Biomarkers for Traumatic Brain Injury: Proceedings of a Workshop (2023)
Chapter: 2 How Biomarkers for Traumatic Brain Injury Can Contribute to Care and Research
This chapter covers several areas in which biomarkers can inform traumatic brain injury (TBI) care and research and discusses lessons from the field of cardiology on the validation and incorporation of cardiac biomarkers into practice. The identification and development of biomarkers for TBI has advanced significantly since the turn of the twenty-first century. While these biomarkers offer opportunities in TBI diagnosis, classification, care, monitoring, and research, questions remain as to how to most effectively advance continued biomarker discovery, achieve regulatory clearance, implement biomarkers into clinical workflows, and use these markers in making informed care decisions. The brain is very complex, and one biomarker will not be able to give all the answers. Thus, said Stuart Hoffman, Department of Veterans Affairs, the field needs to continue to understand how different types of biomarkers and biomarker devices can address different needs, as well as how to integrate the information provided by multiple biomarkers.
ORIENTATION TO TBI BIOMARKERS1
Ramon Diaz-Arrastia, University of Pennsylvania, provided an orientation to four of the settings along the care continuum in which biomarkers for TBI could be used: prehospital settings, emergency departments, hospital intensive care units, and rehabilitation and chronic care locations. He shared some of the latest research on the use of biomarkers in these contexts and outlined examples of gaps and future opportunities.
___________________
1 This section is based on a presentation by Ramon Diaz-Arrastia, University of Pennsylvania. Brief biographies for workshop speakers and moderators are found in Appendix C.
Little research has been done on brain injury in prehospital settings, Diaz-Arrastia said, such as at the scene of an accident or the sidelines of a sporting event. However, these are places in which TBI biomarkers could provide important information in making decisions on whether to transfer a patient to an emergency department (ED) for further care, or whether they can safely be sent home. Additionally, prehospital use of biomarkers could inform the decision of whether the patient may be transferred to the nearest emergency department, which may be at a community hospital, or needs to be transferred to a Level 1 trauma center where advanced neurosurgical care is available.2 Biomarkers to inform this decision need to have high sensitivity (ability to identify that a TBI has occurred) so patients can be further evaluated, while the marker’s specificity (ability to rule out a TBI) is less important in this context. The limited existing evidence on prehospital biomarker performance is typically drawn from the studies of sports injuries, Diaz-Arrastia said. For example, a study of 17 professional boxers found that biomarkers of injury were present in their blood 30 minutes after a fight (May, 2021), and levels correlated with the numbers of blows to the head, demonstrating that biomarkers are detectable very early after an injury.
The vast majority of TBI biomarker research, probably greater than 95 percent, has been in ED settings and has often focused on identifying which patients do or do not need to receive a computed tomography (CT) scan to evaluate a potential serious intracranial injury. Recent large studies find that the biomarkers glial fibrillary acidic protein (GFAP) and ubiquitin C-terminal hydrolase L1 (UCH-L1) are useful in the ED setting in predicting normal CT scans and, if widely implemented, may be able to reduce use of cranial CT. The use of biomarkers in the ED may also help inform which patients would benefit from cranial magnetic resonance imaging (MRI) and inform patient and family counseling at discharge on anticipated risks of prolonged symptoms. Patients whose head CT scan is normal can be discharged with limited instructions and resources but may suffer from disabling symptoms in the weeks afterward. GFAP and UCH-L1, together with neurofilament light chain (NfL), demonstrate some prognostic value in patients with more severe TBIs, although these markers were not found to be as useful in predicting which patients with mild injuries were likely to experience incomplete recovery (Korley et al., 2022).
Diaz-Arrastia noted, however, that only about half of people who experience a TBI go to the emergency department. Many never seek medical care
___________________
2 Trauma centers in the United States are often designated by levels, reflecting the types of resources available at the trauma center. Level 1 centers can provide the highest levels of specialty care (ACS, 2022). For descriptions and further information on trauma center levels, see https://www.amtrauma.org/page/traumalevels (accessed December 12, 2022).
or seek care later in an urgent care clinic or a primary care doctor’s office. More research is needed in these settings, he said, including on biomarker profiles beyond the 12- to 24-hour time window to reflect that many people seek medical attention at later time points.
In hospital intensive care units (ICUs), Diaz-Arrastia explained that biomarkers can be useful for identifying patients at risk of secondary brain injury, such as ischemia, intracranial hypertension, and inflammation, as well as helping to select patients for clinical trials of neuroprotective or neurorestorative therapies. An important gap to fill is how to use biomarkers to better understand the likelihood of meaningful recovery after TBI and inform decisions on intensity of care and referral for rehabilitation services. A 2017 study demonstrated a prolonged increase or a secondary peak of the biomarker S100B (S100 calcium-binding protein B) in patients with secondary brain injury and prolonged recovery (see Figure 2-1; Thelin et al., 2017). However, sample sizes for ICU studies are typically small and more research is needed on the kinetics of biomarker production and clearance after injury.
Finally, in rehabilitation and chronic care settings, Diaz-Arrastia said there is still a huge need for biomarkers to help identify patients at risk of late complications from TBI, such as dementia and chronic traumatic encephalopathy, and to further understand the mechanisms of post-TBI comorbidities, such as headaches. Biomarkers in these setting could also
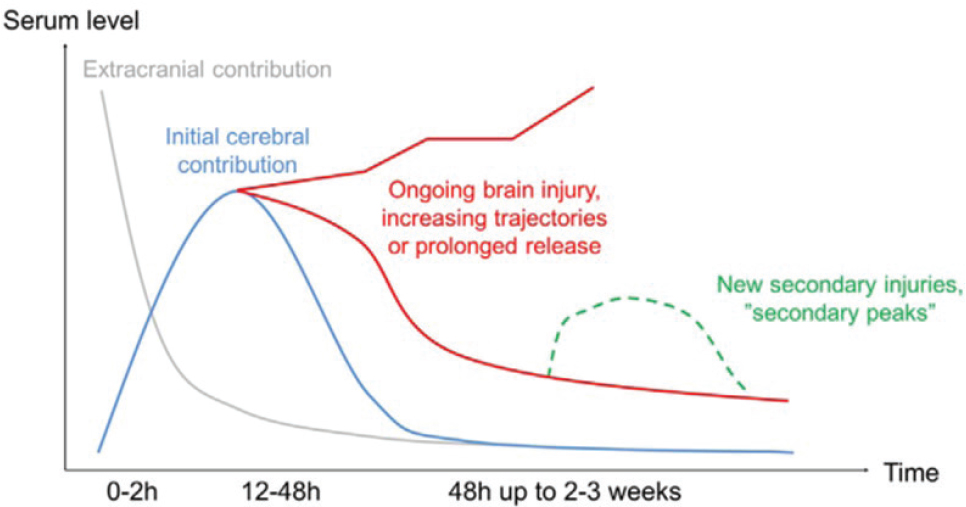
NOTE: h = hours.
SOURCES: Presented by Ramon Diaz-Arrastia, September 29, 2022; Thelin et al., 2017.
help select patients for clinical trials of therapies designed to prevent such late complications. One recent study measured biomarker levels over 5 years and found that elevated NfL was predictive of worse recovery trajectory (Newcombe et al., 2022). The dearth of studies using long-term follow-up remains an important gap, however. The largest longitudinal study of TBI recovery is through the TBI Model Systems network,3 and more systematic collection of blood and imaging data within the network could add value to future longitudinal and community studies, he concluded.
EXAMPLES OF CARE NEEDS DESCRIBED BY THE TBI COMMUNITY
Building on the overview provided by Dr. Diaz-Arrastia, several members of the TBI community shared perspectives on unmet needs and areas in which additional knowledge will be helpful. These areas include addressing TBI needs in populations such as older adults, who have unique injury and recovery patterns, and continuing to advance TBI care along the full care continuum, using the perspective of prehospital and emergency department settings.
Challenges and Opportunities in Geriatric Traumatic Brain Injury4
Adults aged 65 and older have the highest rates of TBI-related hospitalizations and deaths, experience higher morbidity and mortality from TBI, and have slower recovery trajectories than younger patients, noted Rachel Lazarus, a neuroscientist at AARP. For addressing the needs of older adults, she called for a patient-centered approach that focuses on critical junctures in clinical care in which enhanced use of TBI biomarkers would be most valuable. The first juncture is in the acute prehospital setting, she explained. Older adults are less likely to seek medical attention and less likely to be accurately diagnosed than younger patients. In the United States, adults over age 75 account for 31 percent of all TBI-related hospitalizations, she said, but true incidence likely exceeds published reports. Because geriatric TBI patients may have age-related cerebral atrophy, significant bleeding can accumulate with minimum initial symptoms, with severe symptoms suddenly developing as intracranial hemorrhage expands. While the Glasgow Coma Scale is the most widely used tool to identify the severity of a TBI, she noted, it is poorly predictive of morbidity and mortality in older adults, resulting in miscategorization of injuries, potential mistriage to the incorrect medical setting, and delays in essential care.
___________________
3 See https://msktc.org/about-model-systems/TBI (accessed December 20, 2022).
4 This section is based on a presentation from Rachel Lazarus, AARP.
The second juncture at which TBI biomarkers are needed is the stage of decision-making on withdrawal of care, and older adults who present with severe TBI may be incorrectly categorized as “lost causes.” Most in-hospital deaths in geriatric TBI patients occur after elective withdrawal of life-sustaining support, she explained, a decision that is frequently made within 72 hours of injury. However, Lazarus said, evidence shows that lack of neurological improvement within 72 hours of injury in older adults does not sufficiently predict long-term outcome. A subset of older adults with TBI, even severe TBI, can respond well to aggressive intervention and intensive rehabilitation therapies, making age and TBI severity alone poor prognostic markers. There is an important opportunity for imaging and fluid-based biomarkers to provide data to better identify older patients most likely to respond positively to treatment and inform care decisions.
Finally, Lazarus discussed how the current state of care for geriatric TBI reflects a crisis of underrepresentation in research. The past several years have seen enormous progress in development of imaging and blood-based biomarkers for TBI, but exclusion of older adults from many clinical trials makes it difficult to generalize findings on biomarker kinetics and performance, and there is limited knowledge of the interactions among age, drugs taken for other conditions (polypharmacy), comorbidities, and biomarker expression following head injury. Limited studies in older adults have shown mixed results. Biomarker candidates such as S100B, GFAP, and Tau have shown lower specificity in older patients to discriminate among those who have mild TBI with and without CT evidence of intercranial trauma. It is not clear whether the observed differences are caused by differences in injury mechanism, age-related changes in baseline expression of protein biomarkers, age-related changes in biomarker expression after injury, or other factors. Without studies that represent this population, Lazarus argued, older adults will continue to lack evidence-based TBI treatment and management guidelines.
Filling Unmet Needs in Prehospital and Emergency Department Settings5
Jeffrey Bazarian, University of Rochester, shared how TBI biomarkers can address unmet needs in prehospital and emergency department settings. The biggest problem for prehospital providers is knowing where a patient with a potential head injury needs to go, avoiding both under- and overtriage. Overtriage to Level 1 trauma centers can overload the center, while undertriage results in poorer outcome for the patient if they are first transferred to a facility without the needed level of care and subsequently transported elsewhere. Triage decisions are currently made based mainly
___________________
5 This section is based on a presentation from Jeffrey Bazarian, University of Rochester.
on the Glasgow Coma Scale score, which has limitations and can be insufficiently predictive in some patients, such as the older adults noted by Lazarus. The NIH-funded Strategies to Innovate Emergency Care Clinical Trials Network (SIREN)6 would be an excellent platform for studying how TBI biomarkers can assist prehospital providers in making the most appropriate transport decisions, he said.
In the ED, Bazarian identified two unmet needs. Currently, 40 to 70 percent of patients with a head injury receive a CT scan but only 5 to 10 percent need one. Ordering head CT scans when unwarranted exposes a patient to unnecessary radiation and slows ED throughput, with downstream effects on care for all ED patients, not just those with TBI. Existing, well-validated clinical decision rules can help providers select the patients most in need of neuroimaging, but these rules are not always followed, and ED providers need an easy and more practical tool, he argued. The second unmet need is diagnosing patients with a mild TBI. Approximately half of mild TBIs are missed in ED settings, and the use of additional diagnostic biomarkers could significantly alleviate this problem.
But having a readily available biomarker is only the first part of the challenge. Clinical buy-in and adoption are critical. Bazarian laid out the following set of questions that influence the clinical adoption of a biomarker in the ED:
- Will emergency providers order the test? Are they confident in its performance?
- Does the biomarker test affect ED operations and patient throughput? What is needed to get the test functioning as part of ED operations, whether that may be extra staff or test device maintenance?
- Are the test results available in a timely fashion (generally within an hour or less)?
- How do results get into the electronic medical record (EMR), and does this require manual data entry?
- Do patients like the test? Are they okay with being told they do not need a head CT scan based on the biomarker test?
- Is the test cost-effective? Will insurers reimburse for the test, and will CT usage actually be reduced?
Finally, he raised the issue of how to care for ED patients who have a positive biomarker test but a normal head CT scan. TBI clinics to which such patients can be referred for follow-up are just expanding in the United States. As a result, referring all such patients is likely to overwhelm available capacity. Attention must be paid to care system development, to
___________________
6 See https://siren.network (accessed November 29, 2022).
address how, when, and by whom patients can be followed to address any symptoms they experience and support their recovery.
LESSONS FROM CARDIAC BIOMARKERS7
Alan Wu, San Francisco General Hospital, shared advice on the development and implementation of TBI biomarkers (Wu and Peacock, 2020), drawing on lessons from the development and deployment of the cardiac biomarker troponin, which has revolutionized triage for people experiencing chest pain. These lessons reflect a need for
- Understanding the clinical indication for the biomarker test,
- Conducting necessary biological variation studies,
- Standardizing lab assays, and
- Establishing needs and expectation for lab turnaround times.
Wu began by noting that the troponin story has been one of increasing analytic sensitivity. The first troponin test was approved in 1994, and a high-sensitivity troponin assay was approved in 2017, although it has not yet been fully implemented in hospitals around the country. One initial question he posed for the TBI field is whether commercial TBI biomarker assays are yet at the point of sufficient analytic sensitivity to meet the field’s current and future medical needs.
Wu emphasized that the TBI field will need to clearly understand the indications and uses for various biomarkers. One of the problems in the use of troponin, he said, has been that people equate it to being a marker of acute myocardial infarction (MI), but in fact it is a marker of cardiac injury, which can arise from multiple circumstances. The cardiac care field has also experienced changes in specified troponin cutoff concentrations over time, he said, from cutoff values that allowed diagnosis of MI to cutoff values that now enable detection of minor myocardial injury. How to set TBI biomarker cutoff concentrations and interpret test results, accounting for biological variation that results in a range of values across populations, will be critical questions. Rather than compare test values against a population-based reference, Wu suggested that ideally a person should be able to compare themself against their own healthy baseline. However, that personalized baseline does not widely exist for TBI biomarkers.
Wu explained the Index of Individuality and how it affects the understanding and use of biomarkers. This index is the ratio of intraindividual variation to interindividual variation (e.g., how much a biomarker value changes in a given person over time compared to how much it varies
___________________
7 This section is based on a presentation by Alan Wu, San Francisco General Hospital.
among a population of people). Troponin, for example, has a low index of individuality—its normal level varies little within a person over hours, but varies more widely between people (Wu et al., 2009). Population-based reference ranges are of limited use for biomarkers with this profile, because a test result can fall within the population reference range but still be significantly elevated for that specific person, who may need intervention and care. Only limited biological variability studies have yet been done for TBI biomarkers. One study on GFAP found substantial interindividual variability, suggesting that a cutoff value for one person may not be an acceptable cutoff for another (see Figure 2-2; Christenson et al., 2022). Wu encouraged the TBI community to conduct further research on the interindividual variability of biomarker levels and to develop the capacity to reference results against a person’s own baseline. The concept of personal baseline values may not yet be feasible for the general population, he said, but they could be implemented for high-risk occupations, such as football players or military servicemembers, in which biomarker values could be measured before a season or period of deployment starts.
Lastly, Wu highlighted the difficulty of rapid turnaround time for the results of blood biomarker tests, outlining clinical steps necessary to obtain a result within 50 minutes. If the biomarker is being used to rule in or rule out whether a person has a head injury, a turnaround within about an hour is important and would lead to the need for point-of-care testing, maybe via a handheld device or from sensors worn by someone at high risk of a TBI. If the biomarker indication is for long-term risk stratification, on the other hand, longer turnaround times from laboratory testing are sufficient.
DISCUSSION
The discussion began by considering the implementation of TBI biomarkers in clinical practice, including further efforts needed to demonstrate effectiveness and feasibility, to establishing clinical guidelines and policies covering use of TBI biomarkers, and obtaining clinician buy-in. A participant pointed to devices recently cleared by the Food and Drug Administration (FDA) for detecting blood-based biomarkers in the context of acute TBI and asked about using such tests in EDs versus currently standard tools, such as CT scans. Beth McQuiston, Abbott Laboratories, cited work in which the military used a combination of the blood-based biomarkers GFAP and UCH-L1 to rule out 82 percent of people who did not need to be evacuated for a head CT. Bazarian commented that FDA-cleared biomarker assays can provide additional meaning and value to the evaluation of TBI patients in the emergency department, but that doctors do not want to miss a serious intracranial injury and typically overscan. In the absence of recognized clinical practice guidelines or hospital policies, physicians may
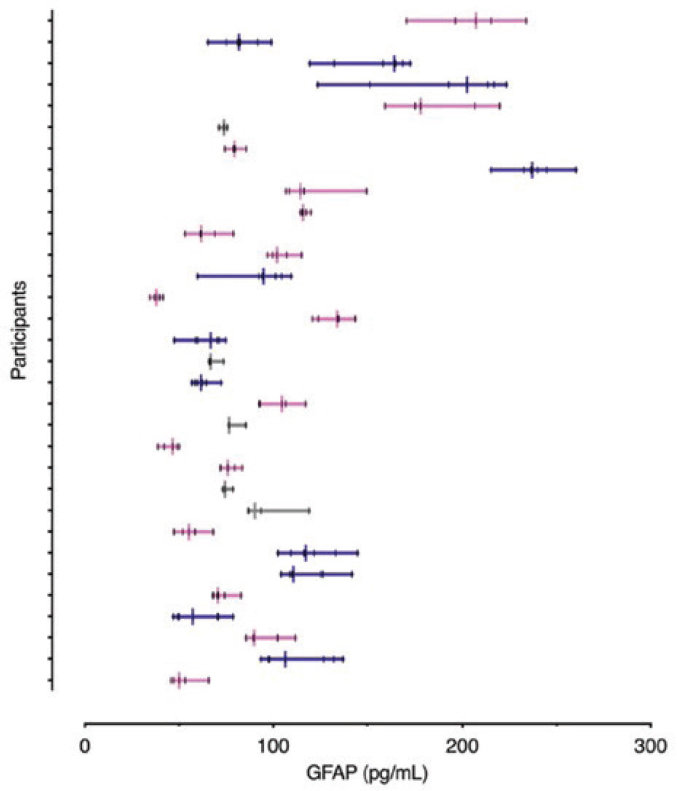
NOTE: Colors indicate the numbers of samples assessed (blue = 7, pink = 5–6, and gray = 3–4).
SOURCES: Presented by Alan Wu, September 29, 2022; Christenson et al., 2022.
be reluctant to rely on a blood biomarker test without a scan to back it up if there is a possibility the injury could be serious. Diaz-Arrastia added that one reason head CTs are ordered in the ED is to identify acute epidural hematoma. Although this is a very rare event, he noted that it is an urgent circumstance that no provider wants to miss, suggesting that blood-based biomarkers may complement, but not fully replace, use of neuroimaging in this care context.
Leslie Prichep, BrainScope Company, added that her experiences with adoption of electrophysiological biomarkers, such as EEG, indicated that people are initially more comfortable incorporating a biomarker to add specificity (to appropriately indicate that a person does not have a TBI), giving them increased confidence in discharging someone who they were planning to discharge anyway. They are less comfortable relying on new biomarkers to provide sensitivity—as in deciding whether or not to order a CT scan. Wu also saw little chance of eliminating head CT scans because it is the standard approach and because neuroimaging is needed to evaluate certain injuries, but he said that a move beyond the use of biomarkers to rule in or rule out a head CT and toward their use for more accurate TBI diagnosis will be a key focus, including the possibility of taking serial blood biomarker measurements to identify change over time. Providers will need to understand how to interpret patients who have negative imaging results but positive biomarker findings. Geoffrey Manley, University of California, San Francisco, added that evidence of elevated blood-based biomarker levels in patients who have a brain injury but a normal head CT scan suggests that biomarkers can add diagnostic value for that patient population.
Wu added that the cardiology field faced some of these standard-of-care issues, and the situation was aided by the American College of Cardiology establishing consensus guidelines on the incorporation of troponin into myocardial infarction care, as well as the types and frequency of tests to conduct. The engagement of relevant professional societies and stakeholders, including ED clinicians, neurology, and laboratory medicine, will be needed to establish similar guidance for using TBI biomarkers, reviewing the evidence and providing information on what the practices should be. Manley noted one important effort in this area. The American College of Surgeons’ Committee on Trauma is currently updating its Trauma Quality Improvement Program (TQIP) best practices for TBI and is introducing a section addressing use of blood-based biomarkers.
The discussion returned to the measurement of biomarker profiles over time, with Diaz-Arrastia noting that NfL levels are elevated at day 1 after injury and continue to increase over subsequent days if there has been substantial axonal injury. This suggests the potential to measure biomarker changes to inform follow-up or referrals to rehabilitation. Several participants indicated that this is an area to be further explored and represents a future evolution for the field.
Participants asked if TBI biomarkers would become available for differentiating among types of injuries, such as blunt trauma, hypoxic injury, or blast injury, and if multiple biomarkers would be available to catch the complexity of injury presentation. Diaz-Arrastia responded that it is very unlikely there will ever be one biomarker or one treatment to address TBI, as it is too heterogeneous. The hope is to develop biomarkers for enhanced
diagnosis and risk stratification, as well as ultimately to guide therapy. Biomarkers are needed to capture the different endophenotypes exhibited by TBI patients.8 In the future, he said, the goal is to be able to order a panel of TBI biomarkers that provide information on such elements as axonal injury, inflammation, and vascular damage. Based on the results, the provider and patient will have a better understanding of injury and treatment needs.
___________________
8 The term endophenotype refers to a subgroup with measurable characteristics or indicators that more precisely characterize the pathophysiology associated with different types of TBI (see, for example, Azad et al., 2022).











