Carving Our Destiny: Scientific Research Faces a New Millennium (2001)
Chapter: The Cognitive Neuroscienceof Numeracy: Exploring the Cerebral Substrate, the Development, and the Pathologies of Number Sense
2
The Cognitive Neuroscience of Numeracy: Exploring the Cerebral Substrate, the Development, and the Pathologies of Number Sense
Stanislas Dehaene
INTRODUCTION
A basic unsolved problem in human cognition concerns how the brain computes meaning. As you read this text, what are the processes unfolding in your mind and brain that allow you to understand the message that I intend to communicate to you? What cerebral events enable you to recognize that the words “Einstein,” “Eighteen,” and “Elephant,” in spite of their visual similarity, point to radically different meanings, which we can access in a fraction of a second? Recent studies in neuropsychology, brain imaging, and infant development have begun to provide a new perspective on this age-old problem. It is now well accepted that lesions to some specific areas of the human brain may cause highly selective semantic deficits (Caramazza, 1996; Hillis and Caramazza, 1995; McCarthy and Warrington, 1988, 1990). For instance, a patient may lose his or her knowledge of animals, but remains able to accurately define inanimate objects, food and vegetables, colors, numbers, or actions (Caramazza and Shelton, 1998). Brain-imaging studies have confirmed that processing of different categories of words such as proper names, animals, or tools is associated with distinct patterns of brain activation that may be similarly localized in different individuals (Damasio et al., 1996; Martin et al., 1995, 1996). Hence, specific cortical areas appear to participate in the representation of different categories of meaning.
Service Hospitalier Frédéric Joliot, INSERM, Orsay, France
In parallel, studies of human infants have revealed that they possess a much richer knowledge base than was initially suspected (Mehler and Dupoux, 1993; Pinker, 1995, 1997). Far from being a blank page, the human mind starts in life with a rich knowledge about objects, colors, numbers, faces, and language. This evidence supports the hypothesis that humans have been endowed by evolution with biologically determined predispositions to represent and acquire knowledge of specific domains (Barkow et al., 1992). Thus, if all languages distinguish between nouns and verbs, or between words for locations, colors, numbers, objects, animals, and persons, it may well be because the architecture of our brain is biologically constrained to focus learning on these domains, using partly dedicated cortical territories (Caramazza and Shelton, 1998).
If this framework has any validity, charting meaning in the human brain requires several steps: first, understanding the basic features specific to a given domain of knowledge; second, discovering whether and how some of these features have been extracted in the course of cerebral evolution and have been internalized in the brains of infants and animals; and third, identifying the neural circuits that underlie them and their internal organization. My research aims at fulfilling these goals in the specific case of arithmetic knowledge and at using this specific domain as a springboard for broader studies of the representation, development, and pathologies of semantic knowledge in the human brain.
THE COGNITIVE NEUROSCIENCE OF NUMERACY: PAST AND PRESENT EVIDENCE
A Biological Basis for Elementary Arithmetic?
In my research of the past decade, I have examined how a special category of words (numbers) are represented in the animal, infant, and adult brain. There are several reasons why numbers are excellent tools for psychological studies of word processing. First, number is a very circumscribed domain. There is only a small list of number words. Their lexicon, syntax, and semantics are clearly defined and have been formalized by mathematicians such as Peano or Russell. As David Marr (1982) remarked several years ago, understanding of any cognitive function requires an initial characterization of its computational demands. It is much easier to characterize what numbers mean than to understand, say, the semantics of verbs or of animal names.
A second advantage of studying numbers is that, for all their simplicity, they constitute one of the most important domains of human cognition. The number system is an indispensable tool for scientific activity. Arithmetic and mathematics in general are a unique achievement of our
species, without which our technology-oriented society could never have developed. Philosophers have been pondering for thousands of years about the relations between mathematics and the human mind and brain. It is very exciting to think that, with the present experimental tools in cognitive neuroscience, we can begin to address this age-old question. It is also clear that this is not just a theoretical issue, but also one with considerable practical import. The loss of calculation and number processing abilities in brain-lesioned patients is a frequent and highly incapacitating deficit. So are developmental dyscalculia and mathematical disabilities in school children. The low level of mathematical competence, or downright innumeracy, particularly in Western countries relative to Asian countries, is a growing concern. By better understanding how mathematics is acquired and represented mentally and cerebrally, we may hope to improve education and rehabilitation methods in arithmetic.
A third advantage of studying the number domain is that there is already a rich database of experiments. I have reviewed many of these experiments in my recent book The Number Sense (Dehaene, 1997). In my opinion, they make the number domain one of the best documented instances of a biologically determined, category-specific domain of knowledge. (In that respect, number knowledge stands on a par with face processing, for which there is also considerable animal, infant, and adult lesion, brain-imaging, and even single-cell recording evidence for a biologically determined ability.) Strong evidence exists that animals and infants possess rudimentary arithmetic knowledge with a clear analogy and continuity with adult knowledge; that specific brain lesions can impair this number sense; and that a specific network of brain areas is active during number processing. This evidence provides support for my hypothesis that specific cerebral networks of the brain, through the course of a long evolutionary history, have internalized the numerical regularities of the world we live in, and it is this internalized representation of numerical quantities that enables us to understand number meaning. In this chapter, I review in turn the infant, animal, and adult behavioral and neurological evidence related to number processing. In each case, I outline what my own contributions to the domain are. Then I turn to the research projects I intend to pursue in the future.
Number Sense in Young Infants
There is now considerable evidence that infants possess some rudimentary knowledge of numbers in the first year of life, independently of language (Antell and Keating, 1983; Bijeljac-Babic et al., 1991; Koechlin et al., 1997; Moore et al., 1987; Simon et al., 1995; Sophian and Adams, 1987; Starkey and Cooper, 1980; Starkey et al., 1983, 1990; Strauss and Curtis,
1981; van Loosbroek and Smitsman, 1990; Wynn, 1992a, 1996; Xu and Carey, 1996). Discrimination of visual numerosity was first demonstrated in six-month- to seven-month-old infants using the classical method of habituation recovery of looking time (Starkey and Cooper, 1980). Following habituation to slides with a fixed number of dots, infants looked longer at slides with a novel number of objects. Discrimination was found for two versus three, and occasionally three versus four, but not four versus six. Recently, however, Xu and Spelke (2000) have established discrimination of 8 versus 16 dots (but not 8 versus 12), thus confirming that infants are not limited to small numbers.
In Starkey and Cooper’s (1980) original experiment, dot density, spacing, and alignment were controlled for. In subsequent studies, the effect was replicated in newborns (Antell and Keating, 1983) and with various stimulus sets, including slides depicting sets of realistic objects of variable size, shape, and spatial layout (Strauss and Curtis, 1981), as well as dynamic computer displays of random geometrical shapes in motion with partial occlusion (van Loosbroek and Smitsman, 1990). Recently, the results have also been extended to the discrimination of visual events, such as a puppet making two or three jumps (Wynn, 1996). Using non-nutritive sucking techniques, it has also been established that four-day-old newborns distinguish two-syllable words from three-syllable words, even when phonemic content, duration, and speech rate are tightly controlled (Bijeljac-Babic et al., 1991). Thus, these results indicate that infants can perceive numerosity in various sensory modalities, independently of other more peripheral physical parameters. There are even experiments suggesting cross-modal numerosity matching in six-month- to eight-month-old infants (Starkey et al., 1983, 1990), although their replicability is disputed (Moore et al., 1987).
Infants use their numerosity perception abilities to predict and make sense of how objects behave in the external world. Wynn (1992a, 1995) presented five-month-old infants with real-life equivalents of the operations 1 + 1 = 2 and 2 − 1 = 1 (see Figure 1). To exemplify the 1 + 1 =2 operation, for instance, five-month-old infants were shown a toy being hidden behind a screen and then a second toy also being brought behind the same screen. To assess whether infants had developed the numerically appropriate expectation of two objects, their looking times were measured as the screen dropped and revealed either one, two, or three objects (objects being surreptitiously added or removed as needed). Infants looked systematically longer at the impossible outcomes of one or three objects than at the expected outcome of two objects. This suggests that they had internally computed an expectation of the outcome of two objects, although the two objects had never been presented together earlier.
In Wynn’s original experiments, there were reasons to be skeptical about whether infants were really attending to number. The surprise reaction could also have been based on non-numerical parameters such as amount of stuff, object identity, or location. My own work with infants was directed at refuting such non-numerical interpretations (Koechlin et al., 1997). My colleagues and I replicated Wynn’s experiments with two groups of infants. One group was tested in exactly the same conditions as Wynn’s. In the other group, objects were placed on a rotating tray so that their location behind the screen was unpredictable. In both cases, infants still reacted to the numerically impossible events 1 + 1 = 1 and 2 − 1 = 2, thus indicating that they attended to a more abstract parameter than object location. A parallel experiment by Simon et al. (1995) showed that the identity of the objects could be surreptitiously modified on some trial, without altering the infants ’ surprise reaction. Together, these experiments suggest that infants encode the scenes they see using an abstract, implicit or explicit representation of the number of objects on the scene, irrespective of their exact identity and location.
Xu and Carey (1996) have examined which cues infants use to infer the presence of one or two objects. When infants under one year of age were presented with two highly discriminable objects alternatively popping out from the same screen, they did not expect to see two objects when the screen drops. Differences in object identity, by themselves, were not used by infants as a valid cue to number. Only if the trajectory followed by the objects was physically incompatible with it being a single object (as, for instance, if one object pops out from one screen on the right while the other object pops out from another spatially separate screen on the left) did preverbal infants infer the presence of two objects. This evidence is compatible with the hypothesis that numerosity is processed by a dedicated, informationally “encapsulated” module (Fodor, 1983) that takes as input information about object location and trajectory, but that is blind to object identity. Although the cerebral substrates of infants’ numerical abilities are unknown, one may speculate that occipito-parietal pathways for spatial visual processing [the “where ” system (Ungerleider and Mishkin, 1982)] may play a crucial role in numerosity extraction, thus explaining the special role of bilateral inferior parietal cortices in number processing in human adults (see below).
Number Sense in Animals
If neural networks of the infant brain are genetically biased toward elementary numerosity perception and arithmetic, one should find precursors of this ability in animals. Indeed, considerable evidence indicates that animals also possess numerosity discrimination, cross-modal numer-
osity perception, and elementary arithmetic abilities comparable to those of human infants (Boysen and Capaldi, 1993; Davis and P érusse, 1988; Dehaene, 1997; Gallistel, 1989, 1990; Gallistel and Gelman, 1992). Like human infants, various animal species including rats, pigeons, raccoons, dolphins, parrots, monkeys, and chimpanzees have been shown to discriminate the numerosity of various sets, including simultaneously or sequentially presented visual objects as well as auditory sequences of sounds (Capaldi and Miller, 1988; Church and Meck, 1984; Mechner, 1958; Mechner and Guevrekian, 1962; Meck and Church, 1983; Mitchell et al. 1985; Pepperberg, 1987; Platt and Johnson, 1971; Rilling and McDiarmid, 1965). Cross-modal extraction of numerosity was observed, for instance, in rats (Church and Meck, 1984). Most such experiments included controls for non-numerical variables such as spacing, size, tempo, and duration of the stimuli (Church and Meck, 1984; Mechner and Guevrekian, 1962; Meck and Church, 1983).
Like human infants, animals also exhibit some abilities for elementary mental arithmetic. They can apply to their internal representations of number simple operations of approximate addition, subtraction and comparison (see Figure 2) (Boysen and Capaldi, 1993; Hauser et al., 1996; Rumbaugh et al., 1987; Woodruff and Premack, 1981). Wynn’s 1 + 1 = 2 and 2 − 1 = 1 experiments with infants, for instance, have been replicated using a very similar violation-of-expectation paradigm with untrained monkeys tested in the wild (Hauser et al., 1996). In some experiments that do require training, there is excellent evidence that animals can generalize beyond the training range. A case in point is the remarkable recent experiment by Brannon and Terrace (1998, 2000). Monkeys were initially trained to press cards on a tactile screen in correct numerical order: first the card bearing one object, then the card bearing two, and so on up to four. Following this training, the monkeys were transferred to a novel block with numerosities five to nine. Although no differential reinforcement was provided, the monkeys readily generalized their smaller-to-larger ordering behavior to this new range of numbers. Such evidence suggests that a genuine understanding of numerosities and their relations can be found in monkeys.
There is also evidence that monkeys (Washburn and Rumbaugh, 1991), chimpanzees (Boysen and Berntson, 1996; Boysen and Capaldi, 1993; Matsuzawa, 1985), and even dolphins (Mitchell et al., 1985) and parrots (Pepperberg, 1987) can learn the use of abstract numerical symbols to refer to numerical quantities. Such symbolic number processing, however, requires years of training and is never found in the wild. Hence, it cannot be taken to indicate that exact symbolic or “linguistic ” number processing is within animals’ normal behavioral repertoire. It does indicate, however, that abstract, presumably nonsymbolic representations of number

FIGURE 2 A chimpanzee spontaneously selects the tray with the largest amount of food, suggesting the availability of approximate addition and comparison operations (reprinted from Rumbaugh et al., 1987).
are available to animals and can, under exceptional circumstances, be mapped onto arbitrary behaviors that can then serve as numerical “symbols.”
Changeux and I have developed a neural network model that reproduces the known features of animal numerical behavior and provides specific hypotheses about its neural basis (Dehaene and Changeux, 1993).
The network’s input consists in a “retina” on which from one to five objects of various sizes can be presented. A first map encodes the locations of objects irrespective of their size and identity, an operation possibly performed by the occipito-parietal pathway. Following this “normalization” operation, each object is represented by an approximately equal number of active neurons, and hence their activity can be summed to yield an estimate of number. Number is therefore represented by the activity of an array of numerosity detector neurons, each responsive to a specific range of input numerosities. Interestingly, number-sensitive neurons similar to those predicted by our model have been recorded by Thompson et al. (1970) in the associative cortex of cats.
The Phylogenetic Continuity of Number Sense
To demonstrate that human abilities for arithmetic have a biological basis with a long evolutionary history, it is not sufficient to demonstrate that animals possess rudimentary number processing abilities. One must also show that this is not a case of parallel evolution, and that there are deep relations between human and animal abilities that suggest a phylogenetic continuity. Indeed, a considerable part of my chronometric work with human adults has been dedicated to documenting two characteristics of number processing that are shared by humans and animals: the distance effect and the size effect (see Figure 3). The distance effect is a systematic, monotonous decrease in numerosity discrimination performance as the numerical distance between the numbers decreases. The size effect indicates that for equal numerical distance, performance also decreases with increasing number size. Both effects indicate that the discrimination of numerosity, like that of many other physical parameters, obeys Fechner’s law.
It is perhaps unsurprising that humans, like other animals, suffer from distance and size effects when processing the numerosity of sets of objects (Buckley and Gillman, 1974; van Oeffelen and Vos, 1982). More interestingly, these effects still hold when human subjects process symbolic stimuli such as Arabic numerals. The time and error rate for humans to compare two numerals increases smoothly, both with number size and with the inverse of the numerical distance between the two numbers. Thus, it is harder to decide that 1 is smaller than 2 than to reach the same decision with 1 and 9; and it is harder still with 8 and 9 (Buckley and Gillman, 1974; Moyer and Landauer, 1967). My own work has helped specify the mathematical equations for comparison time (Dehaene, 1989). I have also explored the interesting case of two-digit number comparison (Dehaene et al., 1990; Hinrichs et al., 1981). One might suppose that, in order to compare 2 two-digit numbers, subjects first compare the leftmost
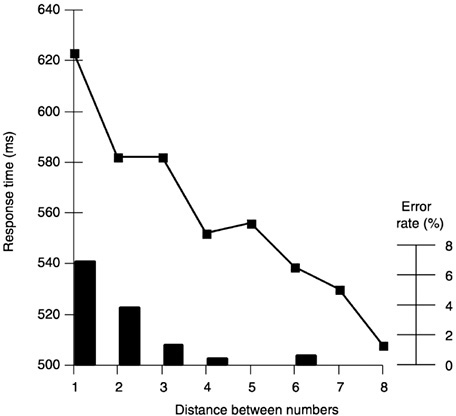
FIGURE 3 The distance effect. The time needed to decide which of two digits is larger, as well as the error rate, decreases as the distance between the two numerals gets larger (redrawn from data in Moyer and Landauer, 1967).
digits and then, if necessary, the rightmost digits. Experiments show, however, that comparison time is still a continuous function of the distance between the two numbers; that the rightmost digit has an effect even when the leftmost digit suffices to respond; and that there is little or no discontinuity in reaction time at decade boundaries. Hence, two-digit numbers seem to be compared as a whole quantity and in an analog fashion.
In general, number comparison data are best explained by supposing that Arabic numerals are immediately coded mentally in an analogical representation of quantities similar to the one found in animals. My theory, which is validated by a considerable number of experimental results, proposes that numbers are not compared as digital symbols, but as analog quantities that can be represented by distributions of activation on an internal continuum, a “number line” obeying Fechner’s law (Dehaene et al., 1990; Restle, 1970). The numerical distance and size effect are a char-
acteristic signature of this mode of representing numbers. The fact that they are found in all sorts of number processing tasks, whether in animals or in humans, with numbers conveyed by symbols or by the numerosity of sets of objects, is crucial to my hypothesis that a “number sense” is part of our evolutionary heritage.
As a matter of fact, distance and size effects have been observed, not only during number comparison, but also during calculation (Ashcraft, 1992), addition or multiplication verification (Ashcraft and Stazyk, 1981), parity judgments (Dehaene et al., 1993), or number reading (Brysbaert, 1995). Interestingly, they are also found when subjects decide whether two digits are the same or different (Duncan and McFarland, 1980). I have shown that, even when subjects try to focus only on the physical differences between two digits, they are slower to respond “different” to two numerically close numbers such as 8 and 9 than to two distant numbers such as 2 and 9 (Dehaene and Akhavein, 1995). Independent evidence suggests that, every time a number is seen, a representation of the corresponding quantity is accessed automatically and irrepressibly (Brysbaert, 1995; Henik and Tzelgov, 1982; LeFevre et al., 1988; Tzelgov et al., 1992; Zbrodoff and Logan, 1986). In my future work, I plan to image this conscious and unconscious access to number semantics using a masked priming situation I have developed (Dehaene et al. 1998c).
Two studies with normal subjects have also provided further proof of the ubiquity of the quantity representation in human number processing. First, Mehler and I have discovered that in all languages, the frequency with which numerals are used in written or spoken texts decreases exponentially with number size, with only local increases for round numbers (Dehaene and Mehler, 1992). This effect can be entirely explained by supposing that language-specific number words are used to access a language-independent quantity representation, a universal compressive (Fechnerian) number line.
Second, a reaction time effect, the spatial-numerical association of response codes (SNARC), indicates that the number line is intimately associated with representations of space (Dehaene et al., 1993). The SNARC effect is the fact that whenever subjects do a bimanual response to numbers they respond faster with the right hand to larger numbers and with the left hand to smaller numbers. My own work used parity judgments, but others have replicated the SNARC effect with symmetry judgments (Huha et al., 1995) or rhyme judgments (Fias et al., 1996). The results indicate that the effect does not depend on the absolute size of the numbers, but only on their relative size. Its direction—small to the left and large to the right—does not change in left-handers, when the hands are crossed, or with mirror-imaged digits or number words. However, it does reverse in Iranian students who recently immigrated to France and who write
from right to left. Thus, there seem to be strong ties between numbers and space, but the direction of this spatial-numerical association is influenced by culture. In my book, The Number Sense (Dehaene, 1997), I speculate that number forms—the vivid, often colorful spatial patterns that some synesthetic subjects claim to experience when thinking about numbers (Galton, 1880; Seron et al., 1992)—are an unusually enriched version of this spatial-numerical association that exceptionally reaches access to consciousness.
Losing Number Sense
One final piece of evidence is required to demonstrate that the elementary understanding and manipulation of numerical quantities is part of our biological evolutionary heritage. One should show that it has a dedicated neural substrate. Animals and humans should share a set of homologous brain areas whose networks are dedicated to representing and acquiring knowledge of numerical quantities and their relations. This hypothesis has guided my neuropsychological and brain-imaging research for almost a decade. Although the demonstration is still far from complete, we now have two strong arguments to suggest that number processing is associated with a specific cerebral network located in the inferior intraparietal area of both hemispheres. First, neuropsychological studies of human patients with brain lesions indicate that the internal representation of quantities can be selectively impaired by lesions to that area (Benton, 1987; Cipolotti et al., 1991; Dehaene and Cohen, 1997; Gerstmann, 1940; Takayama et al., 1994; Warrington, 1982). Second, brain-imaging studies reveal that this region is specifically activated during various number processing tasks (Dehaene, 1996; Dehaene et al., 1996; Kiefer and Dehaene, 1997; Roland and Friberg, 1985; Rueckert et al., 1996).
It has been known for at least 80 years that lesions of the inferior parietal region of the dominant hemisphere can cause number processing deficits (Gerstmann, 1940; Hécaen et al., 1961; Henschen, 1920) (see Figure 4). In some cases, comprehending, producing, and calculating with numbers are globally impaired (Cipolotti et al., 1991). In others, however, the deficit may be selective for calculation and spare reading, writing, spoken recognition, and production of Arabic digits and numerals (Dehaene and Cohen, 1997; Hécaen et al., 1961; Takayama et al., 1994; Warrington, 1982).
My colleague Laurent Cohen and I have recently suggested that the core deficit in left parietal acalculia is a disorganization of an abstract semantic representation of numerical quantities rather than of calculation processes per se (Dehaene and Cohen, 1995; 1997). One of our patients, Mr. Mar (Dehaene and Cohen, 1997), experienced severe difficulties in calculation, especially with single-digit subtraction (75 percent errors). He
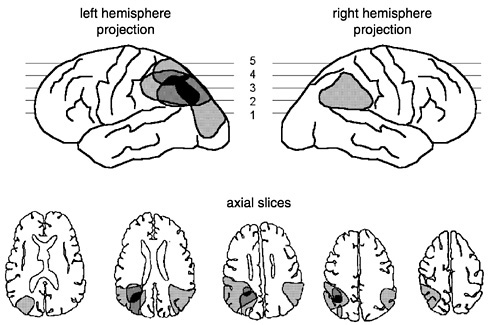
FIGURE 4 Approximate localization of the lesions in patients suffering from acalculia in the context of Gerstmann’s syndrome. Although these patients may still read and write numbers, they exhibit a selective deficit of the comprehension and mental manipulation of numerical quantities (redrawn from Dehaene et al., 1998b).
failed on problems as simple as 3 − 1, with the comment that he no longer knew what the operation meant. His failure was not tied to a specific modality of input or output, because the problems were simultaneously presented visually and read out loud and because he failed in both overt production and covert multiple-choice tests. Moreover, he also erred on tasks outside of calculation per se, such as deciding which of two numbers is the larger (16 percent errors) or what number falls in the middle of two others (bisection task: 77 percent errors). He easily performed analogous comparison and bisection tasks in non-numerical domains such as days of the week, months, or the alphabet (What is between Tuesday and Thursday? February and April? B and D?), indicating that he suffered from a category-specific deficit for numbers. We have now made several observations of patients with dominant-hemisphere inferior parietal lesions and Gerstmann’s syndrome. All of them showed severe impairments in subtraction and number bisection, suggesting disturbance to the central representation of quantities.
A “developmental Gerstmann syndrome,” also called developmental dyscalculia, has been reported in children (Benson and Geschwind, 1970; Spellacy and Peter, 1978; Temple, 1989, 1991). Some children show a highly selective deficit for number processing in the face of normal intelligence, normal language acquisition, and standard education. Paul (Temple, 1989), for instance, is a young boy who suffers no known neurological disease, has a normal command of language, and uses an extended vocabulary, but has experienced exceptionally severe difficulties in arithmetic since kindergarten. At the age of 11, he remained unable to multiply, subtract, or divide numbers and could only add some pairs of digits through finger counting. Paul can easily read nonwords as well as infrequent and irregular words such as colonel. However, he makes word substitution errors only when reading numerals, for instance, reading one as “nine” and four as “two.”
Although there is a dearth of accurate brain-imaging data on such developmental dyscalculia cases, it is tempting to view them as resulting from early damage to inferior parietal cortices that hold an innate, biologically determined representation of numbers. Part of my research project includes exploring the neural substrates of number processing in infants and young children with modern brain-imaging techniques and studying whether these networks are impaired in dyscalculia cases. With two American colleagues, Streissguth and Kopera-Frye, we have shown that children that have been exposed to alcohol in utero suffer from specific deficits of number sense (Kopera-Frye et al., 1996), suggesting that the laying down of numerical networks begins long before birth. Fetal alcohol syndrome (FAS) infants may thus provide a useful group for imaging early impairments of number processing. Developmental dyscalculia can be a devastatingly incapacitating deficit. I have the hope that a better understanding of the neural networks of number sense, and how they dysfunction when lesioned, may lead to better and earlier rehabilitation programs.
Although the inferior parietal region seems to play a crucial role in number sense, it is important to note that it is not the only brain region involved in number processing in adults. The phrenological notion that a single area can hold all the knowledge about an entire domain such as arithmetic has to give way to a more parallel view of number processing in the brain. Multiple brain areas are involved, whether for identifying Arabic numerals, writing them down, understanding spoken number words, retrieving multiplication facts from memory, or organizing a sequence of multidigit calculations. To explore these circuits, Cohen and I have studied tens of brain-lesioned patients with number processing deficits, and we have demonstrated the existence of previously unknown dis-
sociations (Cohen and Dehaene, 1991, 1994, 1995, 1996; Cohen et al., 1994; Dehaene and Cohen, 1991, 1994, 1995, 1997):
-
Split-brain patients: We have now seen three patients with posterior lesions restricted to the corpus callosum and whose posterior hemispheres were consequently disconnected, enabling us to study the numerical functions of the left and right hemispheres (Cohen and Dehaene, 1996). The results indicated that both hemispheres can recognize Arabic digits, convert them to quantities, and compare them. Only the left hemisphere, however, is able to name numerals and to perform exact calculations. Our results have confirmed and extended previous results obtained with surgical cases of callosal lesions (Gazzaniga and Hillyard, 1971; Gazzaniga and Smylie, 1984; Seymour et al., 1994).
-
Large left-hemispheric lesions: Two single cases of patients with large lesions of the posterior left hemisphere have enabled us to confirm that the right hemisphere may not be devoid of numerical abilities. One patient was able to provide approximate answers to calculations he could not perform in exact form (Dehaene and Cohen, 1991). The other, despite severe acalculia and number-naming deficits, remained able to access encyclopedic knowledge of quantities, dates, and other familiar numbers (Cohen et al., 1994).
-
Pure alexia: In two cases of patients with pure alexia, the visual input to the left hemisphere was destroyed by a left inferior occipito-temporal lesion. Yet again, we were able to confirm the presence of intact Arabic digit identification and comparison abilities, presumably supported by right-hemispheric circuits (Cohen and Dehaene, 1995).
-
Neglect dyslexia for number: One single case of a patient with a peculiar Arabic number reading deficit enabled us to study the cognitive mechanisms underlying number reading (Cohen and Dehaene, 1991).
-
Subcortical acalculia: One patient, a retired school teacher, with a lesion to the left lenticular nucleus failed to recite the multiplication table, together with prayers, poetry, nursery rhymes, or even the alphabet (Dehaene and Cohen, 1997). Number sense was preserved, however, as indicated by relatively preserved comparison, subtraction, and interval bisection abilities. Hence, there was a double dissociation with the deficits observed in parietal acalculia and Gerstmann’s syndrome. We suggested that a left cortico-subcortical circuit, connected to perisylvian language areas, contributes to the rote retrieval of arithmetic tables together with other verbal and nonverbal routine operations.
Many additional acalculia cases have been reported by other groups (Campbell and Clark, 1988; Caramazza and McCloskey, 1987; Cipolotti
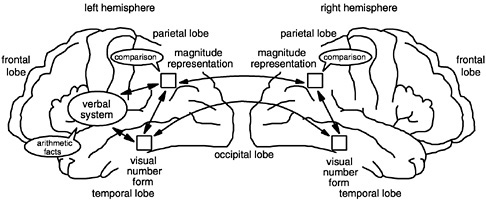
FIGURE 5 Schematic diagram of the main brain areas thought to be involved in simple number processing and their functional interactions (redrawn from Dehaene and Cohen, 1995).
and Butterworth, 1995; Cipolotti et al., 1994, 1995; Dagenbach and McCloskey, 1992; Deloche and Seron 1982a, 1982b, 1984, 1987; Macaruso et al., 1993; McCloskey, 1992, 1993; McCloskey and Caramazza, 1987; McCloskey et al., 1985, 1986, 1991a, 1991b, 1992; Noel and Seron, 1993; Seron and Deloche, 1983; Sokol et al., 1989, 1991; Warrington, 1982). Based on them, Cohen and I have proposed a tentative model of the cerebral circuits implicated in calculation and number processing: the triple-code model (Dehaene, 1992; Dehaene and Cohen, 1995) (see Figure 5). This model helps us make sense of the numerical deficits that patients exhibit consecutive to a focal brain lesion. It also does a fair job of accounting for chronometric data in normal subjects.
Functionally, the model rests on three fundamental hypotheses. First, numerical information can be manipulated mentally in three formats: an analogical representation of quantities, in which numbers are represented as distributions of activation on the number line; a verbal format, in which numbers are represented as strings of words (e.g., thirty seven); and a visual Arabic number form representation, in which numbers are represented as a string of digits (e.g., 37). Second, transcoding procedures enable information to be translated directly from one code to the other. Third, each calculation procedure rests on a fixed set of input and output codes. For instance, we have seen how number comparison works on numbers coded as quantities on the number line. Likewise, the model postulates that multiplication tables are memorized as verbal associations between numbers represented as a string of words and that multidigit operations are performed mentally using the visual Arabic code.
Anatomically, our neuropsychological observations have enabled us
to flesh out the model and to associate tentative anatomical circuits to each function. We speculate that the mesial occipito-temporal sectors of both hemispheres are involved in the visual Arabic number form; that the left perisylvian areas are implicated in the verbal representations of numbers (as with any other string of words); and, most crucially, that the inferior parietal areas of both hemispheres are involved in the analogical quantity representation. Note that the redundant representation of the visual and quantity codes in the left and right hemispheres can explain why pure alexia patients (Cohen and Dehaene, 1995) or patients with major left-hemispheric lesions (Cohen et al., 1994; Dehaene and Cohen, 1991; Grafman et al., 1989) remain able to compare two Arabic numerals. It also explains that in callosal patients, number comparison remains feasible by both hemispheres (Cohen and Dehaene, 1996; Seymour et al., 1994). However, the unilateral, left-hemispheric lateralization of the verbal code and of the calculation abilities that depend on it explains why a left-hemispheric lesion suffices to abolish these abilities and why the right hemisphere of callosal patients cannot read numbers aloud or calculate with them.
Imaging Number Sense in the Living Human Brain
Functional brain-imaging techniques now provide new tests of the organization of cognitive processes. In itself, information about cerebral localization is not particularly interesting to cognitive scientists. Once a specific brain area has been localized, however, it becomes possible to ask what are the parameters that make it more or less active; what tasks it is responsive to; and what aspects of the stimulus, task, or response do not affect its state of activity. With dynamic methods such as electroencephalography or magnetoencephalography, it is also possible to record how quickly and for how long a region activates. In all these respects, the triple-code model makes specific predictions. Most critically, it predicts that the left and right inferior parietal areas should be active during various quantitative number processing tasks, and that their activation should depend purely on quantitative parameters such as number size and numerical distance, but not on the input or output modality or on the notation used for numbers. I have tested these predictions using positron emission tomography (PET) (Dehaene et al., 1996), electroencephalography (event-related potentials or ERPs; Dehaene, 1996; Dehaene et al., 1994; Kiefer and Dehaene, 1997); and, more recently, functional magnetic resonance imaging (fMRI) (Chochon et al., 1999; Dehaene et al., 1999).
Roland and Friberg (1985) were the first to monitor blood flow changes during calculation as opposed to rest. When subjects repeatedly subtracted 3 from a given number, activation increased bilaterally in the
inferior parietal and prefrontal cortex. These locations were later confirmed using fMRI (Burbaud et al., 1995; Rueckert et al., 1996). Because the serial subtraction task imposes a heavy load on working memory, it was not clear which of these activations were specifically related to number processing. However, my own experiments using simpler tasks have confirmed that the inferior parietal area is specifically involved in number processing. In a PET study of multiplication and comparison of digit pairs, my colleagues and I again found bilateral parietal activation confined to the intraparietal region (Dehaene et al., 1996). This confirmed results obtained with a coarser resolution electroencephalogram method (Inouye et al., 1993) as well as with single-unit recordings in neurological patients (Abdullaev and Melnichuk, 1996).
In more detailed studies, I have begun to examine the prediction that inferior parietal cortex activation reflects the operation of an abstract quantity system largely independent of input and output modalities as well as of the specific arithmetic task involved. In one study, I used high-density recordings of ERPs during a number comparison task to study the cerebral basis of the distance effect. An additive-factor design was used in which three factors were varied: number notation (Arabic or verbal), numerical distance (close or far pairs), and response hand (right or left). The results revealed that inferior parietal activity was modulated by the numerical distance separating the numbers to be compared, but not by the notation used to present them (Dehaene et al., 1996). Notation did have a significant effect around 150 ms after the visual presentation of the stimuli, indicating bilateral processing for Arabic digits, but unilateral left-hemispheric processing for visual number words (in agreement with the triple-code model). By about 200 ms, however, ERPs were dominated by a parietal distance effect with a significant lateralization to the right, and without further influence of notation. The response hand effect, which emerged as early as 250 ms after the stimulus, provided an upper bound on the duration of the numerical comparison process.
A similar study of number multiplication showed that inferior parietal activity lasts longer during multiplication of two large digits than during multiplication of two small digits, again regardless of the modality of presentation of the operands (auditory or visual) (Kiefer and Dehaene, 1997). The main difference with my previous study was that the ERP effects, though always bilateral, were stronger over the left inferior parietal area during multiplication, but stronger over the right parietal area during number comparison. We recently replicated this modulation by task demands using fMRI (Chochon et al., 1999). Relative to letter reading, digit comparison yielded greater activity in the right inferior parietal area, multiplication greater activity in the left, and subtraction a bilateral increase (Figure 6). In spite of these small variations, however, an exten-

FIGURE 6 Bilateral activation of the intraparietal sulcus, postcentral sulcus, and prefrontal areas during single-digit subtraction in a single subject (data from Chochon et al., 1999).
sive bilateral intraparietal network was common to all three tasks. In agreement with neuropsychological observations of inferior parietal acaculia, I have suggested that this network represents the cerebral basis of the number sense.
FRONTIERS IN THE COGNITIVE NEUROSCIENCE OF NUMERACY
Outline of Future Research Projects
In the next decade, I plan to continue exploring the cerebral bases of human cognitive functions using number processing as a prime example. My research projects are headed in two main directions.
First, I plan to further test and validate the Dehaene–Cohen triple-code model of number processing. Despite the considerable progress that
has been made in understanding the cerebral bases of numeracy in the past decade, the present state of our knowledge cannot be considered definitive. Most importantly, although we are beginning to understand the organization of the adult cerebral networks for number processing —and further experiments are needed in that domain too—we still have little or no information as to how these networks develop. Dehaene-Lambertz and I are planning to run brain-imaging studies to determine if, when, and how inferior parietal circuits are involved in number processing during infancy and childhood. Our experiments, based on ERP recordings as well as on the newer technique of near-infrared spectroscopy (NIRS), should help us image the normal development of number sense and its disorganization in dyscalculia. We also have a very poor understanding of how the posterior cerebral networks for verbal, Arabic, and quantitative number processing are coordinated during the execution of complex sequential operations. Imaging will help us understand the anatomical organization of numerical representations, particularly in the intraparietal sulcus, and their relations to prefrontal cortices.
Second, the detailed knowledge that we have accrued in the numerical domain will be used to bring new light on more general issues in cognitive neuroscience. I shall use my expertise in the number domain to study the differences between conscious and unconscious processing of numerical information. My students and I have developed a masked priming task in which we have clear behavioral evidence of unconscious access to number semantics. We plan to use brain-imaging techniques to image the processing of unconscious masked primes and thereby to throw some light on what distinguishes conscious and unconscious processing in the human brain. Part of my research will also assess the existence of category-specific representations of word meaning. I shall examine whether other categories of meaning than numbers, such as body parts, animals, foods, and so on, can yield to a similar analysis using convergent behavioral, neuropsychological, and neuroimaging methods.
Research Setting
My research is conducted in the Service Hospitalier Frédéric Joliot (SHFJ) in Orsay, about 15 miles south of Paris. The center is equipped with two PET scanners, one cyclotron, two fMRI scanners, equipment for high-density recordings of ERPs from 128 electrodes, and a major computer network. SHFJ is run by the French Atomic Energy Commission, which provides excellent technical support for developments in imaging and computer science. Within it, my team specializes on the cognitive neuroscience of higher-level functions of the human brain.
My team also benefits from collaborations with two other colleagues.
First, Dehaene-Lambertz, whose research focuses on early language development and infant brain-imaging studies, collaborates on studies of numerical development. She is running 64-channel ERP studies of infants and young children with her own equipment set up in a neuropediatrics unit near Paris, with good recruitment of normal, brain-lesioned, and retarded infants and children. Second, my colleague, Laurent Cohen, a neurologist and an M.D./Ph.D., will continue in our longstanding collaboration on neuropsychology studies of patients with language and/or calculation impairments. Dr. Cohen does clinical work and research at the Hopital de la Salpêtrière in Paris, a well-known hospital with a specialization in neurology and neuropsychological dating back to the foundational studies of Broca, Charchot, or Pinel. Patients recruited at Salpêtrière will be tested behaviorally and using fMRI and ERP techniques, either at SHFJ or using Salpêtrière’s own resources (fMRI and whole-head magneto-encephalography).
Brain-imaging research is costly and fast changing. The James S. McDonnell Centennial Fellowship will considerably help to achieve the brain-imaging and the developmental parts of the research project. In the near future, Dehaene-Lambertz and I will explore the suitability of novel equipment for NIRS for brain-imaging studies of infant cognition. This novel imaging technique is sensitive to brain activation like fMRI, but it is much lighter and less invasive so that it can be applied readily to infants and young children. With it, I hope to image the development of the anatomical networks of number sense since infancy.
Imaging the Development of Number Sense
One of the most important gaps in our current understanding of number sense is the absence of any data concerning the cerebral underpinnings of infant numerical abilities and of children’s mathematical development. In The Number Sense (Dehaene, 1997), I speculated that infant and animal abilities for number processing indicate an early functioning of the cerebral networks of number sense in the inferior parietal lobe. I also suggested that acquisition of number words and of the counting sequence is accompanied by the development of systematic connections between the quantity representation in the parietal lobe and the rest of the language system. Dehaene-Lambertz and I will directly put these hypotheses to a test.
Event-Related Potential Studies in Infants
In the very near future, we will start running studies of infant number processing using high-density recordings of ERPs from 64 channels, a
technique with which we now have considerable experience (e.g., Dehaene-Lambertz and Dehaene, 1994). We shall start by replicating numerosity discrimination studies in the auditory modality, and later in the visual modality. ERPs will be recorded as infants are habituated to repeated sequences comprising an identical number of sounds (e.g., two), but with varied tempo, spacing, and identity of the individual sounds. Occasionally, sequences comprising a novel number of sounds (e.g., three) will be presented. ERPs will be monitored for the presence of a response to numerical novelty. A similar design, again controlling for all non-numerical parameters, will be used in the visual modality. A preliminary pilot study with adults indicates that we may find an auditory mismatch negativity similar to that found in other auditory mismatch situations (Näätänen, 1990), but also possibly an amodal parietal difference common to auditory and visual stimuli, and hence plausibly related to numerosity processing.
If this infant study works in a similar way, we also plan to replicate Wynn’s infant addition and subtraction paradigms using ERPs, looking for cerebral correlates of infant’s surprise response to an unexpected numerosity. In older children, between two and four years of age, we also hope to run ERP studies of how the child’s growing vocabulary of number words connects to number meaning. Wynn (1992b) has identified a stage in numerical development during which children can recite number words and yet fail to relate them to specific quantities (for instance they can count “one, two, three,” but fail to relate the final word “three” to the cardinal of the counted set; and they cannot point to a card with a specific number of objects, such as two). Imaging children listening to number words before and after that stage should enable us to test the hypothesis that the crucial change, at that stage, is the connection of the left-hemispheric verbal system to the nonverbal bilateral parietal quantity system.
Near-Infrared Spectroscopy
In the past four years, NIRS has been used in cerebral functional activation studies to monitor changes of cerebral oxygenation during sensory, motor, or cognitive tasks in human adults (Gratton et al., 1995; Kleinschmidt et al., 1996; Meek et al., 1995), including mathematical problem solving (Hoshi and Tamura, 1997). Briefly, laser light is shone through the scalp at several wavelengths, and scattered light is then detected through photodiodes only a few centimeters away. The intensity of light scattered at specific wavelengths is a direct reflection of oxyhemoglobin and deoxyhemoglobin concentration in the underlying tissue. Hence, NIRS is sensitive to the very same parameters that fMRI measures (brain oxygen-level dependent contrast). The equipment is so much lighter, how-
ever, as to be portable. Furthermore, the cost is low, and the signals can be sampled at up to 50 Hz, allowing for high temporal resolution studies. Ongoing research by Gratton and Fabiani at the University of Missouri suggests that the technique might also be directly sensitive to neuronal activity rather than to subsequent hemodynamic changes, allowing for an even greater temporal resolution. The only serious disadvantage is that, currently, a single emittor-photodiode pair can only detect activation in a single, relatively large brain volume of a few cubic centimeters.
There are several reasons why NIRS may become the ideal technique for brain-imaging studies in infants and in young children. First, fMRI and PET studies of young subjects are ethically questionable and impractical because of the requirements to suppress head motion and remain still but cognitively active. Second, ERP studies have an excellent temporal resolution but are notoriously poor at reconstructing the localization of brain activity. NIRS seems to be the only technique to date that can provide unquestionable functional brain localization information in very young subjects. Third, infants have a small translucent skull and small jaw muscles that should permit much cleaner signal acquisition than in adults. Fourth, NIRS is applicable only to a depth of 1–2 cm, a severe limitation for adult heads but one that should not prevent imaging the entire depth of the cortex in infants.
The Centennial Fellowship will help evaluate the feasibility of NIRS studies of infant and child development in the domains of language and arithmetic. Following pilot studies with simple auditory and visual stimulation, I hope to be able to run studies similar to the above ERP studies of numerosity discrimination in infants using NIRS. In the near future, only a technique such as NIRS seems to have the potential to directly test the hypothesis that the inferior parietal lobe is active during number processing as early as during infancy. But, as novel imaging techniques constantly emerge, I intend to use part of the grant money to follow all new developments in brain imaging that would be suited to noninvasive cognitive brain-imaging studies in young children.
Imaging the Anatomy of Number Sense as a Function of Expertise
The impact of education on the cerebral networks of number sense can also be tested in more classical studies using fMRI and ERPs in adults. My colleagues and I have already performed fMRI and ERP studies in which two operations—single-digit multiplication, which is learned through rote verbal memorization; and subtraction, which is performed through nonmemorized quantity manipulations—correlate with strikingly distinct cerebral activation (Chochon et al., 1999). In another very recent experiment, we showed that exact and approximate calculations
also rely on strikingly distinct circuits (Dehaene et al., 1999). I envision several extensions of this work. First, we can explicitly train adult subjects on various number facts using different strategies, and we can image the concomittent changes in brain activation. Second, we can image activation in the same basic arithmetic tasks in populations with various levels of expertise such as literary versus mathematically oriented students, or even possibly adults with prodigious calculation abilities as compared with normal adults. All of these studies should help us understand how the cerebral architecture for number processing changes with education and expertise. Third, a fascinating experiment would involve imaging cerebral activity during calculation and number processing in adults with or without number forms (Galton, 1880), thus addressing directly whether synesthesia for numbers corresponds to a genuine enrichment of the cerebral map for numbers.
Fourth, and perhaps most importantly, we may also image the cerebral correlates of specific deficits of number sense, either by comparing normal children with children with specific retardation in arithmetic (dyscalculia) or by studying adult brain-lesioned patients before and after recovery of calculation abilities. Once a reliable test of young children’s or even infants’ numerical abilities is developed using ERPs or NIRS, we may begin imaging populations of children at risk of developing dyscalculia, such as FAS or premature children.
Sequences of Calculation and Frontal Cortex
Once we begin to better understand the cerebral networks underlying simple calculations, and how they vary with age, education, and expertise, we can begin to address the issue of their coordination. Multidigit calculations such as 23 + 48 call for the sequential execution of several operations that are partially contingent on previous results (3 + 8 = 11, then 2 + 4 + carry 1 = 7). Prefrontal cortex and anterior cingulate cortex have been associated with the working memory and executive control systems that are needed for attentive, sequential task execution and task control. Indeed, these areas are active during serial subtraction problems (Chochon et al., 1999; Moonen et al., 1990; Roland and Friberg, 1985). I plan to study their specific contributions to calculation using a combination of brain-imaging and computational modeling.
In one experiment, subjects would perform different sequences of internal operations on the same pairs of sequentially presented digits: (a) subtracting the two digits (27 → “five”), thus engaging the quantity system; (b) multiplying the two digits (27 → “fourteen”), thus engaging rote memory for arithmetic facts; (c) subtracting, then multiplying (e.g., for the pair 2 7, first compute 7 − 2, then 2 × 7); (d) multiplying, then subtracting;
(e) subtracting, then multiplying the obtained result (e.g., 7 − 2 = 5, then 5 × 7 = 35); (f) multiplying, then subtracting the obtained result (e.g., 2 × 7 = 14, then 14 − 7 = 7). Activations would be imaged both with fMRI and with ERPs. The triple-code model predicts that ERPs should show the successive activation of parietal and left perisylvian networks in the task-imposed order. Furthermore, the sequence requirement (when having to execute two operations) and the working memory requirement (when having to hold an intermediate result online for further calculations) should yield a detectable additional activation in prefrontal and cingulate areas. Ultimately, other experiments could be designed to explore the contribution of anterior areas to more-complex operations such as those engaged when computing, say, 24 × 17 mentally.
Computational modeling of neural networks will also be used to explore the role of various prefrontal, cingulate, and basal subcortical circuits in executive functions. For the past 10 years, Changeux and I have developed neuronal models of functions associated with the prefrontal cortex. We have successively modeled the delayed-response task (Dehaene and Changeux, 1989), the Wisconsin Card Sorting Test (Dehaene and Changeux, 1991), the putative role of working memory in early numerical development (Dehaene and Changeux, 1993), and most recently the Tower of London Test (Dehaene and Changeux, 1997). The latter model provides a general architecture for the internal generation of a hierarchical sequence of actions aimed at achieving a certain goal. In the future, we hope to extend it to the case of simple arithmetic problems. The main advantage of this form of modeling is that it obliges us to specify in all detail the putative organization and connectivity patterns of functional subsystems such as planning, working memory, error correction, backtracking, or evaluation that are usually given only a loose definition in most "boxological" cognitive models. Hence, the model should provide more-detailed predictions for neuropsychological and brain-imaging studies.
Conscious and Unconscious Number Processing
Studies of number processing can also be relevant to more-general issues in cognitive neuroscience. A considerable controversy in human cognition concerns the extent to which a masked word may contact its semantic representation, or whether consciousness is necessary for semantic processing. Recently, with Ph.D. students Koechlin and Naccache, I have designed an experimental situation in which subjects demonstrably process up to a very high level a numerical information that they claim not to have seen (Dehaene et al., 1998c). Using this masked priming situation, I hope to bring new light to bear on the conditions under which
information may or may not enter consciousness and to identify the relevant networks of cerebral areas using brain-imaging techniques.
Our experiment consists in presenting on a computer screen a random string of consonants (e.g., QfFmPg), followed by a number in Arabic or in verbal notation (the prime; e.g., “two” or “2”), then another consonant string, and finally a second number (the target; e.g., “four” or “4”). When the prime is presented for a very short duration such as 43 ms, subjects cannot report it and claim to be aware only of the second target number. When, however, they are asked to decide if the target is larger or smaller than 5, their response time varies with the prime. On congruent trials where both prime and target fall on the same side of 5 (both larger or both smaller), responses are faster than on incongruent trials when they fall on opposite sides of 5. The effect, which is totally independent of prime and target notation, proves that the prime number has been unconsciously identified, associated with a certain quantity, and categorized as larger or smaller than 5. Unlike most semantic priming experiments, the effect is extremely robust (20 ms; all subjects show a positive priming effect). It can be considered as a normal subject form of “blindsight” (Weiskrantz, 1997).
This experiment opens up remarkable possibilities for studying the cerebral bases of unconscious semantic processing. First, we can record ERPs during the task. Our results to date indicate that ERPs are sufficiently sensitive to detect changes in cerebral activity associated with the prime only. For instance, we detect distinct activation patterns for primes in Arabic or in verbal notation. We also detect differences between congruent and incongruent trials. We even detect a motor readiness potential contralateral to the hand that the subject should use if he had to respond to the prime. It thus becomes possible to follow the sequence of cerebral processing of the prime from visual to semantic to motor areas. We have shown that single-event fMRI can detect small but significant differences in activation as a function of whether the prime “votes” for the left or right hand, a result which confirms the ERP lateralized readiness potential effect (Dehaene et al., 1998c).
If brain imaging continues to prove sensitive to subliminal cerebral processes, this will open up new perspectives. We plan to replicate our experiments while varying the task. This should enable us to explore which tasks can or cannot be executed unconsciously on masked primes. We might thus define a taxonomy of tasks that do or do not require conscious guidance. Brain imaging might then tell us whether this dissociation can be related to the activation of specific anatomical areas such as the anterior executive system (prefrontal cortex, anterior cingulate). My recently developed “global workspace” model of the neuronal networks
underlying conscious effortful tasks will provide a theoretical framework for this research (Dehaene et al., 1998a).
Numbers Among Other Categories of Words
One final issue I would like to address in future research is the notion of category-specific representations. As I mentioned in the introduction, numbers provide probably one of the best demonstrations of a biologically determined domain of knowledge, with evidence of precursor knowledge in animals and human infants, category-specific deficits in brain-lesioned patients, and a reproducible neural substrate as seen in brain imaging. In the next 10 years, I plan to extend the research strategy used with numbers to other categories of words. My colleagues shall study the core knowledge of several plausible categories of words, including animals, foods, colors, and numbers, but we shall focus particularly on the category of body parts, for which there is already some neuropsychological evidence for category-specific deficits (e.g., Dennis, 1976; Sirigu et al., 1991).
Behaviorally, we plan to run chronometric experiments with normal subjects aiming at determining the internal organization of the adult representation of the body. Suppose that subjects are asked to determine which of two body parts (say, knee and ankle) is higher—a task analogous to number comparison. We again expect to find a distance effect. Subjects should be faster for more-distant pairs, indicating that they have an internal representation of the body schema. Indeed, a distance effect on the body schema has been reported in a slightly different task setting (Lakatos and Shepard, 1997). By determining the precise characteristics of this distance metric, examining whether there is an automatic intrusion of a distance effect even in same –different judgments, and examining whether it depends on the input modality (name versus picture), we should be able to characterize this representation, its abstractness, and the automaticity of its access, just like we did with number.
Neuropsychologically, Cohen and I will be looking for cases of autotopoagnosia, or selective deficits of body knowledge, as well as cases of selective naming or comprehension deficits for body part names. As in our previous case studies in the number domain, we shall then characterize the processing level at which the deficit occurs by studying whether it generalizes to different input–output modalities and different semantic or nonsemantic tasks.
Finally, using brain imaging in adults, we shall also examine the presence of areas specifically activated during body part comparison as op-
posed to number comparison. In an experiment we are currently undertaking, 19 names of body parts are matched in frequency, length, and number of syllables to 19 number names. In different blocks, the words are presented auditorily or visually, and in English or in French, to bilingual subjects, while brain activity is recorded with 3-Tesla fMRI. Using a conjunction analysis in the statistical package SPM96, the results allow us to test the prediction that there are brain regions associated with number meaning or with body part meaning that are active only for a certain category of word, but are irrespective of the modality or language used to access that category. Our preliminary results do indeed show such activity in the intraparietal region for numbers and in the left parietal cortex for body parts. In the future, we shall also replicate these results with ERPs. By examining at what point the ERPs to different categories of words begin to diverge, we shall estimate how quickly the semantic representation is accessed in different modalities and languages of input. I have successfully used this approach in a previous ERP experiment in which I showed striking ERP differences to proper names, animal names, action verbs, and numerals by 250–300 ms following visual word presentation (Dehaene, 1995).
CONCLUSIONS AND IMPLICATIONS
The present project can have a significant impact on our understanding of brain function and on its application to pressing societal issues. At the fundamental level, it will provide an in-depth understanding of how the networks for number processing develop in infants and in young children and how they are organized in adults as a function of task and expertise. In the past 10 years, number processing has become a central domain of human cognition, with its specialized scientific journal (Mathematical Cognition, edited by Brian Butterworth) and several dedicated congresses. The triple-code model of number processing currently provides a useful integrative framework for this domain. I intend to continue developing my research on elementary arithmetic to make it one of the best demonstrations of a biologically determined, category-specific domain, with evidence ranging from infant and animal studies to neuropsychology, brain imaging, and neuronal modeling. Starting from the numerical field, forays will also be made into other central issues in cognitive science, including conscious and unconscious processing and the nature and origins of category-specific representations.
Calculation and arithmetic are also a domain of central importance to society. Dyscalculia is a frequent and poorly understood deficit in school children, with causes that range from neurological damage to an acquired phobia for mathematics. Even in children or adults who have followed a
normal school curriculum, innumeracy is a growing concern. Although my research is mostly oriented toward the fundamental goal of understanding brain function, there may eventually be important applications in the domain of developmental dyscalculia, innumeracy, and their early diagnosis and rehabilitation. The triple-code model of number processing is already being applied by developmental psychologists such as Robbie Case to better understand normal and impaired numerical development. Cognitive tests are also being incorporated in batteries used for the diagnosis and rehabilitation of acalculia, not only in adults, but also in younger brain-lesioned patients. In the future, it seems likely that brain-imaging tools such as the ones I am helping develop will be used to understand the normal development of the cerebral networks for arithmetic and its variability and occasional failure. Once reliable imaging paradigms are developed, especially in young children and infants, they will be readily applicable to at-risk populations such as premature or FAS babies. They will also help understand and facilitate the recovery of adult patients with acalculia stemming from focal brain damage.
Finally, is it unreasonable to hope that, by improving our understanding of how the brain acquires mathematics, one may also discover better ways of teaching it? Quite the contrary, I believe that this is an achievable goal. Educational methods with a sound scientific basis are perhaps the most important contribution that cognitive neuroscience has to make to society. Our current understanding of numerical cognition already sheds some light on issues such as how one should teach the multiplication table or whether and how electronic calculators should be used in classrooms (Dehaene, 1997). Although these conclusions are still speculative, many educational developments are to be expected as the field of numerical cognition matures in the next 10 years.
REFERENCES
Abdullaev, Y. G., and K. V. Melnichuk. 1996. Counting and arithmetic functions of neurons in the human parietal cortex. NeuroImage 3:S216.
Antell, S. E., and D. P. Keating. 1983. Perception of numerical invariance in neonates. Child Development 54:695–701.
Ashcraft, M. H. 1992. Cognitive arithmetic: A review of data and theory. Cognition 44:75–106.
Ashcraft, M. H., and E. H. Stazyk. 1981. Mental addition: A test of three verification models. Memory and Cognition 9:185–196.
Barkow, J. H., L. Cosmides, and J. Tooby, eds. 1992. The Adapted Mind: Evolutionary Psychology and the Generation of Culture . New York: Oxford University Press.
Benson, D. F., and N. Geschwind. 1970. Developmental Gerstmann syndrome. Neurology 20:293–298.
Benton, A. L. 1987. Mathematical disability and the Gerstmann syndrome. In G. Deloche and X. Seron, eds. Mathematical Disabilities: A Cognitive Neuropsychological Perspective . Hillsdale, N.J.: Lawrence Erlbaum Associates.
Bijeljac-Babic R., J. Bertoncini, and J. Mehler. 1991. How do four-day-old infants categorize multisyllabic utterances. Developmental Psychology 29:711–721.
Boysen, S. T., and G. G. Berntson. 1996. Quantity-based interference and symbolic representations in chimpanzees (Pan trogolodytes). Journal of Experimental Psychology: Animal Behavior Processes 22:76–86.
Boysen, S. T., and E. J. Capaldi. 1993. The Development of Numerical Competence: Animal and Human Models. Hillsdale, N.J.: Erlbaum.
Brannon, E. M., and H. S. Terrace. 1998. Ordering of the numerosities 1 to 9 by monkeys. Science 282 (5389):746–749.
Brannon, E. M., and H. S. Terrace. 2000. Representation of the numerosities 1–9 by rhesus macaques (Macaca mulatto). Journal of Experimental Psychology: Animal Behavior Processes 26:31–49.
Brysbaert, M. 1995. Arabic number reading: On the nature of the numerical scale and the origin of phonological recoding. Journal of Experimental Psychology: General 124:434–452.
Buckley, P. B., and C. B. Gillman. 1974. Comparison of digits and dot patterns. Journal of Experimental Psychology 103:1131–1136.
Burbaud, P., P. Degreze, P. Lafon, J.-M. Franconi, B. Bouligand, B. Bioulac, J.-M. Caille, and M. Allard. 1995. Lateralization of prefrontal activation during internal mental calculation: A functional magnetic resonance imaging study. Journal of Neurophysiology 74:2194–2200.
Campbell, J. I. D., and J. M. Clark. 1988. An encoding complex view of cognitive number processing: Comment on McCloskey, Sokol & Goodman (1986). Journal of Experimental Psychology: General 117:204–214.
Capaldi, E. J., and D. J. Miller. 1988. Counting in rats: Its functional significance and the independent cognitive processes that constitute it. Journal of Experimental Psychology: Animal Behavior Processes 14:3–17.
Caramazza, A. 1996. The brain’s dictionary. Nature 380:485–486.
Caramazza, A., and M. McCloskey. 1987. Dissociations of calculation processes. In G. Deloche and X. Seron, eds. Mathematical Disabilities: A Cognitive Neuropsychological Perspective . Hillsdale, N.J.: Lawrence Erlbaum Associates.
Caramazza, A., and J. R. Shelton. 1998. Domain-specific knowledge systems in the brain: The animate-inanimate distinction. Journal of Cognitive Neuroscience 10:1–34.
Chochon, F., L. Cohen, P. F. van de Moortele, and S. Dehaene. 1999. Differential contributions of the left and right inferior parietal lobules to number processing. Journal of Cognitive Neuroscience 11:617–630.
Church, R. M., and W. H. Meck. 1984. The numerical attribute of stimuli. In H. L. Roitblat, T. G. Bever, and H. S. Terrace, eds. Animal Cognition. Hillsdale, N.J.: Erlbaum.
Cipolotti, L., and B. Butterworth. 1995. Toward a multiroute model of number processing: Impaired number transcoding with preserved calculation skills. Journal of Experimental Psychology: General 124:375–390.
Cipolotti, L., B. Butterworth, and G. Denes. 1991. A specific deficit for numbers in a case of dense acalculia. Brain 114:2619–2637.
Cipolotti, L., B. Butterworth, and E. K. Warrington. 1994. From one thousand nine hundred and forty-five to 1000945. Neuropsychologia 32:503–509.
Cipolotti, L., E. K. Warrington, and B. Butterworth. 1995. Selective impairment in manipulating arabic numerals. Cortex 31:73–86.
Cohen, L., and S. Dehaene. 1991. Neglect dyslexia for numbers? A case report. Cognitive Neuropsychology 8:39–58.
Cohen, L., and S. Dehaene. 1994. Amnesia for arithmetic facts: A single case study. Brain and Language 47:214–232.
Cohen, L., and S. Dehaene. 1995. Number processing in pure alexia: The effect of hemispheric asymmetries and task demands. NeuroCase 1:121–137.
Cohen, L., and S. Dehaene, 1996. Cerebral networks for number processing: Evidence from a case of posterior callosal lesion. NeuroCase 2:155–174.
Cohen, L., S. Dehaene, and P. Verstichel. 1994. Number words and number non-words: A case of deep dyslexia extending to arabic numerals. Brain 117:267–279.
Dagenbach, D., and M. McCloskey. 1992. The organization of arithmetic facts in memory: Evidence from a brain-damaged patient. Brain and Cognition 20:345–366.
Damasio, H., T. J. Grabowski, D. Tranel, R. D. Hichwa, and A. R. Damasio. 1996. A neural basis for lexical retrieval. Nature 380:499–505.
Davis, H., and R. Pérusse. 1988. Numerical competence in animals: Definitional issues current evidence and a new research agenda. Behavioral and Brain Sciences 11:561–615.
Dehaene, S. 1989. The psychophysics of numerical comparison: A re-examination of apparently incompatible data. Perception and Psychophysics 45:557–566.
Dehaene, S. 1992. Varieties of numerical abilities. Cognition 44:1–42.
Dehaene, S. 1995. Electrophysiological evidence for category-specific word processing in the normal human brain. NeuroReport 6:2153–2157.
Dehaene, S. 1996. The organization of brain activations in number comparison: Event-related potentials and the additive-factors methods. Journal of Cognitive Neuroscience 8:47–68.
Dehaene, S. 1997. The Number Sense. New York: Oxford University Press.
Dehaene, S., and R. Akhavein. 1995. Attention, automaticity and levels of representation in number processing. Journal of Experimental Psychology: Learning, Memory, and Cognition 21:314–326.
Dehaene, S., and J. P. Changeux. 1989. A simple model of prefrontal cortex function in delayed-response tasks. Journal of Cognitive Neuroscience 1:244–261.
Dehaene, S., and J. P. Changeux. 1991. The Wisconsin Card Sorting Test: Theoretical analysis and modelling in a neuronal network. Cerebral Cortex 1:62–79.
Dehaene, S., and J. P. Changeux. 1993. Development of elementary numerical abilities: A neuronal model. Journal of Cognitive Neuroscience 5:390–407.
Dehaene, S., and J. P. Changeux. 1997. A hierarchical neuronal network for planning behavior. Proceedings of the National Academy of Sciences of the United States of America 94:13,293–13,298.
Dehaene, S., and L. Cohen. 1991. Two mental calculation systems: A case study of severe acalculia with preserved approximation. Neuropsychologia 29:1045–1074.
Dehaene, S., and L. Cohen. 1994. Dissociable mechanisms of subitizing and counting: Neuropsychological evidence from simultanagnosic patients. Journal of Experimental Psychology: Human Perception and Performance 20:958–975.
Dehaene, S., and L. Cohen. 1995. Towards an anatomical and functional model of number processing. Mathematical Cognition 1:83–120.
Dehaene, S., and L. Cohen. 1997. Cerebral pathways for calculation: Double dissociation between rote verbal and quantitative knowledge of arithmetic. Cortex 33:219–250.
Dehaene, S., and J. Mehler. 1992. Cross-linguistic regularities in the frequency of number words. Cognition 43:1–29.
Dehaene, S., E. Dupoux, and J. Mehler. 1990. Is numerical comparison digital?: Analogical and symbolic effects in two-digit number comparison. Journal of Experimental Psychology: Human Perception and Performance 16:626–641.
Dehaene, S., S. Bossini, and P. Giraux. 1993. The mental representation of parity and numerical magnitude. Journal of Experimental Psychology: General 122:371–396.
Dehaene, S., M. I. Posner, and D. M. Tucker. 1994. Localization of a neural system for error detection and compensation. Psychological Science 5:303–305.
Dehaene, S., N. Tzourio, V. Frak, L. Raynaud, L., Cohen, J. Mehler, and B. Mazoyer. 1996. Cerebral activations during number multiplication and comparison: A PET study. Neuropsychologia 34:1097–1106.
Dehaene, S., M. Kerszberg, and J. P. Changeux. 1998a. A neuronal model of a global workspace in effortful cognitive tasks. Proceedings of the National Academy of Sciences of the United States of America 95:14,529–14,534.
Dehaene, S., G. Dehaene-Lambertz, and L. Cohen. 1998b. Abstract representations of numbers in the animal and human brain. Trends in Neuroscience 21:355–361.
Dehaene, S., L. Naccache, G. Le Clec’H, E. Koechlin, M. Mueller, G. Dehaene-Lambertz, P. F. van de Moortele, and D. Le Bihan. 1998c. Imaging unconscious semantic priming. Nature 395:597–600.
Dehaene, S., E. Spelke, R. Stanescu, P. Pinel, and S. Tsivkin. 1999. Sources of mathematical thinking: Behavioral and brain-imaging evidence. Science 284:970–974.
Dehaene-Lambertz, G., and S. Dehaene. 1994. Speed and cerebral correlates of syllable discrimination in infants. Nature 370:292–295.
Deloche, G., and X. Seron. 1982a. From one to 1: An analysis of a transcoding process by means of neuropsychological data. Cognition 12:119–149.
Deloche, G., and X. Seron. 1982b. From three to 3: A differential analysis of skills in transcoding quantities between patients with Broca’s and Wernicke’s aphasia. Brain 105:719–733.
Deloche, G., and X. Seron. 1984. Semantic errors reconsidered in the procedural light of stack concepts. Brain and Language 21:59–71.
Deloche, G., and X. Seron. 1987. Numerical transcoding: A general production model. In G. Deloche and X. Seron, eds. Mathematical Disabilities: A Cognitive Neuropsychological Perspective . Hillsdale N.J.: Lawrence Erlbaum Associates.
Dennis, M.. 1976. Dissociated naming and locating of body parts after left anterior temporal lobe resection: An experimental case study. Brain and Language 3:147–163.
Duncan, E. M., and C. E. McFarland. 1980. Isolating the effects of symbolic distance and semantic congruity in comparative judgments: An additive-factors analysis. Memory and Cognition 8:612–622.
Fias, W., M. Brysbaert, F. Geypens, and G. d’Ydewalle. 1996. The importance of magnitude information in numerical processing: Evidence from the SNARC effect. Mathematical Cognition 2:95–110.
Fodor, J. A. 1983. The Modularity of Mind. Cambridge, Mass.: MIT Press.
Gallistel, C. R., 1989. Animal cognition: The representation of space, time, and number. Annual Review of Psychology 40:155–189.
Gallistel, C. R.. 1990. The Organization of Learning. Cambridge, Mass.: MIT Press.
Gallistel, C. R., and R. Gelman, 1992. Preverbal and verbal counting and computation. Cognition 44:43–74.
Galton, F. 1880. Visualised numerals. Nature 21:252–256.
Gazzaniga, M. S., and S. A. Hillyard. 1971. Language and speech capacity of the right hemisphere. Neuropsychologia 9:273–280.
Gazzaniga, M. S., and C. E. Smylie. 1984. Dissociation of language and cognition: A psychological profile of two disconnected right hemispheres. Brain 107:145–153.
Gerstmann, J.. 1940. Syndrome of finger agnosia disorientation for right and left agraphia and acalculia. Archives of Neurology and Psychiatry 44:398–408.
Grafman, J., D. Kampen, J. Rosenberg, A. Salazar, and F. Boller. 1989. Calculation abilities in a patient with a virtual left hemispherectomy. Behavioural Neurology 2:183–194.
Gratton, G., M. Fabiani, D. Friedman, M. A. Franceschini, S. Fantini, P. Corballis, and E. Gratton. 1995. Rapid changes of optical parameters in the human brain during a tapping task. Journal of Cognitive Neuroscience 7:446–456.
Hauser, M. D., P. MacNeilage, and M. Ware. 1996. Numerical representations in primates. Proceedings of the National Academy of Sciences of the United States of America 93:1,514–1,517.
Hécaen, H., R. Angelergues, and S. Houillier. 1961. Les variétés cliniques des acalculies aucours des lésions rétro-rolandiques: Approche statistique du problème. Revue Neurologique 105:85–103.
Henik, A., and J. Tzelgov. 1982. Is three greater than five?: The relation between physical and semantic size in comparison tasks. Memory and Cognition 10:389–395.
Henschen, S. E. 1920. Klinische und Anatomische Beitraege zur Pathologie des Gehirns. Stockholm: Nordiska Bokhandeln.
Hillis, A. E., and A. Caramazza. 1995. Representation of grammatical categories of words in the brain. Journal of Cognitive Neuroscience 7:396–407.
Hinrichs, J. V., D. S. Yurko, and J. M. Hu. 1981. Two-digit number comparison: Use of place information. Journal of Experimental Psychology: Human Perception and Performance 7:890–901.
Hoshi, Y., and M. Tamura. 1997. Near-infrared optical detection of sequential brain activation in the prefrontal cortex during mental tasks. NeuroImage 5:292–297.
Huha, E. M., D. B. Berch, and R. Krikorian. 1995. Obligatory Activation of Magnitude Information During Non-Numerical Judgments of Arabic Numerals. Poster presented at the annual meeting of the American Psychological Society, New York.
Inouye, T., K. Shinosaki, A. Iyama, and Y. Mastumoto. 1993. Localisation of activated areas and directional EEC patterns during mental arithmetic. Electroencephalography and Clinical Neurophysiology 86:224–230.
Kiefer, M., and S. Dehaene. 1997. The time course of parietal activation in single-digit multiplication: Evidence from event-related potentials. Mathematical Cognition 3:1–30.
Kleinschmidt, A., H. Obrig, M. Requardt, K. D. Merboldt, U. Dirnagl, A. Villringer, and J. Frahm. 1996. Simultaneous recording of cerebral blood oxygenation changes during human brain activation by magnetic resonance imaging and near-infrared spectroscopy. Journal of Cerebral Blood Flow and Metabolism 16:817–826.
Koechlin, E., S. Dehaene, and J. Mehler. 1997. Numerical transformations in five month old human infants. Mathematical Cognition 3:89–104.
Kopera-Frye, K., S. Dehaene, and A. P. Streissguth. 1996. Impairments of number processing induced by prenatal alcohol exposure. Neuropsychologia 34:1187–1196.
Lakatos, S., and R. N. Shepard. 1997. Time-distance relations in shifting attention between locations on one’s body. Perception and Psychophysics 59(4):557–566.
LeFevre, J., J. Bisanz, and L. Mrkonjic. 1988. Cognitive arithmetic: Evidence for obligatory activation of arithmetic facts. Memory and Cognition 16:45–53.
Macaruso, P., M. McCloskey, and D. Aliminosa. 1993. The functional architecture of the cognitive numerical-processing system: Evidence from a patient with multiple impairments. Cognitive Neuropsychology 10:341–376.
Marr, D. 1982. Vision: A Computational Investigation into the Human Representation and Processing of Visual Information. New York: W. H. Freeman.
Martin, A., J. V. Haxby, F. M. Lalonde. C. L. Wiggs, and L. G. Ungerleider. 1995. Discrete cortical regions associated with knowledge of color and knowledge of action. Science 270:102–105.
Martin, A., C. L. Wiggs, L. G. Ungerleider, and J. V. Haxby. 1996. Neural correlates of category-specific knowledge. Nature 379:649–652.
Matsuzawa, T. 1985. Use of numbers by a chimpanzee. Nature 315:57–59.
McCarthy, R. A., and E. K. Warrington. 1988. Evidence for modality-specific meaning systems in the brain. Nature 334:428–430.
McCarthy, R. A., and E. K. Warrington. 1990. Cognitive Neuropsychology: A Clinical Introduction. San Diego, Calif.: Academic Press.
McCloskey, M. 1992. Cognitive mechanisms in numerical processing: Evidence from acquired dyscalculia. Cognition 44:107–157.
McCloskey, M. 1993. Theory and evidence in cognitive neuropsychology: A radical response to Robertson, Knight, Rafal, and Shimamura (1993). Journal of Experimental Psychology: Learning 19:718–734.
McCloskey, M., and A. Caramazza. 1987. Cognitive mechanisms in normal and impaired number processing. In G. Deloche and X. Seron, eds. Mathematical Disabilities: A Cognitive Neuropsychological Perspective . Hillsdale, N.J.: Lawrence Erlbaum Associates.
McCloskey, M., A. Caramazza, and A. Basili. 1985. Cognitive mechanisms in number processing and calculation: Evidence from dyscalculia. Brain and Cognition 4:171–196.
McCloskey, M., S. M. Sokol, and R. A. Goodman. 1986. Cognitive processes in verbal-number production: Inferences from the performance of brain-damaged subjects. Journal of Experimental Psychology: General 115:307–330.
McCloskey, M., D. Aliminosa, and S. M. Sokol. 1991a. Facts, rules and procedures in normal calculation: Evidence from multiple single-patient studies of impaired arithmetic fact retrieval. Brain and Cognition 17:154–203.
McCloskey, M., W. Harley, and S. M. Sokol. 1991b. Models of arithmetic fact retrieval: An evaluation in light of findings from normal and brain-damaged subjects. Journal of Experimental Psychology: Learning 17:377–397.
McCloskey, M., P. Macaruso, and T. Whetstone. 1992. The functional architecture of numerical processing mechanisms: Defending the modular model. In J. I. D. Campbell, ed. The Nature and Origins of Mathematical Skills. Amsterdam: Elsevier.
Mechner, F. 1958. Probability relations within response sequences under ratio reinforcement. Journal of the Experimental Analysis of Behavior 1:109–121.
Mechner, F., and L. Guevrekian. 1962. Effects of deprivation upon counting and timing in rats. Journal of the Experimental Analysis of Behavior 5:463–466.
Meek, W. H., and R. M. Church. 1983. A mode control model of counting and timing processes. Journal of Experimental Psychology: Animal Behavior Processes 9:320–334.
Meek, J. H., C. E. Elwell, M. J. Khan, J. Romaya, J. S. Wyatt, D.T. Delpy, and S. Zeki. 1995. Regional changes in cerebral haemodynamics as a result of a visual stimulus measured by near-infrared spectroscopy. Proceedings of the Royal Society of London Series B 261:351–356.
Mehler, J., and E. Dupoux. 1993. What Infants Know. The New Cognitive Science of Early Development . Oxford, U.K.: Blackwell.
Mitchell, R. W., P. Yao, P. T. Sherman, and M. O’Regan. 1985. Discriminative responding of a dolphin (Tursiops truncatus) to differentially rewarded stimuli. Journal of Comparative Psychology 99:218–225.
Moonen, C. T. W., P. C. M. Vanzijl, J. A. Frank, D. LeBihan, and E. D. Becker. 1990. Functional magnetic resonance imaging in medicine and physiology. Science 250:53–61.
Moore, D., J. Benenson, J. S. Reznick, M. Peterson, and J. Kagan. 1987. Effect of auditory numerical information on infant’s looking behavior: Contradictory evidence. Developmental Psychology 23:665–670.
Moyer, R. S., and T. K. Landauer. 1967. Time required for judgements of numerical inequality. Nature 215:1519–1520.
Näätänen, R. 1990. The role of attention in auditory information processing as revealed by event-related potentials and other brain measures of cognitive function. Behavioral and Brain Sciences 13:201–288.
Noel, M. P., and X. Seron. 1993. Arabic number reading deficit: A single case study. Cognitive Neuropsychology 10:317–339.
Pepperberg, I. M. 1987. Evidence for conceptual quantitative abilities in the African grey parrot: Labeling of cardinal sets. Ethology 75:37–61.
Pinker, S. 1995. The Language Instinct. London: Penguin.
Pinker, S. 1997. How the Mind Works. New York: Norton.
Platt, J. R., and D. M. Johnson. 1971. Localization of position within a homogeneous behavior chain: Effects of error contingencies. Learning and Motivation 2:386–414.
Restle, F. 1970. Speed of adding and comparing numbers. Journal of Experimental Psychology 91:191–205.
Rilling, M., and C. McDiarmid. 1965. Signal detection in fixed ratio schedules. Science 148:526–527.
Roland, P. E., and L. Friberg. 1985. Localization of cortical areas activated by thinking. Journal of Neurophysiology 53:1219–1243.
Rueckert, L., N. Lange, A. Partiot, I. Appollonio, I. Litvar, D. Le Bihan, and J. Grafman. 1996. Visualizing cortical activation during mental calculation with functional MRI. NeuroImage 3:97–103.
Rumbaugh, D. M., S. Savage-Rumbaugh, and M. T. Hegel. 1987. Summation in the chimpanzee (Pan troglodytes). Journal of Experimental Psychology: Animal Behavior Processes 13:107–115.
Seron, X., and G. Deloche. 1983. From 4 to four: A supplement to from three to 3. Brain 106:735–744.
Seron, X., M. Pesenti, M. P. Noël, G. Deloche, and J.-A. Cornet. 1992. Images of numbers or when 98 is upper left and 6 sky blue. Cognition 44:159–196.
Seymour, S. E., P. A. Reuter-Lorenz, and M. S. Gazzaniga. 1994. The disconnection syndrome: Basic findings reaffirmed. Brain 117:105–115.
Simon, T. J., S. J. Hespos, and P. Rochat. 1995. Do infants understand simple arithmetic? A replication of Wynn (1992). Cognitive Development 10:253–269.
Sirigu, A., J. Grafman, K. Bressler, and T. Sunderland. 1991. Multiple representations contribute to body knowledge processing: Evidence from a case of autotopoagnosia. Brain 114:629–642.
Sokol, S. M., R. Goodman-Schulman, and M. McCloskey. 1989. In defense of a modular architecture for the number-processing system: Reply to Campbell and Clark. Journal of Experimental Psychology: General 118:105–110.
Sokol, S. M., M. McCloskey, N. J. Cohen, and D. Aliminosa. 1991. Cognitive representations and processes in arithmetic: Inferences from the performance of brain-damaged subjects. Journal of Experimental Psychology: Learning 17:355–376.
Sophian, C., and N. Adams. 1987. Infants’ understanding of numerical transformations. British Journal of Developmental Psychology 5:257–264.
Spellacy, F., and B. Peter. 1978. Dyscalculia and elements of the developmental Gerstmann syndrome in school children. Cortex 14:197–206.
Starkey, P., and R. G. Cooper. 1980. Perception of numbers by human infants. Science 210:1033–1035.
Starkey, P., E. S. Spelke, and R. Gelman. 1983. Detection of intermodal numerical correspondences by human infants. Science 222:179–181.
Starkey, P., E. S. Spelke, and R. Gelman. 1990. Numerical abstraction by human infants. Cognition 36:97–127.
Strauss, M. S., and L. E. Curtis. 1981. Infant perception of numerosity. Child Development 52:1146–1152.
Takayama, Y., M. Sugishita, I. Akiguchi, and J. Kimura. 1994. Isolated acalculia due to left parietal lesion. Archives of Neurology 51:286–291.
Temple, C. M. 1989. Digit dyslexia: A category-specific disorder in development dyscalculia. Cognitive Neuropsychology 6:93–116.
Temple, C. M. 1991. Procedural dyscalculia and number fact dyscalculia: Double dissociation in developmental dyscalculia. Cognitive Neuropsychology 8:155–176.
Thompson, R. F., K. S. Mayers, R. T. Robertson, and C. J. Patterson. 1970. Number coding in association cortex of the cat. Science 168:271–273.
Tzelgov, J., J. Meyer, and A. Henik. 1992. Automatic and intentional processing of numerical information. Journal of Experimental Psychology: Learning, Memory, and Cognition 18:166–179.
Ungerleider, L. G., and M. Mishkin. 1982. Two cortical visual systems. In D. J. Ingle, M. A. Goodale, and R. J. Mansfield, eds. Analysis of Visual Behavior. Cambridge, Mass.: MIT Press.
van Loosbroek, E., and A. W. Smitsman. 1990. Visual perception of numerosity in infancy. Developmental Psychology 26:916–922.
van Oeffelen, M. P., and P. G. Vos. 1982. A probabilistic model for the discrimination of visual number. Perception and Psychophysics 32:163–170.
Warrington, E. K. 1982. The fractionation of arithmetical skills: A single case study. Quarterly Journal of Experimental Psychology 34A:31–51.
Washburn, D. A., and D. M. Rumbaugh. 1991. Ordinal judgments of numerical symbols by macaques (Macaca mulatto). Psychological Science 2:190–193.
Weiskrantz, L. 1997. Consciousness Lost and Found: A neurophyschological exploration. New York: Oxford University Press.
Woodruff, G., and D. Premack. 1981. Primative (sic) mathematical concepts in the chimpanzee: Proportionality and numerosity. Nature 293:568–570.
Wynn, K. 1992a. Addition and subtraction by human infants. Nature 358:749–750.
Wynn, K. 1992b. Children’s acquisition of the number words and the counting system. Cognitive Psychology 24:220–251.
Wynn, K. 1995. Origins of numerical knowledge. Mathematical Cognition 1:35–60.
Wynn, K. 1996. Infants’ individuation and enumeration of actions. Psychological Science 7:164–169.
Xu, F., and S. Carey. 1996. Infants’ metaphysics: The case of numerical identity. Cognitive Psychology 30:111–153.
Xu, F., and E. S. Spelke. 2000. Large number of discrimination in 6-month-old infants. Cognition 74(1):B1–B11.
Zbrodoff, N. J., and G. D. Logan. 1986. On the autonomy of mental processes: A case study of arithmetic. Journal of Experimental Psychology: General 115:118–130.
