Carving Our Destiny: Scientific Research Faces a New Millennium (2001)
Chapter: The Minds of Humans and Apes Are Different Outcomes of an Evolutionary Experiment
1
The Minds of Humans and Apes Are Different Outcomes of an Evolutionary Experiment
Daniel J. Povinelli
Is there anything truly distinctive about the human mind? Charles Darwin did not think so. Indeed, in trying to counter a perceived weakness of his theory, Darwin (1871, p. 445) found it necessary to claim that there was “no fundamental difference” between the mental faculties of humans and other animals. After all, if he could establish that our species did not possess any unique mental features, then there would be less need to invoke a Divine account of our origin. And so, in an extended survey of the animal kingdom, Darwin marshaled an impressive array of anecdotes to defend a claim that he considered self-evident—animals behave like us, therefore they think like us. Rather than viewing it as one kind of mind among many, Darwin presented the human mind as a brighter, more talkative version of other minds; minds that could themselves be arranged in linear order according to how closely they approximated the complexity of the human mind. Darwin’s argument fell on the sympathetic ears of George John Romanes, who, shortly after Darwin’s death, outlined a method for a new science whose purpose would be to compare the mental structures of humans and animals: “Starting from what I know of the operations of my own individual mind, and the activities which in my own organism they prompt, I proceed by analogy to infer from the observable activities of other organisms what are the mental operations that underlie them” (Romanes, 1882, pp. 1–2). Comparative psychology was thus born of an argument by analogy. Indeed, Romanes’ method was
Cognitive Evolution Group, University of Louisiana at Lafayette
really just a restatement of what David Hume (1739–1740) had proclaimed a century earlier: Where humans and animals share similar behavior, so too must they share similar minds.
In this chapter, I explore the possibility that Hume, Darwin, and Romanes were wrong. The results of our comparative research program with chimpanzees and human children suggest that the human species may have evolved fundamentally new psychological abilities—abilities that arose not as graded improvements of general cognitive faculties, but as specializations that were woven into existing ancestral neural systems. These innovations in human cognition may have done more than just provide our species with additional psychological capacities. They may have forever altered the way in which we are able to think about and interpret our social behaviors—behaviors that for the most part evolved long before we did. In short, there may be a far more subtle and complex relationship between what we think and how we behave than what was suspected by Hume, Darwin, and Romanes, or the generation of psychologists who followed in their intellectual footsteps.
EVOLUTIONARY HISTORY OF A COGNITIVE SPECIALIZATION
In cultures around the world, humans exhibit a stubborn penchant for explaining behavior in terms of internal mental states (see Avis and Harris, 1991; Lillard, 1998a, 1998b; Povinelli and Godfrey, 1993; Vinden and Astington, 2000). Even in our own culture, where behaviorists admonished us against doing so for the better part of a century, we refuse to stop reflecting on our thoughts, wants, desires, and beliefs, or worrying about similar states in others. But do other species similarly suppose that behavior is a product of an internal set of mental states? Perhaps no other issue strikes so clearly at the heart of what it may mean to be human. Premack and Woodruff (1978, p. 515) framed the question succinctly by asking whether chimpanzees have a “theory of mind.” “A system of inferences of this kind,” they observed, “may properly be regarded as a theory because such [mental] states are not directly observable, and the system can be used to make predictions about the behavior of others.” Premack and Woodruff suggested an affirmative answer to the question of whether chimpanzees possess such a theory of mind, but it is now clear that this is an issue that is far too complex to be resolved by the results of any single experiment. Over the past nine years, we have developed a diverse set of experimental procedures for investigating various facets of theory of mind, and our results point to a very different conclusion compared with the one reached by Premack and Woodruff. Our work points to the possibility that humans have evolved a cognitive specialization for reasoning about mental states.
Some will immediately object to the idea that there are human cognitive specializations, first, by invoking parsimony and, second, by alleging that any such suggestions are anthropocentric. After all, have we not now seen the remarkable behavioral similarities between humans and chimpanzees, such as their abilities to make and use tools, to deceive and manipulate each other, and to recognize themselves in mirrors? And is this not enough to convince us that chimpanzee behavior is governed by an understanding of the social and physical world fundamentally similar to our own?
But before rushing to the conclusion that similarity in behavior implies comparable similarity in mind, we might pause and consider the development of our own minds. Indeed, the most immediate consequence of Premack and Woodruff’s (1978) initial report was to excite a flurry of research into the development of young children’s understanding of mental states. And, despite the relatively young state of the field, it seems increasingly clear that there are several major transitions in the human understanding of the mental world. No longer can we think of the three-year-old’s mind as a duller, incomplete version of the five-year-old’s. Rather, psychological development seems to entail successive changes in children’s representational capacities so that children of different ages possess different kinds of understandings (e.g., Karmiloff-Smith, 1992). At the same time, observations of their spontaneous interactions with their family and peers reveal a general similarity in their emotions, perceptions, and basic behavioral patterns. Thus, Hume’s argument by analogy begins to break down even when we restrict our study to human beings. Worse yet, just as behavioral similarities between children of different ages can obscure psychological differences, so too may behavioral similarities between humans and apes obscure psychological differences.
TOWARD THE SYSTEMATIC STUDY OF THE EVOLUTION OF THEORY OF MIND
In my doctoral dissertation, I reported the results of several experiments I conducted to compare the social intelligence of monkeys, apes, and humans (Povinelli, 1991). But I finished this work with a nagging sense of dissatisfaction. There were few experimental studies of theory of mind in other species, and it seemed that very little progress had been made. In reflecting on the inconclusive nature of this early research, it became apparent that a new approach to comparing human and ape cognition was needed. Several kinds of projects were possible, each with its own strengths and weaknesses. For example, one could take a single chimpanzee, raise it in the company and culture of humans, and test the animal as it developed. Indeed, this has been the traditional approach to
studying ape cognition. But would it be possible to maintain the objectivity of such a project? And even if it were, would anyone else be convinced of its objectivity? Finally, would such a project meet one of the central requirements of any scientific undertaking —could it be meaningfully replicated? Another approach was to test large numbers of captive chimpanzees living in social groups. But there were serious problems here as well. For example, without sufficient exposure to humans, how could we be confident that the animals were comfortable enough with our testing procedures that they would perform at the upper limits of their abilities?
In the end, I settled on the following course. I selected 7 two-year-old chimpanzees who had been reared together in a nursery with human caretakers (see Figure 1). These seven animals were transferred to an indoor–outdoor compound that was connected to a specially designed testing facility (see Figure 2). This facility allowed us to test each ape in turn for 10–20 minutes at a time. Thus, while the others played, one of them could be transferred into an outside waiting area, which was connected by a shuttle door to an indoor testing room. Typically, this animal waited outside as a trial was set up indoors. When the shuttle door opened, the animal was free to enter the laboratory and respond to the task. A Plexiglas panel separated the apes from the humans. We used this panel for two reasons, even when the apes were young enough that it was unnecessary. First, it offered unambiguous response measures for many of our tests (i.e., which hole they reached through); but more important, we wanted to establish a predictable setting and routine that we could use with these animals throughout their youth and later into their adolescence and adulthood as they reached their full size and strength. That this approach has paid off is clear from the fact that as young adults our animals still eagerly participate in testing two to three times a day. To be sure, our project has had its limitations, but as will become clear, it has had its unique set of strengths as well.
This, then, was our approach: Rear a cohort of chimpanzees together, while simultaneously exposing them to human culture; follow them through their juvenile years into adulthood, and compare their social understanding to that which develops in human children. However, such work would require as fair a set of comparisons with young children as possible, and so we established a center where similar experiments with young children could be conducted. And although I do not discuss it in this chapter, this center has also allowed us to probe other, underexplored aspects of children’s developing understanding of themselves in time—developments that may ultimately prove crucial in defining both the similarities and the differences between humans and apes.
For the past nine years, our cohort of seven apes has participated in dozens of experiments investigating their understanding of mental states

FIGURE 1 Seven chimpanzees (Group Megan) who have formed the basis for our long-term project exploring the nature of chimpanzees’ reasoning about the social and physical world. These chimpanzees began this project when they were 30–42 months old. They were between the ages of four and five years old when these photographs were taken.
such as attention, intention, desire, knowledge, and belief, as well as their understanding of self (see Povinelli and Eddy, 1996a, 1996b, 1996c, 1997; Povinelli and O’Neill, 2000; Povinelli et al., 1993, 1994, 1997, 1998, 1999; Reaux et al., 1999; Theall and Povinelli, 1999). Where informative, we have also tested large numbers of other chimpanzee subjects (from infants to adults). In concert with our work with young children, the results of these studies have allowed us, in the very least, to gain a clearer perspective on the question that motivated me to pursue this research in the first place: Are there unique aspects of human cognition, and if so, how do they emerge from among the complex set of developmental pathways that we share in common with chimpanzees and other primates?
UNDERSTANDING VISUAL PERCEPTION: A CASE STUDY
The Mind of the Eyes
Although we have investigated and compared numerous aspects of chimpanzees ’ and children’s understanding of self and other, we have focused much of our effort on determining whether apes (like us) interpret the eyes as windows into the mind. In short, one of the main agendas of our work has addressed what chimpanzees know about “seeing.” This is critical for questions concerning the evolution of theory of mind because of the importance we place on the eyes in our understanding of attention and knowledge. In one sense, the eyes occupy center stage in human folk psychology. Humans understand the perceptual act of seeing as far more than just a physical relation between eyes and objects and events in the world, and this understanding arises fairly early in development. For example, a four-year-old girl observes someone lift the lid of a box and look inside and automatically infers that the person knows its contents. In contrast, she does not make this assumption when someone merely touches the box. Thus, even though she cannot see the information entering a person’s eyes and then forming the mental state of knowing the box’s contents, she (like an adult) assumes that something very much like that has happened. This may seem trivial, until we try the same test with three-year-olds and discover that they appear oblivious to the psychological difference between the person who looked into the box and the person who only touched it (Gopnik and Graf, 1988; O’Neill and Gopnik, 1991; O’Neill et al., 1992; Perner and Ogden, 1988; Povinelli and deBlois, 1992b; Ruffman and Olson, 1989; Wimmer et al., 1988).
Does this mean that three-year-olds have no understanding of the mentalistic aspects of visual perception? Not necessarily. Even though they may not appreciate that seeing (or other forms of perception) causes internal mental states, they do seem to understand that seeing is “about”
(or refers to) events and objects in the external world. In short, they seem to realize that someone who is looking at an object or event is subjectively connected to it by something that we label “attention.” Many years ago, Flavell and his colleagues conducted an impressive series of studies that revealed at least two clear levels in the development of preschoolers’ understanding of seeing (see Flavell et al., 1978, 1980, 1981; Lempers et al., 1977; Masangkay et al., 1974). The first level is exhibited by children aged two to three who possess a firm understanding of whether or not someone can see something. For example, sit across a table from a child and show the child a picture of a turtle so that it is right side up from the child’s perspective, but upside down from yours. Three-year-olds know that you can see the turtle, and that if you put your hands over your eyes you can no longer see it. But they do not seem to understand seeing at a second, more sophisticated level: They fail to understand that, from your perspective, the turtle appears different (i.e., upside down).
Even infants display some knowledge about visual gaze. In a landmark report over two decades ago, Scaife and Bruner (1975) demonstrated that very young infants will turn and look in the direction they see someone else looking. Since then, a number of carefully controlled studies have explored the emergence of gaze following (or, as Scaife and Bruner called it, “joint visual attention”). Although there is disagreement about the exact timing of its development, some capacities related to gaze following may emerge as early as six months (Butterworth and Cochran, 1980; Butterworth and Jarrett, 1991; Corkum and Moore, 1994; Moore et al., 1997). By 18 months of age, however, the ability is well consolidated, as toddlers will (a) follow an adult’s gaze into space outside their own visual field, (b) precisely locate the target of that gaze, and (c) reliably follow the gaze in response to eye movements alone (without accompanying movements of the head).
Although gaze following is of obvious practical utility, its psychological standing remains less clear. Some, such as Baron-Cohen (1994) of Cambridge University, interpret gaze following as prima facie evidence that infants are explicitly aware of a psychological connection between self and other. In other words, infants turn to follow their mother ’s gaze because they know that she is looking at something, that she sees something, that something has engaged her attention. This account grants infants their first (albeit limited) glimpse into the visual psychology of other people. Other researchers are more cautious, maintaining that gaze following (especially in very young infants) may have little to do with an appreciation of internal psychological states (Butterworth and Jarrett, 1991; Moore, 1994; Povinelli and Eddy, 1994, 1996a, 1996b; Tomasello, 1995). Processes such as hard-wired reflex systems, learned behavioral contingencies, and attentional cuing have all been offered as possible explanations of the emergence of this behavior. There is, finally, a middle ground that inter-
prets the behavior as a causal precursor to a later-emerging, more explicit representation of attention in toddlers. Early gaze following is seen as a fairly automatic response, which later provides a context for developing an understanding that attention is something distinct from action itself.
Gaze following is not the end of the story. Our information processing about the eyes of others is connected to emotional as well as cognitive systems. Making eye contact, for example, is a highly significant emotional experience for infants long before they appreciate the attentional aspect of seeing (Hains and Muir, 1996; Symons et al., 1998; Wolff, 1963). And it is not just human infants who display such sensitivity. Stare into the eyes of many species of Old World monkeys and note their immediate, hostile reaction (Perrett et al., 1990; Redican, 1975). In contrast, mutual gaze plays a more flexible role in humans and other great apes. Here, mutual gaze is an important factor in mediating both agonistic and affiliative social interactions (de Waal, 1989; Gómez, 1990; Goodall, 1986; Köhler, 1927; Schaller, 1963). In chimpanzees, for example, establishing mutual gaze seems to be especially important during “reconciliatory” social interactions that immediately follow conflicts (de Waal, 1989). In general, sensitivity to the presence of eyes appears to be widespread among animals, presumably because the presence of a pair of eyes is linked with the presence of a predator. In a well-known example, Blest (1957) studied the function of so-called “eye spots ” on the wings of butterflies. He experimentally demonstrated that butterflies with such spots are less likely to be eaten by predatory birds (see also Burger et al., 1991; Burghardt and Greene, 1988; Gallup et al., 1971; Ristau, 1991).
But does the evolved sensitivity to the presence, direction, and movement of the eyes indicate an understanding of their connection to internal mental states, even simple ones such as attention? Does the bird who averts from striking a butterfly after being flashed a set of eye spots entertain the notion of having been “seen”? In this case, our intuition may reply, “no,” but perhaps only because birds are involved. And what about gaze following in human infants, or possibly other species? Gaze following inhabits a contentious middle ground where intuitions clash. Clearly, then, we need to move beyond intuition and instead ask whether we can experimentally disentangle alternative psychological accounts of gaze following. In this way, we can begin to make real progress toward understanding whether other species (especially those most closely related to us) reason about visual perception in the mentalistic manner that we do.
Gaze Following: First Steps Toward Understanding Attention?
To begin, we might ask if chimpanzees possess a gaze-following system that is homologous to that of humans, and if so, the extent of similarity between the two. Following suggestive accounts from field research-
ers (e.g., Byrne and Whiten, 1985), several years ago we conducted the first experimental studies of gaze following in nonhuman primates (see Povinelli and Eddy, 1996a, 1996b, 1997; Povinelli et al., 1999). Initially, we simply sought to determine whether chimpanzees follow gaze at all. Our procedure involved our subjects entering the laboratory and using their natural begging gesture to request an apple or banana from a familiar experimenter who sat facing them. On most trials, the experimenter immediately handed the subjects the food. On probe trials, however, the subjects were randomly administered three conditions that had been carefully choreographed ahead of time. On control trials, the experimenter looked at them for five seconds and then handed them a reward as usual. These trials allowed us to determine the subjects’ baseline levels of glancing to specific locations in the room. On eyes-only trials, the experimenter turned his or her head and looked at a target above and behind the chimpanzee for five seconds (see Figure 3a). On eyes-only trials, the experimenter diverted only his or her eyes to the same target, keeping the rest of the head motionless. The results depicted in Figure 4 reveal several things. First, the chimpanzees virtually never looked above and behind themselves on the control trials. In clear contrast, not only did they follow our gaze on the trials involving whole head motion, they even did so in response to eye movements alone. We have now replicated and extended this effect on a number of occasions and have demonstrated that chimpanzees follow gaze with at least the sophistication of 18-month-old human children (see Table 1).
These findings were important because they experimentally established that chimpanzees, like humans, are extremely interested in where others are looking. More recently, researchers in Perrett’s laboratory at the University of St. Andrews, and Tomasello’s laboratory at Emory University, confirmed our speculation that gaze following might be widespread among primates (Emery et al., 1997; Tomasello et al., 1998). Indeed, it seems likely that gaze following evolved through the combined effects of dominance hierarchies (needing to keep track of who is doing what to whom) and predation (exploiting the reactions of others to discover the location of potential predators). Having established this similarity, we next turned to the more central question of how this similarity might help us to understand whether chimpanzees interpret visual perception as a projection of attention.
Knowing That You Cannot See Through Walls
A chimpanzee following your gaze tugs almost irresistibly at a natural inclination to assume that he or she is trying to figure out what you have just seen. But what excludes the possibility that they are simply look-

FIGURE 3 Gaze following in a five-year-old chimpanzee in response to eyes and head condition.
ing where you are looking, without entertaining ideas about your internal attentional state? To intelligently choose between these different accounts of gaze following, we need to flesh them out a bit more clearly and determine if they generate different predictions about how apes (and human infants) might respond in more revealing circumstances.
In a first account, we envisioned that chimpanzees and other nonhuman primate species (and even human infants) understand “gaze” not as a projection of attention, but as a directional cue (i.e., a vector away from the eyes and face). Thus, perhaps the ancestors of the modern primates merely evolved an ability to use the head–eye orientation of others to
move their own visual systems along a particular trajectory. And, once their visual system encountered something novel, the operation of the primitive orienting reflex would ensure that two chimpanzees, for example, would end up attending to the same object or event, without either
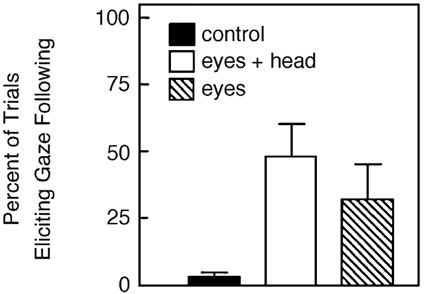
FIGURE 4 Results of the initial study of gaze following in juvenile chimpanzees.
TABLE 1 Behavioral Evidence that Humans and Chimpanzees Possess a Homologous Psychological System Controlling Gaze Following
|
Behavior |
18-Month-Old Human Infants |
Juvenile and Adult Chimpanzees |
|
Respond to whole head movement? |
yes |
yes |
|
Respond to eye movement alone? |
yes |
yes |
|
Left and right specificity? |
yes |
yes |
|
Follow gaze outside immediate visual field? |
yes |
yes |
|
Scan past distractor targets? |
yes |
yes |
|
Account for opaque barriers? |
? |
yes |
|
SOURCES: Butterworth and Cochran (1980), Butterworth and Jarrett(1991), Call et al. (1998), Povinelli and Eddy (1996a, 1996b, 1997),Povinelli et al. (1999), Tomasello et al. (1998). |
||
ape representing the other’s internal attentional state (Figure 5a). In contrast to this psychologically sparse interpretation, we considered a second account: Apes might follow gaze because they appreciate its connection to internal attentional states. Although I fear being misunderstood, it is necessary to have a shorthand label for these alternative kinds of accounts, and so I hereafter refer to them as the “low-level” and “high-level” accounts, respectively. The so-called high-level model stipulates that chimpanzees form concepts about internal mental states (such as attention) and use these concepts to help interpret the behavior of others. In contrast, the low-level model supposes that chimpanzees cogitate about behavioral propensities, not mental states.
In considering how to distinguish between these explanations of gaze following, it occurred to us that if nonhuman primates reason about the attentional aspect of gaze, this might reveal itself most clearly when they witness another animal’s gaze being obstructed by an opaque barrier such as a tree, or in the case of captivity, a wall. If the high-level account of their gaze-following abilities were correct, and if an observing animal were to witness another animal in the situation depicted in Figure 5b, the observing animal should be capable of understanding that the other ape cannot see through the obstruction. If so, the observing ape should look around the barrier to determine what the other ape had seen. In contrast, the low-level account predicted that the observing chimpanzee would project a vector away from the other ape’s face and scan along this path until something novel triggered an orienting reflex, and, if nothing novel were present, stop scanning altogether.
We tested these possibilities in our laboratory (see Figure 6a and Figure 6b; Povinelli and Eddy, 1996b). To begin, we covered half of the Plexiglas panel with an opaque partition. Thus, the chimpanzees could still enter the test lab, approach an experimenter, and request some fruit by begging through a hole in the Plexiglas. However, the partition blocked the apes’ view into a small area of the room behind the partition —an area into which only the human could see. On most trials, the subjects simply entered the lab, gestured to the human, and were handed the food. However, several experimental conditions allowed us to test the accounts described above. In one condition, as soon as the chimpanzee reached through the Plexiglas, the experimenter looked at the subject while executing a choreographed series of irrelevant movements for precisely five seconds. This condition (along with several others) allowed us to measure the subject’s ambient levels of glancing to various locations in the room (Figure 6a). In contrast, on the crucial test trials the experimenter leaned and glanced at a predetermined target on the front of the partition (see Figure 6b). Would the apes attempt to look around the partition to the experimenter ’s side, or would they follow a vector away from the

FIGURE 6 Laboratory recreation of a natural context (see Figure 5b) for investigating whether chimpanzees understand that gaze cannot pass through opaque barriers.
experimenter’s face until they wound up looking behind themselves at the back wall of the test lab? In short, would they (at some level) process the fact that the experimenter’s vision was blocked by the partition? The accounts described above generated very different predictions about the answer our apes would provide.
As can be seen in Figure 7, the chimpanzees’ reactions were unambiguous—they behaved in the manner predicted by the high-level expla-

FIGURE 7 Effect of experimenter’s movements on where chimpanzees looked on the opaque barrier test (see Figure 6).
nation. Instead of blindly turning their heads toward the back of the test unit, the apes looked around to the front of the partition (Figure 7b), exactly as if they understood that the experimenter could not see through it. Thus, despite the fact that the experimenter was never really glancing at anything, and hence the apes were never reinforced for looking around the partition, they nonetheless did so on both the first and the second trials, until gradually stopping altogether. In contrast, they almost never looked around the partition when the experimenter merely engaged in irrelevant movements (Figure 7a).
These results troubled us, but not because we were committed to low-level models of chimpanzee cognition. Rather, prior to conducting these gaze-following studies, we had conducted over a dozen experiments exploring whether our apes had a concept of “seeing”—experiments that suggested an answer very different from the high-level account of gaze following (see Povinelli, 1996; Povinelli and Eddy, 1996a; Reaux et al., 1999). In what follows, I summarize these experiments and explore whether they are truly incongruent with the results of our gaze-following studies and, indeed, whether we really need the high-level model to explain our apes’ abilities to “understand” that gaze cannot pass through opaque barriers.
Knowing Who Can See You
Our first approach to asking our apes about “seeing” was to determine if they understood the psychological distinction between someone who could see them and someone who could not. To answer this, we again capitalized on one of their most common communicative signals—the begging gesture. Chimpanzees use this gesture in a number of different communicative contexts. For example, a chimpanzee may see you walk past his or her compound with some food and immediately reach toward you with his or her palm up (to “request” the item, as it were). This seemed like an ideal, natural context in which to ask our animals whether the deployment of their gestures was mediated by a notion of “seeing.”
We began by training the apes to enter the laboratory and gesture through a hole directly in front of a single, familiar experimenter who was either standing or sitting to their left or right. On every trial that they gestured through the hole directly in front of the experimenter, this person praised them and handed them a food reward. In short order, the apes were all reliably gesturing through the correct hole toward the experimenter (Figure 8a, Figure 8b and Figure 8c).
Of course, we already knew that chimpanzees were inclined to direct their begging gesture toward us. What we really wanted to know was
how the chimpanzees would react when they entered the test lab and encountered not a single experimenter, but two experimenters, one who could see them (and therefore respond to their gestures) and one who could not. With this in mind, we created several clear cases (at least from our human point of view) of seeing versus not seeing. Although we carefully designed and rehearsed these scenarios, we did not simply pluck them out of thin air. Rather, we studied our animals’ spontaneous play and modeled our scenarios after several of their natural behaviors. At the time, one of their favorite pastimes was to use objects in their enclosure to obstruct their vision. For example, they would place large plastic buckets over their heads and then carefully move around their compound until they bumped into something. Occasionally, they would stop, lift the bucket to peek, and then continue along on their blind strolls. Although buckets were the favored means of obstructing their vision, they also used plastic bowls, burlap sacks, pieces of cardboard, and even their hands to produce the same effect. From a common-sense point of view, it seemed obvious that they knew exactly what they were doing—preventing themselves from seeing. In the end, we tested the animals using the conditions depicted in Figure 9a, Figure 9b, Figure 9c and Figure 9d in sessions of 10 trials. Eight of these trials were of the easy variety (with a single experimenter). The other two were critical probe trials of the seeing and not seeing conditions.
So how did the animals react when they encountered two familiar experimenters, for example, one blindfolded, the other not? They entered the lab, but then (measurably) paused. And yet, having apparently noted the novelty of the circumstance, they were then just as likely to gesture to the person who could not see them as to the person who could. The same effect occurred on the buckets and hands over the eyes conditions: The chimpanzees displayed no preference for gesturing toward the experimenter who could see them. In contrast, on the easy surrounding trials, the apes gestured through the hole directly in front on the single experimenter on 98 percent of the trials. Thus, despite their general interest and motivation, when it came to the seeing and not seeing conditions, the animals appeared oblivious to the psychological distinction between the two experimenters.
There was, however, one exception. Unlike the blindfolds, buckets, and hands over the eyes trials, on back versus front trials (in which one person faced toward the ape and the other faced away; see Figure 9a) the animals seemed to have the right idea: “Gesture to the person who can see.” But why the discrepancy? Why should the apes perform well from their very first trial forward on a condition in which one of the experimenters was facing them and the other was facing away, but then not on any of the other conditions? In defense of the high-level account, it could be that the back and front condition was simply the easiest situation in

FIGURE 9 Conditions used to explore chimpanzees’ understanding of “seeing” versus “not seeing”: (a) back and front, (b) buckets, (c) hands over eyes, and (d) blindfolds.
which to recognize the difference between seeing and not seeing. Despite the fact that the animals had measurably paused before making their choices in the other conditions as well, and despite the fact that we had observed them adopt these other postures in their play, the idea that back and front was just a more natural distinction seemed plausible. But there was another, more mundane potential explanation of the results. Compare Figure 8a, Figure 8b and Figure 8c with Figure 9a. Perhaps on the back and front trials the apes were merely doing what they had learned to do—enter the test lab, look for someone who happened to be facing forward, and then gesture in front of him or her. Rather than reasoning about who could see them, perhaps the apes were simply executing a procedural rule that we had inadvertently taught them during testing.
At this point there were several ways of distinguishing between these possibilities. If the high-level account were correct (that is, if the back and front condition was simply the most natural case of seeing and not seeing), then the apes ought to perform well on other, equally natural conditions. Here is a situation that our apes experience daily. One ape approaches a group of others who are facing away from her. As she gets closer, one of the other apes turns and looks at her. Now, although she notices this behavior, does she understand that this other ape is psycho-
logically connected to her in a way that the others are not? The new condition that this consideration inspired (“looking over the shoulder,” Figure 10) was of interest in its own right, but we had an even stronger motivation for testing the apes on such a condition. Recall that the low-level account could explain our apes’ excellent performance on the back and front condition by positing that they were simply being drawn to the forward-facing posture of a person. But in this new, looking over the shoulder condition, there was no forward-facing posture —just the face of one experimenter and the back of the other one’s head. Thus, the low-level account generated the seemingly implausible prediction that the apes would perform well on the back and front condition, but randomly on the looking over the shoulder trials.
To our surprise, this is exactly what the apes did. In full support of the low-level model, on the looking over the shoulder trials the apes did not prefer to gesture to the person who could see them. In direct contrast, they continued to perform without difficulty on the back and front trials. This result made a deep impression on us. No longer was it possible to dismiss the results of the other conditions by supposing that the animals thought we were peeking from under the buckets or blindfolds, or between our fingers. No, here we had made “peeking” clear and explicit, and yet the apes still performed according to the predictions of the low-level model. The experimental dissection of the front of the experimenter from the face using a posture they must witness every day (looking over

FIGURE 10 Looking over the shoulder condition.
the shoulder) sobered us to the possibility that perhaps our apes genuinely might not understand that the experimenters had to see their gesture in order to respond to it. Furthermore, the results seemed to imply that even for the back and front condition, the apes might have no idea that the experimenter facing away was “incorrect”—rather, someone facing away was simply a posture with a lower valence. They were, after all, perfectly willing to choose the person in this posture on fully half of the looking over the shoulder trials.
We had difficulty accepting the implications of these results. After all, we had witnessed our apes using their begging gesture on hundreds of occasions. Was it really possible that a gesture so instantly recognizable to us could be understood so differently by them? Although their behavior continued to be best explained by the low-level model, chimpanzees are alert, cognizing organisms, extremely attuned to the behaviors that unfold around them. So it would be truly surprising if they failed to learn anything after repeated experience on our tests. To be fair, we had intentionally kept the number of test trials in each experiment to a minimum (typically four) in order to minimize their rate of learning. After all, at this point we were interested in what they understood, not in what they could learn through trial and error.
Nonetheless, we were skeptical of our results and decided to examine their reactions to several other conditions, such as one involving screens (Figure 11). Before testing, we familiarized the subjects with these screens

FIGURE 11 Screens condition.
by playing games in which we held the screens in front of our faces, or we let the apes play with the screens themselves. Despite this, when it came to testing, the apes were just as likely to choose the person who could not see them as the person who could. However, after several additional experiments involving the screens condition, their performances began to improve, until finally they were reliably gesturing to the person who could see them. Indeed, follow-up studies revealed the interesting (although not completely unexpected) fact that our apes’ correct responding had generalized from the screens condition to the looking over the shoulder condition as well.
At this point, it was possible to walk away from these studies concluding that the apes had simply learned another procedural rule: “Gesture in front of the person whose face is visible.” However, nothing seemed to eliminate the possibility that, although they did not do so immediately, they might have finally figured out what we were asking them—“Oh! It’s about seeing!” We devised several additional procedures for distinguishing between these possibilities. First, we administered the original set of conditions to the apes (blindfolds, buckets, etc.). The high-level account predicted that, because they had finally learned that the task was about seeing, the apes would perform excellently on all of them. The low-level account also predicted excellent performance—except in the blindfolds condition (where blindfolds covered the eyes of one person versus the mouth of the other). Why the blindfolds condition? Because in this condition, an equal amount of the face of each person was visible (see Figure 9d). Although it is perfectly obvious to us that one person in this condition can see and the other cannot, if our apes had merely acquired a set of arbitrary procedural rules about the presence or absence of a face, they would be forced to guess who was correct, choosing the person whose eyes were covered as often as the person whose mouth was covered. And, to our surprise, that is precisely what they did.
Another test confronted the chimpanzees with two experimenters whose eyes and faces were both clearly visible. However, only one of them had his or her head directed toward the ape. The other appeared (to us) distracted, with her head directed above and behind the chimpanzee (see Figure 12). The high-level model predicted that the apes would gesture to the experimenter who was visually attending, whereas the low-level model, because the eyes and faces were present in both cases, predicted that they would gesture at random. The results were striking. The apes entered, looked, and then followed the distracted experimenter ’s gaze into the rear corner of the ceiling. Nevertheless, the subjects were then just as likely to gesture to the distracted person as toward the person who was looking in their direction. It was as if they processed the infor-
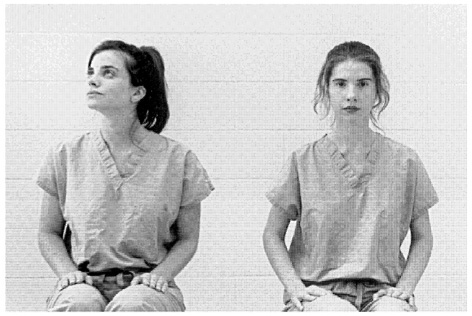
FIGURE 12 Distracted versus attending condition.
mation about the distracted experimenter’s direction of gaze, without interpreting its attentional significance.
Finally, we tested the apes on the most subtle version of this task we could imagine: eyes open versus closed. Although they initially had no preference for the person whose eyes were open, after a number of trials their performance improved. However, even here, additional control tests revealed that when the eyes and face were pitted against one another (see Figure 13), the face rule was more important! In short, through trial and error learning (probably aided by an innate sensitivity to the face, eyes, and overall posture of others) our apes appeared to have learned a hierarchically organized set of procedural rules: (1) gesture to the person whose front is facing forward, if both fronts are present (or absent); (2) gesture to the person whose face is visible, if both faces are visible (or occluded); (3) gesture to the person whose eyes are visible. “Seeing,” then, did not appear to be a concept recruited by the chimpanzees to help them decide to whom they should gesture.
Validating the Task
In reflecting on these results, we considered the possibility that we had underestimated the difficulty of our task. It was possible that our chimpanzees might understand the attentional aspect of seeing, but that
our task might require a more sophisticated understanding of visual perception than we had thought (for example, the connection between seeing and knowing). Worse yet, our task might be tapping into capacities unrelated to the question of seeing. Fortunately, findings from several laboratories had converged to suggest that an understanding of seeing as attention is beginning to be consolidated in young children by about 30 months of age (Gopnik et al., 1995; Lempers et al., 1977). Thus, we reasoned that if our tests were measuring an understanding of the attentional aspect of seeing, then 30–36-month-old children ought to perform quite well on them. On the other hand, if the tasks required an understanding of the connection between seeing and knowing (which develops at around four years of age in human children), then younger children should perform poorly. We investigated this by training two-year-, three-year-, and four-year-old children, over a three- to five- week period, to gesture to familiar adult experimenters to request brightly colored stickers (see Povinelli and Eddy, 1996b, Experiment 15). We then tested them on several of the conditions we had used with the apes (screens, hands over the eyes, and back versus front). Unlike the apes, the children were correct in most or all of the conditions from their very first trial forward—even the majority of the youngest ones that we tested.
Longitudinal Reflections
All of this may seem puzzling, especially given my above account of how the chimpanzees seemed to enjoy obstructing their own vision. Were there crucial methodological limitations in our seeing and not seeing tests that somehow prevented our apes from displaying an (existing) understanding of seeing? Although possible, consider the following example. An ape feels an irritation on its arm and scratches it. The explanation of this action seems simple: The animal produced a behavior (scratching) that is associated with a reinforcing experience (the cessation of itching). And, as much as our folk psychology resists the idea, the “peekaboo” games we observed our apes playing can be explained in a similar manner: They place a bucket over their heads because it produces an interesting, pleasurable experience. Thus, the experience of visual occlusion need not be represented any more explicitly than any other sensation (e.g., the soothing that results from scratching). Such an account could reconcile the seemingly incongruous aspects of our data: Our animals’ natural ability to produce visual deprivation in their play behavior alongside their bemusement when asked to explicitly reason about such visual deprivation in others.
But a more troubling question remained. By participating in our tests, our apes had clearly learned relationships about their gestures, on the one
hand, and the experimenters’ postures, faces, and eyes, on the other. Indeed, they had learned them so well that they could rapidly generalize this understanding to new conditions. This left us in a difficult quandary: How were we to distinguish between the following, very different explanations of these effects? One possibility was that the subjects had not initially possessed a concept of visual attention (or only a weak one), but after extensive experience with our tests they constructed the concept (or at least learned how to apply it in this particular context). But a second possibility refused to quietly go away. Perhaps the subjects neither entered nor emerged from our tests with such a concept, but rather, as a consequence of the differential reinforcement they received, they relearned the set of procedural rules described above —rules that allowed them to behave exactly as if they were reasoning about seeing.
Fortunately, at least one way of distinguishing between these possibilities presented itself 13 months after we completed the studies described above. In the context of a different protocol, we readministered several of the original conditions that our apes had learned very well (e.g., screens, buckets; see Povinelli, 1996). But despite what they had learned a year earlier, the animals responded at chance levels! Indeed, it was only after 48 massed trials of the screens condition that the animals’ performance began to creep up to levels significantly above chance.
Although a retention of performance would not have been particularly informative, their failure to do so was. Consider again the history of the children who participated in our tests. Surely they had received numerous semistructured experiences of “seeing” versus “not seeing” before coming into our lab (so many so, in fact, that their first trials were more equivalent to our apes’ final trials). However, imagine them returning to our lab a year later. The contrast between the enduring understanding that they had apparently constructed during their second year of life, and the failure of our apes to retain what they had learned a year earlier, suggests in a rather dramatic manner that despite superficial similarities in their performances, the two species might nonetheless have parted company conceptually. Indeed, this distinction is even more sobering when one considers that our apes were not idle during this intervening year. On the contrary, they participated in numerous studies that explored their understanding of attention in other ways. One reading of these results is that far from serving to assist them, at some level these other studies may actually have interfered with their performance on this task.
We were so struck by the apes’ weak retention that when they turned eight to nine years of age, we administered a final series of experiments using these same procedures (see Reaux et al., 1999). Again, however, the apes initially responded randomly. We decided to force the issue by training the subjects to perform at above-chance levels. Although not immedi-
ately, the apes did relearn how to solve many of these problems within about 16 trials. Indeed, some of our chimpanzees learned to solve them all. Did this mean that now, finally, they had constructed an understanding of seeing?
We considered two conditions that several of our apes had learned extremely well: eyes open and closed and looking over the shoulder. We realized that by mixing these conditions together—that is, by taking the correct posture from the looking over the shoulder condition and combining it with the incorrect posture from the eyes open and closed condition (see Figure 14)—we could pit the two alternatives against each other. After all, if the apes had truly constructed a concept of seeing, then they would be expected to perform excellently in this new, mixed condition. But if, instead, they had simply learned a set of hierarchical rules concerning the fronts, faces, and eyes of the experimenters, then not only would they not prefer the (correct) person looking over her shoulder, they would display a significant preference for the (incorrect) person facing forward with her eyes closed. This is because the rule-based model that had so neatly accounted for our apes’ previous behavior explicitly stipulated that the forward-facing posture was more important than whether the eyes were open or closed. The outcome was remarkable. Despite their contin-

FIGURE 14 Mixed condition combining incorrect option from eyes open versus closed and correct option from looking over the shoulder.
ued excellent performance on the eyes open and closed and looking over the shoulder conditions, when we mixed these treatments together as described above, just as the low-level model predicted, the apes gestured to the incorrect experimenter—on every trial!
Gaze Following and “Seeing:” Toward a Reconciliation
Although we were impressed by the utility of the low-level model in predicting our apes’ behavior on the seeing and not seeing tests, we pondered the model’s apparent underestimation of their ability to appreciate that gaze cannot pass through opaque partitions. One possible reconciliation was to question the generality of the seeing and not seeing tests. For example, perhaps the apes just had trouble simultaneously reasoning about the different visual perspectives of two persons, or had difficulty understanding themselves as objects of visual attention. With these ideas in mind, we explored whether they would show better evidence of understanding the attentional aspect of visual perception in situations more directly involving their gaze-following abilities (see Povinelli et al., 1999). First, we taught our apes, and three-year-old children, to search under two opaque cups for a hidden treat. Next, we occasionally kept them ignorant as to the treat’s location, but instead let them witness the experimenter gazing either at the correct cup (at target) or above the correct cup (above target) (see Figure 15a and Figure 15b). We reasoned that if the subjects understood the referential significance of the gaze of the experimenter, they ought to select the correct cup on the at-target trials, but should choose randomly between the two cups on the above-target trials. The latter prediction is the key one, because organisms with a theory of attention (for example, human children) should interpret the distracted experimenter as being psychologically (attentionally) disconnected from the cups —conveying no information about the location of the reward.
As expected, the three-year-old children selected the cup at which the experimenter was looking on the at-target trials, but chose randomly between the two cups on the above-target trials. This result provided crucial evidence that our theory of what the task was measuring was correct. In direct contrast, however, the chimpanzees did not discriminate between the at-target and above-target trials. Rather, they entered the test unit, moved to the side of the apparatus in front of the experimenter’s face, and then chose the cup closest to them. Did the apes simply not notice the direction of the experimenter’s gaze on the above-target trials, thereby confusing these with the at-target trials? Hardly. They followed the experimenter’s gaze by looking above and behind themselves on over 71 percent of the above-target trials as compared with only 16 percent of the

FIGURE 15 Conditions used to explore whether chimpanzees understand the referential aspect of gaze (a) at target and (b) above target.
at-target trials. Thus, unlike three-year-old children, our apes behaved according to the predictions of a model that assumed that despite their excellent gaze-following abilities, they do not understand how gaze is related to subjective states of attention.
How, then, do we reconcile these findings with the earlier results con-
cerning our animals’ abilities to take into account whether someone’s gaze is blocked by opaque barriers? Any such reconciliation must begin by abandoning Hume’s argument by analogy and instead explore the possibility that identical behaviors may be generated or attended by different psychological representations. For example, in the case of their apparent understanding of how another’s attention is blocked by opaque partitions, perhaps our original low-level model was too simplistic. Consider the following alternative explanation that accounts for their behavior without invoking an understanding of attention. Given that these animals possess a strong propensity to follow gaze, it seems quite plausible to suppose that this system is modulated by general learning mechanisms. Thus, with sufficient experience of following the “gaze” of others in the real world, primates may quickly learn how “gaze” interacts with objects and obstructions. In particular, they may simply learn that when they follow someone else’s gaze to an opaque barrier, the space behind the barrier is no longer relevant. We have not yet tested this model against its alternatives. Instead, I use it to illustrate that even our apes’ seemingly deep understanding of gaze on these opaque barrier tests deserves further empirical scrutiny.
BEYOND SEEING
It would be misleading to leave matters at this. After all, as surprised as we have been by what our apes appear to be telling us, if this were the sum total of our research on chimpanzees’ understanding of mental states, it might only suggest that their species conceive of visual perception in a very different manner than our own. Although this would be interesting in its own right, other aspects of our research reveal a far more complicated (and intriguing) picture. Below, I briefly summarize just a few of these other projects. Almost without exception, our animals have consistently refused to be easily subsumed under the rubric of theory of mind.
Comprehending Pointing
The ability of chimpanzees to use their gestures to “indicate” or “choose” among people or things might lead one to wonder if this does not, by itself, indicate that they understand something about the mental state of attention. Indeed, we have even occasionally glossed our apes’ actions as “pointing.” Although we have done so with caution, it seems possible that they understand the referential significance of the gesture—that is, that they understand that gestures can be used to redirect attention toward particular locations or objects. We explored this possibility by teaching our adolescent chimpanzees (across dozens of trials) to pick a

FIGURE 16 (a) With sufficient experience, chimpanzees learn to select a box to which the experimenter is pointing. (b) and (c) Alternative means of testing whether chimpanzees understand the referential significance of pointing or instead rely on the distance of the experimenter’ s hand from the correct box.
box to which their caregiver was pointing ( Figure 16a; see Povinelli et al., 1997). Having trained them to do so, we considered several alternative ways in which they might be interpreting the task. On the one hand, they might have understood the idea of reference all along, but simply have needed some experience to apply it in this context. On the other hand, they might have learned a rule such as, “Look in the box closest to the caregiver’s hand.” We pitted these ideas against each other by confronting the apes, as well as very young children, with numerous configurations of someone pointing to two boxes. Figure 16b and Figure 16c provide two of the most important contrasts. In one case, the experimenter ’s hand is equidistant from both boxes; in the other case, although the pointing gesture is referencing the correct box, the experimenter is physically closer to the incorrect box. In each of these cases (as well as several others), the apes performed according to the predictions of the distance-based model (“Choose the box closest to the experimenter’s finger”). In contrast, 26-month-old children performed excellently even in the most difficult conditions ( Figure 16a, Figure 16b, Figure 16c and Figure 17a, Figure 17b, Figure 17c ).
Distinguishing Intended from Unintended Actions
The distinction between intended and unintended (accidental or inadvertent) behavior is a core aspect of our human folk psychology and appears to be a distinction that is appealed to even in cultures very different from our own. Do chimpanzees interpret actions as being based on underlying intentions and hence distinguish between intended and unintended behavior? We investigated this by having our apes request juice or food from two strangers (see Povinelli et al., 1998). After several successful interactions with each person, we staged one of two events, each associated with one of the strangers. One of them accidentally spilled the juice intended for the ape, whereas the other slowly and deliberately poured the juice onto the floor, or, in another experiment, ate the food themselves. We wanted to know if, despite the fact that they failed to receive food from both of the strangers, the animals would distinguish between the two categories of action. When it came to the critical trials in which they had to choose one of the two strangers to give them more juice, the chimpanzees did not avoid the one who had intentionally wasted the juice. Young children, in contrast, begin to make such distinctions by about three years of age (e.g., Shultz et al., 1980; Yuill, 1984).
Cooperating with Others
Partially as a result of Crawford’s early work in Robert Yerkes’ laboratory, chimpanzees gained a reputation as being able to intentionally
cooperate with each other. Crawford (1941) trained two apes to pull separate ropes to retrieve a box that was too heavy to manage alone. Although it is often overlooked, just to achieve this result required a large amount of training. Slowly, however, the animals began to attend to each other, and after about 50 sessions or so they began watching each other, eventually even exhibiting limited instances of touching each other when one wandered “off task.” But was this evidence that they understood the intentional state of the partner or the mere conventionalization of behavior? Recently, O’Neill and I explored these possibilities. After training two “expert” apes to cooperate on a rope-pulling task, we then paired them (on several occasions), one by one, with familiar peers who were ignorant about the nature of the task (Povinelli and O’Neill, 2000). The results, per-

FIGURE 17 Two-year-old child responds to experimenter’s pointing gesture correctly in (a) and (b) easy and (c) difficult conditions.
haps more so than any others we have obtained, were truly captivating. There was not a single instance in which one of the expert apes attempted to direct the attention of the ignorant animals to the relevant features of the task. The expert apes were not oblivious to the need to have the other animal near the ropes, as they looked at the naive animal at levels far exceeding the frequency with which the naive animal looked at them. Indeed, there were a number of noteworthy occasions in which the naive partner approached and inspected the ropes, followed by the expert partner rushing forward to pull the rope he or she had typically used, while at the same time ignoring the other apes ’ actions. The expert partners appeared to realize that another animal needed to be near the ropes, but not that the other ape possessed his or her own subjective understanding of the situation.
REINTERPRETING BEHAVIOR
The traditional reaction to the remarkable behavioral similarity between humans and apes has been to conclude the presence of comparable psychological similarity. This was the solution counseled by Hume, and later adopted by Darwin and Romanes. In the remainder of this chapter, I sketch a different view, one more consistent with the mosaic of similarity and difference between humans and chimpanzees revealed by our research. But to appreciate this alternative view, we must reconsider the argument by analogy, and in particular its key assumption that introspection can successfully penetrate through the multiple levels of representation of the world formed by our neural circuitry, and retrieve the true causal connections between our thoughts and our actions.
Imagine our planet long before humans or the great apes had evolved. Imagine millions of generations of social primates evolving ever-more sophisticated brains to instantiate ever-more complex rules of social interactions, rules demanded by the complexities of group living. But, for the sake of argument, suppose that these rules did not appeal to mental states. Clever brains, in Humphrey’s (1980) turn of phrase, but blank minds. Finally, suppose that one lineage, the human one, evolved a cognitive specialization linked to social understanding. Perhaps it evolved as a separate, domain-general faculty (in which case we might expect even more far-reaching differences between humans and chimpanzees than those discussed here); perhaps it evolved as a more specific system for social computations; finally, perhaps it evolved in conjunction with selection for a specialized language capacity. But however it arose, it seems likely that it did not replace the ancestral psychology of the social primates, but rather was woven into it. Like a tapestry into which new colors were added, new systems or subsystems may have been created while conserv-
ing an ancestral repertoire of social behaviors. Just as the morphological structures and systems of modern organisms reflect the baggage of developmental constraints laid down in the Cambrian, so too do psychological structures carry the ancient alongside the new.
This view essentially defeats Hume’s hope that the spontaneous behavior of animals could serve as portals into their minds. Hume’s error was twofold. First, he incorrectly assumed that an accurate introspective assessment of the mental states that accompany our own behaviors could yield an accurate inference as to the psychological cause of those behaviors. Furthermore, he compounded this error by advocating that we use the results of this introspective diagnosis to infer the mental states in other species by analogy. It is true that humans form internal representations of mental states such as desires, knowledge, and beliefs, and that these representations provide us with a useful means of anticipating what others will do. However, the basic blueprint of the behaviors with which we can respond may have evolved long before those higher-level representations were possible. Thus, the differences between humans and other species may stem first and foremost from underlying cognitive—not behavioral —specializations.
It may seem that I am advocating a form of dualism (such as epiphenomenalism) in which the representation of mental states accompany but play no causative role in behavior. On the contrary, I assume that humans evolved the capacity to represent the mental states of self and other because of their useful, causal connection to behavior. But until now we may have been thinking in the wrong way about this connection. As we have seen in the case of gaze following, two species may share a very similar behavior, controlled by similar low-level mechanisms, but interpret that behavior in very different ways. Does this mean that the human trait of representing the attentional aspect of gaze is superfluous? No, because although it may not be directly linked to the act of following gaze, the representation of attention (and other mental states) may play a profound role at a higher level of behavioral organization. Once humans evolved the capacity to represent other organisms not just as behaving beings, but as beings who attend, want, and think, it became necessary to use already-existing behaviors to respond to the world of the mind, as well as the world of behavior. This distinction may be especially important in those psychological arenas in which we seem to differ most from other species such as culture, pedagogy, and ethics. Determining the correct causal connections between individual behavioral elements and the mental states that ultimately control their expression may simply be a project beyond the reach of introspection alone (for a detailed critique of the argument by analogy, see Povinelli and Giambrone, in press).
More recently, we have begun to explore the differences that might
exist between humans and chimpanzees in other psychological realms, such as understanding the physical world (see Povinelli, 2000). The results of this research suggest that here, too, chimpanzees are alert, cognizing beings—creatures that think and learn about the regularities of the interactions of physical objects in the world. And yet at the same time they appear to understand those interactions in ways that are very different from us. Unlike two-year- or three-year-old human children, chimpanzees do not appear to understand the causal structure that links one set of events to another. Just as they appear not to reason about unobservable mental states such as desires, perceptions, and beliefs, this new research suggests that they do not reason about unobservable physical causes such as gravity, force, or physical connection. Collectively, our work suggests the broader conclusion that chimpanzees may simply not have the ability to form ideas about phenomena that are so abstract that they are, in principle, unobservable. This may set direct limits on their ability to reason about virtually every aspect of the world around them.
My view, then, is that there may be profound psychological differences between humans and chimpanzees. Some will see this as a distinctly negative outlook, one that denies apes their rightful psychological standing alongside humans. But in the final analysis, evolution is about differences as well as similarities—indeed, were there no differences among species, there would have been no reason to postulate an evolutionary process in the first place. On this view, what could be a more constructive, more exciting project than to continue to explore the ways in which humans and chimpanzees have accommodated both the psychological structures of their mammalian ancestry and the innovations they continue to accumulate along their separate evolutionary journeys?
ACKNOWLEDGMENTS
This research was supported in part by National Science Foundation Young Investigator Award SBR-8458111. I thank James E. Reaux, Anthony Rideaux, and Donna Bierschwale for professional support in the testing of chimpanzees. I also thank Todd Preuss and Steve Giambrone for their generous effort in reviewing and editing the manuscript. Original artwork is by Donna Bierschwale. Photographs are by Donna Bierschwale and Corey Porché.
REFERENCES
Avis, J., and P. L. Harris. 1991. Belief-desire reasoning among Baka children: Evidence for a universal conception of mind. Child Development 62:460–467.
Baron-Cohen, S. 1994. How to build a baby that can read minds: Cognitive mechanisms in mindreading. Current Psychology of Cognition 13:513–552.
Blest, A. D. 1957. The function of eyespot patterns in the Lepidoptera. Behaviour 11:209–255.
Burger, J., M. Gochfeld, and B. G. Murray, Jr. 1991. Role of a predator’s eye size in risk perception by basking black iguana, Ctenosaura similis. Animal Behaviour 42:471–476.
Burghardt, G. M., and H. W. Greene. 1988. Predator simulation and duration of death feigning in neonate hognose snakes. Animal Behaviour 36:1842–1844.
Butterworth, G., and E. Cochran. 1980. Towards a mechanism of joint visual attention in human infancy. International Journal of Behavioral Development 3:253–272.
Butterworth, G., and N. Jarrett. 1991. What minds have in common is space: Spatial mechanisms serving joint visual attention in infancy. British Journal of Developmental Psychology 9:55–72.
Byrne, R. W., and A. Whiten. 1985. Tactical deception of familiar individuals in baboons (Papio ursinus). Animal Behaviour 33:669–673.
Call, J., B. A. Hare, and M. Tomasello. 1998. Chimpanzee gaze following in an object-choice task. Animal Cognition 1:89–99.
Corkum, V., and C. Moore. 1994. Development of joint visual attention in infants. Pps. 61–83. in C. Moore and P. Dunham, eds. Joint Attention: Its Origins and Role in Development. Hillsdale, N.J.: Lawrence Erlbaum.
Crawford, M. P. 1941. The cooperative solving by chimpanzees of problems requiring serial responses to color cues. Journal of Social Psychology 13:259–280.
Darwin, C. 1871. The Descent of Man. Reprinted. New York: Modern Library, 1982.
de Waal, F. B. M. 1989. Peacemaking Among Primates. Cambridge, Mass.: Harvard University Press.
Emery, N. J., E. N. Lorincz, D. I. Perrett, M. W. Oram, and C. I. Baker. 1997. Gaze following and joint attention in rhesus monkeys (Macaca mulatto). Journal of Comparative Psychology 111:286–293.
Flavell, J. H., S. G. Shipstead, and K. Croft. 1978. What young children think you see when their eyes are closed. Cognition 8:369–387.
Flavell, J. H., E. R. Flavell, F. L. Green, and S. A. Wilcox. 1980. Young children’s knowledge about visual perception: Effect of observer’s distance from target on perceptual clarity of target. Developmental Psychology 16:10–12.
Flavell, J. H., B. A. Everett, K. Croft, and E. R. Flavell. 1981. Young children’s knowledge about visual perception: Further evidence for the level 1–level 2 distinction. Developmental Psychology 17:99–103.
Gallup, G. G., Jr., R. F. Nash, and A. L. Ellison, Jr. 1971. Tonic immobility as a reaction to predation: Artificial eyes as a fear stimulus for chickens. Psychonomic Science 23:79–80.
Gómez, J. C. 1990. The emergence of intentional communication as a problem-solving strategy in the gorilla. Pp. 333–355 in S. T. Parker and K. R. Gibson, eds. Language and Intelligence in Monkeys and Apes: Comparative Developmental Perspectives. Cambridge, U.K.: Cambridge University Press.
Goodall, J. 1986. The Chimpanzees of Gombe: Patterns of Behavior. Cambridge, Mass.: Belknap, Harvard University Press.
Gopnik, A., and P. Graf. 1988. Knowing how you know: Young children’s ability to identify and remember the sources of their beliefs. Child Development 59:1366–1371.
Gopnik, A., A. N. Meltzoff, and J. Esterly. 1995. Young Children’s Understanding of Visual Perspective-Taking. Poster presented at the First Annual Theory of Mind Conference, Eugene, Oregon.
Hains, S. M. J., and D. W. Muir. 1996. Effects of stimulus contingency in infant–adult interactions. Infant Behavior and Development 19:49–61.
Hume, D. 1739–1740/ 1911. A Treatise of Human Nature, 2 vols. London: Dent.
Humphrey, N. K. 1980. Nature’s psychologists. Pp. 57–75 in B. D. Josephson and V. S. Ramachandran, eds. Consciousness and the Physical World. New York: Pergamon Press.
Karmiloff-Smith, A. 1992. Beyond Modularity: A Developmental Perspective on Cognitive Science. Cambridge, Mass.: MIT Press.
Köhler, W. 1927. The Mentality of Apes, 2nd ed. New York: Vintage Books.
Lempers, J. D., E. R. Flavell, and J. H. Flavell. 1977. The development in very young children of tacit knowledge concerning visual perception. Genetic Psychology Monographs 95:3–53.
Lillard, A. S. 1998a. Ethnopsychologies: Cultural variations in theory of mind. Psychological Bulletin 123:3–32.
Lillard, A. S. 1998b. Ethnopsychologies: Reply to Wellman (1998) and Gauvain (1998). Psychological Bulletin 123:43–46.
Masangkay, Z. S., K. A. McKluskey, C. W. McIntyre, J. Sims-Knight, B.E. Vaughn, and J. H. Flavell. 1974. The early development of inferences about the visual precepts of others. Child Development 45:357–366.
Moore, C. P. 1994. Intentionality and self–other equivalence in early mindreading: The eyes do not have it. Current Psychology of Cognition 13:661–668.
Moore, C., M. Angelopoulos, and P. Bennett. 1997. The role of movement and the development of joint visual attention. Infant Behavior and Development 20:83–92.
O’Neill, D. K., and A. Gopnik. 1991. Young children’s ability to identify the sources of their beliefs. Developmental Psychology 27:390–397.
O’Neill, D. K., J. W. Astington, and J. H. Flavell. 1992. Young children’s understanding of the role that sensory experiences play in knowledge acquisition. Child Development 63:474–490.
Perner, J., and J. Ogden. 1988. Knowledge for hunger: Children’s problems with representation in imputing mental states. Cognition 29:47–61.
Perrett, D., M. Harries, A. Mistlin, J. Hietanen, P. Benson, R. Bevan, S. Thomas, M. Oram, J. Ortega, and K. Brierly. 1990. Social signals analyzed at the single cell level: Someone is looking at me, something touched me, something moved! International Journal of Comparative Psychology 4:25–55.
Povinelli, D. J. 1991. Social Intelligence in Monkeys and Apes. Ph.D. dissertation, Yale University, New Haven, Conn.
Povinelli, D. J. 1996. Growing up ape. Monographs of the Society for Research in Child Development 61(2, Serial No. 247):174–189.
Povinelli, D. J. 1999. Social understanding in chimpanzees: New evidence from a longitudinal approach. Pp. 195–225 in P. Zelazo, J. Astington, and D. Olson, eds. Developing Theories of Intention: Social Understanding and Self-Control . Hillsdale, N.J.: Erlbaum.
Povinelli, D. J. 2000. Folk physics for apes: The chimpanzee’s theory of how the world works. Oxford: Oxford University Press.
Povinelli, D. J., and S. deBlois. 1992a. On (not) attributing mental states to monkeys: First, know thyself. Behavioral and Brain Sciences 15:164–166.
Povinelli, D. J., and S. deBlois. 1992b. Young children’s (Homo sapiens) understanding of knowledge formation in themselves and others. Journal of Comparative Psychology 106:228–238.
Povinelli, D. J., and T. J. Eddy. 1994. The eyes as a window: What young chimpanzees see on the other side. Current Psychology of Cognition 13:695–705.
Povinelli, D. J., and T. J. Eddy. 1996a. What young chimpanzees know about seeing. Monographs of the Society for Research in Child Development 61(3, Serial No. 247):v–152.
Povinelli, D. J., and T. J. Eddy, 1996b. Chimpanzees: Joint visual attention. Psychological Science 7:129-135.
Povinelli, D. J., and T. J. Eddy. 1996c. Factors influencing young chimpanzees’ (Pan troglodytes) recognition of attention. Journal of Comparative Psychology 110:336–345.
Povinelli, D. J., and T. J. Eddy. 1997. Specificity of gaze-following in young chimpanzees. British Journal of Developmental Psychology 15:213–222.
Povinelli, D. J., and S. Giambrone. 2000. Inferring other minds: Failure of the argument by analogy. Philosophical Topics, 27:161–201.
Povinelli, D. J., and L. R. Godfrey. 1993. The chimpanzee’s mind: How noble in reason? How absent of ethics? Pp. 227–324 in M. Nitecki and D. Nitecki, eds. Evolutionary Ethics. Albany, N.Y.: SUNY Press.
Povinelli, D. J., and D. K. O’Neill. 2000. Do chimpanzees use their gestures to instruct each other? In S. Baron-Cohen, H. Tager-Flusberg, and D. J. Cohen, eds. Understanding other Minds: Perspectives from Autism, 2nd ed. Oxford, U.K.: Oxford University Press.
Povinelli, D. J., A. B. Rulf, K. R. Landau, and D. T. Bierschwale. 1993. Self-recognition in chimpanzees (Pan troglodytes): Distribution, ontogeny, and patterns of emergence. Journal of Comparative Psychology 107:347–372.
Povinelli, D. J., A. B. Rulf, and D. Bierschwale. 1994. Absence of knowledge attribution and self-recognition in young chimpanzees (Pan troglodytes). Journal of Comparative Psychology 180:74–80.
Povinelli, D. J., J. E. Reaux, D. T. Bierschwale, A. D. Allain, and B. B. Simon. 1997. Exploitation of pointing as a referential gesture in young children, but not adolescent chimpanzees. Cognitive Development 12:423–461.
Povinelli, D. J., H. K. Perilloux, J. E. Reaux, and D. T. Bierschwale. 1998. Young and juvenile chimpanzees’ (Pan troglodytes) reactions to intentional versus accidental and inadvertent actions. Behavioral Processes 42:205–218.
Povinelli, D. J., D. T. Bierschwale, and C. G. Cech. 1999. Comprehension of seeing as a referential act in young children, but not juvenile chimpanzees. British Journal of Developmental Psychology 17:37–60.
Premack, D., and G. Woodruff. 1978. Does the chimpanzee have a theory of mind? Behavioral and Brain Sciences 1:515–526.
Reaux, J. E., L. A. Theall, and D. J. Povinelli. 1999. A longitudinal investigation of chimpanzees’ understanding of visual perception. Child Development 70:275–290.
Redican, W. K. 1975. Facial expressions in nonhuman primates. Pp. 103–194 in L. A. Rosenblum, ed., Primate Behavior, Vol. 4. New York: Academic Press.
Ristau, C. A. 1991. Before mindreading: Attention, purposes and deception in birds? Pp. 209–222 in A. Whiten, ed. Natural Theories of Mind: Evolution, Development and Simulation of Everyday Mindreading. Cambridge, U.K.: Basil Blackwell.
Romanes, G. J. 1882. Animal Intelligence. London: Keagan Paul.
Ruffman, T. K., and D. R. Olson. 1989. Children’s ascriptions of knowledge to others. Developmental Psychology 25:601–606.
Scaife, M., and J. Bruner. 1975. The capacity for joint visual attention in the infant. Nature 253:265–266.
Schaller, G. B. 1963. The Mountain Gorilla: Ecology and Behavior. Chicago: University of Chicago Press.
Shultz, T. R., D. Wells, and M. Sarda. 1980. Development of the ability to distinguish intended actions from mistakes, reflexes, and passive movements. British Journal of Social and Clinical Psychology 19:301–310.
Symons, L. A., S. M. J. Hains, and D. W. Muir. 1998. Look at me: Five-month-old infants’ sensitivity to very small deviations in eye-gaze during social interactions. Infant Behavior and Development 21:531–536.
Theall, L. A., and D. J. Povinelli. 1999. Do chimpanzees tailor their attention-getting behaviors to fit the attentional states of others? Animal Cognition 2:207–214.
Tomasello, M. 1995. The power of culture: Evidence from apes. Human Development 38:46–52.
Tomasello, M, J. Call, and B. Hare. 1998. Five primate species follow the visual gaze of conspecifics. Animal Behaviour 55:1063–1069.
Vinden, P. G., and J. W. Astington. 2000. Culture and understanding other minds. Pp. 503–519 in S. Baron-Cohen, H. Tager-Flusberg, and D. Cohen, eds. Understanding other Minds: Perspectives from Autism and Cognitive Neuroscience. Oxford, U.K.: Oxford University Press.
Wimmer, H., G. J. Hogrefe, and J. Perner. 1988. Children’s understanding of informational access as a source of knowledge. Child Development 59:386–396.
Wolff, P. H. 1963. Observations on the early development of smiling. Pp. 113–138 in B. M. Foss, ed. Determinants of Infant Behavior II. London: Methuen & Co.
Yuill, N. 1984. Young children’s coordination of motive and outcome in judgements of satisfaction and morality. British Journal of Developmental Psychology 2:73–81.



