Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 15 (2013)
Chapter: 3 Phenyl Mercaptan Acute Exposure Guideline Levels
3
Phenyl Mercaptan1
Acute Exposure Guideline Levels
PREFACE
Under the authority of the Federal Advisory Committee Act (FACA) P.L. 92-463 of 1972, the National Advisory Committee for Acute Exposure Guideline Levels for Hazardous Substances (NAC/AEGL Committee) has been established to identify, review, and interpret relevant toxicologic and other scientific data and develop AEGLs for high-priority, acutely toxic chemicals.
AEGLs represent threshold exposure limits for the general public and are applicable to emergency exposure periods ranging from 10 minutes (min) to 8 hours (h). Three levels—AEGL-1, AEGL-2, and AEGL-3—are developed for each of five exposure periods (10 and 30 min and 1, 4, and 8 h) and are distinguished by varying degrees of severity of toxic effects. The three AEGLs are defined as follows:
AEGL-1 is the airborne concentration (expressed as parts per million or milligrams per cubic meter [ppm or mg/m3]) of a substance above which it is predicted that the general population, including susceptible individuals, could experience notable discomfort, irritation, or certain asymptomatic, nonsensory effects. However, the effects are not disabling and are transient and reversible upon cessation of exposure.
____________________
1This document was prepared by the AEGL Development Team composed of Cheryl Bast (Oak Ridge National Laboratory), Gary Diamond (SRC, Inc.), Chemical Manager Glenn Leach (National Advisory Committee [NAC] on Acute Exposure Guideline Levels for Hazardous Substances), and Ernest V. Falke (U.S. Environmental Protection Agency). The NAC reviewed and revised the document and AEGLs as deemed necessary. Both the document and the AEGL values were then reviewed by the National Research Council (NRC) Committee on Acute Exposure Guideline Levels. The NRC committee has concluded that the AEGLs developed in this document are scientifically valid conclusions based on the data reviewed by the NRC and are consistent with the NRC guidelines reports (NRC 1993, 2001).
AEGL-2 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience irreversible or other serious, long-lasting adverse health effects or an impaired ability to escape.
AEGL-3 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience life-threatening health effects or death.
Airborne concentrations below the AEGL-1 represent exposure concentrations that could produce mild and progressively increasing but transient and nondisabling odor, taste, and sensory irritation or certain asymptomatic, nonsensory effects. With increasing airborne concentrations above each AEGL, there is a progressive increase in the likelihood of occurrence and the severity of effects described for each corresponding AEGL. Although the AEGL values represent threshold concentrations for the general public, including susceptible subpopulations, such as infants, children, the elderly, persons with asthma, and those with other illnesses, it is recognized that individuals, subject to idiosyncratic responses, could experience the effects described at concentrations below the corresponding AEGL.
SUMMARY
Phenyl mercaptan is used as an intermediate in the manufacture of pesticides, pharmaceuticals, and amber dyes, and is also used as a mosquito larvicide. It is an odorous, colorless liquid. The disagreeable odor has been described as penetrating, repulsive, and garlic-like (Shertzer 2012).
Phenyl mercaptan depresses the central nervous system and affects the respiratory center, similar to hydrogen sulfide, producing death by respiratory paralysis. Clinical signs of exposure are ocular and mucous membrane irritation, headache, dizziness, staggering gait, nausea, and vomiting. Paralysis of the locomotor muscles has also been observed. Its primary mechanism of action appears to be interference with cytochrome oxidase.
AEGL-1 values are not recommended for phenyl mercaptan because of insufficient data.
No robust data on phenyl mercaptan consistent with the definition of AEGL-2 were available. Therefore, AEGL-2 values were based on a 3-fold reduction in the AEGL-3 values. These calculations are considered estimated thresholds for inability to escape and are appropriate because of the steep concentration-response relationship for phenyl mercaptan toxicity.
AEGL-3 values were based on a calculated LC01 (lethal concentration, 1% lethality) of 10.3 ppm in rats exposed to phenyl mercaptan for 4 h (Fairchild and Stokinger 1958). A total uncertainty factor of 10 was applied: a factor of 3 for interspecies differences and a factor of 3 for intraspecies variability. Those factors are considered sufficient because the mechanism of action (cytochrome oxidase inhibition) is not expected to vary greatly between or within species. Alt-
hough an interspecies or intraspecies uncertainty factor of 10 might normally be applied because of limited data, a total uncertainty factor of 30 would yield AEGL values that are inconsistent with those derived for the structural and mechanistic analogs ethyl mercaptan, methyl mercaptan, and hydrogen sulfide, all of which have a more robust data set than phenyl mercaptan. Values were scaled across time using the equation Cn × t = k; default values of n = 3 when extrapolating to shorter durations and n = 1 when extrapolating to longer durations were used to derive values protective of human health (NRC 2001). AEGL values for phenyl mercaptan are presented in Table 3-1.
1. INTRODUCTION
Phenyl mercaptan is used as an intermediate in the manufacture of pesticides, pharmaceuticals, and amber dyes, and is also used as a mosquito larvicide. It is an odorous, colorless liquid. The disagreeable odor has been described as penetrating, repulsive, and garlic-like (Shertzer 2012).
Phenyl mercaptan is produced commercially by reducing benzenesulfonyl chloride with zinc dust in sulfuric acid or by reacting hydrogen sulfide with chlorobenzene (Shertzer 2012). In 1981, the total U.S. production of phenyl mercaptan was probably greater than 2.27 × 106 grams and the total U.S. imports were probably greater than 3.78 × 107 grams (HSDB 2009). The 1983 National Occupational Exposure Survey reported that 879 U.S. workers (692 males, 187 females) were exposed to phenyl mercaptan (RTECS 2009).
The physical and chemical properties of phenyl mercaptan are presented in Table 3-2.
2. HUMAN TOXICITY DATA
2.1. Acute Lethality
No information concerning human lethality from acute exposure to phenyl mercaptan was available.
2.2. Nonlethal Toxicity
2.2.1. Odor Threshold and Awareness
Katz and Talbert (1930) exposed six human subjects to phenyl mercaptan at a range of concentrations via a nosepiece. In tests evaluating odor intensity or throat and nasal irritation, the subjects were exposed to a single inhalation of phenyl mercaptan. For ocular irritation tests, the eye was exposed to phenyl mercaptan for 10 seconds. Vapor concentrations were determined by weighing vaporizers containing the phenyl mercaptan before and after a series of odor measurements, and dividing the loss in weight by the volume of air passed through the vaporizer. The subjects described the odor as very disagreeable, repulsive, and persistent.
The odor intensity of phenyl mercaptan is presented in Table 3-3. Faint nasal irritation was observed at 85 ppm and faint ocular irritation was observed at 45 ppm; moderate, strong, and intolerable ocular irritation were reported at 110, 250, 580 ppm, respectively. Nasal and ocular irritation were not reported at 0.4 or 18 ppm, respectively. Throat irritation and headache were noted, but the concentrations at which these effects occurred were not reported.
TABLE 3-1 AEGL Values for Phenyl Mercaptan
| Classification | 10 min | 30 min | 1 h | 4 h | 8 h | End Point (Reference) |
| AEGL-1a (nondisabling) | NR | NR | NR | NR | NR | Insufficient data |
| AEGL-2 (disabling) | 1.0 ppm (4.5 mg/m3) |
0.70 ppm (3.2 mg/m3) |
0.53 ppm (2.4 mg/m3) |
0.33 ppm (1.5 mg/m3) |
0.17 ppm (0.77 mg/m3) |
One-third reduction of AEGL-3 values |
| AEGL-3 (lethal) | 3.0 ppm (14 mg/m3) |
2.1 ppm (9.5 mg/m3) |
1.6 ppm (7.2 mg/m3) |
1.0 ppm (4.5 mg/m3) |
0.52 ppm (2.3 mg/m3) |
LC01 in rats (Fairchild and Stokinger 1958) |
Abbreviations: LC01, lethal concentration, 1% lethality; NR, not recommended.
aThe absence of AEGL-1 values does not imply that concentrations below AEGL-2 values will be without effect.
TABLE 3-2 Physical and Chemical Data on Phenyl Mercaptan
|
Common Name |
Phenyl Mercaptan |
Reference |
|
Synonyms |
Benzenethiol; thiophenol; mercaptobenzene |
HSDB 2009 |
|
CAS registry no. |
108-98-5 |
HSDB 2009 |
|
Chemical formula |
C6H5SH |
HSDB 2009 |
|
Molecular weight |
110.18 |
HSDB 2009 |
|
Physical state |
Water-white liquid |
HSDB 2009 |
|
Odor |
Garlic-like, penetrating, repulsive |
Shertzer 2012 |
|
Melting point |
-14.9°C |
|
|
Boiling point |
168.3°C |
|
|
Density/Specific gravity |
1.0728 at 25°C |
HSDB 2009 |
|
Solubility |
835 mg/L at 25°C in water; very soluble in alcohol; miscible with ether, benzene, and carbon disulfide |
HSDB 2009 |
|
Saturated vapor concentration |
2,539 ppm (11,428 mg/m3) at 25°C |
Calculated |
|
Vapor pressure |
1.93 mm Hg at 25°C |
HSDB 2009 |
|
Conversion factors in air |
1 mg/m3 = 0.22 ppm |
NIOSH 2011 |
TABLE 3-3 Odor Intensity of Phenyl Mercaptan
|
Intensity |
Description |
Concentration (ppm) |
|
0 |
No odor |
0.000005 |
|
1 |
Detectable |
0.00025 |
|
2 |
Faint |
0.014 |
|
3 |
Median, easily noticeable |
0.72 |
|
4 |
Strong |
38 |
|
5 |
Most intense |
2,000 |
Source: Adapted from Katz and Talbert 1930.
Amoore and Hautala (1983) reported an odor threshold for phenyl mercaptan of 0.00094 ppm. This value is the geometric mean of published odor threshold values. AIHA (1989) reported odor thresholds of 0.00003-0.0003 ppm for phenyl mercaptan.
2.3. Developmental and Reproductive Toxicity
Developmental and reproductive studies of human exposure to phenyl mercaptan were not available.
2.4. Genotoxicity
Genotoxicity studies of human exposure to phenyl mercaptan were not available.
2.5. Carcinogenicity
Carcinogenicity studies of human exposure to phenyl mercaptan were not available.
2.6. Summary
Data concerning human exposure to phenyl mercaptan are limited to odor threshold data. Data on acute lethality, developmental and reproductive toxicity, genotoxicity, and carcinogenicity in humans were not available.
3. ANIMAL TOXICITY DATA
3.1. Acute Lethality
3.1.1. Mice
Fairchild and Stokinger (1958) exposed groups of 5-10 Swiss-derived male mice (body weight 25-28 g) to phenyl mercaptan at 20, 31, 41, 52, or 79
ppm for 4-h, followed by a 15-day observation period. Vapor generation was achieved by either bubbling a stream of nitrogen gas through a midget fritted-glass bubbler, which contained liquid phenyl mercaptan, or by passage of nitrogen into a borosilicate glass nebulizer containing the phenyl mercaptan. Target concentrations were maintained in an 18-L glass chamber by varying the ratio of volume flow of air and phenyl mercaptan containing compressed nitrogen. Phenyl mercaptan concentrations were measured during exposure periods by absorption of vapors in either isopropyl alcohol or acetone containing an excess of silver nitrate and titrating the uncombined silver amperometrically. Chamber concentrations during tests were uniform after the first 30 min; mean variation for all exposures was approximately 4%. Clinical signs included increased respiration and restlessness (hyperactivity), uncoordinated movement, staggering gait, muscular weakness, partial skeletal muscle paralysis beginning in the hind limbs, light to severe cyanosis, tolerance of a prone position, and mild to heavy sedation; however, concentration-response data were not provided. Animals exposed to “maximal lethal concentrations” typically died from respiratory arrest during exposure or shortly after removal from the chamber. Animals exposed to “minimal lethal concentrations” typically died while in a semiconscious condition of “long duration”. Surviving animals often remained in a semi-conscious state of sedation and lethargy 4- to 6-h post-exposure before showing signs of recovery. An LC50 (lethal concentration, 50% lethality) value of 28 ppm was calculated by the investigators. A BMC01 (benchmark concentration with 1% response) of 26.5 ppm and BMCL05 (benchmark concentration, 95% lower confidence limit with 5% response) of 18.5 ppm were also calculated. LC05 and LC01 values could not be calculated by the method of Litchfield and Wilcoxon (1949) because there were no data on at least two concentrations the resulted in mortality between 0% and 100%. Mortality data for phenyl mercaptan are presented in Table 3-4.
An oral LD50 (lethal dose, 50% lethality) of 267 mg/kg was reported for male albino mice (Hazleton Laboratories 1951).
3.1.2. Rats
Fairchild and Stokinger (1958) exposed groups of 5-10 Wistar-derived male rats (body weight 180-220 g) to phenyl mercaptan at 20, 31, 41, 52, 79, or 132 ppm for 4 h, followed by a 15-day observation period. Vapor generation and test chamber analysis was similar to that described for experiments in mice (see Section 3.1.1). Clinical signs included increased respiration and restlessness (hyperactivity), uncoordinated movement, staggering gait, muscular weakness, partial skeletal muscle paralysis beginning in the hind limbs, light to severe cyanosis, tolerance of a prone position, and mild to heavy sedation; however, no concentration-response data were provided for clinical signs. Animals exposed to “maximal lethal concentrations” typically died from respiratory arrest during exposure or shortly after removal from the chamber. Animals exposed to “minimal lethal concentrations” typically died while in a semi-conscious condition of
“long duration”. Surviving animals often remained in a semi-conscious state of sedation and lethargy 4- to 6-h post-exposure before showing signs of recovery. An LC50 value of 33 ppm was calculated by the investigators. A BMC01 of 17.7 ppm and a BMCL05 of 13.4 ppm were also calculated. An LC05 value of 15.5 ppm and LC01 value of 10.3 ppm were calculated by the method of Litchfield and Wilcoxon (1949). Mortality data on phenyl mercaptan are presented in Table 3-4.
Groups of five male and five female albino rats were exposed to phenyl mercaptan at 244, 346, or 595 ppm for 1 h, followed by a 14-day observation period (Stauffer Chemical Company 1969). Clinical signs included ocular edema and erythema, and slight nasal discharge; investigators did not report whether these effects were observed in all test groups. “Acute depression” (no additional information provided) was reported in the 244-ppm group, and dyspnea, gagging, fasciculation, and cyanosis were reported in the 346- and 595-ppm groups while the animals were in the exposure chamber. There were no treatment-related deaths in the 244-ppm group, and animals appeared normal during gross pathologic examination. Treatment-related death was occurred in 3/10 animals at 346 ppm and 10/10 animals at 595 ppm. Decedents exhibited areas of hemorrhage in the lungs, while survivors in the 346-ppm group appeared normal during gross examination. The authors calculated an LC50 of 422 ppm. No further experimental details were available.
Fairchild and Stokinger (1958) also administered phenyl mercaptan by oral gavage, intraperitoneal injection, or dermal application to Wistar-derived male rats, followed by a 15-day observation period. An oral LD50 of 46.2 mg/kg, an intraperitoneal LD50 of 9.8 mg/kg, and a dermal LD50 of 300 mg/kg were reported.
3.1.3. Rabbits
Fairchild and Stokinger (1958) administered single dermal applications of phenyl mercaptan at 67, 134, or 213 mg/kg to groups of three New Zealand white rabbits, followed by a 72-h observation period. None of the rabbits in the 67-mg/kg group died, 2/3 rabbits died within 72 h in the 134-mg/kg group, and 3/3 rabbits in the 213-mg/kg group died within 4 h of administration.
3.2. Nonlethal Toxicity
No nonlethal animal toxicity data on phenyl mercaptan were available.
3.3. Repeated-Exposure Studies
Seven adult male albino rats and 12 adult male albino mice were exposed in chambers in which 3.2% of the atmosphere was saturated with phenyl mercaptan for 6 h on the first exposure day and for 8 h/day on the next 3 days (Haz-
leton Laboratories 1951). There were no overnight exposures. Exposures were conducted in a stainless steel chamber with a 30 L/min flow rate.
Mice exhibited excitement, preening, and slight salivation during the first 6 h exposure period. Seven mice were dead the following morning, but the surviving five mice appeared normal (Group A). A second group of 13 adult male albino mice was added to the experiment (Group B). All mice were then exposed 8 h/day for three consecutive days. Of the five remaining mice from group A, two died on day 2 of exposure, two died on day 4, and the fifth died 3 days after the final exposure. Hemorrhagic lungs, irritation of the intestines, and spotted livers and kidneys were found at necropsy. Group B mice also exhibited preening, lacrimation, and salivation immediately after exposure started, and subsequently were lethargic and appeared unkempt. Eleven of the 13 Group-B mice died; deaths occurred between day 1 of exposure and 3 days after the final exposure. Hemorrhagic lungs, irritated intestines, and spotty livers and kidneys were noted in both decedents and animals killed three days after the final exposure.
Rats exhibited preening, lacrimation, and marked salivation during exposure to phenyl mercaptan, followed by unkempt appearance and lethargy. One rat died overnight after the final exposure, and another died 3 days after the final exposure. Hemorrhagic lungs, intestinal irritation, and mottled livers and kidneys were found in the decedents. Surviving rats sacrificed 3 days after the final exposure showed gas-filled and irritated stomachs and intestines, pale brown kidneys, small spleens, mottled livers, and irritated eyes. An odor of phenyl mercaptan was noted when the abdominal cavity was opened.
TABLE 3-4 Mortality in Mice and Rats Exposed to Phenyl Mercaptan for 4 Hours
|
Concentration (ppm) |
Mice |
Rats |
|
20 |
0/10 |
0/5 |
|
31 |
7/10 |
5/10 |
|
41 |
10/10 |
4/6 |
|
52 |
10/10 |
5/5 |
|
79 |
5/5 |
10/10 |
|
132 |
– |
10/10 |
|
BMC01 |
26.5 ppm |
17.7 ppm |
|
BMCL05 |
18.5 ppm |
13.4 ppm |
|
LC01 |
Not applicable |
10.3 ppm |
|
LC05 |
Not applicable |
15.5 ppm |
|
LC50 |
28 ppm |
33 ppm |
Source: Adapted from Fairchild and Stokinger 1958.
3.4. Developmental and Reproductive Toxicity
Groups of 25 pregnant CD rats were administered phenyl mercaptan by gavage in corn oil at doses of 0, 20, 35, or 50 mg/kg/day on gestation days 6-15 (NTP 1994a). Four high-dose dams died. There was no treatment-related effect on pregnancy rates; dose-related clinical signs in dams were limited to rooting behavior after gavage administration. Maternal body weight, body weight gain, and food consumption were decreased in high-dose dams. High-dose animals showed decreased gravid uterine weight, increased post-implantation loss, decreased live litter size, decreased fetal body weight per litter, and increased incidence of external fetal malformations.
Groups of 15-26 pregnant New Zealand white rabbits were administered phenyl mercaptan in corn oil at doses of 0, 10, 30, or 40 mg/kg/day on gestation days 6-19 (NTP 1994b). On gestation day 30, fetuses were removed from the does and examined. Two does died during the study, one each in the 10- and 30-mg/kg/day groups. No consistent maternal clinical signs were found, and only transient decreases in body weight were noted at 30 and 40 mg/kg/day. There were no treatment-related effects on gravid uterine weight, number of implantation sites/litter, preimplantation loss, live litter size, sex ratio, fetal body weight, or fetal malformations.
In a multigenerational study, male and female Sprague-Dawley rats (F0) were administered phenyl mercaptan by gavage in corn oil at doses of 9, 18, or 35 mg/kg/day for a 16-week cohabitation period (NTP 1996). During that time, any litters born to F0 animals were killed on postnatal day 1. Litters born after 17 weeks (F1) were raised until postnatal day 21, and then selected weanlings were administered phenyl mercaptan at the same doses as their parents. On postnatal day 81, F1 animals were allowed to cohabitate for 1 week and were killed after their litters (F2) were delivered. There were dose-related increases in hepatic weight (20-50% in males, 11-36% in females) and renal weight (30-104% in males, 8-20% in females) in all treatment groups of both parental generations. Decreased body weight (7-15%) was observed only in parental males at 35 mg/kg/day. Decreases in sperm motility (5-6%) were noted at 18 and 35 mg/kg/day. Decreases in pup body weight were sporadic, but were generally more pronounced at the higher doses.
3.5. Genotoxicity
Phenyl mercaptan was negative in an Ames Salmonella typhimurium assay with strains TA98 and TA100 (LaVoie et al. 1979).
3.6. Carcinogenicity
Carcinogenicity studies of phenyl mercaptan in animals were not available.
3.7. Summary
Animal toxicity data on phenyl mercaptan were limited. Inhalation lethality studies in rats and mice were available that suggest a steep concentration-response curve. In mice exposed to phenyl mercaptan for 4 h, mortality was 0% at 20 ppm, 70% a 31 ppm, and 100% at 41 ppm. The 4-h mouse LC50 value was 28 ppm (Fairchild and Stokinger 1958). In the rat, the 4-h LC50 value was 33 ppm, whereas the 4-h LC01 value was 10.3 ppm (Fairchild and Stokinger 1958). Clinical signs were indicative of central nervous system depression and respiratory arrest and included changes in respiration, restlessness (hyperactivity), uncoordinated movement, staggering gait, muscular weakness, skeletal muscle paralysis, light to severe cyanosis, and coma. Repeated inhalation exposure studies in rats and mice reported signs of irritation during exposure and at necropsy. Mottling of the liver and kidneys were also noted at necropsy (Hazleton Laboratories 1951). Data on reproductive and developmental effects of phenyl mercaptan were only available from oral exposure studies. Maternal and fetal effects were found in rats (NTP 1994a), whereas no treatment-related effects were noted in rabbit does or fetuses (NTP 1994b). Phenyl mercaptan was not a reproductive toxicant in a multigenerational study of rats (NTP 1996). Phenyl mercaptan was not mutagenic in an Ames bacterial reverse mutation assay, and no carcinogenicity studies were available.
4. SPECIAL CONSIDERATIONS
4.1. Metabolism and Disposition
Adult rats were orally administered 35S-labeled phenyl mercaptan at a dose of 6 mg/kg (McBain and Menn 1969). One hour after administration, excreted urine was extracted with benzene and the aqueous layer was acidified with sulfuric acid and extracted with ether. The benzene-soluble and water-soluble products were analyzed by thin-layer chromatography and gas-liquid chromatography. The only benzene-soluble metabolite identified was 35S-methylphenyl sulfone. Trace amounts of methylphenyl sulfoxide were also identified. The authors concluded that phenyl mercaptan readily undergoes S-methylation, followed by oxidation of phenylsulfide to methylphenyl sulfone.
4.2. Mechanism of Toxicity
Mercaptans act similarly to hydrogen sulfide and cyanide by interrupting electron transport through inhibition of cytochrome oxidase (NIOSH 1978). As a result of the electron transfer blockage, oxidative phosphorylation and aerobic metabolism may be compromised, peripheral tissue PO2 increases, and the unloading gradient for oxyhemoglobin decreases. High concentrations of oxyhemoglobin are thus found in the venous return, resulting in flushed skin and mu-
cous membranes. Lactic acidemia occurs as a result of the increased demand placed on glycolysis. Additionally, repeated-exposure studies of phenyl mercaptan suggest that certain effects, such as renal effects, might be due to the phenol moiety.
4.3. Structure-Activity Relationships
Rat lethality data suggest that the acute inhalation toxicity of phenyl mercaptan is much greater than other mercaptans or hydrogen sulfide (see Table 3-5).
4.4. Concurrent Exposure Issues
Because cyanide, hydrogen sulfide, methyl mercaptan, ethyl mercaptan, and phenyl mercaptan are all cytochrome oxidase inhibitors, an interaction might be possible if individuals were simultaneously exposed to two or more of these compounds (Smith 1991). These interactions could result in lower lethal exposure concentrations for phenyl mercaptan.
TABLE 3-5 Comparative Toxicity of Mercaptans
| Compound | Rat Intraperitoneal LD50 (mg/kg) | Rat Oral LD50 (mg/kg) | 4-h Inhalation LC50 (ppm) | Reference | |
| Rats | Mice | ||||
| Hydrogen sulfide | – | – | 444 | – | Tansy et al. 1981 |
| Methyl mercaptan | – | – | 675 | 1,664 | Horiguchi 1960 (mice); Tansy et al. 1981(rats) |
| Ethyl mercaptan | 226 | 682 | 4,420 | 2,770 | Fairchild and Stokinger 1958 |
| Propyl mercaptan | 515 | 1,790 | 7,200 | 4,010 | Fairchild and Stokinger 1958 |
| Isobutyl mercaptan | 917 | 7,168 | >25,000 | >25,000 | Fairchild and Stokinger 1958 |
| tert-Butyl mercaptan | 590 | 4,729 | 22,200 | 16,500 | Fairchild and Stokinger 1958 |
| n-Butyl mercaptan | 399 | 1,500 | 4,020 | 2,500 | Fairchild and Stokinger 1958 |
| n-Hexyl mercaptan | 396 | 1,254 | 1,080 | 528 | Fairchild and Stokinger 1958 |
| Phenyl mercaptan | 9.8 | 46.2 | 33 | 28 | Fairchild and Stokinger 1958 |
| Benzyl mercaptan | 373 | 493 | >235 | 178 | Fairchild and Stokinger 1958 |
| tert-Octyl mercaptan | 12.9 | 83.5 | 51 (males) | 47 (males) | Fairchild and Stokinger 1958 |
4.5. Species Differences
Because of the limited data available on phenyl mercaptan, a definitive assessment of species variability is not possible. Fairchild and Stokinger (1958) reported similar 4-h LC50 values in rats (33 ppm) and mice (28 ppm). However, the latency period for death was shorter in mice than rats. Exposure of rats to phenyl mercaptan at 41 ppm resulted in no deaths during exposure or 24 h postexposure; in contrast, 4/10 mice died during exposure at 41 ppm and 7/10 died within the first 24 h post-exposure.
4.6. Concentration-Exposure Duration Relationship
The concentration-time relationship for many irritant and systemically-acting vapors and gases may be described by the equation Cn × t = k, where the exponent n ranges from 0.8 to 3.5 (ten Berge et al. 1986). Data were inadequate to derive an empirical value of n for phenyl mercaptan. To obtain conservative and protective AEGL values in the absence of a chemical-specific scaling exponent, temporal scaling was performed using default values of n = 3 when extrapolating to shorter durations and n = 1 when extrapolating to longer durations.
5. DATA ANALYSIS FOR AEGL-1
5.1. Human Data Relevant to AEGL-1
Human data on phenyl mercaptan were not available for deriving AEGL-1 values.
5.2. Animal Data Relevant to AEGL-1
Animal data on phenyl mercaptan were not available for deriving AEGL-1 values.
5.3. Derivation of AEGL-1
AEGL-1 values for phenyl mercaptan are not recommended because of insufficient data. The absence of AEGL-1 values does not imply that concentrations below AEGL-2 values are without effect.
6. DATA ANALYSIS FOR AEGL-2
6.1. Human Data Relevant to AEGL-2
Human data on phenyl mercaptan were not available for deriving AEGL-2 values.
6.2. Animal Data Relevant to AEGL-2
Fairchild and Stokinger (1958) reported clinical signs of uncoordinated movement, partial muscle paralysis, and mild to heavy sedation in rats exposed to phenyl mercaptan for 4 h; however, no concentration-response data were provided which could be used to identify an AEGL-2 effect level. Stauffer Chemical Company (1969) reported “acute depression” in rats exposed to phenyl mercaptan at 244 ppm for 1 h. This study was considered unsuitable for deriving AEGL-2 values because 244 ppm is seven times higher than the 4-h LC50 of 33 ppm estimated by Fairchild and Stokinger (1958).
6.3. Derivation of AEGL-2 Values
No inhalation studies of phenyl mercaptan with concentration and duration information consistent with the definition of AEGL-2 were available. Therefore, AEGL-2 values were based on a 3-fold reduction in the AEGL-3 values. These calculations were considered estimated thresholds for serious or irreversible effects or inability to escape. The calculations are appropriate because of the steep concentration-response curve for lethality. AEGL-2 values for phenyl mercaptan are presented in Table 3-6, and calculations are presented in Appendix A.
7. DATA ANALYSIS FOR AEGL-3
7.1. Human Data Relevant to AEGL-3
Human data on phenyl mercaptan were not available for calculating AEGL-3 values.
7.2. Animal Data Relevant to AEGL-3
A 4-h LC50 value of 28 ppm, BMCL05 value of 18.5 ppm, and BMC01 value of 26.5 ppm were calculated for mice exposed to phenyl mercaptan (Fairchild and Stokinger 1958). A 4-h LC50 value of 33 ppm, BMCL05 value of 13.4 ppm, BMC01 value of 17.7 ppm, LC05 value of 15.5 ppm, and LC01 value of 10.3 ppm were calculated for rats (Fairchild and Stokinger 1958). A 1-h LC50 value of 422 ppm in rats was also calculated (Stauffer Chemical Company 1969).
TABLE 3-6 AEGL-2 Values for Phenyl Mercaptan
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 1.0 ppm (4.5 mg/m3) |
0.70 ppm (3.2 mg/m3) |
0.53 ppm (2.4 mg/m3) |
0.33 ppm (1.5 mg/m3) |
0.17 ppm (0.77 mg/m3) |
7.3. Derivation of AEGL-3 Values
The 4-h LC01 of 10.3 ppm for rats (Fairchild and Stokinger 1958) is the lowest of the predicted lethality thresholds for phenyl mercaptan (see Table 3-4), and was used to derive AEGL-3 values. The rat data were selected because the BMC01 and BMCL05 values were lower than the corresponding values in mice, and LC01 and LC05 values could not be calculated from the mouse data (see Section 3.1.1).
A total uncertainty factor of 10 was applied: 3 for interspecies differences and 3 for intraspecies variability. Those factors were considered sufficient because the mechanism of action (cytochrome oxidase inhibition) is not expected to vary greatly between or within species. Although an interspecies or intraspecies uncertainty factor of 10 might normally be applied because of limited data, a total uncertainty of 30 would yield AEGL values that are inconsistent with the AEGL values for the structural and mechanistic analogs ethyl mercaptan, methyl mercaptan, and hydrogen sulfide, all of which have a more robust data set than phenyl mercaptan. Rat lethality data (see Section 4.3) suggest that the acute inhalation toxicity of phenyl mercaptan is approximately 140-fold greater than ethyl mercaptan, 20-fold greater than methyl mercaptan, and 13-fold greater than hydrogen sulfide. A total uncertainty factor of 30 would yield AEGL-3 values that suggest phenyl mercaptan is 450- to 650-fold more toxic than ethyl mercaptan, 120-fold more toxic than methyl mercaptan, and 77- to 180-fold more toxic than hydrogen sulfide. However, using a lower total uncertainty factor of 10 yields AEGL-3 values that suggest phenyl mercaptan is 150- to 230fold more toxic than ethyl mercaptan, 29- to 42-fold more toxic than methyl mercaptan, and 25- to 58-fold more toxic than hydrogen sulfide. Also, the AEGL-3 point of departure (10.3 ppm) is approximately one-third the 4-h LC50 in rats (33 ppm). Thus, a total uncertainty factor of 10 yields values that are protective and are more consistent with relative toxicity data.
The concentration-exposure time relationship for many irritant and systemically-acting vapors and gases may be described by the equation Cn × t = k, where the exponent n ranges from 0.8 to 3.5 (ten Berge et al. 1986). To obtain conservative and protective AEGL values in the absence of an empirically derived chemical-specific exponent, temporal scaling was performed using default values of n = 3 when extrapolating to shorter durations (10 min, 30 min, and 1 h) and n = 1 when extrapolating to longer durations (8 h). AEGL-3 values for phenyl mercaptan are presented in Table 3-7, and calculations are presented in Appendix A.
Time scaling from the 4-h point of departure to the 10-min AEGL-3 value is supported by the 1-h rat lethality data (Stauffer Chemical Company 1969). The estimated 1-h lethality threshold for rats is 141 ppm (one-third of the LC50 value [422 ppm ÷ 3 = 141 ppm]). Time scaling to the 10-min duration, using n = 3, and applying a total uncertainty factor of 10 would yield a 10-min value of 26 ppm, suggesting that the 10-min AEGL value of 3.0 ppm is protective.
TABLE 3-7 AEGL-3 Values for Phenyl Mercaptan
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 3.0 ppm (14 mg/m3) |
2.1 ppm (9.5 mg/m3) |
1.6 ppm (7.2 mg/m3) |
1.0 ppm (4.5 mg/m3) |
0.52 ppm (2.3 mg/m3) |
8. SUMMARY OF AEGLS
8.1. AEGL Values and Toxicity End Points
Table 3-8 presents AEGL values for phenyl mercaptan. AEGL-1 values are not recommended because of insufficient data. Data on phenyl mercaptan were also inadequate for deriving AEGL-2 values, so AEGL-2 values were estimated by taking one-third of the AEGL-3 values. These calculations are considered thresholds for the inability to escape, and are appropriate because of the steep concentration-response curve for phenyl mercaptan. AEGL-3 values were based on the LC01 of 10.3 ppm in rats exposed to phenyl mercaptan for 4 h (Fairchild and Stokinger 1958).
8.2. Comparisons with Other Standards and Guidelines
Standards and guidance levels for workplace and community exposures to phenyl mercaptan are presented in Table 3-9.
The data requirements for establishing other standards and guidelines differ from those of AEGLs. The documentation for those values does not provide sufficient detail to understand the quantitative basis of the TLV®-TWA established in 2004 by of the American Conference of Governmental Industrial Hygienists or the earlier TLV-TWA (which was the basis for the Dutch MAC). The NIOSH REL ceiling value was derived in 1978 (NIOSH 1978) as follows:
Because benzenethiol [phenyl mercaptan] is not only more toxic than the other thiols (Fairchild and Stokinger, 1958) but also has a comparatively marked potential for causing eye and organ damage, e.g., at 0.72 ppm, at one-third the concentration of ethanethiol (2.1 ppm), as indicated by Katz and Talbert (1930), NIOSH recommends that the concentration of benzenethiol in the workplace air should not exceed 0.1 ppm (0.45 mg/cu m) as a ceiling concentration for any 15-min period.
8.3. Data Adequacy and Research Needs
Data on acute inhalation exposure to phenyl mercaptan in humans and animals are sparse, and the few studies available are old and poorly reported. There were insufficient data to establish a chemical-specific time-scaling exponent for phenyl mercaptan.
TABLE 3-8 AEGL Values for Phenyl Mercaptan
| Classification | 10 min | 30 min | 1 h | 4 h | 8 h |
| AEGL-1a (nondisabling) | NR | NR | NR | NR | NR |
| AEGL-2 (disabling) | 1.0 ppm (4.5 mg/m3) |
0.70 ppm (3.2 mg/m3) |
0.53 ppm (2.4 mg/m3) |
0.33 ppm (1.5 mg/m3) |
0.17 ppm (0.77 mg/m3) |
| AEGL-3 (lethal) | 3.0 ppm (14 mg/m3) |
2.1 ppm (9.5 mg/m3) |
1.6 ppm (7.2 mg/m3) |
1.0 ppm (4.5 mg/m3) |
0.52 ppm (2.3 mg/m3) |
aThe absence of AEGL-1 values does not imply that concentrations below AEGL-2 values are without effect.
TABLE 3-9 Standards and Guidelines for Phenyl Mercaptan
| Guideline | Exposure Duration | ||||
| 10 min | 30 min | 1 h | 4 h | 8 h | |
| AEGL-1 | NR | NR | NR | NR | NR |
| AEGL-2 | 1.0 ppm (4.5 mg/m3) |
0.70 ppm (3.2 mg/m3) |
0.53 ppm (2.4 mg/m3) |
0.33 ppm (1.5 mg/m3) |
0.17 ppm (0.77 mg/m3) |
| AEGL-3 | 3.0 ppm (14 mg/m3) |
2.1 ppm (9.5 mg/m3) |
1.6 ppm (7.2 mg/m3) |
1.0 ppm (4.5 mg/m3) |
0.52 ppm (2.3 mg/m3) |
| TLV-TWA (ACGIH)a | 0.1 ppm (0.45 mg/m3) |
||||
| REL-C (NIOSH)b | 0.1 ppm ( 0.5 mg/m3) |
0.1 ppm (0.5 mg/m3) |
0.1 ppm (0.5 mg/m3) |
0.1 ppm (0.5 mg/m3) |
0.1 ppm (0.5 mg/m3) |
| MAC (The Netherlands) c | 0.5 ppm (2 mg/m3) |
||||
aTLV-TWA (threshold limit value-time weighted average, American Conference of Governmental Industrial Hygienists [ACGIH 2012]) is the time-weighted average concentration for a normal 8-h workday and a 40-h workweek, to which nearly all workers may be repeatedly exposed, day after day, without adverse effect.
bREL-C (recommended exposure limit-ceiling, National Institute for Occupational Safety and Health [NIOSH 2011]) is a ceiling value that should not be exceeded at any time during a workday.
cMAC (maximaal aanvaarde concentratie [maximal accepted concentration], Dutch Expert Committee for Occupational Standards, The Netherlands [MSZW 2004]), is defined analogous to the ACGIH TLV-TWA.
9. REFERENCES
ACGIH (American Conference of Governmental Industrial Hygienists). 2012. Phenyl Mercaptan (CAS Reg. No. 108-98-5). Threshold Limit Values for Chemical Substances and Physical Agents and Biological Exposure Indices. American Conference of Governmental Industrial Hygienists, Cincinnati OH.
AIHA (American Industrial Hygiene Association). 1989. Odor Thresholds for Chemicals with Established Occupational Health Standards. American Industrial Hygiene Association, Fairfax, VA.
Amoore, J.E., and E. Hautala. 1983. Odor as an aid to chemical safety: Thresholds compared with threshold limit values and volatilities for 214 industrial chemicals in air and water dilution. J. Appl. Toxicol. 3(6):272-290.
Fairchild, E.J., and H.E. Stokinger. 1958. Toxicologic studies on organic sulfur compounds. I. Acute toxicity of some aliphatic and aromatic thiols (mercaptans). Am. Ind. Hyg. Assoc. J. 19(3):171-189.
Hazleton Laboratories. 1951. Acute Oral, Acute and Chronic Dermal and Vapor Toxicity Study of Thiophenol, October 20, 1951. Submitted to EPA by Zeneca Specialties, Wilmington, DE, with Cover Letter Dated 05/06/94. EPA Document No. 86940000968. Microfiche No. OTS0557378.
Horiguchi, M. 1960. An experimental study on the toxicity of methyl mercaptan in comparison with hydrosulfide [in Japanese]. J. Osaka City Med. Cent. 9:5257-5293.
HSDB (Hazardous Substances Data Bank). 2009. Thiophenol (CAS Reg. No. 108-98-5). TOXNET, Specialized Information Services, U.S. National Library of Medicine, Bethesda, MD [online]. Available: http://toxnet.nlm.nih.gov/cgi-bin/sis/htmlgen?HSDB [accessed Sept. 2012].
Katz, S.H., and E.J. Talbert. 1930. Intensities of Odors and Irritating Effects of Warning Agents for Inflammable and Poisonous Gases. Technical Report No. 480. Washington, DC: U.S. Government Printing Office.
LaVoie, E., L. Tulley, E. Fow, and D. Hoffmann. 1979. Mutagenicity of aminophenol and nitrophenol ethers, sulfides, and disulfides. Mutat. Res. 67(2): 123-131.
Litchfield J.T., and F. Wilcoxon. 1949. A simplified method of evaluating dose-effect experiments. J. Pharmacol. Exp. Ther. 96(2):99-113.
McBain, J.B., and J. Menn. 1969. S-methylation, oxidation, hydroxylation, and conjugation of thiophenol in the rat. Biochem. Pharmacol. 18(9):2282-2285.
MSZW (Ministerie van Sociale Zaken en Werkgelegenheid). 2004. Nationale MAC-lijst 2004: Benzeenthiol. Den Haag: SDU Uitgevers [online]. Available: http://www.lasrook.net/lasrookNL/maclijst2004.htm [accessed June 27, 2013].
NIOSH (National Institute for Occupational Safety and Health). 1978. Criteria for a Recommended Standard. Occupational Exposure to n-Alkane Monothiols, Cyclohex-anethiol, and Benzenethiol. DHEW (NIOSH) Publication No. 78-213. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, Atlanta, GA [online]. Available: http://www.cdc.gov/niosh/pdfs/78-213a.pdf [accessed June 27, 2013].
NIOSH (National Institute for Occupational Safety and Health). 2011. NIOSH Pocket Guide to Chemical Hazards (NPG): Benzenethiol. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, Atlanta, GA [online]. Available: http://www.cdc.gov/niosh/npg/npgd0050.html [accessed June 27, 2003].NRC (National Research Council). 1993. Guidelines for Developing Community Emergency Exposure Levels for Hazardous Substances. Washington, DC: National Academy Press.
NRC (National Research Council). 2001. Standing Operating Procedures for Developing Acute Exposure Guideline Levels for Hazardous Chemicals. Washington, DC: National Academy Press.
NTP (National Toxicology Program). 1994a. Final Report on the Developmental Toxicity of Thiophenol (CAS Reg. No. 108-98-5) in Sprague-Dawley CD Rats on Gestational Days 6 through 15. NTP TER92133. NTIS No. PB94-155009 U.S. Depart-
ment of Health and Human Services, Public Health Services, National Institutes of Health, National Institute of Environmental Health Sciences, Research Triangle Park, NC.
NTP (National Toxicology Program). 1994b. Final Report on the Developmental Toxicity of Thiophenol (CAS Reg. No. 108-98-5) in New Zealand White Rabbits. TER92134. NTIS No. PB94-201183. U.S. Department of Health and Human Services, Public Health Services, National Institutes of Health, National Institute of Environmental Health Sciences, Research Triangle Park, NC.
NTP (National Toxicology Program). 1996. Final Report on the Reproductive Toxicity of Thiophenol (CAS Reg. No. 108-98-5) Administered by Gavage to Sprague-Dawley CD Rats. RACB94001. U.S. Department of Health and Human Services, Public Health Services, National Institutes of Health, National Institute of Environmental Health Sciences, Research Triangle Park, NC.
RTECS (Registry of Toxic Effects of Chemical Substances). 2009. Benzenethiol. RTECS No. DC0525000. National Institute for Occupational Safety and Health [online]. Available: http://www.cdc.gov/niosh-rtecs/DC802C8.html [accessed June 27, 2013].
Shertzer, H.G. 2012. Organic sulfur compounds. Pp. 1039-1076 in Patty’s Toxicology, 6th Ed., E. Bingham and B. Cohrssen, eds. New York: Wiley.
Smith, R.P. 1991. Toxic responses of the blood. Pp. 276-278 in Casarett and Doull’s Toxicology: The Basic Science of Poisons, 4th Ed., M.O. Amdur, J. Doull, and C.D. Klaassen, eds. New York: Pergamon Press.
Stauffer Chemical Company. 1969. Acute Inhalation LC50 Study of Thiophenol in Male and Female Rats. Toxicology Lab Report T-1403, October 13, 1969. Submitted to EPA by Zeneca Specialties, Wilmington, DE, with Cover Letter Dated 05/06/94. EPA Document No. 86940000970. Microfiche No. OTS0557380.
Tansy, M.F., F.M. Kendall, J. Fantasia, W.E. Landin, R. Oberly, and W. Sherman. 1981. Acute and subchronic toxicity studies of rats exposed to vapors of methyl mercaptan and other reduced-sulfur compounds. J. Toxicol. Environ. Health 8(1-2):71-88.
ten Berge, W.F., A. Zwart, and L.M. Appelman. 1986. Concentration-time mortality response relationship of irritant and systemically acting vapours and gases. J. Hazard. Mater. 13(3):301-309.
APPENDIX A
DERIVATION OF AEGL VALUES FOR PHENYL MERCAPTAN
Derivation of AEGL-1 Values
AEGL-1 values are not recommended for phenyl mercaptan because of insufficient data. The absence of AEGL-1 values does not imply that concentrations below AEGL-2 values will be without effect.
Derivation of AEGL-2 Values
In the absence of relevant data to derive AEGL-2 values and because phenyl mercaptan has a steep concentration-response curve, AEGL-3 values were divided by 3 to estimate a threshold for inability to escape.
|
10-min AEGL-2: |
120 ppm ÷ 3 = 40 ppm |
|
10-min AEGL-2: |
3.0 ppm ÷ 3 = 1.0 ppm |
|
30-min AEGL-2: |
2.1 ppm ÷ 3 = 0.70 ppm |
|
1-h AEGL-2: |
1.6 ppm ÷ 3 = 0.53 ppm |
|
4-h AEGL-2: |
1.0 ppm ÷ 3 = 0.33 ppm |
|
8-h AEGL-2: |
0.52 ppm ÷ 3 = 0.17 ppm |
Derivation of AEGL-3 Values
|
Key study: |
Fairchild, E.J., and H.E. Stokinger. 1958. Toxicologic studies on organic sulfur compounds. I. Acute toxicity of some aliphatic and aromatic thiols (mercaptans). Am. Ind. Hyg. Assoc. J. 19(3):171-189. |
|
Toxicity end point: |
Estimated lethality threshold for rats, 4-h LC01 of 10.3 ppm |
|
Time scaling: |
Cn × t = k (default values of n = 3 for extrapolating to shorter durations and n = 1 for extrapolating to longer durations) |
|
Uncertainty factors: |
3 for interspecies differences |
|
10-min AEGL-3: |
C3 × 0.167 h = 4,371 ppm- h |
|
30-min AEGL-3: |
C3 × 0.5 h = 4,371 ppm- h |
|
1-h AEGL-3: |
C3 × 1 h = 4,371 ppm- h |
|
4-h AEGL-3: |
10.3 ppm ÷ 10 = 1.0 ppm |
|
8-h AEGL-3: |
C1 ÷ 8 h = 41.2 ppm -h |
APPENDIX B
ACUTE EXPOSURE GUIDELINE LEVELS FOR PHENYL MERCAPTAN
Derivation Summary
AEGL-1 VALUES
AEGL-1 values for phenyl mercaptan are not recommended because of insufficient data. The absence of AEGL-1 values does not imply that concentrations below AEGL-2 values are without effect.
AEGL-2 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 1.0 ppm (4.5 mg/m3) |
0.70 ppm (3.2 mg/m3) |
0.53 ppm (2.4 mg/m3) |
0.33 ppm (1.5 mg/m3) |
0.17 ppm (0.77mg/m3) |
Data adequacy: Data inadequate to derive AEGL-2 values. AEGL-3 values were divided by 3 to estimate thresholds for the inability to escape.
AEGL-3 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 3.0 ppm (14 mg/m3) |
2.1 ppm (9.5 mg/m3) |
1.6 ppm (7.2 mg/m3) |
1.0 ppm (4.5 mg/m3) |
0.52 ppm (2.3 mg/m3) |
Reference: Fairchild, E.J., and H.E. Stokinger. 1958. Toxicologic studies on organic sulfur compounds. I. Acute toxicity of some aliphatic and aromatic thiols (mercaptans).
Am. Ind. Hyg. Assoc. J. 19(3):171-189.
Test Species/Strain/Sex/Number: Rats, Wistar, 5-10 males per group
Exposure route/Concentrations/Durations: Inhalation; 0, 20, 31, 41, 52, 79, or 132 ppm for 4 h
|
Effects: |
Mortality in Rats |
|
20 |
0/5 |
|
31 |
5/10 |
|
41 |
4/6 |
|
52 |
5/5 |
|
79 |
10/10 |
|
132 |
10/10 |
|
LC50 = 33 ppm |
|
|
LC01 = 10.3 ppm |
|
AEGL-3 VALUES
End point/Concentration/Rationale: Estimated lethality threshold in rats, 4-h LC01 of 10.3 ppm
Uncertainty factors/Rationale:
Intraspecies: 3
Interspecies: 3
Interspecies and intraspecies uncertainty factors of 3 (total uncertainty factor of 10) are considered sufficient because the mechanism of action for phenyl mercaptan toxicity (cytochrome oxidase inhibition) is not expected to vary greatly between or within species. Although an interspecies or intraspecies uncertainty factor of 10 might normally be applied because of limited data, a total uncertainty factor of 30 would yield AEGL values that are inconsistent with the AEGL values derived for the structural and mechanistic analogs ethyl mercaptan, methyl mercaptan, and hydrogen sulfide, all of which have a more robust data set than phenyl mercaptan. For example, rat lethality data suggest that the acute inhalation toxicity of phenyl mercaptan is approximately 140-fold greater than that of ethyl mercaptan, 20-fold greater than methyl mercaptan, and 13- fold greater than hydrogen sulfide. The 4-h rat LC50 value for phenyl mercaptan was 33 ppm (Fairchild and Stokinger 1958), whereas the 4-h rat LC50 value for ethyl mercaptan was 4,740 ppm (Fairchild and Stokinger 1958), the 4-h LC50 value for methyl mercaptan was 675 ppm (Tansy et al. 1981), and the 4-h LC50 value for hydrogen sulfide was 444 ppm (Tansy et al. 1981). Using a total uncertainty factor of 30 would yield values that suggest phenyl mercaptan is 450- to 650-fold more toxic than ethyl mercaptan, 120-fold more toxic than methyl mercaptan, and 77- to 180-fold more toxic than hydrogen sulfide. However, a lower total uncertainty of 10 yields AEGL-3 values that suggest phenyl mercaptan is 150- to 230-fold more toxic than ethyl mercaptan, 29- to 42-fold more toxic than methyl mercaptan, and 25- to 58-fold more toxic than hydrogen sulfide. Also, the AEGL-3 point of departure (10.3 ppm) is approximately one-third the 4-h LC50 in rats (33 ppm). Thus, factors of 3 for interspecies difference and intraspecies variability are protective and are more consistent with relative toxicity data.
Modifying factor: Not applicable
Animal-to-human dosimetric adjustment: Insufficient data
Time scaling: Cn × t = k; default values of n = 1 for extrapolation from shorter to longer durations (8 h) and n = 3 for extrapolation from longer to shorter durations (10 min, 30 min, and 1 h) were used. Time scaling from the 4-h point of departure to the 10-min AEGL-3 value is supported by the 1-h rat lethality data (Stauffer Chemical Company 1969). The estimated 1-h rat lethality threshold is 141 ppm (one-third of the LC50 value [422 ppm ÷ 3 = 141 ppm]). Time scaling to 10-min using an exponent of n = 3 and applying a total uncertainty factor of 10 would yield a 10-min value of 26 ppm, suggesting that the 10-min AEGL-3 value of 3.0 ppm is protective.
Data adequacy: The study was well conducted and used a sufficient number of animals. The selected end point represents an estimate threshold for lethality.
APPENDIX C
CATEGORY PLOT FOR PHENYL MERCAPTAN
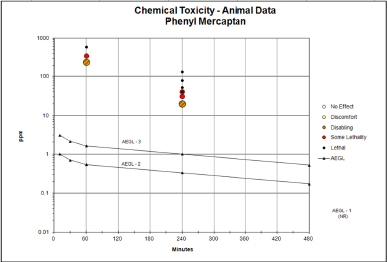
FIGURE C-1 Category plot of toxicity data and AEGL values for phenyl mercaptan. The decimal point is lost on this log-scale plot.
TABLE C-1 Data Used in Category Plot for Phenyl Mercaptan
| Source | Species | Sex | No. Exposures | ppm | Minutes | Category | Comments |
| AEGL-1 | NR | 10 | AEGL | ||||
| AEGL-1 | NR | 30 | AEGL | ||||
| AEGL-1 | NR | 60 | AEGL | ||||
| AEGL-1 | NR | 240 | AEGL | ||||
| AEGL-1 | NR | 480 | AEGL | ||||
| AEGL-2 | 1.00 | 10 | AEGL | ||||
| AEGL-2 | 0.70 | 30 | AEGL | ||||
| AEGL-2 | 0.53 | 60 | AEGL | ||||
| AEGL-2 | 0.33 | 240 | AEGL | ||||
| AEGL-2 | 0.17 | 480 | AEGL | ||||
| AEGL-3 | 3.00 | 10 | AEGL | ||||
| Source | Species | Sex | No. Exposures | ppm | Minutes | Category | Comments |
| AEGL-3 | 2.10 | 30 | AEGL | ||||
| AEGL-3 | 1.60 | 60 | AEGL | ||||
| AEGL-3 | 1.00 | 240 | AEGL | ||||
| AEGL-3 | 0.52 | 480 | AEGL | ||||
| Fairchild and Stokinger 1958 | Mouse | Male | 1 | 20 | 240 | 2 | |
| Mouse | Male | 1 | 31 | 240 | SL | Mortality (7/10) | |
| Mouse | Male | 1 | 41 | 240 | 3 | Mortality (10/10) | |
| Mouse | Male | 1 | 52 | 240 | 3 | Mortality (10/10) | |
| Mouse | Male | 1 | 79 | 240 | 3 | Mortality (5/5) | |
| Rat | Male | 1 | 20 | 240 | 2 | ||
| Rat | Male | 1 | 31 | 240 | SL | Mortality (5/10) | |
| Rat | Male | 1 | 41 | 240 | SL | Mortality (4/6) | |
| Rat | Male | 1 | 52 | 240 | 3 | Mortality (5/5) | |
| Rat | Male | 1 | 79 | 240 | 3 | Mortality (10/10) | |
| Rat | Male | 1 | 132 | 240 | 3 | Mortality (10/10) | |
| Stauffer Chemical Company 1969 | Rat | Both | 1 | 244 | 60 | 2 | |
| Rat | Both | 1 | 346 | 60 | SL | Mortality (3/10) | |
| Rat | Both | 1 | 595 | 60 | 3 | Mortality (10/10) | |
For category: 0 = no effect, 1 = discomfort, 2 = disabling, 3 = lethal; SL = some lethality.























