Strategies to Enhance NIH-Funded Pediatric Research: Optimizing Child Health (2026)
Chapter: 3 Current NIH Pediatric Research Portfolio
3
Current NIH Pediatric Research Portfolio
We will look to the National Institute of Child Health and Human Development for a concentrated attack on the unsolved health problems of children and of mother–infant relationships. This legislation will encourage imaginative research into the complex processes of human development from conception to old age. . . . For the first time, we will have an institute to promote studies directed at the entire life process rather than toward specific diseases or illnesses.
—John F. Kennedy, October 17, 19621
DELINEATING PEDIATRIC RESEARCH AT NIH
At the National Institutes of Health (NIH) “pediatric research” is defined in different ways, depending on the needs of different institutes or centers (ICs), consortia, or reporting groups. As noted in Chapter 1, NIH defines a child for the purposes of research participation as any individual younger than 18 years. An internal memo from NIH characterizes pediatric research as “studies in all categories of biomedical research (basic, clinical, epidemiologic, behavioral, prevention, treatment, diagnosis, as well as outcomes and health services) that relate to diseases, conditions, or the health/development of neonates, infants, children, and adolescents up to age 21” (Gitterman et al., 2023, p. 797). The contrast between the two reflects the distinction between necessary legal protections for individual human participants in research and a developmental approach to defining
___________________
the scope of “pediatric research.” Also discussed in Chapter 1, the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) reported to the committee that “pediatric research” may encompass several distinct areas of study: research in which participants do not specifically exclude children, research in which participants specifically include children, or research involving children with a clinical or public health focus on conditions or developmental processes that typically originate and manifest in childhood. (It is also important to note that this research nearly always involves the participation of caregivers as guardians of the pediatric research participants—whether they are reporting information to investigators or ensuring safe transportation to clinical trial sites, successful pediatric research could not occur without them.) While each of the various definitions in use has merit, it is essential to have a singular definition across NIH to better describe and evaluate pediatric research.
Recommendation 3-1: The National Institutes of Health should adopt and apply a consistent agency-wide definition of pediatric research. The definition should recognize that pediatric research may include studies that (1) involve data or specimens from or about infants, children, adolescents, or young adults; (2) inform the prevention, diagnosis, or treatment of conditions affecting these populations; (3) advance the health, well-being, or resilience of children, adolescents, or young adults; or (4) investigate childhood origins of adult health conditions.
NIH DATA SYSTEMS FOR MONITORING PEDIATRIC RESEARCH SUPPORT
NIH maintains several systems for public reporting on its funded research projects; these systems can be found on the Research Portfolio Online Reporting Tools (RePORT) website.2 One of these systems is the RePORT Expenditures and Results module (RePORTER), which allows users to search NIH-funded research projects using a wide range of fields, such as principal investigator, project location or congressional district, funding IC, funding mechanism, award type, project number, spending category (of the Research, Condition, and Disease Categorization [RCDC]; see below), and much more. Users can also view publications and patents resulting from NIH funding as well as research funded by other federal
___________________
2 Both systems are found at https://report.nih.gov, but the “Categorical Spending webpage,” as it is frequently identified in this report, can be found at https://report.nih.gov/funding/categorical-spending#/. It is sometimes referred to as the RCDC webpage. Advanced searches in RePORTER can be performed at https://reporter.nih.gov/advanced-search.
agencies, such as the Administration for Children and Families, the Agency for Healthcare Research and Quality, the Centers for Disease Control and Prevention, the Health Resources and Services Administration, the Food and Drug Administration, and the Department of Veterans Affairs (NIH RePORT, n.d.-a, 2025). Another system, which is the focus of this chapter, is the RCDC system, developed specifically to comply with legislative mandates to report the spending of appropriated funds on specific research topics, known as categories. These categories do not constitute a taxonomy, and many of them overlap. Both of these systems feature bulk export options for the public to download the data (RePORTER has a separate webpage for ExPORTER, which “provides bulk administrative data found in RePORTER to the public for detailed analyses or to load into their own data systems . . . generally in CSV formats”), although the number of search fields and amount of information included differ between the systems. The Categorical Spending webpage exports offer 11 fields when exporting bulk data by spending category, including spending category, funding IC, project number and title, principal investigator name, and funding amount. By contrast, RePORTER has 46 fields, which include the 11 fields in Categorical Spending webpage exports along with other fields providing much more extensive detail about each project.3 As part of its charge, the committee was asked to examine “the methods and rationale involved in categorizing projects as ‘Pediatrics’ research using the [RCDC] process.” This chapter presents the committee’s analysis of the RCDC reporting system as it appears to the public.
How the RCDC System Works
Initiated in 2008 in response to the NIH Reform Act,4 RCDC ensures uniform coding of research grants and activities across NIH ICs so that the public can analyze NIH appropriations and identify trends and gaps in research funding (NASEM, 2024). RCDC categories can be created at the request of Congress, the White House, advocacy groups, and NIH leadership (NIH, n.d.).
Assigning a grant to one or more of the RCDC spending categories involves several steps. First, the NIH Automated Indexing Service scans each research project’s title, abstract, specific aims section, and public health
___________________
3 See https://report.nih.gov/funding/categorical-spending#/ to export data by RCDC term within the Categorical Spending webpage and https://reporter.nih.gov/exporter to export RePORTER project data by fiscal year.
4 The National Institutes of Health Reform Act, Public Law 109-482, 109th Cong., January 15, 2007.
relevance section for “concepts” that are included in the RCDC Thesaurus. The thesaurus is based on several established biomedical vocabularies and can be “anything ranging from specific diseases, gene names, types of diagnostic tools, therapeutic techniques, to patient populations” (NIH RePORT, n.d.-c). A project typically has many concepts assigned to it. Once this text analysis is done, a project index is produced with the full list of concepts and the weighted number of times each appears in the text.
The project index is then evaluated against the RCDC “category fingerprints.” Each “category fingerprint” includes a set of concepts (see Figure 3-1) and may also incorporate administrative business rules. The set of concepts in the fingerprint is identified by a panel of scientific experts drawn from the NIH ICs. These scientific experts set the parameters for each category and review example projects to determine whether that research should be publicly reported as belonging to a category. This process results in a weighted set of concepts known as a fingerprint. When there is sufficient overlap between the concepts indexed by the project and the category fingerprint, the project will generally be assigned to the category. The threshold for determining sufficient overlap is developed empirically to maximize the validity and reliability of the category—that is, to minimize the occurrence of false positive and false negative results. After this process is completed, projects in the resulting categories are validated by IC experts to ensure that the fingerprint is identifying projects correctly according to the parameters determined by the scientific reviewers. The validation process also includes an artificial intelligence component as a secondary quality check and to minimize burden on the scientific experts while ensuring that the experts’ review contributes to the category validity.
RCDC categories are neither exclusive nor exhaustive. A single project can have multiple RCDC categories assigned to it. For example, “pediatrics,” “childhood injury,” and “unintentional childhood injury” are all distinct categories (Gitterman et al., 2022), and a project studying unintentional poisonings among children would likely be categorized under all three. This is in part because the categories can reflect conditions or diseases of significant public or congressional interest. By nature, the categories overlap, and the categories do not encompass all types of biomedical research (NIH Grants & Funding, 2025a; NIH RePORT, n.d.-c). As of fiscal year (FY) 2024, RCDC had 327 total categories of research, condition, and disease areas. For FY 2024, the committee found there were 310 spending categories assigned to projects that were also assigned to the “Pediatric” category.
The RCDC system describes the Pediatric category as including “research on health conditions that originate during the embryonic period
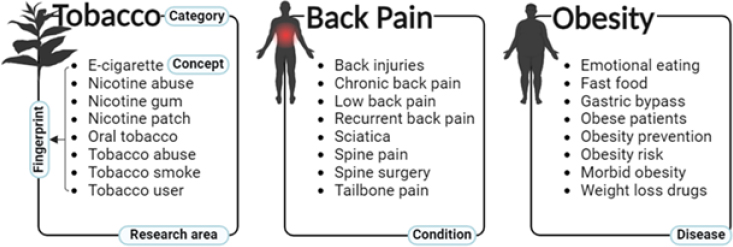
NOTE: RCDC = Research, Condition, and Disease Categorization.
SOURCE: NIH (n.d.).
to age 21, [along with] congenital and developmental diseases” (NIH Grants & Funding, 2025b). Some categories use business rules beyond text analysis to include or exclude projects—for example, using metadata-based rules such as grant codes, funding announcements, or NIH institute (NIH, n.d.). Category relationships are employed to include projects from “narrower” categories into broader ones (referred to as a child–parent relationship) without changing category fingerprints. For example, the following child categories are automatically assigned to the parent Pediatric category: Adolescent Sexual Activity, Child Abuse and Neglect Research, Childhood Obesity, Conditions Affecting the Embryonic and Fetal Periods, Congenital Heart Disease, Congenital Structural Anomalies, Duchenne/Becker Muscular Dystrophy, Pediatric—AIDS, Infant Mortality, Pediatric Cancer, Pediatric Cardiomyopathy, Perinatal Period—Conditions Originating in Perinatal Period, Rett Syndrome, Spinal Muscular Atrophy, Youth Violence, Childhood Injury, and Underage Drinking (NIH Grants & Funding, 2025b).
GAPS AND OPPORTUNITIES FOR THE RCDC PEDIATRIC SPENDING CATEGORY
Per the committee’s statement of task, the committee evaluated the RCDC pediatric spending category in order to assess its utility for public reporting of pediatric research funded by NIH. RCDC is a consistent NIH-wide classification framework which standardizes how research areas are defined and reported across ICs. The Categorical Spending website is user-friendly, allowing for the selection of individual RCDC categories to be examined. In addition, the website supports data downloads of these
category-specific analyses by year. The categorization process is scalable and adaptable. Because the RCDC categorization process uses automated text mining rather than relying on manual review, as NIH’s portfolio expands the categorization process can handle an increase to volume without loss of performance. In addition, RCDC categories can be added as needed based on congressional and IC priorities and public input. Finally, the system allows projects to be tagged under multiple categories, effectively capturing the interdisciplinary nature of biomedical research.
However, a few gaps limit the utility of the Categorical Spending webpage and interface. Regarding the technical aspects of the system, the Categorical Spending webpage supports single-spending-category downloads only, even though the majority of projects are tagged with multiple categories. Thus, it lacks functionality to query RCDC category intersections (e.g., Pediatric × ADHD).
The content of the Categorical Spending webpage could also be enhanced. The committee found that important analytical fields are missing. It is important to note that many of these fields exist in some form when using the RePORTER webpage advanced search function. Nevertheless, it would be useful to be able to access these fields in the Categorical Spending webpage directly. Clear designation of whether a project is intramural or extramural research is lacking, and a designation for clinical, basic, and translational research is missing. There is an RCDC spending category for clinical research but not for basic or translational sciences (NIH Grants & Funding, 2025b). (There is a term for basic behavioral and social science research, but this does not cover the full range of basic science.) Therefore, the fields available in the Categorical Spending webpage were insufficient to identify projects that are classified as basic, clinical, or translational research. The committee was attentive particularly to the inability to place research into the basic, clinical, and translational categories as these categories were also used in several NIH presentations on the subject, although other important research categories were also absent. In RePORTER, various research categories, including basic, clinical, translational, dissemination and implementation sciences, and health services research, can be found under the Project Terms field, though there is no equivalent search field within the interface or data export in the Categorical Spending webpage. Some of these terms may overlap (i.e., the spending category Clinical Research includes health services research), and, at a minimum, it would be useful to be able to distinguish basic science research from clinical research in the context of the Categorical Spending webpage.
It is not currently possible to determine funding by category with the sort of specificity that might be useful for policy making, prioritization of resource distribution, or other impactful decisions made by industry, academia, government, and nonprofit leaders. As an example, one might
hope that funding is reasonably commensurate with the burden of disease, injury, and mortality in each stage of child and adolescent development. Knowledge about NIH funding during major stages of development would provide data to confirm that this is the case or to adjust funding patterns otherwise. Similarly, scholars recognize the value of conducting a range of research that falls into the basic to clinical to translational science paradigm, working toward interventions that improve human health. The current categorization system does not classify research into those categories, preventing an analysis of spending along the basic to translational research spectrum.
It is also not feasible to disaggregate projects by sub-categories such as child development stages (e.g., infant, child, adolescent). When describing pediatric developmental stages, the American Academy of Pediatrics uses the following categories: prenatal, birth–12 months, 1–3 years, 3–5 years, 5–12 years, 12–18 years, and 18–21 years (AAP, n.d.). For research purposes, it is the committee’s view that adding a category for neonates (0–28 days) is critical as more research for this specific age group is greatly needed. For example, very little is known about the efficacy, safety, and dosage for medications for newborns, and, as a result, over 90 percent of medication for them is prescribed off-label or off-license (Modi, 2023). Additionally, for research purposes, the committee feels it important to extend the 18–21 category to 18–24 years as significant biological and neuropsychological maturation and social transitions occur during the early adulthood period (Hochberg and Konner, 2019; Sawyer et al., 2018), with some aspects of executive function developing well into the 20s (Ferguson et al., 2021; Shulman et al., 2015).
The committee also observed the use of eponyms in the classification system. Because eponyms are often local or historical, they may be familiar in one region but confusing or unknown to clinicians and patients elsewhere. They can also complicate reporting, since coders have to memorize arbitrary names instead of terms that encode pathophysiology. Using descriptive terminology for conditions improves clarity, supports communication, and better aligns with modern mechanism-based medicine. The committee found variations and inconsistencies in spelling or phrasing of RCDC terms (despite a standard RCDC term list), creating redundant or mismatched category names, which requires manual cleaning and makes automated processing by external researchers difficult. Data for RePORTER come from the Electronic Research Administration’s (eRA’s) IRDB (IMPAC II Reporting Database—NIH grant data for reporting) as well as from iEdison (inventions), Clinicaltrials.gov, NIH Intramural Database, and PubMed, while data from RCDC appear to come from eRA alone; this discrepancy likely accounts for some differences or lack of updates between older category names assigned to projects (NIH RePORT, n.d.-b).
When the committee merged its data between its RCDC and RePORTER search, it found several discrepancies:
- Year-to-year variation: Minor grammatical or word order changes that occurred over time in the RCDC were not reflected retroactively (e.g., ADD updated to ADHD).
- Category updates and renaming: Some RCDC categories have changed names (e.g., Pneumonia & Influenza became Pneumonia and Influenza), but these updates are not consistently reflected in older RePORTER data.
It would, of course, be useful if the RCDC system were a complete and exhaustive taxonomy of research, condition, and disease categories of projects funded by NIH, but this effort would prove to be immensely time-consuming for NIH staff, and, absent that, the committee is issuing the recommendation below to specifically enhance pediatric research development.
Conclusion 3-1: The National Institutes of Health (NIH) Research, Condition, and Disease Categorization system is a valuable resource for public reporting of NIH-sponsored research projects. The system’s value would be improved by enhancing its specificity and validity for pediatric research as well as its transparency and technical application.
Recommendation 3-2: The National Institutes of Health (NIH) should improve the Research, Condition, and Disease Categorization (RCDC) system to better assess funding for pediatric research by (a) adding the new spending categories and (b) improving the Categorical Spending webpage in the NIH Research Portfolio Online Reporting Tools (RePORT).
The following new spending categories should be updated:
- Add pediatric developmental stages: during pregnancy; neonate (≤28 days); infant (<1 year old); early childhood, age 1–2 years (1- to 2-year-olds) and age 3–5 years (3- to 5-year-olds); middle childhood (6- to 11-year-olds); adolescence (12- to 17-year-olds); and young adults (18- to 24-year-olds).
- Add a Basic Science spending category and ensure its validity.
- Add new terms for pediatric health conditions to better reflect the breadth of pediatric health conditions. The process to determine these new terms should include child health expertise from each institute and center.
- To improve specificity of diagnostic labeling, consider switching eponyms to indicate diseases—e.g., change Cooley’s Anemia to “Beta Thalassemia Major.”
To improve the Categorical Spending (RCDC) webpage in RePORT, NIH should
- Publish a reference paper that clearly and comprehensively describes the RCDC classification methodology with sufficient detail that it can be replicated or a similar assessment can be done by the scientific community. Update the methodology report whenever the methodology changes.
- Evaluate the validity of key RCDC terms, such as “pediatric,” and update the classification to improve accuracy and remove possible biases.
- Allow more than one RCDC term to be exported in a specific project. (This functionality currently exists in RePORTER, but not RCDC.)
- Enable download of all projects funded by NIH for a given year.
- Add a flag to clearly identify extramural; intramural; and, specifically, Clinical Center research and ensure that this information is available for all research projects.
- Add a search field that links projects to their respective notice of funding opportunity, program announcement, program announcement with special review, request for application, notice of special interest, or request for proposal.
Pediatric Research Project Audit
To evaluate the classification accuracy of the RCDC category Pediatric and examine a sample of pediatric research projects funded during the 10-year study period, the committee randomly sampled 20 projects per year assigned the Pediatric category for each of the 10 years (total number of projects was 200; the total number of projects in a given year ranged from about 9,000 to 12,000). A single committee member reviewed the title, abstract, and public health relevance statement for the 200 projects and summarized the findings. Projects for loan repayment and training grants were excluded. Each project was assessed for its pediatric relevance (whether or not it was a study that focused on pediatric health conditions that affect children and youth <21 years old), whether or not it could be considered pediatric basic science (i.e., research not involving data collected from human subjects), and the specific health conditions addressed by the project. Data were entered into a REDCap form and analyzed.
This audit found that 12 of 200 (6 percent) projects categorized as pediatric research by the RCDC system had no public-facing evidence of being pediatric research as determined by the pediatric scientific expert conducting the review of abstracts. Some of the incorrectly classified projects
focused exclusively on maternal health during pregnancy and appeared to exclude pediatric outcomes or child health assessment. Examples include a study on interpersonal psychotherapy for major depression following a perinatal loss, a study on promoting maternal mental health and well-being for mothers of infants in neonatal intensive care, and a study that examined mindfulness-based interventions for poor maternal sleep quality during pregnancy. While it can be argued that any of these subjects may affect the health of a fetus or infant, such studies were excluded when they did not appear to assess those outcomes.
Of the 188 projects classified as pediatric research, 83 (44 percent) had a component of or were fully characterized as pediatric basic science. These projects included animal studies, work with human cell lines, stem cell research, and other methods that did not directly collect or analyze data from human subjects but did address health conditions relevant to children and adolescents. Half (50 percent) of the 188 projects indicated in publicly accessible materials that they would enroll children and youth <21 years old.
Data from the NIH Office of Budget
The NIH Office of Budget oversees financial planning and reporting across all NIH ICs. As part of its external reporting, the office provides publicly accessible data that detail NIH’s historical and current funding trends (NIH Office of Budget, n.d.-b). Available data include annual budget request summaries; annual spending by IC, mechanism, and research category; and archived appropriations data (NIH Office of Budget, n.d.-a). These data were used by the committee to obtain the expenditures for NIH overall and by IC, excluding the line item for administrative services.
It may pose a challenge for NIH to implement the changes suggested in Recommendation 3-2 to the RCDC system, as they will each require financial and staffing resources. However, the committee maintains that updates to RCDC are paramount to enhancing the value and impact of pediatric research supported by NIH and to improving access and transparency to policy makers and the public. Improved spending categories would allow NIH and external stakeholders to see the true scale of investment in pediatric research. It would support data-driven policy and funding decisions as well as prioritization of allocated resources by industry, nonprofit entities, and academia. In addition, with enhanced transparency, data, and accountability, NIH could track, monitor, and demonstrate through targeted and broad communications the positive outcomes of its investments in pediatric research. Finally, if these proposed changes to the RCDC system were implemented, they would highlight underfunded or overlooked areas in pediatric research and enable strategic prioritization of research initiatives. In short,
accurate, consistent data would make it easier to assess and understand the impact of NIH investments on pediatric health outcomes over time.
DATA ON PEDIATRIC RESEARCH FUNDING BY NIH
Overview
Part of the committee’s charge in its statement of task was to use its review of the RCDC process to inform recommendations on improving NIH’s overall support of pediatric research. A dataset of NIH-funded pediatric research projects spanning a 10-year study period, from FY 2015 through FY 2024, was constructed by exporting data from the Categorical Spending webpage. Projects with the RCDC category of Pediatric were considered to be pediatric research. RCDC files were linked to records in RePORTER using the Application ID, which uniquely identifies a project. This linkage was done to obtain all RCDC categories assigned to each project. The Categorical Spending webpage allows for search and export of research projects only one category at a time, and virtually all projects are assigned multiple RCDC categories. The funding IC variable in RePORTER was used selectively when it provided more specific information than RCDC. For example, some projects in RCDC do not list a funding IC because they are tagged under NIH-wide initiatives. Those same projects in RePORTER are tied to specific ICs (e.g., the National Institute of Diabetes and Digestive and Kidney Diseases, the National Institute of Allergy and Infectious Diseases, and NICHD). In those cases, the committee pulled the funding IC from RePORTER. Data were accessed between May 2 and July 15, 2025.
To adjust research project expenditures for inflation, actual dollars were converted to constant 2020 dollars using the Biomedical Research and Development Price Index (BRDPI) published by the Bureau of Economic Analysis of the Department of Commerce (BLS, n.d.; NIH Office of Budget, 2025). Specifically, nominal outlays for each fiscal year (FY 2015 to FY 2024) were multiplied by the ratio of the annual average BRDPI for 2020 to the BRDPI for the corresponding fiscal year, thereby removing the effects of general price inflation and facilitating comparisons over time. Certain multi-project grant mechanisms—such as P01, P50, P30, P60, U19, and U54—include multiple independently funded subprojects. In the NIH reporting systems, each subproject and supplement is assigned its own identifier and reported as a unique project (NIH, n.d.). For the committee’s analysis, each subproject was considered as a separate project.
Overall Spending Trends
NIH expenditures for research, excluding administrative costs, rose from $28.5 billion in FY 2015 to $46.1 billion in FY 2024, a 62 percent increase (see Figure 3-2). However, inflation-adjusted growth in expenditures flattened beginning in FY 2020, and real growth over the 10-year period was just 26 percent.
Similar trends occurred with pediatric research (see Figure 3-3). Actual pediatric research expenditures increased from $3.5 billion in FY 2015 to $6.2 billion in FY 2024, a 70 percent increase. In parallel with overall NIH research funding, a plateau in inflation-adjusted funding was observed beginning in FY 2020, and real growth during the 10-year period was just 32 percent.
The total pediatric research expenditures, based on the pediatric RCDC data, in FY 2024 were $87 per child under 18 years old in the United States. As a proportion of NIH non-administrative expenditures, pediatric research accounted for a constant level of about 13 percent from FY 2015 to FY 2024 (see Figure 3-4). This contrasts with the fact that children and
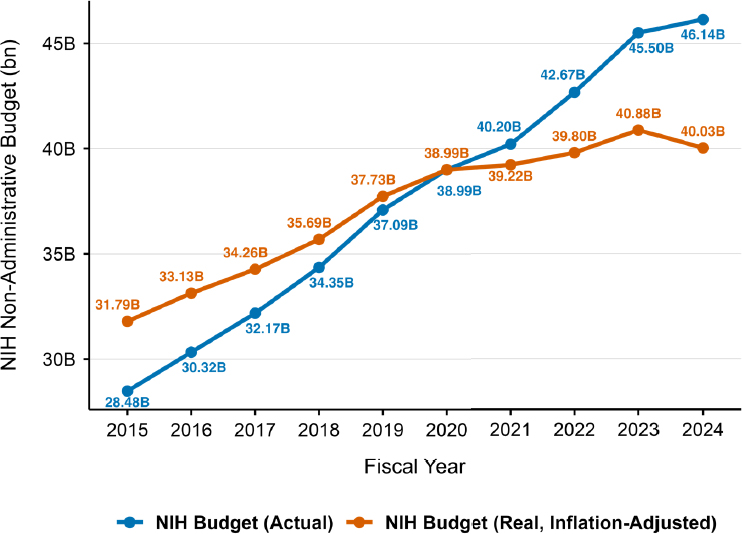
NOTE: FY = fiscal year; NIH = National Institutes of Health.
SOURCE: Committee’s analysis of the data from the NIH Office of Budget.
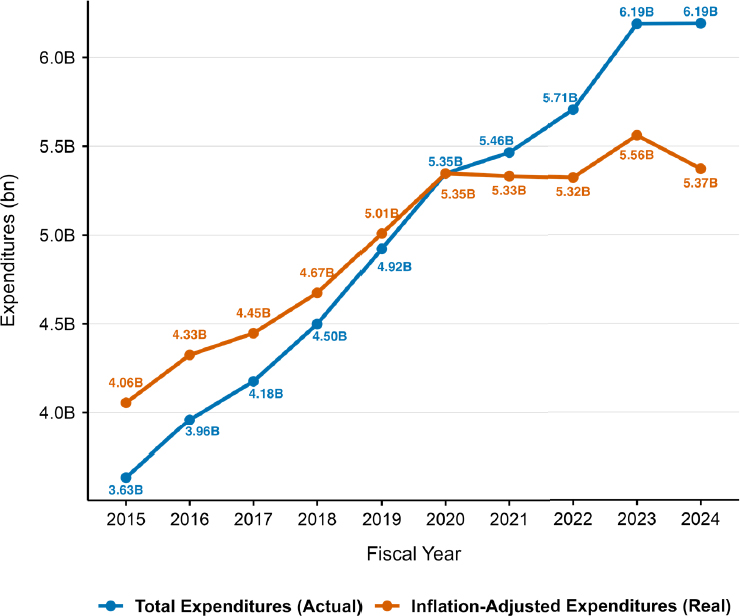
NOTE: FY = fiscal year; RCDC = Research, Condition, and Disease Categorization.
SOURCE: Committee’s analysis of data from the RCDC system.
adolescents under 18 years of age in 2024 accounted for about 22 percent of the U.S. population (Forum on Child and Family Statistics, 2024a,b).
During the 10-year period, the number of pediatric research projects rose from 9,079 to 12,527, a 38 percent increase. The flattening of pediatric research inflation-adjusted funding since FY 2020 led to a decline in real dollars per project from $455,000 in FY 2020 to $429,000 in FY 2024, resulting in a decline in purchasing power for NIH-funded pediatric research projects.
On an annual basis, intramural pediatric research conducted by federal employees and federal researchers within NIH’s laboratories and facilities accounted for between 9 percent and 11 percent annually of pediatric research expenditures across the 10-year period.
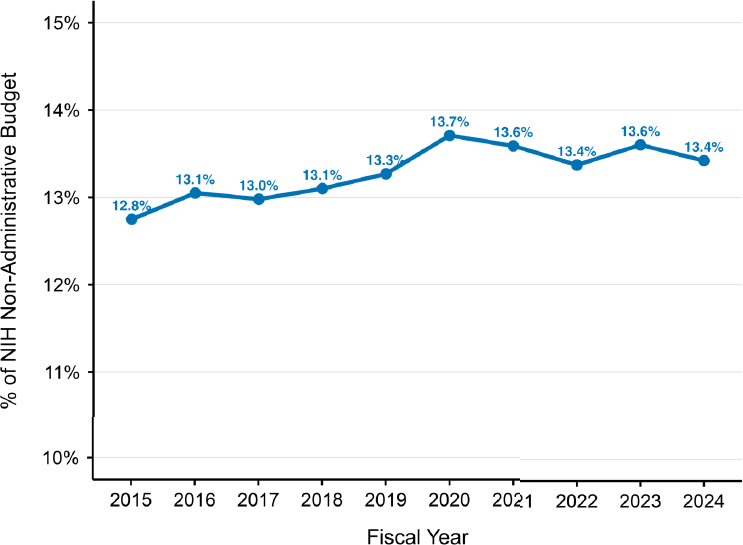
NOTE: FY = fiscal year; NIH = National Institutes of Health; RCDC = Research, Condition, and Disease Categorization.
SOURCE: Committee’s analysis of data from the RCDC system and data from NIH Office of Budget.
Conclusion 3-2: Over the decade analyzed in this report, pediatric research consistently accounted for about 13 percent of non-administrative National Institutes of Health (NIH) spending. Growth in actual dollars largely reflected agency-wide expansion rather than a strategic re-balancing toward pediatric health. Although NIH pediatric research dollars rose 70 percent in nominal terms between FY 2015 and FY 2024 and outpaced overall NIH growth (62 percent), the inflation-adjusted trend flattened after FY 2020. Real dollars per project fell from $455,000 in FY 2020 to $429,000 in FY 2024, signaling a loss of purchasing power that may erode pediatric research project scope.
Grantee Institution and IC
Each grantee institution was assigned to one of four categories: academic, which included universities, medical schools, and research centers affiliated with universities; children’s hospitals, both freestanding (e.g., Seattle Children’s Hospital) and within hospitals (e.g., Benioff Children’s
Hospital at the University of California, San Francisco); general or specialty hospitals not primarily pediatric, such as Veterans Affairs hospitals and Kaiser Permanente; and all other organizations (e.g., nonprofit organizations, transfers between federal agencies). Additionally, joint appointments are also common.
The committee recognized the limitations of this assignment. Some investigators are employed at children’s hospitals, but their grants are managed by partner institutions, such as degree-granting institutions. For example, the Lucile Packard Children’s Hospital is a freestanding children’s hospital, but its grants are managed by Stanford University.
The number of institutions that received funding for pediatric research increased from 754 in FY 2015 to 887 in FY 2024. In FY 2024, the distribution of institution type was heavily weighted toward academic institutions (see Figure 3-5). Children’s hospitals as the grantee institution accounted for 10 percent of the pediatric projects, and children’s hospitals
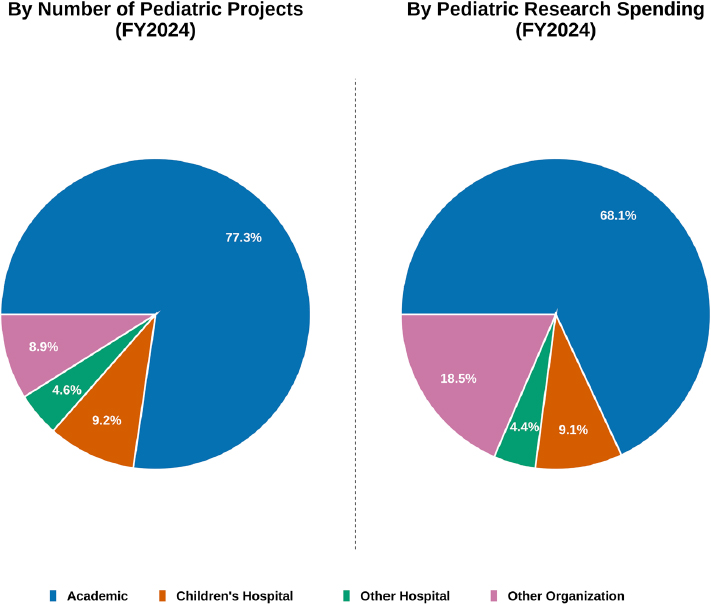
NOTE: FY = fiscal year; RCDC = Research, Condition, and Disease Categorization; RePORTER = National Institutes of Health Research Portfolio Online Reporting Tools Expenditures and Results module.
SOURCE: Committee’s analysis of data from the RCDC and RePORTER systems.
and other organizations each accounted for 10 percent of pediatric research expenditures.
Principal investigator academic departments were categorized into basic science, non-pediatric clinical, and pediatric (see Table E-1 for full list). It is noteworthy that 40 percent of projects had no departmental information. This high rate of missing data is likely because hospitals, including children’s hospitals, do not have department information that is reported to NIH, and not all institutions report their departmental affiliations for their faculty and staff. This table refers to contact principal investigators and does not include the multiple appointments that some researchers may have. Of the 60 percent of projects with available data, non-pediatric clinical departments accounted for the majority (52–54 percent) of pediatric research (see Figure 3-6). Pediatric departments accounted for 12–14 percent of pediatric research. In FY 2024, 19 percent of pediatric research expenditures were from either pediatric departments or children’s hospitals. Pediatric research led by principal investigators in basic science departments was 13–15 percent of the total. International principal investigators were responsible for less than 2 percent of project expenditures.
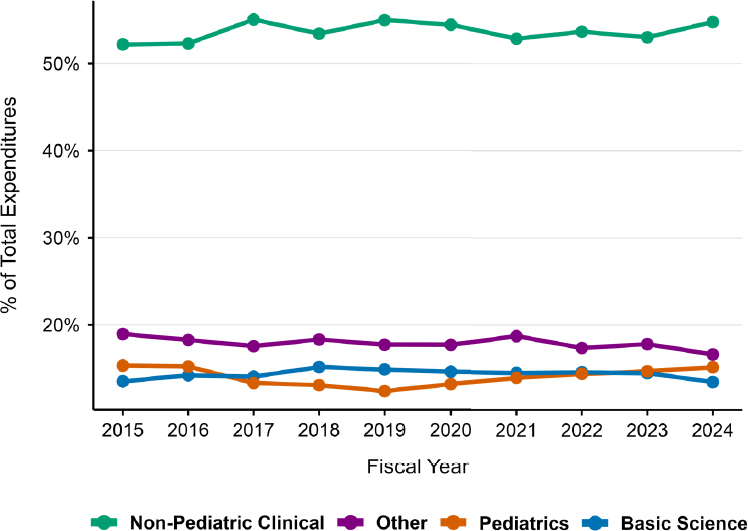
NOTE: FY = fiscal year; RCDC = Research, Condition, and Disease Categorization; RePORTER = National Institutes of Health Research Portfolio Online Reporting Tools Expenditures and Results module.
SOURCE: Committee’s analysis of data from the RCDC and RePORTER systems.
NICHD Pediatric Research Funding
NICHD provides leadership within NIH on children’s health research. Although it funds just 17 percent of NIH-funded pediatric research, it provides the largest amount of funding in absolute terms of any IC ($998 million in FY 2024), both for research and for pediatric research training. From FY 2015 to FY 2024, NICHD funding as a proportion of the overall NIH research budget was approximately 4 percent. It is worth noting that this contrasts with what several NIH-funded investigators reported in their responses to the committee’s call for perspectives (see Box 3-1).
Conclusion 3-3: The Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) is an essential leader for pediatric research within the National Institutes of Health (NIH) and funds the plurality of all pediatric research at NIH, although all funding institutes and centers (ICs) support some level of pediatric research. The diversity of ICs that fund pediatric research and training suggests a continued need for NICHD leadership, which houses a substantial amount of pediatric expertise that other ICs can leverage.
The proportion of each IC’s total research budget spent on pediatric research varied widely, as shown by the black circles corresponding with the secondary y-axis in Figure 3-7. Although every IC funded some pediatric research, there was wide variation in the level of funding. Aggregating the total pediatric research funding across FY 2015–FY 2024, ICs were categorized into five tiers (see Figure 3-7, bars). Tier 1 included a single institute, NICHD, which funded the plurality of research, accounting for 17 percent of total pediatric research expenditures across NIH. Tier 2 ICs included the National Cancer Institute (11 percent), National Heart, Lung, and Blood Institute (10 percent), National Institute of Mental Health (NIMH) (10 percent), and National Institute of Allergy and Infectious Diseases (9 percent). Combined, Tier 1 and Tier 2 ICs accounted for the majority (57 percent) of pediatric research funding. Using each IC’s pediatric research expenditures divided by its total research budget (summed across the 10-year period FY 2015–FY 2024), the five ICs with the highest pediatric shares were NICHD (60 percent), NIMH (26 percent), the National Institute of Dental and Craniofacial Research (26 percent), the National Institute on Alcohol Abuse and Alcoholism (25 percent), and the National Institute on Minority Health and Health Disparities (21 percent).
Spending categories assigned by RCDC are critically important as they enable analysis of patterns of funding for disorders, injuries, and illnesses that affect children and youth. In a study conducted by Rees and colleagues,
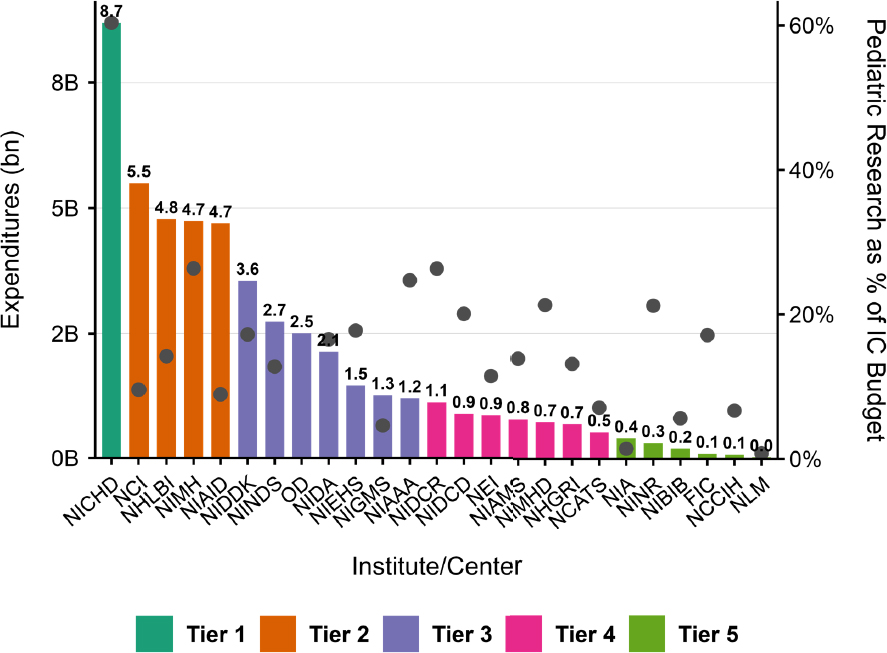
NOTES: The bars represent expenditures by the billion corresponding with the left y-axis. The dots represent the proportion of each IC total research budget that went to pediatric research corresponding with the right y-axis. The bars represent expenditures by the billion corresponding with the left y-axis. The dots represent the proportion of each IC total research budget that went to pediatric research corresponding with the right y-axis. FIC = Fogarty International Center; IC = institute and center; NCATS = National Center for Advancing Translational Sciences; NCCIH = National Center for Complementary and Integrative Health; NICHD = Eunice Kennedy Shriver National Institute of Child Health and Human Development; NCI = National Cancer Institute; NEI = National Eye Institute; NHGRI = National Human Genome Research Institute; NHLBI = National Heart, Lung, and Blood Institute; NIA = National Institute on Aging; NIAAA = National Institute on Alcohol Abuse and Alcoholism; NIAID = National Institute of Allergy and Infectious Diseases; NIAMS = National Institute of Arthritis and Musculoskeletal and Skin Diseases; NIBIB = National Institute of Biomedical Imaging and Bioengineering; NIDA = National Institute on Drug Abuse; NIDCD = National Institute on Deafness and Other Communication Disorders; NIDCR = National Institute of Dental and Craniofacial Research; NIDDK = National Institute of Diabetes and Digestive and Kidney Diseases; NIEHS = National Institute of Environmental Health Sciences; NIGMS = National Institute of General Medical Sciences; NIMH = National Institute of Mental Health; NIMHD = National Institute on Minority Health and Health Disparities; NINDS = National Institute of Neurological Disorders and Stroke; NINR = National Institute of Nursing Research; NLM = National Library of Medicine. OD = Office of the Director.
SOURCE: Committee’s analysis of data from the Research, Condition, and Disease Categorization system.
pediatric research project expenditures obtained from the RCDC were correlated with a variety of population health statistics (Rees et al., 2021). The authors found moderately strong correlations of 0.63 for disability-adjusted life-years, 0.56 for years of potential life lost, and 0.68 for hospital days.
To estimate the level of funding for research that includes a focus on the social influences on health, projects were identified that included at least one of the following RCDC categories of research: social determinants of health (published in 2020), health disparities and racial or ethnic minority health (published in 2023), health disparities research (published in 2023), or racial and ethnic minority health (published 2023). In FY 2024, 29 percent of all pediatric research included a focus on social influences on health (and were accordingly assigned to one of those spending categories).
Conclusion 3-4: National Institutes of Health (NIH) pediatric research funding is broad but concentrated. Every NIH institute and center (IC) supports pediatric projects, yet five of them (the Eunice Kennedy Shriver National Institute of Child Health and Human Development; National Cancer Institute; National Heart, Lung, and Blood Institute; National Institute on Mental Health; National Institute of Allergy and Infectious Diseases) account for the majority of expenditures. No single IC exclusively funds any major health condition category, underscoring both the cross-cutting nature of pediatric research funding and the need for strong inter-IC coordination.
CURRENT PEDIATRIC RESEARCH AT THE CLINICAL CENTER
The NIH Clinical Center (CC) plays a critical role in enabling pediatric research, particularly for children with rare, complex, or currently untreatable diseases. The CC hosts clinical trials and natural history studies for rare and complex pediatric diseases and offers highly specialized wraparound care tailored for children involved in research protocols. Pediatric research at the CC includes gene, stem cell, and cellular therapies with an emphasis on deep phenotyping, genotyping, and novel therapeutic strategies. The CC hosts an 18-bed inpatient pediatric unit, 14-bed pediatric day hospital, 4-bed pediatric behavioral health unit with 2-bed day hospital, and dedicated outpatient clinics; it also provides access to pediatric subspecialties (e.g., endocrinology, gastroenterology, infectious disease) via NIH institutes or partner hospitals and a growing pediatric intensive care unit capability, with 24/7 anesthesia, child life specialists, and pediatric
BOX 3-1
Insights from Grantees: Perceived Funding Discrepancies Between ICs
“Pediatric research is not prioritized across all ICs. NICHD has a limited budget and cannot be the sole or even primary source of funding for pediatric research. Child health at all stages influences adult health. Pediatric populations should be included across all ICs and life course research prioritized.”
— Researcher funded by NICHD, NIAID, NCI, NIGMS, and NCATS
“Investigator-initiated R01s for children’s health-related research often gets assigned to NICHD, but then the low paylines of the NICHD means even if it was well scored, it will not get funded.”
— Recipient of NIH funding for 30 years from NIDA, NIAAA, and NICHD
“Cross-agency mechanisms are a great way to address needs. Pediatric research by its nature is human focused and would benefit from set aside mechanisms that recognize the need to recruit from established clinical networks, such as groups of children’s hospitals working together.”
— Pediatric researcher with more than 30 years of NIH funding
“To better support pediatric researchers, NIH could prioritize mechanisms that explicitly include adolescents and emerging adults, encourage cross-institute initiatives focused on life course transitions, and expand funding for long-term cohort studies that can track early exposures through adult outcomes.”
– Recipient of NIH funding from NINDS
“There are significant opportunities to strengthen pediatric research by embracing a life course approach. Facilitating cross-institute collaborations, harmonizing age-related eligibility criteria across RFAs, and supporting cohort extensions into young adulthood would enable a more complete understanding of how early-life exposures shape long-term health.”
– Neonatal pediatric researcher and recipient of NIH funding
NOTE: NCATS = National Center for Advancing Translational Sciences; NCI = National Cancer Institute; NIAAA = National Institute on Alcohol Abuse and Alcoholism; NIAID = National Institute of Allergy and Infectious Diseases; NIDA = National Institute on Drug Abuse; NIGMS = National Institute of General Medical Sciences; NINDS = National Institute of Neurological Disorders and Stroke; RFAs = requests for application.
SOURCE: These quotes were collected from the committee’s call for perspectives from NIH grant applicants and should not necessarily be considered representative. Responses were collected by staff and synthesized by theme and anonymized to the committee (see Appendix C for the questions posted in the call for perspectives).
hospitalists on staff.5 As of the first quarter of 2025, there were over 1,500 active research protocols at the CC; approximately one-third included children under the age of 18, and 80 protocols included children under 2 years old.6 From 2018 to 2024, 7.7 percent to 13.5 percent of CC patients were pediatric (see Table 3-1). The percentage of children was at an all-time high in FY 2018 and dropped significantly in FY 2020 and FY 2021, due to the pandemic. While the number of pediatric patients has increased since the pandemic, it has not returned to pre-pandemic levels. It is also worth noting that there was a greater decline in pediatric patients than in adult patients during this time.
In 2019 the CC issued a strategic plan, People, Places, Capabilities: The NIH Clinical Center at 65, which identified pediatric health research as an area for expansion (NIH Clinical Center, n.d.). Following discussions with the CC governing board and the NIH Pediatric Research Consortium, a working group was established and charged with identifying pediatric scientific priorities particularly suited to the CC (NIH Clinical Center, 2023). A 2023 report by eight pediatric department chairs who constituted the Children’s Working Group assessed the possibility of increasing volumes of children cared for at the CC and of lowering the age of patients from 3 years to 6 months, while maintaining patient safety (CWG, 2023). It supported the expansion in the numbers and ages of children studied at the CC, while recognizing the challenges in providing adequate staffing and maintaining competence for the care of critically ill children.
Conclusion 3-5: The current portfolio of studies at the National Institutes of Health Clinical Center has been limited by the clinical acuity of participants and the availability of pediatric critical and emergency care service lines.
The findings of the working group formed the basis of the Clinical Center Pediatric Research Strategic Plan (PRSP), which was released in 2023 (NIH Clinical Center, 2023). These priorities included natural history studies, gene therapy and precision medicine, pharmacokinetics and pharmacogenomics, and metabolic and nutrition studies (see Box 3-2 for the full list of priorities) (NIH Clinical Center, 2023). In addition to these scientific priorities, the PRSP also emphasized the need for infrastructure enhancements, including a pediatric protocol development core, child-friendly imaging equipment, institutional review board (IRB) and ethical review
___________________
5 In total, the hospital has 200 inpatient beds, 11 operating rooms, 93 day hospital stations, critical care services and research labs, an ambulatory care research facility for outpatient visits, two onsite pharmacies, a blood bank, and a complex array of imaging and diagnostic services (NIH Clinical Center, 2024).
6 Webinar recording can be accessed at https://www.nationalacademies.org/projects/HMD-HCS-24-07/event/44604.
TABLE 3-1 Clinical Center’s Pediatric Capacity from FY 2018 to FY 2024
| Summary | FY 2018 | FY 2019 | FY 2020 | FY 2021 | FY 2022 | FY 2023 | FY 2024 |
|---|---|---|---|---|---|---|---|
| #peds patients | 3,377 | 3,209 | 1,764 | 1,684 | 1,821 | 2,120 | 2,269 |
| #total CC patients | 25,072 | 24,851 | 20,937 | 21,904 | 18,335 | 20,086 | 21,757 |
| %CC patients <18 yr | 13.47% | 12.86% | 8.42% | 7.69% | 9.93% | 10.55% | 10.43% |
NOTE: CC = NIH Clinical Center; FY = fiscal year.
SOURCE: Hadigan (2025).
enhancements, and workforce development (NIH Clinical Center, 2023). Improvements across these domains would serve to streamline and standardize protocols (facilitating data comparisons), enhance access to novel technologies (facilitating inclusion of pediatric populations), and deepen pediatric expertise within the CC IRB (reducing unnecessary regulatory barriers where appropriate) (NIH Clinical Center, 2023). The PRSP recommended expanding training opportunities for pediatricians at the CC and developing a directory of all pediatric research being conducted at the CC (NIH Clinical Center, 2023). While the number of pediatricians at the CC has increased (about 250 pediatricians in 2022), the strategic plan reiterated the potential for further training opportunities for pediatricians to “increase collaboration and competency” (NIH Clinical Center, 2023, p. 10).
BOX 3-2
Pediatric Scientific Priorities Identified in the 2023 PRSP
- Expanding the scope of natural history studies to support research on the continuum from diagnosis to treatment throughout the disease trajectory within the same disease
- Building a unified clinical and scientific infrastructure to support studies of gene therapy, chimeric antigen receptor T-cell therapy, and other cell therapies
- Exploring the efficacy of precision medicine interventions in rare, nonmalignant diseases
- Increasing the number of pharmacokinetic and pharmacodynamic studies to improve rational medication use and proper dosing in children
- Performing metabolic phenotyping across a variety of pediatric conditions as well as linking metabolic phenotyping in nutrition studies and assessing the impact of diet on immune phenotypes and metabolism
- Developing a cohort of all pediatric patients at the CC to measure physical and mental health and disease across disorders, along with a deeply phenotyped pediatric cohort of healthy volunteer children to establish a standard set of control samples that can be used across studies
- Increasing support for research studies in pregnant and lactating people
SOURCE: NIH Clinical Center (2023).
Key CC Partnerships
The CC is an essential partner to the NIH Intramural Research Program (IRP). Historically, the IRP has been NIH’s internal research program and has included researchers conducting basic, translational, and clinical research, as well as training programs, in federal laboratories across six research campuses and 23 ICs, including the CC (Aitken et al., 2021; Schor, 2023). More than 10 percent of NIH’s budget supports intramural research at NIH laboratories, and approximately 41 percent (492/1,200) of all NIH intramural researchers are principal investigators at the CC (NIH Clinical Center, 2024; NIH Intramural Research Program, 2022; NIH Office of Budget, 2023).
The CC has beneficial partnerships with institutions outside of NIH as well. It currently collaborates with the Children’s National Hospital and Walter Reed National Military Medical Center and is exploring further partnerships with regional pediatric centers for “safety, transfer, protocol integrity, and joint protocol development to enable pediatric trials” (NIH Clinical Center, 2023, p. 12). Additionally, the Opportunities for Collaborative Research at the CC program fosters “collaborations between extramural and intramural investigators using the U01 Collaborative Agreement mechanism to take advantage of the unique resources and patient populations available at the Clinical Center” (NIH Office of Clinical Research Education and Collaboration Outreach, n.d.).
The NIH Undiagnosed Diseases Program (UDP) is an exemplar of successful pediatric research initiatives, led by the CC, the National Human Genome Research Institute, and the NIH Office of Rare Diseases Research (NHGRI, 2025). The NIH UDP was launched in May 2008 to meet the needs of individuals and families with undiagnosed rare or multisystem diseases. These patients remained undiagnosed despite exhaustive evaluations, which for most pediatric patients in the program lasted more than 5 years (Tifft and Adams, 2014). It is part of the Undiagnosed Diseases Network, and 40 percent of the patients seen in the UDP are in the pediatric age group.
Conclusion 3-6: The National Institutes of Health Clinical Center, as the site of most intramural clinical research activities, has facilitated the performance of studies that have not been easily performed in the extramural research community—for example, long-term longitudinal studies and first-in-human gene therapy in rare diseases.
Recommendation 3-3: The National Institutes of Health (NIH) Clinical Center (CC) should continue to prioritize the current level of pediatric research, particularly on topics and using methods that are difficult to
pursue in extramural environments (e.g., longitudinal studies of rare diseases and early-stage interventional trials) as well as on the long-term outcomes of interventions. Potential mechanisms include
- Increasing capacity while ensuring safety by overcoming operational constraints at the CC. Younger and more acute patient populations should be supported in early-phase interventional and observational protocols.
- Continued, expanded, or new partnerships with children’s hospitals, academic medical centers, schools, public health departments, and other large health centers across the country to best support pediatric populations.
- Offering pediatric education and training opportunities at the CC to continue to develop the specialized workforce needed to support this research.
- Collecting and presenting data in the annual report to the NIH Pediatric Research Consortium (or its equivalent) on the proportion of pediatric research studies, support, and usage by each institute and center at the CC.
REFERENCES
AAP (American Academy of Pediatrics). n.d. Ages & stages. https://www.healthychildren.org/english/ages-stages/pages/default.aspx (accessed December 9, 2025).
Aitken, S. F., D. A. Cruz Walma, O. D. Glover, B. D. Kear, A. E. Lee, Z. D. Michel, and T. D. Vo. 2021. Early pathways to research at the NIH. Journal of Dental Education 85(1):11–15.
BLS (Bureau of Labor Statistics). n.d. Data tools. https://www.bls.gov/data/ (accessed August 14, 2025).
CWG (Children’s Working Group). 2023. Pediatric care at the NIH Clinical Center hospital. https://ccrhb.od.nih.gov/documents/02172023_pediatric_working_group_report.pdf (accessed December 9, 2025).
Ferguson, H. J., V. E. A. Brunsdon, and E. E. F. Bradford. 2021. The developmental trajectories of executive function from adolescence to old age. Scientific Reports 11(1):1382.
Forum on Child and Family Statistics. 2024a. America’s children at a glance. https://www.childstats.gov/americaschildren/glance.asp (accessed December 9, 2025).
Forum on Child and Family Statistics. 2024b. America’s children: Special issue 2024, maternal and infant health and well-being. https://www.childstats.gov/americaschildren/ (accessed December 9, 2025).
Gitterman, D. P., W. W. Hay, and W. S. Langford. 2022. 2022: The National Institute of Health and responding to new forms of childhood adversity. Children’s Health Care 52(2):117–122.
Gitterman, D. P., W. W. Hay, Jr., and W. S. Langford. 2023. Making the case for pediatric research: A life-cycle approach and the return on investment. Pediatric Research 93(4):797–800.
Hadigan, C. 2025. Response to NASEM Pediatric Group inquiry, July 24, 2025.
Hochberg, Z. E., and M. Konner. 2019. Emerging adulthood, a pre-adult life-history stage. Frontiers in Endocrinology (Lausanne) 10:918.
Modi, N. 2023. The future of perinatal research. European Journal of Pediatrics 182(5): 1935–1939.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2024. Discussion of policies, systems, and structures for research on women’s health at the National Institutes of Health: Proceedings of a workshop–in brief. Washington, DC: The National Academies Press.
NHGRI (National Human Genome Research Institute). 2025. NIH Undiagnosed Diseases Program (UDP). https://www.genome.gov/Current-NHGRI-Clinical-Studies/NIH-Undi-agnosed-Diseases-Program (accessed September 4, 2025).
NICHD (Eunice Kennedy Shriver National Institute of Child Health and Human Development). 2019. NICHD history. https://www.nichd.nih.gov/about/history (accessed October 31, 2025).
NIH (National Institutes of Health). n.d. RCDC: Categorization process. https://report.nih.gov/funding/categorical-spending/rcdc-process (accessed August 14, 2025).
NIH Clinical Center. n.d. Strategic plan introduction: People, places, capabilities. https://www.cc.nih.gov/strategic (accessed April 18, 2025).
NIH Clinical Center. 2023. Clinical Center pediatric research strategic plan. https://www.cc.nih.gov/sites/default/files/assets/home/pdf/CC-Peds-Research-Strategic-Plan.pdf (accessed December 9, 2025).
NIH Clinical Center. 2024. NIH Clinical Center data report. https://www.cc.nih.gov/sites/default/files/assets/about/pdf/2024CCDataReport.pdf (accessed December 9, 2025).
NIH Grants & Funding. 2025a. New FY 2024 NIH categorical spending data available, including an updated women’s health research category to improve accuracy and consistency. https://grants.nih.gov/news-events/nih-extramural-nexus-news/2025/06/new-fy-2024-nih-categorical-spending-data-available-including-an-updated-womens-health-research-category-to-improve-accuracy-and-consistency (accessed November 1, 2025).
NIH Grants & Funding. 2025b. RCDC categories at a glance. https://grants.nih.gov/funding/explore-data-on-funded-projects/rcdc-categories-at-a-glance (accessed August 14, 2025).
NIH Intramural Research Program. 2022. Principal investigators. https://irp.nih.gov/pi (accessed April 18, 2025).
NIH Office of Budget. n.d.-a. History. https://officeofbudget.od.nih.gov/history.html (accessed November 1, 2025).
NIH Office of Budget. n.d.-b. NIH Office of Budget homepage. https://officeofbudget.od.nih.gov/index.htm (accessed August 20, 2025).
NIH Office of Budget. 2023. Congressional justification of the NIH request for the fiscal year (FY) 2024 budget. https://www.nimh.nih.gov/about/budget/fy-2024-budget-congressional-justification (accessed December 17, 2025).
NIH Office of Budget. 2025. Price indexes. https://officeofbudget.od.nih.gov/gbipriceindexes.html (accessed November 1, 2025).
NIH Office of Clinical Research Education and Collaboration Outreach. n.d. Opportunities for collaborative research at the Clinical Center (U01). https://ocreco.od.nih.gov/new_u01/new_u01.html (accessed December 10, 2025).
NIH RePORT. n.d.-a. Exporter. https://reporter.nih.gov/exporter (accessed November 1, 2025).
NIH RePORT. n.d.-b. Frequently asked questions (FAQs). https://report.nih.gov/faqs (accessed November 1, 2025).
NIH RePORT. n.d.-c. RCDC thesaurus. https://report.nih.gov/funding/categorical-spending/rcdc-process (accessed August 14, 2025).
NIH RePORT. 2025. RePORTER. https://reporter.nih.gov/ (accessed August 14, 2025).
Rees, C. A., M. C. Monuteaux, V. Herdell, E. W. Fleegler, and F. T. Bourgeois. 2021. Correlation between National Institutes of Health funding for pediatric research and pediatric disease burden in the U.S. JAMA Pediatrics 175(12):1236–1243.
Sawyer, S. M., P. S. Azzopardi, D. Wickremarathne, and G. C. Patton. 2018. The age of adolescence. Lancet Child & Adolescent Health 2(3):223–228.
Schor, N. F. 2023. The NIH Intramural Research Program: Opportunities for training and career development in neuroscience and beyond. Annals of the Child Neurology Society 1(1):38–43.
Shulman, E. P., K. P. Harden, J. M. Chein, and L. Steinberg. 2015. Sex differences in the developmental trajectories of impulse control and sensation-seeking from early adolescence to early adulthood. Journal of Youth and Adolescence 44(1):1–17.
Tifft, C. J., and D. R. Adams. 2014. The National Institutes of Health Undiagnosed Diseases Program. Current Opinion in Pediatrics 26(6):626–633.



























