Mendel in the Kitchen: A Scientist's View of Genetically Modified Foods (2004)
Chapter: 14 Sharing the Fruits
14
SHARING THE FRUITS
I believe that scientists, as a privileged group of citizens, have more than an academic responsibility to advance science. They must also accept a higher social responsibility and, wherever possible, use science to help solve the important problems not of industry, but of humanity.
—Ingo Potrykus (2001)
Writing in the New Yorker magazine in April 2000, Michael Specter tells the story of the San Marzano tomato. It’s a plum tomato, from outside Naples, Italy, where the soil is sweetened by the ash of Mount Vesuvius. “According to Neapolitan tradition, pizza was invented as a vehicle for the consumption of the San Marzano,” Specter writes. Yet farmers of this prized tomato are losing the battle against a virus, the tomato fern leaf virus, better known as the cucumber mosaic virus or CMV. It “has taken over the fields,” farmer Eduardo Angelo Ruggiero told Specter. “The tomato was born here. Now I think it’s dying here. We understand that genetics could help, but the question is political. I myself have mixed feelings. I am afraid that if we grow tomatoes differently they will taste like every other tomato in the world. But there is also a truth. We have lost 90 percent of our production in the past decade.” In 20 years the Naples region dropped from being the number one tomato producer in Italy in 1980 to number four or five. Ruggiero himself has stopped growing tomatoes. His children, who had planned to continue the family business, “will have to do it somewhere else,” Specter writes.
In Adams County, Pennsylvania, in March 2000, farmer Jim Lerew of Lerew Brothers Orchard had to bulldoze and burn all 150 acres of his peach and nectarine trees. His neighbor, Jim Lott of Bonnie Brae Fruit Farm had to destroy 25,000 trees—227 acres of orchard. Lott’s trees would have produced $400,000-worth of fruit that year, he estimated. In each case the reason was plum pox virus. The tree-burning was the USDA’s attempt to contain the attack: Lerew was the first farmer in America to identify plum pox as the reason his peach trees looked sick.
Known as sharka (from the Bulgarian for “pox”), plum pox is well known—and hated—in Europe. Some hundred million European peach, nectarine, apricot, plum, cherry, and almond trees are infected. Sharka was found in Chile, also a major fruit producer, in 1992 and is now widespread in that country. Its symptoms resemble those of a nutritional deficiency or insect damage. Often only a few leaves on an infected tree show signs of sickness, yet the fruit is discolored, misshapen, sour, and small. Eventually a sick tree stops bearing fruit altogether. In an infected orchard, crop losses can reach 100 percent. For Pennsylvania that could mean the loss of a $22.5 million industry. If the disease were to spread throughout the United States, losses could climb to $1.8 billion.
For Lerew and Lott the problem is more personal. It could mean losing their family farms. According to Lerew, “Financial losses are higher than a year of lost production. It takes six years for a new fruit tree to start producing in volume and it costs a lot to remove and replace the tree itself.” The government paid Pennsylvania fruit growers $1,000 an acre to destroy their orchards. For Bonnie Brae Farms that was $227,000 for trees that each year produce $400,000 worth of fruit.
Throughout Pennsylvania more than 900 acres of orchards were bulldozed down and burned in the spring of 2000. For the farmers, deciding what to do next is difficult. Replant with peaches or switch to plums, and they risk a recurrence of the pox. The disease is incurable, and no cultivars are yet resistant. Apples are a choice—the pox does not affect them—but many of these farmers already have some acres in apples. They had planted peaches in the first place to lengthen their harvest season, and to give more job security to the migrant workers
who pick the crops. Picking apples is a three-month job. Add in peaches and plums, and the job grows to seven or eight months long. Other crops are not suitable for these hillside farms, nor do the farmers have the capital to buy the equipment needed to plant and harvest something new. The most attractive option to the owner of a burned orchard might well be the ready cash to be had by selling out to a housing developer.
Pennsylvania has invested $5.1 million in its efforts to eradicate plum pox virus. The USDA has allocated nearly as much. Part of the money is supporting research on how to control the pest, which has two known vectors, two ways of traveling from place to place: humans and aphids. “Undoubtedly the human vectors are much more effective in spreading the disease,” says plant pathologist Fred Gildow of Penn State. “Legally imported fruit tree cultivars are tested for disease at USDA quarantine nurseries before being released for public use,” Gildow remarks. The carriers of plum pox virus, therefore, most likely came in illegally, “by accident or out of ignorance,” by someone who failed to inform Customs that they had visited a farm or carried a twig in their luggage.
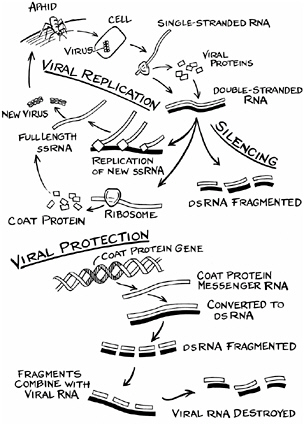
Like the cucumber mosaic virus, the plum pox virus is naturally carried from plant to plant by aphids. An aphid picks up the virus while tasting a plant with its stylet, a syringe-like mouth part. If the sap tastes right, the aphid settles down to feed. If not, it retracts its stylet—now contaminated with the virus—and flies off to taste another plant, which then becomes infected. The virus itself is a single molecule of RNA wrapped in a protective protein coat. Injected into a plant cell, the RNA takes over the cell’s protein-making machinery and instructs it to make virus particles instead.
In Europe scientists studying the plum pox life cycle have counted 3.5 million aphids of 40 different species in each acre of orchard; 20 species of aphid are known to carry plum pox virus. Each tree is visited by 50,000 to 300,000 aphids every year. It takes only one aphid to infect—and kill—a tree.
Insecticides cannot kill every aphid in an orchard. Nor are the harmful species necessarily those that colonize peach and plum trees. In France and Spain the culprit is usually a “migrant aphid,” one that
How a plant virus spreads, and how plants resist their attack
does not usually live in peach trees and is simply passing through the orchard looking for a suitable host. The bird cherry-oat aphid, for example, prefers to feed on small grains; when the grains are harvested, the aphid migrates and can be found in large numbers in peach orchards in late summer. In France the bird cherry-oat aphid carries plum pox virus. Whether it does in Pennsylvania has not yet been tested. Nor is it easy for a farmer to tell just when the bird cherry-oat aphid might be passing through and it is time to spray. Aphids are tiny and look much alike. Besides, as a USDA scientist noted, insecticides don’t kill on contact: the migrant aphid can still infect a tree before it dies.
“The key is to stop them before they take flight, or try to disrupt their life cycle,” says Vernon Damsteegt, a plant pathologist at the USDA’s Foreign Disease-Weed Laboratory in Maryland. One way is to keep orchards completely weed-free, denying the aphids a place to overwinter. To be effective, the weed-free zone would have to be larger than
an aphid usually flies after tasting (and rejecting) a plant. According to Gildow, 100 yards is generally the maximum distance a virus will be carried from an infected plant to a new host. A buffer zone of 300 yards should keep an orchard pox-free. Keeping farms weed-free, however, has its own drawbacks.
![]()
In 2003, the Philosophical Transactions of the Royal Society in Britain published the results of what it called the Farm Scale Evaluations and the press called the GM crop trials. The trials included 66 fields of sugar beets, 68 cornfields, and 67 canola fields, each planted half with a genetically modified herbicide-tolerant variety and half with a conventional crop that was not herbicide tolerant. Teams of scientists from three research centers counted and compared the number of species of weeds and of weed seeds, as well as of beetles, butterflies, and other invertebrates found inside and along the edges of each half-field throughout the year. They concluded that some genetically modified crops (canola and sugar beet) reduced biodiversity, while GM corn increased it.
The difference was that the chemicals currently used to keep weeds out of conventional cornfields (atrazine and its derivatives) are quite effective—better at killing weeds than the glufosinate used with the genetically modified corn. The herbicides used with conventional canola and sugar beet, on the other hand, are less effective than glufosinate when used with GM canola or than glyphosate when used with GM sugar beets. The studies thus, as the Scientist reported, “are less about GM crops directly than about the herbicides used to manage weeds in GM and conventional varieties.” GM corn was said to increase biodiversity because the herbicide used on GM corn (glufosinate) did not kill as many weeds as the herbicide used on non-GM corn (atrazine).
Herbicide-tolerant crops can be created through conventional breeding. Herbicide-tolerant varieties of canola, corn, and wheat created through chemical mutagenesis and somaclonal variation are currently sold. Whether the crop is genetically modified by mutagenesis
or by molecular techniques, the crop-herbicide package that is most effective will result in the fewest weeds—which is, indeed, what farmers are hoping for when they buy weed-killer. But the fewer the weeds, these studies found, the fewer the butterflies, beetles, and other invertebrates living in and around the field—except for those that feed on dead plant matter.
Because 70 percent of the land area of the United Kingdom is either planted in crops or grazed by farm animals, it’s understandable that the British scientists and press should define biodiversity as the number of butterflies, beetles, and other invertebrates that live on the weeds growing in and alongside farmers’ fields. Yet such a definition puts the Pennsylvania peach farmer in a quandary. Aphids are among those other invertebrates that the British believe farmers should protect as part of desirable biodiversity. Aphids carry the plum pox virus that can wipe out a Pennsylvania orchard and leave the farmer with little alternative but to quit farming and sell out. A housing development or a shopping center would most likely be lower in biodiversity than an orchard, even one enclosed by a 300-yard weed-free zone.
Yet there is a way to have both farmland biodiversity—weeds and their invertebrate residents—and orchards free of plum pox. The answer resides in three plum trees growing under quarantine in a USDA orchard, three trees genetically modified to be resistant to the plum pox virus.
Named C-5, the resistant plum tree was developed by Ralph Scorza, a USDA horticulturalist known for breeding the Bluebyrd plum. C-5 contains a gene, provided by French researcher Michel Ravelonandro, that codes for the plum pox virus’s coat protein. Coupled with a strong promoter, the coat protein gene provides the plant with the equivalent of an immune response—although it works very differently from the immune response in humans.
Inside the cells of the resistant plum variety, the coat protein DNA is transcribed into messenger RNA. So much messenger RNA is made, in fact, that it activates an oversupply shutdown mechanism. The mRNA is destroyed and the coat protein is never made. When the plum is infected by the pox virus, the virus uncoats its RNA, ready to instruct the cell to make more viral RNA and more coat protein to wrap it in.
But the cell is ready. It recognizes the RNA and destroys it. The virus-resistant papaya, which in 1998 saved the Hawaiian papaya industry, is protected by this same post-transcriptional gene silencing mechanism. Dennis Gonsalves, who developed the resistant papaya, provided Scorza with the Agrobacterium vector used to transform the plum.
The C-5 plum passed the new gene—and the resistance—to its offspring, so it could be used to breed other plums by crossbreeding or grafting. In fact, when cuttings were grafted onto a rootstock infected with the plum pox virus, C-5 depressed the virus so much that aphids could no longer pick it up and spread it from tree to tree. The rootstock was cured. As of September 2001 the C-5 plum had been tested in three European countries for five years and it was still resistant. But it remains “under development” and has not been commercialized. Once it is on the market, the USDA’s magazine Agricultural Research wrote, plum farmers “can breathe a sigh of relief, knowing they have a backup tree to plant. And U.S. consumers may prefer a transformed plum to no plum at all.”
Relief perhaps very much like what Hawaiian papaya growers felt when they learned that American consumers would, indeed, prefer a transformed Hawaiian papaya to no Hawaiian papaya at all. Three years after the virus-resistant papaya seeds were first given out, Hawaiian papaya production—and sales—were back up to what they had been in 1994, before the virus moved into the prime papaya-growing region of Puna. The 2001 harvest of more than 55 million pounds was worth $14 million to Hawaiian farmers.
The industry recovered in spite of what became known in the media as The Great Papaya Scandal, or “How Prof. Joe Cummins uncovered the great scandal of how U.S. regulatory agencies approved a GM papaya even though it carries a viral gene known to be a potential allergen.” Cummins, a retired professor, had previously been a coauthor with Mae-Wan Ho of the 1999 opinion piece, “Cauliflower Mosaic Viral Promoter—A Recipe for Disaster.” He read a scientific paper that reported a slight similarity between the papaya ringspot virus coat protein and a protein known to be allergenic. Apparently without understanding that post-transcriptional gene silencing prevents the coat protein from being made, he became alarmed that papayas might now
be full of allergy-causing proteins. He contacted an official at the FDA, who told him the resistant papaya was the regulatory responsibility of the EPA. He learned that the EPA had granted the papaya an exemption from regulation—no doubt because a virus-resistant papaya contains much less viral coat protein than the virus-infected papayas people had been eating. Cummins, still unaware of the facts, began his crusade. If he had understood why the plants were protected from the virus, he wouldn’t have had a story.
In 2002 Dennis Gonsalves and his research team were awarded the Alexander von Humboldt Award for making “the most significant contribution to American agriculture” in the previous five years. In 2003—the year Hawaii’s genetically modified papayas were approved for sale in Canada—Gonsalves won the Leadership in Science Public Service Award from the American Society of Plant Biologists. The citation pointed out that Gonsalves was born and raised on a sugar plantation in Hawaii. “What inspired our team,” Gonsalves said, “was the knowledge that we had to apply the best science we could to solve very real problems for farmers and families who were desperate for a solution.”
The Italian farmers growing the prized San Marzano tomatoes are also desperate for a solution. But for many of them the only solution possible is to stop growing San Marzanos. Francesco Sala of the University of Milan reported in April 2003 that “already today, the San Marzano accounts for just 3 percent of Campania tomatoes, while 20 years ago this figure was 30 percent. Today a lot of growers use an American hybrid in its place which yields well and has a good taste, but is not the same thing. In short, the San Marzano is a product in extinction.”
Italian researchers, back in 1993, used the same basic method with which Gonsalves developed Hawaii’s new papaya, and Scorza Pennsylvania’s new plum, to modify the San Marzano tomato to be resistant to the tomato fern leaf virus. But whether a transformed San Marzano tomato is preferable to no San Marzano tomato at all was still to be learned 10 years later because, says Leonardo Vingiani, the director of the Italian biotechnology association, Assobiotec, the “necessary authorisation” for field trials had not yet been granted.
Assobiotec’s Sergio Dompé writes in the magazine Slow: The International Herald of Taste, published by the Slow Food movement,
“Could the name be the problem?” The words “genetic engineering” and “biotechnology,” he argues, call up “a glaring contradiction between life and technology, the natural and the artificial, that generates concern and apprehension.” He compares acceptance of genetically modified foods to the acceptance of nuclear magnetic resonance—the medical procedure that suddenly became noncontroversial when its name was changed to magnetic resonance imaging. “The moral of the story: Inappropriate words, such as a misunderstood adjective or a bold juxtaposition, often influence our view of reality, feeding our suspicions and unspoken fears even when there is no justification.”
Italians, he says, fear that biotechnology will standardize or “dumb down” their traditional foods and flavors, jeopardizing the country’s heritage. “This is a very serious accusation,” Dompé says. It is also, he continues, “as unjust as it is serious. It is a myth.”
In Italy, he writes, rare plant diseases can be devastating. Neither conventional farming practices nor organic methods, for example, have been able to save the San Marzano plum tomato—which Dompé calls “a national treasure on a par with Piedmontese wines”—from the attack of the cucumber mosaic virus. “In this case,” Dompé says, “genetic engineering has offered more than just the promise of a solution. It has already proved able to save the crop by inducing virus-resistant characteristics. The fact that it is not yet possible to apply this solution in the field (because it is based on ‘genetic modification’), suggests that sometimes the risks for typical Italian products do not derive from biotechnology but from unreasonable resistance to their use. It would indeed be a tragedy to condemn this tomato variety to extinction, simply because of preconceived ideas.”
![]()
The loss of the San Marzano tomato will be a tragedy to those who prize its distinctive flavor. Pizza might never be the same. If lack of the San Marzano drives small family farms like Eduardo Angelo Ruggiero’s out of business—as a virus threatened to do in Hawaii and still threatens in Pennsylvania—more people will be affected. Some will miss eating fresh locally grown produce. Farm families must find another
income. Environmentalists who hope to keep the land open and undeveloped will lose another bulwark against the shopping center and housing complex.
For Florence Wambugu the issues surrounding the virus-resistant sweet potato are in some ways the same—she hopes to keep Kenyan women producing a good crop of sweet potatoes—and in other ways very different. In Kenya the failure of the sweet potato crop does not mean just the lack of a prized taste or even a monetary setback for otherwise well-off and well-fed farmers. In Kenya the word tragedy retakes its true meaning. Lacking sweet potatoes, a family can starve. Interviewed by the New Scientist magazine in 2000, Wambugu explained, “The sweet potato is a major staple crop. It is always there in the backyard if there is nothing else to eat.”
Sweet potato supplies more food energy and more nutrients than any other crop that could be cultivated in the same amount of (mostly marginal) land. Yet the yield per acre in Africa is one-third that in China and less than half the global average. In 1999 Matin Qaim of the University of Bonn authored a study of the potential economic effects of genetically modified virus-resistant sweet potatoes in Kenya. Sweet potato, he reported, is cultivated on 75,000 hectares in Kenya, mostly in plots of less than half an acre with two crops a year. Forty percent is kept for the farm family to eat. The other 60 percent is sold, harvested a little at a time, “whenever cash is needed to meet the basic household requirements.” It is considered a woman’s crop. Although men might help with the plowing, the weeding and harvesting are left to the women—as are any sales, either to traders who come to the farm periodically or by carrying baskets on their heads to the nearest village. If yield could be increased, those women and their children would have more food and more money.
A genetically modified virus-resistant sweet potato, Qaim predicted, could increase yields by 18 percent. (Wambugu believes that yields could double.) If yields increased 18 percent, farm incomes would rise by 28 percent, Qaim calculated, for a total of $5.4 million across the country. “Because women often control the revenues from sweet potato sales,” he wrote, “there is a high probability that a significant proportion of the additional income would be spent on food.”
The difference could be critical to a hungry child. According to political scientist Robert Paarlberg, 30 percent of the children in Africa are chronically malnourished due to “lagging productivity on small farms.” In Africa, he says, more than anywhere else in the world, the problem of hunger is due to farming methods that simply do not produce enough food. In some parts of Africa, he writes in The Politics of Precaution: Genetically Modified Crops in Developing Countries, “yields are actually declining from their already low levels.” Although farmers cut down nearly 5 million hectares of forest a year to expand their fields and pastures, the amount of food produced per person has not increased since 1970. Even counting food aid and commercial imports, each person has less food to eat now than in 1980.
Africa’s farm production problem is not the result of modern technology. The agricultural advances of the past half century have largely passed Africa by. As Florence Wambugu says, Africa “missed” the Green Revolution. It can’t afford to miss the next one. Asked by an interviewer for the New Scientist, “Surely what African farmers really need is fertilisers and better irrigation? Won’t putting money into GM technology divert attention from these more basic needs?”, Wambugu answered: “I think that is like saying Africans don’t need aircraft, we should go by road. Or that we should be denied computers until everybody has bought a typewriter and mastered it. We are part of a global community. Of course, we need to look at why existing agricultural technologies have had so little impact in Africa. Africa needs to pick and choose technologies, to learn which ones are compatible.”
Wambugu’s own choice of technologies is influenced by both her upbringing and her education. “As one in a family of nine children growing up on a small farm in Kenya’s highlands,” she says, telling her story in the Washington Post, “I learned firsthand about the enormous challenge of breaking the cycle of poverty and hunger in rural Africa. In fact, the reason I became a plant scientist was to help farmers like my mother, who sold the only cow our family owned to pay for my secondary education. This was a sacrifice in more ways than one because I, like most children in Kenya, was needed on the farm.”
It was, as she puts it, “to solve a national problem” that Wambugu joined the Kenya Agricultural Research Institute upon receiving her
degree in botany from the University of Nairobi. There she worked with scientists from the International Potato Center on the problem of viruses in sweet potatoes. To learn more, she traveled to North Dakota State University for a master’s degree in plant pathology. She earned her Ph.D. in virology and biotechnology from the University of Bath in England in 1991, doing the fieldwork for her dissertation on sweet potato viruses at home in Kenya.
Using traditional plant breeding methods, she tried to improve the sweet potato’s resistance to viruses and pests. “But after years of hard work and frustration,” as she puts it, “I finally realized I would not be able to develop a virus-resistant potato through traditional plant breeding.” It was then that she was invited to visit Monsanto.
Roger Beachy, then at Washington University, had collaborated with researchers at Monsanto in the late 1980s to produce the first virus-resistant plant by inserting a virus coat protein gene. In 2003 Beachy and Monsanto’s Robert Fraley and Stephen Rogers received a patent on this technique, which is the one used to create virus-resistant papaya, plum, and San Marzano tomato, as well as squash, sweet potato, cassava, and several other crops. Rob Horsch, currently Vice President for Product and Technology Cooperation at Monsanto, recalled, “It was Ernie Jaworski that got the idea that it looks like this stuff—both biotechnology broadly and virus coat protein gene specifically—would work. He thought it ought to be shared.” Jaworski’s studies of how Roundup worked had led Monsanto into the business of plant breeding in the early 1980s.
Horsch continued, “Ernie talked with Joel Cohen at the U.S. Agency for International Development (USAID) and hatched a plan to do some kind of root or tuber virus protection project.” They chose roots or tubers because these vegetables are susceptible to the most severe virus problems, Horsch said, “and because no other technology was solving the problem.” Monsanto wanted to recruit an African scientist to decide which root or tuber, and which virus, the project should focus on. Said Horsch, “We figured an African expert would be better qualified to know what was most needed in Africa, rather than making that choice in St. Louis. As we began the recruiting process, Florence Wambugu’s name popped up.”
Wambugu arrived at Monsanto headquarters in 1992, bringing seven sweet potato varieties from Kenya. She joined Horsch’s lab at Monsanto and worked for two years constructing the vector—the ring of DNA containing the virus coat protein gene, the promoter, and the accessory DNA needed for it to be accepted by a sweet potato plant cell—that would make the plant resistant to the sweet potato feathery mottle virus. When her time was up she returned to Nairobi; since then a series of visiting Kenyan scientists, Horsch says, have been “steadily making progress on this difficult problem.”
Monsanto donated to the Kenya Agricultural Research Institute (KARI) not only the improved sweet potato plants but also the license to use for free whatever patented genes or techniques were needed to create them. KARI has since sublicensed the technology back to the Donald Danforth Plant Science Center in St. Louis. The center, whose director is now Roger Beachy, has become the Kenyans’ primary technical partner in place of Monsanto.
In 2000 Horsch visited the first field trials of genetically engineered sweet potatoes in Kenya. “The first field test revealed what virtually all first tests reveal,” he has said. “More work is still needed to produce a satisfactory product. The commercial biotech products my company began selling in 1996,” he explained, “are the result of going back to the drawing board four or five times.”
According to Lawrence Kent of the Danforth Center, the field trials didn’t do well because the virus gene came from an American strain of the virus, not a local Kenyan strain. “Even Monsanto knew there was a good chance it would fail, but they wanted to test the system,” he explained. After the disappointing performance of the first gene, Monsanto created three new vectors and donated them to both KARI and the Danforth Center. The new vectors use a virus isolated in Kenya combined with a new technique, called codon optimization, that increases the level of expression of the coat protein gene. “With sweet potato, the problem is mostly scientific at this point,” Kent said. Only testing will show if the new gene gives sufficient protection against the virus.
Thirteen years after Florence Wambugu arrived at Monsanto headquarters, a virus-resistant sweet potato is still not growing in Kenyan
farmers’ fields. “The project has not moved rapidly,” a USAID spokesperson told Science, “because KARI doesn’t have the expertise to manage transgenic crop development.”
Robert Paarlberg provides a gloss on that term expertise. “It is extremely difficult for politically cautious leaders in poor countries to be seen welcoming GM seeds if they are coming from a private corporate lab in the United States,” he says. “In fact, a strong pattern emerges.” In researching his book, The Politics of Precaution, he learned that one reason Kenya has not approved the virus-resistant sweet potato is that the technology came from Monsanto. One reason Brazil hasn’t approved Roundup Ready soybeans and India hasn’t approved Bt cotton is that they are Monsanto products. And one reason China did approve Monsanto’s Bt cotton was that it could at the same time approve varieties made by Chinese scientists.
For Horsch, who is now in charge of a project called Growing Partnerships at Monsanto, “It also illustrates the dilemma between doing it and trying to build up the local capacity.” By capacity, he means both training and access to the necessary tools and techniques. KARI’s plant biotechnology laboratories, according to a Danforth Center scientist, are now first-rate. “They have established transformation capacity at KARI that’s equal to ours,” said Nigel Taylor. “They’re doing very well now. Really it’s a collaboration now, not a technology transfer.” And yet the decision to help Florence Wambugu and her fellow Kenyan scientists learn how to create virus-resistant plants has, said Horsch, “come at a price.” He explained, “The unfortunate consequence is the product is still not done. If Monsanto had decided to do it ourselves, the product would have been done three to four years ago. Sweet potato was both the wrong crop—the Rockefeller Foundation picked rice to fund—and too experimental to attract much development assistance money from public sources.”
Yet Horsch also alludes to Paarlberg’s insight when he concedes that the project was finally transferred completely to the Danforth Center because, “There was the need for the program to stand on its own merits, and not be seen as a Monsanto project.”
Agricultural economist Lawrence Kent, a former Peace Corps volunteer with 10 years’ experience working in Africa, was hired by the
Danforth Center to help it increase its collaborations with African scientists. “Living in Africa,” he said, “I saw that better seeds and other improved agricultural technologies could make a big impact on people’s lives.” He was working in Egypt when he first became aware of the Danforth Center. “I saw they had good science and a good mission, but they were missing a step: transferring their technology to Africa. In Africa you have to really work the process. I proposed that they hire me to help, and Roger Beachy agreed.”
Among Kent’s most difficult tasks is getting approval for a field trial. One problem is that the regulatory system is not in place. “Lots of countries are working on it, but Kenya is about the only country in sub-Saharan Africa, other than South Africa, that has an application form for a field trial ready for you to fill out. Even in Kenya, there are no permanent bodies to give approvals. They have to convene a committee each time. They all hear these stories from Europe. They have these half-issues and doubts in their minds.”
Wambugu gave her opinion in an essay published by the Washington Post in 2001: “So the question becomes, why aren’t these types of biotechnology applications more readily available to African farmers? I believe blame lies with critics who claim that Africa has no chance to benefit from biotechnology and that our people will be exploited by multinationals. These critics, who have never experienced hunger and death on the scale we sadly witness in Africa, are content to keep Africans dependent on food aid from industrialized nations while mass starvation occurs.”
Asked by New Scientist magazine in 2001 if she was “an apostle of Monsanto in Africa,” she replied, “Some people say I am fighting for the company. But I say I am a stakeholder in the technology. It is 20 years of my life. I believe in the benefits it has for our people. So I fight for the credibility of the technology.”
Two years later, when she continued the discussion with an interviewer for the Canadian Broadcasting Corporation, virus-resistant sweet potatoes were still not on the market. “GM sweet potato has nothing to do with Monsanto as a product,” she told the television audience. “It is not of any value to any company that I know of and I think, if anything, it has served a really good example on how African
countries can help themselves by partnering and going back and developing their own system so they can make decisions.”
The interviewer asked, “Dr. Wambugu, you’re trying to persuade us all that genetically modified foods are the way to go for agriculture in Africa, right?” Wambugu answered, “I don’t think that my business is to persuade anyone specifically on GM. What we’re looking for in Africa is all kinds of technologies that can help us to increase food production. And GM is only one of these technologies.”