Schrödinger's Rabbits: The Many Worlds of Quantum (2004)
Chapter: 2 Clinging to the Classical
CHAPTER 2
CLINGING TO THE CLASSICAL
What is the real-life manifestation of the problem that has gotten scientists in such a spin? It started relatively innocuously about a century ago, with a new twist in an ancient debate—about whether light was composed of waves or particles.
This question had been considered settled at the end of the 18th century, through an ingenious experiment by the British natural philosopher Thomas Young, which involved passing light through slits. When a wave passes through a narrow slit, it tends to spread out on the other side. You can see this happen when a water wave passes through the gap in a harbor wall. It does not just continue on its original straight-line track, but spreads out so that all the boats in the harbor end up bobbing up and down. Light behaves in just this way when it passes through a narrow slit.
Particles don’t generally do the same, but it’s easy to envision how they could be made to. Suppose you were rolling bowling balls toward a narrow gap in a fence. It would be easy to place some springy twigs around the gap so that the bowling balls were deflected by random angles as they passed through. Then a stream of bowling balls being rolled toward the gap would spread out over a range of angles on the far side, just as a wave does. It was evident to Young and others that if
light consisted of a stream of particles, these might be scattered when passing close to solid matter (as when passing through a narrow slit) by something analogous to the springy-twigs effect. So the spreading is not in itself convincing evidence whether light consists of waves or particles.
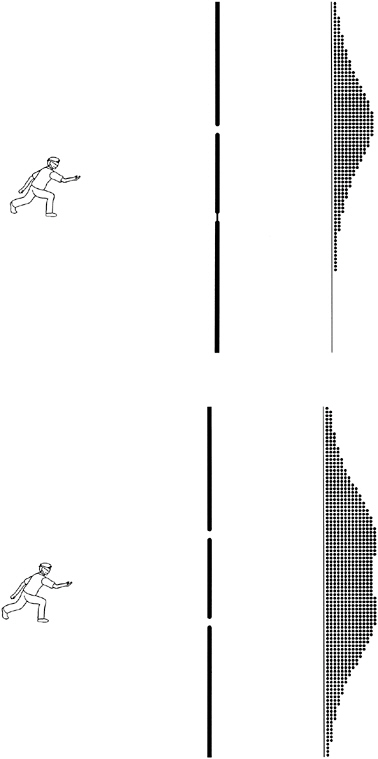
However, a cleverer experiment involving two slits appeared definitive. Imagine a blindfolded man rolling bowling balls toward a fence in which either or both of two narrow gates might be open. The gates have springy twigs placed so that any ball passing through a gate is deflected by a random angle; behind the gates is a line of catchment trenches into which the balls fall. It is fairly obvious that the effect of opening both gates is that each trench gets the sum of the balls it would have gotten if only the left gate was open and those it would have gotten if only the right gate was open, as shown in Figure 2-1. Certainly, closing a gate can never increase the number of balls going into a given trench. The bowling balls are of course behaving like particles.
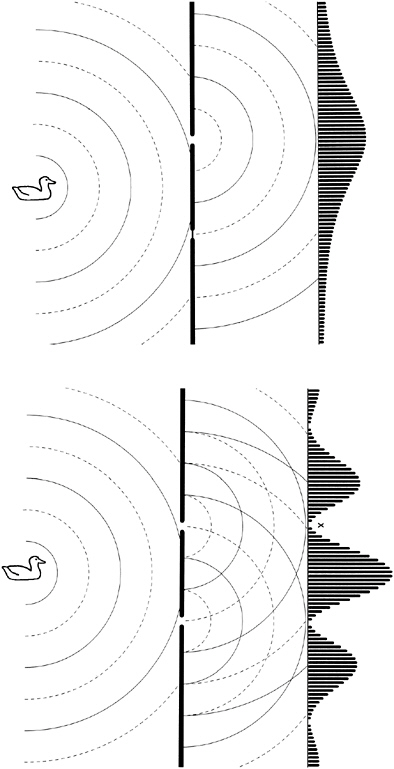
But now suppose we do a similar experiment with waves. For example, we could flood the bowling green and generate water waves of a particular wavelength, as shown in Figure 2-2. As waves strike the barrier at the back, water slops over it (more where the waves are higher, obviously), gradually filling the catchment trenches.
When only one gate is open at a time, the accumulation of water after an appropriate number of waves have been generated is very similar to the result obtained with the bowling balls, as shown at the top of Figure 2-2. But when both gates are opened simultaneously, something quite different happens. Now some trenches that got quite a lot of water when only one gate was open get less, or even none at all.
A little thought reveals why. At points like X, the peak of a wave from one gate always coincides with the trough of a wave from the other. (Peaks are shown as solid lines, troughs as dotted lines.) This leaves the net water depth unchanged at all times, so no water flows over the barrier. The waves from the two gates are said to cancel at such points, and this phenomenon is called interference. This is behavior that particles cannot possibly exhibit; opening an extra gate never reduces the quantity of balls reaching any trench. Young realized that this was a neat way to distinguish waves from particles. When he tried
the two-slit experiment with light, the results corresponded to Figure 2-2. A pattern of light and dark stripes was visible at the back of the apparatus, and points like X received no light at all. An age-old debate appeared to have been settled; light definitely consisted of waves.
![]()
But more than 100 years later, at the start of the 20th century, this picture was thrown into confusion. By then, it was known that solid matter was composed of the tiny particles the Greeks had hypothesized, called atoms, and moreover that atoms were composed of positively charged central nuclei and negatively charged particles called electrons. Electrons could be detached from their parent atoms and made to flow about within a solid material, as when an electric current flows down a wire, and even sprayed into empty space, as happens inside a TV tube. It had become possible to do experiments that involved light interacting directly with electrons. This is not a history book, so I am going to describe only the most definitive of these experiments, which is now called the Compton effect.
Back in the 1920s, Compton arranged to spray electrons into a vacuum, and then shine a bright light of a particular color onto them at right angles as shown in Figure 2-3. It had long been known that light radiation carries momentum as well as energy, so that light shining on a surface exerts a slight pressure. The pressure is small by ordinary standards; if you hold your cupped hands up to the Sun, the force on your palms is about a millionth of an ounce. Nevertheless, light pressure is strong enough to propel a kind of spacecraft called a solar sail, and certainly strong enough to deflect a beam of lightweight particles like electrons.
If light consisted of waves, it would be reasonable to expect that all the electrons would be deflected by a similar amount, as on the left of Figure 2-3. But what really happens is quite different, as shown on the right. Most of the electrons are completely unaffected. But an occasional electron is deflected by a large angle. This is characteristic of two streams of particles intersecting. Think of the electrons as cannon shells and the photons as lighter but faster machine-gun bullets. If a cannon shell happens to be hit by a bullet, it is deflected quite sharply,
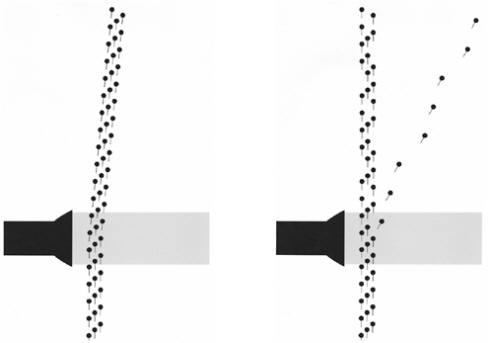
FIGURE 2-3 Stream of electrons intersects a beam of light: two possible outcomes.
but all those cannon shells that are not hit proceed on exactly their original course. Compton’s result implied that light consisted of bullet-like particles. If a particle of light happened to hit a particular electron, then that electron was deflected. These particles of light are nowadays called photons.1
How could this be? When light is traveling, it behaves like a wave, spreading out to explore every possible route open to it as a wave does, even if these routes are centimeters (or, for that matter, kilometers) apart, as in a two-slit experiment. But when light strikes something, it appears at very specific points, like hailstones striking a pavement rather than floodwater washing across it.
One obvious possibility was that light is indeed composed of photons, but the photons are so numerous that they somehow interact, jostling one another so as to give rise to wavelike behavior. After all, the kind of wave most familiar to us, a water wave, is just the visible result of many tiny particles moving together, pushing against one another as they do so. Just as atoms are very small physical things, pho-
tons are very tiny packets of energy. A lightbulb emits about 1020 (that stands for one followed by 20 zeros, 100 billion billion) photons of visible light every second. This is roughly the same as the number of atoms in 1 cubic millimeter of solid matter. Perhaps just as billions of air molecules jostling one another can produce a sound wave, and billions of water molecules jostling one another can create a geometrically perfect ripple on the surface of a liquid, billions of photons jostling one another could produce light’s wavelike action?
Nobody was very happy with this picture, though. The problem is that there are not really enough photons around to produce wavelike interactions. That might sound paradoxical—1020 is a huge number—but let’s do some figuring. Photons travel so fast that a photon emitted from a lightbulb in an ordinary room has a lifetime of only a few billionths of a second before it hits something or escapes through a window, meaning that there are some 1012 photons present in the room at any time. That’s a density of only about 10 photons per cubic millimeter, compared to 1016 air molecules per cubic millimeter.
Another way to look at it is that if we put a soap bubble with a radius of 1 meter and a thickness of 1 wavelength of visible light around the bulb, its skin would contain only 100,000 photons at any instant—only 1 per square centimeter. Yet if photons really were particles, they would have to be tiny things. An appropriate unit of measure to use here is the Angstrom, 1 ten-billionth of a meter. The atoms in a typical solid are 2 or 3 Angstroms apart. When a photon hits a solid, it usually interacts with just a single atom. A particlelike photon would therefore presumably be, at most, 1 Angstrom in diameter. Could such a tiny thing really jostle other corpuscles millimeters or even centimeters away from it? The problem gets worse when you take into account that, even with naked-eye observation, light’s wavelike behavior can be seen at illumination levels thousands of times less than a brightly lit room, when individual photons are centimeters or even meters apart.
In fact, photon jostling can be ruled out altogether. With slightly more modern technology than Young’s, we can lower the level of illumination inside a two-slit apparatus to the point where there can only be a single photon in it at any given time, and place sensitive photo-
graphic film at the back. We leave the experiment to run for a while, then develop the film. The pattern of light and dark stripes is still visible on the film. Somehow each and every photon, a thing so tiny that it interacts with just one atom when it strikes a solid surface, has had its trajectory influenced by the presence and position of both slits. How could each photon possibly have explored, or somehow been aware of, both possible routes? Figure 2-4 shows the contrasting pictures of light as consisting of waves on the one hand, and photons on the other. The left picture shows light as it typically behaves in flight, the right as it typically behaves when it hits something.
Many textbooks describe this as behavior that cannot be explained in terms of any classical picture, a picture in which some kind of behind-the-scenes machinery does definite things at definite locations and times. But that is an oversimplification. Let us demonstrate a determination that is going to guide us throughout this book. We are going to stick stubbornly to the notion that we will explain what is going on in a commonsense, visualizable way. There is such a way to explain the behavior of light going through a two-slit apparatus, and Einstein, among others, was fond of it.
The concept is called pilot waves. Suppose that any light source
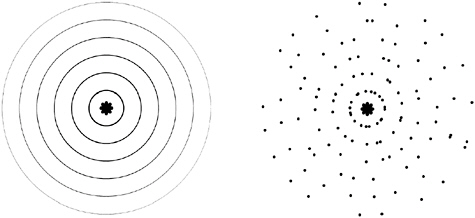
FIGURE 2-4 Two contrasting pictures of light from a point source. Is it emitted as concentric waves, like the ripples from a fisherman’s float bobbing up and down in the water, or as individual photons flung off in random directions like sparks from a firework?
actually emits two kinds of thing. The first are waves as shown on the left of Figure 2-4; however, the waves themselves are completely invisible and imperceptible to us. But the light source also emits photons, as shown on the right. The trajectories of the photons are guided by their interactions with the invisible waves.
Let us return to the bowling-green picture of Figures 2-1 and 2-2. Suppose we flood the bowling green as in Figure 2-2—but now throw a bowling ball into the water. The ball’s motion generates a gentle wave, and the ball travels along with the wave, being guided by it. The bowling ball can obviously go through only one of the gaps in the fence, but the wave goes through both, and continues to guide the ball to its final destination. Although the bowling ball is always in one place, the wave has explored both possible routes, and a pattern like that in Figure 2-2, but with the trenches now full of bowling balls rather than water, can arise quite naturally. We have solved the wave-particle paradox! (We’ll assume that the bowling balls are light enough to float. You might like to think of the ball as a surfer riding a wave, who prefers to be at the highest point of the wave. He is not perfectly successful, but is most likely to be found where the wave is highest, least likely where it is lowest.) 2
![]()
As Compton experimented further with his electron-deflecting apparatus, he confirmed another property of photons. Increasing the intensity of the light increased the number of electrons knocked aside, but not the amount by which each electron was deflected. The greater intensity increased the number of photon-particles, but not the amount of momentum carried by each. On the other hand, changing the color of the light did change the angle by which each electron was deflected. Blue photons knock electrons aside at almost twice the angle that red photons do, indicating that each blue photon carries twice as much momentum or “punch” as a red one.
It had long been known that the color of light is really just the way we perceive its wavelength. For example, blue light has a wavelength of approximately 4,000 Angstroms, and red light approximately 7,000 Angstroms. Compton’s result verified that the momentum of indi-
vidual photons is related to the wavelength of the light involved—the shorter the wavelength, the more the momentum and energy carried by each individual bullet of light. The actual formula is this:
Wavelength = 6.62 × 10−34/Momentum
(The quantity 6.62 × 10−34 stands for 6.62 divided by the number 1 with 34 zeros written after it, that is, .000000000000000000000000000000000662. This quantity appears in many equations of modern physics, and is known as Planck’s constant.)
This leads to a curious thought. Why should this formula apply only to particles of light, and not to particles of matter as well? If it does apply to solid objects, then the wavelength associated with large things like bowling balls will be incredibly tiny. But the wavelength associated with minute things, like atoms when they are moving slowly, will be quite large. It turns out that when we repeat the bowling-ball experiment of Figure 2-1 on a small enough scale, using individual atoms as the balls, then the results are again like those of Figure 2-2. An atom that can sometimes get to X when one gate is open cannot do so when both gates are open! Just as the waves of light can also behave as discrete particles, so the discrete particles of solid matter can also behave as if they were waves.
Once confirmed, the wavelike behavior of matter solved some tough problems that had confronted the early atomic theorists. An early model of the atom—still seen in pictures today—resembled a tiny solar system, with electrons circling the central nucleus like planets circling the Sun. But whereas real solar systems are all slightly different from one another, atoms of the same type all behave in exactly the same way. Take the most basic atom, hydrogen, a single electron circling a single proton. Rather than orbiting the proton at any arbitrary distance, as a planet could, the electron can occupy only certain orbits or energy levels. When the electron switches between two orbits, the amount of energy emitted is therefore always one of a few exactly predictable quantities. This cannot be explained by a purely particle-like electron. If the electron has a wave associated with it, however, then the math predicts that only certain wavelengths will be stable, and therefore describe allowed orbits for the electron, just as a
bell can vibrate stably only at certain frequencies corresponding to its harmonics.
This triumph, explaining the quantization of atomic energy levels, is what gives quantum theory its name. But I would like to stress that this wavelike behavior does not apply just to tiny objects like atoms and molecules, but to objects at any scale. To illustrate, I am tempted to ask you to imagine a wall with two slits in it, and a gun capable of firing a cat toward the arrangement, but cats (even hypothetical ones) have already suffered enough in the cause of quantum physics, and Stephen Hawking has threatened to shoot people who mention Schrödinger’s cat to him, so I will choose an alternative. I have visited a Rolls-Royce factory where they test their jet engines’ ability to survive bird impacts. The apparatus they use is a kind of catapult that fires oven-ready chickens (an accurate model for the largest kind of birds that an aircraft is likely to hit, and available in a range of sizes at the local supermarket) at random angles toward an engine on a test rig. Suppose we remove the jet engine and replace it with a brick wall with two slits in it. Every time a chicken gets through to the far side of the hangar beyond the wall, we make a chalk mark at that point. Eventually we would expect to see a pattern like that of Figure 2-2. With chickens, the scale of the pattern would be incredibly fine, far too fine to measure practicably, but it would be there.
With lightweight particles like electrons, however, the experiment can easily be done. If the experiment shown in Figure 2-1 is done with a source of electrons of appropriate momentum, and hence wavelength (which works out to be electrons traveling at about 1 mile per second, a rather modest speed for an electron), we get an interference pattern as shown in Figure 2-2, at exactly the same scale as one produced by visible light. While they are flying through free space, electrons behave like spread-out waves. Only when they hit something do they remanifest themselves as pointlike objects. Yet we know from other experiments that electrons are much tinier even than atoms. In fact, they are perfectly pointlike insofar as anyone has ever been able to detect. How can this be?
By now I am sure there is an answer on the tip of your tongue—pilot waves! Every time you let fly with an electron (or for that matter
with an oven-ready chicken) the action also generates an invisible wave, which guides the subtle motion of the object. This possibility was taken seriously by many physicists at one time, and still is by a few. But guide waves for solid objects raise conceptual difficulties that are not present (or at least not so apparent) when photons are involved.
In the case of a photon, the point where the guide wave comes into existence is well defined. It is created together with its photon when radiant energy is emitted, and effectively dies (or at least ceases to have significant effects on the rest of the universe) when that photon is absorbed. The photon then momentarily appears at one definite point in space—following the period of travel on the guide wave when its whereabouts were unknown—and expires, donating its energy at that particular point. The image of a hapless surfer finally splatted against a harbor wall is unavoidable. After that, of course, it does not matter what happens to the pilot wave. Its only discernible effect ever was to guide the photon; once the photon is gone, you can think of it as passing on to infinity without any subsequent effect on the rest of the universe.
Particles like protons and electrons, by contrast, have very long lifetimes, typically comparable to the age of the universe, during which their initial guide waves presumably continue to exist, spreading farther and farther throughout space. But we do not need to destroy an electron or a proton in order for it to turn up in some definite place during that time.
What causes a particle like an electron to become localized, and appear in one place rather than another? The theoretical answer to that question is deep and problematic. But the immediate empirical answer could not be more straightforward. The electron’s location becomes definite when an experimenter measures it! Until such a measurement is made, the electron could be anywhere on its guide wave; afterward, its location can be known (at least temporarily) to an arbitrarily high degree of precision. This sudden localization is a form of what is called quantum collapse.
Such measurement has a curious side effect. It effectively knocks the particle you are measuring off its guide wave. If the blobs in Figure 2-1 represent particles, such as electrons or oven-ready chickens, then any attempt to measure the trajectories of the particles destroys the
interference pattern shown in Figure 2-2; instead we again get a result like that in Figure 2-1. It seems that any kind of stuff (whether light or solid matter) can behave either as waves or as particles, but never as both at the same time. If we look at the particles, to try to see which slit they are going through, the wave effects disappear.
At first this sounds like a very strange effect. But what do we really mean when we say that we “look at” the particles? In experimental practice, this translates as: We shine a bright light on them. With normal levels of light, we can see which way an oven-ready chicken is going; with sufficiently bright light, we can even see which way electrons are going. When we do the two-slit experiment with electrons, a perfect interference pattern appears only if the experiment is done in the dark. The brighter the light shone on the electrons, the fainter the interference pattern produced. This washing out of the pattern has nothing to do with whether anyone is watching—be it a so-called conscious observer, a cat, or a camera. We already know that light can affect electrons. There is no reason to assume that anything mystical is going on. It just so happens that the point at which the light becomes bright enough that we can start to tell which way each electron is going is also the point at which the interference pattern starts to disappear.
There is a curious corollary to the wavelike behavior of particles. We find that however bright a light we shine on a small particle like an electron, we can never pin it down perfectly, in the sense of simultaneously knowing its exact position and its exact motion precisely. This, as many readers will recognize, is Heisenberg’s famous uncertainty principle in action. But there is a way to explain this, too, in terms of guide waves. A particle can never be completely divorced from a guide wave—in terms of our poetic surfboarder analogy, the surfer always determinedly climbs back on and finds a new wave, however often he is knocked off the old one. Trying to measure the position of the surfer-particle exactly is like trying to squeeze the entire guide wave into a very small space. Much as when the soap in the bathtub tries to escape as you close your hands about it, amplifying the effect of any waves in the tub, so trying to squash a particle’s guide wave into a small space tends to induce it to a higher speed.
Just as water waves can make a floating cork bob about a great deal while having no discernible effect on a big ship, Heisenberg’s un-
certainty principle is much more noticeable with small things, like electrons and atoms, than with large things like bowling balls and cats. In this respect, Heisenberg uncertainty is analogous to the phenomenon called Brownian motion: When small things like pollen grains floating in air are observed under a powerful microscope, they jitter around because the number of air molecules which are at all times striking them from different sides is subject to statistical variations. Just as you do not always get exactly 10 heads and 10 tails when you toss a coin 20 times, in any given millisecond the pollen grain might be struck by slightly more atoms on one side than the other. For objects large enough to see with the naked eye, however, Brownian motion becomes negligible. Heisenberg uncertainty is a bit like Brownian motion at a yet smaller scale, as if atoms themselves were being knocked around by particles even tinier and harder to discern.
So, where are the famous conceptual difficulties of the quantum world? All the phenomena we have encountered so far—the two-slit experiment, Heisenberg uncertainty, even the dreaded quantum collapse—can be explained merely by postulating some kind of fine structure to space that is too delicate to measure directly, at least with present-day instruments. This hypothetical fine structure (the technical term for it is “hidden local variables”) supports waves that can influence the motion of both photons and more solid particles and make small objects judder about so as to complicate the measurement of their positions and motions. Abrupt collisions jolt particles loose from the waves they are currently associated with.
We are doing very well at drawing a purely classical picture of quantum behavior. Where has the weirdness gone?