Sharing Health Data: The Why, the Will, and the Way Forward (2022)
Chapter: 5 Case Study: COVID-19 Evidence Accelerator (EA)
5
CASE STUDY: COVID-19 EVIDENCE ACCELERATOR
Interviewees: Carla Rodriguez-Watson, PhD, MPH, Director of Research, Reagan-Udall Foundation; and Jeff Allen, PhD, Executive Director, Friends of Cancer Research
ABSTRACT
The COVID-19 Evidence Accelerator (EA) is a research collaborative comprised of 230 public and private entities spanning the health care ecosystem. The project was launched in April 2020 at the behest of the U.S. Food and Drug Administration (FDA) to understand the characteristics of the novel SARS CoV-2 virus and clinical progression of COVID-19, and to provide rigorously developed evidence regarding the diagnosis, treatment, and prevention of the disease. To that end, the EA provides a unique venue for participants to crowdsource real-world evidence (RWE), instead of data, which is the focus of many case studies in this publication; investigate a prioritized list of scientific questions; and compare results without researchers having to expend capital to procure and maintain external datasets. The added benefit of this approach is that researchers can exchange knowledge and ideas corroboratively without exposing themselves to privacy breaches or dealing with consent or control issues with data—concerns raised by the research community in the progenitor publication to this one. Given the urgency to attend to the rapidly evolving COVID-19 crisis, EA used existing information-sharing infrastructure, research protocols, and the prestige of its host organizations, the Reagan-Udall Foundation and Friends of Cancer Research, to quickly attain
critical mass and jumpstart its work. Work within the EA is guided by a code of conduct that emphasizes a commitment to scientific integrity, ruthless transparency, and respect for individual privacy. While the EA began with workstreams in COVID-19 therapeutics and diagnostics, it has recently expanded to COVID-19 vaccines and post-acute sequelae of SARS-CoV-2. It is also being leveraged to explore possibilities for the development of novel treatments in substance use disorder. Nonetheless, irrespective of use case, the EA’s approach to information sharing offers a model for how concerns about privacy risks and loss of competitive advantage that often stymie data sharing efforts can be addressed.
BACKGROUND
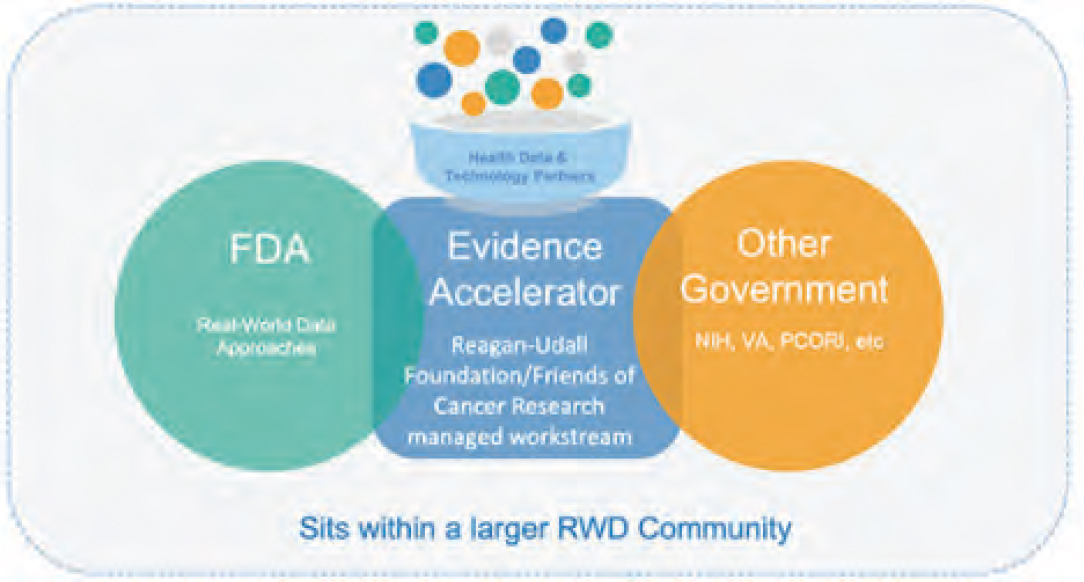
The COVID-19 Evidence Accelerator is a multi-stakeholder collaborative created at the request of the U.S. FDA in April 2020 to understand and address the rapidly developing COVID-19 pandemic through the sharing and leveraging of RWE (FDA, 2020). The EA is managed through a partnership between the Reagan-Udall Foundation (Foundation) and Friends of Cancer Research (Friends). Established by Congress as an independent 501(c)(3), the Foundation uses its neutral position to facilitate dialogue between the FDA and other public and private entities. Comparably, Friends is a nonprofit think tank that seeks to accelerate the discovery and development of new cancer treatments through public-private convenings and research partnerships.
At the time of writing, the EA consists of 230 participating organizations engaged in any or all of its three workstreams. The Diagnostic Evidence Accelerator workstream focuses on addressing diagnostic and serological questions, such as those related to the real-world performance of COVID-19 diagnostic tests. The Thera-
SOURCE: Roe, L., A. Abernethy, J. Franklin, E. Sigal, J. Allen, A. Bhat, C. Rodriguez-Watson, and S. Winckler. 2021. Accelerating Evidence Generation by Convening Diverse Stakeholders Across the Real-World Data Ecosystem. Available at: https://evidenceaccelerator.org/sites/default/files/2021-08/Ev%20Acc%20Lab%20Focus%20Science%20Poster%202021%20FInal%20with%20OCC%20review.pdf (accessed December 6, 2021).
peutic Evidence Accelerator workstream is devoted to expediting the identification of effective therapies for mitigating COVID-19 symptoms. The Vaccine Evidence Accelerator focuses on questions of vaccine performance. Representation in the collaborative spans the health care ecosystem: health care systems, national insurers, health care technology companies, pharma and biopharmaceuticals, laboratories, academics, and various branches of the federal government engage and share their expertise within the EA. Participating organizations range in size, have access to diverse sources of data, and serve diverse populations.
In addition, the EA operates within a larger national data community by interfacing with FDA-adjacent initiatives, such as the Sentinel Program, Biologics Safety and Effectiveness System, and the National Evaluation System for Health Technology; domestic activities, such as the Patient-Centered Outcomes Research Institute (PCORI); and interacts with international forums, such as the

SOURCE: Reagan-Udall Foundation and Friends of Cancer Research. 2021. COVID-19 Evidence Accelerator. Available at: https://evidenceaccelerator.org (accessed November 26, 2021).
Observational Health Data Science and Informatics program (see Figure 3).
At its inception, the EA was not externally financed, but in July 2020, the project secured support from a federal grant and a private foundation. As activities of the EA have expanded, the EA is seeking additional funding that will support additional staff to advance new projects.
DESCRIPTION
The EA convenes experts via Lab Meetings to rapidly share information related to real-world studies of COVID-19. During the Lab Meetings, participants share preliminary findings on research, data analytics, and methods relevant to addressing the COVID-19 pandemic in three discrete areas (see Figure 4). In addition, findings from parallel analyses (described below) are conveyed to this broad community. EA Lab Meetings provide a “safe collaborative
space” for key players (nearly 300 organizations, at the time of writing) across the health data ecosystem to assimilate and evaluate data generated from across the country. Given the importance of information sharing in the wake of the COVID-19 pandemic, meeting summaries are posted on the EA website (Reagan-Udall Foundation and Friends of Cancer Research, 2021).
The second main EA activity is gathering various data analytics experts to explore discrete research questions across a variety of real-world data sources (e.g., insurance claims, EHRs, registries). In the parallel analysis approach, analytic partners (Accelerators) contribute to the rapid development of master protocols to illustrate the use of various treatments or diagnostics, as well as characterize the natural history of certain components of COVID-19. Some of these protocols, particularly those around several potential COVID-19 therapeutics, have been reported on EA’s website and are hosted on a cloud-based file storage site (Reagan-Udall Foundation and Friends of Cancer Research, 2020). Master protocols enable parallel analyses of the same question across different data systems to quickly test reproducibility of results. Unlike most of the cases highlighted in this Special Publication, data exchange is not a feature of the EA. Hence, there is no need for a data coordinating center or data use agreements (DUAs) with research partners, although research partners who engage in data sharing external to the EA may employ DUAs. Each analytic partner applies the master protocol to their own data and performs their own analysis. Therefore, only aggregated results are shared among collaborators for knowledge generation. The benefits of this method are that it mitigates privacy risks and addresses concerns about the loss of competitive advantage that can arise from sharing identifiable data points.
To render results in an easily comparable format and reduce the burden of data handling, each master protocol consists of detailed analysis plans and uniform data tables, ensuring results are synthesized and presented uniformly. A key undertaking common to all three workstreams is to identify appropriate common data elements upfront, which contributes to the acceleration capability.
Outside of COVID-19, this approach was originally piloted to explore population characteristics, outcomes, and novel endpoints

SOURCE: Reagan-Udall Foundation and Friends of Cancer Research. 2021. COVID-19 Evidence Accelerator. Available at: https://evidenceaccelerator.org (accessed November 26, 2021).
for cancer treatments across multiple data sets. The EA has recently expanded to include post-acute sequelae of SARS–CoV-2 and an oncology working group, which are exploring the feasibility to evaluate potential differences in response to cancer treatments for patients who have previously had COVID-19. The EA is also being leveraged to explore possibilities for the development of novel treatments in substance use disorder.
An outcome of this two-fold work (convening experts and pursuing discrete regulatory questions) has been the development of principles (see Figure 5) to guide the EA’s work, which underscore the importance of producing results that are reliable, credible, and usable by regulators and the health care community.
EA’s ethos can be characterized as a “coalition of the willing.” There is no minimum time commitment or fee to participate in the collaborative, and organizations can enter and exit at will. While, on the periphery, health systems may have pre-existing DUAs with third-party analytics companies, collaborations within the EA are not governed by legal documents or a codified decision-making
process. Projects presented for IRB review under the EA have been determined to be exempt. In the absence of an enforcement mechanism, EA abides by the aforementioned set of principles that recognize the sense of urgency “to act fast” without sacrificing data privacy and scientific integrity. Another key operational tenet is embracing convergence and discordance to facilitate understanding about the nuances of the underlying datasets, such as how are the data gathered, the context in which they are gathered, and methods and perspectives of interpreting the data to the extent the data is harmonized.
While Friends and the Foundation guide and provide overall support for the project’s activities, leadership for identifying research questions and resolving issues come about organically. The EA invites collaborators’ input into decisions about which questions should be of priority and how and where resources should be allocated. Final decisions about priority scientific questions rest with the FDA.
The EA is unique in part because of the mutually reinforcing relationship between the value it presents to collaborators and the factors contributing to its success. Born out of the critical need to understand the rapidly evolving natural history of COVID-19, the EA offers a frictionless knowledge sharing environment in which collaborators can share results quickly without being encumbered by bureaucracy. The benefit is derived from leveraging the tools and cachet of the EA’s host organizations (Friends, Foundation, and the FDA). Given the urgency to rapidly construct a data sharing apparatus to keep pace with the quickly evolving nature of the pandemic, the project builds upon the data-sharing and analysis efforts of the oncology-related pilot studies conducted by Friends and numerous data partners. For example, the experience of Friends offered a model in how to formulate common research protocols and apply a parallel learning model. The EA’s data-sharing infrastructure borrows from the groundwork laid by the Foundation’s post-market safety surveillance work as well. EA leaders attribute these elements as key to the project’s success.
The prefabricated infrastructure of the EA coupled with the urgency of COVID-19 endows the effort with momentum by attracting a multitude of research partners who bring to bear their ca-
pabilities and expertise. It is noteworthy to acknowledge that the credibility of the FDA, Friends, and the Foundation created an added stimulus for organizations to join. EA organizers stated how the project quickly became a favorable venue for COVID-19 researchers, especially early in the pandemic (April–May 2020), which spurred others to join for fear of regret of missing an advantageous opportunity.
In turn, the cumulative strengths of the research partners have helped the collaborative develop stable research practices and rigorously generated evidence at a time when the quality and credibility of notable scientific studies of COVID-19 were being challenged. These aspects enhance the attractiveness of the EA, setting it apart as a low-cost, low-risk information sharing solution for entities concerned with the steep financial investment to obtain and maintain high-quality data, which has been noted as a deterrent to data sharing (Whicher et al., 2020).
FUTURE DIRECTIONS
In addition to capitalizing on the resources and efforts of others, EA leaders underscore the significance of a “just do it” mentality, which at times can serve as the best antidote to the inertia of the health care industry. While early attempts to launch the project included missteps, through an iterative process driven by a willingness to learn from others and a commitment to transparency, EA leaders were able to reach a workable steady state within five months. For example, project leaders learned that a step-wise approach to analysis allowed the EA to review data and push out preliminary results to a wider audience. One of the interviewees for this case study aptly observed, “There are decisions being made every day based on limited evidence, so if we can do something, anything, to help we will do it.” While the statement was made in reference to COVID-19 decision making, the kernel of this statement is at the heart of accelerating continuous learning in health and health care and guides EA’s operations to this day as it catalogues lessons learned and tackles new research questions.









