Biological Threats in the Age of Emerging Biotechnology: Proceedings of a Workshop Series (2026)
Chapter: 2 Biotechnology-Based Case Studies
2
Biotechnology-Based Case Studies
Planning Committee members developed three biotechnology-focused case studies, or scenarios, to understand the policy landscape, key products, and status of technology development that constituted the focus of each discussion. Espona outlined the goals for each scenario: (a) understand the governing frameworks that apply to that particular type of biotechnology research and data management and access; (b) understand the impacts of these frameworks on biotechnology development; (c) examine how the biotechnology governing frameworks enable responsible innovation; and (d) understand the ecosystem of biotechnology and data, and risks and hazards, outside of traditional dual-use1 life sciences research concepts.
Two scenarios examined omics data involving in silico modeling and bioinformatics approaches. The first focused on using multi-omic data to affect human health, and the second on using multi-omic data to affect crop improvement. The field of multi-omics has revolutionized researchers’ ability to understand and influence human health and agriculture. Integrating these datasets through in silico modeling and other bioinformatics approaches enables a deeper understanding of the molecular mechanisms underlying diseases while informing the development of personalized medicine,
___________________
1 According to U.S. Government Policy for Oversight of Dual Use Research of Concern and Pathogens with Enhanced Pandemic Potential published in May 2024, “dual use research” is research conducted for legitimate purposes that generates knowledge, information, technologies, and/or products that can be utilized for benevolent or harmful purposes. Additionally, “dual use research of concern (DURC)” is life sciences research that, based on current understanding, can be reasonably anticipated to provide knowledge, information, products, or technologies that could be misapplied to do harm with no, or only minor, modification to pose a significant threat with potential consequences to public health and safety, agricultural crops and other plants, animals, the environment, materiel, or national security.
and has the potential to revolutionize agriculture by enhancing crop improvement strategies.
The third scenario focused on DNA synthesis technologies. Foundational DNA synthesis technologies included solid-phase synthesis, which used phosphonamidite chemistry to add individual bases one at a time, or enzymes, which synthesized short fragments of DNA. However, more recent innovations have led to faster, cheaper, and more accurate assembly of large pieces of DNA. Companies are developing thermal methods that use temperature to control DNA synthesis; double-stranded oligonucleotide pools to create libraries of larger DNA fragments; microarrays, including silicon chips that can synthesize and elongate tens of thousands of DNA fragments; and amplification techniques that use precisely designed templates and primers. As these and other techniques are developed, companies are seeking to scale up production – e.g., by using microarray chips to synthesize numerous DNA strands simultaneously.
Despite their promise, these emerging biotechnologies raise data security, ethical, legal, regulatory, and social concerns because of their dual-use potential and the risks associated with data misuse. Some of the issues raised in discussing each scenario included: human health multi-omics and privacy issues; crop improvement multi-omics, biodiversity, and food security issues; and DNA synthesis biosecurity and data security concerns. More scenario details are available in Appendix B: Committee Developed Scenarios.
The planning committee believed a scenario-based approach would highlight the complex risk and benefit relationships surrounding each biotechnology. The planning committee prepared a series of questions designed to examine the cultural and economic factors at play in development of policies and laws in each country, to elucidate trends in biotechnology research and development, and to facilitate discussion of biotechnology-specific risks and benefits. With this standard set of questions, the planning committee anticipated exploring both general and biotechnology-specific considerations while discussing defined steps for assessing and mitigating exploitation of biotechnology-related research and development activities. These risk and benefit questions are listed in Appendix B. Summaries of the three scenarios are below, and the discussions resulting from the scenarios are summarized in Chapters 3 and 4.
HUMAN HEALTH MULTI-OMICS2
The discussion of human health multi-omics was conducted with experts from India. Participants discussed opportunities and challenges facing human health multi-omics research in India through two facilitated discussions, the first on frameworks and guidance and the second on risk and benefit questions. Multi-omics is revolutionizing researchers’ ability to understand and influence human health outcomes, but it also carries significant ethical, legal, and social implications because of its dual-use potential and the risks associated with data security, privacy, and misuse, said Espona. She emphasized the importance of considering the risks of this technology without losing sight of its benefits. Berger asked participants to critically evaluate the existing risk and benefit frameworks and their applicability to advances in multi-omics for human health, and to consider what information is needed to accurately assess the risks and benefits of this research and to reap the benefits while reducing the risks. To start the discussion, National Academies staff reviewed some examples of multi-omics tools and databases in India.
Genome Projects in India
Genome India is a collaboration of 20 academic and research institutions building a comprehensive catalogue of genetic variations that reflects the unique diversity of the Indian population, with the goal of driving a genomics-based health revolution (Department of Biotechnology, n.d.). Genome India established a robust biobank consisting of 20,000 samples from 83 diverse populations, and whole-genome sequencing was completed for 10,000 of those samples.
Another program, IndiGen, aims to sequence the complete genomes of thousands of individuals representing diverse ethnic groups in India to enable genetic epidemiology (IndiGen, n.d.). Intended uses of the population genome data include developing public health technologies and making carrier screening more affordable.
The IndiGenomes (n.d.) resource is based on genomic data from over 1,000 genomes sequenced across India through IndiGen, representing diverse geographies and ethnicities. IndiGenomes provides access to over 55 million genetic variants comprising single nucleotide variants and indels.3 The Indian Biological Data Center (IBDC) manages data portals for the government of India (Regional Centre for Biotechnology, n.d.). Examples of omics portals for depositing and accessing data include a Nucleotide Data Archive (open and controlled access) (IBDC, n.d.-a), a Structural Data Archive (IBDC,
___________________
2 This section is based on the presentation of the Planning Committee and National Academies staff.
3 Abbreviation for insertion or deletion. Refers to the random removal or addition of nucleotides from a DNA sequence. Source: https://innovativegenomics.org/glossary/indel/.
n.d.-b), a Metabolome Data Archive (IBDC, n.d.-c), a Proteome Databank (IBDC, n.d.-d), and Biological Images Archive (IBDC, n.d.-e). Although it is not human health multi-omic data, IBDC also manages a Crop Phenome Database.
Additional databases and portals are managed by nonprofit groups, such as the Indian Cancer Genome Atlas (ICGA) Foundation (ICGA, n.d.). An updated version of the ICGA Data Policy (2025) recently was released. The policy “ensures that ICGA’s collection, processing, storage, and sharing of data aligns with the laws of India, including the Digital Personal Data Protection (DPDP) Act, 2023, and international best practices such as the FAIR principles.”
Participants engaged in two facilitated discussions. The first discussion asked framework and policy development questions: What frameworks exist to evaluate multiomics analyses for human health studies? Have these frameworks been developed for one or more types of data? What are the main drivers of policy and regulation development? Are the frameworks or policies driven by laws and regulations? If legal and regulatory frameworks do not exist, why have they not been developed? The second discussion asked participants to consider risk and benefit questions (see Appendix B).
MULTI-OMIC ANALYSIS FOR CROP IMPROVEMENT
The discussion of crop improvement multi-omics was conducted with experts from Mexico, Peru, and Argentina. Through two presentations and a lively conversation, participants discussed opportunities and challenges facing multi-omics research for crop improvement across Latin America, ranging from technical details to larger issues that play out on institutional and societal levels with this biotechnology. Luis Herrera-Estrella, Texas Tech University, discussed his work using advanced molecular techniques to study crop plants, first in his native Mexico and more recently in the United States. He described research aimed at crop improvement in chia plants, new techniques that can vastly speed up the improvement of chia and other food crops, and the challenges to implementing these techniques in Mexico. Lena Gálvez Ranilla, Universidad Catolica de Santa Maria, discussed her research on metabolomics, with an emphasis on characterizing the diversity and health-relevant properties of Andean grains.
The Case of Chia (Salvia hispanica)4
Multi-omics typically combines genome sequencing and transcriptomics with other omics, such as the proteome, metabolome, and epigenome of an organism, said Herrera-Estrella. In plants, ionomics (analysis of the ion composition) is useful for understanding the plant’s physiology and its contribution to human nutrition. Phenomics employs imaging systems to perform high-throughput phenotyping of plants, providing information about plant growth, morphology, and chemical composition in response to different environments over time.
These omics data are vital for advancing crop improvement in general, said Herrera-Estrella, and they are particularly useful for orphan crops (crops that have regional importance but are not traded internationally), many of which have not been fully domesticated. Crop omics analysis can enable breeding studies aimed at improving yield, stress tolerance, and nutritional content. Combining omics analyses with other advanced technologies opens the door to even more powerful opportunities, he added. For example, by integrating omics with machine learning tools and gene editing, researchers can discover which omics data are associated with which phenomic traits, such as drought tolerance; identify the regulatory networks that determine these traits; and make precise modifications to the genome for crop improvement.
Multi-Omics Studies Reveal Promising Pathways for Enhancement of Chia Crops
Herrera-Estrella and colleagues have generated an improved chia genome chromosome-level reference (Alejo-Jacuinde et al., 2023). Using metabolomics and transcriptomics, they identified the genes involved in the biosynthesis of rosmarinic acid. They also studied the genes involved in synthesizing mucilage and, by applying machine learning strategies, identified the transcription factors that regulate mucilage production.
Once major transcription factors that regulate a pathway of interest have been identified, they can be targeted for mutational studies aimed at crop improvement, said Herrera-Estrella. This strategy has been used to re-generate modern, domesticated tomato from wild tomato, “reproduc[ing] what took centuries for our ancestors in Mesoamerica” in just a few years, he said. Chia has multiple potential targets for crop improvement. For example, chia could be grown at wider latitudes by reducing photo-sensitivity and improving cold tolerance, as its growth is currently limited to regions with long days. Better drought tolerance, larger seeds, improved nitrogen and phosphorus use efficiency (to reduce the need for fertilizer), and resistance to fungal pathogens are more ideas for chia crop improvement.
___________________
4 This section is based on the presentation of Dr. Luis Herrera-Estrella, Director of Institute of Genomics for Crop Abiotic Stress Tolerance and Full Professor of the Department of Plant and Soil Science at Texas Tech University.
Engineering Mutations for Crop Improvement: Gene Editing can Dramatically Improve Speed and Precision
Herrera-Estrella highlighted how gene editing enables the generation of precise, targeted genetic changes in crops and compared it to the older mutation breeding method (Figure 2-1).
Mutation breeding uses physical radiation or chemical mutagens to generate multiple random mutations, in an accelerated version of natural mutagenesis, followed by selection to isolate a mutant with the desired trait. This century-old method generated widely consumed products, including pink grapefruit, seedless oranges, and varieties of barley, chrysanthemum, rice, soybean, maize, and wheat. The mutagenic process generates hundreds to thousands of off-target mutations, but “to date, nobody cares… the only thing is that the crop looks normal, that it has a high yield, and it has a desirable trait,” he said.
Mutation breeding creates DNA lesions randomly throughout the genome, whereas gene editing uses an RNA guide to target a precise location (Li, Sun, and Gao, 2024). Gene editing generates precise, site-directed mutations with few off-target mutations, making it relatively innocuous compared to mutation breeding, said Herrera-Estrella. Furthermore, gene editing is versatile. In principle, it can be applied to any plant species and target any gene to produce any type of crop improvement trait. Available gene-edited crop varieties include apples and potatoes with reduced polyphenol oxidase production (reducing browning and lengthening shelf life), he said, while gene-edited
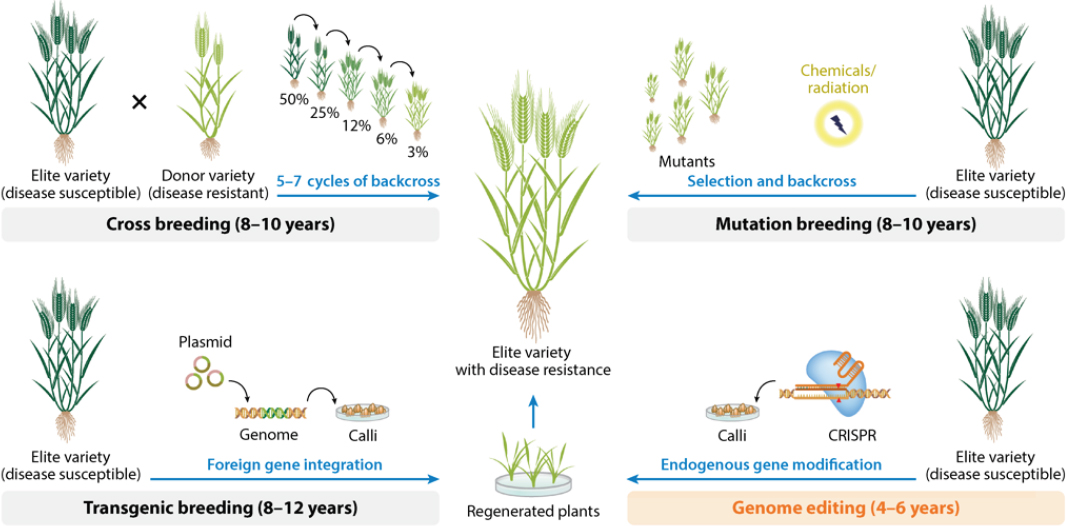
SOURCE: Presented by Dr. Luis Herrera-Estrella on September 17, 2025. Used with permission of Annual Reviews, Inc., from Chen et al. 2019. © 2019 by Annual Reviews, http://www.annualreviews.org; permission conveyed through Copyright Clearance Center, Inc.
berries are soon to be commercialized (FDA, 2024). Mexico is the main producer of avocados in the world, and browning of avocados reduces their shelf life. The genes responsible for browning are known, said Herrera-Estrella, and gene editing methods could be used to generate avocados that do not brown or that have smaller pits.
Surmounting the Technical Limitations to Gene Editing in Orphan Crops
Technical bottlenecks impede gene editing of chia, said Herrera-Estrella. Typically, gene editing is done in cells in tissue culture and then the plant has to be regenerated and produce viable seeds (Altpeter et al., 2016). The methods for accomplishing this are crop-specific, and chia (like most orphan crops) lacks a well-developed tissue culture system. Even for a well-studied crop like maize, some genotypes have established systems for tissue culture and regeneration, while other genotypes do not. Because the system is not easily transferable from one crop to another (even when they are closely related), these tools need to be developed for each individual crop.
Herrera-Estrella and colleagues are employing a new method of gene editing that creates a targeted mutation directly within the cells of a plant growing in soil, circumventing the need for tissue culture (Maher et al., 2020). Herrera-Estrella collaborates with Gunvant Patil, who developed a wound-inducible system for activating the expression of specific transgenes promoting new shoot generation when a plant is wounded. An agrobacterium delivers the CRISPR-Cas system and guide RNAs into cells in the wound site, generating new shoots that are gene-edited but not transgenic.
Advances on the Research of Peruvian Andean Maize Diversity Using Metabolomic Platforms5
Maize is an important food for Peru, with over 50 genetically diverse strains, which may be distinguished by their pigmentation, ecological adaptations, and cultural aspects associated with their use or preparation, said Gálvez Ranilla. Germplasm banks have 3,931 entries aimed at ex situ6 conservation of Peruvian maize (MINAM, 2018), but gaps exist in both in situ and ex situ preservation. Though many samples are “definitely not the same as thousands of years ago,” she said, some native strains still exist in her locality of Arequipa in the southern Andes, although the scientific information about these strains is inadequate.
Research on Peruvian Maize Diversity: Cabanita Andean Maize
To better understand the diversity of Peruvian maize, Gálvez Ranilla and colleagues focused on Cabanita, a traditional Peruvian indigenous maize that has been ground since
___________________
5 This section is based on the presentation of Dr. Lena Gálvez Ranilla, Principal Investigator, Universidad Catolica de Santa Maria, Peru.
6 Ex situ definition: Outside, offsite, or away from the natural location; in this case, seed banks.
pre-Inca times and still provides essential income for many indigenous communities. Researchers collected 58 Cabanita accessions from different provinces within the Arequipa region and analyzed their carotenoid profiles (Fuentes-Cardenas et al., 2022).
Cabanita samples have varied pigmentations, broadly divisible into red, white, and orange maize categories, with pigmentation only covering the top half of the kernel. To characterize Cabanita maize metabolic profiles, the researchers studied representative white, red, and orange types at the kernel maturational stages of milky, dough, and mature (Figure 2-2) (Ranilla et al., 2023). Plants were cultivated under controlled conditions and antioxidants (phenolic compounds and carotenoids) were analyzed by targeted methods, while primary metabolites (e.g., sugars, organic acids, amino acids) were analyzed by gas chromatography–mass spectrometry. In vitro functional tests were used to further examine each sample’s antioxidant potential and anti-hyperglycemic potential.
All Cabanita maize types produced phenolic compounds, the levels of which tended to decrease with kernel maturity, said Gálvez Ranilla. Anthocyanins were detected only in the red type and luteolin derivatives only in the orange type. Carotenoids were highest in the orange type and decreased with maturity, as did total in vitro antioxidant capacity. Distinct patterns of primary metabolite production emerged among the three maize types, with the highest overall concentration of primary metabolites in the white type. In general, levels of primary metabolites tended to be highest in the earliest stage and decreased with maturation.
The two measures of anti-hyperglycemic potential differed in their distribution: a -amylase inhibitory activity was highest in the white type and decreased dramatically

NOTE: White, Red, and Orange maize pigmentations at different stages of maturation (from left to right: level S1 milky, S2 doughy, and S3 mature).
SOURCE: Presented by Dr. Lena Gálvez Ranilla on September 17, 2025. From Ranilla, et al., 2023. License: CC BY 4.0. http://creativecommons.org/licenses/by/4.0/
with maturity, while α-glucosidase inhibitory activity was not dependent on maize type, though it decreased over the course of kernel maturation. These results are from in vitro preparations of kernels in their raw state, cautioned Gálvez Ranilla, and the tests need to be repeated following traditional preparation of the kernels for consumption. She noted, however, that Cabanita maize is traditionally consumed at the physiologically mature stage, while the levels of metabolites important for health tend to be higher at earlier stages.
Both Herrera-Estrella and Gálvez Ranilla discussed the benefits of their research, and this is summarized in Chapter 4: Challenges and Opportunities.
DNA SYNTHESIS
The discussion of DNA synthesis was conducted with experts from India, Mexico, Peru, and Argentina. Through two presentations, participants discussed opportunities and challenges facing DNA synthesis screening efforts internationally and across the four project countries involved in this workshop series. Becky Mackelprang, Engineering Biology Research Consortium (EBRC), reviewed EBRC’s ongoing workshops on International Best Practices for Nucleic Acid Synthesis Screening;7 and Mayra Ameneiros, Tessa Alexanian, Ricardo Chavez, and Suryesh Namdeo from International Biosecurity and Biosafety Initiative for Science (IBBIS) presented on safeguarding modern bioscience and biotechnology for DNA synthesis.
Noting how the crop multi-omics discussion illuminated ways in which over-regulation can impede the progress of science, Espona reiterated that “it’s not all about risks and hazards,” and the goal should be to balance risks against the benefits of these new technologies. Several years ago, the ability to synthesize long strands of DNA seemed like science fiction, but in recent years this capability has become exponentially faster and cheaper, said Espona. This technology offers many potential benefits, but it also presents significant ethical, legal, and social concerns because of its dual-use potential and risks related to data security, privacy, and misuse. Balancing these risks with benefits will require better communication among the stakeholders, she said, to enhance mutual understanding and design better regulations that benefit society.
___________________
7 Description, goals, and additional project information here: https://ebrc.org/focus-areas-security-buildinginternational-best-practices-for-robust-synthetic-nucleic-acid-screening/ (accessed on December 8, 2025).
Examples of International Best Practices for Nucleic Acid Synthesis Screening
The EBRC is a community focused on advancing biotechnology “to support human and planet well-being,” said Mackelprang, and its interest in security stems from the understanding that misuse of this technology will lead to “terrible pushback” on its positive uses. Nucleic acid synthesis screening is critical for securing the bioeconomy, she said, and relatively small measures can have a big impact, such as ensuring that companies screen the DNA they synthesize. Implementing universal screening may be necessary to be truly effective as a safeguard against misuse. To facilitate this, she said, EBRC is developing international best practices for nucleic acid synthesis screening.8
Mackelprang shared that EBRC has two major objectives for this project: (a) develop an internationally relevant set of best practices that works within the contexts of different countries; and (b) establish an informal global network of stakeholders who can adapt these practices in response to changes in the risk landscape. EBRC and the National Institute of Standards and Technology began the groundwork for the project in 2024, engaging stakeholders to develop the Office of Science and Technology Policy (OSTP) Framework for Nucleic Acid Synthesis Screening (OSTP, 2024).
Mackelprang cautioned that screening is complicated. Companies are responsible for determining whether the customer is a legitimate member of the research community, if the sequence is from a pathogen or toxin of concern, and if so, if the customer has a legitimate need for that sequence. If the provider is not certain, they can request additional information from the customer–though what information to request is unclear, and whether to trust the customer is a judgement call. To address these nuanced challenges, EBRC is interviewing stakeholders around the world, conducting workshops, and drawing on local expertise. Its goal is to modify the United States’ best practices, specifically to make them internationally relevant and adaptable, she said. Mackelprang presented findings from workshops and interviews with over 40 stakeholders worldwide.
Mackelprang also explained EBRC’s assessment for how advanced, regional bioeconomies can address local and global challenges across sectors. Figure 23 shows bioeconomy applications of the Design, Build, Test, Learn (DBLT) Cycle that EBRC uses to guide those efforts.
Technical & Operational Realities
Many small providers of synthetic nucleic acids “are unaware that this [biosecurity] is even an issue,” said Mackelprang, and any policy–particularly one that includes enforcement–must include screening standards or best practices, “otherwise it’s just impossible for them to do it correctly.” Many providers lack the tools and training
___________________
8 Description, goals, and additional project information here: https://ebrc.org/focus-areas-security-buildinginternational-best-practices-for-robust-synthetic-nucleic-acid-screening/ (accessed on October 24, 2025).
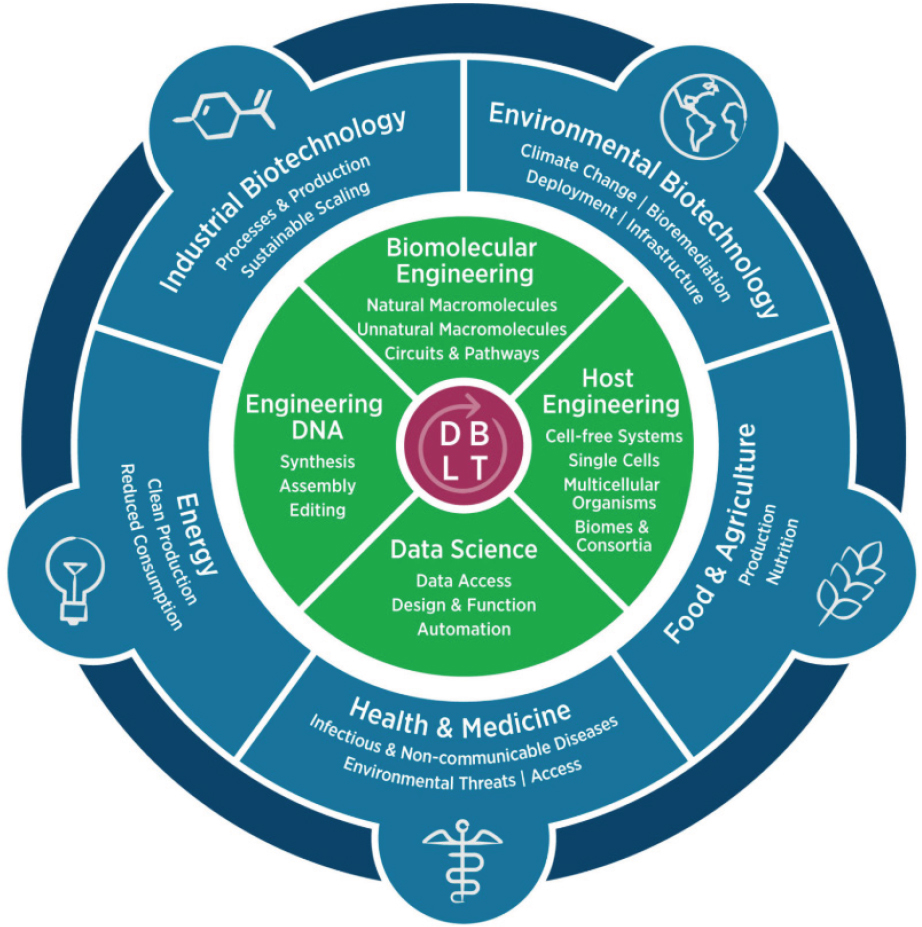
NOTE: The Design, Build, Test, Learn Cycle supports bioeconomy advancement through innovation in key technical themes applied to application sectors.
SOURCE: Presented by Dr. Becky Mackelprang on September 18, 2025; used with permission from Engineering Biology Research Consortium, © 2019.
needed to implement screening, she added, emphasizing the importance of free tools produced by IBBIS, SecureDNA,9 and others, in addition to commercially available tools. Providers may benefit from being trained in the proper use of these tools. Screening can be expensive, she said, and the involvement of distributors and resellers complicates the responsibility for screening.
Another challenge, said Mackelprang, is follow-up screening for flagged orders: how does a provider develop the necessary level of trust to fill an order for a sequence
___________________
9 See https://securedna.org (accessed on October 24, 2025).
of concern? She illustrated this challenge by mapping sequences and customers in two dimensions, according to various levels of customer concern and sequence concern (Figure 2-4). Sequence concern ranges from benign molecular tools to the most potentially hazardous toxins or virulence factors, while customer concern ranges from molecular biologists working at established research institutions to individuals receiving materials at a residential address or post office box. A provider has to think about both the sequence and the customer before deciding whether to fill the order. Areas in green are unlikely to require any follow-up, she said, while those in red clearly do. The tricky cases are the ones in the orange middle ground. How to handle these cases is unclear to providers, she said, and conducting these assessments becomes particularly difficult “when
NOTE: The horizontal axis represents concerns for potential sequences one could order from the most benign (green, left) to the most potentially harmful and hazardous (red, right). For the vertical axis, the severity ranges from least (top) to most (bottom) cause for concern from a potential customer, for example, research, training, purpose for ordering specific sequences, access to facilities. Included in the matrix are a few examples, though there are almost an infinite number of sequences and an infinite number of customer characteristics to be considered in tandem. For example, EBRC data suggest that providers find it unclear to decipher next steps when considering examples in the middle ground, such as a DIY researcher requesting a potential sequence of concern from an unregulated agent or a start-up customer requesting an obfuscated sequence of concern. Abbreviations: U.S. Centers for Disease Control and Prevention (CDC); Green fluorescent protein (GFP); virulence factor (VF).
SOURCE: Presented by Becky Mackelprang on September 18, 2025; created from unpublished data in Engineering Biology Research Consortium interviews, 2025. Project page: https://ebrc.org/focus-areas-security-building-international-best-practices-for-robust-synthetic-nucleic-acid-screening/.
you have almost an infinite number of sequences and an infinite number of customer characteristics.”
Mackelprang highlighted operational realities, which include different perceptions of risk that factor into the assessments conducted by different vendors. AI-enabled bio-design tools further complicate screening, she said, because they offer the potential to alter a sequence of concern in a way that could evade detection while retaining the same function. She cited a recent finding that AI tools could generate protein structures with dangerous functions that might not be caught by screening, which complicates efforts to develop best practices that can stand the test of time (Wittman et al., 2024).
Policy & Regulation
For genetically modified organisms, dangerous pathogens, and toxins, most countries have strong biosafety policies, said Mackelprang, but these policies do not regulate nucleic acids. Many countries lack domestic synthesis capacity and rely on imports, she added, and this raises the question of how to impose a requirement for screening. “Do you say, any provider operating in our country has to demonstrate that they screen? That any researcher ordering nucleic acids has to purchase them from folks that screen?” Different policy approaches might be preferable, depending on a country’s capacity.
Cultural & Sociological Considerations
Governments have difficulty identifying the most appropriate agency to take the lead for regulating synthetic DNA, said Mackelprang. This issue could belong to the bioeconomy, responsible research and innovation, security, or biodefense, depending on local culture and sociology. The perception of threat also varies between countries and regions, with some more concerned about insider threats and “lone wolf attacks” (like a disgruntled scientist), while others might be more concerned about cults or ideological groups, organized crime, or nation states. Different threat models can lead to different strategies for implementing screening, she added.
International Alignment
“This has to be an international conversation,” said Mackelprang, noting that current policies and practices around nucleic acid synthesis screening have been shaped by actors from the International Gene Synthesis Consortium (IGSC), the United States, and Europe, and these “don’t always fit neatly in other contexts.” Furthermore, given the challenge that providers face when different countries or regions operate under different policies, she emphasized the need for stakeholders to address this issue together, on an international level.
EBRC Best Practices for Nucleic Acid Synthesis Screening
The international best practices for nucleic acid synthesis screening that were identified by EBRC fell into six categories, said Mackelprang, as shown in Box 2-1.
Although creating best practices is crucial, this step is “only the beginning,” said Mackelprang. Building a robust, global network of stakeholders who communicate about the challenges that arise over time is essential in order for these practices to be useful, she said. The EBRC best practices will be translated into multiple languages and will undergo continuous updating. She encouraged participants to reach out to EBRC and participate in its workshops.
BOX 2-1 EBRC Best Practices for Nucleic Acid Synthesis Screening
Dr. Becky Mackelprang, Engineering Biology Research Consortium (EBRC), presented the following best practices for nucleic acid synthesis screening. The six best practice areas included policy, sequence screening, customer screening, screening assessment, law enforcement reporting, and benchtop synthesis.
- Policy: Countries with nucleic acid synthesis capabilities should implement policies that encourage or require screening. These policies need to be clear, detailed, and implementable. The role of import/export policies should be clarified, and these policies should not be a barrier to doing research.
- Sequence screening: Providers should screen across all six reading frames* of a sequence to identify sequences of concern. Screening should be robust to obfuscation by AI.
- Customer screening: Providers should screen customers against “denied parties” lists and conduct basic customer identity verification. Customers can facilitate screening by disclosing that their order contains a sequence of concern.
- Screening assessment: Screening processes should be assessed. Assessments can serve as a motivators to encourage companies to screen and as a tool to identify areas of challenge that companies are facing.
- Law enforcement reporting: Providers should have a plan to report suspicious orders to the appropriate law enforcement agency. This requires “having somebody in law enforcement who can understand these issues and not…overreact,” she said. The enforcement body’s responsibility for reporting international orders needs to identified in the country’s policy.
- Benchtop equipment: Manufacturers of benchtop nucleic acid synthesizers should screen customers for a legitimate need. Customers need to ensure that access to the machine is controlled, and the machines themselves should be able to screen sequences before producing them. Manufacturers might require use of proprietary reagents to block unidentified users from gaining access.
*The six possible open (protein-coding) reading frames (ORFs) of a double-stranded nucleic acid sequence (definition by Nelson, C. W., Z. Ardern, and X. Wei. 2019. “OLGenie: Estimating Natural Selection to Predict Functional Overlapping Genes.” bioRxiv: 2019.12.14.876607. https://doi.org/10.1101/2019.12.14.876607. Available under a CC-BY-NC-ND 4.0 International license. http://creativecommons.org/licenses/by-nc-nd/4.0/)
SOURCE: Presented by Becky Mackelprang, Engineering Biology Research Consortium (EBRC), on September 18, 2025.
Safeguarding Modern Bioscience and Biotechnology for DNA Synthesis
The IBBIS10 team’s presentation explored the framework landscape governing DNA synthesis in the participating countries. Alexanian surveyed the potential ways in which synthetic biology could be used to cause harm. Together with Ameneiros, she reviewed strategies that can prevent people from doing harm with biotechnology. IBBIS is an independent non-profit foundation whose overall mission is “safeguarding modern bioscience and biotechnology so it can advance and flourish safely and responsibly,” said Alexanian, and it aims to: (a) increase the share of DNA synthesis orders for which sequences and customers are screened; (b) map DNA synthesis providers, their screening practices, and associated national laws and regulations; and (c) support international standards that are inclusive and rigorous. These standards serve the entire world rather than just a small set of countries, she added, so that bioscience and biotechnology may flourish “for everyone, everywhere in the world.”11
Alexanian noted that synthetic DNA is central to modern biotechnology, figuring prominently in CRISPR-mediated gene editing, mutation library generation, polymerase chain reaction (PCR) diagnostics, and vaccine development. In manufacturing, industry, agriculture, and industrial biotechnology, scientists “order synthetic DNA constantly,” she said. As biology becomes easier to engineer and its promise is unlocked, harm-prevention strategies will become even more crucial, she added.
Synthetic nucleic acids have become exponentially cheaper over the last two decades, enabling the production of nucleotide sequences that are longer and more complex, said Alexanian. Prices fell 100-fold (from roughly ten dollars to five cents per base pair) between 2000 and 2020 (Carlson, 2025; Potomac Institute for Policy Studies, 2018).12 Researchers can purchase expensive benchtop devices that will print a DNA sequence in the lab in a matter of hours. The ability to order large DNA sequences cheaply increases the potential for non-state actors to order dangerous DNA (Piper, 2020; Randerson, 2006); however, DNA is not dangerous on its own, noted Alexanian. Intentional misuse can happen—for example, by reconstructing a harmful virus through stitching together multiple synthetic DNA sequences (Kupferschmidt, 2017), which can be done using published protocols (Thi Nhu Thao et al., 2020); or by engineering existing pathogens to become more dangerous, as has been done for both avian influenza (Davis et al., 2014) and mousepox (Lewis et al., 2019). The chief aim of synthesis screening, she said, is to prevent well-resourced non-state actors and domestic terrorists from accessing the necessary materials. Although the possibility of obtaining mail-order smallpox DNA has been a concern for two decades, the conversation about DNA synthesis
___________________
10 See https://ibbis.bio (accessed on October 23, 2025).
11 See https://ibbis.bio/our-work/international-screening-standards/ (accessed on October 24, 2025).
12 From the September 18, 2025 presentation by Tessa Alexanian.
screening is gaining steam in response to a changing risk landscape (ISO, 2019; 2024), added Alexanian. Recent years have seen the introduction of AI tools for genetic design, the availability of long synthesis and benchtop synthesis, the spread of biofoundries and biomanufacturing, and the growth of the global bioeconomy, all of which are driving additional interest in safeguarding DNA synthesis (NASEM, 2025; Responsible AI x Biodesign, 2024), she said.
DNA Synthesis Screening for the Private Sector: Notes from the World Economic Forum
The private sector is leading in innovation and resources devoted to biotechnology but is often left out of the conversation when it comes to international conventions or global biosecurity efforts, said Ameneiros. DNA is seen as a key risk area by global organizations, including the World Economic Forum (WEF). The WEF Global Risks Report (2025) evaluates the potential for low-probability and high-impact risk over the next decade. This report devotes a section to “Losing Control of Biotech.” Advances in AI, combined with other emerging technologies like DNA-based computing, raise new risks regarding the harmful potential of DNA synthesis. The WEF report emphasizes the need for standardized DNA synthesis screening methods, while industry efforts are underway to build “clean libraries” (collections of DNA constructs that have been rigorously screened and certified as safe) (Park and Turczyk, 2024). Several tools are available for identifying high-risk DNA sequences. Biotechnology’s potential for huge benefits alongside the risks makes screening particularly important, said Ameneiros, noting that “these approaches show that responsible innovation and security can advance together, making DNA synthesis both safer and sustainable at the same time.”
DNA Synthesis and Customer Screening: Balancing Access and Security
DNA synthesis screening is a combination of sequence screening and customer screening, said Alexanian. In the last two years, many new software tools and biosecurity standards specifically geared to screening DNA sequences for potential risks have advanced the field considerably. Over 50 countries regulate the export of genomes from certain listed pathogens, she said, so automated sequence screening is necessary to comply with these export controls. Sequence analysis can also be embedded in machines that perform benchtop synthesis. For a producer of custom DNA, “We don’t want to exhaustively screen every single order,” she said; rather, they should have a system capable of flagging potentially risky sequences, like pathogen genomes and virulence factors, while allowing non-risky sequences to pass. Routine lab work, such as developing vaccines from controlled viruses or anesthetics from toxins, involves dual-use sequences, noted Alexanian, and the mere fact that a sequence is dual-use “is not an indication of their intention.” Therefore, dual-use checklists could be combined with customer screening,
she said. She suggested the Netherland Biosecurity Office’s Dual-Use Quickscan13 and checklists produced by IBBIS14 and other providers.15
For customer screening, some researchers need to access risky DNA, Alexanian said. A construct based on a legitimacy screen that collects information such as affiliation, shipping address (institutional vs. residential) may address the question “does the customer’s work require access to sequences of concern?” For customer screening, Alexanian noted that “it’s very difficult to determine intent.” She suggested using decision support tools for customer screening,16 specifically for assessing customer provided information and building trust with a customer. Sequence screening and customer screening could be fine-tuned “to get that balance of access and security,” she said.
Closing the Information Gap: Tracking Worldwide Suppliers of Synthetic DNA and Their Screening Practices
One of the most important questions regarding DNA synthesis, said Ameneiros, is “can a non-state actor potentially order dangerous DNA?” To answer this, it may be necessary to know which companies sell synthetic DNA and what their screening practices are. Efforts to identify DNA suppliers have tended to focus on particular regions, producing data that is “often fragmented, incomplete, inconsistent, and sometimes it’s not accurate at all,” she said. “The reality is that the production and the transfer of synthetic DNA are poorly understood at the global level.”
To help guide policy and practice, IBBIS developed a Global DNA Synthesis Screening Map,17 said Ameneiros. She noted IBBIS’ efforts to balance transparency with safety—for example, by identifying areas where screening could be improved without revealing that a particular company does not screen. The good news, she said, is that a worldwide biosecurity ecosystem is starting to emerge, with new national and regional policies and regulations. Commercial and non-profit entities around the world are developing sequence screening tools, and international workshops are underway to define standards and shape future policies. At the same time, she added, a lot of room for improvement exists when it comes to legal frameworks.
Scientists are programmed to see the benefits of research, said Ameneiros, but to reap these benefits, safeguarding biotechnology from its inherent risks is important. The scientific community has a role to play, she said, “to help providers, policymakers, industry, and scientists to build the trust, transparency, and safeguards needed to ensure that synthetic DNA remains a tool for progress, and not a path to risk.”
___________________
13 See https://www.biosecuritycentral.org/resource/training-materials/dual-use-quickscan/ (accessed on October 24, 2025).
14 See https://ibbis.bio/our-work/common-mechanism/get-started/ (accessed on October 24, 2025).
15 See https://genesynthesisscreening.centerforhealthsecurity.org/for-providers-benchtop-manufacturers/list-of-companies-and-available-tools-to-assist-in-screening-orders (accessed on October 24 2025).
16 See https://ibbis.bio/our-work/customer-screening/ (accessed on October 24, 2025).
17 See https://ibbis.bio/our-work/global-dna-synthesis-map/ (accessed on December 18, 2025).
Members of the IBBIS team presented their findings on DNA synthesis policy and practice in Argentina, Mexico, Peru, and India. In each country, said Ameneiros, the IBBIS study’s main objectives were as follows: (a) understand how synthetic DNA technologies are being used and who is using them; (b) identify and map the key synthetic DNA providers; (c) investigate these providers’ implementation of sequence and customer screening; (d) identify the government authorities with oversight over synthetic nucleic acids; and (e) review the relevant national policies, regulations, and guidelines, including import- and export-relevant legal instruments. To collect the data, IBBIS drew upon a combination of open-source research, surveys, stakeholder interviews, and collaborations with local partners. This study was specific to synthetic nucleic acids used for research, and not for clinical or therapeutic purposes. In this study, synthetic oligonucleotides were defined as single- or double-stranded DNA or RNA nucleic acid sequences shorter than 200 nucleotides or base pairs. Synthetic genetic constructs (or synthetic DNA constructs) were defined as double-stranded DNA sequences longer than 200 base pairs.
Policies and guidelines around synthesis screening are discussed in Chapter 3: Current Frameworks and Policy Challenges. Mackelprang and the IBBIS presenters discussed opportunities revealed by their research, which are described in Chapter 4: Challenges and Opportunities.

















