Biological Threats in the Age of Emerging Biotechnology: Proceedings of a Workshop Series (2026)
Chapter: 3 Current Frameworks and Policy Challenges
3
Current Frameworks and Policy Challenges
The planning committee and participants discussed international and country-specific issues during the four workshop meetings. The first meeting reviewed existing policies and guidance; considered if risk, hazard, and/or benefit assessments were included in the policies and guidance using committee definitions (see Box 1-2); and asked committee members and participants to discuss gaps. The following three meetings used scenarios to further examine the gaps in existing policies and guidance regarding emerging biotechnologies. The broader policy landscape and scenario-specific landscapes are discussed in this chapter.
The first meeting’s goal was to review existing biosecurity and biosafety frameworks, both in terms of how they assess and mitigate harmful use and how they support responsible science and innovation, focusing on countries involved in this workshop series (India, Peru, Mexico, and Argentina). Espona emphasized the hope of biotechnology-related research and praised the workshop for increasing the dialogue among far-flung communities with related interests. She also highlighted the concept of dual-use life sciences research, which tends to be less familiar to scientists than to security experts. Namdeo said that biotechnology is becoming an important driver of overall economic growth in India, where the bioeconomy is on track to reach $300 billion. Innovative tools like synthetic biology, genome editing, and AI-enabled biological models raise novel biosafety and biosecurity concerns. As the emerging risks of new biotechnologies are addressed, he said, they become safer and public trust is increased. Namdeo said the ultimate goal is to minimize the risks that this research poses to safety and security while maximizing its benefits.
The first meeting’s opening session revealed how differently various countries are approaching the bioeconomy, with different frameworks, activities, documents, and priorities, said Espona. Communities use diverse strategies to address the potential problems related to their particular bioeconomic activities and interests. Espona emphasized the importance of having meetings like this that facilitate collaboration among experts from different countries with different perspectives, because addressing the multiple issues around the biotechnology demands a multidisciplinary and multicultural approach.
REVIEW OF EXISTING FRAMEWORKS AND GUIDANCE
The planning committee searched international frameworks, framework-related journal articles and reviews, and articles on the bioeconomy, focusing on those with relevance to Latin America, India, and/or the United States. Country-specific biosafety guidelines and policy analyses related to the bioeconomy were also included. Meeting participants were encouraged to share their suggestions for additional documents that could contribute to this analysis. Given the relative novelty of the term “bioeconomy,” frameworks and policies specific to biotechnology or fields like neuroscience, AI, or the environment were included. Documents were categorized as international, regional, or country-specific, and their content was assessed based on the definitions used by the planning committee. The process of gathering and comparing these documents was “complicated and difficult,” said Espona, because each participating country uses different terminology to name the bioeconomy, and each document analyzes the bioeconomy from a different perspective. “If it’s about biosafety, biosecurity, or dual use, you don’t have ‘bioeconomy’ or ‘sustainability,’ and the opposite applies as well,” said Espona, who noted that even when the participating countries differ in their focus, “we share the same issues regarding the documents.”
Notable Features of the Collected Documents
Three of the documents were classified as international frameworks for biosafety in the life sciences (Figure 3-1). These international frameworks help to frame the concepts even when they are not specific to the bioeconomy, said Espona. Although more international work may be needed, she noted that the WHO (2022) “Global Guidance Framework for the Responsible Use of the Life Sciences” covered most of the topics that would be discussed throughout this workshop series.
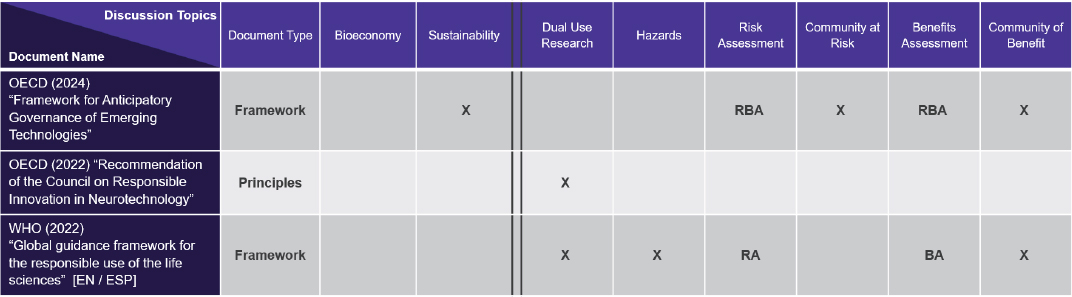
NOTE: (BA) Benefits Assessment; (EN) English; (ESP) Spanish; (RA) Risk Assessment; (RBA) Risk-Benefit Assessment; (X) notes discussion in document on column topic.
SOURCE: Presented by National Academies staff on December 10, 2025.
No single document found to date addresses all the aspects of biotechnologies that were discussed during this workshop series. Journal articles and reviews on risk/benefit assessments did not specify hazards (Figure 3-2).
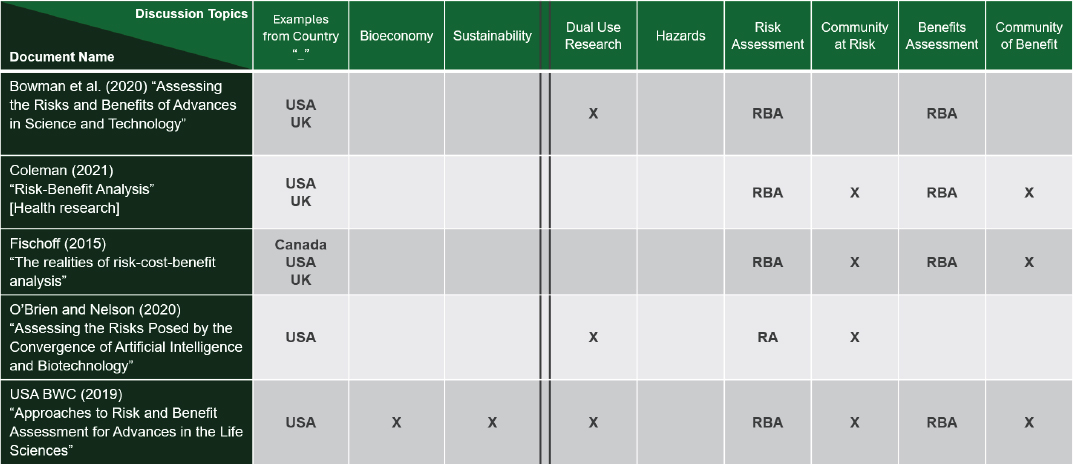
NOTE: Abbreviations (RA) Risk Assessment; (RBA) Risk-Benefit Assessment; (X) notes discussion in document on column topic.
SOURCE: Presented by National Academies staff on December 10, 2025.
Among seven international documents that specifically named the bioeconomy in their titles, only one addressed risk assessment, another benefits assessment, and none addressed dual-use life sciences research or hazards (Figure 3-3).
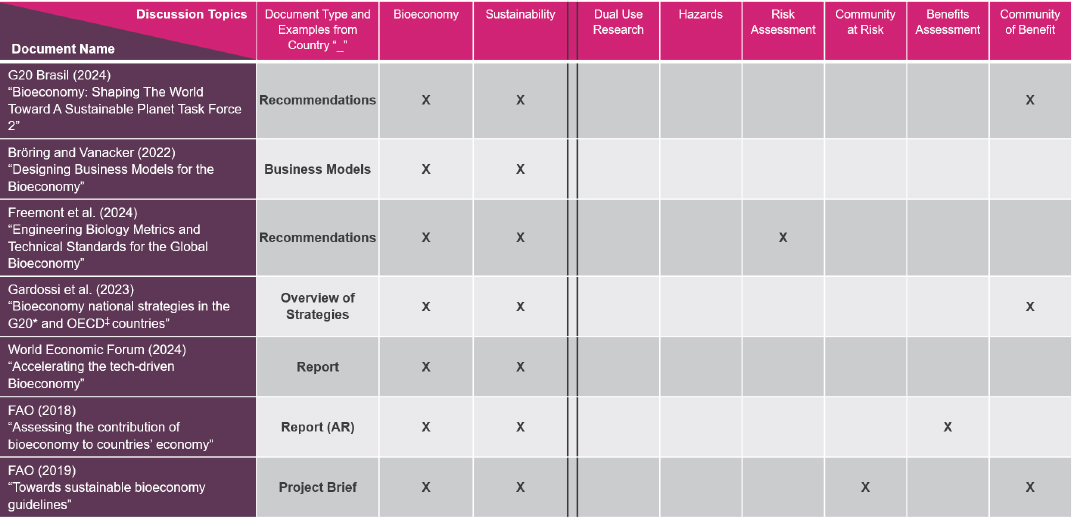
NOTE: *G20 countries include Argentina, India, and Mexico. ‡ OECD countries include Mexico. Abbreviations (FAO) Food and Agriculture Organization of the United Nations; (AR) Argentina; (X) notes discussion in document on column topic.
SOURCE: Presented by National Academies staff on December 10, 2025.
Biotechnology-related documents from the United States are more comprehensive than those from the other countries, said Espona, with widespread discussion of dual use research and risk/benefits assessment, including analyses of the communities at risk and those most likely to benefit. Although a “work in progress,” the U.S. documents can serve as a model for the other countries, she suggested.
International Agreements vs. Local Interests and Legislation
Each country has its own legal framework and national interests, and often making changes is a challenge, said Uriel Alejandro López Lemus, Gene SL. He noted that past administrations in Mexico promoted the development of transgenic products, but many Mexicans do not like transgenics and only want native strains (ISAAA, 2025; USDA GAIN–Mexico, 2025), even though they reduce yield. This aversion is expressed in the legislative branch, and “if legislators are not interested, we will have a barrier to overcome.” However, he added, international trade agreements can impact this interest.
Communication issues occur when non-scientists discuss scientific topics, said Espona. Each country represented in this series of workshops plays a different role internationally. Each has different challenges and priorities, as well as diverse perspectives, biases, and understandings among stakeholder communities. But this workshop series can stimulate the dialogue that is needed to make the most of available technologies and reduce the impact of political transitions on scientific progress, she said.
COUNTRY-SPECIFIC LANDSCAPES
Each of the five countries involved in this conversation has particular interests relevant to safety and security of the biotechnology, said Berger.
India: Frameworks Governing Human Health Multi-Omics and DNA Synthesis
India has a growing set of national-level policies related to the bioeconomy, which spans multiple sectors, said Berger. These sectors include pharmaceutical and agricultural development, biotechnology services, and bioindustry, with considerable vaccine-related work. Regional and US-India partnerships are aimed at building India’s biomanufacturing capabilities and its research and development capacity, and policies include protection of intellectual property and trade secrets and guidance on scaling up production, she said.
The committee reviewed two relevant policy, guidance, or framework documents from India (Figure 3-4). Recently published guidelines for the establishment of biosafety level 3 (BSL3) labs address issues of dual-use research and risk assessment. Many meeting participants suggested the planning committee visit the Department of Biotechnology’s (DBT) India Biosafety knowledge portal in the Rules Act and Guidance section as a resource for policies. More policies from India were discussed during the second meeting, which focused on the use of multi-omic data.
India: Multi-Omics Research for Human Health
Committee member Nidhi Adlakha, Regional Centre for Biotechnology, outlined four framework documents published by the Indian government that are relevant to the security of multi-omics research, and specifically to the human health multi-omics scenario, as follows:
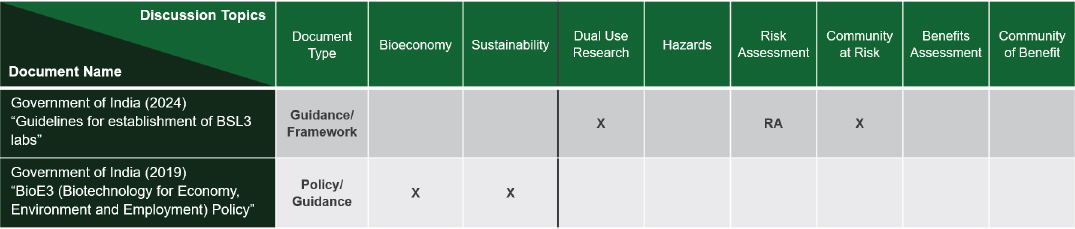
NOTE: Abbreviations (RA) Risk Assessment; (X) notes discussion in document on column topic.
SOURCE: Presented by National Academies staff on December 10, 2025.
- BioE3 (Biotechnology for Economy, Environment and Employment) Policy1
- Biotech-PRIDE (Promotion of Research and Innovation through Data Exchange) Guidelines2
- National Guidelines for the Establishment and Certification of Biosafety Level-3 (BSL-3) Containment Facility, 20243
- Guidelines for sustaining DBT-Bio-Banks and Cohorts4
Of these four documents, only the National Guidelines for the Establishment and Certification of BSL-3 Containment Facility discusses dual-use research, risk assessment, or the community at risk, said Adlakha. BioE3 and Biotech-PRIDE focus on the economic benefit and sustainability of biotechnology research in India. BioE3 has supported the interconnection of multiple human genome databases, with the goal of using them for personalized medicine, while Biotech-PRIDE aims to drive innovation by facilitating data exchange with safeguards (DBT, 2025). India has biological data repository centers where all genomic, transcriptomic, and metabolomic data can be deposited and accessed upon request (see Chapter 2), she added, and additional frameworks and guidelines are available through the DBT biobanks.
With India ramping up its database development technology and governance, Indian participants considered what existing resources may be useful as this effort proceeds. Adlakha asked whether any of the participating countries had guidelines that specifically secured the dissemination of multi-omics data related to human health. Argentina has “a huge gap,” said Espona.
International policies tend to address genomic information far more than the other omics, noted Berger. Among these policies, Europe’s General Data Protection Regulation (GDPR)5 focuses on individual privacy and security, as do other policies, and includes genetic data (European Union, n.d.; Mitchell et al., 2020). An international consortium, the Global Alliance for Genomics and Health, has created standards for protecting not only individuals’ private data but the datasets themselves (GA4GH, 2022). The Global Alliance model is designed to ensure that members share genomic data equally (GA4GH, n.d.), said Berger. Another type of model is a federated system that allows researchers to analyze data from different datasets without keeping the data themselves, she added, noting that India is using that model for its genomics data (Ghosh, 2025).
___________________
1 See https://bmi.dbtindia.gov.in/pdf/folder.pdf (accessed on December 18, 2025).
2 See https://dbtindia.gov.in/sites/default/files/Biotech%20Pride%20Guidelines%20July%202021_0.pdf (accessed on September 25, 2025).
3 Department of Biotechnology and Indian Council of Medical Research. 2024. National Guidelines for the Establishment and Certification of Biosafety Level3 (BSL-3) Containment Facility. https://ibkp.dbtindia.gov.in/Content/Rules. (accessed on December 18, 2025).
4 See https://dbtindia.gov.in/sites/default/files/uploadfiles/Biobank-Cohort-%20sustainability%20guidelines.pdf (accessed on September 25, 2025).
5 See https://gdpr.eu/ (accessed on December 18, 2025).
The Indian research community has a strong commitment to open access science, said Shambhavi Naik, Takshashila Institution, and this informs the Biotech-PRIDE guidelines and DBT policies, which require that raw research data be made available within a year of publication. Safeguards are necessary because “open access means open to misuse,” she said. Naik offered two examples that illustrate India’s approach to enabling multi-omics analysis while safeguarding data. Through the Genome India project, whole-genome sequencing was recently completed for 10,000 human genomes (GenomeIndia, n.d.). Access to the sequence data is layered, and researchers who wish to gain access must partner with a DBT-based laboratory. Second, the National Health Authority and Ayushman Bharat Digital Health Mission is working to develop a national health database for India while securing the data from hacking (NHA, n.d.). Risks to biosecurity based on accessing private data are real, she said, and she cited the United Kingdom General Data Protection Regulation (UK GDPR)6, which directs the management of data in the UK biobank, as model guidance on how access may be protected.
India: Frameworks Governing DNA Synthesis
In a recorded talk, Namdeo shared IBBIS’s findings about nucleic acid synthesis screening in India. India’s biotechnology sector is large and growing rapidly–from $10 billion in 2014 to $165 billion in 2024 and projected to reach $300 billion by 2030, he said. India also has a large biotech and pharma sector that produces around 65 percent of the world’s vaccines and a huge research ecosystem overall, with thousands of labs and hundreds of thousands of researchers working across industry and academia. As noted in the human health multi-omics discussion, the government is pushing to expand India’s bioeconomy and foster high-performance biomanufacturing through the BioE3 (Biotechnology for Economy, Environment, and Employment) Policy.7 This all translates into a high demand for synthetic DNA by academia, industry, healthcare providers, and agriculture companies.
India has no law or regulation that deals specifically with DNA synthesis, said Namdeo. India has no biosecurity policy, and its Guidelines for Biosafety8 do not require companies within India to conduct sequence or customer screening, nor do they reference a “sequences of concern” list. However, he said, existing regulations related to environmental protection, genetically modified organisms, lab biosafety, and national security “could be applied to governing DNA synthesis with some regulatory and administrative tweaking.” These policies include the Environment (Protection) Act,9 Regulations
___________________
6 See https://www.gov.uk/data-protection (accessed on December 19, 2025).
7 See https://birac.nic.in/biomanufacturing.php (accessed on October 24, 2025).
8 See guidelines here: https://dbtindia.gov.in/guidelines-biosafety (accessed on October 24, 2025).
9 See https://www.indiacode.nic.in/bitstream/123456789/6196/1/the_environment_protection_act%2C1986.pdf for the original 1986 Environmental (Protection) Act (accessed on October 24, 2025).
and Guidelines for Recombinant DNA Research and Biocontainment,10 and Rules for the Manufacture, Use, Import, Export and Storage of Hazardous Micro Organisms/Genetically Engineered Organisms or Cells,11 which regulates the production of genetically modified products and specifies conditions for the import and export of nucleic acids. India is a member of the Australia Group, and Namdeo suggested that Australia Group regulations, along with other international norms and standards, could play a key role in addressing the dual-use issue related to export controls.
India’s Guidelines for Biosafety also could be useful, said Namdeo, suggesting that their detailed biocontainment and standards requirements could be modified to require safe and secure use of nucleic acids. Similarly, the Weapons of Mass Destruction Act12 and the Unlawful Activities (Prevention) Act,13 which has anti-terror provisions that could be used against non-state actors who attempt to use synthetic DNA to develop bioweapons (typically defined as pathogens and toxins), could also play a role in regulating nucleic acid synthesis. However, he added, several gaps remain, notably the absence of any specific provision for customer or sequence screening, the relative lack of concern for biosecurity in many current policies, and uneven implementation of institutional biosafety standards.
Namdeo identified 24 companies in India that sell synthetic nucleic acids, most of which are clustered in the cities of New Delhi, Bangalore, and Hyderabad. Seven or eight of these companies conduct in-house synthesis, while the remainder act as distributors for multinational (mostly US-based) companies or are subsidiaries of multinational companies. “There was no evidence of sequence screening” in any of the companies that do in-house synthesis, and none of them are members of the International Gene Synthesis Consortium (IGCS),14 he said. Only two of these in-house nucleic acid synthesis companies use an order form that collects the information needed for verifying and screening customers. Although most of the U.S.-based companies are IGSC members, the IBBIS team did not determine whether they conduct sequence and customer screening for their Indian orders.
Namdeo noted that companies might have multiple concerns related to screening, including the extra cost, fear of over-regulation, and the possible impact on their business if their competitors do not screen. For these reasons, he said, “it’s very difficult to find people to interview, it’s very difficult to find people who are willing to share that information about the practices they have, from the private sector side.” Government
___________________
10 See https://ibkp.dbtindia.gov.in/DBT_Content_Test/CMS/Guidelines/20181115134719867_Regulations-Guidelines-for-Reocminant-DNA-Research-and-Biocontainment-2017.pdf (accessed on December 5, 2025).
11 See http://geacindia.gov.in/resource-documents/biosafety-regulations/acts-and-rules/Rules-for-the-manufacture-use-import-export-and-storage-1989.pdf (accessed on October 24, 2025)
12 See https://www.indiacode.nic.in/handle/123456789/2017 (accessed on October 24, 2025).
13 See https://www.indiacode.nic.in/handle/123456789/1470 (accessed on October 24, 2025).
14 See https://genesynthesisconsortium.org (accessed on October 24, 2025).
can play a big role influencing market forces. “There are not very strong market forces to encourage best practices” in India, he said, but this can change, and it could be beneficial for industry to be engaged in the process to ensure that security is balanced with economic feasibility.
“Very few people across the stakeholders that we have, from industry, academy, or government, know or seriously think about biosecurity,” said Namdeo, who suggested developing programs that raise this awareness. Progress in this area might include a national biosecurity policy that mandates DNA synthesis screening, he said, with a sequence-of-concern list. He suggested integrating international norms, such as the IGSC protocol, into Indian practice.
Given industry’s focus on cost, Namdeo encouraged the promotion of open-source and freely available screening tools, like the IBBIS common mechanism, among industry players. He also suggested offering tax credits or other incentives for adopting safe and secure practices, which “could go a long way towards voluntary screening of the nucleic acids they synthesize.” More broadly, he said, dialogue is needed between government and industry aimed at balancing the need for security with the drive for innovation. By reducing risk, these efforts have the potential to position India as a leader in safe and secure growth of the bioeconomy.
Latin America Overview
Mexico’s investments in the bioeconomy focus on biodiversity, sustainability, economic growth, and national security, said Berger. In contrast to the United States, which focuses its bioeconomy on technological innovation, countries in Central and South America tend to place a greater emphasis on natural products, sustainability, and biodiversity, she said. Argentina has national-level policies for the bioeconomy that focus on sustainable development. These policies address efforts to assess, communicate, and monitor the risks and benefits of more advanced areas, including nanotechnology, gene editing, synthetic biology, and artificial intelligence. Peru’s interests in the bioeconomy are focused on biodiversity, sustainability, and economic growth, she added.
Espona noted that documents related to Mexico, Peru, Argentina, and other Latin American countries address the bioeconomy largely from a sustainability perspective, with relatively little focus on dual-use life sciences research or biosecurity and biosafety risk/benefit (Figure 3-5). In addition, Mexico and Peru currently have laws prohibiting research and use of genetically modified organisms (Figure 3-6).
In Argentina, genetic data are classified as sensitive information and regulated under the Personal Data Protection Law (PDPL, Law 25,326), which requires individual consent for data collection, storage and use, said Ameneiros. This law complements specific regulations on gene databanks, such as the National Genetic Data Bank (BNDG),
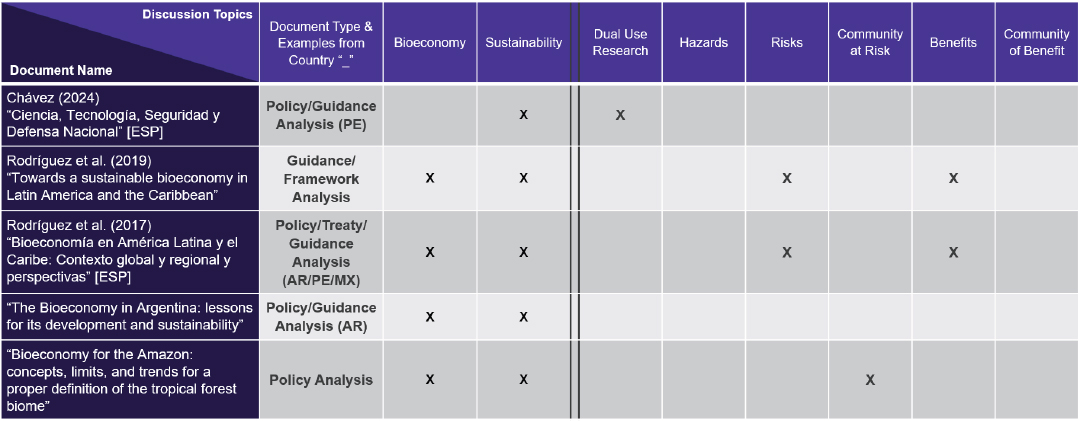
NOTE: * Mexico has legislation prohibiting research and use of genetically modified organisms. * Peru has legislation prohibiting research and use of genetically modified organisms until 2035. Abbreviations (EN) English; (ESP) Spanish; (AR) Argentina; (PE) Peru; (MX) Mexico; (X) notes discussion in document on column topic.
SOURCE: Presented by National Academies staff on December 10, 2025.
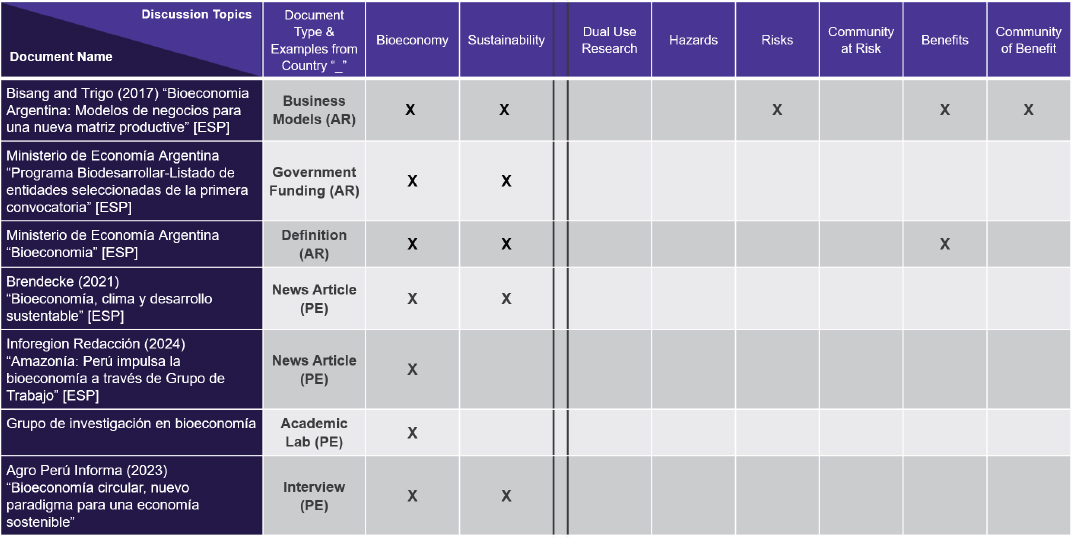
NOTE: (AR) Argentina; (ESP) Spanish; (PE) Peru; (X) notes discussion in document on column topic.
SOURCE: Presented by National Academies staff on December 10, 2025.
that aim to safeguard the use of genetic information. The PDPL is used primarily for human rights investigations and criminal registries, she added, which reflects Argentina’s history.
Status, Challenges, and Future of the Mexican Bioeconomy
In a prerecorded presentation, Luis Ochoa Carrera, Michigan State University, discussed Mexico’s bioeconomy status along with opportunities to enhance its governance and implementation. Mexico’s bioeconomy holds “immense potential” and ranks among the world’s top five nations in biodiversity, he said. Its wealth of biological resources “could drive innovation in agriculture, pharmaceuticals, bioenergy, and beyond, [but] fragmented policies, limited funding, and lack of alignment with global best practices have slowed progress,” he said, and Mexico’s bioeconomy remains underdeveloped. Ochoa Carrera mentioned several different bioeconomic sectors and the challenges they face in Mexico. Agriculture and forestry are key sectors, but efforts to incorporate sustainable agricultural practices like biofertilizers and pest-resistant crops are hindered by regulatory bottlenecks. Mexico has seen growth in biotechnology startups and research efforts, particularly in pharmaceutical biofuels15 and bio-based plastics, but they are challenged by intellectual property constraints and limited access to venture capital. Mexico also has seen innovations in food security, including research into alternative sources of protein and reducing agricultural waste, but “scaling these solutions requires robust policy support, and of course, investment,” he said.
National Regulations Governing Mexico’s Bioeconomy
Mexico has several laws and regulations addressing the bioeconomy, said Ochoa Carrera. The Biosafety law for GMOs regulates the use of genetically modified organisms, with a focus on health and the environment (Cámara de Diputados del H. Congreso de la Unión, 2005). The General Law on Ecological Balance and Environmental Protection (LGEEPA) addresses the environmental risks associated with industrial activities (Cámara de Diputados del H. Congreso de la Unión, 2025). Two official Mexican norms regulate biotechnological processes and products, emphasizing safety in pharmaceuticals and agriculture (Secretaría de Agricultura, Ganadería y Desarrollo Rural, 1999; Secretaría de Medio Ambiente y Recursos Naturales, 2014). But gaps in enforcement, limited inter-agency coordination, and inadequate public-private collaboration undermine the effectiveness of these laws, said Ochoa Carrera.
___________________
15 Integrated biorefining emerges as a strategic response, enabling the co-production of biofuels and pharmaceutical compounds through biomass valorization (see Liu, T., M. He, R. Shi, H. Yin, and W. Luo. 2025. “Biofuel–Pharmaceutical Co-Production in Integrated Biorefineries: Strategies, Challenges, and Sustainability.” Fermentation 11 (6): 312. https://doi.org/10.3390/fermentation11060312.)
Mexico and Peru: Frameworks Governing Crop Improvement Multi-Omics
Participants discussed the challenges limiting the use of multi-omics for crop improvement in Latin America and offered suggestions for overcoming these challenges.
Mexico and Peru: Restrictions for Using Multi-Omics and Gene Editing for Crop Improvement
Like other mega-diverse countries, Peru is a party to both the 1992 Convention on Biological Diversity (Secretariat of the Convention on Biological Diversity, 2011) and the 2010 Nagoya Protocol (Secretariat of the Convention on Biological Diversity, 2010), international treaties aimed at preserving biological diversity and encouraging fair access and benefit-sharing mechanisms for technologies derived from the use of genetic resources (Comisión de la Comunidad Andina, 1996), said Gálvez Ranilla. The Peruvian Ministry of the Environment established many of the rules governing access to genetic resources (MINAM, 2021), and it manages this process through four different national institutions, each of which has its own distinctive application process, paperwork, and waiting times, creating a difficult situation for researchers, she said.
Herrera-Estrella observed two major obstacles to applying gene-editing methods for orphan crops in Mexico and other developing countries. One is social and political. Mexicans consume more maize per capita than any country in the world, he said, and according to Mayan legend, man was created out of maize, highlighting the long link between maize and indigenous cultures. A strong fight against transgenic maize in Mexico is fueled by cultural concerns, he said, and this spills over to other crops. A recent amendment to the Mexican constitution added the passage: “Mexico is the center of origin and diversity of maize, which is an element of national identity…maize cultivation must be free of any modification produced with techniques that go beyond the natural barriers of reproduction or recombination, such as transgenesis.” The implication is that transgenesis, gene editing, and perhaps mutation breeding and hybrids will be outlawed for maize. “This is very dangerous, because if it applies to maize, it will apply to all crops in Mexico,” said Herrera-Estrella. He warned against similar legislation being passed in other countries, “because then people start to believe that gene editing is evil and we should not use any of these things.”
The second obstacle to the use of gene editing for crop improvement is a lack of resources: “we don’t have the tools to do gene editing, we don’t have the tissue culture, we don’t have the characterization of genetic variation to provide the alleles we need,” said Herrera-Estrella.
Are Traditional Maize Strains Threatened by Transgenesis?
Maria Melina Florez-Cuadros, Universidad Peruana Cayetano Heredia, asked Herrera-Estrella to address the concern that transgenic strains of maize could drive out the older,
heirloom strains. Mexico is the center of origin and domestication of maize, answered Herrera-Estrella, with 16 indigenous strains and about 60,000 creole varieties. This diversity started being lost in the 1960’s, he said, for two reasons: (a) migration to the US, which left small family farms deserted; and (b) the introduction of improved (non-transgenic) varieties from the US, which cross-bred with the native creole varieties. “Those original varieties no longer exist,” he said, “so it’s a fairy tale, saying that we have to preserve the maize variety that was generated by our ancestors 5,000 years ago.” Farmers select for types of seed based on how the corn will be used, he added, and they are constantly planting new varieties to allow for cross-breeding and testing for improved yield. “These creole varieties are not the same as 5,000 years ago, not the same as 1,000 years ago, not the same as 100 years ago, not the same as 50 years ago, so this idea of preserving these pristine original varieties is not really true,” he said.
The way to preserve diversity is through seed banks, added Herrera-Estrella. The International Maize and Wheat Improvement Center (CIMMYT) seed bank16 has over 28,000 unique seed collections of maize, many strains are in the Svalbard Global Seed Vault17 in Norway, and Mexico also has a national system of seed banks.18 Additionally, he said, genomic DNA from a vast number of varieties could be preserved and reintroduced into a corn cell at a later date to get the plant back. In contrast, to maintain maize growing in situ, “you have to hire farmers to produce these varieties far from other maize… and pay them very well.” However, the Mexican government does not pay farmers to cultivate the traditional varieties, and little incentive exists for any farmer to grow an heirloom strain that has a fraction of the yield obtained from a hybrid. “It’s complex. It’s not simple. It’s not a problem of transgenics,” he said.
Banking seeds is important for maize, potatoes, and many other crops that are native to Latin America, said Espona. “Collect, standardize, put them in a safe place…that is the first step, and we should communicate that better to the policymakers… it will be like a bank preserving all of that in a way that is safe and secure for future generations,” she said. Although current goals may be to generate strains that are better in different spectra of daylight or weather conditions, Espona added, “we don’t know what will happen in 20 years, what we are going to need. The old crops…have this memory that we should be taking care of.”
Governments need to be invested in preserving their native resources and making the best use of them, said Herrera-Estrella. Right now, he said, in Mexico, a lot of talk exists about how “Mexican creole maize is so fantastic, we should preserve it,” but he
___________________
16 See https://www.cimmyt.org/work/genetic-resources/ (accessed on October 9, 2025).
17 See https://www.regjeringen.no/en/topics/food-fisheries-and-agriculture/svalbard-global-seed-vault/id462220/ (accessed on October 9, 2025).
18 See https://www.academia.edu/124552722/Community_seed_banks_in_Mexico_an_in_situ_conservation_ strategy (accessed on November 12, 2025).
was denied funding to sequence the Creole genome to determine how diverse the varieties actually were, which is essential for deciding which strains to preserve.
Preservation of native Peruvian maize strains is also a challenge. Gálvez Ranilla conducts participatory research with Indigenous communities to promote in situ conservation of the native strains they are growing, in tandem with ex situ conservation, but this work is very difficult. Government regulators do not seem interested in collaborating with academia and industry to develop a strategy for preservation, she said, and the research groups that manage ex situ seed banks do not cooperate with other groups. Nonetheless, a small group of researchers from southern Peru is trying to develop Cabanita maize into a model that could be applied in the future to the preservation and characterization of other maize strains.
Four Priority Work Lines for Omics Research on Crop Biodiversity in Peru
In 2018, a team of Peruvian and UK researchers discussed priority work lines for integrating omics-based research into studies of crop diversity in Peru,19,20 said Gálvez Ranilla. The researchers used SWOT (strengths, weaknesses, opportunities, and threats) analysis21 to identify four priorities:
- Access to genetic resources for both researchers and private companies that want to create products using native resources. Despite revisions to the regulations four years ago, the bureaucratic process remains “very, very hard… waiting times are very, very high, and this compromises our work,” she said, calling for the regulations to be revisited.
- Standardization of curation, collection, and conservation among the various research groups.
- Proper integration and application of omics data (which is currently limited by capacity).
- Capacity building of specialists trained in bioinformatics and data integration.
These changes have to come from the government, said Gálvez Ranilla. “We hope that our government, in some moment, could be open to the Peruvian scientific community…we are open to work together with them,” she said.
___________________
19 See workshop project: https://investigacion.ucsm.edu.pe/taller/english.html and https://www.ukri.org/what-we-do/browse-our-areas-of-investment-and-support/latin-american-biodiversity-programme/. Spanish language workshop descriptions: https://investigacion.ucsm.edu.pe/taller/.
20 See project: “Integrating omic-based technologies for the valorization of Peruvian crop biodiversity” Contract: 114-2018-FONDECYT. This work was funded by a grant from the Newton-Paulet Fund.
21 See https://www.mindtools.com/amtbj63/swot-analysis (accessed on October 10, 2025).
The Increasing Difficulty of Doing Scientific Research in Mexico
Although Mexico always had limited resources for research, Herrera-Estrella was able to maintain his laboratory there for several decades. This changed about seven years ago, he said, when the administration drastically reduced its support for research and “changed the whole concept of why we do science…Rather than say that we want to contribute to human knowledge and develop tools to see if we can address problems, research had to be focused on solving the social problems of Mexico. That was the only thing.”
Mexico, Argentina, and Peru: Frameworks Governing DNA Synthesis
Chavez described the sourcing and regulation of synthetic nucleic acids in Mexico, Argentina, and Peru. Although all three countries obtain synthetic oligonucleotides from a mix of domestic and international producers, these countries rely heavily on international vendors for synthetic DNA constructs (>200 bp). This landscape is likely to change in both Mexico and Argentina, where local companies soon may be producing synthetic DNA constructs, he said.
Most international providers of synthetic nucleic acids are based in the United States or Canada, with others based in Germany, the Netherlands, or South Korea, said Chavez, and the channel for accessing these products is either through direct contact with the provider or via national resellers or authorized distributors. The seven main international providers to Mexico, Argentina, and Peru are considered self-attesting with the OSTP Framework for Nucleic Acid Synthesis Screening (OSTP, 2024) or the IGSC Harmonized Screening Protocol (IGSC, 2024), he said. Although this is a positive finding, he noted that “other companies are also providing these products to these countries, without a clear attestation to any of the voluntary guidelines.” Furthermore, he added, none of these three countries–indeed, no country–has a current legal requirement for either customer or sequence screening.
Six international frameworks are relevant to synthetic nucleic acid regulation, said Chavez, presenting a chart that indicated each framework’s status in Mexico, Argentina, and Peru (Figure 3-7). As referenced in Chapter 1, the Biological Weapons Convention (n.d.) prohibits the development, production, and stockpiling of biological weapons, and requires their destruction or diversion to peaceful purposes, and United Nations Security Council Resolution 1540 (2004) obligates states to adopt and enforce measures to prevent the proliferation of nuclear, chemical, and biological weapons to non-state actors. The Convention on Biological Diversity addresses conservation, sustainable use, and fair benefit sharing of biodiversity (Secretariat of the Convention on Biological Diversity, 2011), and the Cartagena Protocol on Biosafety governs the transboundary movement of living modified organisms (Secretariat of the Convention on Biological Diversity, 2003). All three countries subscribe to these four agreements. In addition, Mexico and
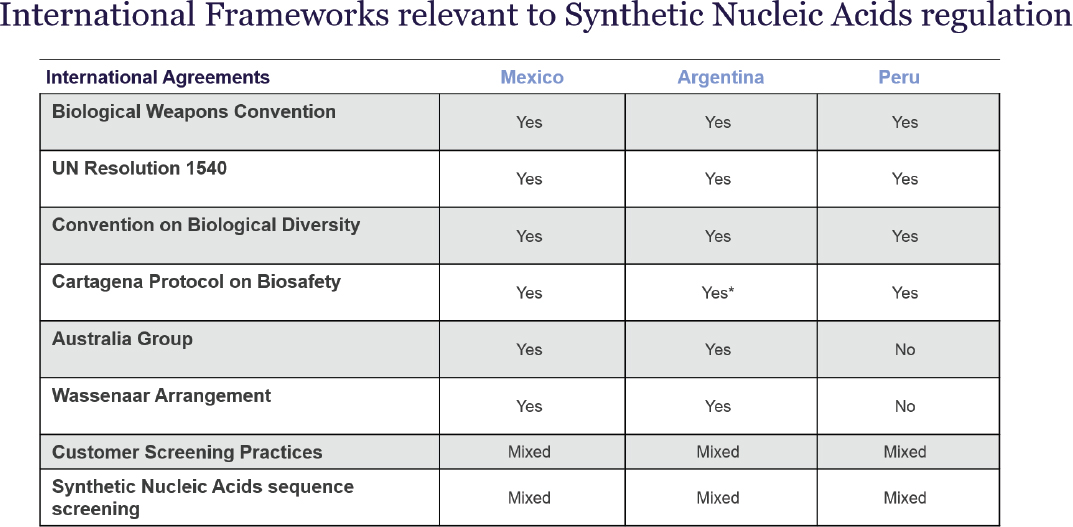
NOTE: *Argentina has signed the Cartagena Protocol on Biosafety, however it has not been ratified within the country, as can be observed here: https://bch.cbd.int/protocol/parties#tab=0 Customer and sequence screening constitute relevant best practices, and they are not international agreements. This was clarified during the presentation.
SOURCE: Presented by Dr. Ricardo Chávez and Mayra Ameneiros on September 18, 2025; created from data in IBBIS Interviews.
Argentina, but not Peru, belong to the Australia Group, which lists certain pathogens and associated genetic elements whose export is controlled and may require a license or be prohibited to certain destinations (The Australia Group, n.d.-a; n.d.-b), and the Wassenaar Arrangement (n.d.), which promotes responsible transfer of conventional arms and dual-use goods and technologies. The IBISS team was unable to find any other legal instruments that might restrict the export of synthetic nucleic acids, said Chavez.
In all three countries, intended use governs the synthetic nucleic acids regulatory procedure, while no specific regulation covers domestic production, said Chavez.
Mexico, Argentina, and Peru: DNA Synthesis Import Screening
Chavez described regulation of synthetic nucleic acids for import screening in Mexico, Argentina and Peru. In Mexico, screening is governed by five legal instruments in addition to the customs and penal codes, and several agreements and guidelines (Table 3-1). Six Mexican government authorities have purview over these laws, agreements, and guidelines. In Argentina, the regulatory framework consists of six key legal instruments (in addition to customs and penal codes) and several decrees, resolutions, and guidelines. Six distinct Argentinian governmental authorities have framework oversight, said Chavez (Table 3-1). Peru’s key legal instrument governing synthetic nucleic acids is the
Moratorium on Living Modified Organisms (El Congreso de la República, 2021), which bans the production and commercialization of GMOs, though research still is allowed, said Chavez. In addition to the moratorium, three additional laws, several decrees, resolutions, guidelines, customs, and penal codes comprise Peru’s synthetic nucleic acids regulatory framework, which are overseen by six different governmental authorities (Table 3-1).
TABLE 3-1 Import Screening Process Steps for Mexico, Argentina, and Peru
| Import Step | Import control step description | ||
|---|---|---|---|
| Mexico | Argentina | Peru | |
| 1 | 1,2,3,4Tariff classification and NTR screening | 6Determine eligibility for the research-imports regime | 19,20Tariff classification (HS) of the shipment |
| 2 | Sectoral import authorization (only if listed in 26/12/2020 accords): 1Human, 2Animal,2Plant Health and 3Environmental Listing | Classify intended use (case-based sectoral gate): 7,8Human biomedical, 9Veterinary, 10Plant (11confined GMO activities), and 12General academic research | 19,21,22,23,24Check OVM moratorium scope and “restricted merchandise” lists (border screening trigger) |
| 3 | 4Customs import clearance (pedimento and validation) | 9,10,11,13,14Confined/contained use authorization (only if agricultural/veterinary/plant GMO work is intended) | 19,21,22,23,25,26 If “restricted merchandise” applies: prearrival/arrival documentary control via VUCE |
| 4 | Confined-use notification before starting GMO work (“aviso de utilización confinada”): 1Human, 2Animal, 2Plant Health and 1,2General Academic Research | 15,16File import via the Single Window for Foreign Trade (VUCEA) | 19 Customs declaration and clearance (non-restricted or once sector steps are complete) |
| 5 | 5Laboratory waste management (RPBI) | 17,18Customs control and release | Post-clearance: before any OVM work, obtain “confined use” authorization 27Human biomedical, 28Veterinary, 28Plant (phytosanitary) and 27,28General Academic Research |
NOTE:
Mexican Authorities
1 Secretaría de Salud (COFEPRIS)
2 Secretaría de Agricultura y Desarrollo Rural (SENASICA)
3 Secretaría de Medio Ambiente y Recursos Naturales (SEMARNAT)
4 Agencia Nacional de Aduanas de México (ANAM)
5 Secretaría de Salud
Argentinian Authorities
6 Secretaría de Innovación, Ciencia y Tecnología (SICyT, Jefatura de Gabinete de Ministros—JGM)
7 Ministerio de Salud (MSAL)
8 Administración Nacional de Medicamentos, Alimentos y Tecnología Médica (ANMAT)
9 Servicio Nacional de Sanidad y Calidad Agroalimentaria (SENASA)
10 Secretaría de Agricultura, Ganadería y Pesca (SAGyP, Ministeriode Economía —MECON)
11 Comisión Nacional Asesora de Biotecnología Agropecuaria (CONABIA)
12 Ventanilla Únicade Comercio Exterior (VUCE)
13 Dirección de Biotecnología
14 Comité Asesoren Bioinsumos de Uso Agropecuario (CABUA)
15 Jefatura de Gabinete de Ministros (Comité de Implementación de VUCEA)
16 Secretaría de Industria y Comercio (MECON)
17 Authority: Dirección General de Aduanas (DGA),
18 Agencia de Recaudación y Control Aduanero (ARCA)
Peruvian Authorities
19 Superintendencia Nacional de Aduanas y de Administración Tributaria (SUNAT)
20 Ministerio de Economía y Finanzas (MEF)
21 Ministerio del Ambiente (MINAM)
22 Servicio Nacional de Sanidad Agraria (SENASA)
23 Organismo Nacional de Sanidad Pesquera (SANIPES)
24 Organismo de Evaluación y Fiscalización Ambiental (OEFA)
25 Ministerio de Comercio Exterior y Turismo (MINCETUR)
26 Ventanilla Únicade Comercio Exterior (VUCE)
27 DIGESA (MINSA) as Órgano Sectorial Competente (OSC)
28 INIA (MIDAGRI) as Órgano Sectorial Competente (OSC)
SOURCE: Adapted from a table presented by Ricardo Chavez and Mayra Ameneiros on September 18, 2025; created from data in IBBIS interviews.
The IBBIS team traced the required stepwise process for importing synthetic nucleic acids that need to be handled in containment. Certain import process steps in Mexico, Argentina, and Peru will vary depending on whether the intended use is for biomedical, veterinary, plant, or general academic research, said Chavez, noting that the regulations may be ambiguous in places.
Mexico, Argentina, and Peru: DNA Synthesis Export Screening
Mexico’s export of synthetic nucleic acids is also a multi-step process, said Chavez. He noted that Mexico’s participation in the Wassenaar Arrangement and Australia Group means that anyone intending to export synthetic nucleic acids with dual-use potential must obtain a specific export license and notify the relevant authority. However, he cautioned, “even though [licensing and notification] is mandated by law, it depends entirely on the user to submit these notifications beforehand.” This has made it difficult to confirm that screening is actually taking place, he said. The export procedures for Argentina are similar to those for Mexico, requiring the user to request a license before exporting dual-use goods, said Chavez.
Mexico, Argentina, and Peru: DNA Synthesis Import and Export Screening Observations
In Mexico, stakeholders displayed uneven awareness of sequence or customer screening, and some assumed minimal or no screening by suppliers, said Chavez. They favored company-led screening, provided that it safeguarded intellectual property and did not extend the already long import timelines. Determining if authorized distributors complied with the regulations that the main provider or parent company attested to was
TABLE 3-2 Export Screening Process Steps for Mexico, Argentina, and Peru.
| Export Step | Export control step description | ||
|---|---|---|---|
| Mexico | Argentina | Peru | |
| 1 | Dual-use control screening (genetic elements/pathogen-related sequences)1 | Screen for export-control sensitivity (dual-use/pathogen-related sequences)5 | Tariff classification (HS) of the shipment14,15 |
| 2 | Export license (“permiso previo de exportación”) if controlled1 | Obtain prior export license (only if controlled under sensitive-exports regime)5 | Check if the export is “restricted merchandise” (sector authorization required before shipment)14 |
| 3 | Sectoral export NTRs (health/environment) screening2,3 | Sectoral checks: Human medicines/biologicals6,7 or Veterinary products8 | If “restricted merchandise” applies: sector authorization via VUCE14,20,21 |
| 4 | Customs export clearance (pedimento and validation)4 | File export via the Single Window for Foreign Trade (VUCEA13) 10,9 | Export customs declaration, manifest, and departure14 |
| 5 | (No fifth step in Mexico) | Customs control and exit11,12 | Enforcement backstop (administrative/environmental/criminal) 14,19 |
NOTE:
Mexican Authorities
1 Secretaría de Economía (SE) Mexico
2 Secretaría de Salud (COFEPRIS)
3 Secretaría de Medio Ambiente y Recursos Naturales (SEMARNAT)
4 Agencia Nacional de Aduanas de México (ANAM)
Argentinian Authorities
5 Comisión Nacional de Control de Exportaciones Sensitivas y Material Bélico (CONCESYMB)
6 Administración Nacional de Medicamentos, Alimentos y Tecnología Médica (ANMAT)
7 Ministerio de Salud (MSAL)
8 Servicio Nacional de Sanidad y Calidad Agroalimentaria (SENASA)
9 Jefatura de Gabinete de Ministros (Comité de Implementación de VUCEA)
10 Secretaría de Industria y Comercio (MECON)
11 Dirección General de Aduanas (DGA)
12 Agencia de Recaudación y Control Aduanero (ARCA)
13 Implementing Unit of the Argentine Single Window for Foreign Trade (VUCEA)
Peruvian Authorities
14 Superintendencia Nacional de Aduanas y de Administración Tributaria (SUNAT)
15 Ministerio de Economía y Finanzas (MEF)
16 Ministerio del Ambiente (MINAM)
17 Servicio Nacional deSanidad Agraria (SENASA)
18 Organismo Nacional de Sanidad Pesquera (SANIPES)
19 Organismo de Evaluación y Fiscalización Ambiental (OEFA)
20 Ministerio de Comercio Exteriory Turismo (MINCETUR)
21 Ventanilla Únicade Comercio Exterior (VUCE)
SOURCE: Adapted from a table presented by Ricardo Chavez and Mayra Ameneiros on September 18, 2025; created from data in IBBIS interviews.
difficult. Orders could be held unpredictably at customs (with buffer agent composition22 sometimes triggering additional delays), but high-volume purchases from domestic vendors could also be “throttled.” Stakeholders interviewed by IBBIS are currently only meeting the minimum legal requirements for synthetic nucleic acid synthesis, he said, and this is “reinforcing the demand for clear rules that do not lengthen the customs clearance process.”
In Argentina, stakeholders revealed that, although academic labs dominate the demand for synthetic nucleic acids, start-up biotechnology companies, which currently number about 200, increasingly are ordering these products, said Chavez. Meanwhile, researchers remain largely dependent on foreign vendors. Argentina has a registry of institutions that was intended to reduce taxes and fast-track scientific imports (Argentina.gob.ar, n.d.), but actual clearance times can extend to three months, he said, and the policy has limited transparency. Jurisdictional fragmentation sometimes leads agencies to pass cases among themselves when they cannot determine which agency oversees approval for a specific shipment of synthetic DNA, further stalling import authorizations. This problem was also encountered in Mexico and Peru. Chronic delays, along with unpredictable release windows of 30-50 days (with associated storage fees and cold-chain surcharges), have led researchers to adopt informal workarounds to sustain their operations, said Chavez, “underscoring the systemic frictions between researchers, entrepreneurs, and the regulatory bodies that exist within this field.”
Peruvian providers typically cannot produce any plasmids longer than 2,000 base pairs, and importing larger genetic constructs is difficult, said Chavez. As a result, stakeholders report ordering from resellers in other countries, sometimes with delivery to the individual rather than the institution. Shipments clearing customs can take anywhere from three days to two months, with long wait times threatening sample viability. Peru has no statutory requirement for sequence screening as do Mexico and Argentina, and stakeholders perceived little or no customer screening. Rather, customs reviews the goods based on their intended use, which may cause confusion regarding which authority is responsible for clearing them. On the governance side, Chavez noted that Peru has plans to establish a BWC-related national focal body.
Mexico, Argentina, and Peru: Synthesis Screening Policy Opportunities
The IBBIS team evaluated their findings in light of a 2023 Regional Biosecurity Threat Assessment by the Organization of American States (OAS) (Binder, Williams and Sin, 2023), said Chavez. Some key insights of the OAS report mirrored IBBIS’ results. Governance gaps constitute the main challenge, as weaknesses in legislation or enforcement can be exploited by threat actors; “failure to enable prevention, detection, regulation,
___________________
22 A buffering agent provides a stable pH.
and prosecution of illicit biological activities materially increases national biosecurity risk,” he said. Second, non-compliance carries reputational and economic costs, with states that are perceived as insecure being put at risk of losing bioscience collaborations and investment.
Chavez noted three key opportunity areas for improving the biosafety and biosecurity of synthetic nucleic acids in Mexico, Argentina, and Peru. The first, he said, is addressing existing framework ambiguity by identifying the agencies in charge of regulating each type of imported product as well as domestic production. Chavez suggested enhancing these frameworks by using procurement leverage with attestation, defining sequences of concern, recommending baseline sequence screening thresholds to suppliers, requiring baseline legitimacy verification of customers, setting minimal record-keeping standards, establishing a single national reporting point, and adopting a red-flags schedule.
The second improvement opportunity addresses the importance of implementing sequence screening of imports. Chavez suggested that all three countries consider using the Single Window for Foreign Trade (VUCE),23 which has been adopted by more than 120 member countries of the World Trade Organization to comply with Article 10.4 of the World Trade Organization Trade Facilitation Agreement (International Trade Centre, 2020). The VUCE has the potential to house the implementation of synthetic nucleic acid screening for imports and exports, he said, which can both reduce risk and ensure compliance with current regulatory frameworks and international commitments. VUCE adoption could also address current paperwork challenges, said Chavez, noting that in some cases the import process still requires e-mail and/or paper-based submission or even has the researcher appear in person.
The third key opportunity mentioned by Chavez involves human resources training and new technologies to aid the regulatory process. Training regulatory staff to better understand emerging technologies related to synthetic nucleic acids could help avoid misinterpretation, he said, and improve relations between researchers and regulatory agencies. Artificial intelligence tools could potentially be used to assist in the risk assessment process and reduce approval time. Chavez noted that Panama is developing a digitalization and traceability tool for the importation of medicines and research-use-only supplies.
Mexico: The Particular Challenges Facing Dual-Use Research
Dual-use research of concern (DURC) poses a unique challenge to Mexico’s bioeconomy, which the current frameworks do not adequately address, said Ochoa Carrera. To
___________________
23 See https://www.vuce.gob.pe (accessed on October 24, 2025).
mitigate DURC risks, he said, the following measures may be essential: (a) expanding the biosafety and biosecurity protocols to include DURC-specific risks, with an emphasis on responsible use of these technologies; (b) creating advanced monitoring systems to track high-risk biological agents and technologies; and (c) developing training programs that foster a culture of responsible research among scientists and institutions. Ochoa Carrera pointed out several vulnerabilities that characterize the relationship between researchers and potential DURC activities in Mexico. One is limited awareness; many researchers lack understanding of DURC implications, which increases the likelihood of unintentional misuse. Another is insufficient oversight, which he attributed to a lack of robust mechanisms for identifying and mitigating DURC risks at the institutional level.
Ochoa Carrera had several suggestions for raising awareness of DURC while maintaining support for research. Raising awareness requires a multi-pronged approach, he said. Educational campaigns and nationwide initiatives can promote responsible science to researchers, policy makers, and the general public, with workshops, conferences, and platforms that foster dialogue on DURC mitigation strategies. All stakeholders could be engaged in these activities. He suggested establishing a national DURC committee to oversee biosecurity measures and provide guidance on emerging risks.
Ochoa Carrera emphasized the importance of balancing innovation with safety, noting that the highest quality, safety, and security standards may be necessary “to keep the science rolling.” He also suggested investing adequately in bioeconomy-related research and infrastructure and fixing the policy fragmentation that created disjointed frameworks across sectors. Opportunities that can “take us to the next level” include international partnerships, which can be leveraged to strengthen frameworks and build capacity; public-private collaborations with industry stakeholders aimed at driving both innovation and compliance; and interdisciplinary integration, “combining expertise across different sectors to address biosecurity challenges holistically,” he said.
Strategies for Addressing DURC Challenges in Mexico
Ochoa Carrera suggested four strategies that Mexico can adopt to help secure its bioeconomy, as follows:
- Promoting institutional oversight by establishing dedicated biosecurity committees within research institutions to monitor DURC-related projects.
- Fostering international cooperation, partnering with global organizations to exchange best practices and implement joint training programs.
- Continuously monitoring technologies and equipment with DURC potential, such as CRISPR and synthetic biology. Bioinformatics and AI tools also need
- oversight to prevent misuse, e.g., by facilitating the design of pathogens. Effective use of these tools requires a balanced approach, said Ochoa Carrera, and scientists should be taught to use them wisely.
- Strengthening legal frameworks for promoting safer science, which includes amending existing laws, including updating the biosafety law for GMOs to address DURC risks, developing a national biosecurity strategy that outlines clear pathways for enhancing biosecurity across sectors, and increasing penalties for noncompliance.






















