Toxicologic Assessment of Jet-Propulsion Fuel 8 (2003)
Chapter: Appendix B JP-8 Final Risk Assessment: Contents, Executive Summary, and Introduction (TIEHH 2001)
Table of Contents
Table of Contents Section 1 |
| |||
EXECUTIVE SUMMARY Section 2 |
| |||
INTRODUCTION Section 3 |
| |||
METHODS Section 4 |
| |||
RESULTS Section 5 |
| |||
SUMMARY REPORTS OF INDIVIDUAL EXPOSURE SUBPROTOCOLS |
| |||
Quantification of Dermal Exposure to Jet Fuel Section 6 |
| |||
Assessment of JP8 in Blood Section 7 |
|
Direct Measurement of Total Body Burden of JP8 Jet Fuel (Breath) Section 8 |
| |||
Measurement of Benzene and Naphthalene in Air and Breath in the U.S. Air Force as an Indicator of JP8 Exposure Section 9 |
| |||
Non-invasive Assessment of exposure to the jet fuel, JP8 Section 10 |
| |||
Urinary Benzene, Naphthalene, 1- and 2-Hydroxy-naphthalene as Biomarkers of Acute (Short-term) Exposure to JP8 Section 11 |
| |||
SUMMARY REPORTS OF INDIVIDUAL EFFECTS SUBPROTOCOLS |
| |||
Neurobehavioral - Interim Report Section 12 |
| |||
Eyeblink Conditioning Response Test Used to Assess Performance in JP8 Exposed Air Force Personnel Section 13 |
| |||
Postural Balance Measurements Section 14 |
| |||
Gene-environment Interactions and Exposure to JP8 Jet Fuel Section 15 |
| |||
Sensitive Early Indicators of Hepatic and Kidney Damage in Workers Exposed to Jet Fuel Section 16 |
| |||
The Human Glutathione-S-Transferase M1 (GSTM1) Polymorphism as a Risk Factor for Acute Toxicity from Jet Fuel Exposure Section 17 |
|
Lynn Frame, Richard Dickerson, Tatiana Khmyl, Wang Li, and Natalie Porter |
| |||
The Effects of JP8 Jet Fuel on Serum Endocrine Concentrations in Men: Risk Assessment of Acute Exposure to Jet Fuel Section 18 |
| |||
The Effects of Heat Stress on Air Force Employees Conducting Fuel Cell Maintenance Activities on Air Force Jets Section 19 |
| |||
The Effects of JP8 Jet Fuel on Immune Cell Counts of Tank Entry Workers Section 20 |
| |||
Protein Adducts as Biomarkers of Exposure to Jet Fuel Section 21 |
| |||
Health Events Comparisons Section 22 |
| |||
Self-reported Health Status Section 23 |
| |||
JP8 Health Effects Data Derived from Ambulatory Data System Records Section 24 |
| |||
FINAL RISK ASSESSMENT: MODELING Section 25 |
| |||
FINAL RISK ASSESSMENT: MODELING Section 26 |
| |||
Statistical Analysis of Risk and Exposure Data Collected for the Risk Assessment of Acute Exposure to Jet Fuel Study |
| |||
UNCERTAINTIES Section 27 |
|
EXECUTIVE SUMMARY
The following report represents the final report of preliminary results of the protocol to assess the health and performance effects of acute exposure to Jet Fuel number 8. Texas TechUniversity, Institute of Environmental and Human Health, in conjunction with the United StatesAir Force, hosted this protocol with funding from Strategic Environmental Research and Development Program. Additional collaborators include the University of Cincinnati, and the Oregon Health Sciences University, the University of Texas, the University of North Carolina,Johns Hopkins University, the US Navy Toxicology Laboratory at Wright-Patterson AFB, OH,the NIOSH, and EPA/NERL herein referred to as the JP-8 Research Team.
Jet Propellant type 8 (JP8) jet fuel is the recognized battlefield fuel for all military operations for the United States, well beyond the year 2025, and represents the single largest source of chemical exposure to Department of Defense (DOD) personnel. Currently, DOD and its NATO partners use approximately 5 billion gallons of JP8 annually. The commercial equivalent, Jet-A, is the primary jet fuel used by aircraft in the US. Worldwide use of kerosene-based jet fuel is over 58 billion gallons per year.
The study was conducted at multiple Air Force installations. Dyess AFB, TX, served as the beta test site for participant selection, specimen collection, and exposure assessment. The lessons learned from the Dyess AFB beta test allowed the JP8 Research Team to improve data collection processes and study logistics, thus reducing the operational study impact at other Air Force bases involved in the study. Data was collected at the following sites: Davis Monthan AFB, AZ, Seymour Johnson AFB, NC, Langley AFB, VA, Pope AFB, NC, Little Rock AFB, AR, and Hurlbert Field, FL. Specimens and data
collected from these locations were analyzed at established laboratory facilities operated by the universities and government agencies involved in the study.
Two groups of airmen were enrolled. Those designated as JP8 exposed consisted of active duty Air Force personnel who routinely worked with or are exposed to JP8 in the performance of their duties. Most exposed volunteers worked in Aircraft Fuel Cell Maintenance shops. These workers routinely performed maintenance activities requiring entry into aircraft fuel tanks. Other exposed volunteers worked in either the Fuels Specialty or Fuels Transportation shops. In order to qualify for the study, exposed volunteers were required to have least 9 months of persistent exposure to jet fuel (such as fuel tank entry at least one hour twice weekly).
Unexposed volunteers were intended to represent the population of active duty Air Force personnel assigned to the Air Force installation where the study was being conducted. They consisted of active duty personnel assigned to the same Air Force installation as the JP8 exposed volunteers, but who do not have routine contact with JP8 or other fuels during the performance of their duty. A wide variety of job classifications were represented in the unexposed group. Since nearly all JP8 exposed volunteers were enlisted personnel, attention was paid to ensure, for the most part, that Air Force officers were not selected to participate. In rare cases, officers were included where the researchers felt their inclusion would not bias the analysis.
Broadly, JP8 exposure was measured both externally in the environment immediately surrounding enrolled workers and internally through the use of several body burden measures. The impact of exposure was evaluated using a series of neurological, hormonal and immunological measures. Cytotoxic and genotoxic effects of JP8 exposure were also evaluated. The activity of Glutathione-S-Transferase (G-S-T), a gene-regulated enzyme associated with increased susceptibility to multiple oxidative stressors including jet fuel and linked to adverse health outcomes, was also measured. Self-reported health problems, health care visit frequency, and early indicators of liver and kidney damage were investigated as part of the study.
The characterization of JP8 health risks, conducted by Texas Tech University, and the identification of uncertainties accounted for exposure measures of JP8 and measures of effect. The preliminary risk characterization attempted to determine the association between the various measures of effect used in this study and assesses the overall impact, by JP8 dose, on workers exposed to the fuel.
INTRODUCTION
Risk Assessment of Acute Exposure to Jet Fuel
Jet Propellant type 8 (JP8) jet fuel is the recognized battlefield fuel for all military operations for the United States, well beyond the year 2025, and represents the single largest source of chemical exposure to Department of Defense (DOD) personnel. Currently, DOD and its NATO partners use approximately 5 billion gallons of JP8 annually. The commercial equivalent, Jet-A, is the primary jet fuel used by aircraft in the US. Worldwide use of kerosene-based jet fuel is over 58 billion gallons per year.
Over the past 20 years, JP8 largely replaced JP4 as the primary aircraft fuel for US military aircraft. JP4, which is chemically similar to gasoline, is highly volatile. Explosive fires in both occupational and operational settings were experienced in military aircraft powered by JP4. JP8, although chemically similar to kerosene, is much less volatile. It is a much safer fuel to handle and less likely to propagate an explosion during instances when military aircraft fuel tanks suffer artillery or small arms damage during operational situations.
As JP8 was phased into the military inventory, exposed personnel began voicing concerns about the potential health effects of exposure. Aircraft groundcrew members reported objectionable odors, skin irritation, dizziness and the persistent taste of jet fuel long after exposure. These concerns prompted the Air Force Surgeon General to task the Air Force Institute for Environment, Safety and Occupational Health Risk Analysis (AFIERA) and the Air Force Research Laboratory (AFRL) to address personal exposure and toxicological hazards from JP8.
A reference report by the Center for Disease Control and Prevention’s (CDC) Agency for Toxic Substances and Disease Registry (ATSDR), “Toxi-
cology Profile for Jet Fuels (JP-5 and JP8),” in 1997 indicated that the toxicities of jet fuel and their mechanisms are not well-defined. According to ATSDR, data gaps exist on dose-response, reproductive system, developmental effects, immune system, neurological system, biomarkers of exposure and effect, rates of absorption, distribution and excretion of, and toxicokinetics in current research of human health effects from jet fuel exposure. Recently, JP8 jet fuel was selected as a priority hazardous chemical requiring establishment of an acute exposure limit by the Environmental Protection Agency’s (EPA) National Advisory Committee for Acute Exposure Guidelines for Hazardous Substances (NAC-AEGL), a subcommittee of the Office of Pollution Prevention and Toxics, US EPA. The NAC-AEGL further identified data gaps in the toxicology profile of jet fuel as submitted by the ATSDR. Recommendations from the NAC-AEGL include measuring total body burden, identifying biomarkers of exposure, conducting an epidemiology study of worst-case exposed populations, conducting neurological assessment, establishing reference dose (RfD) and risk assessment of exposure from JP8. In addition, a 1996 report by the National Research Council’s Committee on Toxicology (COT) identified data gaps in occupational exposure assessments, breath analysis, quantitative neurological effects and hepatotoxicity.
The COT report recommended the following:
Obtain information on exposures during operational procedures, including exposures to respirable aerosols of unburned fuels.
Conduct studies on the possible effects of high-level acute and low-level chronic exposures to military fuel vapors on CNS, including the effects on performance of military personnel.
Conduct further research on the effect of military fuel vapors on hepatotoxicity in experimental animals.
Based on the Air Force Surgeon General’s tasking and ATSDR, COT and NAC-AEGL recommendations, AFIERA initiated a program to evaluate all environmental, safety and occupational health aspects of jet fuel and began collaboration with the National Institute for Occupational Safety and Health (NIOSH), the Environmental Protection Agency, National Exposure Research Laboratory (EPA-NERL), the National Institute for Environmental Health Sciences (NIEHS), and selected academic institutions to resolve open issues regarding JP8. The USAF JP8 Environmental, Safety and Occupational Health Integrated Process Team (IPT), formed in 1996 and in coordination with the Air Force Office of Scientific Research (AFOSR), conducted and funded animal toxicology studies on aerosol exposure, dermal flux and adsorption, biomarkers and neurological assessments. The IPT has also conducted
and funded occupational exposure studies to include ambient vapor and aerosol exposure assessment, breath sampling, and heat stress assessment.
Based on exposure data from previous AFIERA studies, fuel tank repair operations of single-point-entry fuel bladders containing fire suppressant foam were determined as the worst-case exposure situations. The highest exposure results were measured in operations performed inthe C-130 Hercules transport aircraft’s auxiliary fuel tanks.
The studies conducted by AFIERA and other investigators, including toxicology studies supported by the AFOSR, validated the need for research on JP8 impact on workers in occupational settings. In particular, studies of the acute effects of exposure were considered most important. Based on these assessments, this study, entitled Risk Assessment of Acute Exposure to Jet Fuel, was developed and initiated. Prior to this study, no occupational exposure cohort studies had been conducted to assess the effects from acute exposure to JP8 jet fuel. Further, no acute exposure or risk assessment studies had attempted to link quantitative neurological measurements to ambient exposure, biomarkers, and total body burden. This study breaks new ground by correlating ambient exposure with human body burden and neurological performance measures. The results of this study are intended to aid in establishing limits for exposure in both occupational and community settings. The study helps to determine specific occupational exam requirements, personal protective equipment requirements and methods for monitoring exposure. Additionally, by correlating ambient exposure measures with health and performance outcomes, we hope to use the data obtained from this study to extrapolate the extent of community risks associated with ubiquitous, low-level jet fuel exposure.
The study was conducted in conjunction with Texas Tech University, the University of Cincinnati, and the Oregon Health Sciences University. Additional collaborators include the University of Texas, the University of North Carolina, Johns Hopkins University, the US Navy Toxicology Laboratory at Wright-Patterson AFB, OH, the NIOSH, and EPA/NERL.
The study’s purpose was to assess the influence of acute exposure to jet fuel on the health, safety and operational capability of the Air Force population and gain insight into the risk posed by JP8 on the general local population.
The specific aims were to
Compare exposure levels of a selected worst-case exposed cohort to the generally unexposed base workforce.
Determine level of body burden of jet fuel within each exposure group.
Analyze biological specimens from each subject group for jet-fuel-linked specific biomarkers of exposure and effect.
Perform an epidemiology analysis of each subject group.
Assess the impact of JP8 exposure on performance and health outcomes.
Perform a risk analysis for environmental and occupational communities based on collected sample data.
The primary hypotheses addressed through this study are the following:
Is exposure to JP8 detrimental to the health and safety of flightline workers? Does a low-level ambient exposure to jet fuel have an adverse impact on the general community at an Air Force installation?
The study was conducted at multiple Air Force installations. Dyess AFB, TX, served as the beta test site for participant selection, specimen collection, and exposure assessment. The lessons learned from the Dyess AFB beta test allowed the JP8 Research Team to improve data collection processes and study logistics, thus reducing the operational study impact at other Air Force bases involved in the study. Data was collected at the following sites: Davis Monthan AFB, AZ, Seymour Johnson AFB, NC, Langley AFB, VA, Pope AFB, NC, Little Rock AFB, AR, and Hurlbert Field, FL. Specimens and data collected from these locations were analyzed at established laboratory facilities operated by the universities and government agencies involved in the study.
General Methods
The Risk Assessment of Acute Exposure to Jet Fuel study measured JP8 exposures in an operational environment and assessed the impact of exposure on the performance and health of those enrolled in the study. JP8 exposure was measured both externally in the environment immediately surrounding enrolled workers and internally through the use of several body burden measures. The impact of exposure was evaluated using a series of neurological, hormonal and immunological measures. Cytotoxic and genotoxic effects of JP8 exposure were also evaluated. The activity of Glutathione-S-Transferase (G-S-T), a gene-regulated enzyme associated with increased susceptibility to multiple oxidative stressors including jet fuel and linked to adverse health outcomes, was also measured. Self-reported health problems, health care visit frequency, and early indicators of liver and kidney damage were investigated as part of the study.
Study Logistics
The Jet Fuel Research Team, a group of approximately 30 researchers from six academic institutions, two government agencies and two military services, traveled to six Air Force bases in the continental United States to conduct the study. Visits were coordinated in advance to obtain Commander permission to conduct the study. Commanders were briefed in person or by video teleconference prior to the visit to provide information on the rationale for the study, the study goals, milestones to be accomplished during the visit, and the logistics associated with conducting the study on their base. The study was conducted during a two-week period at each study site. One Air Force base was visited every month between April and September 2000. A beta test was conducted prior to the initial site visit to test the logistics of moving people and equipment and synchronizing the timing of multiple specimen collections and testing applications.
Study Subject Recruitment:
Recruitment at each study site was initiated prior to study team arrival and continued throughout the first week of the visit. Subjects were recruited for the study through several vehicles. Since the primary exposure group for the study were workers from shops where contact with jet fuel routinely occurs, the supervisors of such shops as Aircraft Fuel Systems Maintenance, Fuels Transportation, and Fuels Specialty were directly contacted to gain support for the study and solicit volunteers. Members of the fuels community, particularly aircraft fuel systems maintenance personnel, supervisors and commanders, showed high interest in the project and large numbers of workers from these shops volunteered for the study.
Additional recruitment efforts consisted of briefings at Commanders Calls, and informational press releases and solicitation advertisements in local military installation newspapers. At some study locations, First Sergeants were contacted to help gain support for the study. A financial incentive of $50.00 was provided by Texas Tech University to compensate subjects for their participation outside of regular duty hours. Those who completed all requested tests and provided all requested specimens received $50.00. Any subject who dropped out prior to completing the study received $10.00.
Recruitment was successful at all study locations. At several locations, volunteers were turned away after a sufficient number of subjects was achieved. While the study actively recruited females, few women work in jobs where jet fuel exposure occurs. The unexposed to exposed ratio for women
was increased 2:1 as originally planned 4:1 in an attempt to improve the ability to detect differences in effect.
Study Subject Enrollment:
Two groups of airmen were enrolled. Those designated as JP8 exposed consisted of active duty Air Force personnel who routinely worked with or are exposed to JP8 in the performance of their duties. Most exposed volunteers worked in Aircraft Fuel Cell Maintenance shops. These workers routinely performed maintenance activities requiring entry into aircraft fuel tanks. Other exposed volunteers worked in either the Fuels Specialty or Fuels Transportation shops. In order to qualify for the study, exposed volunteers were required to have least 9 months of persistent exposure to jet fuel (such as fuel tank entry at least one hour twice weekly).
Unexposed volunteers were intended to represent the population of active duty Air Force personnel assigned to the Air Force installation where the study was being conducted. They consisted of active duty personnel assigned to the same Air Force installation as the JP8 exposed volunteers, but who do not have routine contact with JP8 or other fuels during the performance of their duty. A wide variety of job classifications were represented in the unexposed group. Since nearly all JP8 exposed volunteers were enlisted personnel, attention was paid to ensure, for the most part, that Air Force officers were not selected to participate. In rare cases, officers were included where the researchers felt their inclusion would not bias the analysis.
All volunteers were informed of the nature of the study and the potential risks associated with participation. By groups of approximately 50, volunteers were given a 30 to 45 minute briefing by an occupational medicine physician. The script used for the briefing had undergone extensive review and testing prior to employment. Groups of researchers and potential volunteers were asked to comment on the briefing during the beta-testing portion of the study. In addition, volunteers at each study site were asked to comment of the acceptability and completeness of the briefing. Without exception, the members of the JP8 study team and study volunteers considered the standardized briefing highly acceptable.
In addition to the briefing, study volunteers were asked to complete a questionnaire designed to obtain information on specific criteria that could disqualify them from participating in the study. Exclusion criteria consisted of conditions that would impact the validity of either study effects or exposure measures. Those criteria were as follows:
Alcohol use within 24 hours prior to entering the study period
Injury requiring medical attention within the last 6 months
History of melanoma
History of congenital night blindness
History of lung or ovarian cancer
History of adult cerebral vascular accident
History of diabetes
History of scoliosis
Major visual impairment
Clinical diagnosis of seizures
On medical profile
Pregnancy
Currently taking any medications determined by an occupational medicine physician to be disqualifying. Such medications included:
Hypertension medication
Antacids or medication for heartburn
Diet pills or other stimulants
Tranquilizers or muscle relaxants
Antidepressive medication
Psychotherapeutic medication
Large doses of megavitamins containing high levels of antioxidants
Each volunteer underwent a personal interview with either an occupational or preventive medicine board certified physician where the volunteer’s completed questionnaire was reviewed and specific volunteer questions were addressed. After the physician determined the volunteer was eligible to participate in the study, the volunteer and the physician completed an informed consent document. The new enrollee was then given appointments for study testing. Each enrollee was assigned a unique study code consisting of the first three letters of his or her assigned Air Force base, e.g. Pope AFB = POP, and a randomly generated number between 1,000 and 9,999. A reference log consisting of enrollee’s social security number, subject code, and exposure group classification was created, maintained, and safeguarded by the occupational medicine physician. All researchers throughout specimen collection, performance testing, and data analysis phases of the study used the study codes for recording information relative to the enrollee. The use of study codes helped maintain subject confidentiality and assisted in blinding researchers to enrollee exposure status. At the end of the study, the reference log was forwarded to Texas Tech University for permanent storage.
Specimen and Data Collection
In most cases, all exposure measurements and performance/health effects testing were conducted during one subject’s workday. Enrollees typically reported for testing on an appointed morning. Each subject was asked about their alcohol and tobacco consumption during the 24 hours prior to testing and whether he or she was experiencing cold or allergy symptoms. Those with cold or allergy symptoms and those who had consumed alcohol within 24 hours were rescheduled to another day whenever possible. Tobacco use was recorded.
From those who met morning test entry parameters, specimens of blood, breath, urine and epidermal skin were collected. Samples of the cells from the interior of the cheek were also collected for later testing. The enrollees completed a series of tests designed to measure various neurological parameters. Prior to returning to work, each volunteer was fitted with equipment designed to collect samples of the air within their breathing zones during the work period. Enrollees were also fitted with equipment designed to measure their heart rate and core body temperature throughout the workday.
After undergoing morning testing, the enrollees returned to their usual workplace and performed routine duties for a period of at least 4 hours. During the time the enrollees were at work, members of the research team collected environmental measures. While most of the environmental samples were collected in or near the Aircraft Fuel System Maintenance Shop, a representative number of samples were gathered from other locations to ensure that those enrollees categorized as unexposed were, in fact, unexposed to jet fuel or similar chemicals.
In the afternoon, enrollees returned to the study site where environmental and vital status monitoring equipment was removed. Post-workday specimens of blood, breath, urine and epidermis were collected and a series of tests similar to those conducted in the morning were repeated. Questionnaires were applied to the enrollees to obtain information regarding the level of mental and physical exertion experienced during the day and details of the individual’s activities throughout the work period. Questionnaires designed to capture information on self-reported symptoms, lifestyle risk (such as smoking and drinking), and the use of personal protective equipment were also applied.
After completing all specimen collection and testing, the enrollees received their study stipend and were released. At the end of the week, the researchers departed the base. Of note, the researchers collected information on the exact time of day each specimen was collected and each test was performed for each enrollee using a subject-time-series log. These time-series data were made available to all investigators to aid in analysis.
One test, the electroretinogram (ERG), was not accomplished during the typical data collection week. The ERG, a method of measuring retinal function, was administered to a subset of enrollees during the week prior to the normal data collection period. Studies in animals chronically-exposed to JP8 have shown selective cellular damage to cells located in the retina and cerebellum. Since any retinal changes detectable by the ERG would be the result of chronic exposure, repeat ERG testing (pre and post work period) was unnecessary. Approximately 20 subjects at each base were selected to complete the ERG.
Exposure measures:
The JP8 exposure measures conducted as part of the JP8 study are briefly discussed below. A more detailed explanation of each exposure measure is provided in the abstracts included in this report.
Biological measures:
Blood: Each subject submitted two 40-ml blood specimens—one specimen during the morning test period and one in the afternoon. Trained phlebotomists from the Air Force Research Laboratory (AFRL) at Wright-Patterson AFB collected all blood specimens. Each blood specimen was divided into three aliquots. Texas Tech University conducted quantitative analysis for the enzyme Glutathione-S-Tranferase (G-S-T) in blood. Researchers from Brooks AFB, in collaboration with researchers from the EPA, conducted analysis for JP8 markers. Scientists from the University of North Carolina analyzed blood specimens for metabolites of benzene and naphthalene. NIOSH and Navy collaborating scientists conducted additional biomarker analyses. A small amount of residual blood from each subject was provided to AFRL for physiologically-based pharmacokinetic (PBPK) modeling of jet fuel metabolism.
Urine: Urine samples collected prior to and after the sampling period were divided into two aliquots. Researchers from the University of North Carolina analyzed urine for the presence of metabolites for benzene and naphthalene. NIOSH conducted analysis of urine samples for the presence of renal biomarkers of exposure.
Breath: Three breath samples were typically collected before and after the work period. Using devices called SUMMA canisters, a scientist from the EPA collected breath samples from selected enrollees and analyzed the specimens
for the presence of JP8 markers. Breathe samples, collected using a 75-ml glass bulb collection device, were processed by University of North Carolina scientists to identify the presence of benzene and naphthalene. Using a third breath collection method, a researcher from Johns Hopkins University obtained pre-and post-work samples from selected enrollees and performed an analysis to quantify the amount of JP8 constituents contained in each specimen.
Skin Exposure Sampling: Epidermal specimens were collected prior to and following the work period using a dermal taping method. The skin specimens were analyzed for the presence of naphthalene by researchers from the University of North Carolina.
Body Temperature Monitoring: Internal body temperature, a potential confounding variable in the association between jet fuel constituent metabolism and performance/health measures, was monitored during the enrollee’s work period. Selected subjects were asked to swallow a small pill-like sensor. The device provided continuous monitoring of body core temperature during the enrollee’s work period. Other enrollees were asked to wear an aural or skin temperature probe. All enrollees wore Polar Band heart rate monitors around the chest area, and activity sensors on the wrist.
Performance/Health Measures:
Enrollees were asked to submit to a series of performance and health effects measures. Tests included the Global Assessment System for Humans/ Behavioral Assessment and Research System (GASH/BARS), the Postural Sway Test, the Eye Blink Conditioned Response Test and the Electroretinogram (ERG). Subjects were also asked complete an electronically administered questionnaire. Medical records were reviewed for pertinent health events occurring during the preceding year.
Global Assessment System for Humans (GASH)/ Behavioral Assessment and Research System (BARS): The GASH/BARS system consists of a series of computer-based neurobehavioral tests designed to measure motivation, response speed, coordination, grip strength, complex mental functioning, memory, and attention. Subjects completed the GASH/BARS test series prior to and after the work period. Data from the subjects’ Air Force Qualifying Test (AFQT) were also obtained from the Air Force Personnel Center and used to support the GASH/BARS analysis. AFQT exam scores were coded using subject codes to protect subject confidentiality and ensure study blinding.
Electroretinogram (ERG): The ERG is a device designed to measure the electrical response of the eye to brief, high intensity flashes. In this study, the ERG was used to determine the association between JP8 exposure and retinal Mueller cell function. Subjects who volunteered for this protocol underwent an ERG as part of their evaluation. Since the hypothesized retinal changes are associated with chronic JP8 exposure, the ERG procedure was accomplished only once on the enrollee selected. In addition to comparisons between the exposed and unexposed groups, ERG results were compared with normative data.
Postural Sway: A team of researchers from the University of Cincinnati conducted a series of tests to assess the enrollee’s balance. During the test, subjects were asked to perform a series of procedures while standing on a platform designed to measure changes in balance. The procedures included standing on the platform alone and with a foam pad between the platform and enrollee’s feet while performing a series of procedures with their eyes open or closed. Each enrollee also answered a short list of questions prior to postural sway testing.
Eye Blink Conditioned Response (ECR): The eye blink response is a reflex that can be classically conditioned. The ECR is considered a sensitive measure of more global issues of brain functioning, and is appropriate for assessing robust and/or subtle changes in neural processing that one might expect from repeated exposure to jet fuel vapors. Enrollees completed the ECR during pre- and post-work periods. Navy technicians conducted this procedure on selected enrollees. The Navy Neurotoxicology Group at Wright-Patterson AFB analyzed the results of ECR tests.
Risk Factor Questionnaire. Each subject volunteer completed a series of questions designed to assess self-reported symptoms, and exposure to potentially confounding factors, such as alcohol and tobacco. Questions regarding hobbies and work-shift history were also addressed. Subjects also completed a series of standardized questions from a copyrighted questionnaire termed the SF-36. Both questionnaires were administered electronically after completion of the GASH/BARS.
Medical Records Review: Epidemiologists from Texas Tech University and AFIERA reviewed the medical records of those enrolled in the study. The epidemiologists recorded health care events occurring during the year prior to the study period using broad disease categories. Associations between health care event frequency and JP8 exposure were tested using these data.
Analysis
Data collection:
Members of the JP8 research team collected all exposure specimens and outcome data. For the most part, the researchers associated with specific sub-protocols (such as the Postural Sway Test) included in the overall JP8 study were responsible for applying tests, analyzing specimens and collecting data specific to their sub-protocols. The exceptions to this rule were time-series logs, study eligibility, exertion and daily activity questionnaires, and blood specimen collection. The blood specimens were collected by AFRL phlebotomists, divided into aliquots and provided to other researchers. The questionnaires and logs were collected by AFIERA personnel, entered into spread-sheets and provided to all researchers to aid in their analyses.
In total, 339 Air Force active duty members were enrolled in the study. Of those enrolled, 324 completed all required tests and submitted all required specimens. Some enrollees were not able to complete the entire study due to unavoidable circumstances. Eight enrollees completed only the ERG, 3 only completed a questionnaire, 2 completed only the ERG and a questionnaire, and 2 enrollees completed all but one or two of the required tests.
Initial Exposure Classification:
Using information provided by the enrollees and exposure stratification assignment at the time of enrollment, those enrolled in the study were categorized into one of four groups. The exposure categories were based on the probability of JP8 exposure in the completion of normal operational duties. The exposure categories and decision tree for enrollee categorization are listed below.
The categorization scheme was developed and employed for two reasons. First, as pointed out in the abstracts following this section, processing specimens is time-consuming. Six months after data collection, some exposure data, particularly measures of JP8 in blood, remained unavailable to researchers. Measures of JP8 effects, particularly neurological test results, were however available for analysis shortly after the end of the data collection phase. The categorization scheme was employed to allow those measuring JP8 effects to obtain a preliminary assessment of degree to which JP8 impacted human performance. The second reason for employing the categorization scheme relates to chronic measure of effects. Effects measures, such as the ERG, medical visit history and self-reported symptoms, are not necessarily related to acute JP8 exposure, but may be influenced by chronic exposure to jet fuel.
CATEGORY | CRITERIA | ||
HI | Classified as exposed by study bioenvironmental engineer? YES | ||
| AND BY SELF REPORT | ||
| Does your current job routinely bring you into physical contact with jet fuel? YES What is your primary AFSC? 2A6X4 Does working in your primary AFSC bring you into physical contact with jet fuel? YES Age under 35? YES | ||
HI MOD | Classified as exposed by study bioenvironmental engineer? YES | ||
| BY SELF REPORT | ||
| Does your current job routinely bring you into physical contact with jet fuel? YES Does working in your primary AFSC bring you into physical contact with jet fuel? YES | ||
| AND | ||
| What is your primary AFSC? |
| 2FOXX 2T3XX 2E4XX |
| OR | ||
| Classified as exposed by study bioenvironmental engineer? YES | ||
| BY SELF REPORT | ||
| Does your current job routinely bring you into physical contact with jet fuel? YES/ NO Does working in your primary AFSC bring you into physical contact with jet fuel? YES/NO MUST ANSWER NO TO ONE OF THE ABOVE QUESTIONS | ||
| AND | ||
| What is your primary AFSC? | 2A6X4 | |
| OR Primary AFSC = 2A6X4 and Age 35+ | ||
MOD | Classified as exposed by study bioenvironmental engineer? NO | ||
| BY SELF REPORT | ||
| Does your current job routinely bring you into physical contact with jet fuel? YES/ NO Does working in your primary AFSC bring you into physical contact with jet fuel? YES/NO MUST ANSWER YES TO ONE OF THE ABOVE | ||
| AND | ||
| What is your primary AFSC? | ANY BUT | 2A6X4 2FOXX 2T3XX 2E4XX |
LOW | Classified as exposed by study bioenvironmental engineer? NO | ||
| BY SELF REPORT | ||
| Does your current job routinely bring you into physical contact with jet fuel? NO Does working in your primary AFSC bring you into physical contact with jet fuel? NO | ||
| AND | ||
| What is your primary AFSC? | ANY BUT | 2A6X4 2FOXX 2T3XX 2E4XX |
Since the categorization scheme assesses the probability of occupational JP8 exposure, it serves well as a means for stratifying enrollees based on chronic exposure.
While the categorization scheme provides a method for determining JP8 exposure, body burden measures employed in this study provide a direct method for quantifying exposure. Direct body burden measures should be far superior to categorization in assessing acute exposure. Three strategies were employed to hasten the availability of JP8 body burden measures.
Strategy One. Prioritization of Specimen Processing
While the processing of all samples is needed to achieve sufficient sample size to determine statistically significant differences in health and performance effects based on JP8 body burden, investigators could estimate the strength of JP8 effects by conducting analyses using a representative sample of the data. To provide this sample as quickly as possible, a stratified random sample of the study enrollees was selected and all investigators were notified to process specimens from these enrollees first. A weighted sample consisted of 110 enrollees chosen at random after stratifying all enrollees by exposure category. Research may refer to this prioritized sample in the abstracts that follow.
Source: Egeghy, University of North Carolina
Strategy Two. Selection of a Single Continuous Measure of Exposure
In January 2001, a group of JP8 researchers, including the three researchers responsible for measuring JP8 in breath specimens, met to consider whether one single breath measure could sufficiently provide a continuous measure of JP8 body burden for use by other researchers involved in the study. The group reviewed available breath data and found reasonable agreement between the three breath measures with correlation coefficients in the 0.7 to 0.8 range. As graphically represented below, breath measures via EPA and John Hopkins methodologies though highly correlated, were not available for all subjects. The breath measures provided by the University of North Carolina provided data on two constituents of JP8: benzene and naphthalene. Though not as highly correlated as the EPA and Johns Hopkins data, the naphthalene data was generally in agreement with the other breath measures and was available on nearly all enrollees. The exposure measurement team of researchers agreed to provide naphalene measurement data to all collaborating researchers for use in assessing the impact of JP8 acute exposure. The abstracts that follow will, in some cases, refer to these measures in their analyses.
Strategy Three.
Preliminary analysis of JP8 exposure using post-workday naphthalene breath samples failed to demonstrate any association between exposure and
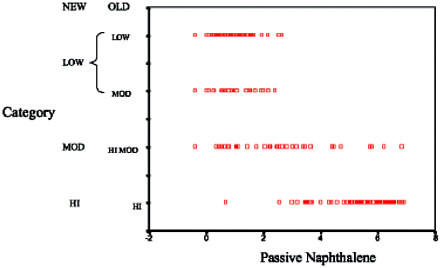
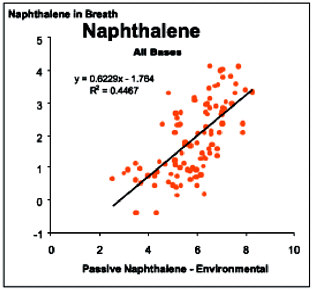
the three primary neurologic tests used in the study. Not only was no association demonstrated, the findings noted during analysis using the exposure categories described under Strategy One were lost. Further, an analysis performed by Egeghy and graphically displayed below showed weak correlations (R2 = -0.44) between post-workday naphthalene specimens and subject-specific environmental naphthalene samples.
Based on these findings, the research team investigated the correlation between environmental naphthalene samples and the previously developed exposure categories. Initial analysis showed much higher correlations (R2 = -0.83). Investigation of outliers revealed the highest environmental naphthalene measures among the LOW exposure category involved subjects tested on Monday and Tuesday at Davis Monthan AFB. Study notes showed investigators were concerned about the possibility of secondary exposure among LOW exposed subjects during post-sampling periods on Monday and Tuesday. LOW exposed subjects returned to the sample collection site at the same time as HI exposed subjects. Further, because of the building design, a strong fuel odor was reported to the research team. Measures were taken to prevent secondary exposure during the following days at Davis Monthan AFB and at subsequent study locations. Based on these findings, the LOW exposed subjects from Monday and Tuesday at Davis Monthan AFB were eliminated from the preliminary analyses.
Outlier analysis also revealed one subject LAN9356 was miscoded. The subject, originally coded as moderately exposed (MOD), actually worked in Aircraft Fuel System Repair Shop and handled fuel-soaked fire suppression foam on the day of the test. After the subject’s exposure code was changed and the Davis Monthan ABF LOW-exposed subjects from Monday and Tues-
day were removed from the dataset, correlation coefficients improved to R2 = -0.86. Since the LOW and MOD categories were indistinguishable with respect to environmental naphthalene measures, the categories were collapsed into one category term LOW. The HI MOD category was renamed MOD and the HI category remained unchanged. Researchers may refer to this new categorization scheme in the abstracts included in the report.
Risk Characterization:
The full characterization of JP8 health risks, to be conducted by Texas Tech University, will take into account exposure measures of JP8 and measures of effect. The risk characterization will attempt to determine the association between the various measures of effect used in this study and assess the overall impact, by JP8 dose, on workers exposed to the fuel. Details regarding the JP8 risk analysis are provided at the conclusion of this report.
General Results
Enrollment Results
Potential subjects for the Risk Assessment of Acute Exposure to Jet Fuel study were solicited from six Air Force bases in the continental United States. Of the approximately 450 candidates who responded to recruitment efforts, 394 received the study briefing and completed the exclusion criteria questionnaire. From these candidates, 339 subjects met minimal enrollment criteria and entered the study. Enrollment percentages by study site are listed below.
Of the 284 males and 55 females that began the study, 15 withdrew before the study was completed. Eight completed only the electroretinograph (ERG) test, 3 completed only the Risk Factor Questionnaire (RFQ), and 2 completed both the ERG and RFQ before dropping out. Two additional subjects withdrew with after completing nearly all parts of the study. Most of the withdrawals (7) were due to a tropical storm that arrived at Hurlbert Field the week of the study. The storm forced researchers to cancel the final data collection day of the study.
Details regarding the enrolled subjects are included in the table below. A total of 284 men and 55 women were enrolled. Subject ages ranged from 18 to 44 years, with an average age of 26.1 and a median age of 24 years. The exposure categories used in the table below represent the revised categorization discussed in the General Methods section. The LOW categories include subjects with no or rare exposure to JP8. Subjects in the MOD category do not have daily exposure to JP8, but may periodically perform tasks requiring fuel
Study Site | Number Considered | Number Enrolled | Percent Enrolled |
Davis Monthan | 74 | 65 | 87.8 |
Seymour Johnson | 70 | 49 | 70.0 |
Langley | 66 | 59 | 89.4 |
Pope | 56 | 60 | 93.3 |
Little Rock | 64 | 49 | 76.6 |
Hurlbert Field | 64 | 57 | 89.1 |
Total | 394 | 339 | 86.0 |
Reasons for ineligibility included: | |
Preexisting medical condition | 20 |
Contraindicated prescription drugs or over-the-counter vitamins | 17 |
Recent surgery | 3 |
Not enough time-on-station | 3 |
TDY during the week of the study | 4 |
On quarters or profile | 2 |
Pregnant | 2 |
Candidate opted out | 4 |
TOTAL | 55 |
exposure. The HI category includes only personnel assigned to Aircraft Fuels System Maintenance shops.
Exposure categories were reasonably comparable with respect to right-handedness, race, height, weight and body mass index. The percentage of smokers, alcohol, caffeine, and processed meat users is approximately the same in all categories. Subjects report approximately the same number of hours worked in a week and engage in same amount of physical activity off duty. While subjects in the highest JP8 exposure categories were, on average, younger than those in the other exposure categories, the biological significance of these age differences is questionable. No differences are seen in the months on the base and in the current job. In two different physical exertion measures, however, the amount of physical work required to perform duties associated with their job, is significantly greater (P-value < 0.001) for those in the HI category. No differences are seen in the mental exertion required. Male sub-
| Risk Assessment of Acute Exposure to Jet Fuel | |||||
| Personal Characteristics | |||||
| Exposure Status | |||||
| Males = 284 |
| Females = 55 |
| ||
| HI | MOD | LOW | HI | MOD | LOW |
Age (mean) | 24.6 | 26.8 | 27.6 | 22.6 | 33.8 | 24.8 |
Age by group (%) |
| |||||
Under 20 | 3.5 | 2.6 | 3.1 | 20.0 | 0.0 | 5.0 |
20 to 24 | 57.4 | 48.7 | 42.3 | 50.0 | 0.0 | 55.0 |
25 to 29 | 27.8 | 20.5 | 20.0 | 20.0 | 20.0 | 25.0 |
30 to 34 | 4.3 | 7.7 | 15.4 | 0.0 | 20.0 | 7.5 |
35 to 39 | 5.2 | 17.9 | 13.8 | 0.0 | 60.0 | 5.0 |
40 and over | 1.7 | 2.6 | 5.4 | 0.0 | 0.0 | 2.5 |
Right Handed (%) | 83.5 | 76.9 | 83.8 | 100.0 | 80.0 | 95.0 |
Caucasian (%) | 80.0 | 74.4 | 72.8 | 80.0 | 100.0 | 70.0 |
Height in inches (mean) | 70.5 | 70.5 | 70.7 | 66.2 | 66.2 | 65.0 |
Weight in lb (mean) | 178.0 | 182.3 | 186.7 | 143.7 | 151.4 | 145.0 |
Body mass index (mean) | 28.4 | 27.7 | 27.2 | 31 | 29.7 | 29.5 |
Smoke at least 1/4 pack per day (%) | 43.9 | 47.4 | 32.3 | 50.0 | 60.0 | 30.8 |
Alcohol users (%) | 61.7 | 66.7 | 74.2 | 75.0 | 100.0 | 64.1 |
Daily caffeine users (%) | 51.9 | 66.7 | 53.5 | 75.0 | 60.0 | 28.2 |
Eat processed meats >1 time per week (%) | 43.5 | 35.9 | 40.3 | 37.5 | 20.0 | 28.9 |
Months of the job | 53.8 | 49.2 | 57.5 | 31.9 | 34.6 | 30.55 |
Months on current base | 33.3 | 28.5 | 33.2 | 19.8 | 43.2 | 26.9 |
Engage in physical activity 1-2 times per week (%) | 62.5 | 66.7 | 60.7 | 100.0 | 100.0 | 61.1 |
Work 8-10 hours per day at job (%) | 94.4 | 84.6 | 85.9 | 75.0 | 100.0 | 89.7 |
A great deal of physical work is required as part of job (%) | 13.9 | 5.1 | 7.0 | 25.0 | 0.0 | 0.0 |
Physical exertion score (mean) | 10.7 | 9.3 | 7.0 | 10.2 | 8.4 | 5.6 |
Mental exertion score (mean) | 3.9 | 3.5 | 4.0 | 3.5 | 4.0 | 3.9 |
Find job very stressful (%) | 26.2 | 5.3 | 19.5 | 12.5 | 20.0 | 5.1 |
jects in the HI category are more likely to report a great deal of stress associated with their job. Due to much smaller sample size, comparisons of personal characteristics among females are less stable than those of males.
JP8 exposure results and the impact of JP8 exposure on various performance tests and health outcome measures are reported in the abstract found in this report.



























