Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 15 (2013)
Chapter: 6 Methyl Isothiocyanate Acute Exposure Guideline Levels
6
Methyl Isothiocyanate1
Acute Exposure Guideline Levels
PREFACE
Under the authority of the Federal Advisory Committee Act (FACA) P.L. 92-463 of 1972, the National Advisory Committee for Acute Exposure Guideline Levels for Hazardous Substances (NAC/AEGL Committee) has been established to identify, review, and interpret relevant toxicologic and other scientific data and develop AEGLs for high-priority, acutely toxic chemicals.
AEGLs represent threshold exposure limits for the general public and are applicable to emergency exposure periods ranging from 10 minutes (min) to 8 hours (h). Three levels—AEGL-1, AEGL-2, and AEGL-3—are developed for each of five exposure periods (10 and 30 min and 1, 4, and 8 h) and are distinguished by varying degrees of severity of toxic effects. The three AEGLs are defined as follows:
AEGL-1 is the airborne concentration (expressed as parts per million or milligrams per cubic meter [ppm or mg/m3]) of a substance above which it is predicted that the general population, including susceptible individuals, could experience notable discomfort, irritation, or certain asymptomatic, nonsensory effects. However, the effects are not disabling and are transient and reversible upon cessation of exposure.
____________________
1 This document was prepared by the AEGL Development Team composed of Robert Young (Oak Ridge National Laboratory), Heather Carlson-Lynch (SRC, Inc.), Chemical Manager Susan Ripple (National Advisory Committee [NAC] on Acute Exposure Guideline Levels for Hazardous Substances), and Ernest V. Falke (U.S. Environmental Protection Agency). The NAC reviewed and revised the document and AEGLs as deemed necessary. Both the document and the AEGL values were then reviewed by the National Research Council (NRC) Committee on Acute Exposure Guideline Levels. The NRC committee has concluded that the AEGLs developed in this document are scientifically valid conclusions based on the data reviewed by the NRC and are consistent with the NRC guidelines reports (NRC 1993, 2001).
AEGL-2 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience irreversible or other serious, long-lasting adverse health effects or an impaired ability to escape.
AEGL-3 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience life-threatening health effects or death.
Airborne concentrations below the AEGL-1 represent exposure concentrations that could produce mild and progressively increasing but transient and nondisabling odor, taste, and sensory irritation or certain asymptomatic, nonsensory effects. With increasing airborne concentrations above each AEGL, there is a progressive increase in the likelihood of occurrence and the severity of effects described for each corresponding AEGL. Although the AEGL values represent threshold concentrations for the general public, including susceptible subpopulations, such as infants, children, the elderly, persons with asthma, and those with other illnesses, it is recognized that individuals, subject to idiosyncratic responses, could experience the effects described at concentrations below the corresponding AEGL.
SUMMARY
Methyl isothiocyanate (MITC) is a colorless crystalline solid that occurs primarily as a decomposition product of pesticides applied as soil fumigants. MITC injected into soil rapidly vaporizes. A level of distinct odor awareness (LOA) of 27 ppm for MITC was calculated.
The database for MITC includes a controlled human clinical study that evaluated odor threshold and ocular irritation, acute and repeated-exposure inhalation studies in rats, and oral studies of reproductive and developmental toxicity in rats and rabbits. MITC is a potent, direct-acting irritant to the eyes and respiratory tract. Death results from acute pulmonary congestion and hemorrhage. Developmental studies indicate that MITC was not teratogenic but caused delayed growth at maternally toxic concentrations. Although MITC is an alkylating agent, most genotoxicity studies reported negative results. Carcinogenicity studies of MITC administered orally to rats and mice did not find a significant neoplastic response.
AEGL-1 values are based on a study of human volunteers (Russell and Rush 1996). This study met the criteria for using data on human subjects outlined in the Standing Operating Procedures for AEGLs (NRC 2001, Section 2.3.2). Slight and transient ocular irritation was reported by subjects exposed to MITC at a concentration of 0.8 ppm. Blinking rate was slightly increased, but there was no tearing or redness of the eye. Thus, 0.8 ppm was considered the highest concentration without notable discomfort and was used as the point of departure for deriving AEGL-1 values. An intraspecies uncertainty factor of 3 was applied, because MITC appears to have a direct-acting irritant mechanism
of toxicity and metabolic and physiologic differences are unlikely to play a major role (NRC 2001). Also, the range of human sensitivity to ocular irritants is approximately two-fold (Kjaergaard et al. 1992). Because 0.8 ppm was tested for up to 4 h, that concentration was used for all the AEGL-1 exposure durations. Furthermore, there is adaptation to the slight irritation that defines the AEGL-1. The AEGL-1 values are supported by no-effect concentrations in repeated-exposure studies with rodents (Rosskamp et al. 1978; Klimisch 1987).
No acute clinical studies or acute toxicology studies in laboratory animals were identified that were relevant deriving AEGL-2 values. The degree of ocular irritation observed in the study by Russell and Rush (1996) was not of sufficient severity to impair escape. In the absence of data that address AEGL-2 end points, the AEGL-3 for MITC values were divided by 3 to derive the respective AEGL-2 values. This approach is appropriate for chemicals with evidence of a steep concentration-response curve (NRC 2001).
The point of departure for AEGL-3 values was the highest nonlethal concentration of 94 ppm in a study of rats exposed for 4 h (Jackson et al. 1981). Interspecies and intraspecies uncertainty factors of 3 each are generally applied to chemicals that are direct-acting irritants (NRC 2001). However, such an approach in this instance would result in values inconsistent with the human volunteer study by Russell and Rush (1996). Therefore, interspecies and intraspecies uncertainty factors of 1 and 3, respectively, were applied. Time-scaling was performed using the equation Cn × t = k, with default values of n = 3 for extrapolation to shorter durations and n = 1 for extrapolating to longer durations (NRC 2001). The 10-min AEGL-3 value was set equal to the 30-min AEGL-3 value because of uncertainties associated with extrapolating a 4-h point of departure to a 10-min value.
AEGL values for MITC are presented in Table 6-1.
1. INTRODUCTION
MITC is a colorless crystalline solid that is used as a soil fumigant (Lam et al. 1993; HSDB 2012). It is produced by the action of carbon disulfide on methylamine or by reacting sodium methyldithiocarbamate with ethyl chlorocarbonate (HSDB 2012). Metam sodium (sodium N-methyldithiocarbamate), which decomposes to MITC, is the third most commonly used agricultural pesticide in the United States (Pruett et al. 2001); however, production data were not located. MITC injected into soil immediately vaporizes (Nihon Schering 1990).
Metam sodium and dazomet are propesticides (compounds that are converted to pesticides) which hydrolyze in soil to MITC as the ultimate toxicant (Lam et al. 1993). Upon dilution with water, metam sodium decomposes to MITC which evolves as a gas of hydrogen sulfide and lesser amounts of methylamine and carbon disulfide. Ditrapex® and Trapex® contain 20% MITC (Nihon Schering 1990). Ditrapex® contains 40% 1,2-dichloropropene, a nematicide. MITC is phytotoxic and planting is delayed until the soil fumigant has decomposed completely.
TABLE 6-1 AEGL Values for Methyl Isothiocyanatea
| Classification | 10 min | 30 min | 1 h | 4 h | 8 h | End Point (Reference) |
| AEGL-1 (nondisabling) | 0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
No evidence of notable discomfort (ocular irritation) at several time points in humans (Russell and Rush 1996) |
| AEGL-2 (disabling) | 21 ppm (63 mg/m3) |
21 ppm (63 mg/m3) |
17 ppm (51 mg/m3) |
10 ppm (30 mg/m3) |
5.3 ppm (16 mg/m3) |
One-third of AEGL-3 values |
| AEGL-3 (lethal) | 63 ppm (190 mg/m3) |
63 ppm (190 mg/m3) |
50 ppm (150 mg/m3) |
31 ppm (94 mg/m3) |
16 ppm (47 mg/m3) |
Nonlethal concentration in rats (Jackson et al. 1981) |
aA level of distinct odor awareness (LOA) of 27 ppm was calculated for MITC (see Appendix A). The LOA is defined as the concentration above which it is predicted that more than half of the exposed population will experience at least a distinct odor intensity, and about 10% of the population will experience strong odor intensity. Calculation of the LOA does not imply that exposure below the LOA is without effects.
MITC belongs to the chemical class mustard oils. It has been considered as a possible military poison (Verschueren 2001). The toxicity of MITC was reviewed by Nihon Schering (1990), NRA (1997), and Rubin et al. (2003). MITC has a pungent, horseradish-like odor at room temperature; its vapors irritate mucous membranes and it is a potent lacrimator (Nihon Schering 1990).
The chemical and physical properties of MITC are presented in Table 6-2.
2. HUMAN TOXICITY DATA
2.1. Odor Threshold and Odor Awareness
MITC has a pungent horseradish-like odor at room temperature (Nihon Schering 1990). Odor thresholds reportedly range from approximately 0.1 ppm (Nesterova 1969) to 5 ppm (Verschueren 2001). The odor threshold determined in a controlled clinical study was 1.7 ppm (Russell and Rush 1996; EPA 2006a). Using the data of Russell and Rush (1996), a level of distinct odor awareness (LOA) of 27 ppm was calculated for MITC (see Appendix A). The LOA represents the concentration above which it is predicted that more than half of the exposed population will experience at least a distinct odor intensity, and about 10% of the population will experience a strong odor intensity.
The propesticide metam sodium hydrolyzes into MITC and hydrogen sulfide, and the odor of hydrogen sulfide might be present at metam sodium application sites.
TABLE 6-2 Chemical and Physical Properties of Methyl Isothiocyanate
|
Parameter |
Value |
|
Synonyms |
Isothiocyanatomethane; methyl mustard oil; Trapex |
|
CAS registry no. |
556-61-6 |
|
Chemical formula |
C2H3NS |
|
Molecular weight |
73.12 |
|
Physical state |
Colorless crystals |
|
Melting point |
36°C |
|
Boiling point |
119°C |
|
Density/specific gravity (water =1) |
1.0691 at 37°C |
|
Solubility in water |
7.6 g/L at 25°C |
|
Vapor density (air =1) |
2.53 |
|
Vapor pressure |
3.54 mm Hg at 25°C |
|
Saturated vapor concentration (calculated from vapor pressure) |
~27,000 ppm (~82,000 mg/m3) |
|
Flammability limits |
Lower 2.5%; Upper 30%/MITC-Fume |
|
Conversion factors (calculated) |
1 ppm = 2.99 mg/m3 |
Source: HSDB 2012.
2.2. Accidents and Community Exposures
In 1991, a railroad tank car in California derailed and spilled 19,000 gallons of metam sodium into the Sacramento River (Alexeeff et al. 1994). The hydrolysis product MITC was released to the air. Many individuals downriver of the incident reported odors. Over 240 individuals complained of ocular and throat irritation, dizziness, and shortness of breath. Ambient air concentrations, measured on the fourth day after the accident (12-h integrated samples) ranged from 0.2 to 37 ppb. Average concentrations reported on the fifth through tenth day ranged from below the limit of detection (<1 ppb) to 2.6 ppb. Estimates of peak concentrations during the first two days were 140-1,600 ppb for exposures of a few minutes to 1 h; these estimates were for areas within 500 meters of the river.
Cone et al. (1994) assessed the occurrence of persistent respiratory disorders among adults exposed as a result of the metam sodium spill. Exposures were most likely to a mixture of metam sodium hydrolysis products which include MITC, hydrogen sulfide gas, methylamine, and carbon disulfide. Among a group of 197 persons referred for health evaluation, 20 were identified as having persistent irritant-induced asthma and 10 were identified as having persistent exacerbation of pre-existing asthma. Cases of irritant-induced asthma met the
following criteria: onset of upper respiratory symptoms within 24 h of exposure; onset of lower respiratory symptoms within 1 week and persisting for more than 3 months; no prior history of respiratory illness (confirmed by medical records); location and activity consistent with exposure during and for 1 week after the spill; and nonspecific airway hyper-responsiveness demonstrated by methacholine challenge. Most (17/20) of the irritant-induced asthma cases met the criteria for reactive airway distress syndrome (RADS).
O’Malley et al. (2004) documented illnesses among residents of a community near a potato field where metam sodium was applied with a sprinkler. Air concentrations of MITC were estimated with air models using application information and meteorological data. Concentrations were estimated to range from 0.5 ppm to just over 1 ppm (1-h time-weighted averages). Peak concentrations at 1 and 3 min were estimated to be 4 and 7 ppm, respectively. No data were reported regarding concentrations of other hydrolysis products of metam sodium, which includes a mixture of known irritants. Residents were interviewed to obtain information on symptoms and proximity to the potato field during pesticide application. Among those closest to the application site (≤0.5 miles), symptoms consisted of irritation of the eyes or upper respiratory tract (burning of eyes, nose, or throat) in 51/135, non-specific systemic symptoms such as headache, nausea, diarrhea, abdominal pain, or malaise in 22/135, “systemic irritant” response (not otherwise specified) in 45/135, and respiratory irritation (but not asthma or lower respiratory irritation) in 6/135. Frequency of complaints decreased with distance from the application site.
O’Malley et al. (2005) reported illnesses related to soil incorporation (shank application) of metam sodium near a rural community in California. Several hours after application of 25,000 pounds of metam sodium to a 100-acre field, 250 nearby residents experienced ocular and upper-respiratory irritation, non-specific systemic symptoms, and lower-respiratory-tract complaints. Some residents sought medical treatment. After the incident, residents were interviewed directly or via medical records in order to correlate symptoms with area and activity. The most serious illnesses were associated with individuals who had pre-existing lung diseases, such as asthma and emphysema. MITC concentrations were estimated based on field treatment, projected emissions, and weather conditions. Modeling results indicted 1-h MITC concentrations in the affected areas of 0.8-1.0 ppm, with peak concentrations between 2.4 and 3.2 ppm.
Bretaudeau Deguigne et al. (2011) described a series of 106 case reports of exposure to metam sodium at a poison control center in France. Most (96) cases were accidentally exposed via inhalation, and the most commonly reported symptoms attributed to MITC exposure were irritation of the eyes (76/96 inhalation exposures) and throat and nose (65/96). Exposure concentrations were not reported. Of the 96 exposed, only four had cough or dyspnea; the investigator reported that there were no cases of persistent irritant-induced asthma or exacerbation of asthma.
2.3. Clinical Studies
In order to determine the thresholds for odor detection and ocular irritation, healthy adult volunteers were exposed to measured concentrations of MITC in a laboratory setting (Russell and Rush 19962; reviewed in EPA 2006a). This study met the criteria for use of data from human subjects discussed in the Standing Operating Procedures for AEGLs (NRC 2001, Section 2.3.2). In the olfactory threshold study, 33 individuals (16 males, 17 females; age range of 1834 years) were exposed to three reference control odorants: pyridine, acetic acid, and n-butyl alcohol, as well as MITC. The odorants were dispensed in a controlled double-blind fashion through one of three presentation ports. A technician chose the odorant and the subject was responsible for determining from which port the odorant was dispersed. There was a 30-sec rest period between odorant presentations. Each volunteer was tested over a range of concentrations for each odorant until a threshold, determined under a standard procedure, was satisfactorily ascertained. The observed odor threshold for MITC ranged from 0.2 to 8 ppm, with a geometric mean of 1.7 ppm.
The ocular irritation study was conducted with 70 adult volunteers (38 males, 32 females; age range of 18-67 years). Four exposure durations of 1 min, 14 min, 4 h, and 8 h were used. In the 1-min trial, subjects were exposed to MITC at concentrations up to 3.3 ppm. In the 14-min trial, 9-10 subjects were exposed at 0 (air only), 0.6, 1.9, or 3.3 ppm. In the 4-h trial, there was both an air and acetic acid control; concentration of MITC tested were 0.23 ppm (12 subjects) or 0.8 ppm (9 subjects). In the 8-h study, 12 subjects were exposed to air only, seven were exposed to acetic acid, and 16 subjects were exposed to MITC at 0.22 ppm. Subjects were permitted two 15-min rest periods and a lunch break during the 8-h study. An olfactometer was used to dispense the test materials through a manifold system. A total hydrocarbon analyzer was used to monitor the flow of test material through the tubing; samples were collected on carbon tube samplers, desorbed, and measured with gas chromatography. The subjects were exposed via goggles. No additional details of the methods were available.
Subjective irritation was measured on a Likert scale in which irritation was rated from no irritation to a feeling the subject would like to end the exposure; the mid-point was described as similar to that of cutting a single mild onion. Subjective irritation, blink rates, and tearing were assessed at several time points. Baseline responses were determined pre-exposure and from exposure to the air control. Visual acuity and ocular morphology were assessed at the beginning and end of each exposure (methods not provided). The results of this study are summarized in Table 6-3. The no-observed-effect levels (NOELs) for the 1-
____________________
2 Russell and Rush (1996) is an unpublished report prepared by the University of California and Western Research Center and submitted to U.S. EPA’s Office of Chemical Safety and Pollution Prevention. The report is not publicly available. A U.S. EPA AEGL staff member with FIFRA clearance reviewed the original report and confirmed the details provided in the U.S. EPA (2006a) Data Evaluation Record for the study.
and 14-min exposures were 3.3 and 0.6, respectively. The NOEL range for the two longer duration exposures was 0.22-0.23 ppm. The lowest-observed-effect level (LOELs) for the 14-min and 1- through 8-h intervals were 1.9 and 0.8 ppm, respectively. During the first hour of the 4-h trial at 0.8 ppm, subjective irritation was rated at 25 ± 14% on a scale of 1 to 100. During the second hour of the 4-h trial at 0.8 ppm, blink rates increased from 3 ± 9/min in the control group to 16 ± 11/min in the 0.8-ppm group. Thereafter, blink rate did not increase with exposure duration. No statistically significant positive tearing responses were observed; photographs of the participant’s eyes failed to show notable, exposure-related changes. Comments from the subjects indicated that recovery began immediately after removal of the goggles and was complete within 20 min at the highest concentration.
2.4. Community Exposures
Lee et al. (2002) reported ambient concentrations of MITC (and other pesticides or pesticide breakdown products) in California during months associated with pesticide application. With 2 weeks of air monitoring data, generally collected from samplers placed atop the roofs of community building, mean concentrations of MITC were 2.1 μg/m3 (0.7 ppb) in urban communities (range not reported) and 4.9 μg/m3 (1.6 ppb) in rural communities (range up to 18 μg/m3 [6 ppb]). The 15-day maximum concentration was 8.4 μg/m3 (2.8 ppb). The publication reported little information on the areas sampled.
TABLE 6-3 Ocular Irritation in Human Subjects
| Exposure Duration | NOELa (ppm) | LOEL (ppm) | Description |
| 1 min | 3.3 | – | |
| 4 min | 0.60 | 1.9 | Subjective ocular irritationb |
| 14 min | 0.60 | 1.9 | Subjective ocular irritation |
| 1 h | 0.23 | 0.8 | Subjective ocular irritation |
| 1.5 h | 0.22 | – | |
| 2 h | 0.23 | 0.8 | Subjective ocular irritation, |
| increased blink rate. | |||
| 3 h | 0.23 | 0.8 | Subjective ocular irritation, |
| increased blink rate. | |||
| 3.5 h | 0.22 | – | |
| 4 h | 0.23 | 0.8 | Subjective ocular irritation |
| 6 h | 0.22 | – | |
| 8 h | 0 22 | – | |
aThe 0.22- and 0.23-ppm concentrations were tested on different days.
bOcular irritation did not include redness or tearing.
Source: Rubin et al. 2003.
Merriman and Hebert (2007) reported the results of an air monitoring study measuring MITC concentrations in the air of an agricultural region of Washington State during the fall season when metam sodium fumigation is typically done. Five residential sites and one commercial site were selected for monitoring. Twenty-four-hour samples comprised of two 12-h day and night subsamples were collected over the course of 1 month (September 26 to October 25, 2005); a total of 201 samples were collected. The frequency of measurements above the detection limit (0.01 ppb) was 199/201. The maximum 12-h time-weighted average concentration of MITC was 67 μg/m3 (22 ppb); the average over the 30-day sampling period was 10 μg/m3 (3.3 ppb).
2.5. Developmental and Reproductive Toxicity
No studies of developmental or reproductive toxicity of MITC in humans were found.
2.6. Genotoxicity
Negative results were obtained with MITC at concentrations of 3.0 or 5.0 μg/mL in an in vitro cytogenetic chromosome aberration test using cultured human lymphocytes (Rubin et al. 2003). MITC induced micronuclei and DNA strand breaks in cultured human hepatoma cells at concentrations that were cytotoxic (Kassie et al. 2001).
2.7. Chronic Toxicity and Carcinogenicity
No studies of the chronic toxicity or potential carcinogenicity of MITC in humans were found.
2.8. Summary
In studies with human volunteers, the odor threshold for MITC ranged from 0.2 to 8 ppm, with a geometric mean of 1.7 ppm (Russell and Rush 1996). Volunteers were exposed to referent odorants including n-butyl alcohol. In a study with 70 volunteers, ocular irritation was examined at discrete time intervals (Russell and Rush 1996). No ocular irritation was observed in association with MITC at 3.3 ppm (the highest concentration tested) for 1 min, 0.6 ppm for 14 min, or 0.22-0.23 ppm for 1-8 h. When exposed at 1.9 ppm for 14 min or at 0.8 ppm for 1-8 h, subjects reported ocular irritation slightly less than that associated with cutting a single mild onion. MITC failed to induce signs of genotoxicity in an in vitro test for chromosome damage in cultured human lymphocytes (Rubin et al. 2003), and induced strand breaks in human hepatoma cells at cytotoxic concentrations (Kassie et al. 2001).
No human studies addressing the potential for reproductive and developmental toxicity or chronic toxicity and carcinogenicity were found.
3. ANIMAL TOXICITY DATA
Data on MITC were reviewed by Nihon Schering (1990), Alexeeff et al. (1994), NRA (1997), HSDB (2012), and Rubin et al. (2003). MITC is a primary ocular irritant when instilled into the eye of rabbits (100 mg), causing severe inflammation with corneal opacity, iritis, and conjunctival swelling (Nihon Schering 1990). In studies with several species, cats were the most sensitive species, exhibiting irritation of the ocular mucosa (Nesterova 1969). No details of those studies were available.
3.1. Acute Lethality
Clark and Jackson (1977) exposed groups of five male and five female Sprague-Dawley CFY rats whole-body to five concentrations of MITC ranging from 600 to 3,100 mg/m3 (200 to 1,037 ppm) for 1 h. The test material administered in the study was a pesticide formulation that contains MITC as an active ingredient. The conversion used to estimate the MITC concentration in the chamber (the concentration reported by secondary sources) was not clearly explained in the original report. Hyperactivity that began within 5 min of exposure was observed in all MITC-treated groups. Ocular irritation, dyspnea, and hypoactivity were observed during the remainder of the exposure period. Most rats exposed at 3,100 mg/m3 (1,037 ppm) died; death was preceded by convulsions. No deaths occurred at 630 mg/m3 (210 ppm). The 1-h LC50 was 1,900 mg/m3 (635 ppm). Necropsy of animals that died revealed pulmonary congestion and hemorrhage of the lungs.
Jackson et al. (1981; reviewed in EPA 2006b)3 exposed groups of five male and five female Sprague-Dawley rats to six different concentrations of MITC for 4 h (concentrations not specified). The test material was MITC. Clinical signs noted during exposure included closure of the eyes, lacrimation, and peripheral vasodilation in all rats and a hunched posture in the majority of the animals. Peripheral vasodilation persisted for several hours after exposure. Opacity of the eyes was observed in rats exposed at ≥ mg/m3 (≥167 ppm). No mortality or gross pathologic changes were observed at 282 mg/m3 (94 ppm), but lung weight was increased and lung rales were observed on day 1 and reoccurred on day 6 post-exposure. The calculated 4-h LC50 was 180 ppm for
____________________
3 Jackson et al. (1981) is an unpublished report prepared by Huntingdon Research Center and submitted to U.S. EPA’s Office of Chemical Safety and Pollution Prevention. The report is not publicly available. A U.S. EPA AEGL staff member with FIFRA clearance reviewed the original report and confirmed the details provided in the U.S. EPA (2006b) Weight-of-Evidence Discussion for the study.
both sexes. Necropsy and histopathologic examination of the animals that died revealed congestion, edema, bronchiolitis, interstitial pneumonitis, and intra-alveolar hemorrhage of the lungs, accompanied by increased lung weight and focal hepatic necrosis. Distention of the stomach and intestines was attributed to swallowing air (gasping) prior to death.
Ullman (1985) reported 100% mortality in Wistar rats within 30 min of exposure to MITC at 10 ppm. These results conflict with those of the other acute toxicity studies, as well as with the repeated-exposure studies with the same strain of rats (see Section 3.3). Nesterova (1969) reported no deaths in rats (strain unidentified) exposed to MITC at 26 ppm for 4 h, whereas 80-100% of mice died at 25-26 ppm. No details of the methods of the Nesterova (1969) were reported. With the exception of these two studies, the acute lethality data for MITC are presented in Table 6-4.
3.2. Nonlethal Acute Toxicity
No acute toxicity studies other than those summarized in Section 3.1 were found.
3.3. Repeated Exposure Studies
Groups of five male and five female SPF Wistar/Chubb:THOM rats were exposed whole-body to MITC for 6 h/day, 5 days/week for 28 days (Klimisch 1987; EPA 2006b). Measured concentrations of MITC were 0, 1.7, 6.8, and 34 ppm. Beginning on the third exposure day and continuing throughout the study, rats exposed at 6.8 or 34 ppm exhibited eyelid closure, somnolence, and ruffled fur during each daily exposure. No clinical signs were observed in the 1.7-ppm group. Additional clinical signs observed in the 34-ppm group included reddish nasal discharge, salivation, ocular discharge, and dyspnea. Except for ruffled fur and respiratory distress in the 34-ppm group, clinical signs resolved between exposures. Body weight and several clinical-chemistry parameters were reduced in the high-exposure group at sacrifice. Histopathologic examination revealed rhinitis in the nasal cavity, atrophy of the olfactory epithelium, metaplasia of the nasal respiratory epithelium, tracheal epithelial proliferation, bronchial pneumonia and bronchial and bronchiolar epithelial proliferation, and emphysema. The no-observed-adverse-effect level was 1.7 ppm.
TABLE 6-4 Acute Lethality Data on Methyl Isothiocyanate
| Species | Concentration (ppm) | Exposure Duration | Effect | Reference |
| Rat | 210 | 1 h | No mortality | Clark and Jackson, 1977 |
| 635 | 1 h | LC50 | ||
| Rat | 94 | 4 h | No mortality | Jackson et al. 1981 |
| 180 | 4 h | LC50 | ||
Groups of 10 male and 10 female Wistar were exposed nose-only to MITC (95.69% pure) at concentrations of 0, 1, 10, or 45 ppm for 4 h/day, 5 days/week for 12-13 weeks (Rosskamp et al. 1978; EPA 2006b). No clinical signs were observed in the 1- or 10-ppm groups. Reduced physical activity accompanied by salivation and nasal discharge was recorded throughout the study in the 45-ppm group. Body weight was reduced only in the 45-ppm group, and body weight gain was reduced by 11-15% in the 10-ppm group and by 47-63% in the 45-ppm group. Histologic examination of the nasal passages was not performed. The results of repeated exposure studies of MITC are summarized in Table 6-5.
MITC was immunotoxic in repeated exposure, oral studies. Daily administration of MITC at 15-55 mg/kg by gavage for 5 days to B6C3F1 mice resulted in a decrease in thymus weight and cellularity and changed peripheral white blood cell populations (Keil et al. 1996). MITC was administered in Hanks balanced salt solution. Oral administration (gavage, water vehicle) of metam sodium to female B6C3F1 mice at 300 mg/kg for 10 or 14 days decreased thymus weight, increased spleen weight, increased bone marrow cellularity, reduced mature lymphocyte subpopulations in the thymus, depleted major subpopulations of thymocytes, and reduced body weight (Pruett et al. 1992).
3.4. Developmental and Reproductive Toxicity
No inhalation studies that evaluated the potential for MITC to induce reproductive or developmental toxicity were found.
The results of two- and three-generation oral reproductive studies with the rat and oral development toxicity studies with the rat and rabbit are summarized in Table 6-6. Parameters of reproductive performance were not altered by treatment in any generation. In the three-generation study, the irritant effects of MITC resulted in lesions of the stomach in all treatment groups. In the developmental toxicity studies, doses that affected fetal parameters also produced maternal toxicity. No teratogenic effects were evident.
TABLE 6-5 Results of Repeated Exposure Studies of Methyl Isothiocyanate in Rats
| Concentration (ppm) | Exposure Duration | Effect | Reference |
| 1.7 | 6 h/d, 5 d/wk | No clinical signs. | Klimisch 1987 |
| 6.8 | for 28 d | Eyelid closure, somnolence, ruffled fur. | |
| 34 | Eyelid closure, somnolence, ruffled fur, nasal discharge, salivation, ocular discharge, dyspnea, nasal and lung lesions. | ||
| 1 | 4 h/d, 5 d/wk for 12-13 wk | No clinical signs. | Rosskamp et al. 1978 |
| 10 | No clinical signs. | ||
| 45 | Apathetic appearance, salivation, nasal discharge reduced body weight. | ||
TABLE 6-6 Developmental and Reproductive Studies of Oral Exposure to Methyl Isothiocyanate
| Species | Exposure | Effect | Reference |
| Developmental Toxicity | |||
| Rat | 2, 10, or 50 ppm in drinking water, 70-77 days prior to mating, two generations | No effect on fertility, reproductive performance, or development or growth of offspring; reduced water consumption (10 and 50 mg/kg). | Barker 1987 |
| Rat | 1, 3, 10, or 30a mg/kg by gavage, starting at 28 days of age until litters delivered, three-generations | No mortality at 1, 3, or 10 mg/kg; no reproductive or developmental effects; stomach lesions in dams. | Pflaum et al. 1978 |
| Reproductive Toxicity | |||
| Rat | 1, 5, or 25 mg/kg by gavage, GDs 6-15 | Fetal growth retardation at 25 mg/kg, secondary to decreases in maternal food intake and body weight. | Irvine 1983 |
| Rat | 3, 10, or 30 mg/kg by gavage, GDs 6-15 | No effect on reproductive indices; some fetal growth retardation at 30 mg/kg. | Hellwig and Hildebrand 1987 |
| Rabbit | 1, 3, or 5 mg/kg by gavage, GDs 7-19 | Reduced fetal weight and shorter crown to rump length at 5 mg/kg, secondary to maternal toxicity evidenced by reduced food consumption and lower weight gain. | Irvine 1984 |
| Rabbit | 1, 3, or 10 mg/kg by gelatin capsules, GDs 6-18 | Maternal deaths, fetal toxicity, reduced fetal body weight and survival at 10 mg/kg/day; possible maternal toxicity at 3 mg/kg/day (2 deaths vs. 1 in controls). | Ladd and Smith 1976 |
| Rabbit | 1, 3, or 10 mg/kg by gavage, GDs 6-28 | No clinical signs; no maternal or fetal toxicit observed | Becker et al. 1986 |
a 30-mg/kg group was terminated at week 5.
Abbreviations: GD, gestation day.
A three-generation oral study was conducted with Charles River CD rats (Pflaum et al. 1978). Beginning at 28 days of age in all generations, groups of 10 male and 20 female rats were administered MITC at 0, 3, 10, or 30 mg/kg by gavage. At week 5, the 30-mg/kg group was terminated and a 1 mg/kg group added. Dosing was 5 days/week until the animals were killed. Each generation was allowed to reach maturity, mate, and produce two litters. The first litter was killed at weaning, and the second litter became the parental animals for the next generation. Each parental generation was killed after littering twice. There were no mortalities or changes in body weight or body weight gain in any generation.
Reproductive indices, gross fetal abnormalities, and organ weights were comparable among all groups. Histopathologic examination of each parental generation revealed lesions of the forestomach, which were dose-related in incidence and severity.
Groups of 24-28 mated CD rats were given MITC by gavage in corn oil at doses of 0, 1, 5, or 25 mg/kg/day on gestation days (GD) days 6-15 (Irvine 1983). There were no differences in pregnancy incidence among the groups and there were no clinical signs except for staining of fur in the high-dose group. For dams, there was a dose-related reduction in food intake, but it was statistically significant only in the high-dose group; this group also had a non-significant reduction in body weight gain. Gross necropsy revealed thickening of the stomach wall, occasionally accompanied by adhesions of viscera to the stomach (24/27 in the 25-mg/kg group, 1/28 in the 5-mg/kg group). Fetuses of dams exposed at 25-mg/kg/day were smaller in body weight and had reduced crown-rump length with delayed skeletal ossification compared with other groups. Fetal growth retardation in the 25-mg/kg group was associated with decreased maternal food intake and reduced weight gain.
Similar to the above study, mated Wistar rats were administered MITC (in corn oil) by gavage at 0, 3, 10, and 30 mg/kg on days 6-15 of gestation (Hellwig and Hildebrand 1987). Dams were killed on GD 20. There were no deaths or clinical signs other than reddish coloration of the snout in some high-dose dams. There were no significant differences in reproductive or developmental parameters (numbers of corpora lutea, implantations, live fetuses, and sex distribution). The number of fetuses weighing less than 75% of the mean fetal weight per litter was increased in the high-dose group. There was no evidence of teratogenicity.
New Zealand white rabbits were administered MITC (in corn oil) by gavage at 0, 1, 3, or 5 mg/kg on GDs 7-19 (Irvine, 1984). At 5 mg/kg, maternal food consumption was reduced and accompanied by a decrease in body weight gain. Fetal body weight was reduced at 5 mg/kg, but litter sizes were larger.
Maternal deaths of rabbits (7/17 does) were observed following administration of MITC (in gelatin capsules) at 10 mg/kg on GDs 6-18 (Ladd and Smith 1976). All does that died had enlarged gall bladders and multiple red foci on their liver surfaces. Maternal toxicity was also observed at 3 mg/kg, as evidenced by two deaths and reduced body weight gain in survivors. Survivors gained weight comparable to that of the control rabbits over GDs 18-29. Delayed ossification of the sternum was observed in fetuses of all the dose groups.
Becker et al. (1986) administered MITC (in corn oil) by gavage at 0, 1, 3, or 10 mg/kg to groups of 16 mated chinchilla rabbits on GDs 6-18. Mean maternal body weight was reduced over the dosing period, but body weights of the low-dose group were higher than those of the control group because of increased food consumption. There was no effect of treatment on the mean number of implantations, number of live fetuses, embryonic or fetal death, or pre-implantation and post-implantation losses. Fetal weight and sex distribution were comparable among groups. There was no evidence of teratogenicity.
3.5. Genotoxicity
Mutagenicity and genotoxicity studies of MITC were reviewed and summarized by NRA (1997) and Rubin et al. (2003). Although some of the studies did not report details, there was no clear evidence of mutagenicity or genotoxicity attributable to MITC treatment. MITC was tested for mutagenicity, with and without metabolic activation, in Salmonella typhimurium strains TA98, TA100, TA1535, TA1537, and TA1538 in several laboratories. MITC was also studied for mutagenicity in Escherichia coli strain wP2 hcr and at the HGPRT locus in cultured Chinese hamster V79 cells. There was no consistent evidence of mutagenicity in any of these studies.
MITC failed to induce sister chromatid exchanges in cultured Chinese hamster V79 cells, but gave positive results at some time points in an in vitro chromosome aberration test in the same system. MITC was negative in two rec assays using Bacillus subtilis, and it failed to induce unscheduled DNA synthesis in an assay with cultured primary rat hepatocytes. In an in vivo micronucleus test with CD-1 mice, MITC (110 mg/kg) did not show evidence of clastogenesis.
MITC (100 μg/mL) caused a marginal increase in mutations in S. typhimurium TA100 and TA98 (Kassie et al. 2001). Treatment with MITC induced repairable DNA damage in E. coli, and addition of a metabolic activation system reduced these effects. An in vivo micronucleus test with mice treated by gavage with MITC at 90 mg/kg produced only a marginal response.
3.6. Chronic Toxicity and Carcinogenicity
No chronic toxicity or carcinogenicity studies of inhalation exposure to MITC were found. Two-year bioassays with the rat and mouse in which MITC was administered via the drinking water were available.
MITC was administered to groups of 60 male and 60 female Sprague-Dawley rats in the drinking water at concentrations of 2, 10, or 50 ppm for 104 weeks (Brown 1981). Vapor loss from the water bottles was minimized with a redesign of the water bottles. Average doses over the 2 years were 0, 0.08, 0.37, or 1.60 mg/kg/day for males and 0, 0.12, 0.56, or 2.65 mg/kg/day for females. Additional groups of 10 rats of each sex were similarly treated and killed after 52 weeks. No histopathologic lesions were present in any organ at 52 weeks. Survival was similar among the groups. Water intake and body weight were decreased in males in the 50-ppm group, an effect the investigators attributed to MITC making the water unpalatable. Food intake was comparable to controls. No oncogenic response was found at any dose.
In a 106-week study, MITC was administered in drinking water at concentrations of 0, 5, 20, 80, or 200 ppm to groups of 70 male and 70 female ICR-JCR mice (Sato 1980). Calculated doses, based on drinking water analysis and water consumption were 0, 0.68, 2.74, 9.82, or 21.34 mg/kg/day for males and 0, 0.76, 3.04, 10.81, or 24.06 mg/kg/day for females. Mortality was comparable for all
groups (35-56%). No specific clinical signs were observed, but ruffled hair and dull coat were noted at 80 and 100 ppm. Body weight gain in males and females at 200 ppm and in males at 80 ppm was decreased. There were no differences in food consumption. Observed changes in hematology and clinical chemistry parameters were either transient or not dose related. There was no abnormal tissue histopathology. No differences in tumor types or time to appearance of tumors were found between the treatment groups.
3.7. Summary
Two acute inhalation toxicity studies with rats were available. The 1- and 4-h LC50 values were 635 and 180 ppm, respectively (Clark and Jackson 1977; Jackson et al. 1981). Respective highest nonlethal concentrations were 210 and 94 ppm. The study by Clark and Jackson (1977) administered a pesticide formulation containing MITC to the animals, and estimated MITC exposure concentrations; however, the means of estimating the MITC exposures was not clearly explained in the report. In repeated exposure studies, no clinical signs were observed in rats exposed to MITC at 1 or 1.7 ppm (Rosskamp et al. 1978; Klimisch 1987). Signs of ocular irritation were observed at 6.8 ppm in one study (6 h/day) but not at 10 ppm (4 h/day) in another study. No deaths were observed in rats after a 28-day exposure to MITC at 34 ppm (6 h/day, 5 days/week) or after 1213 weeks at 45 ppm (4 h/day, 5 days/week).
Only oral studies were available that evaluated the reproductive and developmental toxicity of MITC. Two reproductive studies were conducted in rats, and the developmental toxicity studies were conducted in rats and rabbits. Parameters of reproductive performance were not altered by MITC treatment in any generation. In a three-generation reproduction study, the irritant effect of MITC resulted in gastric lesions in all treatment groups. In the developmental toxicity studies, doses that affected fetal parameters also produced maternal toxicity. No evidence that MITC was teratogenic was found.
No clear or consistent evidence of mutagenicity or genotoxicity was found when MITC was tested in a variety of standard short-term bacterial and mammalian cell systems. No evidence of carcinogenicity was found in rats or mice after chronic oral exposure to MITC.
4. SPECIAL CONSIDERATIONS
4.1. Metabolism and Disposition
In rats, MITC is conjugated with glutathione and excreted in the urine as the corresponding mercapturic acid. Metabolism of MITC was studied in rats and mice after intraperitoneal injection (Lam et al. 1993). MITC was labeled with 13CH3, 14CH3, or 13CS; metabolites were identified with 13C-nuclear magnetic resonance and quantified by high-performance liquid chromatography and radiocar-
bon counting. Mice excreted 80% of the 14C-radiocarbon label in the urine; feces were a minor route of elimination. Recovery of 14CO2 was 3.8%. The carcass contained 6% of the radiolabel after 48 h. The liver and kidneys generally had highest 14C tissue values. For the rat, identification of S-(N-methylthiocarbamoyl) glutathione in the bile and S-(N-methylthiocarbamoyl)mercapturic acid in the urine indicates that direct conjugation with glutathione is the primary detoxification mechanism. The mercapturate was a minor metabolite in the urine of mice. Mennicke et al. (1983) also identified the dithiocarbamidic acid esters in rat urine after oral administration of MITC.
Pharmacokinetic parameters were similar following oral administration of MITC (Hawkins et al. 1987). Rats were given 14C-radiolabeled MITC at 4.4 or 33 mg/kg by gavage. By 24 h, 88-96% of the dose was absorbed. The thyroid, liver, kidneys, whole blood, and adrenals were relatively high sites of accumulation. Tissue concentrations at 168 h did not exceed 2.3% of the administered dose. The metabolites N-acetyl-S-(N-methylthiocarbamoyl)-L-cysteine and the corresponding cysteine conjugate were identified in the urine.
4.2. Mechanism of Toxicity
Clinical signs and respiratory tract pathology consistent with the actions of a primary irritant have been observed in laboratory studies with rodents. Humans exposed to MITC complained of burning eyes and skin, nausea, sore throat, salivation, coughing, and shortness of breath. Dourson et al. (2010) proposed that trigeminal nerve stimulation occurs first, followed by ocular irritation, and finally respiratory effects. The available data, while limited, do not provide clear evidence that MITC exposure is associated with systemic toxicity.
4.3. Structure-Activity Relationships
The isothiocyanates (R-N=C=S) are less toxic than the isocyanate congeners (R-N=C=O). Sulfur is less electronegative than oxygen and hydrogen bonding of sulfur takes place less readily than with oxygen (Finar 1986). In general, the thiols are less water soluble than the corresponding alcohols, likely due to their inability to form hydrogen bonds with water.
4.4. Other Relevant Information
4.4.1. Species Variability
Acute and repeat inhalation studies with MITC were all conducted with the rat. In oral reproductive and developmental studies with the rat and rabbit, no clear differences in species sensitivity were evident.
4.4.2. Susceptible Populations
No data on subpopulations that may be particularly sensitive to MITC exposure were found. MITC is a direct-acting respiratory irritant. As such, individuals with respiratory diseases such as asthma or emphysema might be more sensitive.
4.4.3. Concentration-Exposure Duration Relationship
No information on a concentration-exposure duration relationship was located. Acute rodent studies were conducted for two durations (1 or 4 h). Although an n value of approximately 1 can be calculated from these two studies, there is much uncertainty in extrapolating from two data points. The concentration-exposure duration relationship for many irritant and systemically-acting vapors and gases has been described by the equation Cn × t = k, where the exponent n values range from 0.8 to 3.5 (ten Berge et al. 1986). In the absence of a chemical-specific, empirical exponent, default values of n = 3 and n = 1 when extrapolating to shorter and longer durations, respectively, is used (NRC 2001). This method will yield the most conservative AEGL estimates.
4.4.4. Concurrent Exposure Issues
MITC is a decomposition product of metam sodium, which can also yield other breakdown products including hydrogen sulfide, methyl isocyante, carbon disulfide, and methyl amine, depending on the soil pH and environmental conditions (O’Malley et al. 2004). Thus, exposure to MITC may occur concurrently with exposure to these other compounds, resulting in symptoms that may not be solely related to MITC toxicity.
5. DATA ANALYSIS FOR AEGL-1
5.1. Summary of Human Data Relevant to AEGL-1
A study conducted with human volunteers measured ocular irritation, considered the most sensitive irritant response to MITC (Russell and Rush 1996). Ocular irritation was determined in 70 volunteers, tested in groups of 9-16 individuals, at discrete time intervals of 1 and 14 min and 4 and 8 h. No ocular irritation was reported in association with exposure at 3.3 ppm for 1 min, 0.6 ppm for 14 min, or 0.22-0.23 ppm for 1-8 h. When exposed to MITC at 1.9 ppm for 14 min or 0.8 ppm for 1-8 h, subjects reported ocular irritation slightly less than that associated with cutting a single mild onion. The study was well-designed and well-conducted.
5.2. Summary of Animal Data Relevant to AEGL-1
Acute animal studies were generally conducted at much higher concentrations than the clinical study, and subjective responses cannot be ascertained in animals. In repeated-exposure studies, no clinical signs were observed in rats exposed to MITC at 1.7 ppm for 6 h/day, 5 days/week for 28 days (Klimisch 1987) or in rats exposed at 1 ppm for 4 h/day, 5 days/week for 12-13 weeks (Rosskamp et al. 1978).
5.3. Derivation of AEGL-1
The study with human volunteers (Russell and Rush 1996) was the most appropriate for derivation of AEGL-1 values. Although the study examined only ocular irritation (through the use of goggle exposures), that effect is believed to be a more sensitive indicator of MITC exposure (occurring more quickly and at lower concentrations) than nasal or respiratory irritation (Dourson et al. 2010). No ocular irritation was reported in association with exposure to MITC at 3.3 ppm for 1 min, 0.6 ppm for 14 min, or 0.22-0.23 ppm for 1-8 h. When exposed at 1.9 ppm for 14 min or 0.8 ppm for 1-8 h, subjects reported ocular irritation slightly less than that associated with cutting a single mild onion. The effect at 0.8 ppm was a slight, transient, and subjective effect. Blinking rate was slightly increased, but there was no tearing or redness of the eye. A concentration of 0.8 ppm was considered the highest concentration that was not associated with notable discomfort and was selected as the point of departure. An intraspecies uncertainty factor of 3 was applied to protect sensitive individuals. According to NRC (2001), “in those cases in which the mode or mechanism of action is such that the response elicited by exposure to the chemical by different subpopulations is unlikely to differ, an intraspecies uncertainty factor of 3-fold is generally used. Typically, this response involves a direct-acting mechanism of toxicity in which metabolic or physiologic differences are unlikely to play a major role." Additional support for an uncertainty factor of 3 is provided by the inclusion of young adults (subjects ranged in age from 18-67 years, mean of 32 years) in the group of subjects in the study by Russell and Rush (1996). Kjaergaard et al. (1992) evaluated the range of human sensitivity to ocular irritation by carbon dioxide among 158 volunteers, and observed no differences based on gender or smoking status, but observed that young adults (<40 years of age) were more sensitive than the elderly. The threshold for ocular irritation among sensitive individuals was within a factor of 2 of the average response of young adults (Kjaergaard et al. 1992).
Because 0.8 ppm was tested for up to 4 h, this concentration was used for all AEGL-1 exposure durations (see Table 6-7). Furthermore, there is adaptation to the slight irritation that defines the AEGL-1 effects. The 0.8-ppm value is supported by no-effect concentrations in repeated-exposure studies with rodents. No signs of ocular irritation or other clinical signs were observed in rats exposed
at 1.7 ppm for 6 h/day, 5 days/week for 28 days (Klimisch 1987) or in rats exposed at 1 ppm for 4 h/day, 5 days/week for 12-13 weeks (Rosskamp et al. 1978). Application of total uncertainty factors of 3 or 10 to the animal data would bring the values (0.33-0.56 ppm or 0.10-0.17 ppm, respectively) close to or below the no-effect level of 0.22-0.23 ppm observed in the clinical study. The AEGL-1 calculations are presented in Appendix B, a derivation summary is presented in Appendix C, and a category plot of the relationship between AEGL values and toxicity data is presented in Appendix D.
6. DATA ANALYSIS FOR AEGL-2
6.1. Summary of Human Data Relevant to AEGL-2
Ocular irritation may be sufficient to impair escape, so is a relevant AEGL-2 end point. Russell and Rush (1996) observed several measures of ocular irritation in human volunteers exposed to MITC at 0.8 ppm for up to 4 h. With 3 and 4 h of exposure, all nine subjects appeared to respond positively on the Likert scale (indicating irritation), with mean Likert scale responses of 39 ± 19% and 39 ± 26%, respectively (compared with 5 ± 6% and 4 ± 6% in controls). If 50% on the Likert scale was considered equivalent to the irritation from the cutting of a single mild onion, and the mean response at 0.8 ppm was below this level, this degree of ocular irritation would not be expected to impair escape. No other human studies were available for development of AEGL-2 values.
6.2. Summary of Animal Data Relevant to AEGL-2
No acute exposure studies with laboratory animals relevant to deriving AEGL-2 values were identified. In repeated exposure studies, rats exposed to MITC at 6.8 ppm for 6 h/day, 5 days/week for 28 days showed signs of ocular irritation and general discomfort during daily exposure periods, beginning on the third exposure day and continuing throughout the study (Klimisch 1987). These signs were reversible between exposures. Rats exposed at 10 ppm for 4 h/day, 5 days/week for 12-13 weeks did not have clinical signs (Rosskamp et al. 1978). These concentrations are close to the derived 4- and 8-h AEGL-2 values (10 and 5.3 ppm, respectively [see below]). No clinical signs were seen at 1 or 1.7 ppm.
TABLE 6-7 AEGL-1Values for Methyl Isothiocyanate
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
6.3. Derivation of AEGL-2
In lethality studies with rats, the concentration-response curve for MITC was steep. In a 1-h study with rats (Clark and Jackson 1977), the highest nonlethal concentration of 210 ppm is about one-third the LC50 of 635 ppm. In a 4-h study with rats (Jackson et al. 1981), the highest nonlethal concentration of 94 ppm is about one-half the LC50 of 180 ppm. In the absence of data that address AEGL-2 end points and with evidence of a steep concentration-response curve, the AEGL-3 values were divided by 3 to derive AEGL-2 values (NRC 2001). AEGL-2 values are presented Table 6-8; the calculations are presented in Appendix B, and a category graph of the relationship between AEGL values and toxicity data are presented in Appendix D.
Although acute studies that address AEGL-2 end points are not available, repeat-exposure studies (Rosskamp et al. 1978; Klimisch, 1987) support the AEGL-2 values.
7. DATA ANALYSIS FOR AEGL-3
7.1. Summary of Human Data Relevant to AEGL-3
No human studies were available to derive AEGL-3 values for MITC.
7.2. Summary of Animal Data Relevant to AEGL-3
One- and 4-h inhalation studies were conducted in rats. The 1- and 4-h LC50 values were 635 ppm (Clark and Jackson 1977) and 180 ppm (Jackson et al. 1981), respectively. No deaths occurred at 210 ppm for 1 h or at 94 ppm for 4 h.
7.3. Derivation of AEGL-3
The study by Clark and Jackson (1977) used a pesticide formulation containing MITC as the test material, and the calculation used to estimate the MITC concentration in the chamber was not specified. In light of the uncertainties associated with this study, it was not used to derive AEGL-3 values. The POD for the AEGL-3 values is the highest 4-h nonlethal concentrations of 94 ppm in the study of rats (Jackson et al. 1981). Interspecies and intraspecies uncertainty factors of 3 each are usually applied to chemicals that are direct-acting irritants
TABLE 6-8 AEGL-2 Values for Methyl Isothiocyanate
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 21 ppm (63 mg/m3) |
21 ppm (63 mg/m3) |
17 ppm (51 mg/m3) |
10 ppm (30 mg/m3) |
5.3 ppm (16 mg/m3) |
(NRC 2001). However, application of a total uncertainty factor of 10 would result in AEGL-3 values that are inconsistent with the human exposure study (Russell and Rush 1996). Therefore, interspecies and intraspecies uncertainty factors of 1 and 3, respectively, were applied. Reduction of an uncertainty factor is appropriate when the weight of the evidence indicates that a higher uncertainty factor would result in AEGL values at odds with human data (NRC 2001). Time scaling was performed using the equation Cn x t = k. Because the available data were insufficient for establishing an empirical value of n, default values of n = 3 and n = 1 when extrapolating to shorter and longer durations, respectively, were used (NRC 2001). Because of the uncertainty associated with extrapolating a 4-h POD to a 10-min value, the 10-min AEGL-3 value was set equal to the 30-min AEGL-3 value.
The longer-term AEGL-3 values are supported by repeated-exposure studies. Rats tolerated MITC at 45 ppm (a concentration higher than the 4- and 8-h AEGL-3 values of 27 and 13 ppm, respectively) for 4 h/day, 5 days/week for 1213 weeks (60-65 exposures) without any deaths (Rosskamp et al. 1978). AEGL-3 values for MITC are presented in Table 6-9, the calculations are presented in Appendix B, and a category graph of the relationship between AEGL-3 values and toxicity data are presented in Appendix D.
If the 1-h nonlethal concentration of 210 ppm from the Clark and Jackson (1977) study were used to derive 10-min, 30-min, and 1-h AEGL-3 values, estimates of 130, 88, and 70 ppm, respectively, would result. The estimates are comparable (slightly higher) than those estimated from the 4-h POD (63, 63, and 50 ppm, respectively).
8. SUMMARY OF AEGLs
8.1. AEGL Values and Toxicity End Points
The AEGL values for MITC are presented in Table 6-10.
8.2. Comparison with Other Standards and Guidelines
No exposure standards or guidelines for MITC were identified.
TABLE 6-9 AEGL-3 Values for Methyl Isothiocyanate
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 63 ppm (190 mg/m3) |
63 ppm (190 mg/m3) |
50 ppm (150 mg/m3) |
31 ppm (94 mg/m3) |
16 ppm (47 mg/m3) |
TABLE 6-10 AEGL Values for Methyl Isothiocyanate
| Classification | 10 min | 30 min | 1 h | 4 h | 8 h |
| AEGL-1 (nondisabling) | 0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
| AEGL-2 (disabling) | 21 ppm (63 mg/m3) |
21 ppm (63 mg/m3) |
17 ppm (51 mg/m3) |
10 ppm (30 mg/m3) |
5.3 ppm (16 mg/m3) |
| AEGL-3 (lethal) | 63 ppm (190 mg/m3) |
63 ppm (190 mg/m3) |
50 ppm (150 mg/m3) |
31 ppm (94 mg/m3) |
16 ppm (47 mg/m3) |
8.3. Data Adequacy and Research Needs
Data from clinical studies and studies with laboratory animals were adequate to develop AEGL values for MITC. However, the available acute animal data are limited to two studies in rats (Clark and Jackson 1977; Jackson et al. 1981) and included one study that administered a pesticide formulation containing MITC as the test material (Clark and Jackson 1977). Additional information on acute toxicity of MITC in other species would be helpful. Similarly, although available animal data and human exposure incidents suggest that ocular irritation is a more sensitive indicator of exposure than upper-respiratory-tract irritation, more rigorous investigation is needed to support this finding.
9. REFERENCES
Alexeeff, G.V., D.J. Shusterman, R.A. Howd, and R.J. Jackson. 1994. Dose-response assessment of airborne methyl isothiocyanate (MITC) following a metam sodium spill. Risk Anal. 14(2):191-198.
Barker, L. 1987. Technical Methylisothiocyanate (MITC): Two Generation Oral (Drinking Water) Reproduction Study in the Rat. Study No. Tox/87/203-21, NOR-AM Chemical Company; Prepared by Hazleton Laboratories, UK (as cited in Rubin et al. 2003).
Becker, H., P. Schafroth, W. Vogel, C. Terrier, and K. Sachsse. 1986. Embryotoxicity (Including Teratogenicity) Study with MITC ZNT-No. 85/231-2 in the Rabbit. Project No. 056687, RZ-No. 86/395, RCC Research & Consulting Company AG, Itingen, Switzerland. September 1986 (as cited in NRA 1997).
Bretaudeau Deguigne, M., L. Lagarce, D. Boels, and P. Harry. 2011. Metam sodium intoxication: The specific role of degradation products - methyl isothiocyanate and carbon disulphide - as a function of exposure. Clin. Toxicol. (Phila.) 49(5):416-422.
Brown, B. 1981. Methylisothiocyanate: A Chronic Oral (Drinking Water) Toxicity Study and Carcinogenicity Study in the Rat. Schering Study No. 2611-14/1R, Hazleton Laboratories Europe, Ltd. (as cited in Rubin et al. 2003).
Clark, G.C., and G.C. Jackson. 1977. Methylisothiocyanate: Acute Inhalation Toxicity, 1 Hour LC50 in Rats. SHG 132/77372. Huntingdon Research Centre, UK. Schering Report T6 (as cited in NRA 1997).
Cone, J.E., L. Wugofski, J.R., Balmes, R. Das, R. Bowler, G. Alexeeff, and D. Shusterman. 1994. Persistent respiratory health effects after a metam sodium pesticide spill. Chest 106(2):500-508.
Dourson, M.L., M.J. Kohrman-Vincent, and B.C. Allen. 2010. Dose response assessment for effects of acute exposure to methyl isothiocyanate (MITC). Regul. Toxicol. Pharmacol. 58(2):181-188.
EPA (U.S. Environmental Protection Agency). 2006a. Data Evaluation Record: M.J. Russell and T.J. Rush (1996) Methyl Isothiocyanate: Determination of Human Olfactory Detection Threshold and Human No Observable Effect Level for Eye Irritation. Report No. RP 96-049B, Sensory Testing Laboratory, University of California, Davis [online]. Available: http://www.epa.gov/osa/hsrb/files/meeting-materials/may-2-3-2006-public-meeting/mitc_der.pdf [accessed Aug. 21, 2013].
EPA (U.S. Environmental Protection Agency). 2006b. Human Studies Review Board: Weight of Evidence Discussion for Methyl Isothiocyanate [MITC]). Memorandum from Anna Lowit, Toxicologist, Health Effects Division, to Tina Levine, Director, Health Effects Division, Office of Prevention, Pesticides and Toxic Substances, U.S. Environmental Protection Agency, Washington, DC. April 13, 2006 [online]. Available: http://www.epa.gov/osa/hsrb/files/meeting-materials/may-2-3-2006-public-meeting/7-MITC-weight_of_evidence.pdf [accessed Aug. 21, 2013].
Finar, I.L. 1986. Aliphatic compounds of sulphur, phosphorus, silicon and boron. Chapter 14 in Organic Chemistry, Vol. 1. The Fundamental Principles, 6th Ed. New York, NY: Longman.
Hawkins, D., L.F. Elsom, and G. Girkin. 1987. The Biokinetics and Metabolism of 14C-Metam Sodium in the Rat. Metam-Sodium Task Force. Study No. BASF 88/0030. Huntington Research Centre, Ltd (as cited in Rubin et al. 2003).
Hellwig, J. and B. Hildebrand. 1987. Report on the Study of the Prenatal Toxicology of MITC in Rats after Oral Administration (Gavage). Project No. 34R0231/8537, BASF Aktiengesellschaft, Department of Toxicology, Federal Republic of Germany. September 2, 1987 (as cited in NRA 1997).
HSDB (Hazardous Substances Data Bank). 2012. Methyl Isothiocyanate (CAS Reg. No. 556-61-6). TOXNET, Specialized Information Services, U.S. National Library of Medicine, Bethesda, MD [online]. Available: http://toxnet.nlm.nih.gov/cgi-bin/sis/htmlgen?HSDB [accessed Aug. 21, 2013].
Irvine, L.F.H. 1983. Methyl Isothiocyanate (MITC): Oral (Gavage) Teratology Study in the Rat. Hazleton and Schering Report 3191-14/10; Schering Report T53 (as cited in NRA 1997).
Irvine, L.F.H. 1984. Methyl Isothiocyanate (MITC): Oral (Gavage) Teratology Study in the New Zealand White Rabbit. Hazleton and Schering Report No. 3687-14/30. Schering Report T74 (as cited in NRA 1997).
Jackson, G.C., G.C. Clark, D.E. Prentice, R.M. Read, C. Gopinath, and C. Cherry. 1981. Methyl Isothiocyanate: Acute Inhalation Toxicity in Rats. 4 Hour Exposure. RZ No. 81/082, 378/801109 Huntingdon Research Centre, Huntingdon, England. Submitted to U.S. EPA’s Office of Chemical Safety and Pollution Prevention.
Kassie, F., B. Laky, E. Nobis, M. Kundi, and S. Knasmuller. 2001. Genotoxic effects of methyl isothiocyanate. Mutat. Res. 490(1):1-9.
Keil, D.E., E.L. Padgett, D.B. Barnes, and S.B. Pruett. 1996. Role of decomposition products in sodium methyldithiocarbamate-induced immunotoxicity. J. Toxicol. Environ. Health 47(5):479-492.
Kjaergaard S., O.F. Pedersen, and L. Molhave. 1992. Sensitivity of the eyes to airborne irritant stimuli: Influence of individual characteristics. Arch. Environ. Health 47(1): 45-50.
Klimisch, H.J. 1987. Study of the Subchronic Inhalation Toxicity of Methyl Isothiocyanate in Wistar Rats (4 Weeks Study). BASF Project 87/0244: 4OI0231/8539. BASF Aktiengesellschaft, Ludwigshafen, Federal Republic of Germany (as cited in EPA 2006b).
Ladd, R., and P.S. Smith. 1976. Teratogenic Study with Methylisothiocyanate in Albino Rabbits. Report No. 651-07457, Schering Report T23, Industrial Bio-Test Laboratories Inc., IL (as cited in NRA 1997).
Lam, W.W., J.H. Kim, S.E. Sparks, G.B. Quistad, and J.E. Casida. 1993. Metabolism in rats and mice of the soil fumigants metham, methyl isothiocyanate, and dazonet. J. Agric. Food Chem. 41(9):1487-1502.
Lee, S., R. McLaughlin, M. Harnly, R. Gunier, and R. Kreutzer. 2002. Community exposures to airborne agricultural pesticides in California: Ranking of inhalation risks. Environ. Health Perspect. 110(12):1175-1184.
Mennicke, W.H., K. Gorler, and G. Krumbiegel. 1983. Metabolism of some naturally occurring isothiocyanates in the rat. Xenobiotica 13(4):203-207.
Merriman, J.H., and V.R. Hebert. 2007. Methyl isothiocyanate residential community air assessment for South Franklin County, Washington. Bull. Environ. Contam. Toxicol. 78(1):19-23.
Nesterova, M.F. 1969. Standards for carbathion in working zone air [in Russian]. Gig. Sanit. 34(5):191-196.
Nihon Schering. 1990. Summary of toxicity data on methyl isothiocyanate (MITC). J. Pestic. Sci. 15:297-304.
NRA (National Registration Authority). 1997. Metham Sodium, Dazomet and Methylisothiocyanate (MITC), Volume III, NRA Special Review Series 97.2. Canberra, Australia: National Registration Authority.
NRC (National Research Council). 1993. Guidelines for Developing Community Emergency Exposure Levels for Hazardous Substances. Washington, DC: National Academy Press.
NRC (National Research Council). 2001. Standing Operating Procedures for Developing Acute Exposure Guideline Levels for Hazardous Chemicals. Washington, DC: National Academy Press.
O’Malley, M., T. Barry, M. Verder-Carlos, and A. Rubin. 2004. Modeling of methyl isothiocyanate air concentrations associated with community illnesses following a metam sodium sprinkler application. Am. J. Ind. Med. 46(1):1-15.
O’Malley, M., T. Barry, M. Ibarra, M. Verder-Carlos, and L. Mehler. 2005. Illnesses related to shank application of metam-sodium, Arvin, California, July 2002. J. Agromedicine 10(4):27-42.
Pflaum, J., W. Thompson, C. Salamon, S. Smith, D. Arnold, R.J. Arceo, and G.E. Gordon. 1978. Three-generation Reproductive Study with Methyl Isothiocyanate in Albino Rats. Report No. 623-07393, Schering Report T24. Industrial Bio-Test Laboratories Inc., IL (as cited in NRA 1997).
Pruett, S.B., D.B. Barnes, Y.C. Han, and A.E. Munson. 1992. Immunological characteristics of sodium methyldithiocarbamate. Fundam. Appl. Toxicol. 18(1):40-47.
Pruett, S.B., L.P. Myers, and D.E. Keil. 2001. Toxicity of metam sodium. J. Toxicol. Environ. Health B Crit. Rev. 4(2):207-222.
Rosskamp. G., G. Schobel, A. Bhargava, P. Staben, and G. Schuppler. 1978. Methyl Isothiocyanate: ZK 3.318: A 12-13 Week Inhalation Study in the Rat, Project ID 374/77. Schering AG (as cited in EPA 2006b).
Rubin, A.L., M. Silva, J. Gee, T. Moore, and T. Thongsinthusak. 2003. Risk Characterization Document: Methyl Isothiocyanate (MITC) Following the Agricultural Use of Metam Sodium. Medical Toxicology Branch, Department of Pesticide Regulation, California Environmental Protection Agency, Sacramento, CA. July 25, 2003 [online]. Available: http://www.cdpr.ca.gov/docs/risk/rcd/mitc_sb950.pdf [accessed Aug. 20, 2013].
Russell, M.J., and T.I. Rush. 1996. Methyl Isothiocyanate: Determination of Human Olfactory Detection Threshold and Human No Observable Effect Level for Eye Irritation. Report No. RR 96-049B, Sensory Testing Laboratory, University of California, Davis, CA. Submitted to U.S. EPA’s Office of Chemical Safety and Pollution Prevention.
Sato, R. 1980. Two-year Chronic Toxicity and Oncogenicity Study with Methyl Isothiocyanate in Albino Mice (106 Week Final Report). Schering Study No. T52. Nippon Experimental Medical Research Institute Co (as cited in Rubin et al. 2003).
ten Berge, W.F., A. Zwart, and L.M. Appleman. 1986. Concentration-time mortality response relationship of irritant and systemically acting vapours and gases. J. Hazard. Mater. 13(3):301-309.
Ullman, L. 1985. 4-Hour Vapor Inhalation Toxicity (LC50) Study with Methylsenfoel (MITC) in Rats. Degassa AG. Study No. 042660. Research and Consulting Company, AG (as cited in Rubin et al. 2003).
van Doorn, R., M. Ruijten and T. van Harreveld. 2002. Guidance for the Application of Odor in Chemical Emergency Responses, Version 2.1, August 29, 2002. Presented at the NAC/AEGL Meeting, September 2002, Washington, DC.
Verschueren, K. 2001. Methylisothiocyanate. Pp. 1507-1508 in Handbook of Environmental Data on Chemicals, Vol. 2, 4th Ed. New York: John Wiley & Sons.
APPENDIX A
DERIVATION OF THE LEVEL OF DISTINCT ODOR AWARENESS FOR METHYL ISOTHIOCYANATE
The level of distinct odor awareness (LOA) represents the concentration above which it is predicted that more than half of the exposed population will experience at least a distinct odor intensity, and about 10% of the population will experience a strong odor intensity. The LOA should help chemical emergency responders in assessing the public awareness of the exposure from odor perception. The LOA derivation follows the guidance of van Doorn et al. (2002).
The odor detection threshold (OT50) for MITC was reported to be 1.7 ppm (Russell and Rush 1996). This value is the geometric mean of detection thresholds that ranged from 0.2 to 8 ppm among a panel of 33 individuals.
The concentration (C) leading to an odor intensity (I) of distinct odor detection (I = 3) is derived using the Fechner function:
I = kw × log (C ÷ OT50) + 0.5
For the Fechner coefficient, the default of kw = 2.33 was used due to the lack of chemical-specific data:
3 = 2.33 × log (C ÷ 1.7) + 0.5, which can be rearranged to
log (C ÷ 1.7) = (3 - 0.5) ÷ 2.33 = 1.07, and results in
C = (101.07) × 1.7 = 20 ppm
The resulting concentration is multiplied by an empirical field correction factor. The factor takes into account that in everyday life, factors such as sex, age, sleep, smoking, upper airway infections, allergy, and distraction, may increase the odor detection threshold by up to a factor of 4. In addition, it takes into account that odor perception is very fast (about 5 seconds) which leads to the perception of concentration peaks. A factor of one-third is applied to adjust for peak exposure. Adjustment for distraction and peak exposure lead to a correction factor of 4 ÷ 3 = 1.33.
LOA = C × 1.33 = 20 ppm × 1.33 = 27 ppm
The LOA for MITC is 27 ppm.
APPENDIX B
DERIVATION OF AEGL VALUES FOR METHYL ISOTHIOCYANATE
Derivation of AEGL-1 Values
|
Key study: |
Russell, M.J., and T.I. Rush. 1996. Methyl Isothiocyanate: Determination of Human Olfactory Detection Threshold and Human No Observable Effect Level for Eye Irritation. Report No. RR 96-049B, Sensory Testing Laboratory, University of California, Davis, CA. Unpublished report submitted to U.S. EPA’s Office of Chemical Safety and Pollution Prevention. |
|
Toxicity end point: |
Lowest-observed-effect level (0.8 ppm) for ocular irritation in the clinical study was a NOAEL for ocular irritation according to the definition of the AEGL-1 value; exposure durations of 1-480 min. |
|
Time scaling: |
None, because NOAELs for each exposure duration were used. |
|
Uncertainty factors: |
3 for intraspecies variability; based on direct-acting irritant mechanism of toxicity in which metabolic and physiologic differences are unlikely to play a major role (NRC 2001), and based on data showing approximately 2-fold range of human sensitivity to ocular irritants (Kjaergaard et al. 1992). |
|
Modifying factor: |
Not applicable |
|
Calculations: |
|
|
10-min AEGL-1: |
C = 0.8 ÷ 3 ppm = 0.27 ppm |
|
30-min AEGL-1: |
C = 0.8 ÷ 3 ppm = 0.27 ppm |
|
1-h AEGL-1: |
C = 0.8 ÷ 3 ppm = 0.27 ppm |
|
4-h AEGL-1: |
C = 0.8 ÷ 3 ppm = 0.27 ppm |
|
8-h AEGL-1: |
C = 0.8 ÷ 3 ppm = 0.27 ppm |
Derivation of AEGL-2 Values
In the absence of data for deriving AEGL-2 values for MITC and because MITC has a steep exposure-response curve in lethality studies (Clark and Jackson 1977; Jackson et al. 1981), AEGL-2 values were calculated by dividing the AEGL-3 values by 3 (NRC 2001).
|
10-min AEGL-2: |
63 ppm ÷ 3 = 21 ppm |
|
30-min AEGL-2: |
63 ppm ÷ 3 = 21 ppm |
|
1-h AEGL-2: |
50 ppm ÷ 3 = 17 ppm |
|
1-h AEGL-2: |
50 ppm ÷ 3 = 17 ppm |
|
4-h AEGL-2: |
31 ppm ÷ 3 = 10 ppm |
|
8-h AEGL-2: |
16 ppm ÷ 3 = 5.3 ppm |
Derivation of AEGL-3 Values
|
Key study: |
Jackson, G.C., G.C. Clark, D.E. Prentice, R.M. Read, C. Gopinath, and C. Cherry. 1981. Methyl Isothiocyanate: Acute Inhalation Toxicity in Rats. 4 Hour Exposure. RZ No. 81/082, Huntingdon Research Centre, Huntingdon, England. Unpublished report submitted to U.S. EPA’s Office of Chemical Safety and Pollution Prevention. |
|
Toxicity end points: |
NOAEL of 94 ppm for lethality in rats during 4-h exposure |
|
Time scaling: |
Cn × t = k; default values of n = 3 and n = 1 for scaling to shorter and longer exposure durations, respectively (NRC 2001). 30-min and 1-h values: (94 ppm ÷ 3)3 × 240 min = 7.383 × 106 ppm-min 8-h value: (94 ppm ÷ 3)1 × 240 min = 7,520 ppm-min |
|
Uncertainty factors: |
3 for intraspecies variability; humans are not expected to vary greatly in their response to a direct-acting irritant (NRC 2001). Application of a higher uncertainty factor would result in concentrations inconsistent with human clinical studies. |
APPENDIX C
ACUTE EXPOSURE GUIDELINE LEVELS FOR METHYL ISOTHIOCYANATE
Derivation Summary
AEGL-1 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
0.27 ppm (0.81 mg/m3) |
Key Reference: Russell, M.J., and T.I. Rush. 1996. Methyl Isothiocyanate: Determination of Human Olfactory Detection Threshold and Human No Observable Effect Level for Eye Irritation. Report No. RR 96-049B, Sensory Testing Laboratory, University of California, Davis, CA. Unpublished report submitted to U.S. EPA’s Office of Chemical Safety and Pollution Prevention.
Test species/Gender/Number): Human volunteers, male and female, 9-16 per group
Exposure route/Concentration/Duration: Inhalation; 0, 0.60, 1.9, or 3.3 ppm for 1 or 14 min; 0, 0.23, or 0.8 ppm for 4 h; 0 or 0.22 ppm for 8 h
|
Effects: |
||
|
|
No ocular irritation |
Mild ocular irritation |
|
|
|
|
|
1 min |
3.3 ppm |
— |
|
14 min |
0.60 ppm |
1.9 ppm |
|
1 h |
0.23 ppm |
0.8 ppm |
|
4 h |
0.23 ppm |
0.8 ppm |
|
8 h |
0.22 ppm |
— |
End point/Concentration/Rationale: Ocular irritation was mild (less than that associated with cutting one mild onion) and, thus, did not represent the notable discomfort that constitutes an AEGL-1 effect (NRC 2001).
Uncertainty factors/Rationale:
Total uncertainty factor: 3
Interspecies: 1, because human data were used
Intraspecies: 3, based on direct-acting irritant mechanism of toxicity in which metabolic and physiologic differences are unlikely to play a major role (NRC 2001), and based on data showing an approximately 2-fold range of human sensitivity to ocular irritants (Kjaergaard et al. 1992).
Modifying factor: None
Animal-to-human dosimetric adjustment: Not applicable
Time scaling: None; the POD was tested for durations of 1-4 h with no increase in severity of effect; therefore, the irritant effects of MITC are not expected to become more severe with increasing duration at this concentration.
Data adequacy: The study was well conducted and used an adequate numbers of healthy individuals. Ocular irritation is considered the most sensitive end point in studies with MITC.
AEGL-2 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 21 ppm (63 mg/m3) |
21 ppm (63 mg/m3) |
17 ppm (51 mg/m3) |
10 ppm (30mg/m3) |
5.3 ppm (16 mg/m3) |
Data adequacy: Data on MITC were inadequate for deriving AEGL-2 values. When data are lacking and the concentration-response curve is steep, AEGL-2 values may be derived by dividing the AEGL-3 values by 3 (NRC 2001). A steep concentration-response curve has been demonstrated for MITC. In a 1-h study with rats (Clark and Jackson 1977), the highest nonlethal concentration of 210 ppm is about one-third the LC50 of 635 ppm. In a 4-h study with rats (Jackson et al. 1981), the highest nonlethal concentration of 94 ppm is about one-half the LC50 of 180 ppm.
AEGL-3 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 63ppm (190 mg/m3) | 63 ppm (190 mg/m3) |
50 ppm (150 mg/m3) |
31 ppm (94 mg/m3) |
16 ppm (47 mg/m3) |
Key reference: Jackson, G.C., G.C. Clark, D.E. Prentice, R.M. Read, C. Gopinath, and C. Cherry. 1981. Methyl Isothiocyanate: Acute Inhalation Toxicity in Rats. 4 Hour Exposure. RZ No. 81/082, Huntingdon Research Centre, Huntingdon, England. Unpublished report submitted to U.S. EPA’s Office of Chemical Safety and Pollution Prevention.
Test species/Strain/Number: Rat; Sprague-Dawley; groups of 5 per sex
Exposure route/Concentration/Duration: Inhalation; six concentrations for 4 h
Effects: No mortality at 94 ppm; LC50 = 180 ppm
End point/Concentration/Rationale: 94 ppm, highest 4-h nonlethal concentration
Uncertainty factors/Rationale:
Total uncertainty factor:
3 Interspecies: 1
Intraspecies: 3, considered sufficient to protect the sensitive population with respiratory diseases; application of larger uncertainty factors would conflict with results of clinical studies.
Modifying factor: None
Animal-to-human dosimetric adjustment: Not applicable
Time scaling: Cn × t = k; default values of n = 3 and n = 1 for scaling to shorter and longer exposure durations, respectively (NRC 2001). Due to uncertainty in extrapolating a 4-h point of departure to a 10-min value, the 10-min AEGL-3 was set equal to the 30-min AEGL-3 value.
Data adequacy: The 4-h study used multiple concentrations and an adequate numbers of animals. The values are supported by repeated-exposure studies performed in different laboratories.
APPENDIX D
CATEGORY PLOT FOR METHYL ISOTHIOCYANATE
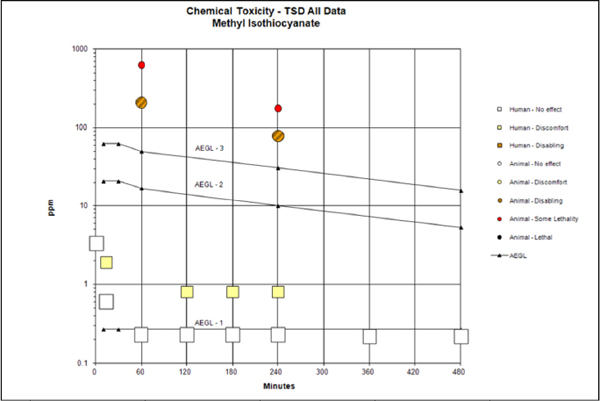
FIGURE D-1 Category plot of animal and human data and AEGL values for methyl isothiocyanate.
TABLE D-1 Data Used in the Category Plot for Methyl Isothiocyanate
| Source | Species | ppm | Minutes | Category | Comments |
| AEGL-1 | 0.27 | 10 | AEGL | ||
| AEGL-1 | 0.27 | 30 | AEGL | ||
| AEGL-1 | 0.27 | 60 | AEGL | ||
| AEGL-1 | 0.27 | 240 | AEGL | ||
| AEGL-1 | 0.27 | 480 | AEGL | ||
| AEGL-2 | 21 | 10 | AEGL | ||
| AEGL-2 | 21 | 30 | AEGL | ||
| AEGL-2 | 17 | 60 | AEGL | ||
| AEGL-2 | 10 | 240 | AEGL | ||
| AEGL-2 | 5.3 | 480 | AEGL | ||
| AEGL-3 | 63 | 10 | AEGL | ||
| AEGL-3 | 63 | 30 | AEGL | ||
| AEGL-3 | 50 | 60 | AEGL | ||
| Source | Species | ppm | Minutes | Category | Comments |
| AEGL-3 | 31 | 240 | AEGL | ||
| AEGL-3 | 16 | 480 | AEGL | ||
| Russell and Rush 1996 | Human | 3.3 | 1 | 0 | No ocular irritation |
| Human | 0.60 | 14 | 0 | No ocular irritation | |
| Human | 0.23 | 60 | 0 | No ocular irritation | |
| Human | 0.23 | 120 | 0 | No ocular irritation | |
| Human | 0.23 | 180 | 0 | No ocular irritation | |
| Human | 0.23 | 240 | 0 | No ocular irritation | |
| Human | 0.22 | 360 | 0 | No ocular irritation | |
| Human | 0.22 | 480 | 0 | No ocular irritation | |
| Human | 1.9 | 14 | 1 | Subjective ocular irritation | |
| Human | 0.8a | 120 | 1 | Subjective ocular irritation; | |
| increased blink rate | |||||
| Human | 0.8a | 180 | 1 | Subjective ocular irritation; | |
| increased blink rate | |||||
| Human | 0.8a | 240 | 1 | Subjective ocular irritation | |
| Clark and Jackson 1977 | Rat | 210 | 60 | 2 | 1-h highest nonlethal value |
| Rat | 635 | 60 | SL | 1-h LC50 | |
| Jackson et al. 1981 | Rat | 94 | 240 | 2 | 4-h highest nonlethal value |
| Rat | 180 | 240 | SL | 4-h LC50 | |
Categories: 0 = no effect, 1 = discomfort, 2 = disabling, SL = some lethality, 3 = lethal.
aNote: the discomfort associated with human exposure at 0.8 ppm (Russell and Rush 1996) was below the threshold for notable discomfort that constitutes an AEGL-1 effect.

































