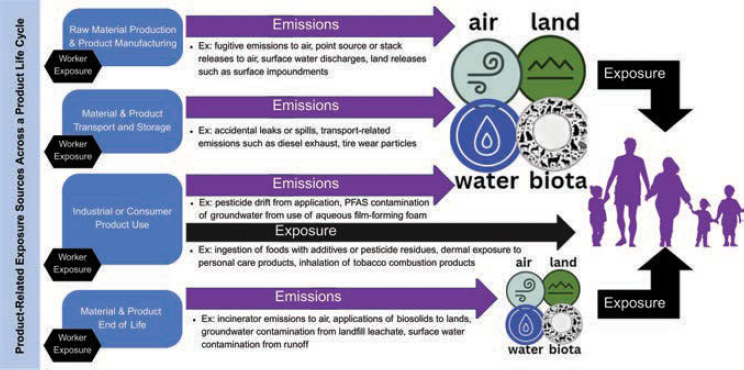
Exposure Sources
Human chemical exposure sources can be broadly grouped into near-field sources that are close to the exposed individual, such as consumer product use or occupational exposures, and far-field sources wherein individuals are exposed to chemicals via environmental media following release or use far away. As shown in Figure 2 above, which provides a broad overview of major types of exposure sources across a product’s life cycle, there are sources of intended exposure (e.g., dermal exposure to cosmetics or consumption of food additives) and unintended exposure (e.g., accidental releases or spills that reach environmental media such as air or water).
There are many chemicals for which intentional use will lead to human exposures, including substances added to food (and indeed food itself),11
11. The natural constituents of food include not only substances that have nutritional value, but also hundreds of thousands of other natural chemicals. This includes a range of compounds generally considered beneficial for human health (e.g., phytonutrients such as flavonoids and
cosmetics, personal care products, fibers and the colorants added to them, and medical products of many types, including pharmaceuticals; many of these uses are carefully regulated under applicable federal and state laws. In addition to exposure to chemicals that are used to manufacture a product, additional chemical exposures can arise during typical use of a product, such as the anticipated generation of tobacco combustion products from use of cigarettes or of thermal transformation productions from use of electronic nicotine delivery systems. Direct contact with products obviously results in some level of exposure. Here direct contact may mean ingestion via swallowing or other mechanisms, such as inhalation or contact with the skin. There are also some exposures that result from intentional use but are not as well understood, including those related to insertion of some medical devices, such as exposure to silicone and metals from certain types of implants. Generally, however, anticipated exposures resulting from intentional use are more readily quantifiable than those associated with unintended exposures.
Unintended exposures include exposure to known chemicals as well as to unknown chemicals, such as those resulting from unanticipated releases, impurities, or environmental reactions that result in the formation of new substances. Many unintended exposures result from deliberate uses of certain chemicals that, although not intended to lead to human exposures, will inevitably do so. Some amount of a pesticide applied aerially may drift into a nearby residential area, components of food packaging materials may migrate into food, and many types of household products are not intended for direct human ingestion or contact, but exposures nonetheless occur indirectly. As with packaging components, materials used in certain medical devices can contain chemicals that may ultimately leach or volatilize from the material and result in exposure, including plasticizers like phthalates in intravenous tubing and catheters. Ultimately, many exposures to a broad range of environmental contaminants and agents used in occupational settings are unintended; these are often exposures that may not be entirely mitigated by engineering controls and protective equipment, but can also include accidental releases. Unintended exposures are generally more difficult to identify and quantify than are intended exposures; such scenarios require significant expertise to appropriately characterize exposure(s).
polyphenols) as well as those that are harmful, including both toxins (toxic compounds naturally produced by living organisms, such as the cyanogenic glycosides produced by a variety of plants consumed as food, including almonds, cassava, sorghum, and stone fruits), and toxic chemicals taken up from the environment (such as arsenic or lead).
Exposure Routes
An exposure route is the way that a contaminant enters an individual at the point of contact.12 Inhalation of air containing a substance of interest (vapors as well as particulates) is one of the major routes of exposure.13 The internal exposure from inhalation is a function of not only the air concentration of the substance being breathed but also the breathing rate of the individual. The physical form of the substance in air will also influence what happens to the substance during inhalation. For example, chemicals that are in the vapor phase will remain in that physical state and will move to the lungs, where a certain fraction will pass through the lungs and enter systemic circulation. The extent to which different chemical substances pass through the lungs is dependent in large part upon their physical properties, particularly solubilities in both fatlike materials and water. Passage through cell membranes (of the cells lining the lungs) requires that substances have a degree of both fat solubility and water solubility. Certain fibrous materials (including but not limited to asbestos), particulate matter (including but not limited to combustion products), and dusts may also move through the airways and may reach the lungs, but to varying degrees. It is anticipated that at least some such materials will be trapped in the nose and excreted.
Generally, only very fine particles reach the lower lung area and are available for absorption systemically. Some particles may be deposited in the upper regions of the respiratory tract and then carried by certain physical processes to the pharynx and then be coughed up or swallowed. Thus, a variety of inhaled substances can enter the body through the gastrointestinal (GI) tract or the respiratory tract.14
Ingestion is another major route of exposure to substances in environmental media.15 Ingestion exposure can occur from intentional consumption of food,
12. EPA Guidelines, supra note 6.
13. See, e.g., Byers v. Lincoln Elec. Co., 607 F. Supp. 2d 840 (N.D. Ohio 2009) (welder inhaled toxic manganese fumes); O’Connor v. Boeing North Am., Inc., No. CV 00-0186 DT RCX, 2005 WL 6035256 (C.D. Cal. 2005) (alleged failure to monitor ambient air emissions of radioactive particles); In re FEMA Trailer Formaldehyde Prod. Liab. Litig., No. MDL 07-1873, 2009 WL 2382773 (E.D. La. 2009) (trailer residents exposed to formaldehyde).
14. Joseph V. Rodricks, From Exposure to Dose, in Calculated Risks: The Toxicity and Human Health Risks of Chemicals in Our Environment 28 (2d ed. 2006), https://doi.org/10.1017/CBO9780511535451.
15. See, e.g., Foster v. Legal Sea Foods, Inc., No. CCB-03-2512, 2008 WL 2945561 (D. Md. 2008) (hepatitis A allegedly contracted from eating undercooked mussels); Winnicki v. Bennigan’s, No. CIV.A. 01-3357 (JAG), 2006 WL 319298 (D.N.J. 2006) (alleged foodborne illness contracted from defendant’s restaurant led to renal failure and death); Palmer v. Asarco Inc., No. 03-CV -0498-CVE-PJC, 2007 WL 2254343 (N.D. Okla. 2007) (children allegedly ingested dust and soil contaminated with lead); Rhodes v. E.I. du Pont de Nemours & Co., 253 F.R.D. 365 (S.D. W. Va. 2008) (suit for medical monitoring costs because exposure to perfluorooctanoic acid (PFOA) in drinking water allegedly caused an increased risk of developing certain diseases in the future).
water, other liquids, and certain other substances designed for consumption, such as medicines or supplements. Ingestion exposure can also occur from the intentional or inadvertent nondietary ingestion16 of soil, dust, or other substances, or of chemical residues on surfaces or objects that are contacted via hand-to-mouth or object-to-mouth activity (especially relevant for young children); this includes incidental ingestion of substances such as cosmetics, personal care products, and certain medications applied to the mouth or lips. They are swallowed, enter the GI tract, and to greater or lesser degrees are absorbed into the bloodstream at various locations along that tract. This is often referred to as the oral route of exposure.
Dermal exposure, the remaining major route of exposure for substances in products and the environment, reflects contact with the largest organ of the body, the skin.17 The skin is composed of two layers, with a thin outer layer of squamous keratinocytes (called the epidermis or the stratum corneum) that is highly hydrophobic and provides the protective barrier function of skin. Beneath the epidermis is a much thicker living layer of cells including blood vessels, nerves, hair follicles, and sweat glands. As with the GI tract and the lungs, substances are absorbed through the skin to greater or lesser degrees, depending on their physical and chemical characteristics. Other factors also influence dermal absorption, including the specific surface involved, as the skin is not uniform in thickness or in its composition of skin surface fluids, its temperature, and whether occlusion (e.g., trapping material between skin and clothing) has occurred. The uptake of chemicals through these two skin layers is governed by diffusion, and therefore regulated by Fick’s law, which states that the rate of diffusion across a barrier will be directly proportional to the concentration gradient. Regardless of exposure route(s) of interest, it is important to note that, depending on the nature of the stressor present at an absorption boundary, in some cases harm can occur directly within the respiratory or GI tracts or on the skin before absorption occurs.18
Exposure Pathways
An exposure pathway reflects the course a stressor takes from the source to the point at which it reaches the human receptor of interest (e.g., individual, group,
16. Such nondietary ingestion occurs to some extent in all individuals. However, there are individuals for whom this can be more considerable, such as those with pica behavior or similar conditions.
17. See, e.g., United States v. Chamness, 435 F.3d 724 (7th Cir. 2006) (evidence that methamphetamine and the ingredients used in its manufacture are toxic to the eyes, mucous membranes, and skin supported sentencing enhancement for danger to human life).
18. Rodricks, From Exposure to Dose, supra note 14.
population).19 Exposure pathways analysis allows the identification of all the routes by which stressors from a given source may enter the body, because it identifies all relevant media of human contact into which the stressors migrate. To ensure thoroughness in the assessment, all conceivable pathways should be explicitly identified, with the understanding that ultimately some pathways will be found to contribute negligibly to the overall exposure. The simplest pathways are those described as direct exposure. For example, a substance, such as a noncaloric sweetener or an emulsifier, once added to food follows a simple and direct pathway to the people who ingest the food. The same can be said for pharmaceuticals, cosmetics, and other personal care products. Calculating exposure to such substances, as shown below in the section titled “Quantification of Exposure to Chemical Stressors,” is generally a straightforward process. Even in such cases, however, complexities can arise. For example, in the case of certain personal care products that are applied to the skin, there is a possibility of inhalation exposures to any substance in those products that can readily volatilize (move from a liquid to a gaseous state) at room temperatures. One physical characteristic of chemicals that exposure scientists need to understand is their capacity to volatilize. Not all chemicals are readily volatile, but inhalation routes can be significant for those that are volatile, regardless of their sources.20
Indirect pathways of exposure can range from the relatively simple to the highly complex. Many packaging materials are polymeric chemicals—very large molecules synthesized by causing very small molecules to chemically bind to each other (or to other small molecules) to make very long chemical chains. These polymers (polyethylene, polyvinyl chloride, polycarbonates, and others) tend to be physically very stable and chemically quite inert (meaning they have very low toxicity potential). But it is generally not possible to synthesize polymers without very small amounts of the starting chemicals (those small molecules, usually called monomers) remaining in the polymers. The small molecules can often migrate from the polymer into materials with which the polymer
19. See, e.g., SPPI-Somersville, Inc. v. TRC Cos., No. 07-5824 SI, 2009 WL 2612227, at *16 (N.D. Cal. 2009) (groundwater contamination claim was dismissed because there was no current pathway to exposure); United States v. W.R. Grace Co., 504 F.3d 745 (9th Cir. 2007) (affirming exclusion of report, but not expert testimony based on the report, identifying which pathways of asbestos exposure were most associated with lung abnormalities); Grace Christian Fellowship v. KJG Invs. Inc., 2009 WL 2460990, at *12 (E.D. Wis. 2009) (preliminary injunction was denied because the plaintiff did not establish that a complete pathway currently existed for toxicants to enter the building); Nat’l Exposure Rsch. Lab’y, U.S. Env’t Prot. Agency, Scientific and Ethical Approaches for Observational Exposure Studies, Doc. No. EPA 600/R-08/062 (2008), https://perma.cc/QM3KTAY6; U.S. Env’t Prot. Agency, Exposure Factors Handbook (2011), https://perma.cc/D8AR-5FT5.
20. Inhalation exposures to nonvolatile chemicals can occur if they are caused to move into the air as dusts. See Nat’l Rsch. Council, Human Exposure Assessment for Airborne Pollutants: Advances and Opportunities (1991), https://doi.org/10.17226/1544.
comes into contact. If those materials are foods or consumer products, people consuming those foods or otherwise using those products will be exposed.
Some amount of the pesticides applied to food crops may remain behind in treated foods and be consumed by people.21 This last pathway can become more complicated when treated crops are used as feed for animals that humans consume (meat and poultry and farm-raised fish) or from which humans obtain food (milk and eggs). Exposure scientists who study these subjects thus need to understand what paths pesticides follow when they are ingested by farm animals used as food. The same complex indirect pathways arise for some veterinary drugs used in animals from which humans obtain food.22 In the realm of environmental contamination, pathways can multiply, at which point the problem of exposure assessment can become even more complex, as exposure must be aggregated across relevant pathways. Example sources of environmental contamination include air emissions from manufacturing facilities and from numerous sources associated with the combustion of fuels and other organic materials.23 Similar emissions that reach water supplies, including ground water used for drinking water or for agricultural applications, can result in human exposures through drinking water and food.24 Contaminants of drinking water that are volatile can enter the air when water is used for bathing, showering, and cooking. In particular, contamination of air in homes and other buildings because of the presence of volatile chemical contaminants in the water beneath those structures, a process referred to as vapor intrusion, is of increasing concern.25 Wastes from industrial processes and many kinds of consumer wastes can similarly result in releases to air and water.26 In some cases, emissions to air can lead to the deposition of contaminants in soils and household dusts; this type of contamination is usually
21. Other pathways for pesticide exposure include spraying homes or fields. See, e.g., Kerner v. Terminix Int’l, Co., No. 2:04-CV-735, 2008 WL 341363 (S.D. Ohio 2008) (pesticides allegedly misapplied inside home); Brittingham v. Collins, No. CIV. AMD 06-1952, 2008 WL 678013 (D. Md. Feb. 26, 2008) (crop-dusting plane sprayed plaintiff’s decedent); Haas v. Peake, 525 F.3d 1168 (Fed. Cir. 2008), overruled by Procopio v. Wilkie, 913 F.3d 1371 (Fed. Cir. 2019) (veteran claimed exposure to Agent Orange).
22. Patricia Frank & James H. Schafer, Animal Health Products, in Regul. Toxicology 70 (Shayne C. Gad ed., 2d ed. 2001).
23. See, e.g., Nat. Res. Def. Council, Inc. v. EPA, 489 F.3d 1250 (D.C. Cir. 2007) (vacating EPA rule for solid waste incinerators); Kurth v. ArcelorMittal USA, Inc., No. 2:09-CV-108RM, 2009 WL 3346588 (N.D. Ind. 2009) (defendant manufacturers allegedly emitted toxic chemicals, endangering schoolchildren); Am. Indus. Hygiene Ass’n, Guideline on Occupational Exposure Reconstruction (Susan Marie Viet et al. eds., 2008).
24. United States v. Sensient Colors, Inc., 580 F. Supp. 2d 369, 373 (D.N.J. 2008) (leaching lead threatened to contaminate ground water used for drinking).
25. Interstate Tech. & Regul. Council (ITRC), Vapor Intrusion Pathway: A Practical Guideline (Jan. 2007), https://perma.cc/N3ED-9DJT.
26. Am. Farm Bureau Fed. v. EPA, 559 F.3d 512 (D.C. Cir. 2009) (EPA outdoor air pollution standards).
associated with nonvolatile substances. Some such substances may remain in soils for very long periods; others may migrate from their sites of deposition and contaminate ground water, whereas others may degrade relatively quickly. All such issues regarding the movement of chemicals from their sources through the environment to reach human populations come under the heading of chemical fate and transport.27 Transport concerns the processes that cause chemicals to follow certain pathways from their sources through the environment, and fate concerns their ultimate disposition—that is, the medium in which they finally reside and the length of time that they might reside there. Fate and transport scientists have models available to estimate the amount of chemical that will be present in that final environmental medium, often referred to as the exposure medium.28 Some discussion of the nature of these models is offered below in the sections titled “Exposure Assessment Considerations for Chemical Stressors” and “Quantification of Exposure to Chemical Stressors.” One final feature of pathways analysis that should be noted concerns the fact that some chemicals degrade rapidly when they enter the environment, others slowly, and some not at all, or only exceedingly slowly.
The study of environmental persistence of different chemicals is a significant feature of exposure science; its goal is to understand the chemical nature of the degradation products and the duration of time the chemical and its degradation products persist in any given environmental medium. Most inorganic chemicals are highly persistent; metals that become contaminants may change their chemical forms in small ways (lead sulfide may convert to lead oxide), but the metal persists forever (although it may migrate from one medium to another). Most organic chemicals degrade in the environment as a result of their exposure to light, to microorganisms present in soils and sediments, and to other environmental substances. But many organic substances (e.g., polyfluorinated substances such as perfluorooctanoic acid (PFOA), polychlorinated biphenyls (PCBs), and chlorinated dioxins such as dichloro-diphenyl-trichloroethane (DDT)) are quite resistant to degradation and may persist for unexpectedly long periods (although even these ultimately degrade). Exposure assessors also need to be aware of the possibility that the degradation products of certain chemicals may be as toxic, or even more toxic, than the chemicals themselves. The once widely used solvents trichloroethylene and perchloroethylene (tetrachloroethylene) are commonly found in ground water. Under certain conditions, these compounds degrade by processes that lead to the replacement of some chlorine atoms by hydrogen atoms. One product of such degradation is the more hazardous chemical vinyl
27. The common phrase used by exposure scientists is “fate and transport.” In fact, transport takes place and has to be understood before fate is known.
28. In the context of exposure science, the term “final” refers to the medium through which people become exposed. A chemical may in fact continue to move to other media after that human exposure has occurred.
chloride (vinyl chloride monomer or chloroethene), the presence of which in drinking water should not be ignored in an assessment.
A description of pathways is the critical step in characterizing exposure and, especially for environmental contaminants, must be done with thoroughness. Are all conceivable pathways accounted for? Have some pathways been eliminated from consideration, and if so, why? Are any environmental degradation products of concern? Only with adequate description can adequate quantification (see section titled “Quantification of Exposure to Chemical Stressors” below) be accomplished.
Introduction to Exposure Assessment
The primary objective of an exposure assessment is to estimate exposure to the stressor(s) of concern to the human receptor. The completion of an exposure assessment provides the information needed (the amount or concentration and duration of exposure) by epidemiologists and toxicologists, who will have information on the adverse health effects of the chemicals involved and on the relationships between those effects and the resulting dose.29 It is important to note that exposure assessment, and underlying exposure data, can directly contribute to sources of error in epidemiological studies, as discussed in the section titled “Sources of Error in Epidemiologic Studies” of the Reference Guide on Epidemiology, in this manual.30 Examples of such sources of error are included in that reference guide and include issues such as methods for imputation of measurements below a method limit of detection (see also “Data for Exposure Assessment: Measurements and Models” below).
As discussed in the next section, exposure assessments can be directed at exposures that occurred in the past, those that are currently occurring, or those that will occur in the future should certain actions be taken (e.g., the entry of a new product into the consumer market or the installation of new air pollution controls). Some aspects of exposure assessment may vary, based on application. Note that various regulatory programs publish their own guidance documents.31 As a field, exposure science has moved beyond the classical, source-oriented
29. See Steve C. Gold et al., Reference Guide on Epidemiology, and David L. Eaton et al., Reference Guide on Toxicology, in this manual. See also, e.g., White v. Dow Chem. Co., 321 Fed. App’x 266 (4th Cir. 2009) (plaintiff must show more than possible exposure; must show concentration and duration); Anderson v. Dow Chem. Co., 255 Fed. App’x 1 (5th Cir. 2007) (lawsuit dismissed because uncontested data showed that magnitude and duration of exposure was insufficient to cause adverse health effects).
30. See Steve C. Gold et al., Reference Guide on Epidemiology, in this manual, for a discussion on sources of error in epidemiology studies.
31. See, e.g., U.S. Env’t Prot. Agency, Exposure Factors Handbook (2011), https://perma.cc/D8AR-5FT5.
approaches (those that measure a stressor at its source and then estimate how much reaches an individual or population) to incorporate more human receptor-oriented approaches (those that measure or model a stressor at the individual or population level). Exposure assessments have also become more sophisticated given advances in technologies: These range from dramatic improvements in sensitivity and specificity of targeted analytical methods and incorporation of nontargeted analytical methods to the use of remote and personal sensors/dosimeters in the measurement of exposure, as well as more robust modeling approaches that have allowed for development of much more refined exposure profiles.
Exposure assessment is generally intended to answer the following questions:
- Who has been or could become exposed to a specific stressor(s) arising from one or more specific sources? Is it the entire general population, or is it a specific subpopulation (e.g., those residing near a certain manufacturing or hazardous waste facility; infants and children; or workers)?32
- What specific stressors comprise the exposures?
- What are the pathways from the source of the stressor to the exposed population? Pathways include direct product use, or those so-called indirect pathways in which the stressor moves through one or more environmental media to reach the media to which people are exposed (air, water, foods, soils, and dusts). Understanding pathways is necessary to understanding exposure routes (below) and quantifying exposures.
- By what routes are people exposed? Routes include ingestion, inhalation, and dermal contact.33 Identifying exposure routes is important because those routes affect the magnitude of ultimate exposures and because they often affect health outcomes.
- What is the magnitude and duration of exposure incurred by the population of interest? Magnitude (often reported as concentration, amount, or intensity) is the amount of a stressor entering the body or contacting the surface of the body, usually over some specified period of time (often over a day).34 Duration refers to the number of days over which exposure occurs. Has exposure changed over time? Note that
32. See, e.g., Hackensack Riverkeeper, Inc. v. Del. Ostego, 450 F. Supp. 2d 467 (D.N.J. 2006) (river and bay users alleged that hazardous waste runoff and emissions polluted the water); Bradford v. Citgo Petroleum Corp., 237 So. 3d 648 (La. Ct. App. 2018) (affirming jury award where the trial court was given “significant circumstantial evidence” that tied plaintiffs living/working/socializing in areas around the CITGO facility to exposures to the subject chemical spills and emissions).
33. Note that additional routes of exposure (e.g., injection, ocular exposure) may be relevant for some pharmaceuticals, diagnostics, and medical devices.
34. Shorter periods of time may be used when the concern is very short-term exposures to chemicals that have extremely high toxicity.
- exposures can be intermittent or continuous and can be highly variable, especially for some air contaminants.
Exposure Assessment Considerations for Chemical Stressors
Problem Formulation: Why Conduct a Given Exposure Assessment?
What an exposure assessment is intended to inform will guide the direction of the assessment itself, including the level of the complexity involved (e.g., screening-level assessment). A specific goal of a given exposure assessment might be identifying exposed individuals, groups, or populations, screening chemicals for potential exposure, or identifying source(s) of contamination. Exposure assessments are carried out for a wide variety of reasons, including use in risk assessment, identifying trends in measurements, mitigation efforts, regulatory decision making, priority setting, and epidemiological studies. For example, an assessment completed as part of a regulatory action may involve certain types of legal considerations (e.g., statutory requirements, mandates under a regulator program), whereas an assessment completed as part of an epidemiologic investigation would not.
Context
The development of exposure estimates can follow different methodological approaches depending on the needs of the evaluation to be carried out. For example, if the interest is directed toward personal exposure, the level of exposure should be measured or estimated at the point of contact with the subject (e.g., dermal, inhalation exposure). If exposure is associated with a specific scenario, as is common with near-field exposures, the exposure can be estimated or measured and, to subsequently refine the estimate, combined with information relating to the frequency and duration of the exposure. An exposure estimate can also be carried out with a retrospective approach (e.g., development of historical profile of exposure, reconstruction of exposure from levels of the chemical or metabolites in an individual) or prospective approach (e.g., estimate of present or future exposures). The prospective approach is typically used for regulatory purposes. The retrospective approach can be used, for example, to characterize exposure based on measures or estimates of concentrations in environmental media but can also be used to estimate past occupational exposures for an individual or a population in relation to specific tasks or professional roles held. Such
retrospective exposure assessments are common in toxic tort claims. Note that regardless of whether a prospective or retrospective approach is warranted, a traditional exposure assessment can be designed to consider an exposure resulting from a single identified source and pathway, or from combined exposures to a single chemical resulting from multiple sources, and/or multiple routes and pathways (typically referred to as aggregate exposure).
An aggregate exposure assessment approach is commonly used when humans can be exposed to a single contaminant in various ways. For example, if residues of the same pesticide could be found on multiple foods, in water, and/or in products used in and around the home, then an individual might have exposure via dermal contact, inhalation, ingestion, and other routes. Note that a cumulative exposure assessment can also be undertaken if there is a need to estimate exposure to multiple stressors by multiple routes and/or multiple pathways. Such cumulative exposure assessments are typically conducted for contaminants that produce toxic responses by the same mode of action, or when a population in a specific location is exposed to a variety of stressors.35 The United States Environmental Protection Agency (EPA) has developed guidance for these assessments,36 particularly because cumulative, community-based assessments that can characterize exposures or risks that disproportionately and unfairly affect certain communities are a cornerstone of assessments of environmental justice issues.
Data for Exposure Assessment: Measurements and Models
As discussed previously, exposure science has moved from a focus on classical source-oriented approaches to incorporate more receptor-oriented approaches. Exposure measures have significantly evolved from limited descriptions of a chemical or other stressor in environmental media, food, consumer products, or biological specimens. Biomonitoring data in particular are increasingly available, both in terms of chemical coverage and diverse populations. Biomonitoring refers to the measurement of cellular, biochemical, analytical, or molecular measures obtained from biological media (e.g., tissues, cells, fluids) that can be used to monitor the presence of (1) a chemical in the human body, (2) biological
35. See, e.g., Diné Citizens Against Ruining Our Env’t v. Haaland, 59 F.4th 1016 (10th Cir. 2023) (finding that Bureau of Land Management acted arbitrarily and capriciously by failing to account for the cumulative impact of hazardous air pollutant emissions from the more than 3,000 wells; these emissions could result in long-term exposure for individuals living in or visiting the San Juan Basin).
36. See, e.g., U.S. Env’t Prot. Agency, Exposure Assessment Tools by Tiers and Types—Aggregate and Cumulative, https://perma.cc/LC2L-28K5.
responses, or (3) adverse health effects.37 These data are typically combined with pharmacokinetic models that simulate the distribution and movement of chemicals within a living system to reconstruct or estimate the amount of chemical to which a person was exposed. Biomonitoring data are particularly useful for reconstruction of aggregate and cumulative exposure as these measures reflect exposure to chemical(s) of interest from all sources, routes, and pathways as well as uptake and accumulation. However, that does mean that these measures are not source or pathway specific and, depending on the scenario and other available data, it may not be possible to identify specific sources or routes of exposure—which limits utility for certain types of assessments.
Biomarkers relevant to exposure assessment include biomarkers of exposure, biomarkers of effect, and biomarkers of susceptibility. The type of biomarker and the biological matrix in which it can be measured are dependent not only on the chemical of interest and its properties, but on inter-individual variability of intrinsic and extrinsic factors that can modify relevant processes, such as metabolism and excretion (e.g., cigarette smoking induces the activity of human enzymes involved in metabolism of various exogenous substances; genetic polymorphisms in such enzymes).
Biological half-life, the length of time required for the concentration of a particular substance to decrease to half of its starting dose in the body, can be highly variable across species and among individuals of the same species. Consideration of biological half-life is particularly important when evaluating biomonitoring data as some chemicals, such as bisphenol A (BPA) and analogues, have an overall biological half-life on the order of hours, while other chemicals, such as dioxins and polychlorinated biphenyls (PCBs), range from years to over a decade. Matrix-specific biological half-life can further complicate this. For example, lead is cleared relatively quickly from the blood and soft tissues with a half-life of one to two months, but is cleared much more slowly from bones, with a half-life of years to decades. As such, biomarkers can be of varying utility depending on the chemical of interest, window of exposure, and sampling period. More information on application of biomonitoring in exposure assessment can be found elsewhere, including the EPA website.38
The EPA has developed working definitions for the types of biomarkers summarized briefly here using examples from pesticide risk assessment.39 Biomarkers of exposure are used to assess the amount of a chemical that is present within the body. Measures include the chemical itself (the most specific biomarker of exposure), chemical metabolites (specificity varies; if a nonspecific metabolite is
37. Nat’l Rsch. Council, Human Biomonitoring for Environmental Chemicals (2006), https://doi.org/10.17226/11700.
38. U.S. Env’t Prot. Agency, Exposure Assessment Tools by Approaches—Exposure Reconstruction (Biomonitoring and Reverse Dosimetry), https://perma.cc/FCQ4-P7P9.
39. U.S. Env’t Prot. Agency, Defining Pesticide Biomarkers, https://perma.cc/5UT2-XEQ8.
measured, additional information is needed to determine to which specific chemical the original exposure occurred), and endogenous surrogates. Biomarkers may also include a response within the body that is highly characteristic of a chemical or class of chemicals (this is the least specific biomarker of exposure, as there are many factors that can influence endogenous responses, such as inhibition of butyrylcholinesterase). Biomarkers of effect are indicators of a change in biologic function in response to an exposure (e.g., blood cholinesterase is typically depressed following exposure to organophosphate pesticides), and thus more directly related to insight into the potential for adverse health effects compared with biomarkers of exposure. Biomarkers of susceptibility are factors that result in certain individuals being more sensitive to a given exposure; these biomarkers are therefore more directly related to the potential for adverse health effects than biomarkers of exposure. Examples of biomarkers of susceptibility include genetic factors, nutritional status, lifestyle, and age, among others.
For decades the U.S. Centers for Disease Control and Prevention (CDC) was the leading source of biomonitoring data (with over 300 chemicals measured in biological specimens, such as blood or urine from a nationally representative population sample as part of the National Health and Nutrition Examination Survey). However, there are now thousands of individual studies reporting biomonitoring data available for consideration. Those conducting biomonitoring studies have radically diversified, and they include academic research centers across the United States, state and local biomonitoring programs, and even nongovernmental organizations. There are now even direct-to-consumer tests for select environmental contaminants facilitated via lab draws or a home testing kit. Note that not all biomonitoring data will be of sufficient quality for use in exposure assessment; the CDC has developed specific guidance regarding assessing the quality of biomonitoring data. Information is available on the CDC National Biomonitoring Program website.40
Advances in technologies—from the use of remote and personal sensor systems to new molecular technologies and computational modeling—allow for rapid development of comprehensive exposure profiles. The use of low-cost sensors is also expanding participatory science, including community-based exposure characterization,41 which coincides with greater availability of personal data from sources such as social media and wearable devices. Such data are increasingly used for exposure assessment, as they are able to provide insight into exposure on
40. National Biomonitoring Program, U.S. Ctrs. for Disease Control & Prevention, https://perma.cc/CYC7-EX5F.
41. See, e.g., Nat’l Inst. Env’t Health Sci., Community-Engaged Research and Citizen Science, https://perma.cc/XGX6-ZV7H; U.S. Env’t Prot. Agency, Participatory Science for Environmental Protection, https://perma.cc/Y4E3-TSMT; U.S. Env’t Prot. Agency, Citizen Science Opportunities for Monitoring Air Quality (Sept. 2016), https://perma.cc/JVH3-YP9A; U.S. Gen. Servs. Admin., Federal Crowdsourcing and Citizen Science Catalog, https://perma.cc/T4XM-HKS7.
a timescale of minutes so that peak exposures can be determined. Similarly, remote sensing technology, including satellite sensors, is being applied to map gaseous and particulate air pollution levels on a population and global scale. Nontargeted analytical methods, those that assign chemical formulas and structures to unknown compounds without the use of reference standards or target substance lists, are increasingly used, along with similar “suspect screening” methods, to characterize chemical unknowns in environmental media and in biological media. Such advances in exposure science, particularly those in analytical chemistry, alongside developments of hundreds of new databases and tools,42 have also allowed for the untargeted discovery of thousands of chemicals in environmental media, in products, and in humans.
Measurement Considerations
Although at first glance it might seem that direct measurements of environmental concentrations would provide the most reliable data, there are limits to what can be gained through this approach. It is important to recognize that there are a variety of factors influencing accuracy and applicability of available environmental measurement data, including but not limited to proximity to sources, activities of the studied individuals, time of day, season, and weather conditions.
- How can we be sure that the physical samples of media taken are representative of the media of interest for exposure assessment? Standard methods are available to design sampling plans that have specified probabilities of being representative, but they can never provide complete assurance. Generally, when the presence of a chemical is likely to be highly homogeneous, there is a greater chance of achieving a reasonably representative sample than is the case when it is highly heterogeneous. In the latter circumstance, obtaining a representative sample, even when very large numbers of samples are taken, may be unachievable.
- How can we be sure that the samples taken represent the presence of a chemical over long periods? Sampling events may provide a good snapshot of current conditions, but in circumstances in which concentrations could be changing over time, and where the health concerns involve long-term exposures, snapshots could be highly misleading. This type of problem may be especially severe when attempts are being made to reconstruct past exposures, based on snapshots taken in the present.
- How can we be sure that the analytical work was done properly? Most major laboratories that routinely engage in this type of analysis have
42. Over 700 tools are listed in the U.S. Environmental Protection Agency’s ExpoBox (A Toolbox for Exposure Assessors). See https://perma.cc/T6FB-PS3V.
- developed standard operating procedures and quality control procedures. Laboratory certification programs of many types also exist to document performance. When analytical work is performed in certified, highly experienced laboratories, there is a reasonably high likelihood that the analytical results are reliable. But it is very difficult to confirm reliability when analytical work is done in laboratories or by individuals who cannot provide evidence of certification or of long-standing quality control procedures.
- How are data showing the absence of the chemical to be interpreted? In most circumstances involving possible presence of a chemical in either biological matrices or environmental media, the analysis of some (and sometimes many) of the samples will fail to quantify or detect the chemical of interest. The analytical chemist will often report such samples as nondetect (ND). But an ND should never be considered evidence that the concentration of the chemical of interest (or other target analyte, such as a metabolite) is truly zero. In fact, most chemists will (and should) report that the chemical is below the limit of detection (LOD) or below the limit of quantification (LOQ). Every analytical method has a nonzero limit of detection that refers to the lowest chemical concentration that can be distinguished from a concentration of zero with reasonable confidence. A critical issue is that detection limits vary between assay methods, between laboratories, and even within a laboratory over time.43 Thus, for each sample for which a concentration is reported as ND or below LOD, all that can be known with confidence is that the concentration of the chemical, if present, is somewhere below that limit. If there is clear evidence that the chemical is present in some of the samples (i.e., its concentration exceeds the method’s LOD), then it is usually assumed that all the samples of the same medium reported as ND or below LOD will actually contain some level of the chemical. In studies using less sensitive methods to detect a chemical in an environmental or biological matrix, the number of samples with levels below the LOD can be substantial. Studies in which the chemical is detected at relatively low frequencies can lose statistical power by omitting samples below the LOD or by imputing samples below the LOD (e.g., assigning a value of zero, the LOD, or some function of the LOD), which can introduce artificial patterns into the data. These approaches can introduce bias into the analysis to varying degrees, based on the sample size, the proportion of observations below the LOD, and the imputation approach used.44
43. See, e.g., Dana B. Barr et al., A Survey of Laboratory and Statistical Issues Related to Farmworker Exposure Studies, 114 Env’t Health Persp. 961 (2006), https://doi.org/10.1289/ehp.8528.
44. See, e.g., U.S. Env’t Prot. Agency, Guidance for Data Quality Assessment: Practical Methods for Data Analysis (2000), https://perma.cc/SB85-7QKS. Relevant references from the peer-reviewed
Sampling and measurement are useful, but it is critical to recognize that these are nonetheless limited in important ways. The alternative involves modeling. In fact, a combination of both approaches—one acting as a check on the other—is often the most useful and reliable.
Model Considerations
A model is “a simplification of reality that is constructed to gain insights into select attributes of a particular physical, biological, economic, or social system.”45 The simplest model is a conceptual model, from which mathematical processes of the process represented in the conceptual model can be derived (with simplifying assumptions as appropriate). In an exposure assessment, models can be used to extrapolate monitoring data to populations that were not directly analyzed, to reconstruct past exposures, and even to predict future exposures. Models are tools the assessor uses to analyze and characterize processes that are too complex for capturing completely by empirical data or for which empirical data are not available. In the context of chemical exposure assessment, there are a range of models available, including:
- Deterministic modeling (i.e., physical modeling) in which a model describes the relationship between variables mathematically on the basis of knowledge of the physical, chemical, and/or biological mechanisms that are relevant.
- For example, air particle concentrations in the indoor environment might be modeled by variables such as the air exchange rate, total volume of rooms, and the sources.
- Stochastic modeling (i.e., statistical modeling) in which the statistical relationships are modeled between variables. Such models do not necessarily
literature include the following: Dana B. Barr et al., A Survey of Laboratory and Statistical Issues Related to Farmworker Exposure Studies, 114 Env’t Health Persp. 961 (2006), https://doi.org/10.1289/ehp.8528; Haiying Chen et al., A Distribution-Based Multiple Imputation Method for Handling Bivariate Pesticide Data with Values Below the Limit of Detection, 119 Env’t Health Persp. 351 (2011), https://doi.org/10.1289/ehp.1002124; Dennis Helsel, Much Ado About Next to Nothing: Incorporating Nondetects in Science 54 Annals Occupational Hygiene 257 (2010), https://doi.org/10.1093/annhyg/mep092; Dennis R. Helsel, More than Obvious: Better Methods for Interpreting Nondetect Data, 39 Env’t Sci. & Tech. 419A (2005), https://doi.org/10.1021/es053368a; Dennis R. Helsel, Less than Obvious: Statistical Treatment of Data Below the Detection Limit, 24 Env’t Sci. & Tech. 1766 (1990), https://doi.org/10.1021/es00082a001; Jay H. Lubin, Epidemiologic Evaluation of Measurement Data in the Presence of Detection Limits, 112 Env’t Health Persp. 1691 (2004), https://doi.org/10.1289/ehp.7199.
45. Nat’l Rsch. Council, Models in Environmental Regulatory Decision Making (2007), https://doi.org/10.17226/11972.
- require fundamental knowledge of the underlying physical, chemical, and/or biological relationships between the variables.
- For example, use of the EPA’s Stochastic Human Exposure and Dose Simulation Model (SHEDS) to estimate a product intake fraction for chemicals in consumer products, or use of land use regression modeling for air pollution modeling in which the relation between land use characteristics such as population density, traffic density, green space, and ambient air pollution levels are modeled using statistical regression techniques and measurements.
Perhaps the most widely used models are those that track the fate and transport pathways followed by substances emitted into the air. Knowledge of the amounts emitted per unit of time (usually obtainable by measurement) from a given location (a stack of a certain height, for example) provides the basic model input. Information on wind directions and velocities, the nature of the physical terrain surrounding the source, and other factors needs to be incorporated into the modeling. Some substances will remain in the vapor phase after emission, but chemical degradation (e.g., because of the action of sunlight) could affect media concentrations. Some models provide for estimating the distributions of soil concentrations for those substances (particulates of a certain size) that may fall during dispersion. Much effort has been put into developing and validating air dispersion models.46
Similar models are available to track the movement of contaminants in both surface and ground waters. Other types of exposure models currently being employed use geographic information system (GIS) mapping and various regression (e.g., land use regression) and other statistical tools to estimate regional air concentrations for use in exposure models. These approaches, which map locations of high air pollution, have been used in analyses that estimate large-scale population exposures for chemical pollutants such as ozone and fine particulate matter (PM 2.5). The fate and transport modeling issue becomes more complex when attempts are made to follow a chemical’s movement from air, water, and soils into the food chain and to estimate concentrations in the edible portions of plants and animals.47 Most of the effort in this area involves the use of empirical data (e.g., What does the scientific literature tell us about the quantitative relationships between the concentration of cadmium in soil and its concentration in the edible portions of plants grown in that soil?). This type of empirical information, together with general data on chemical absorption into,
46. Id.
47. Exposure scientists specializing in ecological receptors will also use modeling results to evaluate risks to wildlife, plants, and ecosystems.
distribution in, and excretion from living systems, is the usual approach to ascertain concentrations in these food media.48
In addition to environmental fate and transport, a variety of human exposure models are readily available for exposure assessment, particularly for assessment of near-field exposures, such as those from consumer products. Availability, and use, of certain types of exposure models has dramatically increased in recent years. This in part results from the advent of the European Union’s Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) legislation, which requires manufacturers and importers of hazardous substances eventually included in consumer products to assess exposure and risk from using these products in a chemical safety report, if a substance is manufactured or imported in sufficient quantities. Given the number and range of exposure models available, it is not always readily apparent which models have established validity and which have not. In some cases, however, a given model may have standing with authoritative bodies (e.g., developed or approved by the EPA for a given application). Further complicating matters, it is often the case that a model intended for a specific purpose is being used in a new context that may not have been anticipated by the original developer. An expert should be able to explain why a model is being used and why other possible models are not as suitable.
As models are the basis of many exposure assessments presented as evidence,49 if not the major component of such assessments, it is important that an exposure science expert be able to not only describe the model itself and the rationale for its selection but also the scientific basis and underlying assumptions of the model, and the ways in which the model has been validated,50 including comparisons to empirical data as available. If empirical data are available, it may also be reasonable to attempt to reconcile the measurement and modeling results and arrive at the most likely values (or range of likely values). Similarly, it may be useful to have the expert describe the likely size of error associated with model results.
48. Nat’l Rsch. Council, Models in Environmental Regulatory Decision Making (2007), https://doi.org/10.17226/11972.
49. See, e.g., Milward v. Acuity Specialty Prods. Grp., Inc., 969 F. Supp. 2d 101, 108 (D. Mass. 2013), aff’d sub nom. Milward v. Rust-Oleum Corp., 820 F.3d 469 (1st Cir. 2016) (concluding that expert’s exposure assessment was admissible where expert used the Advanced REACH Tool (ART) to calculate plaintiff’s benzene exposure from using paint that contained mineral spirits); Brantley v. Int’l Paper Co., No. CV 2:09-230-DCR, 2017 WL 2292767 (M.D. Ala. May 24, 2017) (concluding that expert’s exposure assessment was admissible where expert reasonably linked the plaintiffs’ claims of property damage to the mill’s emissions through his AERMOD study).
50. The process of developing and validating a new model or modifying and evaluating an existing model is beyond the scope of this reference guide but U.S. Env’t Prot. Agency, Guidance on the Development, Evaluation, and Application of Environmental Models (2009), https://perma.cc/7ARB-67EK, describes the steps in detail. This specifies a number of steps including credible and objective peer review, corroborating the model by evaluating the degree to which it corresponds to the system being modeled, and performing sensitivity and uncertainty analyses.
Other issues pertaining to the sources and reliability of the data used in the application of a model can be similarly pursued.
Exposure Assessment Methods
The specific methods used in an exposure assessment depend on the exposure assessment questions, availability and feasibility considerations, the intended applications of the exposure assessment (e.g., use in a type of risk assessment, comparison to a reference value), and, in some cases, the regulatory or statutory requirements. For example, an exposure assessment can inform risk screening, enforcement, remediation decisions, or program and policy evaluation. However, even within an exposure assessment, the use of a given method is not mutually exclusive. Accordingly, an assessor may choose to use several methods. Human exposures can be characterized using various approaches: measurements or estimates in the environment (e.g., ambient air concentrations), at the point of human contact (e.g., personal monitoring or sensors), or after contaminants have entered the human body (e.g., biomonitoring). For environmental data, models can be used to estimate human exposure by combining information about environmental concentrations with information about an individual human receptor or a receptor population. Concentrations and receptor information are referred to as exposure factors, which relate to contact with an agent (e.g., inhalation rates, activity patterns, time in microenvironments). Biomonitoring data that reflect an internal dose can also be used to estimate exposure, using “exposure reconstruction,” an approach that typically involves use of pharmaco/toxicokinetic models. This section provides an introduction to the three methods most commonly used in exposure assessment: direct measurement, indirect estimation, and exposure reconstruction. The first two methods use information collected or estimated prior to, or at, the point of exposure to predict exposure, while the third, exposure reconstruction, uses information (e.g., measurements of a chemical or metabolite) collected from the body after exposure has already occurred to back-calculate exposure.
Direct Measurement
Direct measurement methods (i.e., point-of-contact methods) use a number of techniques to measure the contact of a person with the chemical concentration in an exposure medium over a specified period of time. Use of direct measurement methods provides an exposure assessor with chemical concentrations or amounts at the interface between the environment and an individual. So long as the techniques are sufficiently accurate, this method is likely to result in the least amount
of uncertainty in estimating exposure concentration, especially over the time period spanned by samples collected. Note that uncertainty can increase in situations in which few measurements are available and short-term sampling data are extrapolated to long-term exposure. Another important consideration is that a direct measurement is not typically source-specific without further data and information. Further, samples from multiple individuals or other units (e.g., households) may be required in order for these measurements to be sufficiently representative of a population of interest. However, these values can be used to validate or verify assessments conducted using an alternate assessment method. Commonly used direct measurement methods include:
- Passive and active monitors to measure inhalation exposure. Such monitors are typically compact and located close to the breathing zone of the individual being studied. For passive sampling studies (e.g., use of a diffusion badge to measure formaldehyde) and active sampling studies (e.g., use of a powered pump to draw air through a filter or similar device in order to measure particulate matter), it is often necessary to extrapolate exposure and subsequently dose from the measured concentration as this is dependent on other factors, including breathing rate (see “Understanding Human Exposure: Key Concepts” above).
- Duplicate diet studies to measure dietary ingestion exposure. In these studies, individuals will prepare or collect duplicate samples of all the foods they consume during a given period, allowing investigators to measure concentrations of a contaminant in the diet, which are typically normalized to body weight. While this method gives an accurate estimate of ingestion exposure to such contaminants (and can capture information such as changes in concentration owing to preparation processes such as cooking), it is typically cost-prohibitive and difficult for participants. Note that in order to estimate the applied, internal, or biologically effective dose, an exposure assessor would still need to make other assumptions regarding exposure (e.g., leverage toxicokinetic information to predict absorption across the GI tract).
- Sampling patches, silicone wristbands, whole-body dosimeters, removal methods, and optimal methods to measure dermal contact with chemicals. Patches or wristbands are materials limited to placement on specific areas of the body to collect a chemical or chemicals of interest. While these sensors are relatively inexpensive and easy to use, they may miss key contact surfaces and introduce error when extrapolating from a small surface area to a larger surface or the whole body. Whole-body dosimeters are coverall suits or full-length cotton garments that are meant to capture any contact with a contaminant in a specific scenario and time frame during which the dosimeter is worn. Removal methods (e.g., rinsing, wiping, tape
- stripping) collect contaminants of concern from the skin at a specific interval. Optical methods include fluorescence analyzers, which have been widely used for measurement of various halogenated compounds. These methods all reflect deposition to the skin, so in order to estimate an absorbed or biologically effective dose, an exposure assessor would still need to make additional assumptions (e.g., leverage toxicokinetic information to predict absorption through the skin).
Indirect Estimation
Indirect estimation, sometimes referred to as scenario evaluation, is a method that estimates exposure after developing a set of facts, assumptions, and inferences about a given exposure scenario, using either measurement or estimation of both the amount of a substance contacted by an individual or population and the frequency and duration of this contact. This method typically relies heavily on assumptions about the sources and releases of a chemical of interest, relevant fate and transport mechanisms, and the concentration at the point of contact with the individual or population. This includes identification of the relevant exposure pathway(s) and route(s) of exposure. Similarly, the human receptor of interest (individual or population) must be characterized to generate inputs regarding physiologic processes and activities/behaviors that influence contact with the chemical (especially those that influence the temporal component—i.e., frequency and duration).
Estimates that quantify intake and uptake rates of the chemical for a given individual or population may also be needed. This process often involves development of a conceptual model, a diagram, or a description that lays out the environmental pathways and routes of exposure in the context of the exposure assessment, distinguishes between what is known/determined and what is assumed/based on default values, and identifies sources of uncertainty. The methods applied for quantification of exposure using indirect estimation will vary depending on the level of complexity and the desired result (e.g., a single value or point estimate of exposure versus a distribution of possible exposure values).
Derivation of a single value for exposure is referred to as a deterministic exposure assessment. These values can be either an estimate of central tendency (e.g., mean, median) that is intended to represent the exposure of the average or typical individual in a population or a high-end exposure within the defined receptor group or population. A high-end estimate is typically a representation of individuals at the upper end of the exposure distribution (e.g., at or above 90th percentile of exposure), which is typically calculated using a combination of high-end and central tendency inputs for given exposure parameters (e.g., intake rate).
Note that, while exposure scenarios can be developed to derive a bounding estimate that captures the highest possible exposure, or a theoretical upper bound, for a given exposure pathway, these are more common in screening-level assessments.51 In contrast, a probabilistic exposure assessment uses distributions of data for various parameters to generate a distribution of potential exposure estimates. Probability distributions (e.g., Monte Carlo simulation) describe the range of values (probability) that might occur in the given population. The output of a probabilistic assessment is a probability distribution of exposures that reflects the combination of the probability distributions of one or more of the model inputs or parameters. These distributions can help characterize variability, uncertainty, or both, depending on the design of the model and its inputs.
Exposure Reconstruction
Exposure reconstruction uses internal body measurements of a chemical or its metabolite(s) rather than external measurements to estimate dose. Such measurements are known as biomarkers, the cellular biochemical, analytical, or molecular measures obtained via biomonitoring from biological specimens (e.g., blood, urine, saliva) that indicate exposure to a chemical.52 Biomonitoring is the process of analyzing such human samples to determine biomarker concentrations. This biomonitoring data can then be combined with computational tools such as pharmacokinetic models to reconstruct or estimate the amount of chemical to which a person was exposed.53 Note that there are circumstances in which biomonitoring data alone may be sufficient to demonstrate exposure, even when there is only a semiquantitative or qualitative determination of the presence of a chemical. Such a situation may arise, for example, when the detection or measured concentration of a chemical is established as an inclusion criterion for a class action. This has occurred recently, with various criteria used across cases dealing with perfluorooctanoic acid (PFOA) exposure, ranging from both detection of PFOA in blood to measurement of PFOA in blood and subsequent comparison to either a specified value or to a concentration that is representative of
51. Note that scenarios developed for bounding estimates are sometimes referred to as “worst case” scenarios in which “everything that can plausibly happen to maximize exposure, dose, or risk does in fact happen. This worst case may occur (or even be observed) in a given population, but since it is usually a very unlikely set of circumstances, in most cases, a worst-case estimate will be somewhat higher than occurs in a specific population.” EPA Guidelines, supra note 6. These are useful for screening-level evaluations because if the highest possible exposure is evaluated and found to be not of concern, the more plausible lower exposures will also not be of concern.
52. EPA Guidelines, supra note 6.
53. Yu-Mei Tan et al., U.S. Env’t Prot. Agency, Biomonitoring—An Exposure Science Tool for Exposure and Risk Assessment (2012), https://perma.cc/8QWH-A2GQ.
general population exposure.54 Pharmacokinetics (PK) or toxicokinetics (TK) characterize the absorption, distribution, metabolism, and excretion of a substance in an organism’s body. In exposure assessment, pharmaco/toxicokinetic (PK/TK) models use data and mathematical equations to evaluate the fate of a chemical in the body after exposure has occurred. These are highly variable in complexity, with the simplest model being a one-compartment, first-order model that assumes immediate distribution within a single compartment, such as blood.
More complex PK/TK models account for an organism’s physiology in their equations and are called physiologically based pharmaco/toxicokinetic (PBPK or PBTK) models. These models require parameters to simulate movement and fate of chemicals within the body, considering transfers between tissues and organs, metabolism, and deposition or storage. These parameters can be physiological, physiochemical, or biochemical in nature. Many physiological parameters for these models (e.g., ventilation rates, metabolic rates) are derived from clinical or laboratory studies of animals as well as humans—largely pharmacological studies given the data requirements for such agents. Inter- and intra-species differences in these parameters should be evaluated prior to application.
Figure 3 compares predictive exposure models with reconstructive models. Whereas predictive models use information upstream from the point of exposure, such as environmental media concentrations and exposure factors to estimate an exposure or dose, the reconstructive PK/TK models use information downstream of the point of exposure, such as internal body measurements like biomarker concentrations (e.g., amount absorbed, amount at or bound to a target organ or target biological receptor within the body, and, in some cases, a biomarker of effect) and physiologic processes to estimate the original exposure and internal dose. While reconstructing exposure using biomonitoring data represents definitive exposure to an individual, it is often not possible without further information to identify specific sources or routes of exposure (e.g., inhalation, ingestion, or dermal) that contributed to the level measured in the body.
54. See, e.g., Hardwick v. 3M Co., 589 F. Supp. 3d 832, 869 (S.D. Ohio 2022), motion to certify appeal granted sub nom. In re E.I. DuPont de Nemours & Co. C-8 Pers. Inj. Litig., No. 22-0305, 2022 WL 4149090 (6th Cir. Sept. 9, 2022), vacated and remanded sub nom. In re E.I. du Pont de Nemours & Co. C-8 Pers. Inj. Litig., 87 F.4th 315 (6th Cir. 2023) (Certifies class as “Individuals subject to the laws of Ohio, who have 0.05 parts per trillion (ppt) of perfluorooctanoic acid (PFOA) (C-8) and at least 0.05 ppt of any other PFAS in their blood serum.”); Benoit v. Saint-Gobain Performance Plastics Corp., 959 F.3d 491, 501 (2d Cir 2020) (plaintiffs must show that they have a “physical manifestation of or clinically demonstrable presence of” PFOA in their blood); Sullivan et al. v. Saint-Gobain Performance Plastics Corp. 226 F. Supp. 3d 288 (D. Vt. 2016) (requires plaintiffs to show that their blood accumulation levels differ from those in the general population and link blood levels with increased risk of disease and appropriate monitoring).

Quantification of Exposure to Chemical Stressors
Calculation of Exposure
It is important to note that quantitative calculations may not always be necessary to sufficiently describe exposure;55 however, these are standard practice. The general approaches to characterize exposure are measurements, model estimates,
55. Causation may sometimes be established even if quantification of the exposure is not possible. See, e.g., Bell v. Abb Grp., Inc., No. 13-CV-1338-SMY-SCW, 2015 WL 11439279, at *2 (S.D. Ill. Sept. 1, 2015) (in denial of Daubert motion, the court found that the opinion of an expert who conducted a qualitative exposure assessment for an individual, based in part on historical data and exposure modeling reliant on historical analyses of asbestos exposure for a similar occupation, and did not intend to quantify exposure levels, was both supported by evidence and rests on authority and, “as such, cannot be fairly characterized as mere ipse dixit”); Lightfoot v. Ga.-Pac. Wood Prods., LLC, No. 7:16-CV-244-FL, 2018 WL 4517616, at *20 (E.D.N.C. Sept. 20, 2018) (In denial of Daubert motion, the court found that qualitative exposure opinions of experts, both with and without quantitative exposure assessments, are both “relevant and reliable for purposes of supporting their specific causation opinions. See Westberry [v. Gislaved Gummi AB, 178 F.3d 257], 264 (‘[T]his clearly is not a case in which the plaintiff was unable to establish any substantial exposure to the allegedly defective product.’)”.); Best v. Lowe’s Home Ctrs., Inc., 563 F.3d 171 (6th Cir. 2009) (doctor permitted to testify as to causation based on differential diagnosis).
or a combination of both. Regardless of whether an assessment is informed by measures or models (or both), the calculation of exposure follows the same principles. The simplest calculations relate to situations in which direct exposures occur. For example, consider the case of a substance directly added to food (and approved by the U.S. Food and Drug Administration (FDA) for such addition). Suppose the chemical is of well-established identity and is approved for use in nonalcoholic beverages at a concentration of 10 milligrams of additive for each liter of beverage (10 mg/L).56 To understand the amount (weight) of the additive ingested each day, it is necessary to know how much of the beverage people consume each day. Data are available for relevant exposure factors (e.g., rates of food consumption in the general population). Typically, those data reflect average consumption rates and also rates at the high end of consumption.
To make sure that the additive is safe for use, the FDA seeks to ensure the absence of risk for individuals who may consume at the high end, perhaps at the 95th percentile of consumption rates.57 Assume that surveys of intake levels for the beverage in our example reveal that the 95th percentile intake is 1.2 L per day for adults. The weight of additive ingested by individuals at the 95th percentile of beverage consumption rate is thus obtained as follows:
10 mg/L × 1.2 L/day = 12 mg/day
For a number of reasons, toxicologists often express exposure and dose as weight of chemical per unit of body weight. For adults having a body weight (bw) of, on average, 70 kilograms (kg), the dose of additive is therefore:
12 mg/day ÷ 70 kg bw = 0.17 mg/kg bw per day58
Chemical exposure resulting from ingestion of other products containing specified amounts of chemicals are calculated in much the same way. It generally would be assumed that the duration of exposure for a substance added to a food or beverage would be continuous and would cover a large fraction of a lifetime, while for other products, particularly pharmaceuticals, exposure durations will vary widely. It will be useful, before proceeding further, to illustrate calculations for exposures occurring by the inhalation and dermal routes.59
56. See infra Appendix A for a discussion of units used in exposure science.
57. Vasilios H. Frankos & Joseph V. Rodricks, Food Additives and Nutrition Supplements, in Regul. Toxicology 133 (Shayne C. Gad ed., 2d ed. 2001).
58. To gain approval for such an additive, FDA would require that no toxic effects are observable in long-term animal studies at doses of at least 17 mg/kg bw per day (100 times the high-end human intake).
59. See, e.g., Henricksen v. ConocoPhillips Co., 605 F. Supp. 2d 1142, 1164 (E.D. Wash. 2009) (benzene exposure on skin and by inhalation); Bland v. Verizon Wireless (VAW) L.L.C., No. 3:06CV00008-CFB, 2007 WL 5681791, at *9 (S.D. Iowa 2007), aff’d, 538 F.3d 893 (8th Cir.
Consider a hypothetical workplace setting in which a solvent is present in the air. Measurement by an industrial hygienist reveals its presence at a weight of 2 mg in each cubic meter (m3) of air. Data on breathing rates reveal that a typical worker breathes in 10 m3 of air each 8-hour workday.60 Thus, the worker exposure will be:
2 mg/m3 × 10 m3/day = 20 mg/day
20 mg/day ÷ 70 kg = 0.29 mg/kg bw per day
As noted earlier, it is likely that only a fraction of this dose will reach and pass through the lungs and enter the bloodstream. As also noted earlier, if the chemical is a fiber or other particle, its dynamics in the respiratory tract will be different than that of a vapor, with a portion of the inhaled dose entering the GI tract.
In contrast to ingestion and inhalation exposure, dermal exposure often is expressed as the weight of chemical per some unit of skin surface area (e.g., per m2 of skin). The body surface area of an average (70 kg, ~154 lb) adult is 1.8 m2. Thus, consider a body lotion containing a chemical of interest. If the lotion is applied over the entire body, then it is necessary to know the total amount of lotion applied and then the total amount of chemical present in that amount of lotion. That last amount will then be divided by 1.8 to yield the dermal exposure in units of milligrams per square meter. If the chemical causes toxicity directly to the skin, that toxicity dose information also will be expressed in milligrams per square meter. Then risk is evaluated by examining the quantitative relationship between the toxic dose (milligrams per square meter) and the (presumably much lower) human dose expressed in the same units. If the chemical can penetrate the skin and produce toxicity within the body, then a dose determination must include an examination of the amount absorbed into the human body.
One final matter concerning exposure estimation, and subsequently dose estimation, concerns the importance of body size and composition, in particular that of infants and growing children. In matters such as food and water intake, and breathing rates, small children are known to take in these media at higher rates per unit of their body weight than do adults.61 Thus, when a small child is exposed to a food contaminant, that child will often experience a greater relative exposure to the contaminant than will an adult consuming food with the same
2008) (inhalation exposure to Freon in “canned air” sprayed into water bottle). For a discussion of the importance of assessment of dose as a measure of exposure, see David L. Eaton et al., “Fundamental Principles of Toxicology,” in Reference Guide on Toxicology, in this manual.
60. The 24-hour inhalation rate outside the workplace setting is ca. 20 m. The lack of direct proportion to time reflects the fact that breathing rates increase under exertion.
61. See, e.g., Northwest Coalition for Alternatives to Pesticides (NCAP) v. E.P.A., 544 F.3d 1043 (9th Cir. 2008) (dispute over how much lower allowable pesticide levels should be to account for children’s greater susceptibility).
level of contaminant. Children also tend to ingest greater amounts of nonfood items, such as soils and dusts, than do adults. In some cases, nursing mothers excrete chemicals in their breastmilk, which forms a unique source of exposure for nursing infants. The exposure scientist generally conducts separate assessments for children—these assessments take into account the possibility of periods of increased exposure during the developmental period.62
Integrated Exposure Assessment
In many cases, the issue of exposure (and subsequent dose) concerns one chemical in one product and only one route of exposure. But numerous variations on this basic scenario are possible: aggregate exposure involving one chemical in several products or environmental media (e.g., lead exposure from lead-based paint, drinking water in contact with leaded pipes, proximity to airports in which leaded aviation gas is used); cumulative exposures involving either many chemicals in one product or environmental medium (e.g., nitrates and arsenic in drinking water); or many chemicals in many environmental media (e.g., exposure to metals from drinking water, air, consumer products, foods). Even though some exposure situations can be complex and involve multiple chemicals through both direct and indirect pathways, the exposure assessment methods and principles described here can be applied. Exposures occurring by different routes can be added together, or they can be reported separately. The decisions on the final exposure or dose estimates and their form of presentation can be made only after discussions with the users of that information—typically the toxicologists and epidemiologists involved in the risk assessment.63 The exposure or dose metrics emerging from the exposure assessment need to match the exposure or dose metrics that are used to describe toxicity risks.
Further, as discussed in the earlier section on exposure assessment methods, it may be useful to consider high-end or even bounding exposures, not only for exposure to chemicals through foods and consumer products but also in environmental settings. This is because, though a worst case may occur (or even be observed) in a given population, it is usually a very unlikely set of circumstances, and such a worst-case estimate will be somewhat higher than what occurs in a specific population. Therefore, if the highest possible exposure is evaluated and
62. For some substances, susceptibility to toxicity is also enhanced during the same periods.
63. See, e.g., Am. Farm Bureau Fed’n v. EPA, 559 F.3d 512 (D.C. Cir. 2009) (challenging EPA’s risk assessment for fine particulate matter); Miami-Dade Cnty. v. U.S. EPA, 529 F.3d 1049 (11th Cir. 2008) (assessment of risk of wastewater disposal methods to drinking water); Kennecott Greens Creek Min. Co. v. Mine Safety & Health Admin., 476 F.3d 946 (D.C. Cir. 2007) (risk assessment of diesel particulate matter to miners); Rowe v. E.I. DuPont de Nemours & Co., No. CIV. 06-1810 (RMB), 2008 WL 5412912, at *12 (D.N.J. 2008) (risk assessment for proposed class).
found to be not of concern, the more plausible lower exposures will also not be of concern. The EPA’s Exposure Factors Handbook is one authoritative source of recommended values for general exposure factors needed for estimation of exposure distributions, including such high-end and bounding estimates.64
Reviewing the relevant recommended values for direct drinking water ingestion from community sources, a 95th percentile value for drinking water ingestion is approximately 3 L per day across consumers of all ages, with the highest 95th percentile intake of approximately 3.4 L per day for adults aged 21 to 30. Thus, for example, it is possible to assert with relatively high confidence that almost no one in the general population consumes more than 3.5 L of water a day and that almost everyone consumes less, though care should be taken to ensure the appropriate exposure factors are used for the population of interest (e.g., athletes or manual laborers may consume even more water). If an exposure or dose calculation assumes a general population water consumption rate of 3.5 L per day, then the risk estimated for that dose is almost certainly an upper limit on the population risk, and regulatory actions based on that risk will almost certainly be highly protective for the general population. For regulatory and public health decision making, such a precautionary approach has a great deal of precedent, although care must be taken to ensure adherence to scientific data and principles.65
This approach becomes problematic, however, if applied to assessments of exposures that may have been incurred in the past by individuals claiming to have been harmed. In such cases, an approach based on attempts to accurately describe the individual’s exposure would be necessary. Whatever the case, the exposure scientist must be careful to ensure an accurate description of the exposure concentration (and resulting dose) so that the users of the information can understand whether they have been provided with high-end or bounding estimates, or central tendency estimates (e.g., mean, median) that may describe more typical exposure for a population.
64. U.S. Env’t Prot. Agency, Exposure Factors Handbook (2011), https://perma.cc/D8AR-5FT5. Note: The EPA has developed a companion software tool, ExpoFIRST, that utilizes data from the Exposure Factors Handbook in development of user-defined exposure scenarios for a given assessment. ExpoFIRST can then be used to calculate deterministic exposure estimates as point estimates for various receptor populations and life stages. This tool is accessible via the EPA ExpoBox (see https://perma.cc/T6FB-PS3V).
65. Nat’l Rsch. Council, Evolution and Use of Risk Assessment in the Environmental Protection Agency: Current Practice and Future Prospects, in Science and Decisions: Advancing Risk Assessment 26 (2009), https://doi.org/10.17226/12209. Those who must comply with regulations that were developed based on a high degree of caution often protest that more accurate assessments should be used as their basis. For several reasons, truly accurate prediction of risk is difficult to achieve (see David L. Eaton et al., Reference Guide on Toxicology, in this manual), while predicting an upper bound on the risk is not. At the same time, unless carefully done and described, upper-bound estimates may be so remote from reality that decisions based on them should be avoided.
Exposure Assessment of Biologic and Physical Stressors
In addition to chemical stressors, humans are exposed to a wide range of nonchemical stressors, including biologic stressors (e.g., bacteria, viruses) and physical stressors (e.g., radiation, heat, noise). Much like chemical stressors, these exposures may arise from typical activities, such as consumption of food, or from specific incidents, such as an industrial accident or a natural disaster. Exposure assessment considerations specific to biological and physical stressors are not enumerated here. For many nonchemical stressors, the general conceptual approach, including calculations, to exposure assessment is comparable to those outlined for chemical stressors in the earlier sections, “Exposure Assessment Considerations for Chemical Stressors” and “Quantification of Exposure to Chemical Stressors.” For example, much like a chemical stressor, exposure assessment for radiation can involve various measurements, including the amount of radioactivity, ambient radiation levels in environmental media such as water, soil, or air, and an internal or absorbed dose (energy deposited per unit mass) that a person has received from a radioactive source. However, it is important to note that there can be substantial differences in the type and scope of information and data required, as well as in the exposure measurements and models used to characterize exposure and inform risk assessment for certain nonchemical stressors.
There are considerable differences even in the approach to description of radiation, with four different but interrelated types of units for measuring radioactivity, exposure, absorbed (internal) dose, and effective dose (also referred to as dose equivalent). Typically, the amount of radioactivity refers to the amount of ionizing radiation released by a material, whether it emits alpha particles, beta particles, gamma rays, x-rays, or neutrons. The quantity of radioactive material is expressed in terms of its radioactivity, which represents how many atoms in the material decay in a given time period, with units of measure most commonly reported in becquerels (Bq, international unit, equivalent to one radioactive decay per second) or Curie (Ci, U.S. unit, approximate number of radioactive decays in one gram of radium per second). Such radioactivity can be measured by a variety of detectors, including the commonly used Geiger counter. For measurement of ambient radiation in the environment, other instrumentation, such as pressurized ionization chambers, are best suited, and levels are reported in units such as roentgen per hour (R/h) or coulomb per kilogram (C/kg).
In contrast, an absorbed radiation dose reflects the amount of energy that a radioactive source deposits in an organism through which the radiation passes. Absorbed radiation can be measured using additional specialized instruments, including alarming dosimeters that are typically used to monitor exposure rates and accumulated dose in real time. The conventional unit for absorbed radiation dose is now the international unit Gray (Gy), though the older term rad
(for radiation absorbed dose) may still be used (one Gy is equivalent to 100 rad).66 Lastly, the dose equivalent, or effective dose, of radiation is a term that addresses an individual’s biological risk given the type of radiation exposure by adjusting the absorbed dose by radiation type and relative organ sensitivity. The conventional unit for dose equivalent is now the sievert (Sv), though the roentgen equivalent man (rem) may still be used in some cases. The dose equivalent is commonly used to set protective levels for groups of people. For example, the annual radiation dose limit for workers in the United States is 0.05 Sv or 5 rem.67
Other physical stressors, such as noise and temperature, have their own terminology and approaches, as do the various types of pathogens. As such, appropriate stressor-specific guidance should be considered when evaluating such an assessment. A variety of guidance documents and tools have been developed to support measurement of specific biological and physical stressors, with various federal agencies and nongovernmental organizations having also developed specific guidance for risk assessment of such stressors, particularly for radiation and microbial exposures. For example, to support the assessment of radiation, the EPA and the Oak Ridge National Laboratory (ORNL) have developed a comprehensive software system specifically for the calculation of tissue dose and subsequent health risk from radionuclides in environmental media. Interpretation of these model results requires specialized knowledge of exposure assessment and the integration period of radiological decay. Health physicists, for example, are individuals who have received such specialized training in nuclear physics, radiation biology, radiation detection, radiation chemistry, and other related sciences. Microbial risk assessment has similar specificity, with the U.S. Department of Agriculture/Food Safety and Inspection Service (USDA/FSIS) and EPA jointly developing a guideline for microbial risk assessment with an emphasis on pathogenic organisms in food and water.68 Accordingly, exposure assessment for a nonchemical stressor should be conducted by an expert sufficiently familiar with the stressor and appropriate means of exposure and risk assessment.69
66. U.S. Ctrs. for Disease Control & Prevention, Nat’l Ctr. for Env’t Health, Measuring Radiation, https://perma.cc/P2BF-QZKD.
67. 10 C.F.R. § 20.1201 (1991), https://perma.cc/SYP4-Y4LT.
68. U.S. Env’t Prot. Agency (EPA) & U.S. Dep’t of Agric./Food Safety & Inspection Serv. (USDA/FSIS), Microbial Risk Assessment Guideline: Pathogenic Organisms with Focus on Food and Water (2012), https://perma.cc/U4QB-RT8J.
69. See, e.g., United States v. Mass. Water Auth., 97 F. Supp. 2d 155, 185 (D. Mass. 2000), aff’d, 256 F.3d 36 (1st Cir. 2001) (The most authoritative risk assessment of one water treatment option was attributed to a scientist with expertise in assessment of microbial risks of drinking water; the court discounted the testimony of another scientist who testified only in general terms about water filtration as the court found him to have “no expertise in water quality issues. He has testified ‘maybe a hundred times as a plaintiff’s expert in asbestosis cases.’”).
How Exposure Science Informs Evaluation of Disease Causation in Individuals
In the evaluation of disease causation for an individual, exposure science is applied to characterize the individual’s contact with a stressor, either qualitatively70 or quantitatively. The resulting exposure assessment is then linked to evidence of disease causation (e.g., a causal link established using epidemiological and toxicological investigations,71 but also possibly an association between exposure and outcome identified by the same means). Note that an individual is generally not faced with the burden of demonstrating causality with absolute proof; rather, the legal standard is typically expressed as a need to show that causality is demonstrable by a preponderance of the evidence (e.g., more likely true than not true that the harm observed was caused by the exposure).72 Establishing general causality, and providing an expert opinion as to whether it is reasonable to conclude that the exposure caused harm to an individual, is the domain of epidemiology, toxicology, and/or medicine.73 Exposure experts are needed to assess the exposures incurred by an individual and to evaluate the quality and validity of such assessments, while appropriate causation experts are called upon to offer testimony on whether those exposures are of a magnitude sufficient to cause the claimed harm.
70. As discussed in the section titled “Quantification of Exposure to Chemical Stressors” above, causation may sometimes be established even if quantification of the exposure is not possible. See, e.g., cases cited supra note 55 (Bell, Lightfoot, Westberry, & Best); Allen v. Martin Surfacing, 263 F.R.D. 47 (D. Mass. 2009).
71. See Steve C. Gold et al., Reference Guide on Epidemiology, and David L. Eaton et al., Reference Guide on Toxicology, in this manual, for discussions on epidemiological and toxicological approaches to disease causation, respectively. Regulations and public health actions are usually driven by findings of excessive risk of harm (although sometimes evidence of actual harm).
72. Did the plaintiff incur exposures to the suspect chemical of sufficient magnitude and duration to make it more likely true than not that the chemical, and not some other factor, was the cause of the plaintiff’s medical condition? See, e.g., Barrett v. Rhodia, Inc., 606 F.3d 975, 984 (8th Cir. 2010) (No appellant expert was qualified to present evidence that Barrett was exposed to toxic levels of hydrogen sulfide gas as opposed to another harmful exposure; the defense presented such an alternative cause supported by expert testimony. Court noted that “[e]xpert testimony ‘based on possibility or speculation is insufficient [to establish causation]; it must be stated as being at least “probable,” in other words, more likely than not.’ Fackler v. Genetzky, 263 Neb. 68, 638 N.W.2d 521, 527–28 (2002).”).
73. Note that a statistical association between exposure and disease does not prove causation, so plausible alternative hypotheses must be eliminated by careful statistical adjustment and/or consideration of all relevant scientific knowledge. Epidemiologic studies can show an association after such adjustment. Such studies, reasonably free of bias and further confounding, provide evidence for but not definitive proof of causation; causal associations can be further strengthened by mechanistic knowledge about how particular agents might produce adverse health effects.
Exposures of concern for individual disease causation can be highly variable in scope, including hazards present in a workplace, contamination of environmental media in a particular location (e.g., release of toxic air contaminants from a facility), and even use of a given product or products. In many cases, exposure to the same stressor may occur simultaneously through multiple media (e.g., air, food, water). This complicates both exposure assessment itself and attribution of exposure to a specific source, which is often relevant. In any such instance, however, assessment of exposure can involve either exposure measurements or modeling (see sections titled “Exposure Assessment Considerations for Chemical Stressors” and “Exposure Assessment of Biologic and Physical Stressors” above). Unless a personal monitoring study has been conducted, at least some modeling is expected regardless of what measured data may be available. Evidence presented should, at minimum, include exposure and dose estimates, and should identify and quantify important source(s) and significant pathway(s) and route(s) of exposure from the source to the individual. As an individual’s exposure is expected to vary over time, often on several different time scales and usually depending on the individual’s pattern of activities and locations, these variables must be described in some level of detail and incorporated into an exposure assessment, in addition to information about the concentration of the exposure in relevant media. This is of critical importance as it is generally expected that there is some degree of association between the intensity and temporal characteristic of the exposure and the biology of the disease; this information can therefore provide information to mitigate a source or end exposure to an individual at risk.
There are two general scenarios in which an individual’s exposure must be characterized. In the first, exposure to a stressor is currently occurring or anticipated to occur. This is typically more straightforward than in the second scenario, most common in toxic tort claims, in which an individual claims to have been harmed by prior exposure to a stressor, often by alleging that some existing medical condition has been caused by exposure, or exposures, occurring in the past. In both scenarios, exposure assessments are used to ascertain whether an individual would be exposed to a stressor, and to what extent.
Assessment of past exposures is especially difficult when considering diseases with very long latency periods.74 By the time disease occurs, documentary proof of exposure and magnitude may have disappeared; however, courts regularly deal with evidence reconstructing the past, and assessment of exposure is another
74. In certain tort and insurance litigation, diseases with a long latency period, especially those attributable to continuous or repeated exposure to a substance, may be referred to as “long-tail harm.” See, e.g., Danaher Corp. v. Travelers Indem. Co., 414 F. Supp. 3d 436 (S.D.N.Y. 2019) (resolution of disputes concerning insurance coverage for asbestos- and silica-related claims against a former subsidiary).
application of this common practice.75 Depending on the stressor, there may be measurable evidence of past exposure at the time of assessment, such as biomonitoring data (more likely for a persistent chemical76 than for a short-lived chemical). However, such a scenario typically requires reconstruction of exposure, an undertaking that also requires appropriate consideration of factors that may have influenced the disease process, or both the exposure and disease process.77 Although there may in some cases be historical measured data available, such as monitoring data in a relevant environmental medium or location (e.g., historic levels of water or air contaminants in a community, which are available for relatively few stressors, mostly chemical or biological), reconstructions of exposure are typically heavily reliant on modeling. Reconstruction of occupational exposures has been a relatively successful pursuit, because often historical industrial hygiene data are available involving the measurement of workplace air levels of chemicals. If it is possible, through the examination of employment records, to understand an individual’s job history, it may be possible to ascertain that individual’s exposure history and develop a retrospective exposure assessment.78 Guidelines for such retrospective occupational exposure
75. Courts have accepted indirect evidence of exposure. For example, differential diagnosis may support an expert’s opinion that the exposure caused the harm. Best v. Lowe’s Home Ctrs., Inc., 563 F.3d 171 (6th Cir. 2009). On occasion, qualitative evidence of exposure is admitted as evidence that the magnitude was great enough to cause harm. See, e.g., Westberry v. Gislaved Gummi AB, 178 F.3d 257 (4th Cir. 1999) (no quantitative measurement required where evidence showed plaintiff was covered in talc and left footprints); Allen v. Martin Surfacing, 263 F.R.D. 47 (D. Mass. 2009) (accounts of symptoms at the time of exposure formed the basis for expert’s opinion that exposure was high enough to cause harm). And courts have accepted the government’s reconstruction of exposure to radiation. Hayward v. U.S. Dep’t of Labor, 536 F.3d 376 (5th Cir. 2008); Hannis v. Shinseki, No. 09-0593, 2009 WL 3157546 (Vet. App. 2009) (no direct measure of veteran’s exposure to radiation was possible but VA’s dose estimate was not clearly erroneous).
76. See, e.g., Benoit v. Saint-Gobain Performance Plastics Corp., 959 F.3d 491, 501 (2d Cir. 2020) (plaintiffs must show that they have a “physical manifestation of or clinically demonstrable presence of” PFOA in their blood); Sullivan et al. v. Saint-Gobain Performance Plastics Corp., 226 F. Supp. 3d 288 (D. Vt. 2016) (requires plaintiffs to show that their blood accumulation levels differ from those in the general population and link blood levels with increased risk of disease and appropriate monitoring).
77. Confounding factors must be carefully addressed. See, e.g., Allgood v. Gen. Motors Corp., No. 102CV1077DFHTAB, 2006 WL 2669337, at *11 (S.D. Ind. 2006) (selection bias rendered expert testimony inadmissible); Am. Farm Bureau Fed’n v. E.P.A., 559 F.3d 512 (2009) (in setting particulate matter standards addressing visibility, the data relied on should avoid the confounding effects of humidity); Avila v. Willits Env’t Remediation Tr., No. C 99-3941 SI, 2009 WL 1813125 (N.D. Cal. 2009) (failure to rule out confounding factors of other sources of exposure or other causes of disease rendered expert’s opinion inadmissible); Adams v. Cooper Indus., Inc., No. CIVA 03-476 JBC, 2007 WL 2219212 (E.D. Ky. 2007) (differential diagnosis includes ruling out confounding causes of plaintiffs’ disease).
78. See, e.g., T.W. Armstrong, Exposure Reconstruction, in Mathematical Models for Estimating Occupational Exposures to Chemicals (Charles B. Keil et al. eds., 2d ed. 2009); Francesca Borghi et al., Retrospective Exposure Assessment Methods Used in Occupational Human Health Risk Assessment:
assessments have been published by various agencies, as well as nongovernmental organizations such as the American Industrial Hygiene Association.79
A retrospective exposure assessment outside of occupational settings is typically more difficult because there are usually many changes over time—for example, changes in technologies of the pollution sources, and also changes in people’s behavior and location over time.
How Exposure Science Informs Evaluation of Risk in Populations
Exposure assessment is one of the core components of regulatory quantitative risk assessment; the quality of exposure information and the exposure assessment itself are critical to the quality and utility of risk assessment. Risk assessments are typically directed at an existing exposure situation, such as the risks incurred by populations residing in the vicinity of a manufacturing or hazardous waste facility, or at the exposure situation expected if certain regulatory actions are taken. In considering exposures in a population, as opposed to an individual, it is critical to recognize that for any one exposure scenario there is inherently a range of exposures to a given stressor that are experienced by the individuals who compose the population. For example, assuming a toxicant is released from a manufacturing facility into the air in a community, some individuals may have a high degree of contact for an extended period (e.g., factory workers exposed to a substance on the job) while other individuals may have a lower degree of contact for a shorter period (e.g., individuals using a recreational site downwind of the facility). These exposures will be a function not only of the substance’s concentration in the air at the respective points of contact, but also of the characteristics of the individual or population (e.g., inhalation rate is variable across life stage and differs substantially with various activities).
To be useful for population risk assessment, an exposure assessment needs to be capable of identifying and quantifying the exposure of the populations that are most highly exposed and the populations that are most vulnerable. The assessment should include all relevant exposure pathways and allow the pathways to be identified and defined individually (for example, to allow for quantification of exposure from water or from food, as well as total exposure). Exposure assessment also needs to consider background exposures to other stressors that could influence exposure or risk within a population.
A Systematic Review, 17 Int’l J. Env’t Rsch. & Pub. Health 6190 (2020), https://doi.org/10.3390/ijerph17176190.
79. Am. Indus. Hygiene Ass’n, Guideline on Occupational Exposure Reconstruction (Susan Marie Viet et al. eds., 2008).
The assessment must also consider inherent uncertainty in the process to determine the level of confidence in the overall risk assessment. Note that in some cases, exposure science can also inform risk to a population in a given scenario through comparison of estimated or measured concentrations of stressors in environmental media (e.g., air, water) to established reference values or standards delineating a level at which no harm is anticipated from exposure. The most common such values are regulatory tolerances for pesticide residues in food, maximum contaminant levels (MCLs) for drinking water contaminants, National Ambient Air Quality Standards (NAAQS), and, for workplace exposure, permissible exposure limits (PELs) or threshold limit values (TLVs).80 Such comparisons are intended to determine whether products and environmental media contain substances at concentrations that meet existing regulatory requirements.
Evaluating the Scientific Quality of an Exposure Assessment
Though there is no standard set of criteria for evaluating the quality of an exposure assessment across all possible stressors, some concepts are universally relevant. Regardless of the stressor, all exposure assessments must reflect at least the two principal dimensions of exposure: its magnitude (often reported as concentration, amount, or intensity) and time. Temporal considerations include duration as well as pattern or frequency (e.g., continuous or intermittent exposure), and any relevant windows of susceptibility. Exposure scientists may offer expert testimony regarding exposures to stressors incurred by individuals or populations. In some cases, expert testimony will include description and quantification of concentrations in environmental media or a description and quantification of body burden, while in others, testimony may rely entirely on models rather than measurements of any kind. In each case, such assessments typically include a description of how and when exposures did or could occur, the identities of the chemicals involved, the routes of exposure, the magnitudes of exposure incurred, and the durations of exposure. These may be qualitative, quantitative, or semiquantitative in nature; an
80. PELs are official standards promulgated by the Occupational Safety and Health Administration. TLVs are guidance values offered by an organization called the American Conference of Governmental Industrial Hygienists. See, e.g., In re Howard, 570 F.3d 752, 754 (6th Cir. 2009) (challenging PELs for coal mine dust); Jowers v. BOC Grp., Inc., 608 F. Supp. 2d 724, 735–36 (S.D. Miss. 2009) (PELs and TLVs for welders’ manganese fume exposure); Int’l Brominated Solvents Ass’n v. Am. Conf. of Governmental Indus. Hygienists, Inc., 625 F. Supp. 2d 1310 (M.D. Ga. 2008) (challenging TLVs for several chemicals); Miami-Dade Cnty. v. U.S. E.P.A., 529 F.3d 1049 (11th Cir. 2008) (MCLs for public drinking water).
exposure assessment does not have to be quantitative to be of use,81 but typically an estimation of at least semiquantitative82 magnitude and duration of incurred exposure is necessary to evaluate plausibility of a causation claim.83
For the purposes of this reference guide, it is assumed that questions regarding disease risk and causation are beyond the bounds of exposure science and are instead within the domains of other sciences, such as epidemiology and toxicology.84 However, if the exposure scientist is also an epidemiologist or toxicologist,85 or has a relevant medical background, he or she may offer additional testimony on the health risks associated with those exposures or even regarding the question of whether such exposures may have caused disease. Below is a set of questions that exposure scientists should be able to answer, with appropriate documentation and scientific reasoning, to support any given exposure assessment.
- Is the purpose of the assessment clear? Is the exposed individual or population specified?
- Has the source(s) of exposure been identified?
- When did the exposure occur: Past? Present? Future? If exposure is occurring now, will it continue to occur? Under what conditions is future exposure anticipated?
- What is known or assumed about the nature and extent of media contact by members of the exposed population? How has this been ascertained?
- What is the assumed frequency and duration of exposure, and what is the basis for these assumptions?
81. As discussed in the section titled “Quantification of Exposure to Chemical Stressors” above, causation may sometimes be established even if quantification of the exposure is not possible. See cases cited supra note 55; Allen v. Martin Surfacing, 263 F.R.D. 47 (D. Mass. 2009) (accounts of symptoms at the time of exposure formed the basis for expert’s opinion that exposure was high enough to cause harm).
82. See, e.g., Rhyne v. United States Steel Corp., 474 F. Supp. 3d 733, 761 (W.D.N.C. 2020) (Expert exposure assessment found to be based on sufficient data and to be the product of a reliable methodology, even though estimates are not based on exact data on how often the plaintiff used the product in question at home. The court notes that “[t]o require expert testimony to be based on exact information as to how frequently plaintiff used a product fifty years ago, as Savogran suggests, would effectively prohibit a plaintiff from ever recovering in a latent disease case.”).
83. See Steve C. Gold et al., Reference Guide on Epidemiology, in this manual; see also section titled “How Exposure Science Informs Evaluation of Risk in Population” above.
84. See Steve C. Gold et al., Reference Guide on Epidemiology, and David L. Eaton et al., Reference Guide on Toxicology, in this manual, for discussions on epidemiological and toxicological approaches to disease causation, respectively.
85. See “Qualifications of Exposure Scientists or Other Exposure Assessors” below, which deals with the question of the qualifications of exposure scientists. In many cases, the work of exposure experts is turned over to health experts to incorporate into their evaluation of risk and disease causation. In some cases, usually the less complex ones, exposure assessments may be undertaken by the health experts.
- What are the pathways from the source to the exposed individuals or population(s)? How has it been established that those pathways exist? (Past? Present? Future?)
- What is the concentration of the chemical in the media with which the exposed individual or population comes into contact? (Past? Present? Future?) What is the basis for this concentration: Direct measurement? Indirect estimation? Exposure reconstruction?
- If the concentration is based on direct measurement, what procedures were followed in obtaining that measurement? Was sampling of environmental media sufficient to ensure that it was representative? If not, why is representativeness not important? If a biomarker was measured, how does it relate to the original exposure—is this the stressor itself or a metabolite? Were validated analytical methods used by an accredited laboratory? If not, how can one ensure that the analytical results are reliable?
- If models were used, what is their reliability? (See “Exposure Assessment of Biologic and Physical Stressors.”) What is the variability over time in concentrations in the media of concern? How has the variability been determined? Has the model been validated? Has the selected model output been compared to those from other similar models, and are they similar (e.g., estimates in the same direction)?
- Models developed by or accepted by authoritative bodies (e.g., the EPA86) are generally considered reliable, at least for the intended applications. Other models should be more carefully evaluated to determine validity and applicability.
- What exposure, over what period of time, by which routes, has been incurred? What calculations support this determination?
- What is the variability among members of the population in their exposure to the chemical of concern? How is this known?
- What is the likely error in the exposure estimates?
- What uncertainties are associated with the exposure/duration findings? Is it a “most likely” estimate, or is it an upper limit? To what fraction of the population is the upper limit likely to apply?
- What has been omitted from the exposure assessment, and why?
These questions are perhaps the minimum that an expert should be able to address when offering testimony. An expert should be able to support such answers with documentation.
86. Such exposure models are enumerated in EPA’s ExpoBox. U.S. Env’t Prot. Agency, EPA ExpoBox (A Toolbox for Exposure Assessors), https://perma.cc/T6FB-PS3V.
Qualifications of Exposure Scientists or Other Exposure Assessors
As discussed generally by Liesa L. Richter and Daniel J. Capra in their reference guide in this manual, The Admissibility of Expert Testimony,87 the court is expected to perform a gatekeeping function with regard to proposed experts involved in the evaluation of exposure-related information and assessment of exposure experts for a given case.
A determination of whether or not a proposed exposure expert is qualified is complicated by the fact that exposure science is a heterogeneous field with various subspecialties (e.g., biomonitoring, environmental monitoring, modeling, remote sensing, use of sensors/dosimeters) as well as specialized applications, such as industrial hygiene/occupational exposures. No single academic degree, research specialty, or career path qualifies an individual as an expert in and of itself. Further complicating evaluation of expert qualifications, a given exposure assessment may involve collaborative efforts among members of various disciplines. However, there are a number of indicia of expertise that can be ascertained for an individual proffering an opinion on exposure, outlined as follows.
Does the Proposed Expert Have an Advanced Degree or Certification in Exposure Science, Industrial Hygiene, Environmental Health, or a Related Field?
Historically, exposure scientists and related experts have possessed a wide range of academic backgrounds, spanning industrial hygiene, environmental and analytical chemistry, chemical and environmental engineering, geology and hydrogeology, toxicology (toxicokinetic applications in particular), epidemiology, and even behavioral sciences (pertaining to those aspects of human behavior that affect exposures). Note that a single course in exposure assessment or risk assessment is unlikely to provide sufficient background for developing expertise in exposure science. An increasing number of schools offer either a graduate degree in the area of exposure science and/or exposure assessment or, more commonly, a concentration or emphasis in these areas within a broader degree program such as Environmental Health Sciences.88 Such a focused graduate degree demonstrates
87. See Liesa L. Richter & Daniel J. Capra, The Admissibility of Expert Testimony, in this manual.
88. While some institutions have dedicated exposure science degree programs, such as the doctoral program in Human Exposure Assessment at Rutgers University, many leading U.S.
that the proposed expert has a substantial background in the basic issues and tenets of exposure science and exposure assessment. There are currently no certification programs available specifically for exposure scientists, although the Europe Regional Chapter of the International Society of Exposure Science (ISES Europe) has developed a program to begin certifying International Registered Exposure Scientists (IRES) by 2026. However, experts may hold other certifications, many as certified industrial hygienists.89
Is the Proposed Expert Established Professionally in the Field?
The success of academic scientists in exposure science, as in other applied sciences, is usually ascertained using the following types of criteria: the quality and number of peer-reviewed publications, the ability to compete for research grants, service on scientific advisory panels and/or working groups, university appointments, and participation or service in professional organizations and societies. Publication of articles in relevant peer-reviewed journals or peer-reviewed technical reports or exposure assessments (as is often more common for governmental experts) generally is construed to indicate some degree of expertise in exposure science. The number of articles, their topics, and whether the individual is the principal or senior author are important factors in determining the expertise of such a scientist.90 Selection for local, national, and international regulatory advisory panels or working groups (including those convened by the
institutions with environmental and public health programs now offer various degrees with a concentration or focused track in exposure science and/or exposure assessment, including Harvard University (Environmental Health Exposures/Exposure Assessment specialty), the Johns Hopkins University (Exposure Sciences and Environmental Epidemiology track), the University of Michigan (Exposure Science-Industrial Hygiene track), and the University of Washington (area of emphasis in Occupational Hygiene/Exposure Science).
89. See, e.g., Allen v. Martin Surfacing, 263 F.R.D. 47 (D. Mass. 2009) (industrial hygienist qualified to testify regarding concentration and duration of plaintiffs’ decedent’s exposure to toluene and other chemicals); Buzzerd v. Flagship Carwash of Port St. Lucie, Inc., 669 F. Supp. 2d 514 (M.D. Pa. 2009) (industrial hygienist qualified to opine on carbon monoxide exposure, but his conclusions were not based on reliable methodology).
90. Examples of reputable, peer-reviewed journals that publish exposure-related research are the Journal of Exposure Science & Environmental Epidemiology; American Journal of Industrial Medicine; Annals of Work Exposures and Health (previously Annals of Occupational Hygiene); Archives of Environmental & Occupational Health (formerly the Archives of Environmental Health); Atmospheric Environment; Biomarkers; Chemosphere; Environmental Health Perspectives; Environment International; Environmental Research; Environmental Science & Technology; Environmental Toxicology and Chemistry; Exposure and Health; Indoor Air; Integrated Environmental Assessment and Management; International Journal of Environmental Research and Public Health; International Journal of Hygiene and Environmental Health; Journal of Occupational and Environmental Hygiene; Journal of Toxicology and Environmental Health; Journal of
EPA, FDA, National Academies, National Institutes of Health (NIH), and World Health Organization (WHO)) also typically implies recognition in the field. Similarly, exposure-focused research grants from government agencies (including the National Institute of Environmental Health Sciences, the National Institute of Occupational Safety and Health, and the EPA) and private foundations are highly competitive. Successful competition for funding may also be construed to indicate competence in this area. A university appointment in exposure science, industrial hygiene/occupational health, environmental health, risk assessment, or a related field signifies relevant expertise, particularly if the university has a graduate education program in that area and the expert provides formal instruction or mentoring of trainees and fellows.91 Participation in professional societies92 is common for advanced practitioners; not only do meetings and events allow for presentation of research and networking activities, but most societies give awards to notable professionals based on scientific contributions to the field.
Does the Proposed Expert Have Experience Relevant to the Topic or Method of Interest?
Given the heterogeneity of exposure science, a given expert’s body of work and training should be carefully evaluated for relevance to the exposure-related issue(s) of interest in a given case. Exposure scientists are increasingly scrutinized not just for the scope of their expertise in general,93 but for their scope of
Occupational and Environmental Medicine; Regulatory Toxicology and Pharmacology; Risk Analysis; Science of the Total Environment; Toxicology; Toxicology Letters; and Toxicological Sciences.
91. See, e.g., Bearden v. Honeywell Int’l, Inc., No. 3:09-CV-1035, 2015 WL 7574344 (M.D. Tenn. Nov. 23, 2015).
92. Note that only one professional society, the International Society of Exposure Science, is dedicated to exposure science and/or exposure assessment. Such membership has been referenced in expert qualifications, see, e.g., Bearden v. Honeywell Int’l, supra note 91. However, other professional organizations and societies relevant to toxicology and risk assessment have established specialty groups or technical sections in exposure science or assessment, including the Society of Environmental Toxicology and Analytical Chemistry’s Exposure Modeling Interest Group, the Society of Toxicology’s Exposure Specialty Section, the Society of Risk Analysis’ Exposure Assessment Specialty Group, and the American Industrial Hygiene Association’s Exposure Assessment Strategies Committee. Of these, only the Society of Toxicology has qualifications-centered requirements for membership, based either on peer-reviewed publications or on the active practice of toxicology or a related discipline that informs toxicology.
93. See, e.g., United States v. Burkich, No. 1:19-CV-3510-MLB, 2022 WL 4236243, at *4 (N.D. Ga. Sept. 14, 2022) (exclusion of opinions offered in the area of patient care following exposure by an exposure expert without a background in healthcare based on determination that the expert is “seeking to testify outside the scope of [her] academic and professional specialty”) (quoting Moore v. Intuitive Surgical, Inc., 995 F.3d 839, 853 n.12 (11th Cir. 2021)).
expertise within the field itself94 as the types of exposure data and exposure issues presented as evidence continue to evolve. For example, a scientist specializing in dispersion modeling of toxic air contaminants may not have sufficient expertise to address exposure and/or dose reconstruction from biomonitoring data. And while some healthcare professionals specializing in occupational and environmental health may have training in certain elements of exposure assessment (particularly those who work in the chemical, petrochemical, and pharmaceutical industries, in which the surveillance of workers exposed to chemicals is a major responsibility), these individuals95 may not be sufficiently familiar with exposures outside of occupational settings or with certain aspects of data collection and analysis. While a review of a proposed expert’s biosketch, curriculum vitae, and/or publication record should give some indication of areas of specialization, additional focused questions may be necessary.
94. See, e.g., In re 3M Combat Arms Earplug Prods. Liab. Litig., No. 3:19MD2885, 2021 WL 948839 (N.D. Fla. Mar. 13, 2021) (expert’s professional experience in the past decade was related to chemical and product exposure and risk assessment, rather than the exposure of interest, which was noise exposure); Jones v. United States, No. 2:16-cv-00435-JRS-DLP, 2019 WL 367622 (S.D. Ind. Jan. 30, 2019) (expert’s training and professional experience, which were in laser and air particulates, were deemed not relevant to provide an opinion regarding the presence of H. pylori in water and the transmission of H. pylori through waterborne sources).
95. See David L. Eaton et al., Reference Guide on Toxicology, in this manual (“Most practicing physicians have little knowledge of environmental and occupational medicine.”).
Appendix A: Units of Exposure and Dose
Choosing the proper units to express concentrations of chemicals in environmental media is crucial for precisely defining exposure. Chemical concentrations in environmental media usually are reported in one of two forms: as numeric ratios, such as parts per million or billion (ppm and ppb, respectively), or as unit weight of the chemical per weight or volume of environmental media, such as milligrams per kilogram (mg/kg) or milligrams per cubic meter (mg/m3). Although concentrations expressed as parts per million or parts per billion are easier for some people to conceptualize, their use assumes that media are always sampled at standard temperature and pressure (25°C and 760 torr, respectively). Consequently, scientists often prefer to express chemical concentrations as weight of chemical per unit weight or volume of media. This method also makes conversions to exposure and dose equivalents, usually expressed in terms of weight of chemical per unit body weight (mg/kg bw), more convenient. To permit the presentation of results without excessive zeroes before or after the decimal point, appropriate units are needed. The choice of units depends on both the medium in which the chemical is present and the amount of chemical measured. For example, if 50 nanograms of chemical were found in one liter of water, the appropriate units would be ng/L, rather than 0.00005 mg/L. If 50 grams were found instead, the appropriate units would be 50,000 mg/L, because milligrams are generally the largest units used to express the mass of a chemical in media (Table 1).
Table 1. Weight of Chemical Per Unit Weight of Medium
| Preferred Unit | Alternative Unit |
|---|---|
| mg/kg | ppm (parts per million) |
| µg/kg | ppb (parts per billion) |
| ng/kg | ppt (parts per trillion) |
| pg/kg | ppq (parts per quadrillion) |
In water or food, concentration expressed by the preferred unit equals concentration expressed by alternative unit; thus, 2 mg/kg = 2 ppm. One mg (10−3 g) per kg (103 g) equals 1 part per million (10−3/103 = 10−6). Similarly, 1 µg (10−6 g) per kilogram (103 g) equals 1 part per billion (10−6/103 = 10−9), and so on (Table 2).
Table 2. Weight of Chemical Per Unit Volume of Medium
| Water | Air |
|---|---|
| mg/L = ppm | mg/m3 ≠ ppm |
| µg/L = ppb | mg/m3 ≠ ppb |
| ng/L = ppt | ng/m3 ≠ ppt |
Note that in air, parts per million and parts per billion have different meanings than they do in water or food; to avoid confusion, it is always preferable to express air concentrations in weight of chemical per unit volume (rather than weight) of air (usually cubic meters, m3).
Glossary of Terms
The following definitions are derived from a range of sources, including the ES21FWG Glossary released in June 2015,96 the U.S. EPA Exposure Factors Handbook,97 various National Academies reports,98 and the official glossary adopted by the International Society of Exposure Science.99
absorbed dose. The amount of a substance that actually enters the body following absorption.
absorption. The penetration of a substance through a barrier (e.g., the skin, the gut, or the lungs).
absorption barrier. Any exposure surface that may retard the rate of penetration of an agent into an organism. Examples of absorption barriers are the skin, respiratory tract lining, and gastrointestinal tract wall.
accuracy. The ability of a method to determine the “true” concentration of the environment sampled, or the theoretical maximum error of measurement.
activity pattern data. Information on human activities used in exposure assessments. These may include a description of the activity, frequency of activity, duration spent performing the activity, and the microenvironment in which the activity occurs.
acute exposure. A contact between a stressor and an individual occurring over a short time, generally less than a day. (Other terms, such as short-term exposure and single dose, are also used.)
agent. A chemical, biological, or physical stressor that contacts a receptor (individual or population).
aggregate exposure. The sum of an individual’s exposures to a single stressor from all sources, routes, and pathways over a period of time.
analyte. A specific chemical moiety being measured, which can be an intact drug, a biomolecule or its derivative, a metabolite, and/or a degradation product in a biologic matrix.
96. ES21 Fed. Working Grp. on Exposure Sci., Glossary of Exposure Science Terms, https://perma.cc/8XCY-YPRD.
97. U.S. Env’t Prot. Agency, Exposure Factors Handbook (2011), https://perma.cc/D8AR-5FT5. Note: The EPA has developed a companion software tool, ExpoFIRST, that utilizes data from the handbook in development of user-defined exposure scenarios for a given assessment. ExpoFIRST can then be used to calculate deterministic exposure estimates as point estimates for various receptor populations and life stages. This tool is accessible via the EPA website. U.S. Env’t Prot. Agency, EPA ExpoBox (A Toolbox for Exposure Assessors), https://perma.cc/T6FB-PS3V.
98. See Nat’l Rsch. Council, Human Biomonitoring for Environmental Chemicals (2006), https://doi.org/10.17226/11700.
99. Valerie Zartarian, Tina Bahadori & Tom McKone, Adoption of an official ISEA glossary, 15 J. Exposure Analysis & Env’t Epidemiology 1 (2005), https://doi.org/10.1038/sj.jea.7500411.
average daily dose (ADD). The average dose received on any given day during a period of exposure, expressed in mg/kg body weight per day. Ordinarily used in assessing noncancer risks.
background level. The amount of an agent in a medium (e.g., water, soil) that is not attributed to the source(s) under investigation in an exposure assessment. Background level(s) can be naturally occurring or the result of human activities.
bias. In exposure measurements, the difference between the average measured mass or concentration and reference mass or concentration expressed as a fraction of reference mass or concentration. In epidemiology, this typically refers to any effect at any stage of investigation or inference tending to produce results that depart systematically from the true values.
bioavailability. The rate and extent to which a chemical (or chemical metabolite) enters the general circulation, thereby permitting access to the site of toxic action.
biological matrix. Discrete material of biological origin that can be sampled and processed in a reproducible manner. Examples are blood, serum, plasma, urine, feces, saliva, sputum, breast milk, semen, and various tissues.
biomarker of effect or biomarker of response. A measurable biochemical, physiologic, behavioral, or other alteration in an individual that, depending on the magnitude, can be recognized as associated with an established or possible health impairment or disease.
biomarker of exposure. A chemical, its metabolite, or the product of an interaction between a chemical and some target molecule or cell, that is measured in an individual.
biomarker of susceptibility. A measurable factor, such as genetic polymorphism, nutritional status, or age, that can result in certain individuals being more sensitive to a given exposure.
biomonitoring. A method used to assess human exposure to chemicals by measuring a chemical, its metabolite, or a reaction product in human tissues or specimens, such as blood and urine.
body burden. The total amount of a chemical present or stored in the body. In humans, body burden is an especially important measure of exposure to chemicals that tend to accumulate in fat cells. These chemicals include DDT, PCBs, and dioxins.
bounding estimate. An estimate of exposure, dose, or risk that is higher than that incurred by the person with the highest exposure, dose, or risk in the population being assessed. Bounding estimates are useful in developing statements that exposures, doses, or risks are “not greater than” the estimated value.
chronic exposure. A continuous, recurring, or intermittent long-term contact between a stressor and a receptor population. (Other terms, such as “long-term exposure,” are also used.)
cumulative exposure. The sum of an individual’s exposures to stressors that affect a single biological target over a period of time.
direct exposure. Exposure of a subject who comes into contact with a chemical via the medium in which it was initially released to the environment. Examples include exposures mediated by cosmetics, other consumer products, some food and beverage additives, medical devices, over-the-counter drugs, and single-medium environmental exposures.
dose. The amount of a substance entering a person, usually expressed for chemicals in the form of weight of the substance (generally in milligrams (mg) or micrograms (µg)) per unit of body weight (generally in kilograms (kg)). The time over which it is received must also be specified. The time of interest is typically one day. If the duration of exposure is specified, dose is actually a dose rate and is expressed as mg or µg/kg per day. In human epidemiological studies or experimental animal models, dose typically indicates the amount of chemical or physical agent that is absorbed into the body, not just the amount or concentration to which a person or animal is externally exposed. Some toxicologists may also refer to the external exposure as an external dose.
dose–response assessment. In risk assessment, an analysis of the relationship between the dose administered to a group and the frequency or magnitude of the biological effect (response).
duration of exposure. Toxicologically, there are four general categories describing duration of exposure: acute (one time), subacute (repeated over a short period, generally less than two weeks), subchronic (repeated, generally up to 90 days), and chronic (repeated, for nearly a lifetime).
environmental media. Air, water, soils, and food; consumer products may also be considered media. Chemicals may be directly and intentionally introduced into certain media. Others may move from their sources through one or more media before they reach the media with which people have contact.
exposure. Contact made between a chemical, physical, or biological agent and the outer boundary of an organism. The opportunity to receive a dose through direct contact with a chemical or medium containing a chemical. See also direct exposure; indirect exposure.
exposure assessment. The process of describing, for a population at risk, the amounts of chemicals to which individuals are exposed, or the distribution of exposures within a population, or the average exposure over an entire population. Also a formal step in the risk assessment process.
exposure frequency. The number of times an exposure occurs in a given period; exposure may be continuous, discontinuous but regular (e.g., once daily), or intermittent (e.g., less than daily, with no standard quantitative definition).
exposure pathway. The connected media that transport a chemical from source to receptor populations.
exposure route. The way an agent enters the body after contact (e.g., by ingestion, inhalation, or dermal absorption).
indirect exposure. Often defined as an exposure involving multimedia transport of chemicals from source to exposed individual. Examples include exposures to chemicals deposited onto soils from the air, chemicals released into the ground water beneath a hazardous waste site, or consumption of fruits or vegetables with pesticide residues.
intake. The amount of contact between an organism and a medium containing a chemical; used for estimating the exposure received from a particular medium. In toxicology, this may also refer to the amount of contact between a biological surface with a medium containing a chemical; used for estimating the dose received from a particular medium.
levels. An alternative term for expressing chemical concentration in environmental media. Usually expressed as mass per unit volume or unit weight in the medium of interest.
lifetime average daily dose (LADD). Total dose received over a lifetime multiplied by the fraction of lifetime during which exposure occurs, expressed in mg/kg body weight per day. Ordinarily used for assessing cancer risk.
limit of detection (LOD). Typically, the smallest amount of analyte distinguishable from background, or the lowest concentration of an analyte that the bioanalytical procedure can reliably differentiate from background noise. See, for example, the FDA’s Bioanalytical Method Validation Guidance for Industry (https://www.fda.gov/regulatory-information/search-fda-guidance-documents/bioanalytical-method-validation-guidance-industry). A common estimate for unbiased analyses, with media blanks not distinguishable from background, is three times the standard error of the calibration graph for low concentrations, divided by the slope (instrument reading per unit mass or per unit concentration of analyte).
limit of quantification (LOQ) or limit of quantitation (LLOQ). Typically refers to the smallest amount of analyte that can be quantitatively determined with acceptable precision and accuracy from background. See, for example, the FDA’s Bioanalytical Method Validation Guidance for Industry (https://www.fda.gov/regulatory-information/search-fda-guidance-documents/bioanalytical-method-validation-guidance-industry) and the
CDC’s NIOSH Manual of Analytical Methods (NMAM), 5th Edition (https://perma.cc/FU2Q-LNXV).
metabolite. Chemical compound that results from biotransformation (i.e., metabolism) of a chemical (e.g., phenol in urine is a metabolite of benzene and is representative of benzene absorption in an exposed worker). Note that often a chemical that enters the body is rapidly metabolized or otherwise difficult to measure or distinguish from external contamination. A metabolite may be more stable and also may be eliminated in urine, making it more accessible and easier to measure.
model. Idealized mathematical expression of the relationship between two or more factors (variables). In toxicology, may also describe the use of animals as a substitute for humans in an experimental system to ascertain an outcome of interest.
particulate matter. General term for complex mixtures of solids and aerosols composed of small droplets of liquid, dry solid fragments, and solid cores with liquid coatings that are suspended in the air. These particles vary widely in size, shape, and chemical composition and are typically defined by their diameter for air quality regulatory purposes and human health risk assessment (e.g., PM 10 for particles with a diameter of 10 microns or less and PM 2.5 for particles with a diameter of 2.5 microns or less).
point-of-contact exposure. Exposure expressed as the product of the concentration of the chemical in the medium of exposure and the duration and surface area of contact with the body surface, for example, mg/cm2-hours. Some chemicals do not need to be absorbed into the body but rather produce toxicity directly at the point of contact—for example, the skin, mouth, GI tract, nose, bronchial tubes, or lungs. In such cases, the absorbed dose is not the relevant measure for toxicity; rather, it is the amount of toxic chemical coming directly into contact with the body surface that would be relevant for toxic effects.
population at risk. A group of subjects with the opportunity to be exposed to a chemical.
receptor population. People who could come into contact with a stressor. See exposure pathway.
risk. The nature and probability of occurrence of an unwanted, adverse effect on human life or health or on the environment.
risk assessment. Characterization of the potential adverse effects on human life or health or on the environment. According to the National Research Council’s Committee on the Institutional Means for Assessment of Health Risk, human health risk assessment includes the following:
- description of the potential adverse health effects based on an evaluation of results of epidemiologic, clinical, toxicologic, and environmental research (hazard identification);
- extrapolation from those results to predict the type and estimate the extent of health effects in humans under given conditions of exposure (dose–response assessment);
- judgments regarding the number and characteristics of persons exposed at various intensities and durations (exposure assessment);
- summary judgments on the existence and overall magnitude of the public-health problem; and
- characterization of the uncertainties inherent in the process of inferring risk (risk characterization).
setting. The place or situation in which a person is exposed to the chemical. Setting is often modified by the activity a person is undertaking—for example, occupational or in-home exposures.
sensor. A technology that includes small, portable, autonomous, low-cost, real-time devices. Sensors are air or water monitoring technologies supporting very large numbers of measurement locations, including wearable, mobile (e.g., on autonomous/robotic platforms), or stationary applications. Key traits of sensor devices include direct measurement of one or more toxicants, toxins, and/or pathogens; portability; low power draw; turnkey operation; and market price supporting large numbers to be purchased by the public as individuals or community groups.
source. The activity or entity from which the chemical is released for potential human exposure.
stressor. Any entity, stimulus, or condition that can modulate normal functions of the organism or induce an adverse response (e.g., agent, lack of food, drought).
subchronic exposure. Contact between a stressor and an individual that is of intermediate duration between acute and chronic exposures.
subject. An exposed individual, whether a human or an exposed animal or organism in the environment. An exposed individual is sometimes also called a receptor, while a group of exposed individuals is sometimes called a receptor population.
systemic dose. A dose of a chemical within the body—that is, not localized at the point of contact. Thus, skin irritation caused by contact with a chemical is not a systemic effect, but liver damage due to absorption of the chemical through the skin is. Often referred to as target site dose.
total dose. The doses received by more than one route of exposure are added to yield the total dose. See also aggregate exposure.
uncertainty. A limited knowledge of the agreement between data, information, or outcomes relative to an unknown truth. The uncertainty of a measurement is the parameter associated with the result of a measurement
that characterizes the dispersion of the values that could reasonably be attributed to the quantity being measured (the measurand).
validated method. A method that meets or exceeds certain sampling and measurement performance criteria (e.g., Guidelines for Air Sampling and Analytical Method Development and Evaluation (NIOSH Technical Report)).
validated model. A model that meets or exceeds certain criteria, including credible and objective peer review, corroborating the model by evaluating the degree to which it corresponds to the system being modeled, and performing sensitivity and uncertainty analyses (e.g., EPA Guidance on the Development, Evaluation, and Application of Environmental Models).
validation. The confirmation that an observation meets a defined standard or objective reference.
References on Law and Exposure Science
In addition to the in-text legal citations, the following articles may be useful as references.
William Anderson & Kieran Tuckley, How Much Is Enough? A Judicial Roadmap to Low Dose Causation Testimony in Asbestos and Tort Litigation, 42 Am. J. Trial Advoc. 39 (2018).
Linda S. Birnbaum et al., Environmental Health Science for Regulatory Decisionmaking, 21 Duke Env’t L. & Pol’y F. 259 (2010).
Caroline Cecot, The Data Gap: Promoting Analysis of Exposure-Related Harms, 69 DePaul L. Rev. 297 (2019).
Caroline Gillie et al., Leveraging Science to Inform Proactive and Reactive Risk Management, 51 Env’t L. Rep. 10198 (2021).
Michael Green, The Education of the Judiciary: The Sciences Addressing Disease Causation, 49 Sw. L. Rev. 492 (2020).
Laura Hall et al., Litigating Toxic Risks Ahead of Regulation: Biomonitoring Science in the Courtroom, 31 Stan. Env’t L.J. 3 (2012).
Jeff B. Kray & Sarah J. Wightman, Contaminants of Emerging Concern: A New Frontier for Hazardous Waste and Drinking Water Regulation, 32(4) Nat. Res. & Env’t 36–40 (2018).
Albert C. Lin, Deciphering the Chemical Soup: Using Public Nuisance to Compel Chemical Testing, 85 Notre Dame L. Rev. 955 (2009).
Jason B. Miller & Ranjit J. Machado, Prospective and Retrospective Exposure Assessment, 22(3) Env’t Claims J. 221–29 (2010).
Megan Noonan, The Doctor Can’t See You Yet: Overcoming the “Injury” Barrier to Medical Monitoring Recovery for PFAS Exposure, 45 Vt. L. Rev. 287 (2020).
Catherine A. O’Neill, Exposed: Asking the Wrong Question in Risk Regulation, 48 Ariz. St. L.J. 703 (2016).
Elizabeth V. Young, Outsourcing the Jury: Barlett v. DuPont and the Role of Alternative Adjudication in Preserving Jury Fairness in Complex Scientific Litigation, 77 Ohio St. L.J. Furthermore: Sixth Cir. Rev. 15 (2016).
References on Exposure Science
Select Guidelines, Reports, and Other Technical Documents
National Academies of Sciences, Engineering, and Medicine, Using 21st Century Science to Improve Risk-Related Evaluations (2017), https://doi.org/10.17226/24635.
National Research Council, Risk Assessment in the Federal Government: Managing the Process (1983), https://doi.org/10.17226/366.
National Research Council, Human Exposure Assessment for Airborne Pollutants: Advances and Opportunities (1991), https://doi.org/10.17226/1544.
National Research Council, Human Biomonitoring for Environmental Chemicals (2006), https://doi.org/10.17226/11700.
National Research Council, Models in Environmental Regulatory Decision Making (2007), https://doi.org/10.17226/11972.
National Research Council, Exposure Science in the 21st Century: A Vision and a Strategy (2012), https://doi.org/10.17226/13507.
Organisation for Economic Co-operation & Development, Descriptions of Existing Models and Tools Used for Exposure Assessment: Series on Testing and Assessment No. 182 (2012).
U.S. Centers for Disease Control and Prevention, National Report on Human Exposure to Environmental Chemicals, https://perma.cc/LS6D-4WWL.
U.S. Environmental Protection Agency, Guidelines for Human Exposure Assessment (Nicolle Tulve et al. eds., 2019), https://perma.cc/WD6E-AGCF.
World Health Organization, International Programme on Chemical Safety, Principles of Characterizing and Applying Human Exposure Models (2005), https://perma.cc/Q36C-3TLZ.
World Health Organization, International Programme on Chemical Safety, Uncertainty and Data Quality in Exposure Assessment (2008), https://perma.cc/Q6L8-X77R.
World Health Organization, International Programme on Chemical Safety, WHO Human Health Risk Assessment Toolkit: Chemical Hazards (2d ed. 2012), Geneva, Switzerland: IPCS Harmonization Project, WHO (2021), https://perma.cc/W9BH-WHDM.
Select Databases and Tools
U.S. Centers for Disease Control and Prevention, National Environmental Public Health Tracking Network (Tracking Network), https://ephtracking.cdc.gov.
U.S. Environmental Protection Agency, CompTox Chemicals, https://perma.cc/Z3W6-JGPR.
U.S. Environmental Protection Agency, ExpoBox (A Toolbox for Exposure Assessors), https://perma.cc/YNT8-9R3N.
Select Articles
The following are select articles that reflect advances in exposure science and exposure assessment and highlight potential applications relevant to litigation and risk assessment. The selected articles focus on exposure-related approaches, considerations, and models rather than reports of specific exposure measures, with an emphasis on articles that demonstrate or inform utility of exposure science to risk assessment.
Kim A. Anderson et al., Preparation and Performance Features of Wristband Samplers and Considerations for Chemical Exposure Assessment, 27 J. Exposure Sci. & Env’t Epidemiology 551–59 (2017), https://doi.org/10.1038/jes.2017.9.
Jon A. Arnot, Mass Balance Models for Chemical Fate, Bioaccumulation, Exposure and Risk Assessment, in Exposure and Risk Assessment of Chemical Pollution—Contemporary Methodology 69–91 (Lubomir I. Simeonov & Mahmoud A. Hassanien eds., 2009), https://doi.org/10.1007/978-90-481-2335-3
Jon A. Arnot et al., Prioritizing Chemicals and Data Requirements for Screening-Level Exposure and Risk Assessment, 102(11) Env’t Health Persp. 1565–70 (2012), https://doi.org/10.1289/ehp.1205355.
Lesa L. Aylward et al., Variation In Urinary Spot Sample, 24 H Samples, and Longer-Term Average Urinary Concentrations of Short-Lived Environmental Chemicals: Implications For Exposure Assessment and Reverse Dosimetry, 27 J. Exposure Sci. & Env’t Epidemiology 582–90 (2017), https://doi.org/10.1038/jes.2016.54.
Hyunkyung Ban et al., Impact of Exposure Factor Selection on Deterministic Consumer Exposure Assessment, 94 Regul. Toxicology & Pharmacology 240–44 (2018), https://doi.org/10.1016/j.yrtph.2018.02.007.
Mark A. Bonnell et al., Fate and Exposure Modeling in Regulatory Chemical Evaluation: New Directions from Retrospection, 20(1) Env’t Sci. Processes & Impacts 20–31 (2018), https://doi.org/10.1039/C7EM00510E.
Francesca Borghi et al., Retrospective Exposure Assessment Methods Used In Occupational Human Health Risk Assessment: A Systematic Review, 17(17) Int’l J. Env’t Rsch. & Pub. Health 6190 (2020), https://doi.org/10.3390/ijerph17176190.
Michael S. Breen et al., A Review Of Air Exchange Rate Models For Air Pollution Exposure Assessments, 24(6) J. Exposure Sci. & Env’t Epidemiology 555–63 (2014), https://doi.org/10.1038/jes.2013.30.
Antonia M. Calafat, The US National Health and Nutrition Examination Survey and Human Exposure to Environmental Chemicals, 215(2) Int’l J. Hygiene & Env’t Health 99–101 (2012), https://doi.org/10.1016/j.ijheh.2011.08.014.
Antonia M. Calafat, Contemporary Issues in Exposure Assessment Using Biomonitoring, 3 Current Epidemiology Rep. 145–53 (2016), https://doi.org/10.1007/s40471-016-0075-7.
Andrew Caplin et al., Advancing Environmental Exposure Assessment Science to Benefit Society, 10(1236) Nature Commc’n 1 (2019), https://doi.org/10.1038/s41467-019-09155-4.
Elaine A. Cohen Hubal et al., Exposure Science and the US EPA National Center for Computational Toxicology, 20 J. Exposure Sci. & Env’t Epidemiology 231–36 (2010), https://doi.org/10.1038/jes.2008.70.
Susan A. Csiszar et al., Stochastic Modeling of Near-Field Exposure to Parabens in Personal Care Products, 27 J. Exposure Sci. & Env’t Epidemiology 152–59 (2017), https://doi.org/10.1038/jes.2015.85.
Yuxia Cui et al., Integrating Multiscale Geospatial Environmental Data into Large Population Health Studies: Challenges and Opportunities, 10(7) Toxics 403 (2022), https://doi.org/10.3390/toxics10070403.
Christiaan Delmaar & Joris Meesters, Modeling Consumer Exposure to Spray Products: An Evaluation of the Consexpo Web and Consexpo Nano Models with Experimental Data, 30 J. Exposure Sci. & Env’t Epidemiology 878–87 (2020), https://doi.org/10.1038/s41370-020-0239-x.
Antonio Di Guardo et al., Environmental Fate and Exposure Models: Advances and Challenges In 21st Century Chemical Risk Assessment, 20 Env’t Sci. Processes & Impacts 58–71 (2018), https://doi.org/10.1039/C7EM00568G.
Peter P. Egeghy et al., Computational Exposure Science: An Emerging Discipline To Support 21st-Century Risk Assessment, 124(6) Env’t Health Persp. 697–702 (2016), https://doi.org/10.1289/ehp.1509748.
Peter Fantke et al., Coupled Near-Field and Far-Field Exposure Assessment Framework for Chemicals in Consumer Products, 94 Env’t Int’l 508–18 (2016), https://doi.org/10.1016/j.envint.2016.06.010.
Peter Fantke et al., Exposure and Toxicity Characterization of Chemical Emissions and Chemicals In Products: Global Recommendations and Implementation In USEtox, 26 Int’l J. Life Cycle Assessment 899–915 (2021), https://doi.org/10.1007/s11367-021-01889-y.
Mark L. Glasgow et al., Using Smartphones to Collect Time–Activity Data for Long-Term Personal-Level Air Pollution Exposure Assessment, 26 J. Exposure Sci. & Env’t Epidemiology 356–64 (2016), https://doi.org/10.1038/jes.2014.78.
W. Greggs et al., Qualitative Approach to Comparative Exposure in Alternatives Assessment, 15(6) Integrated Env’t Assessment & Mgmt. 880–94 (2019), https://doi.org/10.1002/ieam.4070.
Zequin Guo et al., Recent Advances in Non-Targeted Screening Analysis Using Liquid Chromatography-High Resolution Mass Spectrometry to Explore New Biomarkers for Human Exposure, 219 Talanta 121339 (2020), https://doi.org/10.1016/j.talanta.2020.121339.
Lei Huang, A Review of Models for Near-Field Exposure Pathways of Chemicals in Consumer Products, 574 Sci. Total Env’t 1182–208 (2017), https://doi.org/10.1016/j.scitotenv.2016.06.118.
Kristin K. Isaacs, Establishing a System of Consumer Product Use Categories to Support Rapid Modeling of Human Exposure, 30(1) J. Exposure Sci. & Env’t Epidemiology 171–83 (2020), https//doi.org/10.1038/s41370-019-0187-5.
Andrew Larkin & Perry Hystad, Towards Personal Exposures: How Technology Is Changing Air Pollution and Health Research, 4(4) Current Env’t Health Rep. 463–71 (2017), https://doi.org/10.1007/s40572-017-0163-y.
Paul J. Lioy, Exposure Science: A View of the Past and Milestones For the Future, 118(8) Env’t Health Persp. 1081–90 (2010), https://doi.org/10.1289/ehp.0901634.
Paul J. Lioy & Kirk R. Smith, A Discussion of Exposure Science in the 21st Century: A Vision and a Strategy, 121(4) Env’t Health Persp. 405–09 (2013), https://doi.org/10.1289/ehp.1206170.
Lidia Morawska et al., Applications of Low-Cost Sensing Technologies for Air Quality Monitoring and Exposure Assessment: How Far Have They Gone?, 116 Env’t Int’l 286–99 (2018), https://doi.org/10.1016/j.envint.2018.04.018.
Linda Phillips & Jacqueline Moya, The Evolution of EPA’s Exposure Factors Handbook and Its Future as an Exposure Assessment Resource, 23 J. Exposure Sci. & Env’t Epidemiology 13–21 (2013), https://doi.org/10.1038/jes.2012.77.
Linda Phillips et al., EPA’s Exposure Assessment Toolbox (EPA-Expo-Box), 25(2) J. Env’t Informatics 81–84 (2015), https://doi.org/10.3808/jei.201400269.
Stephen M. Rappaport, Implications of the Exposome for Exposure Science, 21 J. Exposure Sci. & Env’t Epidemiology 5–9 (2011), https://doi.org/10.1038/jes.2010.50.
Aduldatch Sailabaht et al., Extension of the Advanced REACH Tool (ART) to Include Welding Fume Exposure, 15(10) me 2199 (2018), https://doi.org/10.3390/ijerph15102199.
Samantha M. Samon et al., Silicone Wristbands As Personal Passive Sampling Devices: Current Knowledge, Recommendations for Use, and Future Directions, 169 Env’t Int’l 107339 (2022), https://doi.org/10.1016/j.envint.2022.107339.
Paul T. Scheepers et al., Application of Biological Monitoring for Exposure Assessment Following Chemical Incidents: A Procedure for Decision-Making, 21(3) J. Exposure Sci. & Env’t Epidemiology 247–61 (2011), https://doi.org/10.1038/jes.2010.4.
Linda S. Sheldon & Elaine A. Cohen Hubal, Exposure as Part of a Systems Approach for Assessing Risk, 117(8) Env’t Health Persp. 1181–94 (2009), https://doi.org/10.1289/ehp.0800407.
Fenna C. Sillé et al., The Exposome: A New Approach for Risk Assessment, 37(1) ALTEX: Alt. Animal Experimentation 3–23 (2020), https://doi.org/10.14573/altex.2001051.
Jon R. Sobus et al., Integrating Tools for Non-Targeted Analysis Research and Chemical Safety Evaluations at the US EPA, 28 J. Exposure Sci. & Env’t Epidemiology 411–26 (2018), https://doi.org/10.1038/s41370-017-0012-y.
Andrea Spinazzè et al., How to Obtain a Reliable Estimate of Occupational Exposure? Review and Discussion of Models’ Reliability, 16(15) Int’l J. Env’t Rsch. Public Health 2764 (2019), https://doi.org/10.3390/ijerph16152764.
Asimina Stamatelopoulou et al., Assessing and Enhancing the Utility of Low-Cost Activity and Location Sensors for Exposure Studies, 190(3) Env’t Monitoring & Assessment 155 (2018), https://doi.org/10.1007/s10661-018-6537-2.
Lindsay W. Stanek et al., Environmental public health research at the US Environmental Protection Agency: A blueprint for exposure science in a connected world, J. Exposure Sci. & Env’t Epidemiology (2024), https://doi.org/10.1038/s41370-024-00720-8.
Susanne Steinle et al., Quantifying Human Exposure to Air Pollution—Moving from Static Monitoring to Spatio-Temporally Resolved Personal Exposure Assessment, 443 Sci. Total Env’t 184–93 (2013), https://doi.org/10.1016/j.scitotenv.2012.10.098.
Yu-Mei Tan et al., Aggregate Exposure Pathways In Support of Risk Assessment, 9 Current Op. in Toxicology 8–13 (2018), https://doi.org/10.1016/j.cotox.2018.03.006.
Susan Viegas et al., Biomonitoring as an Underused Exposure Assessment Tool in Occupational Safety and Health Context—Challenges and Way Forward, 17(16) Int’l J. Env’t Rsch. & Public Health 5884 (2020), https://doi.org/10.3390/ijerph17165884.
Lance A. Wallace et al., Validation of Continuous Particle Monitors for Personal, Indoor, and Outdoor Exposures, 21 J. Exposure Sci. & Env’t Epidemiology 49–64 (2011), https://doi.org/10.1038/jes.2010.15.
John F. Wambaugh et al., New Approach Methodologies for Exposure Science, 15 Current Op. Toxicology 76–92 (2019), https://doi.org/10.1016/j.cotox.2019.07.001.
EunHye Yoo et al., Geospatial Estimation of Individual Exposure to Air Pollutants: Moving from Static Monitoring to Activity-Based Dynamic Exposure Assessment, 105(5) Annals of the Ass’n Am. Geographers 915–26 (2015), http://www.jstor.org/stable/24537962.
Bruce M. Young et al., Comparison of four probabilistic models (CARES, Calendex, ConsExpo, and SHEDS) to estimate aggregate residential exposures to pesticides, 22 J. Exposure Sci. & Env’t Epidemiology 522–32 (2012), https://doi.org/10.1038/jes.2012.54.
Valerie Zartarian et al., Adoption of an Official ISEA Glossary, 15 J. Exposure Sci. & Env’t Epidemiology 1–5 (2005), https://doi.org/10.1038/sj.jea.7500411.
Valerie G. Zartarian & Bradley D. Schultz, The EPA’s Human Exposure Research Program for Assessing Cumulative Risk in Communities, 20 J. Exposure Sci. & Env’t Epidemiology 351–58 (2010), https://doi.org/10.1038/jes.2009.20.
Christopher Zuidema et al., Estimating Personal Exposures from a Multi-Hazard Sensor Network, 30 J. Exposure Sci. & Env’t Epidemiology 1013–22 (2020), https://doi.org/10.1038/s41370-019-0146-1.

































































