Considerations for Returning Individual Genomic Results from Population-Based Surveys: Focus on the National Health and Nutrition Examination Survey: Proceedings of a Workshop (2023)
Chapter: 4 Regulatory Issues in the Return of Genetic Results (Session 3)
4
Regulatory Issues in the Return of Genetic Results (Session 3)
This chapter summarizes the presentations and discussion in Session 3, which took place on December 2, 2022. The session focused on regulatory issues when returning genetic results to research participants. Ingrid A. Holm, Harvard Medical School and Boston Children’s Hospital, moderated the session.1
CLINICAL LABORATORY IMPROVEMENT AMENDMENTS LAB CERTIFICATION AND TEST VALIDATION
Benjamin Solomon, Clinical Director of the National Human Genome Research Institute (NHGRI), spoke next about Clinical Laboratory Improvement Amendments (CLIA) lab certification and test validation. Solomon explained that he would be drawing upon his past experiences running a certified clinical genetics/genomics lab, which ran about 100,000 samples and 200,000 tests per year (primarily exomes/genomes/large panels), as well as his experiences as a principal investigator on several research studies involving whole-genome sequencing in healthy and affected individuals that involved an array of clinical and nonclinical analyses.
___________________
1 Video recordings of the presentations and discussions, along with copies of the presenters’ slides for Session 3, are available online at https://www.nationalacademies.org/event/12-02-2022/workshop-on-considerations-for-returning-individual-genomic-results-from-population-based-surveys-focus-on-the-national-health-and-nutrition-examination-survey-day-1-virtual
CLIA Certification and Validation
Solomon began by providing a list of some of the most frequent categories of clinical lab certifications in genetics/genomics in the United States. Many labs that conduct clinical genetic/genomic testing will have multiple certifications and may have different certifications for different tests or testing areas. CLIA is the main oversight system for this type of laboratory testing; it is administered through the Centers for Medicare & Medicaid Services (CMS), with involvement from the Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC). Solomon noted that the College of American Pathologists (CAP) is another voluntary certification program, which informally is viewed as more stringent and more directly involved in providing information and guidance about best practices in applicable areas, such as a recent set of guidelines around next-generation sequencing data validation. He explained that in addition to some specific FDA-approved assays that are currently available, there are ongoing discussions about FDA possibly becoming more involved in the regulation of genetic and other lab-developed tests, which would lead to a future with more FDA-approved assays, tests, and machines. Solomon noted that certain states, such as New York and Washington, have an additional layer of steps and requirements.
Noting that some research studies like All of Us are not necessarily CLIA approved (since alternative processes may be available in some situations), Solomon explained that the CLIA framework is required if individual results that may be used for health care decisions or are otherwise clinically important are returned. He noted that one untrue myth is that CLIA is involved in determining whether a lab can become commercial or can charge for testing; rather, a commercial lab will face reimbursement and other financial repercussions if they are not meeting CLIA requirements. Solomon stressed that while the CLIA framework includes some clinical components, such as specifying who is involved in the CLIA process, CLIA focuses on analytic validity, and there is not a specific CLIA requirement related to clinical validation.
Solomon next outlined three testing categories for CLIA (this is related to but different from risk categories). The first category is for waived tests, which include very simple assays with little risk of error and therefore little risk of harm. Most genetics and genomics testing is considered moderate to high complexity and therefore falls within the second category. A third and final category includes those specific specialty areas, such as cytogenetics and bacteriology, that have their own requirements.
Solomon highlighted several components of CLIA that may be required depending on the category of testing. These include proficiency testing (e.g., for some nonspecialty areas: biannual self-testing, with voluntary
proficiency testing exchange as available), quality systems, and inspections of paperwork/documentation, procedure manuals, personnel interviews, and patient charts (biennial, usually with 2 weeks’ notice). He explained that CLIA includes some requirements of key personnel, such as the lab director, technical supervisor, clinical consultant, and general supervisor. Solomon noted parenthetically that FDA-related requirements depend on the exact test being performed, such as a specific FDA-approved assay. Presenting the information shown in Box 4-1, Solomon offered an example of personnel requirements in a mid-sized genomics lab.
Solomon stated that from his experience, some of the most important components for high-quality labs are day-to-day tasks, such as making sure there are processes in place for minimizing the chance of sample contamination or mix-ups. He emphasized that laboratories need to have processes in place that allow them to identify, trace back, and correct samples that could get mixed up within the lab. Solomon offered that rather than doing all of its own CLIA-certified testing, a brand-new, research-focused lab conducting a study today may want to consider partnering with a more experienced lab for clinical validation. “There is a heavy lift involved, especially if you’re doing all sorts of different things that may be changing over time, which is the norm in genetics and genomics,” added Solomon.
Processes and Examples
Solomon outlined a hypothetical case of a research study planning to sequence the genomes of 10,000 people, where the consent includes the possible receipt of individual, clinically relevant results related to the genome sequencing. He next offered three models for approaching CLIA-related requirements for the hypothetical study. (However, he mentioned that some studies, such as All of Us, may pursue different routes.) He explained that in the “pure CLIA model” everything (e.g., DNA extraction, sequencing, orthogonal confirmation) is done from the outset in a CLIA-compliant manner in a lab that is CLIA certified. This requires extensive logistic front-loading. Solomon explained that while this model allows for more flexibility downstream to do research analyses, as well as clinical management and reporting (e.g., may obviate needing to do orthogonal confirmation in some situations), it may be difficult to set up and maintain.
Solomon next defined the next model, which he said is used more frequently, as one in which the research-based sequencing and analyses are done with good research practices in a non-CLIA-compliant manner. In this model, if the research study identifies a genetic finding that could be clinically important to return, participants are recontacted to collect a new sample in a CLIA-compliant manner. “Recontact typically involves letting participants know there may be a finding of interest that requires technical validation, then collecting a new sample for variant-specific, CLIA-based orthogonal confirmation and return of results,” said Solomon. He noted that a major challenge is that this model requires ongoing communication and contact with study participants, for which the National Health and Nutrition Examination Survey (NHANES) is not currently set up.
The final example Solomon offered is a “hybrid approach,” such as that used by the Geisinger MyCode project.2 The samples in this study were collected in a CLIA-compliant manner and the analyses were done on a research basis. Data and CLIA-compliant samples were shared with a clinical lab for confirmation and reporting. If samples had not been collected in a CLIA-compliant manner or if there were no samples still available for confirmation, then new samples could be obtained for confirmation/reporting, just as with the previous model. Solomon, who is the editor-in-chief of the journal in which the referenced paper was published, explained that the publication goes “into the nitty gritty in a good way about how much time
___________________
2 Kelly, M. A., Leader, J. B., Wain, K. E., Bodian, D., Oetjens, M. T., Ledbetter, D. H., Martin, C. L., and Strande, N. T. (2021). Leveraging population-based exome screening to impact clinical care: The evolution of variant assessment in the Geisinger MyCode research project. American Journal of Medical Genetics. Part C, Seminars in Medical Genetics, 187(1), 83–94. https://doi.org/10.1002/ajmg.c.31887
does it take to do this, how much does it cost to do this … [and] gives a very concrete and realistic picture of the processes that have been required.”
Solomon concluded his talk with several lessons learned. Partnering with a certified clinical lab with lots of experience in this area can be helpful in terms of both the logistics of sample confirmation and regulatory management, as well as having access to the lab’s databases of variants for comparison. He explained that publicly available databases like ClinVar, while enormously helpful, do not yet have enough data or clear justification for every possible variant, whereas clinical labs may have more specific information, such as clinical findings in a particular patient or family. That is, partnering with a large lab that has done lots of sequencing can help with variant classification in some situations. Solomon highlighted that variants beyond single nucleotide variants, such as certain types of structural variants, can be medically important and often require different assays for confirmation. Finally, Solomon urged the audience to consider up front that even in the best-run labs, issues such as “sample swaps” can happen; he offered a final, broader lesson that “challenging participant-specific situations will arise: have an experienced advisory panel on hand!”
POTENTIAL FDA OVERSIGHT AND APPROVAL
Beth Collins, a contractor for Columbus Technologies and Services working with the National Institutes of Health (NIH) All of Us Research Program to ensure Investigational Device Exemption (IDE) sponsor communications and essential documentation meet compliance under Title 21 of the Code of Federal Regulations (C.F.R.) Part 812,3 spoke next on the topic of FDA regulatory issues that one may encounter when returning genetic research results. Collins explained that their talk would focus on the significant risk and IDEs related to a research sponsor’s responsibilities. Putting their talk into context, Collins presented the “expanded and adapted” timeline shown in Figure 4-1, which started in November 2018 with the FDA indicating that the All of Us return of health-related genomics results is considered a Significant Risk Device Study. Collins noted that unlike many Significant Risk Devices, the medical device used by All of Us was not implantable but still falls under the federal regulations under C.F.R. Title 21. Collins highlighted the many meetings, conversations, and milestones up to the present that have taken place between the All of Us Research Program and the FDA, with Collins explaining, “there are a lot of meetings and questions and evidence that might need to be provided to the FDA prior to the actual IDE application to make sure that it goes smoothly.” Collins explained that the initial meetings with the FDA concluded that an IDE
___________________
3 See https://www.eC.F.R..gov/current/title-21/chapter-I/subchapter-H/part-812?toc=1
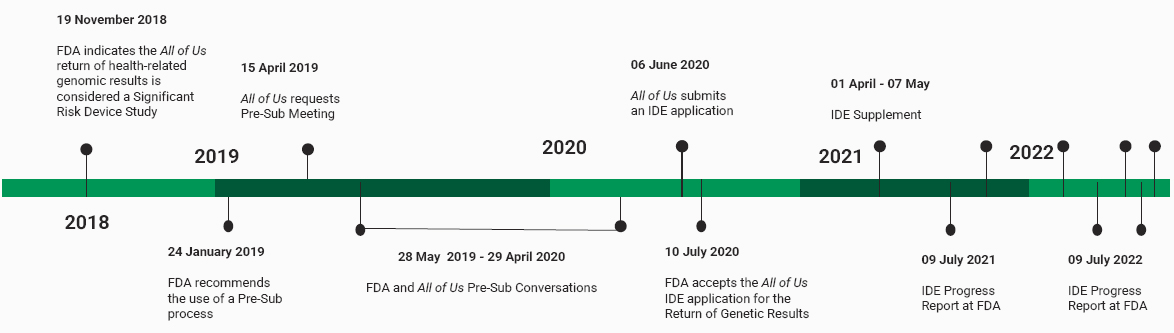
NOTE: FDA = Food and Drug Administration; IDE = Investigational Device Exemption.
SOURCES: Workshop presentation by Beth Collins on December 2, 2022 (slide 2). Adapted and expanded from timeline originally presented in Venner, E., Muzny, D., Smith, J. D., Walker, K., Neben, C. L., Lockwood, C. M., Empey, P. E., Metcalf, G. A., Kachulis, C., The All of Us Research Program Regulatory Working Group, Mian, S., Musick, A., Rehm, H. L., Harrison, S., Gabriel, S., Gibbs, R. A., Nickerson, D., Zhou, A. Y., Doheny, K., Ozenberger, B., Topper, S. E., and Lennon, N. J. (2022). Whole-genome sequencing as an investigational device for return of hereditary disease risk and pharmacogenomic results as part of the All of Us Research Program. Genome Medicine, 14, 34. https://doi.org/10.1186/s13073-022-01031-z
application was required for the health-related return of genetic results to participants to ensure acceptable risk levels.
Looking closely at those activities that occurred after the application was accepted by the FDA in 2020, Collins explained that the “FDA considers the All of Us Research Program Return of Genetic Results (All of Us Research Program gRoR) investigational device to be the entire test system, which includes sample collection, sample preparation, sequencing and genotyping, variant calling, variant interpretation, and generation of the test report provided to study participant.” She shared that because the scope of regulations under C.F.R. Title 21 includes the actual shipment of those devices, which can have implications about the return of results, the study needed to be “a little bit more flexible in defining how we consume these regulations.” Collins explained that as the IDE sponsor (21 C.F.R. § 812.40, General responsibilities of sponsors), the All of Us Research Program is responsible for each of the following—from consent through biospecimen collections, through the embedded quality systems for the return of results:4
- selecting qualified investigators and providing them with the information needed to comply properly with the parameters of the IDE (i.e., genetic testing and return of results),
- ensuring proper monitoring of the investigation,
- ensuring that Institutional Review Board (IRB) review and approval are obtained,
- submitting an IDE application to the FDA, and
- ensuring that any reviewing IRB and the FDA are promptly informed of significant new information about the investigation.
Turning next to 21 C.F.R. 8 § 14.42 (FDA and IRB approval),5 Collins shared that the All of Us IRB is the single IRB of record and maintains compliance with the federal regulations outlined in 21 C.F.R. § 50 (protection of human subjects)6 and 21 C.F.R. § 56 (IRBs).7 This means that the All of Us Research Program maintains approval from both the All of Us IRB and the FDA. As the IDE sponsor, the program must have processes in place to ensure that all monitors and investigators are qualified to perform duties as assigned along the IDE pathway. Collins added that the program also wanted to make sure it had the resources needed “to document and
___________________
4 See https://www.eC.F.R..gov/current/title-21/chapter-I/subchapter-H/part-812/subpart-C/section-812.40
5 See https://www.eC.F.R..gov/current/title-21/chapter-I/subchapter-H/part-812/subpart-C/section-812.42
6 See https://www.eC.F.R..gov/current/title-21/chapter-I/subchapter-A/part-50
7 See https://www.eC.F.R..gov/current/title-21/chapter-I/subchapter-A/part-56
maintain this compliance for the responsible investigators,” underscoring that 21 C.F.R. § 812.150 (reports)8 includes extensive record keeping components from both the investigator and sponsor standpoints and, as is the case for the All of Us Research Program, may be required to keep documentation for both parts.
For the context of this workshop, Collins advised that it is critical to understand the FDA’s requirements for progress reports before the data capturing system is built out. She emphasized that a program must be able capture and quantify any unexpected events that occur. She also highlighted that a study needs to have processes in place to report any withdrawal of IRB or FDA approval. Collins explained that the study must have in place procedures for recording and mitigating strategies in the event a person receives the report of their health without providing consent.
Under 21 C.F.R. § 812.45 (informing investigators),9 the sponsor must supply all investigators participating in the investigation with copies of the investigational plan and report of prior investigations of the device, explained Collins. She stated that because compliance with 21 C.F.R. § 812.46 (monitoring investigations)10 requires monitoring for any unanticipated adverse device effects, the All of Us Research Program includes a mechanism for immediately terminating the part of the study that would require further investigation, and processes for making sure that the studies are reinstated properly with the approval of IDE and the IRB, said Collins. Collins underscored the importance of being “audit-inspection ready at all times” in response to 21 C.F.R. § 812.145 (inspections),11 which includes information about where, when, and how the FDA can conduct inspections. In particular, she highlighted that the FDA may request to look at records that identify participants.
Collins highlighted the other regulations that should be considered while building out an investigational plan and protocol. Collins outlined that 21 C.F.R. Subpart A includes the labeling of investigational devices, prohibition of promotion and other practices, and “making sure that there are research results looking at the ways that we speak to our program’s return of these results.” She outlined that 21 C.F.R. Subpart B speaks to the application process, investigational plan, and confidentiality of data and information. She emphasized that prior to any reviews with the FDA,
___________________
8 See https://www.eC.F.R..gov/current/title-21/chapter-I/subchapter-H/part-812/subpart-G/section-812.150
9 See https://www.eC.F.R..gov/current/title-21/chapter-I/subchapter-H/part-812/subpart-C/section-812.45
10 See https://www.eC.F.R..gov/current/title-21/chapter-I/subchapter-H/part-812/subpart-C/section-812.46
11 See https://www.eC.F.R..gov/current/title-21/chapter-I/subchapter-H/part-812/subpart-G/section-812.145
a study should develop a written protocol and risk analysis looking at how (a) the possible risks might be mitigated and (b) those risks may be updated depending on the research findings, as well as a standard operating procedure for monitoring. Collins’s final slide included additional resources that could be useful as one explores whether they need to go down the pathway of a significant-risk device exemption.
NAVIGATING FEDERAL AND STATE-SPECIFIC LAWS GOVERNING GENETIC TESTING AND THE RETURN OF RESULTS
Katherine Blizinsky, the policy director for the All of Us Research Program, gave the day’s final talk on navigating federal and state-specific laws governing genetic testing and the return of results. Blizinsky began by providing the audience with some disclaimers. She first reminded the audience that although she has extensive experience analyzing and writing laws, she is not an attorney. Blizinsky, therefore, warned that the audience should consider her advice accordingly. She also noted that her talk would be limited to laws that directly apply to the All of Us program and would not be an extensive list of all the laws and policies that might be relevant to the return of genetic results. Specifically, she noted that CLIA, “the IDE process,” and the Health Insurance Portability and Accountability Act (HIPAA) would not be discussed. Blizinsky’s goal in sharing these disclaimers was to emphasize that there may be laws necessary for NHANES’s legal compliance that are not discussed.
Blizinsky described laws regarding the return of genetic information as necessary to provide “specific requirements for the conduct of lawful genetic testing” and the “specific rights conferred to the individual being tested.” Such laws “exist at both the federal and state levels, with federal protections largely setting the stage for further refinements by state-specific statute.” It follows that when considering compliance policies, it is necessary to ensure they satisfy regulatory requirements at both levels. Blizinsky made the important point that because this is a legislative area of “earnest development … we’re likely to see changes in state law and possibly even federal law that will influence how programs like All of Us and NHANES approach the return of genetic results.” To emphasize this point, Blizinsky shared a map highlighting states that already have laws that either directly or indirectly impact the return of genetic results and states where legislation is currently in development (Figure 4-2). Blizinsky suggested that this dynamic environment necessitates regularly tracking federal and state bills to ensure smooth program operations. However, she noted that this task is relatively manageable because states that propose new legislation often borrow language from other states, reducing the risk that it will interrupt programmatic processes.
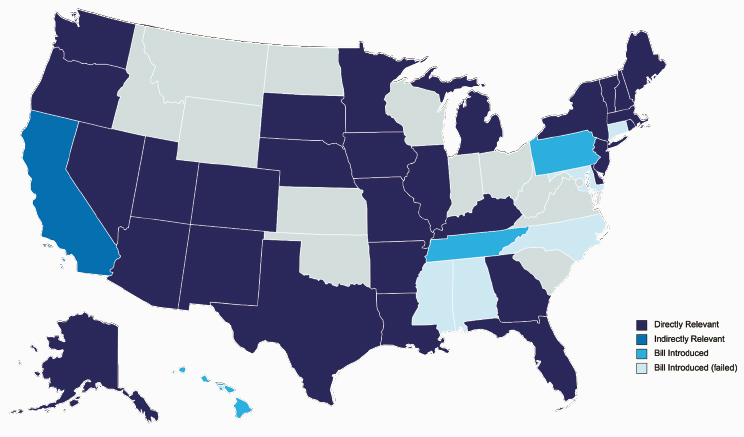
SOURCE: Workshop presentation by Katherine Blizinsky on December 2, 2022 (slide 6).
Federal and State Legislation Stipulating Informed Consent
Most states require, with few exceptions, “that genetic testing be performed with the knowledge at a minimum, or more frequently the explicit informed consent of the individual that’s being tested or their legally authorized representative.” These laws typically describe specific requirements in three predominant areas: (a) “they describe the format and the circumstances under which informed consent can be attained;” (b) they describe the “requirements for the content of a valid consent form;” and (c) they “define default limitations on certain facets of the testing process, including the authorized period of retention of the samples that are given under the auspices of testing, or research, and also the information that’s derived therefrom.”
Laws outlining “valid” consent forms tend to have certain commonalities. For example, these laws often describe the test being performed, the nature and purpose of the test, the effectiveness and limitations of the test, and the implications of the test. The implications of the test most often refer to its risks and benefits, which some states generally mention, whereas others (e.g., Michigan) narrow their scope to “medical risks and benefits.” In addition, states’ consent forms often require an explanation of “the meaning of the test results”; information about how individuals will be notified of their results; who will have access to the samples and the
information; under what circumstances individuals or their family members may be contacted with genetic results; and default limitations of the testing process, such as a limit on the retention of samples and the derived information that results from the research. However, Blizinsky noted that default limitations can almost always “be overridden if the individual undergoing the testing or their representative consents to unique terms.” Therefore, Blizinsky advised explicitly including any necessary deviations from states’ default limitations into the programmatic consent form is “critical to ensure that a program can continue to operate smoothly and within the boundaries that are defined.”
Blizinsky also mentioned a few less common state provisions that dictate valid consent forms. For example, a few states, notably Delaware and New York, “established one-off provisions, including provisions that require consent to include information about potential uses of a genetic test, the possible desire or need for validation of the test, and also the specific conditions or condition that may be indicated by the results that one would receive.” Furthermore, Massachusetts and New York have included stipulations “requiring informed consent to include a discussion of the availability and utility of genetic counseling.” Although neither state law requires genetic counseling to be provided alongside testing by the program, “they do require that entities provide testing, [and] at least point individuals in the right direction for such services.”
In closing her discussion on consent, Blizinsky emphasized that state laws about consent very often contain similar language. Still, she warned that some stipulate additional requirements and never to assume perfect agreement between similar state legislations. In addition, Blizinsky noted that most state consent laws are “well aligned with federal requirements for consent and general informed consent best practices.” As a result, she advised that thoughtfully crafting an initial consent process that meets all state and federal requirements will likely lead to a robust result.
Federal and State Legislation Stipulating Privacy Protection
According to Blizinsky, “Both federal and state laws guarantee participants some level of privacy protection for individuals undergoing genetic testing in the context of research.” These privacy protections can be broken down into three categories: laws requiring the withholding of genetic information, laws outlining the conditions for valid disclosures, and laws outlining the confidential treatment of genetic information.
Blizinsky discussed new stipulations enhancing privacy contained within the 21st Century Cures Act that strengthened privacy protections afforded to individuals participating in research. For example, updates to the certificate of confidentiality statute imposed a federal mandate to
withhold genetic information acquired during research, with some exceptions outlined below. Another statute within the 21st Century Cures Act,12 the Exception 3 Subpart B of the Freedom of Information Act, has language largely similar to the certificate of confidentiality statute.13 However, a significant difference is that it “allows for disclosure leveraged at the discretion of the secretary of HHS [United States Department of Health and Human Services].” Blizinsky emphasized that understanding the distinctions between the privacy protections afforded by each statute is essential for programs that collect genetic information. Specifically, she suggested this understanding will be needed to help guide how programs determine “appropriate data collection and guardianship models” and how to “communicate to participants around privacy protection from risks.”
Blizinsky next focused on when genetic disclosure is allowable, which occurs under both federal and state law. She explained that the aforementioned “certificate of confidentiality statute” details four scenarios for lawful disclosure. First, some state laws designate specific terms for legal disclosure of genetic test results and outline certain circumstances that trigger mandatory disclosure laws, such as where disclosures are necessary to prevent certain types of harm. Second, genetic disclosure is also federally lawful when it is “necessary for the medical treatment of the individual to whom the information, document, or biospecimen pertains and made with the consent of the individual.” Third, disclosures are allowable when they are made with the individual’s consent. There are sometimes laws that allow for disclosures in emergent situations, or other laws that either limit or expand permissibility of disclosures of private or identifiable information. The comprehensive set of laws, both federal and locality specific, must be examined before knowing what is or is not an allowable disclosure under the exceptions at U.S.C. 42 § 241, Part D, Subpart 1(c) (e.g., Protection of privacy of individuals who are research subjects).14
Finally, Blizinsky described participant privacy laws dictating confidentiality protections mainly in the context of state laws. But, again, she noted that different states invoke confidentiality to different degrees, and their laws contain various exceptions. Because of these differences, Blizinsky advised that the level of privacy protections will vary based on the locality in which the participant resides and that collecting valid informed consent requires that each participant understand the protections that apply specifically to them.
___________________
12 Public Law No. 114-255. For more information, see https://www.congress.gov/bill/114th-congress/house-bill/34
13 See https://www.justice.gov/archives/oip/foia-guide-2004-edition-exemption-3
14 For more information, see https://www.govinfo.gov/content/pkg/USCODE-2021-title42/pdf/USCODE-2021-title42-chap6A-subchapII-partA-sec241.pdf
Federal and State Legislation Stipulating Rights to Nondiscrimination
The last category of laws that Blizinsky discussed pertained to participants’ rights to nondiscrimination. The Genetic Information Nondiscrimination Act (GINA) outlines nondiscrimination practices related to employment and insurability. Blizinsky focused on GINA as it relates to insurability nondiscrimination, which prohibits the use of genetic information for purposes of health insurance underwriting. This legislation is a success in that genetic information cannot “be used to make medical insurance cost prohibitive for those who need it.” However, Blizinsky noted, these prohibitions only apply to health insurance and exclude other types of insurance (e.g., life insurance and long-term care insurance). As a result, some states have introduced and passed legislation that extends these (Figure 4-3). Blizinsky highlighted that these changes make insurability a volatile legal area, and “frequent changes in policies must be accounted for in your implementation plan.”
Blizinsky underlined several specific reasons that rapidly changing legislation around genetic insurability complicates human genetic testing research programs. For example, states’ protections against genetic insurance discrimination vary based on (a) who conducts the genetic test (e.g., clinical service or direct-to-consumer test service), (b) the type of insurance being provided (e.g., life, disability, long-term care), (c) whether the insurer can
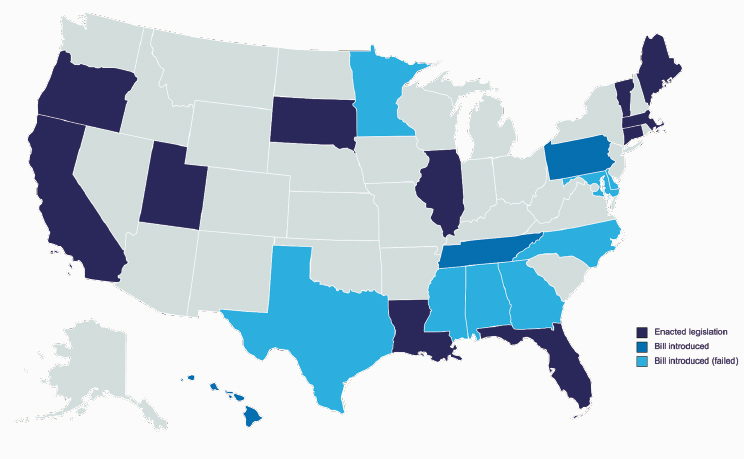
SOURCE: Workshop presentation by Katherine Blizinsky on December 2, 2022 (slide 24).
access and use genetic information (e.g., requesting test results, requiring testing, using test results for insurance underwriting, or using test results for coverage decision-making), and (d) the responsibilities incurred by the entity performing the test (e.g., withholding genetic information under all circumstances or only under certain conditions).
Blizinsky warned that guiding research participants through their state-level nondiscriminatory rights and helping them understand how genetic testing could impact their insurability is particularly challenging. Federal agencies are “ill-equipped to provide guidance on state law, and in most cases, we’re actually barred from doing it. But there also isn’t a particularly good state-level source of information to direct participants to either.” Blizinsky explained that “All of Us has prescribed pointing people to both the state attorneys general and state insurance commissioners, but participants have actually come back and complained to us about the lack of knowledge and assistance that they’ve received from these sources.” She noted that All of Us provides educational materials and FAQs to be “as helpful as possible,” but have found these methods to be insufficient. Despite the tremendous difficulty All of Us has experienced, Blizinsky advised that “sorting out and clearly communicating around individuals’ risks regarding genetic testing and insurability is really important.” Blizinsky shared that there is no clear solution to this problem at present, but she suggested that research programs such as All of Us and NHANES collaborate and “pool our intellectual resources toward a communal solution.”
Major Takeaways: Navigating Federal and State-Specific Laws Governing the Return of Genetic Results
Blizinsky ended her talk by summarizing a few takeaway messages regarding best practices while navigating state and federal laws that inform procedures for returning genetic results. First, she emphasized the existence of relevant laws at both the federal and state levels and the importance of understanding how they interact. Blizinsky also acknowledged the volatility of statutes about genetic information and testing. She suggested that keeping abreast of these legislations is critical for national programs that return genetic information. In most cases, state legislation uses similar language, which makes programmatic adaptation and implementation relatively simple. However, confidentiality protections and insurability nondiscrimination statutes are two notable exceptions where a universal application is not readily possible. State-level differences in confidentiality protections “affect what a research program can promise participants about how they and their genetic information are protected.” Blizinsky advised that this nuance “must be reflected in conversations about risk and the informed consent process and may impact program decisions about process and
implementation.” Finally, she proposed that establishing clear and reliable information regarding insurability risks due to genetic testing is perhaps the most difficult challenge facing national programs that return genetic information. Despite this, she also noted its critical importance. In conclusion, Blizinsky made a final appeal for programs like NHANES or All of Us to work together to help solve these evolving challenges.
QUESTIONS AND REFLECTIONS FOR SESSION 3
Holm led and moderated the discussion for this session. Leslie G. Biesecker, NHGRI at the National Institutes of Health (NIH), noted a difference between NHANES and the All of Us study, which he postulated could have bearing on how much the state laws come into play for each study. Blizinsky noted those sorts of topics should be covered with the relevant legal counsel. She underscored that there are laws that affect participants that a study will be required to comply with and disclose to the participant regardless of whether the participant says yes to participation. She stated that a study is responsible for disclosing those possible effects as part of the risk analysis for participants to make an informed decision about consent and encouraged the audience “not to ignore state law.”
First noting that they would be checking with the NHANES general counsel about a lot of the issues being raised, Alan Simon of the CDC’s National Center for Health Statistics proceeded to a new topic by asking if anyone could expand on the financial and operational structure of the All of Us study. For example, does All of Us provide grants to different institutions that then conduct the research program or is it run by a single contractor on a single contract who is then going out and collecting the data in all of the different areas? Blizinsky explained that the study uses a “mishmash” of grants, Other Transaction agreements, and contracts that are coordinated out of NIH by agency staff, which makes that study unique in terms of the compliance requirements.15
Jeffrey R. Botkin, University of Utah, next raised a question regarding how investigators are communicating genetic findings from non–CLIA certified laboratories. Solomon responded by first pointing out that there are different interpretations of CLIA, and any definitions or descriptions that are used “come through a lens of interpretation.” Solomon next offered that the common practice that he has observed is for a study to recontact participants with something general that does not give specificity but does explain that additional testing would be beneficial to confirm or rule out a possible finding. Solomon stated that “you’re not supposed to give
___________________
15 Other Transaction agreements are awards made under NIH’s Other Transaction Authority. For more information see https://grants.nih.gov/funding/other-transactions.htm
something that would even be a hint at guiding clinical suspicion” until a finding is CLIA-validated and added that no matter how careful the planning, a study can expect to encounter difficult situations. Biesecker next offered that NHGRI interprets the CMS regulations as prohibiting a study from returning a specific research result to a participant, but a study can engage with participants in a way that communicates a concern from a particular test (e.g., genetic sequencing) and that should be evaluated by undertaking particular steps. He noted this is the same as many nongenetic circumstances in that a study wants to provide enough information for a participant to make an informed decision that follows the proper clinical diagnostic process.
Adam H. Buchanan, Geisinger, next asked if Collins had any additional information about why the FDA determined that the All of Us Research Program’s genetic testing qualified as a significant risk device. Collins explained that the decision predated her time with the program, and from her understanding the concern was not related to a particular test but rather concerns about the overall process across the full life cycle, from user comprehension to processes and controls for returning results.
In response to a question raised by Holm about the role of the FDA in the larger landscape of genetic testing, Collins remarked that is still a “transient space.” She recommended early and frequent conversations with the IRB and the FDA. Solomon added that any results received from a commercial genetic testing lab are usually going to be from a CLIA- and possibly CAP-certified test. Solomon noted that with some exceptions there are different nuances for what is or is not FDA approved that vary depending on the locality. The exceptions include a “couple of assays” that are FDA approved, meaning some labs use a device that has been approved by the FDA for a certain type of testing such as a microarray; Solomon provided somatic cancer genomics as an additional specific example. Solomon echoed Collins by noting that the FDA’s role in oversight of genetic and genomics tests is evolving and added, “but right now the vast majority is not what you would call ‘an FDA-approved test’ that you get when you send a test to GeneDx or Invitae, Ambry, Myriad.”
As a point of clarification during the Session 3 discussion, Simon offered that while it is not true that all of the labs NHANES uses are CLIA certified, it is true that any results that get returned to participants were processed in a CLIA-certified lab.
Biesecker commented that “FDA chooses to not require FDA certification for all tests, for example LDTs [laboratory-developed tests]. A good bit of what FDA approval of a test accomplishes is the ability of a company to advertise a test and their claims of accuracy, et cetera.” To this, Solomon explained that one of the reasons that companies may seek FDA approval in the clinical context is because the testing is more likely to receive insurance
coverage and be reimbursed. Matthew Lebo, Mass General Brigham, highlighted two paths for a research study: identify pathogenic variants and then reach out to consent participants to see if they want results, versus running a clinical assay where the return to the patient is part of the assay. Lebo noted that if passed as currently written, the VALID Act16 would give the FDA more oversight, potentially giving more avenues for its role in regulating LDTs. Solomon added that, in the future, the FDA may also decide that they are going to have involvement over testing that was not initially under FDA jurisdiction and pointed to a rapid genome sequencing research project as an example. Holm offered the BabySeq as another example, explaining that the initial project never considered the FDA’s potential involvement but later in 2015, the FDA changed course and said BabySeq did need to be regulated. The FDA ended up determining theirs was a nonsignificant risk device “as long as we didn’t return an adult-onset condition to a baby.”
First noting a distinction between covered and noncovered entities, Botkin raised the final question by asking speakers to weigh in on whether HIPAA is relevant to this discussion. Blizinsky thought that it could be relevant in places depending on how the tests are run, the eventual downstream users of the test, and whether the testing results become part of a participant’s medical record. “But largely if this is a research program conducted by a noncovered entity or a hybrid entity that’s operating in the research arm, the not-covered arm of that entity, HIPAA largely isn’t going to come into play,” said Blizinsky. She explained that All of Us does not currently offer participants access to their raw dataset because they are research results and would require clinical validation before being shared. Rather, All of Us participants are offered a standardized report and are able to send those reports to their physicians and their providers. Simon clarified that HIPAA does not apply to NHANES. Botkin noted that from his view this is somewhat separate from whether NHANES might grant people HIPAA-like access to records if that is considered an important value.
GENERAL END-OF-DAY DISCUSSIONS FOR SESSIONS 1, 2, AND 3
Botkin led and moderated the general discussion for Sessions 1, 2, and 3, which was held at the end of Day 1. Botkin began by returning to some questions and comments that were initially raised in earlier discussions but not yet addressed because of time constraints. The first was a followup question from a webcast viewer who commented during Session 1 that
___________________
16 Verifying Accurate Leading-edge IVCT Development (VALID) Act of 2021, H.R. 4128, 117 Cong. (2019). https://www.congress.gov/bill/117th-congress/house-bill/4128?s=1&r=1&q=%7B%22search%22%3A%5B%22valid+act%22%5D%7D
it would be powerful to include a self-assessment similar to that used by the Behavioral Risk Factor Surveillance System (BRFSS) for adverse childhood experiences, to demonstrate the associative power of developmental experiences to negative outcomes at a population level.17 “The idea is that assessment data for developmental adversity seems likely to shed additional light on population outcomes, especially when combined with polymorphism data to identify population phenotypes. Combining assessment data for developmental adversity could eventually be combined with epigenetic status, especially of key master genes, to leverage insights into individual outcomes,” explained the workshop participant. Botkin offered his perspective that this sort of assessment could potentially prove to be valuable in association with certain types of genetic assessment, before asking others to respond. Simon explained that NHANES does not currently collect information about adverse childhood experiences, noting that is something that has been considered as a possibility before adding his personal opinion that “there are some plusses and minuses to that specific battery.”
The next comment came from another webcast viewer in Session 2 after Holm raised a question for Sapp regarding the diversity and return of results in diverse communities. “Regarding the Kaiser data that showed respondents are willing to participate in genetic research ranged from 22 percent with no results returned to 87 percent if health-related genetic results were returned, it was found that when results were returned, disparities in the willingness to participate disappeared for NH [non-Hispanic] Blacks and Hispanics,” commented the webcast participant. After noting the “impressive increase in the number of people interested in participating,” Botkin shared that preworkshop discussions had included “whether adding some genetic component, even if it didn’t have a clear, strong, major scientific justification, might it be part of a package of feedback to participants that would encourage their participation.” Botkin noted that while this is probably “not a good reason to include a test in a research project,” it could have that sort of secondary effect.
Dana C. Crawford, Case Western Reserve University, noted that the NHANES biospecimen consent process had yielded a decrease in participation among those who otherwise participated in the broader study. Crawford recalled that fewer non-Hispanic Black participants consented for the biorepository part, and added that attitudes may have changed in the interceding 10 years with the rise of direct-to-consumer genetic testing and as genetics and genomics have become more prominent in everyday conversations and awareness. She suggested that NHANES or other groups may want to reexamine that issue.
___________________
17 For more information on the BRFSS, see https://www.cdc.gov/brfss/index.html
Biesecker commented that the structure of NHANES “with its annual accrual of 5,000 individuals and discontinuation of engagement at 16 weeks is a major impediment to its utility as a genetic and genomic research resource,” as well as an impediment to the obligation of returning secondary individual findings. Crawford responded by separating the problem into two issues. She noted that the issue of statistical power is dependent on the research question as well as the frequency of the genetic variant and added, “It takes a long time to accrue the outcomes of interest if it’s a chronic disease.” Crawford saw this as separate from the issue of whether or not NHANES data lend themselves to precision medicine research, which would require repeated measures and long-term follow-up with participants. “NHANES is not designed for that, so it certainly is very limited in those kinds of analyses,” offered Crawford.
Botkin noted that one key advantage of NHANES is the diversity of the recruited population, which stands in contrast to much of the other genomics datasets. He then raised whether there was a way to leverage that advantage as a topic for discussion. He offered an example to help ground the discussion, “Are there polygenic risk assessments for common conditions [such as type 2 diabetes] that you could compare across racial or ethnic groups that would be meaningful for the number of people who are being recruited into this study?… Could that partially fill some of the gap that we’ve heard about in terms of the polygenic risk data and the relatively low amount of information we have from non-European populations?” Acknowledging that she is not an expert on creating polygenic risk scores, Holm offered her perspective that the issue is needing a lot of people of a particular ethnicity, race, or ancestry to generate polygenic risk scores.
Lebo chimed in that “the numbers is probably the biggest thing.” He noted that All of Us, eMERGE, Polygenic Risk Methods in Diverse Populations,18 and other consortiums are trying to increase the number of individuals who are historically under-represented in genomics, specifically polygenic risk scores and the like. He offered that from an implementation standpoint it would be interesting to see if a more representative study like NHANES could perhaps produce more interesting phenotypic data compared with the other studies that are “aiming to do it in probably larger numbers in URM [under-represented minorities].”
Crawford agreed with Holm and Lebo, stating that NHANES is not well suited to developing polygenic risk scores because of the sample size. She reminded the audience that All of Us is purposefully not representative because it has intentionally oversampled by race/ethnicity, social constructs, and “other kinds of groups that are traditionally not well represented in research.” She went on to explain that NHANES may offer an opportunity
___________________
18 For more information, see https://primedconsortium.org/
to evaluate polygenic risk scores from an unbiased population-based sample and setting that was not drawn from a clinical setting, purposefully oversampled (e.g., All of Us), or subject to major selection biases (e.g., Million Veteran program).
A webcast viewer asked discussants to comment on whether NHANES could be a point of referral to other long-term studies that specifically want to study genetic variation in the long term. Crawford offered that she accesses a lot of biobank data and has found those to be biased samples and uses NHANES to better understand those biases. “I do look at the reference ranges if you will of what the distribution would look like in that unbiased representative sample versus what I have in the biobank,” she explained. Botkin offered that NHANES could be a way to funnel actual participants into other studies, such as All of Us, that could follow them longer term. “You’ve already got people who have agreed to participate in research, maybe they’d be willing to have someone contact them periodically for follow-up data,” he said.
Botkin shifted the conversation to the topic of biorepositories, noting that many rely on the regulatory circumstance that research using deidentified data is no longer considered human subjects research and investigators can pretty much do what they want with those samples. “A lot of people of course are concerned about the ethics of that practice, but it is an effective way empirically, I think, to essentially eliminate risk to the people who have contributed samples in the past,” stated Botkin. He noted that NHANES seems to have taken a different approach by restricting investigators’ research with the biospecimens to nonclinical actionable information as opposed to deidentifying before asking whether NHANES had considered loosening the research agenda limitations by deidentifying the dataset.
Simon explained that the NHANES data are not deidentified in the biorepository internally because in order to make the samples really useful they need to be linked to the rest of the data. “So then the question of identifiability becomes a question of are we releasing something to you that in combination with the data makes it potentially identifiable,” explained Simon before noting that was a question that would need more thought. Holms added that her understanding is that All of Us allows people access to the phenotypic and genotypic data along with electronic medical record and other data that have been deidentified for research. Botkin followed up by noting that, in that context, if an investigator finds a clinically actionable finding there is no mechanism for returning the result because the link between the dataset and individual’s data has been broken. Lebo explained that the All of Us researcher workbench uses multitiered access, where investigators must sign agreements and get trained in order to access the detailed data in the workbench but cannot ever download the data. Crawford
noted that NHANES is set up in much the same way and reminded the audience that the biospecimen program is separate from NHANES 2025 and beyond, “which is where the potential genetic testing and return of results would happen.” She highlighted that a key difference is that linked NHANES data can only be accessed in person through the Research Data Center which is “under resourced” and “not set up for genetic analyses.” Crawford clarified that the NHANES data do not include personal identifiers, noting that with genetic data there is always a risk, however small, of loss of privacy.
This page intentionally left blank.





















