Strategies to Enhance NIH-Funded Pediatric Research: Optimizing Child Health (2026)
Chapter: 6 Implementing the Committee's Vision for the Future of Pediatric Health Research at NIH
6
Implementing the Committee’s Vision for the Future of Pediatric Health Research at NIH
As the mother of a child who died from diffuse intrinsic pontine glioma (DIPG), my experience with National Institutes of Health (NIH)-funded pediatric research is shaped by both deep gratitude and painful frustration. I’m grateful for the foundational discoveries NIH has made possible, such as the identification of the H3K27M mutation, which helped define DIPG as a distinct disease. . . . My son at age 4 participated in four early-phase clinical trials. These were not easy decisions; they were desperate ones. The research offered the only possibility of hope. Even though those trials didn’t save his life, we chose them because doing nothing was not an option. We did it to push science forward and to help the children who would come after him—because that is the only way we are going to find a treatment.
— Parent of young child NIH research participant
Our future society is already present among us in the form of children. Imagine a future where every child in the United States is given the chance not just to survive, but to thrive as a child and future adult, bolstered in a health care system rooted in discovery, access to all, and innovation. In this future, National Institutes of Health (NIH)-funded research into child health is not an afterthought but a national priority, built on investments that rival our most ambitious scientific endeavors. In this future, children, from the first days of life, benefit from our comprehensive understanding of the biological, environmental, and social factors that shape life course development of health, well-being, and resilience.
Prevention lies at the heart of this vision. NIH-supported breakthroughs in genomics, environmental monitoring, and digital health converge to
allow researchers and clinicians to identify risks before disease takes hold. Predictive models help families and clinicians intervene early to prevent asthma, diabetes, mental illness, and other chronic conditions long before symptoms appear. Childhood becomes the foundation for a healthy adulthood, with early interventions that transform the trajectory of lives and eliminate disparities that have persisted for generations.
In addition, a new era of therapeutics redefines what is possible for children with serious illness. Gene editing, immune modulation, artificial intelligence (AI), and regenerative medicine lead to cures, not just symptom control, for conditions that once led to lifelong disability or early death. Preventative strategies, treatments, and cures become personalized, safe, and developed from their inception with children in mind. The research and discovery that NIH supports today fuels a tomorrow in which no child’s potential is limited by biology, birthplace, or the inequities of our past systems.
Children today have more opportunities for health and longevity than at any point in history. Yet rising rates of chronic conditions, including obesity, anxiety, and other health challenges, signal that this progress is uneven and fragile. NIH has a unique opportunity to lead a new era of research that illuminates how rapidly evolving exposures and experiences shape child health trajectories. This includes studying the effects of pervasive electronic device use, information ubiquity, and shifting food environments as well as emerging environmental and social determinants. By investing in rigorous child-centered science on these modern exposures, NIH can generate actionable knowledge that empowers families, clinicians, and communities to foster resilience, prevent disease, and optimize lifelong health.
To build this future, the nation will need to act with urgency and intention. Success will require us to center children in the NIH research agendas and foster collaborations that unite families, communities, and researchers. The reward is profound: a generation of children who grow up healthier, stronger, and more fully able to contribute to the United States and the world around them. This chapter reiterates recommendations made in the preceding three chapters, organized under the committee’s four goals discussed in Chapter 1, and describes how this future could look if the recommendations are successfully implemented.
GOALS AND RECOMMENDATIONS FOR NIH-SUPPORTED PEDIATRIC RESEARCH
Pediatric health is a cornerstone of our national well-being. Increases in the U.S. average lifespan during the 20th century were in great part due to declines in child mortality from increased public health and sanitation, and decreased infectious diseases, prematurity, trauma, cancer, and other health issues. These advances would never have happened without the significant
and consistent federal investment, led by NIH (and supported by Congress), in basic, translational, and clinical research conducted by intramural NIH investigators and investigators across the nation in our universities, children’s hospitals, and biotechnology and pharmaceutical industries.
NIH is one of the most prestigious and revered research institutions in the world. As the world’s leading funder of biomedical research, it has the opportunity and responsibility to also be the world’s leader in advancing and innovating pediatric health research. This report provides the evidence and recommendations for how NIH, with the support of Congress, the executive branch, and partner federal agencies, can best fund, support, and coordinate research in pediatric health. This closing chapter recapitulates those recommendations in the context of the committee’s vision and goals.
More than ever before, federal and federally funded biomedical research is under heightened scrutiny. Debates over the size, scope, and organization of NIH and calls for increasing fiscal restraint pose significant challenges to long-term planning, infrastructure development, and the pursuit of bold scientific goals to improve the health of U.S. children. Perhaps paradoxically, these pressures also create an opportunity to reaffirm the unique value of pediatric research to the health and well-being of children and, because our children are our future, to demonstrate the significant return on investment our nation gains in health, prosperity, opportunity, innovation, and growth by prioritizing pediatric health research that is comprehensive, equitable, and groundbreaking.
When investments in pediatric health research are designed with rigor, focused on outcomes, and grounded in the needs of our children and the nation, they should command broad support across the federal government and the public. When these investments are visibly linked to national priorities such as educational readiness, workforce development, economic productivity, national security, and family well-being, there should be support for our children by funding and carrying out basic, clinical, and translational research. Children experience rapid, complex developmental, physical, and psychosocial changes with lifelong implications for their health, well-being, and resilience. The United States needs a new vision for pediatric research that positions pediatric research as a dynamic essential driver of population health across the life course rather than one limited to the pediatric years. This chapter presents the committee’s vision, in which there is an NIH-led pediatric research enterprise that is coordinated, data-enabled, and impact-oriented, designed to deliver lasting health benefits for all children while maximizing efficiency, accountability, and scientific relevance in an era of constrained resources. In this envisioned future, pediatric research that is efficiently stewarded and adequately resourced by NIH generates knowledge that is urgently needed, broadly applicable, and visibly impactful.
The committee was tasked by NIH to examine its portfolio of pediatric health research and to make recommendations on how NIH could improve
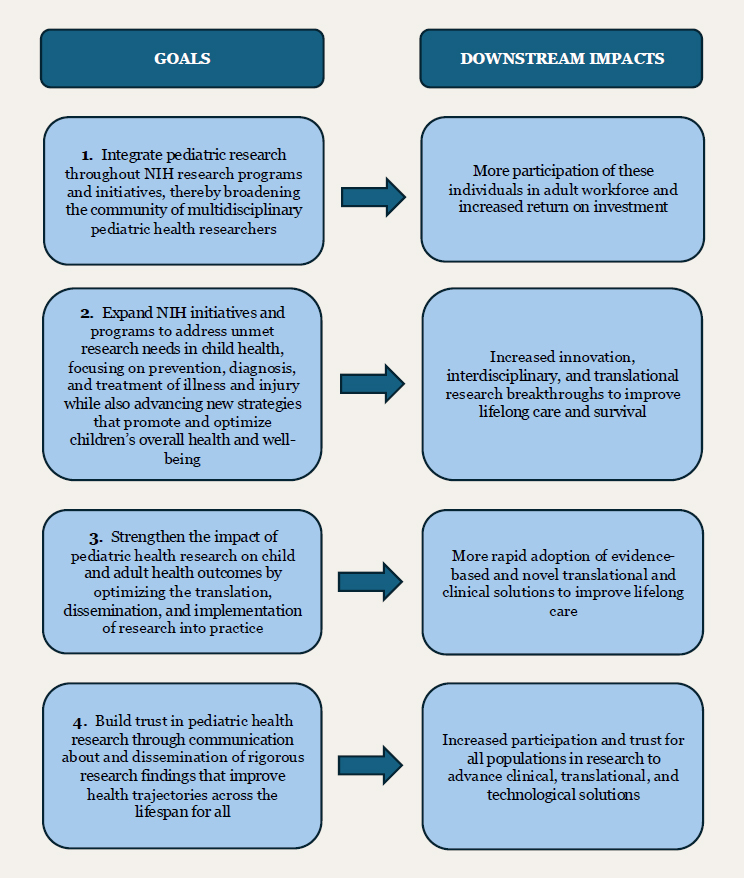
the support, management, and coordination of pediatric research throughout its institutes and centers (ICs). This report has outlined the committee’s areas of focus and has detailed its approach to assessing the effectiveness, strengths, and opportunities of the current NIH pediatric research portfolio. The committee developed a vision for where U.S. pediatric health research should head, and, to actualize that vision, the committee created a set of four goals (see Box 6-1). Figure 6-1 shows the downstream impacts that would occur with the achievement of the four goals.
The committee was tasked with making recommendations “focused on improving NIH’s overall support of child health research” while maintaining strong consideration for the burden on researchers and NIH staff. The committee’s recommendations serve as a series of action points that, if implemented, would help achieve the goals and ultimately improve pediatric health outcomes.
BOX 6-1
The Committee’s Vision and Goals for Pediatric Research at NIH
The United States will be the global leader in promoting child health, preventing childhood diseases and morbidity, reversing recent declines in child health, and using scientific discoveries through a robust, dynamic, and integrated pediatric research program at NIH. These goals will ensure that all children are supported, resilient, and thriving across the lifespan. To accomplish this vision, NIH would do the following:
GOAL 1: Integrate pediatric research throughout NIH research programs and initiatives, thereby broadening the community of multidisciplinary pediatric health researchers.
GOAL 2: Expand NIH initiatives and programs to address unmet research needs in child health, focusing on prevention, diagnosis, and treatment of illness and injury while also advancing new strategies that promote and optimize children’s overall health and well-being.
GOAL 3: Strengthen the impact of pediatric health research on child and adult health outcomes by optimizing the translation, dissemination, and implementation of research into practice.
GOAL 4: Build trust in pediatric health research through communication about and dissemination of rigorous research findings that improve health trajectories across the lifespan for all.
NOTE: NIH = National Institutes of Health.
Goal 1: Integrate pediatric research throughout NIH research programs and initiatives, thereby broadening the community of multidisciplinary pediatric health researchers.
Recommendation 3-1: The National Institutes of Health should adopt and apply a consistent agency-wide definition of pediatric research. The definition should recognize that pediatric research may include studies that (1) involve data or specimens from or about infants, children, adolescents, or young adults; (2) inform the prevention, diagnosis, or treatment of conditions affecting these populations; (3) advance the health, well-being, or resilience of children, adolescents, or young adults; or (4) investigate childhood origins of adult health conditions.
Historically, NIH has not consistently defined pediatric research across ICs. The committee recommends that NIH adopt the definition above to standardize what research is categorized as pediatric and to promote consistency in reporting. Pediatric research may encompass several distinct areas of study: research in which participants do not specifically exclude children, research in which participants specifically include children, or research on children with a clinical or public health focus on conditions or developmental processes that typically originate and manifest in childhood.
Recommendation 3-2: The National Institutes of Health (NIH) should improve the Research, Condition, and Disease Categorization (RCDC) system to better assess funding for pediatric research by (a) adding the new spending categories and (b) improving the Categorical Spending webpage in the NIH Research Portfolio Online Reporting Tools (RePORT).
The following new spending categories should be updated:
- Add pediatric developmental stages: during pregnancy; neonate (≤28 days); infant (<1 year old); early childhood, age 1–2 years (1- to 2-year-olds) and age 3–5 years (3- to 5-year-olds); middle childhood (6- to 11-year-olds); adolescence (12- to 17-year-olds); and young adults (18- to 24-year-olds).
- Add a Basic Science spending category and ensure its validity.
- Add new terms for pediatric health conditions to better reflect the breadth of pediatric health conditions. The process to determine these new terms should include child health expertise from each institute and center.
- To improve specificity of diagnostic labeling, consider switching eponyms to indicate diseases—e.g., change Cooley’s Anemia to “Beta Thalassemia Major.”
To improve the Categorical Spending (RCDC) webpage in RePORT, NIH should
- Publish a reference paper that clearly and comprehensively describes the RCDC classification methodology with sufficient detail that it can be replicated or a similar assessment can be done by the scientific community. Update the methodology report whenever the methodology changes.
- Evaluate the validity of key RCDC terms, such as “pediatric,” and update the classification to improve accuracy and remove possible biases.
- Allow more than one RCDC term to be exported in a specific project. (This functionality currently exists in RePORTER, but not RCDC.)
- Enable download of all projects funded by NIH for a given year.
- Add a flag to clearly identify extramural; intramural; and, specifically, Clinical Center research and ensure that this information is available for all research projects.
- Add a search field that links projects to their respective notice of funding opportunity, program announcement, program announcement with special review, request for application, notice of special interest, or request for proposal.
Currently, it is challenging to study NIH funding of pediatric research because of deficiencies in the Research, Condition, and Disease Categorization (RCDC) system that is used to categorize funding. The two recommendations above add transparency and clarity into the types of funded projects. Grant information in the future will be coded with more specific pediatric codes denoting the developmental stages and common pediatric health conditions that are currently missing in RCDC, making NIH-funded research more transparent and easier to track, thus helping to improve efficiency and accountability. RCDC will also have a code for basic research, making it possible to distinguish it from translational and clinical research.
Recommendation 4-1: All directors of National Institutes of Health institutes and centers that fund research should explicitly incorporate pediatric health and potential downstream life course impacts into their strategic plans, as childhood chronic conditions, exposures, and experiences may have disproportionate health impacts into adulthood. Include metrics such as those focused on investigators (e.g., funding rates and career advancement), scientific discovery (i.e., publications and innovations), impact on child health (e.g., morbidity, mortality), and downstream life course impacts (e.g., premature cardiovascular disease) to measure success in meeting their goals.
The committee recommends that each IC explicitly and transparently consider pediatrics and plan for the ways that they will fund research to improve child health. The committee’s recommendation would result in all 27 NIH ICs including pediatric research in their strategic plans, which act as roadmaps for establishing goals and research priorities. Including pediatrics in the written strategic plan will also help ICs and the NIH Pediatric Research Consortium (NPeRC) identify gaps and redundancy across NIH’s global plan for pediatric research.
Recommendation 4-2: The National Institutes of Health (NIH) director should
- Elevate the NIH Pediatric Research Consortium (NPeRC) (or its equivalent) to the Office of the Director, with membership including senior leaders within each institute or center (IC) with pediatric health and life course expertise who systematically review all relevant components of strategic plans, including progress and impact made in the past year. Each senior leader should then report back to their IC and to the NIH director.
- Provide appropriate infrastructure funding and adequate resources (including personnel) to enable NPeRC (or its equivalent) to accomplish its goals and for its members to participate in its work, which includes (1) identifying and addressing research gaps, (2) enabling a nimble response to emerging threats to pediatric health, (3) promoting greater efficiency and reach of ongoing initiatives through data harmonization, (4) spearheading novel partnerships, (5) providing consultation to ambitious inter-IC initiatives, and (6) ensuring representation and visibility at national scientific meetings and societies.
- Have NPeRC’s membership (or its equivalent) include expertise in pediatric bioethics. NPeRC should also partner with patient advocacy or community representatives and develop a communications plan to disseminate pediatric research findings to both the scientific community and the general public. NPeRC (or its equivalent) should be prepared to coordinate the NIH-wide response to any emerging pediatric research needs.
The inclusion of children in NIH-wide and inter-IC initiatives will be facilitated by an empowered NPeRC or its equivalent. Historically, it has been difficult to identify what NPeRC has done to support and enable NIH-funded pediatric research. Only through extensive requests for information and follow-up questions has the committee been able to identify many of the vitally important contributions NPeRC has made. This recommendation
would lead to an NPeRC that is overseen by the Office of the Director and is given sufficient resources and authority to be able to bring ICs together to foster the inclusion of pediatric research in all NIH-wide initiatives and to sponsor the development of new initiatives focused specifically on children.
Recommendation 5-2: National Institutes of Health (NIH) institutes and centers should award pediatric research grants and budgets that support the specific needs of pediatric studies. In addition, to address the mismatch between study timelines and funding cycles, NIH should
- Allow for extended or extendable award timelines and/or renewable grant mechanisms specifically designed for pediatric research, allowing investigators to sustain long-term studies that align with children’s developmental trajectories;
- Provide higher budget maximums to account for the need for representative recruitment and multi-center studies, when scientifically justified;
- Create supplemental funding opportunities specifically designed for longitudinal or follow-up data collection, which would enhance learning across the life course while capitalizing on previously funded and ongoing studies;
- Provide funding supplements to support multilingual research and community engagement to facilitate representation for generalizable results; and
- Increase access and usage of the Clinical and Translational Science Awards as a mechanism for promoting pediatric research.
Pediatric research should not be seen as a niche within adult medicine, but as foundational science that informs health across the lifespan. Prioritizing child health research is not only an investment in vulnerable populations; it is a strategic imperative for addressing long-term public health, as healthy children grow up to be adults.
– Neonatal pediatric researcher and recipient of NIH grants
Currently, most studies do not include children. Unfortunately, this is despite updated requirements for providing justification for any exclusion of children. In the committee’s vision, it is unacceptable to exclude children from a clinical study simply because it is somewhat difficult for the investigators. The committee envisions that NIH adopts and rigorously applies the following policy: children will be included in all research funded by NIH, unless including them is not scientifically justified. Integrating this policy change into NIH begins with weaving it into the fabric of each IC’s strategic plan. This perspective then guides the development of programs within the ICs, funding opportunity announcements, and intramural research programs at each IC.
We are challenged by time for pediatric research. To acquire enough data on enough patients within a 5-year funding mechanism is very hard. It is even more challenging when you are wanting to track change over time, which is important in child development. Longer-term funding mechanisms would be very appropriate for many studies of pediatric conditions that require longitudinal surveillance.
– NIH grant recipient
Every adult was once a child, and children are our country’s future. Research to support the current and future health of children must be prioritized. The committee envisions that the NIH extramural research program will be organized to ensure that pediatric research is appropriately prioritized. Since most diseases in children are much less common than in adults, pediatric research studies will routinely require multi-center involvement to generate scientifically adequate sample sizes. The extra cost of many such studies is not feasible under the current $500,000 direct-cost annual cap for most R01-level grants; thus, this will be increased to enable rigorous pediatric research to be conducted. The committee proposes that NIH use incentives to allow for longer research timelines to accommodate for developmental changes as they pertain to recruitment and follow-up.
Goal 2: Expand NIH initiatives and programs to address unmet research needs in child health, focusing on prevention, diagnosis, and treatment of illness and injury while also advancing new strategies that promote and optimize children’s overall health and well-being.
Recommendation 4-3: Initiatives within and across the National Institutes of Health (NIH) institutes and centers should coordinate with the NIH Pediatric Research Consortium (NPeRC) (or its equivalent) to include children and/or the downstream life course impact of childhood chronic disease, exposures, and experiences in studies in a strategic and deliberate way. NPeRC (or its equivalent) should support the enforcement of existing policies requiring that studies not exclude children or childhood downstream impacts on the life course without clear and adequate justification.
Initiatives across the ICs of NIH can lead to transformations in the diagnoses, treatment, and prevention of disease and the enhancement of health. The committee envisions that inter-IC initiatives will coordinate with NPeRC to include pediatrics in research from the beginning of studies unless there is a scientific justification for excluding children. Because ethical
considerations are sometimes highlighted as a barrier to the inclusion of children in research, the committee envisions that NIH will provide strong leadership in developing and disseminating foundational ethical guidance for research on pregnant women and children. This includes guidance about when it is acceptable that pregnant women and children are excluded from research. The committee envisions that NIH will be a leader nationally in the translation of this ethical guidance into appropriate regulations, thereby helping researchers conduct ethical, rigorous research with children.
As NIH develops foundational guidance on the ethical considerations of research in pediatrics, the committee recommends that the Office for Human Research Protections develop and disseminate clear guidance on when it is and is not appropriate to ask for assent from children participating in research and that it provide clarity on what state laws may prohibit adolescents from consenting to treatment. Developing a clear foundational ethical framework will support researchers across the United States who work with regulatory boards that have different levels of experience with issues involving children. The development of a national framework will also protect and support the nation’s children who will benefit from participating in and contributing toward safe research.
Recommendation 3-3: The National Institutes of Health (NIH) Clinical Center (CC) should continue to prioritize the current level of pediatric research, particularly on topics and using methods that are difficult to pursue in extramural environments (e.g., longitudinal studies of rare diseases and early-stage interventional trials) as well as on the long-term outcomes of interventions. Potential mechanisms include
- Increasing capacity while ensuring safety by overcoming operational constraints at the CC. Younger and more acute patient populations should be supported in early-phase interventional and observational protocols.
- Continued, expanded, or new partnerships with children’s hospitals, academic medical centers, schools, public health departments, and other large health centers across the country to best support pediatric populations.
- Offering pediatric education and training opportunities at the CC to continue to develop the specialized workforce needed to support this research.
- Collecting and presenting data in the annual report to the NIH Pediatric Research Consortium (or its equivalent) on the proportion of pediatric research studies, support, and usage by each institute and center at the CC.
A core part of NIH is the Clinical Center. The committee envisions that changes at the Clinical Center to accommodate pediatric research will be accelerated. These will include lowering the age for patients to allow admission of neonates and building the infrastructure to care for critically ill children. While some of the changes can occur in the Clinical Center on the NIH campus, the committee notes that it will be advantageous and efficient for other studies to be done in collaboration with research-intensive children’s hospitals, which can conduct the study protocols. The committee recommends that the Clinical Center continue to build up its expertise in all pediatric subspecialties to accomplish its expanded pediatric mission. Importantly, this will include the expansion of subspecialty fellowship programs at the Clinical Center. Additionally, the Clinical Center will collect data on the proportion of pediatric research studies, support, and usage by each IC and report these findings back to NPeRC (or its equivalent).
Goal 3: Strengthen the impact of pediatric health research on child and adult health outcomes by optimizing the translation, dissemination, and implementation of research into practice.
Recommendation 5-1: The National Institutes of Health (NIH) should work to create a culture of ethical inclusion of children in research, focused on protecting children through research rather than from research. To this end, NIH should develop measurable strategies and mechanisms to better implement and enforce existing policies to ensure inclusion of children at the project and proposal level. Strategies and mechanisms should include
- Updating the simplified review framework by explicitly considering the inclusion of children and its implications for health during the life course as a component of evaluating a proposal (i.e., its significance) during review.
-
Continuing to support the NIH Center for Scientific Review in implementing and monitoring structural changes to support pediatric research.
- Requiring and monitoring that pediatric representation is consistent with the number of applications on all relevant standing study sections.
- Ensuring provision of adequate justification for exclusion of children during grant review: If there is a scientific, legal, or ethical reason for the exclusion of children, this justification should be clearly articulated in the grant application
-
- and assessed and documented during the grant review process.
- Elevating prioritization of implications of research on the life course during scientific review: NIH should identify the impact of pediatric research on the life course as an important component of a proposal’s significance section and should do so directly within reviewer guidance. In addition, training for study section reviewers should encourage them to consider whether a proposal adequately addresses the importance of the research for the life course when evaluating the significance or impact of extramural grant applications.
- Improving transparency on inclusion: The Office of the Director of NIH should annually track and report justifications provided for the exclusion of children. This information should be included in the NIH annual report and published on the NIH Pediatric Research Consortium (NPeRC) website, or an NPeRC equivalent website, to track progress on the improved inclusion of children across NIH activities over time.
- Providing clearer institutional guidance about the ethical inclusion of children in research: NPeRC (or NPeRC equivalent), in consultation with the NIH Clinical Center’s Department of Bioethics, should develop a widely accessible toolkit and guidance on ethical issues in pediatric research that have the potential to unduly impede research if misunderstood, such as when to include children in research, provisions for assent, adolescent independent consent, age de-escalation, and payment for research participation.
Additionally, the smaller sample sizes and longer follow-up periods often required for pediatric studies can be perceived as limitations during peer review (and these longer follow-ups are associated with significant costs). From a process perspective, the current structure of study sections can disadvantage pediatric applications when they are reviewed by panels lacking pediatric expertise. This can lead to lower enthusiasm scores and reduced appreciation for the nuances of conducting rigorous research in neonatal or pediatric populations.
– Neonatal pediatric researcher and recipient of NIH grants
Applications to fund pediatric research are at a marked disadvantage when the review committees do not include experts in pediatric research. Having pediatric representation on these panels will ensure that members of the panel understand the unique needs, methods, and features of pediatric
research and can advocate to other review committee members on behalf of these pediatric research issues.
Pediatric research often involves complex regulatory requirements, including parental consent and assent from minors, which can slow down recruitment and add logistical hurdles not encountered in adult-focused research.
– Neonatal pediatric researcher and recipient of multiple NIH grants
The timeline for the translation of research findings supported by NIH to affect clinical care, practice, and policy is long, and the road to this translation is uneven. One of the barriers to translation in pediatric research arises from the fact that many pediatric diseases are classified as rare (affecting fewer than 200,000 individuals), which reduces the financial incentive for industry to develop therapeutic options for these diseases. This is aggravated by the reluctance to enroll children in clinical trials to test the efficacy and adequacy of these therapies. Another barrier, which impacts implementation, is that more than 50 percent of patients in our children’s hospitals are insured only by Medicaid, which often fails to adequately reimburse for the cost of care provided by hospitals and clinicians. This barrier to implementation is compounded by the fact that a majority of U.S. children (53 percent) are of non-White race and ethnicity, which has been the case since 2020. Despite the efforts of health care professionals to provide equal care for all, children who are of minority race and ethnicity,1 are poor, and come from families with limited English proficiency continue to experience access barriers to timely high-quality health care in the United States. Finally, advanced pediatric care is increasingly concentrated in urban centers, creating barriers for the one in five children living in rural communities.
Globally, under-age-5 child mortality has been cut in half in the last 25 years, thanks both to the use of new technologies and the adaptation of effective treatments to low-resource settings. A key to this has been the development of the new field of implementation science, defined by NIH as the “study of methods to promote the adoption and integration of evidence-based practices and interventions into routine health care and public health settings to improve our impact on population health” (NCI, n.d.). For example, at the National Cancer Institute, one goal is to “integrate implementation science into research across the cancer control continuum to improve the relevance to health care systems and population and public
___________________
1 “Minority” as used here refers to the entire U.S. population’s demographic statistics, not just that of U.S. children. See https://www.census.gov/library/visualizations/interactive/race-and-ethnicity-in-the-united-state-2010-and-2020-census.html (accessed October 8, 2025).
health” (NCI, 2022). If NIH were to apply a similar effort to integrate pediatric research into practice by using implementation science, it could help to lower the aforementioned barriers, leading to significant improvements in the health of all children.
Goal 4: Build trust in pediatric health research through communication about and dissemination of rigorous research findings that improve health trajectories across the lifespan for all.
Recommendation 4-2: The National Institutes of Health (NIH) director should
. . .
- Have NPeRC’s membership (or its equivalent) include expertise in pediatric bioethics. NPeRC should also partner with patient advocacy or community representatives and develop a communications plan to disseminate pediatric research findings to both the scientific community and the general public. NPeRC (or its equivalent) should be prepared to coordinate the NIH-wide response to any emerging pediatric research needs.
As a part of this recommendation, the committee suggests that NPeRC have bioethics representation to address concerns with consent and assent in pediatric research and patient advocacy representation to ensure that patients’ needs are understood and addressed. NPeRC’s communications staff will provide the expertise to disseminate pediatric research findings to a range of audiences, thereby promoting trust in pediatric health research, its findings, and the clinical guidelines, therapies, and devices that arise from the research.
For children to thrive, families and communities must be able to trust the research that informs their care. Building that trust starts with making sure that the science itself is rigorous and then sharing it in ways that are clear, non-political, and grounded in fact. When parents, pediatricians, teachers, and policy makers hear directly and consistently about research findings that improve children’s health (whether about nutrition, asthma, vaccines, or mental health), they are better equipped to support the choices that set children on healthier paths. Trust in experts and physicians is not just a professional issue; it is a cornerstone of a healthy nation. When people believe that the information they are receiving is reliable and free of political bias, they are more likely to act on it in ways that benefit children, families, and the broader society.
The challenge today is that the landscape of health information looks very different than it did even a generation ago. Thirty years ago, parents
might have gone to the library and pulled a medical reference book from a card catalogue; today, they are more likely to type a question into a search bar and find themselves scrolling through a flood of websites, videos, AI-generated answers, and social media posts. Because many of these sources lack the imprimatur of subject-matter experts, it can be difficult and frustrating to know what information to trust. This means that investigators must both do excellent science and communicate it in ways that meet families and communities where they are. To build trust in pediatric health research, NIH has an important role in shaping communication strategies that are fact-based, transparent, and accessible, ensuring that credible knowledge rises above the noise and ultimately improves the health of children across their entire life course.
A CODA
The future of pediatric research will be shaped not only by scientific innovation but also by the ability of leaders to steward public resources wisely, build powerful cross-sector coalitions, and keep children firmly at the heart of our national health priorities. NIH is uniquely positioned to lead this effort, but leadership in today’s climate means sharpening focus, aligning research to public concerns, and demonstrating and communicating impact from the laboratory to the community. Renewing our commitment to child health research is not simply an expansion or a transformation per se; instead, it is a reorientation toward prevention and a future in which every child can flourish. America’s children represent our most profound opportunity for shaping a better and healthier society. The earlier in the lifespan that we invest in child health, the greater the return in long-term health, well-being, and vitality throughout the life course of individuals, through generations of families, and in communities across the United States. Investing in pediatric health through prevention, targeted therapeutics, and cures not only improves lifelong health outcomes but also yields significant societal and economic benefits by reducing the burden of disease and fostering healthier communities and a more productive and vital society. National security, U.S. competitiveness, and future generations of Americans depend on our having healthy children.
REFERENCES
NCI (National Cancer Institute). n.d. About implementation science. https://cancercontrol.cancer.gov/is/about (accessed November 9, 2025).
NCI. 2022. Implementation science (IS). https://cancercontrol.cancer.gov/sites/default/files/2020-04/DCCPS-IS-Factsheet.pdf (accessed December 9, 2025).















