The Great Brain Debate: Nature or Nurture? (2004)
Chapter: Part III—The Aging Brain 6 Is Aging of the Brain a Disease?
6
IS AGING OF THE BRAIN A DISEASE?
We have all witnessed the deterioration of our parents or grand-parents as they aged. Too often, it is a sad deterioration with a loss of quality of life as they fail mentally. At the same time, we read about the marvelous advances in medicine that are supposed to make us healthier and live longer. What is going on? Are people living longer? And if so, what are the consequences? Is it likely that we could live to be 150 or even 200 years old, and what would it take for us to achieve that goal if, indeed, this is something we would want to do?
These are the topics I cover in this chapter. I will first describe what seems to be happening in the brain as we age. But this topic, like many others discussed in this book, generates much controversy and the neurobiological facts are disputed. Nevertheless, there are a number of things that can be said.
When I was in medical school, no one talked about Alzheimer’s disease in people over 65 years of age; at that time the failing mental abilities seen in those over 65 were described as
senile dementia and a natural consequence of aging for many. Today the view is different. Alzheimer’s disease is thought of as a real disease regardless of the age when it strikes. But is this an accurate perception?
A number of other age-related degenerative diseases such as Parkinson’s disease and age-related macular degeneration can occur as people age. What causes these diseases? How might we combat them and what types of therapies are possible? Here I shall use as a model an inherited retinal degeneration, called retinitis pigmentosa, which typically attacks people in their twenties and progresses until complete blindness ensues by age 60 or so. It might be the best-understood neurodegenerative disease at the moment, and therapies for its cure, at least in animals, look promising.
Neuronal Changes with Age
I noted in Chapter 1 that virtually all neurons are generated in the developing brain by about six months of age, but the brain is then only about 30 percent of its adult size. The brain grows rapidly in the first three years of life, but does not reach its maximum weight until about age 20, as Figure 1-3B showed. Much of that increase is due to the growth and elaboration of neurons, shown in Figure 1-4, and presumably neuronal circuitry. After age 20, on the other hand, brain weight and volume gradually decline and this continues for as long as we live.
How much brain weight is lost and what does the loss mean? Everyone agrees that the brain shrinks with age—this is seen in both humans and other primates. The figure often given for humans is up to 15 percent shrinkage over a life span of 100 years, but some investigators believe this figure is too high. But what does a loss of even half of 15 percent mean?
Because few new neurons seem to be generated in the adult brain (as discussed in Chapter 5), one possibility is that the shrinkage reflects loss of neurons, and certainly this occurs. If the shrinkage were due entirely to neuronal loss, the number of neurons lost per day would be astonishingly high—about 200,000 per day or
more than 8,000 per hour! This calculation is based on an estimated volume loss of 7 percent over a life span of 100 years with the number of neurons initially present (at one year of age) being 1011 or 100 billion (a conservative estimate).
In the 1950s, Harold Brody of the State University of New York examined the brains of 20 human subjects from newborn to age 95 and reported extensive cell loss—up to 40 percent in several regions of the cortex. However, later studies questioned these results, and some researchers reported either minimal neuronal cell loss or even none in many areas of the cortex. One reason suggested to explain Brody’s results was that cases of Alzheimer’s disease or other dementias might have been included in his sample, and it is well known that these conditions cause massive cell loss.
There is a general agreement that brain shrinkage occurs, so if neuronal cell death is not the major contributor, at least in the cortex, what is going on? Neuronal atrophy is one possibility and there is substantial evidence that this happens with age. Just as neurons in the very young brain grow larger and extend more dendritic branches that go longer distances (shown in Figure 1-4), neurons in the aging brain do the opposite—they shrink and have fewer branches. The atrophy of neurons and neuronal branches almost certainly results also in fewer synapses in the aging brain.
Another factor is loss of white matter in the brain—those regions where axons are most prevalent—and again, there is both human and animal evidence for this. Indeed, recent studies have reported losses of as much as 30 percent of the white matter in aged brains. One suggestion, for which there is increasing evidence, is that the myelin sheaths surrounding axons break down with age. This could account for the decrease in white matter volume and also for some of the cognitive changes that occur with age. For example, myelin promotes more efficient transmission of the electrical signals that travel down axons, and if this transmission is slowed, the ability to process information could also be slowed. And, as I shall describe below, older people process information more slowly.
Biochemical measurements also show significant changes
with age. Total brain protein is down by as much as 30 percent in 80-year-old brains compared to protein in the brains of 20-year-olds, and NMDA receptors have been reported to be lower by about 30 percent in aged brains. This finding might partially explain one of the most common complaints of older people—a loss of memory recall. (Remember the importance of NMDA receptors in memory and learning that was discussed in Chapter 4.)
What can we conclude from present data? Whereas it is probably correct that there are regions of the brain, including certain cortical areas, that show minimal neuronal loss with age, certain other regions—for example, some subcortical areas, the brain-stem, and other regions—do show substantial cell loss, as much as 40-80 percent. In the retina, for example, careful measurements of the photoreceptors, which are quite easy to count accurately, indicate a loss of 30 percent of both rod and cone photoreceptors from teenage years to age 80.
So, many brain regions certainly have substantial cell loss, and all regions are likely to have some. Neuronal loss might also be selective for certain types of neurons. Betz cells, which are very large neurons found in the motor cortex, seem to wither away with age and are pretty much gone by age 80. And white matter, NMDA receptors, and proteins in general seem to be lost. A caution, noted above, is that the dementias are so common in elderly people, especially over age 85, that a few diseased brains might have been included inadvertently in some of the studies these above conclusions are based upon. Conversely, brains that were found to have substantial neuronal cell loss might have been excluded from some of these studies because of a belief that they are diseased—brains from people with early Alzheimer’s, for example.
Cognitive Changes with Aging
Everyone over 50 complains of not remembering things as well as when younger, and it is certainly true, as shown by cognitive studies, that older people have decrements in learning and memory. Not all types of memory seem to be equally affected. Delayed
memory—recall of something learned a while ago, for example, remembering a name—is much more affected than immediate memory—recall of something that has just happened—but with age more time is needed to learn new information and it is harder to stay focused on a memory task. When different brain regions were examined to see if shrinkage of a specific area could be correlated with the loss of memory function in people between ages 55 and 85, the only area that showed a significant decrease in volume (measured by brain imaging techniques) was the hippocampus. We shall return later to the issue of memory loss and hippocampal changes.
Other cognitive deficits have been seen in older people. For example, the manipulation of information slows down, so it takes an older person longer to come up with a response to a complicated scenario. This deficit is thought to be related to changes occurring mainly in the frontal lobes, which are concerned with reasoning, planning, and keeping things in one’s mind. Older people need to reflect more on an issue than do younger adults. On the other hand, vocabulary and language skills do not appear to deteriorate much, if at all, in normal people as they age; neither is IQ or abstract thinking much affected.
When do these changes begin to take place? Already in one’s twenties, changes appear to be occurring. For example, psychologists can measure differences between 30-year-old subjects and people in their late teens or early twenties. Information is processed a bit slower and is held for a shorter time in conscious awareness, and recall is somewhat less efficient in the 30-year-olds. Cognitive scientists agree that aging of the brain begins at about the time brain volume begins to decline, and this decline of cognitive abilities and brain volume continues for the rest of our lives.
How Long Could We Live?
It is common knowledge that average life expectancy has increased spectacularly in the past 100 years. In Europe and the
United States, the average life span was less than 47 years in 1890 and by the 1990s it was more than 75 years. During the decade of 1968-1978, average life expectancy rose at the phenomenal rate of one month per year for all those over 50! In Japan, the figures are even more impressive; by the mid-1990s the average life expectancy for women was about 83 years of age. Japanese men, like men the world over, had a lower average life span of 77 years. Developing countries also showed substantial increases of average life expectancy in the 1990s.
But what about absolute life expectancy? Has that increased? Here the news is quite different. Ancient texts mention individuals living to 120 years of age, and today we occasionally hear of someone that old, but this is very exceptional. The age at death of the longest-living human that is well documented was 122 years, and in the fall of 2003 the then-oldest man in the world died at the age of 114. He was Japanese and the oldest woman alive at that time, also Japanese, was 116 years old. Indeed, if one looks at the trends in human longevity from antiquity to the present day, it has not increased significantly if at all, as shown in Figure 6-1.
Whereas average life expectancy has increased dramatically
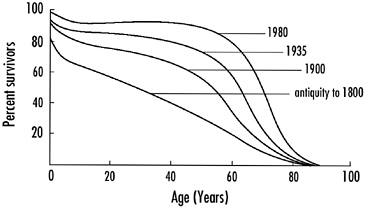
FIGURE 6-1 The changes in life expectancy from ancient times to 1980. Average life expectancy has changed dramatically from about 35 years 200 years ago to more than 75 today. Absolute life span of humans has not increased significantly since antiquity. Only a small percentage of humans live to be more than 100.
as a result of medical advances and improved housing and sanitation, the maximum life span has not. The number of people who live to be 60 has increased from less than 20 percent in the early nineteenth century to more than 80 percent today, but the end-point of life expectancy remains at about 100 years of age.
Although the average life expectancy has been increasing virtually linearly since the 1960s in Western countries, it is expected to level off with an average life expectancy of about 85. In other words, it is likely that we are already coming close to our maximum average life expectancy, if there is a biological limit to absolute life expectancy.
My view is that this limit is real and I suspect it might relate to the brain and its aging. Whereas we can replace hearts, lungs, and livers, we cannot replace brains or even brain cells, at least at the moment, and some believe that we will never be able to replace whole brains. Indeed, as someone glibly pointed out, if whole brain transplants were possible, it would be far better to be the donor rather than the recipient, for obvious reasons! This is why there is so much interest in the possibility of stem cells remaining in the adult brain (discussed in Chapter 5). Indeed, if they are present generally, or even in a relatively few places, and could be induced to generate a variety of new neurons to replace dying or dead ones, one might suppose that we could renew our brains and increase maximal life span.
The transplantation of embryonic stem cells into a brain to replace dead neurons and maintain brain circuitry is another possibility that is receiving much attention. Alternatively, it might be possible to find ways to stop or slow the neurons’ aging processes. All these possibilities are being explored, but at the moment they are still very distant.
But do we even want to extend our life span significantly? Perhaps this is a philosophical question; nevertheless, it has biological implications. Evolution depends on organisms having finite lifetimes, so that different mixes of genes can be expressed in new generations. But are humans still evolving biologically? I am not sure anyone knows.
If we did live 50 percent longer, to 150-180 years, population pressures would be substantially increased, and overpopulation on Earth is already an enormous problem. Would we be willing to restrict population growth to maintain a habitable planet? And, of course, the presumption in all of this is that quality of life would be maintained at a vigorous level for much longer, so retirement would be at 130-150 years of age, rather than between 60 and 70 as it is now for most people in the developed world. How would this affect human creativity, which depends so much on the young. What might be the economic implications if people lived for 150 years? None of this has been thought through. Perhaps it should be.
Alzheimer’s Disease
No disease of the elderly is more feared than Alzheimer’s disease. It strips sufferers of dignity, and eventually they lose those traits that make them unique human beings. As many as 4 million Americans suffer from the disease, and one estimate suggests that by 2040 as many as 14 million people will be affected in this country alone.
Those who suffer from it commonly show a decline in mental abilities in their late fifties or early sixties, beginning with deficits in recent memory, and progressing to a loss of virtually all higher mental functions. Confusion and forgetfulness are the most common symptoms, followed in some patients by difficulty in executing motor acts that previously were simple to do, and even loss of speech. A few people have a much earlier onset of Alzheimer’s disease—but many of these come from families in which the disease is inherited as an autosomal dominant disorder of midlife. Many more people show Alzheimer’s-like symptoms as they age and eventually display the full-blown mental deterioration of the condition. One study showed that at age 65 about 15 percent of the U.S. population is showing Alzheimer’s-like symptoms and by age 85 as much as 50 percent is showing them.
But, you say, my Aunt Marian lived to be 102 and she was as
sharp as a tack almost to the day she died. This is certainly correct, that certain people live to be 100 or a bit more and show virtually no symptoms of the disease. But, unfortunately, it is a relatively rare person who reaches the century mark without evidence of one or another neurodegenerative disease. Alzheimer’s accounts for about 50 percent of the neurodegenerative diseases, and changes found in the brains of Alzheimer’s victims are often found also in the brains of “normal” aged people, although in them they are much less severe.
The striking feature of the brains of Alzheimer’s patients is a tremendous loss of brain volume and massive degeneration of neurons. Significant portions of the cortex atrophy, and PET scanning indicates a marked reduction in brain metabolism. One region of the brain particularly hard hit is a nucleus found on the basal surface of the forebrain, called the nucleus basalis. Most of the synapses that release acetylcholine in the cortex are made by the axon terminals of neurons coming from nucleus basalis neurons. In Alzheimer’s patients as much as 60-90 percent of the enzyme responsible for synthesizing acetylcholine in the brain is lost. One treatment being tried for the disease is an inhibitor of the enzyme that breaks down acetylcholine in the brain, the idea being to raise acetylcholine levels. Its success so far is modest at best. The neurons in the nucleus basalis are thought to play a role in integrating both subcortical and cortical information processing, and thus, degeneration of this nucleus could be key in explaining the cognitive defects in Alzheimer’s disease.
Why do neurons deteriorate and eventually die in Alzheimer’s patients? Neuroscientists are beginning to get a handle on the answer. As was the case for Huntington’s disease, it appears that the accumulation of toxic proteins causes the neurons to die. But unlike Huntington’s disease, the toxic proteins that accumulate are extracellular (outside the neurons) in Alzheimer’s disease. The principal protein that accumulates is β-amyloid, a relatively small protein consisting of 40-42 amino acids that is a natural constituent of the extracellular space in the brain and tends to build up in all of us as we age. However, a prominent feature of brains af-
fected by Alzheimer’s is the presence of many dense accumulations of protein, mainly β-amyloid, in prominent extracellular structures called plaques as shown in Figure 6-2.
The aggregates form because β-amyloid is sticky, especially when it is somewhat misfolded. This occasionally happens with proteins, which are, of course, long chains of amino acids that must be folded correctly to carry out their specific function. In forming plaques, β-amyloid molecules stick to each other and to other proteins, forming a mass that binds to both neurons and glial cells. Having a glob of sticky stuff on a neuron is probably enough to disrupt its function and structure, but the immune system also comes into play. The plaques set off an inflammatory reaction that further complicates matters in the region and results in more cell death and brain damage.
Plaques and β-amyloid are not the only things that accumu-

FIGURE 6-2 The histological changes seen in the brains of Alzheimer’s patients. The arrow in the lower left points to a plaque that consists mainly of misfolded β-amyloid protein encompassing neurons and presumably disrupting their function. The other arrows point to neurons filled excessively with another protein called tau.
late in the brains of Alzheimer’s sufferers. An intracellular protein called tau also accumulates, forming tangles inside neurons. Present evidence suggests that the accumulations of tau in tangles, as well as the loss of neurons, is secondary to the plaque formation. Indeed, it might be that the tangles are a sign that a neuron is dying. On the other hand, in a variant of Alzheimer’s disease called Pick’s disease—which has virtually the same symptoms—intracellular tau protein accumulations are the main pathological feature. Thus, accumulation of tau protein might play a role in causing neurodegeneration.
How does genetics fit into all this? At least five genes predispose people for Alzheimer’s disease. At the moment, we can account for about 50 percent of the Alzheimer’s cases based on a genetic predisposition (excluding those cases of early onset due to an autosomal dominant mutation). What is meant by a genetic predisposition? People with a predisposing gene might not get the disease, but the chances are higher than if they do not have that specific gene variant. For example, relatives of people with Alzheimer’s who have the variant gene have an increased risk of about 38 percent for the disease at age 90. However, about 50 percent of the population shows Alzheimer’s-like symptoms at that age, so of the healthy 50 percent, if they have Alzheimer’s in the family, about 40 percent, or two out of five, will eventually come down with the disease. The other three will stay healthy even with the predisposing gene.
What can be said about the nature of the predisposing genes for Alzheimer’s? Without getting into the details of their specific names or explaining their exact function, all appear to crank up levels of amyloid in the brain. One of the genes codes for the amyloid precursor protein (APP) from which β-amyloid is formed. This predisposing gene might have a small variation (called a polymorphism), which means that the β-amyloid protein it codes for is slightly altered. For example, more often than normal the protein might not fold quite right or perhaps it is more resistant to degradation. Both of these hypothetical scenarios would lead to more β-amyloid accumulation.
Other predisposing genes are involved in the breakdown of APP to β-amyloid and they, too, could result in more β-amyloid accumulation. Yet other predisposing genes seem involved in the breakdown of β-amyloid itself. The changes in the genes are presumably not all that large, and thus their effects are minimal so that it takes decades for the buildup of the amyloid to reach levels high enough to cause Alzheimer’s.
An important point to reiterate is that β-amyloid accumulates in all of us as we age, and many brains of normal older people even show plaques at autopsy. So the key is how much and how fast amyloid accumulates. And, of course, one could have variations in these or other genes that could predispose one to not get the disease. Having a gene that codes for a slightly more efficient enzyme that breaks down β-amyloid is certainly possible in certain people—the Aunt Marians, perhaps, who live to 102 with virtually no cognitive deficits.
Of interest, and perfectly understandable, is the fact that three of the same genes are involved in families that inherit Alzheimer’s as an autosomal dominant disorder that starts at a much earlier age. These families must have a more severe alteration in the genes, which then leads to a much more rapid accumulation of β-amyloid and the midlife onset of the disease.
Parkinson’s Disease and Other Age-Related Neurodegenerative Diseases
Parkinson’s disease is another age-related neurodegenerative disease commonly seen in older people. Indeed, Parkinson’s disease is the second most common neurodegenerative disease, affecting about 1 million Americans over the age of 55. It is also due to degeneration of neurons, but in this case to neurons in a specific midbrain nucleus that innervates a large part of the basal ganglia complex. The basal ganglia are concerned with the initiation and execution of movement. Thus, patients with Parkinson’s disease show specific motor deficits. Initially the disease is marked by a rhythmic tremor of the limbs at rest, but then it progresses to a
rigidity of limb muscles. Eventually, Parkinson’s patients have difficulty initiating movements. They tend to shuffle as they walk and have a mask-like facial expression. Parkinson’s disease sufferers can also develop a dementia.
The neurons that die in Parkinson’s disease release the neuromodulator dopamine in the brain, and a deficiency of dopamine seems to be a major cause of the symptoms. Thus, one therapy consists of giving patients a molecule that is a precursor to dopamine—a molecule that the brain can easily convert to dopamine.
This molecule, L-dopa, is made naturally in the brain from the amino acid tyrosine and with one enzymatic step can be converted to dopamine as shown in Figure 6-3.

FIGURE 6-3 The conversion of the amino acid tyrosine into dopamine requires just two steps and involves an intermediate molecule, L-dopa. The first enzymatic step involves adding a hydroxyl molecule (+OH) to tyrosine at the position shown by arrow 1. The second step is the elimination of a carboxyl molecule (−COO−) from position 2. L-dopa readily crosses into the brain when given to a patient, but dopamine does not; hence L-dopa is given to patients suffering from Parkinson’s disease.
L-dopa therapy doesn’t cure the disease, but it substantially relieves symptoms in many patients for substantial periods. It does not work for all because it causes side effects in some patients. Another treatment, much riskier and more experimental, is the transplantation of dopamine-secreting cells into the brain. This was described briefly in Chapter 5, where I pointed out that this approach has so far met with only modest success.
How much neurodegeneration must occur for the symptoms of Parkinson’s disease to occur? This has been quite convincingly documented, and the findings indicate there must be a loss of about 50 percent of the dopaminergic neurons themselves and a decrease in dopamine content in the basal ganglia of about 75-80 percent before the classic symptoms appear. Thus, a substantial amount of neurodegeneration must happen before the disease becomes apparent.
The causes of Parkinson’s disease are not well understood, although some suggest that it, like Alzheimer’s and Huntington’s diseases, might be caused by the accumulation of a toxic protein. Cytoplasmic inclusion bodies, called Lewy bodies, are found in the dopaminergic neurons early in Parkinson’s disease. One protein, called α-synuclein, is the major component of the Lewy bodies. The function of α-synuclein is not well understood, though it seems to have a role in synaptic function and synaptic plasticity and might be involved in the release of dopamine at dopaminergic synapses. The α-synuclein molecule, like β-amyloid, is sticky and binds to other α-synuclein molecules as well as to other proteins.
With Parkinson’s, as with Alzheimer’s, several genes, including the gene that codes for α-synuclein itself, have been implicated as playing a predisposing role. Two mutations in the α-synuclein gene have been linked to rare forms of Parkinson’s disease, and these mutant genes as well as the normal α-synuclein gene have been expressed in the fruit fly, Drosophila melanogaster, using transgenic techniques. Interestingly, all three expressed genes lead to a Parkinson’s-like disease in fruit flies, causing Lewy bodies to appear in the dopaminergic neurons and producing a late-onset locomotor dysfunction in the flies. There
is, however, little difference in the effects of the three expressed proteins, indicating that normal α-synuclein is almost as toxic as the mutant protein. This agrees with studies on humans in which it has been shown that most Lewy bodies in Parkinson’s patients are made up of normal α-synuclein molecules.
A number of key questions remain. Why does α-synuclein accumulate, why does it accumulate specifically in the dopaminergic neurons, and how does it kill the dopaminergic neurons? And also, what are the triggering events for Parkinson’s disease? There are clearly predisposing genes for some instances of Parkinson’s, but most cases seem not to be explained in this way.
Environment and Neurodegenerative Diseases
The emphasis in the discussion so far has been on the role of predisposing genes in the causes of age-related neurodegenerative diseases. But many cases of these diseases cannot be linked to a genetic factor, so it is believed that environmental factors must be the precipitating cause. Only about 50 percent of the cases of Alzheimer’s disease can be linked to one of the five predisposing genes for it. The cause of the other 50 percent is very much up in the air. The genetic links to Parkinson’s disease are even weaker.
Clearly, brain injury or trauma can lead to excessive brain cell loss and people who have had such injury can show Alzheimer’s-like symptoms. People prone to brain injury, like prizefighters, can develop these symptoms and in such cases are described as punch-drunk. Suffering from a prolonged high fever also can cause substantial neuronal death and result in an Alzheimer’s-like condition. Such brain cell death might not be distributed evenly throughout the brain, but confined to a specific region or set of cells for some unknown reason. The former heavyweight boxing champion Muhammad Ali might be an example of this. He is said to be suffering from Parkinson’s disease. He shows a severe tremor, walks sluggishly, has difficulty initiating movements, and displays an expressionless face. It seems quite possible that his disease is linked to the pounding his head took during his fighting days.
Toxins and toxic substances can also be involved in some cases. A chemical called MPTP, discovered because it was formed as a by-product of defective heroin synthesis by drug dealers in California, led to tragic consequences in a number of young drug users. MPTP breaks down in the body to a neurotoxin, MPP+, which selectively destroys dopaminergic neurons. Those unfortunate to take such defective heroin show symptoms of severe Parkinson’s disease within a few days of ingestion. Other toxins have been shown to induce Parkinson’s-like conditions, so it is possible that both man-made and natural toxic substances can be involved in the disease’s etiology.
Parkinson’s disease is also linked with the great influenza epidemic of 1917. A subgroup of people who survived the epidemic subsequently came down with severe Parkinson’s disease. Here the precipitating cause might have been a virus. These patients were some of the first to be treated with L-dopa by the famed neurologist-author Oliver Sacks. Sacks subsequently wrote a book on this experience, called Awakenings, that was turned into a well-received film.
Finally, the question has been raised as to whether excessive stress can predispose to neurodegenerative disease. When an animal or human is stressed, one of the things that happens is an increased release of glucocorticoids from the adrenal glands. In the short term glucocorticoids are helpful to a stressed animal. Among other things, they promote the breakdown of protein to glucose, helping to make fat available for use and increasing blood flow. With prolonged stress and a prolonged release, the glucocorticoids can have damaging effects, including increased blood pressure, gastric ulcers, and depression of the immune system.
Of particular interest here is the fact that prolonged stress in animals can also cause brain damage, especially to the hippocampus. In Chapter 5, I noted that glucocorticoids decrease the generation of new neurons in the hippocampus, and this can account for some of the effects of prolonged stress. However, hippocampal volume is also reduced in prolonged stress and one study showed a loss of hippocampal CA1 cells in stressed rats.
The situation with regard to stress and the aging human brain is by no means clear, but some studies on aging people suggest a relation between stress hormones and difficulties with memory tasks. In one study, 11 healthy subjects in their sixties or seventies were followed for four years. Of these, six showed increased levels of cortisol, one of the main glucocorticoids released from the adrenal gland in stress. The other five had stable or decreased levels of cortisol. The six whose cortisol levels increased over the four years had difficulty with certain memory tasks such as navigating a maze or remembering a list of words. Those whose cortisol levels remained low or even decreased somewhat performed these tests with no difficulties. A subsequent fMRI study showed that the hippocampi of subjects with higher cortisol levels were smaller by about 14 percent.
Retinitis Pigmentosa: A Model Neurodegenerative Disease
As people age, they often lose not only cognitive function but also visual, auditory, and other sensory functions. They can gradually become isolated from other humans and the environment. A particularly devastating condition is age-related macular degeneration (AMD), which robs older people of their central vision. The macula is a specialized region of the retina having the highest density of photoreceptors. At its center is a small indented area, the fovea, which contains only cone photoreceptors and serves all of our high-acuity vision. We look at things we want to examine closely with that small region of the retina. There are only about 35,000 photoreceptors in the fovea, out of a total of 6 million cones in the entire human retina and perhaps 95 million rods, but if the foveal photoreceptors are lost, which happens in AMD, high-acuity vision is lost, and it is devastating for those affected. They cannot read, watch television, or do any of the things normally sighted people take for granted.
We know very little about AMD, what causes it or predisposes to it. The only environmental link known is smoking: Those who smoke have an increased risk of AMD. Some rare
forms are inherited and those susceptible might get the disease at a young age. In one such form of macular degeneration, called Malattia Leventinese, the mutation has been identified in a gene coding for a secreted protein of unknown function. The mutation results in misfolding of the protein that causes it to be abnormally secreted and to accumulate both intracellularly and extracellularly.
This form of macular degeneration, then, has characteristics of other neurodegenerative diseases such as Huntington’s, Alzheimer’s and Parkinson’s diseases in which excess accumulation of protein, either extracellularly or intracellularly, is the defining feature. However, for most cases of AMD, the link with genetics is tenuous. At present, there are no animal models of the disease, because relatively few animals have a fovea like ours. Higher primates, some birds, reptiles, and fish do, although the foveas of these latter animals are somewhat different. Thus, we can do relatively little for most cases of AMD at present and it severely compromises the quality of life of those who suffer from it. The same is true of those who become deaf in old age. They, too, can become quite isolated from their fellow humans.
An inherited retinal degeneration called retinitis pigmentosa (RP) is not a disease of the aged. Indeed, in most cases its onset is in the late teens or early twenties, progressing to complete blindness in the fifties, sixties, or even later. It is a fairly rare disease, affecting about one in 4,000 people worldwide. It is a disease of the rod photoreceptors, but cone photoreceptors are eventually affected as well. In contrast to AMD, where central vision is lost, RP begins in the periphery of the retina, gradually restricting the visual field. The fovea is the last to go, but eventually it, too, degenerates, leaving the individual completely blind. This disease has been studied intensively for more than 40 years and enormous progress has been made in understanding its causes. Furthermore, progress is being made with therapies for the disease, at least in treating animal models of the disease, of which there are many. Animals that have RP-like diseases include mice, rats, dogs, and cats.
RP, like many neurodegenerative diseases, was thought at one time to be a single disease with a single cause. We now realize that this is a very incorrect view. The first hint that RP represents several different diseases came from genetic studies. Careful analysis of patients and their families with RP first showed that about 50 percent of the RP cases could be linked to a genetic cause, but the genetics is varied. Sometimes the disease is inherited as an autosomal dominant disease—this means that the chances of an offspring having the disease are 50 percent; sometimes it is inherited as a recessive disease—if both parents have the mutant gene, one out of four children will inherit the gene.
There are also cases where the disease’s inheritance is sex linked. The mutant gene is on the X chromosome, but females have two X chromosomes while males have just one. Males show the disease when the gene in their single X chromosome is defective; females need to have the defective gene in both of their X chromosomes. Therefore, males inherit the disease much more frequently than do females in these families. There are some even rarer inheritance patterns in RP, but they need not concern us here. Suffice it to say that the genetic variability is large, but there is much more to come.
Of the 50 percent of RP cases that can be linked to genetics, about 40 percent show an autosomal dominance inheritance. These cases were the first in which the specific genetic defect was discovered in the early 1990s by Thaddeus Dryja and Eliot Berson and their colleagues at the Massachusetts Eye and Ear Infirmary in Boston. They showed that a mutation affecting a single amino acid in the gene coding for rhodopsin (the protein that, when combined with a vitamin A derivative, is light sensitive and initiates vision) was the culprit in one family with RP. This discovery opened the floodgates and within a decade numerous mutations both in the rhodopsin gene and other genes found in photoreceptor cells and important for initiating vision were discovered.
As of this writing (Fall, 2003) more than 100 different mutations in 35 different RP-causing genes have been found. In rhodopsin alone, more than 70 RP-causing mutations have been
identified. Thus, RP is not a single disease, but more than 100 diseases, and this accounts for only about 60 percent of the cases of RP. The lesson here is that it is likely many of the neurodegenerative diseases will be as heterogeneous, and careful classification of them is critical if we are to deal with them. RP might be leading the way in this regard.
Figure 6-4 shows the rhodopsin protein, consisting of a chain of 348 amino acids, each indicated by a circle. The chain weaves in and out of the membrane seven times. The amino acids that, when altered, lead to RP are indicated in black.
At least 70 mutations in the rhodopsin molecule can lead to RP diseases. Some of the mutations lead to a misfolding of the protein, others to alterations in how the molecule is excited by light. Different mutations result in somewhat different diseases in terms of age of onset and progression of the disease, although there is considerable variability in people with the same mutation. This variability or penetrance is not well understood; as discussed in earlier chapters, both genetic and environmental factors could be involved.
With some understanding of the genetic basis of the autosomal dominant form of RP, investigators have turned to therapies and are focusing on two approaches: genetic and pharmaceutical.
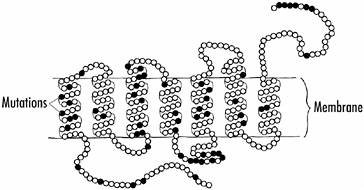
FIGURE 6-4 A schematic drawing of the rhodopsin molecule. Amino acids which when altered as a result of a genetic mutation cause retinitis pigmentosa are indicated by black circles.
Gene therapy has already had spectacular success in a strain of dogs with one rare form of RP. These dogs have a defect in a gene that codes for a protein required to make the correct form of vitamin A to make rhodopsin light sensitive. A good gene is inserted into a virus that is then injected into the eye—into the subretinal space between the photoreceptors and the cells behind, the pigment epithelial cells. In this case, the defective protein is present in the pigment epithelial cells, but this is unimportant for the discussion; the defective protein could as well be in the photoreceptors. The injected virus infects the pigment epithelial cells and the cells begin to make the normal protein—they now have the correct gene—and the dogs show a remarkable recovery. For any of this to happen requires, of course, exact knowledge of the mutated gene, a virus that readily infects the defective cells, and luck!
The second approach is via drug or chemical therapy, and several classes of compounds have been tried. Significant success has been obtained in animals with some forms of RP following the administration of growth factors such as the neurotrophins described in the beginning of Chapter 2. These molecules are important for the survival and growth of neurons in the developing brain, and they also seem to help defective cells from dying or, at the very least, they slow down the deterioration of genetically defective cells. All forms of RP might not respond to growth factors, and infusing the growth factors into the eye is a problem because these are proteins that must be put directly into the eye. That we have animal models with various forms of RP is important to this effort, and it is now possible to make mice with other forms of RP using transgenic techniques, thus providing the opportunity to test drugs on a wide variety of mutations that cause the disease.
Another substance that has been shown effective in RP is vitamin A. Initial studies with a population of human RP patients who had various forms of the disease suggested a very small but positive effect of vitamin A therapy. However, the effect was so minimal that many physicians felt it was not particularly useful. But as transgenic mouse models of various forms of RP have been
developed and the specific genetic defects found in human patients are induced in them, it turns out that some types of RP are helped quite significantly by vitamin A, but others not at all.
The lesson here is that when seeking therapies for a disease, it is critical to know what specific form of the disease you are dealing with. A therapy might be very effective for one or a few forms of a disease, but ineffective for many or most forms. Thus, when testing a therapy on a population with various forms of a disease, the positive effects might be swamped out by the nonresponders. Thus, careful characterization of different forms of the neurodegenerative diseases is essential, and this classification is just beginning for many age-related neurodegenerative diseases such as AMD, Alzheimer’s, and even perhaps Parkinson’s disease.