Review of the Long-Term Operations of the Central Valley Project and the State Water Project (2026)
Chapter: Appendix E: Listed Fish Species
Appendix E
Listed Fish Species
This appendix discusses the seven fish species in the California Bay-Delta that are listed under the Endangered Species Act (ESA) or the California Endangered Species Act (CESA; see Appendix C), along with one nonlisted but economically valuable fish. These species include Delta smelt, longfin smelt, Sacramento River winter-run Chinook salmon, Central Valley spring-run Chinook salmon, Central Valley fall-run Chinook salmon (currently unlisted), California Central Valley steelhead (CCV steelhead), the southern distinct population segment (sDPS) of North American green sturgeon, and white sturgeon (a candidate for listing as of July 2024). For each fish, this chapter briefly describes its life history, monitoring of species and abundance data, conceptual models and quantitative life-cycle models for the species, the effect of the Central Valley Project (CVP) and State Water Project (SWP) operations on the species, the role of hatcheries in maintaining the population, and key uncertainties that lead to science needs for the species.
In 2023, the U.S. Bureau of Reclamation (USBR) and the California Department of Water Resources (CDWR) reinitiated ESA Section 7 consultation for the Long-term Operations of the CVP and SWP. As part of that process, USBR prepared an environmental impact statement (including a Biological Assessment for each fish species) to assess how alternative management actions related to the long-term operations of the CVP and SWP would affect a suite of life stage-specific stressors of the ESA-listed species and their critical habitats. Information in this chapter draws largely on the Biological Assessments for six of the listed species (USBR, 2024a,b,c,d,e,f), and the reader is referred to those documents1 for more detailed information.
DELTA SMELT
The Delta smelt (Hypomesus transpacificus) is a small (60–70 millimeter [mm]), silvery-blue fish in the family Osmeridae (distantly related to salmonids). Delta smelt are endemic to the Sacramento-San Joaquin Delta, where they were historically found from the San Pablo Bay upstream into the Central Valley. They are adapted to a broad range of environmental conditions and use a wide variety of Delta habitats to complete their different life stages (reviewed in USBR, 2024a).
The Delta smelt was once considered one of the most abundant fishes in the Delta and as such was considered an indicator of ecosystem health (Bennett, 2005; Skinner, 1962; Sweetnam et al., 2001). However, the conditions
___________________
1 See https://www.usbr.gov/mp/nepa/nepa_project_details.php?Project_ID=54661.
of the Delta ecosystem have been altered to such a degree that recruitment (the transitioning of a fish from the juvenile life stage to the mature life stage) of Delta smelt is nearly absent, and Delta smelt are now extremely rare, with zero having been caught in many catch indices in recent years (USBR, 2024a). They may be locally, functionally extinct, meaning that there may be a few individuals still living, but the species is no longer able to produce healthy offspring due to low numbers and is no longer playing a significant role in the ecosystem (e.g., Jørgensen, 2002).
In 1991, the U.S. Fish and Wildlife Service (USFWS) proposed to list the Delta smelt under the ESA as threatened with proposed critical habitat; the threatened listing occurred in 1993 (USFWS, 1993) and the designated critical habitat listing for the species occurred in 1994 (USFWS, 1994). The species was listed as endangered under the California ESA in 2009, although it is still only listed as threatened under the federal ESA.
Life History and Stages
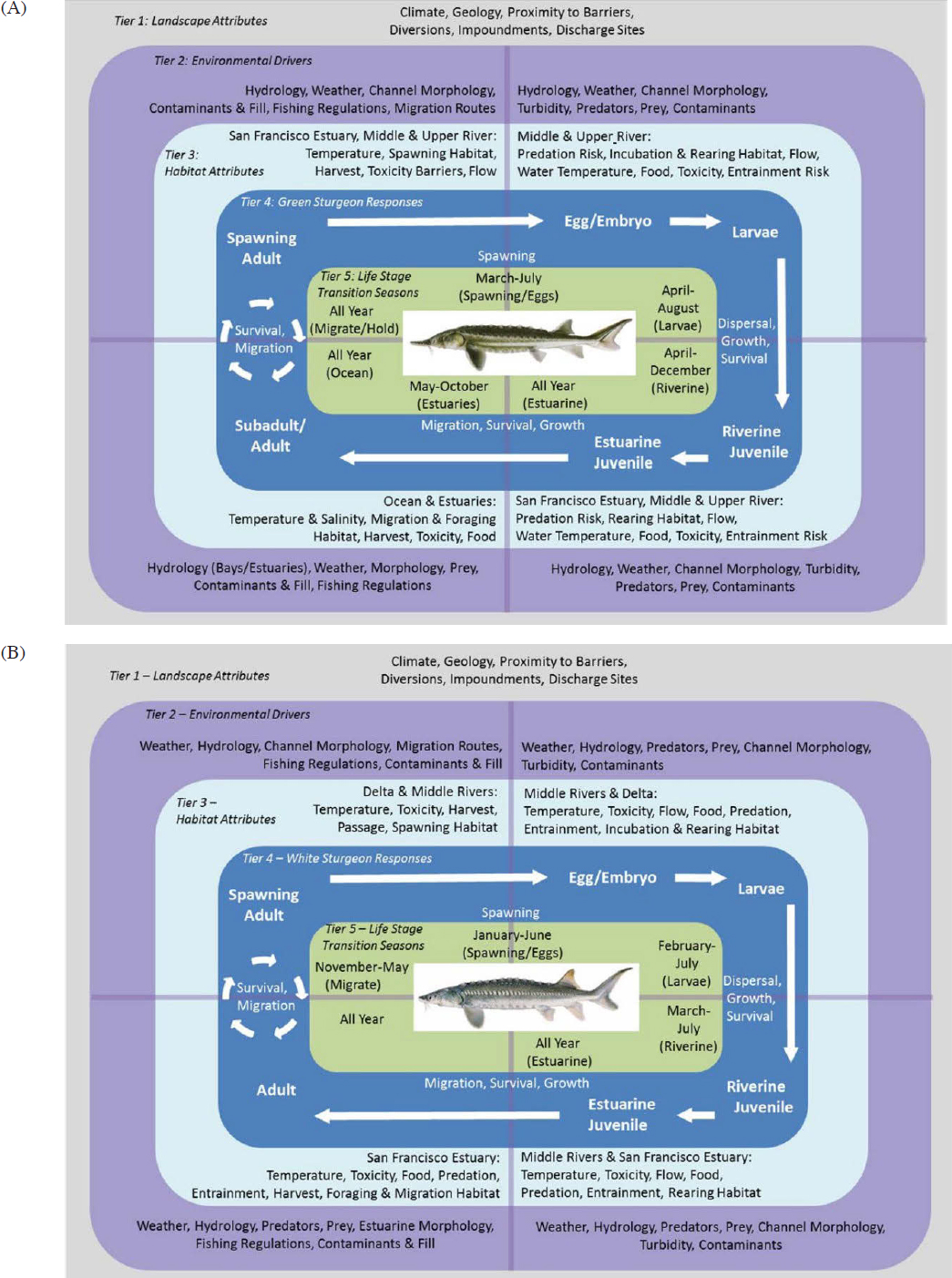
The Delta smelt is primarily an annual species that completes its life cycle in one year, beginning in approximately March to the following March, a window that varies by one to two months (Moyle et al., 2016). Delta smelt has been described as a semi-anadromous species, migrating from brackish low-salinity (1–6 psu) habitats downstream of the confluence of the Sacramento and San Joaquin rivers to tidal freshwater habitats in the Delta (see conceptual model in Figure E-1). However, recent analyses have demonstrated substantial variation in life-history expression (phenotypes) including a freshwater resident, brackish-water resident, and semi-anadromous fish; of the most recent viable cohorts, the greatest proportion were semi-anadromous (Hobbs et al., 2019). The low-salinity

SOURCE: IEP MAST (2015).
zone, which migrates up- and downstream depending upon Delta water outflow (e.g., Dege and Brown, 2004), has been changing in areal magnitude and physical characteristics such as turbidity, salinity, and temperature in ways that appear to be to the detriment of Delta smelt (USBR, 2024a).
Historically, most Delta smelt spawning was thought to occur in upstream freshwater areas that are tidally influenced backwater sloughs and low-velocity channel edgewaters. The eggs are adhesive and are believed to be batch-released over firm substrates or sand.2 The adult fish generally migrate upstream in the winter, following the first flush of turbid freshwater from precipitation, to then spawn in the spring. The first-flush period preceding the winter migration coincides with increased freshwater availability and elevated pumping rates to fill storage reservoirs for the dry season; thus, migration is a critical life stage during which adult Delta smelt can be entrained into the South Delta and possibly be salvaged at the export facilities.
Delta smelt are primarily planktivores, feeding on small, free-floating (pelagic) crustaceans and also sporadically on insect larvae and even larval fish. Traditionally, the main prey of Delta smelt was a copepod (Eurytemora affinis), but they also feed on cladocerans and mysid shrimp (Neomysis mercedis; reviewed in USBR, 2024a).
Monitoring for Delta Smelt and Abundance Data
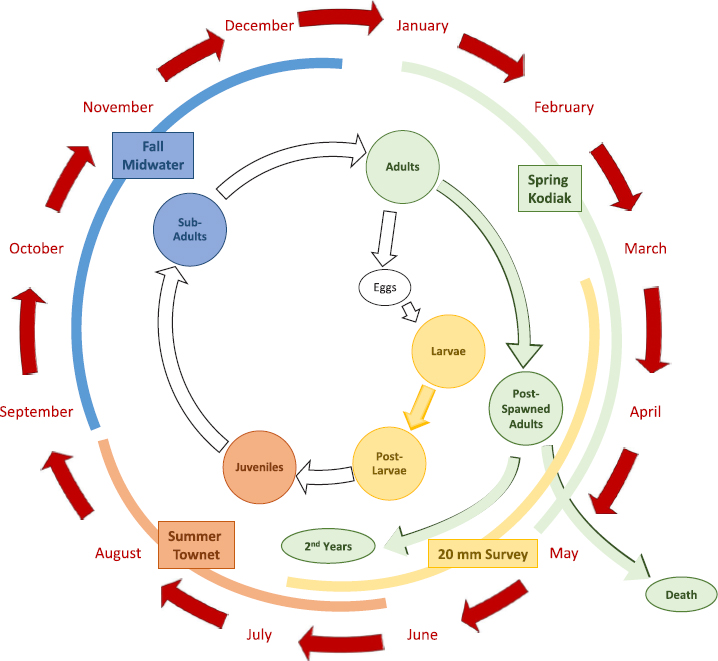
Figure E-2 shows the four Delta-wide monitoring networks of the California Department of Fish and Wildlife (CDFW) that measure for Delta smelt (20mm Survey, Summer Townet, Fall Midwater Trawl, Spring Kodiak Trawl—see Appendix D and Table D-1). Together these survey a wide variety of habitats and regions of the estuary at all times of year, providing a reasonable picture of smelt distribution and abundance. However, the capture efficiencies of the sampling gear involved in the four networks are poorly known and difficult to compare among surveys. With the exception of the Spring Kodiak Trawl surveys, none of the sampling programs was specifically designed to capture Delta smelt, and they all have limitations. For example, the Fall Midwater Trawl is not ideal because Delta smelt will school and avoid the net. Furthermore, to optimize monitoring of Delta smelt, sampling should be done at night.
In late 2016, the Enhanced Delta Smelt Monitoring (EDSM) program was created by USFWS in response to the low numbers of smelt being observed in the other monitoring networks. Its goals are “to (1) estimate the total abundance of Delta smelt on a weekly basis, (2) estimate their spatial distribution at spatial and temporal scales relevant to management, and (3) provide data that support management decisions and address scientific questions to further understanding of sampling efficiency, drivers of Delta smelt population patterns, and other conservation and management-relevant topics.”3 EDSM is a high-effort, year-round monitoring program that samples randomly selected locations throughout the San Francisco Estuary. This sampling technique provides well-distributed site locations to produce better spatial information. Throughout the year, the program targets three Delta smelt life stages (larvae, juvenile, and adult) using multiple gear types. EDSM reports data in near-real time.
Because Delta smelt are now rare, the effectiveness of the monitoring is limited, to the extent that indices of abundance, rather than abundance estimates or absolute numbers, are used. This approach is necessary because their distribution is patchy and mobile, and Delta smelt may at times occupy regions that are difficult to sample.
Abundance
Until the 1980s, the Delta smelt was an abundant fish in the upper estuary, moving with tides and river flows between the freshwater Delta and brackish Suisun Bay. Because Delta smelt is primarily an annual species, it exhibits highly variable year-to-year abundance, although in general Delta smelt appear to demonstrate higher population viability in very wet and cool conditions, such as those present in 2011 (USBR, 2024a). Delta smelt experienced a major decline in the early 1980s, followed by a substantial but brief increase in the late 1990s. This increase, in turn, was followed by an abrupt decline in the early 2000s, part of the pelagic organism decline (Box E-1; Baxter et al., 2008) that has continued to the present.
___________________
2 See https://wildlife.ca.gov/Conservation/Fishes/Delta-Smelt.
3 See https://www.fws.gov/project/enhanced-delta-smelt-monitoring-program.
SOURCE: Moyle et al. (2016).
In 2022, the Spring Kodiak Trawl abundance index was 1.7, which is the fourth lowest on record (reviewed in USBR, 2024a). The 2023 actual abundance was estimated to be 4,656 adults (95% CI: 1,178–12,730); this is the lowest abundance since that survey began in 2002 (Figure E-4). Furthermore, all CDFW relative abundance indices show a declining trend since the early 2000s. The extremely low spawning stock of Delta smelt relative to historical numbers suggests that the population is vulnerable to stochastic events and Allee effects (e.g., not finding mates, toxic spills) as well as continued anthropogenic alteration of the Delta including the direct (e.g., entrainment) and indirect (e.g., predation) impacts of CVP and SWP operation (USBR, 2024a).
Conceptual Models and Quantitative Life-Cycle Models for Delta Smelt
The many conceptual models of the Delta smelt life cycle, and several quantitative life-cycle or population dynamics–type models, generally tend to agree about the status and limiting factors for Delta smelt. Baxter et al. (2015) updated previous conceptual models with new information to (1) provide decision makers with a practical tool for evaluating difficult tradeoffs associated with management and policy decisions, (2) provide scientists with a framework from which they can formulate and evaluate hypotheses using qualitative or quantitative models, and (3) provide the general public with a new way of learning about Delta smelt and their habitat. Baxter et al. (2015) show that while good larval recruitment is essential for setting the stage for a strong year class, increased growth and survival through subsequent life stages are also necessary to achieve and sustain more viable higher-population abundances.
Rose et al. (2013a,b) developed a spatially explicit, individual-based population model of Delta smelt constructed for the upper estuary. Predicted and observed spatial distributions in the fall showed moderately good
BOX E-1
Pelagic Organism Decline
The “pelagic organism decline” refers to the pattern in Fall Midwater Trawl abundance indices observed around year 2000 for four major pelagic fishes of the upper estuary (Delta smelt, longfin smelt, striped bass, and threadfin shad). This sharp decline was surprising because these four species vary widely in life histories and estuary use. In 2005, an Interagency Ecology Program team investigated possible causes of the decline (Sommer et al., 2007), including bottom-up controls (food limitation), top-down pressure (predators), trends (previous abundance), and habitat (including water quality variables). The introduction and spread of non-native grazers (Corbula and Potamocorbula) in the mid-1980s that sequester the pelagic energy, resulting in the loss of rotifers, calanoid copepods, and mysids, seems to have been one major driver of the decline, along with the lack of recovery of pelagic fishes in wet years. According to Sommer et al. (2007), “The grazing effects from Corbula are thought to have resulted in a substantial decline in phytoplankton and calanoid copepods, the primary prey of early life stages of pelagic fishes. As a consequence, comparable levels of flow did not generate the expected levels of fish biomass (as indexed by abundance) after 1986. ... Hence, it appears that the response of these pelagic fishes to environmental conditions has fundamentally changed.”
Potamocorbula clams (P. amurensis), a brackish/saltwater clam species, were introduced to Suisun Bay around 1987 and quickly became established throughout Suisun Bay. (They are rarely observed or are at low abundances east of the Sacramento River/San Joaquin River confluence.) As a dominant grazer, P. amurensis has substantially reduced phytoplankton biomass and copepod nauplii. Corbicula clams (C. fluminea and C. largillierti) were probably introduced to the San Francisco Estuary in the early 20th century and quickly spread throughout the estuary and into the Delta. They are found in both sandy and muddy sediment types. They are simultaneous hermaphrodites; eggs develop into larvae (glochidia) within the brood chambers of females, and after release the glochidia are parasitic on fish gills for weeks to months. Juveniles then detach from fish and develop in the sediments. Because of their rapid reproduction, their effective dispersal, and their efficient filtering of overlying water, the clams have become an aggressive invasive species responsible for a profound shift in biomass within parts of the Delta. Their high filtering rate of particulate organic matter means that they can regulate water clarity, nutrient concentrations, and dynamics within the sediments. Figure E-3 shows the correlation between Corbula increases and decreases in zooplankton and chl-a.
agreement for extremely low- and high-outflow years. A high-outflow year (1998) was predicted to have a high population growth rate, and a low-outflow year (2001) was predicted to have a lower population growth rate due to entrainment and stage-specific survival rates. Smith et al. (2021) developed a state-space life-cycle model that predicts that as outflow declines in the summer, the estimated mortality of juveniles increases, natural mortality in the fall season is negatively associated with turbidity, and natural mortality of late sub-adult and adult stages is negatively associated with food. Similarly, Maunder and Deriso (2011) developed a state-space life-cycle model fitted to Delta smelt abundance indices at four life stages and environmental covariate data from the 1995–2015 period. They predicted that the most important factors affecting population dynamics were food abundance, water temperature, predator abundance, and density dependence. Polansky et al. (2021) used nonlinear state-space modeling to predict the probability of survival of different life stages. They predicted that larval survival is influenced by covariates related to abiotic habitat conditions (e.g., temperature, X2 position, outflow, turbidity) and biological factors (e.g., prey availability, competitors, predators); post-larval survival was influenced by outflow and turbidity;
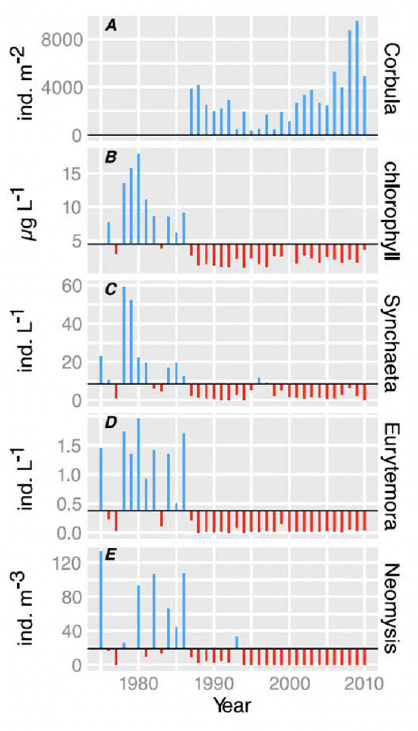
NOTES: (A) Corbula abundance. (B) Phytoplankton biomass as chl-a concentration. (C) Density of the rotifer Synchaeta bicornisa. (D) Density of the copepod Eurytemora affinis. (E) Density of the mysid shrimp Neomysis mercedis. The horizontal line in (B)–(E) is the long-term mean.
SOURCE: Cloern and Jassby (2012). ©2012. American Geophysical Union. All rights reserved.
juvenile survival was influenced by turbidity and temperature; and sub-adult survival was influenced by turbidity in the South Delta, Old and Middle River (OMR) flow, and adult striped bass (Morone saxatilis).
How Project Operations and Climate Change Affect Delta Smelt
Operation of the CVP and the SWP impacts Delta smelt via several often interconnected pathways. There are two types of possible entrainment. First, fish encountering CVP and SWP facilities may be pulled into diversions or the export facilities as they follow net flows (Grimaldo et al., 2009). Second, fish can be routed through specific migratory pathways in the Delta where tidal surfing behaviors (Sommer et al., 2011) draw Delta smelt into areas with increased entrainment risk. Entrainment of adult Delta smelt into the South Delta and the pumping facilities is most likely during the movement of fish from brackish waters to freshwater regions (Grimaldo et al., 2009, 2021; Kimmerer, 2008; Smith et al., 2021). Entrainment into the facilities tends to be highest when OMR flows are negative (i.e., toward the pumps) and when turbidity is high (Smith et al., 2021).

SOURCE: USBR (2024a).
The projects indirectly affect Delta smelt by reducing the size of the low-salinity zone due to reductions in freshwater flow through the lower Sacramento River system in summer and fall. In addition, the projects have altered the hydrology of the Sacramento River in ways that negatively affect Delta smelt, such as trapping sediment behind upstream dams, which increases water clarity. Introduced species, such as the overbite clam and the invasive aquatic weed Egeria densa that traps sediment, have also increased water quality to the detriment of Delta smelt. In particular, increases in dense submerged aquatic vegetation in terminal sloughs (Christman et al., 2023; Moyle et al., 2016; Smits et al., 2024) pose the dual threat of increasing water clarity and blocking access to Delta smelt spawning habitats.
A third major stressor on Delta smelt attributable to the projects are changes in food quality and quantity, potentially due to spatial and temporal mismatches between low-velocity pelagic habitat, nutrients and carbon, primary and secondary production, and the timing and distribution of larval smelt. The Summer-Fall Habitat Action (SFHA) aims to address this mismatch (see Chapter 4). Finally, the Delta smelt is now subject to greater predation, particularly by non-native predators, in slow water habitat created by the CVP and SWP, where Delta smelt may artificially congregate or be concentrated by flow patterns (Brown et al., 2024; Moyle et al., 2016; USBR, 2024a).
These threats and others are summarized in detail by life stage in USBR (2024a) and at an ecosystem level in Brown et al. (2024). One major conclusion is that all life stages are at risk nearly year-round.
Based on 30-year averages, temperatures in the Bay-Delta region are getting warmer and drier; however, extremes are also increasing in both directions, in part increasing the duration of dry spells (see Appendix A; Anderson, 2024). Because Delta smelt appear to survive best in wet and cool years, an increase in warmer, drier conditions could be expected to further decrease population viability. Komoroske et al. (2014) demonstrated that juvenile tolerance limits are close to what occurs currently such that this life stage may be the most susceptible to future climate warming. Furthermore, they predicted that otherwise suitable habitat for all life stages could be lost if high maximum temperatures, which are currently rare, increase in occurrence. Brown et al. (2013) evaluated potential changes in the position of the low-salinity zone, using a habitat suitability index, turbidity, and water temperature based on four 100-year scenarios of climate change. Overall, they found that the availability of suitable habitat would be similar to the worst droughts observed on record. Brown et al. (2016) then demonstrated, using global climate change model output downscaled to the watershed scale and then coupled with ecophysi-
ological metrics, the effects of warming on Delta smelt by integrating localized projected water temperatures with thermal sensitivity metrics (for such things as tolerance, spawning and maturation windows, and sublethal stress thresholds) across life stages. Lethal temperatures occurred under several scenarios, but sublethal effects resulting from chronic stressful temperatures were more common across the estuary. Behavioral avoidance of such stressful temperatures would make a large portion of the potential range of Delta smelt unavailable during the summer and fall. In addition, the Delta smelt maturation window was shortened by 18–85 days, revealing cumulative effects of stressful summer and fall temperatures with early initiation of spring spawning that may negatively impact fitness. Collectively, it appears that climate change will exacerbate the stress of Project operations on Delta smelt.
Hatchery Potential
Multiple generations of Delta smelt have been successfully reared in captivity in recent years. Beginning in 2021, cultured Delta smelt from the University of California (UC), Davis, Fish Conservation and Culture Laboratory have been released into the north Delta. These releases are expected to continue and even increase as production capacity increases (USBR, 2024a). Although these captive fish offer opportunities for supplementation, there are few examples available where supplementing natural populations of fish in the wild from hatchery fish has led to recovery, in the absence of eliminating the limiting factors (e.g., McMillan et al., 2023). If limiting factors can be minimized or eliminated, then the hatchery fish could be critical for restarting the natural population. In addition, the hatchery fish offer an excellent opportunity to treat actions, or components of actions, as natural experiments. As discussed in Chapter 4, tagged hatchery fish could be released in areas targeted for the SFHA. It is unknown how well released hatchery fish thrive and whether or not they reproduce in the wild. Reducing these uncertainties will require many more fish than the hatchery currently produces.
Uncertainties That Complicate Management of Delta Smelt
After updating the Delta smelt conceptual model, Baxter et al. (2015) developed a series of hypotheses for evaluating why Delta smelt abundance increased in 2011. Ongoing research addresses some of these issues, but there have been few definitive conclusions to date. The hypotheses are broken up by life stage and include the following:
- Adults
- How do hydrology and exports interact to influence adults?
- How does hydrology interact with turbidity to affect predation risk?
- How does predator distribution affect predation risk?
- How does variability in prey availability during winter and spring affect growth and fecundity (eggs per clutch and number of clutches)?
- Larvae
- Are larvae numbers positively affected by increased duration of the temperature spawning window?
- Will increased food availability result in increased larval abundance and survival?
- Does high abundance of Mississippi silverside increase predation risk/rate on larval Delta smelt, and can this be counteracted by increased turbidity?
- How do hydrology and water exports interact with one another to influence direction of transport and risk of entrainment?
- Juveniles
- How does high water temperature reduce juvenile growth and survival (bioenergetic stress; reduced distribution)?
- Does the distribution and abundance of striped bass, temperature, and turbidity influence predation risk/rate?
- How are growth and survival affected by food availability?
- Are survival and growth reduced by harmful algal blooms, and if so, how (direct effects such as habitat quality and toxicity or indirect effects such as food quality and quantity)?
LONGFIN SMELT
The longfin smelt (Spirinchus thaleichthys) is a small (<150 mm) pelagic fish native to the Bay-Delta but with a widespread distribution in coastal estuaries stretching from San Francisco to Alaska. Within California, it is also found in Humboldt Bay, Russian River, Eel River, and Klamath River and has been caught as far south as Monterey Bay. The San Francisco Bay-Delta population is the southernmost reproductive population and is considered a genetically distinct population.
Longfin smelt have been historically very abundant and were even considered one of the most abundant species in the San Francisco Bay Estuary. There was even a baitfish commercial fishery in the 1970s. Over the past few decades, abundance of this species has progressively declined, with, in particular, a more precipitous decline after the invasion of the overbite clam in 1980. Longfin smelt are part of the pelagic organism decline in the California Delta (Box E-1); abundances have collapsed to an estimated 1 percent of former levels. This decline resulted in longfin smelt being listed as threatened under the CESA in 2009 (CDFG, 2009a,b). This listing “prohibits unpermitted possession, purchase, sale or take.” USFWS listed the Bay-Delta distinct population segment of longfin smelt as endangered in August 2024,4 and habitat protection was afforded in January 2025.5
Life History and Stages
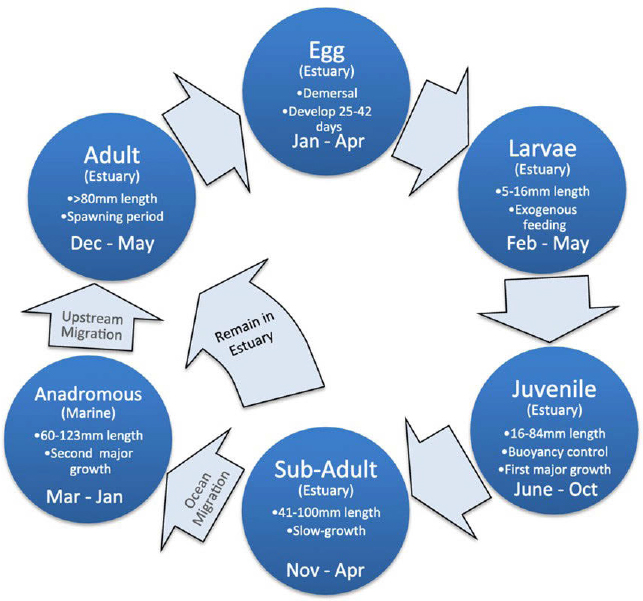
A recent detailed literature review of the life history, reproduction, distribution, diet, and habitat requirements (i.e., temperature, salinity, turbidity, diet) of the longfin smelt can be found in the Species Status Assessment for the Bay-Delta published by USFWS (2022, pp. 9–27). The longfin smelt has a widespread distribution throughout the San Francisco Bay and much of the Delta (Figure E-5). It lives in freshwater, estuarine, and marine habitats at different stages of its life history. It is considered to be an anadromous species that reproduces in freshwater or low-salinity water but spends its late juvenile and adult life in estuarine (Bay) or nearshore marine coastal waters. Longfin smelt generally have a two-year lifespan in the Bay-Delta. Adults migrate to fresher waters in the fall and spawn from about November through April with a peak in February (Figure E-6). Adults generally die after spawning.
The exact spawning regions for the longfin smelt are unknown, but smelt are believed to spawn in freshwater or in very low-salinity waters in the Delta or San Francisco Bay tributaries. Females produce up to 12,000 eggs. Eggs are adhesive and are believed to be deposited on hard substrate, possibly sand, gravel, or other structures as the species does in other areas. Low-salinity regions may be a critical habitat for early life stages of this species. There are ongoing otolith studies to better understand habitat needs for young-of-the-year and to reconstruct growth and salinity history in relation to climate and outflow history at the UC Davis Otolith Geochemistry and Fish Ecology Laboratory (e.g., Hobbs et al., 2010).
When the larvae reach 10–12 mm, they can move up and down in the water column and maintain position relative to the estuarine/freshwater (i.e., X2) interface where zooplankton food production is high. The juvenile stage is reached in about 90 days when the fish are about 20 mm in size. The smelt then spend the late juvenile and adult stage in estuarine or marine coastal waters as far out as the Farallon Islands.
Longfin smelt is generally considered to be a cooler-water species with preferences of 16–18°C (60.8–64.4°F) and spawning temperatures of 7–14.5°C (44.6–58.1°F). They are sensitive to temperatures above 20°C (68°F). The longfin smelt is a pelagic species and feeds largely on zooplankton. Larvae and young juveniles feed mainly on calanoid copepods whereas older juveniles and adults feed mainly on mysids. Longfin smelt are likely prey for Mississippi silversides and striped bass.
Monitoring for Longfin Smelt and Abundance Data
Like Delta smelt, longfin smelt are assessed and caught in most monitoring trawl surveys. New methods to assess larval smelt abundances are being developed to better assess the potential for larval fish entrainment
___________________
4 See https://www.govinfo.gov/content/pkg/FR-2024-07-30/pdf/2024-16380.pdf.
5 See https://www.govinfo.gov/content/pkg/FR-2025-01-15/pdf/2024-29641.pdf.

SOURCE: Lewis et al. (2020). CC BY-NC.
SOURCE: Merz et al. (2013) cited as being adapted from the Delta Regional Ecosystem Restoration Implementation Plan Conceptual Models.
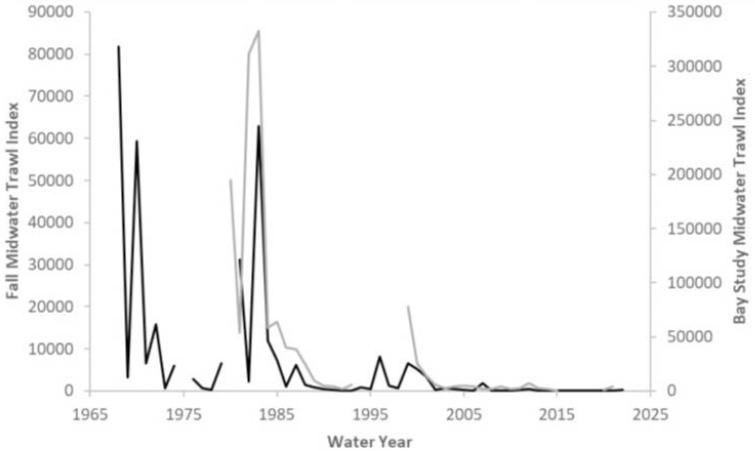
(Mora et al., 2024). Although the abundance of longfin smelt has declined precipitously in recent decades, it is not considered almost functionally extinct like the Delta smelt. Figure E-7 shows longfin smelt data from the Fall Midwater Trawl and the San Francisco Bay Study Midwater Trawl over the past 50 years.
Conceptual Models and Quantitative Life-Cycle Models
Given the complex life history of the longfin smelt, the seasonal changes in distributions and stressors, and the vulnerability of the species to high salinity levels as well as to co-occurring stressors (e.g., higher water temperatures), a mechanistic life-cycle model for this species may be warranted (see Maunder et al. [2015] and Nobriga and Rosenfield [2016] for examples of statistical life-cycle models). Developing such a mechanistic model could help to assess population-level impacts of entrainment, better understand the winter/spring flow impacts, and predict impacts of interannual or climatic effects of changing temperatures on survival. A life-cycle model is currently under development by USFWS and partners (Tobias et al., 2024), with completion expected by 2027.
How Project Operations and Climate Change Affect Longfin Smelt
The recent longfin smelt Species Status Assessment (USFWS, 2022, pp. 28–42) discusses the following stressors in detail: food limitation, temperature, loss of suitable spawning habitat, predation, contaminants, and entrainment. In general, the widespread distribution of longfin smelt means that different life stages are vulnerable to different drivers during different seasons. Recent declines in abundance are believed to have been caused by reduced freshwater flow during incubation and larval rearing, because increases in salinity are known to decrease fertilization success (Rahman et al., 2023). Indeed, the relationship between freshwater outflows and longfin smelt juvenile production (and thus total abundance) is well documented (Kimmerer, 2002; Nobriga and Rosenfield, 2016). However, there is also debate about whether the flow-ecology relationship for this species may have weakened over time (Nobriga and Rosenfield, 2016; Rosenfield and Baxter, 2007; Thomson et al., 2010), coinciding with step-changes in the species population size. Active research is underway focusing on this consequential issue, to parse out the effects of the small population on limited recruitment from the effects of freshwater outflow on
SOURCE: USBR (2024b).
recruitment,6 which may both explain the decrease in longfin smelt production per unit of flow. The unique effects of low-flow conditions on longfin smelt rearing and persistence have also been challenging to quantify, because low-flow years are also typically warmer, and longfin smelt in the Delta are living near their upper limit of 20°C (68°F). Upriver shifts in salinity (e.g., as measured by X2) seem to be the strongest proximal driver of changes in freshwater outflow on longfin smelt recruitment, as suggested by the fact that persistent low-salinity spawning and rearing habitat in high precipitation and outflow years coincide with strong recruitment (Lewis et al., 2020). The Species Status Assessment discusses the potential of climate change and supraseasonal droughts to continue to change flow rates and increase salinity, cause sea level rise, and cause warm water temperatures in the fall—stressors that if not counteracted, will likely compound and accelerate the decline of the species.
Hatchery Potential
The longfin smelt has been successfully bred and raised in the laboratory and so has the potential for the creation of hatcheries (e.g., Hung et al., 2024).
Uncertainties That Complicate Management of Longfin Smelt
A detailed science plan for the longfin smelt has been proposed for 2020–2030 (CDWR et al., 2020). This plan includes a critical discussion of the literature with knowns and unknowns including the nuances about the species’ lifestyle and ecological role in the Delta. Seven priority science areas were identified in the science plan. For each of these topics, the discussion includes past research on the topic, current needs, management applications, and scientific approaches. The seven priority areas were as follows:
- Life-cycle modeling
- Factors affecting abundance, growth, and survival
- Improved distribution monitoring
- Improved larval entrainment monitoring
- Longfin smelt culture
- Fish migration and movements
- Spawning and rearing habitats for longfin smelt
Additional science topics were also identified to stimulate research. These topics were as follows:
- Invasive species effects
- Contaminant effects
- Possible role of diseases in longfin smelt population dynamics
- Identification of the food web for longfin smelt
- Development of new tools (e.g., monitoring, health)
- Measurement of vital rates (e.g., growth, survival, reproduction)
- Climate change effects (e.g., hydrology, temperature)
CENTRAL VALLEY CHINOOK SALMON
Chinook salmon (Oncorhynchus tshawytscha) are anadromous fish native to the North Pacific Ocean. North American populations of Chinook salmon range from Central California to Kotzebue Sound, Alaska, and migrate to spawning areas in streams from near tidewater to 3,200 kilometers upstream (Major et al., 1978; Roni and Quinn, 1995). Adult fish typically range in size from 60 to 90 centimeters (cm) (24 to 36 inches) but may be up to 150 cm (58 inches) in length; they average 4.5 to 22.7 kilograms (kg) (10 to 50 pounds) but may reach 59 kg
___________________
6 See https://caseagrant.ucsd.edu/our-work/research-projects/examining-relationship-between-longfin-smelt-zooplankton-and-flow-san.
(130 pounds) (Roni and Quinn, 1995). Central Valley Chinook salmon are anadromous and semelparous (with the exception of cases where introduced or isolated populations are prevented from ocean migration by dams or other human constructed barriers). They are generally considered to be the largest Pacific salmon but exhibit great variation in size and life-history traits across individuals and populations including the duration of juvenile residence in freshwater, size at age, age at maturity, ocean distribution, migration time, and spawning time (Healey, 1991; Ricker, 1972; Roni and Quinn, 1995).
Chinook can have extended lifespans, in which some fish spend one to five years in the ocean, reaching age eight. More northerly populations tend to have longer lives. Salmon need suitable freshwater spawning habitat including clean, cool, oxygenated, and sediment-free freshwater for egg development. Chinook use larger sediment (gravel) sizes for spawning compared to other Pacific salmon. Riparian vegetation and woody debris help juvenile salmon by providing cover and maintaining low water temperatures in streams.
California’s Central Valley drainages formerly produced immense numbers of Chinook salmon. Four seasonal runs occur in this system—fall, late-fall, winter, and spring runs. Differences in life-history timing and spatial distribution enabled the four runs to maximally use the drainages and diverse mosaic of available habitats. Table E-1 provides a summary of the timing ranges for late fall-, fall-, winter-, and spring-run life-history stages in the Sacramento and San Joaquin basins, respectively, and Figure E-8 shows the typical stages of the Chinook life cycle.
The spatial distribution and diversity in run timings of Central Valley Chinook salmon historically contributed to the region being one of the richest in the world for Chinook salmon production, conservatively estimated to have reached approximately 1 to 2 million spawners annually (Yoshiyama et al., 1998). Sacramento River winter-run Chinook salmon, also known as king salmon, is one of five Evolutionarily Significant Units (ESUs) of Chinook salmon found in California (Moyle, 2002). As of May 16, 1989, the California Fish and Game Commission listed the Sacramento River winter-run Chinook salmon as endangered under the CESA. Subsequently, the Sacramento River winter-run Chinook Salmon ESU was listed as threatened under the federal ESA on August 4, 1989, and then updated to endangered on January 4, 1994.7
The historical range of the Central Valley spring-run Chinook salmon ESU encompassed most of the Central Valley and its rivers: from the upper Sacramento River south through the San Joaquin River, down to the Kings
TABLE E-1 Generalized Life-History Timing of Central Valley Chinook Salmon

SOURCES: Yoshiyama et al. (1998) based on Fisher (1994), USFWS (1995).
___________________
7 See https://wildlife.ca.gov/Conservation/Fishes/Chinook-Salmon/Winter-run.
SOURCE: Winter-run Chinook Salmon Lifecycle model, see https://oceanview.pfeg.noaa.gov/wrlcm/src/lifestage-plots.html.
River (Lindley et al., 2004). The San Joaquin River and its tributaries historically supported the largest run of spring-run Chinook (Yoshiyama et al., 1998), with an estimated 200,000 to 500,000 adults returning to the San Joaquin River annually before the completion of Friant Dam in 1942 (Yoshiyama et al., 2001). Friant Dam and other dams blocked access to nearly all Central Valley spring-run Chinook salmon spawning habitat in the San Joaquin basin, causing severe population declines. At the time of the 1999 listing, Central Valley spring-run Chinook salmon in the San Joaquin basin were considered extirpated; therefore, only Central Valley spring-run Chinook salmon in the Sacramento River and its tributaries were formally listed as threatened under the ESA (Gutierrez et al., 2024).
Although not a state- or federally listed species, fall-run Chinook salmon populations across the Central Valley are severely impacted and vulnerable to extinction (Katz et al., 2012). Over the past two decades, production of fall-run Chinook salmon in the San Joaquin River basin has fallen to very low levels (SEP Group, 2016) and Sacramento River basin production has exhibited some of the lowest returns in recorded history, resulting in multiple closures of both commercial and recreational fisheries.
Life History and Stages
The following provides a generalized overview of Central Valley Chinook salmon life stage needs and associated vulnerabilities with an emphasis on those factors directly or indirectly affected by CVP and SWP operations.
Adult Migration and Spawning
Chinook salmon return from the ocean to spawn in freshwater, notably the rivers of the Central Valley. Adults cease to eat during their spawning migrations and die after spawning. Somatic energy reserves and nutrients are used to complete the upstream journey, attain and defend nest sites, mate, and spawn. Nutrients and energy are also allocated to the production of gametes. Adult migration and gametogenesis are energy-intensive and time sensitive
activities. Therefore, delays to migration and spawning caused by barriers, false pathways, or disorientation can result in death, lost opportunities to spawn, or other forms of reduced reproductive success (SEP Group, 2016).
Chinook salmon typically return to their natal streams to reproduce, a process called homing. A limited percentage of locally adapted wild populations and a larger percentage of hatchery populations also exhibit the opposite behavior (i.e., returning to a non-natal stream to spawn), which is known as straying. Several modes of orientation play a role in successful homing. However, once adult fish enter freshwater, olfactory identification of water emanating from the natal stream is the dominant cue driving salmonid orientation (Healey, 1991; Quinn, 2005). In highly managed watersheds such as those of the Central Valley where large fractions of a river’s flow may be diverted at one or more locations along the migration path, homing success can be influenced by the amount of flow from a particular spawning stream that reaches a migrating adult salmon and the ratio of flow from various source streams in a watershed (Marston et al., 2012; SEP Group, 2016).
Egg Incubation and Emergence
Suitable water temperature is necessary and perhaps most essential for viability of the Chinook salmon egg incubation stage. Indeed, the egg stage has stringent requirements for cold water as an inherent consequence of their large size (for fish eggs). Water temperature and developmental rate are tightly and positively correlated in salmonids (Healey, 1991; Quinn, 2005), such that above certain temperatures, enzymatic function is compromised, resulting in inefficient use of food resources (SEP Group, 2016). Eggs and alevins incubated at temperatures that are too warm produce smaller fry than they would at optimal temperatures (EPA, 2001, 2003). Hatching and emergence success decrease as temperatures rise above the threshold for optimum development. Direct egg mortality due to elevated temperatures is known to occur in the Central Valley (Williams, 2006), and temperature-related mortality and habitat limitation will likely become an increasingly serious challenge for Central Valley salmonids as climate changes (Lindley et al., 2007).
Juvenile Rearing and Migration
The juvenile rearing and migration life stage encompasses all those developmental stages, life-history strategies, behaviors, and phenotypic expressions that occur subsequent to emergence and prior to either ocean entry and/or sexual maturation, including fry, parr, smolt, and yearling developmental stages. Generally, optimal conditions for juvenile Central Valley Chinook salmon rearing involve a balance of (1) water quality conditions (e.g., temperature, dissolved oxygen, contaminant concentrations); (2) physical attributes of habitat (water depth, suitable cover, and substrate); (3) extent of available habitat relative to fish territory size (as a function of fish size, fish density, prey density, and habitat structure); (4) ecosystem and food-web conditions (e.g., prey availability and density, predator density, and competition); and (5) activity levels (as a function of the interaction of 1, 2, 3, and 4 with water velocity)—such that juvenile salmonids can sustain metabolic needs while maximizing growth (Quinn, 2005). However, these conditions vary across a range of sub-habitat types within the riverine landscape used by juvenile salmonids. Various sub-habitats may also be used differently by specific life-history stages of a given salmonid species (Bradford and Higgins, 2001; Merz et al., 2016; Roper et al., 1994), and by individuals within a life-history stage that are developing at different rates (e.g., “young”/small parr may utilize habitats differently than older/larger smolts).
Research from both the Sacramento and San Joaquin basins suggests that fall-run juvenile Chinook salmon experience superior foraging conditions in and prefer off-channel and floodplain rearing habitat to adjacent in-channel habitat, and they exhibit improved growth and survival when rearing on seasonally inundated floodplains (Merz et al., 2016; Sommer et al., 2001a,b, 2005). The interaction of different life stages and histories with different sub-habitats can additionally reinforce cohort and population-level life-history diversity and associated resilience (McClure et al., 2008; Zimmerman et al., 2015). For example, juvenile Chinook salmon rearing on floodplains can experience greater maximum size, diversity in growth, and exposure to environmental pollutants than juvenile salmon reared in the associated river channel (Henery et al., 2010; Jeffres et al., 2008; Sommer et al., 2001a,b, 2005).
Central Valley fall-, winter-, and spring-run Chinook salmon have also been observed rearing in non-natal tributaries as well as in the Delta (Cordoleani et al., 2021a; Limm and Marchetti, 2009; Phillis et al., 2018; Stur-
rock et al., 2015). However, the extent to which these expressions are a response to limited off-channel rearing habitat in their natal streams and/or were strategies exhibited by a significant portion of the population historically is unknown.8
Key Differences in Life Histories of the Runs
Winter-Run Chinook Salmon.
Adult winter-run Chinook salmon return from the ocean in the winter and migrate through the Bay-Delta and up the mainstem Sacramento River to reach the upper Sacramento River. Prior to the building of Shasta Dam, winter-run Chinook reached the Pit, McCloud, and upper Sacramento rivers, and Battle Creek, where summer water temperatures were typically 10–15°C (50–59°F), and it was under these conditions that they held and spawned, their eggs incubated, and their young reared. Since the construction of the dams, winter-run migration has ended in the stretch of the Sacramento River below Keswick Dam where adults hold until spawning, followed by egg incubation and fry emergence in the summer. Given their evolution in coldwater spawning grounds, the winter-run Chinook have stringent temperature and dissolved oxygen requirements as shown in Table E-2.
In addition to these temperature and dissolved oxygen requirements, winter-run Chinook salmon require certain water velocities during holding, spawning, and egg incubation (summarized in USBR, 2024e), as well as clean loose gravel 0.75–4.0 inches in diameter for successful spawning (NMFS, 2014). The outmigration requirements for fry include streamside habitats containing riparian vegetation and associated substrates that provide aquatic and terrestrial invertebrates for food, predator avoidance cover, and slower water velocities for resting (NMFS, 2014).
Spring-Run Chinook Salmon.
Spring-run Chinook salmon evolved to take advantage of the geography and hydrology of the California Central Valley and surrounding mountain ranges where melting snow from high elevations provides cold water for adult and juvenile salmon to survive and thrive during the hot Central Valley summers (Fry, 1961). Adult spring-run Chinook salmon leave the Pacific Ocean to begin their upstream migration to natal streams in late January and early February (Yoshiyama et al., 2001) and enter the Sacramento and San Joaquin rivers from March to September, with peak migration during the spring months (Moyle, 2002; Yoshiyama et al., 1998). They enter rivers in a sexually immature state; they then hold in deep, cold freshwater pools for several months to mature; and then adults spawn from mid-August to early October, with peak spawning occurring in September (Moyle, 2002). Hence, access to coldwater habitats during the summer and early fall months is a critical component of spring-run adult holding, spawning, egg incubation, and juvenile rearing life stages (Gutierrez et al., 2024).
Spring-run Chinook salmon have two distinct juvenile life-history strategies that enable them to take advantage of California’s variable climate: (1) the “sub-yearling” life-history strategy in which juveniles rear and then migrate downstream to the ocean within three to eight months of hatching and (2) the “yearling” life-history strategy in which juveniles rear in the river for a more extended 8- to 12-month period prior to migrating to the ocean. The yearling life-history strategy helps to ensure species’ survival across a range of hydrological conditions and water-year types (Cordoleani et al., 2021b). Yearling juveniles are typically larger in size than sub-yearling juveniles at the time of outmigration. Cordoleani et al. (2021b) found that spring-run Chinook salmon yearlings comprised roughly 10 percent of juveniles but approximately 60 percent of returning adults when averaged across years in Mill and Deer creeks in the Sacramento River basin. The number of returning adults with the yearling juvenile life-history strategy was even higher (77–100 percent) in drought years. Hence, ensuring that habitat conditions are suitable for the expression of the yearling life-history strategy in all water-year types is critical for Central Valley spring-run Chinook salmon survival and recovery, particularly in challenging conditions created by climate change and the varying water-year types of the California Central Valley (Gutierrez et al., 2024).
Fall-Run Chinook Salmon.
Fall-run Chinook salmon were historically the most widely distributed of the Central Valley runs, with more than 30 independent populations (Yoshiyama et al., 2001) across the Sacramento and San Joaquin basins, and that continues to be true under current conditions. Indeed, because of the decline
___________________
8 This paragraph was edited after release of the report to include winter-run salmon.
in spring-run Chinook salmon, fall-run is now the dominant population supporting the California and Southern Oregon commercial salmon fisheries (Huber and Carlson, 2015).
Like the spring-run Chinook, the fall-run Chinook may include a small fraction of juveniles that over-summer and emigrate as yearlings (SEP Group, 2016; Yoshiyama et al., 2001). Run timing for fall-run relative to hydrologic conditions and other run timings likely resulted in their occupying and spawning in lower reaches of the CV rivers closer to the valley floor. For this reason, their spawning habitat was less (although still significantly) impacted by the construction of the rim dams, relative to winter- and spring-run populations.
Monitoring of Chinook and Data on Abundance
A range of monitoring surveys for CV Chinook salmon is targeted at each of the life stages, for each of the run-types, across a broad range of the geographies they occupy. In the Sacramento River system, a significant portion of the monitoring for all run timings is an outgrowth of monitoring to support the conservation of winter-run and is conducted by diverse agencies and entities. A summary of these monitoring efforts is provided in Table E-3. In addition to adult and juvenile monitoring in the Sacramento River, additional monitoring for Chinook salmon occurs via salvage data from the Tracy and Skinner fish collection facilities (discussed in Chapter 3), via seasonal fish assemblage trawls and the Delta Juvenile Fish Monitoring Program (including the Sacramento, Mossdale, and Chipps Island trawls and beach seines), and in commercial and recreational ocean fisheries via coded wire tag.
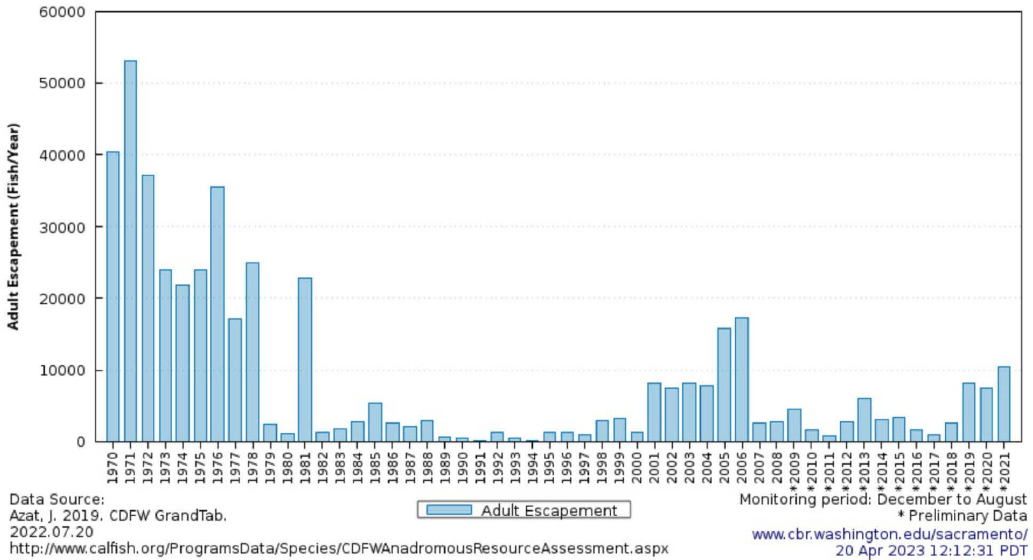
In terms of abundance, winter-run Chinook salmon population estimates were as high as 120,000 fish in the 1960s but declined to less than 200 fish by the 1990s (NMFS, 2014). As shown in Figure E-9, during 1970–2021, the highest escapement values were seen in the early 1970s, followed by low values in the early 1990s, increases in the early 2000s, and varying between ~1,000 and ~10,000 individuals since 2007. For context, the “doubling goal” under the Central Valley Project Improvement Act (CVPIA) targets 110,000 winter-run Chinook salmon. Since 2001, the majority of winter-run Chinook salmon redds have occurred in the first 10 miles downstream of Keswick Dam (USBR, 2024e).
SOURCE: USBR (2024e).
Conceptual Models and Quantitative Life-Cycle Models
There are two primary life-cycle models for Chinook salmon that are used in the Delta, the Winter-Run Life Cycle Model (WRLCM) (Hendrix et al., 2014, 2019) and the CVPIA decision support model (Peterson and Duarte, 2020). Although other models exist, the WRLCM and CVPIA models are perhaps the most widely leveraged for landscape-scale population management and planning, for many reasons. Their development was led by the federal agencies overseeing fish management (National Oceanic and Atmospheric Administration’s National Marine Fisheries Service [NOAA/NMFS] for the WRLCM and USFWS and USBR for the CVPIA) and offered many engagement opportunities for a diverse suite of stakeholders. Their scope is broad, including the complete species life cycle and range of habitats and capturing effects from the individual to the population level. And finally, they were designed to provide decision support in critical management areas related to flow allocations (WRLCM) and habitat investments (both the CVPIA decision support model and WRLCM).
The WRLCM was briefly discussed in Chapter 2, with a focus on its potential application for the Shasta Coldwater Pool Management Action. Figure D-4 shows the processes included in the model.
USBR and USFWS established the Science Integration Team (SIT) to use structured decision making to identify priorities for Chinook salmon, steelhead, and green and white sturgeon. The SIT is a self-selected technical group of agency staff and stakeholders that employ a collaborative and transparent process to identify, compile, and provide access to appropriate data; develop decision support models consistent with CVPIA fish doubling goals (see Appendix C); and provide input into the development of CVPIA priorities for restoration actions and information needs.
The SIT developed and continues to maintain models to support the development of restoration strategies as a basis for guiding CVPIA investments in CV salmon recovery. The stochastic stage-based CVPIA Chinook model tracks the number of Chinook salmon in four juvenile size classes: small, <42 mm total length; medium, 42–72 mm; large, 72–110 mm; and very large, >110 mm, for both natural and hatchery-origin fish (Figure E-10). The model operates on a monthly time step over a simulated 20-year period. Transitions between stages are estimated using submodels that predict survival, growth, and movement as functions of input parameters that represent conditions
| Run and Life Stage | Action | Location | Frequency | Agency | Notes |
|---|---|---|---|---|---|
| Winter-run (WR), fall-run (FR), late-fall-run (LFR) | Rotary screw trap | Red Bluff Diversion Dam | USFWS | Used to determine Juvenile Production Estimate (JPE) for all runs except for SR | |
| WR – Juveniles (fry) | Rotary screw trap | McCloud River, Head of reservoir (McCloud River Bridge) | March–December (or as flows permit) | CDFW, Pacific States Marine Fisheries Commission (PSMFC), Winnemem Wintu Tribe | |
| WR – Juveniles | WR jumpstart monitoring | Battle Creek | USFWS | Coleman National Fish Hatchery | |
| WR – Juveniles | Rotary screw trap | Glenn-Colusa Irrigation District (GCID) bypass channel within the Sacramento River near RM 205 | CDFW, GCID | GCID recently stopped operating the trap. | |
| WR – Juveniles | Livingston Stone Fish Hatchery (LSFH) | Sacramento River near Redding | December–March | USFWS | 200,000–600,000 WR are raised annually at LSFH and released into the river at Caldwell Park in December/January during high flow/turbidity events. |
| WR – Juveniles | Juvenile Salmonid Collection System | McCloud River, Head of reservoir (downstream of Screw Trap) | Mid-August–January | CDWR | |
| WR – Egg-Fry | Survival monitoring | McCloud River, Ah Di Na campground | UC Davis, Winnemem Wintu Tribe, CDFW | ||
| WR – Adults, Juveniles | Monitoring and relocation (seine) | Sutter and Yolo bypasses and related facilities including Tisdale, Fremont, and Sacramento weirs | Multiple times per year (depending on flow and environmental conditions) | CDFW | Juveniles are identified using length-at-date criteria (sometimes larger juveniles are confirmed with genetic analyses). Adults are identified via genetic analyses. |
| WR – Adults | Monitoring and relocation (trap) | Wallace Weir Fish Collection Facility located in the Knights Landing Ridge Cut within the Yolo Bypass | CDFW | Adults are identified via genetic analyses. | |
| WR - Adults | Shallow Redd monitoring | Red Bluff Diversion Dam to Keswick | June–November | CDFW | Shallow = less than 2' water depth when first encountered; informs USBR Keswick Dam operations during incubation |
| WR - Adults | Livingston Stone fish trap | CDFW and PSMFC | WR are captured, and either spawned or released based on a spawning matrix. | ||
| WR - Adults | Carcass Survey | Mainstem Sacramento River, Balls Ferry Bridge to Keswick | 7 days a week, May–September | CDFW, PSMFC, and USFWS | Population estimates are determined using a Cormack Jolly Seber model. |
| WR - Adults | Aerial (helicopter) redd survey | Red Bluff Diversion Dam to Keswick Dam | Weekly May–September | Redd surveys provide expansion factor for redds that fall outside of the carcass survey reach. | |
| SR/Steelhead - Adults | Livingston Stone fish trap | CDFW and PSMFC | Adult SR and steelhead captured; spring-run released either into other SR tributaries or back into the river; more recently, some captured unmarked SR held and spawned at LSFH; the eggs from these SR were raised in hatch partner boxes in Clear Creek below Whiskeytown Dam. Adult steelhead are PIT-tagged and released back into the river at Caldwell Park. | ||
| FR/LFR/SR - Adults | Shallow Redd monitoring | Mainstem Sacramento | Respective spawning seasons | CDFW | Monitored for potential dewatering; only early SR redds (September) are monitored due to overlap with FR spawning in October |
| FR/LFR - Adults | Redd survey | Woodson Bridge to Keswick Dam | Biweekly October–May | Historically began at Princeton and extended up to Keswick Dam. More recently the starting point moved to Woodson Bridge (Corning) as fewer and fewer redds have been observed in the section below Corning. | |
| FR - Adult | Carcass survey | Mainstem Sacramento River, Balls Ferry Bridge to Keswick | 3–5 days a week (FR spawning season) | CDFW and PSMFC | Population estimates are determined using a Cormack Jolly Seber model. |
| All runs & Steelhead - Juveniles | Rotary screw trap | Three locations upstream of the Tisdale Weir near RM 120 | Late August/early September to May/June | CDFW | Fish are identified using length-at-date criteria. |
| All runs & Steelhead - Juveniles | Rotary screw trap | Three locations Knights Landing near RM 88 | Late August/early September to May/June | CDFW | Fish are identified using length-at-date criteria. |
| All runs & Steelhead - Juveniles | Rotary screw trap | Three locations near town of Verona and RM 75 | October to May/June | CDFW | Fish are identified using length-at-date criteria. |
| All runs & Steelhead - Juveniles | Rotary screw trap | Lower Feather River near RM 17 | CDFW | Juvenile WR are consistently captured at this location, assumed to be non-natal rearing. For example, during January–February 2024, 35 juvenile WR (32 unmarked; 3 marked, based on length-at-date criteria) were captured. WR assumed to have originated from LSFH. |
| Run and Life Stage | Action | Location | Frequency | Agency | Notes |
|---|---|---|---|---|---|
| All Runs - Juveniles | Pre- and post-restoration habitat monitoring – Snorkel Surveys | Red Bluff Diversion Dam to Keswick Dam | Year-round, (or when conditions allow) | CDFW, PSMFC, Chico State, and Tussing Enterprises | Juveniles are identified using length at date and enumerated as part of snorkel surveys on proposed and completed habitat restoration projects. |
| All Runs - Juveniles | Stranding and rescue surveys | Los Molinos to Keswick Dam (documented stranding sites) | Following flow reductions from Keswick or flood releases | CDFW and PSMFC | Conducted as a component of operations for Keswick; all salmonid species are identified to species and/or run using the length-at-date criteria. |
NOTE: RM = River Mile.
SOURCE: Table information drawn primarily from Michael Memeo, CDFW Region 1, personal communication, 2025.

NOTE: Each arrow represents a state transition (i.e., a function such as survival or growth). Parameter estimates and sources are in Peterson and Duarte (2020).
SOURCE: https://cvpia.scienceintegrationteam.com/cvpia-sit/resources/documents.
in the Bay-Delta, migratory corridors, and natal tributaries. The decision support models are parameterized and updated using empirical data, existing models, analyses of existing data, and a minimal amount of expert opinion. The decision support models for the three Chinook salmon runs have the same general structure but differ with respect to timing and inputs (Peterson and Duarte, 2020).
Beginning in 2021, Reorienting to Recovery (R2R), a nonregulatory, voluntary, collaborative process lead by nongovernmental organizations and water agencies and focused on the application of life-cycle models and structured transparent processes to develop a widely supported pathway to salmon recovery in the Central Valley, started to apply the CVPIA decision support model. In order to explore approaches to achieving salmon recovery that integrated the range of management action categories applicable to salmon (hydrology, habitat, hatcheries, and harvest—commonly termed “the 4Hs”), the R2R process assembled a diverse group of CV salmon experts and built out the decision support models, which were used by the CVPIA primarily to contemplate habitat actions and to be able to evaluate combinations of actions integrating all 4Hs. The resulting R2R decision support model is also publicly available.
***
The existence of three landscape-scale decision support models for winter-run Chinook salmon in part emanates from a collective evolution in management from a more narrow focus to a more integrative one. Shortly after its development, the WRLCM was viewed primarily as a tool to assess the impact of flow management on winter-run Chinook salmon as it pertained to regulatory compliance and was primarily applied to the evaluation of a range of proposed flow management scenarios. The WRLCM also lacked some of the key parameters (e.g., growth) and relationships (e.g., habitat specific growth and survival and survival to adulthood) that rearing habitat projects were often designed to affect. The CVPIA model, by contrast, was initially designed to provide guidance for CVPIA investments in habitat restoration and enhancement projects with a focus on fall-run Chinook salmon
which, though not listed, make up the majority of California’s commercial and recreational harvest and are the focus of the CVPIA Salmon Doubling Goal.9
Since their initial development, both the CVPIA decision support model and the WRLCM have been expanded and refined, resulting in diversification (and convergence) in the range of decision support applications that they are positioned to inform. For example, the WRLCM has been expanded to include effects of habitat on fish growth and size, and CVPIA decision support models have been developed for winter-run and spring-run Chinook. The R2R decision support model expanded the CVPIA decision support model to include parameters and relationships enabling the integrated evaluation not only of hydraulic and habitat-related actions but also of hatchery and harvest management actions (i.e., “the 4Hs”). The R2R process also applies both the WRLCM and the R2R decision support models in order to compare and contrast performance of the same suite of actions in the two different models.
How Project Operations and Climate Change Affect CV Chinook Salmon
The geographic and temporal diversity and extent of its freshwater habitat use has resulted in CV Chinook salmon being susceptible to impacts from a commensurately diverse range of landscape and ecosystem changes through time including (but not limited to) loss of access to historic spawning and rearing habitat; reduction and degradation of salmon habitat and alterations to streamflows by dams, levees, and water diversions; decreased ecosystem productivity and altered food-web dynamics; increased predation as a result of introduced species and altered hydraulic and habitat conditions; overfishing; hatchery practices (both introgressions10 and competition with natural origin fish); blockage and degradation of streams by mining activities; and climate change (Katz et al., 2012; SEP Group, 2016; Yoshiyama et al., 1998).11 While survival at ocean entry (which itself is strongly related to dynamics during the freshwater portion of the life cycle) and ocean conditions may also play a significant role in survival to adulthood and resulting population dynamics, environmental drivers for freshwater life stages have been shown to have a higher correlation with smolt-to-adult ratios than two marine productivity indices (Michel, 2019).
The four Chinook salmon runs experience different degrees of impact from different stressors generally and from those stressors most closely related (directly or indirectly) to CVP and SWP operations specifically. Of particular interest are stressors that (1) have a significant impact on the species/run timing, (2) are proximate to CVP and SWP operations (e.g., linked either directly or indirectly via a well-established driver-linkage-outcome relationship), and (3) could be significantly reduced or eliminated by changes to CVP and SWP operations alone or in combination with other actions. Stressors meeting these three criteria for winter-, spring- and/or fall-run Chinook salmon in the Central Valley are described below.
Loss of Habitat (Due to Physical Alterations and Infrastructure)
Historically, Chinook salmon adult spawning was widespread throughout the mid and upper river reaches of the Sacramento and San Joaquin basins (Yoshiyama et al., 1998). At least 1,057 miles (mi) of the stream lengths historically available to salmon (or 48 percent of the total habitat, >72 percent of the spawning habitat) have been lost as a function of dams from the original total of 2,183 mi in the Central Valley drainages (Yoshiyama et al., 2001). This impact is especially pronounced for winter- and spring-run Chinook salmon that primarily occupied the higher-elevation portions of the spawning habitat that is more likely to provide the cold spring and snowmelt-fed summer conditions they require for over-summering and the yearling life-history strategy (Cordoleani et al., 2021b; Gutierrez et al., 2024).
The extent of upper elevation habitat blocked from access by dams is mirrored on the valley floor. As much as 80 percent of the floodplain habitat historically accessible to fall-run Chinook salmon has been lost as a function of levees and reduced flows during critical portions of the outmigration window (Katz et al., 2012). Of the
___________________
9 See https://www.fws.gov/project/CVPIA.
10 Introgression is a technical term that refers (in this case) to hatchery fish mating with natural origin fish. It can also be used in other cases where presumed “natural” reproductive behavior has been interrupted—for example, between different run timings as a function of habitat loss. Since their habitat was restricted to below dam reaches, spring-run populations experience significantly higher levels of introgression than fall-run populations.
11 See https://wildlife.ca.gov/Conservation/Fishes/Chinook-Salmon/Winter-run.
remaining portion, the vast majority for the Sacramento drainage is located within the Yolo and Sutter bypasses and activated primarily during flood events. Lack of access to floodplain rearing habitat along channel margins and floodplain habitat being concentrated in the Yolo and Sutter bypasses results in those highly productive habitat types being available to juveniles only during normal and above normal water years, and specifically when the bypasses are inundated, magnifying the difference in rearing habitat quantity and quality experienced by juvenile Chinook salmon in those years.
Degradation in Habitat Quality/Suitability
The disruption of longitudinal connectivity in the river continuum from dams, of latitudinal connectivity with floodplains from levees, and of seasonal hydrodynamics caused by physical structures, diversions, and managed flows has resulted in the disruption of ecosystem and food-web processes in the habitats that remain accessible to CV Chinook salmon. In rivers, the abundance and accessibility of food and the energetic consequences of foraging vary among habitats and through time, providing a shifting mosaic of growth opportunities for fish (Rossi et al., 2024). The flood pulse and seasonal inundation of floodplains drive key hydrologic and geomorphic processes that provide habitat and trophic benefits to river ecosystems and fish species (Junk and Wantzen, 2004; Junk et al., 1989; Poff et al., 2010). Floodplain inundation and the extension of the photic zone it creates have been shown to enhance phytoplankton biomass (Ahearn et al., 2006; Schemel et al., 2004; Sommer et al., 2005), zooplankton growth (Grosholz and Gallo, 2006; Müller-Solger et al., 2002), and drift invertebrate biomass (Benigno and Sommer, 2008; Sommer et al., 2001a,b). Greater frequency of inundation has also been linked to higher levels of invertebrate productivity (Grosholz and Gallo, 2006). It is therefore not surprising that juvenile Chinook salmon rearing in the Yolo Bypass and other CV floodplains and off-channel habitats tend to be larger and in better physical condition than those that rear in the river main channels (Henery et al., 2010; Jeffres et al., 2008; Limm and Marchetti, 2009; SEP Group, 2016; Sommer et al., 2001a).
For outmigrating Chinook salmon in the Yolo Bypass specifically, survival can be similar to that of fish outmigrating through the Sacramento River in dry years (Johnston et al., 2018) despite greater diversity in travel time and growth experienced by fish using the Yolo Bypass (Johnston et al., 2018; Takata et al., 2017), suggesting that it is not only access to the bypass as rearing habitat and migratory pathway but also the extent and duration of Yolo Bypass habitat inundation that support the bioenergetic differences and consequent superior growth observed in Yolo Bypass fish during wetter years (Sommer et al., 2001a; Takata et al., 2017).
For juvenile Chinook salmon rearing and outmigrating in the Sacramento River channel, their survival may be correlated with flow during key periods. In an analysis of flow–survival relationships for Chinook salmon in the Sacramento River across a range of water years, Michel et al. (2021) found a relationship best described by a step function, with three distinct flow thresholds that they defined as minimum (4,259 cubic feet per second [cfs]), historic mean (10,712 cfs), and high (22,872 cfs), where survival varied by threshold: 3.0 percent below minimum, 18.9 percent between minimum and historic mean, 50.8 percent between historic mean and high, and 35.3 percent above high.
Stranding/False Pathways Including Delta Routing
Chinook salmon in the Central Valley evolved to take advantage of the heterogeneity of the habitat both in terms of landform and hydrology. Therefore, impacts to these species resulting from stranding (due to hydraulic variation), false pathways, or detrimental routing are generally attributable to human alteration of the landscape, migration corridors, and hydrograph.
As a function of the cumulative impacts on survival for juvenile Chinook salmon during rearing and outmigration in the river, poor through-Delta survival, which is the final stage of outmigration, can have a disproportionately large influence on survival to adulthood. That is, losing 10 percent of newly emerged fry matters less to adult returns than losing 10 percent of the smolts in the Delta. In recent years, through-Delta survival has been consistently low for San Joaquin River Chinook salmon and more variable for Sacramento River Chinook salmon
(SST, 2017). For San Joaquin River Chinook salmon, since 2002 through-Delta survival has been extremely low (less than 0.2) even in higher flow years (e.g., 2006, 2011), which is not consistent with results of earlier survival studies showing evidence of increased juvenile survival as Delta inflows increased during the migration period (SST, 2017). Perry et al. (2018) found that travel time was inversely related to river inflow in all reaches and that survival was positively related to inflow in reaches that transitioned from bidirectional tidal flows to unidirectional flow with increasing inflows, suggesting that factors affecting through-Delta survival are flow-mediated in the non-tidal reaches at a minimum. In addition, Perry et al. (2018) found that the probability of fish entering the low-survival interior Delta declined as inflow increased.
Interspecies Competition (Including Chinook Runs)
It has long been hypothesized that fall-run redds may be dewatered by the constraining of flow releases for meeting winter-run temperature requirements, and that this could be a primary driver of impacts to fall-run production in the Sacramento River in years where winter-run temperature standards are met and upheld. Stranding of juveniles can also occur during ramp-down periods after large flow releases. Although these phenomena have been observed, the extent of their impact (if any) on cohorts in any given year varies, and information on potential population-level effects is sparse at best.
Entrainment in Diversions
Water export operations contribute to salmonid mortality in the Delta via direct mortality at the pumping facilities. However, direct mortality does not account for the majority of the mortality that juvenile Chinook salmon experience in the Delta (see Chapter 3). The mechanism and magnitude of indirect effects of water project operations on Delta mortality outside the facilities is uncertain. Direct mortality (at the facilities) is a combination of pre-screen and within-facility mortality (including mortality during salvage and transport), and entrainment into the pumps and water conveyance canals (SST, 2017).
Hatchery Potential
Hatchery supplementation remains a key conservation strategy for winter-run Chinook salmon, with two USFWS locations at the Livingston Stone Fish Hatchery just downstream of Shasta Dam and the Coleman National Fish Hatchery along Battle Creek. Coleman also supports populations of fall-run and late-fall-run Chinook salmon. At the time of the 2024 Five-Year Review (NMFS, 2024) the biological status of the winter-run Chinook salmon population had declined since 2016, with the single spawning population on the mainstem Sacramento River at a high risk of extinction. Evidence suggested that the population, experiencing a declining trend in abundance through 2017, had begun to rebuild to a point where the population decline viability criteria might indicate a low risk of extinction for winter-run Chinook salmon, were it not for the influence of the hatchery broodstock remaining at a level above that indicative of a low or moderate extinction risk.
The fall-run stock from both the Sacramento and San Joaquin basin populations is largely dominated by hatchery production and has shown high variability in adult returns, suggesting that hatchery practices are now critical to the sustainability of the fishery (Huber and Carlson, 2015). Nevertheless, spawning of hatchery fish in the wild poses a potentially serious threat to the viability of natural populations (Lindley et al., 2007).
STEELHEAD
Steelhead (Oncorhynchus mykiss) are an anadromous salmon native to tributaries of the Pacific basin in Northeast Asia and North America. They are adapted to cold, nutrient-poor water with seasonally variable food sources (Budy et al., 2019). CCV steelhead (and resident rainbow trout) were historically present in nearly every Central Valley tributary (NMFS, 2014).
The distinct population segment of steelhead found in the California Central Valley is federally listed as threatened. Like the smelt and Chinook previously discussed, designated critical habitat occurs in the areas affected by the seasonal operations of the SWP and the CVP.
There is no commercial fishery for CCV steelhead, but CCV steelhead support a popular and lucrative recreational fishery in Northern California.
Life History and Stages
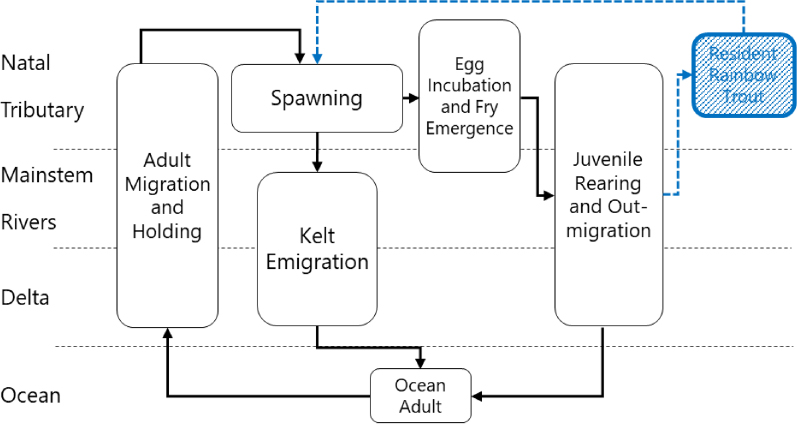
Like other anadromous trout and salmon, steelhead spawn in freshwater, smolts migrate to the ocean to forage for several years, and adults return to their natal streams to spawn. Many populations of CCV steelhead were historically iteroparous (can spawn repeatedly), but iteroparity is now considered to be rare (Moyle, 2002; USBR, 2024c). Adults migrate into freshwater primarily between September and January, adults spawn primarily from March, adults emigrate between March and June, eggs emerge between March and May, and juveniles migrate to the ocean between January and June. However, juvenile steelhead can be found in the Sacramento River year-round (McEwan, 2001; NMFS, 2014; USBR, 2024c). The geographic location of the different life stages is summarized in Figure E-11. Spawning and rearing habitat is typically characterized as perennial streams with clear, cold, fast-flowing water; high dissolved oxygen content; and abundant riffles and gravel (Kershner et al., 2019; USBR, 2024c). Steelhead are different from many other imperiled CV fish in that spawning peaks during lower flows, and wet years are predicted to have lower spawning success.
When steelhead do not migrate to the ocean, they are considered resident, non-listed (ESA) rainbow trout, which complicates conservation and management. There is some indication that the expression of anadromy is decreasing in CCV steelhead, and there are many biological reasons why that pattern is of concern (e.g., CBEC, Inc., 2022). The factors that influence the expression of anadromy versus residency include genetics, individual condition, and water temperature (reviewed in Kendall et al., 2015). Overall, the decision of whether or not to migrate appears to be related to juvenile growth rates and factors that determine those growth rates (Budy et al., 2019).
Steelhead are opportunistic carnivores, and as such, their diet varies by time and space (Budy et al., 2019).
Monitoring of Steelhead and Data on Abundance
CCV steelhead were historically distributed from the upper Sacramento and Pit river systems (upper Sacramento, McCloud, Pit, and Fall rivers) far south to the Kings River (and possibly Kern River systems in wet years) and in both east- and west-side tributaries of the Sacramento River and east-side tributaries of the San Joaquin
SOURCES: USBR (2024c) and adapted from Windell et al. (2017).
River (McEwan, 2001). Currently, CCV steelhead are found in the Sacramento River downstream of Keswick Dam and in the major tributary rivers and creeks in the Sacramento River watershed. Steelhead are present in three tributaries to the San Joaquin River (Stanislaus, Tuolumne, and Merced rivers) as well as in the Calaveras River.
The different life stages of CCV steelhead are monitored in the major rivers and the Delta and using salvage data from the fish collection facilities in the South Delta. Juvenile passage monitoring at Red Bluff Diversion Dam, a primary population metric, is affected by flow and turbidity, which affects trap efficiency. Therefore, comparisons across years can be confounded by monitoring conditions for a given year. Estimates of juvenile passage monitored at the Red Bluff Diversion Dam consistently exceeded 80,000 fish/year from 2002 to 2013, but since 2014, estimates have averaged around 28,000/year (USBR, 2024c). Catch and passage indices exist for Knights Landing, the Sacramento River, Battle Creek, Clear Creek, the American River, and several locations in the Delta. Although variable, most of these indices demonstrate much higher numbers in the early 2000s and much lower numbers more recently (Figure E-12). The observation that CCV steelhead are becoming rare where they were once abundant indicates that an important component of their life-history diversity is being lost (Lindley et al., 2007). In addition, a potential shift to greater rates of residency and lower rates of anadromy is a conservation concern and is a focus of current monitoring (USBR, 2024c).
Conceptual Models and Quantitative Life-Cycle Models
There is currently no quantitative CCV steelhead-specific life-cycle model. For the chapter on steelhead in the Biological Assessment, a habitat suitability modeling approach was used to determine the amount of suitable spawning habitat available at different river flows, and the results are presented as standard Wetted Usable Area (WUA) metrics for different flow scenarios (USBR, 2024c). Based on results from that modeling effort, under all scenarios considered, WUA increases from wetter to drier years.
Trout Unlimited and USFWS have developed a modeling framework to investigate the effects of altered flow and temperature regimes on the expression of steelhead anadromy (promoted or suppressed) in the Lower Yuba River (CBEC, Inc., 2022). The approach is empirical and relates variability in flow and temperature to the level of anadromy. They observed that streams dominated by a resident life-history expression tended to have more stable flows and/or higher flows in the summer. In contrast streams dominated by a migratory life-history expression tended to have more variable flows and/or lower flows in the summer. Temperatures have also become less variable and more stable and warmer in the winter, a pattern thought to be contributing to the increased rates of residency.

SOURCE: USBR (2024c).
How Project Operations and Climate Change Affect Steelhead
The long-term operation of the CVP and SWP impact steelhead via a suite of limiting factors (Perry et al., 2016; USBR, 2024c). First, as with other salmonids, the Projects have blocked steelhead access to 80 percent of its historical habitat and have contributed to the degradation of the remaining accessible habitat. Pumping has led to direct (e.g., mortality due to entrainment) and indirect (e.g., predation, delayed mortality due to collection and transport) impacts (Perry et al., 2016). In water year 2023, a total of 1,015 natural steelhead were “taken” from December 1 to March 31, which exceeded the 50 percent single-year loss threshold for that period. A two-year tagging study of the Salmon Technical Working Group found that 31 percent of steelhead arriving at the export facility ended up in CVP salvage, with the likelihood of that happening increasing with reverse net OMR flow (STWG, 2024).
Other important stressors that derive from project existence and operation include the blocking of coarse sediment from reaching rivers and tributaries, which affects spawning success and can reduce egg-to-fry survival; operation of outdated fish screens resulting in fish entrainment and likely mortality; diversion dams, which can lead to fish stranding and high mortality; fish ladders, which have variable effectiveness at providing passage upstream over dams; simplified habitat due to levee construction and maintenance and disconnection of off-channel habitat; and confounded migratory cues caused by reverse flow in the Central and South Delta. The relative importance of these stressors varies by life stage, with some stressors (such as confounded migration) being highly synergistic with other stressors (e.g., entrainment).
Despite evidence that pumping entrains steelhead, it is not clear that exports are the most important factor affecting steelhead mortality. The Salmon Technical Working Group reviewed recent science including up-to-date analyses of survival through the Delta via acoustic tag networks and concluded that San Joaquin River inflow was the primary environmental driver of through-Delta survival for steelhead and not exports (STWG, 2024). Yet, survival remains low, which is an obvious incongruence.
Many steelhead recovery plan activities are described in USBR (2024c), primarily for the tributaries and headwaters, including but not limited to water temperature management plans, experimental pulse flows to benefit juvenile emigration, temperature-induced anadromy, gravel and large wood supplementation, floodplain and riparian restoration, mitigating predation, and increasing instream flows.
Climate change that leads to increasing water temperatures and altered precipitation patterns is likely to become a substantial stressor for steelhead in the near future. Because water temperature constrains all aspects of trout physiology, cues for migration, and the potential production of the food base, the current and potential future trajectory of climate change is a critical uncertainty for steelhead and will likely include direct (e.g., incipient temperature regime) and indirect effects (i.e., growth potential, oxygen concentrations).
Hatchery Potential
There are many steelhead stocking programs in lakes and tributaries above rim dams, such as the Feather River Hatchery, the Mokelumne River Hatchery, and the Nimbus Fish Hatchery—all run by CDFW (USBR, 2024c). Many populations are largely supported by hatcheries (IEP et al., 2022). Presumably, the combination of hatchery production of steelhead has impacted population genetics, which is a component of viability necessary for delisting. Unlike for the listed Chinook salmon, there are remnant populations of steelhead above all of the major rim dams that in some or all cases may contain ancestral genetics (USBR, 2024c); evidence from other populations suggests these fish could revert to anadromy if/once passage is restored (e.g., Fraik et al., 2021). However, as managed currently, there are no protections for those populations because of their not having an opportunity to express that anadromy.
Uncertainties That Complicate Management of Steelhead
As mentioned previously, there is currently no operational quantitative life-cycle model for steelhead, which limits the ability to assess the relative impacts of different stressors across the life cycle, determine the most limiting factors, and effectively manage long-term operations to reduce impacts on steelhead. NOAA indicates that a life-cycle model may be under development.
The degree to which through-Delta survival of steelhead is directly affected by long-term operations of the projects remains uncertain. What is known is that through-Delta survival is low (< 25 percent), variable annually, and appears to be strongly affected by San Joaquin River inflow (see above). Salvage efficiency goals at the CVP and SWP export facilities are not being met, and predation in the forebays may be a key limiting factor (STWG, 2024).
Although many habitat restoration (e.g., gravel addition) activities have occurred largely in the tributaries and headwaters, the only indication is that the habitat has been enhanced, not that the sub-populations of steelhead have responded to the habitat restoration (USBR, 2024c).
It has been suggested that pulse flows are needed to support different life stages of steelhead, but this recommendation does not appear to be linked to available data or modeling (e.g., WUA) (Rose et al., 2024).
Impacts of Chinook Salmon Decline on Steelhead
During their spawning periods, Chinook salmon eggs have historically been (and continue to be) a major food-source for O. mykiss, especially in the more nutrient-poor, higher-elevation habitat where steelhead historically over-summered. There is reason to believe that the decline in steelhead is closely coupled with the decline in Chinook salmon (especially spring-run Chinook with whom they historically shared habitat), even if buffered by greater life-history plasticity and iteroparity in steelhead.
San Joaquin Steelhead Declines
San Joaquin steelhead populations have declined precipitously to the point where generally only a handful are detected. This decline is likely a function of (1) the San Joaquin basin geography/topography and the Valley portion of rivers (below the dams) being more inhospitable during the warmer times of year than those areas in the Sacramento River for species that over-summer; (2) lower through-Delta survival for species emigrating from the San Joaquin River; and (3) collapse of San Joaquin salmon populations, especially spring-run (see above).
STURGEON
Two species of anadromous sturgeon are the focus of concern in the Bay-Delta because of declining populations. One is Acipenser medirostris, the sDPS of the North American green sturgeon that spawns in the Sacramento River yet spends much of its life in the nearshore marine environment and coastal bays and estuaries along the west coast of North America. In waters inland from the Golden Gate Bridge in California, sDPS green sturgeon are known to range through the estuary and Delta and up the Sacramento, Feather, Stanislaus, San Joaquin, and Yuba rivers (Anderson et al., 2018; Beamesderfer et al., 2004; Israel et al., 2009; Root et al., 2020; Seesholtz et al., 2015). NMFS listed green sturgeon as threatened under the federal ESA on April 7, 2006 (71 Federal Register [FR] 17757), and its critical habitat was designated on October 9, 2009 (74 FR 52300).
The second sturgeon of concern, white sturgeon (Acipenser transmontanus), ranges in the Eastern Pacific from the Gulf of Alaska to Monterey, California, although some are landlocked in the Columbia River drainage, in Montana, and in Shasta Reservoir in California. In addition to residing in the ocean, they also inhabit large rivers and their associated estuaries, including the Sacramento-San Joaquin Delta, where they represent the southernmost spawning population of white sturgeon. Some spawning has recently been documented in the San Joaquin River and may also occur in tributaries including the Feather, Yuba, and Bear rivers.12 White sturgeon are the largest freshwater fish species in North America and can reach 1.6 meters (m) (5.2 feet [ft]) in length, while the maximum length recorded of any age class is 6.1 m (20 ft), with common lengths around 2.1 m (6.9 ft). In one 1995 study, a sample of adults weighed from 34 to 75 kg (75 to 165 lb) (Watson et al., 1995).
Because green sturgeon has been listed as threatened under the ESA for almost two decades, it was the subject of one of the Biological Assessments written during the 2021–2024 reconsultation for the NMFS Biological
___________________
12 See https://wildlife.ca.gov/Conservation/Fishes/Sturgeon/White-Sturgeon.
Opinions (USBR, 2024d). No such assessment exists for white sturgeon, although on June 19, 2024, the California Fish and Game Commission approved white sturgeon as a candidate species for listing under the CESA.13
Life History and Stages
Green sturgeon in the Sacramento-San Joaquin Delta have been well documented since the late 1800s. The life stages of sDPS green sturgeon are schematically shown in Figure E-13 and described in detail in Heublein et al. (2017b). Green sturgeon reach maturity around age 15 and can live to be 60–70 years old and grow to a size of 6.5 feet (USBR, 2024d). Around age 15, adults migrate into the San Francisco estuary from late winter to early spring, with elevated Delta outflow a likely spawning cue for mature adults to enter the river system (Heublein et al., 2009; Moyle et al., 1995). Adult green sturgeon have been found to arrive upstream of the Glenn-Colusa Irrigation District’s water diversion on the upper Sacramento River in April and May to spawn (Moyle, 2002). Generally, adults spawn every three to four years, with a range of two to six years. Following spawning, adults may remain in the Sacramento River basin for up to a year (Miller et al., 2020; Moser et al., 2016), although the upstream extent of historical spawning by green sturgeon in the Sacramento River is unknown (Beamesderfer et al., 2007).
The majority of post-spawn adult green sturgeon will move down river to the Delta during the fall through winter, although some adult green sturgeon move back downstream as early as April and May through the Delta region, either as early post-spawners or as unsuccessful spawners. Fish that over-summer in the upper Sacramento River will move downstream when the river water cools and rain events increase the river’s flow and either hold in the Delta or migrate directly to the ocean (USBR, 2024d).
For juvenile green sturgeon, the mainstem Sacramento River and Delta serve as rearing habitat and a migratory corridor. Some rearing also may occur in the lowest reaches of the lower American River where deep pools
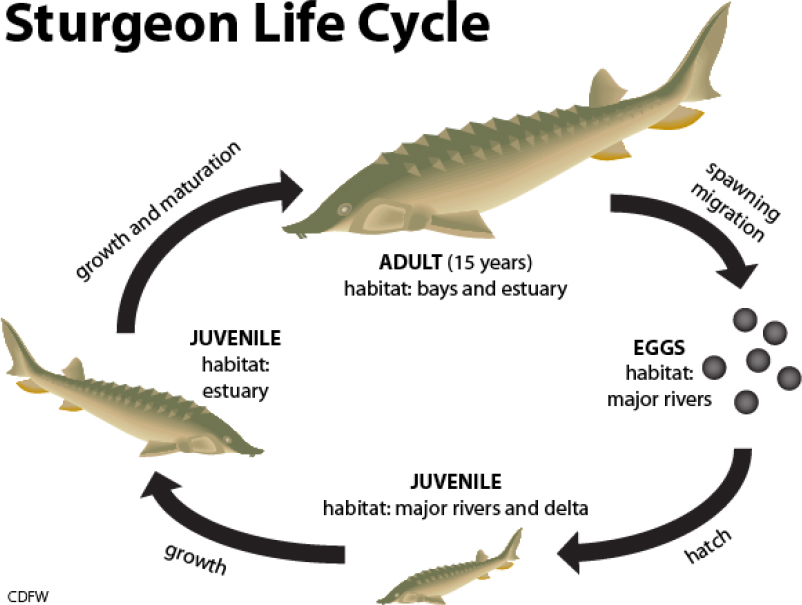
SOURCE: https://wildlife.ca.gov/Conservation/Fishes/Sturgeon.
___________________
13 See https://nrm.dfg.ca.gov/FileHandler.ashx?DocumentID=218091.
occur for rearing of older life stages (Thomas et al., 2013). Juvenile green sturgeon can rear up to four years in the Delta and San Francisco Estuary before entering the ocean as sub-adults (Moyle et al., 1995).
Hildebrand et al. (2016) provide an overview of white sturgeon life-history stages. Although white sturgeon potentially have access to the ocean, use of marine environments is not obligatory to the completion of their life cycle, and most populations show no evidence of consistent or recurring movements to marine environments.
The spawning periodicity of white sturgeon is thought to be two to four years for females and one to two years for males. Kohlhorst (1976) found that (presumably) white surgeon have a spawning period in the Sacramento and San Joaquin rivers between mid-February to late May, with peak activity during March and April. Behavior during spawning is not well known, but it is known that they are communal broadcast spawners, where a female’s eggs are fertilized by many males. When ready to spawn, white sturgeon choose a variety of substrates dependent on the river system, spawning on gravel or rocky substrate in moderate to fast currents, with observed depths of 3–23 m (9.8–75.5 ft) and water velocities at the bottom on a range of 0.6–2.4 m/sec (Marineau et al., 2017). Optimal temperature is 14–16°C (57–61°F), with mortality observed below 8°C (46°F) and above 2°C (36°F) (Coutant, 2004).
Temporally, spawning adult green and white sturgeon can overlap in the general timing of migrations but differ in the median timing of their entry and exit of each reach and the duration spent within each reach (Miller et al., 2020). Spatially, they differ in the location of their spawning reach and route selection in the Delta. Juveniles and sub-adults also differ in their spatial and temporal distributions within the San Francisco Estuary.
Monitoring of Sturgeon and Data on Abundance
Data on green sturgeon abundance come from three primary sources. The first are salvage data from the CVP and SWP pumps. In general, a decrease in sDPS green sturgeon abundance has been inferred from the amount of take observed at the South Delta pumping facilities, although salvage data cannot be used to rigorously quantify abundance. Second, green sturgeon juvenile abundance has been monitored in rotary screw traps at Red Bluff Diversion Dam since 2002 by USFWS. Abundance, measured as catch per unit volume (CPUV), was consistently less than 3 fish/acre-feet before 2011 but near or greater than 3 fish/acre-feet in most years since 2011, including four years during which CPUV exceeded 20 fish/acre-feet (Figure 8-2 in USBR, 2024d). Third, since 2010 acoustic telemetry as well as dual-frequency identification sonar (DIDSON) have been used to locate green sturgeon in the Sacramento River and derive an adult spawner abundance estimate (Mora et al., 2018). These surveys have recently been used to generate an adult sDPS green sturgeon abundance estimate of 2,106 (95% CI: 1,246–2,966; Mora et al., 2018). These estimates do not include the number of spawning adults in the lower Feather or Yuba rivers, where green sturgeon spawning has been confirmed (Seesholtz et al., 2015).
A recent YouTube video from CDFW states that white sturgeon abundance in the Bay-Delta area has declined by 85 percent over the past 25 years.14 Kohlhorst (1980) used commercial passenger fishing boat and tagging records to evaluate white sturgeon population trends in the Sacramento-San Joaquin Estuary. Population estimates suggested that a decrease in abundance occurred between 1967 and 1974 with an increase between 1974 and 1979. The mean size of sturgeon caught increased from 1964 to 1974 and then decreased through 1978. Annual survival rate changed little over this period (84–88 percent with harvest rates of 6–7 percent). Population decline was likely due to poor recruitment from year classes produced after the mid-1950s. Suggested influences on recruitment include degradation of juvenile habitat, environmental contaminants (such as polychlorinated biphenyls), and spawning stock size.
As is common with many sturgeon life histories, low recruitment, delayed age at maturity, and multi-year intervals between spawning events make white sturgeon vulnerable to population interventions (Blackburn et al., 2019). Blackburn et al. (2019) further note that white sturgeon were historically abundant in the Delta, but altered habitat and hydrological conditions coupled with overexploitation are considered to have caused the declines in population abundance and distribution (Hildebrand et al., 2016; Moyle et al., 2011; Nichols et al., 1986). CDFW’s most recent estimate of the five-year average of the harvestable population is about 33,000 fish (CDFW, 2023). Recruitment is influenced by survival at early life stages (Hildebrand et al., 1999; Jackson et al., 2016 at p. 172 citing Kohlhorst et al., 1991; Secor et al., 2002) and as such is dependent on the frequency of high-magnitude spring-summer river flows.
___________________
Conceptual Models and Quantitative Life-Cycle Models for Sturgeon
Life-cycle models for both sturgeon species have been developed (Figure E-14), and they are remarkably similar, with the exception of the locations of various life stages. Modeling of green sturgeon is inherently more complex because of their multi-state movements compared to the mostly local estuarine white sturgeon.
Israel et al. (2009) developed a conceptual life-history model of white sturgeon (Figure E-15) and the factors that affect reproduction, growth, and survival in the Sacramento-San Joaquin rivers and the Delta. This model can be used to organize, visualize, and evaluate how the complex life history of white sturgeon relates to the spatial and temporal variability of riverine and estuarine ecosystems and potential consequences of ecosystem restoration and water management alternatives. The model has a geographic emphasis on the Sacramento River and Delta regions, although the entire distribution of white sturgeon should be considered when population-level responses are of concern, because of the migratory life history of this species.
How Project Operations and Climate Change Affect Sturgeon
The sturgeon conceptual models in Figure E-14 suggest that Project operations negatively affect all life stages of green and white sturgeon in much the same manner as they do Chinook salmon, with an emphasis on blockage from traditional spawning locations, altered flow regimes that misroute fish and prevent access to floodplain habitat, and temperature-dependent mortality that decreases spawning success and egg survival. Finally, larval, juvenile, and adult sturgeon risk entrainment in the South Delta if exports are high enough.
Like the other salmonids, the viability of sDPS green sturgeon is constrained by many factors including a small population size and restriction of spawning sites to a few locations (USBR, 2024d)—the latter of which is a direct result of the CVP and SWP. However, according to USBR (2024d), “there is much uncertainty regarding the scope of threats and the viability of the population based on abundance indices.” The most recent five-year status review for sDPS green sturgeon found that some threats to the species have been eliminated, such as take from commercial fisheries and removal of some passage barriers, but many other threats still exist (NMFS, 2021). According to McElhany et al. (2000), the sDPS does not meet the definition of viable as an independent population having a negligible risk of extinction due to threats from demographic variation, local environmental variation, and genetic diversity changes over a 100-year time frame.
Hatchery Potential
The UC Davis Green Sturgeon Broodstock program is the only captive breeding population of green sturgeon.15 A variety of hatcheries for white sturgeon exist in the region, because of the harvesting of caviar.
___________________
15 See https://fishconservationphysiologylab.faculty.ucdavis.edu/research/sturgeon/.
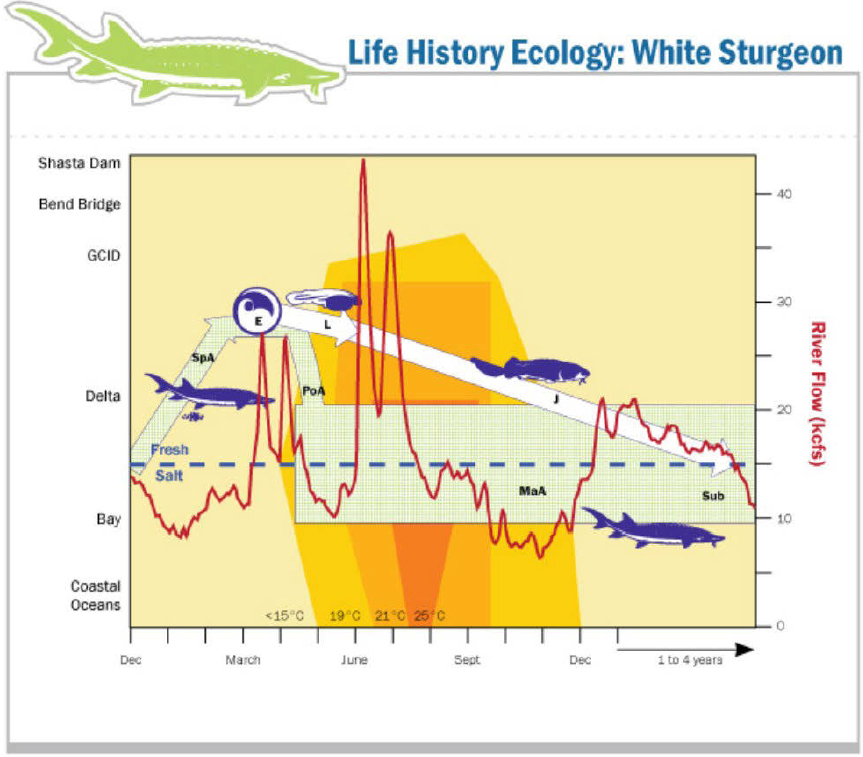
NOTE: Spawning white sturgeon adults (SpA) enter the river in the winter to spawn and outmigrate quickly as post-spawners (PoA). Eggs (E) are broadcast spawned and mature into larvae (L), then enter and remain in the estuary at juveniles (J). Blue line represents 2006 daily average flow at Vernalis (waterdata.usgs.gov). Temperature data are 2006 data from Bend Bridge, GCID, Bethel Island, Port Chicago, and Martinez (cdec.water.gov).
SOURCE: Figure 4 from Israel et al. (2009).
REFERENCES
Ahearn, D. S., J. H. Viers, J. F. Mount, and R. A. Dahlgren. 2006. “Priming the Productivity Pump: Flood Pulse Driven Trends in Suspended Algal Biomass Distribution Across a Restored Floodplain.” Freshwater Biology 51(8):1417–1433. https://doi.org/10.1111/j.1365-2427.2006.01580.x.
Anderson, J. T., G. Schumer, P. J. Anders, K. Horvath, and J. E. Merz. 2018. “Confirmed Observation: A North American Green Sturgeon Acipenser medirostris Recorded in the Stanislaus River, California.” Journal of Fish and Wildlife Management 9(2):624–630. https://doi.org/10.3996/012018-JFWM-006.
Anderson, M. L. 2024. Delta Watershed Climate and Expected Change. Presentation to the National Academies of Sciences, Meeting 1, January 31, 2024, Sacramento, CA.
Baxter, R., R. Breuer, L. Brown, et al. 2008. Pelagic Organism Decline Progress Report: 2007 Synthesis of Results.
Baxter, R., L. R. Brown, G. Castillo, et al. 2015. An Updated Conceptual Model of Delta Smelt Biology: Our Evolving Understanding of an Estuarine Fish (No. 90). Interagency Ecological Program, California Department of Water Resources.
Beamesderfer, R., M. Simpson, G. Kopp, J. Inman, A. Fuller, and D. Demko. 2004. Historical and Current Information on Green Sturgeon Occurrence in the Sacramento and San Joaquin Rivers and Tributaries. SP Cramer & Associates, Inc for State Water Contractors, Sacramento, California. https://www.waterboards.ca.gov/waterrights/water_issues/programs/bay_delta/california_waterfix/exhibits/docs/petitioners_exhibit/dwr/part2/DWR-1130%20Beamesderfer_et_al_2004_green_sturgeon_spawning_locations.pdf.
Beamesderfer, R. C., M. L. Simpson, and G. J. Kopp. 2007. “Use of Life History Information in a Population Model for Sacramento Green Sturgeon.” Environmental Biology of Fishes 79(3):315–337. https://citeseerx.ist.psu.edu/document?repid=rep1&type=pdf&doi=72a669c63d8f95b8cb6c587090b18e9f61f87adf.
Benigno, G. M., and T. R. Sommer. 2008. “Just Add Water: Sources of Chironomid Drift in a Large River Floodplain.” Hydrobiologia 600:297–305. https://doi.org/10.1007/s10750-007-9239-2.
Bennett, W. A. 2005. “Critical Assessment of the Delta Smelt Population in the San Francisco Estuary, California.” San Francisco Estuary & Watershed Science 3(2). https://doi.org/10.15447/sfews.2005v3iss2art1.
Blackburn, S. E., M. L. Gingras, J. DuBois, Z. J. Jackson, and M. C. Quist. 2019. “Population Dynamics and Evaluation of Management Scenarios for White Sturgeon in the Sacramento—Joaquin River Basin.” North American Journal of Fisheries Management 39(5):896–912. https://doi.org/10.1002/nafm.10316.
Bradford, M. J., and P. S. Higgins. 2001. “Habitat-, Season-, and Size-Specific Variation in Diel Activity Patterns of Juvenile Chinook Salmon (Oncorhynchus Tshawytscha) and Steelhead Trout (Oncorhynchus Mykiss).” Canadian Journal of Fisheries and Aquatic Sciences 58(2):365–374. https://doi.org/10.1139/f00-253.
Brown, L. R., D. E. Ayers, B. Bergamaschi, et al. 2024. Physics to Fish—Understanding the Factors That Create and Sustain Native Fish Habitat in the San Francisco Estuary. U.S. Geological Survey Open-File Report 2023-1087. https://doi.org/10.3133/ofr20231087.
Brown, L. R., W. A. Bennett, R. W. Wagner, et al. 2013. “Implications for Future Survival of Delta Smelt from Four Climate Change Scenarios for the Sacramento–San Joaquin Delta, California.” Estuaries and Coasts 36:754–774. https://doi.org/10.1007/s12237-013-9585-4.
Brown, L. R., L. M. Komoroske, R. W. Wagner, et al. 2016. “Coupled Downscaled Climate Models and Ecophysiological Metrics Forecast Habitat Compression for an Endangered Estuarine Fish.” PLoS One 11(1):e0146724. https://doi.org/10.1371/journal.pone.0146724.
Budy, P., K. B. Rogers, Y. Kanno, et al. 2019. “Distribution and Status of Trout and Char in North America.” Chapter 8 in Diversity and Status of Trout and Char of the World, edited by J. L. Kershner, J. E. Williams, and R. E. Gresswell. American Fisheries Society. Symposium Book.
CBEC, Inc. 2022. Lower Yuba River Flow and Temperature Relationships for Supporting Anadromous Steelhead Yuba River Flow Effects Modeling Project Lower Yuba River, California. Prepared for Trout Unlimited and U.S. Fish and Wildlife Service. CBEC Project # 19-1029.
CDFG (California Department of Fish and Game). 2009a. A Status Review of the Longfin Smelt (Spirinchus thaleichthys) in California. Report to the Fish and Game Commission.
CDFG. 2009b. Effects Analysis—State Water Project Effects on Longfin Smelt, California Department of Fish and Game.
CDFW (California Department of Fish and Wildlife). 2023. 2023 Sacramento River Sturgeon Spawning Study Report. CDFW North Central Region, Rancho Cordova, CA. https://nrm.dfg.ca.gov/FileHandler.ashx?DocumentID=214950.
CDWR (California Department of Water Resources), CDFW, State Water Contractors, and USFWS. 2020. Longfin Smelt Science Plan 2020–2030. https://water.ca.gov/-/media/DWR-Website/Web-Pages/Programs/State-Water-Project/Files/ITP/ITP-Longfin-Science-Plan_SWP_12232020_-FINAL.pdf.
Christman, M. A., S. Khanna, J. Z. Drexler, and M. J. Young. 2023. “Ecology and Ecosystem Effects of Submerged and Floating Aquatic Vegetation in the Sacramento–San Joaquin Delta.” San Francisco Estuary and Watershed Science 20(4). https://doi.org/10.15447/sfews.2023v20iss4art3.
Cloern, J. E., and A. D. Jassby. 2012. “Drivers of Change in Estuarine-Coastal Ecosystems: Discoveries from Four Decades of Study in San Francisco Bay.” Reviews of Geophysics 50. https://doi.org/10.1029/2012RG000397.
Cordoleani, F., C. C. Phillis, G. Whitman, A. Sturrock, and C. Johnson. 2021a. Life History Diversity in Central Valley Butte Creek Spring-run Chinook Salmon Population: Implications for Future Management. Technical report, State Water Contractors, Sacramento, CA.
Cordoleani, F., C. C. Phillis, A. M. Sturrock, et al. 2021b. “Threatened Salmon Rely on a Rare Life History Strategy in a Warming Landscape.” Nature Climate Change 11:982–988. https://doi.org/10.1038/s41558-021-01186-4.
Coutant, C. C. 2004. “A Riparian Habitat Hypothesis for Successful Reproduction of White Sturgeon.” Reviews in Fisheries Science 12(1):23–73. https://doi.org/10.1080/10641260490273023.
Dege, M., and L. R. Brown. 2004. “Effect of Outflow on Spring and Summertime Distribution and Abundance of Larval and Juvenile Fishes in the Upper San Francisco Estuary.” American Fisheries Society Symposium 39:49–65.
EPA (U.S. Environmental Protection Agency). 2001. Salmonid Behavior and Water Temperature. Issue Paper. Prepared as part of EPA Region 10 Temperature Water Quality Criteria Guidance Development Project by S.T. Sauter, J. McMillan, and J. Dunham. EPA-910-D-01-001. EPA Region 10.
EPA. 2003. EPA Region 10 Guidance for Pacific Northwest State and Tribal Temperature Water Quality Standards. EPA 910-B-03-002.
Fisher, F. W. 1994. “Past and Present Status of Central Valley Chinook Salmon.” Conservation Biology 8:870–873. https://www.jstor.org/stable/2386533.
Fraik, A. K., J. R. McMillan, M. Liermann, et al. 2021. “The Impacts of Dam Construction and Removal on the Genetics of Recovering Steelhead (Oncorhynchus mykiss) Populations across the Elwha River Watershed.” Genes 12(1):89. https://doi.org/10.3390/genes12010089.
Fry, D. H. 1961. “King Salmon Spawning Stocks of the California Central Valley, 1940-1959.” California Fish and Game 47(1):55–71.
Grimaldo, L. F., W. E. Smith, and M. L. Nobriga. 2021. “Re-Examining Factors That Affect Delta Smelt (Hypomesus transpacificus) Entrainment at the State Water Project and Central Valley Project in the Sacramento–San Joaquin Delta.” San Francisco Estuary & Watershed Science 19(1). https://doi.org/10.15447/sfews.2021v19iss1art5.
Grimaldo, L. F., T. Sommer, N. Van Ark, et al. 2009. “Factors Affecting Fish Entrainment into Massive Water Diversions in a Tidal Freshwater Estuary: Can Fish Losses Be Managed?” North American Journal of Fisheries Management 29(5):1253–1270. https://doi.org/10.1577/M08-062.1.
Grosholz, E., and E. Gallo. 2006. “The Influence of Flood Cycle and Fish Predation on Invertebrate Production on a Restored California Floodplain.” Hydrobiologia 568:91–109. https://doi.org/10.1007/s10750-006-0029-z.
Gutierrez, M., H. Glenn, M. Colombano, C. Ambrose, J. Rennert, and J. Ambrose. 2024. Central Valley Spring-Run Chinook Salmon in the San Joaquin River Basin. U.S. Department of Commerce, NOAA Technical Memorandum NMFSSWFSC-706. https://doi.org/10.25923/t7c1-3229.
Healey, M. C. 1991. “Life History of Chinook Salmon.” Pp. 312–393 in Pacific Salmon Life Histories, edited by C. Groot and L. Margolis. Vancouver: University of British Columbia Press.
Hendrix, N., A. Criss, E. Danner, et al. 2014. Life Cycle Modeling Framework for Sacramento River Winter-Run Chinook Salmon. NOAA-TM-NMFS-SWFSC-530.
Hendrix, N., A. M. K. Osterback, E. Jennings, et al. 2019. Model Description for the Sacramento River Winter-Run Chinook Salmon Life Cycle Model. NOAA. https://oceanview.pfeg.noaa.gov/wrlcm/documents/publications/Hendrix%20et%20al%202019_WRLCM%20Description.pdf.
Henery, R. E., T. R. Sommer, and C. R. Goldman. 2010. “Growth and Methylmercury Accumulation in Juvenile Chinook Salmon in the Sacramento River and its Floodplain, the Yolo Bypass.” Transactions of the American Fisheries Society 139:550–563. https://doi.org/10.1577/T08-112.1.
Heublein, J. C., R. J. Bellmer, R. D. Chase, et al. 2017a. Improved Fisheries Management Through Life Stage Monitoring: The Case for the Southern Distinct Population Segment of North American Green Sturgeon and the Sacramento-San Joaquin River White Sturgeon. NOAA technical memorandum; NOAA-TM-NMFS-SWFSC588. https://repository.library.noaa.gov/view/noaa/15997.
Heublein, J. C., R. Bellmer, R.D. Chase, et al. 2017b. Life History and Current Monitoring Inventory of San Francisco Estuary Sturgeon. NOAA Technical Memorandum. U.S. Department of Commerce, National Oceanic and Atmospheric Administration, National Marine Fisheries Service, Southwest Fisheries Science Center. https://repository.library.noaa.gov/view/noaa/15998.
Heublein, J. C., J. T. Kelly, C. E. Crocker, A. P. Klimley, and S. T. Lindley. 2009. “Migration of Green Sturgeon, Acipenser medirostris, in the Sacramento River.” Environmental Biology of Fishes 84(3):245–225. https://doi.org/10.1007/s10641008-9432-9.
Hildebrand, L., C. McLeod, and S. McKenzie. 1999. “Status and Management of White Sturgeon in the Columbia River in British Columbia, Canada: An Overview.” Journal of Applied Ichthyology 15:164–172. https://doi.org/10.1111/j.1439-0426.1999.tb00227.x.
Hildebrand, L. R., A. D. Schreier, K. Lepla, et al. 2016. “Status of White Sturgeon (Acipenser transmontanus Richardson 1863) Throughout the Species Range, Threats to Survival, and Prognosis for the Future.” Journal of Applied Ichthyology 32:261–312. https://doi.org/10.1111/jai.13243.
Hobbs, J. A., L. S. Lewis, N. Ikemiyagi, T. Sommer, and R. D. Baxter. 2010. “The Use of Otolith Strontium Isotopes (87Sr/86Sr) to Identify Nursery Habitat for a Threatened Estuarine Fish.” Environmental Biology of Fishes 89:557–569.
Hobbs, J. A., L. S. Lewis, M. Willmes, C. Denney, and E. Bush. 2019. “Complex Life Histories Discovered in a Critically Endangered Fish. Scientific Reports 9(1):16772. https://doi.org/10.1038/s41598-019-52273-8.
Huber, E. R., and S. M. Carlson. 2015. “Temporal Trends in Hatchery Releases of Fall-Run Chinook Salmon in California’s Central Valley.” San Francisco Estuary & Watershed Science 13(2). https://doi.org/10.15447/sfews.2015v13iss2art3.
Hung, T, M. M. Rahman, L. S. Lewis, et al. 2024. “Laboratory-bred Longfin Smelt Produced Offspring in the First Year in Captivity.” North American Journal of Aquaculture 86:228–233. https://doi.org/10.1002/naaq.10327.
IEP (Interagency Ecological Program), J. Speegle, R. McKenzie, et al. 2022. “Interagency Ecological Program: Over Four Decades of Juvenile Fish Monitoring Data from the San Francisco Estuary, Collected by the Delta Juvenile Fish Monitoring Program, 1976-2022 ver 11.” Environmental Data Initiative. https://doi.org/10.6073/pasta/57b6c257edd72691702f9731d5fe4172.
IEP MAST, Management, Analysis and Synthesis Team). 2015. An Updated Conceptual Model of Delta Smelt Biology: Our Evolving Understanding of Estuarine Fish. Technical Report 90. Prepared for the San Francisco Bay/Delta Estuary.
Israel, J., A. Drauch, and M. Gingras. 2009. Life History Conceptual Model for White Sturgeon. Unpubl. technical report to Bay Delta Ecosystem Restoration and Improvement Program, Sacramento, CA.
Jackson, Z. J., J. J. Gruber, and J. P. Van Eenennaam. 2016. “White Sturgeon Spawning in the San Joaquin River, California, and Effects of Water Management.” Journal of Fish and Wildlife Management 7(1):171–180. https://doi.org/10.3996/092015-JFWM-092.
Jeffres, C.A., J. J. Opperman, and P. B. Moyle. 2008. “Ephemeral Floodplain Habitats Provide Best Growth Conditions for Juvenile Chinook Salmon in a California River.” Environmental Biology of Fishes 83:449–458. https://doi.org/10.1007/s10641-008-9367-1.
Johnston, M. E., A. E. Steel, M. Espe, T. Sommer, A. P. Klimley, P. Sandstrom, and D. Smith. 2018. “Survival of Juvenile Chinook Salmon in the Yolo Bypass and the Lower Sacramento River, California.” San Francisco Estuary and Watershed Science 16(2). https://doi.org/10.15447/sfews.2018v16iss2art4.
Jørgensen, D. 2002. “Extinction and the End of Futures.” History & Theory: Studies in the Philosophy of History. https://doi.org/10.1111/hith.12258.
Junk, W. J., P. B. Bayley, and R. E. Sparks. 1989. “The Flood Pulse Concept in River-Floodplain Systems.” Canadian Special Publication of Fisheries and Aquatic Sciences 106:110–127.
Junk, W. J., and K. M. Wantzen. 2004. “The Flood Pulse Concept: New Aspects, Approaches and Applications-An Update.” Pp. 114–149 in Second International Symposium on the Management of Large Rivers for Fisheries. Food and Agriculture Organization and Mekong River Commission, FAO Regional Office for Asia and the Pacific.
Katz, J., P. B. Moyle, R. M. Quiñones, J. Israel, and S. Purdy. 2012. “Impending Extinction of Salmon, Steelhead, and Trout (Salmonidae) in California.” Environmental Biology of Fishes 96(10-11):1169–1186. https://doi.org/10.1007/s10641-012-9974-8.
Kendall, N. W., J. R. McMillan, M. R. Sloat, et al. 2015. “Anadromy and Residency in Steelhead and Rainbow Trout (Oncorhynchus Mykiss): A Review of the Processes and Patterns.” Canadian Journal of Fisheries and Aquatic Sciences 72:319–342. https://doi.org/10.1139/cjfas-2014-0192.
Kershner, J. L., J. E. Williams, R. E. Gresswell, and J. Lobón-Cerviá. 2019. Trout and Char of the World. Bethesda, MD: American Fisheries Society.
Kimmerer, W. J. 2002. “Effects of Freshwater Flow on Abundance of Estuarine Organisms: Physical Effects or Trophic Linkages? Marine Ecology Progress Series 243:39–55. https://doi.org/10.3354/meps243039.
Kimmerer, W. J. 2008. “Losses of Sacramento River Chinook Salmon and Delta Smelt to Entrainment in Water Diversions in the Sacramento–San Joaquin Delta.” San Francisco Estuary & Watershed Science 6(2). https://doi.org/10.15447/sfews.2008v6iss2art2.
Kohlhorst, D. W. 1976. “Sturgeon Spawning in the Sacramento River in 1973, as Determined by Distribution of Larvae.” California Fish and Game 62(1):32–40.
Kohlhorst, D. W. 1980. “Recent Trends in the White Sturgeon Population in California’s Sacramento San Joaquin Estuary.” California Fish and Game 66(4):210–219.
Kohlhorst, D. W., L.W. Botsford, J. S. Brennan, and G.M. Cailliet. 1991. “Aspects of the Structure and Dynamics of an Exploited California Population of White Sturgeon (Acipenser transmontanus).” Pp. 277–293 in Acipenser: Actes du premier colloque sur l’esturgeon, edited by P. Williot. CHEMAGREF, Bordeaux, France.
Komoroske, L. M., R. E. Connon, J. Lindberg, et al. 2014. “Ontogeny Influences Sensitivity to Climate Change Stressors in an Endangered Fish.” Conservation Physiology 2(1):cou008, https://doi.org/10.1093/conphys/cou008.
Lewis, L. S., M. Willmes, A. Barros, P. K. Crain, and J. A. Hobbs. 2020. “Newly Discovered Spawning and Recruitment of Threatened Longfin Smelt in Restored and Under-Explored Tidal Wetlands.” Ecology 2020:4–7. https://doi.org/10.1002/ecy.2868.
Limm, M. P., and M. P. Marchetti. 2009. “Juvenile Chinook Salmon (Oncorhynchus Tshawytscha) Growth in Off-Channel and Main-Channel Habitats on the Sacramento River, CA Using Otolith Increment Widths.” Environmental Biology of Fishes 85(2):141–151. https://doi.org/10.1007/s10641-009-9473-8.
Lindley, S. T., R. Schick, B. P. May, et al. 2004. Population Structure of Threatened and Endangered Chinook Salmon ESUs in California’s Central Valley Basin. https://www.noaa.gov/sites/default/files/legacy/document/2020/Oct/07354626200.pdf.
Lindley, S. T., R. S. Schick, E. Mora, et al. 2007. “Framework for Assessing the Viability of Threatened and Endangered Chinook Salmon and Steelhead in the Sacramento-San Joaquin Basin.” San Francisco Estuary & Watershed Sciences 5(1). http://repositories.cdlib.org/jmie/sfews/vol5/iss1/art4.
Major, R. L., J. Ito, S. Ito, and H. Godfrey. 1978. Distribution and Origin of Chinook Salmon (Oncorhynchus Tshawytscha) in Offshore Waters of the North Pacific Ocean. International North Pacific Fisheries Commission Bulletin 38.
Marineau, M. D., S. A. Wright, D. R. Whealdon-Haught, and P. J. Kinze. 2017. “Physical Characteristics of the Lower San Joaquin River, California, in Relation to White Sturgeon Spawning Habitat, 2011–14.” USGS Scientific Investigations Report. https://doi.org/10.3133/sir20175069.
Marston, D., C. Mesick, A. Hubbard, et al. 2012. “Delta Flow Factors Influencing Stray Rates of Escaping Adult San Joaquin River Fall-Run Chinook Salmon (Oncorhynchus tshawytscha).” San Francisco Estuary & Watershed Science 10(4). https://doi.org/10.15447/sfews.2012v10iss4art3.
Maunder, M. N., and R. B. Deriso. 2011. “A State–Space Multistage Life Cycle Model to Evaluate Population Impacts in the Presence of Density Dependence: Illustrated with Application to Delta Smelt (Hyposmesus transpacificus).” Canadian Journal of Fisheries and Aquatic Science 68:1285–1306. https://doi.org/10.1139/F2011-07.
Maunder, M. N., R. B. Deriso, and C. H. Hanson. 2015. “Use of State-Space Population Dynamics Models in Hypothesis Testing: Advantages Over Simple Log-Linear Regressions for Modeling Survival, Illustrated with Application to Longfin Smelt (Spirinchus thaleichthys).” Fisheries Research 164:102–111. https://doi.org/10.1016/j.fishres.2014.10.017.
McClure, M. M., S. M. Carlson, T. J. Beechie, et al. 2008. “Evolutionary Consequences of Habitat Loss for Pacific Anadromous Salmonids.” Evolutionary Applications 1(2):300–318. https://doi.org/10.1111/j.1752-4571.2008.00030.x.
McElhany, P., M. H. Ruckelshaus, M. J. Ford, T. C. Wainwright, and E. P. Bjorkstedt. 2000. “Viable Salmonid Populations and the Recovery of Evolutionarily Significant Units.” Pp. 1–174 in National Marine Fisheries Service.
McEwan, D. 2001. “Central Valley Steelhead.” Pp. 1–44 in Fish Bulletin 179(1): Contributions to the Biology of Central Valley Salmonids, edited by R. L. Brown. Sacramento, CA: California Department of Fish and Game.
McMillan, J. R., B. Morrison, N. Chambers, et al. 2023. “A Global Synthesis of Peer-Reviewed Research on the Effects of Hatchery Salmonids on Wild Salmonids.” Fisheries Management and Ecology 30:446–463. https://doi.org/10.1111/fme.12643.
Merz, J. E., P. S. Bergman, J. F. Melgo, and S. Hamilton. 2013. “Longfin Smelt: Spatial Dynamics and Ontogeny in the San Francisco Estuary, California.” California Fish and Game 99(3):122–148.
Merz, J. E., D. G. Delaney, J. D. Setka, and M. L. Workman. 2016. “Seasonal Rearing Habitat in a Large Mediterranean-Climate River: Management Implications at the Southern Extent of Pacific Salmon (Oncorhynchus spp.).” River Research and Applications 32(6):1220–1231. https://doi.org/10.1002/rra.2969.
Michel, C. J. 2019. “Decoupling Outmigration from Marine Survival Indicates Outsized Influence of Streamflow on Cohort Success for California’s Chinook Salmon Populations.” Canadian Journal of Fisheries and Aquatic Sciences 76(8):1398–1410. https://doi.org/10.1139/cjfas-2018-0140.
Michel, C. J., J. J. Notch, F. Cordoleani, A. J. Ammann, and E. M. Danner. 2021. “Nonlinear Survival of Imperiled Fish Informs Managed Flows in a Highly Modified River.” Ecosphere 12(5):e03498. https://doi.org/10.1002/ecs2.3498.
Miller, E. A., G. P. Singer, M. L. Peterson, et al. 2020. “Spatio-temporal Distribution of Green Sturgeon (Acipenser medirostris) and White Sturgeon (A. transmontanus) in the San Francisco Estuary and Sacramento River, California.” Environmental Biology of Fish 103:577–603. https://doi.org/10.1007/s10641-020-00972-x.
Mora, E. A., R. D. Battleson, S. T. Lindley, et al. 2018. “Estimating the Annual Spawning Run Size and Population Size of the Southern Distinct Population Segment of Green Sturgeon.” Transactions of the American Fisheries Society 147:195–203. https://doi.org/10.1002/tafs.10009.
Mora, V., J. Oceguera Zavala, J. A. Jimenez, and CDFW. 2024. “Expansion into San Pablo Bay: Smelt Larva Survey & 20-mm Survey Updates.” Interagency Ecological Program 2024 Annual Workshop. April 23–25, 2024.
Moser, M. L., J. A. Israel, M. Neuman, et al. 2016. “Biology and Life History of Green Sturgeon (Acipenser medirostris Ayres, 1854): State of the Science.” Journal of Applied Ichthyology 32:67–86. https://doi.org/10.1111/jai.13238.
Moyle, P. B. 2002. Inland Fishes of California: Revised and Expanded. Berkeley, CA: University of California Press.
Moyle, P. B., R. M. Yoshiyama, and J. E. Williams, eds. 1995. Fish Species of Special Concern of California, 2nd edition. Sacramento, CA: California Department of Fish and Game
Moyle, P. B., L. R. Brown, J. R. Durand, and J. A. Hobbs. 2016. “Life History and Decline of a Once-Abundant Species in the San Francisco Estuary.” San Francisco Estuary & Watershed Science 14(2). https://doi.org/10.15447/sfews.2016v14iss2art6.
Moyle, P. B., J. V. E. Katz, and R. M. Quiñones. 2011. “Rapid Decline of California’s Native Inland Fishes: A Status Assessment.” Biological Conservation 144:2414–2423. https://citeseerx.ist.psu.edu/document?repid=rep1&type=pdf&doi=97a8817d3c037028df857f61f76ad375165acf17.
Müller-Solger, A. B., A. D. Jassby, and D. C. Müller-Navarra. 2002. “Nutritional Quality of Food Resources for Zooplankton (Daphnia) in a Tidal Freshwater System (Sacramento-San Joaquin River Delta).” Limnology and Oceanography 475:1468–1476. https://doi.org/10.4319/lo.2002.47.5.1468.
Nichols, F. H., J. E. Cloern, S. N. Luoma, and D. H. Peterson. 1986. “The Modification of an Estuary.” Science 231:567–573. DOI: 10.1126/science.231.4738.567.
NMFS (National Marine Fisheries Service). 2014. “Recovery Plan for the Evolutionarily Significant Units of Sacramento River Winter-run Chinook Salmon and Central Valley Spring-run Chinook Salmon and the Distinct Population Segment of California Central Valley Steelhead. California Central Valley Area Office. July 2014.” https://www.fisheries.noaa.gov/resource/document/recovery-plan-evolutionarily-significant-units-sacramento-river-winter-run.
NMFS. 2021. Southern Distinct Population Segment of North American Green Sturgeon (Acipenser medirostris); 5-Year Review: Summary and Evaluation. California Central Valley Office, Sacramento, CA.
NMFS. 2024. 2024 5-Year Review: Summary & Evaluation of Sacramento River Winter-Run Chinook Salmon. https://doi.org/10.25923/4t2m-kt5217.
Nobriga, M. L., and J. A. Rosenfield. 2016. “Population Dynamics of an Estuarine Forage Fish: Disaggregating Forces Driving Long-Term Decline of Longfin Smelt in California’s San Francisco Estuary.” Transactions of the American Fisheries Society 145:44–58. https://doi.org/10.1080/00028487.2015.1100136.
Perry, R.W., R. A. Buchanan, P. L. Brandes, J. R. Burau, and J.A. Israel. 2016. “Anadromous Salmonids in the Delta: New Science, 2006–2016.” San Francisco Estuary and Watershed Science 14(2). https://doi.org/10.15447/sfews.2016v14iss2art7.
Perry, R. W., A. C. Pope, J. G. Romine, et al. 2018. “Flow-Mediated Effects on Travel Time, Routing, and Survival of Juvenile Chinook Salmon in a Spatially Complex, Tidally Forced River Delta. Canadian Journal of Fisheries and Aquatic Sciences 75(11):1886–1901. https://doi.org/10.1139/cjfas-2017-0310.
Peterson, J. T., and A. Duarte. 2020. “Decision Analysis for Greater Insights into the Development and Evaluation of Chinook Salmon Restoration Strategies in California’s Central Valley.” Restoration Ecology 28(6):1596–1609. https://doi.org/10.1111/rec.13244.
Phillis, C. C., A. M Sturrock, R. C. Johnson, and P. K. Weber. 2018. “Endangered Winter-Run Chinook Salmon Rely on Diverse Rearing Habitats in a Highly Altered Landscape.” Biological Conservation 217:358–362. https://doi.org/10.1016/j.biocon.2017.10.023.
Poff, N. L., B. D. Richter, A. H. Arthington, et al. 2010. “The Ecological Limits of Hydrologic Alteration (ELOHA): A New Framework for Developing Regional Environmental Flow Standards.” Freshwater Biology 55(1):147–170. https://doi.org/10.1111/j.1365-2427.2009.02204.x.
Polansky, L., K. B. Newman, and L. Mitchell. 2021. “Improving Inference for Nonlinear State-Space Models of Animal Population Dynamics Given Biased Sequential Life Stage Data.” Biometrics 77(1):352–361. https://doi.org/10.1111/biom.13267.
Quinn, T. P. 2005. The Behavior and Ecology of Pacific Salmon and Trout. Seattle: University of Washington Press.
Rahman, M. M., L. S. Lewis, N. A. Fangue, R. E. Connon, and T. Hung. 2023. “Effects of Salinity on Fertilization, Hatching, and Larval Performance of Longfin Smelt Spirinchus thaleichthys.” Aquaculture Research 9984382. https://doi.org/10.1155/2023/9984382.
Ricker, W. E. 1972. “Heredity and Environmental Factors Affecting Certain Salmonid Populations.” Pp. 27–160 in The Stock Concept in Pacific Salmon, edited by R. C. Simon and P. A. Larkin. H. R. MacMillan Lectures in Fisheries. Vancouver: University of British Columbia.
Roni, P., and T. P. Quinn. 1995. “Geographic Variation in Size and Age of North American Chinook Salmon.” North American Journal of Fisheries Management 15(2):325–345. https://doi.org/10.1577/1548-8675(1995)015<0325:GVISAA>2.3.CO;2.
Root, S. T., Z. Sutphin, and T. Burgess. 2020. “Green Sturgeon (Acipenser medirostris) in the San Joaquin River, California: New Record.” California Fish and Wildlife 106(4):268–270.
Roper, B. B., D. L. Scarnecchia, and T. J. La Marr. 1994. “Summer Distribution of and Habitat Use by Chinook Salmon and Steelhead within a Major Basin of the South Umpqua River, Oregon.” Transactions of the American Fisheries Society 123(3):298–308. https://doi.org/10.1577/1548-8659(1994)123<0298:SDOAHU>2.3.CO;2.
Rose, K., H. Jager, Q. N. Monsen, Z. Bai, and D. E. Howe. 2024. The Nature Conservancy Peer Review of the Fish and Aquatic Effects Analysis for the Long-Term Operations of the Central Valley Project and State Water Project. A report to the Delta Science Program. https://deltacouncil.ca.gov/pdf/science-program/reports/2024-04-23-long-term-operations-fish-and-aquatic-effects-analysis-final-report.pdf.
Rose, K. A., W. J. Kimmerer, K. P. Edwards, and W. A. Bennett. 2013a. “Individual-Based Modeling of Delta Smelt Population Dynamics in the Upper San Francisco Estuary: I. Model Description and Baseline Results.” Transactions of the American Fisheries Society 142(5):1238–1259. https://doi.org/10.1080/00028487.2013.799518.
Rose, K. A., W. J. Kimmerer, K. P. Edwards, and W. A. Bennett. 2013b. “Individual-Based Modeling of Delta Smelt Population Dynamics in the Upper San Francisco Estuary: II. Alternative Baselines and Good Versus Bad Years.” Transactions of the American Fisheries Society 142(5):1260–1272. https://doi.org/10.1080/00028487.2013.799519.
Rosenfield, J. A., and R. D. Baxter. 2007. “Population Dynamics and Distribution Patterns of Longfin Smelt in the San Francisco Estuary.” Transactions of the American Fisheries Society 136:1577–1592. https://doi.org/10.1577/T06-148.1.
Rossi, G. J., B. R. Bellmore, J. B. Armstrong, et al. 2024. “Foodscapes for Salmon and Other Mobile Consumers in River Networks.” BioScience 74(9):586–600. https://doi.org/10.1093/biosci/biae064.
Schemel, L. E., T. R. Sommer, A. B. Müller-Solger, and W. C. Harrell. 2004. “Hydrologic Variability, Water Chemistry, and Phytoplankton Biomass in a Large Floodplain of the Sacramento River, CA, USA.” Hydrobiologia 513:129–139. https://doi.org/10.1023/B:hydr.0000018178.85404.1c.
Secor, D. H., P. J. Anders, W. E. Van Winkle, and D. A. Dixon. 2002. “Can We Study Sturgeons to Extinction? What We Do and Don’t Know About the Conservation of North American Sturgeons.” American Fisheries Society Symposium 28:3–10. https://filelib.wildlife.ca.gov/FileLib/Adult_Sturgeon_and_Striped_Bass/Sturgeon%20study%20to%20extinction%202002.pdf.
Seesholtz, A. M., M. J. Manuel, and J. P. Van Eenennaam. 2015. “First Documented Spawning and Associated Habitat Conditions for Green Sturgeon in the Feather River, California.” Environmental Biology of Fishes 98(3):905–912. https://doi.org/10.1007/s10641-014-0325-9.
SEP Group. 2016. Conservation Planning Foundation for Restoring Chinook Salmon (Oncorhynchus tshawytscha) and O. Mykiss in the Stanislaus River. https://www.noaa.gov/sites/default/files/legacy/document/2020/Oct/07354626617.pdf.
Skinner, J. E. 1962. An Historical Review of the Fish and Wildlife Resources of the San Francisco Bay Area. Water Projects Report No. 1. Sacramento, CA: California Department of Fish and Game. www.estuaryarchive.org/archive/skinner_1962/.
Smith, W. E., L. Polansky, and M. L. Nobriga. 2021. “Disentangling Risks to an Endangered Fish: Using A State-Space Life Cycle Model to Separate Natural Mortality from Anthropogenic Losses.” Canadian Journal of Fisheries and Aquatic Sciences 78(8):1008–1029. https://doi.org/10.1139/cjfas-2020-0251.
Smits, A.P., J.R. Durand, B.O. Williamshen, K. Luke, and T. O’Rear. 2025. “Drought and Aquatic Vegetation Alter Water Clarity and Fish Assemblages in Freshwater Tidal Ecosystems of the Sacramento-San Joaquin River Delta.” Estuaries and Coasts 48:120. https://doi.org/10.1007/s12237-025-01556-7.
Sommer, T., B. Harrell, M. Nobriga, R. Brown, P. Moyle, W. Kimmerer, and L. Schemel. 2001a. “California’s Yolo Bypass: Evidence That Flood Control Can Be Compatible with Fisheries, Wetlands, Wildlife, and Agriculture.” Fisheries 26:6–16. https://doi.org/10.1577/1548-8446(2001)026<0006:CYB>2.0.CO;2.
Sommer, T. R., M. L. Nobriga, W. C. Harrell, W. Batham, and W. J. Kimmerer. 2001b. “Floodplain Rearing of Juvenile Chinook Salmon: Evidence of Enhanced Growth and Survival.” Canadian Journal of Fisheries and Aquatic Sciences 58:325–333. https://doi.org/10.1139/f00-245.
Sommer, T. R., W. C. Harrell, and M. L. Nobriga. 2005. “Habitat Use and Stranding Risk of Juvenile Chinook Salmon on a Seasonal Floodplain.” North American Journal of Fisheries Management 25:1493–1504. https://doi.org/10.1577/M04-208.1.
Sommer, T., C. Armor, R. Baxter, et al. 2007. “The Collapse of Pelagic Fishes in the Upper San Francisco Estuary.” Fisheries 32:270–277. https://doi.org/10.1577/1548-8446(2007)32[270:TCOPFI]2.0.CO;2.
Sommer, T., F. H. Mejia, M. L. Nobriga, F. Feyrer, and L. Grimaldo. 2011. “The Spawning Migration of Delta Smelt in the Upper San Francisco Estuary.” San Francisco Estuary &Watershed Science 9(2). https://doi.org/10.15447/sfews.2011v9iss2art2.
SST (Salmon Scoping Team). 2017. Effects of Water Project Operations on Juvenile Salmonid Migration and Survival in the South Delta; Volume 1: Findings and Recommendations. Report to the Collaborative Adaptive Management Team.
STWG (Salmon Technical Working Group). 2024. A Review of Recent Science to Improve Our Understanding and Application of Life Cycle and Decision Support Models to Salmon Management in the South Delta. https://csamp.baydeltalive.com/camt_salmon_work/salmon-technical-studies.
Sturrock, A. M., J. D. Wikert, T. Heyne, et al. 2015. “Reconstructing the Migratory Behavior and Long-Term Survivorship of Juvenile Chinook Salmon Under Contrasting Hydrologic Regimes.” PLoS One 10(5):e0122380. https://doi.org/10.1371/journal.pone.0122380.
Sweetnam, D. A., R. D. Baxter, and P. B. Moyle. 2001. “True Smelts.” Pp. 472–479 in California’s Living Marine Resources: A Status Report. California Department of Fish and Game, California Sea Grant Program.
Takata, L., T. R. Sommer, J. Louise Conrad, and B. M. Schreier. 2017. “Rearing and Migration of Juvenile Chinook Salmon (Oncorhynchus Tshawytscha) in a Large River Floodplain.” Environmental Biology of Fishes 100:1105–1120. https://doi.org/10.1007/s10641-017-0631-0.
Thomas, M. J., M. L. Peterson, N. Friedenberg, et al. 2013. “Stranding of Spawning Run Green Sturgeon in the Sacramento River: Post-Rescue Movements and Potential Population-Level Effects.” North American Journal of Fisheries Management 33(2):287–297. https://doi.org/10.1080/02755947.2012.758201.
Thomson, J. R., W. J. Kimmerer, L. R. Brown, et al. 2010. “Bayesian Change Point Analysis of Abundance Trends for Pelagic fishes in the Upper San Francisco Estuary.” Ecological Applications 20:1431–1448. https://doi.org/10.1890/09-0998.1.
Tobias, V., L. Mitchell, L. Polansky, et al. 2024. Building a Longfin Smelt Life Cycle Model. Interagency Ecological Program 2024 Annual Workshop. April 23–25, 2024.
USBR (U.S. Bureau of Reclamation). 2024a. Final EIS Long-Term Operations of the Central Valley Project and State Water Project - Appendix AB Biological Assessment. Chapter 9 – Delta Smelt. https://www.usbr.gov/mp/nepa/includes/documentShow.php?Doc_ID=55337.
USBR. 2024b. Final EIS Long-Term Operations of the Central Valley Project and State Water Project - Appendix AB Biological Assessment. Chapter 10 – Long-fin Smelt. https://www.usbr.gov/mp/nepa/includes/documentShow.php?Doc_ID=55336.
USBR. 2024c. Final EIS Long-Term Operations of the Central Valley Project and State Water Project - Appendix AB Biological Assessment. Chapter 7 – Steelhead. https://www.usbr.gov/mp/nepa/includes/documentShow.php?Doc_ID=55339.
USBR. 2024d. Final EIS Long-Term Operations of the Central Valley Project and State Water Project - Appendix AB Biological Assessment. Chapter 8 – Green Sturgeon. https://www.usbr.gov/mp/nepa/includes/documentShow.php?Doc_ID=55338.
USBR. 2024e. Final EIS Long-Term Operations of the Central Valley Project and State Water Project - Appendix AB Biological Assessment. Chapter 5 – Winter-run Chinook Salmon. https://www.usbr.gov/mp/nepa/includes/documentShow.php?Doc_ID=55341.
USBR. 2024f. Final EIS Long-Term Operations of the Central Valley Project and State Water Project - Appendix AB Biological Assessment. Chapter 5 – Spring-run Chinook Salmon. https://www.usbr.gov/mp/nepa/includes/documentShow.php?Doc_ID=55340.
USFWS (U.S. Fish and Wildlife Service). 1993. “Endangered and Threatened Wildlife and Plants; Final Rule, Determination of Threatened Status of the Delta Smelt.” Federal Register 58:12854–12864.
USFWS. 1994. “Endangered and Threatened Wildlife and Plants; Critical Habitat Determination for the Delta Smelt.” Federal Register 59:65256–65278.
USFWS. 1995. Working paper on restoration needs: Habitat restoration actions to double natural production of anadromous fish in the Central Valley of California. Volumes 1–3. Prepared by the Anadromous Fish Restoration Program Core Group for the U.S. Fish and Wildlife Service, Stockton, California.
USFWS. 2022. Species Status Assessment for the San Francisco Bay Delta Distinct Population Segment of the Longfin Smelt. U.S. Fish and Wildlife Service San Francisco-Bay-Delta Fish and Wildlife Office, Sacramento, California. https://www.fws.gov/sites/default/files/documents/08_12_22_LFS_Final_SSA_508_compliant.pdf.
Watson, L. R., S. C. Yun, J. M. Groff, and R. P. Hedrick. 1995. “Characteristics and Pathogenicity of a Novel Herpesvirus Isolated from Adult and Subadult White Sturgeon Acipensertransmontanus.” Diseases of Aquatic Organisms 22:199–210. https://doi.org/10.3354/dao022199.
Williams, J. G. 2006. “Central Valley Salmon: A perspective on Chinook and Steelhead in the Central Valley of California.” San Francisco Estuary and Watershed Science 4(3). https://doi.org/10.15447/sfews.2006v4iss3art2.
Windell, S., P. L. Brandes, J. L. Conrad, J. W. Ferguson, P. A. L. Goertler, B. N. Harvey, J. Heublein, J. A. Israel, D. W. Kratville, J. E. Kirsch, R. W. Perry, J. Pisciotto, W. R. Poytress, K. Reece, B. G. Swart, and R. C. Johnson. 2017. Scientific Framework for Assessing Factors Influencing Endangered Sacramento River Winter-Run Chinook Salmon (Oncorhynchus tshawytscha) Across the Life Cycle. U.S. Department of Commerce, NOAA Technical Memorandum NMFS-SWFSC-586. http://doi.org/10.7289/V5/TM-SWFSC-586.
Yoshiyama, R. M., F. W. Fisher, and P. B. Moyle. 1998. “Historical Abundance and Decline of Chinook Salmon in the Central Valley Region of California.” North American Journal of Fisheries Management 18:487–521. https://doi.org/10.1577/1548-8675(1998)018<0487:HAADOC>2.0.CO;2.
Yoshiyama, R. M., E. R. Gerstung, F. W. Fisher, and P. B. Moyle. 2001. “Historical and Present Distribution of Chinook Salmon in the Central Valley Drainage of California.” Fish Bulletin 179(1):71–176. https://escholarship.org/content/qt6sd4z5b2/qt6sd4z5b2.pdf#page=94.
Zimmerman, M. S., C. Kinsel, E. Beamer, E. J. Connor, and D. E. Pflug. 2015. “Abundance, Survival, and Life History Strategies of Juvenile Chinook Salmon in the Skagit River, Washington.” Transactions of the American Fisheries Society 144(3):627–641. https://doi.org/10.1080/00028487.2015.1017658.