Mendel in the Kitchen: A Scientist's View of Genetically Modified Foods (2004)
Chapter: 10 The Butterfly and the Corn Borer
10
THE BUTTERFLY AND THE CORN BORER
Because science and politics have become so entangled, it has been hard to pick rationally through the facts.
—Michael Specter (2000)
Victor Davis Hanson is a fifth-generation grape farmer and orchardist in the San Joaquin Valley of California. He is also a professor of classics at California State University. In The Land Was Everything: Letters from an American Farmer, he writes eloquently about “a race that we cannot win, between land farmed and land paved. Our ancestors,” he notes, “beat back the wild to give us these farms—we of a lesser breed were beaten back by new men to give them up. We battle the encroaching suburb; our grandfathers in their youth once fought hunger and typhoid—both of us have received four dollars a box for plums.”
Asphalt is, of course, not his only enemy, nor the one he spends his days battling. “People must realize,” he writes, “that farming is not statistics from the Department of Agriculture nor quotes from some scrubbed agribusiness talking head, but rather is a very unpleasant and brutal task to bring food out of the dirt.” Against the “flyers, hoppers, and crawlers,” for instance, those “pests of biblical magnitude,” he sprays poison. “The trick,” he says, “is merely one of logistics, not of poison per se: providing the dose in enough quantity and strength to kill the swarm before it demolishes the vineyard in question.”
Farmers have long had first-hand knowledge that they were also, while saving the crop, poisoning other creatures who did no harm. Hanson writes, “Organophosphates killed every living thing in the orchard and the vineyard: mites, hoppers, and worms, but also predator spiders, wasps, and ladybugs, even birds, gophers and snakes—and the occasional laborer. Even the lowly toad snug in his hole beneath the vine was not safe. I have seen these cute amphibians still and yellow outside their holes on the day after the spray.”
Still, Hanson sprays.
“Why, you ask, would any sane man, as I have, apply even the less toxic ancestors of parathion—Lannate, Guthion, dimethoate, lindane, carbaryl, Thiodan, or diazon—to kill everything on leaf and tendril?” Because the alternative is to cash it in and let the “new men” pave over the farm.
Not that Hanson hasn’t tried less toxic means. Not that he doesn’t wish, fervently, for a better way. A farmer’s glossary, he says, defines a spray rig as, “a mechanical Faust that can save your crop and you along with it, at the price of your soul.”
Referring to a brand of pesticidal Bt, he writes:
If you dust the organic bacteria Dipel on grape leaves, it will deter, but not stop, a grape-leaf skeletonizer or coddling moth. If, as one should, you sow cover crop, plant berries and flowers, and use manure, you can grow predators to chomp on, but not eradicate, deleterious insects. Is it to be hours of contemplation, repeated application, and constant monitoring, or one shot of caustic Omite that will kill red spider mite for two months? The choice is not really the blinkered farmer’s alone. The decision has already been made by the environmentally correct consumer: He wants fruit plentiful, colorful, hard, and fresh, free from crack and scar—not high-priced and tasty, overripe, leaking, and pockmarked, a hitchhiking gnat or stowaway fly now swirling amid the glistening produce section at the local supermarket. Under no condition must the vine-hopper’s non-toxic excrement dot the grape. An English professor once called me complaining about the grapes he bought at the local organic farmer’s market on campus. “They have black dots all over the bunches,” he raved.
“That’s hopper crap,” I replied.
“Well, who wants to eat it? Not me,” he fumed.
No grapes have been genetically modified to carry their own insecticide (although researchers in California are developing a grape that is protected against a bacterial disease). Yet the crops that produce
toxic Cry proteins, using a gene from the bacterium Bacillus thuringiensis, were designed with Hanson’s concerns in mind. Bt crops are intended to keep “non-target species,” from the lowly toad to the laborer, safe. The first Bt gene was cloned and transferred to E. coli in the early 1980s. By 1990, with some adjusting of the DNA coding sequences, to reflect differences in the abundance of certain kinds of RNA in plants as compared to bacteria, tomato, tobacco, and cotton had been modified to make their own Bt toxins. Bt corn and potatoes were reported in 1993. In field tests it was found that the toxic Cry proteins worked. The only creatures killed by the poison were the ones that ate the crop—which is a fair definition of “pest.”
It is ironic, then, that the fight against genetically modified foods was inflamed by a scientific finding that pollen from Bt corn could, in the laboratory, kill the larvae of a monarch butterfly.
Monarch butterflies have been called “the great fluttering pandas of the insect world.” Like pandas, eagles, wolves, whales, elephants, snow leopards, baby seals, and other charismatic mega-fauna, monarchs are much beloved and much photographed. They are emblematic in many people’s minds of the word “butterfly.” As well as being wonderfully picturesque in their bright orange and black habit, monarch butterflies are also mysterious: they undertake a heroic migration each year from Canada and the United States to a patch of Mexican forest, there to cluster in brilliant bunches in the trees.
How they navigate over such distances and why millions congregate in so small a space are unknown to science. Much of their lifestyle, though, is plain. Monarch butterflies depend on the milkweed plant; they are also known as milkweed butterflies. They lay their eggs on the undersides of the weed’s upper leaves and, when the larvae hatch, the caterpillars eat the milkweed leaves. They eat only milkweed leaves for two weeks, growing and molting several times. Then, when the caterpillar is about 2 inches long, it spins a cocoon, in which it is transformed into the beautiful butterfly—the beautiful and noxious butterfly. For the milkweed plant is poisonous. The monarch larvae accumulate the toxin from the leaves they eat, and the poisons persist in the butterflies. These toxins make the monarch repulsive to most birds and other predators. A bird will bite a monarch once and drop it,
never to bother such an insect again. The ploy is so successful that other butterflies, which evolved into monarch look-alikes, also avoid being eaten, even though they are not themselves toxic.
In May 1999, in a one-page letter published in the journal Nature, John Losey and two colleagues in the entomology department of Cornell University told the world that pollen from Bt corn plants kills the larvae of the monarch butterfly. Losey’s paper was front-page news in the New York Times; the monarch photo accompanying the story was captioned “Bambi of the insect world.” The news traveled around the world, from the BBC to the Boston Globe to the San Francisco Chronicle.
The study immediately turned up the volume of sentiment against genetically modified foods. Greenpeace members in butterfly costumes mimed being “felled by killer corn.” Friends of the Earth, soliciting new memberships, sent a letter asking, “If deadly toxins that kill butterflies are being introduced into our food supply, what effect are these toxins having on you and your family?” The activism itself fueled further coverage in the news, and calculatedly so. Margaret Mellon of the Union of Concerned Scientists recently admitted to a journalist that “We worked hard to make this a high-profile issue because without media attention we knew nothing would be done. We saw the findings as an illustration of how superficial risk assessment for GM foods was.”
Inside the scientific community, Losey and his colleagues were criticized for overstating their case. Their laboratory study was imprecise and—more importantly—seemed far from the conditions a monarch would encounter in the wild. Losey’s team had sprinkled either Bt corn pollen or regular corn pollen onto an array of damp milkweed leaves. Onto each leaf they set five three-day-old caterpillars. They repeated the experiment four times. Of the larvae exposed to Bt corn pollen, 44 percent died; those that lived were smaller than the larvae in the control group. The researchers concluded that Bt corn had “potentially profound implications for the conservation of monarch butterflies.”
“Never mind,” reporter Bill Lambrecht wrote, “that just eleven caterpillars died. Or that the pollen might not have been spread uniformly on the leaves and that they had run out of the pollen, preventing them from broadening the scope of the project.” Losey had not quantified
the amount of pollen on each leaf, nor had he given the caterpillars a choice between plain milkweed leaves and those with the presumably untasty pollen on them.
The Cry protein to be expressed in corn was chosen specifically because it did kill caterpillars. To entomologist Anthony Shelton, also at Cornell University, Losey had no story. “Every entomologist knows,” said Shelton, “if you feed monarch butterfly larvae Bt toxin, whether it be in corn or whether it be on a spray, that insect will die.” Many environmentalists know it as well. They have used the same argument to fight the widespread spraying of forests to kill the alien gypsy moth. Bt is the pesticide of choice to control this voracious caterpillar, which will defoliate whole forests if left unchecked. Spraying Bt from helicopters saves the trees, yet kills most other butterflies and moths, including monarchs. In the case of Bt corn, choosing between the risks to the butterfly and the benefits to people seems far easier. As John Foster, an entomologist at the University of Nebraska, wrote: “There probably was not an entomologist in the world who was not aware that corn pollen containing the Bt gene could harm butterflies—if butterflies ate corn pollen, which they don’t.”
Monarch caterpillars in the wild eat milkweed and only milkweed. In their butterfly stage they sip nectar. The only way corn pollen can harm them is if it falls on a milkweed leaf while they are hungry caterpillars and they inadvertently eat it. Milkweed grows both in and outside of cornfields. Inside the field, it is a weed—one the farmer diligently tries to eradicate. If outside the field, it still needs to be near enough that the heavy corn pollen can reach it in quantity. The two events also have to be synchronized. Losey’s study didn’t mention the fact that corn pollen had to be shed and fall on the milkweed leaves at precisely the same time the butterflies were laying their eggs and caterpillars were hatching.
Because of the unknowns—and the publicity—a remarkable study was launched. It was funded jointly by a pooled grant provided by the Agricultural Research Service of the USDA and the Agricultural Biotechnology Stewardship Technical Committee (whose members were Aventis CropScience, Dow AgroSciences, E. I. DuPont de Nemours and Company, Monsanto, and Syngenta Seeds, Inc.), together with funding
provided from the Canadian Food Inspection Agency, Environment Canada, the Ontario Ministry of Agriculture, Food, and Rural Affairs, the Maryland Agricultural Experiment Station, and the Leopold Center for Sustainable Agriculture.
Researchers from the USDA and several universities looked at all the factors likely to determine the effects of Bt corn pollen on the larvae of monarch butterflies. They performed tests in the laboratory and in actual cornfields. One group measured exactly how toxic the various Cry proteins were to caterpillars, as well as the amount of toxin present in the pollen grains of three widely used Bt corn varieties. Other groups measured how much milkweed grew in, near, and far from cornfields at sites from Ontario west to Wisconsin and Iowa, and down through New York to Maryland. Others counted the number of pollen grains deposited per square centimeter of leaf surface on milkweed plants in cornfields, near cornfields, and far from cornfields. Did the upper leaves of milkweed plants, on which the larvae fed, they asked, receive the same amount of pollen as leaves farther down? How was the pollen density affected by wind and rain? Still others asked whether monarchs laid more eggs on milkweed plants inside cornfields, where milkweed is a weed, or on plants outside cornfields. Finally, they determined the extent to which egg laying coincided with the week to 10 days during which corn plants shed their pollen.
The results of all these studies—including one coauthored by John Losey himself—were published together in September 2001. The timing could not have been worse. “The world might have a different attitude to the safety of genetically modified (GM) crops if it had not been for the terrorist attacks on New York and Washington,” wrote the editors of SciDev.Net in June 2002. SciDev.Net is an Internet news service sponsored by the journals Nature and Science in association with the Third World Academy of Sciences and is funded by the United Kingdom, Canada, and Sweden. “For among the many stories that these attacks obliterated from the newspapers,” the editorial continued, “was a report on six scientific papers published simultaneously in the Proceedings of the National Academy of Sciences (PNAS) on the safety of corn that has been genetically engineered to express a toxin that kills certain pests.”
There were no Greenpeace costumers, no Friends of the Earth letter-writers to ensure that the news reached page one under a photo of the “Bambi of the insect world.” There was no effort by the Union of Concerned Scientists to make it a high-profile issue, as there had been when Losey’s original study was published. Consequently, noted SciDev.Net’s editors, “the image of dead monarch butterflies lingers in the public consciousness as the unacceptable face of GM technology.”
A report by the Pew Initiative on Food and Biotechnology notes that Losey’s initial study and the “firestorm that it unleashed” had good consequences. The EPA made some intelligent changes in its regulatory process. And when the registrations on five Bt-corn varieties came up for renewal in October 2001, “The decision to go ahead was taken after a vigorous debate in which all stakeholders—from industry representatives to environmentalist groups—had been given a chance to examine the Cornell research and its implications in detail, to assess the results of follow-up studies, and to talk through their differences,” according to SciDev.Net. The result, they reported, was “a robust social consensus between all sides of the debate that had previously been lacking.”
News of this robust social consensus did not make it to page one of the world’s newspapers. In 2003, when Prospect Magazine in the U.K. printed “The Butterfly Flap,” journalist Peter Pringle instead summarized the results of the “vigorous debate” as: “Losey and co. had lost a battle.” The PNAS series did uncover some unappreciated facts about monarch butterfly biology. While corn pollen is shed well before the peak of monarch egg laying in Maryland, the two events overlap in more northern areas, such as Minnesota and Ontario. In Ontario, most of the milkweed that supports monarch caterpillars is outside cornfields. In other places, particularly in the Midwest Corn Belt, monarchs laid their eggs on milkweed plants inside cornfields just as often as they did on milkweed outside cornfields. The studies also confirmed, as plant breeders had argued, that corn pollen is rather heavy and most of it comes to rest inside the cornfield. A few steps away from the field (2-3 yards), only a fifth as much pollen landed as did inside the field.
As for the risk of Bt corn, the studies determined that only one of the three commercial varieties produced enough Cry protein to kill
monarch caterpillars—or even to slow their growth—at the actual pollen densities detected in cornfields. At the time of the study, this variety—called event 176 hybrid and sold by Novartis under the name KnockOut and by Mycogen as NatureGard—accounted for only 2 percent of the corn planted. It had already been slated for phaseout. According to Chuck Armstrong of Monsanto, “The product concept was to express Bt in the foliage and the pollen. But insects still got inside the plant and caused the damage. Farmers found one kind of Bt corn, Yieldgard, worked well, and Bt176 didn’t work very well. The monarch butterfly study was just one more nail in the coffin. It wasn’t the reason event 176 went off the market.” In 2001 the registration for event 176 hybrid was allowed to lapse, and it is no longer sold.
In a summary paper in PNAS, Mark Sears of the University of Guelph in Canada and several other authors calculated the worst-case scenarios for Iowa, a major corn-producing state. At one extreme, they assumed that KnockOut and NatureGard would increase to 5 percent of the area planted. When they combined this assumption with the amount of egg-laying-pollen-shed overlap, the actual pollen densities, and the proportions of milkweed both inside and outside cornfields, they found that about 0.4 percent of the monarch population (or 1 monarch out of every 250) would be at risk of exposure to toxic effects from Bt corn.
A second scenario assumed, instead, that KnockOut and NatureGard corn were phased out (as indeed has happened) and the pollen of future Bt varieties would be only as toxic as the two other varieties tested (Syngenta’s Bt11 YieldGard or Attribute, and Monsanto’s Mon810 Yieldgard). Then, even if 80 percent of the corn acreage in Iowa were planted in Bt varieties—and this is the maximum expected if the current stipulation for Bt-free refuges, or refugia, is maintained—only 0.05 percent of the state’s monarch population might be in danger. Five hundredths of a percent means one larva out of every 2,000.
Per Pinstrup-Andersen, then director general of the International Food Policy Research Institute, summed up the monarch butterfly fracas in 2000: “The attention given to this particular case contrasts strangely with the general attitude toward standard agricultural prac-
tices: conventional pesticides, which do not differentiate between friend and foe, kill a great many harmless insects. The fierce denunciation of clearly targeted and contained pest control through Bt plants is perhaps a little out of touch with reality.”
The Associated Press, to its credit, did try to include a reality check in one news story in 1999. It contacted Jeffery Glassberg, president of the North American Butterfly Association, and asked his opinion of the threat of Bt corn. Said Glassberg, “I think there are a lot more dire threats than that to monarchs. In the Midwest, mowing roadsides and using herbicides is probably much more devastating, actually.”
![]()
Anthony Shelton, Losey’s colleague in Cornell’s entomology department, is equally concerned about the side effects of Bt crops. He does not worry about the monarch: “How many monarchs get killed on the windshield of a car?” was his flippant answer to one journalist who pressed the point. But he has warned since 1995, when the first Bt plant (cotton) was commercialized, that the crops’ success could very well spell their failure—and that of Dipel and the other Bt sprays on which organic farmers depend.
Plants and insects, as entomologists well know, wage ongoing chemical battles for survival. Like the milkweed, plants produce toxic chemicals in an effort to keep insects from eating them. Like the monarch, insects evolve ways to evade the chemical tricks of plants. The interests of humans are generally aligned with those of the plants—we don’t want bugs getting to our food first—with one rather large exception. The chemicals that plants make to protect themselves sometimes irritate us as well. The lectins in kidney beans are there to ward off insects, as are the glycoalkaloids in potatoes and cassava, and the psoralens in celery. It is no accident that domesticated crops are less toxic, in general, than their wild ancestors. As Michael W. Pariza, a specialist on cancer risks in food at the University of Wisconsin-Madison, has noted, “In this context, crop breeding can be seen as a means of controlling unwanted wild traits, such as toxin production, in plants destined for the table.”
“Unfortunately,” Pariza adds, “selected traits that make a plant desirable as human food also make it desirable to insects. This, in fact, is why synthetic pesticides are used: they replace the naturally occurring pesticides and related survival traits that have been bred out of food plants.”
Crop breeding has, at times, worked the other way. Jack Harlan, the expert on crop evolution, points out that many Indian tribes prefer to grow highly toxic varieties of cassava. The poisonous varieties, while requiring extensive care when preparing and cooking, were less damaged by insects and disease, and so produced a greater yield. The developers of the Lenape potato, in the 1960s, also selected for high toxicity—without meaning to—while trying to breed in disease and insect resistance. Again, in the 1980s, trying to reduce the need for chemical insecticides, crop breeders used traditional breeding techniques to create a celery that naturally warded off its pests. Ordinary celery contains about 0.8 part per million of psoralen, the plant’s natural pesticide. The new version contained 6.2 parts per million, more than seven times as much. Bugs did not like it a bit. But neither did the celery pickers, on whose hands and arms it caused rashes. The new celery was quickly withdrawn from the market. Later research found that the psoralen in celery was also carcinogenic when fed to rats and that an ordinary celery plant, if under stress from drought or insect damage, will increase its production of the toxin to 25 parts per million, 30 times the normal level.
Chemical insecticides are, from an insect’s point of view, no different from the plant’s own toxins. And their widespread use in modern agriculture has had a highly predictable result: the emergence of insects that are resistant to certain pesticides. Sprayed, they do not die. Sprayed with a double dose, they still might not die. They have won that particular chemical battle. How? Through mutation. Although a mutation in just the right gene to make an insect proof against a toxin is very rare, the number of insects in nature is very large—so large that the heavy use of insecticides rapidly selects for the ones with the right kinds of mutations. All the others die, and only the few that are resistant reproduce and multiply. But, as Victor Davis Hanson has seen in his vineyards, those few are enough. “Mites and hoppers breed geo-
metrically, not arithmetically,” he writes in The Land Was Everything. “Their reproductive cycles overlap, as populations go from hundreds to thousands to millions to billions in a vineyard in only days.” Again, Hanson and his fellow farmers must roll out their Faustian spray rigs. Only this time the poison must be different, for these billions of bugs are resistant to the old one.
Bt crops, in theory, can make farming safer for farmers (and, incidentally, for butterflies). Because the Bt genes protect plants from within, the use of the spray rig, and the accidents that accompany that use, should be less. But farm safety is not, in fact, behind the spectacular success of Bt corn. As professors at Colorado State University explain in their handbook on transgenic crops, “Field corn is not usually sprayed with insecticides because there is some market tolerance for insect damage on this kind of corn.” It is either fed to animals or ground and processed into corn chips, cornflakes, and cornmeal, so wormholes and other blemishes are not generally noticed. Worm-eaten sweet corn, on the other hand, is, like black-spotted grapes, hard to sell. “Sweet corn is sprayed with insecticides frequently, sometimes every two or three days, to ensure that the ears will be attractive at harvest.” But while Bt sweet corn has been approved by the EPA, it is not being grown.
Instead of replacing the chemical pesticides, Bt field corn has shown farmers the true cost of the damage done by the European corn borer. In its moth stage, the corn borer lays eggs on young corn plants, where the larvae hatch. They feed briefly on the leaves, then, as their name implies, bore into the cornstalk. The damage their tunnels do goes unseen and unchecked until, in a heavy rain or wind, the riddled cornstalk topples. Unless a farmer scouts his fields often, an infestation is easily overlooked until too late. Even the efforts of the 5 to 8 percent of farmers who did try to spray against corn borers were often without effect. Once the worm is in the corn, the poison cannot reach it. The corn borer can produce two generations in each crop cycle; in the South, it reaches three generations, or even four. The only sure way to control it is to stop planting corn altogether.
Bt corn first went on sale in 1996; as of June 2002 more than a third of the field corn in the U.S. (as well as more than 70 percent of
the cotton) were Bt crops. Bt varieties were sold not only by Monsanto, but by Syngenta, Aventis, Mycogen (owned by Dow AgroSciences), and Pioneer Hi-Bred (owned by DuPont). Bt corn did not immediately reduce farmers’ pesticide use. As the 2000 report to Congress on the benefits, safety, and oversight of agricultural biotechnology noted, this fact “is often cited by critics of biotechnology as an example of a bioengineered crop that has not met expectations.” Rebecca Goldburg of the Environmental Defense Fund, for example, complained to Congress that “Bt corn largely supplements rather than substitutes for insecticide use on field corn.” And yet few farmers had expected Bt corn to cut their use of sprays. According to a study by Iowa State University in 1998, 82 percent of the farmers in the Midwest who had planted Bt corn that year said their primary reason for doing so was to prevent losses from the corn borer. Those losses, according to the National Center for Food and Agricultural Policy, a nonprofit research organization, were equal to 3.5 billion pounds of corn by 2001, an amount worth $125 million.
Now that the enemy has shown its face, farmers are unlikely to ignore it once again if, for any reason, Bt corn becomes unavailable. Having seen how much more corn each acre yields when corn borers are eliminated, the National Center for Food and Agricultural Policy study suggests, farmers “would be likely to take the pest more seriously and apply insecticides more frequently than in prior years.” In a typical year, the study calculates, farmers would spray an additional 2.6 million pounds of pesticide to kill corn borers if Bt corn were not available.
Why would Bt corn become unavailable? Because of its spectacular success, Shelton and other entomologists have argued. Just like a chemical insecticide, Bt kills sensitive insects. Those insects with rare mutations that make them resistant to the toxin will be a larger fraction of the survivors. With the introduction of Bt genes into so many different kinds of plants (by 1999 18 crop varieties had been approved for field testing, and 16 companies were at work developing more), and with the acreage being planted in Bt crops expanding so rapidly, entomologists feared that Bt crops would select for resistance faster than anyone anticipated. An early estimate was that insects might become resistant in no more than five to seven years—or even less.
![]()
Because there are more than 170 different toxic Cry proteins, some scientists suggest that saying insects will become resistant to Bt is like crying wolf. Biochemist Milton Gordon of the University of Washington, for instance, writes that talking about Bt toxin “as a single compound is very similar to talking about all of the antibiotics that have been discovered and are now being used in humans as a single compound. If the pathogenic bacteria become resistant to one type of antibiotic, it is possible to switch to another type and still get good results. The same is true of Bt.”
And yet for the company that has developed a Bt corn variety, Gordon’s argument is little solace. Once insects become resistant to its toxin, a Bt crop would have no advantage over another, less expensive variety; both would need to be sprayed to protect them against the corn borer. Because developing and testing new Bt crops is fiercely expensive, it was important for companies to invest in strategies that would extend the useful life span of each Bt variety.
The EPA was also concerned. Part of its mission is to limit the environmental risks of pesticides and to promote safer means of pest management. Bt, used by home gardeners and organic farmers for decades, has a reputation for being a benign and environmentally friendly insecticide.
That reputation might be undeserved. Bacillus thuringiensis differs from another soil bacterium, Bacillus cereus, only in that B. thuringiensis contains a plasmid, an extra ring of DNA, that gives it the ability to make toxic Cry proteins, and B. cereus does not. Otherwise the two species are indistinguishable. A team of microbiologists from the University of Oslo who studied the genes of the two bacteria argued in a paper published in Applied Environmental Microbiology in June 2000 that they were, genetically, the same species despite the different names. The name scheme matters, they pointed out, because B. cereus is “an opportunistic pathogen that is a common cause of food poisoning.” The researchers found that a third Bacillus species should also be lumped together with B. cereus and B. thuringiensis: the third species is Bacillus anthracis. “B. anthracis,” they write, “causes the acute
fatal disease anthrax and is a potential biological weapon due to its high toxicity.” But for that one plasmid, the Bt whose spores are sprayed on organic vegetables is the same as anthrax. The plasmid is known to move into and out of Bt quite easily. The fact that the three bacteria belong to the same species is not just a matter of scientific interest: genes, including plasmids, are naturally shared among members of the same species.
That said, Bt remains the safest insecticide on the market, one that farmers are not even required to wash off of their produce before selling it. The loss of one Bt toxin, like the loss of one antibiotic, might not matter. Yet many of the Cry proteins engineered into plants are also found in Bt sprays. A bug that became resistant to the plant would also be unharmed by the spray. Consequently, a 2001 EPA report noted that the loss of a popular Bt toxin, if insects did become resistant to it, could have “serious adverse consequences for the environment”: conventional growers would have to shift to more-toxic pesticides and organic farmers would lose a valuable tool. Keeping pests susceptible to Bt, the EPA decided, was “in the public good.”
“This issue is not new to agriculture,” plant pathologist Jim Cook explains. “Resistance breeding is an ongoing effort for crops just to stay ahead of the ever-evolving populations of pest species.”
In The Living Fields, Jack Harlan illustrates this effort to stay ahead in the battle between wheat and the fungus Puccinia, which causes the disease called rust. “In North America,” he writes, “we managed to plant a carpet of wheat from northern Mexico well into the prairie provinces of Canada.” Although patches of other crops are mixed in, none is large enough to keep the rust spores from traveling on the wind from one wheat field to the next over a total distance of some 2,500 miles. Each year the disease hits first in the south, along the Rio Grande valley. The fungus germinates. Its thread-like mycelia consume the wheat’s stems and leaves, then set loose on the wind great numbers of spores. Some of these blow northward to the next wheat field, where their life cycle starts over. “The rusts cannot overwinter in the northern end of the wheat belt and must arrive each year from the south if wheat is to be rusted,” Harlan explains. Nor can the fungus withstand the summer heat in the south, “so spores must be blown southward to infect winter
wheat and renew the spring migration.” Our 2,500-mile carpet of wheat allows this yearly migration to happen. Says Harlan, “The system must be included among the marvels of the biological world.”
A Canadian-bred wheat called Marquis was planted across the northern plains in 1912 because rust did not harm it. “The rust epidemic of 1916 took it out,” Harlan says. A derivative of Marquis called Ceres was the favorite from 1926 to 1934. “Severe epidemics in 1935 and 1937 took out Ceres. The epidemic of 1935 is estimated to have cost 160,000,000 bushels in the USA alone.” Two other varieties, Thatcher and the aptly named Hope, held rust briefly in check, then in 1953 and 1954 the disease swept over North America again. By growing one favored cultivar—Marquis, Ceres, Thatcher, or Hope—over so many acres, we made it easy for the fungus to adapt. A cultivar lasted as little as four years before the fungus mutated and outfoxed it.
What is different today is that a new cultivar—a new Ceres or Thatcher or Hope—is much more expensive to develop than it was in 1916 or 1954. The costs are particularly high if the new cultivar is a genetically engineered variety, like Bt corn, which must weather extensive safety tests before it can be planted. A four-year life span cannot recoup a company’s outlay.
To protect their investment, the seed companies, with the help of the EPA, sought a workable plan to delay—or if possible, to avoid altogether—the onset of resistance to Bt crops. Their solution was one that Shelton had long studied and lobbied for: the establishment of insect refuges or refugia, areas planted in conventional, non-Bt crops, within or adjacent to each cornfield.
In a refuge the pests—the corn borers in this case—are allowed to multiply. Most of them are sensitive, susceptible to the Bt toxin. A larva with the lucky mutation that makes it resistant to Bt enjoys no advantage there in the refuge, because it is never exposed to Bt. It takes its chances along with its kin and might or might not live long enough to metamorphose into a moth, fly away, and mate.
Next door in the Bt fields, however, most insects die before reaching maturity. Among those few that survive and transform into moths, there’s a much greater chance that one is resistant—simply because the others have succumbed to the toxin. But with all the normal, sensitive
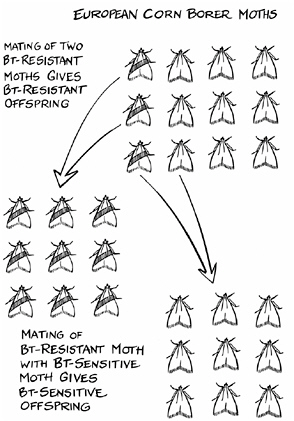
How a refuge delays the onset of resistance to Bt
moths now emerging from the nearby refuge, the odds are that this resistant survivor will mate with one of them. The offspring of such a mating should be sensitive to the toxin, having one resistance allele (the gene from the resistant parent) and one sensitivity allele (from the parent from the refuge). When such a heterozygote eats a Bt plant, it dies, eliminating the resistant allele from the population.
It dies, that is, if the resistance trait is recessive, like the wrinkled peas in Mendel’s famous experiment. When Mendel crossed peas with round seeds and peas with wrinkled seeds, he found that the seeds of three-quarters of the offspring were round. Only if the peas received two alleles for the wrinkled trait would it appear. Roundness was dominant. If the offspring received one roundness allele and one wrinkliness allele, the wrinkliness would seem to disappear, masked by the dominant trait. For the larva of a European corn borer in a Bt cornfield, Mendel’s experiment means
that a heterozygous insect, one with each type of allele, is doomed because resistance, like wrinkliness, is recessive.
This assumption has been tested by a number of researchers. They selected Bt-resistant insects and allowed them to breed in the laboratory. In almost all of the experiments, the resistance trait was, indeed, recessive. One report in 1999, however, suggested that a kind of resistance that is dominant—which wouldn’t be controlled through the use of refuges—can occasionally arise.
To test the refugia strategy under true field conditions, Shelton and his colleagues at Cornell University released diamondback moths into plots of broccoli plants carrying a Bt toxin gene. Shelton chose diamondback moths instead of corn borers for two reasons. First, the diamondback had already shown some resistance to Bt and the trait was known to be recessive. (Because the resistance was apparent by 1993, three years before any Bt crops were on the market, it was clearly a response to the Bt sprays that organic gardeners and others had used for decades.) Second, the diamondback is a warm-climate moth. The harsh New York winters would act like the walls of a greenhouse to contain the experiment: any moths that escaped the test plots would die when the cold weather came.
In 1996 and 1997 Shelton and his colleagues monitored the diamondbacks in their Bt broccoli fields. As they reported in 2000, the refuge strategy worked very well—if it was used carefully. In pure stands of Bt broccoli, with no refuge set aside, the moths quickly became highly resistant to Bt. But the larger the refuge, the longer it took for that resistance to develop. It also mattered where the refuge was. A separate, well-defined refuge, around the edges of the field or even up to a half mile away, was more effective in keeping the level of resistance in the moth population low than one created, for example, by planting every other row with the Bt crop.
The separate refuges worked as expected because although diamondbacks travel only short distances in their leaf-eating larval stage, they cover a much wider territory as winged moths looking for mates. When the two types of plants, Bt and non-Bt, were mixed together randomly in a single plot, the diamondback larvae naturally wandered from one plant to the next. The ordinary, sensitive larvae that moved
onto a Bt plant and started to munch quickly died. The resistant larvae that found the same plant lived. The result was that only the resistant larvae lived long enough to become moths and breed, passing on their resistance genes. If, on the other hand, a patch of non-Bt broccoli were planted far enough away from the Bt broccoli that the larvae could not wriggle from one to the other, many sensitive larvae also lived to adulthood. Once winged, these sought out and mated with their resistant cousins in the Bt broccoli patch and, following Mendel’s laws, the recessive resistance trait was found in many fewer of their offspring. In 2001 the EPA included Shelton’s and other scientists’ recommendations in its rules for registering Bt corn.
In order to buy Bt corn seed, farmers must sign grower agreements or stewardship agreements, which “impose binding contractual obligations on the grower to comply with the refuge requirements”—that is, farmers who plant Bt corn but fail to establish proper insect refuges can be sued. The onus for educating farmers about proper stewardship—and for ensuring their compliance—falls on the seed company. Any company selling Bt corn seed has to monitor the success of its stewardship plan and report to EPA any “statistically significant and biologically relevant” changes in the corn borer’s susceptibility to the Bt toxin its variety expresses. It has to have ready a “remedial action plan” in case resistant insects are detected. And finally, it must submit annual reports to the EPA on its sales, its educational programs, the results of its stewardship plan, and the extent to which its farmers complied with that plan.
In the Corn Belt (but outside cotton-growing regions, where the existence of Bt cotton makes the rules even more exacting), the stewardship agreements between the seed company and the farmer specify a refuge that covers at least 20 percent of the farmer’s cornfields (it’s 50 percent in the South). This refuge can be laid out as whole fields, as blocks within fields (for instance, as a border along the edges), or as strips, at least four rows wide, across the field. If whole fields, they must be within half a mile (closer is better) of a Bt field.
A refuge can be sprayed for European corn borer and similar pests “only if economic thresholds are reached”; these thresholds are to be determined “using methods recommended by local or regional profes-
sionals” such as Cooperative Extension Service agents or crop consultants. Under no circumstances can the spray be any form of Bt. No conventional pesticide has such extensive—and expensive—requirements for managing insect resistance, yet as the case of the diamondback moth shows, insects respond to dusts, powders, and sprays by becoming resistant to the pesticide just as they do when eating Bt corn.
Leaving the refuge unsprayed means farmers must stand by and watch while pests eat 20 percent or more of their cornfields. This kind of compromise, between a short-term sacrifice and a long-term gain, can be hard for a farmer to make. As California grape farmer Victor Davis Hanson writes,
I once sprayed dimethoate, and in between the stinking loads, as the 500-gallon spray tank refilled, I read the Inferno, wondering whether I was in Hell or earning my way. I have dusted organic Dipel six times on vines and watched worms sicken but not die. I have sprayed the toxin Lannate once and watched them drop off before the tractor left the field. Yes, we know it is legal but wrong. Yes, we know it is expedient but nonsustainable for the millennia. Yes, we know that it costs money we don’t have. Yes, we know there will be nemeses to confront for doing what we should not; we know that he who pollutes his land must atone tenfold in the hereafter. But farmers as a last resort use terrible chemicals because they do kill, no questions asked. And there are some times in farmers’ lives when there are insects, millions of them, that must be killed and killed quickly if their brethren are to eat cheaply and plentifully and on schedule—if one man is to feed 99 other Americans 3,000 miles away by next Tuesday.
In 2000 a telephone survey commissioned by the major seed companies, Aventis, Dow, DuPont, Monsanto, and Syngenta, found that 29 percent of the farmers growing Bt corn “broke the rules.” The refuges they planted were either too small or too far away. Many farmers pleaded ignorance of the rules. A third of the Bt corn growers in the Midwest and more than half of those in the South couldn’t say what size of refuge was required in their area; 60 percent couldn’t say how far away the refuge should be; and two-thirds said they didn’t know they couldn’t spray it with Bt. In 2001 the situation improved somewhat: 13 percent of farmers in the Midwest’s Corn Belt and 23 percent of farmers in the South (where the restrictions are tighter because of the proximity of Bt cotton) were “out of compliance.” Still, only just over a third, when quizzed, knew the rules for making a refuge in their
area. The 2002 growing season saw a closing of the knowledge gap—only 12 percent of the 550 Bt corn farmers surveyed remained unaware of the rules—but 14 percent still didn’t comply.
An independent survey by the Center for Science in the Public Interest found that 19 percent of farms did not comply. Thirteen percent planted no refuge at all. “One reason for the discrepancy,” noted the New York Times, “was that the industry surveyed only large farms. The center also looked at small farms, which had a higher rate of non-compliance.” Those small farms—on the scale of Victor Davis Hanson’s fifth-generation family farm—accounted for only 8 percent of the Bt corn grown. Yet, as the Center insisted, the requirement is for each farm, not for each county or region.
Before the Center’s report came out—and before seeds were sold in 2003—the EPA stiffened the rules. Seed companies are now required to enforce the stewardship agreements the farmers sign by conducting on-farm visits. They are required to help the farmer design an appropriate refuge, and they are required to check, the next growing season, to see if the farmer complied. Farmers who are “significantly out of compliance” for two years will no longer be allowed to buy Bt corn.
In spite of the numbers of farmers who have broken the Bt corn rules, the crop is still winning the chemical battle for survival. Approving the re-registrations of five varieties of Bt corn in 2001, the EPA noted, “Available data indicate that after six years of commercialization, no reported insect resistance has occurred to the Bt toxins expressed either in Bt potato, Bt corn, or Bt cotton products.” If the refugia rules are followed, scientists predict that the corn borer will not evolve to be resistant to Bt corn for at least 99 years.
One of Shelton’s collaborators, Richard T. Roush, now at the University of California at Davis, believes that the wider use of some techniques already available to plant breeders could keep insect resistance at bay even longer. He suggests designing plants that express Bt toxins only when they are turned on by an otherwise environmentally harmless chemical spray. Or that express Bt only in certain parts of the plant.
But even without these enhancements “the success of Bt crops exceeds expectations,” concluded a study published in 2003 by entomologist Bruce Tabashnik of the University of Arizona and seven other
scientists, including Shelton. In laboratory and greenhouse tests, three pests have been found that are completely resistant, able to live and reproduce on Bt crops. None of these have yet been found in the field. Even pests living in Bt crop fields that have been continuously monitored for five and six years have shown no signs of an increase in the frequency of resistance.
Discussing the study with a reporter from Nature Biotechnology News, Tabashnik said, “If I’d gotten up seven years ago and said that there would be no evidence of increased Bt resistance after Bt crops were planted on 62 million hectares”—meaning the cumulative number of hectares since Bt crops were released in 1996—“I would have been hooted off the stage. No one predicted that there wouldn’t even be a minor increase, which is extraordinary.”