Mendel in the Kitchen: A Scientist's View of Genetically Modified Foods (2004)
Chapter: 1 Against the Ways of Nature
1
AGAINST THE WAYS OF NATURE
One of the most remarkable features in our domesticated races is that we see in them adaptation, not indeed to the animal’s or plant’s own good, but to man’s use or fancy.
—Charles Darwin (1859)
Golden Rice is a rice rich in beta carotene, the substance that gives carrots their color. Its creator, Swiss scientist Ingo Potrykus, wasn’t trying for a colorful garden curiosity like the Iceberg blackberry, a paradoxically white blackberry created in the 1890s by plant breeder Luther Burbank (better known for creating the Idaho potato and Shasta daisy). Nor was Potrykus trying to complement the contemporary dinner-plate aesthetics of the All Red potato, bred by Robert Lobitz in 1984, or the Graffiti cauliflower, introduced to American seed catalogs from Europe in 2002. The All Red potato is prized by chefs for retaining its cranberry color even when cooked; it makes a striking potato salad. The Graffiti cauliflower’s deep blue-purple hue is an “outrageous bid for attention, the vegetable equivalent of a Versace neckline,” wrote one food critic. It was one of many new vegetables of 2002 “designed to stir excitement and desire in the hearts of consumers.”
Potrykus was not even trying to improve upon an already colorful “sport,” as breeders name those interesting mutant varieties that crop up from time to time; he was not like Richard Hensz, the Texas A&M University researcher who in the 1960s made pink grapefruit more red,
producing the bright and attractive Star Ruby and Rio Red. Potrykus was not trying to create the culinary delight of saffron rice on the stalk.
The parchment-yellow hue of Golden Rice—which, unlike the other colorful crop varieties mentioned, is not yet on the market—was merely a side effect. The color signals the rice’s beta-carotene content. Beta carotene is a precursor of vitamin A. Yellow rice, Potrykus knew, could help the hundred million children who risk blindness and death from vitamin A deficiency each year in countries where rice is the staple food.
There are 22 species of rice. Two are cultivated. The most popular, Oryza sativa, is grown in more than a hundred countries, although most of it is eaten in Asia. There are more than 80,000 varieties of Oryza sativa. Some cultures prefer a sticky rice, some like it drier. Some want slender grains, some round. Some want an aromatic rice, some favor rice with a red or purple hue, which, if not more nutritious (as they believe), at least brings good luck (red, in Asia, is a lucky color). Rice is the staple food for nearly half the world’s people. It provides more than half—sometimes almost all—of the calories eaten by people in Bangladesh, Cambodia, Laos, Myanmar, Thailand, Vietnam, and Indonesia. In those countries, infants being weaned eat very little else.
Yet a grain of white rice, polished the way most people like it, lacks crucial vitamins, among them vitamin A. The first vitamin to be named, vitamin A was discovered in 1915. The livers of animals and fish are a good source but so are green and yellow plants because the body can turn beta carotene into vitamin A. Early signs of vitamin A deficiency include difficulty seeing in dim light. The eyes become unusually dry. And yet by the time the eye’s cells are damaged, many other bodily systems are in distress; someone suffering from vitamin A deficiency often succumbs to intestinal or respiratory disease. Each year as many as 230 million children are at risk of going blind from vitamin A deficiency. The World Health Organization estimates that more than a million children a year die for lack of vitamin A in their diets. To the Rockefeller Foundation, a philanthropy devoted to “applying science to benefit mankind,” creating rice that contains vitamin A seemed a sensible way to help those children.
The idea first surfaced in 1984. At a meeting of rice breeders at the
International Rice Research Institute in the Philippines, Gary Toenniessen of the Rockefeller Foundation had asked, “What gene would you put into rice, if you could put in any gene at all?” The breeders were skeptical that they could put new genes into rice. Genetic engineering was quite a success in medicine—human insulin to treat diabetes was by then being produced almost exclusively in genetically engineered bacteria—but it was hardly routine in plants. The first practical technique had been published only the year before.
But Toenniessen, a microbiologist whose earlier work had centered on integrated pest management and biodegradable alternatives to pesticides, pushed the issue. “Finally, Peter Jennings said ‘yellow endosperm,’” Toenniessen said, retelling the story 20 years later. “Peter was one of the creators of IR8, the rice that started the Green Revolution,” he continued. “He was then working in Latin America. ‘As long as I’ve been a rice breeder,’ Peter said, ‘over 20 years, I’ve been looking for a rice with yellow endosperm, because then it would produce vitamin A.’” If he had found such a rice, with beta carotene tinting the edible part of its kernels, he could have enriched commercial varieties through crossbreeding. But he had not found one. Rice, like all green plants, has beta carotene in its leaves, where the yellow pigment helps in photosynthesis; but none of it accumulates in the grain, unlike in maize (or corn), with its yellow kernels.
“The more I thought about that,” Toenniessen said, “the more it struck me as doable. If Nature has figured out how to do it, then we can figure out how to do it. And Nature does it. Maize produces beta carotene in the endosperm with no deleterious impacts on other parts of the plant.” And maize and rice, both grasses, are quite similar in many ways.
Toenniessen funded a study at Iowa State University to clone the maize genes needed for beta carotene to form in the kernels. He funded a project at the University of Liverpool to analyze rice endosperm to see if the precursors to beta carotene were there. Learning that rice grains do contain the genes for beta-carotene production, he funded work at the City University of New York to see if those silent genes could be turned on. And he organized a workshop, with “everyone in the world” who knew about beta-carotene biochemistry and genetics,
to which he invited several “gene jockeys,” as he calls them, who were willing to work on rice. At the workshop, Ingo Potrykus met Peter Beyer, an expert on beta carotene in daffodils. They put their heads together. In 1990 Toenniessen offered funding, which grew over 10 years to $2 million. In 1999 Potrykus and Beyer patented Golden Rice.
Those first grains were merely a “proof of concept,” the inventors said. More tinkering would be needed before Golden Rice would be of any use in the effort to combat vitamin A deficiency. The prototype rice would be tested to see if it contained allergens, such as the one associated with “daffodil pickers’ rash.” The antibiotic-resistance marker gene the first grains carried would be taken out: it was of use in the laboratory, but not in the field or final product. The amount of beta carotene would be increased, perhaps by using a different promoter to express the genes. And the new genes would be bred into easily grown strains of rice, which would then be thoroughly field-tested. Still, it seemed feasible that Golden Rice could eventually provide 20 to 50 percent of a child’s daily requirement of vitamin A, enough to make up for the typical deficiency and to prevent a child who ate it every day from going blind. According to one collaborator, “We are aiming the benefits of Golden Rice at the poorest of the poor who cannot get anything other than rice, green chilies, and salt, if at all.”
![]()
Ingo Potrykus is typical of many modern plant scientists around the world. His story—the story of Golden Rice—could belong to almost any one of them. He was not a plant breeder like Luther Burbank, working the soil, although greenhouses and growlights were a vital part of his lab. He was a geneticist whose research subject was not the fruit fly, the mouse, or the worm C. elegans; not bacteria or bacteriophages or molds, but plants.
Potrykus began his study of genes in the early 1970s, when he tried to turn a white petunia pink. He wanted to see if a gene could be taken from one plant and inserted into the cells of another, as was then being done routinely in bacteria. His results, in 1972, were baffling and never published. By then enough failures had accumulated that most scientists agreed plants could not exchange genes the way bacteria did. The
only way plant genes would recombine, they concluded, was through pollination: plant sex. Not for several more years would studies of the plant pest Agrobacterium tumefaciens suggest a very different approach to gene transfer, one not so much like plant sex as bacterial sex.
Yet Potrykus persevered, even after the Agrobacterium method was announced in 1983. Twelve years after he had first tried to transfer genes between plant cells, he succeeded, this time using tobacco, which, like petunia, was a favorite plant for such studies. (It has since been eclipsed by a little weed in the mustard family, Arabidopsis thaliana or mouse-ear cress.) In 1984 Potrykus’s lab, then in Basel, Switzerland, published “an ironclad demonstration” and “the first incontrovertible evidence,” according to Paul Lurquin, an American scientist who reviewed the work, that plant cells could take up and incorporate foreign genes directly. Potrykus’s proof, coming within a year of the first report of gene transfer using Agrobacterium, finally answered the question raised in 1967: could genes be introduced into plants? It was, said Lurquin, a “turning point announcing an era of applications rather than one of basic discovery.”
Potrykus decided the application he would pursue was to help developing countries grow more food. Together with a colleague, he set up a new Institute of Plant Sciences in Zurich. Even in the early 1970s, he wrote, there had been “claims (from those working with tobacco and petunia) that the new techniques would contribute to food security in developing countries. Obviously, to contribute, one would have to work with important crop plants and not only talk about them.” He began projects to increase the yields of wheat, corn, barley, and rye by making them more resistant to disease. Two Indian scientists, Karabi Datta and her husband, Swapan Datta, drawn to the new lab, persuaded Potrykus to add rice to his list of experimental crops and to shift his emphasis away from yield alone. Rice, Swapan Datta argued, is food for the poor. When improving it, nutrition should be considered, not just yield, which had been the main objective of the breeding that produced the Green Revolution varieties of the 1960s.
From 1990 to 1999, with funding from the Rockefeller Foundation, Potrykus and his colleagues worked to transfer into rice the genes that give daffodils their brilliant yellow color—the genes needed to
make beta carotene. It was not a simple task. Until then only one or two new genes at a time had been added to a plant. To make beta carotene, four genes had to be added and their protein products had to interact. It proved more difficult than anyone had anticipated. Eventually even Peter Beyer gave up hope—although he didn’t let Potrykus know. “This exemplifies the advantage of my ignorance and naivete,” Potrykus wrote later. “With my simple engineering mind I was throughout optimistic and, therefore, carried the project through. I was naive enough to believe in its success.” But it took nearly 10 years and the concentrated work of some 60 scientists from Germany, Switzerland, Poland, India, Japan, and China before Potrykus saw the first golden grains—at a symposium celebrating his mandatory retirement from the lab when he reached the official Swiss retirement age of 65.
The timing seemed perfect. In France and Ireland, protesters had vandalized seed stores and test plots of what had come to be called genetically modified organisms (GMOs) or genetically modified (GM) food. Austria, Luxembourg, and Norway had banned the planting of GM corn. Food stores in Britain had pronounced themselves GM-free, and fast-food outlets soon followed suit. Britain’s Prince Charles had accused scientists of interfering with God’s plan, warning of a “manmade disaster,” “unforeseen consequences,” and threats to biodiversity. In the U.S., where almost half of the soybeans and cotton and 25 percent of the corn planted in 1998 was genetically modified for insect or herbicide resistance, a lawyer, twelve religious leaders, and nine university scientists had brought suit against the U.S. Food and Drug Administration (FDA) for its “negligent oversight” and “unethical” handling of genetically modified foods.
But Golden Rice, Potrykus thought, was GM with a difference. Publicly funded, it would be given away free to subsistence farmers; multinational corporations would make no profit from the poor. The farmers could save the seeds from their crops, and those seeds would breed true the next season. The rice would provide better nutrition—but otherwise the plant was unchanged. It didn’t require more fertilizer or pesticide. “To grow this rice does not require any additional input,” Potrykus has said. “All a farmer needs to benefit from the technology is one seed.” Because the trait could be bred into any of the
thousands of varieties of Oryza sativa, it need not restrict the rice gene pool. In fact, it would be fairer to speak of golden rices, not one Golden Rice.
Golden Rice would change the international debate. With pride, Potrykus and Beyer applied for a patent, licensed it to Zeneca Agrochemicals, then sent a paper off to the prestigious British journal Nature. The journal rejected it. Potrykus persevered. He garnered letters of support from famous European scientists. Nature declined even to send the paper out for review.
Word of Golden Rice reached Peter Raven, head of the Missouri Botanical Garden and organizer of the International Botanical Congress planned for St. Louis in August 1999. Raven invited Potrykus to speak at the meeting, held a press conference, and encouraged the American journal Science, equal in prestige to Nature, to read the manuscript. Science accepted it, printing it in January 2000 with a glowing commentary. It was a “tour de force.” It “exemplifies the best that agricultural biotechnology has to offer,” the reviewer said. By July Potrykus’s face (looking puzzled but determined, his chin beard and brush mustache mostly white) had made the cover of Time magazine. “This rice could save a million kids a year,” the banner headline read. The industry-sponsored Council for Biotechnology Information (whose slogan was “Good ideas are growing”) produced a new ad—“Biotechnology researchers call it ‘golden rice.’ For the color. For the opportunity.”—and began running it in such venues as the New York Times and the New Yorker magazine.
The backlash was fierce. Potrykus had created a “Frankenfood,” a source of “genetic pollution.” He had sold out to the multinational corporations. (Zeneca Agrochemicals had by then merged with rival Novartis Agribusiness to become Syngenta, “the world’s leading agribusiness.”) Golden Rice was “fool’s gold,” “an intentional deception,” a “gift horse”—even a Trojan horse—for the developing world. It was “a useless application” and “an exercise in how not to do science,” wrote Mae-Wan Ho, a founder of the Institute of Science in Society and scientific advisor to a nongovernmental organization called Third World Network. Golden Rice was “a rip-off of the public trust,” as well as a “major obstruction” to work that would “provide the real solutions to world hunger.” It was “worse than telling them to eat cake.”
As the vitriolic attacks continued, Potrykus began to wonder how he could have been so naive as to think that he could use science to solve a social problem. A press release from the Rural Advancement Foundation International, a “civil society organization” based in Canada, accused him of having “surrendered a decade of public funding to the commercial and PR interests of the biotech industry.” In response, Potrykus plaintively wrote: “That I tried to use the funds for both competitive science and contribution to food security in developing countries was my personal and free decision. I could have used the same funds for studying why the hairs on the leaves of the small weed Arabidopsis thaliana are sometimes two- and sometimes three-forked.” If he had done so, he mused, perhaps no one would have complained.
![]()
The term “Frankenfoods,” with its power to call up the mad scientist Frankenstein and his unnatural monster, was first applied to genetically modified food by a Boston College English professor in a letter to the New York Times in 1992. Frankenfoods has not clarified the debate or contributed to, in Potrykus’s words, “any discussion of a risk/benefit analysis.” Just the opposite. “The Frankenstein myth,” writes critic Jon Turney in Frankenstein’s Footsteps: Science, Genetics, and Popular Culture, “hampers the necessary task of agreeing how to control the new technological powers now being developed in the laboratories.
“It invites an all-or-nothing response to a whole complex of developments, when we should be insisting on our right to choose some, and block others. When we do so, it should be for reasons which we can articulate more clearly than saying either that there are some things humans are not meant to know, or that we should not tamper with nature.”
How did Luther Burbank create a white blackberry? How did Richard Hensz create the grapefruit called Rio Red? Few people know. The history of food plants is not a popular subject. Few people recognize the art and artifice in plant breeding—that each new variety is a product of the human mind and hand. It is hardly common knowl-
edge that farming and science have been intertwined for 200 years, and that well before then, more than 10,000 years ago, the way humans procured their food became distinctly unnatural. Yet to think clearly about Golden Rice, and about genetically modified foods in general, we need to know these things. We need to know that people have been genetically modifying plants for many thousands of years. We need to understand the science behind these genetic changes, and the meanings people give to the terms they use to describe them.
We need to be able to compare, for instance, the risks and benefits of Golden Rice—which brings together genes from two plants belonging to two unrelated species, rice and daffodil—with those of triti-cale—a cross between two unrelated species, wheat and rye. Each was produced in the laboratory: Golden Rice by molecular techniques, triticale through chemical ones. Triticale, says a botany textbook, is the “first successful attempt to synthesize a new crop species from intergeneric hybridization”—meaning that it is the first artificial creation of a hybrid between two plants not only not in the same species, but not in the same genus. Yet triticale flour is available in the natural-foods section of the grocery store. Its label touts its nutritional goodness. Golden Rice, on the other hand, is condemned as Frankenfood and genetic pollution.
We have all eaten a red grapefruit. Who knows—or cares—that the most popular red variety, the Rio Red, was created by exposing grapefruit buds to thermal neutron radiation at Brookhaven National Laboratory in 1968? Nor do we recall that the original pink grapefruit was a mutant that appeared as a sport on the limb of one grapefruit tree in a Florida orchard in 1907 and has been cloned countless times since then to make all the pink grapefruits in the world.
Proponents of genetically modified foods often argue that the molecular techniques lumped under the term genetic engineering or genetic modification are simply an extension of conventional plant breeding. According to Klaus Ammann, curator of the botanical garden at the University of Bern in Switzerland, “The most significant changes in grains and advances in knowledge about crop genomes occurred many years ago. When we eat wheat, we consume varieties mutated by nuclear radiation. It is not known what happened with the
genomes, but we have been eating this wheat for decades, without any type of problem. Today, with more extensive knowledge and new applications of the technologies resulting from genetic engineering, we are faced with a new system where control is greater, more precise, and less risky than that of the old systems.”
Opponents counter that genetically modified food is unlike anything that has been produced before. Dennis J. Kucinich, a U.S. representative from Ohio, argued in a recent video production that genetic engineering “is not the same as conventional growth of food. It’s a manmade process. It has nothing to do with the ways of nature. It’s very violent.” Which view will seem right to you depends on what you consider conventional, and on how you define the ways of nature.
By 1860, for instance, German botanist Julius von Sachs was growing crops hydroponically—in water, not soil. Though his technique was “much disputed at first,” he successfully grew garden beans, field beans, corn, and buckwheat in water, “in a sunny window or in a suitable greenhouse,” produced fertile seeds, and planted and grew the offspring. He set a corn kernel in a box “filled with well-washed damp sawdust” until it germinated and sprouted. “After the seedling has been carefully taken out and washed, it is fastened into a perforated cork … so that only the root dips into the water.” To the water he added various chemicals containing nitrogen, iron, potassium, calcium, magnesium, phosphorus, and sulfur. It was the beginning, he wrote, of a “complete revolution” in agriculture. “We are now able to rear plants artificially,” he reflected in 1887. From “inconspicuous and often scarcely ponderable quantities of vegetable substance,” scientists could produce quantities of food “as large as we choose.”
According to Harvard biologist John Torrey, Sachs’s work was the beginning of plant biotechnology. The next step was straightforward. Once plants had been freed of soil, he writes, “it was natural to ask whether it was possible to grow plant parts independent of each other.” In fact, in 1902, German botanist Gottlieb Haberlandt theorized that each and every plant cell contained all the necessary instructions to grow a complete plant. It should be possible to grow a plant not only from a seed but from a bit of leaf or a root tip. Plant scientists tried growing the tips of tomato and pea roots in all sorts of solutions, in-
cluding yeast extract and beef broth. Eventually they found that the roots required tiny amounts of certain vitamins, in addition to sugar (ordinarily made by the plants themselves through photosynthesis) and the minerals Sachs and later experimenters had identified. These would keep a root tip alive and growing—but only as a root.
To get a shoot and leaves required plant hormones. Charles Darwin and his son Francis, in 1881, had noted an “influence” that caused seedlings to bend toward the light. In 1926 the Dutch plant physiologist Frits W. Went proved it to be one of a class of plant hormones he named auxins (from the Greek, “to increase”), because he found that the hormone controlled how cells lengthen. French scientist Roger Gauthieret discovered that auxins could keep plant tissues growing indefinitely. In the mid-1930s he took chunks of carrot and cultured them in a solution of sugar, minerals, and vitamins. When he added auxins, an unorganized mass of cells grew at the cut ends of the chunk. He called this mass “callus” tissue because it resembled the calluses that grew around wounds on tree trunks. Plucked off and set in a separate petri dish with the standard nutrients, the callus continued growing—in this lumpy, undifferentiated state—as long as it was supplied with auxin. One of Gauthieret’s carrot cultures survived for 40 years. But it was still just a mass of cells, not a plant. Scientists kept searching for the substance that would turn a callus into roots and shoots, leaves and fruit.
In the 1950s F. C. Steward at Cornell University was growing tiny bits of carrot root in a solution containing coconut milk. The mass of carrot cells, just like Gauthieret’s, grew and divided. When Steward swirled the flask, some cells broke free from the mass. These grew and divided and occasionally formed roots. If he transferred them to a solid agar gel, some of them developed shoots. If set in earth the roots and shoots grew into plants that flowered and set seed. The hormone involved in this case was identified in 1954 by Carlos O. Miller, a postdoctoral researcher at the University of Wisconsin, when, in desperation, after trying everything else, he squirted drops from an old bottle marked “Herring Sperm DNA” onto his culture medium, and noticed that it caused his tobacco cells to divide.
Miller and his mentor, Folke Skoog, had, like Steward, originally
tried coconut milk, but they could not purify the growth-promoting factor from it. Knowing that a similar factor existed in herring sperm DNA (but only in old herring sperm—new samples had no effect on the tobacco cells) gave Skoog and Miller the hint they needed to isolate the hormone. They named it kinetin and called the family the cytokinins, because of their role in cytokinesis, or cell division. Cytokinins are derivatives of nucleotides, the building blocks of DNA; the herring sperm DNA worked because it was so old that much of it had broken down into its basic parts. Skoog and Miller showed how the two plant hormones, the auxins and the cytokinins, governed the transformation of a callus into a plant. When the two hormones were in balance, the clump of cells remained a callus. With less cytokinin, roots grew; with more cytokinin, buds and shoots formed.
These new insights into how plant cells grow and differentiate were brought into farming by French botanist Georges Morel. In the late 1950s Morel was trying to rid potatoes of viruses that stunted the plants and caused deformed leaves. Even in heavily infected plants, he found, the very tip of the growing shoot, the meristem, was free of disease. Morel snipped off the meristem (eventually other researchers would show that a sliver thinner than a fingernail was all that was needed) and cultured it. The cells multiplied and, when he let them grow on a solid medium with the right balance of hormones, they turned into thousands of genetically identical plants—clones of the original, but now virus-free. The technique came to be known as tissue culture cloning.
One of Morel’s graduate students, Walter Bertsch, “happened to be an orchid fancier and also happened to be dating a girl who worked at a famous French orchid company,” writes Susan Orlean in her book The Orchid Thief. “This is how orchids came to be the first ornamental plant to be cloned on a large scale.” Orchids had been prized not only for their rare beauty, but also for the skill it took to grow them. “With cloning, they could be almost as common as daisies.” The orchids now routinely sold in grocery stores and discount outlets are all clones produced through tissue culture.
Scientists in Japan learned how to use these techniques to “rescue” rice embryos from hybrid seeds that ordinarily would not germinate.
Soon they were making wide crosses between rice and wild relatives with which the cultivated varieties normally did not interbreed. Another type of tissue culture, the one that Peter Raven of the Missouri Botanical Garden has called “the most elegant,” is protoplast culture. In 1960 English scientist E. C. Cocking used enzymes from bacteria and fungi to strip the sturdy wall off a cell from a tomato’s root tip. In 1968 I. Takebe of Japan, using a tobacco cell, simplified the procedure. Seen through a microscope, these naked protoplasts (named for the Greek for “first,” or “fundamental”) are round green globes, not the squarish boxes of cells inside a plant. Their now-flexible membranes can be disrupted briefly to slip in a molecule, a virus, or even part of a cell, like the DNA-rich nucleus. When cultured under the right conditions, protoplasts regenerate their cell walls and divide, eventually growing into plants, some with new genetic characteristics.
To make new hybrids, scientists began trying to fuse cells of two different plants. In 1978 Georg Melchers of Germany succeeded in making a Tomoffel or Karmate, from the German names of tomato and potato. The half-tomato, half-potato protoplasts grew into plants that flowered but didn’t set seed. The experiment would not have been surprising to Luther Burbank, who noted in 1893 that “tomatoes may be grown from seed pollinated from potato pollen only” and claimed in his catalogs to have thousands of tomato-potato hybrids growing in his plots. Nor has the effort to make a Tomoffel been abandoned. Laboratory specimens were grown to maturity in 1995 and the fertile offspring backcrossed to the parent plants. Crosses between a tomato and a wild eggplant have also been successful.
How does protoplast fusion work? T. Kinoshita and K. Mori of Hokkaido University in Japan reported in 2002 on their efforts to cross cultivated rice, Oryza sativa, with its close relatives in the genus Oryza in order to bring in “important genes for sustainable agriculture, such as resistance to brown planthopper, bacterial leaf blight, and grassy stunt virus.” First they treated the protoplasts they had taken from rice seeds with certain chemicals, then chilled them at 4°C, a temperature just above freezing. This kept the cells from dividing. Then they positioned a wild protoplast next to a cultivated one in a test tube and applied an electrical current. The membranes of the different proto-
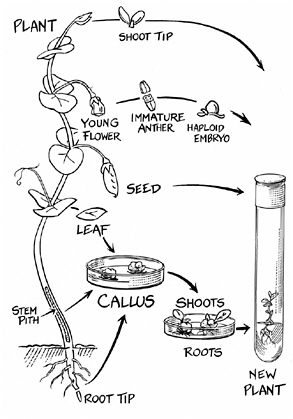
How to regenerate a plant
plasts fused. They exposed the fused cells to gamma radiation, after which they followed standard tissue culture protocols to grow some of the fused cells into mature plants.
Plants have also been grown from immature anthers—the part of a flower that makes pollen. A group of scientists from Hangzhou, China, reported in 2002 that anther culture was an effective way to improve rice cultivars. Their method involved clipping off the flowering tip, or boot, of the rice plant in the latter stages of pollen development. They wrapped the rice boots in aluminum foil and chilled them at 7°C for five days, then disinfected them with successive treatments of alcohol, Clorox, and distilled water. They placed 50 anthers in each of 10 test tubes containing some jelled growth medium, and incubated them at room temperature in the dark for 30 days. Then they irradiated the test tubes with gamma rays. With the gamma-ray treatment
they got almost 500 green plants for every 100 anthers, 300 times the number that grew without irradiation. The bits of callus tissue that began to grow were then transferred into larger tubes and returned to the warm room, where they were grown under constant light for 40 days. The green plantlets that formed were fed nutrients and allowed to grow until their shoot tips reached the tops of the tubes, then their tubes were opened and they were moved into the greenhouse. A week later, they were transplanted into the field.
An interesting puzzle about plants grown from cultured tissues is that they are not all the same. Depending on the type of plant, its age, where the tissue was taken from (root tip, leaf, anther, elsewhere), the balance of hormones and nutrients in the test tube, and the length of time in culture, various types of mutations commonly arise even without the use of radiation or mutagenic chemicals. These mutations, which turn out to be of all the different kinds known to occur, are lumped under the term somaclonal variation (soma is Greek for “body” or “tissue”; a clone is a copy). As one scientist notes, “Uniformity among the plantlets regenerated from callus tissue is considered to be the exception rather than the rule.”
Most of these mutations harm the plant. Plants with such harmful mutations are discarded. But occasionally a good mutation appears, and some have given rise to commercial crops. Clearfield corn, for instance, sold in the U.S. since the early 1990s, is a variety of maize resistant to Patriot, an imidazolinone herbicide. Another variety of maize to come from somaclonal variation tolerates glyphosate, the generic name of Monsanto’s herbicide Roundup. Farmers who plant herbicide-tolerant varieties can spray their fields to kill weeds after the corn has sprouted without killing off the young crop. This practice is popularly thought to be limited to Roundup Ready crops that are genetically modified by molecular techniques. It isn’t.
How natural are these plants? Are tissue culture-derived mutants more natural than plants to which an herbicide-resistance gene has been added? The answer, again, depends on the meaning of the word “natural.” What is quite clear is that all of the techniques of tissue culture just described are considered conventional. Mutant plants arising by somaclonal variation in tissue culture are not given the GM label.
![]()
The use of radiation and chemicals is not limited to encouraging plants to grow from pollen cells in test tubes. At about the same time that Gottlieb Haberlandt first suggested that every plant cell could become a plant, Hugo De Vries, author of the influential book The Mutation Theory, proposed that X-rays could be used to mutate plants for study. In 1927 Hermann Joseph Muller proved that irradiating plants did cause mutations, and in 1928 Louis Stadler published the first paper on the effects of X-rays and radium on plant seeds. Soon other types of radiation, including gamma rays, fast neutrons, and thermal neutrons, were being used to cause mutations in plants. Chemicals were also used to induce new mutations, as well as to allow hybridization of varieties that would not naturally crossbreed.
The chemical colchicine, for instance, was isolated from the autumn crocus or meadow saffron in 1936 and found to be a pesticide. The news that it also doubled a plant’s chromosomes “became the start of a real colchicine craze,” according to one plant breeder. “Within a few years the chromosome doubling technique had been applied to at least 50 plant species.” Applied to a sterile hybrid of rye and durum wheat in the 1950s, colchicine allowed F. G. O’Mara of Iowa State University to create the new, fertile grain triticale, which by the mid-1980s was being grown on more than two million acres in the Soviet Union, Europe (particularly France), the United States, Canada, and South America. The label for triticale flour from Bob’s Red Mill Natural Foods in Milwaukie, Oregon, reads: “Triticale is a hybrid grain—a cross between wheat and rye. It averages 28 percent higher protein than wheat and contains all the essential amino acids, thus making it a more complete protein than the parent grains. It has an interesting nutty flavor and is high in fiber…. Since Triticale Flour has inherited the best qualities of its parental grains—wheat and rye—and comes with a delicious flavor all its own, it needs to be discovered. Right now … by you …” The label makes no mention of the fact that wheat and rye could not naturally hybridize.
Colchicine is also used to make fruits seedless. A favorite fruit produced this way is the seedless watermelon. Colchicine interrupts cell
division, so that the cell makes extra copies of its chromosomes, and thus of the genes on those chromosomes, but fails to divide as it normally should. As a result, the seeds don’t develop and the watermelon is much easier to eat.
Other notable successes of mutation breeding include the most popular variety of wheat used for making pasta in Italy. The durum wheat called Creso is a cross between two earlier types, each created by exposing seeds to neutrons or X-rays. It has “high and stable yield capacity, good adaptability and grain quality.” A third of the area planted in durum wheat in Italy in the 1990s was planted in Creso.
Golden Promise barley, created in 1956 by exposing an earlier variety to gamma rays, “became a standard for malt quality and is still grown today under contract for specialist distillers” of fine beers, including some organic beers. B. P. Forster of the Scottish Crop Research Institute notes that Golden Promise was on the recommended list of cereals for England in 1967 and for Scotland in 1968. “It dominated Scottish barley acreage,” Forster writes, “from the 1970s to the mid-1980s,” even though it was susceptible to powdery mildew and needed to be sprayed with fungicides. Writes Forster, “The introduction of Golden Promise to Scotland revolutionised cereal growing to the benefit of the farmer, maltster, brewer, and distiller (fungicide manufacturers also profited), and enabled Scotland to come to the fore as a producer of malting barley.” Although the mildew problem eventually reduced its popularity, Golden Promise became the breeding stock for 15 other successful cultivars and might yet make a comeback in other countries: Forster recently discovered that it is highly salt-tolerant.
Some California rice is also a product of mutation breeding. A popular variety called Calrose 76, released in 1976, was created by treating seeds with gamma rays from a cobalt-60 source. In the early 1980s the high-yielding Calrose 76 was earning California rice growers an extra $20 million per year.
And, as Swiss botanist Klaus Ammann hinted, of the hundreds of varieties of bread wheat grown around the world almost 200 were created using X-rays, gamma rays, neutrons, or various chemicals to cause mutations. The latest variety, a hard red winter wheat called Above, was mutated so that it would tolerate an herbicide produced by the
BASF corporation. Just like Clearfield corn, Above wheat can be sprayed with herbicide and will not die, letting farmers weed their fields chemically after the crop sprouts. Yet, although the end result is the same as the Roundup Ready crops sold by Monsanto, Above is not considered genetically modified.
Several new crops, including a tasty little lettuce called Icecube and a number of varieties of barley used for making fine beers, were created by exposing seeds or plant parts to the chemical ethyl methane-sulphonate. This chemical is known to cause genetic mutations in people as well, including some that cause cancer. Plant breeders who use it must wear coveralls and breathe through a respirator.
And yet, other than general safety recommendations for laboratory personnel, there is little government oversight of this type of plant breeding, whether for food crops or for ornamentals. When Susan Orlean, chronicling Florida’s orchid industry, visited one backyard breeder, she reported: “One bench was stacked high with small plastic pots. The plants inside them were withered and droopy, a jumbled mess. Mike nodded toward it and said, ‘Failed Antherium project. A species called Elaine. It was created by irradiation. We took the germinating material and radiated it. We hoped to get some interesting mutations, but it didn’t work out that way.’ I asked him what they were going to do with all the loser Elaines. ‘Take all ten thousand of them and toss them in the Dumpster,’ he said.” Less than 1 percent of all observed mutations created through mutation breeding are of value. “Many induced mutations have direct or indirect adverse effects,” one researcher noted. There is no official, recommended procedure for destroying the so-called “losers.”
This lackadaisical oversight has not caused environmental problems even though, according to Henry Miller, a fellow at Stanford University’s Hoover Institution, “Literally millions of genetically altered, but not gene-spliced, plants are field tested each year without governmental oversight or strictures: the average plant breeder of corn, soybean, wheat, or potato, for example, may put into the field 50,000 discrete, new genetic variants per year, many or all of which may be the product of ‘wide crosses,’ hybridization in which genetic material (including that from weedy or poisonous plants) has been transferred
across natural breeding barriers.” Fifty thousand might be a low estimate. In 1959 W. C. Gregory grew a million M2 peanut plants (M2 meaning that they were the second generation after mutagenic treatment of seeds) in North Carolina. Among the million was one that became a commercial cultivar, registered in 1964 as N.C. 4X. And at the Carlsberg Research Laboratory in Copenhagen, as of 1995, Dieter von Wettstein and his colleagues had screened 18.5 million chemically mutated barley plants, looking for one that did not cause chill haze in beer.
Nor, as Ammann notes, has human health suffered from eating bread made from mutated wheat or drinking beer made with mutated barley. “Nowadays, for a number of crops, one even has lost track of the newly induced mutant cultivars,” says A. M. van Harten, who has taught mutation breeding for 25 years in the Netherlands. “Breeders often do not care to distinguish anymore between spontaneous and induced mutants. Part of the reason for this is that the public has become aware of—supposed or real—risks of what is commonly called ‘biotechnology.’ As a consequence, breeders may prefer not to mention anymore that their cultivars arose from mutagenic treatments, as this may negatively influence their customers.” In other cases, he notes, the breeders might not even know that the building blocks, or parent varieties they are using in their breeding programs, were mutation-bred.
None of the crops produced through the use of radiation or chemicals is labeled “mutation bred.” Many, like triticale, are called, instead, natural foods. If a law were passed requiring certain crops to be labeled GM or GMO, these crops would not be included. These procedures are all considered modern standard breeding techniques. “The term itself conveys a sense of slow-paced, gentle manipulations that are in some way natural,” notes David Saul, a biologist at the University of Auckland, New Zealand. “But have no illusions about these procedures,” he continues, “they are often genetically brutal and highly unpredictable.” Some of them have been in use for 80 years or more, he notes. They “are the tools that have wrought the fruit and vegetables found in your typical organic food store.”
According to the Mutant Variety Database (available on the Internet), more than 2,000 crop varieties have been created using
chemical or radiation mutation; of these, half have been released since 1985. These crops, including varieties of wheat, rice, oats, barley, grapefruit, lettuce, and beans, are being grown in gardens and orchards and farm plots around the world. Are these crops any less genetically modified—or any more natural—than Golden Rice?
When W. Navarro Alvarez was trying to breed a salt-tolerant rice in Costa Rica using anther culture, he irradiated seeds of seven commercial rice varieties with gamma rays. He reported in 2002 that “all irradiated seeds were sown in the field and grown” for two generations. Out of 10,000 second-generation plants growing on saline soil he found 64 mutants that were salt tolerant.
Yet in order to continue developing Golden Rice, scientists must work inside a Biosafety Level Four greenhouse, the same level of biosafety required of those who work with the deadly Ebola virus or anthrax. At the International Rice Research Institute in the Philippines, Karabi and Swapan Datta, the Indian couple who first urged Ingo Potrykus to work on rice, are breeding the golden color—and the genes coding for beta carotene—into 10 rice varieties currently grown in Bangladesh, Vietnam, India, the Philippines, and Mozambique. They chose “popular and successful plants within particular environments, plants with which we are totally familiar,” explains Swapan Datta. One, for instance, is a rice called BR29 from Bangladesh. “It has good cooking quality and moderate disease and pest resistance, and it is well and truly adapted to its environment. The farmers are happy with it, the market is happy with it.”
If we condemn Golden Rice because it was created by a “manmade” process, as Congressman Kucinich says, that “has nothing to do with the ways of nature,” what do we do with Creso wheat or triticale or the Rio Red grapefruit or Alvarez’s salt-tolerant rice?
What do we do with Luther Burbank and his Iceberg blackberry? Burbank did not use hydroponics or tissue culture or mutation breeding or genetic engineering to create his new crops, yet they are just as manmade. As Liberty Hyde Bailey, dean of agriculture at Cornell University, wrote in 1901:
His new plants are the results of downright, earnest, long-continued effort…. Before the flower opens he cuts off the petals. Thus the bees are
not attracted, and they have no foothold. Then he applies the pollen with a free hand…. Mr. Burbank gets unusual hybrids because he crosses great numbers of flowers and uses much pollen. He is skillful in the technique. He also dares. He has no traditional limitations. He knows no cross that he may not attempt. He has not studied the books. He has not been taught. Therefore he is free. The professor of horticulture would consider it beyond all bounds of academic and botanical propriety to try to cross an apple on a blackberry; but Luther Burbank would make the attempt as naturally as he would dig a new lily from the fields.
Is this amount of interference, this amount of manipulation, still “natural”?