Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 19 (2015)
Chapter: 5 Tellurium Hexafluoride Acute Exposure Guideline Levels
Acute Exposure Guideline Levels
PREFACE
Under the authority of the Federal Advisory Committee Act (FACA) P.L. 92-463 of 1972, the National Advisory Committee for Acute Exposure Guideline Levels for Hazardous Substances (NAC/AEGL Committee) has been established to identify, review, and interpret relevant toxicologic and other scientific data and develop AEGLs for high-priority, acutely toxic chemicals.
AEGLs represent threshold exposure limits for the general public and are applicable to emergency exposure periods ranging from 10 minutes (min) to 8 hours (h). Three levels—AEGL-1, AEGL-2, and AEGL-3—are developed for each of five exposure periods (10 and 30 min and 1, 4, and 8 h) and are distinguished by varying degrees of severity of toxic effects. The three AEGLs are defined as follows:
AEGL-1 is the airborne concentration (expressed as parts per million or milligrams per cubic meter [ppm or mg/m3]) of a substance above which it is predicted that the general population, including susceptible individuals, could experience notable discomfort, irritation, or certain asymptomatic, nonsensory effects. However, the effects are not disabling and are transient and reversible upon cessation of exposure.
____________________
1This document was prepared by the AEGL Development Team composed of Sylvia Talmage (Oak Ridge National Laboratory), Heather Carlson-Lynch (SRC, Inc.), Chemical Manager George Rusch (National Advisory Committee [NAC] on Acute Exposure Guideline Levels for Hazardous Substances), and Ernest V. Falke (U.S. Environmental Protection Agency). The NAC reviewed and revised the document and AEGLs as deemed necessary. Both the document and the AEGL values were then reviewed by the National Research Council (NRC) Committee on Acute Exposure Guideline Levels. The NRC committee has concluded that the AEGLs developed in this document are scientifically valid conclusions based on the data reviewed by the NRC and are consistent with the NRC guidelines reports (NRC 1993, 2001).
AEGL-2 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience irreversible or other serious, long-lasting adverse health effects or an impaired ability to escape.
AEGL-3 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience life-threatening health effects or death.
Airborne concentrations below the AEGL-1 represent exposure concentrations that could produce mild and progressively increasing but transient and nondisabling odor, taste, and sensory irritation or certain asymptomatic, nonsensory effects. With increasing airborne concentrations above each AEGL, there is a progressive increase in the likelihood of occurrence and the severity of effects described for each corresponding AEGL. Although the AEGL values represent threshold concentrations for the general public, including susceptible subpopulations, such as infants, children, the elderly, persons with asthma, and those with other illnesses, it is recognized that individuals, subject to idiosyncratic responses, could experience the effects described at concentrations below the corresponding AEGL.
SUMMARY
Tellurium hexafluoride is a byproduct of ore refining. It is a colorless gas with a repulsive odor. It decomposes slowly in water to form hydrogen fluoride and tellurium ion. Tellurium hexafluoride is severely irritating and causes respiratory distress, pulmonary edema, and death in animals. In humans, it is reported to cause “garlic” breath, a metallic taste in the mouth, and fatigue. Inhalation of tellurium hexafluoride is expected to cause breathing difficulties in humans.
AEGL-1 values are not recommended for tellurium hexafluoride because of insufficient data. Absence of AEGL-1 values does not imply that exposures below the AEGL-2 concentrations are without adverse effects.
Data were also inadequate for deriving AEGL-2 values. However, the standing operating procedures for determining AEGL values specifies that AEGL-2 values for chemicals with steep concentrations-response curves may be estimated by dividing the AEGL-3 values by 3 (NRC 2001). Lethality data on tellurium hexafluoride demonstrates a steep-concentration response curve. All rabbits, guinea pigs, rats, and mice exposed for 4 h to tellurium hexafluoride at concentrations of 5 ppm or higher died, and all mice exposed at 5 ppm for 1 h died. All animals exposed at 1 ppm for 1 or 4 h survived (Kimmerle 1960).
The point-of-departure for deriving AEGL-3 values was 1 ppm for 4 h, which was the highest concentration of tellurium hexafluoride at which no mortality occurred in rabbits, guinea pigs, rats, and mice (Kimmerle 1960). An interspecies uncertainty factor of 3 was applied because the four species appear to be
similarly sensitive to the acute effects of tellurium hexafluoride; however, that assessment is based on a small number of test animals (one to four animals per group). An intraspecies uncertainty factor of 3 was applied because tellurium hexafluoride is highly irritating and corrosive, and much of its toxicity is probably caused by a direct chemical effect on tissues; that type of portal-of-entry effect is not expected to vary greatly among individuals. A modifying factor of 10 also was applied to account for the sparse database on tellurium hexafluoride and for the potential effects of tellurium. Time scaling of the values were performed using the equation Cn × t = k, where the exponent n ranges from 0.8 to 3.5 (ten Berge et al. 1986). In the absence of data on tellurium hexafluoride to determine an empirical value for n, default values of n = 3 when extrapolating to shorter durations and n = 1 when extrapolating to longer durations were used.
The AEGL values for tellurium hexafluoride are presented in Table 5-1.
1. INTRODUCTION
Tellurium hexafluoride is a colorless gas created by the direct fluorination of tellurium metal (HSDB 2008). It is a byproduct of ore refining, and there are no known uses for it (ACGIH 2001; HSDB 2008). Production data were not found. Tellurium hexafluoride hydrolyzes slowly in water to hydrogen fluoride and telluric acid. Its chemical and physical properties are presented in Table 5-2.
2. HUMAN TOXICITY DATA
2.1. Acute Lethality
No human data on the acute lethality of tellurium hexafluoride were found.
2.2. Nonlethal Toxicity
2.2.1. Odor Threshold and Odor Awareness
Tellurium hexafluoride has a repulsive odor (ACGIH 2001).
2.2.2. Case Reports
Blackadder and Manderson (1975) reported a case of tellurium hexafluoride exposure. Two men, 24 and 26 years old, were exposed when 50 g of tellurium hexafluoride gas leaked from a cylinder while they were doing research. The first man experienced tiredness, a metallic taste in the mouth, and sour garlic odor in his breath, sweat, and urine. He was admitted to the hospital for observation and developed a rash on the hands, arms, and neck after the second
day of observation. He also developed bluish-black patches between the fingers and on the neck and face, which took several weeks to fade. The second man experienced garlic odor of the breath and bluish-black patches on the skin. Liver-function tests, renal-function tests, urinalysis, chest radiographs, blood electrolytes, and blood indices were all normal. The men were not treated and both completely recovered; the garlic odor of the breath and blue-black patches on the skin took several weeks to clear. The skin discoloration was thought to be the result of dermal absorption of tellurium.
TABLE 5-1 AEGL Values for Tellurium Hexafluoride
| Classification | 10 min | 30 min | 1 h | 4h | 8 h | End Point (Reference) |
| AEGL-1 | NRa | NRa | NRa | NRa | NRa | Insufficient data. |
| (nondisabling) | ||||||
| AEGL-2 | 0.0097 ppm | 0.0067 ppm | 0.0053 ppm | 0.0033 ppm | 0.0017 ppm | One-third of the |
| (disabling) | (0.096 mg/m3) | (0.066 mg/m3) | (0.052 mg/m3) | (0.033 mg/m3) | (0.017 mg/m3) | AEGL-3 values (NRC 2001). |
| AEGL-3 (lethal) | 0.029 ppm (0.28 mg/m3) | 0.020 ppm (0.20 mg/m3) | 0.016 ppm (0.16 mg/m3) | 0.010 ppm (0.10 mg/m3) | 0.0050 ppm (0.049 mg/m3) | Highest concentration causing no mortality in rabbits, guinea pig, rats, and mice (Kimmerle 1960). |
aNot recommended. Absence of an AEGL-1 value does not imply that exposure below the AEGL-2 concentration is without adverse effects.
TABLE 5-2 Chemical and Physical Properties of Tellurium Hexafluoride
| Parameter | Value | References |
| Synonyms | Tellurium fluoride (TEF6), (OC-6-11)-; tellurium fluoride (TEF) | HSDB 2008 |
| CAS registry no. | 7783-80-4 | HSDB 2008 |
| Chemical formula | TeF6 | HSDB 2008 |
| Molecular weight | 241.61 | HSDB 2008 |
| Physical state | Colorless gas | HSDB 2008 |
| Melting point | -37.6ºC | ACGIH 2001 |
| Boiling point | -38.9ºC | ACGIH 2001 |
| Vapor density (air = 1) | 8.3 | HSDB 2008 |
| Solubility in water | Decomposes slowly in water to telluric acid | ACGIH 2001 |
| Vapor pressure | >760 torr at 20°C | ACGIH 2001 |
| Flammability limits | Nonflammable gas | NIOSH 2011 |
| Conversion factors | 1 ppm = 9.88 mg/m3 1 mg/m3 = 0.10 ppm | NIOSH 2011 |
2.2.3. Occupational Exposure
Steinberg et al. (1942) examined 49 workers exposed to fumes of tellurium and its oxides for 15 or 22 months. The most commonly reported subjective symptoms were garlic odor of the breath, mouth dryness, metallic taste, somnolence, and garlic odor of the sweat. A small number of subjects occasionally reported loss of appetite and nausea. Somnolence was observed only in the workers with the highest urinary concentrations of tellurium. No alterations in hematologic or urinalysis parameters were observed.
2.3. Neurotoxicity
No human data on the neurotoxicity of tellurium hexafluoride were found.
2.4. Developmental and Reproductive Toxicity
No human data on the developmental or reproductive toxicity of tellurium hexafluoride were found.
2.5. Genotoxicity
No human data on the genotoxicity of tellurium hexafluoride were found.
2.6. Carcinogenicity
No human data on the carcinogenicity of tellurium hexafluoride were found.
2.7. Summary
Human exposure to tellurium hexafluoride or fumes of tellurium oxides caused metallic taste in the mouth; tiredness; sour garlic odor of the breath, sweat, and urine; and bluish-black patches on the skin. Tellurium hexafluoride is a respiratory irritant and humans may experience breathing difficulties after inhaling it (NIOSH 1978; OSHA 1996).
3. ANIMAL TOXICITY DATA
3.1. Acute Lethality
Kimmerle (1960) exposed one rabbit, one guinea pig, two male white rats, and four male white mice per group to tellurium hexafluoride for 1 or 4 h. Exposures were carried out in a 2-m3 chamber. Tellurium hexafluoride was intro-
duced into the chamber through a glass burette and mixed with air by a propeller. The animals were exposed at 1 or 5 ppm (nominal concentrations) for 1 h and 1, 5, 10, 25, 50 or 100 ppm (nominal concentrations) for 4 h. The results are shown in Table 5-3. At 5 ppm for 1 h, severe damage was observed in the respiratory organs of the animals, and all mice died between 24 and 36 h. All animals survived 1-h exposures at 1 ppm. Exposure to tellurium hexafluoride at 1 ppm for 4 h caused respiratory dysfunction in all animals. All animals died from pulmonary edema after exposure at 5 ppm or higher for 4 h.
TABLE 5-3 Results of Acute Toxicity Studies of Tellurium Hexafluoride by Kimmerle (1960)
| Species | Concentration (ppm) | Duration (h) | Effect |
| Rabbit | 1 | 4 | Respiratory dysfunction, pulmonary edema. |
| (1/group) | 5 | Death after 8 h. | |
| 10 | Death after 140 min. | ||
| 25 | Death after 80 min. | ||
| 50 | Death after 60 min. | ||
| 100 | Death after 15 min | ||
| Rabbit | 1 | 1 | Significantly increased respiratory frequency. |
| (1/group) | 5 | Severe damage to respiratory organs. | |
| Guinea pig | 1 | 4 | Respiratory dysfunction, pulmonary edema. |
| (1/group) | 5 | Death after 6 h. | |
| 10 | Death after 120 min. | ||
| 25 | Death after 100 min. | ||
| 50 | Death after 70 min. | ||
| 100 | Death after 30 min. | ||
| Guinea pig | 1 | 1 | Significantly increased respiratory frequency. |
| (1/group) | 5 | Severe damage to respiratory organs. | |
| Rat | 1 | 4 | Respiratory dysfunction, pulmonary edema. |
| (2/group) | 5 | Death after 6 and 24 h. | |
| 10 | Death after 100 and 115 min. | ||
| 25 | Death after 60 and 85 min. | ||
| 50 | Death after 55 and 70 min. | ||
| 100 | Death after 20 and 25 min. | ||
| Rat | 1 | 1 | Significantly increased respiratory frequency. |
| (2/group) | 5 | Severe damage to respiratory organs. | |
| Mouse | 1 | 4 | Respiratory dysfunction, pulmonary edema. |
| (4/group) | 5 | Death within 4-24 h. | |
| 10 | Death within 110-130 min. | ||
| 25 | Death within 75-110 min. | ||
| 50 | Death within 45-70 min. | ||
| 100 | Death within 10-30 min. | ||
| Mouse | 1 | 1 | Significantly increased respiratory frequency. |
| (4/group) | 5 | Death between 24-36 h, severe damage to respiratory organs. | |
3.2. Nonlethal Toxicity
Kimmerle (1960) exposed one rabbit, one guinea pig, two male white rats, and four male white mice per group to tellurium hexafluoride at 1 or 5 ppm for 1 or 4 h, as described in Section 3.1. Significantly increased respiratory frequency (hyperpnea) was observed in all animals exposed at 1 ppm for 1 h. At 5 ppm for 1 h, severe damage was observed in the respiratory organs of the animals; the rabbit, guinea pigs, and rats survived the exposure but recovered very slowly. Exposure to tellurium hexafluoride at 1 ppm for 4 h caused respiratory dysfunction in all animals. The investigator also exposed the same species to tellurium hexafluoride at 1 ppm for 1 h each day for 5 days and found no visible effects in the animals. In the rabbit, liver-function tests were carried out after the end of the repeat-exposure test and again one week later. No hepatic damage was observed.
There are few data on other tellurium compounds. One study reported that one of four guinea pigs died 24 h after a single injection of tellurium oxide. The remaining guinea pigs survived the 1-week observation period (Amdur 1958). No histologic alterations were observed in the livers or kidneys of the surviving animals.
3.3. Developmental and Reproductive Toxicity
No animal data on the developmental or reproductive toxicity of tellurium hexafluoride were found, but a few studies on tellurium and tellurium pulveratum were available.
Oral exposure studies have found that the developing nervous system is sensitive to the toxicity of tellurium. Highly synchronous primary demyelination of peripheral nerves followed by remyelination was observed in developing rats exposed to 1.1% tellurium in the diet (Malczewska-Toth 2012). The demyelination was due to tellurium-induced inhibition of squalene epoxidase activity. Duckett (1970) reported that there were no differences in the size or appearance of the fetuses of dams exposed to tellurium at 3,000 ppm in their diet. Although no anomalies were found by light microscopy of the brains, electron microscopic examination revealed morphologic anomalies in the ependymal layer of tellurium-exposed fetuses; no microvilli were detected in the ventricular plasma-lemma and the number of mitochondria was greatly diminished.
In weanling rats (17 days old) fed a diet containing 1% tellurium pulveratum for at least 3 days, partial or complete paralysis of the hind limbs was observed (Lampert et al. 1970). A gradual recovery started on the tenth day of exposure, and weakness of the hind limbs was only occasionally observed after 20-25 days of exposure. The investigators also noted severe wasting in the animals by the tenth exposure day. Consistent with the clinical signs, increased cellularity and demyelination was observed in the lumbar roots and sciatic nerves, with peak damage occurring after 4 days of exposure; remeylination was observed after 10 days of exposure. The investigators suggested that tellurium-induced
neuropathy was age-specific, as evidenced by remyelination despite continuing exposure. No histologic alterations were observed in the brain or spinal cord and demyelinated axons were occasionally observed in the brachial plexus of some animals; in general, no alterations were observed in the liver.
3.4. Genotoxicity
No data on the genotoxicity of tellurium hexafluoride were found.
3.5. Chronic Toxicity and Carcinogenicity
No data on the chronic toxicity or carcinogenicity of tellurium hexafluoride were found.
3.5. Summary
Only one study of tellurium hexafluoride toxicity in animal models was found. In that study, all rabbits, guinea pigs, rats, and mice exposed to tellurium hexafluoride at 5, 10, 25, 50, or 100 ppm for 4 h died (Kimmerle 1960). All mice exposed at 5 ppm for 1 h died, whereas animals exposed at 1 ppm for 1 h survived. Repeated exposure to tellurium hexafluoride at 1 ppm for 1 h per day for 5 days resulted in no clinical signs or mortality in any species tested. Clinical signs (respiratory distress) and post-mortem findings (pulmonary edema) were consistent with severe irritation in all animals except those exposed at 1 ppm for 1 h, which exhibited only hyperpnea. A limitation of this study is that only a small number of animals were tested. No data on the developmental or reproductive toxicity, genotoxicity, or chronic toxicity or carcinogenicity following inhalation exposure to tellurium hexafluoride were found. Studies of related chemicals (tellurium and tellurium pulveratum) have reported demyelination in peripheral nerves and morphologic alterations in the brain of developing animals after oral exposure (Duckett 1970; Lampert et al. 1970; Malczewska-Toth 2012).
4. SPECIAL CONSIDERATIONS
4.1. Metabolism and Disposition
Little information on the metabolism and disposition of tellurium hexafluoride were found. Tellurium hexafluoride may be hydrolyzed in the moist respiratory tract to hydrogen fluoride and the tellurium ion or telluric acid (HSDB 2008). Tellurium is distributed through the body with higher concentrations found in the kidneys, liver, bone, brain, and testes (IPCS 1998). In the liver, hepatic metabolism creates dimethyl telluride, which is exhaled and has a garlic
odor (IPCS 1998). Tellurium is mainly excreted in the urine with small amounts exhaled as dimethyl telluride (IPCS 1998).
4.2. Mechanism of Toxicity
In the moist respiratory tract, tellurium hexafluoride slowly hydrolyzes into hydrogen fluoride and tellurium ion or telluric acid. Kimmerle (1960) has shown that the toxic effects of inhaled tellurium hexafluoride are consistent with severe irritation and corrosion. Hydrogen fluoride is a severe irritant to the skin, eyes, and respiratory tract. The AEGL values for hydrogen fluoride, however, are orders of magnitude higher than the AEGL values for tellurium hexafluoride, which may indicate that hydrogen fluoride does not play a major role in its toxicity. Penetration of hydrogen fluoride to the lungs produces pulmonary hemorrhage and edema and may result in death (NRC 2004a). The mechanism of toxicity of tellurium is unknown. It has been shown that tellurium inhibits squalene epoxidase, which might interfere with neurotransmission through demyelination. Demyelination has been observed in young animals but not in humans (Anthony et al. 2001).
4.3. Structure-Activity Relationships
Because one mole of tellurium hexafluoride may decompose in moist atmospheres to form up to six moles of hydrogen fluoride, it might be assumed that tellurium hexafluoride may be approximately six times more toxic than hydrogen fluoride on a molar basis. However, the small data set on tellurium hexafluoride suggests that it is much more than six times as toxic as hydrogen fluoride.
The 1-h LC50 values for hydrogen fluoride for the mouse range from 342 to 501 ppm (NRC 2004a). If the acute inhalation toxicity of tellurium hexafluoride was due only to hydrogen fluoride, then 1-h LC50 values for tellurium hexafluoride should have a range of 57-84 ppm for mice. However, 100% mortality occurred in mice exposed to tellurium hexafluoride at 5 ppm for 1 h (Kimmerle 1960). The greater relative toxicity of tellurium hexafluoride might be due to the tellurium moiety and/or the slow hydrolysis rate of tellurium hexafluoride. If the slow hydrolysis rate resulted in more hydrogen fluoride being released in the lung than in the upper respiratory tract, it would result in greater pulmonary damage and likely be more lethal. Mortality in rats exposed to hydrogen fluoride at 1,300 ppm for 30 min by cannulation (to simulate mouth breathing) was 25%, whereas no mortality occurred in rats similarly exposed by nasal breathing (Stavert et al. 1991).
Few toxicity studies are available on other metal hexafluorides, such as uranium hexafluoride and selenium hexafluoride. The relevance of those compounds to tellurium hexafluoride has not been established. Tellurium hexafluoride is analogous to selenium hexafluoride in molecular structure and noble gas
configuration. Both are irritating gases that cause pulmonary edema and death. Tellurium hexafluoride was found to be more toxic in laboratory animals than selenium hexafluoride (Kimmerle 1960); although this comparison is limited by the small number of animals tested for both compounds (one rabbit, one guinea pig, two rats, and four mice). All rabbits, guinea pigs, rats, and mice exposed to selenium hexafluoride at 10 ppm for 4 h died, but survived exposure at 5 or 1 ppm. Animals exposed at 5 ppm exhibited difficulty breathing and pulmonary edema, which resolved during the follow-up period. No effects were observed in animals exposed selenium hexafluoride at 1 ppm. All rabbits, guinea pigs, rats, and mice exposed to tellurium hexafluoride at 5 ppm for 4 h died, but survived exposure at 1 ppm. Animals exposed at 1 ppm exhibited difficulty breathing and pulmonary edema, which resolved during the follow-up period. Unlike tellurium hexafluoride, uranium hexafluoride rapidly hydrolyzes to form hydrogen fluoride and uranyl fluoride (NRC 2004b); thus, the site of toxicity might be different from that of tellurium hexafluoride. Acute inhalation exposure to uranium hexafluoride results in renal damage caused by the uranium moiety (NRC 2004b). However, no evidence that the kidney is a sensitive target of tellurium hexafluoride or other tellurium compounds was found. No histologic alterations were observed in the kidneys of guinea pigs administered a single injection of tellurium oxide and no alterations in urinary glucose or albumin concentrations or urine specific gravity were observed in workers exposed to tellurium and its oxides (Steinberg et al. 1942).
4.4. Other Relevant Information
Although the data on the toxicity of tellurium are sparse, they suggest that it is a neurotoxicant. Somnolence was reported following accidental acute exposure to tellurium hexafluoride (Blackadder and Manderson 1975) and in workers exposed to tellurium and its oxides (Steinberg et al. 1942). Additionally, demyelination of peripheral nerves and morphologic alterations in the brain were observed in developing animals (Duckett 1970; Lampert et al. 1970; Malczewska-Toth 2012).
4.4.1. Species Variability
The study by Kimmerle (1960) suggests that the acute toxicity of tellurium hexafluoride is similar between rabbits, guinea pigs, rats, and mice. Mice might be slightly more sensitive, as they died from exposure to tellurium hexafluoride that the other species survived (5 ppm for 1 h). Although this sensitivity would be expected for a corrosive and severely irritating chemical, a major limitation of the study is that it tested a small number of animals.
4.4.2. Susceptible Populations
The effects of tellurium hexafluoride might be exacerbated in individuals with impaired pulmonary function due to the chemical’s irritant properties (NIOSH 1978). However, no information on the susceptibility of such individuals to tellurium hexafluoride relative to normal individuals was found.
Mortality data on tellurium hexafluoride suggest a steep concentration-response curve, which implies little intraspecies variability. Mortality was 100% in rabbits, guinea pigs, rats, and mice exposed to tellurium hexafluoride at 5 ppm or higher for 4 h. All mice exposed at 5 ppm for 1 h died, but survived exposure at 1 ppm for 1 h (Kimmerle 1960).
4.4.3. Concentration-Exposure Duration Relationship
The concentration-exposure duration relationship for many irritant and systemically acting vapors and gases may be described by the equation Cn × t = k, where the exponent n ranges from 0.8 to 3.5 (ten Berge et al. 1986). In the absence of data on tellurium hexafluoride from which to derive an empirical value for n, temporal scaling was performed using default values of n = 3 when extrapolating to shorter durations and n = 1 when extrapolating to longer durations (NRC 2001).
4.4.4. Concurrent Exposure Issues
No concurrent exposure issues relevant to tellurium hexafluoride were found.
5. DATA ANALYSIS FOR AEGL-1
5.1. Human Data Relevant to AEGL-1
No human data relevant to developing AEGL-1 values for tellurium hexafluoride were identified.
5.2. Animal Data Relevant to AEGL-1
No animal data relevant to developing AEGL-1 values for tellurium hexafluoride were identified.
5.3. Derivation of AEGL-1 Values
AEGL-1 values are not recommended for tellurium hexafluoride because of insufficient data. Absence of AEGL-1 values does not imply that exposures below the AEGL-2 concentrations are without adverse effects.
6. DATA ANALYSIS FOR AEGL-2
6.1. Human Data Relevant to AEGL-2
No human data relevant to developing AEGL-2 values for tellurium hexafluoride were identified.
6.2. Animal Data Relevant to AEGL-2
Kimmerle (1960) exposed one rabbit, one guinea pig, two male white rats, and four male white mice per group to tellurium hexafluoride at 1 or 5 ppm for 1 or 4 h. Hyperpnea was observed in all animals exposed at 1 ppm for 1 h. At 5 ppm for 1 h, severe damage in the respiratory organs of the animals was found; the rabbit, guinea pigs, and rats survived the exposure but recovered very slowly. All of the mice died. Exposure to tellurium hexafluoride at 1 ppm for 4 h caused respiratory dysfunction in all animals.
In a study of the chemical tellurium, morphologic alterations of the brain were found in the fetuses of rats fed tellurium (Duckett 1970). Extrapolating the results of this study to derive AEGL values for tellurium hexafluoride was considered inappropriate.
6.3. Derivation of AEGL-2 Values
Data on tellurium hexafluoride are not consistent with AEGL-2 severity effects. Animals experienced hyperpnea after exposure to tellurium hexafluoride at 1 ppm for 1 h. The standing operating procedures for determining AEGL values specifies that AEGL-2 values for chemicals with steep concentrations-response curves may be estimated by dividing the AEGL-3 values by 3 (NRC 2001). Lethality data on tellurium hexafluoride demonstrates a steep-concentration response curve. All rabbits, guinea pigs, rats, and mice exposed at concentrations of 5, 10, 25, 50, or 100 ppm for 4 h died, and all mice exposed at 5 ppm for 1 h died. All animals exposed at 1 ppm for 1 or 4 h survived (Kimmerle 1960).
AEGL-2 values for tellurium hexafluoride are presented in Table 5-4, and the calculations are presented in Appendix A.
TABLE 5-4 AEGL-2 Values for Tellurium Hexafluoride
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 0.0097 ppm | 0.0067 ppm | 0.0053 ppm | 0.0033 ppm | 0.0017 ppm |
| (0.096 mg/m3) | (0.066 mg/m3) | (0.052 mg/m3) | (0.033 mg/m3) | (0.017 mg/m3) |
7. DATA ANALYSIS FOR AEGL-3
7.1. Human Data Relevant to AEGL-3
No human data relevant to deriving AEGL-3 values for tellurium hexafluoride were identified.
7.2. Animal Data Relevant to AEGL-3
Mortality was 100% in rabbits, guinea pigs, rats, and mice exposed for 4 h to tellurium hexafluoride at 5 ppm or higher (Kimmerle 1960); at 1 ppm, the animals survived but experienced pulmonary edema and respiratory dysfunction. For a 1-h exposure, all mice exposed at 5 ppm died; the other species survived that exposure and recovered slowly from severe damage to the respiratory organs. Animals exposed at 1 ppm for 1 h experienced hyperpnea.
7.3. Derivation of AEGL-3 Values
The highest concentration of tellurium hexafluoride causing no mortality in rabbits, guinea pigs, rats, and mice (1 ppm for 4 h) was used to derive AEGL-3 values (Kimmerle 1960). An interspecies uncertainty factor of 3 was applied because the four test species appeared to similarly sensitive to the acute effects of tellurium hexafluoride (Kimmerle 1960); however, that assessment is based on a small number of test animals (one to four per group). An intraspecies uncertainty factor of 3 was applied because tellurium hexafluoride is highly irritating and corrosive, and much of its toxicity is likely caused by a direct chemical effect on the tissue; that type of portal-of-entry effect is not expected to vary greatly among individuals. The steep concentration-response curve for tellurium hexafluoride implies little intraindividual variability. A modifying factor of 10 also was applied to account for the sparse database on tellurium hexafluoride and for the potential effects of tellurium. Thus, the total adjustment was 100. The concentration-exposure time relationship for many irritant and systemically-acting vapors and gases may be described by the equation Cn × t = k, where the exponent n ranges from 0.8 to 3.5 (ten Berge et al. 1986). Data on tellurium hexafluoride were inadequate for determining an empirical value for n, so default values of n = 3 when extrapolating to shorter durations (10, 30, and 60 min) and n = 1 when extrapolating to longer durations (8 h) were used. The AEGL-3 values for tellurium hexafluoride are presented in Table 5-5, and the calculations are presented in Appendix A.
TABLE 5-5 AEGL-3 Values for Tellurium Hexafluoride
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 0.029 ppm | 0.020 ppm | 0.016 ppm | 0.010 ppm | 0.0050 ppm |
| (0.28 mg/m3) | (0.20 mg/m3) | (0.16 mg/m3) | (0.10 mg/m3) | (0.049 mg/m3) |
Because of the uncertainty associated with extrapolating a point-of-departure based on a 4-h exposure to a 10-min AEGL value, the 30-min AEGL-3 value is typically adopted as the 10-min value. For tellurium hexafluoride, however, this approach was not used and the 10-min value was calculated from the Kimmerle (1960) data. Several laboratory animal species were exposed to tellurium hexafluoride at 1 ppm for 1 h, and only hyperpnea, a nonlife-threatening end point, was observed in those animals.
8. SUMMARY OF AEGLS
8.1. AEGL Values and Toxicity End Points
AEGL values for tellurium hexafluoride are presented in Table 5-6. AEGL-1 values are not recommended because of insufficient data. Data were also insufficient for deriving AEGL-2 values. Because tellurium hexafluoride has been shown to have a steep concentration-response curve, AEGL-2 values were estimated by dividing the AEGL-3 values by 3. AEGL-3 values were based on the highest concentration of tellurium hexafluoride causing no deaths in laboratory animals (Kimmerle 1960).
8.2. Other Standards and Guidelines
AEGL values for tellurium hexafluoride are compared with other guidelines and standards for this chemical in Table 5-7. The time-weighted average exposure concentration for workers is 0.02 ppm (29 CFR Part 1910 [2006]; NIOSH 2011; ACGIH 2013). The American Conference of Governmental Industrial Hygienists established a threshold limit value – time-weighted average of 0.02 ppm (measured as tellurium) on the basis that tellurium hexafluoride is approximately 2.5 times as acutely toxic as ozone and to protect against respiratory effects (ACGIH 2001). The immediately dangerous to life or health value (NIOSH 1994) is based on the acute inhalation toxicity data from the studies by Kimmerle (1960).
TABLE 5-6 AEGL Values for Tellurium Hexafluoride
| Classification | 10 min | 30 min | 1 h | 4 h | 8 h |
| AEGL-1 | NRa | NRa | NRa | NRa | NRa |
| (nondisabling) | |||||
| AEGL-2 | 0.0097 ppm | 0.0067 ppm | 0.0053 ppm | 0.0033 ppm | 0.0017 ppm |
| (disabling) | (0.096 mg/m3) | (0.066 mg/m3) | (0.052 mg/m3) | (0.033 mg/m3) | (0.017 mg/m3) |
| AEGL-3 | 0.029 ppm | 0.020 ppm | 0.016 ppm | 0.010 ppm | 0.0050 ppm |
| (lethal) | (0.28 mg/m3) | (0.20 mg/m3) | (0.16 mg/m3) | (0.10 mg/m3) | (0.049 mg/m3) |
aNot recommended. Absence of an AEGL-1 value does not imply that exposure below the AEGL-2 concentration is without adverse effects.
TABLE 5-7 Standards and Guidelines for Tellurium Hexafluoride
| Exposure Duration | |||||
| Guideline | 10 min | 30 min | 1 h | 4 h | 8 h |
| AEGL-1 | NR | NR | NR | NR | NR |
| AEGL-2 | 0.0097 ppm | 0.0067 ppm | 0.0053 ppm | 0.0033 ppm | 0.0017 ppm |
| AEGL-3 | 0.029 ppm | 0.020 ppm | 0.016 ppm | 0.010 ppm | 0.0050 ppm |
| IDLH (NIOSH)a | – | 1 ppm | – | – | – |
| TLV-TWA | – | – | – | – | 0.02 ppm as Te |
| (ACGIH)b | |||||
| PEL-TWA | – | – | – | – | 0.02 ppm as Te |
| (OSHA)c | |||||
| REL-TWA (NIOSH)d | – | – | – | – | 0.02 ppm as Te |
| MAC (The Netherlands)e | – | – | – | – | 0.02 ppm |
aIDLH (immediately dangerous to life or health, National Institute for Occupational Safety and Health) (NIOSH 1994) represents the maximum concentration from which one could escape within 30 min without any escape-impairing symptoms or any irreversible health effects.
bTLV-TWA (threshold limit value – time-weighted average, American Conference of Governmental Industrial Hygienists) (ACGIH 2013) is the time-weighted average concentration for a normal 8-h workday and a 40-h work week, to which nearly all workers may be repeatedly exposed, day after day, without adverse effect.
cPEL-TWA (permissible exposure limit – time-weighted average, Occupational Health and Safety Administration) (29 CFR Part 1910 [2006]) is defined analogous to the ACGIH TLV-TWA, but is for exposures of no more than 10 h/day, 40 h/wk. dREL-TWA (recommended exposure limit – time-weighted average, National Institute for Occupational Safety and Health) (NIOSH 2011) is defined analogous to the ACGIH TLV-TWA.
eMAC (maximaal aanvaaarde concentratie [maximal accepted concentration – peak category]) (Dutch Expert Committee for Occupational Standards, The Hague, The Netherlands (MSZW 2007) is defined analogous to the to the ACGIH TLV-TWA.
8.3. Data Adequacy and Research
No quantitative human data on tellurium hexafluoride are available, and only a few animal studies have been conducted. A single study of the acute toxicity of tellurium hexafluoride in rabbits, guinea pigs, rats, and mice is available (Kimmerle 1960), but only a few animals were tested and some potentially relevant end points were not evaluated. For example, humans acutely or repeatedly exposed to tellurium compounds frequently report somnolence, but the Kimmerle (1960) study did not examine that end point. A few studies of the related chemicals tellurium and tellurium pulveratum (Duckett 1970; Lampert et al. 1970; Malczewska-Toth 2012) found morphologic alterations in fetuses and demyelination in weanling rats after oral exposure; it is unknown whether reproductive and developmental toxicity would also occur following acute inhala-
tion exposure to tellurium hexafluoride. In the moist respiratory tract, tellurium hexafluoride slowly breaks down into hydrogen fluoride and tellurium; however, the contribution of the hydrolysis products to tellurium hexafluoride toxicity is unknown. No mechanistic data are available for other potential end points, including neurotoxicity and reproductive and developmental toxicity. Additional acute inhalation toxicity studies would help strengthen the basis of the AEGL values.
9. REFERENCES
ACGIH (American Conference of Governmental Industrial Hygienists). 2001. Documentation of the Threshold Limit Values and Biological Exposure Indices: Tellurium Hexafluoride. American Conference of Governmental Industrial Hygienists, Inc. Cincinnati, OH.
ACGIH (American Conference of Governmental Industrial Hygienists). 2013. TLVs and BEIs: Threshold Limit Values for Chemical and Physical Agents and Biological Exposure Indices. American Conference of Governmental Industrial Hygienists, Inc., Cincinnati, OH.
Amdur, M.I. 1958. Tellurium oxide: An animal study in acute toxicity. AMA Arch. Ind. Health 17(6):665-667.
Anthony, D.C., T.J. Montine, W.M. Valentine, and D.G. Graham. 2001. Toxic responses of the nervous system. P. 535-563 in Casarett & Doull’s Toxicology: The Basic Science of Poisons, 6th Ed., C.D. Klaassen, ed. New York: McGraw-Hill.
Blackadder, E.S., and W.G. Manderson. 1975. Occupational absorption of tellurium: A report of two cases. Br. J. Ind. Med. 32(1):59-61.
Duckett, S. 1970. Fetal encephalopathy following ingestion of tellurium. Experientia 26(11):1239-1241.
HSDB (Hazardous Substances Data Bank). 2008. Tellurium Hexafluoride (CAS Reg. No. 7783-80-4). TOXNET, Specialized Information Services, U.S. National Library of Medicine, Bethesda, MD [online]. Available: http://toxnet.nlm.nih.gov/cgi-bin/sis/htmlgen?HSDB [accessed Sept. 10, 2013].
IPCS (International Programme on Chemical Safety). 1998. Tellurium Hexafluoride. UKPID Monograph [online] Available: http://www.inchem.org/documents/ukpids/ukpids/ukpid83.htm [accessed December 9, 2014].
Kimmerle, G. 1960. Comparative studies on the inhalation toxicity of sulfur-, selenium-, and tellurium-hexafluoride [in German]. Arch. Toxikol. 18:140-144.
Lampert, P., F. Garro, and A. Pentschew. 1970. Tellurium neuropathy. Acta Neuropathol. 15(4):308-317.
Malczewska-Toth, B. 2012. Phosphorus, selenium, tellurium, and sulfur. Pp. 841-884 in Patty’s Toxicology, 6th Ed., P.E. Bingham, and B. Cohrssen, eds. Wiley Online Library.
MSZW (Ministerie van Sociale Zaken en Werkgelegenheid). 2007. OEL Database: Tellurium hexafluoride (as Te). The Social and Economic Council of the Netherlands, The Hague [online]. Available: http://www.ser.nl/en/grenswaarden/telluriumhexafluoride%20%20als%20te.aspx [accessed Dec. 9, 2014].
NIOSH (National Institute for Occupational Safety and Health). 1978. Occupational Health Guideline for Tellurium Hexafluoride. National Institute for Occupational Safety and Health, Centers for Disease Control and Prevention, Atlanta, GA. September 1978
[online]. Available: http://www.cdc.gov/niosh/docs/81-123/pdfs/0588.pdf [accessed Sept. 20, 2013].
NIOSH (National Institute for Occupational Safety and Health). 1994. Documentation for Immediately Dangerous to Life and Health Concentrations (IDLHs): Tellurim hexafluoride (as Te) [online]. Available: http://www.cdc.gov/niosh/idlh/7783804.html [accessed Sept. 20, 2013].
NIOSH (National Institute for Occupational Safety and Health). 2011. NIOSH Pocket Guide to Chemical Hazards: Tellurium hexafluoride [online]. Available: http://www.cdc.gov/niosh/npg/npgd0588.html [accessed Sept. 10, 2013].
NRC (National Research Council). 1993. Guidelines for Developing Community Emergency Exposure Levels for Hazardous Substances. Washington, DC: National Academy Press.
NRC (National Research Council). 2001. Standing Operating Procedures for Developing Acute Exposure Guideline Levels for Hazardous Chemicals. Washington, DC: National Academy Press.
NRC (National Research Council). 2004a. Hydrogen fluoride. Pp. 123-197 in Acute Exposure Guideline Levels for Selected Airborne Chemicals, Vol. 4. Washington, DC: The National Academies Press.
NRC (National Research Council). 2004b. Uranium hexafluoride. Pp. 250-291 in Acute Exposure Guideline Levels for Selected Airborne Chemicals, Vol. 4. Washington, DC: The National Academies Press.
OSHA (Occupational Safety and Health Administration). 1996. Occupational Safety and Health Guideline for Tellurium Hexafluoride [online]. Available: http://www.osha.gov/SLTC/healthguidelines/telluriumhexafluoride/recognition.html [accessed Feb. 4, 2009].
Stavert, D.M., D,C. Archuleta, M.J. Behr, and B.E. Lehner. 1991. Relative acute toxicities of hydrogen fluoride, hydrogen chloride, and hydrogen bromide in nose- and pseudo-mouth-breathing rats. Fundam. Appl. Toxicol. 16(4):636-655.
Steinberg, H.H., S.C. Massari, A.C. Miner, and R. Rink. 1942. Industrial exposure to tellurium: Atmospheric studies and clinical evaluation. J. Ind. Hyg. Toxicol. 24:183-192.
ten Berge, W.F., A. Zwart, and L.M. Appelman. 1986. Concentration-time mortality response relationship of irritant and systemically acting vapours and gases. J. Hazard. Mater. 13(3):301-309.
APPENDIX A
DERIVATION OF AEGL VALUES
Derivation of AEGL-1 Values
AEGL-1 values are not recommended because of insufficient data. Absence of an AEGL-1 value does not imply that exposure below the AEGL-2 concentration is without adverse effects.
| Derivation of AEGL-2 Values | |
| Key study: | Kimmerle, G. 1960. Comparative studies on the inhalation toxicity of sulfur-, selenium-, and telluriumhexafluoride [in German]. Arch. Toxikol. 18:140-144. |
| Toxicity end points: | Data were inadequate for deriving AEGL-2 values. However, the standing operating procedures for determining AEGL values specifies that AEGL-2 values for chemicals with steep concentrations-response curves may be estimated by dividing the AEGL-3 values by 3 (NRC 2001). Lethality data on tellurium hexafluoride demonstrates a steep-concentration response curve. All rabbits, guinea pigs, rats, and mice exposed at concentrations of 5, 10, 25, 50, or 100 ppm for 4 h died, and all mice exposed at 5 ppm for 1 h died. All animals exposed at 1 ppm for 1 or 4 h survived (Kimmerle 1960). |
| Calculations: | |
| 10-min AEGL-2: | 0.029 ppm ÷ 3 = 0.0097 ppm |
| 30-min AEGL-2: | 0.020 ppm ÷ 3 = 0.0067 ppm |
| 1-h AEGL-2: | 0.016 ppm÷ 3 = 0.0053 ppm |
| 4-h AEGL-2: | 0.010 ppm÷ 3 = 0.0033 ppm |
| 8-h AEGL-2: | 0.005 ppm÷ 3 = 0.0017 ppm |
| Derivation of AEGL-3 Values | |
| Key studies: | Kimmerle, G. 1960. Comparative studies on the inhalation toxicity of sulfur-, selenium-, and telluriumhexafluoride [in German]. Arch. Toxikol. 18:140-144. |
| Toxicity end point: | Highest concentration causing no mortality in the guinea pig, rabbit, rat, and mouse (1 ppm for 4 h) |
| Uncertainty factors: | Interspecies: 3, because the guinea pig, rabbit, rat, and mouse appear to be similarly sensitive to the acute effects of tellurium hexafluoride; however, this assessment is based on a small number of animals. |
| Intraspecies: 3, because tellurium hexafluoride is highly irritating and corrosive and much of the toxicity is likely caused by a direct chemical effect on the tissues; that type of portal-of-entry effect is not expected to vary greatly among individuals. | |
| Modifying factor: | 10, because of the sparse database on tellurium hexafluoride and the potential effects of tellurium |
| Time scaling: | Cn × t = k; default values of n = 3 for extrapolating to shorter durations and n = 1 for extrapolating to longer durations (NRC 2001) 1 ppm ÷ 100 (total uncertainty factor) = 0.01 ppm (0.01 ppm) 3 × 240 min = 0.00024 ppm-min (0.01 ppm)1 × 240 min = 2.4 ppm-min |
| 10-min AEGL-3: | C3 × 10 min = 0.00024 ppm-min |
| C = 0.029 ppm | |
| 30-min AEGL-3: | C3 × 30 min = 0.00024 ppm-min |
| C = 0.020 ppm | |
| 1-h AEGL-3: | C3 × 30 min = 0.00024 ppm-min |
| C = 0.016 ppm | |
| 4-h AEGL-3: | C1 × 240 min = 2.4 ppm-min |
| C = 0.010 ppm | |
| 8-h AEGL-3: | C1 × 480 min = 2.4 ppm-min |
| C = 0.0050 ppm | |
APPENDIX B
ACUTE EXPOSURE GUIDELINE LEVELS FOR TELLURIUM HEXAFLUORIDE
Derivation Summary
AEGL-1 VALUES
No AEGL-1 values were derived for tellurium hexafluoride because of insufficient data. Absence of AEGL-1 values does not imply that exposure below the AEGL concentrations are without adverse effects.
AEGL-2 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 0.0097 ppm | 0.0067 ppm | 0.0053 ppm | 0.0033 ppm | 0.0017 ppm |
| Data adequacy: Data on tellurium hexafluoride were inadequate for deriving AEGL-2 values. However, the standing operating procedures for determining AEGL values specify that AEGL-2 values for chemicals with steep concentrations-response curves may be estimated by dividing the AEGL-3 values by 3 (NRC 2001). Lethality data on tellurium hexafluoride indicate a steep-concentration response curve. All rabbits, guinea pigs, rats, and mice exposed at concentrations of 5, 10, 25, 50, or 100 ppm for 4 h died, and all mice exposed at 5 ppm for 1 h died. All animals exposed at 1 ppm for 1 or 4 h survived (Kimmerle 1960). | ||||
AEGL-3 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 0.029 ppm | 0.020 ppm | 0.016 ppm | 0.010 ppm | 0.0050 ppm |
| Key reference: Kimmerle, G. 1960. Comparative study of the inhalation toxicity of sulfur, selenium, and tellurium hexafluorides [in German] Arch. Toxikol. 18:140-144. | ||||
| Test species/Strain/Number: Unspecified strains of rabbits (n = 1), guinea pigs (n = 1), rats (n = 2), and mice (n = 4) | ||||
| Exposure route/Concentrations/Durations: Inhalation ; 1, 5, 10, 25, 50, 100 ppm for 4 h | ||||
| Effects: | ||||
| 1 ppm: respiratory dysfunction, pulmonary edema | ||||
| 5 ppm: death after 4-24 h | ||||
| 10 ppm: death after 100-140 min | ||||
| 25 ppm: death after 60-110 min | ||||
| 50 ppm: death after 45-70 min | ||||
| 100 ppm: death after 10-30 min | ||||
| End point/Concentration/Rationale: Highest concentration causing no mortality 1 f 4 h | ||||
| Uncertainty factors/Rationale: | ||||
| Total uncertainty factor: 10 | ||||
| Interspecies: 3, because the guinea pig, rabbit, rat, and mouse appear to be similarly sensitive to the acute effects of tellurium hexafluoride; however, this assessment is based on a small number of animals. | ||||
| Intraspecies: 3, because tellurium hexafluoride is highly irritating and corrosive and much of the toxicity is likely caused by a direct chemical effect on the tissues; that type of portal-of-entry effect is not expected to vary greatly among individuals. | ||||
| Modifying factor: 10, because of the sparse database on tellurium hexafluoride and to account for potential effects of tellurium | ||||
| Animal-to-human dosimetric adjustment: None | ||||
| Time scaling: Cn × t = k; default values of n = 3 for extrapolating to shorter durations and n = 1 for extrapolating to longer durations (NRC 2001). | ||||
| Data adequacy: Tellurium hexafluoride has a sparse database consisting of one lethality study in laboratory animals. | ||||
APPENDIX C
CATEGORY PLOT FOR TELLURIUM HEXAFLUORIDE
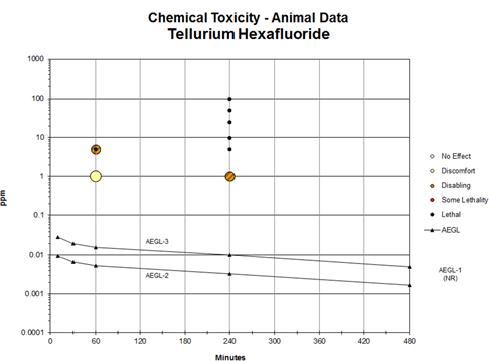
FIGURE C-1 Category plot of toxicity data and AEGL values for tellurium hexafluoride.
TABLE D-1 Data Used in the Category Plot for Tellurium Hexafluoride
| Source | Species | Sex | No. of Exposures | ppm | Minutes | Category | Comments |
| AEGL-2 | 0.0097 | 10 | AEGL | ||||
| AEGL-2 | 0.0067 | 30 | AEGL | ||||
| AEGL-2 | 0.0053 | 60 | AEGL | ||||
| AEGL-2 | 0.0033 | 240 | AEGL | ||||
| AEGL-2 | 0.0017 | 480 | AEGL | ||||
| AEGL-3 | 0.029 | 10 | AEGL | ||||
| AEGL-3 | 0.020 | 30 | AEGL | ||||
| AEGL-3 | 0.016 | 60 | AEGL | ||||
| AEGL-3 | 0.010 | 240 | AEGL | ||||
| AEGL-3 | 0.0050 | 480 | AEGL | ||||
| Kimmerle 1960 | Rabbit | M | 1 | 1 | 240 | 2 | Respiratory dysfunction, pulmonary edema |
| Kimmerle 1960 | Rabbit | M | 1 | 5 | 240 | 3 | Death after 8 h |
| Kimmerle 1960 | Rabbit | M | 1 | 10 | 240 | 3 | Death after 140 min |
| Kimmerle 1960 | Rabbit | M | 1 | 25 | 240 | 3 | Death after 80 min |
| Kimmerle 1960 | Rabbit | M | 1 | 50 | 240 | 3 | Death after 60 min |
| Kimmerle 1960 | Rabbit | M | 1 | 100 | 240 | 3 | Death after 15 min |
| Kimmerle 1960 | Rabbit | M | 1 | 1 | 60 | 1 | Hyperpnea |
| Kimmerle 1960 | Rabbit | M | 1 | 5 | 60 | 2 | Severe damage to respiratory organs |
| Kimmerle 1960 | Guinea pig | M | 1 | 1 | 240 | 2 | Respiratory dysfunction, pulmonary edema |
| Kimmerle 1960 | Guinea pig | M | 1 | 5 | 240 | 3 | Death after 8 h |
| Kimmerle 1960 | Guinea pig | M | 1 | 10 | 240 | 3 | Death after 140 min |
| Source | Species | Sex | No. of Exposures | ppm | Minutes | Category | Comments |
| Kimmerle 1960 | Guinea pig | M | 1 | 25 | 240 | 3 | Death after 80 min |
| Kimmerle 1960 | Guinea pig | M | 1 | 50 | 240 | 3 | Death after 60 min |
| Kimmerle 1960 | Guinea pig | M | 1 | 100 | 240 | 3 | Death after 15 min |
| Kimmerle 1960 | Guinea pig | M | 1 | 1 | 60 | 1 | Hyperpnea |
| Kimmerle 1960 | Guinea pig | M | 1 | 5 | 60 | 2 | Severe damage to respiratory organs |
| Kimmerle 1960 | Rat | M | 1 | 1 | 240 | 2 | Respiratory dysfunction, pulmonary edema |
| Kimmerle 1960 | Rat | M | 1 | 5 | 240 | 3 | Death after 8 h |
| Kimmerle 1960 | Rat | M | 1 | 10 | 240 | 3 | Death after 140 min |
| Kimmerle 1960 | Rat | M | 1 | 25 | 240 | 3 | Death after 80 min |
| Kimmerle 1960 | Rat | M | 1 | 50 | 240 | 3 | Death after 60 min |
| Kimmerle 1960 | Rat | M | 1 | 100 | 240 | 3 | Death after 15 min |
| Kimmerle 1960 | Rat | M | 1 | 1 | 60 | 1 | Hyperpnea |
| Kimmerle 1960 | Rat | M | 1 | 5 | 60 | 2 | Severe damage to respiratory organs |
| Kimmerle 1960 | Mouse | M | 1 | 1 | 240 | 2 | Respiratory dysfunction, pulmonary edema |
| Kimmerle 1960 | Mouse | M | 1 | 5 | 240 | 3 | Death after 8 h |
| Kimmerle 1960 | Mouse | M | 1 | 10 | 240 | 3 | Death after 140 min |
| Kimmerle 1960 | Mouse | M | 1 | 25 | 240 | 3 | Death after 80 min |
| Kimmerle 1960 | Mouse | M | 1 | 50 | 240 | 3 | Death after 60 min |
| Kimmerle 1960 | Mouse | M | 1 | 100 | 240 | 3 | Death abate 15 min |
| Kimmerle 1960 | Mouse | M | 1 | 1 | 60 | 1 | Hyperpnea |
| Kimmerle 1960 | Mouse | M | 1 | 5 | 60 | 3 | Death between 24-36 h |
For category: 0 = no effect, 1 = discomfort, 2 = disabling, SL = some lethality, 3 = lethality.























