Mendel in the Kitchen: A Scientist's View of Genetically Modified Foods (2004)
Chapter: 9 Poisoned Rats or Poisoned Wells
9
POISONED RATS OR POISONED WELLS
The increased public awareness of food allergy has arisen from a combination of three factors: reasoned concern, fear through ignorance, and political motivation.
—Bob Buchanan (2001)
On August 10, 1998, Arpad Pusztai was interviewed on the British TV show “World in Action.” Pusztai studied lectins, sugar-binding proteins found in peas and beans, cereals and potatoes. In his 35 years at the Rowett Research Institute in Aberdeen, Scotland, he had written three books on lectins and published 270 research papers. Because of his expertise, he had been asked to test the safety of a potato variety that had been genetically engineered to produce its own pesticide. That pesticide was a lectin from a flower, the snowdrop.
Pusztai fed the genetically modified potatoes to rats. His experiments, he told the television audience, showed that the GM potatoes damaged the rats’ immune systems and stunted their growth. He himself would not eat GM food, Pusztai said. He found it “very, very unfair to use our fellow citizens as guinea pigs.”
Pusztai’s study made headlines around the world. It came to be called “the poisoned rat debate.” According to a news report in the journal Science, the Rowett Institute was flooded with calls from reporters even before the television show aired. The Institute was faced with “a megacrisis we didn’t remotely anticipate,” said director Philip James. When James examined Pusztai’s experiments, he found them a
“muddle.” Pusztai’s laboratory was sealed, his notebooks were turned over to an audit committee, and Pusztai himself was put on indefinite leave—he was out of a job. The audit committee’s report, released in October 1998, concluded that Pusztai’s data did not support his conclusions.
Forbidden to talk to the press, Pusztai asked a number of scientists to review the audit committee’s report. In February 1999 they posted a memorandum, supported by more than 20 scientists, on the World Socialist Web Site. “Those of us who have known Dr. Pusztai’s work or have collaborated with him,” the memorandum stated, “were shocked by the harshness of his treatment by the Rowett and even more by the impenetrable secrecy surrounding these events. It is an unacceptable code of practice by the Rowett and its director, Professor James, to set themselves up as arbiters or judges of the validity of the data which could have such a profound importance not only for scientists, but also for the public and its health.”
One scientist stressed that “there is enough strong evidence that the work of the audit group was not objective and per se dangerous, not only for Dr. Pusztai, but generally for free and objective science.” Another thought the Rowett Institute’s treatment of Pusztai was “a great injustice.” Even Kenneth Lough, who had been the Rowett’s principal scientific officer from 1956 to 1987, said, “The Institute is at risk in sending the wrong signals to scientists in this field of research that any sign of apparent default [that is, any error in judgment] will be treated with the utmost severity. The awareness will of course act as a strong deterrent to those who wish to conduct research in this vitally important field.”
Pusztai’s study was also reviewed by a committee of six members of the British Royal Society. They sent out material from Pusztai, the Rowett Institute, and other sources to scientists with expertise in statistics, clinical trials, physiology, nutrition, quantitative genetics, growth and development, and immunology. The committee concluded that Pusztai’s experiments were poorly designed; the statistics he used were inappropriate and his results were inconsistent. They recommended that the experiments be repeated and the results be published.
Pusztai jumped to his own defense with a detailed response circu-
lated on the Internet. He and a colleague with whom he had worked for several years published their original study in the medical journal Lancet. For this Lancet also came under criticism. The U.K.’s Biotechnology and Biological Sciences Research Council called the journal “irresponsible.” Lancet’s editor, Richard Horton, stood by his decision. Five of six reviewers had favored publication, and he believed that it was appropriate for the information to be available in the public domain.
The lectin in question is called the Galanthus nivalis agglutinin, after the Latin name of the snowdrop, Galanthus nivalis. It is abbreviated GNA. Like other lectins, it recognizes and binds to sugars on other proteins. Although lectins were first discovered in plants, we now know that there are many different kinds of lectins in animals as well. Many proteins—in all kinds of organisms—are decorated with sugar molecules. Some, called glycoproteins, carry long strings or branches made of several sugar molecules. Each glycoprotein has a different complement of sugar molecules, depending on what it does and where. The sugar signature works like a zip code in the cell, determining where the protein is delivered by the machinery that produces it.
Lectins read these sugar codes. They serve many functions, one of which is to recognize disease-causing bacteria and viruses. For plants, lectins are a defense against insects. GNA, for instance, is mildly toxic to some pests of rice and other important crops. It does not affect ladybird beetles, considered to be beneficial insects, although it does affect parasitic wasps, which are also beneficial. Other lectins, including one called ricin, are quite toxic. When taken up by cells, ricin blocks the synthesis of proteins by inactivating the ribosomes. GNA does not have this property. Pusztai’s own studies showed that rats could safely eat purified GNA. Moreover, he and his colleagues found that GNA protected the rats against infection by Salmonella bacteria, the intestinal bug often found in raw eggs and on uncooked chicken. When the gene coding for GNA was introduced into potatoes and rice, it increased the plants’ resistance to insect pests. The next question was how a plant that produced GNA would affect a human gut. This question Puzstai tried to answer using rats as stand-ins for humans.
People already eat lectins. They are present in most plants, and are especially abundant in seeds, including cereals and beans, and in tubers
such as potatoes. They tend to survive cooking and digestive enzymes. They occasionally cause symptoms of food poisoning. A lectin called phytohemagglutinin or PHA, for example, is a normal component of kidney beans. Allergist David Freed recounts an incident in 1988 when a hospital had a “healthy eating day” in its cafeteria at lunchtime. Thirty-one portions of a dish containing kidney beans were served. Over the next several hours, 11 people experienced typical food-poisoning symptoms, including vomiting and diarrhea. All recovered by the next day, but no pathogen was found in the food. It turned out that the beans contained an abnormally high concentration of PHA.
Studying PHA in the early 1990s, Pusztai and his colleagues found that it caused the cells lining the surfaces of a rat’s intestines to die off and be replaced more quickly than usual. The rats became more susceptible to an overgrowth of the common gut bacterium, Escherichia coli. E. coli is harmless in small numbers, but causes stomach upset if it multiplies. The younger replacement cells on the tiny surface projections, or villi, of the intestines, Pusztai and his colleagues found, had a high proportion of proteins with mannose sugars on the ends of their sugar signatures. Because E. coli has projections, or fimbrae, that recognize and bind to mannose, the bacterium could grow more easily. (Pusztai also found that including the lectin GNA in the rat’s diet reduced the extent of bacterial overgrowth, because the GNA binds to the mannose and keeps the bacteria’s fimbrae from using it.)
As they do in insects, some lectins can get into and through animal cells and enter the bloodstream. Some are potent allergens. So even though GNA appeared to be relatively benign, there was no doubt that a food containing it needed careful testing. Sensibly, the Scottish Office of the Agriculture, Environment, and Fisheries Department commissioned a three-year study in 1995. The University of Durham and the Scottish Crop Research Institute were to provide the transformed potatoes, and the Rowett Institute was to do a chemical analysis of them. Pusztai was also to do both short-term (10-day) and long-term (3-month) rat feeding trials. In these he would compare the effects of eating potatoes from the transgenic plants with the effects of eating potatoes from the parent lines, that is, from the plants that provided the cells transformed by recombinant DNA.
The rats in Pusztai’s study were fed either raw or cooked potatoes. If they ate ordinary, nontransgenic potatoes, their diets were supplemented with pure GNA. The results of the experiment showed that the organs of the rats fed transgenic potatoes weighed significantly less than the same organs from control animals fed nontransgenic potatoes. In this control group—the rats fed purified GNA and ordinary potatoes—the lymphocytes (cells in their immune systems) were more responsive to stimulation by other lectins. By contrast, the lymphocytes were depressed in the animals fed the transgenic potatoes. It seemed that the potatoes expressing the GNA protein were somehow poisonous, while the ordinary potatoes—although spiced with pure GNA—were not.
Since he knew GNA wasn’t toxic, Pusztai jumped to the conclusion that the new gene itself—or perhaps the other DNA introduced along with it—was causing the problem. He went public with his conclusion on “World in Action.” What his experiments actually showed was that the genetically modified potatoes were different from each other, as well as from their parent lines. When the potatoes were chemically analyzed, the researchers measured total protein concentration, as well as the content of several relevant proteins, including GNA and potato lectin. All of these differed between the unmodified and modified potatoes, as well as between the different lines of modified potatoes. A later study on genetically modified potatoes found the same thing. Rather than the effects of the introduced DNA, what Pusztai was most likely seeing were the effects of somaclonal variation, the variation that arises as a result of tissue culture.
Plant breeders have known about somaclonal variation for decades. The techniques of tissue culture come from the 1950s, when Miller and Skoog identified the ingredient in old herring sperm that would make a plant cell divide and form a callus. Callus culture is commonly an intermediate stage between Agrobacterium-mediated transformation and the regeneration of transformed plants. With the right mix of hormones, callus cells divide for a long time; weaned off hormones, the callus turns into a plantlet, with roots, a shoot, and leaves. By 1981 the fact that different regenerated plantlets from a single callus were not identical was well-enough known to be given a name.
Somaclonal variation was both a nuisance and an aid. Through screening, breeders could pick and choose among the changes, discarding the bad ones.
Some of the changes, scientists have since learned, are epigenetic, meaning that the modifications affect the expression of genes, but not their structure. These changes can be caused by differences in how the DNA is methylated. Attaching methyl groups to genes shuts them down, silences them, makes them sleep. Taking the methyl groups off awakens them. These changes, though, tend to right themselves quickly once the plants are propagated sexually or through cuttings. Morestable genetic changes—deletions, insertions, single base changes, and rearrangements—also arise. Therefore, every new plant derived using tissue culture techniques must be evaluated both for how it grows and for its food properties. Potato breeders know to be especially careful of the toxic glycoalkaloids that potatoes naturally produce. These chemicals, which contribute to inflammatory bowel disease, are concentrated—not destroyed—when potatoes are fried.
Pusztai’s conclusion that the variation he observed was due to genetic engineering was unwarranted. His mistake proved costly—and not just to his reputation. Pusztai’s experiments have been attacked for their small sample sizes, for the use of inappropriate statistics, and for the fact that a diet of raw—or even cooked—potatoes is bad for rats, even if supplemented with a bit of extra protein. But oddly enough in the entire poisoned rat debate no one seems to have seen the central flaw in Pusztai’s experiments: the absence of appropriate controls. The control in an experiment is the material that allows a good comparison to be made in order to understand the consequences of the experimental treatment being studied. In Pusztai’s experiments the control potatoes had a different breeding history than the transgenic potatoes, so they couldn’t be compared directly to those being tested. Only the genetically modified potatoes had undergone tissue culture. To blame the new DNA for the potatoes’ effects on his rats, Pusztai needed control plants that had also come out of tissue culture and were exactly the same as the ones being tested—except for the new genes. Pusztai didn’t use such plants.
To a fellow nutritionist, Pusztai’s conclusions might have seemed
justified. A plant breeder, familiar with the tissue culture technique, would have seen instead the signs of somaclonal variation. The tissue culture-derived potatoes were very different from those the experiment had been started with. It is quite likely that it was the changes that occurred in culture that were responsible for Pusztai’s results, not the introduced gene. Perhaps we will never know. But it is quite clear that the expertise battle that sprang up around Pusztai’s experiments obscured an important point. When plants are engineered to express new proteins that could affect human health—and lectins are clearly in this category—the foodstuffs produced from them must be analyzed carefully.
![]()
Plant breeders, too, can have their blind spots. The Lenape potato was on the market—and potato chips were being made out of it—when the toxicity was discovered.
Steven Taylor, head of the University of Nebraska’s Department of Food Science and Technology, attended a conference at which Jim Cook, a plant pathologist at Washington State University, described a new wheat variety. In 1988 Cook had overseen the first field test of a genetically modified organism in the Pacific Northwest, a soil microorganism modified to protect wheat from root disease. In the early 1990s he had chaired the 70-member international working group that produced the report “Safety Considerations for Biotechnology Scale-up of Crop Plants” for the OECD. The new wheat he described at the conference was resistant to rust, a devastating fungal disease, the scourge that Norman Borlaug had studied half a century earlier. The wheat had been modified to produce a chitinase, an enzyme that can attack and break open molecules in the cell walls of invading fungi. Taylor asked if Cook knew that chitinases were major allergens. Cook replied that he did not. “I relay that story,” Taylor writes, “to show how important it is for the people selecting these genes to become more aware of what’s going to happen in the commercialization phase and to, perhaps, address some of those questions early in the process.”
In the past, few plant breeders had much awareness of the fine points of food safety or nutrition. The two disciplines are in different
university departments. There was little overlap until the risks and benefits of genetically modified food began bringing both to the same conferences. John Beard, a nutritionist at Pennsylvania State University, for example, tells the same sort of story about Ingo Potrykus. “I met Potrykus at a conference,” Beard said. “I asked him about the beta carotene in Golden Rice, ‘Is it bioavailable?’ He got this blank look on his face. ‘Is it absorbable?’ I asked. He said, ‘Why wouldn’t it be?’” Beard sent Peter Beyer, Potrykus’s colleague, references on cell culture systems with which nutritionists test a nutrient’s bioavailability, that is, how easily the body can absorb and use it.
This disconnect between the two fields led some nutritionists to sound the alarm against genetically modified food. Marion Nestle, a nutritionist at New York University, for instance, wrote in the New England Journal of Medicine in 1996, “Biotechnology companies might be introducing allergenic proteins from donor organisms into the food supply.” In an article in the Cambridge World History of Food, published in 2002, she elaborates: “Most biotechnology companies are using microorganisms rather than food plants as gene donors. Although these microbial proteins do not appear to share sequence similarities with known food allergens, few of them have as yet entered the food supply. At the present time, their allergenic potential is uncertain, unpredictable, and untestable.”
A plant breeder might quibble that we have, indeed, eaten most of these microbial proteins and genes. People are adventurous eaters. We eat plants and animals, fish, crustaceans, worms, insects, fungi, yeast (the Marmite so beloved in England is a yeast extract), and bacteria of all kinds. Some bacteria we eat accidentally—on unwashed lettuce or spotted apples, for instance. Other kinds give us our wonderful array of yogurts and cheeses. In both cases the bacteria we eat are alive, their genes busily producing microbial proteins. Yet even if the genes biotechnologists use come from yeasts and bacteria that we ordinarily eat, the need to better understand food allergies remains.
Many more people think they have a food allergy than actually do. While a quarter of all Americans will say they are allergic to some food, true allergies are actually relatively uncommon. Only about 2 percent of adults and 5 to 8 percent of children exhibit allergic responses when
given skin tests with a range of proteins. Nonetheless, allergies are a serious health concern. Allergic reactions range from annoying to life-threatening.
An allergic response, whether to a bee sting, pollen, or peanuts, commonly involves immunoglobulin E, a key molecule in the immune system. Immunoglobulin E, or IgE, comes in various shapes, each of which is made to match a particular protein. IgE molecules sit on the surfaces of special cells called mast cells and basophils. Mast cells lie just beneath the surface of body tissues that are exposed to the outside world: skin, gut, nasal passages, lungs, and urinary tract. Basophils are in the blood. When the shape of the IgE molecules matches that of a protein they’re exposed to, the IgE binds to it. In fact, two IgE molecules have to bind to two parts of the protein, called the allergen, thus linking the two IgE molecules together. This cross-linking triggers the mast cells to release packets of highly irritating substances—histamines and leukotrienes—to incite the body to eject the invader in one way or another. A runny nose, itchy eyes, swollen tongue, tight throat, asthma, hives, vomiting, diarrhea, and other discomforts can result.
People with allergies can produce as much as 10 times the average amount of IgE. This makes their mast cells rather jumpy, releasing floods of inflammatory molecules in response to proteins that other
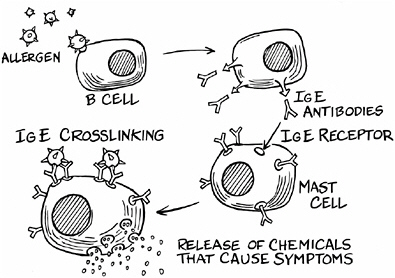
How IgE causes an allergic reaction
people’s mast cells ignore. These include proteins in pollen and bee venom as well as in food. And yet just a handful of the many proteins we eat cause most of the problems. “While virtually any food may cause an allergic reaction in someone, somewhere,” notes Dean D. Metcalfe, chief of the laboratory of allergic diseases at the NIH, “a relatively small number of foods appear to cause over 90 percent of all reported immediate reactions in both children and adults.” In young children, most problems are caused by milk and soybeans. Adults react most to peanuts and other nuts, shrimp and crustaceans, fish, and eggs. Sesame seeds and wheat are sometimes added to this most-allergenic list, as are peaches, plums, apricots, cherries, almonds, celery, and rice. Each of these foods holds hundreds of thousands of proteins. Just one or two of them are usually the IgE triggers, or allergens.
An IgE overreaction can be quite dangerous. Although most people have mild symptoms—hives, diarrhea—allergies can cause anaphylactic shock: difficulty breathing, a rapid and dramatic fall in blood pressure, and cardiovascular complications that can lead to death if not treated immediately. Allergic reactions account for thousands of emergency room visits a year in the U.S. About 135 people die each year as a result of allergic reactions. Although food allergies rarely trigger such extreme responses—the most common cause is bee stings—they can.
For someone who is allergic to a food, avoiding it is the only sure way to be safe. For many American children, that means no peanut butter sandwiches. But avoidance isn’t always so easy. Foodstuffs can show up in surprising places. Peanuts lurk in piecrusts, gravy, and hot chocolate, according to a front-page story in the New York Times about a promising drug for desensitizing people to peanuts. “One person died after eating an egg roll that used peanut butter to hold the roll together,” the Times reported.
The FDA recognized the problem of allergenicity early on. In its first guidelines on genetically modified foods, published in 1992, it warned against taking proteins from known allergens, such as peanuts, and moving them into other plants, such as corn. Someone who was allergic to peanuts would never think to avoid cornflakes. A related concern is that a protein from a bacterium or insect, for instance, when moved into a food like corn, might prove to be an allergen.
A 2001 brochure from the radical environmental group Greenpeace echoes Marion Nestle’s complaint: “Because most genes being introduced into GE [genetically engineered] plants come from sources which have never been part of the human diet, such as bacteria, insects, and viruses, there is no way of knowing whether or not the products of these genes will cause allergic reactions.” John Haglin, a physicist who ran for president of the United States in 2000, raises a new alarm in a video documentary called Genetically Engineered Food: Are We at Risk? “If you can teach a tomato, for example, to produce enough flounder blood, that tomato becomes frost-resistant and is more profitable,” he said. “What a fabulous application of technology—unless you’re allergic to fish, or your children are allergic to fish.”
Such proclamations, coupled with the very real concerns of parents whose children have food allergies, have led many people who are not otherwise against innovations to wonder about the wisdom of genetically modified food. And yet conventional plant breeding and many other farming practices can also affect how much of an allergen a crop contains. All allergens are proteins. Both the amount of a particular protein a plant contains and where in the plant that protein is concentrated are influenced by the weather, by stresses due to disease or pests, and by the plant’s breeding history. None of these factors has had much effect on food allergies. Instead, says a 2000 report issued by the World Health Organization (WHO) and the U.N.’s Food and Agriculture Organization (FAO), what has caused new allergies to appear are foods eaten in unusual quantities and foods newly brought in from foreign lands. Peanut allergies are frequent in the United States, Canada, and parts of Europe, but not in countries where peanuts are rarely eaten, the report points out. “Also, recent food introductions such as kiwi fruit have proven to be additional sources of food allergens.”
In 1962, when the first 2,400 pounds of Chinese gooseberries were sold in Los Angeles under the name kiwi fruit, no one raised the issue of allergenicity. Catherine Woteki is the dean of agriculture at Iowa State University and a former undersecretary of food safety in the USDA. “I can remember the time when there was no kiwi fruit in the U.S.,” she said. When the first shipment was brought in, she recalled, “There were no requirements for allergy testing. None.” Kiwis were
considered “a traditional food,” one with a history of having been eaten in other countries. They were assumed to be safe. That designation has not been changed even though many Americans have found they are allergic to kiwi fruit.
Nor is the allergy issue raised today when rambutans and white apricots are turned up by the self-styled Fruit Detective, David Karp, whose job, according to a profile in the New Yorker magazine is to “range around the country and the world and find exotic fruits, or uncommon varieties of common fruits” for grocers who like to “titillate” their customers with “curiosities.” The white apricots were created by a California farmer who had worked for 30 years to interbreed apricot stock he had imported from Morocco and Iran. The rambutans, “bright-red, golf-ball-size, tendril-covered fruits from Southeast Asia, with translucent, sweet-tart flesh,” were grown in Hawaii. They could not be imported to the mainland until a new technology involving electron beams was devised to destroy the tropical pests they harbor. “The new technology, many in the exotic-fruit world believe, will greatly expand Americans’ awareness of the fruit that the rest of the world eats, and bring a cornucopia of new items to the produce department,” the New Yorker reported. None of these new introductions will be tested for allergenicity.
The number one food allergen, the peanut, is itself a rather recent newcomer to North America. Although the peanut originated in South America, it came to the United States by way of Africa, having been brought to Virginia by slaves. Its popularity didn’t climb until the 1890s, when John Harvey Kellogg, the inventor of cornflakes and granola, began making peanut butter at his health sanitarium. He wanted his patients with poor teeth to enjoy what he called “the noble nut.” The fact that peanut butter can cause fatal allergic reactions was then—and is now—generally overlooked. Peanut butter is more likely to be thought of today as a health food, a staple of children’s lunchboxes.
But the FDA and developers of genetically modified crops are not ignoring the problem of allergenicity. They consider it their most important food safety issue. And while Greenpeace worries that “there is no way of knowing whether or not the products of these genes will
cause allergic reactions,” scientists have made considerable progress in identifying and categorizing what makes proteins allergenic.
Most allergenic proteins have several traits in common. They are generally abundant in the food, representing more than 1 percent of the total protein. They survive cooking. And they are quite stable, resisting being broken down by stomach acid and digestive enzymes. But these are not hard and fast rules and there are exceptions. Some very abundant proteins aren’t allergens. Some proteins are allergenic, but not very abundant. Some unstable proteins—and even unfolded proteins, which should digest very easily—are allergenic. Yet despite our inability to develop criteria with no exceptions, there is a rapidly growing body of information about what makes a protein an allergen. This information includes the amino acid sequences that interact with IgE, how the protein folds, and the extent to which a protein is decorated with sugar molecules.
To see if a protein is likely to be allergenic, for example, its amino acid sequence is compared to those of known allergens in chunks as small as eight amino acids long. The known allergens with which it is compared include not only food allergens, but proteins from pollens, fungal spores, insect venom, and other things people have allergies to. If a sequence of 80 amino acids or more is the same over a third of this stretch, as a sequence in a known allergen, the protein is considered suspect.
The function of the protein is also a factor. As the WHO-FAO report said in 2000, “Certain classes of protein are well known allergens. For example, the 2S high-methionine albumins from Brazil nut, walnut, sunflower seed, and mustard are major allergens from those sources. Thus, other 2S high-methionine albumins should be scrutinized very carefully.” The report suggested that a list be compiled of proteins that scientists agree have “allergenic potential.” Such lists have since been made available for everyone’s use on the Internet.
If the protein comes from a known allergen, such as the peanut, it can be tested against serum from people who are allergic to peanuts. A test tube assay will show if, and how strongly, each person’s IgE reacts to the new protein. If the laboratory test is negative, skin-prick tests—the kind of test done by a doctor to diagnose an allergy in the first
place—can be done. If those are negative, allergic people can be recruited to perform a double-blind, placebo-controlled food challenge, a test in which neither the scientists nor the subjects know who is eating the potentially allergenic food and who is eating the harmless placebo.
What’s missing in this battery of tests is a good animal model—a way of testing a new food on an animal first, instead of asking humans to become guinea pigs. Several animals have been tried: the guinea pig, of course, but also the rat, pig, dog, and mouse. None, as yet, has been shown to react to allergens exactly as a person would. As the University of Nebraska’s Steve Taylor noted in an address published in Nutrition Reviews, “I would indicate to those of you interested in pursuing careers in this field that one of the most desperately needed things is a validated animal model.”
The questions to ask to learn if a new protein will cause food allergies were published by WHO and the FAO in 2000 and 2001. The consensus of the American Medical Association is that by following these rules, “the overall risks of introducing an allergen into the food supply are believed to be similar to or less than that associated with conventional breeding methods.” Samuel B. Lehrer, a professor of medicine at Tulane University and an expert on allergies to shrimp and corn, is more emphatic: “The allergy tests are so extensive that most of our foodstuffs would never pass them,” he wrote.
![]()
When transferring any gene from a known allergen such as peanut, the FDA’s 1992 guidelines considered it “prudent” for the plant breeder “to assume that the transferred protein is the allergen.” The WHO-FAO report, written 10 years later, reinforces this suggestion: “It must be assumed that the novel gene product is allergenic unless proven otherwise.”
Between these two recommendations, an allergen was indeed moved from one food plant into another. The project began because Pioneer Hi-Bred International wanted to increase the nutritional content of its soy-based animal feeds. Soybeans lack certain amino acids
(methionine and cysteine), so supplements must be added to the feeds for the animals fed on them to grow well. Knowing that the 2S albumin of Brazil nuts was a protein with a very high methionine content, Pioneer’s scientists introduced a gene coding for the Brazil nut protein into soybeans.
At the time the 2S high-methionine albumin was not known to be an allergen. Studies done a few years previously in rats and mice had found it to be innocuous, although such studies could not give information about its allergenicity for humans. But because Brazil nuts were an allergenic food, Pioneer Hi-Bred asked a team of scientists—including allergy expert Steven Taylor—to test the new soybeans. When they were mixed with serum from people allergic to Brazil nuts, the IgE reaction was positive. Skin-prick tests were also positive. The 2S high-methionine albumin was a human allergen. Pioneer stopped development of the soybean, even though the product was intended for animals, not people. The soybean was never marketed.
Scientists, as well as consumers, have interpreted the results of this study in two ways. Marion Nestle, in a commentary that accompanied the team’s report in the New England Journal of Medicine, said: “This study highlights gaps in our current knowledge of food allergies.” For plant pathologist Jim Cook, on the other hand, “This is a perfect example of how the system works.” In his testimony to Congress in 1999 Cook said, “It is always cited as how things can go wrong, but it is exactly how good testing in the laboratory can provide for safety.”
Another example often cited as “how things go wrong” is the story of StarLink corn. Yet in this case there was no evidence—and still isn’t—that the added protein was an allergen. A company called Aventis S.A., headquartered in Lyon, France, received approval in 1997 and 1998 to commercialize a variety of corn that contained both an herbicide-resistance gene and a Bt toxin gene. The approval was given by the EPA, whose primary concern is how a new crop will affect the environment. StarLink’s application, for instance, had to explain how the new corn would affect honeybees, earthworms, minnows, mice, ducks, songbirds, and many other creatures, as well as other plants. Yet EPA also requires proof that any pesticide added to—or expressed in—a crop is safe to eat.
Bt-based pesticides have been used for more than 30 years to control a variety of insects, including gypsy moths. They are especially favored by organic farmers, who consider them natural, not synthetic. The toxins, which break down when exposed to sunlight, heat, or drying, come from a bacterium, Bacillis thuringiensis. “While commonly referred to in the singular as ‘Bt,’ B. thuringiensis is actually a large group of subspecies,” writes entomologist Brian Federici of the University of California, Riverside. More than 70 subspecies (also called varieties or strains) have been identified. Each produces one or more types of “Cry” (for crystal-like) proteins in its spores. These proteins are not toxic until they come into contact with an insect’s digestive juices, which are highly alkaline (with a pH reading of 8 to 10).
Eaten by an insect larva, the crystal dissolves. The protein is then broken apart, producing a toxic fragment. The fragment binds to a receptor on the lining of the insect’s gut. If the insect doesn’t have such a receptor—and most don’t—nothing happens. In insects that have these receptors, on the other hand, the cell immediately begins to swell until it bursts. The active toxin binds to proteins on the epithelial cells lining the insect’s midgut, forming pores that let potassium ions escape. Lacking potassium, the insect’s gut cells take up too much water. Within two hours the insect stops feeding; if it has eaten enough of the toxin it becomes paralyzed and soon dies. The bacterium in this way prepares its own habitat. Federici points out that while Bt is usually thought of as a “soil bacterium,” its true ecological niche is the dead insect. After the larva is dead, the bacterium feeds off its body, reproduces, and makes millions of spores.
The toxin has no effect on humans because of the differences between our digestive system and that of an insect. A human’s digestive juices are highly acidic (with a pH reading of 1 to 3), so the crystal is not dissolved and the toxic fragment is not released. Even if it were, it would still not be toxic to a human. Toxins work by interfering with, or changing, a normal part of a cell. Human cells lack the receptor that interacts with the Cry protein in the insect’s gut. For people, a Cry protein is just an extra bit of protein.
The genes that encode Cry proteins are on a large plasmid that B. thuringiensis strains carry. When the plasmid is lost—which can happen naturally or can be made to happen in the laboratory using heat—no
toxins are made. Without the plasmid a Bt bacterium is indistinguishable from another species, Bacillus cereus. As Federici explains, molecular, biochemical, and physiological studies all agree that Bt and B. cereus are actually the same species: “The latter becomes the former when it acquires one or more plasmids that express genes for insecticidal proteins.” Scientists retain the species name B. thuringiensis, though, for its practical use: it helps them classify the more than 20,000 isolates, individual Bt colonies that differ from each other in subtle ways.
More than 170 different Cry proteins have been identified in those 20,000 isolates; 16 of them have been approved by EPA for use in sprays and dusts or powders, of which 30 different formulations are sold. These are made by fermentation—growing bacteria in large vats and separating out the toxic spores, then adding stabilizing agents, spreaders, stickers, and fillers. To make sure they are safe to use, the EPA established a three-tier series of tests. First laboratory animals, including insects, birds, fish, and mammals, are exposed to a hundred times the amount of Bt spores recommended for use in a crop field as a pesticide. If there are any signs of toxicity, that Bt isolate is rejected. If the results are unclear, tier II tests are conducted, in which the animals are exposed multiple times. Tier III tests include two years of feeding trials. “To date,” writes Federici, “none of the registered bacterial insecticides based on Bt have had to undergo tier II testing.” As a result, no limit is set by the EPA for the amount of insecticide residue that can be left on a crop. Bt can be sprayed on tomatoes, for instance, just before they are picked and eaten—the EPA doesn’t even require the vegetables to be washed before they are sold. Writes Federici, “It is important to realize that such a statement cannot be made for just about any synthetic insecticide.”
Most of the insecticidal sprays and powders contain four Bt toxins to control a wider range of pests, because each Cry protein is toxic to only some kinds of insects. Some affect gypsy moths, mosquitoes, blackflies, cabbage loopers, and boll weevils. Others—particularly the ones named Cry1Ab, Cry1Ac, and Cry9C—kill the European corn borer, the number two insect enemy of corn farmers. In bad years the pest has cost farmers an estimated $1 billion, or about 20 bushels per acre.
Several varieties of corn have been genetically engineered to incor-
porate a Bt gene. Most produce Cry1Ab. StarLink corn was the only one to use Cry9C. In spite of the fact that Bt was a familiar pesticide whose action was well known and whose toxicity was well tested, the EPA did not simply assume that Cry1Ab and Cry9c, when introduced into corn, would not be toxic, nor that they were free of allergenic potential. Both proteins were put through tests. Cry1Ab was shown to be easily digested. It was inactivated by cooking. Even at relatively high doses (4,000 milligrams per kilogram), when fed to a laboratory animal it was not toxic. Corn containing Cry1Ab was approved for human use.
StarLink corn, however, was approved only for animal feed. Cry9C did not break down as quickly as Cry1Ab when it was mixed with stomach acid and digestive enzymes. Such resistance to digestion is one of the criteria used to identify potential allergens. This result didn’t mean that Cry9C is necessarily allergenic, only that it was suspect. The EPA required further testing before StarLink corn could be allowed in human foods. Aventis CropScience, the company’s American subsidiary, decided to commercialize its variety as animal feed first, then follow up with more testing and a later petition for it to be used in human foods.
Farmers who bought StarLink seeds were supposed to keep their corn segregated from other varieties by planting a 660-foot buffer strip of another crop between it and any other type of corn. They were also not to sell it for use in human food. Yet most field corn—the kind used for animal feed, as well as chips and taco shells or even gasohol—is stored by grain dealers in the same huge silo-like grain elevators. On a practical basis, the promise Aventis sought to extract was very hard for a farmer to keep. In the opinion of the attorney general of Iowa, it was “irresponsible” for Aventis to market its seed corn with such unrealistic restrictions. He did not believe that most Iowa growers even knew about the rules. An Aventis-sponsored survey of 230 farmers in December 1999, in fact, found 2 farmers who had knowingly sold their harvest for use in human food. Another 29 did not know what their crop had been used for. Aventis did—responsibly—report these results to the EPA, but no action was taken. A few months later a panel of scientists was convened to recommend a way to test StarLink further, in hopes of approving it for human consumption.
Before their suggestions could be acted upon, the Washington Post
reported that a company called Genetic ID had detected the presence of StarLink corn in Kraft Food’s Taco Bell taco shells. As Genetic ID explained, the environmental group Friends of the Earth had contracted with it to test the taco shells. The company claimed to detect the 1 percent StarLink corn in the taco shells using a PCR test that it has not disclosed. Genetic ID’s president, John Fagan, is the author of the book, Genetic Engineering: The Hazards, Vedic Engineering: The Solutions: Health—Agriculture—The Environment. He was one of 26 plaintiffs who sued the FDA in 1998 for inadequate testing of foods modified using biotechnology. Their suit was dismissed by the United States District Court for the District of Columbia on September 29, 2000, 11 days after the StarLink story broke.
The publicity triggered a massive recall of taco shells, corn chips, cornmeal, and other foods containing corn. Aventis offered to purchase the entire 2000 StarLink harvest and stopped all sales of StarLink seed corn. The USDA published a recommended method to distinguish StarLink from other corn, and grain elevators and seed companies started testing their stores. The tests—which were sensitive enough to find one StarLink kernel in a sample of 400 kernels—turned up much more StarLink DNA than expected. StarLink had been grown on only 0.5 percent of all the corn acreage in America in 2000. Yet corn varieties that were not supposed to contain the gene tested positive. Even products made from white corn—StarLink was a yellow variety—tested positive. Farmers in 17 states whose nontransgenic products tested positive threatened to sue. They were backed by their state attorneys general, who negotiated an agreement with Aventis to reimburse growers and grain elevator operators who had suffered losses. Aventis would pay 25 cents per bushel to farmers who grew StarLink, to those who grew other varieties of corn within 660 feet of a StarLink field, and to those whose corn tested positive for Cry9C. The company also agreed to cover losses from transportation, storage, and testing. Aventis expected the settlement would cost it at least $100 million. The top leadership of Aventis in the United States—president, general counsel, and vice president for market development—were fired. The U.S. Department of Agriculture offered another $15 to $20 million to compensate small seed companies whose stocks were contaminated
with Cry9C. By 2002 all StarLink corn was eliminated from the U.S. grain supply.
Meanwhile, calls began coming in to the FDA reporting allergic reactions to taco shells and corn chips. As these calls accumulated, the FDA requested that the Centers for Disease Control (CDC) test the people reporting such reactions to see if their blood serum contained the IgE sensitive to Cry9C, the purported culprit. The CDC determined that 28 of the people who had filed complaints had indeed experienced allergic reactions—but none of them had a form of IgE that would react to the Cry9C protein. They were allergic to something else. This result is not altogether surprising given what is known about allergic reactions. An IgE molecule that matches and binds to Cry9C can be made only after the body has been exposed to Cry9C—repeatedly, and in significant amounts. Because Cry9C had not been in foods before, allergy expert Steven Taylor was quoted as saying, “There is virtually no risk associated with the ingestion of StarLink corn in this situation.”
In July 2001, an EPA committee reviewed the StarLink case. In petitioning for approval for StarLink to be used in human food, Aventis had submitted data that showed that the Cry9C protein broke down more easily when it was wet or heated—both essential steps in making taco shells. The EPA’s scientists confirmed that, indeed, the normal process of preparing corn for that use, called wet milling, removed almost all of the Cry9C. The question of whether that small amount was harmful to health went unanswered, because, as the EPA had reported several months earlier, “Aventis requested voluntary cancellation of their Cry9C StarLink corn registration and this cancellation became effective on February 20, 2001.”
The tests sponsored by Friends of the Earth, and the subsequent publicity and recalls, did not prove that StarLink corn contained an allergen. They proved only that the attempt to separate corn for human food from corn for animal feed had failed. For agreeing to the EPA’s request not to sell StarLink for use in human food until more data were submitted, Aventis lost more than $100 million in cash and an uncalculated amount in farmer confidence and consumer goodwill. Bayer bought Aventis CropScience in 2001; the company is now called Bayer CropScience.
![]()
StarLink corn, the Brazil nut-soy case, and the poisoned rat debate have all received worldwide publicity. Each has been transformed and sensationalized with retelling. StarLink corn and the Brazil nut protein-containing soybean have been credited with causing allergic reactions. Genetically modified potatoes have been accused of poisoning rats. But the facts don’t support the stories. StarLink caused no allergic reactions, the soybean didn’t reach the market, and the new genes in the potatoes were never shown to have caused harm. However titillating, might this sensationalism itself not be the poison?
What is rarely mentioned in debates about genetically modified food is how it has made our food safer.
Entomologist Brian Federici writes, for instance, “One notable improvement in safety that has not received much attention is that Bt maize grain contains lower levels of certain fungal toxins known as fumonisins. These toxins can cause illness and death in horses and pigs, and have been implicated in certain forms of liver and esophageal cancer in humans.” When the European corn borer tunnels into an ear of corn, it opens a passage for Fusarium fungus. The fungus is not easy to see, and infected ears are harvested along with their undamaged neighbors. The toxins have been found in corn around the world. They are especially a problem in countries with warm or subtropical climates. A study in 2000 found that 82 percent of the corn grown in the islands of the central and southern Pacific, 77 percent of the corn in Africa, and 63 percent of the corn in North and South America was contaminated with fumonisins. In corn meant for use as human food, the levels of toxins ranged from 1 milligram per kilogram up to 150 milligrams per kilogram. Horses die of brain damage when they are fed infected corn containing more than 10 milligrams of toxin per kilogram of corn. Rats fed corn tainted with 150 milligrams of toxin per kilogram suffer liver and kidney cancer.
One way to stop the toxins from accumulating in the corn is to limit the holes through which the fungus can get into the plant to multiply. By killing the European corn borer as soon as it starts eating, StarLink corn and the other varieties engineered to produce Bt toxins do just that. Comparisons of Bt corn and conventional varieties in Iowa
found that the Bt corn contained 93 percent less of the toxic fumonisins. Similar results have been reported in France, Italy, and Spain.
Bob Buchanan, a plant pathologist at the University of California, Berkeley, happened onto another way in which genetic engineering can make food safer. Thirty years ago he began studying a small protein called thioredoxin. First discovered in bacteria, it was found to play a part in regulating photosynthesis. Buchanan’s team discovered that thioredoxin somehow alleviates food allergies, as well as enhancing the digestibility, nutritional quality, and baking quality of flour. Working with Buchanan are allergy specialist Oscar Frick of the University of California, San Francisco, and geneticist Peggy Lemaux of U.C. Berkeley. As Buchanan said in 1999, “We seek to gain an understanding as to how thioredoxin from our laboratory, when engineered into cereals by Dr. Lemaux’s group, could benefit humans. In addition to barley and wheat, our agenda includes rice, corn, sorghum, soybean, and peanuts.”
Other researchers are addressing the allergy issue a different way. Samuel Lehrer at Tulane University is identifying the parts of a protein molecule that allow the immune system’s gatekeeper, IgE, to bind to it and set off the cascade of symptoms we call an allergic reaction. “If you can substitute one amino acid in the protein,” he explains, “you can completely abolish IgE binding.” Lehrer’s group is working with shrimp, while another team at the University of Arkansas and Arkansas Children’s Hospital in Little Rock is concentrating on soybeans and peanuts. “The group in Little Rock are farther along,” he reported at the American Association for the Advancement of Science’s annual conference in 2003. “They can almost completely abolish IgE binding. There are still serious issues to address, though,” he added. “We need to demonstrate that these altered proteins don’t contain new binding sites.” Lehrer concluded, “It is our hope that in the not-too-distant future foods can be developed that will substantially reduce the number and severity of food-induced allergic reactions.”
Eliot Herman of the Donald Danforth Plant Science Center in St. Louis has also been trying to breed a less allergenic variety of soybean. He has targeted an enzyme called P34, which is the major allergen in soybeans. In the plant’s leaves and stems it seems to help fight off diseases. Unfortunately for people, it accumulates in the beans. Herman’s
modified soybeans, now in field trials, completely lack P34. The gene that directs the production of the protein in the seed has been silenced. Nothing else about the plant has been changed. Herman writes, “These results show that it is feasible to use biotechnology to suppress a major human allergen in crops, implying that widespread use of this approach can improve the safety of food for sensitive people.”