Exploring Sleep Disturbance in Central Nervous System Disorders: Proceedings of a Workshop (2023)
Chapter: 3 Underlying Mechanisms and Model Systems of Sleep
While advances in the field have led to greater understanding of sleep and circadian rhythms, continued research is needed to explore mechanisms of sleep, said Louis Ptáček. Studying the mechanisms of sleep may provide not only pathways toward addressing sleep disorders, but also insight into the core functions of sleep—“the why of sleep”—which led to its evolution and maintenance, said David Raizen, associate professor of neurology, medicine, and genetics at the Perelman School of Medicine, University of Pennsylvania.
CIRCADIAN TIMING
Sleep is regulated by two major and interacting processes, commonly known as process S and process C, said Erik Herzog, professor of biology at Washington University in St. Louis (Borbély, 1982; Borbély et al., 2016). He explained that process S refers to the homeostatic sleep drive wherein sleep deprivation results in progressive building of sleep pressure. Process C, the circadian process, evolved to help humans and other organisms anticipate the challenges of life on Earth by organizing their behaviors into their appropriate niches around the light/dark cycles. For example, “[for diurnal creatures] the idea that you need to be ready for the day when you’re going to be active, and ready for night when you’re going to be starving and fasting,” said Herzog.
As mentioned in Chapter 1, circadian pacemakers run with a period of nearly 24 hours, synchronize primarily to light/dark cycles, and drive daily rhythms in physiology and behavior, said Herzog. About a dozen mammalian clock genes have key roles in keeping 24-hour time in a transcription/translation negative feedback loop providing the molecular basis for gener-
ating daily rhythms and regulating other genes with circadian rhythms, he said. The clock regulates physiology in individual cells, including neurons and other cell types including glial cells, cells in the liver, cells involved in uterine function, and cells involved in producing cartilage. “The list of circadian cell types just goes on,” said Herzog.
A structure in the brain called the suprachiasmatic nucleus (SCN) has been shown to be a central circadian pacemaker in vertebrates, said Herzog (Ralph et al., 1990). Amita Sehgal noted that the central clock in the SCN synchronizes other clocks that exist in all tissues and cells. Lesioning of the SCN leads to a loss of daily rhythmicity in sleep and other physiological functions, said Herzog. Input from the retina synchronizes the SCN, and synaptic projections from the SCN have been mapped to many structures associated with wake and sleep cycles. Signaling by neurotransmitters like gamma-aminobutyric acid (GABA) and neuropeptides like vasoactive intestinal polypeptide (VIP) keep different cell types in this network synchronized to each other (Sanchez et al., 2022). “We’ve got to think about the system as a functional network of clocks,” said Herzog, adding that environmental factors can tune or disrupt normal GABA and VIP signaling.
Sehgal added that other cell types and neuromodulators—dopamine, norepinephrine—secreted by these different cell types contribute to the regulation of sleep and that these neuromodulators have conserved effects on sleep in flies and mammals.
GENETICS
Gene mutations are key to understanding the molecular mechanisms underlying sleep and the circadian clock, said Sehgal. Although much progress has been made in identifying genetic mechanisms involved in circadian rhythms, “We want to discover new genes involved in sleep,” she said, arguing that an unbiased approach is the best approach. “You let the organism tell you what’s important for sleep rather than going in with any preconceived notions.” Sehgal further argued that flies have distinct research advantages to better understand sleep and circadian disorders. “The fly lends itself beautifully to forward genetics,” she said, where mutagenized animals can be screened for aberrant functions, and then the mutated genes responsible for the phenotype can be cloned. She noted that the first circadian genes were identified in Drosophila through forward genetics (Konopka and Benzer, 1971). These genes were subsequently shown to be mutated in human circadian disorders. Studies by multiple research groups have further shown that sleep-regulating genes are conserved from worms to humans, said Sehgal.
Ying-Hui Fu uses a genes-first approach to study human sleep behavior. Her approach takes advantage of four naturally occurring familial human
sleep–wake phenotypes: (1) the advanced sleep phase of morning larks; (2) the delayed sleep phase in night owls; (3) natural short sleepers who sleep fewer hours than is typical; and (4) natural long sleepers who sleep longer than is typical. These four types can be divided into those with atypical sleep schedules (morning larks and night owls) versus those with atypical sleep duration (short and long sleepers), said Fu.
Over the past 20 years, Fu and colleagues have reported more than a dozen mutations in molecular core clock components. Among these are mutations in the Per3 gene, which results in the morning lark phenotype as well as seasonal affective disorder, said Fu (Zhang et al., 2016). Other mutations found in morning larks have also been linked to other conditions such as migraine. Studying these genes can help clarify the connection between sleep and these other conditions.
For example, another gene called Per2 produces a protein that is under circadian regulation. By studying a mutation in this gene, Fu and colleagues demonstrated posttranslational modifications in the mutation region of the gene that are also under circadian regulation and in some cases are dependent on nutrient intake. Under high glucose concentration in the cell or body, the posttranslational modification O-GlcNAcylation leads to a faster clock, said Fu (Kaasik et al., 2013).
Fu and colleagues have also identified five mutations on four genes associated with the natural short sleep behavior trait. People with this trait only need 4 to 6 hours of sleep per day, stay active during waking hours, and live long, healthy lives, said Fu. By studying these mutations, her team has been able to elucidate the circuitry that regulates sleep duration, which Fu said has turned out to be very complex. Ptáček added that different pathways may lead to the short sleep phenotype and that there may be heterogeneity with regard to different dimensions of sleep, such as sleep architecture and tolerance of sleep pressure.
Fu and colleagues noticed that while natural short sleepers live long, healthy lives, individuals who experience long-term sleep deprivation or disruption have a greater chance of developing a host of diseases, including high blood pressure, cancer, and Alzheimer’s disease. The most logical explanation suggested by this observation, according to Fu, is that even though short sleepers sleep fewer hours, their sleep quality or sleep efficiency must be higher and offers them protection against other diseases. To test this idea, they crossed mice carrying short sleep mutations with two different Alzheimer’s mouse models—one that forms amyloid plaques and the other that forms tau tangles. In both models, the mice with short sleep mutations had dramatically reduced plaque/tangle pathology (Dong et al., 2022). She added that mice with short sleep mutations can also tolerate higher sleep pressure (i.e., the need to sleep). “I think we have shown strong evidence that these short sleep mutations offer protection against
Alzheimer-like diseases,” said Fu. Given that short sleepers do not seem to develop many health problems, Fu believes these mutations may protect against many other diseases as well.
NEURAL CIRCUITRY AND CORTICAL ACTIVITY
Neural circuits that underlie wake, rapid eye movement (REM) sleep, and non-REM sleep are distributed across the brain, as opposed to circadian circuits where the central clock localizes to a discrete structure, the SCN, said Sehgal. Luis de Lecea, professor of psychiatry and behavioral sciences at Stanford University, has been using reverse genetic approaches, starting from the discovery of the preprohypocretin gene, to understand the neural circuitry of sleep–wake regulation. His approach involves identifying markers that define circuits involved in sleep regulation and then implementing a variety of methods to interrogate those circuits. For example, more than 20 years ago, the hypocretin/orexin system was identified as a key node in the regulation of sleep and wakefulness (de Lecea et al., 1998; Sakurai et al., 1998). Subsequent studies showed deficiency of hypocretin/orexin-expressing neurons in the lateral hypothalamus is associated with human narcolepsy (Peyron et al., 2000). “From these studies, we learned that this group of neurons are non-redundant and essential in providing sleep/wake stability,” said de Lecea.
De Lecea’s lab used optogenetics to show that activation of these neurons was sufficient to induce sleep-to-wake transitions. This combination of genetic identification of markers that define a circuit and the functional interrogation of the circuit led to an explosion in identification of circuits and markers that modulate sleep and wakefulness, said de Lecea. Sleep, he said, provides a framework for forward genetic approaches and the circuitry explains how elements are connected in many ways, enabling scientists to explore functional issues in sleep disorders. For example, the hypocretin system integrates signals from the circadian clock, metabolism, emotions, and stress. Understanding of this system has been exploited by the pharmaceutical industry to develop hypocretin receptor antagonists used to treat insomnia and other disorders, said de Lecea. Small molecule agonists are also in development to treat and possibly cure narcolepsy, he added.
De Lecea and colleagues have also used this theoretical framework to interrogate sleep disorders associated with aging. They identified a voltage-dependent potassium channel, modulation of which reduces the excitability of hypocretin neurons (Li et al., 2022). De Lecea suggested that these compounds may reduce aging-related sleep fragmentation.
Another potential sleep intervention strategy under development in de Lecea’s lab targets deep brain structures and neuronal circuits that
modulate sleep and wakefulness using focused ultrasound, which affects neuronal activity through the interaction of mechanical soundwaves with mechanosensitive channels in neurons. A device they designed allows them to identify the parameters that are optimal for neuromodulation using ultrasound. Using this device, they have shown that activation of brain circuits in the central medial thalamus results in long-lasting increases in slow wave activity, said de Lecea.
MODEL SYSTEMS
While electroencephalogram (EEG) recordings have been the primary tool for assessing sleep in humans, Raizen noted that other features can be used to identify a sleep state in animals where EEG is not readily done. These features include:
- Behavioral quiescence—the animal not moving or feeding
- Reduced responsiveness of the animal to the environment
- Ready reversibility of quiescence and reduced responsiveness to the normal state
- Homeostasis—the animal being driven to compensate for inadequate sleep
- Episodic nature of sleep under the control of an internal clock
Model systems enable study of the core functions of sleep, namely, those functions that have been evolutionarily conserved. “We think that evolution [of sleep] happened very early,” said Raizen, noting that sleep states are seen not only in worms and flies, but also in animals as diverse as jellyfish and hydra, which lack a central nervous system.
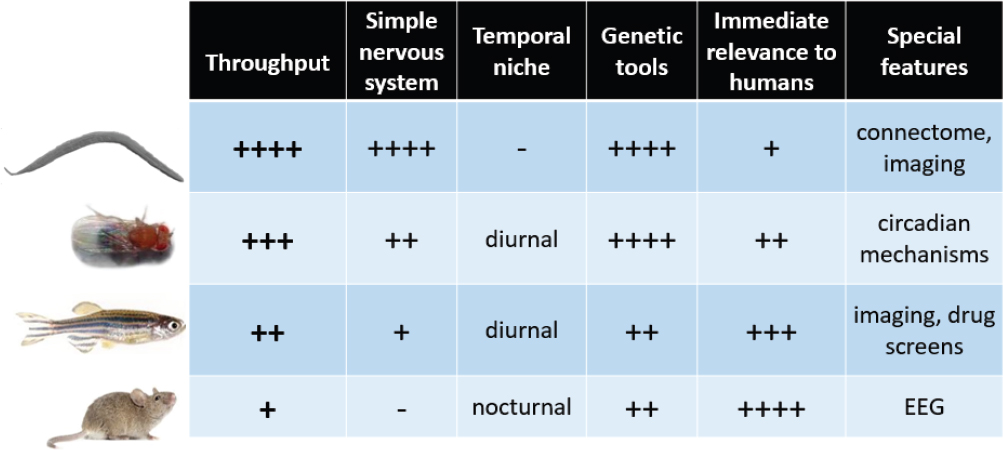
Worms, flies, zebrafish, and mice are all widely used as model systems to study sleep, said Raizen. Each has its relative merits and demerits, as outlined in Figure 3-1. For example, the very short life cycle, simple nervous system, and connectome in worms allows scientists to form hypotheses regarding circuits of sleep. Raizen said the fly has similar advantages, with the additional advantage of strong circadian rhythms. Fish are vertebrates, active during the day, and easily imaged. Of course, mouse models have immediate relevance to human physiology and are amenable to EEG, enabling study of the spectral properties and different types of sleep, said Raizen.
Sleep studies in worms and flies provide experimental power, high throughput, and simplicity, but lack immediate relevance to human sleep, said Raizen. They provide an intermediate step before being translatable to humans. For example, nearly a decade before the groundbreaking paper in 2016 where a forward genetic screen in mice was used to identify genes regulating sleep (Funato et al., 2016), scientists had shown that one of
NOTE: EEG = electroencephalogram
SOURCE: Presented by David Raizen on November 2, 2022; from Kryger et al., 2010.
these genes, Sik3, was clearly involved in the regulation of sleep in the worm Caenorhabditis elegans, said Raizen (van der Linden et al., 2008). Subsequent work, also in C. elegans, has identified the neurons in which this gene functions and has shown where it acts, its molecular target, and which neurons actually promote sleep (Grubbs et al., 2020).
Raizen and colleagues are now conducting forward genetic screens in worms to identify genes involved in sleep disorders that occur in conjunction with other diseases, such as chronic infectious diseases or chronic autoimmune disorders. “Many of the tools that have been developed to study sleep in humans don’t apply to these individuals, and their sleep doesn’t always follow the two-process model,” he said. They are using an approach called saturation mutagenesis, which potentially could lead to the identification of every gene regulating this process, said Raizen.
Although studies in fruit flies and other animal models have demonstrated that sleep is a fundamentally important process across species, Sehgal emphasized that sleep in these organisms is not necessarily the same as sleep in humans. Some, but not all, functions are likely to be conserved across species, and the functions of sleep may also change across the life span. “The hope is that by putting all these models together, we will have a [clearer] understanding of sleep,” she said.
This page intentionally left blank.







