The Great Brain Debate: Nature or Nurture? (2004)
Chapter: Part I—The Developing Brain 1 Building a Brain
1
BUILDING A BRAIN
Understanding how the brain forms is one of biology’s greatest challenges. From a relatively few undifferentiated cells in the young embryo, all of the neurons and glial (supporting) cells arise. The adult human brain contains about 100 billion neurons (a conservative estimate) and perhaps 10 times as many glial cells. Because virtually all neurons and most glial cells form before we are born, an embryo would generate approximately 250,000 cells per minute in the womb if brain cell generation were constant over the nine-month gestation period. However, most neurons are generated in the first four months of gestation, so the number of cells generated per minute during this early period is much higher. Furthermore, many brain regions initially overproduce neurons and the surplus dies during the maturation process. Thus, at various times during the gestation period more than 500,000 cells might be generated per minute!
Our brain begins to form about three weeks after conception. A group of about 125,000 cells forms a distinctive flat sheet along
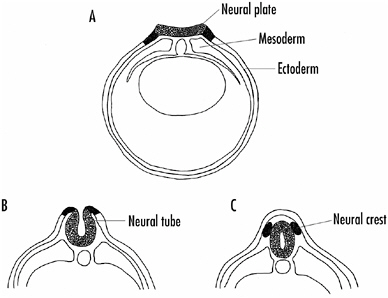
FIGURE 1-1 Formation of the neural plate, neural tube, and neural crest in young embryos.
A: The neural plate cells derive from ectodermal cells on the dorsal surface of the embryo. Signals coming from underlying mesodermal cells induce the dorsal ectodermal cells to become neural plate cells.
B: The neural plate invaginates to become the neural tube, and cells that initially lie laterally along the neural plate form the neural crest.
C: The neural tube becomes the central nervous system (brain and spinal cord), whereas the neural crest forms much of the rest of the nervous system (peripheral nervous system).
the dorsal or back side of the embryo. Known as the neural plate, all the neurons and glial cells derive from this early structure (Figure 1-1A).
Between the third and fourth weeks of development, the neural plate curves inward and creates a groove that slowly closes into a long tube, the neural tube, as shown in Figure 1-1B. The entire central nervous system—that is, the brain and spinal cord—develops from the neural tube. The anterior part of the neural tube becomes the brain proper, the posterior part the spinal cord. By the 40th day of development, three swellings become apparent along the anterior part of the neural tube as shown in Figure 1-2. These eventually form the three major subdivisions of the brain—the forebrain, midbrain, and hindbrain.
During formation of the neural tube, some cells on either side separate to form structures known as the neural crests as shown in Figure 1-1C. Much of the peripheral nervous system—those nerve and glial cells that lie outside the brain and spinal cord—derive from the neural crest cells.
Figure 1-2 shows the development of the human brain from the neural tube. The detailed drawings at the left are enlarged relative to the drawings on the right. By 60 days after conception, the forebrain, midbrain, and hindbrain regions can be readily distinguished. Infolding or wrinkling of the brain’s surface—to increase the cortical area—begins at about seven months.
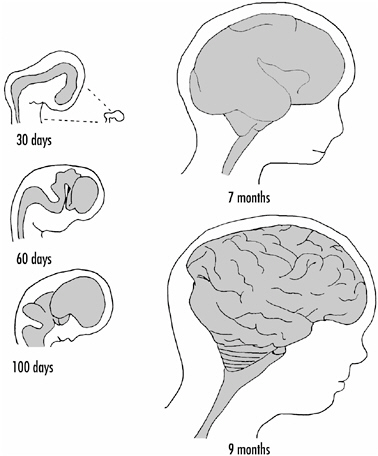
FIGURE 1-2 Development of the human brain from the neural tube. The drawings on the left are enlarged relative to those on the right. The tiny drawing at the top indicates the actual size of the brain at 30 days relative to size of the brain at 7 and 9 months.
At nine months of gestation, the brain overall looks quite adult, but it has far to go. The average newborn human brain weighs less than 400 grams, whereas the typical human adult brain weighs about 1,400 grams. Figure 1-3A shows the brain viewed from the dorsal (top) side in a newborn and at six years of age.
Much of the weight increase occurs during the first three years after birth, but the brain does not reach its maximum weight until about 20 years of age. Thereafter, brain weight declines slowly but steadily. Figure 1-3B shows graphically the average weight of the human male brain (based on measurements made on more than 2,000 normal brains) from birth to age 85. Female brains, on average, are slightly smaller at all ages, probably because women tend to be somewhat smaller than men.
I noted above that virtually all neurons are generated by about birth or certainly by six months of age. Thus, what underlies the remarkable growth of the brain in the first three to five years of life? A number of things are going on, including an increase in the number of glial and other supporting cells, growth of blood vessels, and, importantly, the ensheathing of the long axonal processes of the neurons by myelin. Myelin is formed by glial cells wrapping their cell membrane around axons, creating a highly enriched lipid layer that covers the axons. Myelin insulates the axons, making them more efficient in transmitting the electrical signals that travel their length.
However, the most important factor contributing to brain size increase in the early years is the growth and elaboration of the neurons themselves. Not only do their cell bodies grow in size, they also extend more dendritic branches during brain maturation. The dendrites grow larger and go longer distances as shown in Figure 1-4.
More than 80 percent of total dendritic growth probably occurs after birth. It is on the dendrites of a neuron that most synaptic contacts are made; thus, the elaboration of the dendritic processes of neurons that occurs during the brain growth of the
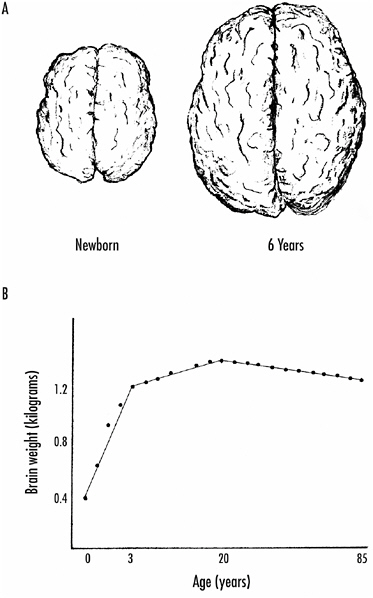
FIGURE 1-3 A: Growth of the human brain from birth (left) to age 6 (right). These are dorsal views showing the cortical surfaces of the brain.
B: Brain weight as a function of age. A rapid increase in brain weight occurs in the first three years. The rate of increase then slows, but the brain does not reach its maximum weight until about 20 years. Thereafter, there is a slow and constant loss of brain weight.
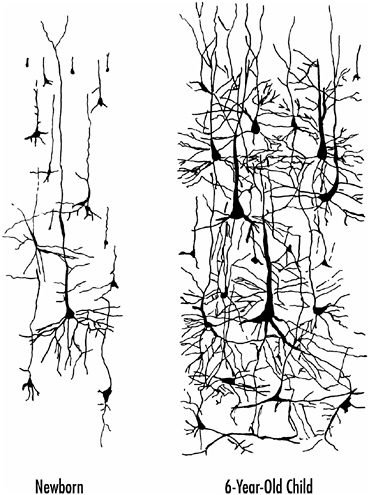
FIGURE 1-4 The elaboration of neurons during brain maturation. Not only do the cell bodies of the neurons increase in size, but there is an enormous increase in the number, extent, and complexity of their branches.
early years implies a substantial increase in the synaptic circuitry of the brain.
There is no question that there is an enormous increase in total numbers of brain synapses, not only prenatally but also postnatally up to at least age 2. But the situation is much more complex than just adding synapses. As we shall see, there is a substantial rearrangement and pruning of synapses during brain development and growth, so not only are many synapses added,
but many others are lost. Indeed, if one simply looks at the total number of synapses, the peak is between six and eight months postnatally, and then total numbers decline. Experience clearly influences the rewiring of brain synapses during brain maturation, but this rewiring is not just limited to the young brain. All of our lives our brains are being changed by our experiences, and these changes are reflected in the synaptic circuitry of the brain. (See Part II, The Adult Brain, for more information on these changes.) It is certain, however, that the young brain is considerably more plastic than is the adult brain, a topic we shall return to in the next chapter.
It is important also to emphasize that not all parts of the nervous system mature simultaneously. Maturation occurs in a roughly tail-to-head gradient. For example, the spinal cord and brain stem (which controls vital body functions such as respiration, heart rate, and gastrointestinal function) are essentially fully organized by birth, and myelination of the axons in these regions is quite complete. Shortly after birth, myelination of axons in the cerebellum (concerned with motor coordination) and midbrain begins, and thereafter—by the end of the first year or early in the second year—it begins in various parts of the forebrain, including the cerebral cortex.
The last brain structure to mature is the cerebral cortex, the seat of higher mental functions, including perception, memory, judgment, and reasoning, but here also maturation of all areas of the cortex does not occur simultaneously. Those cortical areas concerned with sensory processing mature earliest, followed by motor areas. But those areas concerned with the more sophisticated aspects of brain function—the so-called higher-order association areas of the brain, concerned with planning, intentionality, and other aspects of one’s personality—are still myelinating axons and rearranging synapses up until the age of 18 or so! This includes much of the frontal lobes as well as parts of the temporal and parietal lobes of the cortex.
Recent imaging studies have extended our understanding of brain and cortical maturation. Studies have been carried out on
children between the ages of five days and 15 years, using positron emission tomography (PET) scanning to determine glucose utilization by various parts of the brain. Glucose is the primary energy source for neurons (and other cells); the more active a neuron is, the more glucose it uses. It was observed that glucose utilization in newborns is largely limited to the brain stem, parts of the cerebellum, and certain subcortical structures. Very little glucose utilization was observed in the cortex itself, indicating relatively low neuronal activity there. By two or three months of age, glucose utilization increases significantly in some cortical areas, especially in the occipital cortex, which is involved in visual processing and perception. Not until six to eight months is significant activity observed in the frontal lobes, and again some parts of the frontal lobes show more activity than others.
Glucose utilization by the brain increases through early childhood and, interestingly, it peaks between four and seven years of age (depending on brain region), at which point glucose utilization is about twice the level of that in the adult brain. Glucose utilization by the brain then slowly subsides to adult levels through childhood and adolescence. The peaking of glucose utilization by the brain at four to seven years of age perhaps relates to the enormous synaptic plasticity of the brain at these early ages. Many new synapses are being formed, others eliminated, and synaptic circuits refined; but now I am getting ahead of the story. We shall come back to these issues later.
Mechanisms Underlying Brain Development
Let us return to the earliest stages of nervous system development and consider what is known about the underlying biological mechanisms. All neural tissue derives from neural plate cells, as shown in Figure 1-1, but what causes these cells on the dorsal side of the very young embryo to become neural plate cells? Two layers of cells initially make up the very young embryo: ectodermal cells, which cover the surface of the embryo and will eventually form mainly skin, and endodermal cells, which line the embryo
and will form the digestive system and internal organs. A third layer of cells—the mesoderm, which will become muscle, bone, and connective tissue—develops next, and during its formation it migrates between the ectodermal and endodermal layers, initially on the dorsal side of the embryo. It turns out that the migrating mesoderm provides the signal for ectodermal cells along the dorsal surface of the embryo to become neural plate cells.
This aspect of brain development was first shown by two German biologists, Hans Spemann and his student, Hilde Mangold, who, in the 1920s, took mesodermal cells from the dorsal part of the salamander embryo and transplanted them to other parts of the embryo. They found that the transplanted mesodermal cells were capable of inducing any ectodermal cells to become neural plate cells, not just those on the dorsal part of the embryo. Thus, if they transplanted mesodermal cells from one embryo into another, they could induce the formation of two neural plates and, in some cases, the development of two nervous systems in the animal. Conversely, if they prevented mesodermal cells from migrating underneath the ectoderm in the early stages of embryonic development, no neural plate formed and the embryo lacked a nervous system.
How does the mesoderm cause ectodermal cells to become neural plate cells? It has long been suspected that a chemical signal from the mesoderm mediates this induction. For example, if pieces of embryonic ectoderm are cultured in the presence of mesoderm, they will become neural plate cells, but in the absence of mesodermal cells they will not. By placing porous filters between ectodermal and mesodermal layers, it is possible to define the size of the signal molecules, and these experiments suggest that the inducers are small proteins. If the porous filters are too small to allow small proteins to pass through, the ectodermal cells fail to become neural plate cells.
Three small proteins have now been shown to be neural plate inducers in amphibians: noggin, chordin, and follistatin. All of these proteins are thought to be secreted by mesodermal cells. Curiously, they act by binding to another secreted protein, called
BMP, and preventing it from interacting with ectodermal cells which would cause the cells to develop into skin. Thus, it is not the inducers acting directly on the ectodermal cells that causes them to become neural plate cells, but the lack of stimulation by BMP that results in their becoming neural plate cells. Why it works this way is not clear, but this first stage of brain development illustrates a principle seen again and again in brain growth, maturation, and function; namely, the key role of chemical signaling between cells.
Proliferation of Neural Cells
I noted earlier that as many as half a million cells might be generated per minute, on average, for the first four months of gestation in humans. How and when does this happen? Proliferation begins upon closure of the neural tube and initially takes place almost exclusively around its inner surface—an area called the germinal zone. Initially, the neural tube is just one or a few cell layers thick, but it rapidly thickens, enlarging from the inside out. Dividing cells undergo characteristic movements, shown schematically in Figure 1-5.
A cell getting ready to divide is bipolar in shape with a branch extending to the inner surface of the neural tube and another to
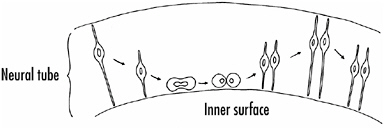
FIGURE 1-5 The generation of new cells along the inner surface (germinal zone) of the neural tube. The nucleus of a dividing cell moves up and down during the proliferation process. DNA synthesis occurs when the nucleus is away from the inner surface; cell division occurs along the inner surface.
or toward the outer surface of the tube. The nucleus of a dividing cell is situated away from the inner surface of the tube while it synthesizes DNA, but then it migrates to the inner surface of the neural tube. The cell withdraws its branches, rounds up, and divides. After cell division, the two daughter cells extend new branches, the nuclei migrate deeper into the tube, and the process repeats.
The genesis of cells destined to become neurons begins as soon as the neural tube forms (at three weeks of development) and reaches a peak in the seventh week, but then is largely completed by 18 weeks. Some neurons are generated later in the fetal stage of life, and some even in the first few postnatal months, but most neurons are generated by just four months of gestation. Glial cells, on the other hand, are generated continuously throughout gestation and even throughout life, though at a low rate.
The proliferation of the neural progenitor cells is under the control both of extrinsic growth factors—specialized proteins that act directly on cells to promote their division—and intrinsic factors—intracellular mechanisms that limit cell division. Cells exit the cell cycle—stop dividing—when the negative signals exceed the positive ones, but what the various signals are and how they are controlled are still poorly understood.
One idea that has been proposed to explain how cells leave the cell cycle is that there is a mechanism leading to asymmetric cell division at some point during proliferation. As long as cells divide symmetrically, they continue to generate more precursor cells. On the other hand, in asymmetrical cell division, one of the cells has less of a particular molecule than the other, causing the cell to leave the cell cycle. Is there any evidence for such a molecule? Recent experiments in mice, in which a protein called β-catenin is altered genetically so that it is more resistant to degradation, resulted in precursor cells excessively reentering the cell cycle rather than leaving it during early brain development. These mice grew grossly enlarged brains. Particularly striking in these animals is the cortex, which developed deep folds in the modified animals. In normal mice, on the other hand, the cortex
is quite smooth, reflecting a much smaller structure. Unchecked neural cell proliferation can also lead to cancers—neuroblastomas—and this happens in the young developing brain. (See Chapter 5 for more discussion of cell proliferation.)
When cells exit the cell cycle, they typically move away from the germinal zone and form a distinct layer distally—called the intermediate zone. Cells in the intermediate zone are mainly young neurons that will never divide again. Where they will reside in the brain and even what kind of neuron they are likely to become are now essentially established. Some cells that migrate from the germinal zone retain the ability to divide, and a number of these cells form important brain structures, including the basal ganglia—subcortical areas that are involved in the initiation of movement. Certain cerebellar cells also proliferate after migration away from the germinal zone, and neural crest cells often divide after they have reached their final destination.
In cold-blooded vertebrates, such as frogs or fish, proliferative cells remain in the adult brain and continue to divide and generate new neurons. A particularly clear example is the retina of fish, which continuously adds neurons during the animal’s life. In other words, the retina continues to grow as the animal grows over its life span. But most neuroscientists believe this is the exception; in most species, especially mammals, new neurons are not often generated in the adult brain.
Recent research has identified germinal cells in at least two regions of the mammalian brain, one is the hippocampus, a region of the brain concerned with the long-term storage of memories. There is some evidence that these germinal cells in the hippocampus can generate new neurons, but whether these new neurons become incorporated in the neural circuitry of the hippocampus is as yet uncertain. The nature and significance of other proliferating cells in the adult mammalian brain are also unclear and the subject of much controversy at present. Some investigators believe that many of these proliferating cells are glial cells. We shall return to this important issue in Chapter 5.
Migration of Young Neurons
From the intermediate zone, the young neurons must migrate, often considerable distances, to take up their final position. How this happens varies from region to region. In some parts of the brain, such as the retina and spinal cord, cells migrate in response to chemical clues, both positive and negative, present in the area. In other parts of the brain, such as the cortex and cerebellum, specialized glial cells, called radial glial cells, provide a scaffolding along which the neurons migrate. The cell bodies of these glial cells reside in the germinal zone, but they extend a branch to the surface of the brain as shown in Figure 1-6.
Electron microscopy has shown in the intact brain that migrating neurons are entwined around radial glial cell branches, and in tissue culture, neurons have been observed migrating along radial glial cell branches. In a mouse mutant that has a cerebellar defect in which the radial glial cells degenerate early, many of the cerebellar neurons do not end up in their proper positions and the animals show severe movement deficits. In normal mice (and other animals) the radial glial cells remain until neuronal migration is complete and then they disappear.
The sequence in which the cells migrate in the developing brain varies among different brain regions. In the cerebral cortex, for example, the first neurons to complete cell division and to migrate form the deepest layer of the cortex (so-called layer 6). Cells that proliferate later form the more superficial layers. In other words, the cortex grows from inside out. In the retina the opposite happens. The first cells generated (the ganglion cells) migrate across the retina where they take up residence, and cells generated later form layers of the retina closer to the germinal zone.
Differentiation of Neurons
Once the young neurons arrive at their final destination, they are first specified. That is, the kind of neuron they will become is
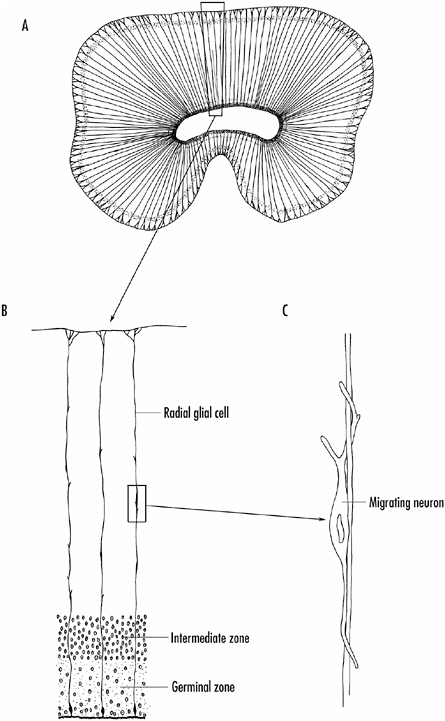
FIGURE 1-6 A: Radial glial cells in the developing cortex of the brain.
B: The radial glial cells extend from the germinal zone on the inner surface of the neural tube to its outer surface.
C: Neurons from the intermediate zone migrate along the processes of the radial glial cells to find their proper position in the brain.
determined. They next undergo differentiation: they extend branches characteristic of the type of neuron they are and begin to make synaptic contacts.
Almost needless to say, neurons are very complex cells of various sizes and extending numerous branches that often extend considerable distances as seen in Figure 1-4. Furthermore, each neuronal cell type usually has a unique branching pattern. What mechanisms underlie the specification and differentiation of neurons? Again, both intrinsic and extrinsic factors are at play. Specific types of neurons tend to be generated at specific times during development, and often all neurons of that type differentiate more or less simultaneously. In the retina, for example, the ganglion cells are specified and differentiate first, followed by cone photoreceptors, amacrine, and horizontal cells. The rod photoreceptors and bipolar cells differentiate last.
What triggers the specification and differentiation of a precursor neuron into a particular cell type? The local environment—the chemical signals the cells encounter—is clearly critical and this depends on the cells’ position in the tissue. In other words, signals from nearby cells determine a cell’s fate. Thus, extrinsic signals are key in the process. However, over time, the options for a cell to become a particular type of neuron are limited. That is, a precursor neuron is receptive to a specific inducing signal for only a particular window of time. Thus, intrinsic mechanisms are also at play in neuronal specification and differentiation and are also clearly important. To summarize, to become a particular type of neuron, a precursor cell must be in the right place at the right time during development.
In some species, especially invertebrates, in which there is little cell migration during development, intrinsic mechanisms play the major role in neuronal specification and differentiation. That is, the type of neuron a precursor cell becomes is determined by inherited developmental directives—the cell’s lineage determines its fate. Thus, if a precursor cell is destroyed during development, the organism develops without the cells that it was to become. Other precursor cells cannot substitute for the deleted cell.
However, induction by position-dependent signals is clearly the major mechanism by which precursor neurons become specified and differentiate, especially in the vertebrate brain. Studies on the developing eye of the fruit fly, Drosophila melanogaster, have provided important insights into how this happens and the molecular mechanisms involved. The fruit fly’s eye is a mosaic eye, consisting of individual photoreceptor units, called ommatidia. Each ommatidium has eight photoreceptor cells that can be individually identified and that are precisely arranged in the structure. During development, R8 (R stands for “retinular cells,” the technical name for these photoreceptor cells) differentiates first. This is followed by R2 and R6, which differentiate together, then R3 and R4, followed by R1 and R5. The last cell to differentiate is R7, which contains a molecule that senses wavelengths of light in the ultraviolet region of the spectrum. The other photoreceptor cells respond to light in the visible spectral region (visible to us, that is).
The strict sequence of development of an ommatidium in the fruit fly eye suggests that earlier cells are responsible for the differentiation of the later cells with R8 taking the lead role. This was learned from experiments in which the early developmental sequence was disturbed. In such cases, the entire ommatidium did not form properly. Even more revealing were experiments on a mutated fly in which the R7 photoreceptor did not form at all—discovered because these flies do not respond to ultraviolet light. This mutant, called “sevenless,” has told us much about the nature of the signaling mechanism and how the cell responds to such extrinsic signals.
The mutated gene in the sevenless fly codes for a protein that extends across the membrane of the cell as shown in Figure 1-7. On the outside of the cell, the protein serves as the receptor for the signal that tells the cell to become an R7 cell. On the inside of the cell, it acts as an enzyme when the receptor part of the molecule is activated. The enzyme part of the protein is a kinase, an enzyme that adds phosphate groups to proteins. Adding phosphate groups typically changes proteins’ properties; activating or
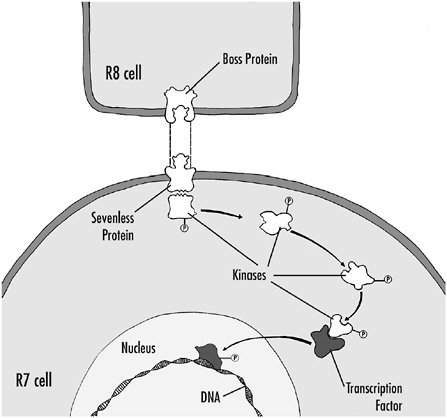
FIGURE 1-7 The interaction of the R8 cell with a precursor cell, and the sequence of events that leads to the differentiation of the precursor cell into an R7 receptor cell. When the boss protein on the R8 cell binds to the sevenless protein on the precursor cell, the sevenless protein acts as a kinase, adding phosphate groups to intracellular proteins. The intracellular proteins might themselves act as kinases, adding phosphate groups to other proteins and activating them. Eventually, proteins, called transcription factors, are phosphorylated. These proteins migrate into the nucleus, bind directly to DNA, and regulate gene expression, thus leading to the differentiation of the cell.
inactivating them if, for example, they are enzymes. Once the receptor part of the sevenless protein activates the intracellular kinase activity of the molecule, a series of biochemical events is initiated that ultimately leads to differentiation of the cell. If the cascade is not initiated—that is, the receptor protein is defective as in the sevenless mutant—the cell follows a default pathway and becomes a nonneural cell. In other words, it differentiates by
way of intrinsic directives, but not into an R7 cell or even into a photoreceptor.
What is known about the signal that activates the sevenless protein? A second mutation in fruit flies in which the developing ommatidium also fails to form an R7 cell has provided an answer. This mutant is called “bride of sevenless” or “boss” and it affects the R8 cell. The defective gene codes for a membrane protein that is found in R8 cells in normal flies. We think that part of this protein extends from the surface of R8 and activates the sevenless receptor on the R7 precursor cells; this membrane protein is the signal (see Figure 1-7). Thus, direct contact between the cells is required for activation of the sevenless protein.
Scientists have also made progress in understanding what happens in the R7 precursor cell after the sevenless receptor protein has been activated. A number of downstream proteins have been identified and many are kinases themselves. Thus, activation of the sevenless kinase leads to activation of a number of other kinase enzymes. Targets of at least some of these kinases turn out to be transcription factors—proteins that migrate into the nucleus of the cell and turn on or off the expression of various genes by interacting directly with the regulatory regions of genes, the so-called promoter regions. The idea is this: when the sevenless receptor-kinase protein is activated by the R8 cell, a number of other kinases and transcription factors are eventually activated that lead to the expression of the appropriate genes needed to turn a precursor cell into a R7 photoreceptor cell.
Scientists believe that in other situations diffusible substances released by nearby cells control the differentiation of precursor cells, but that the same principles as described above apply. Some of these signaling molecules have been identified as small proteins. Some of the so-called growth factors described earlier play this role. These proteins all activate membrane receptors linked to a cascade of intracellular kinases that ultimately turn on or off specific genes. Thus, the general scheme shown in Figure 1-7 probably holds for the differentiation of neurons and glial cells throughout the brain.
How Do Axons Find Their Way?
Once neurons are specified and begin to differentiate, they extend both dendritic and axonal branches. This leads to the formation of synapses between neurons and, ultimately, to the wiring of the brain. A critical question is how axons find their way to their targets and how they know which cells to synapse upon. Often axons must travel considerable distances to reach their target neurons.
Again, chemical signaling is implicated as playing a critical role in cell-cell recognition. The notion is that as neurons differentiate, they become chemically specified; they make specific membrane proteins that extend from their surface and enable innervating axons to recognize them. Experiments that support this chemoaffinity hypothesis date back a century, but it was work carried out in the early 1940s by Roger Sperry at the University of Chicago that established the idea. Sperry studied the projection of retinal ganglion cells to a brain region called the tectum in animals such as fish or frogs. In these cold-blooded animals the tectum is the major target for the ganglion cell axons. The projections from retina to tectum are orderly; ganglion cell axons from one part of the retina innervate a specific region of the tectum. The projections are called topographic and are quite invariant from one animal to another. In other words, a retinal map is impressed on the tectum. The right retina projects to the left tectum and vice versa, and the tectal map is inverted relative to the retinal map, as shown in Figure 1-8.
In cold-blooded vertebrates such as fish and frogs, central nervous system (CNS) axons regenerate after they are severed and remake synaptic contacts (this, unfortunately, does not happen in mammals; see Chapter 5). Sperry took advantage of the regeneration of CNS axons in fish and frogs to show first that, if you sever the optic nerve in a newt (a frog-like animal), the axons regenerate and reform synaptic connections in the tectum. Furthermore, their vision is restored. How specific are these new connections? To test this idea, Sperry severed the optic nerves in frogs, but then
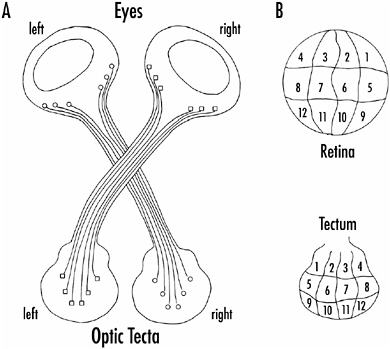
FIGURE 1-8 A: Ganglion cell axons from one eye of a fish or frog project in an orderly fashion to the tectum on the opposite side of the brain.
B: Specific regions of the retina (indicated by numbers) project to specific regions of the tectum, forming what is called a topographic map. The tectal map is inverted relative to the retinal map.
rotated the eyes 180° before reattaching them in the socket. He found that after the optic nerves had regenerated, the animals could see once again, but their visual world was upside down and inverted from right to left! When the animals were feeding, they consistently misdirected their attempts to capture their prey by 180°. If prey they wished to capture was up and to the right, they moved down and to the left, and vice versa.
These experiments clearly showed that the severed ganglion cell axons had grown back to reinnervate the neurons with which they were originally connected. But because the animals’ eyes were inverted, they saw an inverted world and responded in this way. Over time, there was no recovery; the animals were permanently altered (again, mammals are different in this respect; see Chapter 5). Sperry concluded that optic nerve axons can recognize
the cells they are intended to synapse upon; that is, the cells have complementary markers that allow for recognition.
Direct evidence for recognition between retinal and tectal cells has come from the work of Friedrich Bonhoeffer and his colleagues in Germany. They took pieces of retina and tectum and dissociated the cells. By marking cells from the dorsal and ventral regions of the retina and tectum, they showed that cultured cells from the ventral part of the retina adhere preferentially to dorsal tectal cells and vice versa. In other words, the cells could recognize each other regardless of which part of the retina or tectum the cells came from.
How specific is this recognition? Present evidence indicates it is not cell specific, but region specific. That is, the retinal axons are not strictly wired to specific tectal cells. Rather, the ganglion cell axons have a strong affinity for tectal cells from a particular region and they will make connections with cells in that area. If, for example, ganglion cell axons from the chick retina are allowed to grow in a tissue culture dish that is coated with alternating stripes of cell membranes from either the anterior or posterior part of the tectum, axons from the anterior part of the retina (which normally innervate the posterior part of the tectum) in the chick will grow only on the stripes made up of posterior cell membranes, and vice versa. Furthermore, the axons actively avoid the inappropriate cell membrane stripes. From these experiments (and others) has come the realization that there are positive and negative recognition factors at work in this process. That is, some factors attract axons whereas others repel them.
A number of these factors have been identified as small proteins, but almost certainly all of the factors are not yet identified. The assumption is that these cell adhesion or repulsion proteins interact with receptors on the cells and or axons and activate intracellular enzyme cascades similar to those described earlier between the R8 and R7 cells in the developing fruit fly’s eye. Activation of certain receptors and cascades leads to synapse formation between two cells; activation of other receptors and cascades tells the axons to go elsewhere.
Axons grow by way of specialized structures called growth cones that are flattened expansions at the ends of growing axons. A characteristic of growth cones is a prominent array of fine processes that extend from the growth cone as shown in Figure 1-9.
While axons are growing and seeking their way, the growth cone is in constant motion, extending and retracting its fine processes as it explores the area surrounding it. As the growth cone moves along, it adds new membrane to the axon, lengthening it.
The rate and direction of growth cone movement are affected by many factors. The presence of substances in the environment that attract or repel the axons is one key element, and long ago Sperry suggested that a gradient of a chemical substance could
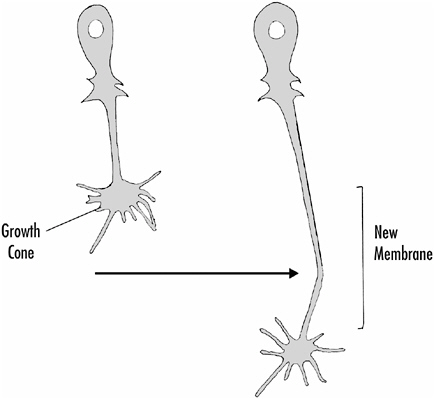
FIGURE 1-9 How an axon grows. The growth cone at the end of a growing axon extends fine processes that explore the surrounding area. The axon adds new membrane near the growth cone, thus extending its length.
guide axons to their target areas. Then cell-to-cell recognition mechanisms would promote synapse formation. He pointed out that two or more chemical gradients at different angles could provide growing axons with quite specific positional information as they grow. But other factors also contribute to growth cone movements, including the substrate on which the growth cone is moving, and even electrical fields in the growth cone’s vicinity. Texture and adhesiveness of substrate, as well as the presence of recognition factors in the substrate, appear to be important.
When axons are required to grow long distances to reach their targets, two other mechanisms have been proposed to play a role. In some situations, guidepost neurons found at intermediate distances along the path the axons follow have been identified. Scientists think that the guidepost neurons provide a secreted attractant chemical that is sensed by the growth cone. Axons grow toward the guidepost neuron, but do not stop when they reach the cell. Rather, they move on to the next guidepost neurons, perhaps because there are repulsion factors on the surface of the guidepost neuron itself. As would be expected, damage to a guidepost neuron can disrupt axonal pathfinding.
A second proposed mechanism depends on the fact that initial axonal pathfinding in the brain occurs early in development, when distances between structures are much shorter than they are later in development or in the adult. Thus, early pioneer axons might serve to mark the path for axons coming along later, or axons migrating later might simply grow along the surfaces of earlier axons.
Synapse Formation
When axons reach their target, they make synaptic contacts with those neurons they recognize. Synapse formation requires reciprocal interaction between growth cone and cell to be innervated. Substances released from the growth cone initiate the formation of postsynaptic structures; conversely, the postsynaptic element provides signals to the growth cone to develop into a mature syn-
apse. Much of the information we have concerning synapse formation has come from study of the innervation of muscle by the axons of motor neurons. This synapse, called the neuromuscular junction (NMJ), is large, employing acetylcholine (ACh) as its transmitter. When a motor neuron axon is active, it releases ACh from its terminals and the ACh diffuses to the muscle membrane, where it interacts with specific proteins that form channels through the membrane. When activated by ACh, these channels allow charged ions to cross the membrane. The movement of charged ions across the membrane results in voltage changes across the muscle membrane, initiating contraction of the muscle. Synapses in the brain work basically the same way, except that the voltage changes that occur across neuronal cell membranes result in the generation of electrical signals that move along axons (rather than contraction as in muscle cells).
The first clues concerning a trophic interaction between a motor axon synapsing on a muscle cell and the muscle itself came from studies on the distribution of the ACh channels along the muscle cell membrane. If ACh is squirted onto a normal adult muscle cell, the sensitivity of the cell to ACh is confined to the synaptic area; that is, the ACh channels are clustered at the synapse. However, if the axon is removed from the muscle—the muscle is deinnervated—the muscle becomes sensitive to ACh all over its surface. That is, in response to the deinnervation, new ACh receptors are synthesized by the muscle cell and they spread all over the cell’s membrane.
Motor axons will reinnervate deinnervated muscle fibers in some animals and when this occurs, the sensitivity of the cell to ACh becomes confined once again to the synaptic region. Thus, reinnervation results in a decreased synthesis of ACh receptors and a clustering of the receptors to the synaptic site area. The same thing happens during development. Initially a muscle cell is sensitive to ACh all over its surface in the young embryo, but once it is innervated by a motor axon, the ACh receptors become clustered to the synaptic region.
These observations suggest that the motor axon terminal re-
leases a substance that causes clustering of ACh receptors. Such a protein has been identified and is called agrin. This protein interacts with agrin receptors on the muscle membrane, which initiates a series of intracellular biochemical reactions resulting in phosphorylation of proteins. So once again, a chain of biochemical reactions similar to that shown in Figure 1-7 is implicated as playing a key role.
Agrin almost certainly contributes more to synapse formation than simply clustering the ACh receptors, but it also is not the whole story. Another protein called ARIA (acetylcholine receptor inducing activity) was isolated from chicken brain and causes an increase in ACh receptor synthesis. Again, it is thought that ARIA is released from the motor axon nerve terminals.
The story is by no means complete and, as noted above, it is believed that chemical signals also go from the postsynaptic cell to the innervating presynaptic axon, and these signals trigger changes in the presynaptic terminal that lead to the formation of a mature synapse. But what these signals are is not known.