Firepower in the Lab: Automation in the Fight Against Infectious Diseases and Bioterrorism (2001)
Chapter: The Application of Mathematical Models in Infectious Disease Research
3
The Application of Mathematical Models in Infectious Disease Research
Roy M. Anderson
The European countries have concerns similar to those of the United States regarding emerging or introduced infectious diseases in at least three specific areas. First, because London and other major cities in Europe serve as hubs for international travel, there is a continual risk of the introduction of new pathogens or new strains of endemic pathogens from other regions of the world. Second, the general public has become increasingly sensitized to the issue of food safety because of outbreaks of highly pathogenic strains of E. coli, and the prion-related “Mad Cow disease,” with its associated disease in humans, new variant Creutzfeldt-Jakob Disease. Third, European governments are increasingly aware of a general vulnerability to acts of bioterrorism involving the deliberate release of dangerous pathogens.
This paper addresses the use of mathematical methods in research on infectious disease problems in terms of their emergence, spread, and control. Mathematical analysis is a scientific approach that can provide precision to complicated fields such as biology, where there are numerous variables and many nonlinear relationships that complicate the interpretation of observed pattern and the prediction of future events. Modern computational power provides us with extraordinary opportunities to sort through the growing volume of data being generated in the fields of biology and medicine. Many areas are ripe for the application of mathematical approaches such as the analysis of host and pathogen genome sequence information, the translation of genome sequence information into three-dimensional protein structure, the impact of global warming on ecological community structure, and analysis of the spread and con-
trol of infectious diseases in human communities. In the field of bio-terrorism, appropriate methods can provide many insights into vulnerabilities and how to reduce these plus plan for the impact of a deliberate release of an infectious agent.
History is instructive. The first application of mathematics to the study of infectious diseases is accredited to the probabilist Daniel Bernoulli. Bernoulli used a simple algebraic formulation to assess the degree to which variolation against smallpox would change mortality in a population subject to an epidemic of the viral disease. The underpinning algebra, converted into modern terminology, is as applicable today in the context of assessing how different control interventions will impact morbidity and mortality as it was in Bernoulli's day. It provides a powerful tool to demonstrate or assess the use or impact of a particular intervention before it is put into practice.
POPULATION GROWTH AND TRAVEL AS FACTORS IN DISEASE SPREAD
Infectious diseases remain the largest single cause of morbidity and premature mortality in the world today. There are dramatic differences in the age distributions of populations throughout the world, and by and large this difference is dominated by mortality induced by infectious agents. The most dramatic example of this at present is the unfolding impact of AIDS in sub-Saharan Africa, where life expectancy in the worst-afflicted countries such as South Africa, Botswana, and Zimbabwe is predicted to fall to around 35 years of age by 2020 (Schwartländer et al., 2000). The evolution and spread of infectious agents are greatly influenced by the population size or density of the host species. In a historical context it is important to note the extraordinary opportunities today for the spread of infectious agents, given the rapid growth of the world's population over the past millennium (see Figure 3.1). Today, the major population growth centers are predominantly in Asia. In the recent past they have been in China, but India is projected to have the largest population in the near future. In the context of the evolution of new infectious agents and their propensity for rapid spread in growing communities, Asia is one location where we need to have high-quality surveillance for clusters in space and time of unusually high rates of morbidity and mortality that may be attributable to the emergence of a new pathogen or new strain of an existing infectious agent.
There are many other aspects to population structure and growth that are of relevance to the spread of infectious agents. One of these is where mega-cities will be located in the future, which again is Asia (see Table 3.1). These cities have very particular characteristics—small cores in the center
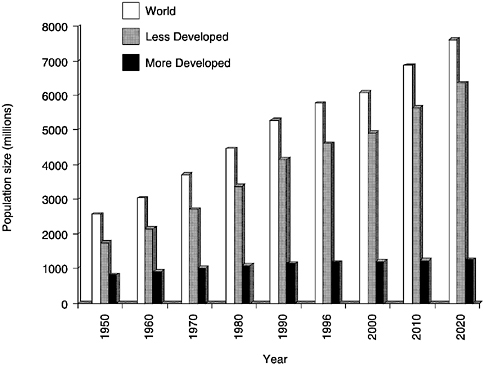
FIGURE 3.1 Projected world population size, 1950-2020.
with a degree of wealth and good health care in close proximity to very large surrounding peri-urban populations of extreme poverty, dense over-crowding, and poor sanitation and hygiene. These latter areas are important sites for the evolution of new infectious agents.
TABLE 3.1 Worldwide Increase in Megacities (number in different regions)
1970 | 1994 | 2000 | 2015 | |
Less Developed Regions | ||||
Africa | 0 | 2 | 2 | 3 |
Asia | 2 | 10 | 12 | 19 |
Latin America | 3 | 3 | 4 | 5 |
More Developed Regions | ||||
Europe | 2 | 2 | 2 | 2 |
Japan | 2 | 2 | 2 | 2 |
North America | 2 | 2 | 2 | 2 |
The rapid growth of international travel is also of great importance to the spread infectious disease. Of particular importance is travel by air, where transportation of infected individuals to the farthest corners of the globe can occur well within the time frame of the incubation period of virtually all infectious agents. The number of passengers carried by international airlines is growing linearly (see Figure 3.2). Closer examination of travel patterns show that the nodes and hubs of air travel are concentrated in particular centers, and cities in Asia increasingly serve as hubs for much international travel today.
EVOLUTION OF INFECTIOUS AGENTS
Quantitative study of the evolution of infectious disease is not well developed at present. Although in historical terms population genetics is a highly developed mathematical subject, the application of mathematical tools to the study of the evolution of, for example, influenza or antibody resistance is a neglected field with much research needed to provide predictive methods to help in policy formulation.
Evolutionary changes in infectious agents can occur rapidly under an intense selective pressure such as widespread drug therapy due to their short generation times relative to the human host. For example, a beta-lactamase-producing bacteria that is resistant to antibiotic treatment can,

FIGURE 3.2 Development of worldwide scheduled air traffic, 1970 to 1995 (predicted annual growth annual growth of 5.7 percent from 1997 to 2001 with 1.8 billion passengers in 2001.
in a period of a few years, rise from a very low prevalence to a frequency of more than 80 percent. An example is given in Figure 3.3 for the spread of resistance in bacteria infecting children in Finland. It is difficult to understand the precise pattern of the emergence of drug resistance without correlating changes in frequency with the intensity of the selective pressure. That intensity is related to the volume of drug use in a defined community, the data for which are not always publicly available. Two time series—drug volume consumption and the frequency of resistance—are essential to interpret these patterns. Once this relationship is determined, predictions can be made, with the help of simple mathematical models, of how reducing the volume of drug use will affect the frequency of resistance. More generally, in the study of the evolution of infectious agents it is of great importance to quantify the intensity of the selective pressure concomitant with recording changes in the genetic constitution of the pathogen population.
PLOTTING EPIDEMIC CURVES
The plague killed one-third of Europe's population in the seventeenth century epidemic, and at that time it was one of the most pathogenic
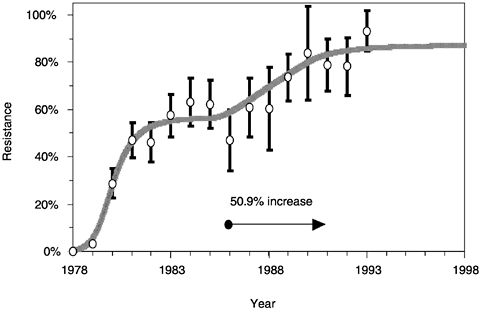
FIGURE 3.3 Frequency of TEMβ-lactamase-producing isolates of Morexella catarrhalis in Finnish children. Source: Nissinen et al. (1995).
organisms ever experienced by humankind. At the end of the last millennium an agent emerged that is both more pathogenic and has a greater potential to spread. The pathogen is HIV, which has a higher case fatality rate of nearly 100 percent, which is to be compared with a figure of roughly 30 percent for the plague. Mathematical models, specifically those describing epidemic curves, can serve as useful predictive tools to study the time course and potential magnitude of epidemics such as that of HIV.
An epidemic of an agent with a long incubation period, such as that found in HIV-1 infection and the associated disease AIDS, can be analyzed by constructing a set of two differential equations. One of the equations represents changes over time in the variable X denoting the susceptible population, and the other presents changes in Y denoting the infected population. The pair of equations can be solved by analytical or numerical methods, depending on the nonlinearities in their structures, to produce graphs that show changes in prevalence and incidence of infection over time. An example is plotted in Figure 3.4, which shows a simple bell-shaped epidemic curve that settles to an endemic state and the associated change in the prevalence of infection, which rises in a sigmoid fashion to a semistable state. The slight decline from the endemic prevalence is caused by AIDS-induced mortality.
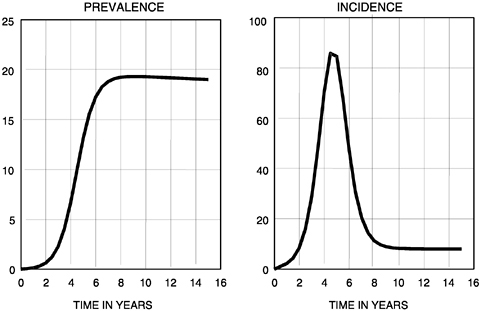
FIGURE 3-4 Relationship between prevalence and incidence in an HIV-1 epidemic.
Observed patterns are very similar to those predicted by this simple two-equation model. A specific example is provided by the time course of the HIV-1 epidemic in intravenous drug users in New York City. Simple epidemic theory suggests that saturation occurs in the course of the epidemic (as defined by the incidence of new infections), such that a natural decline occurs after an initial period of rapid spread due to a limitation in the number of susceptible individuals who are capable of contracting infection.
Concomitantly, a rise in the prevalence of an infection that persists in the human host will be sigmoid in form. In the absence of this knowledge of epidemic pattern, the decline in incidence could be falsely interpreted as the impact of interventions to control spread. This is exactly what happened in many published accounts of changes in the incidence of HIV-1 in different communities. Epidemiologists often interpreted a decline in incidence as evidence of intervention success. That may or may not be. The key issue in interpretation is to dissect the natural dynamic of the epidemic from the impact of any intervention. This example is a very simple illustration of the power of model construction and analysis in helping to interpret the pattern and course of an epidemic.
The principal factor determining the rate of spread of an infectious agent is the basic reproductive ratio, R0. It is defined as the average number of secondary cases generated by one primary case in a susceptible population. The components of R0 specify the parameters that control the typical duration of infection in the host and those that determine transmission between hosts (Anderson and May, 1991). An illustration of how R0 determines chains of transmission in a host population is illustrated diagrammatically in Figure 3.5.
If this chain of transmission events is an expanding one, the quantity R0 is on average greater than unity in value and an epidemic occurs. If R0 < 1, the chain stutters to extinction. The quantity R0 can be expressed in terms of a few easily measurable demographic parameters, such as the life expectancy of the host, the average age at which people are infected, and the average duration of protection provided by maternal antibodies.
Once such measurements are made for a given infection in a defined community, it is possible to derive estimates of the degree of control intervention required to suppress R0 below unity in value and hence eradicate infection and associated disease. In the case of vaccine-preventable infections such as measles and pertussis, such calculations enable estimates to be made of the critical vaccine coverage required to block transmission and the optimal age at which to immunize. For example, the simplest and most accurate way to estimate the average age at infection prior to control is via a cross-sectional serological profile, which records the decay in maternally derived antibodies and the rise in immunity due to infection.

FIGURE 3.5 Diagrammatic illustration of chains of transmission between hosts.
An illustrative example is presented in Figure 3.6, which records a cross-sectional serological survey for antibodies to the measles virus, with an average age at infection of approximately 5 years.
Although these methods are easily applied to derive estimates of transmission intensity (R0) and vaccine coverage required to block transmission, it is extraordinary how rarely they are used in public health practice. This may be due to a degree of mistrust of mathematical methods among public health scientists, perhaps related to the simplifications made in model construction. However, if the mathematical models are developed in close association with empirical measurement, and complexity is added slowly in a manner designed to promote understanding of which variables are key in determining the observed pattern, the methods can be very valuable in designing public health policy.
LIMITS OF MODELS
The real world is often much more complex than that portrayed in the simple assumptions that form a set of differential equations. Additional

FIGURE 3.6 Age-stratified serological profile for measles virus infection.
sets of derivatives are typically needed to capture this complexity, beyond the rate of change with respect to time recorded, for example, in Figure 3.3. The rate of change with respect to age is important (see Figure 3.5), as is the rate of spread of an epidemic with respect to space. Including time, age, and space results in three derivatives within a set of partial differential equations, necessitating significant computational power to generate numerical solutions of the spatial diffusion, age distribution, and time course of an epidemic. Today, however, such approaches are possible, and the resultant models can be powerful predictive tools when used in conjunction with good biological and epidemiologic data that record the key parameters.
These methods can be used in a wide variety of circumstances involving much biological complexity. One illustration is their use to assess the potential value of a vaccine against the chicken pox virus (and the associated disease, zoster, in immunocompromised individuals and the elderly). The mathematical models used in analyzing this problem are both deterministic and stochastic and were able to show that cases of zoster in the elderly contributed approximately 7 percent of the total transmission
intensity (i.e., primary infection plus transmission from zoster cases). Models were parameterized using data from small isolated island communities where it is possible to show that the virus would not be able to persist endemically without the help of transmission from zoster cases. This was informative from a public health perspective because it demonstrated that any immunization program targeted at the chicken pox virus and based on the immunization of young children would take a long time (i.e., many decades) to have its full impact because zoster in older people would still cause a degree of transmission among those not immunized.
Mathematical models have been applied most widely for the antigenically stable, vaccine-preventable childhood diseases. They have proved capable of predicting observed changes in both the average age of infection and the interepidemic period as a result of immunization programs. They have also proved reliable in predicting how a given level of immunization will change the herd immunity surface in a population. This surface represents changes in the proportion seropositive with respect to age and time (see Figure 3.7). Infection by the rubella virus provides a useful illustration of this predictive power. In this case the average age of infection
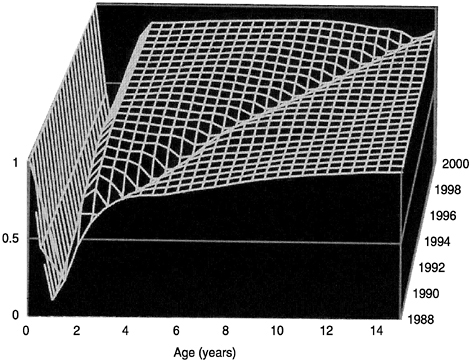
FIGURE 3.7 Herd immunity profile across age classes and through time.
was about 8 to 9 years prior to immunization in most developed countries. As we immunize a greater and greater fraction of the population, the age profile shifts, with the average age of infection increasing. It is important to understand that in a public health context this shift in the age distribution is not a failure of the immunization program but rather a natural consequence of the intervention. In the case of rubella this shift in the age distribution has important public health implications since infection during pregnancy can induce a serious disease—congenital rubella syndrome—in the unborn infant. Immunization programs targeted at the young must be at a sufficiently high level of coverage so as not to shift many young women into the pregnancy-age classes still susceptible to infection than was the case prior to the start of vaccination. Mass immunization induces a perturbation in the dynamics of the transmission system, which reduces the net force or rate of transmission of the infectious agent such that those who failed to be immunized have a lower exposure to the infectious agent. This is the so-called indirect effect of mass immunization. Normally, this is beneficial; however, in the case of rubella, infection at an older age carries an increased risk of serious morbidity (in this case for the unborn child of a pregnant women who contracts the infection).
CONSTRUCTING COST-BENEFIT MODELS
New techniques, such as saliva-based antibody tests, can facilitate the construction of herd immunity profiles for many infectious agents, including influenza. Using such epidemiologic data, model development can go one stage further and ask questions about vaccine efficacy and cost effectiveness of different control interventions. Virtually all published cost-benefit analyses in this area are incorrect because they typically only take into account the direct benefit of immunization in protecting the individual who is immunized. The indirect benefit is the herd immunity effect, as discussed in the preceding section, where those still susceptible experience a reduced rate of infection due to the immunity created by the vaccination of others. The percentage gain from the indirect effect of herd immunity is often quite dramatic, particularly at high vaccine uptake. This point is illustrated in Figure 3.8 for measles infection. It means that many immunization programs for the common vaccine-preventable infections are much more cost beneficial in terms of reducing morbidity and mortality than is currently appreciated.
Much of current methodology is designed for the antigenically stable infectious agents. Many important pathogens, particularly bacterial pathogens, exist as sets of antigenically related strains. Strain structure often varies between different communities and any significant change over time that is due to unknown selective pressures. Strain structure is best
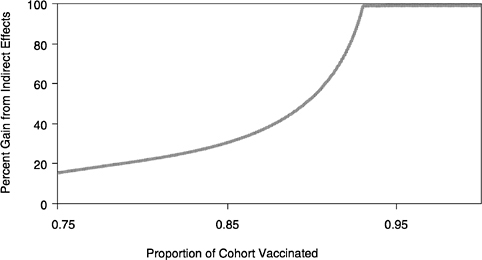
FIGURE 3.8 Percent gain in immunity from the indirect effects of herd immunity.
measured in terms of some easily identifiable phenotypic property. In the case of vaccine development, one such property would be recognition of the antigens of the pathogens recognized by the human immune system. Consider the case of a single dominant antigen coded for by a defined gene. If the pathogen expresses several epitopes on a particular gene and these epitopes are variable between strains, or where there is recombination taking place, the model must account for the dynamics of an evolving system in which different strains may have antigenically distinct combinations of epitopes. For example, if there were a whole series of phenotypes (= strains) that share some but not all alleles coding for the different epitopes, exposure to one strain may not confer immunity to another strain if that phenotype has a different set of alleles. If an organism shares all its alleles with another strain, the immune system is likely to confer complete protection against either strain from past exposure to either strain. If it shared only one out of, say, three alleles, there may be a degree of cross-protection but not complete protection.
These multistrain systems are dynamically very complex, and cycles plus chaos may be generated for fluctuations in prevalence over time by even simple models that represent cross-immunity generated by exposure to pathogens that share alleles. Much more research is required in this area to generate predictive tools to further our understanding of the likely impact of a new generation of vaccines targeted at antigenically
complex bacterial pathogens, such as the pneumococcal organisms where there is a degree of cross-immunity between different strain types. It is important to understand these complex systems not only for the assessment of vaccine impact but also to understand fluctuations in strain structure over time in unimmunized populations.
THE CASE OF MAD COW DISEASE
Mad Cow disease (i.e., bovine spongiform encephalopathy [BSE]) is induced by a transmissible etiological agent in the form of an abnormal form of the prion protein. In the mid 1980s the disease developed as a major epidemic in cattle in the United Kingdom and it still persists in Great Britain but at a low and slowly decreasing rate of incidence (see Figure 3.9). The epidemic was created by the recycling of material from infected cattle in animal feeds given to cattle from the late 1970s through the 1980s. The concern for human health first arose in 1996 with the first report of a new strain of Creutzfeldt-Jakob disease (vCJD) in humans, which appeared to be related to BSE. Subsequent research has suggested that exposure to the etiological agent of BSE induces vCJD. The prion diseases can be diagnosed on the onset of characteristic symptoms, but no

FIGURE 3.9 BSE epidemic in Great Britain (incidence/year).
reliable test exists at present to detect infection with the etiological agent prior to the onset of disease. The diseases are characterized by long incubation periods. In cattle the average incubation period is 5 years, while that of vCJD in humans is thought to be much longer.
Given a time course of the incidence of infection of BSE in cattle plus knowledge of the incubation period distribution, an important epidemiologic task is to estimate by back-calculation methods the numbers of infected animals that might have entered the human food chain via the consumption of infected beef or cattle products. These back-calculation methods were first used in the study of the AIDS epidemic to estimate the prevalence of HIV infection given observed time series of AIDS cases. The problem is more complicated in the case of BSE given the short life expectancy of cattle, which is on the order of 2 to 3 years from birth. This observation implies that most infected animals are slaughtered prior to the onset of symptoms of disease. Mathematical models that meld cattle demography with epidemiologic details of the incubation period distribution of BSE plus the major transmission route (horizontal and vertical) are required to assess the degree of exposure of the human population in Great Britain to the BSE agent.
Using such models, recent research has detailed the degree of exposure, which records temporal changes in the incidence of BSE, the incidence of new infections, and the prevalence of infected animals (Anderson et al., 1996). Taking the analysis further, to estimate the potential size and duration of the vCJD epidemic in humans is much more difficult due to the many unknowns surrounding this new disease. The major ones are the incubation period distribution and the infectivity of a unit quantity of contaminated beef. To complicate matters further, the density of abnormal prion protein in an infected cow changes significantly over the incubation period of the disease. It rises to very high levels in the brain and other neural tissues in the period immediately before the onset of symptoms of disease. Models can be constructed to make predictions of the possible future course of the epidemic, given the past history of exposure to BSE-contaminated material plus the observed time course of the incidence of vCJD. However, care must be taken to carry out many simulations, which embed methods for varying the values of the key unknown variables over ranges designed to cover sensible limits to these values. Such work requires considerable numerical analysis to generate prediction bounds on the future course of the epidemic. These bounds are very wide and do not eliminate either very small or very large epidemics. The major policy conclusion from this research is simply that the future is very uncertain and will remain so for many years. However, this conclusion is still useful since in its absence some scientists or policymakers may have been tempted to opt for one or other of the small or large epidemic scenarios and plan
accordingly. The only sensible conclusion at present is that a large epidemic spread over many decades cannot be ruled out. The methods developed for the analyses of the BSE and vCJD epidemics may be of use in the future in studying the spread of other new infectious diseases.
EXPERIMENTAL SYSTEMS IN BIOLOGY
An exciting and developing area in the application of mathematics in biology and medicine is in understanding how the immune system interacts with the pathogen in an individual host. All the techniques that are used in epidemiologic population-based studies can be applied rigorously to the study of the population dynamics and evolution of pathogens in the human host. In addition, the opportunities for measurement and quantification of key parameters is in some sense much greater in this area due to new molecular and other methods for quantifying pathogen burden and associated immunological parameters such as cell life expectancies and rates of cell division.
One of the most important areas of application has been in HIV research, where a series of studies have used simple deterministic models to study HIV pathogenesis, viral life and infected cell life expectancies, the rate of evolution of drug resistance, and the dynamics of the immune system under invasion by the virus (e.g., Ho et al., 1995; Ferguson et al., 1999). Similar methods are now being used to study bacterial pathogens and other viral infections. The human immune system is very complex and nonlinear, especially in its response to replicating pathogens. There seems to be little doubt that mathematical models will play an important role in helping to unravel this complexity. Furthermore, melding this type of research with studies of the pharmacokinetics and pharmacodynamics of therapeutic agents designed to kill pathogens or inhibit their replication should provide new insights into the design of more effective therapies or the design of better ways to use existing drugs. A particular area of importance in this context will be the study of how best to slow the evolution of drug resistance, both for antivirals and antibacterial agents.
SUMMARY
Today, sophisticated mathematical models can be developed to estimate the size and speed of spread of an emerging epidemic, irrespective of the nature of the infectious agent or the size and spatial distribution of the affected population. These models not only can help in the development of policy options for containment but can also assist in the development of guidelines for effective population treatment programs.
REFERENCES
Anderson, R., and R. May. 1991. Infectious Diseases of Humans: Dynamics and Control. Oxford: University of Oxford Press.
Anderson, R. M., C. A. Donnelly, N. M. Ferguson, M. E. J. Woolhouse, C. Watt, et al. 1996. Transmission dynamics and epidemiology of BSE in British cattle. Nature, 382:779-788.
Ferguson, N. M., F. de Wolf, A. C. Ghani, C. Fraser, C. Donnelly, et al. 1999. Antigen driven CD4+ T-cell and HIV-1 dynamics: Residual viral replication under HAART Proceedings of the National Academy of Sciences, 21:96(26):15167-15172.
Ho, D., A. U. Neumann. A. S. Perelson, W. Chen, J. M. Leonard, and M. Markowitz. 1995. Rapid turnover of plasma virions and CD4 lymphocytes in HIV-1 infection Nature, 373:123-126.
Nissinen A, M. Leinonen, P. Huovinen, E. Herva, M.L. Katila, S. Kontiainen, O. Liimatainen, S. Oinonen, A.K. Takala, P.H. Makela. 1995. Antimicrobial resistance of Streptococcus pneumoniae in Finland, 1987-1990. Clinical Infectious Diseases, 20(5):1275-1280.
Schwartländer, B., G. Garnett, N. Walker, and R. Anderson. 2000. AIDS in the new millennium. Science, 289:64-67.