Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 15 (2013)
Chapter: 1 Ethyl Mercaptan Acute Exposure Guideline Levels
1
Ethyl Mercaptan1
Acute Exposure Guideline Levels
PREFACE
Under the authority of the Federal Advisory Committee Act (FACA) P.L. 92-463 of 1972, the National Advisory Committee for Acute Exposure Guideline Levels for Hazardous Substances (NAC/AEGL Committee) has been established to identify, review, and interpret relevant toxicologic and other scientific data and develop AEGLs for high-priority, acutely toxic chemicals.
AEGLs represent threshold exposure limits for the general public and are applicable to emergency exposure periods ranging from 10 minutes (min) to 8 hours (h). Three levels—AEGL-1, AEGL-2, and AEGL-3—are developed for each of five exposure periods (10 and 30 min and 1, 4, and 8 h) and are distinguished by varying degrees of severity of toxic effects. The three AEGLs are defined as follows:
AEGL-1 is the airborne concentration (expressed as parts per million or milligrams per cubic meter [ppm or mg/m3]) of a substance above which it is predicted that the general population, including susceptible individuals, could experience notable discomfort, irritation, or certain asymptomatic, nonsensory
____________________
1This document was prepared by the AEGL Development Team composed of Cheryl Bast (Oak Ridge National Laboratory), Gary Diamond (SRC, Inc.), Chemical Manager Iris Camacho (U.S. Environmental Protection Agency and National Advisory Committee [NAC] on Acute Exposure Guideline Levels for Hazardous Substances), and Ernest V. Falke (U.S. Environmental Protection Agency). The NAC reviewed and revised the document and AEGLs as deemed necessary. Both the document and the AEGL values were then reviewed by the National Research Council (NRC) Committee on Acute Exposure Guideline Levels. The NRC committee has concluded that the AEGLs developed in this document are scientifically valid conclusions based on the data reviewed by the NRC and are consistent with the NRC guidelines reports (NRC 1993, 2001).
effects. However, the effects are not disabling and are transient and reversible upon cessation of exposure.
AEGL-2 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience irreversible or other serious, long-lasting adverse health effects or an impaired ability to escape.
AEGL-3 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience life-threatening health effects or death.
Airborne concentrations below the AEGL-1 represent exposure concentrations that could produce mild and progressively increasing but transient and nondisabling odor, taste, and sensory irritation or certain asymptomatic, nonsensory effects. With increasing airborne concentrations above each AEGL, there is a progressive increase in the likelihood of occurrence and the severity of effects described for each corresponding AEGL. Although the AEGL values represent threshold concentrations for the general public, including susceptible subpopulations, such as infants, children, the elderly, persons with asthma, and those with other illnesses, it is recognized that individuals, subject to idiosyncratic responses, could experience the effects described at concentrations below the corresponding AEGL.
SUMMARY
Ethyl mercaptan is an odorous, colorless liquid. The disagreeable odor has been described as penetrating, persistent, and garlic- or leek-like, similar to decaying cabbage. It is found in illuminating gas, in “sour” gas in West Texas oil fields, and in petroleum distillates from which it may be separated by chemical or physical methods. It is used as an intermediate and starting material in the manufacture of plastics, insecticides, and antioxidants, and as an odorant to serve as a warning property for natural gas (O’Neil et al. 2006).
Ethyl mercaptan depresses the central nervous system and affects the respiratory center, similar to hydrogen sulfide, producing death by respiratory paralysis. Clinical signs of exposure are ocular and mucous membrane irritation, headache, dizziness, staggering gait, nausea, and vomiting. Paralysis of locomotor muscles has also been observed. Its primary mechanism of action appears to be interference with cytochrome oxidase.
AEGL-1 values for ethyl mercaptan were based on a no-effect level of 10 ppm for respiratory changes associated with odor avoidance in rabbits exposed for 20 min (Shibata 1966a). Two uncertainty factors of 3 were applied to account for interspecies differences and intraspecies variability, and are considered sufficient because use of the full factor of 10 for either type of uncertainty would yield AEGL-1 values of 0.3 ppm or less, concentrations that are inconsistent with human data. A single AEGL-1 value was used across exposure durations
because prolonged exposure to ethyl mercaptan is unlikely to result in an enhanced effect.
The level of distinct odor awareness (LOA) for ethyl mercaptan is 1.4 x10-4 ppm (see Appendix C for LOA derivation). The LOA represents the concentration above which it is predicted that more than half of the exposed population will experience at least a distinct odor intensity, and about 10% of the population will experience a strong odor intensity. Because of its relatively high vapor pressure (442 mm Hg at 20ºC) (NIOSH 2011), ethyl mercaptan has the potential to generate toxic air concentrations very quickly in the event of a spill. The LOA should help chemical emergency responders assess the public awareness of exposure to ethyl mercaptan from its odor.
No robust data on ethyl mercaptan consistent with the definition of AEGL-2 were available. Therefore, the AEGL-2 values for ethyl mercaptan were based on a 3-fold reduction in the AEGL-3 values. This calculation is considered an estimate of a threshold for irreversible effects and is appropriate because of the steep concentration-response curve for ethyl mercaptan toxicity.
AEGL-3 values are based on a calculated 4-h LC01 (lethal concentration, 1% lethality) of 2,250 ppm in mice (Fairchild and Stokinger 1958). The corresponding 4-h LC01 value for rats is 3,808 ppm. An intraspecies uncertainty factor of 3 was applied, and is considered sufficient because of the steepness of the lethality concentration-response curve which implies limited individual variability. An interspecies uncertainty factor of 3 was also applied because the limited data suggest that the mouse is the most sensitive species. Although an interspecies uncertainty factor of 10 might normally be applied because of the limited data, a total uncertainty factor of 30 would yield AEGL-3 values that are inconsistent with the total data set (the values would be in the range of AEGL-3 values for hydrogen sulfide [NRC 2010]). Furthermore, the 30-min AEGL-3 value would be 150 ppm, a value that is inconsistent with the finding that a single human exposed to ethyl mercaptan at 112 ppm for 20 min exhibited only a slightly irregular and decreased breathing rate (Shibata 1966b). Thus, the total uncertainty factor is 10. The 30-min AEGL-3 value was adopted as the 10-min value because of the uncertainty associated with extrapolating a 4-h point of departure to a 10-min value.
AEGL values for ethyl mercaptan are presented in Table 1-1.
1. INTRODUCTION
Ethyl mercaptan is used as an intermediate and starting material in the manufacture of plastics, insecticides, and antioxidants, and as an odorant to serve as a warning property for natural gas (O’Neil et al. 2006).
Ethyl mercaptan is an odorous, colorless liquid. The disagreeable odor has been described as penetrating, persistent, and garlic- or leek-like, similar to decaying cabbage (O’Neil et al. 2006). It is found in illuminating gas, in “sour” gas in West Texas oil fields, and in petroleum distillates from which it may be separated by chemical or physical methods (O’Neil et al. 2006).
TABLE 1-1 AEGL Values for Ethyl Mercaptan
| Classifiaction | 10 min | 30 min | 1 h | 4 h | 8 h | End point (Reference) |
| AEGL-1 (nondisabling) | 1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
No-effect level for respiratory changes associated with odor avoidance in rabbits (Shibata 1966a). |
| AEGL-2 (disabling) | 150 ppm (380 mg/m3) | 150 ppm (380 mg/m3) | 120 ppm (310 mg/m3) | 77 ppm (200 mg/m3) | 37 ppm (94 mg/m3) | 3-fold reduction of AEGL-3 values. |
| AEGL-3 (lethal) | 450 ppm (1,100 mg/m3) | 450 ppm (1,100 mg/m3) | 360 ppm (910 mg/m3) | 230 ppm (580 mg/m3) | 110 ppm (280 mg/m3) | LC01 in mice (Fairchild and Stokinger 1958). |
Abbreviation: LC01, lethal concentration, 1% lethality.
Ethyl mercaptan is produced commercially by the reaction of sodium ethyl sulfate with potassium hydrosulfide, or catalytically from ethanol and hydrogen sulfide (O’Neil et al. 2006). The total production of methane, ethane, propane, butane, octane, nonane, decane, hexadecane, and miscellaneous thiols was 264,797,000 pounds in 1976, and an estimated 23,130 U.S. workers were exposed to ethyl mercaptan from 1972-1974 (NIOSH 1978).
The physical and chemical properties of ethyl mercaptan are presented in Table 1-2. Because of its relatively high vapor pressure (442 mm Hg at 20ºC), ethyl mercaptan has the potential to generate toxic air concentrations very quickly in the event of a spill.
2. HUMAN TOXICITY DATA
2.1. Acute Lethality
No information concerning human lethality from acute exposure to ethyl mercaptan was found.
2.2. Nonlethal Toxicity
2.2.1. Odor Threshold and Odor Awareness
Katz and Talbert (1930) conducted two trials, each exposing six human subjects to a range of ethyl mercaptan concentrations via a nosepiece. The subjects described the odor as that of decayed cabbage and very disagreeable. A description of the odor intensity of ethyl mercaptan is presented in Table 1-3. No ocular or nasal irritation was reported in subjects exposed to ethyl mercaptan at concentrations up to 1,000 ppm for less than 10 seconds.
TABLE 1-2 Physical and Chemical Data for Ethyl Mercaptan
|
Parameter |
Value |
Reference |
|
Synonyms |
Ethanethiol; ethyl sulfhydrate; |
HSDB 2011 |
|
CAS registry no. |
75-08-1 |
HSDB 2011 |
|
Chemical formula |
C2H5SH |
HSDB 2011 |
|
Molecular weight |
62.14 |
HSDB 2011 |
|
Physical state |
Colorless liquid |
O’Neil et al. 2006 |
|
Odor |
Garlic-, leek-, or skunk-like |
O’Neil et al. 2006; |
|
Melting point |
-147.8°C |
HSDB 2011 |
|
Boiling point |
35.1°C |
HSDB 2011 |
|
Flash point |
-48.3°C (closed cup) |
HSDB 2011 |
|
Density/Specific gravity |
0.8315 at 25°C |
HSDB 2011 |
|
Solubility |
15,603 mg/L at 25°C in water, |
HSDB 2011 |
|
Saturated vapor concentration (neat) |
7.0 × 105 ppm |
Calculated |
|
Vapor pressure |
442 mm Hg at 20°C |
HSDB 2011 |
|
Incompatibility |
Strong oxidizers |
NIOSH 2011 |
|
Conversion factors in air |
1 mg/m3 = 0.39 ppm |
NIOSH 2011 |
TABLE 1-3 Odor Intensity of Ethyl Mercaptan
| Intensity | Description | Concentration (ppm) | |
| Trial 1 | Trial 2 | ||
| 0 | No odor | 2.1 ×10-5 | 6.0 × 10-6 |
| 1 | Detectable | 9.7 × 10-4 | 2.6 × 10-4 |
| 2 | Faint | 4.5 × 10-2 | 1.1 × 10-2 |
| 3 | Median, easily noticeable | 2.1 × 100 | 4.9 × 10-1 |
| 4 | Strong | 9.7 × 101 | 2.1 × 101 |
| 5 | Most intense | 4.5 × 103 | 9.20 × 102 |
Source: Adapted from Katz and Talbert 1930.
Wilby (1969) exposed three individuals to ethyl mercaptan at 12 concentrations representing a 100-fold range. An odor recognition threshold was determined for each subject on the basis of three trials. The mean odor-threshold concentration for ethyl mercaptan was 4.0 × 10-4 ppm, with a standard deviation of 2.6 × 10-4 ppm and a coefficient of variation of 0.65. No other effects were noted.
Blinova (1965) conducted a series of experiments whereby a total of nine human subjects inhaled ethyl mercaptan through a mask connected to a 1,000-L chamber in which a known concentration of ethyl mercaptan had been established. No other information on atmosphere generation or analytic methods was provided. The concentration range of minimum perceptible odor was 2.2 × 10-3 to 1.1 × 10-2 ppm, and the range of imperceptible odor (olfactory fatigue) was reported as 1.8 × 10-3 to 7.2 × 10-3 ppm. Other experimental protocols and results from this study are summarized in Table 1-4.
NIOSH (1978) cites an Italian study wherein humans (no details provided) experienced olfactory fatigue and mucosal irritation during experimental exposure to ethyl mercaptan at 4 ppm (1 mg/m3) for 3 h/day for 5 days. These effects were transient with cessation of exposure. Subjects exposed at 0.4 ppm did not experience these effects (Gobbato and Terribile 1968). This Italian-language study provides support for the effect levels reported by Blinova (1965).
Amoore and Hautala (1983) reported an odor threshold of 7.6 × 10-4 ppm for ethyl mercaptan. This value is the geometric mean calculated from reliable published odor threshold values.
Nagata (2003) reported an odor threshold of 8.7 × 10-6 ppm for ethyl mercaptan. This value was determined by a validated method and included a butanol standard for comparison. Therefore, it is considered most appropriate for calculation of the level of distinct odor awareness (LOA).
TABLE 1-4 Effects of Ethyl Mercaptan in Humans
| Concentration (ppm) | Duration | Subjects | Effects |
| 4.0 | 3 h/d for 10 d | 1 female | Odor, olfactory fatigue, mucosal irritation |
| 0.4 | 3 h/d for 10 days (one month after above exposure) | 1 female | None |
| 4.0 | 3 h/d for 5 d | 2 subjects (sex not reported) | Odor, olfactory fatigue, mucosal irritation |
| 4.0 | 3 h/d for 5 d (one month after above exposure) | 2 subjects (sex not reported) | Same as above, but less pronounced |
| 0.4 | 3 h/d for 5 d | 2 subjects (sex not reported) | None |
| 0.4 | 3 h/d for 5 d (one month after above exposure) | 2 subjects (sex not reported) | None |
Source: Blinova 1965.
The LOA for ethyl mercaptan is 1.4 × 10-4 ppm (see Appendix C for LOA derivation). The LOA represents the concentration above which it is predicted that more than half of the exposed population will experience at least a distinct odor intensity, and about 10% of the population will experience a strong odor intensity. The LOA should help chemical emergency responders in assessing the public awareness of the exposure to ethyl mercaptan from its odor; however, the potential for odor fatigue should also be considered (Shertzer 2012).
2.2.2. Case Report
Twenty-eight male and two female high school students (16- to 18-years old), whose classroom was connected by a door to a chemical storeroom, were accidentally exposed to ethyl mercaptan vapor during morning classes (Pichler 1918). The class was dismissed approximately 1 h after the students began complaining about a bad odor emanating from the adjacent room. Ten students (eight male and two female) complained of dull headache, general discomfort, and abdominal pain, and three students vomited and had diarrhea. All symptoms resolved by the afternoon, and the students reportedly slept normally that night. The class met in the same room the next day for 3 h. Even though the classroom and storeroom had been ventilated, eight of the students with symptoms the previous day developed headaches, but to a lesser degree. Two of the students did not return to school for several days. Examination of one male student showed “changes” around the eyes and a palpable liver, and protein, erythrocytes, and a few leukocytes were detected in the urine. There were no epithelial cells or casts in the urine and the other urinary parameters returned to normal within 5-6 weeks. It was estimated that 3 g of ethyl mercaptan had vaporized in 325-m3 rooms resulting in an approximate concentration of 4 ppm.
2.2.3. Experimental Study
Shibata (1966b) exposed two adult men to ethyl mercaptan at 50 ppm for 20 min and one adult man to 112 ppm for 20 min. Respiration frequency, pulse rate, and blood pressure were monitored continuously for 10 min before and throughout exposure. In one subject exposed at 50 ppm, breathing frequency decreased immediately with exposure and returned to the pre-exposure inhalation rate after termination of exposure. The second subject exposed at 50 ppm experienced no change in breathing rate. The subject exposed at 112 ppm had a slightly irregular and decreased breathing rate. Minute volume and tidal volume increased in all three subjects. Pulse rate increased slightly in only one subject (50 ppm), and there was no effect on blood pressure and no electrocardiographic abnormalities in any subject. The only subjective response was odor recognition only during the first few breaths, suggesting that olfactory fatigue and accommodation occurred.
2.3. Developmental and Reproductive Toxicity
Developmental and reproductive studies of human exposure to ethyl mercaptan were not available.
2.4. Genotoxicity
Genotoxicity studies of human exposure to ethyl mercaptan were not available.
2.5. Carcinogenicity
Carcinogenic studies of human exposure to ethyl mercaptan were not available.
2.6. Summary
Data on human exposure to ethyl mercaptan are limited. Case reports of deaths from accidental exposure to ethyl mercaptan were not available. Nonlethal toxicity data include a case report where high school students accidentally exposed to ethyl mercaptan experienced reversible dull headache, general discomfort, abdominal pain, vomiting, and diarrhea. Other data included odor-detection (identification) and olfactory-fatigue data but no accompanying health effects information, and a study showing slight changes in breathing rate in three individuals exposed to ethyl mercaptan for 20 min. Atmospheric generation and exposure concentration parameters were not described in detail for any of the human studies. Data on developmental and reproductive toxicity, genotoxicity, and carcinogenicity in humans were not available.
3. ANIMAL TOXICITY DATA
3.1. Acute Lethality
3.1.1. Mice
Fairchild and Stokinger (1958) exposed groups of 10 Swiss-derived male mice (body weight 25-28 g) to ethyl mercaptan at 2,600, 3,150, 3,573, 4,438, or 4,832 ppm for 4 h, followed by a 15-day observation period. Vapor generation was achieved by either bubbling a stream of nitrogen gas through a midget fritted-glass bubbler, which contained liquid ethyl mercaptan, or by passage of nitrogen into a borosilicate glass nebulizer containing the ethyl mercaptan. Target concentrations were maintained in an 18-L glass chamber by varying the ratio of volume flow of compressed air and compressed nitrogen. Ethyl mercaptan concentrations during exposure periods were measured by absorption of vapors in
either isopropyl alcohol or acetone containing an excess of silver nitrate and titrating the uncombined silver amperometrically. Chamber concentrations during tests were uniform after the first 30 min; mean variation for all exposures was approximately 4%. Clinical signs included increased respiration and restlessness (hyperactivity), uncoordinated movement, staggering gait, muscular weakness, partial skeletal muscle paralysis beginning in the hind limbs, light to severe cyanosis, tolerance of a prone position, and mild to heavy sedation. Animals exposed to “maximal lethal concentrations” typically died from respiratory arrest during exposure or shortly after removal from the chamber. Animals exposed to “minimal lethal concentrations” typically died while in a semiconscious condition of “long duration”. Surviving animals often remained in a semiconscious state of sedation and lethargy for 4- to 6-h post-exposure before showing signs of recovery. An LC50 value (lethal concentration, 50% lethality) of 2,770 ppm, LC05 value (lethal concentration, 5% lethality) of 2,498 ppm, and LC01 value of 2,250 ppm were calculated by the method of Litchfield and Wilcoxon (1949). A BMC01 (benchmark concentration with 1% response) of 1,921 ppm and BMCL05 (benchmark concentration, 95% lower confidence limit with 5% response) of 1,545 ppm were also calculated. Mortality data are summarized in Table 1-5.
3.1.2. Rats
Fairchild and Stokinger (1958) exposed groups of five or six Wistar-derived male rats (body weight 180-220 g) to ethyl mercaptan at 2,600, 3,150, 3,573, 4,438, 4,832, 4,868, 5,100, or 5,125 ppm for 4 h, followed by a 15-day observation period. Vapor generation and test chamber analysis is similar to that described for studies in mice (see Section 3.1.1). Clinical signs included increased respiration and restlessness (hyperactivity), incoordinated movement, staggering gait, muscular weakness, partial skeletal muscle paralysis beginning in the hind limbs, light to severe cyanosis, tolerance of a prone position, and mild to heavy sedation. Animals exposed to “maximal lethal concentrations” typically died from respiratory arrest during exposure or shortly after removal from the chamber. Animals exposed to “minimal lethal concentrations” typically died while in a semiconscious condition of “long duration”. Surviving animals often remained in a semiconscious state of sedation and lethargy for 4- to 6-h post-exposure before showing signs of recovery. An LC50 value of 4,420 ppm, LC05 value of 4,120 ppm, and LC01 value of 3,808 ppm were calculated by the method of Litchfield and Wilcoxon (1949). Mortality data are summarized in Table 1-5.
Fairchild and Stokinger (1958) also administered ethyl mercaptan by oral gavage or intraperitoneal injection to Wistar-derived male rats, followed by 15-day observation periods. An oral LD50 (lethal dose, 50% mortality) of 682 mg/kg and an intraperitoneal LD50 of 226 mg/kg were reported.
TABLE 1-5 Mortality in Mice and Rats Exposed to Ethyl Mercaptan for 4 Hours
|
Concentration (ppm) |
Mice |
Rats |
|
2,600 |
4/10 |
0/5 |
|
3,150 |
7/10 |
0/5 |
|
3,573 |
10/10 |
0/5 |
|
4,438 |
10/10 |
1/5 |
|
4,832 |
10/10 |
4/6 |
|
4,868 |
– |
2/5 |
|
5,100 |
– |
5/5 |
|
5,125 |
– |
2/6 |
|
LC01 |
2,250 ppm |
3,808 ppm |
|
LC05 |
2,498 ppm |
4,120 ppm |
|
LC50 |
2,770 ppm |
4,420 ppm |
Source: Adapted from Fairchild and Stokinger 1958.
3.2. Nonlethal Toxicity
3.2.1. Rats
Groups of three to five male Holtzman or Sprague-Dawley rats (weighing 285-325 g) were individually exposed in a 4-L glass desiccator to ethyl mercaptan at concentrations of 2.7-3.8% (approximately 27,000-38,000 ppm) for 15 min or less (Zieve et al. 1974). The target concentrations were achieved by injecting the required amount of ethyl mercaptan through a rubber septum in the lid of the chamber. The concentration of ethyl mercaptan in the chamber atmosphere was not analyzed, rather concentrations were calculated from the dose injected. A CD50 value (concentration causing coma induction in 50% of animals, as measured by complete loss of the righting reflex) of 3.3% (33,000 ppm) was determined. No rats lost the righting reflex at ethyl mercaptan concentrations of about 3.0% (30,000 ppm), but all rats lost the righting reflex at about 3.7% (37,000 ppm). The rats exhibited a brief excitement phase before becoming “groggy”. At the CD50, the excitement phase lasted about 2 min, the groggy and lethargic phase lasted about 1 min, and finally frank coma ensued within 1 to 2 min. At lower concentrations, the excitement and pre-coma phases were prolonged and at higher concentrations, the entire sequence occurred more quickly. When rats were removed from exposure immediately after becoming comatose, the coma generally did not last more than 30 min and the rats appeared and remained alert and active on recovery. Blood concentrations of ethyl mercaptan found in comatose animals were greater than 200 nmoles/mL; however, there was no clear concentration-response relationship between inhaled concentrations and blood levels.
No mortality was observed in rats exposed head only to ethyl mercaptan at 991 ppm for 4 h or in rats exposed whole body at 27 ppm for 4 h (Shertzer 2012).
3.2.2. Rabbits
Shibata (1966a) exposed groups of two male rabbits (weighing 3 kg) to ethyl mercaptan at 10, 100, or 1,000 ppm by breathing mask for 20 min. Breathing rate (measured by observed thorax movement) and minute expiratory volume (measured by wet spirometry) were monitored throughout the exposure periods. Tidal volume was then calculated by dividing the minute expiratory volume by the breathing rate. At 100 and 1,000 ppm, respiratory rate and expiratory volume were decreased and tidal volume was increased. Approximate changes in respiratory function parameters (estimated from graphs) at the end of the exposure period in the 1,000-ppm group were: 20% decrease in expiratory volume, 40% decrease in respiratory rate, and 40% increase in tidal volume. Approximate changes in the 100 ppm group were: 10% decrease in expiratory volume, 10% decrease in respiratory rate, and 20% increase in tidal volume. The respiratory changes in rabbits at 1,000 and 100 ppm for 20 min are suggestive of odor avoidance. At 10 ppm, respiratory rate and ventilation rate showed unstable fluctuation and tidal volume was increased slightly during the last half of the exposure period. All respiratory indicators returned to pre-exposure levels by the end of the 35-min observation period, except for the respiratory rate of animals exposed at 1,000 ppm, which was still decreased by approximately 25%. The changes in breathing rate and tidal volume in rabbits exposed to ethyl mercaptan at 100 ppm or higher for 20 min are similar to the effects reported in the human study by the same investigator (Shibata 1966b). However, the authors note (Shibata 1966b, translated by OPPT):
It is difficult to compare the rabbit study and human study. The rabbit’s body weight is about one-twentieth that of humans and the expiratory volume is about one-ninth that of humans; therefore, rabbit’s expiratory volume per body weight is greater than humans, which indicates that the rabbit’s inspiratory volume is about twice as much as that of humans if they are exposed to the same concentration of gas. Therefore rabbits would be affected more than humans. The differences in respiratory center sensitivity of rabbits and humans should also be considered as well.
Fairchild and Stokinger (1958) instilled ethyl mercaptan (0.1mL) into the conjunctival sac of the right eye of one male New Zealand white rabbit. The left eye served as a control. Slight to moderate irritation was observed and resolved within 48 h.
3.3. Repeated-Exposure Study
Shibata ((1966a) exposed four male rabbits (weighing 3 kg) to ethyl mercaptan at 1,000 ppm by breathing mask for 20 min/day for 9 days in a 10-day period. No significant treatment-related effects on urinary sulfate, urine volume, erythrocyte or leukocyte counts, or body weight were found.
3.4. Developmental and Reproductive Toxicity
Developmental or reproductive toxicity studies of animal exposure to ethyl mercaptan were not available.
3.5. Genotoxicity
Ethyl mercaptan was negative in an Ames Salmonella typhimurium assay (Hazleton Laboratories 1984). It was positive in a sister-chromatid-exchange assay in cultured Chinese hamster ovary cells with or without metabolic activation (Hazleton Laboratories 1984), and in a forward mutation assay in cultured mouse lymphoma cells without activation (Hazleton Laboratories 1983).
3.6. Carcinogenicity
Carcinogenicity studies of ethyl mercaptan in animals were not available.
3.7. Summary
Animal toxicity data for ethyl mercaptan are limited. Lethality studies are available for rats and mice, and suggest a steep concentration-response curve for ethyl mercaptan. For example, lethality was 40% and 100% in rats exposed for 4 h to ethyl mercaptan at 2,600 ppm and 3,573 ppm, respectively. The 4-h LC50 value for rats was 4,420 ppm, and the 4-h LC01 value was 3,808 ppm. In mice, the 4-h LC50 value was 2,770 ppm, and the 4-h LC01 value was 2,250 ppm (Fairchild and Stokinger 1958). Clinical observations were indicative of central nervous system depression and respiratory arrest, and included changes in respiration, restlessness (hyperactivity), incoordinated movement, staggering gait, muscular weakness, skeletal muscle paralysis, light to severe cyanosis, and coma. Coma-induction data also suggest a steep concentration-response curve for ethyl mercaptan. No coma-induction was observed in rats exposed at 30,000 ppm for up to 15 min, but coma was induced in 50% of rats exposed at 33,000 ppm and 100% of rats exposed at 37,000 ppm (Zieve et al. 1974). The limited genotoxicity data are equivocal. No reproductive or developmental toxicity data or carcinogenicity studies were available.
4. SPECIAL CONSIDERATIONS
4.1. Metabolism and Disposition
Snow (1957) demonstrated that ethyl mercaptan was rapidly absorbed and distributed evenly throughout the body tissues of mice and guinea pigs after oral or subcutaneous administration. Excretion occurred mainly via the kidney as inorganic sulfate. Organic metabolites, ethyl methyl sulfone, and an unidentified product accounted for 10-20% of the sulfur excreted in the urine. There was little fecal excretion, but approximately 14% of the dose was excreted in the breath. It was hypothesized that oxidation converted the thiol to the sulphide and then to the sulfone.
Ethyl mercaptan is a metabolite of the human body and is excreted in the breath of normal individuals; patients with advanced liver disease excrete it at higher concentrations. Chen et al. (1970) measured ethyl mercaptan in the breath of normal subjects and in patients with liver cirrhosis or in hepatic coma after fasting and after ingestion of methionine (8-12 g),. Concentrations in the breath were 1.1-12.3 ng/L in seven normal, fasting subjects, and increased about 1.5fold after daily ingestion of methionine for 7 days. In cirrhotic patients, the average ethyl mercaptan concentration was 11.5 ng/L. After ingestion of methionine, there was no significant increase in the amount of ethyl mercaptan in the breath of patients with liver disease.
4.2. Mechanism of Toxicity
Ethyl mercaptan acts similar to hydrogen sulfide and cyanide by interrupting electron transport through inhibition of cytochrome oxidase. Ethyl mercaptan decreased Na,K-ATPase in the rat brain (Foster et al. 1974). Vahlkamp et al. (1979) investigated the effects of ethyl mercaptan in vitro in isolated rat hepatocytes, isolated mitochondria from rat liver and brain, and submitochondrial particles from ox heart. Ethyl mercaptan inhibited gluconeogenesis and ureogenesis from various substrates in rat hepatocytes, decreased cellular ATP content, and caused an increase in the reduction state of mitochondria. It also inhibited respiration in rat liver mitochondria with several substrates, in the presence of ADP and phosphate or in the presence of an uncoupling agent, and inhibited respiration in rat brain mitochondria. In submitochondrial particles of ox heart, ethyl mercaptan inhibited electron transfer between cytochrome c and oxygen, and purified cytochrome c oxidase was inhibited by ethyl mercaptan in a non-competitive manner.
As a result of the electron transfer blockage, oxidative phosphorylation and aerobic metabolism are compromised, peripheral tissue PO2 increases, and the unloading gradient for oxyhemoglobin decreases. High concentrations of oxyhemoglobin are thus found in the venous return, resulting in flushed skin and mucous membranes. Lactic acidemia occurs as a result of the increased demand placed on glycolysis.
4.3. Structure-Activity Relationships
Rat lethality data suggest that the acute toxicity of ethyl mercaptan is much less than that of methyl mercaptan (approximately 6-fold lower) or hydrogen sulfide (approximately 10-fold lower) (see Table 1-6). For example, the 4-h LC50 value for ethyl mercaptan was 4,420 ppm, whereas the corresponding values for methyl mercaptan and hydrogen sulfide were 675 ppm and 444 ppm, respectively (Tansy et al. 1981).
TABLE 1-6 Comparative Toxicity of Selected Mercaptans
| Compound | Rat Intraperitoneal LD50 (mg/kg) | Rat Oral LD50 (mg/kg) | 4-h Inhalation LC50 (ppm) | Reference | |
| Rats | Mice | ||||
| Hydrogen sulfide | – | – | 444 | – | Tansy et al. 1981 |
| Methyl mercaptan | – | – | 675 | 1,664 | Horiguchi 1960 (mice); Tansy et al. 1981 (rats) |
| Ethyl mercaptan | 226 | 682 | 4,420 | 2,770 | Fairchild and Stokinger 1958 |
| Propyl mercaptan | 515 | 1,790 | 7,200 | 4,010 | Fairchild and Stokinger 1958 |
| Isobutyl mercaptan | 917 | 7,168 | >25,000 | >25,000 | Fairchild and Stokinger 1958 |
| tert-Butyl mercaptan | 590 | 4,729 | 22,200 | 16,500 | Fairchild and Stokinger 1958 |
| n-Butyl mercaptan | 399 | 1,500 | 4,020 | 2,500 | Fairchild and Stokinger 1958 |
| n-Hexyl mercaptan | 396 | 1,254 | 1,080 | 528 | Fairchild and Stokinger 1958 |
| Phenyl mercaptan | 9.8 | 46.2 | 33 | 28 | Fairchild and Stokinger 1958 |
| Benzyl mercaptan | 373 | 493 | >235 | 178 | Fairchild and Stokinger 1958 |
| tert-Octyl mercaptan | 12.9 | 83.5 | 51 (males) | 47 (males) | Fairchild and Stokinger 1958 |
4.4. Concurrent Exposure Issues
Because cyanide, hydrogen sulfide, methyl mercaptan, and ethyl mercaptan are all cytochrome oxidase inhibitors, an interaction might be possible if individuals were simultaneously exposed to two or more or more of these chemicals (Smith 1991). Such interactions could result in lower lethal exposure concentrations for ethyl mercaptan.
Ethyl mercaptan may also have a role in facilitating the toxic effects of ammonia and fatty acids relative to hepatic failure in humans (Zieve et al. 1974).
4.5. Species Differences
Because of the limited data available on ethyl mercaptan, a definitive assessment of species variability is not possible. However, the data suggest that mice are approximately 1.6-fold more sensitive than rats to lethality from inhalation exposure to ethyl mercaptan.
4.6. Concentration-Exposure Duration Relationship
The concentration-exposure time relationship for many irritant and systemically-acting vapors and gases may be described by the equation Cn × t = k, where the exponent n ranges from 0.8 to 3.5 (ten Berge et al. 1986). Data were inadequate to empirically derive a chemical-specific scaling exponent for ethyl mercaptan. So, temporal scaling was performed using default values of n = 3 for extrapolation to shorter durations and n = 1 for extrapolation to longer durations. See Appendix A.
5. DATA ANALYSIS FOR AEGL-1
5.1. Human Data Relevant to AEGL-1
One of two adult males exposed to ethyl mercaptan at 50 ppm for 20 min had decreased respiratory frequency (Shibata 1966b). Mucosal irritation occurred in one female exposed at 4 ppm for 3 h/day for 10 days and in two males exposed at 4 ppm for 3 h/day for 5 days (Blinova 1965); no effects were reported at 0.4 ppm. These findings are supported by the study of Gobbato and Terribile (1968). That study reported that mucosal irritation and olfactory fatigue returned to normal after cessation of exposure.
5.2. Animal Data Relevant to AEGL-1
Rabbits exposed to ethyl mercaptan at 100 or 1,000 ppm for 20 min exhibited decreased respiratory rate and expiratory volume, indicative of odor avoidance, but no significant effects was observed at 10 ppm (Shibata 1966a).
5.3. Derivation of AEGL-1 Values
AEGL-1 values for ethyl mercaptan were based on a no-effect level of 10 ppm for respiratory changes associated with odor avoidance in rabbits exposed for 20 min (Shibata 1966a). Two uncertainty factors of 3 were applied to account for interspecies differences and intraspecies variability. These values are sufficient because a full factor of 10 for either uncertainty would yield AEGL-1 values of 0.3 ppm or less, concentrations that are inconsistent with human data. A single AEGL-1 value was used for all exposure durations because prolonged exposure to ethyl mercaptan is not expected to result in an enhanced effect. AEGL-1 values for ethyl mercaptan are presented in Table 1-7, and calculations are presented in Appendix A.
The AEGL-1 value of 1 ppm may appear to be too low in the context of the acute lethality information on ethyl mercaptan (see Table 1-6). However, the respiratory effects on which the AEGL-1 values are based appear be the result of odor avoidance rather than evidence of early effects that lead to lethality. In this case, comparison with LOA values is more applicable than a comparison with 4-h LC50 values in rats. The LOA for ethyl mercaptan is 1.4 × 10-4 ppm. The LOA for the related chemical methyl mercaptan (1.9 × 10-2 ppm) is greater than the LOA for ethyl mercaptan, which supports AEGL-1 values for ethyl mercaptan (1 ppm) being lower than the AEGL-1 values for methyl mercaptan (11 pm, see Chapter 2). Even though ethyl mercaptan has an extremely unpleasant odor, olfactory desensitization or olfactory fatigue may occur at high concentrations. Therefore, odor and symptoms of irritation may not adequately provide warning of high concentrations of ethyl mercaptan (Shertzer 2012).
6. DATA ANALYSIS FOR AEGL-2
6.1. Human Data Relevant to AEGL-2
Ten students complained of dull headache, general discomfort, and abdominal pain, and three students vomited and had diarrhea after accidentally being exposed to ethyl mercaptan at school (Pichler 1918). However, no definitive information on concentration or duration of exposure were available.
6.2. Animal Data Relevant to AEGL-2
No animal data on ethyl mercaptan relevant for deriving AEGL-2 values were available.
TABLE 1-7 AEGL-1 Values for Ethyl Mercaptan
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
6.3. Derivation of AEGL-2 Values
No relevant inhalation data on ethyl mercaptan consistent with the definition of AEGL-2 were available. Therefore, the AEGL-2 values were based on a one-third reduction in the AEGL-3 values, which this is considered an estimate of a threshold for inability to escape and is considered appropriate because of the steep concentration-response curve for ethyl mercaptan (see Table 1-5; NRC 2001). The AEGL-2 values are supported by the 15-min no-effect level of 30,000 ppm for loss of righting reflex in rats reported by by Zieve et al. (1974), and a 20-min no-effect level of 112 ppm reported for a single human (Shibata 1966b). AEGL-2 values for ethyl mercaptan are presented in Table 1-8, and calculations are presented in Appendix A.
7. DATA ANALYSIS FOR AEGL-3
7.1. Human Data Relevant to AEGL-3
No human data on ethyl mercaptan relevant to AEGL-3 values were available.
7.2. Animal Data Relevant to AEGL-3
A 4-h LC50 value of 2,770 ppm, LC05 value of 2,498 ppm, and LC01 value of 2,250 ppm were calculated for mice, and a 4-h LC50 value of 4,420 ppm, LC05 value of 4,120 ppm, and LC01 value of 3,808 ppm were calculated for rats (Fairchild and Stokinger 1958).
7.3. Derivation of AEGL-3
The LC01 of 2,250 ppm in mice exposed to ethyl mercaptan for 4 h (Fairchild and Stokinger 1958) was used to derive AEGL-3 values. The mouse data were chosen over the rat data because mice are more sensitive (LC01, LC05, and LC50 estimates were lower in mice than in rats; see Table 1-5) and because more mice were tested.
An intraspecies uncertainty factor of 3 was considered sufficient to account for intraindividual variability because of the steep concentration-response curve for lethality (see Table 1-5), which implies limited individual variability. An interspecies uncertainty factor of 3 was also be applied because the limited data suggest that the mouse is the most sensitive species (see Section 4.5). Although an interspecies uncertainty factor of 10 might normally be applied because of the limited data, a total uncertainty factor of 30 would yield AEGL-3 values approaching or equivalent to the AEGL-3 values for hydrogen sulfide. For example, if a total uncertainty factor of 30 is applied, an 8-h
TABLE 1-8 AEGL-2 Values for Ethyl Mercaptan
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 150 ppm (380 mg/m3) |
150 ppm (380 mg/m3) |
120 ppm (310 mg/m3) |
77 ppm (200 mg/m3) |
37 ppm (94 mg/m3) |
AEGL-3 value for ethyl mercaptan would be 37 ppm, which is slightly higher than the 8-h AEGL-3 value for hydrogen sulfide of 31 ppm (NRC 2010). Because a robust database exists for hydrogen sulfide and because data suggest that ethyl mercaptan is less toxic than hydrogen sulfide (see Table 1-6), it would be inconsistent with the total data set to have AEGL-3 values for ethyl mercaptan that are in the range of the AEGL-3 values for hydrogen sulfide. Furthermore, use of a total uncertainty factor of 30 would yield a 30-min AEGL-3 value of 150 ppm, which is inconsistent with the finding that a single human exposed to ethyl mercaptan at 112 ppm for 20 min exhibited only a slightly irregular and decreased breathing rate (Shibata 1966b). The conservative AEGL-3 values for short exposure periods are supported by the Zieve et al. (1974) study, wherein ethyl mercaptan at 30,000 ppm for 15 min produced no loss of righting reflex in rats and 33,000 ppm produced loss of righting reflex in 100% of rats. Thus, the total uncertainty factor is 10.
The concentration-time relationship for many irritant and systemically-acting vapors and gases is described in Section 4.6. The 30-min AEGL-3 value is adopted as the 10-min value because of the uncertainties associated with extrapolating a 4-h exposure to a 10-min value. AEGL-3 values for ethyl mercaptan are presented in Table 1-9, and calculations are presented in Appendix A.
8. SUMMARY OF AEGLS
8.1. AEGL Values and Toxicity End Points
Table 1-10 summarizes the AEGL values for ethyl mercaptan. AEGL-1 values were based on a no-effect level for respiratory changes associated with odor avoidance in rabbits exposed for 20 min. In the absence of relevant data and because of the steep concentration-response curve, AEGL-2 values were calculated as one-third of the AEGL-3 values. These calculations are estimated thresholds for the inability to escape. AEGL-3 values are based on the LC01 of 2,250 ppm in mice exposed to ethyl mercaptan for 4 h (Fairchild and Stokinger 1958).
8.2. Comparisons with Other Standards and Guidelines
Standards and guidance levels for workplace and community exposures to ethyl mercaptan are presented in Table 1-11.
TABLE 1-9 AEGL-3 Values for Ethyl Mercaptan
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 450 ppm (1,100 mg/m3) |
450 ppm (1,100 mg/m3) |
360 ppm (910 mg/m3) |
230 ppm (580 mg/m3) |
110 ppm (280 mg/m3) |
TABLE 1-10 AEGL Values for Ethyl Mercaptan
| Classification | 10 min | 30 min | 1 h | 4 h | 8 h |
| AEGL-1 (nondisabling) | 1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
| AEGL-2 (disabling) | 150 ppm (380 mg/m3) |
150 ppm (380 mg/m3) |
120 ppm (310 mg/m3) |
77 ppm (200 mg/m3) |
37 ppm (94 mg/m3) |
| AEGL-3 (lethal) | 450 ppm (1,100 mg/m3) |
450 ppm (1,100 mg/m3) |
360 ppm (910 mg/m3) |
230 ppm (580 mg/m3) |
110 ppm (280 mg/m3) |
TABLE 1-11 Standards and Guidelines for Ethyl Mercaptan
| Exposure Duration | |||||
| Guideline | 10 min | 30 min | 1 h | 4 h | 8 h |
| AEGL-1 | 1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
| AEGL-2 | 150 ppm (380 mg/m3) |
150 ppm (380 mg/m3) |
120 ppm (310 mg/m3) |
77 ppm (200 mg/m3) |
37 ppm (94 mg/m3) |
| AEGL-3 | 450 ppm (1,100 mg/m3) |
450 ppm (1,100 mg/m3) |
360 ppm (910 mg/m3) |
230 ppm (580 mg/m3) |
110 ppm (280 mg/m3) |
| IDLH (NIOSH)a | 500 ppm (1300 mg/m3) |
||||
| TLV-TWA (ACGIH®)b | 0.5 ppm (1.3 mg/m3) |
||||
| REL-C (NIOSH)c | 0.5 ppm (1.3 mg/m3) |
0.5 ppm (1.3 mg/m3) |
0.5 ppm (1.3 mg/m3) |
0.5 ppm (1.3 mg/m3) |
0.5 ppm (1.3 mg/m3) |
| PEL-C (OSHA)d | 10 ppm (25 mg/m3) |
10 ppm (25 mg/m3) |
10 ppm (25 mg/m3) |
10 ppm (25 mg/m3) |
10 ppm (25 mg/m3) |
| MAK (Germany)e | 0.5 ppm (1.3 mg/m3) |
||||
| MAC (The Netherlands)f | 0.4 ppm (1 mg/m3) |
||||
aIDLH (immediately dangerous to life or health, National Institute for Occupational Safety and Health [NIOSH 1994]) represents a maximum concentration from which, in the event of respirator failure, one could escape within 30 min without experiencing any escape-impairing or irreversible health effects.
bTLV-TWA (threshold limit value - time weighted average, American Conference of Governmental Industrial Hygienists [ACGIH 2004, 2012]) is the time-weighted average concentration for a normal 8-h workday and a 40-h workweek, to which nearly all workers may be repeatedly exposed, day after day, without adverse effect.
cREL-C (recommended exposure limit - ceiling, National Institute for Occupational Safety and Health [NIOSH 2011]) is a ceiling value that should not be exceeded at any time during a workday.
dPEL-C (permissible exposure limit - ceiling, Occupational Safety and Health Administration (29 CFR 1910.1000 [2006]) is the concentration that should not be exceeded at any time.
eMAK (maximale Arbeitsplatzkonzentration [maximum workplace concentration], Deutsche Forschungsgemeinschaft [German Research Association] DFG 2012) is defined analogous to the ACGIH TLV-TWA.
fMAC (maximaal aanvaarde concentratie [maximal accepted concentration], Dutch Expert Committee for Occupational Standards, The Netherlands (MSZW 2004) is defined analogous to the ACGIH TLV-TWA.
The AEGL-1 values exceed the recommended exposure limit (REL) of the National Institute for Occupational Safety and Health (NIOSH), the threshold limit value (TLV) of the American Conference of Governmental Industrial Hygienists, the Dutch maximal accepted concentration (MAC), and the German maximum workplace concentration (MAK) by a factor of two. The basis of the MAC and MAK values are not available, but the ACGIH (2004) documentation states that the TLV value is based on the very limited human data indicating that irritation of the mucous membranes, lacrimation, and the central nervous system effects were seen at 4 ppm, but is “more experience-based than experimentally derived”. Similarly, the NIOSH REL (1978) value was derived:
based on the effects in humans—headache, nausea, and irritation—of exposure to the chemical at 4 ppm for 4 h/day (Blinova 1965)…. The minimal effects of olfactory fatigue and mucosal irritation (Gobbatto and Terribile 1968) observed when individuals were exposed to 4 ppm ethanethiol ceased when the inhalation exposure was stopped, and no effects were observed at 0.4 ppm exposure. Because there is no evidence that adherence to the TLV of 0.5 ppm has resulted in any cases of toxicity, NIOSH recommends that the concentration of C1-C12, C16, C18 alkane thiols, or cyclohexanethiol, or any combination of these thiols, in the workplace air should not exceed 0.5 ppm as a ceiling concentration for any 15-min period (pp. 83-84).
The 30-min AEGL-3 value of 450 ppm is very similar to the NIOSH immediately dangerous to life or health value of 500 ppm (NIOSH 1994). That value is based on acute inhalation toxicity in animals as reported by Fairchild and Stokinger (1958), the same study used to derive AEGL-3 values.
8.3. Data Adequacy and Research Needs
The database on acute inhalation studies in animals is sparse, and the few available studies are dated and poorly described. Human studies of short-term
exposure to ethyl mercaptan lack data on exposure concentrations. There were insufficient data to establish a chemical-specific time-scaling relationship for ethyl mercaptan.
9. REFERENCES
ACGIH (American Conference of Governmental Industrial Hygienists). 2004. Ethyl mercaptan (CAS Reg. No.75-08-1). Documentation of the Threshold Limit Values and Biological Exposure Indices. American Conference of Governmental Industrial Hygienists, Cincinnati, OH.
ACGIH (American Conference of Governmental Industrial Hygienists). 2012. Ethyl mercaptan (CAS Reg. No.75-08-1). Threshold Limit Values and Biological Exposure Indices. American Conference of Governmental Industrial Hygienists, Cincinnati, OH.
Amoore, J.E., and E. Hautala. 1983. Odor as an aid to chemical safety: Thresholds compared with threshold limit values and volatilities for 214 industrial chemicals in air and water dilution. J. Appl. Toxicol. 3(6):272-290.
Blinova, E.A. 1965. On the normalization of concentrations of substances with strong odors in the air and work places [in Russian]. Gig. Sanit. 30(1):18-22.
Chen, S., L. Zieve, and V. Mahadevan. 1970. Mercaptans and dimethyl sulfide in the breath of patients with cirrhosis of the liver. Effect of feeding methionine. J. Lab. Clin. Med. 75(4):628-635.
DFG (Deutsche Forschungsgemeinschaft). 2012. List of MAK and BAT Values: Maximum Concentrations and Biological Tolerance Values at Workplace. Report No. 48. Wiley-VCH [online]. Available: http://onlinelibrary.wiley.com/doi/10.1002/9783527666034.oth01/pdf [accessed June 21, 2013].
Fairchild, E.J., and H.E. Stokinger. 1958. Toxicologic studies on organic sulfur compounds. I. Acute toxicity of some aliphatic and aromatic thiols (mercaptans). Am. Ind. Hyg. Assoc. J. 19(3):171-189.
Foster, D., K. Ahmed, and L. Zieve. 1974. Action of methanethiol on NA, K-ATPase: Implications for hepatic coma. Ann. NY Acad. Sci. 242:573-576.
Gobbato, F., and P.M. Terribile. 1968. Toxicological property of mercaptans [in Italian]. Folia Med. 51: 329-341 (as cited by NIOSH 1978).
Hazleton Laboratories, Inc. 1983. Mouse Lymphoma Forward Mutation Assay: Ethyl Mercaptan (Ethanethiol). Final Report, April 28, 1983. Submitted to EPA by Phillips Petroleum Company, Bartlesville, OK, with Cover Letter Dated 8/24/92. EPA Document No. 88-920010738. Microfiche No. OTS0571884.
Hazleton Laboratories, Inc. 1984. In vitro Sister Chromatid Exchange in Chinese Hamster Ovary Cells: Ethyl Mercaptan. Final Report, December 11, 1984. Submitted to EPA by Phillips Petroleum Company, Bartlesville, OK, with Cover Letter Dated 8/24/92. EPA Document No. 88-920010738. Microfiche No. OTS0571884.
Horiguchi, M. 1960. An experimental study on the toxicity of methyl mercaptan in comparison with hydrosulfide [in Japanese]. J. Osaka City Med. Cent. 9:5257-5293 (as cited in NIOSH 1978).
HSDB (Hazardous Substances Data Bank). 2011. Ethyl Mercaptan (CAS Reg. No. 7508-1). TOXNET, Specialized Information Services, U.S. National Library of Medicine, Bethesda, MD [online]. Available: http://toxnet.nlm.nih.gov/cgi-bin/sis/htmlgen?HSDB [accessed Sept. 2012].
Katz, S.H., and E.J. Talbert. 1930. Intensities of Odors and Irritating Effects of Warning Agents for Inflammable and Poisonous Gases. Technical Report No. 480. Washington, DC: U.S. Government Printing Office.
Litchfield, J.T., and F. Wilcoxon. 1949. A simplified method of evaluating dose-effect experiments. J. Pharmacol. Exp. Ther. 96(2):99-113.
MSZW (Ministerie van Sociale Zaken en Werkgelegenheid). 2004. Nationale MAC-lijst 2004: Ethaanthiol. Den Haag: SDU Uitgevers [online]. Available: http://www.lasrook.net/lasrookNL/maclijst2004.htm [accessed June 21, 2013].
Nagata, Y. 2003. Measurement of odor threshold by triangle odor bag method. Pp. 118127 in Odor Measurement Review. Office of Odor, Noise and Vibration, Environmental Management Bureau, Ministry of the Environment, Government of Japan. September 2003 [online]. Available: http://www.env.go.jp/en/air/odor/measure/02_3_2.pdf [accessed June 20, 2013].
NIOSH (National Institute for Occupational Safety and Health). 1978. Criteria for Recommended Standard. Occupational Exposure to n-Alkane Monothiols, Cyclohexanethiol, and Benzenethiol. DHEW(NIOSH) No. 78-213. U.S. Department of Health Education and Welfare, Public Health Service, Center for Disease Control, National Institute for Occupational Safety and Health, Atlanta, GA. September [online]. Available: http://www.cdc.gov/niosh/pdfs/78-213a.pdf [accessed June 20, 2013].
NIOSH (National Institute for Occupational Safety and Health). 1994. Documentation for Immediately Dangerous to Life or Health Concentrations (IDLHs): Ethyl mercaptan. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, Atlanta, GA [online]. Available: http://www.cdc.gov/niosh/idlh/75081.html [accessed June 20, 2013].
NIOSH (National Institute for Occupational Safety and Health). 2011. NIOSH Pocket Guide to Chemical Hazards: Ethyl mercaptane. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, Cincinnati, OH [online]. Available: http://www.cdc.gov/niosh/npg/npgd0280.html [accessed June 20, 2013].
NRC (National Research Council). 1993. Guidelines for Developing Community Emergency Exposure Levels for Hazardous Substances. Washington, DC: National Academy Press.
NRC (National Research Council). 2001. Standing Operating Procedures for Developing Acute Exposure Guideline Levels for Hazardous Chemicals. Washington, DC: National Academy Press.
NRC (National Research Council). 2010. Hydrogen sulfide. Pp. 173-218 in Acute Exposure Guideline Levels for Selected Airborne Chemicals, Vol. 9. Washington, DC: The National Academies Press.
O’Neil, M.J., P.E. Heckelman, C.B. Koch, and K.J. Roman, eds. 2006. The Merck Index, 14th Ed. Whitehouse Station, NJ: Merck.
Pichler, K. 1918. Intoxication due to inhalation of ethyl mercaptan [in German]. Zen-tralbl. Inn. Med. 39: 689-693.
Ruijten, M.W.M.M., R. van Doorn, and A.P. van Harreveld. 2009. Assessment of Odour Annoyance in Chemical Emergency Management. RIVM Report 609200001/2009. RIVM (National institute for Public Health and the Environment), Bilthoven, The Netherlands [online]. Available: http://www.rivm.nl/bibliotheek/rapporten/609200001.pdf [accessed Dec. 13, 2010].
Shertzer, H.G. 2012. Organic sulfur compounds. Pp. 1039-1078 in Patty’s Toxicology, 6th Ed., E. Bingham, and B. Cohrssen, eds. New York: Wiley.
Shibata, Y. 1966a. Studies on the influence of ethylmercaptan upon the living body: Part 2. On the respiratory function and clinical findings in rabbits which inhaled ethyl mercaptan gas [in Japanese]. Shikoku Acta Med. 22(6):834-843.
Shibata, Y. 1966b. Studies on the influence of ethylmercaptan upon the living body: Part 3. Inhalation experiment of ethyl mercaptan gas in the human body [in Japanese]. Shikoku Acta Med. 22(6):844-850.
Smith, R.P. 1991. Toxic responses of the blood. Pp. 276-278 in Casarett and Doull’s Toxicology: The Basic Science of Poisons, 4th Ed., M.O. Amdur, J. Doull, and C.D. Klaassen, eds. New York: Pergamon Press.
Snow, G.A. 1957. The metabolism of compounds related to ethanethiol. Biochem. J. 65(1):77-82.
Tansy, M.F., F.M. Kendall, J. Fantasia, W.E. Landin, R. Oberly, and W. Sherman. 1981. Acute and subchronic toxicity studies of rats exposed to vapors of methyl mercaptan and other reduced-sulfur compounds. J. Toxicol. Environ. Health 8(1-2):71-88.
ten Berge, W.F., A. Zwart, and L.M Appelman. 1986. Concentration-time mortality response relationship of irritant and systemically acting vapours and gases. J. Hazard. Mater. 13(3):301-309. Vahlkamp, T., A.J. Meijer, J. Wilms, and R.A. Chamuleau. 1979. Inhibition of mitochondrial electron transfer in rats by ethane thiol and methanethiol. Clin. Sci. 56(2):147-156.
Wilby, F.V. 1969. Variation in recognition odor threshold of a panel. J. Air Pollut. Control Assoc. 19(2): 96-100.
Zieve, L., W.M. Doizaki, and J. Zieve. 1974. Synergism between mercaptans and ammonia or fatty acids in the production of coma: A possible role for mercaptans in the pathogenesis of hepatic coma. J. Lab. Clin. Med. 83(1):16-28.
APPENDIX A
DERIVATION OF AEGL VALUES FOR ETHYL MERCAPTAN
Derivation of AEGL-1 Values
Derivation of AEGL-1 Values
|
Key study: |
Shibata, Y. 1966a. Studies on the influence of ethylmercaptan upon the living body: II. On the respiratory function and clinical findings in rabbits which inhaled ethyl mercaptan gas. Shikoku Acta Med. 22(6): 834-843. |
|
Toxicity end point: |
No-effect level for respiratory changes associated with odor avoidance in rabbits, 10 ppm for 20 min |
|
Scaling: |
Values held constant across time |
|
Uncertainty factor: |
3 for interspecies differences |
|
All AEGL-1 durations: |
10 ppm ÷ 10 = 1.0 ppm |
Derivation of AEGL-2 Values
In the absence of relevant data to derive AEGL-2 values and because ethyl mercaptan has a steep concentration-response curve, AEGL-3 values were divided by 3 to estimate a threshold for inability to escape.
|
10-min AEGL-2: |
450 ppm ÷ 3 = 150 ppm |
|
30-min AEGL-2: |
450 ppm ÷ 3 = 150 ppm |
|
1-h AEGL-2: |
360 ppm ÷ 3 = 120 ppm |
|
4-h AEGL-2: |
230 ppm ÷ 3 = 77 ppm |
|
8-h AEGL-2: |
110 ppm ÷ 3 = 37 ppm |
Derivation of AEGL-3 Values
|
Key study: |
Fairchild, E.J., and H.E. Stokinger. 1958. Toxicologic studies on organic sulfur compounds. I. Acute toxicity of some aliphatic and aromatic thiols (mercaptans). Am. Ind. Hyg. Assoc. J. 19(3):171-189. |
|
Toxicity end point: |
4-h LC01 of 2,250 ppm was used as an estimated lethality threshold in mice. |
|
Time scaling: |
Cn × t = k (default values of n = 3 for extrapolating to shorter durations and n = 1 for extrapolating to longer durations); time scaling not performed for the 10-min AEGL-3 value because of the uncertainty in extrapolating a 4 h point of departure to a 10-min value. |
|
Uncertainty factors: |
3 for interspecies differences |
|
10-min AEGL-3: |
450 ppm (30-min AEGL-3 value adopted) |
|
30-min AEGL-3: |
C3 × 0.5 h = 4.56 × 1010 ppm-h |
|
1-h AEGL-3: |
C3 × 1 h = 4.56 × 1010 ppm-h |
|
4-h AEGL-3: |
C3 × 4 h = 4.56 × 1010 ppm-h |
|
8-h AEGL-3: |
C1 × 8 h = 9,000 ppm-h |
APPENDIX B
ACUTE EXPOSURE GUIDELINE LEVELS FOR ETHYL MERCAPTAN
Derivation Summary
AEGL-1 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
1.0 ppm (2.5 mg/m3) |
Key reference: Shibata, Y. 1966a. Studies on the influence of ethyl mercaptan upon the living body: II. On the respiratory function and clinical findings in rabbits which inhaled ethyl mercaptan gas. Shikoku Acta. Med. 22(6):834-843.
Test species/Strain/Sex/Number: Rabbits, males, 2/group
Exposure route/Concentrations/Durations: Inhalation; 10, 100, 1,000 ppm for 20 min
Effects:
10 ppm: Unstable fluctuation in respiratory rate.
100 ppm: Decreased respiratory rate (10%) and expiratory volume (10%); increased tidal volume (20%).
1,000 ppm: Decreased respiratory rate (40%) and expiratory volume (20%); increased tidal volume (40%).
End point/Concentration/Rationale: No-effect level for respiratory changes associated with odor avoidance, 10 ppm
Uncertainty factors/Rationale: Use of the full factor of 10 for either interspecies differences or intraspecies variability would yield AEGL-1 values of 0.3 ppm or lower, concentrations that are inconsistent with human data.
Interspecies: 3
Intraspecies: 3
Modifying factor: Not applicable
Animal-to-human dosimetric adjustment: Not applicable
Time scaling: Values held constant across time because effects are not expected to vary greatly over time.
Data adequacy: The study was considered adequate for derivation of AEGL-1 values.
AEGL-2 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 150 ppm (380 mg/m3) |
150 ppm (380 mg/m3) |
120 ppm (310 mg/m3) |
77 ppm (200 mg/m3) |
37 ppm (94 mg/m3) |
Data adequacy: Data inadequate to derive AEGL-2 values. AEGL-3 values were divided by 3 to estimate thresholds for the inability to escape. This calculation is supported by the steep concentration-response curve for ethyl mercaptan (lethality in mice exposed for 4 h was 40% at 2,600 ppm, 50% at 2,770 ppm, and 100% at 3,573 ppm; in rats, the LC01 value was 3,808 and the LC50 value was 4,420 ppm).
AEGL-3 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 450 ppm (1,100 mg/m3) |
450 ppm (1,100 mg/m3) |
360 ppm (910 mg/m3) |
230 ppm (580 mg/m3) |
110 ppm (280 mg/m3) |
Reference: Fairchild, E.J., and H.E. Stokinger. 1958. Toxicologic studies on organic sulfur compounds. I. Acute toxicity of some aliphatic and aromatic thiols (mercaptans).
Am. Ind. Hyg. Assoc. J. 19(3):171-189.
Test species/Strain/Sex/Number: Mice, Swiss-derived, male, 10/group
Exposure route/Concentrations/Durations: Inhalation; 0, 2,600, 3,150, 3,573, 4,438, or 4,832 ppm for 4 h
|
Effects: |
Mortality |
|
0 |
0/10 |
|
2,600 |
4/10 |
|
3,150 |
7/10 |
|
3,573 |
10/10 |
|
4,438 |
10/10 |
|
4,832 |
10/10 |
LC50 = 2,770 ppm
LC01 = 2,250 ppm10/10
End point/Concentration/Rationale: Estimated lethality threshold in mice, 4-h LC01 of 2,250 ppm
Uncertainty factors/Rationale:
Intraspecies: 3, considered sufficient because of steep concentration-response curve (lethality in mice exposed for 4 h was 40% at 2,600 ppm, 50% at 2,770 ppm, and 100% at 3,573 ppm; in rats, the LC01 value was 3,808 and the LC50 value was 4,420 ppm), which implies limited individual variability.
Interspecies: 3, because the mouse is the most sensitive species. Also, although an interspecies uncertainty factor of 10 might normally be applied because of limited data, AEGL-3 values calculated with a total uncertainty factor of 30 would lead to values approaching or equivalent to the AEGL-3 values for hydrogen sulfide. For example, if a total uncertainty factor of 30 is applied, an 8-h AEGL-3 value for ethyl mercaptan would be 37 ppm, which is slightly higher than the 8-h AEGL-3 value for hydrogen sulfide of 31 ppm (NRC 2010). Because a robust database exists for hydrogen sulfide and because data suggest that ethyl mercaptan is less toxic than hydrogen sulfide (4-h LC50 is 4,420 ppm for ethyl mercaptan and 444 ppm for hydrogen sulfide), it would be inconsistent with the total data set to have AEGL-3 values for ethyl mercaptan that are in the range of the AEGL-3 values for hydrogen sulfide. Furthermore, use of a total uncertainty factor of 30 would yield a 30-min AEGL-3 value of 150 ppm, which is inconsistent with the finding that a single human exposed to ethyl mercaptan at 112 ppm for 20 min exhibited only a slightly irregular and decreased breathing rate (Shibata 1966b).
Modifying factor: Not applicable
Animal-to-human dosimetric adjustment: Insufficient data
Time scaling: Cn × t = k; default value of n = 3 was used for extrapolation to the shorter durations (30 min, 1 h, and 4 h) and n = 1 for extrapolation to the longer duration (8 h). The 30-min value was adopted as the 10-min AEGL-3 value because of the uncertainty associated with extrapolating a 4-h exposure to a 10-min value.
Data adequacy: The study was well conducted and used a sufficient number of animals.
APPENDIX C
DERIVATION OF THE LEVEL OF DISTINCT ODOR AWARENESS FOR ETHYL MERCAPTAN
The level of distinct odor awareness (LOA) represents the concentration above which it is predicted that more than half of the exposed population will experience at least a distinct odor intensity, and about 10% of the population will experience a strong odor intensity. The LOA should help chemical emergency responders in assessing the public awareness of the exposure on the basis of odor perception. The LOA derivation follows the guidance of Ruijten et al. (2009).
The odor detection threshold (OT50) for ethyl mercaptan was reported to be 0.0000087 ppm (Nagata 2003).
The concentration (C) leading to an odor intensity (I) of distinct odor detection (I = 3) is derived using the Fechner function:
I = kw × log (C ÷ OT50) + 0.5
For the Fechner coefficient, the default of kw = 2.33 was used due to the lack of chemical-specific data.
3 = 2.33 × log (C ÷ 0.0000087) + 0.5
log (C ÷ 0.0000087) = ([3 - 0.5] ÷ 2.33)
log (C ÷ 0.0000087) = 1.07
C = (10107) × 0.0000087
C = 0.000102 ppm
The resulting concentration is multiplied by an empirical field correction factor. It takes into account that, in everyday life, factors such as sex, age, sleep, smoking, upper airway infections, and allergy, as well as distractions, increase the odor detection threshold by a factor of 4. In addition, it takes into account that odor perception is very fast (about 5 seconds), which leads to the perception of concentration peaks. On the basis of current current knowledge, a factor of 1/3 is applied to adjust for peak exposure. Adjustment for distraction and peak exposure lead to a correction factor of 4 ÷ 3 = 1.33.
LOA = C ×
1.33 LOA = 0.000102 ppm × 1.33
LOA = 0.00014 ppm
APPENDIX D
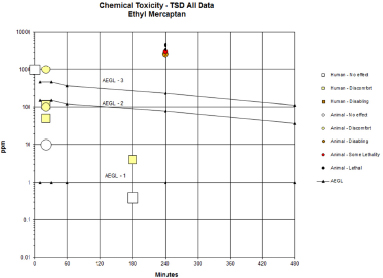
FIGURE D-1 Category plot of toxicity data and AEGL values for ethyl mercaptan.
TABLE D-1 Data Used in Category Plot of AEGL Values for Ethyl Mercaptan
| Source | Species | Sex | No. of Exposures | ppm | Min | Category |
| AEGL-1 | 1 | 10 | AEGL | |||
| AEGL-1 | 1 | 30 | AEGL | |||
| AEGL-1 | 1 | 60 | AEGL | |||
| AEGL-1 | 1 | 240 | AEGL | |||
| AEGL-1 | 1 | 480 | AEGL | |||
| AEGL-2 | 150 | 10 | AEGL | |||
| AEGL-2 | 150 | 30 | AEGL | |||
| AEGL-2 | 120 | 60 | AEGL | |||
| AEGL-2 | 77 | 240 | AEGL | |||
| AEGL-2 | 37 | 480 | AEGL | |||
| AEGL-3 | 450 | 10 | AEGL | |||
| AEGL-3 | 450 | 30 | AEGL | |||
| AEGL-3 | 360 | 60 | AEGL | |||
| AEGL-3 | 230 | 240 | AEGL | |||
| AEGL-3 | 110 | 480 | AEGL | |||
| Shibata 1966a | Rabbit | Male | 1 | 10 | 20 | 0 |
| Rabbit | Male | 1 | 100 | 20 | 1 | |
| Rabbit | Male | 1 | 1,000 | 20 | 1 | |
| Fairchild and Stokinger 1958 | Mouse | Male | 1 | 2,600 | 240 | 2 |
| Mouse | Male | 1 | 3,150 | 240 | SL | |
| Mouse | Male | 1 | 3,573 | 240 | 3 | |
| Mouse | Male | 1 | 4,438 | 240 | 3 | |
| Mouse | Male | 1 | 4,832 | 240 | 3 | |
| Katz and Talbert 1930 | Human | Male | 1 | 0.00002 | 0 | |
| Human | Male | 1 | 920 | 1 | ||
| Wilby 1969 | Human | Male | 1 | 0.0004 | 0 | |
| Blinova 1965 | Human | Male | 1 | 4 | 180 | 1 |
| Pichler 1918 | Human | Male | ||||
| Shibata 1966b | Human | Male | 1 | 50 | 20 | 1 |
| Human | Male | 1 | 112 | 20 | 1 | |
| Amoore and Hautala 1983 | Human | 1 | 0.0008 | 1 | ||
| Nagata 2003 | Human | 1 | 0.00001 | 1 | ||
| Zieve et al. 1974 | Rats | |||||
| Rats | ||||||
| Blinova 1965 | Human | 1 | 0.4 | 180 | 0 | |
| NIOSH 1978 | Human | 5 | 4.0 | 180 | 1 | |
| Human | 5 | 0.4 | 180 | 0 | ||
| Katz and Talbert 1930 | Human | 1 | 1 000 0 | 0 17 | 0 | |
For category: 0 = no effect, 1 = discomfort, 2 = disabling, 3 = lethal; SL = some lethality.






























