Strategies to Enhance NIH-Funded Pediatric Research: Optimizing Child Health (2026)
Chapter: 5 Supporting Pediatric Research and Addressing Challenges
5
Supporting Pediatric Research and Addressing Challenges
Pediatric research projects offer unique opportunities to reverse the current pediatric health crisis and support a healthy generation of children; the future health of those children as adults; and, ultimately, a prosperous United States. Despite this, pediatric research is often undervalued compared with adult research (Speer et al., 2023). This undervaluation can be seen in the relatively limited funding allocated to pediatric research, the lower prestige of working in pediatric fields, and the greater difficulty in translating findings into long-term benefits for children (Stoll and Taegtmeyer, 2018; Subasri et al., 2023). Furthermore, the full impact of pediatric research is not always realized because studies often focus on short-term outcomes rather than impacts on the life course (Long and Fink, 2021). This is shortsighted because the health decisions made in childhood have profound consequences on an individual’s overall health trajectory, and understanding these long-term impacts is crucial to improving public health.
In addition to being undervalued, pediatric research faces a number of other challenges due both to the complexities of studying children and their environment and the infrastructure required to support such research. Numerous barriers impede the successful execution and advancement of pediatric research. These barriers are of several types: ethical and legal concerns related to the inclusion of pediatric patients in clinical trials, intrinsic complexities related to the interplay between complex environmental influences and child development, methodological challenges and issues largely faced by researchers, structural and procedural challenges faced by research funders, and financial limitations. There are multiple strategies to overcome these barriers and thus to promote the conduct of important
pediatric research. In this chapter, both the barriers to and facilitators of pediatric research are discussed, along with recommendations to overcome those barriers and strengthen facilitators.
ETHICAL AND LEGAL IMPEDIMENTS
Despite decades of recognition of the problem, there remains an urgent need to correct the longstanding default approach of “adults first” in research funded by the National Institutes of Health (NIH) (Gitterman et al., 2018). Children’s vulnerability to harm has been used as a reason not to conduct pediatric research or to wait for data from relevant studies with adults. While there are some types of research where delayed inclusion of children may be appropriate (e.g., research with new molecular entities that have not yet been tested in humans), evidence suggests that the common practice of using a default of exclusion has harmed the interests of all children, because pediatric clinicians often lack the necessary information about how best to treat their patients. As discussed in Chapter 2, Congress and NIH have taken steps toward the increased inclusion of children in research, most notably releasing the Inclusion of Children policy in 1998 and the Inclusion Across the Lifespan policy in 2019 (NIH, 1998, 2017). Noting that 10–20 percent of research at the time inappropriately excluded children and that most drugs on the market do not have any labeling for use in children, the U.S. Senate concluded that “considerations of justice” call for the greater inclusion of children in biomedical research (NIH, 1998). As the Inclusion of Children policy explains, “It is the policy of NIH that children (i.e., individuals under the age of 21) must be included in all human subjects research, conducted or supported by the NIH, unless there are scientific and ethical reasons not to include them” (NIH, 1998). The Inclusion Across the Lifespan policy expands on this by clarifying that NIH research should be inclusive for people of all ages, including children and older adults. Yet as discussed in Chapter 3, the promise of these policies to improve the inclusion of children in research has not materialized. Instead, the amount of pediatric research funding at NIH has stalled in real terms since 2020.
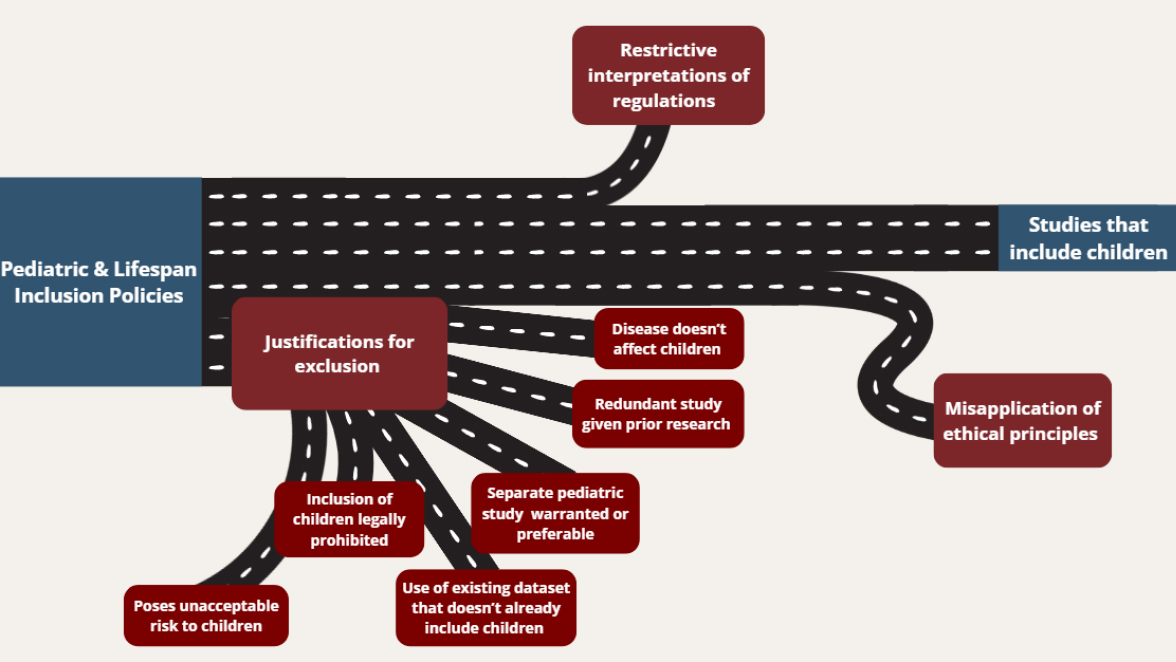
One of the reasons that these policies have not dramatically improved the inclusion of children in research may be that they have several exceptions and limited enforcement mechanisms. Investigators submitting grants to NIH are required to include a description of their plans for including individuals across the lifespan and presenting a justification for any exclusion (NIH, 2025). However, investigators deciding whether to include children can use any of six justifications for exclusion provided by these policies (see Table 5-1). The committee was unable to find any data on the frequency of using these justifications for excluding children from research.
| Potential justifications for the exclusion of children from research | Examples |
|---|---|
| Disease or condition does not occur in children or is not relevant to them. | Alzheimer’s disease research. |
| Studying children will be redundant of current or previous research. | A drug under study has already been studied and approved for use in children. |
| A separate age-specific study in the excluded age group is warranted and preferable (but consider taking account of age in study design). | A clinical trial designed to promote self-monitoring of blood glucose levels in adolescents with type 1 diabetes proposes to include only adolescents. |
| The study will collect or analyze data on pre-enrolled study participants; data for children are not part of this dataset. | A study that began prior to implementation of the NIH Policy and Guidelines on the Inclusion of Children proposes follow-up to examine long-term outcomes of individuals with the condition. The original study excluded children, and similar data are not available from a cohort that includes children. |
| Laws or regulations prohibit including children. | Regulations for protection of human subjects allow consenting adults to accept a higher level of risk than is permitted for children. |
| Study poses an unacceptable risk to the excluded group, such that their participation would be seen as unethical by institutional review boards, peer reviewers, or NIH staff. | Children are excluded from a Phase I study for a treatment that includes significant risk, including death. Evidence suggests the potential benefits to children do not outweigh the risks. |
NOTE: NIH = National Institutes of Health.
SOURCE: Adapted from NIH (2017).
While the exceptions to justify exclusion are important and reasonable as written, they may be interpreted broadly in a way that unduly restricts the inclusion of children (see Figure 5-1). One example provided is that researchers could exclude children for conditions seen in older adults, such as Alzheimer’s disease. However, pediatric research may yield insights into the precursors of Alzheimer’s disease (Assuras et al., 2025). Perhaps the broadest potential justification for exclusion is when “[a] separate, age-specific study in children is warranted and preferable” (NIH, 1998). Existing NIH guidance gives clear and specific reasons why this statement may be acceptable, such as if a condition is relatively rare in children and is already being studied in a nationwide pediatric disease research network. However, it is unclear whether this exception is actually used in the relatively narrow way in which it was intended. Additionally, studies that collect even limited information in children may offer insights that can inform future research.
As will be discussed further below, another justification for the exclusion of children is when pediatric research is not legally or ethically permissible, and the policies provide examples focused on concerns about research risks. While concerns about research risk are sometimes an important reason to exclude children from certain types of research, such as first-inhuman research or risky interventions that have limited potential benefit for children, the risk/benefit categories in federal regulations illustrate that even high-risk research can ethically and legally include children in some cases. For instance, risky research may be approvable when it includes children with serious, life-threatening conditions and the research has a high potential of offering benefits not attainable outside of the research.1 There are some examples of early and responsible inclusion of children in research, several of which were discussed in Chapter 4. As another example, in pediatric rheumatology, researchers recognized the limited therapeutic options available for children. When new biological therapies transformed treatment for adults, researchers collaborated with the Food and Drug Administration (FDA) to include children early in the research cycle with novel study designs in which each child had the opportunity to respond to the drug being studied and participate in an extension arm if they were benefiting from therapy (Ruperto, 2016).
The inclusion of children in research can therefore be consistent with existing federal regulations in ways that may not always be recognized by applicants or reviewers. Accordingly, further guidance may be needed to ensure that these exceptions are more narrowly understood so that children are more routinely included in research when it is scientifically and ethically appropriate to do so, in a manner consistent with existing policy (see Figure 5-2). It is important that scientific review groups tasked with identifying whether a particular justification is acceptable or unacceptable take care to prevent overuse of these exceptions.
Conclusion 5-1: The National Institutes of Health policies requiring the inclusion of children in research related to questions relevant across the lifespan lack adequate mechanisms for enforcement and implementation to ensure their consistency with existing policy. Comprehensive data to monitor the implementation of these policies are not systematically collected.
Beyond their function as exceptions to policies promoting the inclusion of children in research, research ethics principles and regulations may also lead to the undue exclusion of children from research, particularly when they are misapplied or misunderstood (Shah, 2021). U.S. federal regulations
___________________
1 45 CFR § 46.407 (2025).

governing research recognize that it can be important to include children in research and are designed to strike a balance between the inclusion of children and their exposure to risk. These regulations apply to research that involves biomedical interventions, psychological or behavioral research, or social science methods (Shah, 2021).
Notably, the U.S. federal regulations governing research with children have not been updated since their passage in 1983 and have some limitations that could benefit from reform (HHS, 1983; Ross, 2006; Shah and Wendler, 2010). Given the challenges associated with regulatory reform (Berkman et al., 2017), however, this committee focused on other legal and ethical promoters for pediatric research and impediments to pediatric research that may be easier to address. For instance, some regulatory provisions are ambiguous and can be interpreted in different ways by review committees, which can lead to the unnecessary restriction of pediatric research.
The interpretation of requirements that can generate barriers for pediatric research is discussed in greater detail below. First, however, existing
regulatory requirements for research with children are presented. After discussing barriers created by ambiguity or restrictive interpretations of existing regulations, the section closes with a discussion of ethical safeguards that are commonly used but not required by regulation, highlighting how these can sometimes be impediments to pediatric research that is ethically appropriate.
Regulatory and Legal Requirements That Apply to Research with Children
Regulatory requirements in pediatric research are more stringent than those for adult trials. Children are considered a vulnerable population, which means additional safety, ethical, and legal safeguards must be in place when conducting pediatric research. Children are generally defined by law as individuals who are not old enough to have the capacity to make their own decisions (see Box 5-1). As discussed in Box 5-1, U.S. federal regulations defer to state and local laws on who is considered a child.
The U.S. federal regulations permit institutional review boards (IRBs) to approve three categories of pediatric research involving low risks or where the benefits outweigh the risks:2
- Pediatric research with minimal risk,
- Pediatric research in which the prospect of direct benefit outweighs risk, and
- Pediatric research involving a minor increase over minimal risk with no prospect of direct benefit.3
Ambiguity In the Interpretation of Requirements That Impede Pediatric Research
One key source of ambiguity in the regulatory requirements for research with children is the issue of how to identify the risk level. While the regulations define minimal risk as “the risks of everyday life or of routine examinations or tests,” they do not define other levels of risk or benefit.4 As discussed above, to protect pediatric participants federal regulations impose different requirements based on the risks posed by the research and whether the risks are justified by the prospect of direct benefit. Moreover, difficulties
___________________
2 Research involving greater than minimal risk and no prospect of direct benefit to individual subjects but likely to yield generalizable knowledge about the subject’s disorder or condition; 45 CFR § 46.406 (2025).
3 45 C.F.R. 46, Subpart D.
4 Definitions for purposes of this policy; 45 CFR § 46.102i (2025).
in recruiting pediatric participants mean that many studies must enroll at multiple sites in order to obtain enough participants to answer a scientific question definitively. One study found wide variability in IRB judgments about the risks of common biomedical research procedures (Shah, 2004). A more recent study similarly found variability in identifying what counts as minimal risk in certain research procedures involving pregnant people, along with a reluctance to approve studies that would count as minimal risk and be approvable under existing regulations (White et al., 2021). The available data suggest that IRBs tend to use restrictive and variable interpretations of risk and benefit as well as rigid approaches to assent and consent that may not reflect the developmental capacities of pediatric populations (Kodish et al., 2004). As a result, studies may be delayed, redesigned, or prevented altogether. Although waiting to include children may be appropriate for some early studies that pose high risk and uncertain benefit, particularly if they are testing interventions that are not urgently needed for children, these barriers can cause delay when research poses minimal risk and has strong potential for benefit.
Confusion about what is required for research decision making can also lead to delays in including children. Under the U.S. regulations governing research, children should give their agreement to participate (“assent”) if they are capable, and parents or guardians are also asked to consent. For anything that does not fit in these categories, the Secretary of the Department of Health and Human Services can convene a special panel that can review and approve the research.5 For research with older children, the question of when they can provide their own consent is difficult to determine (Leibson and Koren, 2015). As noted in Box 5-1, the U.S. federal regulations differ from state and local laws on who can consent and allow independent adolescent consent in a range of circumstances, but most state and local laws do not directly address consent to research. This leaves IRBs to decide when the laws that allow adolescents to consent for themselves based on their status, condition, or maturity can be applied to informed consent for research. The existing patchwork of laws and the room left for interpretation can make it challenging to determine when adolescents can participate in research without parental permission, particularly for studies that are conducted in multiple states (Gilbert et al., 2015). This also leads to skewed data from the studies that are conducted, making it challenging to include the populations at greatest risk or in greatest need of intervention and to know whether interventions work for them (Liu et al., 2017).
The vagueness of assent requirements can lead to challenges and delay for pediatric research. A lack of clarity about assent may lead some
___________________
5 Research not otherwise approvable which presents an opportunity to understand, prevent, or alleviate a serious problem affecting the health or welfare of children; 45 CFR § 46.407 (2025).
BOX 5-1
U.S. Federal Regulations for Pediatric Research Definition of a Child
The U.S. federal regulations governing research involving children as participants define children as “persons who have not attained the legal age for consent to treatments or procedures involved in the research, under the applicable law of the jurisdiction in which the research will be conducted” (45 CFR 46.402[a]). Laws that determine who can consent for themselves to treatments or procedures vary across U.S. states and even cities in some cases (Fogler and Lerman, 2017). In general, jurisdictions define an age of majority when people are considered able to consent for themselves; for most states in the United States, this age is 18 (Guttmacher Institute, n.d.). States typically also have a patchwork of exceptions allowing minors to consent for themselves before they reach the age of majority for several reasons, including the importance of ensuring prompt treatment for communicable diseases to promote public health, the need to remove barriers to treating children who are not living with their parents, and the recognition that children do not suddenly become able to make their own decisions on the day they turn 18. The three main categories of exceptions allowing minors to consent are based on condition (e.g., sexually transmitted infection), status (e.g., marriage), and designation (e.g., a determination by a judge or clinician that a minor is mature enough to make their own decisions or emancipation from one’s parents through legal proceedings) (Guttmacher Institute, n.d.). In the United States, states generally do not have laws that specifically address consent for research, and the IRB must determine when laws addressing consent for treatment can be applied to research (HHS, n.d.).
investigators to believe that research with children is more complicated than adult research and may lead to a sense that it is easier to start with research in adults in all cases. Confusion about assent could also cause researchers to obtain assent when it is not necessary. One prominent group of scholars has argued that assent should not be obtained in two cases: (1) the child lacks capacity to assent for various reasons, or (2) “the research offers benefits to the child that are substantially greater than those available outside the research and so joining the study is unequivocally in the child’s best interests” (Joffe et al., 2023, p. 464). These authors do note that children should still be given developmentally appropriate information about the research and engaged by research teams, even if they are not capable of assent and it does not make sense to ask them for their agreement to participate (Joffe et al., 2023).
State and local laws also impose mandatory reporting requirements for certain categories of health professionals that can introduce barriers for pediatric research (Kingsley et al., 2024). These requirements are typically justified by the importance of protecting individuals or the public’s health. They range from requiring people designated as mandatory reporters to share information with departments of public health about certain communicable diseases to requiring that information about child abuse or neglect be shared with child protective services (Kingsley et al., 2024). For example, pediatric researchers in Pennsylvania may encounter challenges to conducting research related to adverse childhood experiences (ACEs) due to the state’s mandated reporting laws, which apply to all licensed health professionals. This legal obligation can introduce complex ethical and logistical considerations, as inquiring about participants’ or their parents’ ACE exposure may generate requirements to report to state authorities, thereby complicating both study design and participant engagement.
Additionally, some requirements imposed by agencies such as the FDA for adult research may apply to pediatric research and thus raise special challenges. For example, the FDA recommends pregnancy testing for phase I and II clinical trials in any person who has reached menarche, which applies to children who have gone through puberty (FDA, 2025; Thomas and Reeves, 2023). This requirement can be complicated to implement in pediatric research if parents are uncomfortable with their young child undergoing pregnancy testing. The length of follow-up required can also be challenging to determine for pediatric research, depending on the outcome of interest. For instance, studies testing cooling for birth asphyxia and other interventions thought to have neuroprotective effects may require endpoints that are longer in pediatrics in order to capture whether there are developmental delays. Although adult studies sometimes address this issue by developing surrogate endpoints, surrogate endpoints are not available for many pediatric outcomes of interest and may not be appropriate (Green et al., 2019).
Conclusion 5-2: More flexibility in approaches to assent could give investigators the ability to tailor the provision of information and level of engagement to a particular child’s abilities.
This variability can be a significant problem for multi-site studies, as it can extend the time needed to conduct research. While single IRBs are now mandated under the Common Rule, there is local variation in familiarity with procedures and relevant variation in state laws, as discussed further below, which may make it hard to determine how much local review is still needed. In some cases, local IRBs may be hesitant to rely on a single IRB and insist on conducting their own review, and IRBs may have differing
interpretations about the risk level of a given study, which can lead to requirements that vary based on the site (Hu et al., 2022). More work is needed to implement the process of single IRB review for pediatric research, along with additional guidance for local IRBs on ethical issues that are widely misunderstood or are addressed in variable ways. Additionally, the process for approving studies that do not fit into existing risk/benefit categories is slow and cumbersome and could benefit from greater transparency and efforts to increase efficiency (Jennings et al., 2025).
There has been significant controversy concerning whether children whose biological samples were included in biobanks should reconsent to continued use of their samples when they turn 18. While some researchers conducting studies with stored samples opt to reconsent participants who reach the age of majority, others de-identify the sample instead by destroying the information connecting the sample to a particular person. De-identification is one way to preserve privacy but makes it difficult or impossible to share findings that may emerge at a later date, some of which may be clinically actionable or even life saving. In contrast to the standard approaches to stored sample research, some have argued that reconsenting or de-identification may not always be ethically necessary. Parents make many decisions for their children that can influence their lives even as adults, ranging from the schools they attend to the medicine they take, and people who reach the age of majority are not routinely asked to revisit all of these decisions. Given the burdens associated with reconsent, it is unclear that the practice of reconsenting should always be required (Berkman et al., 2018).
Other Ethical Safeguards That Can Stymie Pediatric Research
Another source of ambiguity is deciding when ethical concerns about pediatric participants should be addressed by incorporating additional safeguards, such as extended safety monitoring. These additional requirements may sometimes be justified to protect participants and future pediatric patients but can increase the cost and complexity of pediatric studies. In the worst-case scenario, additional requirements for pediatric research can lead to efforts and funding being directed toward adult-focused research if risks and other ethical complexities are perceived as easier to surmount.
A further contributor to delayed pediatric inclusion may be the common practice of enrolling older children before younger children (i.e., “age de-escalation”) without questioning when it makes sense to do so (Harbin et al., 2023). As discussed in Chapter 4, the practice of age de-escalation has been raised for studies even when there is little difference in the risks of research for children across different ages, such as in the All of Us study. Furthermore, the forgone benefit of not receiving access to research may
be higher for younger children in some cases if they have greater disease severity and few treatment options. Age de-escalation may be an important safeguard in some cases but should not be applied across the board.
One final ethical issue that may stymie pediatric research is the perceived need to limit payment to avoid undue inducement or coercion (Gelinas et al., 2018). Undue inducement is the concern that people deciding on research participation will be so blinded by the money that they disregard the risks or act in ways that go against their values. Coercion is often mistakenly raised as a concern that offering payment will force someone to participate. Offering payment expands the options available to people and does not involve a threat to take away what they are entitled to or a way to force them to do something against their will (Wertheimer and Miller, 2008).
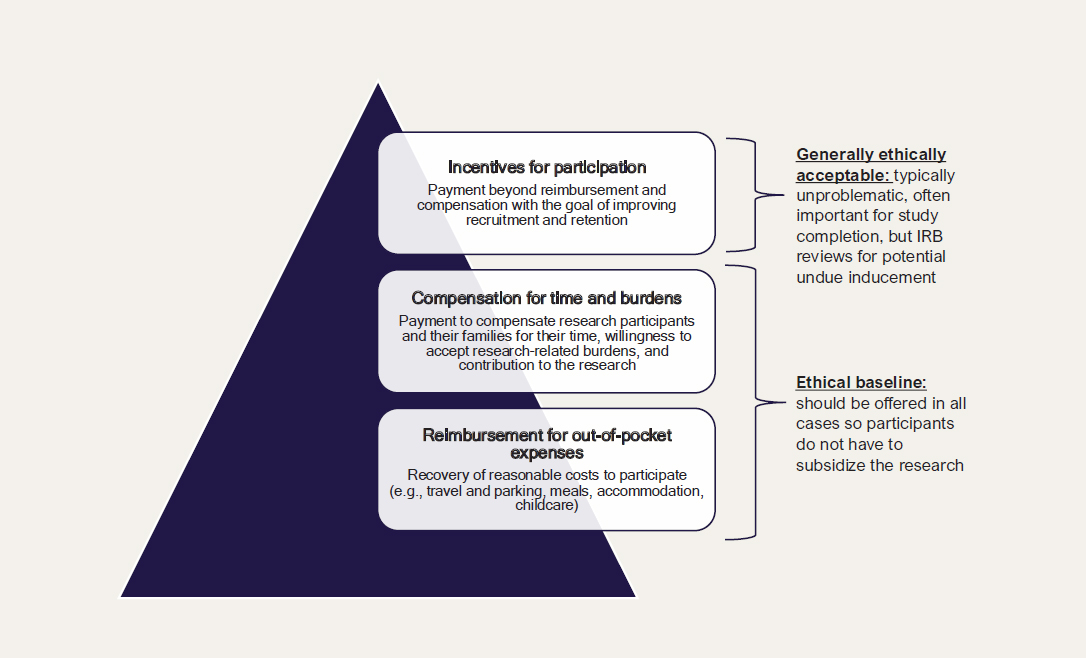
In fact, payments in pediatric research can be valuable to (1) reimburse caregivers for the costs they incur to participate in research, (2) compensate caregivers and children for their time, or (3) show gratitude to and incentivize caregivers and participants to take on the burdens of research participation (see Figure 5-3). First, reimbursement is important because caregivers may have to pay for childcare, transportation, and parking in order to attend research visits. Without consideration of the costs of research participation, families are essentially being asked to subsidize the research.
Second, paying caregivers and participants more than is needed to cover their costs is not coercive, but it may serve to compensate them for the time spent on research and make it possible for them to choose to contribute to research rather than doing something else. Finally, incentives can be a way to ensure that families feel their contributions are valued and keep coming back to research visits over time (Bierer et al., 2021; Shah, 2021).
While high payments may lead to worries about undue inducement or concerns that parents might pressure their children to participate in research studies, there are other ways to address this concern rather than reducing compensation (Diekema, 2018; Gelinas et al., 2018; Wendler et al., 2002). Research should be reviewed and determined to be ethically acceptable, consent and assent (if appropriate) should be obtained, and dissent should be respected unless the research offers potential therapeutic benefits that the child could not otherwise receive. With those safeguards in place, it may be less important to worry about paying families too much for participation and more important to worry about not paying them enough (Largent and Lynch, 2017).
As these examples illustrate, the reflexive application of ethical dogmas—as opposed to the thoughtful application of ethical principles—can lead to delayed inclusion of children in research. Exclusion is one response to vulnerability. Another potential response is responsible inclusion. Research can be designed to account for and respect the vulnerabilities and needs
NOTE: IRB = institutional review board.
SOURCE: Content from Gelinas et al. (2018).
of different children, implementing safeguards for children based on their vulnerabilities. As has been recognized with other groups previously considered “vulnerable,” such as pregnant people, it is time to recognize the importance of protecting children through research, not from it (Lyerly et al., 2021). Ultimately, shifting the default toward inclusion to realize the promise of existing federal policies that promote the inclusion of children, along with ensuring that ethical safeguards are applied appropriately, will help advance the health and well-being of all children.
SCIENTIFIC CHALLENGES TO CONDUCTING PEDIATRIC RESEARCH
Pediatric researchers face numerous scientific challenges due to the inherent physiological and behavioral differences between children and adults, the complexity of diseases that affect children, and the limitations in current research models and scientific approaches in fully unraveling the intricacies of pediatric diseases (EPA, 2025). These barriers hinder the ability to generate accurate and meaningful data and to refine pediatric disease models that are used to understand the mechanisms of diseases affecting children and to test potential therapies. Without the best data and models, it is much more difficult to inform the care and treatment of pediatric populations.
Biological Differences Between Children and Adults
There are significant differences in developmental variability, growth, and maturation between children and adults. Developmental variability impacts infants and young children as well as adolescents, who have developing organ systems, immune responses, and metabolic rates that are different from adults. Infants, children, and adolescents also have developing psychosocial characteristics, including cognitive skill, patterns of social engagement, and perceptual and motor abilities. In the case of disease and injury, the risk, progression, and treatment responses may vary across these developmental stages, making it difficult and sometimes inappropriate to translate adult disease models to pediatric populations. It is also important to take into account growth and maturation in basic science projects and prevention/intervention programming, as pediatric models often need to account for age-specific factors such as hormonal changes, brain development, and skeletal maturation, which may not be well represented in animal models or adult human models. For example, humans accrue approximately 25 percent of peak bone mass during the 2-year period encompassing peak height velocity (Wojtys, 2020). Threats to bone health during that period
may have very different lifelong implications than the same threats a few years later when skeletal maturation is complete.
Limited Availability of Pediatric Participants and Samples
The relatively small size of certain pediatric populations, particularly those with rare diseases or specific genetic conditions, can make it difficult to conduct large-scale studies. Without enough participants, the power of studies is limited because small sample sizes make it challenging to generate statistically significant findings and more difficult to identify trends or differences that might be important in understanding pediatric conditions. Furthermore, small trials may have limited generalizability because findings from a small homogenous group of children may not apply to the broader pediatric population. Collecting tissue samples or clinical data from children presents significant challenges, particularly for very young children and neonates with heightened vulnerability to the potential risks associated with invasive procedures, which must be carefully addressed in the research design. The combination of small participant pools and limited material for analysis means there may be an insufficient amount of data for accurate modeling. To generate sufficient sample sizes and statistically meaningful data, multi-center studies are not only common but often essential in pediatric research studies. A previous Institute of Medicine report drew a similar conclusion:
Because most children today are healthy, children suffering from serious conditions such as cancer or heart disease are relatively few in number compared with the number of adults who have such conditions. For any one research location, the numbers available for a study are usually quite low. For many pediatric studies, the relative scarcity of potential study participants means that it often takes considerable effort and some creativity to enroll and retain sufficient numbers of children who meet the criteria for study participation. It may also mean that studies must extend for quite long periods just to secure enough participants. (IOM, 2004, p. 80)
Genetic and Environmental Variability
Health outcomes in children are shaped by a complex interplay among environmental exposures, family factors, and genetics, and these influences vary significantly across populations. There are numerous genetic diseases that manifest early in life. Pediatric models, thus, need to incorporate a wide variety of genetic mutations, which can make the models complex and difficult to study. Environmental factors (e.g., infections, nutrition, toxins) can influence disease development and response to treatment in children, but
these factors are difficult to incorporate into animal or laboratory models. Pediatric research must take into account a wide range of environmental determinants—air and water pollution, nutritional access, housing quality, neighborhood safety, the availability of health care services, and so on—which often interact with inherited genetic risk factors in ways that are not yet fully understood. These factors do not operate in isolation; rather, they are embedded within broader social and structural contexts that influence children’s development and disease risk. ACEs, which have been established to have long-lasting effects on a child’s health into adulthood, are linked to “to the social and economic environments in which some families live” (CDC, 2025). Risk factors for ACEs include, but are not limited to, children with few or no friends, families with low incomes, and families with adults who have low levels of education (CDC, 2025). Collecting data from diverse populations is essential to uncovering how different environmental and genetic factors converge to influence the onset, severity, and progression of pediatric diseases. Such insights are critical not only for improving diagnostic accuracy and therapeutic interventions but also for advancing health equity and tailoring prevention strategies to the needs of specific communities.
Complexity and Rarity of Pediatric Diseases
Based on the limited number of individuals affected, nearly all pediatric diseases can be considered as rare diseases. A rare disease is defined by FDA and the National Cancer Institute as affecting fewer than 200,000 individuals per year (FDA, 2024; NCI, n.d.). Furthermore, children are also affected by rare and heterogeneous conditions with complex manifestations. Disorders like childhood cancers, genetic syndromes, or rare metabolic disorders may present differently at different developmental stages, making it difficult to develop a one-size-fits-all model. Pediatric diseases often involve multiple organ systems, making it hard to replicate their full complexity in a single model. This can hinder the development of the treatments needed to address systemic effects.
Challenges in Translating Animal Models to Humans
Basic and translational science studies using animal model organisms, such as simple organisms (e.g., zebrafish, fruit fly, and nematode), mice, or large mammals, play a crucial role in advancing pediatric research (Ogden et al., 2021; Tain et al., 2025). While differences exist between species in immune systems, metabolism, and developmental trajectories, these models provide indispensable opportunities to study biological mechanisms that cannot be ethically or practically examined in children. Certain
investigations, such as the long-term follow-up of early-life interventions, intergenerational impacts, or life course studies, are feasible only in animal models, given the lifespan limitations of human studies.
Animal research has been foundational for numerous pediatric breakthroughs, including prenatal–neonatal therapies, gene therapy for monogenic disorders, and interventions for metabolic diseases (Farooqi and Xu, 2024; Tain et al., 2025), necrotizing enterocolitis (Kovler et al., 2020), and cystic fibrosis (Ogden et al., 2021). Even studies in complex neurodevelopmental disorders, when designed carefully, allow researchers to establish cause-and-effect relationships by isolating specific genes or factors before translating findings to children, who are influenced by diverse genetic, environmental, and social contexts.
Although complementary studies in human tissues, cells, or organoids can provide additional insights, organoids mainly reflect early embryonic development and cannot fully capture the developmental stages of childhood organs or the long-term effects of interventions. Therefore, animal models remain essential for determining in vivo effects, safety, and the mechanisms of novel therapies before human trials.
To maximize translational relevance, it is recommended that research teams conducting animal studies include pediatricians or pediatric clinician–scientists. Their expertise ensures that preclinical work aligns with child health priorities and can be effectively translated into clinical interventions. This approach mirrors successful maternal studies funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), where inclusion of obstetricians was required to ensure relevance.
Maintaining a robust pipeline from animal research to clinical application has been a distinguishing feature of U.S. pediatric research, contributing to life-saving interventions such as surfactant therapy, nitric oxide treatments, fish oil supplementation, and targeted gene therapies. Continued careful integration of preclinical animal research into pediatric studies is essential to sustain innovation, improve child health outcomes, and uphold the United States’ preeminent position in translating basic science into clinical breakthroughs.
Difficulty in Assessing Pediatric Treatment Responses
Children and adults metabolize drugs differently due to differences in liver enzymes, kidney function, and many other physiological factors. Therefore, in many cases pharmacokinetic data from adult models cannot be directly applied to pediatric populations (Fernandez et al., 2011). Furthermore, the tools used to measure disease progression or treatment efficacy in adults may not be suitable for use in children. For example, in
developmental diseases or pediatric cancers, standard biomarkers or imaging techniques may not be as effective for assessing outcomes in children.
METHODOLOGIC BARRIERS TO CONDUCTING PEDIATRIC RESEARCH
Developing research studies that include human participants requires navigating a number of methodologic considerations, including participant recruitment (as discussed further in the section addressing financial barriers), the diversity of participants, and data collection and measurement. Each of these takes on an additional degree of complexity when developing pediatric health research. These methodologic challenges are more than important considerations for good study design. Left unaddressed, they generate structural barriers to pediatric representation in NIH-funded research that hinder advances in pediatric health.
Psychosocial Factors
Disease can have a significant impact on normal developmental processes. Chronic illness in childhood affects cognitive, emotional, and social development, and these impacts on development can complicate the study of disease progression and treatment (Turkel and Pao, 2007). The psychological burden of disease may affect children’s ability to participate in clinical trials or models. Furthermore, pediatric disease models often need to consider long-term quality-of-life outcomes, which can be harder to measure and predict in young patients and require long-term follow-up research.
Cognitive Development
Children continuously undergo cognitive development, which complicates research assessment and treatment. When measuring pain, for example, young children have difficulty expressing verbally the level of pain they are experiencing. Infants are unable to do so at all, except through crying and nonverbal cues (Thrane et al., 2016). When measuring depression, children may express depressive thoughts in ways that differ from adult expression (Mullen, 2018). Young children are unable to compare themselves with peers who are functioning with different mental health (Levy-Friedman and Kogut, 2024). Therefore, when gathering data from children that can be used to understand, prevent, and treat their illnesses and diseases, it is often necessary to use innovative, complicated, and creative assessment and treatment strategies, which are frequently more burdensome than those used for adults (Jonsson et al., 2017).
Inclusion of Rural and Underserved Populations in Pediatric Clinical Studies
Ensuring that the results of pediatric research are generalizable across diverse populations is critical for creating applicable, impactful findings. Children from rural areas and those living in poverty face distinct health risks and barriers to accessing health care. These children are often underrepresented in clinical trials, which means that their specific needs and responses to treatment may not be well understood. Expanding access to clinical trials for these populations is essential to ensuring that all children, regardless of their geographic location or socioeconomic status, are included in research and benefit from advances in pediatric research. To increase the participation of rural and underserved children, pediatric researchers need to meet children in their communities, homes, and local clinics. This may require developing or using infrastructure that is local to these underrepresented populations, such as collaborating with regional hospitals and outpatient clinics, community centers, K–12 schools, and religious establishments; establishing satellite locations; and, when feasible, using telehealth and medical devices that can collect, store, and transmit health information securely from participants to researchers.
Ensuring that pediatric research is inclusive and generalizable can be particularly challenging. Trials may unintentionally exclude certain groups, such as children with disabilities or those from underserved communities as well as pregnant women, leading to a lack of representativeness in research findings. The geographic concentration of pediatric clinical studies also contributes to greater direct and indirect costs by requiring a greater number of sites to meet enrollment benchmarks. For pediatric health studies to succeed, they must be provided sufficient funds to meet these challenges to pediatric recruitment. Failing to enable broad representation in studies can lead to gaps in pediatric representativeness, which can cause pediatric harm in real-world clinical practice (Sullivan-Bolyai et al., 2007; Winter et al., 2018).
Family Management Considerations and Gendered Roles
Family dynamics play an important role in pediatric research because the involvement of parents or caregivers is crucial in the recruitment and ongoing participation of children in research. In one study, parents reported that the primary motivators for study participation were health benefits for the child and altruism. The most common barriers to participation were the risk to the child and the burdens of participating (Nathe et al., 2023).
Gendered Roles and Bias
In many households, socialized expectations and norms about gender and caregiving roles can limit parent participation in research. Across the globe, even in countries well known for policies supporting gender equality, mothers engage in more parenting responsibilities than do fathers (Mark et al., 2025). This reflects a broader trend in which women report bearing more of the mental load when it comes to parenting and household tasks more generally. Parenting, general domestic labor performance, and breakdown among heads of a household can be affected not only by socialized gender roles but also by class status, racialized and ethnic identity, and economic and public policies. Overall, women and mothers are more likely than men to quit their jobs and stay at home for a child with a complex medical condition (Werner, 2023); and at specialty outpatient pediatric clinics, 74 percent of mothers attended with their children compared with 44 percent of fathers (Mark et al., 2025). In a systematic review of paternal involvement in pediatric obesity prevention trials, Morgan and colleagues reported participation rates by fathers as low as 6 percent (Morgan et al., 2017). This evidence base is further limited by the lack of research on historically underrepresented groups. Increased involvement from fathers in pediatric research will provide a more complete picture of child health and increase opportunities for family-centered research.
Impact of Caregiving on Participation
Caregiving responsibilities, especially in the context of complex pediatric conditions, can limit a family’s ability to participate in studies (MacNeil et al., 2025; Page et al., 2020). These challenges are exacerbated by social or financial difficulties, including the need for transportation, access to technology, language barriers, childcare, and time away from work. Addressing these barriers requires that researchers consider flexible trial designs that accommodate the varying schedules and needs of families and that funding is sufficient to cover the cost of transportation, childcare, and missed work.
Data Acquisition Challenges
Pediatric researchers face several data collection challenges that are not typically problems for health research involving adults. First, many pediatric research studies require longitudinal data collection to be able to observe relevant outcomes across the developmental course. Creating the capacity for such longitudinal data collection and storage is more labor-intensive and costly than the typical R01 (5 years of funding) allows (Flores-Toro et
al., 2023; Oken et al., 2023). Second, data collection in pediatric studies must evolve as participants transition from childhood to adulthood (Oken et al., 2023; Stepien et al., 2021). Third, many research tools have not been validated across all age groups. As a result, existing research initiatives may not adequately translate to pediatric populations (Collins et al., 2022). Fourth, many outcomes in childhood are best understood within the broader context of family and household factors, where parental health, caregiving practices, and family stressors strongly shape child development and well-being (Masarik and Conger, 2017; Osher et al., 2018; Szatmari et al., 2016; Ward and Lee, 2020). Capturing these interdependencies requires approaches that extend beyond the individual child to include parents, caregivers, or siblings as part of the analytic unit. However, pediatric research lacks established standards for dyad- or family unit–based data collection. The absence of common frameworks limits comparability across studies, constrains longitudinal follow-up as children mature into adulthood, and hampers the development of interventions that target both the child and family system. National data systems such as Medicare and the Veterans Administration are less available for pediatric populations than for adult populations. Establishing robust methods for dyadic and family data collection—such as harmonized questionnaires, parallel outcome measures across participants, and models that account for interdependent data—will be essential for advancing pediatric research and strengthening its translational impact.
These pediatric research–specific challenges around data collection and infrastructure complicate the incorporation of pediatric research into NIH-wide efforts that use adult-oriented frameworks. Advancing pediatric health outcomes through NIH-wide initiatives will require accommodating the longer data collection times needed and establishing improved standards for data collection that can accommodate pediatric research needs. For example, surrogate endpoints (i.e., biomarkers) in pediatric research for FDA studies could shorten the time required to collect enough data for the FDA to consider approving a drug for use in children (Green et al., 2019). In addition to modifying data collection standards, the development of standardized measurement protocols for pediatric research will be important for comparing and harmonizing studies as well as for following children across multiple developmental stages. A valuable resource, funded by NIH, is the Phenotypes and Exposures (PhenX) Toolkit, which includes a pediatric development research domain with a wide variety of freely available protocols (Enlow et al., 2022; RTI International, 2025). NICHD has a Data and Specimen Hub (DASH) that includes protocols, manuals of operation, case report forms, and data. Some of the studies collect family, lifestyle, demographic, and other types of data (NICHD, n.d.).
Conclusion 5-3: There is a need to invest further in pediatric-oriented measurement science at the National Institutes of Health (NIH), which would expand the tools available to support data collection for all age ranges in NIH-wide initiatives.
Adjustments to Study Design
Research designs tailored for adults often need to be adjusted for studying children, because the physiology, pharmacokinetics, cognitive skill, and responses to treatment of children differ significantly from those of adults. Developing age-appropriate research requires specialized knowledge and infrastructure. For example, implementing a pediatric physical therapy program may require child-sized equipment, sensory-friendly spaces, and trained staff who can adapt exercises to different developmental stages. Pediatric clinical trials require cautious and careful determination of safety factors and drug dosing to account for the physiological and pharmacokinetic properties of children, which may impact participants’ level of risk.
Addressing Barriers to Pediatric Health Research Through Community Engagement
To establish partnerships, researchers have to meet children where they are and collaborate with community organizations to connect with children and their families. Because enrolling participants in pediatric research requires asking parents or guardians to enroll their children in a study that may pose risk, pediatric research arguably requires greater trust in research institutions than does adult research. Entire communities may be deterred from participating in pediatric research if researchers do not, for example, practice cultural humility and linguistically appropriate engagement. The use of interpretation and translation services is essential for engaging children and families who are non-English-speaking or for whom English is not their primary language (Chong et al., 2025). Some communities have historical or ongoing reasons not to trust the health care system and the research enterprise (George et al., 2014). Many of these concerns and issues of mistrust may be mitigated by using community engagement tools (Reber et al., 2025; Sprague Martinez et al., 2018). Meaningfully engaging communities through building lasting partnerships is increasingly recognized as a component of research that is ethically important and that can help to address these barriers to research (Emanuel et al., 2004; Sprague Martinez et al., 2018).
Community engagement in research is not only valuable in and of itself, but it can also serve a number of goals, including increasing trust, transparency, and buy-in; understanding how to minimize risks of research based on the community’s understanding of the local context and culture; addressing
misinformation; and ensuring that research is responsive to community needs (Hood et al., 2010; Shah et al., 2020). Approaches to community engagement vary widely and can be conceptualized on a spectrum from no community engagement to community control (Arnstein, 1969). Sharing information and seeking input with communities (community notification and consultation) are more limited forms of community engagement, while strategies for coproduction and cocreation, such as community-based participatory research, are considered the highest level of community engagement. More than one strategy may be used within a single study, and the level of community engagement needed or even desired by the community may vary from one study to another. More robust engagement in research could help to increase trust and transparency, ensure that researchers can address actual (as opposed to hypothetical) community concerns, and ultimately ensure the success of pediatric research.
The creation of patient and family advisory councils (PFACs, also called patient advisory boards) is one increasingly common approach to community engagement. This approach can be particularly relevant for studies that include children with complex health conditions (Richard et al., 2020). While PFACs can provide essential insights into study relevance, burden, and feasibility, their use in pediatric research remains uneven (Lewis et al., 2025). Some NIH requests for application (RFAs) have added community partners to the application (e.g., community-partnered nursing research centers, the Environmental Health Sciences Core Centers Program) (NIEHS, 1999, 2024; NINR, 2025). Making the use of PFACs routinely could better align research with the lived experiences and priorities of young participants and their families. Increasing access to guidance on recruiting PFAC members, educating families about the value of pediatric health research, building trust between families and researchers, and providing training for all parties on how to engage in a cooperative research protocol will facilitate the inclusion of PFACs into pediatrics research.
Addressing Contextual Barriers Rhrough a Focus on Dissemination and Implementation Research
Lessons from implementation and dissemination research may also offer new ways to lower barriers to the successful implementation of interventions that promote pediatric health. Implementation and dissemination research can help ensure that an intervention accounts for the context in which it will be implemented, whether that is a clinical or community setting; is delivery by a clinician, teacher, or caregiver; or differs based on what is developmentally appropriate (Smith and Polaha, 2017). NIH defines implementation science as the “study of methods to promote the adoption and integration of evidence-based practices and interventions into routine
health care and public health settings to improve our impact on population health” (NCI, n.d.). When investigators imbue their research with a focus on translation, implementation, and dissemination, they can also promote trust-building among the communities with whom they are conducting research, by integrating co-creation and co-production approaches into their research, including soliciting feedback on research questions with the intervention context in mind (Israels et al., 2024).
Through contextually tailored dissemination and implementation strategies such as stakeholder engagement, adaptation of interventions to local needs, and iterative evaluation, researchers can enhance the relevance, uptake, and sustainability of pediatric health innovations across diverse populations. Table 5-2 shows that there has been an increase in pediatric dissemination and implementation funding at NIH from fiscal year (FY) 2021 through FY 2024. Incorporating considerations about implementation and dissemination for interventions and treatments for pediatric populations can mitigate gaps between new research findings and actual use in clinical or community settings (Halle, n.d.; Samanta, 2020; Wittmeier et al., 2015).
Conclusion 5-4: The translation, implementation, and dissemination of new research findings into practice and policy are needed to improve the health of all children.
STRUCTURAL AND PROCEDURAL CHALLENGES AND OPPORTUNITIES
Structural and procedural obstacles to conducting pediatric research include challenges involving grant mechanisms, scientific review processes, and administrative hurdles. Conducting pediatric research often requires additional time for recruitment, participant consent, and independent review.
TABLE 5-2 R18 or Pediatric Dissemination and Implementation Funding, FY 2021–FY 2024
| FY | # of Eligible Pediatric Projects | Eligible Amount ($M) | % of Total Pediatric |
|---|---|---|---|
| FY 2021 | 451 | 187.3 | 3.43 |
| FY 2022 | 581 | 244.7 | 4.29 |
| FY 2023 | 700 | 280.2 | 4.53 |
| FY 2024 | 829 | 318.9 | 5.15 |
NOTES: FY = fiscal year. Summary of inclusion criteria: Activity code R18 (“Research Demonstration and Dissemination Projects”), OR Research, Condition, and Disease Categorization (RCDC) category includes “Dissemination and Implementation Research” or “Dissemination/Implementation.”
The need to establish a network of study sites along with forming collaborations with people and institutions in underrepresented communities can also extend the timeline for a pediatric research study. Additionally, pediatric clinical studies may require extended follow-up periods to track long-term outcomes in children. Given children’s developmental trajectories, measuring outcomes at 6 or 12 months after intervention may be inadequate to assess the true effects of an intervention. These extended study timelines often do not align with the typical period of an NIH award.
Grant Mechanisms
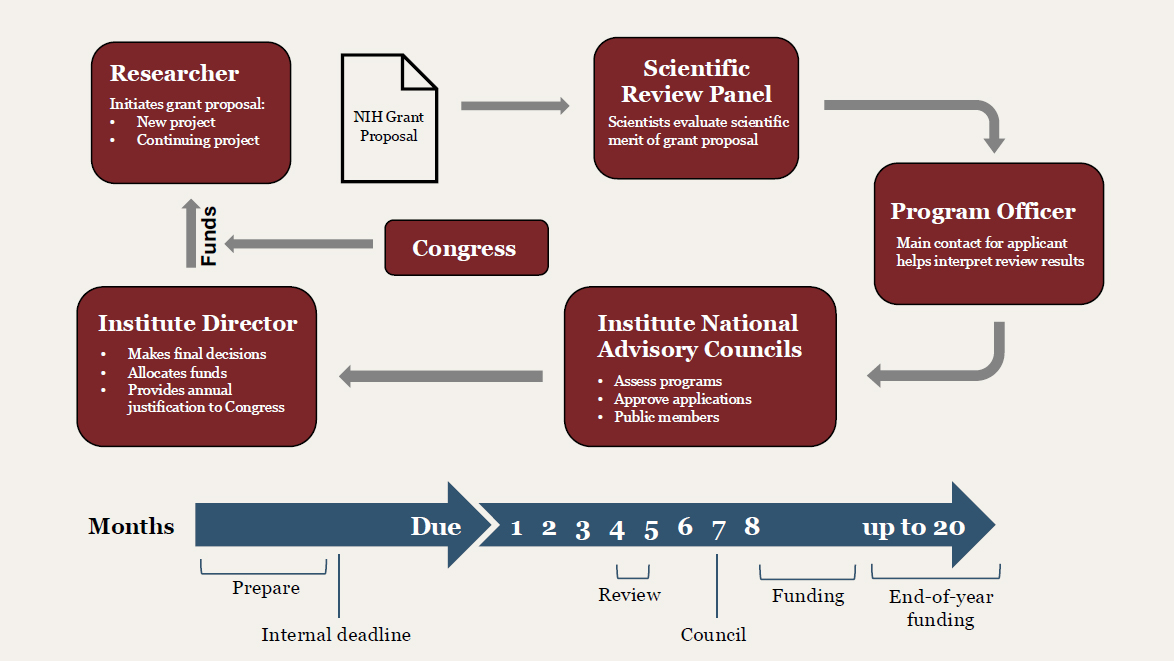
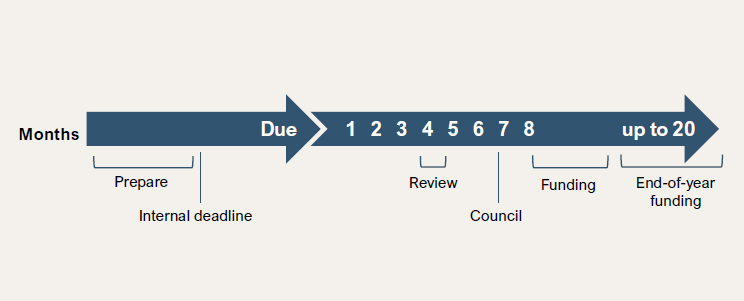
Most grant mechanisms have a timeline of 9 or more months from application submission to receipt of a notice of grant award (NCI, 2020; NIAID, 2024). In addition, most initial submissions are not funded, so multiple application submissions are required, which extends the already long timeline to attain extramural support (see Figures 5-4 and 5-5). When support is obtained, several NIH institutes implement procedures for reducing the total individual award so that more awards can be made to additional investigators. Although this increases the number of funded proposals, it reduces the proposed budget for many projects.
The very long review process precludes rapid revision and resubmission. NIH funding requires planning and a significant amount of effort on the part of investigators and their institutions. These issues are experienced by all investigators who seek extramural funding from NIH, not just those conducting pediatric research. However, studies that include children may have an extended period of review because there is a need to address additional safeguards for children as a vulnerable population (Kastner et al., 2015; Stewart et al., 2025). For example, studies that include children often cannot be exempted from IRB review or have an expedited review process (NIH, n.d.). Consent may take longer, and assent may be required. In addition, review bodies generally have a greater level of comfort with research that includes adult participants than they do with research that involves children.
The Scientific Review Process
The Center for Scientific Review (CSR) was established by NIH in 1946 to oversee a fair and independent review process of grant applications and to ensure that the most promising research is chosen for funding (NIH CSR, 2025). The CSR maintains established expert scientific review committees, colloquially known as “study sections,” that evaluate the significance, innovation, rigor, reproducibility, and methodological approach of research
NOTE: NIH = National Institutes of Health.
SOURCE: NIH (2019).
proposals from extramural investigators. These study sections are composed of established investigators from institutions across the United States.
One obstacle for submitted pediatric-focused research proposals is that while expertise in pediatrics on study section rosters is critical for proposals that include enrollment, interventions, and observations of children, the amount of pediatric expertise can vary widely on a study section. Study section reviewers who lack experience with the intricacies around children as study subjects may be ill-equipped to evaluate the merits of pediatric research proposals. The significance and impact of a pediatric study could be missed by a non-pediatric expert. In addition, for basic or translational research proposals, the importance and relevance for a pediatric disease and its mechanisms may not be evident to a reviewer who lacks pediatric research expertise. For these reasons, pediatric-focused clinical research proposals, including clinical trials, require rigorous evaluation by pediatric investigators, ideally clinician–investigators.
There is considerable effort among CSR staff to maintain a diverse range of expertise on any given study section, and CSR uses ad hoc reviewers quite liberally to ensure expertise on study sections; however, it is difficult to estimate how all award applications involving pediatric research may align with study sections that have adequate pediatric expertis.6 In an effort to develop a crude estimate of how well pediatrics is represented in study sections, the committee examined the rosters of permanent members of regular standing study sections and special emphasis panels (NIH CSR, 2023). The committee counted study section members as having pediatrics expertise if their primary affiliations were with a department of pediatrics at a medical school or they were employed at a children’s hospital. It is
___________________
6 Presentation by Valerie Durrant to the Committee on Strategies to Enhance Pediatric Health Research Funded by NIH, March 14, 2025.
worth noting that these percentages do not include individuals with primarily affiliations at hospitals that are primarily adult but might include a pediatric unit or those who might have secondary (or other) affiliations in pediatrics departments since it would not have been obvious in their affiliation that they have a pediatric affiliation or expertise.
The data in Tables G-1 and G-2 in Appendix G also very likely under-represent what the committee would consider pediatric expertise, such as study sections focused on prevention science, which would inherently have both membership expertise in pediatrics and research applications heavily skewed toward child and adolescent populations, as was noted by Dr. Valerie Durrant in an open session with the committee (Durrant, 2025). The committee also did not have access to the number of pediatric-related applications in each study section and whether the applications reviewed in the study section required pediatric expertise. Additionally, Table G-1 does not include the count of ad hoc reviewers with pediatric expertise, so the percentage of reviewers with pediatric expertise on the study section may be inexact. It is also worth noting that the two tables include two different timeframes, with Table G-1 consisting of standing members participating in meetings for 1 year while Table G-2 includes panel members participating in one meeting occurrence. Special emphasis panels may use aggregate rosters, which can skew the proportion of reviewers with pediatric expertise, as these rosters include reviewers from various panels. Despite these clear limitations to this evaluation, the results offer an initial look at where pediatric expertise could be more significantly represented. Among the 187 active (as of April 2025) regular study sections, pediatric representation was low, with almost half (88/187; 47 percent) of them having no pediatric expertise represented in their primary affiliation (see Table G-1 in Appendix G). Only 24 study sections (13 percent) had three or more members representing pediatrics. The Reproductive, Perinatal, and Pediatric Health study section had the greatest number of pediatrics experts: nine of its 20 members (45 percent). Among the 3,459 permanent members of study sections at NIH, 204 (6 percent) represented pediatrics. Pediatrics was also poorly represented among the special emphasis panels, with 6 percent of the standing members (324 of 5,702 members) having pediatric expertise. Among the 138 special emphasis panels, 37 of them (27 percent) had no representatives from pediatrics (see Table G-2 in Appendix G). The potential lack of pediatrics expertise on study sections and special emphasis panels could make it difficult for pediatrics-focused submissions to be evaluated fairly if reviewers cannot fully understand the merits and mechanics of the proposals (see Box 5-2). Consequently, the distribution of pediatrics research across institutes and centers (ICs) could be affected, and the representation of pediatrics-focused principal investigators on NIH-wide initiatives could be lessened.
BOX 5-2
The Challenge of Having Insufficient Pediatric Representation on Study Sections
“From a process perspective, NIH review panels may not always include reviewers with pediatric expertise, which can lead to undervaluation of the unique scientific and clinical significance of pediatric-focused proposals.”
– Early career researcher and recipient of NIH grant funding
“Additionally, the smaller sample sizes and longer follow-up periods often required for pediatric studies can be perceived as limitations during peer review (and these longer follow-ups are associated with significant costs). From a process perspective, the current structure of study sections can disadvantage pediatric applications when they are reviewed by panels lacking pediatric expertise. This can lead to lower enthusiasm scores and reduced appreciation for the nuances of conducting rigorous research in neonatal or pediatric populations.”
– Neonatal pediatric researcher and recipient of NIH grant funding
“Review processes seem dominated by scientists/researchers with an adult focus. These reviewers may not appreciate the significance and impact of pediatric-focused projects. Research review of pediatric projects by pediatric-focused scientists would be better.”
– Pediatric oncologist who received grant funding from NIH
SOURCE: These quotes were collected from the committee’s call for perspectives from NIH grant applicants; responses were collected by staff and synthesized by theme and anonymized to the committee (see Appendix C for the questions posted in the call for perspectives).
Conclusion 5-5: Representation of pediatric expertise on study sections is variable, which can create circumstances in which pediatric research proposals are reviewed by panels lacking pediatric expertise, potentially leading to inadequate reviews and lower prioritization.
Administrative Hurdles
Administrative burdens, including complex federal requirements, associated with the application processes for funding can be significant. NIH could improve processes to better support pediatric researchers to assist with regulatory requirements that are more challenging in pediatrics,
including informed consent and assent. Given the special challenges facing pediatric researchers described above, further education of investigators and engagement with NIH program officers is key. “Navigators” could assist investigators who possess varying levels of expertise. Navigators could assist students, postdoctoral fellows, junior faculty, mid-level faculty, and senior faculty with respect to opportunities and processes. In addition, they could advise on the ever-changing landscape for proposals and on compliance with and adherence to policies and procedures, especially with regard to how they impact children and pediatric research. The navigators could better educate investigators on how to best use the resources that lead to success (engagement with program officer, the RFA process, etc.). The navigators would have expertise relative to pediatric research and could better guide applicants through the process. NICHD previously conducted a young investigator meeting with highlighted speakers and a mock study section; the feedback was very positive, and this could be a potential path for young investigator engagement.
In conclusion, pediatric research faces significant scientific and methodological barriers, such as difficulties in translating animal models to bedside care, challenges recruiting diverse sets of study participants, and the ecosystem of care which consists of medical, health, social, and environmental aspects of child growth and development. Overcoming these limitations requires a multifaceted strategy that will include improving recruitment and inclusion efforts, developing more accurate models of pediatric health, and valuing the long-term impact of pediatric research for both individuals and society. In Chapter 6 the committee expands on these ideas, presenting a vision of what pediatric health research could become both at NIH and via extramural research funded by NIH.
Recommendation 5-1: The National Institutes of Health (NIH) should work to create a culture of ethical inclusion of children in research, focused on protecting children through research rather than from research. To this end, NIH should develop measurable strategies and mechanisms to better implement and enforce existing policies to ensure inclusion of children at the project and proposal level. Strategies and mechanisms should include:
- Updating the simplified review framework by explicitly considering the inclusion of children and its implications for health during the life course as a component of evaluating a proposal (i.e., its significance) during review.
- Continuing to support the NIH Center for Scientific Review in implementing and monitoring structural changes to support pediatric research.
-
- Requiring and monitoring that pediatric representation is consistent with the number of applications on all relevant standing study sections.
- Ensuring provision of adequate justification for exclusion of children during grant review: If there is a scientific, legal, or ethical reason for the exclusion of children, this justification should be clearly articulated in the grant application and assessed and documented during the grant review process.
- Elevating prioritization of implications of research on the life course during scientific review: NIH should identify the impact of pediatric research on the life course as an important component of a proposal’s significance section and should do so directly within reviewer guidance. In addition, training for study section reviewers should encourage them to consider whether a proposal adequately addresses the importance of the research for the life course when evaluating the significance or impact of extramural grant applications.
- Improving transparency on inclusion: The Office of the Director of NIH should annually track and report justifications provided for the exclusion of children. This information should be included in the NIH annual report and published on the NIH Pediatric Research Consortium (NPeRC) website, or an NPeRC equivalent website, to track progress on the improved inclusion of children across NIH activities over time.
- Providing clearer institutional guidance about the ethical inclusion of children in research: NPeRC (or NPeRC equivalent), in consultation with the NIH Clinical Center’s Department of Bioethics, should develop a widely accessible toolkit and guidance on ethical issues in pediatric research that have the potential to unduly impede research if misunderstood, such as when to include children in research, provisions for assent, adolescent independent consent, age de-escalation, and payment for research participation.
FINANCIAL BARRIERS FACED BY PEDIATRIC HEALTH RESEARCHERS
Insufficient Funding to Cover the Higher Costs of Pediatric Research
Funding for pediatric research often does not align with the actual costs required for pediatric trials, which can be more expensive and logistically complex than adult trials. (See Box 5-3 for some perspectives from recent NIH award applicants when asked to discuss their experience regarding any structural impediments to securing NIH funding for pediatric research; see Appendix C for the full list of questions posed in the call for perspectives.)
Pediatric Recruitment and Retention Challenges
For several reasons, recruiting and retaining children in clinical trials is more difficult than recruiting and retaining adults. First, parents or guardians are essential participants in recruitment and study processes. Caregiver distrust of academic research can challenge pediatric recruitments and often requires educating parents and fostering trust in the validity and value of clinical pediatric research (Cunningham-Erves et al., 2019; Rajakumar et al., 2009). Second, the participation of children is often contingent on the availability of family support, which can limit who can participate in studies. Third, pediatric clinical research often happens at select urban academic medical centers, which can make it difficult for families living in rural and underserved areas to participate (Lim et al., 2011). Fourth, inclusion of non-English-speaking participants is essential to ensuring a representative sample. This requires multilingual recruitment strategies (Beauchemin et al., 2024), especially since 22 percent of children live in homes in which a language other than English is spoken (Annie E. Casey Foundation, 2025). Fifth, it is simply difficult to obtain enough participants in pediatric studies for several reasons.
The pediatric population available for study for most specific diseases is significantly smaller than the available population for adult studies since most children are healthy. For example, while about half of adults in the United States have hypertension, it affects only about 4 percent of children. Thus, most pediatric research studies, especially for rarer conditions or diseases, require multiple study sites to recruit enough participants of the eligible population. Rare disease studies are a cornerstone of pediatric research, but low prevalence precludes recruitment and enrollment targets on par with standards in adult populations (Batshaw et al., 2014; Rees et al., 2019).
More centers mean greater infrastructure costs. A multi-site study may also require a consortium agreement among the institutions, which also
NOTE: CKID = Chronic Kidney Disease in Children; CRIC = Chronic Renal Insufficiency Cohort.
SOURCE: Data from or derived from CRIC and CKID studies.
BOX 5-3
Financial Impediments
“NIH has had the same cap on R03, R21, and R01 modular grants for >20 years despite inflation. This is absurd. Applicants can propose less and less research in order to fit the budget caps. These caps need to be raised to be more realistic. NIH needs to stop pretending research costs haven’t risen in 20 years!”
– Experienced researcher (preclinical research in developmental neuroscience) with grants from the National Institute on Environmental Health Sciences, National Institute on Mental Health, and NIDA
“Conducting clinical trials in pediatrics is extremely difficult with the current NIH caps on direct costs. Since RA [research assistant], coordinator, grad student, [and] technician salaries have increased over time, it leaves less and less money to conduct the research, provide stipends for participants, and ensure that there are robust facilities and infrastructure to support this work. Unless we figure out a way to share resources across an institution or between institutions, it will become more and more difficult to conduct clinical trials or interventions that can change lifestyle behaviors that promote a healthier life.”
– Researcher studying childhood obesity treatment, parenting interventions, and related topics for over 15 years with NIH grant funding
SOURCE: These quotes were collected from the committee’s call for perspectives from NIH grant applicants; responses were collected by staff and synthesized by theme and anonymized to the committee (see Appendix C for the questions posted in the call for perspectives).
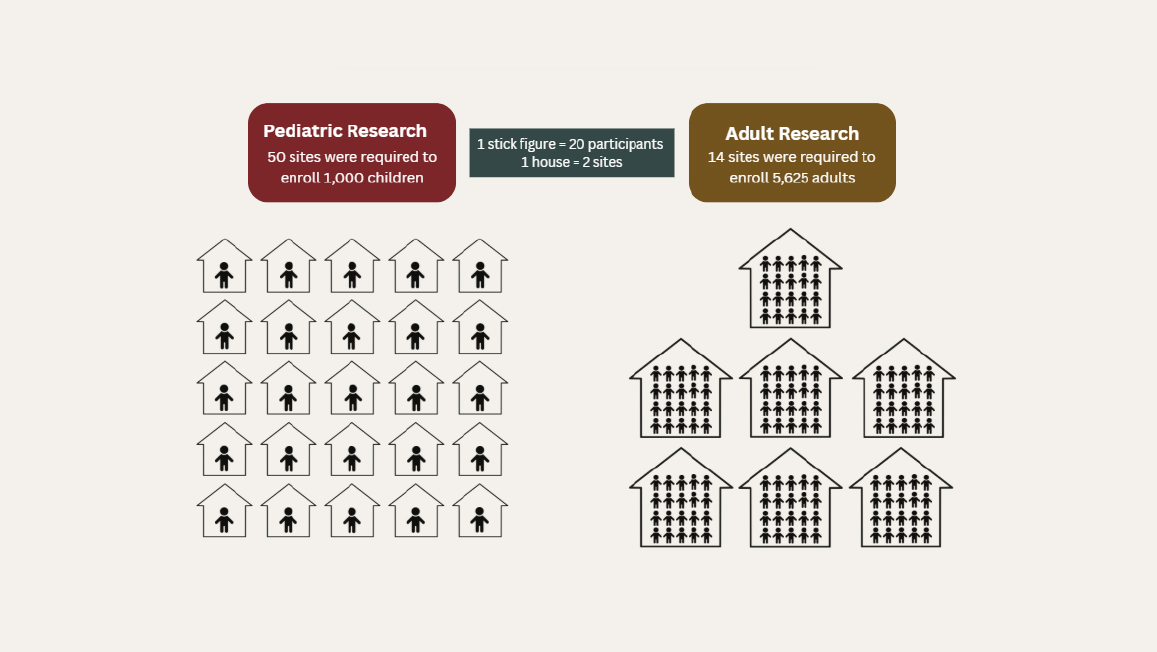
increases cost and study complexity. Including more institutions in studies requires an increase in baseline infrastructure costs and necessitates multi-center IRBs, which are costly. As an example, the Chronic Renal Insufficiency Cohort (CRIC) study run by the National Institute of Diabetes and Digestive and Kidney Diseases included 13 clinical sites and enrolled more than 5,625 adults (NIDDK, 2024), compared with the concurrent Chronic Kidney Disease in Children (CKID) study, which required 50 clinical sites to enroll 1,100 children over a nearly 20-year period (NIDDK, 2025) (see Figure 5-6). Hospitals and research centers must contend with financial constraints when implementing large-scale research studies, further limiting their capacity to compensate parents for their time and potential lost wages.
Timelines and endpoints may face a critical window depending on the age of recruitment with regard to a child’s development and the need for longer follow-up that accounts for developmental changes. A 12-month follow-up may be appropriate for many adult studies, but this is often inadequate for children. This is particularly true for studies of drugs (IOM, 2004). Delays in funding or interrupted funding may limit the ability to follow up with study cohorts.
Due to the accelerated pace of development in childhood and the fact that child development has critical periods that create the potential for delayed outcomes from interventions, advancing the science of pediatric medicine at times may require multiple sequential studies to ensure safety and efficacy across pediatric populations. For example, the NICHD Neonatal Research Network’s whole-body cooling trial for hypoxic–ischemic encephalopathy demonstrated significantly reduced death and disability in infants (Shankaran et al., 2005) but could not confirm whether the benefits would persist at school age, a critical downstream outcome. To answer this important question, additional funding was obtained from NICHD for a 7-year follow-up following the results at 18 to 22 months. This additional investment confirmed the benefits of whole-body cooling in reducing mortality in 6- to 7-year-old children with hypoxic–ischemic encephalopathy (Shankaran et al., 2012).
Another financial barrier for pediatric research is the lengthier decision-making process as compared with adult research. For example, the informed consent process requires that at least one parent, and sometimes both parents and guardians, agree to the child participating in the research. If only one parent is present at a time, that parent may need to ask the other parent and may solicit advice from family members and friends before consenting on behalf of the child (Nelson et al., 2013). Further time may be needed for child assent, which is the process of explaining the study to the child and obtaining the child’s agreement to participate. Another expense in pediatrics research is compensating parents or caregivers for missed work time and travel to the enrollment site. Additional research time may be needed if the data collection includes parents, siblings, and the children enrolled in the study, which is often the case for pediatric studies. This too adds to the cost of the research.
One example of a need for extended follow-up was the Antenatal Late Preterm Steroids (ALPS) study from the Maternal Fetal Medicine Units Network, which showed that the administration of antenatal steroids (betamethasone) significantly reduced the need for respiratory treatment among infants born at late preterm (34–36 weeks of gestation) (Gyamfi-Bannerman et al., 2016). However, low blood sugar, or hypoglycemia, was
more common in the infants exposed to betamethasone, raising a concern over safety. The initial proposal for the ALPS study included a 2-year follow-up, but both funding challenges and the uncertain outcome of the parent trial prevented this plan from coming to fruition. Participants in the parent trial were asked to consent to the possibility of future contact with the researchers (Gyamfi-Bannerman, 2024). Given the desire for long-term follow-up to monitor low blood sugar and its impacts on children who received betamethasone before delivery, funding was secured for further assessment, and no differences were found at 6 years of age between the exposed and unexposed children (Gyamfi-Bannerman et al., 2016, 2024). These results offered reassuring data regarding the lack of impact of hypoglycemia on the study participants. The ALPS study and the whole-body cooling trial are good examples of why it is necessary to have supplemental funds available for long-term follow-up assessment of efficacy and safety in pediatric clinical studies.
Conclusion 5-6: Pediatric research faces unique barriers that increase the duration, cost, and complexity of studies. These barriers include, but are not limited to, the need for extended monitoring to capture developmental outcomes; multi-site collaboration to ensure representative and adequately sized samples; and, at times, challenges arising from misunderstood or misapplied ethical standards, such as the common practice of enrolling older children before younger ones (“age de-escalation”) without considering when it serves as a protection and when it does not. These barriers contribute to a culture of overprotection and undue delay in the inclusion of children in research.
To address these additional hurdles faced by pediatric research, structural solutions to accelerate research become even more important. One such attempt to do so is awards made by the National Center for Advancing Translational Sciences (NCATS). NCATS has a Clinical and Translational Science Awards (CTSA) program that supports a network of institutions working on the translation of research discoveries into improved care (NCATS, 2025). While information provided by NCATS shows that nearly all CTSAs recipients engage in pediatric research, only 5 to 7 percent of trainees and scholars focus on pediatric research, and merely 20 percent of pilot awards involve pediatric research. There are 16 CTSA program hubs which are led by principal researchers or co-principal investigators who are also pediatricians. Ninety-one percent of the hubs include children’s hospitals. However, only about 6 percent of the CTSA publications are pediatric in nature (Llewellyn et al., 2020). Given that children make up 22 percent of the U.S. population, it would seem that more resources should be directed toward additional pediatric studies in the CTSA program. It
was difficult for the committee to discern the actual dollar amounts that are directed toward pediatric research through the CTSA program. The current program announcement asks for inclusion or exclusion of children as required by statute to determine justification in terms of scientific goals and research strategy (e.g., HHS, 2025). The guidance for the reviewers states, “Women, members of racial and ethnic minority groups, and individuals across the lifespan (including children and older adults) must be included in all clinical research studies, as appropriate for the scientific goals of the work proposed” (NIH, 2025). The reviewers are asked to justify inclusion as acceptable or unacceptable. Stressing the importance of inclusion of pediatric research to reviewers of CTSA programs, along with financial incentives for pediatric research, may encourage greater inclusion of pediatric research.
Recommendation 5-2: National Institutes of Health (NIH) institutes and centers should award pediatric research grants and budgets that support the specific needs of pediatric studies. In addition, to address the mismatch between study timelines and funding cycles, NIH should:
- Allow for extended or extendable award timelines and/or renewable grant mechanisms specifically designed for pediatric research, allowing investigators to sustain long-term studies that align with children’s developmental trajectories;
- Provide higher budget maximums to account for the need for representative recruitment and multi-center studies, when scientifically justified;
- Create supplemental funding opportunities specifically designed for longitudinal or follow-up data collection, which would enhance learning across the life course while capitalizing on previously funded and ongoing studies;
- Provide funding supplements to support multilingual research and community engagement to facilitate representation for generalizable results; and
- Increase access and usage of Clinical and Translational Science Awards as a mechanism for promoting pediatric research.
Appropriations for NIH Funding
There are research projects that may require 10- to 15-year-long financial commitments from NIH. Mechanisms for such long-term funding are not yet in place. There would need to be some assessments over time to ensure that milestones are being met by the investigators.
NIH funding is dependent on timely appropriations. When appropriations are delayed, as has been the case for most of the last two decades,
ICs are hesitant to fund grants above a certain percentile (known as the payline) in an effort to ensure fairness over the fiscal year and to stay within their eventual budget. An additional impact of delayed appropriations is that many ICs impose reductions in the applicant’s requested budget. For example, in 2024 the average budget reduction to an R01 from NICHD was approximately 14 percent (NICHD, 2024). Since NICHD was the largest funder of pediatric research in 2023 ($993 million), these project budget reductions can disproportionately affect NICHD’s pediatric grantees. Delays in appropriations also slow the onset of award notices and, in turn, research projects, which hampers potential discoveries and delays their clinical and therapeutic benefits. A mechanism that guarantees yearly funding of ICs at the start of the fiscal year would make research more efficient and streamlined and would likely accelerate the pace of discovery. An additional challenge is that many NIH ICs have reductions in the requested budgets they submit to Congress.
THE STRATEGIC IMPORTANCE OF INVESTING IN PEDIATRIC RESEARCH
Investing in pediatric research is a crucial precursor to advancing pediatric health, itself a strategic necessity for the United States. A healthy generation of children is the foundation of a resilient, innovative, and secure nation, directly influencing the state of the nation’s future workforce, military readiness, economic productivity, and societal stability. A robust pediatric research enterprise will enable the kind of preventive interventions, early diagnostics, and targeted therapies that can dramatically alter life course trajectories, improving short- and long-term health outcomes (Halfon et al., 2022; NASEM, 2024).
As introduced in Chapter 1, U.S. children face nearly universal risk for chronic disease and mental health issues when compared with peers in other high-income nations (Forrest et al., 2025). This is a sobering indicator of systemic vulnerabilities in U.S. health care and social support systems, which are discussed in detail in the recent National Academies of Sciences, Engineering, and Medicine report Launching Lifelong Health by Improving Health Care for Children, Youth, and Families (NASEM, 2024).
According to a recent U.S. military report, more than 70 percent of 18-year-olds are ineligible for service without a waiver due to chronic health and mental conditions (DOD, 2022). Moreover, half of all chronic mental disorders begin at 14 years of age, and 80 percent of obese adolescents will become obese adults (Bibbins-Domingo et al., 2007; Kessler et al., 2005; Simmonds et al., 2016). Chronic diseases and mental health conditions that begin in childhood often persist into adulthood, compounding health care costs and reducing quality of life. Without a healthy youth
population, the nation’s capacity to maintain a robust military, respond to global challenges, and sustain economic competitiveness is compromised.
Investment in evidence-based health and social services and, importantly, the research needed to create them benefits children and their families and leads to greater adult productivity by way of a healthier labor force (NASEM, 2024). Children who grow up without the burden of chronic disease are more likely to thrive academically, socially, and professionally (Maslow et al., 2011). This is especially critical in an era when precision medicine and genetic therapies are rapidly advancing fields in which pediatric research has already yielded groundbreaking successes, such as curative gene therapy for spinal muscular atrophy (Finkel et al., 2017). The impact of NIH-supported pediatric research and its discoveries cannot be overstated: lifespan-extending treatments for cystic fibrosis, vaccines that have prevented countless deaths and hospitalizations, and tailored therapies leading to a 90 percent cure rate for acute lymphoblastic leukemia. For example, a Centers for Disease Control and Prevention analysis found that among approximately “117 million children born during 1994–2023, routine childhood vaccinations will have prevented approximately 508 million lifetime cases of illness, 32 million hospitalizations, and 1,129,000 deaths, at a net savings of $540 billion in direct costs and $2.7 trillion in societal costs” (Zhou et al., 2024).
The benefits of investment in pediatric research are exponential in the number of increased and healthier life-years across the life course to which they contribute. Healthier children and adolescents will become the next generation of Americans.
REFERENCES
Annie E. Casey Foundation. 2025. Children who speak a language other than English at home in United States. https://datacenter.aecf.org/data/tables/81-children-who-speak-a-language-other-than-english-at-home#detailed/1/any/false/1096,871,870,573,869/any/396,397 (accessed November 8, 2025).
Arnstein, S. R. 1969. A ladder of citizen participation. Journal of the American Institute of Planners 35(4):216–224.
Assuras, S., K. Courtney, M. Maxfield, S. Shagalow, S. Sherer, J. J. Manly, and C. S. Widom. 2025. Childhood maltreatment confers long-term risk for cognitive impairment: A prospective investigation. Journal of Prevention of Alzheimers Disease 12(9):100303.
Batshaw, M. L., S. C. Groft, and J. P. Krischer. 2014. Research into rare diseases of childhood. JAMA 311(17):1729–1730.
Beauchemin, M. P., M. Ortega, S. J. Santacroce, J. M. Robles, J. Ruiz, A. G. Hall, J. M. Kahn, C. Fu, M. Orjuela-Grimm, G. C. Hillyer, S. Solomon, W. Pelletier, R. Montiel-Esparza, L. J. Blazin, C. Kline, A. E. Seif, P. Aristizabal, L. E. Winestone, and M. C. Velez. 2024. Clinical trial recruitment of people who speak languages other than English: A Children’s Oncology Group report. JNCI Cancer Spectrum 8(4):pkae047.
Berkman, B. E., D. Wendler, H. K. Sullivan, and C. Grady. 2017. A proposed process for reliably updating the common rule. American Journal of Bioethics 17(7):8–14.
Berkman, B. E., D. Howard, and D. Wendler. 2018. Reconsidering the need for reconsent at 18. Pediatrics 142(2):e20171202.
Bibbins-Domingo, K., P. Coxson, M. J. Pletcher, J. Lightwood, and L. Goldman. 2007. Adolescent overweight and future adult coronary heart disease. New England Journal of Medicine 357(23):2371–2379.
Bierer, B. E., S. A. White, L. Gelinas, and D. H. Strauss. 2021. Fair payment and just benefits to enhance diversity in clinical research. Journal of Clinical and Translational Science 5(1):e159.
CDC (Centers for Disease Control and Prevention). 2025. About adverse childhood experiences. https://www.cdc.gov/aces/about/index.html (accessed December 10, 2025).
Chong, A., J. Claydon, S. Chhina, M. Sadarangani, and Q. Doan. 2025. Strategies to reduce language barriers in clinical research: A national survey of pediatric health researchers. Ethics, Medicine and Public Health 33:101122.
Collins, K. C., O. Burdall, J. Kassam, G. Firth, D. Perry, and M. Ramachandran. 2022. Health-related quality of life and functional outcome measures for pediatric multiple injury: A systematic review and narrative synthesis. Journal of Trauma and Acute Care Surgery 92(5):e92–e106.
Cunningham-Erves, J., J. Deakings, T. Mayo-Gamble, K. Kelly-Taylor, and S. T. Miller. 2019. Factors influencing parental trust in medical researchers for child and adolescent patients’ clinical trial participation. Psychology, Health & Medicine 24(6):691–702.
Diekema, D. S. 2018. Payments for participation of children in research. In E. Kodish and R. M. Nelson (eds.), Ethics and research with children: A case-based approach. New York: Oxford University Press.
DOD (Department of Defense). 2022. Qualified military available technical report. https://www.esd.whs.mil/Portals/54/Documents/FOID/Reading%20Room/Personnel_Related/23-F-1060_QMA_Technical_Report_Mar_2022.pdf (accessed November 8, 2025).
Emanuel, E. J., D. Wendler, J. Killen, and C. Grady. 2004. What makes clinical research in developing countries ethical? The benchmarks of ethical research. Journal of Infectious Diseases 189(5):930–937.
Enlow, M. B., R. J. Chung, M. A. Parisi, S. K. Sagiv, M. A. Sheridan, A. Stroustrup, R. J. Wright, L. A. Cox, J. Beverly, T. Hendershot, D. R. Maiese, and C. M. Hamilton. 2022. Standard measurement protocols for pediatric development research in the PhenX Toolkit. Research Triange Park, NC: RTI Press. https://www.ncbi.nlm.nih.gov/books/NBK589470/ (accessed November 8, 2025).
EPA (Environmental Protection Agency). 2025. Children are not little adults! https://www.epa.gov/children/children-are-not-little-adults (accessed November 7, 2025).
Farooqi, I. S., and Y. Xu. 2024. Translational potential of mouse models of human metabolic disease. Cell 187(16):4129–4143.
FDA (Food and Drug Administration). 2024. Rare diseases at FDA. https://www.fda.gov/patients/rare-diseases-fda (accessed September 12, 2025).
FDA. 2025. Evaluation of sex differences in clinical investigations: Guidance for industry. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/evaluation-sex-differences-clinical-investigations (accessed November 7, 2025).
Fernandez, E., R. Perez, A. Hernandez, P. Tejada, M. Arteta, and J. T. Ramos. 2011. Factors and mechanisms for pharmacokinetic differences between pediatric population and adults. Pharmaceutics 3(1):53–72.
Finkel, R. S., E. Mercuri, B. T. Darras, A. M. Connolly, N. L. Kuntz, J. Kirschner, C. A. Chiriboga, K. Saito, L. Servais, E. Tizzano, H. Topaloglu, M. Tulinius, J. Montes, A. M. Glanzman, K. Bishop, Z. J. Zhong, S. Gheuens, C. F. Bennett, E. Schneider, W. Farwell, D. C. De Vivo, and E. S. Group. 2017. Nusinersen versus sham control in infantile-onset spinal muscular atrophy. New England Journal of Medicine 377(18):1723–1732.
Flores-Toro, J. A., S. Jagu, G. T. Armstrong, D. F. Arons, G. J. Aune, S. J. Chanock, D. S. Hawkins, A. Heath, L. J. Helman, K. A. Janeway, J. E. Levine, E. Miller, L. Penberthy, C. W. M. Roberts, E. R. Shalley, J. F. Shern, M. A. Smith, L. M. Staudt, S. L. Volchenboum, J. Zhang, J. C. Zenklusen, D. R. Lowy, N. E. Sharpless, J. M. Guidry Auvil, A. R. Kerlavage, B. C. Widemann, G. H. Reaman, W. A. Kibbe, J. H. Doroshow, and the Childhood Cancer Data Initiative Working Group. 2023. The Childhood Cancer Data Initiative: Using the power of data to learn from and improve outcomes for every child and young adult with pediatric cancer. Journal of Clinical Oncology 41(24):4045–4053.
Fogler, E. C., and A. Lerman. 2017. Balancing state and federal informed consent law. https://www.healthlawadvisor.com/balancing-state-and-federal-informed-consent-law (accessed November 7, 2025).
Forrest, C. B., L. J. Koenigsberg, F. Eddy Harvey, M. G. Maltenfort, and N. Halfon. 2025. Trends in U.S. children’s mortality, chronic conditions, obesity, functional status, and symptoms. JAMA 334(6):509–516.
Gelinas, L., E. A. Largent, I. G. Cohen, S. Kornetsky, B. E. Bierer, and H. Fernandez Lynch. 2018. A framework for ethical payment to research participants. New England Journal of Medicine 378(8):766–771.
George, S., N. Duran, and K. Norris. 2014. A systematic review of barriers and facilitators to minority research participation among African Americans, Latinos, Asian Americans, and Pacific Islanders. American Journal of Public Health 104(2):e16–e31.
Gilbert, A. L., A. S. Knopf, J. D. Fortenberry, S. G. Hosek, B. G. Kapogiannis, and G. D. Zimet. 2015. Adolescent self-consent for biomedical human immunodeficiency virus prevention research. Journal of Adolescent Health 57(1):113–119.
Gitterman, D. P., W. S. Langford, and W. W. Hay. 2018. The uncertain fate of the National Institutes of Health (NIH) pediatric research portfolio. Pediatric Research 84(3):328–332.
Green, D. J., H. Sun, J. Burnham, X. I. Liu, J. van den Anker, J. Temeck, L. Yao, S. K. McCune, and G. J. Burckart. 2019. Surrogate endpoints in pediatric studies submitted to the U.S. FDA. Clinical Pharmacology & Therapeutics 105(3):555–557.
Guttmacher Institute. n.d. Notes. https://www.guttmacher.org/sites/default/files/graphics/gr030406t/gr030406_note.html (accessed November 7, 2025).
Gyamfi-Bannerman, C. 2024. Antenatal late preterm steroids: The evolution of the ALPS trial. Clinical Obstetrics and Gynecology 67(2):399–410.
Gyamfi-Bannerman, C., E. A. Thom, S. C. Blackwell, A. T. Tita, U. M. Reddy, G. R. Saade, D. J. Rouse, D. S. McKenna, E. A. Clark, J. M. Thorp Jr, E. K. Chien, A. M. Peaceman, R. S. Gibbs, G. K. Swamy, M. E. Norton, B. M. Casey, S. N. Caritis, J. E. Tolosa, Y. Sorokin, J. P. VanDorsten, L. Jain, and the NICHD Maternal–Fetal Medicine Units Network. 2016. Antenatal betamethasone for women at risk for late preterm delivery. New England Journal of Medicine 374(14):1311–1320. See correspondence at New England Journal of Medicine 375:485–487 and correction to the correspondence at New England Journal of Medicine 388(18):1728.
Gyamfi-Bannerman, C., R. G. Clifton, A. T. N. Tita, S. C. Blackwell, M. Longo, J. A. de Voest, T. M. O’Shea, S. Z. Bousleiman, F. Ortiz, D. J. Rouse, T. D. Metz, G. R. Saade, K. M. Rood, K. D. Heyborne, J. M. Thorp, Jr., G. K. Swamy, W. A. Grobman, K. S. Gibson, Y. Y. El-Sayed, G. A. Macones, and the Eunice Kennedy Shriver Maternal-Fetal Medicine Units Network. 2024. Neurodevelopmental outcomes after late preterm antenatal corticosteroids: The ALPS follow-up study. JAMA 331(19):1629–1637.
Halfon, N., S. A. Russ, and E. L. Schor. 2022. The emergence of life course intervention research: Optimizing health development and child well-being. Pediatrics 149(Suppl 5):e2021053509C.
Halle, T. G. n.d. How implementation science and improvement science can work together to improve early care and education. In Getting it right: Using implementation research to improve outcomes in early care and education. New York: Foundation for Child Development. Pp. 223–258. GettingitRight_UsingImplementationResearchtoImproveOutcomesinECE_Chapter10_2020.pdf (accessed November 8, 2025).
Harbin, A., N. Laventhal, and M. Navin. 2023. Ethics of age de-escalation in pediatric vaccine trials: Attending to the case of COVID-19. Vaccine 41(9):1584–1588.
HHS (Department of Health and Human Services). n.d. Research with children FAQs. https://www.hhs.gov/ohrp/regulations-and-policy/guidance/faq/children-research/index.html (accessed April 29, 2025).
HHS. 1983. Additional protections for children involved as subjects in research—Department of Health and Human Services. Final rule. Federal Register 48(46):9814–9820.
HHS. 2025. Clinical and Translational Science Award. https://grants.nih.gov/grants/guide/pafiles/PAR-24-272.html (accessed August 19, 2025).
Hood, N. E., T. Brewer, R. Jackson, and M. E. Wewers. 2010. Survey of community engagement in NIH-funded research. Clinical and Translational Science 3(1):19–22.
Hu, A., J. L. Holl, and M. V. Raval. 2022. Pediatric specific challenges of the single institutional review board mandate. Trials 23(1):224.
IOM (Institute of Medicine). 2004. Ethical conduct of clinical research involving children. Washington, DC: The National Academies Press.
Israels, T., B. Atwiine, C. Duffy, J. Sichali, G. Afungchwi, K. Pritchard-Jones, D. Nyirenda, and V. Mwapasa. 2024. From “intuitive” pragmatic interventions to a systematic approach—Using implementation science to improve childhood cancer survival in low-income settings: The CANCaRe Africa experience. Pediatric Hematology Oncology Journal 9(1):42–44.
Jennings, L. N., R. Escandon, D. S. Diekema, R. M. Nelson, D. L. Snyder, and L. M. Johnson. 2025. Improving the pediatric research federal review process. Pediatrics 156(2): e2024069858.
Joffe, S., A. J. Allen, J. M. Davis, E. Koppelman, S. Z. Kornetsky, G. M. V. Ku, V. A. Miller, J. Preston, L. D. Shah, and B. E. Bierer. 2023. Establishing a global regulatory floor for children’s decisions about participation in clinical research. Pediatric Research 94(2): 462–465.
Jonsson, U., I. Alaie, A. Lofgren Wilteus, E. Zander, P. B. Marschik, D. Coghill, and S. Bolte. 2017. Annual research review: Quality of life and childhood mental and behavioural disorders—A critical review of the research. Journal of Child Psychology and Psychiatry 58(4):439–469.
Kastner, B., S. Behre, N. Lutz, F. Burger, S. Luntz, K. Hinderhofer, M. Bendszus, G. F. Hoffmann, and M. Ries. 2015. Clinical research in vulnerable populations: Variability and focus of institutional review boards’ responses. PLOS ONE 10(8):e0135997.
Kessler, R. C., P. Berglund, O. Demler, R. Jin, K. R. Merikangas, and E. E. Walters. 2005. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Archives of General Psychiatry 62(6):593–602.
Kingsley, J., B. Andraka-Christou, S. K. Shah, P. Spicer, S. Newman, and P. N. Ossorio. 2024. Navigating ethical and legal challenges in the Healthy Brain and Child Development Study: Lessons learned from the Ethics, Law, Policy Working Group. Developmental Cognitive Neuroscience 70:101460.
Kodish, E., M. Eder, R. B. Noll, K. Ruccione, B. Lange, A. Angiolillo, R. Pentz, S. Zyzanski, L. A. Siminoff, and D. Drotar. 2004. Communication of randomization in childhood leukemia trials. JAMA 291(4):470–475.
Kovler, M. L., C. P. Sodhi, and D. J. Hackam. 2020. Precision-based modeling approaches for necrotizing enterocolitis. Disease Models & Mechanisms 13(6):dmm044388.
Largent, E. A., and H. F. Lynch. 2017. Paying research participants: The outsized influence of “undue influence.” IRB: Ethics & Human Research 39(4):1–9.
Leibson, T., and G. Koren. 2015. Informed consent in pediatric research. Paediatric Drugs 17(1):5–11.
Levy-Friedman, B., and T. Kogut. 2024. Children’s self-evaluation of their prosociality when comparing themselves with a specific versus abstract other. Child Development 95(1): 24–33.
Lewis, B., C. Cochran, E. Marquez, N. Bhandari, J. Pharr, S. Upadhyay, and S. Shoemaker. 2025. Use of hospital patient and family advisory councils: A scoping study. Journal of Patient Experience 12:23743735251316995.
Lim, C. S., K. W. Follansbee-Junger, M. S. Crawford, and D. M. Janicke. 2011. Treatment outcome research in rural pediatric populations: The challenge of recruitment. Journal of Pediatric Psychology 36(6):696–707.
Liu, C., R. B. Cox, Jr., I. J. Washburn, J. M. Croff, and H. C. Crethar. 2017. The effects of requiring parental consent for research on adolescents’ risk behaviors: A meta-analysis. Journal of Adolescent Health 61(1):45–52.
Llewellyn, N., D. R. Carter, D. DiazGranados, C. Pelfrey, L. Rollins, and E. J. Nehl. 2020. Scope, influence, and interdisciplinary collaboration: The publication portfolio of the NIH Clinical and Translational Science Awards (CTSA) program from 2006 through 2017. Evaluation & the Health Professions 43(3):169–179.
Long, D. A., and E. L. Fink. 2021. Transitions from short to long-term outcomes in pediatric critical care: Considerations for clinical practice. Translational Pediatrics 10(10): 2858–2874.
Lyerly, A. D., R. Beigi, L. G. Bekker, B. H. Chi, S. E. Cohn, D. D. Diallo, J. Eron, R. Faden, E. Jaffe, A. Kashuba, M. Kasule, C. Krubiner, M. Little, J. Mfustso-Bengo, L. Mofenson, V. Mwapasa, L. Mworeko, L. Myer, M. Penazzato, A. Rid, R. Shapiro, J. A. Singh, K. Sullivan, M. Vicari, J. Wambui, A. White, M. Wickremsinhe, and L. Wolf. 2021. Ending the evidence gap for pregnancy, HIV and co-infections: Ethics guidance from the PHASES project. Journal of the International AIDS Society 24(12):e25846.
MacNeil, M., B. Benoit, T. Disher, A. J. Newman, and M. Campbell-Yeo. 2025. Challenges and best practices for recruiting families of children with intellectual disabilities for health research. Journal of Intellectual Disabilities 29(3):784–797.
Mark, L., V. Mellqvist, J. Michelsen, H. Imberg, K. Bjorkman, N. Johansson, and M. Svedberg. 2025. Gender equality in caregiver attendance for children with chronic diseases: A Swedish longitudinal observational study. BMJ Public Health 3(1):e001584.
Masarik, A. S., and R. D. Conger. 2017. Stress and child development: A review of the family stress model. Current Opinion in Psychology 13:85–90.
Maslow, G. R., A. A. Haydon, C. A. Ford, and C. T. Halpern. 2011. Young adult outcomes of children growing up with chronic illness: An analysis of the National Longitudinal Study of Adolescent Health. Archives of Pediatric and Adolescent Medicine 165(3):256–261.
Morgan, P. J., M. D. Young, A. B. Lloyd, M. L. Wang, N. Eather, A. Miller, E. M. Murtagh, A. T. Barnes, and S. L. Pagoto. 2017. Involvement of fathers in pediatric obesity treatment and prevention trials: A systematic review. Pediatrics 139(2):e20162635.
Mullen, S. 2018. Major depressive disorder in children and adolescents. Mental Health Clinician 8(6):275–283.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2024. Launching lifelong health by improving health care for children, youth, and families. Washington, DC: The National Academies Press.
Nathe, J. M., T. T. Oskoui, and E. M. Weiss. 2023. Parental views of facilitators and barriers to research participation: Systematic review. Pediatrics 151(1):e2022058067.
NCATS (National Center for Advancing Translational Sciences). 2025. Clinical and Translational Science Awards (CTSA) program. https://ncats.nih.gov/research/research-activities/ctsa (accessed August 19, 2025).
NCI (National Cancer Institute). n.d. Rare disease. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/rare-disease (accessed September 12, 2025).
NCI. 2020. The grants process: The lifecycle of a grant. https://www.cancer.gov/grants-training/policies-process/overview/grants-process.pdf (accessed November 8, 2025).
Nelson, D. K., D. Skinner, S. Guarda, S. Choudhury, J. Sideris, L. Barnum, K. Ten Haagen, Q. Heyward, and D. B. Bailey, Jr. 2013. Obtaining consent from both parents for pediatric research: What does “reasonably available” mean? Pediatrics 131(1):e223–e229.
NIAID (National Institute of Allergy and Infectious Diseases). 2024. Timeline for funding decisions. https://www.niaid.nih.gov/grants-contracts/timelines-funding-decisions (accessed December 10, 2025).
NICHD (Eunice Kennedy Shriver National Institute of Child Health and Human Development). n.d. Data and Specimen Hub. https://dash.nichd.nih.gov/ (accessed December 10, 2025).
NICHD. 2024. NICHD funding strategies for fiscal year 2024. https://www.nichd.nih.gov/grants-contracts/process-strategies/strategies/2024 (accessed November 8, 2025).
NIDDK (National Institute of Diabetes and Digestive and Kidney Diseases). 2024. Chronic Renal Insufficiency Cohort study (CRIC). https://repository.niddk.nih.gov/study/15 (accessed November 8, 2025).
NIDDK. 2025. The Chronic Kidney Disease in Children cohort study (CKID). https://repository.niddk.nih.gov/study/39 (accessed November 8, 2025).
NIEHS (National Institute of Environmental Health Sciences). 1999. Community-based prevention and intervention research. https://grants.nih.gov/grants/guide/rfa-files/RFAES-99-012.html (accessed November 8, 2025).
NIEHS. 2024. Environmental Health Sciences Core Centers Program (P30 clinical trials optional). https://grants.nih.gov/grants/guide/rfa-files/RFA-ES-25-002.html (accessed November 8, 2025).
NIH (National Institutes of Health). n.d. Exempt human subjects research infographic. https://grants.nih.gov/sites/default/files/exempt-human-subjects-infographic.pdf (accessed November 8, 2025).
NIH. 1998. NIH policy and guidelines on the inclusion of children as participants in research involving human subjects. https://grants.nih.gov/grants/guide/notice-files/not98-024.html (accessed June 3, 2025).
NIH. 2017. Revision: NIH policy and guidelines on the inclusion of individuals across the lifespan as participants in research involving human subjects. https://grants.nih.gov/grants/guide/notice-files/NOT-OD-18-116.html (accessed June 4, 2025).
NIH. 2019. Illustrated application and grant timelines. https://www.niaid.nih.gov/grantscontracts/timelines-illustrated (accessed July 29, 2025).
NIH. 2025. Guidance for the review of participant inclusion in clinical research and clinical trials. https://grants.nih.gov/grants/peer/guidelines_general/Review_Human_Subjects_Inclusion.pdf (accessed September 12, 2025).
NIH CSR (Center for Scientific Review). 2023. Regular standing study sections and continuing Special Emphasis Panels (SEPs). https://public.csr.nih.gov/StudySections/StandingStudySections (accessed April 29, 2025).
NIH CSR. 2025. About CSR. https://public.csr.nih.gov/AboutCSR (accessed April 29, 2025).
NINR (National Institute for Nursing Research). 2025. Community-partnered nursing research centers (P20 clinical trial optional). https://grants.nih.gov/grants/guide/pa-files/PAR-25-439.html (accessed November 8, 2025).
Ogden, H. L., H. Kim, K. A. Wikenheiser-Brokamp, A. P. Naren, and K. S. Mun. 2021. Cystic fibrosis human organs-on-a-chip. Micromachines (Basel) 12(7):747.
Oken, E., T. M. Bastain, N. Bornkamp, C. V. Breton, R. C. Fry, D. R. Gold, M. F. Hivert, S. Howland, D. J. Jackson, C. C. Johnson, K. Jones, M. Killingbeck, T. M. O’Shea, M. Ortega, D. Ownby, F. Perera, J. V. Rollins, J. B. Herbstman, and program collaborators for Environmental Influences on Child Health Outcomes. 2023. When a birth cohort grows up: Challenges and opportunities in longitudinal developmental origins of health and disease (DOHaD) research. Journal of Developmental Origin of Health and Disease 14(2):175–181.
Osher, D., P. Cantor, J. Berg, L. Steyer, and T. Rose. 2018. Drivers of human development: How relationships and context shape learning and development. Applied Developmental Science 24(1):6–36.
Page, B. F., L. Hinton, E. Harrop, and C. Vincent. 2020. The challenges of caring for children who require complex medical care at home: “The go between for everyone is the parent, and as the parent that’s an awful lot of responsibility.” Health Expectations 23(5):1144–1154.
Rajakumar, K., S. B. Thomas, D. Musa, D. Almario, and M. A. Garza. 2009. Racial differences in parents’ distrust of medicine and research. Archives of Pediatrics & Adolescent Medicine 163(2):108–114.
Reber, L., Z. Griggs, M. J. Patel, E. Ruiz, S. Tamirisa, E. Huan, S. Pennington, and H. Kitzman. 2025. “Misled, misinformed, under-informed”: Socio-ecological determinants for rebuilding trust in clinical research. Ethics, Medicine and Public Health 33:101142.
Rees, C. A., N. Pica, M. C. Monuteaux, and F. T. Bourgeois. 2019. Noncompletion and non-publication of trials studying rare diseases: A cross-sectional analysis. PLOS Medicine 16(11):e1002966.
Richard, J., R. Azar, S. Doucet, and A. Luke. 2020. Pediatric patient and family advisory councils: A guide to their development and ongoing implementation. Journal of Patient Experience 7(6):1476–1481.
Ross, L. F. 2006. Children in medical research: Access versus protection. New York: Oxford University Press.
RTI International. 2025. Research domain—Pediatric development. https://www.phenxtoolkit.org/domains/view/300000 (accessed November 8, 2025).
Ruperto, N. 2016. SP0063 clinical trial design in pediatric rheumatology. Annals of the Rheumatic Diseases 75:16–17.
Samanta, D. 2020. Improving management of infantile spasms by adopting implementation science. Neuropediatrics 51(6):377–388.
Shah, S. 2004. How do institutional review boards apply the federal risk and benefit standards for pediatric research? JAMA 291(4):476–482.
Shah, S., and D. Wendler. 2010. Interpretation of the subjects’ condition requirement: A legal perspective. Journal of Law, Medicine & Ethics 38(2):365–373.
Shah, S. K. 2021. The ethics of pediatric research. In A. S. Iltis and D. MacKay (eds.), The Oxford handbook of research ethics. New York: Oxford University Press. Pp. 683–698.
Shah, S. K., F. Miller, and H. Fernandez Lynch. 2020. The role of community engagement in addressing bystander risks in research: The case of a Zika virus controlled human infection study. Bioethics 34(9):883–892.
Shankaran, S., A. R. Laptook, R. A. Ehrenkranz, J. E. Tyson, S. A. McDonald, E. F. Donovan, A. A. Fanaroff, W. K. Poole, L. L. Wright, R. D. Higgins, N. N. Finer, W. A. Carlo, S. Duara, W. Oh, C. M. Cotten, D. K. Stevenson, B. J. Stoll, J. A. Lemons, R. Guillet, A. H. Jobe, and the National Institute of Child Health and Human Development Neonatal Research Network. 2005. Whole-body hypothermia for neonates with hypoxic–ischemic encephalopathy. New England Journal of Medicine 353(15):1574–1584.
Shankaran, S., A. Pappas, S. A. McDonald, B. R. Vohr, S. R. Hintz, K. Yolton, K. E. Gustafson, T. M. Leach, C. Green, R. Bara, C. M. Petrie Huitema, R. A. Ehrenkranz, J. E. Tyson, A. Das, J. Hammond, M. Peralta-Carcelen, P. W. Evans, R. J. Heyne, D. E. Wilson-Costello, Y. E. Vaucher, C. R. Bauer, A. M. Dusick, I. Adams-Chapman, R. F. Goldstein, R. Guillet, L. A. Papile, R. D. Higgins, and the Eunice Kennedy Shriver NICHD Neonatal Research Network. 2012. Childhood outcomes after hypothermia for neonatal encephalopathy. New England Journal of Medicine 366(22):2085–2092.
Simmonds, M., A. Llewellyn, C. G. Owen, and N. Woolacott. 2016. Predicting adult obesity from childhood obesity: A systematic review and meta-analysis. Obesity Reviews 17(2):95–107.
Smith, J. D., and J. Polaha. 2017. Using implementation science to guide the integration of evidence-based family interventions into primary care. Families, Systems, & Health 35(2):125–135.
Speer, E. M., L. K. Lee, F. T. Bourgeois, D. Gitterman, W. W. Hay, J. M. Davis, and J. R. Javier. 2023. The state and future of pediatric research—An introductory overview. Pediatric Research January 24:1–5. https://doi.org/10.1038/s41390-022-02439-4 [Epub ahead of print].
Sprague Martinez, L., K. Carolan, A. O’Donnell, Y. Diaz, and E. R. Freeman. 2018. Community engagement in patient-centered outcomes research: Benefits, barriers, and measurement. Journal of Clinical and Translational Science 2(6):371–376.
Stepien, K. M., B. Kiec-Wilk, C. Lampe, T. Tangeraas, G. Cefalo, N. Belmatoug, R. Francisco, M. Del Toro, L. Wagner, A. G. Lauridsen, S. Sestini, N. Weinhold, A. Hahn, C. Montanari, V. Rovelli, C. M. Bellettato, L. Paneghetti, C. van Lingen, and M. Scarpa. 2021. Challenges in transition from childhood to adulthood care in rare metabolic diseases: Results from the first multi-center European survey. Frontiers in Medicine (Lausanne) 8:652358.
Stewart, B., P. Lepola, G. F. Egger, F. Ali, A. J. Allen, A. K. Croker, A. J. Davidson, P. Dicks, S. N. Faust, D. Green, C. Hovinga, A. V. Klein, R. Langham, H. Nakamura, L. Pioppo, S. Ramroop, M. Sakiyama, I. Sanchez Vigil de la Villa, J. Sato, D. L. Snyder, M. A. Turner, S. Zaidi, K. Zimmerman, and T. Lacaze-Masmonteil. 2025. Requirements and special considerations for drug trials with children across six jurisdictions: 2. Ethics review in the regulatory approval process. Frontiers in Medicine 12:1539787.
Stoll, B. J., and H. Taegtmeyer. 2018. Challenges for today’s pediatric physician–scientists. JAMA Pediatrics 172(3):220–221.
Subasri, M., C. Cressman, D. Arje, L. Schreyer, E. Cooper, K. Patel, W. J. Ungar, M. Barwick, A. Denburg, and R. Z. Hayeems. 2023. Translating precision health for pediatrics: A scoping review. Children 10(5):897.
Sullivan-Bolyai, S., C. Bova, J. A. Deatrick, K. Knafl, M. Grey, K. Leung, and A. Trudeau. 2007. Barriers and strategies for recruiting study participants in clinical settings. Western Journal of Nursing Research 29(4):486–500.
Szatmari, P., K. Chawarska, G. Dawson, S. Georgiades, R. Landa, C. Lord, D. S. Messinger, A. Thurm, and A. Halladay. 2016. Prospective longitudinal studies of infant siblings of children with autism: Lessons learned and future directions. Journal of the American Academy of Child & Adolescent Psychiatry 55(3):179–187.
Tain, Y. L., Y. J. Lin, and C. N. Hsu. 2025. Animal models for studying developmental origins of cardiovascular–kidney–metabolic syndrome. Biomedicines 13(2):452.
Thomas, R., and M. Reeves. 2023. Mandatory reporting laws. Treasure Island, FL: StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK560690/ (accessed November 7, 2025).
Thrane, S. E., S. Wanless, S. M. Cohen, and C. A. Danford. 2016. The assessment and nonpharmacologic treatment of procedural pain from infancy to school age through a developmental lens: A synthesis of evidence with recommendations. Journal of Pediatric Nursing 31(1):e23–e32.
Turkel, S., and M. Pao. 2007. Late consequences of chronic pediatric illness. Psychiatric Clinics of North America 30(4):819–835.
Ward, K. P., and S. J. Lee. 2020. Mothers’ and fathers’ parenting stress, responsiveness, and child wellbeing among low-income families. Children and Youth Services Review 116:105218.
Wendler, D., J. E. Rackoff, E. J. Emanuel, and C. Grady. 2002. The ethics of paying for children’s participation in research. Journal of Pediatrics 141(2):166–171.
Werner, K. M. 2023. The gender gap in caring for children with medical complexity. Journal of Perinatology 43(7):835–836.
Wertheimer, A., and F. G. Miller. 2008. Payment for research participation: A coercive offer? Journal of Medical Ethics 34(5):389–392.
White, A., C. Grady, M. Little, K. Sullivan, K. Clark, M. Ngwu, and A. D. Lyerly. 2021. IRB decision-making about minimal risk research with pregnant participants. Ethics in Human Research 43(5):2–17.
Winter, S. S., J. M. Page-Reeves, K. A. Page, E. Haozous, A. Solares, C. Nicole Cordova, and R. S. Larson. 2018. Inclusion of special populations in clinical research: Important considerations and guidelines. Journal of Clinical and Translational Research 4(1):56–69.
Wittmeier, K. D., T. P. Klassen, and K. M. Sibley. 2015. Implementation science in pediatric health care: Advances and opportunities. JAMA Pediatrics 169(4):307–309.
Wojtys, E. M. 2020. Bone health. Sports Health 12(5):423–424.
Zhou, F., T. C. Jatlaoui, A. J. Leidner, R. J. Carter, X. Dong, J. M. Santoli, S. Stokley, D. C. Daskalakis, and G. Peacock. 2024. Health and economic benefits of routine childhood immunizations in the era of the Vaccines for Children program—United States, 1994–2023. Morbidity and Mortality Weekly Report 73(31):682–685.
This page intentionally left blank.