Strategies to Enhance NIH-Funded Pediatric Research: Optimizing Child Health (2026)
Chapter: 2 Background: History, Funding Landscape, and NIH Structure
2
Background: History, Funding Landscape, and NIH Structure
HISTORY OF NIH-SUPPORTED PEDIATRIC RESEARCH
The National Institutes of Health (NIH) began in 1887 as the Hygienic Laboratory for the study of bacteria (NIH Almanac, 2025). After several decades of growth, it was officially designated by Congress in 1930 as the National Institute of Health. Eighteen years later, it became the National Institutes of Health (NIH). NIH gained prominence and grew rapidly during the middle of the 20th century. Between 1947 and 1957, NIH grant awards grew 25-fold, from about $4 million annually to over $100 million. Similarly, the overall NIH budget grew from $8 million in 1947 to over $1 billion in 1966. Government funding allowed NIH to rapidly become a global leader in supporting research on human health, including the health of children.
Throughout the 20th century—and thanks in large part to NIH support for scientific inquiry—medical science advanced quickly. Penicillin, discovered as an antibacterial agent in 1928 by Alexander Fleming, became widely used and available during and after World War II. Jonas Salk developed the polio vaccine in 1954. The 1955 discovery by Joe Hin Tjio that humans had a total of 46 chromosomes led to recognition of genetic disorders, including substantial research on children’s intellectual and developmental disability created by chromosomal abnormalities and genetic mutations. In 1961 Robert Guthrie developed a simple blood test to screen for phenylketonuria in newborns, the first newborn test for a genetic disorder.
Because of these advances, the nation’s attention became focused on
understanding and improving the health of children. The National Institute of Child Health and Human Development (NICHD) was created in 1962, representing the first institute at NIH not focused on a specific disease or organ system. An early focus at NICHD was on intellectual and developmental disabilities, inspired by the Kennedy family and especially Eunice Kennedy Shriver, who had a personal dedication, given her sister Rosemary’s disability.1 Another early focus at NICHD was the consideration of biobehavioral and social science aspects of children’s health. Researchers recognized the multiple systems that impact a child’s development and thus their physical and mental health, and so pediatric health research began to consider family and community systems that might impact the healthy development of children across the country. See Appendix D for a more detailed history of pediatric health and research.
Child health in the United States experienced another concentrated flurry of activity and successful initiatives between the 1980s and 2000s. Some of the exemplary NIH-enabled research advances that improved the health of children are found in Table 2-1. For each advance summarized in Table 2-1, the NIH-supported translational process began decades before widespread clinical uptake. For example, the discovery of cell-free fetal DNA in maternal plasma in the late 1990s eventually enabled today’s noninvasive prenatal testing, and the identification of the SMN1 gene in 1995 launched a 20-year trajectory culminating in gene therapy for spinal muscular atrophy. Similar multi-decade arcs link early NIH investments to today’s front-line therapies and public health recommendations.
Initiatives like the National Children’s Study, a prospective longitudinal study to collect a broad range of environmental exposures on child health, provided important lessons for the success of future studies (IOM, 2014). Reflections on the study led to a recognition of the need for leadership buy-in, a clearer study design, more expertise in relevant scientific and survey disciplines, and stronger oversight and review structures (IOM, 2014).
FEDERAL LAWS AND POLICIES SUPPORTING AND PROTECTING CHILDREN IN HEALTH RESEARCH: A BRIEF HISTORY
Though children have been included in research for decades, they are considered a vulnerable group because they lack the capacity to provide their own informed consent and protect their own interests, putting them at increased risk of being harmed or wronged (CIOMS, 2016; Hurst, 2008). In addition, biological and behavioral differences between children and adults
___________________
1 In 2007, NICHD was renamed the Eunice Kennedy Shriver National Institute for Child Health and Human Development to commemorate her founding role.
TABLE 2-1 Pediatric Health Achievements Funded by NIH
| Innovations enabled by NIH funding | Approximate start of NIH-supported researcha | Consequent improvements to child health |
|---|---|---|
| Cell-free prenatal screening (NHGRI, 2024) | Late 1990s - discovery of cell-free fetal DNA in maternal plasma and early NIH-supported non-invasive prenatal testing development | Opportunities to diagnose and treat congenital anomalies in children before birth; invasive amniocentesis rarely necessary (Turriff et al., 2024). |
| Advances in prevention and treatment of neonatal conditions (NICHD, 2025d) | 1960s - early NIH investments in neonatal intensive care, surfactant, and antenatal steroids that laid the foundation for current survival at 22–23 weeks | Babies born as early as 22 weeks survive (Edwards et al., 2024). |
| Newborn screening for genetic diseases (NIH Office of Strategic Coordination, 2025b) | 1960s - NICHD-supported development and validation of newborn screening for PKU and other inherited conditions | Interventions for genetic diseases can be started within hours of birth (CDC, 2024). |
| New approaches to treat and cure childhood cancer (NCI, n.d., 2023; NIH Office of Strategic Coordination, 2025a) | 1950s - creation of NCI pediatric cooperative groups and early multi-agent chemotherapy trials that launched modern childhood cancer treatment | Increased 5-year survival rates for children to over 85%, with more than 80% of children expected to survive into adulthood (Hunger et al., 2012; Moke et al., 2018; NASEM, 2021). |
| Safe Sleep campaigns (NICHD, n.d.) | 1990s - NIH-supported SIDS epidemiology and sleep-position studies | 50% reduction in infant deaths from SIDS (Moon et al., 2022a, 2022b). |
| Innovations enabled by NIH funding | Approximate start of NIH-supported researcha | Consequent improvements to child health |
|---|---|---|
| Reduction in maternal transmission of HIV to their infants (Connor et al., 1994) | Late 1980s - NIH AIDS Clinical Trials Group studies of antiretrovirals in pregnancy and delivery culminating in ACTG 076 (published 1994) | Infants born to mothers with HIV are routinely born without the virus (Irshad et al., 2023; Nesheim et al., 2018). |
| New therapies for chronic diseases of childhood (e.g., cystic fibrosis) (NHLBI, 2024) | 1980s - CFTR gene discovery and NIH-supported studies of CF pathophysiology that enabled CFTR-modulator and other targeted therapies | Life expectancy for children born with cystic fibrosis was once in the single digits and is now over 60 years (CFF, n.d.). |
| New vaccines (e.g., malaria, measles, HIB, rotavirus) (NIH, 2024a; Sanaria, 2025) | 1960s - early NIH-supported development and trials of childhood vaccines such as measles, followed by HIB conjugate, rotavirus, and malaria vaccines over subsequent decades | >99% decrease in measles, HIB, and rotavirus in vaccinated children (Dempsey and Zimet, 2015; Esposito et al., 2014; Kao et al., 2014; Khabbaz et al., 2014; Temoka, 2013). |
| Development of coalitions to study diseases impacting children (e.g., childhood cancer, autism) (Children’s Oncology Group, n.d.; NICHD, 2022) | 1950s–2000s - from early NCI cooperative pediatric oncology groups to later NIH-supported rare disease research networks such as CARRA and similar consortia | Children across the United States benefit from specialty centers through early diagnosis, optimal interventions/treatments, and education. More children across the United States are appropriately diagnosed and receiving timely care. |
| Gene therapy for spinal muscular atrophy type 1 (NINDS, 2024) | Mid-1990s - discovery of the SMN1 gene and start of NIH-supported SMA biology and gene-targeted therapy research, leading to AAV gene therapy and antisense drugs | Life-saving treatment that improves survival, preserves motor function, and reduces lifelong disability (Mendell et al., 2017). |
| Innovations enabled by NIH funding | Approximate start of NIH-supported researcha | Consequent improvements to child health |
|---|---|---|
| Cryotherapy and laser surgery for retinopathy of prematurity (Early Treatment for Retinopathy of Prematurity Cooperative Group, 2003) | Mid-1980s - NEI-funded CRYO-ROP trial launched in 1986, followed by NIH-supported laser photocoagulation trials in the 1990s | Provided an intervention for a disease that previously had no treatment. Reduced rates of childhood blindness and visual impairment in premature infants (Chow et al., 2022; Cryotherapy for Retinopathy of Prematurity Cooperative Group and National Eye Institute, 1988; Lumsden and Urv, 2023). |
| Whole body cooling for hypoxic–ischemic encephalopathy (Shankaran et al., 2005) | Late 1990s - planning and initiation of NICHD Neonatal Research Network whole-body hypothermia trials, with first major results published in 2005 | Reduced risk of death and severe neurodevelopmental disability. Improves survival and cognitive outcomes in affected infants (Shankaran et al., 2012). |
| The development and clinical implementation of clinical whole-genome sequencing (NCATS, 2024) | Early 2010s - NIH-funded pilots of rapid clinical whole-genome sequencing (e.g., NSIGHT and NICU rWGS studies) in critically ill infants and newborns | Dramatically improved clinicians’ ability to diagnose children with rare diseases in both intensive care and outpatient settings (Brlek et al., 2024). |
| Prenatal surgery for myelomeningocele | Early 2000s - NICHD-funded MOMS trial of prenatal vs. postnatal repair, launched in 2003 with NEJM results in 2011 | Reduced the need for shunting and improved motor outcomes in young children (Adzick et al., 2011; Houtrow et al., 2020). |
| Antenatal corticosteroid therapy (NICHD, 2025g) | Early 1970s - first randomized trials of antenatal corticosteroids for fetal lung maturation, followed by NICHD Neonatal Research Network trials and implementation studies in the 1990s | Decreases neonatal morbidity and mortality, with substantial savings in health care costs. The use of antenatal corticosteroids for fetal maturation is a rare example of a technology that yields substantial cost savings in addition to improving health (Gilstrap et al., 1995; McGoldrick et al., 2020). |
| Innovations enabled by NIH funding | Approximate start of NIH-supported researcha | Consequent improvements to child health |
|---|---|---|
| Very early antiretroviral therapy for infants with HIV | Early 2010s - NIH-supported studies of combination ART initiated within hours/days of birth (e.g., “Mississippi baby” case and subsequent early-ART cohorts) | Advances have made it possible to achieve viral remission in infants (Persaud et al., 2024). |
a Decades are approximate and refer to the period when NIH-supported work that was clearly on the translational path to the listed advance began (e.g., gene discovery, first randomized control trials, or launch of NIH cooperative trials), not necessarily the very first experiment performed in each area.
NOTE: AAV = adeno-associated virus; ART = antiretroviral therapy; CARRA = Childhood Arthritis and Rheumatology Research Alliance; CF = cystic fibrosis; CFTR = cystic fibrosis transmembrane regulator; CRYO-ROP = Multicenter Trial of Cryotherapy for Retinopathy of Prematurity; HIB = Haemophilus influenzae type B; NCI = National Cancer Institute; NEI = National Eye Institute; NEJM = New England Journal of Medicine; NICHD = Eunice Kennedy Shriver National Institute of Child Health and Human Development; NICU = neonatal intensive care unit; NIH = National Institutes of Health; PKU = phenylketonuria; rWGS =rapid whole-genome sequencing; SIDS = sudden infant death syndrome; SMA = spinal muscular atrophy.
may make children more likely to suffer harm from the use of experimental interventions, unless care is taken to protect their safety and well-being.
Without established systems of oversight and ethical frameworks for research involving human subjects, questionable and unethical conduct often occurred in early research studies with children. Historical examples of ethically problematic research include cases in which children were exposed to considerable risk, often without informed permission from their parents or caregivers or sufficient scientific justification for doing so. In 1796, Edward Jenner proved the concept of vaccination by exposing individuals to cowpox and subsequently inoculating them with smallpox. Among these individuals were his own 1-year-old son and James Phipps, the son of his gardener. While the importance of the study was clear and ultimately provided Phipps with protection against smallpox, it is unclear whether his parents understood the risks or gave their permission for James to participate in the experiment (Zildzic and Zunic, 2023). In the 1930s, a researcher conducted a study with children which has been referred to as the “monster” study, in which the goal was to induce children to stutter or exacerbate existing stuttering (Silverman, 1988). Horrific and sometimes fatal studies involving children, including studies with young twins, were conducted by the Nazis during World War II. In response to these atrocities, the first international ethical guidance for
research was developed, referred to as the Nuremberg Code, which required voluntary informed consent from all research participants, thereby ruling out the participation of children (Diekema, 2006; Holocaust Memorial Museum, n.d.). In the 1950s and 1960s, controversial research was conducted at the Willowbrook State School in which healthy children with cognitive disabilities were deliberately infected with hepatitis. The principal investigator said that the study was done to address an existing widespread hepatitis infection in the institution and noted that inoculation led to mild disease (Krugman, 1986). Others questioned the ethics of this study, particularly related to the level of risk, whether it was necessary to do the study with institutionalized children with disabilities, and whether parents were freely able to decide to enroll or exclude their children (Beecher, 1966; Diamond, 1973).
In response to significant public concern about the Willowbrook hepatitis study and similar research involving children, Congress passed the National Research Act in 1974. This act created the National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research. The National Commission wrote the highly influential Belmont Report, which laid out principles for the ethical conduct of research for all participants, including a special focus on whether research with children could be ethically acceptable. The National Commission ultimately found that pediatric research is important and necessary to promote child health and well-being and that specific safeguards could help ensure that research with children is conducted ethically. The National Commission’s work led ultimately to new U.S. federal regulations, which created institutional review boards to oversee research and provided a framework for the protection of children based on risk/benefit considerations and parental permission requirements to protect children and respect parental authority (Shah, 2021). The National Commission’s work and subsequent regulations and guidelines led to the modern system of research ethics oversight that serves as a bulwark against many types of unethical research with children.
Following the National Commission’s determination that pediatric health research is important and necessary to improve child health, the U.S. Congress took steps to encourage and incentivize pediatric research. In 2002 the Best Pharmaceuticals for Children Act directed NIH to prioritize pediatric trials on medications and devices being used off-label in children. It also gave pharmaceutical companies that added pediatric labeling to their products an extension of patent exclusivity (NASEM, 2012). In 2003 the Pediatric Research Equity Act required pediatric assessments of new drugs and biologics with new active ingredients, indications, dosages, and routes of administration (Bourgeois et al., 2012). The Gabriella Miller Kids First Research Act, passed in 2013, required the director of NIH to allocate $12.6 million annually from the 10-year Pediatric Research for fiscal year (FY) 2014–FY 2024 Initiative Fund to institutes and centers (ICs) to
fund grants for pediatric research on childhood cancer and structural birth defects.2 The Gabriella Miller Kids First Research Act extended funding until FY 2028.3
NIH implemented its Inclusion Across the Lifespan policy in 2019 to ensure that individuals of all ages, including children and older adults, are appropriately represented in NIH-funded research that includes human participants. This policy mandates that researchers include participants across the lifespan unless there are scientific or ethical reasons for exclusion. Applicants must provide a rationale for any age-related exclusions, and progress reports are required to include data on participant age at enrollment (NIH, 2025b). By requiring the inclusion of children in research, the policy is intended to generate data that can inform better health care decisions for pediatric populations. This approach acknowledges that many adult health conditions have roots in early life and that early interventions can have long-term health benefits.
Despite these changes in federal law and NIH policy, however, many barriers remain for pediatric research that make it difficult to address the national pediatric health crisis (Forrest et al., 2025). A deeper discussion of the intersection of ethical considerations and pediatric health research along with the committee’s conclusions and recommendations can be found in Chapter 5.
THE ORGANIZATION OF NIH AND ITS CAPACITY AND SUPPORT FOR PEDIATRIC HEALTH RESEARCH
The organizational structure of NIH includes central leadership from the Office of the Director (OD) and 27 semi-autonomous ICs (see Figure 2-1). Twenty-two ICs fund intramural research (NIH Intramural Research Program, 2022a),4 and 24 ICs5 support extramural research. In addition, NIH supports a variety of strategic initiatives that cut across diseases, systems, and populations (NIH, n.d.).
Each IC has its own research agenda, often focusing on specific diseases or body systems (NIH, 2024b). Table 2-2 lists the ICs, the year each was established, and their primary focus areas.
All of the ICs support pediatric research, although the absolute dollar amount as well as the proportion of each budget varies widely among them.
___________________
2 Gabriella Miller Kids First Research Act, 2014, H.R.2019, 113th Congress (2013–2014).
3 Gabriella Miller Kids First Research Act 2.0, 2025, H.R.3391, 118th Congress (2023–2024).
4 Twenty-two ICs provide funding for intramural research while the Clinical Center serves to house intramural research (see Figure 2-1).
5 Twenty-four of the 27 ICs provide research funding, and the National Institute of General Medical Sciences, one of the funding ICs, exclusively funds basic research not conducted at the Clinical Center.
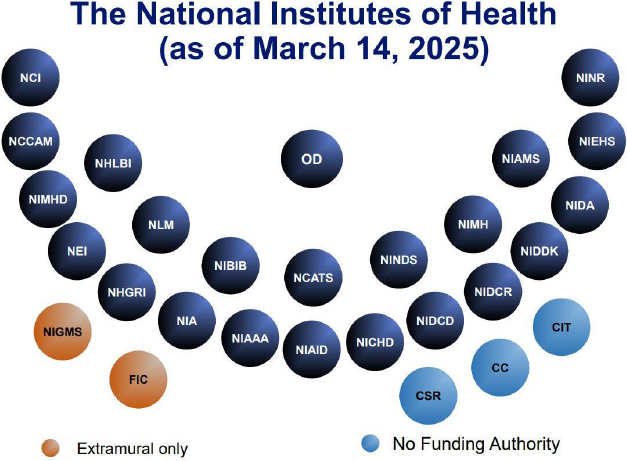
NOTE: CC = Clinical Center; CIT = Center for Information Technology; CSR = Center for Scientific Review; FIC = Fogarty International Center; MIAMS = National Institute of Arthritis and Musculoskeletal and Skin Diseases; NCATS = National Center for Advancing Translational Sciences; NCCIH = National Center for Complementary and Integrative Health; NCI = National Cancer Institute; NEI = National Eye Institute; NHGRI = National Human Genome Research Institute; NHLBI = National Heart, Lung, and Blood Institute; NIA = National Institute on Aging; NIAAA = National Institute on Alcohol Abuse and Alcoholism; NIAID = National Institute of Allergy and Infectious Diseases; NIBIB = National Institute of Biomedical Imaging and Bioengineering; NICHD = Eunice Kennedy Shriver National Institute of Child Health and Human Development; NIDA = National Institute on Drug Abuse; NIDCD = National Institute on Deafness and Other Communication Disorders; NIDCR = National Institute of Dental and Craniofacial Research; NIDDK = National Institute of Diabetes and Digestive and Kidney Diseases; NIEHS = National Institute of Environmental Health Sciences; NIGMS = National Institute of General Medical Sciences; NIMHD = National Institute on Minority Health and Health Disparities; NINDS = National Institute of Neurological Disorders and Stroke; NINR = National Institute of Nursing Research; NLM = National Library of Medicine; OD = Office of the Director.
SOURCE: Presentation by Diana Bianchi, M.D., director of the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), at the Open Session: Committee on Strategies to Enhance Pediatric Health Research Funded by NIH, hosted by the National Academies of Sciences, Engineering, and Medicine on March 14, 2025.
TABLE 2-2 The 27 NIH ICs, Year of Establishment, and Primary Focus Area, as of March 21, 2025
| Institute/Center | Year Established | Primary Focus Area |
|---|---|---|
| National Cancer Institute (NCI) | 1937 | Cancer research and training |
| Center for Scientific Review (CSR)a,b | 1946 | Peer review of NIH grant applications |
| National Heart, Lung, and Blood Institute (NHLBI) | 1948 | Heart, lung, and blood diseases |
| National Institute of Allergy and Infectious Diseases (NIAID) | 1948 | Infectious, immunologic, and allergic diseases |
| National Institute of Dental and Craniofacial Research (NIDCR) | 1948 | Dental, oral, and craniofacial health |
| National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) | 1948 | Diabetes, metabolic, digestive, and kidney diseases |
| National Institute of Mental Health (NIMH) | 1949 | Mental illnesses |
| National Institute of Neurological Disorders and Stroke (NINDS) | 1950 | Neurological disorders |
| NIH Clinical Center (CC)b | 1953 | Clinical research facility and patient care |
| National Library of Medicine (NLM) | 1956 | Biomedical information resources |
| Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) | 1962 | Child health, development, and reproductive health |
| National Institute of General Medical Sciences (NIGMS)a | 1962 | Basic biomedical research |
| Center for Information Technology (CIT)a, b | 1964 | Biomedical computing and IT |
| Fogarty International Center (FIC)a | 1968 | Global health research and training |
| National Eye Institute (NEI) | 1968 | Eye diseases and vision research |
| National Institute of Environmental Health Sciences (NIEHS) | 1969 | Environmental health sciences |
| National Institute on Alcohol Abuse and Alcoholism (NIAAA) | 1970 | Alcohol-related research |
| National Institute on Aging (NIA) | 1974 | Aging and age-related diseases |
| National Institute on Drug Abuse (NIDA) | 1974 | Drug abuse and addiction |
| Institute/Center | Year Established | Primary Focus Area |
|---|---|---|
| National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS) | 1986 | Arthritis and musculoskeletal and skin diseases |
| National Institute of Nursing Research (NINR) | 1986 | Scientific basis of nursing practice |
| National Institute on Deafness and Other Communication Disorders (NIDCD) | 1988 | Communication disorders including hearing and speech |
| National Human Genome Research Institute (NHGRI) | 1989 | Human genome structure and function |
| National Center for Complementary and Integrative Health (NCCIH) | 1999 | Complementary and integrative health practices |
| National Institute of Biomedical Imaging and Bioengineering (NIBIB) | 2000 | Biomedical imaging and bioengineering |
| National Institute on Minority Health and Health Disparities (NIMHD) | 2010 | Minority health and health disparities |
| National Center for Advancing Translational Sciences (NCATS) | 2011 | Translational science and therapeutic development |
a ICs that do not fund intramural research.
b ICs that do not fund extramural research.
NOTE: ICs = institutes and centers; IT = information technology; NIH = National Institutes of Health.
SOURCE: NIH (2024b).
NICHD allocates the largest portion of its budget to pediatric research (historically around 60 percent) and is the largest contributor to NIH’s pediatric portfolio (17 percent for FY 2024 and approximately 18 percent in FY 2021). Other ICs, including several with budgets substantially larger than NICHD, such as the National Institute of Allergy and Infectious Diseases (NIAID), the National Cancer Institute (NCI), the National Institute of Mental Health, and the National Heart, Lung, and Blood Institute (NHLBI), also allocate significant funds to pediatric health research (see Figure 3-7) (NICHD, 2025a,b,c,e,f). For example, the NCI funds extensive research on pediatric cancers, including initiatives to mitigate shortages of pediatric cancer drugs as part of the Cancer Moonshot initiative; the NHLBI supports studies on congenital heart defects and blood disorders, such as sickle cell disease, aiming to enhance diagnosis and treatment in pediatric populations; and the NIAID funds research on pediatric infectious diseases, including studies on vaccines and treatments for conditions such as pediatric HIV/AIDS. There is robust support of pediatric research across
most ICs, with all the ICs funding some research categorized as “pediatric,” according to the NIH Research, Condition, and Disease Categorization system. Chapter 3 provides a more in-depth analysis of pediatric health research funding across the ICs.
The NIH Clinical Center: A Critical Resource for Advancing Health Across the Life Course
The NIH Clinical Center (CC) is a vibrant hospital system hosting more than 2,000 health care professionals and investigators, who engage in scientific research to advance human health (NIH, 2021; Sekar, 2025). Opened in 1953, the CC’s primary innovation was the co-location of patient care and research laboratories, thus facilitating research intended to advance diagnosis, disease characterization, and treatment effectiveness (NIH Clinical Center, n.d.-a). Originally conceived as an inpatient facility, the CC added an outpatient infrastructure in the 1980s (NIH Clinical Center, n.d.-b). For more information about the CC and its evolution, please see Appendix D.
The CC’s Vision for Pediatric Research
The CC is uniquely positioned to conduct research in several key areas of pediatric science due to its specialized infrastructure, collaborative networks, and focus on underserved populations, making the CC a key component of any NIH strategy to advance innovative pediatric research, which is discussed in detail in Chapter 3.
The NIH Pediatric Research Consortium
The NIH Pediatric Research Consortium (NPeRC) was established in 2018 to enhance collaboration and coordination in pediatric research across NIH’s ICs (NICHD, 2025e). The NPeRC membership includes representatives from 25 ICs, five offices, and two research programs (All of Us and Environmental Influences on Child Health Outcomes; both discussed in depth in Chapter 4). NPeRC focuses on harmonizing pediatric research activities, identifying gaps and opportunities in the pediatric research portfolio, and taking advantage of existing resources and expertise. It currently operates without dedicated funding and does not directly support research projects or initiatives. The consortium convenes several times a year to discuss scientific opportunities and potential new areas of collaboration, including efforts to enhance research training for the next generation of pediatricians. Among its accomplishments, NPeRC has facilitated discussions on common areas of interest and needs in pediatric research, which have contributed to the development of a child health agenda within NIH.
NPeRC has also collaborated with the Helping to End Addiction Long-term Initiative to address pediatric pain research and has engaged in efforts to bolster research on improving the transition from pediatric to adult health care. Additional findings, conclusions, and recommendations about the NPeRC program can be found in Chapter 4.
Intramural Pediatric Research at NIH
NIH’s Intramural Research Program (IRP) funds biomedical research conducted by NIH-employed investigators within its own laboratories and the CC (Cryotherapy for Retinopathy of Prematurity Cooperative Group and National Eye Institute, 1988). With around 1,200 principal investigators and over 4,000 postdoctoral fellows, the IRP is unique: rather than relying on competitive grants like the extramural program, it receives direct appropriations from Congress, making up approximately 11 percent of the NIH budget, which in recent years has translated to around $4 billion annually (NIH, 2025a; NIH Intramural Research Program, 2022c). This internal funding mechanism makes it possible to carry out long-term, high-risk/high-reward projects, which are often resource-intensive (such as longitudinal or population-based studies) and which would be challenging to support through standard extramural grant mechanisms.
To ensure accountability and scientific excellence, IRP investigators undergo rigorous review cycles approximately every 4 years. These reviews are conducted by external boards of scientific counselors (HELP, 2024; NIH Intramural Research Program, 2022d). Reviews include in-person site visits and focus on both past accomplishments and future research plans. Evaluation criteria include three factors: importance of the research, rigor and feasibility, and expertise and resources. Final recommendations are presented to the NIH deputy director for intramural research, who discusses it with the relevant scientific director. When decisions on funding levels, staffing, and promotions are finalized, they are provided to the relevant IC council.
Ten percent of intramurally funded projects focus on pediatrics, based on available data (see the data analyses in Chapter 3). Some recent intramural research includes identifying new therapies for pediatric cancers, understanding the relationship between maternal folic acid levels and childhood asthma risk, and creating robotic exoskeletons to improve walking posture among children with cerebral palsy (NIH Intramural Research Program, 2016, 2020, 2022b).
Extramural NIH-Supported Pediatric Research
Extramural research applications go through a rigorous process to receive funding from NIH. Once a research application has been submitted,
it first undergoes an administrative review to confirm that the application complies with submission requirements and eligibility criteria. Next, the application is assigned to a study section, a panel of experts in relevant scientific fields who evaluate the scientific merit of the proposal. During the peer review, reviewers provide scores and written critiques focusing on several criteria, including significance, innovation, and approach. The application is assigned a percentile score that ranks its overall scientific quality relative to other applications in the same review cycle. In parallel, the application is assigned to the relevant NIH IC, which in turn assigns a program officer who manages a relevant portfolio of research. That program officer does not play any direct role in the peer review process to maximize integrity and independence of review but is available to an applicant following completion of peer review to help interpret the review results. Program staff consider program relevance, strategic priorities, and budget considerations when making funding recommendations. An application considered for funding advances to the IC’s advisory council or board, where it undergoes a second level of review (NIH Grants & Funding, 2025). The advisory board/council considers the IC’s goals and needs and advises the IC director concerning funding decisions. Based on committee discussions with IC leaders, most ICs prioritize those grants with the most meritorious scores, irrespective of pre-conceived “targets” for types of research (e.g., ICs do not currently have pre-determined target proportions of “pediatric”-focused grants to fund) (NIH Office of Extramural Research, 2024).
Investigator-initiated awards are a core component of the NIH funding portfolio. These grants are designed to support researchers who propose their own innovative ideas and hypotheses without predefined project constraints (NIGMS, 2025; NIH Grants & Funding, n.d.). These awards provide flexibility for researchers to explore novel scientific questions, develop new methodologies, or test emerging theories aligned with their expertise and interests. Typically awarded through R programs (research project grants such as R01 or R21), these grants are highly competitive and require detailed proposals demonstrating scientific rigor, significance, and innovation. When specific funds have been set aside for particular research topics (e.g., pediatric asthma), specific requests for application are developed, with submitted grants going through a process similar to that of investigator-initiated awards. Similarly, notices of special interest (NOSIs) allow ICs to solicit research applications that focus on particular topics (NIAID, n.d.).6 For NOSIs, no specific funds are prospectively set aside for funded applications. In addition to NIH ICs, the OD has numerous funding opportunities known
___________________
6 Going forward, NIH will no longer use NOSIs. Instead, NIH will use Highlighted Topics, which are not replacing NOSIs but will serve a similar purpose. More information is available at https://grants.nih.gov/funding/find-a-fit-for-your-research/highlighted-topics.
as NIH Director’s awards (e.g., Early Independence Award, Transformative Research Award, New Innovator Award, and Pioneer Award) that can be used to fund investigator-initiated pediatric research (NIH Office of Strategic Coordination, 2024a, 2024b, 2024c, 2024d).
Research Center Grants Supporting Pediatrics
NIH-supported research center grants have played an important role in advancing pediatric research by promoting multidisciplinary collaboration, resourcing shared infrastructure, and supporting long-term scientific programs focused on child health. These center grants—such as the P30 (Center Core), P50 (Specialized Center), and U54 (Specialized Collaborative Center)—have enabled institutions to bring together basic, clinical, and translational scientists around shared thematic priorities in pediatrics, including rare diseases, developmental disorders, pediatric cancers, and environmental influences on child health. By supporting centralized cores, career development opportunities, and pilot project funding, NIH center grants nurture innovation and sustain research ecosystems that might otherwise be difficult to maintain through individual investigator-initiated grants alone. Many of these centers have also emphasized community engagement and training of the next generation of pediatric investigators, thereby ensuring that the infrastructure not only generates high-impact science but also builds capacity for the future of child health research. Some exemplary centers and their contributions to child health are listed in Table 2-3. Additional information about how ICs and the OD prioritize and fund research centers to promote child health can be found in Chapter 4.
THE BROADER LANDSCAPE: OTHER PEDIATRIC RESEARCH FUNDERS
The U.S. pediatric research funding landscape is complex. Beyond NIH, there is an array of other federal, state, and private funders. Some pediatric research involves collaborations between NIH ICs and other federal agencies, such as the Centers for Disease Control and Prevention, Agency for Healthcare Research and Quality, Health Resources and Services Administration, Substance Abuse and Mental Health Services Administration, Food and Drug Administration, Environmental Protection Agency, National Institute of Justice, National Science Foundation, Department of Defense, Department of Transportation, Department of Education, the Indian Health Service, and other agencies within the Department of Health and Human Services. State and local governments as well as private funders such as health systems, foundations, industry, and voluntary health associations provide critical supplementary support for pediatric health research. NIH,
TABLE 2-3 NIH Research Center Awards
| Exemplary Center | Focus | Highlighted Contribution to Child Health |
|---|---|---|
| Intellectual and Developmental Disabilities Research Centers (Kennedy Krieger, 2025) | Intellectual and developmental disabilities research infrastructure | Improved diagnosis, support, and clinical care of children with neurodevelopmental disorders |
| Child Health Research Career Development Awards (CHRCDAs) (NICHD, 2023) | Career development for pediatric researchers | Trained pediatric physician-scientists who drive innovations in child health research |
| Pediatric Clinical and Translational Research Centers (UPMC CHP, 2025) | Translation of basic research into clinical applications in children | Enabled early-phase trials and translational studies tailored to pediatric populations |
| Pediatric Environmental Health Specialty Units (PEHSU, 2025) | Focus on addressing environmental health issues affecting children | Provided expertise to prevent and respond to environmental exposures affecting children |
| Pediatric HIV/AIDS Cohort Study (PHACS, 2025) | Research on long-term effects of HIV and its treatment in children | Improved long-term care and outcomes for children affected by perinatal HIV and its treatments |
| Pediatric Heart Network (Pediatric Heart Network, n.d.) | Multi-center studies on congenital and acquired heart disease in children | Developed standards of care for diagnosis, treatment, and outcomes in children with congenital and acquired heart disease |
NOTE: NIH = National Institutes of Health.
however, remains the primary funder of pediatric research through its extramural funding, collaborations, and extensive IRPs.
Changes in the federal budget and policies may influence the future funding environment for biomedical and public health research, including pediatric health research. Congressionally approved cuts to Medicaid will likely have profound effects on pediatric research. More than half of the patients treated in clinical programs in academic pediatric departments and children’s hospitals are covered by Medicaid or the Children’s Health Insurance Program (CHIP), and the approved reductions in reimbursement will represent the loss of tens to hundreds of millions of dollars for these institutions. Thus, the ability of these institutions to support pediatric research
at the level they have done in the past will be severely compromised. The contributions of these other funders cannot replace the sustained, targeted NIH funding for pediatric health that has led to many significant breakthroughs in the past and is essential for ensuring that the United States remains a leader in pediatric health research into the future.
REFERENCES
Adzick, N. S., E. A. Thom, C. Y. Spong, J. W. Brock, 3rd, P. K. Burrows, M. P. Johnson, L. J. Howell, J. A. Farrell, M. E. Dabrowiak, L. N. Sutton, N. Gupta, N. B. Tulipan, M. E. D’Alton, D. L. Farmer, and MOMS Investigators. 2011. A randomized trial of prenatal versus postnatal repair of myelomeningocele. New England Journal of Medicine 364(11):993–1004.
Beecher, H. K. 1966. Ethics and clinical research. New England Journal of Medicine 274(24):1354–1360.
Bourgeois, F. T., S. Murthy, C. Pinto, K. L. Olson, J. P. A. Ioannidis, and K. D. Mandl. 2012. Pediatric versus adult drug trials for conditions with high pediatric disease burden. Pediatrics 130(2):285–292.
Brlek, P., L. Bulic, M. Bracic, P. Projic, V. Skaro, N. Shah, P. Shah, and D. Primorac. 2024. Implementing whole genome sequencing (WGS) in clinical practice: Advantages, challenges, and future perspectives. Cells 13(6):504.
CDC (Centers for Disease Control and Prevention). 2024. About newborn screening. https://www.cdc.gov/newborn-screening/about/index.html (accessed April 4, 2025).
CFF (Cystic Fibrosis Foundation). n.d. Understanding changes in life expectancy. https://www.cff.org/managing-cf/understanding-changes-life-expectancy (accessed October 30, 2025).
Children’s Oncology Group. n.d. Home. https://www.childrensoncologygroup.org/ (accessed December 9, 2025).
Chow, S. C., P. Y. Lam, W. C. Lam, and N. S. K. Fung. 2022. The role of anti-vascular endothelial growth factor in treatment of retinopathy of prematurity—A current review. Eye (London) 36(8):1532–1545.
CIOMS (Council for International Organizations of Medical Sciences). 2016. International ethical guidelines for health-related research involving humans. Geneva: Council for International Organizations of Medical Sciences.
Connor, E. M., R. S. Sperling, R. Gelber, P. Kiselev, G. Scott, M. J. O’Sullivan, R. VanDyke, M. Bey, W. Shearer, R. L. Jacobson, E. Jimenez, E. O’Neill, B. Bazin, J.-F. Delfraissy, M. Culnane, R. Coombs, M. Elkins, J. Moye, P. Stratton, and J. Balsley, for the Pediatric AIDS Clinical Trials Group Protocol 076 Study Group. 1994. Reduction of maternal–infant transmission of human immunodeficiency virus type 1 with zidovudine treatment. New England Journal of Medicine 331(18):1173–1180.
Cryotherapy for Retinopathy of Prematurity Cooperative Group and National Eye Institute. 1988. Multicenter trial of cryotherapy for retinopathy of prematurity. Preliminary results. Archives of Ophthalmology 106(4):471–479.
Dempsey, A. F., and G. D. Zimet. 2015. Interventions to improve adolescent vaccination: What may work and what still needs to be tested. Vaccine 33(Suppl 4):D106–D113.
Diamond, E. F. 1973. The Willowbrook experiments. The Linacre Quarterly 40(2):133–137.
Diekema, D. S. 2006. Conducting ethical research in pediatrics: A brief historical overview and review of pediatric regulations. The Journal of Pediatrics 149(1 Suppl):S3–S11.
Early Treatment for Retinopathy of Prematurity Cooperative Group. 2003. Revised indications for the treatment of retinopathy of prematurity: Results of the Early Treatment for Retinopathy of Prematurity randomized trial. Archives of Ophthalmology 121(12):1684–1694.
Edwards, E. M., D. E. Y. Ehret, R. F. Soll, and J. D. Horbar. 2024. Survival of infants born at 22 to 25 weeks’ gestation receiving care in the NICU: 2020–2022. Pediatrics 154(4):e2024065963.
Esposito, S., P. Durando, S. Bosis, F. Ansaldi, C. Tagliabue, G. Icardi, and the ESCMID Vaccine Study Group. 2014. Vaccine-preventable diseases: From paediatric to adult targets. European Journal of Internal Medicine 25(3):203–212.
Forrest, C. B., L. J. Koenigsberg, F. Eddy Harvey, M. G. Maltenfort, and N. Halfon. 2025. Trends in U.S. children’s mortality, chronic conditions, obesity, functional status, and symptoms. JAMA 334(6):509–516.
Gilstrap, L. C., R. Christensen, W. H. Clewell, M. E. D’Alton, E. C. Davidson, Jr, M. B. Escobedo, D. K. Gjerdingen, J. Goddard-Finegold, R. L. Goldenberg, D. A. Grimes, T. N. Hansen, R. E. Kauffman, E. B. Keeler, W. Oh, E. J. Susman, M. G. Vogel, M. E. Avery, P. L. Ballard, R. A. Ballard, P. Crowley, T. Garite, G. D. V. Hankins, A. H. Jobe, J. G. Koppe, J. E. Maher, I. R. Merkatz, S. Shankaran, K. N. Simpson, J. C. Sinclair, T. A. Slotkin, H. W. Taeusch, Jr, L. L. Wright, D. Alexander, M. A. Berberich, M. Bracken, L. Cooper, L. Culpepper, J. M. Elliott, J. H. Ferguson, F. Frigoletto, D. B. Gail, W. H. Hall, M. D. Jones, Jr, B. Medoff-Cooper, G. B. Merenstein, J. M. Whalen, C. Lenfant, and A. S. Hinshaw. 1995. Effect of corticosteroids for fetal maturation on perinatal outcomes: NIH consensus development panel on the effect of corticosteroids for fetal maturation on perinatal outcomes. JAMA 273(5):413–418.
HELP (Senate Committee on Health, Education, Labor, and Pensions). 2024. NIH in the 21st century: Ensuring transparency and American biomedical leadership. https://www.help.senate.gov/imo/media/doc/nih_modernization_5924pdf.pdf (accessed October 30, 2025).
Holocaust Memorial Museum. n.d. The Doctors Trial: The medical case of the subsequent Nuremberg proceedings. https://encyclopedia.ushmm.org/content/en/article/the-doctors-trial-the-medical-case-of-the-subsequent-nuremberg-proceedings (accessed April 24, 2025).
Houtrow, A. J., E. A. Thom, J. M. Fletcher, P. K. Burrows, N. S. Adzick, N. H. Thomas, J. W. Brock, 3rd, T. Cooper, H. Lee, L. Bilaniuk, O. A. Glenn, S. Pruthi, C. MacPherson, D. L. Farmer, M. P. Johnson, L. J. Howell, N. Gupta, and W. O. Walker. 2020. Prenatal repair of myelomeningocele and school-age functional outcomes. Pediatrics 145(2):e20191544.
Hunger, S. P., X. Lu, M. Devidas, B. M. Camitta, P. S. Gaynon, N. J. Winick, G. H. Reaman, and W. L. Carroll. 2012. Improved survival for children and adolescents with acute lymphoblastic leukemia between 1990 and 2005: A report from the Children’s Oncology Group. Journal of Clinical Oncology 30(14):1663–1669.
Hurst, S. A. 2008. Vulnerability in research and health care; describing the elephant in the room? Bioethics 22(4):191–202.
IOM (Institute of Medicine). 2014. The National Children’s Study 2014. Washington, DC: The National Academies Press.
Irshad, U., H. Mahdy, and T. Tonismae. 2023. HIV in pregnancy. Treasure Island, FL: StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK558972/ (accessed October 30, 2025).
Kao, C. M., R. J. Schneyer, and J. A. Bocchini, Jr. 2014. Child and adolescent immunizations: Selected review of recent U.S. recommendations and literature. Current Opinion in Pediatrics 26(3):383–395.
Kennedy Krieger. 2025. Intellectual and Developmental Disabilities Research Center (ID-DRC). https://www.kennedykrieger.org/research/centers-labs-cores/intellectual-and-developmental-disabilities-research-center-iddrc (accessed August 14, 2025).
Khabbaz, R. F., R. R. Moseley, R. J. Steiner, A. M. Levitt, and B. P. Bell. 2014. Challenges of infectious diseases in the USA. Lancet 384(9937):53–63.
Krugman, S. 1986. The Willowbrook hepatitis studies revisited: Ethical aspects. Reviews of Infectious Diseases 8(1):157–162.
Lumsden, J. M., and T. K. Urv. 2023. The Rare Diseases Clinical Research Network: A model for clinical trial readiness. Therapeutic Advances in Rare Disease 4:26330040231219272.
McGoldrick, E., F. Stewart, R. Parker, and S. R. Dalziel. 2020. Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database of Systematic Reviews 12(12):CD004454.
Mendell, J. R., S. Al-Zaidy, R. Shell, W. D. Arnold, L. R. Rodino-Klapac, T. W. Prior, L. Lowes, L. Alfano, K. Berry, K. Church, J. T. Kissel, S. Nagendran, J. L’Italien, D. M. Sproule, C. Wells, J. A. Cardenas, M. D. Heitzer, A. Kaspar, S. Corcoran, L. Braun, S. Likhite, C. Miranda, K. Meyer, K. D. Foust, A. H. M. Burghes, and B. K. Kaspar. 2017. Single-dose gene-replacement therapy for spinal muscular atrophy. New England Journal of Medicine 377(18):1713–1722.
Moke, D. J., M. J. Oberley, D. Bhojwani, C. Parekh, and E. Orgel. 2018. Association of clinical trial enrollment and survival using contemporary therapy for pediatric acute lymphoblastic leukemia. Pediatric Blood & Cancer 65(2):10.1002/pbc.26788.
Moon, R. Y., R. F. Carlin, I. Hand, and the Task Force on Sudden Infant Death Syndrome and the Committee on Fetus and Newborn. 2022a. Evidence base for 2022 updated recommendations for a safe infant sleeping environment to reduce the risk of sleep-related infant deaths. Pediatrics 150(1):e2022057991.
Moon, R. Y., R. F. Carlin, I. Hand, and the Task Force on Sudden Infant Death Syndrome and the Committee on Fetus and Newborn. 2022b. Sleep-related infant deaths: Updated 2022 recommendations for reducing infant deaths in the sleep environment. Pediatrics 150(1):e2022057990.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2012. Safe and effective medicines for children: Pediatric studies conducted under the Best Pharmaceuticals for Children Act and the Pediatric Research Equity Act. Washington, DC: The National Academies Press.
NASEM. 2021. Childhood cancer and functional impacts across the care continuum. Washington, DC: The National Academies Press.
NCATS (National Center for Advancing Translational Sciences). 2024. Current RDCRN consortia. https://ncats.nih.gov/research/research-activities/rdcrn/consortia (accessed April 29, 2025).
NCI (National Cancer Institute). n.d. Childhood Cancer Data Initiative (CCDI). https://www.cancer.gov/research/areas/childhood/childhood-cancer-data-initiative (accessed October 30, 2025).
NCI. 2023. Childhood Cancer STAR Act. https://www.cancer.gov/about-nci/legislative/recentpublic-laws/star-act (accessed December 20, 2025).
Nesheim, S. R., L. F. FitzHarris, M. A. Lampe, and K. M. Gray. 2018. Reconsidering the number of women with HIV infection who give birth annually in the United States. Public Health Reports 133(6):637–643.
NHGRI (National Human Genome Research Institute). 2024. IDENTIFY study. https://www.genome.gov/Clinical-Research/Current-NHGRI-Clinical-Studies/IDENTIFY-Study (accessed October 30, 2025).
NHLBI. 2024. Cystic fibrosis research. https://www.nhlbi.nih.gov/research/cystic-fibrosis#NHLBI-research-that-really-made-a-difference (accessed April 29, 2025).
NIAID (National Institute of Allergy and Infectious Diseases). n.d. Homepage. https://www.niaid.nih.gov/ (accessed April 29, 2025).
NICHD. 2022. Autism Centers of Excellence Program. https://www.nichd.nih.gov/research/supported/ace (accessed December 20, 2025).
NICHD. 2023. Child Health Research Career Development Award Program. https://www.nichd.nih.gov/research/supported/chrcda (accessed August 14, 2025).
NICHD. 2025a. Division of Intramural Research. https://www.nichd.nih.gov/about/org/dir (accessed April 29, 2025).
NICHD. 2025b. Division of Population Health Research. https://www.nichd.nih.gov/about/org/dir/dph (accessed April 29, 2025).
NICHD. 2025c. National Center for Medical Rehabilitation Research. https://www.nichd.nih.gov/about/org/ncmrr (accessed April 29, 2025).
NICHD. 2025d. Neonatal Research Network (NRN). https://www.nichd.nih.gov/research/supported/nrn (accessed December 20, 2025).
NICHD. 2025e. NIH Pediatric Research Consortium (NPeRC). https://www.nichd.nih.gov/research/supported/nperc (accessed April 29, 2025).
NICHD. 2025f. Office of the NICHD Director (OD). https://www.nichd.nih.gov/about/org/od (accessed April 29, 2025).
NICHD. 2025g. Science update: Partial steroid course can enhance preterm infant lung development, NICHD-funded study suggests. https://www.nichd.nih.gov/newsroom/news/022625-antenatal-steroids (accessed December 20, 2025).
NICHD. n.d.-a. Homepage. https://www.nichd.nih.gov/ (accessed April 29, 2025).
NICHD. n.d.-b. Safe to Sleep. https://safetosleep.nichd.nih.gov/ (accessed December 20, 2025).
NIGMS (National Institute of General Medical Sciences). 2025. Investigator-initiated research grants. https://www.nigms.nih.gov/about/overview/BBCB/biomedicaltechnology/Pages/InvestigatorInitiatedResearchGrants (accessed September 10, 2025).
NIH (National Institutes of Health). n.d. Organizational chart. https://oma.od.nih.gov/IC_Organization_Chart/OD%20Organizational%20Chart.pdf (accessed April 29, 2025).
NIH Clinical Center. n.d.-a. Places. https://www.cc.nih.gov/strategic/places (accessed April 18, 2025).
NIH Clinical Center. n.d.-b. Strategic plan introduction: People, places, capabilities. https://www.cc.nih.gov/strategic (accessed April 18, 2025).
NIH. 2021. Report of the director National Institutes of Health (2019–2021). https://dpcpsi.nih.gov/sites/default/files/2023-09/FY19-21%20Triennial_Report_FINAL_508C.pdf (accessed December 22, 2025).
NIH. 2024a. Candidate malaria vaccine provides lasting protection in NIH-sponsored trials. https://www.nih.gov/news-events/news-releases/candidate-malaria-vaccine-provides-lasting-protection-nih-sponsored-trials (accessed December 20, 2025).
NIH. 2024b. List of institutes and centers. https://www.nih.gov/institutes-nih/list-institutes-centers (accessed April 29, 2025).
NIH. 2025a. Budget. https://www.nih.gov/about-nih/organization/budget (accessed August 5, 2025).
NIH. 2025b. Inclusion across the lifespan in human subjects research. https://grants.nih.gov/policy-and-compliance/policytopics/inclusion/lifespan#:~:text=All%20human%20subjects%20research%20supported,reasons%20not%20to%20include%20them (accessed April 24, 2025).
NIH Almanac. 2025. Timelines: Chronology of events. https://www.nih.gov/about-nih/nih-almanac/chronology-events (accessed December 20, 2025).
NIH Grants & Funding. n.d. Parent announcements (for unsolicited or investigator-initiated applications). https://grants.nih.gov/funding/nih-guide-for-grants-and-contracts/parent-announcements (accessed September 10, 2025).
NIH Grants & Funding. 2025. Second level: Advisory council review. https://grants.nih.gov/grants-process/review/second-level (accessed December 20, 2025).
NIH Intramural Research Program. 2016. Folic acid levels impact the newborn epigenome and childhood asthma risk. https://irp.nih.gov/accomplishments/folic-acid-levels-impact-the-newborn-epigenome-and-childhood-asthma-risk (accessed December 20, 2025).
NIH Intramural Research Program. 2020. First therapy approved for children with genetic tumor syndrome. https://irp.nih.gov/accomplishments/first-therapy-approved-for-children-with-genetic-tumor-syndrome (accessed December 20, 2025).
NIH Intramural Research Program. 2022a. Our programs. https://irp.nih.gov/about-us/our-programs (accessed December 20, 2025).
NIH Intramural Research Program. 2022b. A robotic exoskeleton helps kids learn to walk better outside the clinical setting. https://irp.nih.gov/accomplishments/a-robotic-exoskel-eton-helps-kids-learn-to-walk-better-outside-the-clinical (accessed December 20, 2025).
NIH Intramural Research Program. 2022c. What is the IRP? https://irp.nih.gov/about-us/what-is-the-irp (accessed August 5, 2025).
NIH Intramural Research Program. 2022d. IRP review process. https://irp.nih.gov/our-research/irp-review-process#:~:text=Subsequently%2C%20the%20SD%20determines%20funding,meet%20expectations%2C%20funds%20are%20redeployed (accessed September 10, 2025).
NIH Office of Extramural Research. 2024. NIH grants policy statement. https://grants.nih.gov/grants/policy/nihgps/html5/section_2/2.4.3_national_advisory_council_or_board_review.htm (accessed September 10, 2025).
NIH Office of Strategic Coordination. 2024a. NIH Director’s Early Independence Award. https://commonfund.nih.gov/earlyindependence (accessed April 29, 2025).
NIH Office of Strategic Coordination. 2024b. NIH Director’s New Innovator Award. https://commonfund.nih.gov/newinnovator (accessed April 29, 2025).
NIH Office of Strategic Coordination. 2024c. NIH Director’s Pioneer Award. https://commonfund.nih.gov/pioneer (accessed April 29, 2025).
NIH Office of Strategic Coordination. 2024d. NIH Director’s Transformative Research Award. https://commonfund.nih.gov/tra (accessed April 29, 2025).
NIH Office of Strategic Coordination. 2025a. Gabriella Miller Kids First Pediatric Research (Kids First). https://commonfund.nih.gov/KidsFirst (accessed December 20, 2025).
NIH Office of Strategic Coordination. 2025b. Venture programs. https://commonfund.nih.gov/venture/beacons (accessed December 20, 2025).
NINDS. 2024. Nusinersen (spinraza®) – spinal muscular atrophy (sma). https://www.ninds.nih.gov/about-ninds/what-we-do/impact/ninds-contributions-approved-therapies/nusinersen-spinrazar-spinal-muscular-atrophy-sma#:~:text=Results%20from%20these%20and%20other,resembling%20timelines%20in%20healthy%20children.&text=The%20deve (accessed February 10, 2026).
Pediatric Heart Network. n.d. About the Pediatric Heart Network. https://www.pediatricheart-network.org/about-phn/ (accessed August 14, 2025).
PEHSU (Pediatric Environmental Health Specialty Units). 2025. Home. https://pehsu.net/ (accessed August 14, 2025).
Persaud, D., Y. Bryson, B. S. Nelson, C. Tierney, M. F. Cotton, A. Coletti, J. Jao, S. A. Spector, M. Mirochnick, E. V. Capparelli, D. Costello, J. Szewczyk, N. Nicodimus, L. Stranix-Chibanda, A. R. Kekitiinwa, V. Korutaro, C. Reding, M. N. Carrington, S. Majji, D. E. Yin, P. Jean-Philippe, and E. G. Chadwick. 2024. HIV-1 reservoir size after neonatal antiretroviral therapy and the potential to evaluate antiretroviral-therapy-free remission (IMPAACT P1115): A phase 1/2 proof-of-concept study. Lancet HIV 11(1):e20–e30.
PHACS (Pediatric HIV/AIDS Cohort Study). 2025. Pediatric HIV/AIDS Cohort Study. https://phacsstudy.org/ (accessed September 10, 2025).
Sanaria. 2025. A single-dose breakthrough: PfSPZ-LARC vaccines offer transformative protection against malaria. https://sanaria.com/2025/02/12/a-single-dose-breakthrough-pfspz-larc-vaccines-offer-transformative-protection-against-malaria/ (accessed December 20, 2025).
Sekar, K. 2025. The National Institutes of Health (NIH): Background and congressional issues. https://www.congress.gov/crs-product/R41705 (accessed December 22, 2025).
Shah, S. K. 2021. The ethics of pediatric research. In The Oxford handbook of research ethics. Pp. 683–698.
Shankaran, S., A. R. Laptook, R. A. Ehrenkranz, J. E. Tyson, S. A. McDonald, E. F. Donovan, A. A. Fanaroff, W. K. Poole, L. L. Wright, R. D. Higgins, N. N. Finer, W. A. Carlo, S. Duara, W. Oh, C. M. Cotten, D. K. Stevenson, B. J. Stoll, J. A. Lemons, R. Guillet, A. H. Jobe, and National Institute of Child Health and Human Development Neonatal Research Network. 2005. Whole-body hypothermia for neonates with hypoxic–ischemic encephalopathy. New England Journal of Medicine 353(15):1574–1584.
Shankaran, S., A. Pappas, S. A. McDonald, B. R. Vohr, S. R. Hintz, K. Yolton, K. E. Gustafson, T. M. Leach, C. Green, R. Bara, C. M. Petrie Huitema, R. A. Ehrenkranz, J. E. Tyson, A. Das, J. Hammond, M. Peralta-Carcelen, P. W. Evans, R. J. Heyne, D. E. Wilson-Costello, Y. E. Vaucher, C. R. Bauer, A. M. Dusick, I. Adams-Chapman, R. F. Goldstein, R. Guillet, L. A. Papile, R. D. Higgins, and Eunice Kennedy Shriver NICHD Neonatal Research Network. 2012. Childhood outcomes after hypothermia for neonatal encephalopathy. New England Journal of Medicine 366(22):2085–2092.
Silverman, F. H. 1988. The “monster” study. Journal of Fluency Disorders 13(3):225–231.
Temoka, E. 2013. Becoming a vaccine champion: Evidence-based interventions to address the challenges of vaccination. South Dakota Medicine 68–72.
Turriff, A. E., C. M. Annunziata, A. A. Malayeri, B. Redd, M. Pavelova, I. S. Goldlust, P. S. Rajagopal, J. Lin, and D. W. Bianchi. 2024. Prenatal cfDNA sequencing and incidental detection of maternal cancer. New England Journal of Medicine 391(22):2123–2132.
UPMC CHP (University of Pittsburgh Medical Center Children’s Hospital of Pittsburgh). 2025. The Pediatric Clinical and Translational Research Center (PCTRC). https://www.chp.edu/research/our-facilities/pctrc (accessed August 14, 2025).
Zildzic, M., and L. Zunic. 2023. The most influential scientists in the development of public health: Edward Jenner (1749-1823). International Journal on Biomedicine and Healthcare 11(1):58–61. https://doi.org/10.5455/IJBH.2023.11.58-61.





















