Oil in the Sea IV: Quick Guide for Practitioners and Researchers (2023)
Chapter: Fates: Where Does the Oil Go?

Fates: Where Does the Oil Go?
When oil spills into the ocean, an array of physical, chemical, and biological processes determine its fate. Because many oil components do not mix with and are lighter than seawater, oil spills form slicks on the sea surface and droplets (or bubbles in the case of natural gas) in the ocean water column. Oil may sink to the bottom when it adheres to heavier particles. The interactions between oil and seawater, marine organisms, and the atmosphere will change the oil’s composition and physical properties and begin to break it down. Because these processes change the chemical composition of the oil, its physical properties can also change, in turn altering the environmental fate. The major processes are listed in Table 2 and shown in Figure 12.
IMAGE SOURCE: National Oceanic and Atmospheric Administration.
TABLE 2 Major Processes Affecting Oil Fate
| Process | Definition | Promoting Conditions |
|---|---|---|
| Spreading on water | Movement of oil on the sea surface that creates thin, floating pools of slicks and sheens | Low-viscosity oils spread more quickly than those with high viscosity |
| Dispersion | Incorporation of spilled oil into seawater | Oil concentration decreases as oil mixes with seawater, with the degree of mixing influenced by turbulence and ocean currents |
| Aerosolization | Transfer of liquid petroleum from the sea to surface to the atmosphere by particle formation | Wind, waves, and white capping |
| Emulsification | Formation of a mixture consisting of small droplets of oil and water | The amount of oil at the sea surface, wind, and wave action (turbulence), and the use of dispersants |
| Evaporation | Transfer of the slick or more volatile substances from the oil slick to the air by vaporization | Higher surface area of oil exposed to the air, high fraction of volatile compounds in the oil, warm temperatures, wind and wave action (turbulence) |
| Microbial degradation | Metabolism and degradation of organic compounds in the oil by microorganisms such as bacteria | Presence of nutrients, such as nitrogen and phosphorus, and the presence of a wide range of microorganisms |
| Process | Definition | Promoting Conditions |
|---|---|---|
| Dissolution | Transfer of water-soluble compounds in oil into the surrounding water | High fraction of water-soluble compounds in the oil spilled, turbulence, and large interfacial area between petroleum fluid and the water |
| Photo-oxidation | Chemical reactions occurring in the liquid-phase petroleum and weathered petroleum as a result of sunlight in the presence of oxygen | Oil at the surface of the water, in the photic zone, and on shorelines and beaches; sunlight; formation of free radicals which in turn react with oxygen to produce reactive oxygen species |
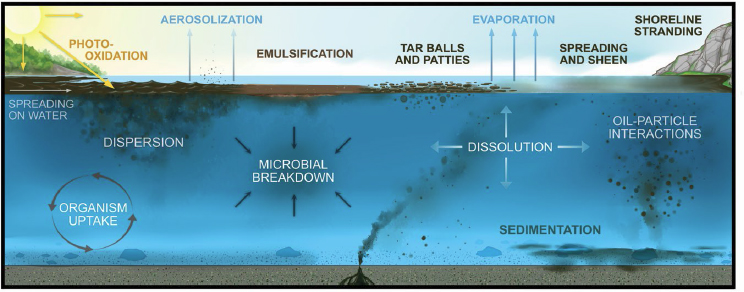
SOURCE: Image provided courtesy of the American Petroleum Institute, produced by Iron Octopus Productions, Inc.
Recent Advances in Understanding Oil Fate
Since the publication of Oil in the Sea III by the National Research Council in 2003, the following major technological advances have improved our ability to monitor and predict the fates of spilled oil in the ocean.
PHOTO-OXIDATION AS AN IMPORTANT FACTOR IN OIL FATE
Over the past two decades, there has been a paradigm shift in understanding the importance of photochemical reactions—chemical changes in oil caused by sunlight—in the fate of spilled oil. Observations and experiments carried out during the DWH oil spill, the availability of sophisticated new chemical methods for analyzing photo-oxidation reaction products and combining the resulting new knowledge with pre-2003 research have yielded new insights that point to the role of photo-oxidation early in the weathering and breakdown of slicks on water. Photo-oxidation has also been shown to affect oil coating sand and rocks on shorelines and vegetation in marshes. These findings have generated new questions about the quantity, identity, fate, and toxicity of photo-oxidation reaction products.
THE ROLE OF ‘OMICS IN STUDYING MICROBIAL COMMUNITIES IN THE SEA
Revolutionary advances in high-throughput DNA sequencing, bioinformatic software, and sequence databases (collectively termed ‘omics technologies) have enhanced the ability to detect and analyze microbial communities, including those that affect the fate of oil in the ocean.
‘Omics technologies were used extensively during and following the DWH incident, leading to many important discoveries such as the identification of new oil-degrading bacteria, microbial metabolic pathways, and a further understanding of microbial communities and the ways they may change after an oil input.
The classical method for identifying and enumerating microbes is to collect samples and cultivate individual species on artificial media as clones (i.e., colonies on agar plates). This is a slow, labor-intensive, and highly biased procedure that isolates fewer than 1 percent of the total microbes in a sample (only those capable of growing on laboratory media in pure culture). With ‘omics techniques, microbial identity and activity can be studied without having to cultivate organisms or separate them from their community partners. Speed is also a factor: with high-throughput sequencing methods and, recently, miniaturized hand-held DNA sequencers, analyses that once required sample transport, significant laboratory infrastructure, and days or weeks of data acquisition can now be accomplished in hours and used for nearly real-time analysis of community structure and activity (see Box 4). This allows repeated temporal and spatial surveys of dynamic environments with statistical replication.
NEW INSIGHTS ON THE MICROBIAL COMMUNITIES INVOLVED IN OIL BIODEGRADATION
Certain ubiquitous microbes—particularly bacteria—can biodegrade petroleum compounds, using them as high-energy substrates for growth (see Box 5). Microbial communities have been called the “first responders” to oil spills and, given appropriate conditions, can begin degrading the oil within hours or days. They can also serve as “final responders,” continuing to biodegrade oil components that remain after other natural processes wane and human interventions are complete.
Samples taken from the deep dispersed oil plume from the DWH spill revealed the magnitude of oil biodegradation in cold, deep ocean water. Natural Gulf microbiota were capable of significant biodegradation of several oil components under low-temperature and high-pressure conditions and carried out the degradations much more quickly than previously thought possible.
Insights into Oil Fate Afforded by the Deepwater Horizon Oil Spill
Prior to 2010, the majority of oil spill observations focused on the fate of oil spilled at the sea surface, but the DWH oil spill took place deep underwater, highlighting many oil behaviors and fates occurring in the ocean water column. Following are some insights gleaned from that spill.
THE IMPORTANCE OF PHOTO-OXIDATION PROCESSES IN OIL FATE
Laboratory experiments, field sampling and analysis, field observations, and modeling to demonstrate that during and after the DWH oil spill, photo-oxidation of oil compounds in the surface slick converted many compounds to reaction products and was a significant quantitative fate for these compounds. The reaction products may then undergo further degradation by microbial or other biological processes, although this has yet to be determined.
THE PHYSICS OF “DEAD OIL” VERSUS “LIVE OIL”
The oil released from the DWH spill was hot, and under the high pressure of the deep sea. This caused a large fraction of the normally gaseous components to be dissolved in the liquid oil, and the resulting mixture of oil and gas under high pressure is considered a “live oil.” Likewise, “dead oil” refers to liquid petroleum that has released enough of the dissolved gases that it is in equilibrium at standard
conditions. Significant new research has been conducted at a wide range of temperatures and pressures to understand the interactions of live oil and gas with the sea. Methods to connect equations-of-state with analyzed oil properties and models describing bubble and droplet breakup have evolved tremendously, and laboratory data have been used to calibrate and validate these new models.
THE EFFECTS OF SSDI
As well as being used to break up surface oil spills, chemical dispersants may also be used on subsea spills to make smaller oil droplets that can disperse into the water column. This method of applying dispersants to subsea spills—typically near the release source—is referred to as SSDI. By reducing the interfacial tension of the released oil and reducing the size of the oil droplets, SSDI helps dissipate the dissolved hydrocarbons into the deep sea, thereby reducing the amount of volatile compounds that reach the sea surface and the atmosphere. Further demonstration of the efficacy of SSDI remains to be determined through laboratory research and modeling exercises under different conditions of well pressure, depth, type of gas–oil mixture, type and dosage regime of dispersant, and physical oceanography conditions.
NEW MODELS FOR BUBBLE AND DROPLET SIZE
A key parameter controlling the behavior of oil in the sea is the bubble and droplet size. Experiments and analysis following the DWH spill now have yielded predictive models for droplet size under diverse spill conditions, including modeling of the effects of dispersants.
THE IMPORTANCE OF OIL–MINERAL AGGREGATES IN SUBMERGENCE AND SINKING OF MACONDO 252 OIL FROM THE DWH OIL SPILL
Although the role of oil–particle interactions in causing oil to submerge has been known for decades, light oils have been considered to be non-sinking. The presence of high concentrations of suspended sand particles in the nearshore Gulf of Mexico and their interaction
with weathered Macondo oil led to a significant proportion of oil being sedimented to the seafloor in the shallow nearshore waters. This illustrated the importance of oil–mineral aggregates in determining oil fate.
THE ROLE OF MARINE SNOW IN TRANSPORTING SPILLED OIL TO THE SEAFLOOR
Marine snow is a common natural phenomenon in the sea and occurs when a shower of organic material—including dead animals and plants, soot, and dust—falls from upper waters to the deep water (see Figure 13). When marine snow mixes with oil, it can create a sediment called marine oil snow. During and shortly after the DWH spill, an
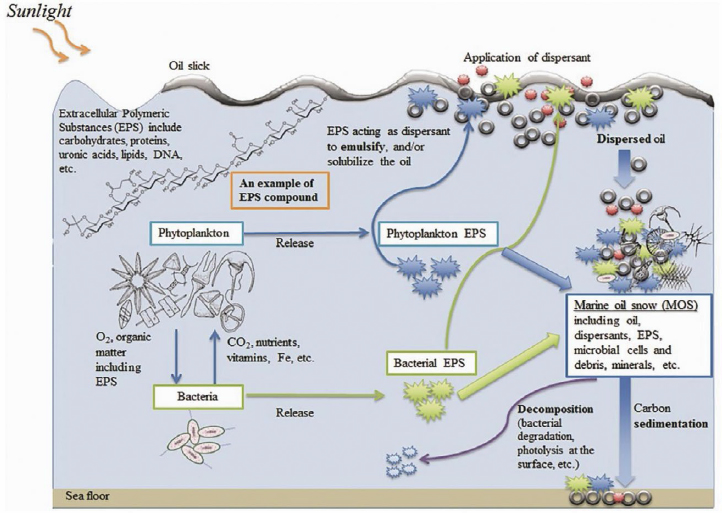
NOTE: EPS adsorbs and disperses oil, thus enhancing its solubility and bioavailability; provides a physical structure for microbes to assemble and degrade oil efficiently; and traps colloidal and particulate matter.
SOURCE: Quigg, A., U. Passow, W. C. Chin, C. Xu, S. Doyle, L. Bretherton, M. Kamalanathan, A. K. Williams, J. B. Sylvan, and Z. V. Finkel. 2016. The role of microbial exopolymers in determining the fate of oil and chemical dispersants in the ocean. Limnology and Oceanography Letters 1(1):3–26. CC BY.
unexpectedly large amount of marine oil snow formed very quickly in the oil-contaminated surface waters of the Gulf of Mexico and had a major role in the fate of the spilled oil and its transport to the seafloor. The formation of the marine oil snow, and its subsequent sedimentation and flocculant accumulation (MOSSFA), was the first such significant event documented. This process potentially has implications for both surface and subsurface oil spills elsewhere, as it has not previously been considered to be a significant fate for oil. Its global importance and the role of subsurface dispersant injection on MOSSFA remain to be quantified.
Research Gaps: Fate of Oil in the Sea
Although major progress has been made on many important topics related to the fate of oil in the sea, the following major research gaps remain.
HOW TO SCALE MODELS TO PREDICT BUBBLE AND DROPLET SIZE IN FIELD-SCALE EVENTS
With new laboratory facilities and methods, significant progress has been made in measuring droplet size distributions for oil jet breakup and dispersion of floating oil, and these data have been used to develop and test models for oil droplet size distribution. However, these data were generated in reduced-scale laboratory experiments, which cannot accurately recreate all the parameters of field-scale oil spill events. Therefore, field-scale data for oil and gas breakup and dispersion are still needed.
INTRICACIES OF PHOTO-CHEMICAL REACTIONS AND THE ROLE OF DISPERSANT ADDITION
With the renewed appreciation of photo-oxidation as a significant process affecting oil chemistry, more research is needed to focus on interactions of photo-chemical products with the physical and chemical properties of oil, its behavior in the water column and on shorelines (e.g., emulsification and adherence to mineral surfaces), and its effect on biodegradation.
MECHANISMS OF ANAEROBIC HYDROCARBON BIODEGRADATION
Aerobic biodegradation of oil components has been well studied for decades, but the range and kinetics of anaerobic hydrocarbon biodegradation, relevant to seafloor and estuarine sediments and fine-grained shoreline sediments, are less well known.
HOW MARINE OIL SNOW IS FORMED, AND ITS IMPACT ON THE ENVIRONMENT
Observations found that part of the natural biological response to the DWH oil spill resulted in vast quantities of oil-associated marine oil snow forming throughout the water. However, the mechanisms controlling the formation of marine oil snow, and its impact on the environment, remain largely unknown.
BEHAVIOR AND FATES OF NEW OR UNCONVENTIONAL OILS
Two classes of unconventional oils are due to be transported by ship in increasing volumes within the next decade: diluted bitumen (dilbit) products and low sulfur fuel oil and very low LSFOs and VLSFOs. Whereas some research has been conducted on the submergence and sinking potential of dilbit in various environments, there has not yet been a major marine spill of this two-component blend, and the fates of the diluent versus the weathered dilbit warrant further large-scale open-air experimentation to provide insight into potential behavior and fates.
DISTILLING NEW KNOWLEDGE INTO ALGORITHMS THAT CAN IMPROVE OIL SPILL FATE AND TRAJECTORY MODELS
As understanding of oil fate and transport in the sea improves, researchers will now need to convert the new insights into operational algorithms for oil fate and trajectory modeling. This will include developing new modeling algorithms, adding the new algorithms to models, and validating their predictions, ideally using in situ measurements.










